Abstract
Two-component signal transduction systems (TCS) are a main strategy used by bacteria to sense and adapt to changes in their environment. In the legume symbiont Rhizobium leguminosarum biovar viciae VF39, mutation of chvG, a histidine kinase, caused a number of pleiotropic phenotypes. ChvG mutants are unable to grow on proline, glutamate, histidine, or arginine as the sole carbon source. The chvG mutant secreted smaller amounts of acidic and neutral surface polysaccharides and accumulated abnormally large amounts of poly-ß-hydroxybutyrate. Mutation of chvG caused symbiotic defects on peas, lentils, and vetch; nodules formed by the chvG mutant were small and white and contained only a few cells that had failed to differentiate into bacteroids. Mutation of chvG also destabilized the outer membrane of R. leguminosarum, resulting in increased sensitivity to membrane stressors. Constitutive expression of ropB, the outer membrane protein-encoding gene, restored membrane stability and rescued the sensitivity phenotypes described above. Similar phenotypes have been described for mutations in other ChvG-regulated genes encoding a conserved operon of unknown function and in the fabXL genes required for synthesis of the lipid A very-long-chain fatty acid, suggesting that ChvG is a key component of the envelope stress response in Rhizobium leguminosarum. Collectively, the results of this study demonstrate the important and unique role the ChvG/ChvI TCS plays in the physiology, metabolism, and symbiotic competency of R. leguminosarum.
INTRODUCTION
The Gram-negative cell envelope is critical for cell survival, and a complex and dynamic structure ensures the bacterium's successful adaptation to sudden environmental changes (32). The complexity of the cell envelope is particularly evident from the study of rhizobia, a group of soil-dwelling bacteria that form mutualistic symbioses with leguminous plants. These bacteria are uniquely adapted to survive in the rhizosphere as well as intracellularly within legume plant cells (12). During symbiosis, the cell envelope of rhizobia undergoes significant changes, and several cell envelope components are critical for a successful transition from a free living cell to an intracellular bacteroid (2, 4, 9, 12, 43). While significant attention has been focused on characterizing individual cell envelope components in free-living cells and during plant infection, a major challenge moving forward is to elucidate how rhizobia integrate multiple signals from their environment to coordinate an appropriate adaptive response to their cell envelope.
Two-component signal transduction systems (TCSs) represent a broadly conserved strategy among bacteria for sensing and responding to external stimuli. TCSs are typically comprised of a membrane-associated sensor kinase and a cytoplasmic response regulator. The most extensively studied TCS in the Rhizobiaceae are the ChvG/ChvI system from Agrobacterium tumefaciens and the homologous ExoS/ChvI system from Sinorhizobium meliloti, where ChvG (ExoS) is the sensor kinase and ChvI is the response regulator (5, 7). These TCSs play important roles in both pathogenic and beneficial host-microbe interactions (2, 5, 7, 8, 19, 43). Mutation of either chvG or chvI in A. tumefaciens results in avirulence toward several different host plants, including infection of stems of Zinnia elegans and Lycopersicon esculentum and of Bryophyllum daigremontianum leaves (5). The ChvG/ChvI TCS in A. tumefaciens positively regulates a number of acid-inducible genes, including aopB, katA, virB, and virE, leading to the hypothesis that the Agrobacterium ChvG/ChvI acts as a pH sensor that regulates virulence genes in response to the acidic conditions encountered during the early stages of infection (19). In S. meliloti, a number of studies have shown that ExoS and ChvI are required for successful symbiosis with alfalfa, the host legume (2, 43, 45). The ExoS/ChvI TCS in S. meliloti is required for production of succinoglycan and galactoglucan, the two symbiotically active exopolysaccharides (EPSs) (2, 7, 43, 45). These EPSs are required for proper infection thread initiation and elongation (7, 45). It remains to be determined whether the ExoS/ChvI TCS is a direct or indirect regulator of EPS production in S. meliloti. In addition, mutation of homologs of chvG and chvI in closely related alphaproteobacterial animal pathogens such as Brucella abortus (BvrR/BvrS) and Bartonella spp. (BatR/BatS) leads to a reduction in invasiveness, prevention of intracellular replication, and lysosomal fusion, resulting in a loss of virulence in mice (30, 35). It is clear from these studies that the ChvG/ChvI TCS represents an important regulatory system whose proper function is required during host infection.
The ChvG/ChvI TCS is also critical for proper physiological functioning of free-living bacteria. In A. tumefaciens, mutation of chvG or chvI leads to alterations in the outer membrane, resulting in increased sensitivity to detergents and hydrophobic antibiotics (5, 19). Mutations in these genes also lead to loss of growth on complex media and minimal media containing cas-amino acids (5). Functional ChvG and ChvI are also required for growth at acidic pH, an observation that is consistent with the speculation that this TCS acts as a sensor to respond to acid stress in A. tumefaciens (19).
Until recently, the ExoS/ChvI TCS was assumed to be essential in S. meliloti, due to the inability to isolate null mutations in the genes encoding either ExoS or ChvI (22). However, two recent studies have reported null mutations in both of these genes. Bélanger et al. (2) used a merodiploid-facilitated mutation strategy to create null mutants of both exoS and chvI. In addition to confirming the role of ExoS and ChvI in symbiosis and exopolysaccharide production, they reported significant metabolic defects in the mutants, which had a 75% reduction in the number of carbon sources able to support growth compared to the wild type (2). A separate chvI deletion mutant was found to be acid sensitive and hypermotile, have an altered lipopolysaccharide (LPS) profile, and produce less poly-ß-hydroxybutyrate (PHB) (43). Furthermore, a total of 59 genes encoding members of a number of diverse functional groups, including EPS synthesis proteins, outer membrane proteins, catabolism-related proteins, and transporters, have been identified as putative targets for direct regulation by ChvI (6). The difficulty in isolating null mutants, the pleiotropic phenotypes displayed by mutants, and the large chvI regulon clearly emphasize the physiological importance of this TCS.
To date, characterization of the ChvG (ExoS)/ChvI TCS in the Rhizobiaceae has focused on S. meliloti and A. tumefaciens. Recently, we reported isolation of a chvG mutant in Rhizobium leguminosarum VF39 and described its role in regulating the ropB outer membrane protein-encoding gene and an operon required for cell envelope integrity and control of cell morphology (RL3499-RL3502) (10, 38). We have also found that ChvG is important for induction of the fabXL genes required for biosynthesis of the lipid A very-long-chain fatty acid during growth under membrane stress conditions (E. M. Vanderlinde and C. K. Yost, unpublished data). In this paper, we have identified novel phenotypes for a chvG mutant that are distinct from those observed for other rhizobia. We have found that the role of chvG in regulating polysaccharide production, PHB accumulation, and carbon source utilization in R. leguminosarum differs significantly from that of S. meliloti. We have also shown that constitutive expression of ropB restores tolerance of detergents, hydrophobic antibiotics, and hydrogen peroxide in the chvG mutant by restoring outer membrane stability, suggesting that the mechanism for loss of membrane integrity in the chvG mutant is due, in part, to loss of ropB expression. The chvG mutant in R. leguminosarum also has many of the same phenotypes observed for homologs in other bacteria, including defective nodulation, acid sensitivity, and loss of growth on complex media. Our data suggest that chvG plays an important role in Rhizobium leguminosarum physiology by independently and coordinately regulating both cellular metabolism and cell envelope structure.
MATERIALS AND METHODS
Strains, media, and growth conditions.
Strains and plasmids used in the study are summarized in Table 1. Escherichia coli strains were cultured using Luria-Bertani (LB) medium (31), supplemented as necessary with the following concentrations of antibiotics: ampicillin, 100 μg/ml; spectinomycin, 100 μg/ml; and tetracycline, 10 μg/ml. R. leguminosarum cells were cultured using tryptone-yeast extract (TY) (3) or Vincent's minimal medium (VMM) with 10 mM mannitol (41). The VMM, prepared according to the method of Vincent (41), contained the following ingredients: K2HPO4 (1 g/liter); KH2PO4 (1 g/liter); KNO3 (0.6 g/liter); 1 g or 10 g of carbon source/liter, as indicated; FeCl3 (0.1 g/liter); MgSO4 (2.5 g/liter); CaCl2 (1 g/liter); biotin (0.01 g/liter); thiamine (0.01 g/liter); and calcium pantothenate (0.01 g/liter). R. leguminosarum cultures were grown as required with the following concentrations of antibiotics: gentamicin, 30 μg/ml; neomycin, 100 μg/ml; tetracycline, 5 μg/ml; and streptomycin, 500 μg/ml. Unless otherwise indicated, all media were buffered to pH 7.0. Growth was routinely assessed following incubation at 30°C for 2 days.
Table 1.
Bacterial strains and plasmids
| Strain or plasmid | Relevant characteristicsa | Source or reference |
|---|---|---|
| E. coli strains | ||
| TOP10 | F−mcrAΔ(mrr hsdRMS mcrBC) Φ80lacZΔM15 ΔlacX74 recA1 araΔ139Δ(ara-leu)7697 galU galK rpsL (Smr) endA1 nupG | Invitrogen |
| S17-1 | RP4 tra region, mobilizer strain recA derivative of MM294A with RP4-2 (Tc::Mu, Km::Tn7) integrated into the chromosome | 33 |
| DH5α | endA1 hsdR17 supE44 thi-1 recA1 gyrA96 relA1 Δ(argF-lacZYA) ΔlacU169 Φ80lacZ ΔM15 | Invitrogen |
| R. leguminosarum strains | ||
| VF39 | Biovar viciae, Smr | 29 |
| DF20 | VF39SM, chvGΩKm mutant, unable to grow on complex media, Smr Nmr | 10 |
| DF20pDF38 | VF39SM, chvGΩKm mutant complemented in trans with a functional copy of chvG cloned downstream of the constitutive trp promoter in the broad-host-range plasmid, pDG71, Smr Nmr Tcr | 10 |
| DF20pEV91 | VF39SM, chvGΩKm mutant, complemented in trans with a functional copy of ropB cloned downstream of the constitutive trp promoter in the broad-host-range plasmid, pDG71, Smr Nmr Tcr | This study |
| Plasmids | ||
| pCR2.1 Topo | TOPO TA cloning vector, 4.0 kb, Kmr Apr | Invitrogen |
| pFUS1par | Broad-host-range vector with promoterless gusA for transcriptional fusions, par stabilized, Tcr | 46 |
| pDG71 | Broad-host-range derivative of pHC41, constitutively expressed tryptophan promoter and gfp(mut3), Tcr | 11 |
| pNdvB | pFUS1par, ndvB::gusA, Tcr | 39 |
| pDF38 | Broad-host-range vector pDG71 containing a functional copy of chvG downstream of tryptophan promoter, Tcr | 10 |
| pEV91 | Broad-host-range vector pDG71 containing a functional copy of ropB downstream of tryptophan promoter, Tcr | 40 |
| pEV109 | pFus1par with pssA::gusA transcriptional fusion, Tcr | This study |
| pEV117 | pFus1par with phbC::gusA transcriptional fusion, Tcr | This study |
| pEV118 | pFus1par with phaZ::gusA transcriptional fusion, Tcr | This study |
Ap, ampicillin; Km, kanamycin; Nm, neomycin, Sm, streptomycin; Tc, tetracycline.
Growth on different carbon sources and at acidic pH.
To assess the growth of the chvG mutant on different carbon sources, the wild-type, chvG mutant, and chvG-complemented strains were streaked onto VMM containing 0.1% of each tested carbon source. The plates were incubated at 30°C for 3 days, which is the amount of time necessary for the chvG mutant to yield visualizable colonies on VMM with mannitol, which was used as a positive control. The relative levels of growth of the chvG mutants were evaluated semiquantitatively such that +++ represents growth equivalent to that observed on a mannitol plate, ++ represents roughly 50% to 80% of the amount of growth observed on a mannitol plate, + represents roughly 20% to 50% of the amount of growth observed on a mannitol plate, +/− represents scarce growth, and − represents no growth.
To determine the effect of pH on the growth of the chvG mutant, viable plate counts of the wild-type, chvG mutant, and chvG-complemented strains were determined on VMM with mannitol buffered to pH 5.0, pH 7.0, or pH 9.0. The log reduction in growth compared to growth at pH 7.0 was then calculated as described by Vanderlinde et al. (38).
Quantification of EPS and neutral polysaccharides.
Briefly, cells from a fresh plate of VMM with 1% mannitol as the carbon source were resuspended in 50 ml of VMM broth to an optical density at 600 nm (OD600) of approximately 0.1 and filtered onto S-Pak 0.45-μm-pore-size TypeHA membranes with a Millipore vacuum manifold (Millipore Inc, Bedford, MA) using Microfil filtration funnels. Filters were placed on VMM with 1% mannitol, glucose, or sorbitol, as indicated, and incubated at 30°C for 48 h. Membranes were placed in sterile tubes containing 10 ml of sterile double-distilled water (dH2O) and subjected to vortexing to remove cells. Secreted EPS and neutral polysaccharides were quantified as described previously (39, 40).
Construction of gusA transcriptional fusions.
A directional cloning approach was used to construct gusA transcriptional fusions. PCR was used to amplify the putative promoter region of the following genes of interest: pssA (386 bp; pssaF [GTCTCGAGTGTCGAAACGGCA] and pssaR [CGATCGCTGTGGATAACAGG]), phbC (496 bp; phbcF [TACTCGAGTAGCCGTCCTGGTATTTTGC] and phbcR [GCTGCTTGTCCGAGATTTTC]), and phaZ (496 bp; phazF [GACTCGAGTCCTCGTCTCCTACCTCAGC] and phazR [TATGGGAGAACGGATTCAGC]). The forward primers contained XhoI linkers (underlined) to assist with cloning. The PCR products were cloned into pCR2.1 TOPO using reagents and protocols supplied by the manufacturer (Invitrogen, Burlington, Ontario, Canada). The promoter fragments were subsequently excised using XhoI and EcoRI and cloned into vector pFUS1par containing a promoterless gusA reporter gene and a par stabilization locus, creating plasmids pEV109 (pssA), pEV117 (phbC), and pEV118 (phaZ) (46). Restriction mapping and DNA sequencing were used to confirm the proper orientation and sequence fidelity of the cloned PCR amplicons. Plasmids were subsequently transformed into the E. coli S17-1 mobilizer strain and conjugated into R. leguminosarum strain VF39 (wild type) or DF20 (chvG mutant) to measure gene expression as described below.
β-Glucuronidase (gusA) reporter gene assays.
The enzyme assays for β-glucuronidase activity were carried out as described previously (21, 46).
Nile blue staining of intracellular granules.
The staining procedure was a modification of a protocol described previously (25). Nile blue A is a lipophilic stain that fluoresces when bound to polyhydroxyalkanoates (PHAs) (23, 25). Cells were grown on standard VMM agar supplemented with 1 ml of 1% Nile blue A per liter, and the amount of fluorescence was observed using a UV light box. To quantify the amount of fluorescence accumulated over time, cells from a fresh plate of VMM with 1% mannitol as the carbon source were resuspended in 50 ml of VMM broth to an OD600 of approximately 0.1 and filtered onto S-Pak 0.45-μm-pore-size TypeHA membranes with a Millipore vacuum manifold (Millipore Inc, Bedford, MA) using Microfil filtration funnels. Membranes were subsequently divided into quarters, placed onto VMM Nile blue A medium prepared as described above, and incubated at 30°C. After 2, 3, 4, and 5 days, a quarter of the membrane was removed and placed into a sterile tube with 2 ml of sterile water. Cells were removed from the filter by vortexing vigorously for 2 min. The fluorescence of a 200-μl aliquot of cells was measured using excitation and emission wavelengths of 490 nm and 580 nm, respectively, in a BioTek Synergy HT microplate reader, with Gen5 analysis software (BioTek Instruments Inc, Winooski, VT). To standardize the data, viable plate counts for the cell suspensions were also determined. Data are reported as relative fluorescent units (RFU) per CFU. To visualize fluorescent granules within individual cells, smears were made from day 5 cell suspensions and stained with crystal violet. Cells were visualized under white and UV light with an Olympus BX51 microscope (Olympus, Center Valley, PA). For fluorescence microscopy, an Olympus WU filter cube with the following specifications was used: dichroic mirror DM400, excitation filter BP330-385, and barrier filter BA420 (Olympus, Center Valley, PA).
Plant assays.
Nodulation assays were carried out with peas (Pisum sativum cv. Trapper), vetch (Vicia nigricans), and lentils (Lens culinaris L. cv. CDC Sovereign) as the host legumes. Seeds were germinated and planted as described previously (47). Following germination, seeds were inoculated with approximately 1 × 109 cells of VF39, DF20, or DF20pDF38. Plants were grown at ambient temperature with a 16-h photoperiod for 6 weeks prior to harvesting. Nodules were visually inspected for defects. To examine bacteroids of the different strains, individual nodules were placed into 50 μl of sterile water and crushed. A 5-μl aliquot was spotted onto a slide and stained with crystal violet. Bacteroids were visualized with an Olympus BX51 microscope (Olympus, Center Valley, PA). The plant shoot dry weights were determined by removing the roots just above the cotyledon and drying the shoots overnight at 60°C prior to weighing.
Antibiotic and detergent sensitivity assays.
Antibiotic and detergent sensitivity assays were carried out using disk diffusion assays. Strains were inoculated into VMM overlays and poured onto solid VMM with mannitol. Discs containing 50 μg of polymyxin B, 37.5 μg of H2O2, 1000 μg of sodium dodecyl sulfate (SDS), or 40 μg of erythromycin were applied to the inoculated VMM plates, and zones of inhibition were measured after incubation at 30°C for 2 days.
N-Phenyl-1-naphthylamine (NPN) uptake assay of outer membrane permeability.
A modified NPN uptake assay was used to assess the relative permeability of the outer membrane of the different strains used in this study (13). Fresh, isolated colonies were scraped from a VMM plate and resuspended in 5 mM HEPES buffer (pH 7.2) to an OD600 of ∼0.5. The following were then combined in a microtiter plate: 95 μl of HEPES buffer (pH 7.2), 5 μl of 0.5 mM 1-N-phenyl-1-naphthylamine (NPN) in acetone, and 100 μl of cells. The fluorescence was then measured at 2-min intervals for 20 min using excitation and emission wavelengths of 355 nm and 405 nm, respectively, in a BioTek Synergy HT microplate reader, with Gen5 analysis software (BioTek Instruments Inc, Winooski, VT). To standardize the data, viable plate counts for the cell suspensions were also determined. Data are reported as relative fluorescent units (RFU) per CFU. Control samples were set up as follows: (i) buffer (100 μl) plus cells (100 μl) and (ii) buffer (95 μl) plus NPN (5 μl). The relative fluorescence of control samples containing either (i) buffer (100 μl) plus cells (100 μl) or (ii) buffer (95 μl) plus NPN (5 μl) was measured as described above. The average background fluorescence of buffer plus cells was 634 ± 2.66 RFU, and the average background fluorescence of buffer plus NPN was 28,552 ± 438 RFU.
RESULTS
Growth of the chvG mutant is impaired on certain amino acids and at acidic pH.
The chvG null mutant in Rhizobium leguminosarum bv. viciae VF39 is unable to grow on complex media (10). This observation is consistent with phenotypes observed for mutations in chvG orthologs in both S. meliloti and A. tumefaciens (2, 5). Bélanger et al. (2) observed that mutants of exoS or chvI of S. meliloti were unable to grow on 48 of the 64 carbon sources tested, suggesting an important role for the TCS in central metabolism (2). Therefore, we tested growth of the R. leguminosarum chvG mutant on a variety of different carbon sources. A total of 45 different carbon sources were tested, including amino acids, hexoses, pentoses, polyols, oligosaccharides, and dicarboxylic acids. Of the 45 carbon sources tested, only 22 were able to support growth of the wild type (VF39). The results for growth of the chvG mutant on these 22 carbon sources are summarized in Table 2. All assays were performed on solid media, because the chvG mutant is unable to grow in broth culture. The mutant grew on all carbon sources tested that would support growth of the wild type, with the exception of glutamate, histidine, arginine, and proline (Table 2). Growth on all four amino acids was restored to wild-type levels through complementation of the chvG mutation with pDF38, the chvG-expressing plasmid. The chvG mutant was able to grow on VMM containing mannitol and E, H, P, or R when these amino acids were used as a sole nitrogen source (data not shown).
Table 2.
Growth of the chvG mutant on different carbon sourcesa
| Carbon source | Category | Growth of indicated strain |
||
|---|---|---|---|---|
| VF39 (wild type) | DF20 (chvG mutant) | DF20pDF38 (chvG complement) | ||
| Sucrose | Oligosaccharide | +++ | +++ | +++ |
| Trehalose | Oligosaccharide | +++ | +++ | +++ |
| Maltose | Oligosaccharide | +++ | +++ | +++ |
| Raffinose | Oligosaccharide | +++ | +++ | +++ |
| Ribose | Pentose | ++ | + | ++ |
| Xylose | Pentose | +++ | ++ | +++ |
| Arabinose | Pentose | +++ | +++ | +++ |
| Glucose | Hexose | +++ | +++ | +++ |
| Galactose | Hexose | +++ | +++ | +++ |
| Mannose | Hexose | +++ | +++ | +++ |
| Glycerol | Polyol | +++ | ++ | +++ |
| Mannitol | Polyol | +++ | +++ | +++ |
| Sorbitol | Polyol | +++ | +++ | +++ |
| Adonitol | Polyol | +++ | +++ | +++ |
| Dulcitol | Polyol | +++ | ++ | +++ |
| Inositol | Polyol | +++ | +++ | +++ |
| Erythritol | Polyol | +++ | +++ | +++ |
| Alanine | Neutral, nonpolar amino acid | +++ | ++ | +++ |
| Proline | Neutral, nonpolar amino acid | +++ | + | +++ |
| Arginine | Basic, polar amino acid | +++ | − | +++ |
| Histidine | Basic, polar amino acid | +++ | +/− | +++ |
| Glutamate | Acidic, polar amino acid | +++ | + | +++ |
Strains were streaked onto solid VMM with 0.1% of the indicated carbon source, and growth was assessed visually after 48 h of incubation at 30°C. Only carbon sources that supported growth of wild-type R. leguminosarum VF39 are shown. Data presented are representative of the results of at least three separate trials.
Homologs of chvG found in A. tumefaciens and S. meliloti are required for growth at acidic pH (19, 43). Likewise, the chvG mutant in R. leguminosarum VF39 had a >7.0 log reduction in growth in media buffered to pH 5 compared to growth on the same media buffered to pH 7.0. The wild-type and complement strains grew equally well on media at either pH 5.0 or pH 7.0. No growth defects were observed for any of the strains grown at an alkaline pH of 9.0. To determine whether medium acidification due to metabolism of the E, H, P, and R amino acids contributes to the inability of the chvG mutant to grow on these amino acids as sole carbon sources, the pH of VMM with each amino acid was monitored during growth of the wild-type and chvG-complemented strains. The pH of the medium remained neutral throughout (data not shown).
The chvG mutant maintains exopolysaccharide production but does so at lower levels than the wild type.
In S. meliloti, ExoS and ChvI are required for production of both succinoglycan and galactoglucan, the two major exopolysaccharides secreted by this bacterium (2, 7). The direct role of ChvG/ChvI in regulation of succinoglycan in A. tumefaciens has not been reported. However, mutation of exoR, a periplasmic protein that inhibits ChvG, results in hypermucoid cells in A. tumefaciens strain C58, providing indirect evidence that ChvG and ChvI regulate succinoglycan production in A. tumefaciens (36). R. leguminosarum strains do not produce succinoglycan or galactoglucan. Instead, R. leguminosarum secretes an acidic EPS with an octasaccharide repeating unit of glucose, glucuronic acid, and galactose at a molar ratio of 5:2:1, as well as several different neutral polysaccharides, including cyclic (ß-1,2) glucans and glucomannan (34, 44). The R. leguminosarum chvG mutant had a 1.7-fold decrease in EPS production compared to the wild type when each was grown on mannitol (Table 3). To determine whether the nature of the carbon source had an effect on the amount of EPS produced by the mutant, we also quantified the amount of EPS produced by cells grown on sorbitol and glucose. Sorbitol was chosen because it is a structurally similar polyol that produces phenotypically drier colonies than mannitol, and glucose was chosen because it is a structurally distinct hexose sugar that produces hypermucoid colonies that are phenotypically similar to those formed on mannitol. As expected, cells grown on sorbitol produced approximately 4-fold-smaller amounts of EPS than cells grown on mannitol and glucose (Table 3). However, the chvG mutant produced, on average, 2.12- and 2.05-fold less EPS on glucose and sorbitol, respectively, compared to the wild type. Results for secretion of neutral polysaccharides by the chvG mutant were similar to those found for EPS. The chvG mutant, on average, secreted 1.62-, 2.25-, and 1.75-fold-lower concentrations of neutral polysaccharides than the wild type when grown on mannitol, glucose, and sorbitol, respectively (Table 3).
Table 3.
EPS and neutral polysaccharide secretion by the chvG mutant grown on mannitol, glucose, or sorbitola
| Strain | Avg (± SD) amount of polysaccharide secreted (mg/mg dry cell wt) |
|||||
|---|---|---|---|---|---|---|
| Mannitol |
Glucose |
Sorbitol |
||||
| EPS | Neutral polysaccharide | EPS | Neutral polysaccharide | EPS | Neutral polysaccharide | |
| VF39 (wild type) | 2.45 ± 0.172 | 76.9 ± 9.17 | 2.14 ± 0.084 | 138 ± 18.1 | 0.437 ± 0.120 | 35.0 ± 6.02 |
| DF20 (chvG mutant) | 1.41 ± 0.286* | 47.4 ± 11.6* | 1.01 ± 0.138* | 61.3 ± 0.259* | 0.213 ± 0.049* | 19.9 ± 2.20* |
| DF20pDF38 (chvG complement) | 1.94 ± 0.453 | 79.7 ± 8.63 | 1.723 ± 0.449 | 75.4 ± 0.607 | 0.406 ± 0.093 | 36.9 ± 9.08 |
Polysaccharides were quantified as described in Materials and Methods. Data are presented as averages (± standard deviations) of the amounts of secreted polysaccharide (EPS or neutral polysaccharide) determined in at least three independent replicate experiments. An asterisk indicates a statistically significant difference in the amount of polysaccharide secreted compared to the wild-type strain (P < 0.005; Student's t test).
To determine whether chvG alters polysaccharide secretion through regulation of gene expression, a pssA::gusA transcriptional fusion was mated into the chvG mutant and VF39, and expression was measured on VMM with mannitol, glucose, or sorbitol. PssA is a glucosyl transferase that initiates EPS synthesis by transferring glucose-6-phosphate from UDP glucose to an isoprenylphosphate lipid carrier attached to the cytosolic surface of the inner membrane (34). Expression of pssA was slightly reduced in the chvG mutant compared to expression in R. leguminosarum VF39 (Table 4). However, there was a less than 1.5-fold difference in expression for each of the carbon sources tested. Similarly, expression of ndvB, a glycosyltransferase required for synthesis of cyclic (ß-1,2) glucan, a major neutral polysaccharide secreted by R. leguminosarum, was slightly reduced in the chvG mutant. Taken together, these results suggest that, while mutation of chvG has an effect on polysaccharide secretion in R. leguminosarum, it is unlikely to occur through direct gene regulation of known polysaccharide synthesis genes.
Table 4.
Expression of phbC, phaZ, pssA, and ndvB in the chvG mutant grown on mannitol, glucose, or sorbitola
| Gene | Avg (± SD) GusA activity (Miller units) |
|||||
|---|---|---|---|---|---|---|
| Mannitol |
Glucose |
Sorbitol |
||||
| VF39 (wild type) | DF20 (chvG mutant) | VF39 (wild type) | DF20 (chvG mutant) | VF39 (wild type) | DF20 (chvG mutant) | |
| pssA | 7,166 ± 1,467 | 4,394 ± 2,617 | 8,047 ± 712 | 5,724 ± 42 | 4,600 ± 401 | 3,340 ± 222 |
| ndvB | 747 ± 51 | 520 ± 95 | 1,317 ± 70 | 929 ± 18 | 856 ± 69 | 761 ± 34 |
| phbC | 706 ± 138 | 923 ± 20 | 736 ± 48.2 | 735 ± 49.5 | 1,058 ± 64.5 | 1,152 ± 125.9 |
| phaZ | 493 ± 48.4 | 370 ± 39.5 | 458 ± 49.6 | 339 ± 75.2 | 549 ± 44.5 | 387 ± 121 |
Data represent averages (± standard deviations) of the results at least three independent trials. No statistically significant differences were observed between the expression of the wild type and that of the chvG mutant.
The chvG mutant hyperaccumulates granules that stain with Nile blue A.
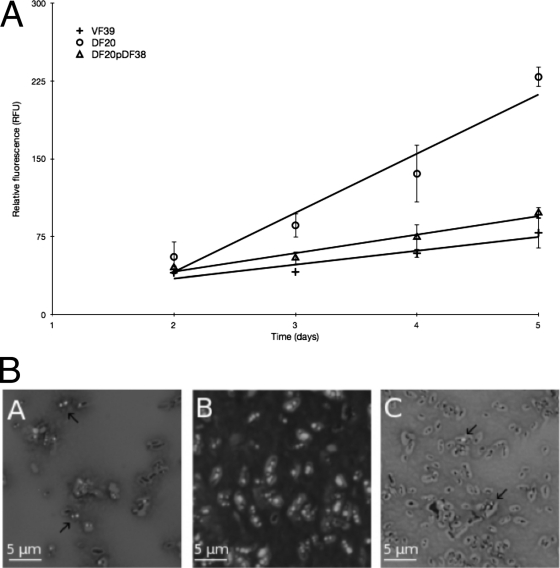
Fast-growing rhizobia, such as R. leguminosarum VF39, are known to accumulate PHB granules in free-living and bacteroid cells (37). In wild-type cells, PHB acts as a carbon storage polymer for cells experiencing high concentrations of a carbon source but nutrient limitation with respect to an essential element such as nitrogen. To observe whether the chvG mutant accumulated more PHB or a related PHA molecule, cells were grown on VMM containing the fluorescent, lipophilic dye Nile blue A and mannitol as the carbon source. Following incubation for several days, colonies were observed under UV light. The chvG mutant colonies were significantly more fluorescent than colonies formed by the wild-type and complement strains (data not shown). To quantify fluorescence in the different strains, cells were grown on media containing Nile blue A and mannitol, and the relative fluorescence was measured over time (Fig. 1A). After 2 days of growth, the levels of fluorescence of the wild-type and mutant cells were very similar, with an increase of only 1.4-fold in fluorescence for the chvG mutant compared to the wild type. However, at day 5, the mutant accumulated 2.9 times more fluorescence than the wild type.
Fig 1.
(A) Accumulation of PHB granules over time. Cells were grown on VMM with mannitol and Nile blue A, and fluorescence was measured after 2, 3, 4, and 5 days of incubation at 30°C. Viable plate counts were used to standardize the fluorescence data. (B) Fluorescence microscopy of PHB granules in cells grown on VMM with mannitol and Nile blue A for 5 days at 30°C. Panel A, wild type; panel B, chvG mutant; panel C, chvG complement.
To further confirm that the increased fluorescence in the mutant was due to staining of intracellular granules, cells grown with Nile blue A and mannitol were observed under a fluorescence microscope (Fig. 1B). The granules fluoresced strongly under conditions of UV excitation, indicating they had been stained with the Nile blue A. Close to 100% of the observed chvG mutant cells had many large, strongly fluorescent granules that occupied the majority of the cytoplasmic space. In comparison, roughly 50% of the observed wild-type and complement cells had smaller fluorescent granules, while the remaining cells lacked any fluorescence. In addition, the granules did not stain with dyes specific for other polymers, such as starch (data not shown). Collectively, these results support the conclusion that the chvG mutant accumulates excessive amounts of PHB or of another closely related PHA polymer.
To determine the effect of the carbon source on PHB accumulation by the chvG mutant, the Nile blue A assay was repeated for cells grown on glucose or sorbitol (Table 5). For all carbon sources tested, the fluorescence of the chvG mutant cells after 6 days of incubation at 30°C was higher than that of the wild-type and complement cells; however, the magnitude of difference between the mutant and the wild type varied depending on the carbon source. For example, growth in mannitol resulted in an 8.5-fold difference between the mutant and wild type and growth in glucose resulted in a 3-fold increase in fluorescence, while growth in sorbitol resulted in a 1.6-fold increase in fluorescence.
Table 5.
PHB accumulation by the chvG mutant grown on mannitol, glucose, or sorbitola
| Strain | Avg (± SD) amt of fluorescence at 580 nm |
||
|---|---|---|---|
| Mannitol | Glucose | Sorbitol | |
| VF39 (wild type) | 141 ± 14.5 | 426 ± 54 | 1,416 ± 66 |
| DF20 (chvG mutant) | 1,199 ± 114* | 1,303 ± 51* | 2,285 ± 62* |
| DF20pDF38 (chvG complement) | 426 ± 22 | 488 ± 11 | 1,575 ± 42 |
PHB accumulation was measured using Nile blue A fluorescence as described in Materials and Methods. Data represent averages (± standard deviations) of the results of at least three independent replicate experiments. An asterisk indicates a statistically significant difference in PHB accumulation compared to the wild type (P < 0.001; Student's t test).
Expression of gusA transcriptional fusions to genes for synthesis (phbC) and degradation (phaZ) of PHB was measured in the wild-type and chvG mutant strains on mannitol, sorbitol, and glucose to determine whether the increase in PHB accumulation in the chvG mutant was due to alterations in gene expression (Table 4). We found no significant differences in expression of the fusions on any of the carbon sources tested.
The chvG mutant is unable to form an effective symbiosis with peas, lentils, or vetch.
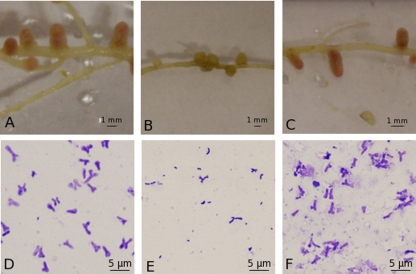
Functional ChvG is required for successful infection in other host-associated Rhizobiales species, including S. meliloti, A. tumefaciens, and B. abortus (2, 5, 7, 35, 43). Likewise, plants inoculated with the R. leguminosarum VF39 chvG mutant were small and yellow and displayed symptoms of nitrogen starvation compared to the large, green, healthy appearance of plants inoculated with the wild-type and complemented strains. Dry weights of peas, lentils, and vetch inoculated with the mutant were similar to the weights of uninoculated control plants and were less than half the weights of plants inoculated with the wild-type strain (Table 6). Nodules formed by the chvG mutant were small, round, and white, whereas nodules formed by the wild-type and complemented strains were large, oblong, and red (Fig. 2). Nodules formed by the wild-type and complemented strains contained numerous, large, Y-shaped bacteroids, while nodules formed by the chvG mutant contained few, rod-shaped cells (Fig. 2).
Table 6.
Dry weights of legume plants inoculated with VF39, DF20, or DF20pDF38a
| Host plant | Dry wt (g ± SD) of plant inoculated with: |
|||
|---|---|---|---|---|
| VF39 (wild type) | DF20 (chvG mutant) | DF20pDF38 (chvG complement) | Uninoculated | |
| Pisum sativum cv. Trapper (pea) | 0.580 ± 0.129 | 0.237 ± 0.063* | 0.364 ± 0.010 | 0.205 ± 0.080* |
| Lens culinaris L. cv. CDC Sovereign (lentil) | 0.394 ± 0.069 | 0.162 ± 0.043* | 0.349 ± 0.058 | 0.123 ± 0.022* |
| Vicia nigricans (vetch) | 0.256 ± 0.030 | 0.097 ± 0.032* | 0.192 ± 0.022 | 0.050 ± 0.011* |
Six-week-old plants were dried and weighed as described in Materials and Methods. Data are presented as grams per plant and represent averages (± standard deviations) of the results of at least three independent replicate experiments. An asterisk indicates a statistically significant difference in weight compared to plants inoculated with the wild type (P < 0.001; Student's t test).
Fig 2.
Nodules (A to C) and bacteroids (D to F) from P. sativum cv. Trapper plants inoculated with the wild type (A and D), chvG mutant (B and E), or chvG complement (C and F). Similar results were obtained for L. culinaris L. cv. CDC Sovereign and Vicia nigricans.
The chvG mutant has increased outer membrane permeability and is sensitive to membrane stressors.
R. leguminosarum ChvG is a positive regulator of the outer membrane protein-encoding gene ropB in R. leguminosarum (10). Cells lacking RopB are sensitive to detergents and other stressors of the outer membrane, such as hydrophobic antibiotics (10). Therefore, we used disc diffusion assays to determine whether the chvG mutant was also sensitive to various outer membrane stressors (Table 7). The chvG mutant displayed an increased sensitivity to all of the compounds tested. In addition, the chvG mutant had a 1.3-fold increase in the zone of inhibition around a disc of hydrogen peroxide, indicating the chvG mutant is more sensitive to oxidative stress than the wild-type strain.
Table 7.
Zone-of-inhibition assays to assess sensitivity of the chvG mutant to erythromycin, polymyxin B, SDS, and hydrogen peroxidea
| Inhibition zone (mm) |
||||
|---|---|---|---|---|
| VF39 (wild type) | DF20 (chvG mutant) | DF20pDF38 (chvG complement) | DF20pEV91 (chvG mutant, ropBcon) | |
| Erythromycin | 11.7 ± 0.578 | 19.0 ± 1.00* | 12.0 ± 0.00 | 14.3 ± 0.578 |
| Polymyxin B | 12.7 ± 0.578 | 18.3 ± 0.578* | 14.3 ± 0.578 | 14.7 ± 0.578 |
| SDS | 9.67 ± 0.578 | 18.3 ± 1.53* | 9.33 ± 0.578 | 12.3 ± 0.578 |
| H2O2 | 20.7 ± 1.53 | 27.0 ± 3.00* | 19.7 ± 3.21 | 15 ± 0.00 |
Data are presented as averages (± SD) of the zones of inhibition (in millimeters) determined in at least three independent replicate experiments. An asterisk indicates a statistically significant difference in zone of inhibition compared to the wild type (P < 0.005; Student's t test).
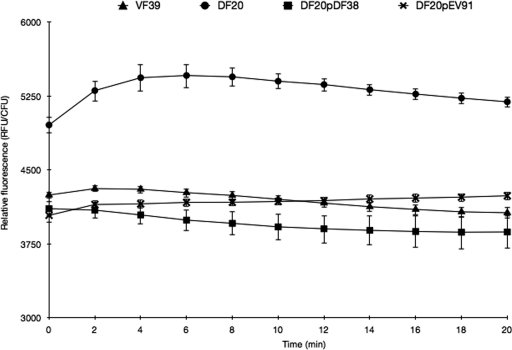
The outer membrane integrity of the chvG mutant was measured by an NPN uptake assay (13). NPN is a hydrophobic dye that fluoresces strongly when bound to the inner phospholipid bilayer of Gram-negative bacteria. An intact outer membrane acts as a permeability barrier, preventing NPN from accessing the inner membrane and resulting in low fluorescence. However, a damaged outer membrane allows entry of the dye and results in much higher relative fluorescence (13). The wild-type and complement strains of R. leguminosarum VF39 were able to prevent uptake of the dye (Fig. 3), and prior to normalization based on CFU, the wild-type and complement strains had fluorescence readings close to the background levels determined for controls containing only buffer and NPN, as described in Materials and Methods. The chvG mutant strain, however, had high fluorescence that peaked after 6 min of incubation, at which point there was a 30% increase in relative fluorescence compared to the wild-type strain (Fig. 3).
Fig 3.
N-Phenyl-1-naphthylamine (NPN) uptake assay to measure outer membrane permeability. Cells from VMM with mannitol were resuspended in 5 mM HEPES buffer (pH 7.2) to an approximate OD600 of 0.5. dH2O (95 μl), 0.5 mM NPN (5 μl), and cells (100 μl) were combined, and the fluorescence was measured every 2 min for 20 min. Viable plate counts were used to standardize the fluorescence data.
To determine whether loss of outer membrane integrity in the chvG mutant was due to the lack of ropB expression, we complemented the chvG mutant with a plasmid constitutively expressing ropB from a trp promoter in the pDG71 plasmid (11). Constitutive expression of ropB restored the resistance of the chvG mutant to erythromycin, polymyxin B, and SDS by 64.4%, 64.3%, and 69.5%, respectively (Table 7). Resistance to hydrogen peroxide was also restored to levels that exceeded the resistance of the wild-type strain (Table 7). In addition, the amount of fluorescence of chvG mutant cells constitutively expressing ropB in the presence of NPN was similar to levels observed for wild-type cells, suggesting that the restored presence of RopB alone can sufficiently restabilize the outer membrane in the chvG mutant to prevent uptake of the fluorescent dye (Fig. 3). Growth of the chvG mutant on VMM with glutamate, proline, arginine, or histidine as the sole carbon source was not restored by constitutive ropB expression (data not shown). In addition, constitutive ropB expression did not complement the decrease in polysaccharide secretion, growth at acidic pH, or PHB accumulation phenotypes described above (data not shown).
DISCUSSION
The ChvG sensor kinase in R. leguminosarum impacts both cellular metabolism and cell envelope structure. Unlike the S. meliloti chvG mutant, the R. leguminosarum mutant's growth defect was restricted to four amino acids: glutamate, histidine, arginine, and proline. The amino acids E, H, P, and R are structurally different; however, they all enter the central metabolic pathway through glutamate and the tricarboxylic acid (TCA) cycle by the action of either glutamate dehydrogenase or the γ-aminobutyric acid (GABA) shunt (17). The chvG mutant was able to grow on alanine, which was the only other amino acid able to support growth of R. leguminosarum VF39 as a sole carbon source. Alanine enters the central metabolic pathway through glycolysis. There are several possible explanations for the inability of the chvG mutant to grow on E, H, P, or R. We have determined that acidification of the medium or membrane stress caused by the charged amino acids is not responsible for the growth defects. It could be that alterations in the cell envelope structure were interfering with import of the amino acids E, H, P, and R into the cytosol of the chvG mutant and therefore preventing growth. However, we do not think that the growth issue is related to an amino acid transport defect in the chvG mutant, since the mutant can grow in VMM when the individual amino acids are used as sole nitrogen sources (data not shown). The seemingly most parsimonious explanation is that the chvG mutant has alterations in the activity of glutamate dehydrogenase or enzymes in the GABA shunt pathway that prevent its growth on E, H, P, and R. When considering metabolic defects associated with the chvG mutant, it is notable that the gene directly downstream of chvG is a putative hprK-like gene. This hprK homolog in S. meliloti is involved in carbon metabolism and assimilation (26, 27). We used reverse transcription-PCR (RT-PCR) to confirm that chvG and hprK are cotranscribed in R. leguminosarum and that the chvG mutant (DF20) does not express hprK (data not shown). Notably, we were able to fully complement all of the chvG mutant phenotypes with chvG alone; therefore, we do not believe that loss of hprK expression had any significant direct effect on the metabolic defects described above. However, our results do not rule out an indirect role for HprK in controlling carbon metabolism in the chvG polar mutant. Further experimentation would be required to determine the function of the hprK homolog in R. leguminosarum.
In addition to altering carbon source utilization, mutation of chvG also impacts accumulation of PHB and secretion of EPS and neutral polysaccharides. We found that the chvG mutant accumulated significantly more PHB than the wild type on several different carbon sources. The mutant also secreted smaller amounts of EPS and neutral polysaccharides on all carbon sources tested. There were no differences in expression of the genes phbC and phaZ, which encode enzymes for PHB synthesis and degradation, respectively, between the wild-type strain and the chvG mutant. There was also a less than 1.5-fold decrease in the expression of pssA and ndvB in the chvG mutant compared to the wild type. From these results, it is clear that ChvG was not influencing PHB accumulation or polysaccharide secretion through direct regulation. In rhizobia, PHB is synthesized from acetyl-coenzyme A (acetyl-CoA); however, the mechanisms controlling regulation of acetyl-CoA partitioning are not completely understood (37). Factors known to influence the accumulation of PHB include oxygen concentration, cytosolic redox potential, and the activity of the TCA cycle (37). Since the metabolic defects of the chvG mutant localize to the TCA cycle, we speculate that mutation of chvG affects partitioning of acetyl-CoA such that an excess amount is channeled into PHB accumulation at the expense of other cellular processes, such as cell division and polysaccharide synthesis. This hypothesis is further supported by our observation that the growth rate of the chvG mutant is substantially lower than that of the wild-type VF39 strain (data not shown).
The results we observed for PHB accumulation and polysaccharide secretion in the R. leguminosarum chvG mutant are distinctly different from those reported for S. meliloti and A. tumefaciens. Wang et al. (43) reported a decrease in PHB accumulation by a chvI loss-of-function deletion mutant grown in liquid yeast mannitol broth (YMB) medium. Our attempts to grow the R. leguminosarum chvG mutant in broth culture were unsuccessful; therefore, it is difficult to conclusively determine whether the differences in PHB accumulation in R. leguminosarum and S. meliloti were due to differences in the function of chvG or were due simply to the different growth conditions used.
Both A. tumefaciens and S. meliloti produce the exopolysaccharide succinoglycan in a manner dependent on the presence of functional ChvG (ExoS)/ChvI (2, 7, 36). Null mutants of exoS and chvI in S. meliloti do not produce succinoglycan (2). ChvI is believed to be a positive regulator of the exo genes required for synthesis of succinoglycan, although direct binding of ChvI to the exo gene promoters has not been confirmed (45). In contrast, the EPS produced by R. leguminosarum is comprised of a distinct octasaccharide repeating unit of glucose, gluronic acid, and galactose and is synthesized by genes encoding glucosyltransferases that are not orthologous to the exo genes required for succinoglycan synthesis (34). In R. leguminosarum bv. trifolii, the pssA gene is positively regulated by the transcriptional regulator RosR (15). Mutants of rosR have phenotypes similar to those described for chvG, including alterations in membrane stability, decreased levels of ropB expression, and symbiotic defects, suggesting that there may be overlap between the regulons of ChvG/ChvI and RosR (16).
R. leguminosarum bv. viciae has a relatively large range of host legumes and can infect plants belonging to the genera of Pisum, Vicia, Lens, and Lathyrus (1). Plants inoculated with the chvG mutant formed small, white, defective nodules. Similar phenotypes were observed for pea, lentil, and vetch plants; therefore, mutation of chvG does not affect host range and likely plays a central non-host-specific role during symbiosis. The pleiotropic nature of the chvG mutation makes defining the specific mechanism for the nodulation defect challenging. The cell envelope integrity of the chvG mutant is severely compromised, and the mutant is sensitive to acid and oxidative stress; therefore, it is possible that the chvG mutant is not able to adapt to the stressful conditions encountered during symbiosis, thus impeding cell division of the mutant in the infection thread and preventing entry of the bacteria into the nodule. However, a number of other mutants, including those lacking very-long-chain fatty acid-modified lipid A or the outer membrane protein RopB, have very similar free-living phenotypes, including destabilized outer membranes and acid sensitivity, and yet are able to form effective symbioses, suggesting that these defects do not necessarily prevent proper nodule formation (10, 40).
In addition to the stresses encountered during nodule formation, the metabolism of R. leguminosarum undergoes substantial changes in the initial stages of nodule development and during bacteroid differentiation. For example, during the early stages of nodulation, enzymes for GABA metabolism and the decarboxylating arm of the TCA cycle are significantly upregulated (18, 28). In addition, glutamate and glutamate dehydrogenase are thought to play important roles in ammonia assimilation within bacteroids (20, 24). Therefore, it is possible that the putative defects in glutamate catabolism observed in the chvG mutant were negatively impacting symbiosis. Future research will focus on comparing the activities of glutamate dehydrogenase, glutamate decarboxylase, GABA aminotransferase, and succinate semialdehyde dehydrogenase, the enzymes involved in glutamate catabolism (28), in strain VF39 and the chvG mutant in free-living conditions and during symbiosis.
RopB is important for maintaining outer membrane stability in R. leguminosarum (10); therefore, it is not surprising that a chvG mutant is sensitive to several membrane stressors. We confirmed the importance of ropB in stabilizing the outer membrane in the chvG mutant by supplying constitutively expressed ropB in trans, showing that it restored tolerance of the chvG mutant to membrane stressors. We also confirmed that RopB was directly affecting the stability of the outer membrane by demonstrating that it prevented uptake of the fluorescent dye NPN. We have observed similar results for ropB complementation in mutants with a conserved operon of unknown function (RL3499-RL3502) (38) and in mutants that lack the unusual lipid A very-long-chain fatty acid (Vanderlinde and Yost, unpublished). Notably, these genes are also regulated by chvG, providing evidence that the ChvG/ChvI TCS plays an important role in regulating cell envelope dynamics and interactions between different cell envelope components (37, 38) (Vanderlinde and Yost, unpublished). The envelope stress response in R. leguminosarum is poorly understood; however, our results demonstrate that ChvG, RopB, the very-long-chain fatty acid, and RL3499-RL3502 are all critical for membrane stress tolerance, implicating them as core components of the envelope stress response in R. leguminosarum.
In conclusion, our results have identified previously undescribed phenotypes associated with chvG mutations. Of particular note is the newly reported increased polyhydroxybutyrate production in the chvG mutant. Furthermore, the restoration of the ability of the chvG mutant to tolerate membrane stressors by constitutively expressing ropB has provided new insights into the mechanisms affecting outer membrane integrity in a chvG mutant. Finally, although chvG mutants within the Rhizobiaceae share several common phenotypes, the observed differences in metabolic defects associated with the R. leguminosarum chvG mutant relative to S. meliloti highlight the unique differences between these rhizobia in chvG regulatory networks.
ACKNOWLEDGMENTS
We gratefully acknowledge Trevor Charles and Ricardo Nordeste for providing unpublished data and Michael Hynes for helpful comments.
This research has been supported by a Natural Sciences and Engineering Research Council (NSERC) grant to C.K.Y. E.M.V. was supported by a Canada Graduate Scholarship from NSERC.
Footnotes
Published ahead of print 9 December 2011
REFERENCES
- 1. Allen ON, Allen EK. 1981. The Leguminosae—a source book of characteristics, uses and nodulation. Macmillan Publishers Ltd, London, United Kingdom [Google Scholar]
- 2. Bélanger L, Dimmick KA, Fleming JS, Charles TC. 2009. Null mutations in Sinorhizobium meliloti exoS and chvI demonstrate the importance of this two-component regulatory system for symbiosis. Mol. Microbiol. 74:1223–1237 [DOI] [PubMed] [Google Scholar]
- 3. Beringer J. 1974. R factor transfer in Rhizobium leguminosarum. J. Gen. Microbiol. 84:188–198 [DOI] [PubMed] [Google Scholar]
- 4. Campbell G, Reuhs B, Walker G. 2002. Chronic intracellular infection of alfalfa nodules by Sinorhizobium meliloti requires correct lipopolysaccharide core. Proc. Natl. Acad. Sci. U. S. A. 99:3938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Charles TC, Nester EW. 1993. A chromosomally encoded two-component sensory transduction system is required for virulence of Agrobacterium tumefaciens. J. Bacteriol. 175:6614–6625 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Chen E, Fisher R, Perovich V, Sabio E, Long S. 2009. Identification of direct transcriptional target genes of ExoS/ChvI two-component signaling in Sinorhizobium meliloti. J. Bacteriol. 191:6833–6842 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Cheng HP, Walker GC. 1998. Succinoglycan production by Rhizobium meliloti is regulated through the ExoS-ChvI two-component regulatory system. J. Bacteriol. 180:20–26 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Doherty D, Leigh JA, Glazebrook J, Walker GC. 1988. Rhizobium meliloti mutants that overproduce the R. meliloti acidic calcofluor-binding exopolysaccharide. J. Bacteriol. 170:4249–4256 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Ferguson G, Roop I. 2002. Deficiency of a Sinorhizobium meliloti bacA mutant in alfalfa symbiosis correlates with alteration of the cell envelope. J. Bacteriol. 184:5625–5632 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Foreman DL, Vanderlinde EM, Bay DC, Yost CK. 2010. Characterization of a gene family of outer membrane proteins (ropB) in Rhizobium leguminosarum bv. viciae VF39SM and the role of the sensor kinase ChvG in their regulation. J. Bacteriol. 192:975–983 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Gage DJ. 2002. Analysis of infection thread development using Gfp- and DsRed-expressing Sinorhizobium meliloti. J. Bacteriol. 184:7042–7046 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Gibson KE, Kobayashi H, Walker GC. 2008. Molecular determinants of a symbiotic chronic infection. Annu. Rev. Genet. 42:413–441 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Helander I. 2000. Fluorometric assessment of Gram-negative bacterial permeabilization. J. Appl. Microbiol. 88:213–219 [DOI] [PubMed] [Google Scholar]
- 14. Reference deleted.
- 15. Janczarek M, Kutkowska J, Piersiak T, Skorupska A. 2010. Rhizobium leguminosarum bv. trifolii rosR is required for interaction with clover, biofilm formation and adaptation to the environment. BMC Microbiol. 10:284–307 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Janczarek M, Skorupska A. 2004. Regulation of pssA and pssB gene expression in Rhizobium leguminosarum bv. trifolii in response to environmental factors. Antonie Van Leeuwenhoek 85:217–227 [DOI] [PubMed] [Google Scholar]
- 17. Kanehisa M. 2000. KEGG: Kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 28:27–30 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Karunakaran R, et al. 2009. Transcriptomic analysis of Rhizobium leguminosarum biovar viciae in symbiosis with host plants Pisum sativum and Vicia cracca. J. Bacteriol. 191:4002–4014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Li L, et al. 2002. A global pH sensor: Agrobacterium sensor protein ChvG regulates acid-inducible genes on its two chromosomes and Ti plasmid. Proc. Natl. Acad. Sci. U. S. A. 99:12369–12374 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Mendoza A, Valderrama B, Leija A, Mora J. 1998. NifA-dependent expression of glutamate dehydrogenase in Rhizobium etli modifies nitrogen partitioning during symbiosis. Mol. Plant Microbe Interact. 11:83–90 [DOI] [PubMed] [Google Scholar]
- 21. Miller JH. 1972. Experiments in molecular genetics. Cold Spring Harbor Laboratory, Cold Spring Harbor, NY [Google Scholar]
- 22. Osterås M, Stanley J, Finan TM. 1995. Identification of Rhizobium-specific intergenic mosaic elements within an essential two-component regulatory system of Rhizobium species. J. Bacteriol. 177:5485–5494 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Ostle AG, Holt JG. 1982. Nile blue A as a fluorescent stain for poly-beta-hydroxybutyrate. Appl. Environ. Microbiol. 44:238–241 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Patriarca EJ, Tatè R, Iaccarino M. 2002. Key role of bacterial NH(4)(+) metabolism in Rhizobium-plant symbiosis. Microbiol. Mol. Biol. Rev. 66:203–222 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Pierce L, Schroth M. 1994. Detection of Pseudomonas colonies that accumulate poly-β-hydroxybutyrate on Nile blue medium. Plant Dis. 78:683–685 [Google Scholar]
- 26. Pinedo CA, Bringhurst RM, Gage DJ. 2008. Sinorhizobium meliloti mutants lacking phosphotransferase system enzyme HPr or EIIA are altered in diverse processes, including carbon metabolism, cobalt requirements, and succinoglycan production. J. Bacteriol. 190:2947–2956 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Pinedo CA, Gage DJ. 2009. HPrK regulates succinate-mediated catabolite repression in the Gram-negative symbiont Sinorhizobium meliloti. J. Bacteriol. 191:298–309 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Prell J, Bourdes A, Karunakaran R, Lopez-Gomez M, Poole P. 2009. Pathway of γ-aminobutyrate metabolism in Rhizobium leguminosarum 3841 and its role in symbiosis. J. Bacteriol. 191:2177–2186 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Priefer U. 1989. Genes involved in lipopolysaccharide production and symbiosis are clustered on the chromosome of Rhizobium leguminosarum biovar viciae VF39. J. Bacteriol. 171:6161–6168 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Quebatte M, et al. 2010. The BatR/BatS two-component regulatory system controls the adaptive response of Bartonella henselae during human endothelial cell infection. J. Bacteriol. 192:3352–3367 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Sambrook J, Fritsch EF, Maniatis T. 1989. Molecular cloning: a laboratory manual, 2nd ed Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY [Google Scholar]
- 32. Silhavy TJ, Kahne D, Walker S. 2010. The bacterial cell envelope. Cold Spring Harb. Perspect. Biol. 2:a000414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Simon R, Priefer U, Pühler A. 1983. A broad host range mobilization system for in vivo genetic engineering: transposon mutagenesis in Gram-negative bacteria. Nat. Biotechnol. 1:784–791 doi:10.1038/nbt1183-784 [Google Scholar]
- 34. Skorupska A, Janczarek M, Marczak M, Mazur A. 2006. Rhizobial exopolysaccharides: genetic control and symbiotic functions. Microb. Cell Fact. 5:7–26 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Sola-Landa A, et al. 1998. A two-component regulatory system playing a critical role in plant pathogens and endosymbionts is present in Brucella abortus and controls cell invasion and virulence. Mol. Microbiol. 29:125–138 [DOI] [PubMed] [Google Scholar]
- 36. Tomlinson AD, Ramey-Hartung B, Day TW, Merritt PM, Fuqua C. 2010. Agrobacterium tumefaciens ExoR represses succinoglycan biosynthesis and is required for biofilm formation and motility. Microbiology 156:2670–2681 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Trainer MA, Charles TC. 2006. The role of PHB metabolism in the symbiosis of rhizobia with legumes. Appl. Microbiol. Biotech. 71:377–386 [DOI] [PubMed] [Google Scholar]
- 38. Vanderlinde EM, Magnus SA, Tambalo DD, Koval SF, Yost CK. 2011. Mutation of a broadly conserved operon from Rhizobium leguminosarum biovar viciae (RL3499-RL3502) causes defects to cell morphology and envelope integrity. J. Bacteriol. 193:2684–2694 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Vanderlinde EM, et al. 2010. Identification of a novel ABC transporter required for desiccation tolerance, and biofilm formation in Rhizobium leguminosarum bv. viciae 3841. FEMS Microbiol. Ecol. 71:327–340 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Vanderlinde EM, et al. 2009. Rhizobium leguminosarum biovar viciae 3841, deficient in 27-hydroxyoctacosanoate-modified lipopolysaccharide, is impaired in desiccation tolerance, biofilm formation and motility. Microbiology 155:3055–3069 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Vincent VM. 1970. A manual for the practical study of root-nodule bacteria. Blackwell, Oxford, United Kingdom [Google Scholar]
- 42. Reference deleted.
- 43. Wang C, et al. 2010. Sinorhizobium meliloti 1021 loss-of-function deletion mutation in chvI and its phenotypic characteristics. Mol. Plant Microbe Interact. 23:153–160 [DOI] [PubMed] [Google Scholar]
- 44. Williams A, et al. 2008. Glucomannan-mediated attachment of Rhizobium leguminosarum to pea root hairs is required for competitive nodule infection. J. Bacteriol. 190:4706–4715 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Yao S, Luo L, Har K, Becker A. 2004. Sinorhizobium meliloti ExoR and ExoS proteins regulate both succinoglycan and flagellum production. J. Bacteriol. 186:6042–6049 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Yost CK, Del Bel KL, Quandt JR, Hynes MF. 2004. Rhizobium leguminosarum methyl-accepting chemotaxis protein genes are down-regulated in the pea nodule. Arch. Microbiol. 182:505–513 [DOI] [PubMed] [Google Scholar]
- 47. Yost CK, Rochepeau P, Hynes MF. 1998. Rhizobium leguminosarum contains a group of genes that appear to code for methyl-accepting chemotaxis proteins. Microbiology 144:1945–1956 [DOI] [PubMed] [Google Scholar]