Abstract
To detect gatifloxacin (GAT) residue in swine urine, an electrochemical immunoassay was established. An indirect competitive immunoassay was developed, in which the coating antigen is immobilized in an enzyme-linked immunosorbent assay (ELISA) plate and GAT residue from the sample competes with the limited binding sites in added anti-GAT antibody. Horseradish peroxidase (HRP) conjugated to goat anti-rabbit IgG was used as the enzymatic label. A carbon fiber working electrode was constructed and current signals were detected by using hydrogen peroxide as a substrate and hydroquinone as an electrochemical mediator. The electrochemical immunoassay was evaluated by analysis of GAT in buffer or swine urine and an average value of half inhibition concentration (IC50) of 8.9 ng/ml was obtained. Excellent specificity of the antibody was achieved with little cross-reaction with lomefloxacin (3.0%), ciprofloxacin (3.0%), and ofloxacin (1.9%) among commonly used (fluoro)quinolones. In conclusion, the immunoassay system developed in this research can be used as a rapid, powerful and on-site analytical tool to detect GAT residue in foods and food products.
Keywords: Gatifloxacin, Drug residue, Antibody, Carbon fiber electrode, Electrochemical immunoassay
1. Introduction
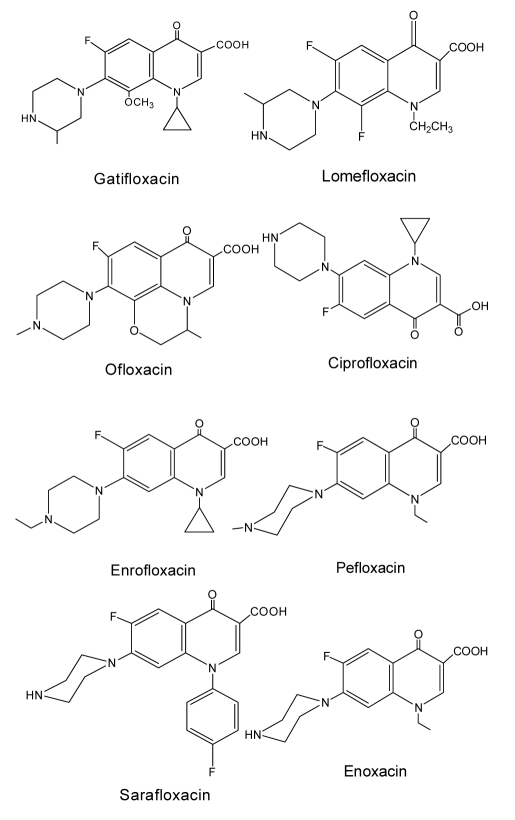
Gatifloxacin (GAT, Fig. 1) is a fourth generation synthetic antibacterial (fluoro)quinolone agent. It has been widely used in clinical medicine and as a veterinary drug in animal husbandry (Hooper, 1998; Blondeau et al., 2000; Weigel et al., 2002; Kays et al., 2005) for the treatment of acute bacterial exacerbation of chronic bronchitis, acute sinusitis, community acquired pneumonia, uncomplicated urinary tract infections (cystitis), and complicated urinary tract infection (Perry et al., 1999). However, the presence of GAT in food samples constitutes a potential hazard for consumers and may cause abnormalities in glucose metabolism (Bhasin et al., 2005). As a consequence, the US Food and Drug Administration (FDA) has claimed that the potential risk associated with GAT administration must be emphasized in the drug description. In May 2006, Bristol-Myers Squibb (BMS), the developer of GAT, even asked suppliers to stop selling GAT due to increasing concerns about the side effects. In China, GAT was found to inhibit hens from laying eggs (Amin et al., 2007). Due to the important effects of GAT on human and animal health, extensive research has been conducted to generate sensitive and specific methods for rapid and cost-effective GAT detection in food and biological samples.
Fig. 1.
Structures of the gatifloxacin and related (fluoro)quinolones
The main analytical techniques used for GAT analysis in foodstuffs include high pressure liquid chromatography (HPLC) (Overholser et al., 2003), liquid chromatography-mass spectrometry (LC-MS) (Vishwanathan et al., 2001), high-performance thin layer chromatography (HPTLC) (Suhagia et al., 2006), liquid chromatography (LC) (Amin et al., 2007), capillary electrochromatography (Fierens et al., 2000), and fluorescence methods (Espinosa-Mansilla et al., 2006). The chromatographic techniques usually require extraction, per-concentration steps, and complicated cleanup procedures. Moreover, expensive and sophisticated equipment is required, which must be run by skilled operators. As a consequence, routine and efficient residue monitoring by the official agencies has become problematic. To protect consumer health, official laboratories should be able to test a large number of samples using rapid, sensitive, cost-effective, high-throughput and on-site analytical methods.
Immunoassay, on the other hand, has proven to be an excellent on-site analytical strategy as a sensitive, timely, inexpensive and high-throughput technique that can be used to detect veterinary drugs in a variety of food matrices (Zhou et al., 2005; Liu et al., 2007). Immunoanalytical techniques have been reported to detect GAT by an enzyme-linked immunosorbent assay (ELISA) using ultraviolet (UV) or fluorescence absorbance as a quantitative parameter (Santoro et al., 2006; Zhao et al., 2007). In recent years, electrochemical immunoassays have attracted attention because of their excellent sensitivity, simplicity, inexpensive construction, suitability for mass production, and their variety of labels, including enzymes and electro-active molecules. For the development of hand-held devices which can be used for daily measurements, electrochemical immunoassay is a promising alternative to the existing ELISA tests. Also, electrochemical immunoassays are preferable for opaque matrices (Laschi et al., 2003; Conneely et al., 2007; Piermarini et al., 2007). The aim of this work was to develop an electrochemical immunoassay to detect GAT in biological samples. To our knowledge, there are no published reports of an electrochemical immunoassay for GAT detection. In this research, an indirect competitive immunoassay was established in which the coating antigen is immobilized onto an ELISA plate and GAT from a sample competes with the limited binding sites in the anti-GAT antibody. Horseradish peroxidase (HRP) conjugated to goat anti-rabbit IgG was added as an enzymatic label. A carbon fiber working electrode was fabricated to detect current signals on the ELISA plate caused by the electroreaction with hydrogen peroxide as the substrate and hydroquinone (HQ) as the electrochemical mediator. The current response was used to indicate the competition of GAT with coating antigen for binding with the anti-GAT antibody (Ni et al., 2006). The electrochemical immunoassay was evaluated by analysis of GAT in buffer or swine urine. This electrochemical immunoassay shows good performance in terms of sensitivity and specificity and could be used as a screening method to detect GAT residues in foods and food products.
2. Materials and methods
2.1. Chemicals and materials
GAT, lomefloxacin, ofloxacin, ciprofloxacin, fleroxacin, enrofloxacin, enoxacin, norfloxacin, pefloxacin, sarafloxacin, danofloxacin and pipemidic acid were obtained from the Institute of Veterinary Medicine of China (Beijing, China). 1-Ethyl-3-(dimethylaminopropyl) carbodiimide hydrochloride (EDC), N-hydroxysuccinimide (NHS), bovine serum albumin (BSA), and ovalbumin (OVA) were purchased from Sigma-Aldrich (St. Louis, MO, USA). Goat anti-rabbit IgG-HRP conjugate was provided by the Military Medical Institute (Beijing, China). HQ was purchased from Hongye Chemical Technology (Tianjin, China). Dimethylformamide (DMF), hydrogen peroxide [H2O2, 30% (mass fraction)], and other reagents were purchased from Guangmang Chemical Co. (Jinan, China).
Sodium phosphate buffer (PBS, pH 7.4) consisted of NaCl (138 mmol/L), KH2PO4 (1.5 mmol/L), Na2HPO4·12H2O (7 mmol/L), and KCl (2.7 mmol/L). Washing buffer (PBST) comprised the PBS solution containing 0.05% (v/v) Tween 20. Carbonate buffer (pH 9.6) containing 15 mmol/L Na2CO3 and 35 mmol/L NaHCO3 was used as a coating buffer. The blocking buffer was 0.1 mol/L PBS mixed with 0.01 g/ml of OVA and 0.05% of Tween 20. Ultrapure deionized water with resistivity >18 MΩ∙cm (Milli-Q) was used throughout this study. The 2.0 mmol/L H2O2 was used as the substrate solution. The mediator solution was 2.0 mmol/L HQ solution, prepared by dissolving 17.6 mg HQ in 20 ml of PBS.
2.2. Apparatus
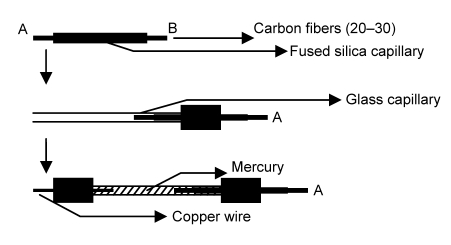
Amperometric signal was monitored by an electrochemical potentiometer (CHI 800, Shanghai Chenhua Instrument Ltd., China). The electrochemical system consisted of a three-electrode system in which a carbon fiber electrode was used as a working electrode (Fig. 2), a platinum wire was used as an auxiliary electrode, and filamentous silver was used as a reference electrode. The electrochemical reaction was carried out in one well of a polystyrene 12-well microtiter plate (Bio Basic Inc., Ontario, Canada).
Fig. 2.
Fabrication of carbon fiber working electrode
2.3. Construction of carbon fiber working electrode
Carbon fiber working electrodes are widely used as probes for biochemical detection in the wells of microplates (Jin et al., 2003; He et al., 2003). Our electrode was constructed as described in Fig. 2. First, 20–30 carbon fibers (6 μm diameter) soaking in acetone were carefully inserted into a fused silica capillary (250 μm i.d., 375 μm o.d., 0.6 cm length) by gluing end A using ethyl α-cyanoacrylate. The fused silica capillary was then inserted into a glass capillary (2 mm i.d., 3 mm o.d., 3 cm length) with end B sealed using epoxy AB adhesive. Mercury (2.0 cm height) was then injected into one end of the glass capillary while at the other end, copper wire was inserted and sealed with epoxy AB adhesive. Before each use, the end A was sonicated for 10 min in ethanol and deionized water (Milli-Q water, >18 MΩ∙cm).
2.4. Preparation of polyclonal antibody for GAT
The immunogen cationized BSA (cBSA)-GAT and coating antigen cOVA-GAT for GAT were prepared via the carbodiimide-modified active ester method, with EDC and NHS used as crosslinking agents. Two male New Zealand white rabbits were subcutaneously immunized to obtain polyclonal antibody for GAT. The synthesis procedure and immunization design were the same as those reported in our previous study (Zhao et al., 2007). The purified antiserum was aliquotted and stored at −70 °C.
2.5. Immunoassay procedure
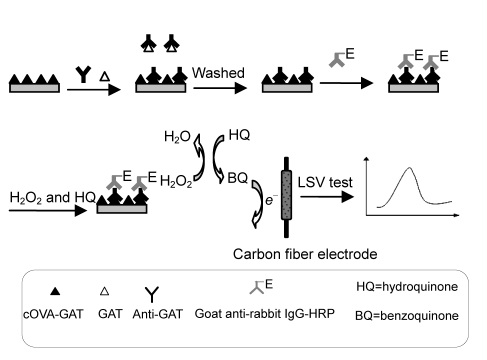
The immunoassay was developed on the basis of an indirect competition scheme (Fig. 3), in which GAT and goat anti-rabbit IgG-HRP compete for the binding sites of the anti-GAT antibody. First, the coating antigen cOVA-GAT (10 μg/ml, 100 μl/well) was immobilized onto the surface of ELISA plates for the capture of the antibody. The microplates were incubated overnight at 4 °C and were then blocked with 250 μl/well of blocking buffer, followed by incubation for 2 h at 37 °C. Between each step, the microplates were washed three times with washing buffer. Then, 100 μl of anti-GAT antibody was added, followed by addition of buffer or competitor solution (GAT samples or similar compounds analyzed), and the plates were incubated for 2 h. Goat anti-rabbit IgG-HRP (1:1000, 100 μl/well) was added. The competitive immunoreaction was then allowed to proceed for 70 min at 37 °C. Then HQ mediator solution and H2O2 substrate solution were added (50 μl/well). The HRP enzymatic reaction progressed for another 10 min at room temperature. Then, the carbon fiber working electrode was inserted and amperometric signals were determined at −0.15 V. The result was expressed in percent inhibition as follows: I/I 0×100%, where I is the peak current in the linear sweep voltammetry (LSV) profile for the well containing competitor and I 0 is that for the well without competitor.
Fig. 3.
Electrochemical immunoassay procedure
3. Results and discussion
3.1. Optimization of the assay
As ideal chemical sensors, microelectrodes have attracted widespread research interest and have been studied intensively in many fields (Wightman et al., 1988; Huang et al., 2001) to facilitate rapid measurement of chemical compounds. Carbon fiber microelectrodes are important components of the wider microelectrode family. Several electrochemical assays for rapid analysis with carbon fiber microelectrodes have been described, including amperometry, differential pulse voltammetry, and cyclic voltammetry (Pihel et al., 1996; Blanco et al., 1999; Netchiporouk et al., 2001). The carbon fiber electrodes used in this study were fabricated using a traditional preparation method. The competitive immunoassay was performed on ELISA microplate wells, in which GAT from the sample and the immobilized coating antigen compete for the binding sites of the antibody. When the analyte molecular recognition event happens, the extent of the reaction is evaluated by the electrochemical detection of the mediator (HQ) based directly on the activity of the enzyme tracer (goat-anti-rabbit IgG-HRP). Hydrogen peroxide was used to facilitate the enzymatic reaction (Lei et al., 2004; Hervás et al., 2009). After the reaction was allowed to proceed for 10 min, the electrode was inserted and LSV evaluation was employed to examine current signals for each group with the potential fixed. The response was determined at −0.15 V (previously optimized) on the carbon fiber working electrode where the reduction of the enzymatically oxidized mediator HQ took place.
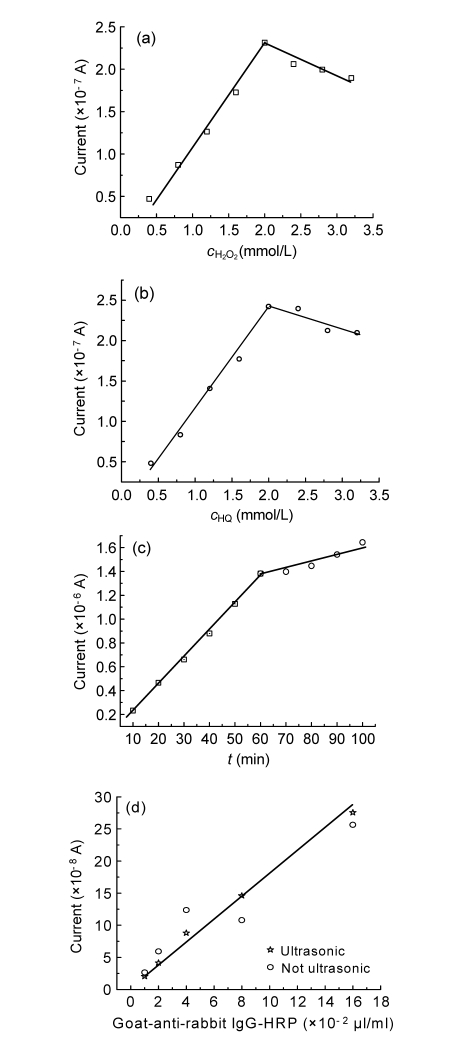
The concentrations of substrate (H2O2), mediator (HQ), and enzyme tracer (HRP) are important factors that affect the sensitivity of the immunoassay, and were optimized by testing current values for each group. The tested concentration of H2O2 was in the range of 0.4−3.2 mmol/L, using 1 μl/ml of IgG-HRP and 2.0 mmol/L of HQ (Fig. 4a). The current response increased gradually when the concentration of H2O2 increased until a peak was reached at 2.0 mmol/L, then the current measured started to decrease as the concentration of H2O2 continued to increase (Fig. 4a). As the concentration of H2O2 increased, electron transfer was enhanced. However, when the concentration of H2O2 increased further from 2.0 to 3.2 mmol/L, the reaction was inhibited by the excess of H2O2. Thus, a concentration of 2.0 mmol/L of H2O2 was selected as the optimal concentration throughout the research. The optimization result for the peroxide concentration is in good agreement with Ordóñez and Fàbregas (2007).
Fig. 4.
Optimization of analytical conditions
The influences of (a) H2O2 (2.0 mmol/L HQ), (b) HQ (2.0 mmol/L H2O2), (c) incubation time (2.0 mmol/L HQ and 2.0 mmol/L H2O2), and (d) ultrasonic for the working electrode (2.0 mmol/L HQ, 2.0 mmol/L H2O2, and 10 min for incubation) on current response in the competitive assay are shown. The concentration of IgG-HRP conjugate was 1 μl/ml for protocols (a) to (c). The experiments were carried out in PBST (pH 7.4) containing NaCl (138 mmol/L), KH2PO4 (1.5 mmol/L), Na2HPO4·12H2O (7 mmol/L), KCl (2.7 mmol/L), and Tween 20 (0.05%), with the applied potential of −0.15 V
It was reported that reducing substrates, such as HQ, have a low specificity for catalyzing the oxidation of H2O2 (Zhang et al., 2008). To find out the most suitable quantity of HQ, a series of concentrations in the range of 0.4–3.2 mmol/L of HQ were evaluated, with the concentrations of H2O2 and IgG-HRP fixed at 2.0 mmol/L and 1 μl/ml, respectively. The maximum current was observed when the concentration of HQ was 2.0 mmol/L (Fig. 4b). The decrease in current when the concentration of HQ was more than 2.0 mmol/L was probably caused by the limited enzyme-substrate kinetics (Lei et al., 2004). The optimized concentration of HQ, therefore, was determined to be 2.0 mmol/L and was used throughout this research.
The effect of the immunochemical incubation time was also investigated due to its importance. The concentrations of both H2O2 and HQ were fixed at 2.0 mmol/L, and the concentration of goat-anti-rabbit IgG-HRP conjugate was fixed at 1 μl/ml. The current was measured at 10 min intervals (Fig. 4c). Although, the current measured can be increased to a high value, we eventually chose a 10-min incubation time because the current measured at this time was high enough to make a judgment. More importantly, the experimental time can thereby be shortened significantly.
Non-specific adsorption due to the direct adsorption of enzyme tracer onto the working electrode surface could produce a residual background signal when the electrode is moved to the next well. This effect was evaluated by comparing the relationship of current response with IgG-HRP concentration while ultrasonic and control electrodes were used. The results (Fig. 4d) showed that non-specific adsorption in non-ultrasonic groups markedly affected the linear relationship for GAT detection. Therefore, in our research, the working electrode was sonicated in PBST for 10 min to stabilize the background before each measurement.
3.2. Sensitivity and specificity of the test
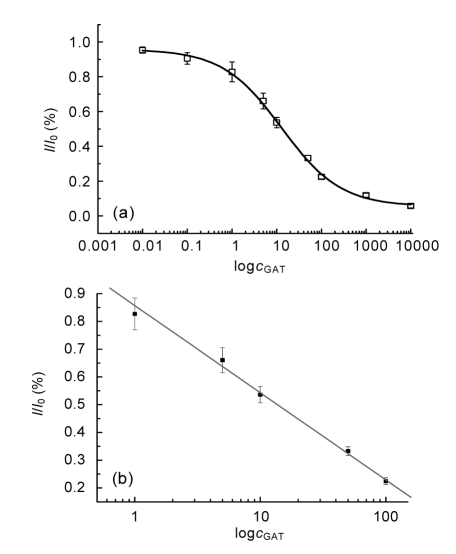
The electrochemical immunoassay was developed under the optimized condition (2.0 mmol/L H2O2, 2.0 mmol/L HQ, and 1 μl/ml IgG-HRP). Antiserum with a dilution factor of 1:8 000 was utilized as the primary antibody and co-incubated with GAT from 0.01 to 1 000 ng/ml to compete with the coating antigen for binding the antiserum. The competitive inhibition curve and standard calibration curve (Y=0.86−0.31X, R 2=0.99) are shown in Figs. 5a and 5b, respectively. The GAT concentration with 50% inhibition (IC50 value) was estimated to be 10.5 ng/ml and the linear concentration range was from 1 to 100 ng/ml.
Fig. 5.
Competitive curve (a) and standard curve (b) of GAT determination with GAT as competitor in PBST
c GAT: concentration of GAT (ng/ml)
The specificity of the antibody was evaluated by testing the cross-reactivity with various (fluoro) quinolones. The cross-reactivity studies were carried out with the electrochemical immunoassay by adding various competitors in concentrations from 0.01 to 1000 ng/ml (Lu et al., 2006). The results confirmed that the anti-GAT antibody was highly specific and showed little cross-reactivity with lomefloxacin, ciprofloxacin, or ofloxacin (Table 1).
Table 1.
IC50 values and cross-reactivity of anti-GAT antibody toward selected (fluoro)quinolones by the electrochemical immunoassay
| Compound | IC50 (ng/ml) | Cross-reactivity (%) |
| Gatifloxacin | 10.5 | 100.0 |
| Lomefloxacin | 280 | 3.0 |
| Ciprofloxacin | 345 | 3.0 |
| Ofloxacin | 548 | 1.9 |
| Enrofloxacin | >1000 | <1 |
| Sarafloxacin | >1000 | <1 |
| Flerfloxacin | >1000 | <1 |
| Pipemidic acid | >1000 | <1 |
| Pefloxacin | >1000 | <1 |
| Norfloxacin | >1000 | <1 |
| Enoxacin | >1000 | <1 |
3.3. Detection of GAT in swine urine
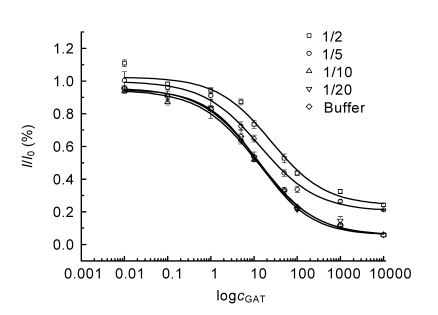
Urine, composed of various electrochemical interferents, such as uric acid, ascorbic xanthine, ascorbic acid, and p-aminophenol (Jamal et al., 2005), was selected to study the matrix effect (Yang et al., 2006). Swine urine was centrifuged at 15 000 r/min for 15 min and the supernatant obtained was diluted with PBST. The competitive curves with final GAT concentrations of 0.01, 0.1, 1, 5, 10, 50, 100, 1 000 and 10 000 ng/ml were run in PBST and in various dilutions (1:2, 1:5, 1:10, and 1:20) of the extraction to determine the matrix effect (Fig. 6). The IC50 values with GAT as the competitor were 66.07, 29.36, 12.37, and 12.59 ng/ml when the dilution factors were 1:2, 1:5, 1:10, and 1:20, respectively. It was possible that electrochemical interferents would restrain the electron transfer, resulting in an increase in background current signal. The results were in good agreement with those observed in a milk system (Zhao et al., 2007). The average IC50 value of a 1:10 dilution was close to that in PBST (8.9 ng/ml), and was chosen for performance of the immunoassay. Based on the standard curve obtained in 1:10 dilution, swine urine was used as a real system to test the applicability of the electrochemical immunoassay (Table 2). A series of GAT solutions with concentrations of 1, 5, 10, 50, and 100 ng/ml were added to the urine sample, and the recovery rate and coefficient of variation (CV) were calculated. The CVs were below 13.38% and the recovery rates were between 93% and 118%, indicating the reliable accuracy of the test when applied to swine urine (Table 2).
Fig. 6.
Competitive curves of GAT in PBST and various dilutions of 1:2, 1:5, 1:10, and 1:20 of the extracted swine urine
c GAT: concentration of GAT (ng/ml)
Table 2.
Recovery rate and coefficient of variation (CV) of swine urine spiked with GAT and detected by the electrochemical method
| GAT level (ng/ml) | n | Measured GAT level (ng/ml) | Recovery (%) | CV (%) |
| 1 | 3 | 1.13±0.09 | 112.54 | 8.16 |
| 5 | 3 | 4.69±0.62 | 93.72 | 13.38 |
| 10 | 3 | 11.74±0.38 | 117.36 | 3.27 |
| 50 | 3 | 48.30±1.62 | 96.60 | 3.35 |
| 100 | 3 | 105.06±2.27 | 105.06 | 2.16 |
4. Conclusions
In this work, a carbon fiber working electrode was prepared and a sensitive electrochemical immunoassay was developed to detect GAT residue in swine urine. The average IC50 value for this test was found to be 8.9 ng/ml, and there was little cross-reactivity with other common (fluoro)quinolones. The utilization of the assay was also tested by using it to detect GAT in spiked swine urine samples. Satisfactory results were obtained (CV<13.38%; recovery rates<20%).
In summary, the potential application of the electrochemical immunoassay for the detection of GAT residue in swine urine was demonstrated. The method developed could be commercialized in the form of a hand-held device to detect GAT residue in foods.
Footnotes
Project supported by the National High-Tech R&D Program (863) of China (Nos. 07AA10Z435 and 2007AA06A407), the National Natural Science Foundation of China (No. 20675048), the Fundamental Research Funds for the Central Universities (No. 65011121), and the Shandong Provincial Natural Science Foundation (No. Y2008B31), China
References
- 1.Amin AS, Gouda AAEF, El-Sheikh R, Zahran F. Spectrophotometric determination of gatifloxacin in pure form and in pharmaceutical formulation. Spectrochim Acta A. 2007;67(5):1306–1312. doi: 10.1016/j.saa.2006.09.041. [DOI] [PubMed] [Google Scholar]
- 2.Bhasin R, Arce FC, Pasmantier R. Hypoglycemia associated with the use of gatifloxacin. Am J Med Sci. 2005;330(5):250–253. doi: 10.1097/00000441-200511000-00010. [DOI] [PubMed] [Google Scholar]
- 3.Blanco MH, Quintana LH, Hernandez L. Determination of dihydrozeatin riboside in apples by anodic stripping voltammetry with a carbon fiber microelectrode. Fresenius J Anal Chem. 1999;364(3):254–260. doi: 10.1007/s002160051333. [DOI] [Google Scholar]
- 4.Blondeau JM, Laskowski R, Bjarnason J, Stewart C. Comparative in vitro activity of gatifloxacin, grepafloxacin, levofloxacin, moxifloxacin and trovafloxacin against Gram-negative and Gram-positive organisms. Int J Antimicrob Agents. 2000;14(1):45–50. doi: 10.1016/S0924-8579(99)00143-0. [DOI] [PubMed] [Google Scholar]
- 5.Conneely G, O′Mahony D, Lu H, Guilbault GG, Pravda M, Aherne M. An immunosensor for the detection of stanozolol in bovine urine. Anal Lett. 2007;40(7):1280–1293. doi: 10.1080/00032710701326650. [DOI] [Google Scholar]
- 6.Espinosa-Mansilla A, de la Pena AM, Gomez DG, Lopez FS. Determination of fluoroquinolones in urine and serum by using high performance liquid chromatography and multiemission scan fluorimetric detection. Talanta. 2006;68(4):1215–1221. doi: 10.1016/j.talanta.2005.07.035. [DOI] [PubMed] [Google Scholar]
- 7.Fierens C, Hillaert S, van den Bossche W. The qualitative and quantitative determination of quinolones of first and second generation by capillary electrophoresis. J Pharmaceut Biomed Anal. 2000;22(5):763–772. doi: 10.1016/S0731-7085(99)00282-4. [DOI] [PubMed] [Google Scholar]
- 8.He ZH, Gao N, Jin WR. Capillary electrophoretic enzyme immunoassay with electrochemical detection using a noncompetitive format. J Chromatogr B. 2003;784(2):343–350. doi: 10.1016/S1570-0232(02)00823-1. [DOI] [PubMed] [Google Scholar]
- 9.Hervás M, López MA, Escarpa A. Electrochemical immunoassay using magnetic beads for the determination of zearalenone in baby food: an anticipated analytical tool for food safety. Anal Chim Acta. 2009;653(2):167–172. doi: 10.1016/j.aca.2009.09.024. [DOI] [PubMed] [Google Scholar]
- 10.Hooper DC. Clinical applications of quinolones. BBA-Gene Struct Expr. 1998;1400(1-3):45–61. doi: 10.1016/S0167-4781(98)00127-4. [DOI] [PubMed] [Google Scholar]
- 11.Huang WH, Pang DW, Tong H, Wang ZL, Cheng JK. A method for the fabrication of low-noise carbon fiber nanoelectrdes. Anal Chem. 2001;73(5):1048–1052. doi: 10.1021/ac0008183. [DOI] [PubMed] [Google Scholar]
- 12.Jamal M, Crowe MA, Magner E. Characterization of the composition of bovine urine and its effect on the electrochemical analysis of the model mediator, p-aminophenol. Anal Chim Acta. 2005;554(1-2):79–85. doi: 10.1016/j.aca.2005.08.065. [DOI] [Google Scholar]
- 13.Jin WR, Li XJ, Gao N. Simultaneous determination of tryptophan and glutathione in individual rat hepatocytes by capillary zone electrophoresis with electrochemical detection at a carbon fiber bundle-Au/Hg dual electrode. Anal Chem. 2003;75(15):3859–3864. doi: 10.1021/ac0207022. [DOI] [PubMed] [Google Scholar]
- 14.Kays MB, Overholser BR, Lagvankar S, Goldman M, Sowinski KM. Effect of ensure on the oral bioavailability of gatifloxacin in healthy volunteers. Pharmacotherapy. 2005;25(11):1530–1535. doi: 10.1592/phco.2005.25.11.1530. [DOI] [PubMed] [Google Scholar]
- 15.Laschi S, Mascini M, Scortichini G, Franek M, Mascini M. Polychlorinated biphenyls (PCBs) detection in food samples using an electrochemical immunosensor. J Agric Food Chem. 2003;51(7):1816–1822. doi: 10.1021/jf0208637. [DOI] [PubMed] [Google Scholar]
- 16.Lei CX, Hu SQ, Gao N, Shen GL, Yu RQ. An amperometric hydrogen peroxide biosensor based on immobilizing horseradish peroxidase to a nano-Au monolayer supported by sol-gel derived carbon ceramic electrode. Bioelectrochemistry. 2004;65(1):33–39. doi: 10.1016/j.bioelechem.2004.06.002. [DOI] [PubMed] [Google Scholar]
- 17.Liu Y, Zhang CZ, Yu XY, Zhang ZY, Zhang X, Liu RR, Liu XJ, Gong ZM. Development and evaluation of immunoassay for zeranol in bovine urine. J Zhejiang Univ-Sci B. 2007;8(12):900–905. doi: 10.1631/jzus.2007.B0900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lu SX, Zhang YL, Liu JT, Zhao CB, Liu W, Xi RM. Preparation of anti-pefloxacin antibody and development of an indirect competitive enzyme-linked immunosorbent assay for detection of pefloxacin residue in chicken liver. J Agric Food Chem. 2006;54(19):6995–7000. doi: 10.1021/jf061309q. [DOI] [PubMed] [Google Scholar]
- 19.Netchiporouk L, Shram N, Salvert D, Cespuglio R. Brain extracellular glucose assessed by voltammetry throughout the rat sleep-wake cycle. Eur J Neursci. 2001;13(7):1429–1434. doi: 10.1046/j.0953-816x.2001.01503.x. [DOI] [PubMed] [Google Scholar]
- 20.Ni YN, Wang YR, Kokot S. Simultaneous determination of three fluoroquinolones by linear sweep stripping voltammetry with the aid of chemometrics. Talanta. 2006;69(1):216–225. doi: 10.1016/j.talanta.2005.09.032. [DOI] [PubMed] [Google Scholar]
- 21.Ordóñez SS, Fàbregas E. New antibodies immobilization system into a graphite-polysulfone membrane for amperometric immunosensors. Biosens Bioelectron. 2007;22(6):965–972. doi: 10.1016/j.bios.2006.03.022. [DOI] [PubMed] [Google Scholar]
- 22.Overholser BR, Kays MB, Sowinski KM. Determination of gatifloxacin in human serum and urine by high-performance liquid chromatography with ultraviolet detection. J Chromatogr B. 2003;798(1):167–173. doi: 10.1016/j.jchromb.2003.09.020. [DOI] [PubMed] [Google Scholar]
- 23.Perry CM, Barman Balfour JA, Lamb HM. Gatifloxacin. Drugs. 1999;58(4):683–696. doi: 10.2165/00003495-199958040-00010. [DOI] [PubMed] [Google Scholar]
- 24.Piermarini S, Micheli L, Ammida NHS, Palleschi G, Moscone D. Electrochemical immunosensor array using a 96-well screen-printed microplate for aflatoxin B-1 detection. Biosens Bioelectron. 2007;22(7):1434–1440. doi: 10.1016/j.bios.2006.06.029. [DOI] [PubMed] [Google Scholar]
- 25.Pihel K, Walker QD, Wightman RM. Overoxidized polypyrrole-coated carbon fiber microelectrodes for dopamine measurements with fast-scan cyclic voltammetry. Anal Chem. 1996;68(13):2084–2089. doi: 10.1021/ac960153y. [DOI] [PubMed] [Google Scholar]
- 26.Santoro MIRM, Kassab NM, Singh AK, Kedor-Hackmam ERM. Quantitative determination of gatifloxacin, levofloxacin, lomefloxacin and pefloxacin fluoroquinolonic antibiotics in pharmaceutical preparations by high-performance liquid chromatography. J Pharmaceut Biomed Anal. 2006;40(1):179–184. doi: 10.1016/j.jpba.2005.06.018. [DOI] [PubMed] [Google Scholar]
- 27.Suhagia BN, Shah SA, Rathod IS, Patel HM, Shah DR, Marolia BP. Determination of gatifloxacin and ornidazole in tablet dosage forms by high-performance thin-layer chromatography. Anal Sci. 2006;22(5):743–745. doi: 10.2116/analsci.22.743. [DOI] [PubMed] [Google Scholar]
- 28.Vishwanathan K, Bartlett MG, Stewart JT. Determination of gatifloxacin in human plasma by liquid chromatography/electrospray tandem mass spectrometry. Rapid Commun Mass Spectrom. 2001;15(12):915–919. doi: 10.1002/rcm.322. [DOI] [PubMed] [Google Scholar]
- 29.Weigel LM, Anderson GJ, Tenover FC. DNA gyrase and topoisomerase IV mutations associated with fluoroquinolone resistance in Proteus mirabilis. Antimicrob Agents Chemother. 2002;46(8):2582–2587. doi: 10.1128/AAC.46.8.2582-2587.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wightman RM, May LJ, Michael AC. Detection of dopamine dynamics in the brain. Anal Chem. 1988;60(13):769–779. doi: 10.1021/ac00164a001. [DOI] [PubMed] [Google Scholar]
- 31.Yang ZY, Kolosova AY, Shim WB, Chung DH. Development of monoclonal antibodies against pirimiphos-methyl and their application to IC-ELISA. J Agric Food Chem. 2006;54(13):4551–4556. doi: 10.1021/jf0606196. [DOI] [PubMed] [Google Scholar]
- 32.Zhang HL, Lai GS, Han DY, Yu AM. An amperometric hydrogen peroxide biosensor based on immobilization of horseradish peroxidase on an electrode modified with magnetic dextran microspheres. Anal Bioanal Chem. 2008;390(3):971–977. doi: 10.1007/s00216-007-1748-3. [DOI] [PubMed] [Google Scholar]
- 33.Zhao C, Liu W, Ling H, Lu S, Zhang Y, Liu J, Xi R. Preparation of anti-gatifloxacin antibody and development of an indirect competitive enzyme-linked immunosorbent assay for the detection of gatifloxacin residue in milk. J Agric Food Chem. 2007;55(17):6879–6884. doi: 10.1021/jf070978g. [DOI] [PubMed] [Google Scholar]
- 34.Zhou M, Guan C, Chen G, Xie X, Wu S. Determination of theophylline concentration in serum by chemiluminescent immunoassay. J Zhejiang Univ-Sci B. 2005;6(12):1148–1152. doi: 10.1631/jzus.2005.B1148. [DOI] [PMC free article] [PubMed] [Google Scholar]