Abstract
Several craniofacial pain conditions including temporomandibular joint disorders (TMJD) are more prevalent in women than men. The basis for sex differences in deep craniofacial pain is not known. The present study compared the magnitude of ascending projections from TMJ-responsive neurons in trigeminal brainstem to the ventrolateral periaqueductal gray (vlPAG) or posterior nucleus of the thalamus (Po) in males and female rats. Fluorogold (FG) was injected into vlPAG or Po and TMJ-responsive neurons were identified by Fos-like immunoreactivity (Fos-LI) after mustard oil injection. TMJ-evoked Fos-LI was similar in males and females; however, significant differences in cell counts were seen for FG single-labeled and Fos/FG double-labeled neurons in trigeminal brainstem. After vlPAG injections, the number of FG-labeled neurons in trigeminal subnucleus interpolaris (Vi), ventral interpolaris/caudalis transition (vl-Vi/Vc) and dorsal paratrigeminal region (dPa5) was greater in females than males. The percentage of Fos/FG double-labeled neurons in vl-Vi/Vc and dPa5 after vlPAG injection also were greater in females than males. By contrast after Po injections, males displayed a greater number of FG-labeled neurons in superficial laminae of Vc and C1–2 and deeper laminae at C1–2 than females. The percentage of Fos/FG double-labeled neurons in superficial laminae of Vc after Po injection also was greater in males than females.
These data revealed significant sex differences in ascending projections from TMJ-responsive neurons in trigeminal brainstem. Such differences may influence the ability of males and females to recruit autonomic reflexes and endogenous pain control circuits relevant for TMJ nociception.
Keywords: sex difference, temporomandibular joint, trigeminal brainstem, periaqueductal gray, posterior thalamus
Temporomandibular joint disorders (TMJD) represent a heterogeneous family of conditions that present with pain in the jaw joint and muscles of mastication. The signs and symptoms of TMJD suggest a central neural dysfunction resulting in pain amplification; however, the central neural pathways that underlie TMJD pain are not well defined (Diatchenko et al. 2006; Maixner 2009). Although women are more likely to develop TMJD than men (Bereiter and Okamoto 2011), little is known about brain pathways that process noxious sensory information from deep craniofacial tissues in males and females. Sensory afferent nerves that supply the TMJ and muscles of mastication terminate at multiple rostrocaudal levels of the trigeminal brainstem nuclear complex (TBNC) and upper cervical dorsal horn (Shigenaga et al. 1988; Dessem et al. 2007). C-fos immunohistochemical studies have confirmed that select regions of the TBNC that receive direct input from the TMJ, e.g., dorsal paratrigeminal region (dPa5) and superficial laminae of Vc and upper cervical dorsal horn (C1–2), encode the intensity of an intra-TMJ stimulus in an estrogen-dependent manner (Bereiter 2001; Okamoto et al. 2008). Anatomical tract-tracing combined with c-fos methods have been used to report that TBNC neurons responsive to jaw muscle (Ikeda et al. 2003; Sugiyo et al. 2005) or TMJ injury (Yamazaki et al. 2008) projected to higher brain centers associated with nociceptive processing; however, only male animals were included in these studies.
The aim of the present study was to determine if ascending projections from TMJ-responsive neurons in the TBNC displayed sex differences. Cell counts were made after Fluorogold (FG) injections into the ventrolateral periaqueductal gray (vlPAG) or posterior thalamus (Po) in adult male and female rats. The PAG-rostroventromedial medulla (RVM) complex plays a pivotal role in pain modulation (Tracey and Mantyh 2007), including pain evoked by stimulation of jaw muscles (Kupers et al. 2004) and facial skin (Mainero et al. 2007). The vlPAG preferentially integrates noxious sensory input from deep somatic and visceral tissues (Clement et al. 2000) and, more recently, the anatomical and functional organization of vlPAG-RVM pathways was shown to display sex-related differences that may underlie sexually dimorphic responses to opioid analgesics in spinal pain models (Loyd and Murphy 2006). The Po receives significant direct input from the TBNC (Iwata et al. 1992; Gauriau and Bernard 2004a). Spinothalamic input to Po derives mainly from nociceptive neurons in superficial and deep laminae (Zhang and Giesler 2005), while Po neurons that encode noxious sensory information project strongly to cortical regions that integrate affective and sensory aspects of pain (Gauriau and Bernard 2004b). Stimulation of specialized craniofacial tissues such as tooth pulp (Zhang et al. 2006) and dura (Burstein et al. 2010) activate Po neurons, while cornea- (Hirata et al. 2000) and TMJ-responsive neurons (Takeshita et al. 2001) recorded from superficial laminae of trigeminal subnucleus caudalis (Vc) are driven antidromically by stimulation sites in the Po, but not from sites in the ventral posterior medial thalamic nucleus (VPM). Although considerable evidence suggests that the vlPAG and Po contribute to the proposed “TMJ pain matrix” in the brain (Bereiter and Okamoto 2011), it is not known if ascending projections from TMJ-responsive neurons in the lower brainstem to these regions are similar in males and females.
EXPERIMENTAL PROCEDURES
The animal protocols were approved by the Institutional Animal Care and Use Committee of the University of Minnesota and conformed to the established guidelines set by The National Institutes of Health guide for the care the use of laboratory animals (PHS Law 99–158, revised 2002).
Estradiol (E2) treatment in females
Ovariectomized (OvX) female rats were injected daily with 17β-estradiol-3-benzoate (Sigma, St. Louis, MO, USA), (E2, 3 µg/kg) dissolved in 200 µl sesame oil for 2 days prior to the day of perfusion. Our rationale for comparing males to low E2-treated females was two-fold. First, we were interested in sex differences in projections of TMJ-responsive neurons rather than the effect of estrogen status per se. Females under low estrogen conditions and males produced similar Fos-like immunoreactivity (Fos-LI) responses in TBNC regions after TMJ stimulation (Bereiter 2001). Second, E2 was given as a low maintenance dose to normalize the elevation of hypothalamic and pituitary hormones resulting from OvX surgery (Ajika et al. 1972). Recent studies in humans indicated that elevated levels of pituitary hormones significantly influenced pain sensation (Tajar et al. 2011). In some cases blood samples were collected at the time of perfusion and measured for E2 by immunoassay (Invitrogen, Camarillo, CA). Plasma E2 levels averaged 6.3 ± 0.9 pg/ml for males (n = 4) and 7.6 ± 1.2 pg/ml for OvX E2-treated females (n = 7). Vaginal smears were assessed from all female rats and a low E2 status (>90% small nucleated leukocytes) was confirmed on the day of perfusion.
Animal preparation and Fluorogold tracer injection
Weight-matched (280–390 g) female (n = 16) and male rats (n = 18, Sprague-Dawley, Harlan, Indianapolis, IN) were anesthetized with pentobarbital sodium (55 mg/kg, ip), placed in a stereotaxic frame and the skull positioned so that the bregma and lambda landmarks had equal dorsoventral coordinates. Lidocaine (2% 0.1 ml) was injected into the scalp prior to exposing the skull by dorsal incision and performing a craniotomy over the right hemisphere (2 × 2 mm). A small opening was cut in the dura and a glass micropipette (40–80 µm tip diameter) filled with 4% Fluorogold (FG, Fluorochrome, LLC, Denver, CO), dissolved in sterile saline, was lowered into position to the posterior thalamus/ triangular area (Po/PoT; male, n = 8; female, n = 8) or the caudal ventrolateral/lateral periaqueductal gray area (vlPAG; male, n = 10; female, n = 8). The stereotaxic coordinates according to Paxinos and Watson (1997) were: Po, 3.2–4.8 mm caudal to bregma, 2.1–2.4 mm lateral to the midline and depth of 5.0–5.4 mm; and PAG, 7.3–8.0 mm caudal to bregma and 0.8–1.0mm lateral to midline, depth 5.1–5.7 mm. A single FG (50–80 nl) injection was delivered slowly (~10 nl/min) into the target area from a 1 µl microsyringe. The micropipette was left in the place for 30 min after injection and the scalp wound was washed with sterile saline and closed with nylon suture.
Temporomandibular joint (TMJ) stimulation
Seven days after FG injection, rats were anesthetized with pentobarbital sodium (60 mg/kg) and half of the animals from each FG injection group was prepared for local stimulation of the TMJ region, while the remaining half remained anesthetized for 2 h without further treatment (referred to as “controls” in the text). For rats receiving TMJ stimulation, the preauricular skin overlying the left TMJ was shaved and a small skin incision was made. The selective small fiber excitant, mustard oil (MO, 20% allyl isothiocyanate in mineral oil, 25 µl), was injected into the TMJ via a 26-gauge needle. Two hours after MO injection, rats were given a bolus injection of pentobarbital sodium (100 mg/kg, i.p.) and perfused transcardially with 100 ml saline, followed by 4% paraformaldehyde in cold 0.01M phosphate buffered saline (PBS, pH 7.4). The brain and brainstem including upper cervical spinal cord were removed, post-fixed for 2 h and transferred to 30% sucrose (w/v) in 0.01M PBS for 2–3 days at 4°C.
Fos protein/FG double labeling
Transverse frozen sections (50 µm) were cut at the level of the posterior diencephalon and midbrain to identify injection sites. At the lower brainstem, alternate sections from 2 mm rostral to the obex through the C2 spinal segment were saved, transferred serially to multiwell tissue culture plates containing 0.01M PBS and processed first for the protein product of the immediate early gene, c-fos (Fos-LI), and then for FG by immunohistochemistry. Sections were incubated in 1% hydrogen peroxide with 0.75% Triton X-100 (Sigma, St. Louis, MO) in PBS for 90 min, 3% normal goat serum (Sigma) in PBS for 30 min, and reacted with polyclonal rabbit anti-Fos antibody (Ab-5; 1:15,000 dilution, Calbiochem, Gibbstown, NJ) overnight at room temperature with gentle agitation. To visualize Fos-LI, sections were incubated with goat anti-rabbit biotinylated secondary IgG (1:400, 90 min, Vector, Burlingame, CA), peroxidase-conjugated avidin-biotin complex (1:100, 60 min, Vectastain ABC Kit; Vector), 0.04% diaminobenzidine-tetra (DAB substrate Kit DAB-Ni; Vector), 0.2% nickel ammonium sulfate (DAB substrate Kit DAB-Ni; Vector) and 0.006% hydrogen peroxide for 10 min. After Fos-LI, sections were rinsed in PBS followed by 3% normal goat serum in PBS for 30 min, and incubated for overnight with rabbit polyclonal anti-FG antibody (1:20,000, Chemicon, Temecula, CA) at 4 °C. Sections then were washed in PBS (3x), incubated with goat anti-rabbit biotinylated secondary IgG (1:400, 90 min, Vector), peroxidase-conjugated avidin-biotin complex (1:100, 60 min, Vectastain ABC Kit; Vector) at room temperature, 0.04% diaminobenzidine-tetra (DAB substrate Kit DAB-Ni; Vector) and 0.006% hydrogen peroxide without nickel ammonium sulfate for 10 minutes. Sections were rinsed in PBS, mounted on gelatin-coated slides, dehydrated and coverslipped. Fos-LI appeared as darkly stained regularly shaped nuclei, while FG appeared as a brown cytoplasmic reaction product. Omission of primary antisera eliminated specific staining for Fos-LI and FG. Photomicrograph images were captured (Olympus model BX51, Central Valley, PA) and adjusted in Adobe Photoshop 7.0 for brightness and contrast balance. Microinjection sites were reconstructed from fluorescent-labeled tissue (DAPI filter, U-MWU2, Olympus) using an X-Cite epifluorescent attachment (EXFO, Mississauga, Ont).
Data analysis
The sections were mounted serially on slides and cell counts for Fos-LI and FG-positive neurons were made without prior knowledge of treatment. Transverse sections (60–70 per rat) were categorized according to the rostrocaudal level, from +2 mm to −6 mm, relative to the obex. The obex is a surface landmark defined by the caudal end of the fourth ventricle approximately 500 µm rostral to the most caudal tip of the trigeminal subnucleus interpolaris (Yoshida et al. 1991). Rostrocaudal levels were determined based on well-established structural landmarks for the lower brainstem and upper cervical spinal cord of adult rats (Molander et al. 1989; Yoshida et al. 1991; Strassman and Vos, 1993).
Fos- and FG-positive cells were counted at X 40 magnification. Cells counted as Fos-positive had a black regularly shaped nucleus, whereas cells counted as FG-positive had a well defined nucleus, that may or may not have expressed Fos-LI, surrounded by a brown-stained peri-nuclear cytoplasmic region with dendritic processes. Since section thickness (50 µm) exceeded by more than 1.5 the estimated soma size for neurons in the brain regions of interest, no correction was made for cell counts (Clarke 1992). The average number of Fos-and FG-positive cells per section was compared for the following trigeminal brainstem regions: 1) subnucleus interpolaris (Vi, +2 to +1 mm relative to obex); 2) dorsal paratrigeminal region (dPa5, +0.5 to −0.5 mm relative to obex), comprised of cells along the dorsolateral edge of the main body of TBNC plus a nuclear grouping within the spinal trigeminal tract; 3) dorsal half at the subnucleus interpolaris/caudalis transition region (dm-Vi/Vc, +0.5 to −0.5 mm to obex), the most rostral pole of Vc (Yoshida et al. 1991); 4) ventral Vi/Vc transition region (vl-Vi/Vc,, +0.5 to −0.5 mm); 5) main portion of Vc (mid-Vc, −1 to −4 mm); and 6) upper cervical dorsal horn (C1–2, −5 to −6 mm). At the Vc and C1–2 regions separate counts were made for cells in superficial (I–II) and deeper laminae (III–V) according to established landmarks (Molander et al. 1989). Separate counts were made for cells in nucleus tractus solitarii (NTS, +0.5 to −0.5 mm to obex) and caudal ventrolateral medulla (CVLM, +0.5 to −0.5 mm to obex). Total cell counts were assessed per 1 mm (average cell count per 50 µm section × 20, see Figs 1 and 4) or per brain region (cell counts per 50 µm section × 20 × rostrocaudal dimension in mm, see Tables 1, 2 and 3). Statistical comparisons were made by ANOVA and individual comparisons by Newman-Keuls after ANOVA.
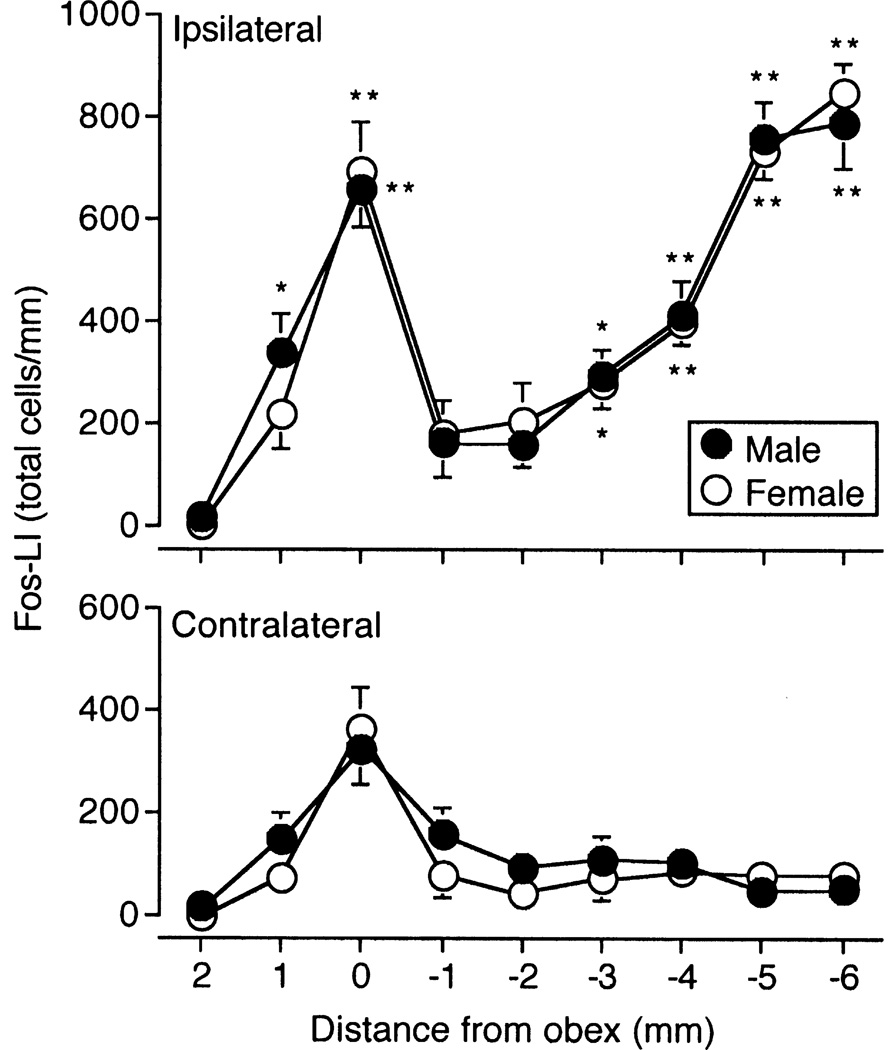
Figure 1.
Average number of Fos-positive neurons produced in the caudal trigeminal brainstem nuclear complex (TBNC) and upper cervical dorsal horn after unilateral injection of the small fiber excitant, mustard oil (20% solution, 25 µl), into the temporomandibular joint (TMJ) region of male and female rats. Results expressed as average total number of cells per mm (± SEM) as a function of distance from the obex. *P < 0.05, **P < 0.01 ipsilateral versus contralateral. N = 8–10 rats per group.
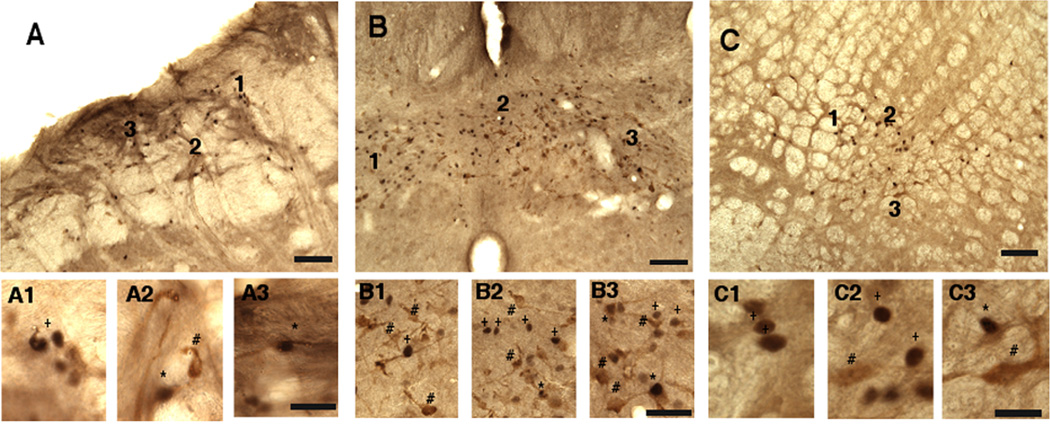
Figure 4.
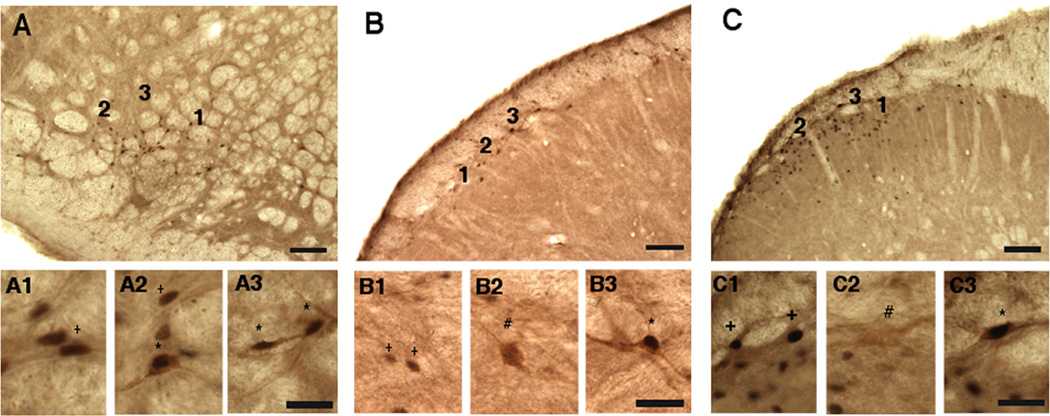
Examples of Fos, FG and Fos/FG double-labeled neurons from a male rat after vlPAG injection of FG and TMJ stimulation with mustard oil. A. Field view of the dorsal paratrigeminal region (dPa5). A1–3: enlarged images of numbered regions in A. B. Field view of the commissural nucleus tractus solitarii (NTS). B1–3: enlarged images of numbered regions in B. C. Field view of the caudal ventrolateral medulla (CVLM). C1–3: enlarged images of numbered regions in C. Symbols: + = Fos, # = FG, and *= Fos/FG double-labeled neuron. All examples are from the side ipsilateral to the TMJ stimulus. Calibration: low magnification (A, B, C) = 100 µm; high magnification (A1, A2, and A3) = 50 µm.
Table 1.
Total cell counts for Fos/FG double-labeled, Fos-positive, and FG-positive neurons in rats that received Fluorogold injection into ventrolateral PAG and TMJ stimulation.
| Vi | dm-Vi/Vc | vl-Vi/Vc | dPa5 | |
|---|---|---|---|---|
| Male-ipsi | ||||
| Fos/FG | 6.2 ± 2.6 | 2.1 ± 1.0 | 8.0 ± 1.7 | 2.8 ± 1.3 |
| Fos | 356 ± 93.1 | 200.3 ± 35.3 | 193 ± 47.8 | 254 ± 56.8 |
| FG | 472 ± 51.8 | 105.7 ± 11.5 | 69.3 ± 6.0 | 9.33 ± 3.0 |
| Female-ipsi | ||||
| Fos/FG | 8.0 ± 2.8 | 1.1 ± 0.6 | 19.0 ± 5.5 a | 12.0 ± 4.3 b |
| Fos | 199 ± 116.5 | 117 ± 18.7 | 277 ± 18.7 | 290 ± 56.7 |
| FG | 764 ± 50.3 b | 131 ± 10.4 | 93.0 ± 10.7 a | 41.0 ± 14.2 a |
| Male-contra | ||||
| Fos/FG | 0.0 ± 0.0 | 0.8 ± 1.0 | 3.1 ± 2.2 | 0.0 ± 0.0 |
| Fos | 172 ± 65.2 ** | 94.1 ± 21.4 ** | 74.3 ± 17.4 * | 156 ± 37.3 |
| FG | 370 ± 70.6 | 79.3 ± 16.2 | 48.7 ± 3.0 * | 5.3 ± 1.7 |
| Female-contra | ||||
| Fos/FG | 1.3 ± 1.0 | 0.7 ± 0.5 | 5.1 ± 1.3 ** | 3.1 ± 1.9 ** |
| Fos | 64.0 ± 32.1 | 92.5 ± 42.1 | 65.5 ± 5.7 ** | 179 ± 23.6 |
| FG | 355 ± 81.4 ** | 92.0 ± 36.7 * | 58.5 ± 10.8 ** | 27.0 ± 10.3 * |
| Vc I–II | Vc III–V | C1–2 I–II | C1–2 III–V | |
| Male-ipsi | ||||
| Fos/FG | 62.3 ± 24.3 | 2.0 ± 0.1 | 48.7 ± 10.2 | 0.7 ± 0.7 |
| Fos | 814 ± 252 | 149 ± 7.3 | 1415 ± 116 | 194.7 ± 23.0 |
| FG | 412 ± 45.4 | 165 ± 22.0 | 103 ± 23.7 | 173.3 ± 21.6 |
| Female-ipsi | ||||
| Fos/FG | 84.0 ± 34.7 | 0.0 ± 0.0 | 46.0 ± 7.4 | 0.0 ± 0.0 |
| Fos | 1082 ± 323 | 255 ± 27.2 | 1572 ± 23.4 | 205 ± 23.8 |
| FG | 433 ± 23.4 | 298 ± 29.5 b | 130 ± 20.8 | 165 ± 11.2 |
| Male-contra | ||||
| Fos/FG | 18.0 ± 10.9 * | 0.0 ± 0.0 | 2.2 ± 0.9 ** | 0.0 ± 0.0 |
| Fos | 335 ± 46.4 * | 64.0 ± 9.8 | 138 ± 24.5 ** | 48.7 ± 5.4 |
| FG | 210 ± 18.8 ** | 64.1 ± 9.6 ** | 18.7 ± 9.6 ** | 63.3 ± 3.2 ** |
| Female-contra | ||||
| Fos/FG | 7.4 ± 5.7 ** | 0.0 ± 0.0 | 6.1 ± 3.5 ** | 0.0 ± 0.0 |
| Fos | 208 ± 52.6 ** | 84.0 ± 6.6 | 168 ± 27.7 ** | 60.0 ± 2.6 |
| FG | 227 ± 89.1 * | 93.0 ± 10.1 ** | 40.0 ± 14.9 ** | 66.0 ± 5.5 ** |
Total cell counts per brain region, mean ± SEM. See Methods for number of sections/ CNS region. N = 4–6 rats per treatment group. Ipsi = ipsilateral to TMJ stimulus (left side); contra = contralateral to TMJ stimulus (right side). FG injected into PAG contralateral to TMJ stimulus.
P < 0.05,
P < 0.01 ipsilateral versus contralateral;
= P < 0.05,
= P < 0.01 male versus female.
Table 2.
Total cell counts for Fos/FG double-labeled, Fos-positive, and FG-positive neurons for rats that received Fluorogold injection into posterior thalamus and TMJ stimulation.
| Vi | dm-Vi/Vc | vl-Vi/Vc | dPa5 | |
|---|---|---|---|---|
| Male-ipsi | ||||
| Fos/FG | 12.1 ± 5.9 | 5.6 ± 4.3 | 6.6 ± 3.0 | 6.5 ± 4.0 |
| Fos | 330 ± 146 | 178 ± 43.4 | 225 ± 49.3 | 182 ± 22.8 |
| FG | 1987 ± 354 | 551 ± 105 | 171 ± 13.1 | 64.0 ± 25.5 |
| Female-ipsi | ||||
| Fos/FG | 0.0 ± 0.0 | 0.6 ± 0.5 | 6.0 ± 1.8 | 10.1 ± 4.2 |
| Fos | 49.0 ± 31.7 | 65.5 ± 21.5 | 160.0 ± 45.3 | 211 ± 9.0 a |
| FG | 1631 ± 629 | 536 ± 185 | 124.0 ± 45.8 | 56.0 ± 7.5 |
| Male-contra | ||||
| Fos/FG | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.7 ± 0.1 | 0.0 ± 0.0 * |
| Fos | 146 ± 104 | 105 ± 43.7 * | 103 ± 65.5 * | 32.0 ± 16.6 ** |
| FG | 160 ± 84.9 * | 72.9 ± 16.5 * | 24.5 ± 10.8 ** | 8.1 ± 3.3 ** |
| Female-contra | ||||
| Fos/FG | 1.3 ± 1.0 | 0.6 ± 0.5 | 2.2 ± 2.0 | 3.1 ± 1.2 * |
| Fos | 83.0 ± 69.8 | 56.5 ± 48.6 | 108 ± 34.7 | 69.1 ± 37.7 ** |
| FG | 250 ± 76.9 * | 57.0 ± 17.4 * | 42.0 ± 16.3 * | 26.0 ± 9.6 * |
| Vc I–II | Vc III–V | C1–2 I–II | C1–2 III–V | |
| Male-ipsi | ||||
| Fos/FG | 84.0 ± 13.1 b | 0.0 ± 0.0 | 53.0 ± 16.4 | 0.0 ± 0.0 |
| Fos | 830 ± 250 | 221 ± 22.7 | 1704 ± 252 | 269 ± 44.1 |
| FG | 693 ± 35.2 b | 393 ± 37.7 | 118 ± 17.9 a | 163 ± 20.1 a |
| Female-ipsi | ||||
| Fos/FG | 26.0 ± 3.5 | 0.0 ± 0.0 | 19.0 ± 5.9 | 0.0 ± 0.0 |
| Fos | 699 ± 72.8 | 197 ± 29.5 | 1591 ± 121 | 120 ± 10.7 |
| FG | 335 ± 112 | 295 ± 71.2 | 88.0 ± 2.3 | 106 ± 19.2 |
| Male-contra | ||||
| Fos/FG | 1.6 ± 1.6 ** | 0.0 ± 0.0 | 0.0 ± 0.0 ** | 0.0 ± 0.0 |
| Fos | 213 ± 60.9 ** | 86.0 ± 3.4 | 68.0 ± 42.0 ** | 47.0 ± 2.8 |
| FG | 90.0 ± 11.6 ** | 50.0 ± 3.4 ** | 9.1 ± 5.9 ** | 42.0 ± 5.0 ** |
| Female-contra | ||||
| Fos/FG | 1.6 ± 1.0 ** | 0.0 ± 0.0 | 2.0 ± 1.2 ** | 0.0 ± 0.0 |
| Fos | 180 ± 54.5 ** | 82.0 ± 8.1 | 129 ± 16.7 ** | 55.1 ± 2.6 |
| FG | 122 ± 22.9 * | 75.1 ± 11.1 ** | 8.2 ± 3.4 ** | 64.8 ± 10.0 |
Total cell counts per brain region, mean ± SEM. See Methods for number of sections/CNS region. N = 4–6 rats per treatment group. Ipsi = ipsilateral to TMJ stimulus (left side); contra = contralateral to TMJ stimulus (right side). FG injected into PO contralateral to TMJ stimulus.
P < 0.05,
P < 0.01 ipsilateral versus contralateral;
= P < 0.05,
= P < 0.01 male versus female.
Table 3.
Total cell counts for Fos/FG double-labeled, Fos-positive, and FG-positive neurons in the CVLM and NTS after Fluorogold injections into vlPAG or PO and TMJ stimulation.
| PAG | CVLM | NTS |
|---|---|---|
| Male-ipsi | ||
| Fos/FG | 98.0 ± 12.1 b | 48.0 ± 11.4 a |
| Fos | 369 ± 31.5 | 515 ± 57.3 |
| FG | 211 ± 23.7 | 197 ± 47.3 |
| Female-ipsi | ||
| Fos/FG | 86.0 ± 12.4 a | 60.0 ± 10.2 a |
| Fos | 350 ± 11.9 | 600 ± 119 |
| FG | 228 ± 25.9 | 282 ± 84.7 |
| Male-contra | ||
| Fos/FG | 82.7 ± 12.6 a | 107 ± 28.3 b ** |
| Fos | 293 ± 27.5 | 595 ± 89.6 |
| FG | 157 ± 16.1 * | 493 ± 118 ** |
| Female-contra | ||
| Fos/FG | 83.0 ± 22.2 a | 152 ± 9.4 b ** |
| Fos | 329 ± 16.7 | 615 ± 98.6 |
| FG | 202 ± 25.8 | 644 ± 116 ** |
| PO | CVLM | NTS |
| Male-ipsi | ||
| Fos/FG | 37.0 ± 9.2 | 12.0 ± 2.3 |
| Fos | 406 ± 19.4 | 437 ± 18.4 |
| FG | 100 ± 25.1 | 73.0 ± 13.4 |
| Female-ipsi | ||
| Fos/FG | 32.0 ± 1.6 | 12.0 ± 1.6 |
| Fos | 410 ± 29.1 | 424 ± 142 |
| FG | 92.4 ± 6.6 | 61.0 ± 7.7 |
| Male-contra | ||
| Fos/FG | 21.0 ± 5.7 * | 24.0 ± 8.2 * |
| Fos | 355 ± 41.6 | 415 ± 46.0 |
| FG | 66.0 ± 15.7 | 103 ± 35.2 |
| Female-contra | ||
| Fos/FG | 22.0 ± 2.6 * | 24.0 ± 4.3 |
| Fos | 416 ± 63.3 | 420 ± 115 |
| FG | 69.0 ± 2.5 | 80.0 ± 21.5 |
Total cell counts per brain region, mean ± SEM. See Methods for number of sections/CNS region. N = 4–6 rats per treatment group. Ipsi = ipsilateral to TMJ stimulus (left side); contra = contralateral to TMJ stimulus (right side). FG injected into PAG and PO on the right side.
P < 0.01 ipsilateral versus contralateral;
= P < 0.05,
= P < 0.01 versus similar group in Po injection series.
Results
Fos-like immunoreactivity (Fos-LI) produced by TMJ stimulation
Injection of the small fiber excitant, MO, into the left TMJ produced a distinct rostrocaudal distribution of Fos-positive neurons within the ipsilateral (Fig 1, upper panel) and contralateral TBNC (Fig 1, lower panel) and was equivalent for males and females (F1,16 = 0.11, P > 0.10). This pattern of Fos expression was consistent with earlier results (Bereiter 2001; Okamoto et al. 2008) and confirmed that the low dose E2 regimen used here was not sufficient to enhance TMJ-evoked Fos-LI in the TBNC. Analyses of TMJ-evoked Fos-LI within individual brainstem regions found only minor sex differences (Tables 1, 2 and 3). Controls, i.e., animals that received only FG injections into vlPAG or Po, but no TMJ stimulus, had only scattered Fos-positive neurons in the TBNC, mainly at vl-Vi/Vc, and only rarely were any of these neurons also FG-positive (data not shown).
FG-labeled neurons after vlPAG injections
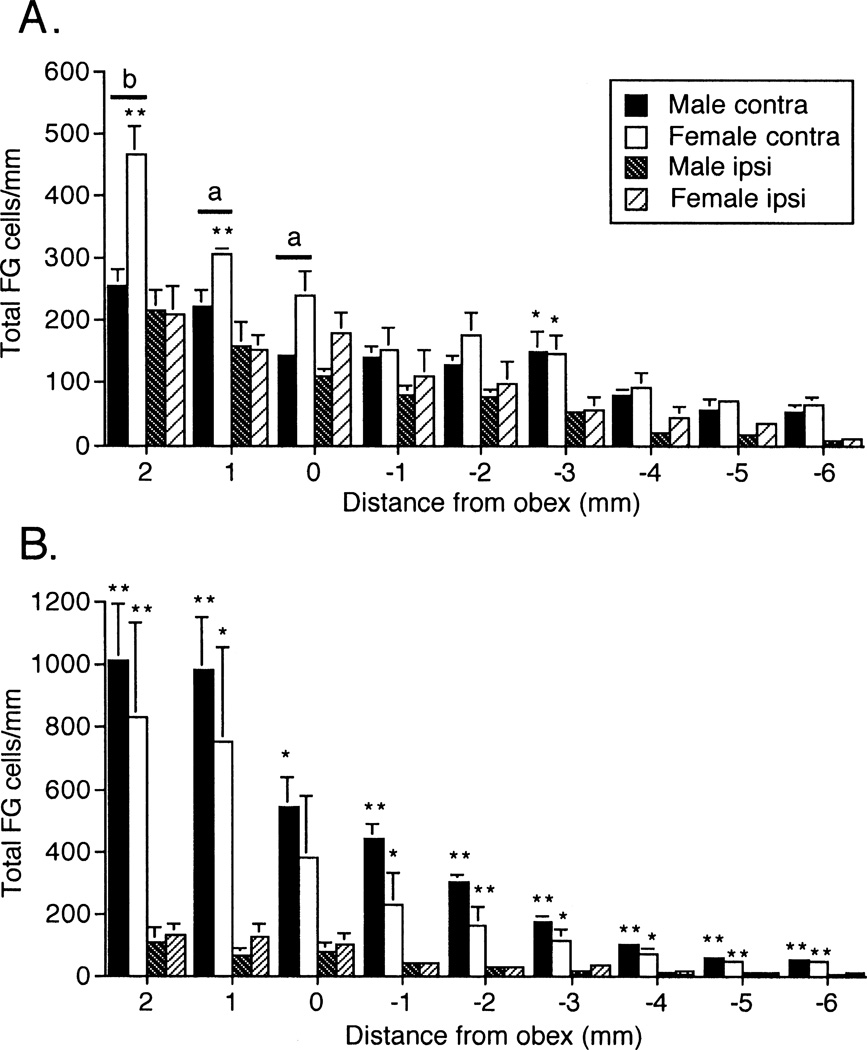
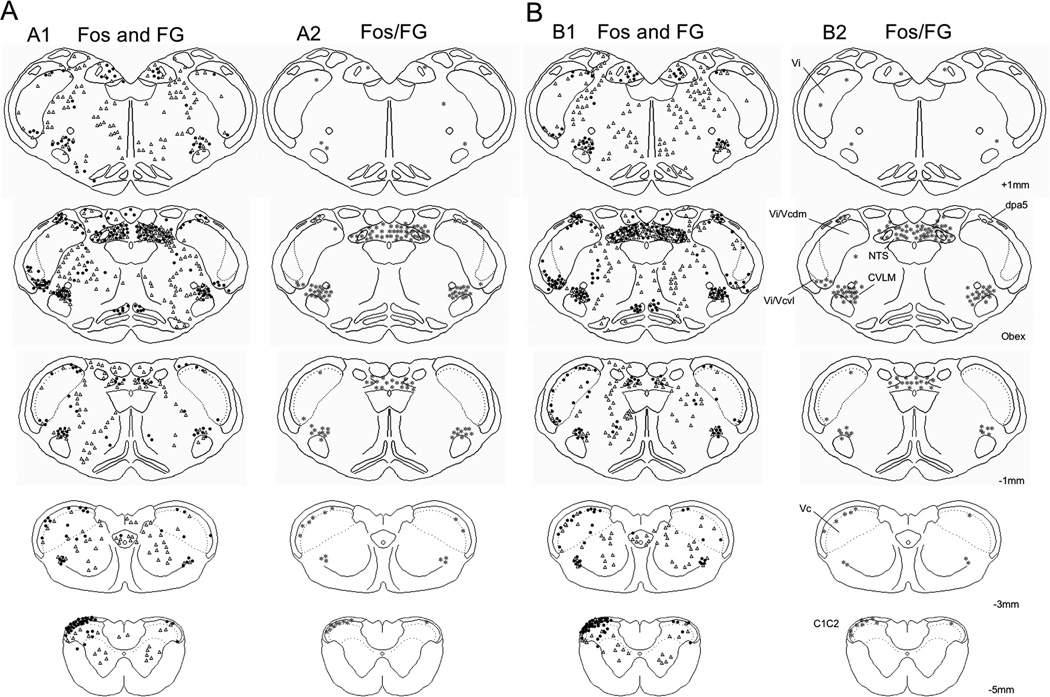
Microinjections of FG were directed at the vlPAG of the right hemisphere; however, as seen in Fig 2A (see example in Fig 2B), FG spread to regions immediately dorsal to the vlPAG in most cases. The greatest number of FG neurons was seen at TBNC regions near the level of the obex, especially at caudal Vi, and although higher counts were found contralateral to the injection, substantial numbers of FG neurons were seen ipsilaterally (Fig 3A). Analyses of cell counts within the TBNC across all rostrocaudal levels revealed significantly more FG neurons in females than males contralateral (F1,8 = 7.04, P < 0.05), but not ipsilateral (F1,8 = 0.58, P > 0.10) to the vlPAG injection. Within specific TNBC regions contralateral to FG injections females displayed greater numbers in Vi (F1,14 = 9.53, P < 0.01), vl-Vi/Vc (F1,14 = 7.24, P < 0.025, Fig 5A), dPa5 (F1,10 = 9.55, P < 0.025, Fig 4A) and in deep laminae of Vc (F1,8 = 15.08, P < 0.005) than males. No sex differences were seen for FG-labeled neurons in superficial laminae at the Vc and C1–2 regions after vlPAG injections (Table 1, see examples in Figs 5B, 5C). Many FG neurons were found bilaterally in the CVLM and NTS (see examples in Figs 4B, 4C) and in similar numbers for males and females (Table 3). Numerous FG-labeled neurons also were found medial to the TBNC in the lower brainstem after vlPAG injections but were not quantified (Figs 6A1 6B1). The micrographic examples shown in Figures 4 and 5 were from males, however, no qualitative differences in the appearance or general distribution of FG- and/or Fos-labeled neurons were seen in either the vlPAG or Po injection series between males and females.
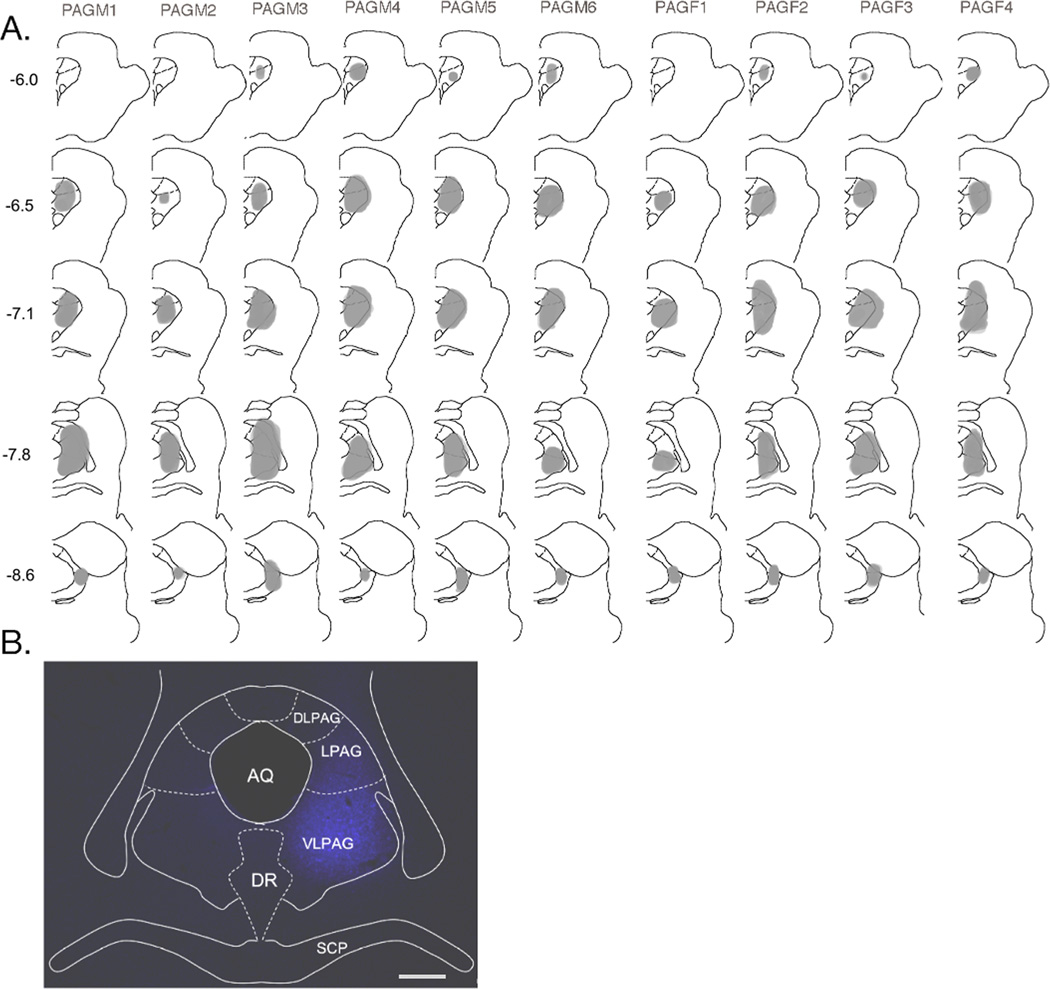
Figure 2.
Fluorogold (FG) injection sites in the periaqueductal gray (PAG). A. Drawings display the spread of FG (shaded regions) for injections in males (PAGM1–6) and female rats (PAGF1–4). Each vertical column represents the results for one experimental animal. Numbers to the left of the first column indicate the rostrocaudal distance from bregma according to the rat atlas of Paxinos and Watson (1997). B. Micrograph example of an injection site in the PAG. Abbreviations: AQ, aqueduct; DR, dorsal raphe; DLPAG, dorsolateral PAG; LPAG, lateral PAG; SCP, superior cerebellar peduncle; VLPAG, ventrolateral PAG. Calibration = 200 µm.
Figure 3.
Average total number of FG-positive neurons per mm within the TBNC and upper cervical dorsal horn of male and female rats. A. Total number of FG neurons after vlPAG injections of FG. B. Total number of FG neurons after PO injections of FG. Abbreviations: contra, contralateral to injection; ipsi, ipsilateral to injection. *P < 0.05, **P < 0.01 ipsilateral versus contralateral; a = P < 0.05, b = P < 0.01 male versus female. N = 8–10 rats per group.
Figure 5.
Examples of Fos, FG and Fos/FG double-labeled neurons from a male rat after vlPAG injection of FG and TMJ stimulation with mustard oil. A. Field view of the ventral interpolaris/caudalis transition (vl-Vi/Vc) transition region. A1–3: enlarged images of numbered regions in A. B. Field view of superficial laminae of Vc. B1–3: enlarged images of numbered regions in B. C. Field view of the upper cervical dorsal horn. C1–3: enlarged images of numbered regions in C. Symbols: + = Fos, # = FG, and *= Fos/FG double-labeled neuron. All examples are from the side ipsilateral to the TMJ stimulus. Calibration: low magnification (A, B, C) = 100 µm; high magnification (A1, A2, and A3) = 50 µm.
Figure 6.
Camera lucida drawing of an example of Fos, FG and Fos/FG double-labeled neurons in the caudal brainstem and upper cervical spinal cord after FG injection into the vlPAG. A. Example from a male rat. A1, Fos (●) and FG (Δ) labeling; A2, Fos/FG double-labeled neurons (*). B. Example from a female rat. B1, Fos (●) and FG (Δ) labeling; B2, Fos/FG double-labeled neurons (*). Numbers to the side of the right most column refer to the distance in mm relative to obex. All drawings are from the side ipsilateral to the TMJ stimulus.
FG-labeled neurons after Po injections
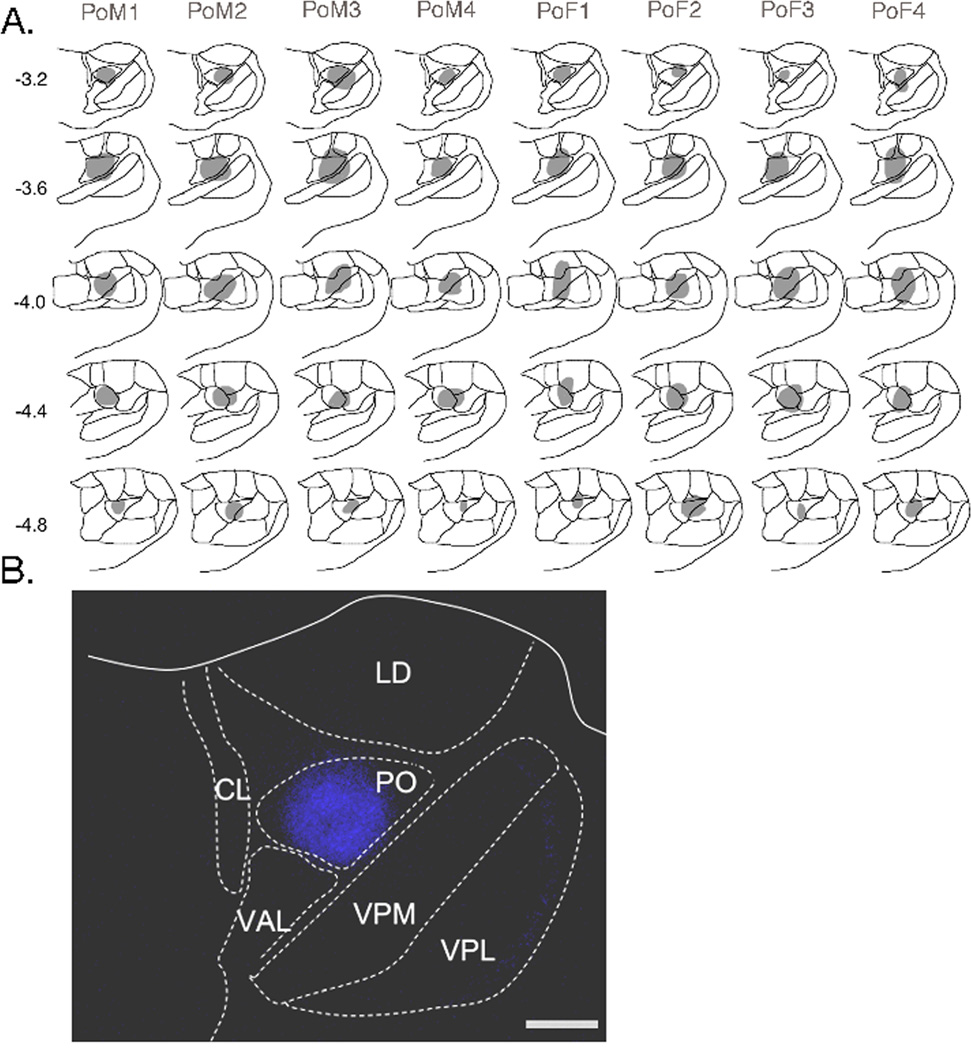
Microinjections of FG directed at Po also included the posterior thalamic triangular area (PoT) in most cases (see Fig 8A and example in Fig 8B). The greatest number of FG-labeled neurons was found at periobex regions of the TBNC, especially Vi, with the vast majority being contralateral to the Po injection (Fig 3B). Comparison of FG cell counts across all rostrocaudal levels of the TBNC revealed no significant sex differences contralateral (F1,6 = 0.74, P > 0.10) or ipsilateral (F1,6 = 0.48, P > 0.10) relative to the Po injection. However, within specific TBNC regions contralateral to FG injections males had significantly more FG neurons in superficial laminae of Vc (F1,12 = 13.7, P < 0.005), superficial laminae at C1–2 (F1,12 = 5.1, P < 0.05) and deep laminae at C1–2 (F1,12 = 7.25, P < 0.025) than females. No sex differences were seen for FG-labeled neurons in periobex regions of the TBNC (Table 2). Many FG neurons were found bilaterally in the CVLM and NTS after Po injections and in equal numbers for males and females (Table 3).
Figure 8.
Fluorogold injection sites in posterior nucleus of the thalamus (Po). A. Drawings display the spread of FG (shaded regions) for injections in males (PoM1–4) and E2-treated female rats (PoF1–4). Each vertical column represents the results for one experimental animal. Numbers to the left of the first column indicate the rostrocaudal distance from bregma according to the rat atlas of Paxinos and Watson (1997). B. Micrograph example of an injection site in the Po. Abbreviations: CL, centrolateral thalamic n.; LD, lateral dorsal thalamic n.; Po, posterior thalamic n.; VAL, ventral anterolateral n.; VPM, ventral posterior medial thalamic n.; VPL, ventral posterior lateral thalamic n. Calibration = 200 µm.
The overall results of single-labeled FG neurons after vlPAG versus Po injections revealed several notable features. First, the average total number of FG-labeled neurons found bilaterally within the TBNC after Po injection (3245 ± 296 cells per rat, n = 16) was significantly greater than after vlPAG injection (2053 ± 149 cells per rat, n = 18; F1,32 = 13.8, P < 0.001). Second, the pattern of FG-labeled neurons within the TBNC indicated a predominantly contralateral projection to the Po, while the projection to the vlPAG was more bilateral. Third, the average total number of FG-labeled neurons found bilaterally within the CVLM after vlPAG injection (414 ± 23 cells per rat, n = 18) was more than twice that seen after Po injection (168 ± 13 cells per rat, n = 16, F1,32 = 81.1, P < 0.001). Similarly, the average total number of FG-labeled neurons found bilaterally within the NTS after vlPAG injection (796 ± 78 cells per rat, n = 18) was more than twice that seen after Po injections (225 ± 34 cells per rat, n = 16, F1,32 = 43.5, P < 0.001). As seen in Table 3 projections from the CVLM and NTS to either the vlPAG or Po were bilateral. Lastly, numerous FG-labeled neurons were seen in reticular areas medial to the TBNC after vlPAG injection (Fig 6A1, B1), whereas very few were seen in these areas after Po injection (Fig 9A1, B1).
Figure 9.
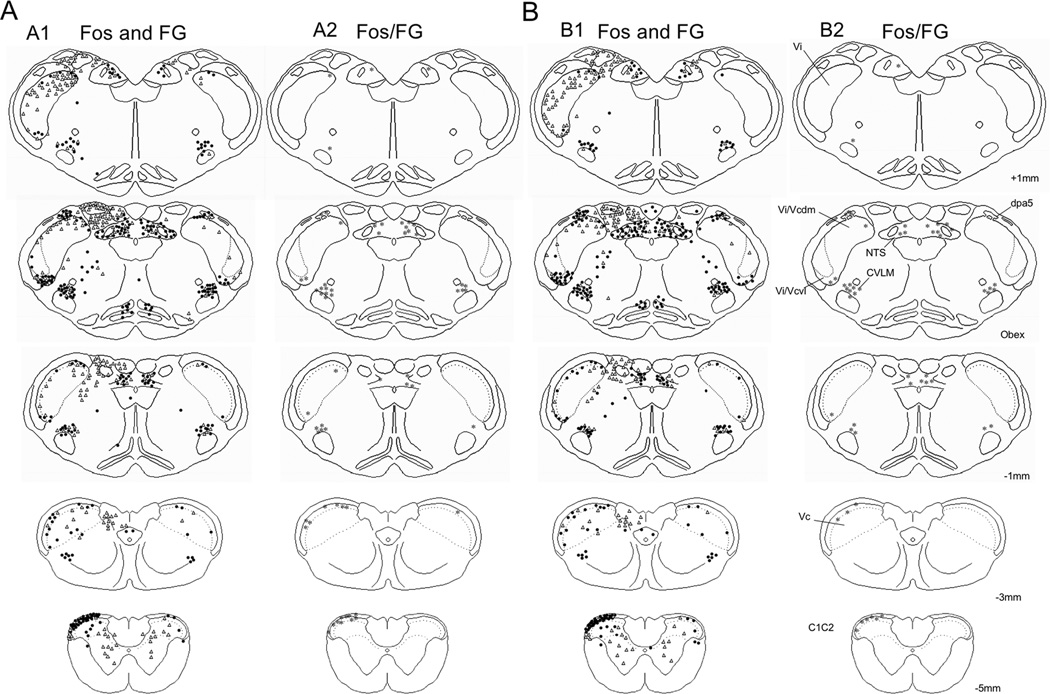
Camera lucida drawing of an example of Fos, FG and Fos/FG double-labeled neurons in the caudal brainstem and upper cervical spinal cord after FG injection into the PO. A. Example from a male rat. A1, Fos (●) and FG (Δ) labeling; A2, Fos/FG double-labeled neurons (*). B. Example from a female rat. B1, Fos (●) and FG (Δ) labeling; B2, Fos/FG double-labeled neurons (*). Numbers to the side of the right most column refer to the distance in mm relative to obex. All drawings are from the side ipsilateral to the TMJ stimulus.
Fos/FG double-labeled neurons after vlPAG injections
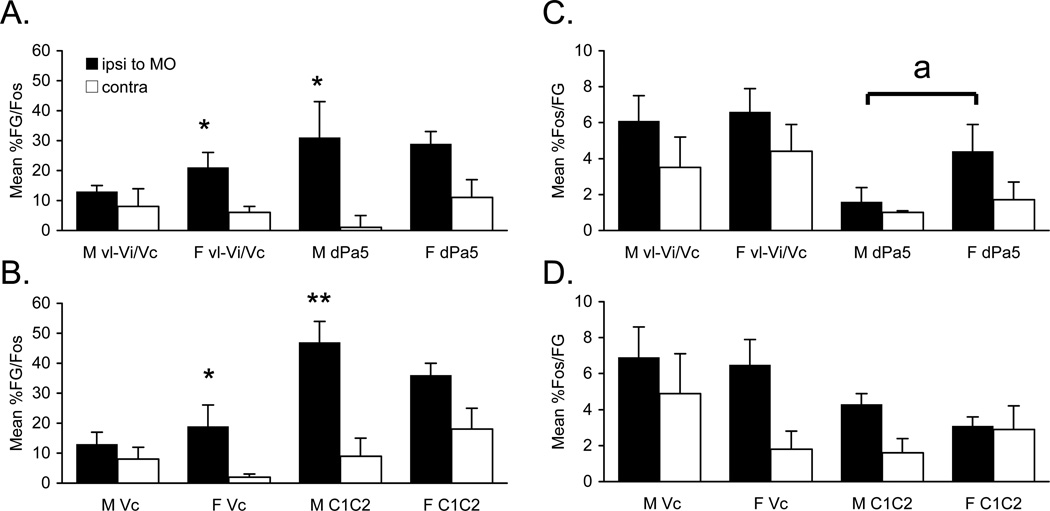
The number of Fos/FG double-labeled neurons after TMJ stimulation and vlPAG injection was greater in females than males at the vl-Vi/Vc transition (F1,14 = 6.59, P < 0.05) and the dPa5 region (F1,14 = 10.4, P < 0.01) ipsilateral to the TMJ stimulus, but not at other regions of the lower brainstem (Tables 1 and 3). However, when expressed as a percentage of FG neurons that also were Fos-positive there were no sex differences at any region (Fig 7A, 7B). Conversely, when Fos-positive neurons were expressed as a percentage of FG-labeled cells, the percentage found at the dPa5 was significantly greater in females than males (F1,14 = 5,59, P < 0.05; Fig 7C), despite a similar number of Fos-positive neurons alone. Numerous Fos/FG double-labeled neurons also were found at the CVLM and NTS (Figs 6A2, 6B2); however, no sex differences were noted for these regions (Table 3, upper panels). These data suggested that TMJ-responsive neurons at periobex regions of the TBNC were more likely to project to the vlPAG in females than males, whereas those at Vc and C1–2 were equally likely to project to vlPAG.
Figure 7.
Double-labeled cells after vlPAG injection and TMJ stimulation in males (M) and females (F). A. Percentage of FG-labeled neurons also labeled for Fos in vl-Vi/Vc and dPa5. B. Percentage of FG-labeled neurons also labeled for Fos in superficial laminae of Vc and C1–2. C. Percentage of Fos-positive neurons also labeled for FG in vl-Vi/Vc and dPa5. D. Percentage of Fos-positive neurons also labeled for FG in superficial laminae of Vc and C1–2. *P < 0.05, **P < 0.01 ipsilateral versus contralateral; a = P < 0.05 male versus female. Ipsi and contra refer to ipsilateral and contralateral to the TMJ stimulus.
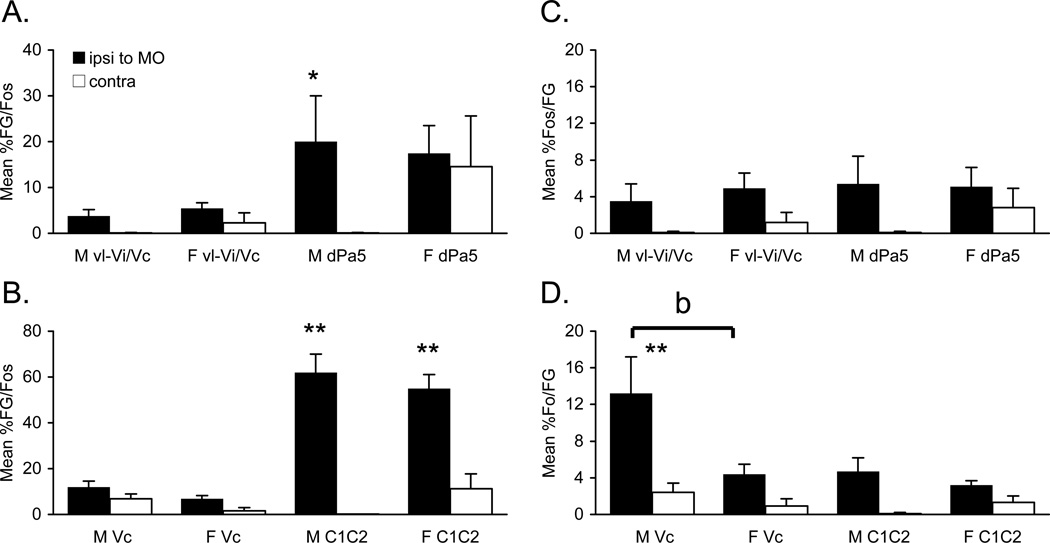
Fos/FG double-labeled neurons after Po injections
The number of Fos/FG double-labeled neurons seen after TMJ stimulation and Po injection was greater for males than females at superficial laminae of Vc (F1,12 = 24.7, P < 0.001) ipsilateral to the TMJ stimulus, but not at other regions of the lower brainstem (Tables 2 and 3). However, when expressed as a percentage of FG neurons that also were Fos-positive there were no sex differences at any region (Fig 10A, 10B). By contrast, the percentage of Fos-positive neurons at superficial laminae of Vc that also were FG-labeled cells was significantly higher in males than females (F1,12 = 24.7, P < 0.001; Fig 10C, 10D). As noted above for vlPAG injections, nearly all Fos/FG double-labeled neurons after Po injections located outside the borders of the TBNC were within the CVLM and NTS (Figs 9A2, 9B2). The number of Fos/FG double-labeled cells in CVLM and NTS was similar numbers for males and females (Table 3, lower panels). It was further noted that the number of Fos/FG double-labeled neurons in CVLM (F3,14 = 11.6, P < 0.001) and NTS (F3,14 = 9.05, P < 0.005) after Po injection was less than that seen after vlPAG injections. These data suggested that TMJ-responsive neurons in superficial laminae at Vc were more likely to project to Po in males than females, while projections from periobex regions of the TBNC were equally likely in males and females.
Figure 10.
Double-labeled cells after Po injection and TMJ stimulation in males (M) and females (F). A. Percentage of FG-labeled neurons also labeled for Fos in vl-Vi/Vc and dPa5. B. Percentage of FG-labeled neurons also labeled for Fos in superficial laminae of Vc and C1–2. C. Percentage of Fos-positive neurons also labeled for FG in vl-Vi/Vc and dPa5. D. Percentage of Fos-positive neurons also labeled for FG in superficial laminae of Vc and C1–2. *P < 0.05, **P < 0.01 ipsilateral versus contralateral. Ipsi and contra refer to ipsilateral and contralateral to the TMJ stimulus.
Discussion
These data indicated significant sex differences in number of neurons projecting to the vlPAG and Po from different levels of the TBNC. Projections to vlPAG from periobex regions of the TBNC (i.e., dPa5, vl-Vi/Vc) were significantly greater in females than males, whereas those projecting to the Po from more caudal portions of the TBNC (i.e., Vc and C1–2), were greater in males. Results from Fos/FG double labeling indicated TMJ-responsive neurons at the dPa5 were more likely to project to vlPAG in females than males, whereas TMJ-responsive neurons in superficial laminae at Vc were more likely to project to Po in males than females.
TMJD is the most common form of non-dental pain and cognitive, psychophysical and autonomic factors likely contribute to sensory dysfunction (Diatchenko et al. 2006; Maixner 2009). Animal experiments designed to investigate the basis for TMJ nociceptive processing in the lower brainstem have been limited by two main factors. First, the innervation of the TMJ region is complex consisting of trigeminal, vagal and upper cervical sensory fibers (Uddman et al. 1998; Casatti et al. 1999) that converge and terminate at multiple levels of the TBNC (Nishimori et al. 1986; Capra 1987; Shigenaga et al. 1988; Dessem et al. 2007). Fos-LI coupled with anatomical tracing is well suited for this analysis since it allows quantification and functional mapping of complex networks at the single neuron level (Hunt 1987; Menetrey et al. 1989). Second, despite evidence that persistent TMJD is more prevalent in females than males (see Bereiter and Okamoto 2011), the vast majority of studies have used only male animals. Sex-specific differences in modulatory pathways for trigeminal (Nag and Mokha 2006) and spinal nociception (Tershner et al. 2000; Mitrovic et al. 2003; Mogil et al. 2003) are well documented. However, few studies have compared ascending spinal pathways related to nociceptive processing for possible sex differences (Murphy et al. 2009) and none have involved trigeminal pathways.
The organization of afferent input to the TBNC is quite different from spinal cord in that trigeminal afferents project to multiple, and spatially discrete, regions of the TBNC, while spinal afferents terminate in contiguous spinal segments (Takemura et al. 2006; Bereiter et al. 2009). Several studies using Fos-LI as a neuronal activity marker found that acute stimulation of the TMJ region (Hathaway et al. 1995; Zhou et al. 1999) or masticatory muscles (Imbe et al. 1999; Ikeda et al. 2003; Ro et al. 2004) produced a distinct labeling pattern in the TBNC of male rats characterized by high density labeling in superficial laminae of caudal Vc and C1–2 ipsilateral to the stimulus, bilateral labeling at the vl-Vi/Vc transition and lesser, though significant, labeling in the ipsilateral dPa5. Although the functional significance of Fos-LI produced at these different TBNC regions is not yet certain, converging lines of evidence based on stimulus-effect curves, response to analgesic drugs and efferent projection targets have greatly aided data interpretation. Stimulus intensity encoding, seen as increased Fos-LI, was most pronounced in superficial laminae of Vc and C1–2, to a lesser extent at dPa5, but was not seen at the vl-Vi/Vc transition region (Bereiter 2001; Okamoto et al. 2008). Analgesic drugs markedly reduced Fos-LI in superficial laminae of Vc and C1–2 after TMJ or jaw muscle stimulation, while only weak or no reduction was seen at vl-Vi/Vc and dPa5 (Bereiter and Bereiter 2000; Ro et al. 2004). The present study found that ~12% of Fos-positive neurons in superficial laminae of Vc projected to the contralateral Po in males compared to < 4% of neurons in females. Previously, TMJ input to neurons in superficial but not deeper laminae of Vc/C1–2 was significantly affected by estrogen status based on Fos-LI (Bereiter 2001) and single unit recording data (Tashiro et al. 2007). Since we held estrogen status low in this study, a sex-specific difference in the magnitude of ascending projections from caudal Vc to Po was indicated. These data also revealed nearly twice the number of FG-labeled neurons in superficial compared to deep laminae at Vc and C1–2 in males, whereas FG-labeled neurons were more equally distributed across superficial and deeper laminae in females (see Table 2). Thus, one interpretation of these data was that ascending projections to Po from Vc were more widely distributed across laminae in females than males. Anterograde tracing studies also found that medullary or spinal dorsal horn projections to Po were more dense after injection into superficial versus deeper laminae in male rats (Iwata et al. 1992; Gauriau and Bernard 2004a). Similarly, antidromic stimulation of Po activated a higher percentage of spinal dorsal horn neurons in superficial than deep laminae in male rats (Zhang and Giesler 2005). Although neurons in superficial and deep laminae likely serve different aspects of nociception (Braz et al. 2005; Eckert et al. 2006), it is not yet known if sex differences impact this contribution. A notable finding was the dense projection to Po from Vc and C1–2 in this study versus the sparse projection to VPM seen after masseter muscle inflammation (Ikeda et al. 2003). This agreed well with previous recording studies in which antidromic stimulation in Po, but not in the ventral posterior medial thalamic nucleus, was able to activate TMJ-responsive neurons in superficial laminae of caudal Vc in male rats (Takeshita et al. 2001).
In contrast to results after Po injection in which projections from caudal laminated portions of the TBNC were generally greater in males, vlPAG injects resulted in greater FG-labeling of TBNC neurons, mainly at periobex levels, in females than males. Females displayed more single labeled FG neurons in caudal Vi, vl-Vi/Vc and dPa5 and more double-labeled Fos/FG neurons in vl-Vi/Vc and dPa5 than males. The present study also confirmed previous results that the marginal zone of caudal Vc projected substantially to the vlPAG in male rats (Menetrey et al. 1982; Clement et al. 2000; however, see Beitz 1982). The role of the vl-Vi/Vc in deep craniofacial pain remains uncertain. The Fos-LI response at the vl-Vi/Vc and caudal Vc after TMJ and masseter muscle stimulation differed markedly and suggests different contributions to nociception. First, intensity coding, as seen by enhanced Fos-LI with increasing TMJ stimulus intensity, was weak at the vl-Vi/Vc transition, while proportional increases were seen at the caudal Vc (Bereiter 2001; Okamoto et al. 2008). Second, analgesic drug treatment reduced slightly (Bereiter and Bereiter 2000) or not at all the Fos-LI response to noxious stimuli at vl-Vi/Vc, while Fos-LI at caudal Vc was markedly reduced (Ro et al. 2004; Okamoto et al. 2007). Third, TMJ-evoked Fos-LI at the vl-Vi/Vc transition was similar for males and females, while high estrogen states greatly enhanced Fos-LI at the caudal Vc (Bereiter 2001). Similarly, among OvX females treated with high or low dose E2, Fos-LI produced at caudal Vc after TMJ stimulation was enhanced compared low dose E2 treatment, while Fos-LI at the vl-Vi/Vc was not affected (Okamoto et al. 2008). Fourth, Fos-LI was produced bilaterally at the vl-Vi/Vc in the absence of overt TMJ or muscle stimulation by different anesthetic regimens (Strassman and Vos 1993; Zhou et al. 1999). Although a few Fos-positive neurons were produced in the absence of somatic stimulation, none were found to project to higher brain centers (Ikeda et al. 2003; present study). Collectively, these results suggested that TMJ-responsive neurons at the vl-Vi/Vc do not contribute directly to sensory-discriminative aspects of TMJ nociceptive processing.
Possible alternative roles for TMJ-responsive neurons at vl-Vi/Vc that project to vlPAG include recruitment endogenous pain controls and control of autonomic and homeostatic reflexes. Dysfunction in endogenous pain controls has been proposed as a contributing factor to pain expression in TMJD (King et al. 2009). Both the total number of single-labeled FG neurons and Fos/FG double-labeled neurons after vlPAG injection at the vl-Vi/Vc were greater in females than males (Table 1). Loyd and Murphy (2006) found that the magnitude of direct projections between the vlPAG and RVM was greater in females than males; however, interestingly, the magnitude of Fos/FG double-labeled PAG-RVM projection neurons after noxious hindlimb stimulation was greater in males. Jaw muscle inflammation produced Fos-LI at vl-Vi/Vc neurons, some of which projected directly to the RVM (Sugiyo et al. 2005) and thalamic nucleus submedius in male rats (Ikeda et al. 2003), consistent with the notion that the vl-Vi/Vc is involved in pain and/or autonomic modulation. Indeed, direct inhibition of vl-Vi/Vc reduced secondary hyperalgesia in male rats after jaw muscle inflammation (Shimizu et al. 2009), while blockade of the vl-Vi/Vc inhibited cardiorespiratory reflexes to nasal stimulation (Panneton and Yavari 1995) and prevented ocular surface stimulus-evoked lacrimation (Hirata et al. 2004). These studies suggested that the vl-Vi/Vc transition likely contributes to trigeminal nociception by recruitment of endogenous pain and autonomic control circuits.
The number of FG single-labeled and Fos/FG double-labeled neurons after vlPAG injection at the dPa5 was greater in females than males (Table 1). The afferent and efferent organization of the dPa5 region differs considerably from the vl-Vi/Vc transition in that it receives direct sensory input from trigeminal, upper cervical and vagus nerve fibers and projects to multiple medullary and pontine regions that control autonomic outflow including the NTS and CVLM (Menetrey and Basbaum 1987; Saxon and Hopkins 1998; Caous et al. 2001). Similar to the response at caudal Vc, TMJ-evoked Fos-LI at the dPa5 increased with greater stimulus intensity, was mainly ipsilateral to the stimulus and was enhanced by high estrogen conditions (Bereiter 2001; Okamoto et al. 2008). Lesion of the dPa5 prevented cardiovascular reflex responses to noxious stimuli (Yu et al. 2002), while recording studies found that dPa5 neurons encoded facial skin stimulus intensity and became sensitized after TMJ injury (Yamazaki et al. 2008). Although the dPa5 likely is critical for somatic autonomic integration in the trigeminal system (Saxon and Hopkins 1998; Caous et al. 2001), these studies were performed in male animals, and thus, it is not known if sex differences influence the contribution of dPa5 to TMJ nociceptive processing.
The significance of greater projections from periobex regions of the TBNC to the vlPAG in females compared to males is not known. However, control of endogenous pain regulatory systems and cardiovascular function are closely linked (Lovick 1993; Bruehl and Chung 2004) and altered control of autonomic function is a feature of several chronic pain conditions that are more prevalent in women than men such as Raynaud’s disease (Heidrich 2010) and complex regional pain syndrome (Schwartzman et al. 2009) in addition to TMJD (Bragdon et al. 2002; Mohn et al. 2008). In animal studies, trigeminal nerve-evoked changes in masseter muscle blood flow were significantly different in females and male rats (Ishii et al. 2009); however, the basis for this difference is not certain.
In summary, sex differences in FG-labeled neurons observed after vlPAG injections were mainly at periobex regions (Vi, vl-Vi/Vc, dPa5), while sex differences after Po injections were seen only in the laminated portions of the TBNC (Vc and C1–2). It is well established that women are more likely than men to develop several craniofacial pain conditions including TMJD (Bereiter and Okamoto 2011). Although the basis for this female prevalence is not known, the present study suggests that differences in ascending projections from second-order neurons in TBNC to higher brain centers that integrate nociceptive information may contribute.
Highlights.
Projections of TMJ-responsive brainstem neurons were different in males and females.
Projections to PAG were greater in females than males.
Projections to posterior thalamus were greater in males than females.
Acknowledgements
The authors thank Randall Thompson for excellent technical assistance. This study was supported by NIH grant DE12758 and the Office of Research on Women’s Health.
Abbreviations
- CVLM
caudal ventrolateral medulla
- C1–2
upper cervical spinal cord
- dPa5
dorsal paratrigeminal region
- E2
estradiol
- FG
Fluorogold
- Lam I/II
superficial laminae
- Lam III/V
deep laminae
- MO
mustard oil (allyl isothiocyanate)
- NTS
nucleus tractus solitarii
- OvX
ovariectomized
- Po
posterior thalamic nucleus
- RVM
rostroventromedial medulla
- TBNC
trigeminal brainstem nuclear complex
- TMJ
temporomandibular joint
- Vc
trigeminal subnucleus caudalis
- Vi
trigeminal subnucleus interpolaris
- Vi/Vc
trigeminal interpolaris/caudalis transition region
- vlPAG
ventrolateral periaqueductal gray
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Ajika K, Krulich L, Fawcett CP, McCann SM. Effects of estrogen on plasma and pituitary gonadotropins and prolactin, and on hypothalamic releasing and inhibiting factors. Neuroendocrinology. 1972;9:304–315. doi: 10.1159/000122062. [DOI] [PubMed] [Google Scholar]
- Bandler R, Shipley MT. Columnar organization in the midbrain periaqueductal gray: modules for emotional expression? Trends Neurosci. 1994;17:379–389. doi: 10.1016/0166-2236(94)90047-7. [DOI] [PubMed] [Google Scholar]
- Beitz AJ. The organization of afferent projections to the midbrain periaqueductal gray of the rat. Neuroscience. 1982;7:133–159. doi: 10.1016/0306-4522(82)90157-9. [DOI] [PubMed] [Google Scholar]
- Bereiter DA. Sex differences in brainstem neural activation after injury to the TMJ region. Cells Tissues Organs. 2001;169:226–237. doi: 10.1159/000047886. [DOI] [PubMed] [Google Scholar]
- Bereiter DA, Bereiter DF. Morphine and NMDA receptor antagonism reduce c-fos expression in spinal trigeminal nucleus produced by acute injury to the TMJ region. Pain. 2000;85:65–77. doi: 10.1016/s0304-3959(99)00246-8. [DOI] [PubMed] [Google Scholar]
- Bereiter DA, Hargreaves KM, Hu JW. Trigeminal mechanisms of nociception: peripheral and brainstem organization. In: Basbaum A, Bushnell MC, editors. Science of Pain. vol. 5. New York: Elsevier; 2009. pp. 435–460. [Google Scholar]
- Bereiter DA, Okamoto K. Neurobiology of estrogen status in deep craniofacial pain. Int Rev Neurobiol. 2011;97:251–284. doi: 10.1016/B978-0-12-385198-7.00010-2. [DOI] [PubMed] [Google Scholar]
- Bragdon EE, Light KC, Costello NL, Sigurdsson A, Bunting S, Bhalang K, Maixner W. Group differences in pain modulation: pain-free women compared to pain-free men and to women with TMD. Pain. 2002;96:227–237. doi: 10.1016/S0304-3959(01)00451-1. [DOI] [PubMed] [Google Scholar]
- Braz JM, Nassar MA, Wood JN, Basbaum AI. Parallel "pain" pathways arise from subpopulations of primary afferent nociceptor. Neuron. 2005;47:787–793. doi: 10.1016/j.neuron.2005.08.015. [DOI] [PubMed] [Google Scholar]
- Burstein R, Jakubowski M, Garcia-Nicas E, Kainz V, Bajwa Z, Hargreaves R, Becerra L, Borsook D. Thalamic sensitization transforms localized pain into widespread allodynia. Ann Neurol. 2010;68:81–91. doi: 10.1002/ana.21994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bruehl S, Chung OY. Interactions between the cardiovascular and pain regulatory systems: an updated review of mechanisms and possible alterations in chronic pain. Neurosci Biobehav Rev. 2004;28:395–414. doi: 10.1016/j.neubiorev.2004.06.004. [DOI] [PubMed] [Google Scholar]
- Caous CA, de Sousa Buck H, Lindsey CJ. Neuronal connections of the paratrigeminal nucleus: a topographic analysis of neurons projecting to bulbar, pontine and thalamic nuclei related to cardiovascular, respiratory and sensory functions. Auton Neurosci. 2001;94:14–24. doi: 10.1016/s1566-0702(01)00338-1. [DOI] [PubMed] [Google Scholar]
- Capra NF. Localization and central projections of primary afferent neurons that innervate the temporomandibular joint in cats. Somatosens Res. 1987;4:201–213. doi: 10.3109/07367228709144607. [DOI] [PubMed] [Google Scholar]
- Casatti CA, Frigo L, Bauer JA. Origin of sensory and autonomic innervation of the rat temporomandibular joint: a retrograde axonal tracing study with the fluorescent dye Fast Blue. J Dent Res. 1999;78:776–783. doi: 10.1177/00220345990780031001. [DOI] [PubMed] [Google Scholar]
- Clarke PG. How inaccurate is the Abercrombie correction factor for cell counts? Trends Neurosci. 1992;15:211–212. doi: 10.1016/0166-2236(92)90036-8. [DOI] [PubMed] [Google Scholar]
- Clement CI, Keay KA, Podzebenko K, Gordon BD, Bandler R. Spinal sources of noxious visceral and noxious deep somatic afferent drive onto the ventrolateral periaqueductal gray of the rat. J Comp Neurol. 2000;425:323–344. doi: 10.1002/1096-9861(20000925)425:3<323::aid-cne1>3.0.co;2-z. [DOI] [PubMed] [Google Scholar]
- Dessem D, Moritani M, Ambalavanar R. Nociceptive craniofacial muscle primary afferent neurons synapse in both the rostral and caudal brain stem. J Neurophysiol. 2007;98:214–223. doi: 10.1152/jn.00990.2006. [DOI] [PubMed] [Google Scholar]
- Diatchenko L, Nackley AG, Slade GD, Fillingim RB, Maixner W. Idiopathic pain disorders--pathways of vulnerability. Pain. 2006;123:226–230. doi: 10.1016/j.pain.2006.04.015. [DOI] [PubMed] [Google Scholar]
- Eckert WA, 3rd, Julius D, Basbaum AI. Differential contribution of TRPV1 to thermal responses and tissue injury-induced sensitization of dorsal horn neurons in laminae I and V in the mouse. Pain. 2006;126:184–197. doi: 10.1016/j.pain.2006.06.032. [DOI] [PubMed] [Google Scholar]
- Gauriau C, Bernard JF. A comparative reappraisal of projections from the superficial laminae of the dorsal horn in the rat: the forebrain. J Comp Neurol. 2004a;468:24–56. doi: 10.1002/cne.10873. [DOI] [PubMed] [Google Scholar]
- Gauriau C, Bernard JF. Posterior triangular thalamic neurons convey nociceptive messages to the secondary somatosensory and insular cortices in the rat. J Neurosci. 2004b;24:752–761. doi: 10.1523/JNEUROSCI.3272-03.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hathaway CB, Hu JW, Bereiter DA. Distribution of Fos-like immunoreactivity in the caudal brainstem of the rat following noxious chemical stimulation of the temporomandibular joint. J Comp Neurol. 1995;356:444–456. doi: 10.1002/cne.903560311. [DOI] [PubMed] [Google Scholar]
- Heidrich H. Functional vascular diseases: Raynaud's syndrome, acrocyanosis and erythromelalgia. VASA Zeitschrift fur Gefasskrankheiten. 2010;39:33–41. doi: 10.1024/0301-1526/a000003. [DOI] [PubMed] [Google Scholar]
- Hirata H, Okamoto K, Tashiro A, Bereiter DA. A novel class of neurons at the trigeminal subnucleus interpolaris/caudalis transition region monitors ocular surface fluid status and modulates tear production. J Neurosci. 2004;24:4224–4232. doi: 10.1523/JNEUROSCI.0381-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hirata H, Takeshita S, Hu JW, Bereiter DA. Cornea–responsive medullary dorsal horn neurons: modulation by local opioid agonists and projections to thalamus and brainstem. J Neurophysiol. 2000;84:1050–1061. doi: 10.1152/jn.2000.84.2.1050. [DOI] [PubMed] [Google Scholar]
- Hunt SP, Pini A, Evan G. Induction of c-fos-like protein in spinal cord neurons following sensory stimulation. Nature. 1987;328:632–634. doi: 10.1038/328632a0. [DOI] [PubMed] [Google Scholar]
- Ikeda T, Terayama R, Jue S, Sugiyo S, Dubner R, Ren K. Differential rostral projections of caudal brainstem neurons receiving trigeminal input after masseter inflammation. J Comp Neurol. 2003;465:220–233. doi: 10.1002/cne.10836. [DOI] [PubMed] [Google Scholar]
- Imbe H, Dubner R, Ren K. Masseteric inflammatory-induced Fos protein expression in the trigeminal interpolaris/caudalis transition zone: contribution of somatosensory-vagal-adrenal integration. Brain Res. 1999;845:165–175. doi: 10.1016/s0006-8993(99)01913-7. [DOI] [PubMed] [Google Scholar]
- Ishii H, Niioka T, Izumi H. Difference between male and female rats in cholinergic activity of parasympathetic vasodilatation in the masseter muscle. Arch Oral Biol. 2009;54:533–542. doi: 10.1016/j.archoralbio.2009.02.008. [DOI] [PubMed] [Google Scholar]
- Iwata K, Kenshalo DR, Dubner R, Nahin RL. Diencephalic projections from the superficial and deep laminae of the medullary dorsal horn in the rat. J Comp Neurol. 1992;321:404–420. doi: 10.1002/cne.903210308. [DOI] [PubMed] [Google Scholar]
- King CD, Wong F, Currie T, Mauderli AP, Fillingim RB, Riley JL., 3rd Deficiency in endogenous modulation of prolonged heat pain in patients with Irritable Bowel Syndrome and Temporomandibular Disorder. Pain. 2009;143:172–178. doi: 10.1016/j.pain.2008.12.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kupers RC, Svensson P, Jensen TS. Central representation of muscle pain and mechanical hyperesthesia in the orofacial region: a positron emission tomography study. Pain. 2004;108:284–293. doi: 10.1016/j.pain.2003.12.029. [DOI] [PubMed] [Google Scholar]
- Lovick TA. Integrated activity of cardiovascular and pain regulatory systems: role in adaptive behavioural responses. Progr Neurobiol. 1993;40:631–644. doi: 10.1016/0301-0082(93)90036-r. [DOI] [PubMed] [Google Scholar]
- Loyd DR, Murphy AZ. Sex differences in the anatomical and functional organization of the periaqueductal gray-rostral ventromedial medullary pathway in the rat: a potential circuit mediating the sexually dimorphic actions of morphine. J Comp Neurol. 2006;496:723–738. doi: 10.1002/cne.20962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mainero C, Zhang WT, Kumar A, Rosen BR, Sorensen AG. Mapping the spinal and supraspinal pathways of dynamic mechanical allodynia in the human trigeminal system using cardiac-gated fMRI. NeuroImage. 2007;35:1201–1210. doi: 10.1016/j.neuroimage.2007.01.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maixner W. Temporomandibular joint disorders. In: Mayer E, Bushnell M, editors. Functional pain syndromes: presentation and pathology. Seattle: IASP Press; 2009. pp. 55–69. [Google Scholar]
- Menetrey D, Basbaum AI. Spinal and trigeminal projections to the nucleus of the solitary tract: a possible substrate for somatovisceral and viscerovisceral reflex activation. J Comp Neurol. 1987;255:439–450. doi: 10.1002/cne.902550310. [DOI] [PubMed] [Google Scholar]
- Menetrey D, Chaouch A, Binder D, Besson JM. The origin of the spinomesencephalic tract in the rat: an anatomical study using the retrograde transport of horseradish peroxidase. J Comp Neurol. 1982;206:193–207. doi: 10.1002/cne.902060208. [DOI] [PubMed] [Google Scholar]
- Menetrey D, Gannon A, Levine JD, Basbaum AI. Expression of c-fos protein in interneurons and projection neurons of the rat spinal cord in response to noxious somatic, articular, and visceral stimulation. J Comp Neurol. 1989;285:177–195. doi: 10.1002/cne.902850203. [DOI] [PubMed] [Google Scholar]
- Mitrovic I, Mitrovic MM, Bader S, Stoffel M, Jan LY, Basbaum AI. Contribution of GIRK2-mediated postsynaptic signaling to opiate and a2-adrenergic analgesia and analgesic sex differences. Proc Nat Acad Sci. 2003;100:271–276. doi: 10.1073/pnas.0136822100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mogil JS, Wilson SG, Chesler EJ, Rankin AL, Nemmani KV, Lariviere WR, Groce MK, Wallace MR, Kaplan L, Staud R, Ness TJ, Glover TL, Stankova M, Mayorov A, Hruby VJ, Grisel JE, Fillingim RB. The melanocortin-1 receptor gene mediates female-specific mechanisms of analgesia in mice and humans. Proc Natl Acad Sci U S A. 2003;100:4867–4872. doi: 10.1073/pnas.0730053100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mohn C, Vassend O, Knardahl S. Experimental pain sensitivity in women with temporomandibular disorders and pain-free controls: the relationship to orofacial muscular contraction and cardiovascular responses. Clinical J Pain. 2008;24:343–352. doi: 10.1097/AJP.0b013e318162eaf4. [DOI] [PubMed] [Google Scholar]
- Molander C, Xu Q, Rivero-Melian C, Grant G. Cytoarchitectonic organization of the spinal cord in the rat: II. The cervical and upper thoracic cord. J Comp Neurol. 1989;289:375–385. doi: 10.1002/cne.902890303. [DOI] [PubMed] [Google Scholar]
- Murphy AZ, Suckow SK, Johns M, Traub RJ. Sex differences in the activation of the spinoparabrachial circuit by visceral pain. Physiol Behav. 2009;97:205–212. doi: 10.1016/j.physbeh.2009.02.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nag S, Mokha SS. Activation of alpha2-adrenoceptors in the trigeminal region produces sex-specific modulation of nociception in the rat. Neuroscience. 2006;142:1255–1262. doi: 10.1016/j.neuroscience.2006.07.012. [DOI] [PubMed] [Google Scholar]
- Nishimori T, Sera M, Suemune S, Yoshida A, Tsuru K, Tsuiki Y, Akisaka T, Okamoto T, Dateoka Y, Shigenaga Y. The distribution of muscle primary afferents from the masseter nerve to the trigeminal sensory nuclei. Brain Res. 1986;372:375–381. doi: 10.1016/0006-8993(86)91148-0. [DOI] [PubMed] [Google Scholar]
- Okamoto K, Bereiter DF, Thompson R, Tashiro A, Bereiter DA. Estradiol replacement modifies c-fos expression at the spinomedullary junction evoked by temporomandibular joint stimulation in ovariectomized female rats. Neuroscience. 1986;156:729–736. doi: 10.1016/j.neuroscience.2008.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okamoto K, Imbe H, Kimura A, Donishi T, Tamai Y, Senba E. Activation of central 5HT2A receptors reduces the craniofacial nociception of rats. Neuroscience. 2007;147:1090–1102. doi: 10.1016/j.neuroscience.2007.05.012. [DOI] [PubMed] [Google Scholar]
- Panneton WM, Yavari P. A medullary dorsal horn relay for the cardiorespiratory responses evoked by stimulation of the nasal mucosa in the muskrat Ondatra zibethicus: evidence for excitatory amino acid transmission. Brain Res. 1995;691:37–45. doi: 10.1016/0006-8993(95)00597-j. [DOI] [PubMed] [Google Scholar]
- Paxinos G, Watson C. The Rat Brain in Stereotaxic Coordinates. New York: Academic Press; 1997. [Google Scholar]
- Ro JY, Capra NF, Masri R. Contribution of peripheral N-methyl-D-aspartate receptors to c-fos expression in the trigeminal spinal nucleus following acute masseteric inflammation. Neuroscience. 2004;123:213–219. doi: 10.1016/s0306-4522(03)00465-2. [DOI] [PubMed] [Google Scholar]
- Saxon DW, Hopkins DA. Efferent and collateral organization of paratrigeminal nucleus projections: an anterograde and retrograde fluorescent tracer study in the rat. J Comp Neurol. 1998;402:93–110. [PubMed] [Google Scholar]
- Schwartzman RJ, Erwin KL, Alexander GM. The natural history of complex regional pain syndrome. Clinical J Pain. 2009;25:273–280. doi: 10.1097/AJP.0b013e31818ecea5. [DOI] [PubMed] [Google Scholar]
- Shigenaga Y, Sera M, Nishimori T, Suemune S, Nishimura M, Yoshida A, Tsuru K. The central projection of masticatory afferent fibers to the trigeminal sensory nuclear complex and upper cervical spinal cord. J Comp Neurol. 1988;268:489–507. doi: 10.1002/cne.902680403. [DOI] [PubMed] [Google Scholar]
- Shimizu K, Guo W, Wang H, Zou S, LaGraize SC, Iwata K, Wei F, Dubner R, Ren K. Differential involvement of trigeminal transition zone and laminated subnucleus caudalis in orofacial deep and cutaneous hyperalgesia: the effects of interleukin-10 and glial inhibitors. Mol Pain. 2009;5:75. doi: 10.1186/1744-8069-5-75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strassman AM, Vos BP. Somatotopic and laminar organization of Fos-like immunoreactivity in the medullary and cervical dorsal horn induced by noxious facial stimulation in the rat. J Comp Neurol. 1993;331:495–516. doi: 10.1002/cne.903310406. [DOI] [PubMed] [Google Scholar]
- Sugiyo S, Takemura M, Dubner R, Ren K. Trigeminal transition zone/rostral ventromedial medulla connections and facilitation of orofacial hyperalgesia after masseter inflammation in rats. J Comp Neurol. 2005;493:510–523. doi: 10.1002/cne.20797. [DOI] [PubMed] [Google Scholar]
- Tajar A, McBeth J, Lee DM, Macfarlane GJ, Huhtaniemi IT, Finn JD, Bartfai G, Boonen S, Casanueva FF, Forti G, Giwercman A, Han TS, Kula K, Labrie F, Lean ME, Pendleton N, Punab M, Silman AJ, Vanderschueren D, O'Neill TW, Wu FC. Elevated levels of gonadotrophins but not sex steroids are associated with musculoskeletal pain in middle-aged and older European men. Pain. 2011;152:1495–1501. doi: 10.1016/j.pain.2011.01.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takemura M, Sugiyo S, Moritani M, Kobayashi M, Yonehara N. Mechanisms of orofacial pain control in the central nervous system. Arch Histol Cytol. 2006;69:79–100. doi: 10.1679/aohc.69.79. [DOI] [PubMed] [Google Scholar]
- Takeshita S, Hirata H, Bereiter DA. Intensity coding by TMJ-responsive neurons in superficial laminae of caudal medullary dorsal horn of the rat. J Neurophysiol. 2006;86:2393–2404. doi: 10.1152/jn.2001.86.5.2393. [DOI] [PubMed] [Google Scholar]
- Tashiro A, Okamoto K, Milam SB, Bereiter DA. Differential effects of estradiol on encoding properties of TMJ units in laminae I and V at the spinomedullary junction in female rats. J Neurophysiol. 2007;98:3242–3253. doi: 10.1152/jn.00677.2007. [DOI] [PubMed] [Google Scholar]
- Tershner SA, Mitchell JM, Fields HL. Brainstem pain modulating circuitry is sexually dimorphic with respect to mu and kappa opioid receptor function. Pain. 2000;85:153–159. doi: 10.1016/s0304-3959(99)00257-2. [DOI] [PubMed] [Google Scholar]
- Tracey I, Mantyh PW. The cerebral signature for pain perception and its modulation. Neuron. 2000;55:377–391. doi: 10.1016/j.neuron.2007.07.012. [DOI] [PubMed] [Google Scholar]
- Uddman R, Grunditz T, Kato J, Sundler F. Distribution and origin of nerve fibers in the rat temporomandibular joint capsule. Anat Embryol. 1998;197:273–282. doi: 10.1007/s004290050137. [DOI] [PubMed] [Google Scholar]
- Yamazaki Y, Ren K, Shimada M, Iwata K. Modulation of paratrigeminal nociceptive neurons following temporomandibular joint inflammation in rats. Exp Neurol. 2008;214:209–218. doi: 10.1016/j.expneurol.2008.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoshida A, Dostrovsky JO, Sessle BJ, Chiang CY. Trigeminal projections to the nucleus submedius of the thalamus in the rat. J Comp Neurol. 1991;307:609–625. doi: 10.1002/cne.903070408. [DOI] [PubMed] [Google Scholar]
- Yu YG, Caous CA, Balan AC, Rae GA, Lindsey CJ. Cardiovascular responses to sciatic nerve stimulation are blocked by paratrigeminal nucleus lesion. Auton Neurosci. 2002;98:70–74. doi: 10.1016/s1566-0702(02)00035-8. [DOI] [PubMed] [Google Scholar]
- Zhang S, Chiang CY, Xie YF, Park SJ, Lu Y, Hu JW, Dostrovsky JO, Sessle BJ. Central sensitization in thalamic nociceptive neurons induced by mustard oil application to rat molar tooth pulp. Neuroscience. 2006;142:833–842. doi: 10.1016/j.neuroscience.2006.06.063. [DOI] [PubMed] [Google Scholar]
- Zhang X, Giesler GJ., Jr Response characterstics of spinothalamic tract neurons that project to the posterior thalamus in rats. J Neurophysiol. 2005;93:2552–2564. doi: 10.1152/jn.01237.2004. [DOI] [PubMed] [Google Scholar]
- Zhou Q, Imbe H, Dubner R, Ren K. Persistent Fos protein expression after orofacial deep or cutaneous tissue inflammation in rats: implications for persistent orofacial pain. J Comp Neurol. 1999;412:276–291. doi: 10.1002/(sici)1096-9861(19990920)412:2<276::aid-cne7>3.0.co;2-9. [DOI] [PubMed] [Google Scholar]