Abstract
Heroin dependence is a major health and social problem associated with increased morbidity and mortality that adversely affects social circumstances, productivity, and healthcare and law enforcement costs. In the UK and many other Western countries, both methadone and buprenorphine are recommended by the relevant agencies for detoxification from heroin and for opioid maintenance therapy. However, despite obvious benefits due to its unique pharmacotherapy (eg, greatly reduced risk of overdose), buprenorphine has largely failed to overtake methadone in managing opioid addiction. The experience from the developing world (based on data from India) is similar. In this article we compare the advantages and disadvantages of the use methadone and buprenorphine for the treatment of opioid addiction from both a developed and developing world perspective; and explore some of the reasons why buprenorphine has not fulfilled the expectations predicted by many in the addictions field.
Keywords: Addiction, buprenorphine, detoxification, maintenance, methadone, opiate
Introduction
Illicit heroin use is an international problem. Opiate use and/or dependence is destructive on many levels. To mention a few, it is associated with high rates of hepatitis C and HIV infection, overdose and criminality.[1–3] Globally, it is estimated that there are 13 million injecting drug users, including 9.2 million who use heroin, according to the World Health Organization.[4] Based on the United Nations Drug Control Program of 2000 global illicit drug trends for persons older than 15 years, the estimated prevalence of “problematic” opioid use in countries such as France, Germany, Italy, Netherlands, Spain, and UK was 0.11%.[5] For Southeast Asian countries, such as India and Bangladesh the “problematic” opioid use prevalence rate was 0.15%. Furthermore, the United Nations Office on Drugs and Crime reports the prevalence of opioid use at least once in the year 2008 as 0.4%–0.5% in Western Europe and North America, and 0.2%–0.4% in Southeast Asia.[6] Prevalence of opioid dependence seems to be concentrated mainly in younger age groups and higher in men compared with women.[5]
In the UK, USA, Australia, and parts of Europe, methadone is the most commonly used medication to treat heroin dependence. The National Institute of Health and Clinical Excellence (NICE) in the UK recommends both buprenorphine and methadone as a first line treatment for both medically assisted withdrawal from heroin or other opiates or opioids and for maintenance purposes, with due consideration given to service user preference. NICE also recommends lofexidine, an alpha-2 adrenergic agonist, for mild and uncertain dependence. Clinical practice guidelines for management of opioid dependence in India adopt similar recommendations, that is, both methadone and buprenorphine as pharmacotherapy.[7]
Methadone is a full opioid agonist and overdose-related deaths (mostly mediated though respiratory depression) are a significant problem.[8,9] Buprenorphine, a partial opioid agonist, has become an increasingly popular choice in clinical practice in recent years in a number of developed countries, such as UK, France, USA, and Australia. Due to its unique pharmacological mode of action buprenorphine has been heralded as a new dawn for the treatment of heroin dependence with claims of less abuse potential and lower overdose risk. However, in the UK at least, these claims do not appear to have stood up to scrutiny. In this article we will:
Compare buprenorphine and methadone in terms of efficacy;
Examine the relative safety of both drugs; and
Explore the issues relating to when clinicians should consider prescribing one over the other.
We conducted a literature review using MEDLINE (with the search terms: “methadone,” “heroin,” “buprenorphine,” “opiate,” “opioid,” and “treatment”) to generate the papers we used to inform our conclusions relating to these issues. In addition, the UK treatment guidelines (eg, NICE) were reviewed and the authors’ familiarity with field was used to guide the exercise. This article is not meant to be an extensive review of the literature; it is summary of the “methadone vs buprenorphine for treatment of opiate dependence” argument from both a Western perspective and from a “developing” nation point-of-view. We chose India as a comparator from the developing world as there are reasonable data regarding opiate abuse from this country, which has a similar prevalence of dependence despite being one of the world's producers of opium.
Mode of action of opioid drugs
Opiate drugs, such as heroin (diamorphine), are natural derivates from opium, whereas opioids, such as methadone and buprenorphine, are synthetic derivates of opiates. There are a number of opioid receptor subtypes in the central nervous system and the effect of their activation is shown in Table 1. For both buprenorphine and methadone mμ and delta receptor subtypes mediate adenyl cyclase activity and activation of inwardly rectifying potassium channels leading to a release of endogenous opioids, such as endorphins and enkephalins, thus both are effective at bringing about analgesia and euphoria, but to differing degrees.
Table 1.
Activity of opioid receptor subtypes

Mode of action of buprenorphine
Due to its unique pharmacologic profile, buprenorphine has, in principle, a number of advantages over methadone for use as an opioid replacement therapy. For example, it has low intrinsic activity at mμ receptors. This means that buprenorphine is not as potent as a full mμ agonist, like methadone, and causes less analgesia and euphoria but nonetheless ameliorates withdrawal symptoms [Table 2]. Anecdotal clinical experience is in keeping with these findings and recipients of buprenorphine often complain about missing the “high” of methadone.
Table 2.
Comparison of buprenorphine and methadone in the treatment of opioid dependence
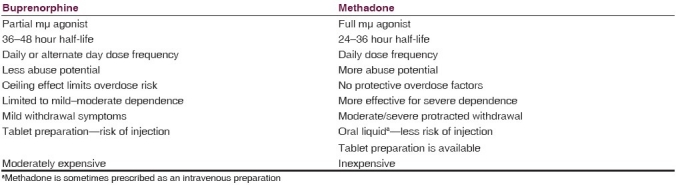
As a partial agonist, buprenorphine has a “ceiling effect,” that is, after a certain point taking more will not increase any of the effects of the drug. In addition, buprenorphine has a high affinity for the mμ receptor, which means that it reduces the effects of additional opioid use. As a potent kappa antagonist, buprenorphine has less dysphoriant effects than methadone and, therefore, may be better tolerated according to an open-label study.[10]
Efficacy of opioid replacement therapies
Maintenance therapy
Twenty-four randomized controlled trials (RCTs) comparing buprenorphine to methadone in the maintenance treatment of opioid dependence with a total number of 4497 participants were included in a 2008 Cochrane systematic review and meta-analysis.[11] The main outcome measures were treatment retention and suppression of illicit opioid use. Given in flexible doses buprenorphine was statistically significantly less effective than methadone in retaining patients in treatment (relative risk = 0.8) but, for those who remained in the trials, equivalent in suppressing heroin use. However, low dose methadone retained more patients than low or medium doses of buprenorphine (although medium dose buprenorphine suppressed illicit opioid use more effectively than low dose methadone). Medium dose buprenorphine did not have any advantage over medium dose methadone in terms of retention and was less effective in suppressing illicit opioid use. Studies from the All India Institute of Medical Sciences performed by Rajat Ray and colleagues, were also reviewed as they have done further work to compare differential doses of buprenorphine among opiate-dependent subjects.[12,13]
Detoxification
Fewer data exist for direct comparisons of buprenorphine vs methadone for detoxification from heroin dependence. Gowing and colleagues conducted a Cochrane systematic review 18 studies involving 1356 participants of buprenorphine for management of opioid withdrawal.[14] However, only 14 studies were RCTs and just 4 compared buprenorphine with methadone. Nonetheless, buprenorphine was equivalent to methadone in ameliorating severity of opioid withdrawal but symptoms may resolve more quickly, and there was a trend toward greater completion of treatment relative to methadone (relative risk = 1.3, P value = 0.08).
Cost-effectiveness
A number of studies have addressed the issue of the cost-effectiveness of buprenorphine and methadone. A comparison of these studies is problematic because they use different parameters to evaluate cost-effectiveness.[15] Where comparison is possible, flexible dose methadone maintenance (UK£14,000; US$23,100; Indian Rupees 1,030,400) performs somewhat better in terms of quality-adjusted life years than buprenorphine (UK£27,000; US$44,550; Indian Rupees 1,987,200) when both are compared to no drug therapy for heroin users. Table 2 compares buprenorphine to methadone in terms of pharmacology, preparation, effects, and costs.
Safety of opioid replacement therapies
The safety features of pharmacotherapies for heroin dependence must be weighed up against the benefits of continued illicit drug use. All opioid replacement therapies considered here can potentially cause death but continued heroin use is associated with high morbidity. For example, Caplehorn and Drummer found that methadone maintenance saved two lives for every one lost in a year.[8] There are a number of safety domains that could be considered here but these will be limited to the most significant, being:
Overdose potential;
Abuse potential; and
Serious side effects, for example, cardiovascular problems.
Overdose liability
Buprenorphine causes less respiratory depression than methadone due to its ceiling effect and, thus, has lower overdose potential.[16] In a recent pooled analysis of RCTs of opioid maintenance therapy buprenorphine showed no significant differences in serious adverse events compared with methadone.[15] However, when the authors conducted an indirect comparison from population cross-sectional studies there was less mortality associated with buprenorphine than methadone use. Two caveats need to be added here. Firstly, when used in combination with other respiratory depressants, such as alcohol or benzodiazepines, buprenorphine use can result in sedation, coma, and death.[17] Secondly, patients who have not been educated about the pharmacology of buprenorphine and use additional opioids seeking a “high” are at risk of overdose when the effects of buprenorphine wear off.
Abuse potential
Like all opioid drugs, buprenorphine has the potential for misuse. For instance, in opioid users in Glasgow in Scotland in the 1980s and 1990s, buprenorphine prescribed for pain management was obtained and used illicitly.[18] In countries where buprenorphine has been used widely for the treatment of opioid dependence, such as France, diversion has been a problem in 20% of recipients.[19] Furthermore, there is evidence suggesting that buprenorphine diversion is an increasing problem.[20] Although, where toxicological measures are recorded nationally diversion seems to be less of a problem—the average quarterly ratio of abuse cases per 1000 Subutex (the trade name for buprenorphine when used to treat opioid dependence) prescriptions dispensed in the USA was only 0.08.[21]
Nonetheless, buprenorphine diversion is a particular problem when it leads to use by the intravenous route, since as a preparation buprenorphine is formulated as a tablet and is associated with a number of complications, such as phlebitis. The higher prevalence of buprenorphine diversion—10 times more so than methadone, according to a recent Australian study[22] —may be due to its sublingual tablet formulation and difficulty associated with supervising its consumption compared to that of an oral liquid, such as methadone linctus or mixture. The recent adoption of a combined buprenorphine–naloxone tablet (Suboxone) may go some way to countering the effects of diversion—when taken sublingually buprenorphine avoids first pass metabolism (unlike naloxone, an opioid antagonist), but if Suboxone is injected naloxone blocks the effect of buprenorphine thus negating any “high.” However, once established at mμ receptor sites, there is some evidence that buprenorphine has been difficult to antagonize with naloxone.[23]
Serious side effects
Although buprenorphine has been used as an analgesic for over three decades it is a relatively new compound in terms of the treatment of opioid dependence, certainly in comparison with methadone. As with all new treatments, unknown medical complications may become apparent as clinical experience and usage of buprenorphine develops. Reports already exist of buprenorphine-induced hepatitis, in particular when used intravenously, and national guidelines now recommend regular monitoring of liver function, especially when commencing treatment.[24] However, alternative treatments are not without serious side effects; it has only recently been established that methadone, probably when prescribed in high doses, can cause cardiac problems, such as Torsades de Pointes.[25]
Indian perspective
India has a substantial opiate dependence problem with reports of up to 2 million addicts in the Sub-continent (although this must be considered in the light of a huge population pushing 1 billion people).[26] Although men are overwhelmingly more likely to use opiates in India compared to Europe and North America, and the urban-rural differential rates of use are not as marked. Official guidelines are very similar to those used in the UK and other developed nations, and the same treatments—both psychosocial and pharmacological—are available, but the health care system that delivers these interventions is different.[7] Both methadone and buprenorphine are available and used for opiate detoxification and maintenance purposes, but the enthusiasm for the latter agent in not shared quite to the same degree as in the West. Furthermore, there are differences in the licensing of the two compounds between India and Europe/North America.
Choosing between buprenorphine and methadone
Severity of dependence
Many factors are relevant when choosing the correct agent for opioid maintenance or detoxification, such as patient/clinician's preference and local guidelines. However, the patient's degree of opiate dependence is a key factor. Due to its weaker efficacy, buprenorphine is probably best restricted those with mild–moderate dependence, whereas methadone can be used with all levels of dependence.
Risk of divergence
If the risk of divergence is considered high with a given patient then one should consider methadone prescription due to the previously highlighted difficulties supervising the consumption of buprenorphine.
Precipitated withdrawal
Buprenorphine's high affinity for the mμ receptor means that it will displace any existing circulating opioids. This presents a unique problem when initiating buprenorphine treatment for opioid dependence: the phenomenon of precipitated withdrawal. Therefore, inducting heroin users requires caution and transferring patients from methadone runs a particular risk of an extended precipitated withdrawal syndrome because of methadone's long half-life; so much so that prescribing guidelines do not recommend switching patients who are on greater than 60 mg of methadone to buprenorphine[27] and the Department of Health recommends that the dose of methadone should be no more than 30 mg methadone/day.[24]
Special considerations
The safety of opioid replacement for the management of heroin dependence in pregnancy is an important issue given the potential adverse effects on the fetus of the prescribed compound and/or continued illicit drug use. Buprenorphine is not licensed for this purpose and, given the substantial experience of its use in pregnancy, methadone remains the opioid of choice for many clinicians in these circumstances. That said, there is growing evidence for the safety of buprenorphine in pregnancy compared to methadone, albeit from poorly powered studies,[28] and it is reported to be associated with fewer neonatal withdrawal symptoms.
Conclusion
Despite the obvious benefits conferred by the ceiling effect, buprenorphine prescriptions have failed to overtake methadone in the UK. Therefore, it seems that other issues are more important. There is significant evidence showing that better treatment outcomes are associated with high activity at the mμ receptor, for example, “the narcotic blockade” achieved with high doses of methadone.[29] Therefore, it seems likely that buprenorphine's weaker mμ activity accounts for its poorer performance compared to methadone in clinical trials. It is the authors’ view based on clinical experience that the user misses the “gouch” (the colloquial term in the UK coined by users for the slight “head-nodding” effect of heroin and methadone) associated with methadone compared with the relatively “clear-headed” state associated with buprenorphine, and this plays a significant role in the relative lack of uptake of the latter.
Buprenorphine is probably the safer agent. However, its relative advantage over methadone in these safety domains is somewhat tempered by the emerging evidence of problematic diversion and the risks associated with the intravenous use of crushed tablets.
A decade or so after the introduction of buprenorphine into clinical practice in the UK, the drug has not fulfilled all the dreams of those who heralded its arrival (ie, becoming the mainstay replacement therapy instead of methadone). The same can be said of India. The higher cost of buprenorphine (approx. £2.80 for 8 mg tablet) compared to methadone (approx. £2.50 for 500 mL) may mean that health care providers favor the cheaper more familiar methadone. However, buprenorphine is a useful addition to the armory of pharmacotherapies available to substance use clinicians. This viewpoint is supported by the NICE and guidelines in India.[30,26]
Acknowledgments
The author would like to thank Drs. Kim Wolff from the Institute of Psychiatry, London, and Neeraj Tripathi from the St. Mary's Senior House Officer rotational training program in psychiatry, London, for their input into previous drafts of this article.
Footnotes
Source of Support: Nil.
Conflict of Interest: Much of the information sources for this article come from an unpublished essay the first author wrote as part of course work in achieving a Masters of Science degree in Public Health and Clinical Aspects of Addiction from King's College London.
References
- 1.Shooting up: Infections among injecting drug users in the United Kingdom 2005. London: Health Protection Agency; 2006. Health Protection Agency. [Google Scholar]
- 2.Ghodse AH, Sheehan M, Taylor C, Edwards G. Deaths of drug addicts in the United Kingdom 1967-81. Br Med J (Clin Res Ed) 1985;290:425–8. doi: 10.1136/bmj.290.6466.425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Parker H, Newcombe R. Heroin use and acquisitive crime in an English community. Br J Sociol. 1987;38:331–50. [PubMed] [Google Scholar]
- 4.World Health Organization, Department of mental health and substance misuse. Proposal for the inclusion of buprenorphine in the WHO model list of essential medicines. WHO. 2004. [Last accessed: 2011]. Available from: http://www.who.int/substance_abuse/activities/buprenorphine_essential_medicines.pdf .
- 5.United Nations Drug Control Programme. [Last accessed on 2000]. Available from: http://www.un.org/Docs/SG/SG-Rpt/ch3c-5.htm .
- 6.United Nations Office on Drugs and Crime. World Drug Report. 2009:152–6. [Google Scholar]
- 7.Clinical practice guidelines for management of opioid dependence. Indian Psychiatr Soc. 2004 [Google Scholar]
- 8.Caplehorn JR, Drummer OH. Mortality associated with New South Wales methadone programs in 1994: lives lost and saved. Med J Aust. 1999;170:104–9. doi: 10.5694/j.1326-5377.1999.tb127675.x. [DOI] [PubMed] [Google Scholar]
- 9.Wolff K. Characterization of methadone overdose: clinical considerations and the scientific evidence. Ther Drug Monit. 2002;24:457–70. doi: 10.1097/00007691-200208000-00001. [DOI] [PubMed] [Google Scholar]
- 10.Rothman RB, Gorelick DA, Heishman SJ, Eichmiller PR, Hill BH, Norbeck J, et al. An open-label study of a functional opioid kappa antagonist in the treatment of opioid dependence. J Subst Abuse Treat. 2000;18:277–81. doi: 10.1016/s0740-5472(99)00074-4. [DOI] [PubMed] [Google Scholar]
- 11.Mattick R, Kimber J, Breen C, Davoli M. Buprenorphine maintenance versus placebo or methadone maintenance for opioid dependence. Cochrane Database Syst Rev. 2003:CD002207. doi: 10.1002/14651858.CD002207. [DOI] [PubMed] [Google Scholar]
- 12.Ray R, Pal H, Kumar R, Maulick P, Mangla R. Post-marketing surveillance of buprenorphine. Pharmacoepidemiol Drug Saf. 2004;13:615–9. doi: 10.1002/pds.975. [DOI] [PubMed] [Google Scholar]
- 13.De S, Jain R, Ray R, Dhawan A, Varghese ST. Assessment of differential doses of buprenorphine for long term pharmacotherapy among opiate dependent subjects. Indian J Physiol Pharmacol. 2008;52:53–63. [PubMed] [Google Scholar]
- 14.Gowing L, Ali R, White J. Buprenorphine for the management of opioid withdrawal. Cochrane Database Syst Rev. 2006:CD002025. doi: 10.1002/14651858.CD002025.pub3. [DOI] [PubMed] [Google Scholar]
- 15.Connock M, Juarez-Garcia A, Jowett S, Frew E, Liu Z, Taylor RJ, et al. Methadone and buprenorphine for the management of opioid dependence: a systematic review and economic evaluation. (iii-iv).Health Technol Assess. 2007;11:1–171. doi: 10.3310/hta11090. [DOI] [PubMed] [Google Scholar]
- 16.Wolff K, editor. Substance Misuse: Substitution drugs (methadone and buprenorphine) Oxford: Elsevier; 2005. [Google Scholar]
- 17.Mégarbane B, Hreiche R, Pirnay S, Marie N, Baud FJ. Does high-dose buprenorphine cause respiratory depression?: possible mechanisms and therapeutic consequences. Toxicol Rev. 2006;25:79–85. doi: 10.2165/00139709-200625020-00002. [DOI] [PubMed] [Google Scholar]
- 18.Hammersley R, Cassidy MT, Oliver J. Drugs associated with drug-related deaths in Edinburgh and Glasgow, November 1990 to October 1992. Addiction. 1995;90:959–65. doi: 10.1046/j.1360-0443.1995.9079598.x. [DOI] [PubMed] [Google Scholar]
- 19.Auriacombe M, Fatséas M, Dubernet J, Daulouède JP, Tignol J. French field experience with buprenorphine. Am J Addict. 2004;13(Suppl 1):S17–28. doi: 10.1080/10550490490440780. [DOI] [PubMed] [Google Scholar]
- 20.Jenkinson RA, Clark NC, Fry CL, Dobbin M. Buprenorphine diversion and injection in Melbourne, Australia: an emerging issue? Addiction. 2005;100:197–205. doi: 10.1111/j.1360-0443.2004.00958.x. [DOI] [PubMed] [Google Scholar]
- 21.Smith MY, Bailey JE, Woody GE, Kleber HD. Abuse of buprenorphine in the United States: 2003-2005. J Addict Dis. 2007;26:107–11. doi: 10.1300/J069v26n03_12. [DOI] [PubMed] [Google Scholar]
- 22.Winstock AR, Lea T, Sheridan J. Prevalence of diversion and injection of methadone and buprenorphine among clients receiving opioid treatment at community pharmacies in New South Wales, Australia. Int J Drug Policy. 2008;19:450–8. doi: 10.1016/j.drugpo.2007.03.002. [DOI] [PubMed] [Google Scholar]
- 23.Cowan A, Lewis JW, editors. Buprenorphine: combating drug abuse with a unique opioid. Chichester: Wiley-Liss; 1995. [Google Scholar]
- 24.Misuse and Dependence: UK Guidelines on Clinical Management. London: Department of Health; 2007. Department of Health. [Google Scholar]
- 25.Krantz MJ, Rowan SB, Schmittner J, Bucher Bartelson B. Physician awareness of the cardiac effects of methadone: results of a national survey. J Addict Dis. 2007;26:79–85. doi: 10.1300/J069v26n04_10. [DOI] [PubMed] [Google Scholar]
- 26.Ganguly K. The pattern and process of drug and alcohol use in India. ICMR Bull. 2008. [Last accessed on 2008]. Available from: http://www.icmr.nic.in/bulletin/english/2008/bulljan-mar08.pdf .
- 27.Taylor D, Paton C, Kerwin R. The Maudsley prescribing Guidelines. 9th ed. London: Informa Healthcare; 2007. [Google Scholar]
- 28.Fischer G, Ortner R, Rohrmeister K, Jagsch R, Baewert A, Langer M, et al. Methadone versus buprenorphine in pregnant addicts: a double-blind, double-dummy comparison study. Addiction. 2006;101:275–81. doi: 10.1111/j.1360-0443.2006.01321.x. [DOI] [PubMed] [Google Scholar]
- 29.Ball J, Ross A. The effectiveness of methadone maintenance treatment: Patients, programs, services and outcome. New York: Springer-Verlag; 1991. [Google Scholar]
- 30.National Institute of Health and Clinical Excellence. Drug misuse: psychosocial interventions: NICE guideline 51. [Last accessed in 2011]. Available from: http://guidance.nice.org.uk/CG51 .


