Abstract
Most anti-vascular endothelial growth factor (VEGF) therapies in diabetic macular edema are not as robust as in proliferative diabetic retinopathy. Although the VEGF appears to be a good target in diabetic macular edema, the anti-VEGF therapies appear to be of transient benefit as the edema recurs within a few weeks, and repeated injections are necessary. There is new evidence that indicates ‘retinal inflammation’ as an important player in the pathogenesis of diabetic retinopathy. There are common sets of inflammatory cytokines that are upregulated in both the serum and vitreous and aqueous samples, in subjects with diabetic retinopathy, and these cytokines can have multiple interactions to impact the pathogenesis of the disease. The key inflammatory events involved in the blood retinal barrier (BRB) alteration appear to be: (1) Increased expression of endothelial adhesion molecules such as ICAM1, VCAM1, PECAM-1, and P-selectin, (2) adhesion of leukocytes to the endothelium, (3) release of inflammatory chemokines, cytokines, and vascular permeability factors, (4) alteration of adherens and tight junctional proteins between the endothelial cells, and (5) infiltration of leukocytes into the neuro-retina, resulting in the alteration of the blood retinal barrier (diapedesis). VEGF inhibition itself may not achieve neutralization of other inflammatory molecules involved in the inflammatory cascade of the breakdown of the BRB. It is possible that the novel selective inhibitors of the inflammatory cascade (like angiopoietin-2, TNFα, and chemokines) may be useful therapeutic agents in the treatment of diabetic macular edema (DME), either alone or in combination with the anti-VEGF drugs.
Keywords: Blood-retinal barrier, Chemokines, Cytokines, Diabetic Retinopathy, Inflammation, Steroids, Vascular Endothelial Growth Factor
INTRODUCTION
In recent times there has been a significant increase in the number of people with diabetes worldwide, and nearly 366 million people are estimated to be diabetic (IDF Atlas, 2011). By 2030, the number is expected to double, and this global increase will have a tremendous impact on the prevalence of diabetic complications.1 Diabetic retinopathy (DR) is an important microvascular complication and is the leading cause of blindness in the working-age population (20-64 years old) of developed countries, and each year around new cases of blindness, of 12%, are recorded.2,3 It is estimated that around 40% of the people with diabetes, aged over 40 years have some degree of retinopathy that includes approximately 8.2% with vision-threatening retinopathy.4,5 Landmark clinical trials such as The Diabetes Control and Complications Trial (DCCT) Research Group, 1993, and the United Kingdom Prospective Diabetes Study (UKPDS) Group, 1998, have established that hyperglycemia is the decisive risk factor for the development of diabetic retinopathy.6,7 Furthermore, the presence of diabetic retinopathy is an important risk factor for the development of diabetic micro- and macrovascular complications.8,9 Recent clinical and laboratory investigations have identified inflammation as an important molecular mechanism in the development and progression of diabetic retinopathy, and this review will discuss the role of inflammation and novel therapeutic targets in diabetic retinopathy.
MOLECULAR MECHANISMS OF DIABETIC RETINOPATHY
Activation of multiple cellular pathways, primarily mediated by hyperglycemia, is considered to play an important role in the pathogenesis of diabetic retinopathy. The metabolic effects of hyperglycemia result in microvascular damage of the retina that leads to vascular leakage (non-proliferative diabetic retinopathy (NPDR)) and ischemia-induced retinal neovascularization (proliferative diabetic retinopathy (PDR)). The mechanism by which the diabetic risk factor initiates the vascular disruption in retinopathy remains unclear, and several pathways have been implicated. The hyperglycemia-induced pathogenesis of diabetic retinopathy is related to four main biochemical alterations such as: (i) increased polyol pathway flux; (ii) increased advanced glycation end-product (AGE) formation; (iii) activation of protein kinase C (PKC) isoforms; and (iv) increased hexosamine pathway flux.10 These pathways terminate in increased oxidative stress, inflammation, and vascular dysfunction that result in eventual increased permeability, vascular occlusion, and local ischemia. Further, these events lead to the upregulation of proangiogenic and inflammatory factors such as the vascular endothelial growth factor (VEGF), insulin-like growth factor (IGF), angiopoietins (Ang-2), stromal derived factor-1 (SDF-1), basic fibroblast growth factor-2 (bFGF), hepatocyte growth factor (HGF), tumor necrosis factor (TNF), and interleukin-6. VEGF is the factor that has been extensively studied in the pathogenesis of DR, and new pharmacotherapies predominantly target the VEGF molecule.11 Furthermore, the use of anti-VEGF therapy in diabetic macular edema is not as robust as in retinal angiogenesis (PDR). The anti-VEGF therapies appear to be of transient benefit as the edema recurs within a few weeks, and repeated injections are necessary.12 This creates a stronger case to analyze the additional molecular pathways to be a potential target in DR, and emerging studies indicate ‘retinal inflammation’ as an important player in the pathogenesis of DR.
INFLAMMATION IN DIABETIC RETINOPATHY
The role of inflammation in the development and progression of diabetic retinopathy has been studied for a long time, but in the last 10 years there has been enormous interest in the molecular mechanisms to develop novel therapeutic approaches. In the literature, as early as in 1960, Powell and Field observed that diabetics treated with anti-inflammatory agents like ‘salicylate’ had a lower incidence rate of diabetic retinopathy.13 This study suggests that inflammation may play an important role in the pathogenesis of diabetic retinopathy. Earlier studies by Lutty and his group identified the important role of leukocytes in the development of diabetic retinopathy14,15 and a subsequent study has established diabetic retinopathy as an ‘inflammatory disease’.16
It is important to note that inflammation starts very early and within one week of experimental diabetes, leukocytes accumulate in the vasculature of the retina.16 The important components of the inflammatory phenotype that are widely observed in diabetic retinopathy [Table 1] progress toward increased retinal vascular permeability (diabetic macular edema) and neovascularization (proliferative diabetic retinopathy).
Table 1.

INFLAMMATORY BIOMARKERS IN DIABETIC RETINOPATHY
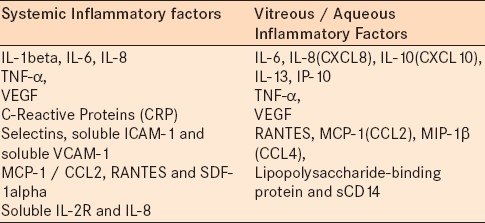
The important evidence for inflammation as a component of retinal dysfunction in diabetes is established through clinical studies. The traditional risk factors for diabetic retinopathy include duration of diabetes, hyperglycemia, hypertension, and dyslipidemia, which explains only some degree of variance in the risk of diabetic retinopathy.39 Studies have shown the association of multiple systemic inflammatory factors in the progression of diabetic retinopathy, and a further analysis of diabetic vitreous samples has provided insights into novel proinflammatory markers in the process.40,41 These novel inflammatory risk factors identified in the human population not only serve as potential biomarkers, but they also give insights into the development of potential molecular targets for treating diabetic retinopathy.
Systemic factors
Systemic inflammatory factors are considered to help in the prediction and progression of diabetic retinopathy. The systemic biomarkers that have been extensively studied and are well known are: C-reactive protein (CRP) and vascular cell adhesion molecules (selectins, soluble ICAM-1, and soluble VCAM-1) in microvascular diabetic complications.42–45 Clinical studies have identified that the elevated serum concentrations of IL-1beta, TNF-alpha, and VEGF, correlate with the presence and severity of diabetic retinopathy.46 VEGF is an important proinflammatory factor that is considered to be the key molecule in the pathogenesis of diabetic retinopathy, and its serum levels are seen to be associated with the development of diabetic retinopathy.47 Studies have shown that the levels of MCP-1, IL-1beta, and IL-6, IL-8, TNF-alpha (P = 0.002) and VEGF (P < 0.0001) are elevated in PDR, and interestingly in one study the serum levels of TNF-alpha (P = 0.05) and VEGF (P = 0.047) are correlated with the levels of HbA1c.48,49 Additionally, studies have also shown that the serum chemokines, Regulated upon Activation, Normal T cell Expressed, and Secreted (RANTES) and SDF-1alpha are significantly elevated in patients with non-proliferative diabetic retinopathy compared to those who have less severe retinopathy.50 In addition, studies have suggested a role for serum sIL-2R, IL-8, and TNF-alpha in the pathophysiology and progression of diabetic retinopathy.51 Also the elevated neutrophil count is associated with the presence and severity of retinopathy.52 All these potentially proinflammatory molecules may act together in the development of diabetic retinopathy.
Local factors
Elevated levels of the aqueous and vitreous chemokines and cell adhesion molecules are seen to be elevated in subjects with diabetic retinopathy. Immunostaining in human diabetic eyes has shown that TNF-α, MCP-1, and RANTES are positive in the diabetic eyes, and further ICAM-1 staining is strongly positive throughout the diabetic retina.50 In a comprehensive study using a cytokine array, Suzuki et al. have shown that the levels of IL-6, IL-8 (CXCL8), IL-10 (CXCL10), IL-13, IP-10, MCP-1(CCL2), MIP-1β (CCL4), PDGF, and VEGF in the vitreous fluid are significantly higher in any sign of a diabetic retinopathy group.53 Studies have shown that the elevated vitreous IL-8 levels in diabetic subjects are associated with deteriorating visual acuity in diabetic retinopathy.54 Elevation of the aqueous concentration of MCP-1 / CCL2, IP-10, and IL-8 levels in eyes with severe NPDR suggests that inflammatory changes precede the development of neovascularization in PDR.55 The positive correlation between the aqueous levels of IL-6 and macular thickness indicates that IL-6 may play a central role in the development of diabetic macular edema.56 Vitreous fluid levels of VEGF, ICAM-1, IL-6, and MCP-1 have been shown to be related to the retinal vascular permeability and severity of DME [24). Additionally, in PDR, it has been shown that the levels of IL-6, IL-8, MCP-1, and VEGF, in the vitreous, were significantly higher.57 Lipopolysaccharide-binding protein and sCD14 are also shown to be elevated in the vitreous fluid of patients with PDR, and are thus considered to play an important role in the innate immune response triggered by the inflammatory injury characteristic of PDR.58
The important observation one can infer from the above set of studies are: (1) There are common sets of inflammatory cytokines that are upregulated in both serum and vitreous / aqueous samples in subjects with retinopathy, (2) the same kind of cytokines are involved in the pathogenesis of both PDR and NPDR [Table 2] and these cytokines can have multiple interactions to impact the pathogenesis of diabetic retinopathy.54,55,59 Despite the important function of VEGF as an angiogenic and inflammatory factor, studies have observed that a selective blocking of VEGF using bevacizumab injections (anti-VEGF) has no influence on the control of other immunogenic cytokines, such as, MCP-1 and IL-6, in the aqueous humor of DME.60 In addition to the presence of inflammatory factors in the serum and vitreous samples in diabetic retinopathy, studies have reported that the tears in diabetics exhibited elevated levels of IP-10 and MCP-1.61 The importance of inflammatory chemokines in the progression of diabetic retinopathy is further supported from genetic evidences. Studies have shown the association between diabetic retinopathy and monocyte chemoattractant protein (MCP)-1 / CCl2 gene polymorphism, and this study suggests that the A-2518G polymorphism is a susceptible allele for diabetic retinopathy.62 In addition, the EE genotype of the K469E polymorphism of the ICAM-1 has been shown to be a risk factor for PDR in type 2 diabetes.63 The (CCTTT)15 allele in the promoter of the inducible nitric oxide synthase (iNOS) gene, and the (GT)13 promoter polymorphism in the tumor necrosis factor β (TNFb) gene are also shown to be increased risk factors for diabetic retinopathy64
Table 2.
Inflammatory Biomarkers in Diabetic Retinopathy
ROLE OF INFLAMMATION IN THE ALTERATION OF BLOOD RETINAL BARRIER
Schroder et al., first described leukostasis in the retinal vasculature as an important phenomenon for the increased neutrophils and monocytes that have been shown to be associated with retinal vascular abnormalities in diabetic rat models.65 Increased density of neutrophil levels have been shown in the diabetic retinal vasculature, particularly around the optic disc and macula.66 Miyamoto et al., have shown that retinal vascular permeability and nonperfusion are associated with retinal leukostasis in the rat model of streptozotocin-induced diabetes.19 One of the earlier events in diabetic retinal inflammation is the adhesion of leukocytes to the microvasculature. Increased leukocyte adhesion results in loss of endothelial cells and breakdown of the blood-retinal barrier.25,67 The leukocyte adhesion in the diabetic retina is facilitated by the increased expression of adhesion molecules such as, ICAM-1 and P-selectin, on the endothelium and its leukocyte counter-receptor CD18.15,17 The inhibition of leukocyte adhesion prevents the loss of pericytes and the formation of acellular capillaries, leading to suppression of the blood-retinal barrier breakdown in animal models of diabetic retinopathy.25 From these observations, the key inflammatory events involved in the endothelial barrier alteration appear to be: (1) Increased expression of endothelial adhesion molecules such as ICAM1, VCAM1, PECAM-1, P-Selectin, and so on, (2) Adhesion of leukocytes to the endothelium, (3) release of inflammatory cytokines, vascular permeability factors, and growth cytokines, (4) alteration of adherens and tight junctional proteins (e.g., VE-Cadherin, ZO-1, Claudin etc.), and (5) infiltration of leukocytes (diapedesis) into the retina resulting in the alteration of the blood retinal barrier [Figure 1].
Figure 1.

Diabetes through hyperglycemia causes upregulation of adhesion molecules such as ICAM-1 and VCAM-1 in the endothelial lining of the retinal microvasculature. Further activation of leukocytes in diabetes leads to their attachment to endothelial cells. Attachment of the leukocytes augments microvessel damage through the secretion of proinflammatory molecules (VEGF, TNF-α, Ang-2, proteinases, chemokines) and leukostasis, which leads to the alteration of the blood retinal barrier, resulting in diabetic macular edema (DME)
Alteration of the inner blood retinal barrier (BRB) is the hallmark of diabetic retinopathy. The inner BRB is made of endothelial cells in the inner retina and the endothelial cells form a very tight monolayer, formed by the adherens and the tight junction proteins. The principal proteins found in the endothelial tight junctions are occludin, claudin-5, and JAMs, and the adherent junctional protein VE-Cadherin, which creates a tight seal so that water-soluble molecules cannot easily penetrate between the cells. The blood-retinal barrier integrity is also maintained tightly by the presence of pericytes and astrocytes around the endothelium. The normal BRB has been shown to be impermeable to leukocytes, but studies have shown that the activation of leukocytes leads to the transient breakdown of the BRB, resulting in the augmentation of the proinflammatory reaction in the retina. The role of leukocyte migration from the blood into the retinal tissue is considered to be important for the breakdown of the blood retinal barrier. It has been shown that diapedesis occurs in the retina when both the leukocytes and the endothelium are activated.68 The process of diapedesis is mediated through the alteration of adherent and tight junctional proteins in the endothelial cells, and further studies have shown that the cell junctional molecules dissociate during intercellular leukocyte migration. Inflammatory cytokines such as TNF-α and VEGF in the diabetic retina have previously been shown to alter the vascular permeability by decreasing the levels of tight junction proteins (occludin and ZO-1) and the adherens junction protein (VE-cadherin).69–72 Observations from our laboratory suggest a possible mechanism by which diabetes contributes to BRB breakdown through the proteolytic degradation of VE-cadherin.73 This brings out the possibility that the endothelial cell-associated MMPs or leukocyte proteases may help in leukocyte migration through the alteration of VE-cadherin molecules.
INFLAMMATORY MEDIATORS AS THERAPEUTIC TARGETS
Vascular endothelial growth factor
VEGF has been the most important factor that has been investigated extensively in relation to the alteration of the blood retinal barrier. The VEGF levels are significantly elevated in patients with DME when compared with non-diabetic eye conditions.28,74 VEGF is a potent vasoactive cytokine that causes increased vascular permeability. It affects endothelial tight junction proteins, resulting in extravasation of fluid and retinal edema. VEGF induces the phosphorylation of VE-cadherin, occludin, and ZO-1, and thus causes a disruption of the barrier.75 VEGF also stimulates increased leukostasis in the microvessels of the retina, and the sticky leukocytes release cytokines or may migrate via the transendothelial route, which causes BRB breakdown.76,77
The majority of drugs that are in the clinical trials in DME are drugs that target the molecule, VEGF. Drugs that directly inhibit the VEGF molecule include the anti-VEGF aptamer, pegaptanib (Macugen, OSI Pharmaceuticals), the monoclonal antibody fragment Ranibizumab (Lucentis, Genentech Inc., San Francisco), and the full length antibody bevacizumab (Avastin, Genentech Inc.). Other treatment modalities include soluble VEGF receptor analogs, VEGF-Trap (Regeneron Pharmaceuticals, Tarrytown, NY), small interfering RNAs (siRNAs) bevasiranib (Opko Health Inc., Miami, FL), and rapamycin (Sirolimus, MacuSight Inc., Union City, CA). The DRCR Network study (Protocol I) has shown that after two years, the mean change in the visual acuity letter score from the baseline was 3.7 letters greater in the ranibizumab + prompt laser group, 5.8 letters greater in the ranibizumab + deferred laser group, and 1.5 letters worse in the triamcinolone + prompt laser group.78 However, response to the anti-VEGF treatments in DME is variable, and is not as robust as in PDR or neovascular glaucoma. This clinical observation indicates that the pathogenesis of DME is multifactorial, and many other factors beyond VEGF may play a role in this process.
Angiopoietin-2
Angiopoietins represent another family of inflammatory growth factors that bind to the receptor tyrosine kinase Tie2, and are important modulators of angiogenesis. The vitreous levels of angiopoietin-2 are significantly elevated in patients with clinically significant macular edema (CSME),79 indicating the role of Ang-2 in the alteration of the blood-retinal barrier. In a recent study, we have shown increased expression of Ang-2 mRNA and proteins in the retinas of diabetic animals.80 Intravitreal injection of Ang-2 in non-diabetic rats shows a three-fold increase in retinal vascular permeability. Ang-2 also induces loss of VE-cadherin mediated through the phosphorylation mechanism.80 A recent study through cellular experiments shows that Ang-2 promotes adhesion of monocytes by sensitizing endothelial cells toward TNF-alpha and modulates the TNF-alpha-induced expression of endothelial cell adhesion molecules, ICAM-1. This finding identifies Ang-2 as an autocrine regulator of endothelial cell inflammatory responses. Ang-2 thereby acts as a switch of vascular responsiveness, exerting a permissive role for the augmentation of proinflammatory cytokines.81 Thus, Ang-2 appears to serve as an important therapeutic target in DME. At least in tumor models, Ang-2 inhibitors have been found to very effective in preventing tumor growth through the modulation of monocyte infiltration and angiogenesis.82 Clinical trials using Ang-2 inhibitors are ongoing in patients with advanced solid tumors and renal cell carcinoma.
Proteinases
There is increasing evidence implicating MMPs (matrix metalloproteinases) as major regulators of innate and acquired immunity.83 Furthermore, studies in knockout mice have shown that MMPs play an important role in acute as well as chronic inflammation.84 Studies have also shown that the proteolytic alteration of chemokines by MMPs is important for its activation. Several members of the CCL / monocyte chemoattractant protein (MCP) family of chemokines are cleaved by MMPs rendering them proactive molecules that amplify the inflammation. In addition, MMPs orchestrate the recruitment of leukocytes as an essential component of tumor-associated inflammation.83 It is now evident that MMPs also play an important role in the pathogenesis of diabetic retinopathy. The vitreous level of proteinases, like MMP9, has been shown to be higher in diabetic subjects with DR than in diabetic subjects without DR.85 Furthermore, in the animal model of diabetes, both MMP2 and MMP9 are elevated in the retinas.86 We have shown that retinal vascular permeability in diabetic animals is significantly increased, coincident with a decrease of cell–cell junctional protein, VE-cadherin. This increased vascular permeability could be inhibited with an MMP inhibitor.73 These observations suggest a possible mechanism by which diabetes contributes to BRB breakdown through the proteolytic degradation of VE-cadherin. This may indicate a role for extracellular proteinases in the alteration of the BRB seen in diabetic retinopathy.73 High glucose can activate many soluble mediators such as AGE, ROS, and inflammatory cytokines, which can increase MMP expression and activity in a diabetic state. Retinal inflammation attracts increased leukocytes to the retina, which then bind to the vascular endothelium. The binding of leukocytes to the endothelial cells activates cellular proteinases such as elastase, which may clip off VE-cadherin and its associated protein from the cell surface, resulting in endothelial monolayer alteration.87 Based on the findings in our diabetic animal studies, it appears that proteinases may serve as important therapeutic targets in DME.
Chemokines
Chemokines levels in diabetic retinopathy have been shown to be significantly elevated, and in various studies, the most common chemokine that is significantly elevated in serum and vitreous is identified to be CCL2.88,89 The chemokine ligand 2 (CCL2), also known as monocyte chemotactic protein-1 (MCP-1) is considered to play an important role in vascular inflammation by inducing leukocyte recruitment and activation. Hyperglycemia has been shown to increase the CCL2 / MCP-1 generation from retinal vascular endothelial cells, pigmented epithelial cells, and Muller's glial cells.90 Furthermore, the vitreous CCL2 level has been reported to be increased in patients with diabetic retinopathy,91,92 and the CCL2 gene polymorphism has been indicated as a potential risk factor for diabetic retinopathy.62 Studies from our laboratory have indicated that a genetic knock out of the CCL2 gene in diabetic mice prevented blood retinal barrier alteration (ARVO abstract, 2011). Our preliminary animal studies indicate that selective inhibition of the CCL2 gene can prevent the alteration of the BRB in diabetes, and further studies using selective inhibitors of CCL2 and CCR2 are in progress. There is systemic use of CCR2 inhibitors in various clinical trials, in several inflammatory diseases like atherosclerosis, multiple sclerosis, rheumatoid arthritis, and systemic lupus erythematosus (SLE).
CONCLUSIONS
Diabetic macular edema is a multifactorial process that involves many factors beyond VEGF. Many clinical trials in DME have shown that in spite of repeated anti-VEGF drug injections, more than 50% of the patients with DME still have persistent macular edema (> 250 microns in thickness) at the end of a one-year study (DRCR Protocol I). The effect of anti-VEGF treatment in DME appears to be less robust compared to its effect in angiogenesis (PDR). It is possible that inflammatory mediators like Ang-2, proteinases, and chemokines may be involved in the pathophysiology of BRB breakdown in diabetic retinopathy. The VEGF inhibition itself may not achieve neutralization of other inflammatory molecules involved in the cascade of the breakdown of BRB. Steroids that target many inflammatory molecules including Ang-2, TNF-α, and chemokines, work very effectively in patients with DME.93 However, the steroids have side effects like increased intraocular pressure and cataract formation that limit their use in DME. The novel selective inhibitors of the inflammatory cascade, including those for molecules like Ang-2, TNF-α, proteinases, and CCL2 may be useful therapeutic agents in the treatment of DME in the future, either alone or in combination with the currently used anti-VEGF drugs.
Footnotes
Source of Support: Nil
Conflict of Interest: None declared.
REFERENCES
- 1.Shaw JE, Sicree RA, Zimmet PZ. Global estimates of the prevalence of diabetes for 2010 and 2030. Diabetes Res Clin Pract. 2010;87:4–14. doi: 10.1016/j.diabres.2009.10.007. [DOI] [PubMed] [Google Scholar]
- 2.From the Centers for Disease Control and Prevention: Blindness caused by diabetes-- Massachusetts, 1987-1994. JAMA. 1996;276:1865–6. [PubMed] [Google Scholar]
- 3.Aiello LM. Perspectives on diabetic retinopathy. Am J Ophthalmol. 2003;136:122–35. doi: 10.1016/s0002-9394(03)00219-8. [DOI] [PubMed] [Google Scholar]
- 4.Ciulla TA, Amador AG, Zinman B. Diabetic retinopathy and diabetic macular edema: pathophysiology, screening, and novel therapies. Diabetes Care. 2003;26:2653–64. doi: 10.2337/diacare.26.9.2653. [DOI] [PubMed] [Google Scholar]
- 5.Klein R, Klein BE, Moss SE, Cruickshanks KJ. The Wisconsin Epidemiologic Study of Diabetic Retinopathy: XVII.The 14-year incidence and progression of diabetic retinopathy and associated risk factors in type 1 diabetes. Ophthalmology. 1998;105:1801–15. doi: 10.1016/S0161-6420(98)91020-X. [DOI] [PubMed] [Google Scholar]
- 6.The Diabetes Control and Complications Trial Research Group: The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med. 1993;329:977–86. doi: 10.1056/NEJM199309303291401. [DOI] [PubMed] [Google Scholar]
- 7.UK Prospective Diabetes Study (UKPDS) Group: Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33) Lancet. 1998;352:837–53. [PubMed] [Google Scholar]
- 8.Villar G, García Y, Goicolea I, Vázquez JA. Determinants of development of microalbuminuria in normotensive patients with type 1 and type 2 diabetes. Diabetes Metab. 1999;25:246–54. [PubMed] [Google Scholar]
- 9.Reaven PD, Emanuele N, Moritz T, Klein R, Davis M, Glander K, et al. Proliferative diabetic retinopathy in type 2 diabetes is related to coronary artery calcium in the Veterans Affairs Diabetes Trial (VADT) Diabetes Care. 2008;31:952–7. doi: 10.2337/dc07-1926. [DOI] [PubMed] [Google Scholar]
- 10.Brownlee M. The pathobiology of diabetic complications: A unifying mechanism. Diabetes. 2005;54:1615–25. doi: 10.2337/diabetes.54.6.1615. [DOI] [PubMed] [Google Scholar]
- 11.Pober JS, Sessa WC. Evolving functions of endothelial cells in inflammation. Nat Rev Immunol. 2007;7:803–15. doi: 10.1038/nri2171. [DOI] [PubMed] [Google Scholar]
- 12.Sang DN, D’Amore PA. Is blockade of vascular endothelial growth factor beneficial for all types of diabetic retinopathy? Diabetologia. 2008;51:1570–3. doi: 10.1007/s00125-008-1078-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Powell ED, Field RA. Diabetic retinopathy and rheumatoid arthritis. Lancet. 1964;2:17–8. doi: 10.1016/s0140-6736(64)90008-x. [DOI] [PubMed] [Google Scholar]
- 14.Lutty GA, Cao J, McLeod DS. Relationship of polymorphonuclear leukocytes to capillary dropout in the human diabetic choroid. Am J Pathol. 1997;151:707–14. [PMC free article] [PubMed] [Google Scholar]
- 15.McLeod DS, Lefer DJ, Merges C, Lutty GA. Enhanced expression of intracellular adhesion molecule-1 and P-selectin in the diabetic human retina and choroid. Am J Pathol. 1995;147:642–53. [PMC free article] [PubMed] [Google Scholar]
- 16.Adamis AP. Is diabetic retinopathy an inflammatory disease? Br J Ophthalmol. 2002;86:363–5. doi: 10.1136/bjo.86.4.363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Adamis AP, Berman AJ. Immunological mechanisms in the pathogenesis of diabetic retinopathy. Semin Immunopathol. 2008;30:65–84. doi: 10.1007/s00281-008-0111-x. [DOI] [PubMed] [Google Scholar]
- 18.Antonetti DA, Barber AJ, Bronson SK, Freeman WM, Gardner TW, Jefferson LS, et al. Diabetic retinopathy: seeing beyond glucose-induced microvascular disease. Diabetes. 2006;55:2401–11. doi: 10.2337/db05-1635. [DOI] [PubMed] [Google Scholar]
- 19.Miyamoto K, Khosrof S, Bursell SE, Rohan R, Murata T, Clermont AC, et al. Prevention of leukostasis and vascular leakage in streptozotocin-induced diabetic retinopathy via intercellular adhesion molecule-1 inhibition. Proc Natl Acad Sci U S A. 1999;96:10836–41. doi: 10.1073/pnas.96.19.10836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Joussen AM, Poulaki V, Le ML, Koizumi K, Esser C, Janicki H, et al. A central role for inflammation in the pathogenesis of diabetic retinopathy. FASEB J. 2004;18:1450–2. doi: 10.1096/fj.03-1476fje. [DOI] [PubMed] [Google Scholar]
- 21.Aiello LP, Avery RL, Arrigg PG, Keyt BA, Jampel HD, Shah ST, et al. Vascular endothelial growth factor in ocular fluid of patients with diabetic retinopathy and other retinal disorders. N Engl J Med. 1994;331:1480–7. doi: 10.1056/NEJM199412013312203. [DOI] [PubMed] [Google Scholar]
- 22.Adamis AP, Miller JW, Bernal MT, D’Amico DJ, Folkman J, Yeo TK, et al. Increased vascular endothelial growth factor levels in the vitreous of eyes with proliferative diabetic retinopathy. Am J Ophthalmol. 1994;118:445–50. doi: 10.1016/s0002-9394(14)75794-0. [DOI] [PubMed] [Google Scholar]
- 23.Demircan N, Safran BG, Soylu M, Ozcan AA, Sizmaz S. Determination of vitreous interleukin-1 (IL-1) and tumour necrosis factor (TNF) levels in proliferative diabetic retinopathy. Eye (Lond) 2006;20:1366–9. doi: 10.1038/sj.eye.6702138. [DOI] [PubMed] [Google Scholar]
- 24.Funatsu H, Yamashita H, Shimizu E, Kojima R, Hori S. Relationship between vascular endothelial growth factor and interleukin-6 in diabetic retinopathy. Retina. 2001;21:469–77. doi: 10.1097/00006982-200110000-00009. [DOI] [PubMed] [Google Scholar]
- 25.Yuuki T, Kanda T, Kimura Y, Kotajima N, Tamura J, Kobayashi I, et al. Inflammatory cytokines in vitreous fluid and serum of patients with diabetic vitreoretinopathy. J Diabetes Complications. 2001;15:257–9. doi: 10.1016/s1056-8727(01)00155-6. [DOI] [PubMed] [Google Scholar]
- 26.Canataroglu H, Varinli I, Ozcan AA, Canataroglu A, Doran F, Varinli S. Interleukin (IL)-6, interleukin (IL)-8 levels and cellular composition of the vitreous humor in proliferative diabetic retinopathy, proliferative vitreoretinopathy, and traumatic proliferative vitreoretinopathy. Ocul Immunol Inflamm. 2005;13:375–81. doi: 10.1080/09273940490518900. [DOI] [PubMed] [Google Scholar]
- 27.Brooks HL, Jr, Caballero S, Jr, Newell CK, Steinmetz RL, Watson D, Segal MS, et al. Vitreous levels of vascular endothelial growth factor and stromal-derived factor 1 in patients with diabetic retinopathy and cystoid macular edema before and after intraocular injection of triamcinolone. Arch Ophthalmol. 2004;122:1801–7. doi: 10.1001/archopht.122.12.1801. [DOI] [PubMed] [Google Scholar]
- 28.Funatsu H, Yamashita H, Ikeda T, Nakanishi Y, Kitano S, Hori S. Angiotensin II and vascular endothelial growth factor in the vitreous fluid of patients with diabetic macular edema and other retinal disorders. Am J Ophthalmol. 2002;133:537–43. doi: 10.1016/s0002-9394(02)01323-5. [DOI] [PubMed] [Google Scholar]
- 29.Patel JI, Hykin PG, Gregor ZJ, Boulton M, Cree IA. Angiopoietin concentrations in diabetic retinopathy. Br J Ophthalmol. 2005;89:480–3. doi: 10.1136/bjo.2004.049940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Watanabe D, Suzuma K, Suzuma I, Ohashi H, Ojima T, Kurimoto M, et al. Vitreous levels of angiopoietin 2 and vascular endothelial growth factor in patients with proliferative diabetic retinopathy. Am J Ophthalmol. 2005;139:476–81. doi: 10.1016/j.ajo.2004.10.004. [DOI] [PubMed] [Google Scholar]
- 31.Limb GA, Chignell AH, Green W, LeRoy F, Dumonde DC. Distribution of TNF alpha and its reactive vascular adhesion molecules in fibrovascular membranes of proliferative diabetic retinopathy. Br J Ophthalmol. 1996;80:168–73. doi: 10.1136/bjo.80.2.168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Meleth AD, Agrón E, Chan CC, Reed GF, Arora K, Byrnes G, et al. Serum inflammatory markers in diabetic retinopathy. Invest Ophthalmol Vis Sci. 2005;46:4295–301. doi: 10.1167/iovs.04-1057. [DOI] [PubMed] [Google Scholar]
- 33.Rungger-Brandle E, Dosso AA, Leuenberger PM. Glial reactivity, an early feature of diabetic retinopathy. Invest Ophthalmol Vis Sci. 2000;41:1971–80. [PubMed] [Google Scholar]
- 34.Zeng XX, Ng YK, Ling EA. Neuronal and microglial response in the retina of streptozotocin-induced diabetic rats. Vis Neurosci. 2000;17:463–71. doi: 10.1017/s0952523800173122. [DOI] [PubMed] [Google Scholar]
- 35.Krady JK, Basu A, Allen CM, Xu Y, LaNoue KF, Gardner TW, et al. Minocycline reduces proinflammatory cytokine expression, microglial activation, and caspase-3 activation in a rodent model of diabetic retinopathy. Diabetes. 2005;54:1559–65. doi: 10.2337/diabetes.54.5.1559. [DOI] [PubMed] [Google Scholar]
- 36.Esser P, Heimann K, Wiedemann P. Macrophages in proliferative vitreoretinopathy and proliferative diabetic retinopathy: Differentiation of subpopulations. Br J Ophthalmol. 1993;77:731–3. doi: 10.1136/bjo.77.11.731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Barber AJ, Lieth E, Khin SA, Antonetti DA, Buchanan AG, Gardner TW. Neural apoptosis in the retina during experimental and human diabetes. Early onset and effect of insulin. J Clin Invest. 1998;102:783–91. doi: 10.1172/JCI2425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Mizutani M, Kern TS, Lorenzi M. Accelerated death of retinal microvascular cells in human and experimental diabetic retinopathy. J Clin Invest. 1996;97:2883–90. doi: 10.1172/JCI118746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Nguyen TT, Alibrahim E, Islam FM, Klein R, Klein BE, Cotch MF, et al. Inflammatory, hemostatic, and other novel biomarkers for diabetic retinopathy: the multi-ethnic study of atherosclerosis. Diabetes Care. 2009;32:1704–9. doi: 10.2337/dc09-0102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Schmidt AM, Hori O, Chen JX, Li JF, Crandall J, Zhang J, et al. Advanced glycation endproducts interacting with their endothelial receptor induce expression of vascular cell adhesion molecule-1 (VCAM-1) in cultured human endothelial cells and in mice: A potential mechanism for the accelerated vasculopathy of diabetes. J Clin Invest. 1995;96:1395–403. doi: 10.1172/JCI118175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Schram MT, Chaturvedi N, Schalkwijk C, Giorgino F, Ebeling P, Fuller JH, et al. EURODIAB Prospective Complications Study. Vascular risk factors and markers of endothelial function as determinants of inflammatory markers in type 1 diabetes: the EURODIAB Prospective Complications Study. Diabetes Care. 2003;26:2165–73. doi: 10.2337/diacare.26.7.2165. [DOI] [PubMed] [Google Scholar]
- 42.Olson JA, Whitelaw CM, McHardy KC, Pearson DW, Forrester JV. Soluble leucocyte adhesion molecules in diabetic retinopathy stimulate retinal capillary endothelial cell migration. Diabetologia. 1997;40:1166–71. doi: 10.1007/s001250050802. [DOI] [PubMed] [Google Scholar]
- 43.Matsumoto K, Sera Y, Ueki Y, Inukai G, Niiro E, Miyake S. Comparison of serum concentrations of soluble adhesion molecules in diabetic microangiopathy and macroangiopathy. Diabet Med. 2002;19:822–6. doi: 10.1046/j.1464-5491.2002.00799.x. [DOI] [PubMed] [Google Scholar]
- 44.Nguyen TT, Alibrahim E, Islam FM, Klein R, Klein BE, Cotch MF, et al. Inflammatory, hemostatic, and other novel biomarkers for diabetic retinopathy: the multi-ethnic study of atherosclerosis. Diabetes Care. 2009;32:1704–9. doi: 10.2337/dc09-0102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Klein BE, Knudtson MD, Tsai MY, Klein R. The relation of markers of inflammation and endothelial dysfunction to the prevalence and progression of diabetic retinopathy: Wisconsin epidemiologic study of diabetic retinopathy. Arch Ophthalmol. 2009;127:1175–82. doi: 10.1001/archophthalmol.2009.172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Kaul K, Hodgkinson A, Tarr JM, Kohner EM, Chibber R. Is inflammation a common retinal-renal-nerve pathogenic link in diabetes? Curr Diabetes Rev. 2010;6:294–303. doi: 10.2174/157339910793360851. [DOI] [PubMed] [Google Scholar]
- 47.Ozturk BT, Bozkurt B, Kerimoglu H, Okka M, Kamis U, Gunduz K. Effect of serum cytokines and VEGF levels on diabetic retinopathy and macular thickness. Mol Vis. 2009;15:1906–14. [PMC free article] [PubMed] [Google Scholar]
- 48.Koleva-Georgieva DN, Sivkova NP, Terzieva D. Serum inflammatory cytokines IL-1beta, IL-6, TNF-alpha and VEGF have influence on the development of diabetic retinopathy. Folia Med (Plovdiv) 2011;53:44–50. doi: 10.2478/v10153-010-0036-8. [DOI] [PubMed] [Google Scholar]
- 49.Maier R, Weger M, Haller-Schober EM, El-Shabrawi Y, Wedrich A, Theisl A, et al. Multiplex bead analysis of vitreous and serum concentrations of inflammatory and proangiogenic factors in diabetic patients. Mol Vis. 2008;27(14):637–43. [PMC free article] [PubMed] [Google Scholar]
- 50.Meleth AD, Agrón E, Chan CC, Reed GF, Arora K, Byrnes G, et al. Serum inflammatory markers in diabetic retinopathy. Invest Ophthalmol Vis Sci. 2005;46:4295–301. doi: 10.1167/iovs.04-1057. [DOI] [PubMed] [Google Scholar]
- 51.Doganay S, Evereklioglu C, Er H, Türköz Y, Sevinç A, Mehmet N, et al. Comparison of serum NO, TNF-alpha, IL-1beta, sIL-2R, IL-6 and IL-8 levels with grades of retinopathy in patients with diabetes mellitus. Eye (Lond) 2002;16:163–70. doi: 10.1038/sj.eye.6700095. [DOI] [PubMed] [Google Scholar]
- 52.Woo SJ, Ahn SJ, Ahn J, Park KH, Lee K. Elevated Systemic Neutrophil Count in Diabetic Retinopathy and Diabetes: A Hospital-Based Cross-sectional Study of 30,793 Korean Subjects. Invest Ophthalmol Vis Sci. 2011;52:7697–703. doi: 10.1167/iovs.11-7784. [DOI] [PubMed] [Google Scholar]
- 53.Suzuki Y, Nakazawa M, Suzuki K, Yamazaki H, Miyagawa Y. Expression profiles of cytokines and chemokines in vitreous fluid in diabetic retinopathy and central retinal vein occlusion. Jpn J Ophthalmol. 2011;55:256–63. doi: 10.1007/s10384-011-0004-8. [DOI] [PubMed] [Google Scholar]
- 54.Petrovic MG, Korošec P, Košnik M, Hawlina M. Association of preoperative vitreous IL-8 and VEGF levels with visual acuity after vitrectomy in proliferative diabetic retinopathy. Acta Ophthalmol. 2010;88:e311–6. doi: 10.1111/j.1755-3768.2010.02030.x. [DOI] [PubMed] [Google Scholar]
- 55.Yoshimura T, Sonoda KH, Sugahara M, Mochizuki Y, Enaida H, Oshima Y, et al. Comprehensive analysis of inflammatory immune mediators in vitreoretinal diseases. PLoS One. 2009;4:e8158. doi: 10.1371/journal.pone.0008158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Oh IK, Kim SW, Oh J, Lee TS, Huh K. Inflammatory and angiogenic factors in the aqueous humor and the relationship to diabetic retinopathy. Curr Eye Res. 2010;35:1116–27. doi: 10.3109/02713683.2010.510257. [DOI] [PubMed] [Google Scholar]
- 57.Murugeswari P, Shukla D, Rajendran A, Kim R, Namperumalsamy P, Muthukkaruppan V. Proinflammatory cytokines and angiogenic and anti-angiogenic factors in vitreous of patients with proliferative diabetic retinopathy and eales’ disease. Retina. 2008;28:817–24. doi: 10.1097/IAE.0b013e31816576d5. [DOI] [PubMed] [Google Scholar]
- 58.Hernández C, Ortega F, García-Ramírez M, Villarroel M, Casado J, García-Pascual L, et al. Lipopolysaccharide-binding protein and soluble CD14 in the vitreous fluid of patients with proliferative diabetic retinopathy. Retina. 2010;30:345–52. doi: 10.1097/iae.0b013e3181b7738b. [DOI] [PubMed] [Google Scholar]
- 59.Wakabayashi Y, Usui Y, Okunuki Y, Kezuka T, Takeuchi M, Goto H, et al. Correlation of vascular endothelial growth factor with chemokines in the vitreous in diabetic retinopathy. Retina. 2010;30:339–44. doi: 10.1097/IAE.0b013e3181bd2f44. [DOI] [PubMed] [Google Scholar]
- 60.Funk M, Schmidinger G, Maar N, Bolz M, Benesch T, Zlabinger GJ, et al. Angiogenic and inflammatory markers in the intraocular fluid of eyes with diabetic macular edema and influence of therapy with bevacizumab. Retina. 2010;30:1412–9. doi: 10.1097/IAE.0b013e3181e095c0. [DOI] [PubMed] [Google Scholar]
- 61.Liu J, Shi B, He S, Yao X, Willcox MD, Zhao Z. Changes to tear cytokines of type 2 diabetic patients with or without retinopathy. Mol Vis. 2010;16:2931–8. [PMC free article] [PubMed] [Google Scholar]
- 62.Katakami N, Matsuhisa M, Kaneto H, Matsuoka TA, Imamura K, Ishibashi F, et al. Monocyte chemoattractant protein-1 (MCP-1) gene polymorphism as a potential risk factor for diabetic retinopathy in Japanese patients with type 2 diabetes. Diabetes Res Clin Pract. 2010;89:e9–12. doi: 10.1016/j.diabres.2010.04.006. [DOI] [PubMed] [Google Scholar]
- 63.Petrovic MG, Osredkar J, Saraga-Babic M, Petrovic D. K469E polymorphism of the intracellular adhesion molecule 1 gene is associated with proliferative diabetic retinopathy in Caucasians with type 2 diabetes. Clin Experiment Ophthalmol. 2008;36:468–72. doi: 10.1111/j.1442-9071.2008.01785.x. [DOI] [PubMed] [Google Scholar]
- 64.Uthra S, Raman R, Mukesh BN, Rajkumar SA, Kumari P, Lakshmipathy P, et al. Diabetic retinopathy: Validation study of ALR2, RAGE, iNOS and TNFB gene variants in a south Indian cohort. Ophthalmic Genet. 2010;31:244–51. doi: 10.3109/13816810.2010.523037. [DOI] [PubMed] [Google Scholar]
- 65.Schröder S, Palinski W, Schmid-Schönbein GW. Activated monocytes and granulocytes, capillary nonperfusion, and neovascularization in diabetic retinopathy. Am J Pathol. 1991;139:81–100. [PMC free article] [PubMed] [Google Scholar]
- 66.Kim SY, Johnson MA, McLeod DS, Alexander T, Hansen BC, Lutty GA. Neutrophils are associated with capillary closure in spontaneously diabetic monkey retinas. Diabetes. 2005;54:1534–42. doi: 10.2337/diabetes.54.5.1534. [DOI] [PubMed] [Google Scholar]
- 67.Joussen AM, Murata T, Tsujikawa A, Kirchhof B, Bursell SE, Adamis AP. Leukocyte-mediated endothelial cell injury and death in the diabetic retina. Am J Pathol. 2001;158:147–52. doi: 10.1016/S0002-9440(10)63952-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Muller WA. Leukocyte-endothelial-cell interactions in leukocyte transmigration and the inflammatory response. Trends Immunol. 2003;24:327–34. doi: 10.1016/s1471-4906(03)00117-0. [DOI] [PubMed] [Google Scholar]
- 69.Wang J, Xu X, Elliott MH, Zhu M, Le YZ. Müller cell-derived VEGF is essential for diabetes-induced retinal inflammation and vascular leakage. Diabetes. 2010;59:2297–305. doi: 10.2337/db09-1420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Li J, Wang JJ, Chen D, Mott R, Yu Q, Ma JX, et al. Systemic administration of HMG-CoA reductase inhibitor protects the blood-retinal barrier and ameliorates retinal inflammation in type 2 diabetes. Exp Eye Res. 2009;89:71–8. doi: 10.1016/j.exer.2009.02.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Murakami T, Felinski EA, Antonetti DA. Occludin phosphorylation and ubiquitination regulate tight junction trafficking and vascular endothelial growth factor-induced permeability. J Biol Chem. 2009;284:21036–46. doi: 10.1074/jbc.M109.016766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Harhaj NS, Felinski EA, Wolpert EB, Sundstrom JM, Gardner TW, Antonetti DA. VEGF activation of protein kinase C stimulates occludin phosphorylation and contributes to endothelial permeability. Invest Ophthalmol Vis Sci. 2006;47:5106–15. doi: 10.1167/iovs.06-0322. [DOI] [PubMed] [Google Scholar]
- 73.Navaratna D, McGuire PG, Menicucci G, Das A. Proteolytic degradation of VE-cadherin alters the blood-retinal barrier in diabetes. Diabetes. 2007;56:2380–7. doi: 10.2337/db06-1694. [DOI] [PubMed] [Google Scholar]
- 74.Caldwell RB, Bartoli M, Behzadian MA, El-Remessy AE, Al-Shabrawey M, Platt DH, et al. Vascular endothelial growth factor and diabetic retinopathy:pathophysiological mechanisms and treatment perspectives. Diabetes Metab Res Rev. 2003;19:442–55. doi: 10.1002/dmrr.415. [DOI] [PubMed] [Google Scholar]
- 75.Antonetti DA, Barber AJ, Hollinger LA, Wolpert EB, Gardner TW. Vascular endothelial growth factor induces rapid phosphorylation of tight junction proteins occludin and zonula occluden 1. A potential mechanism for vascular permeability in diabetic retinopathy and tumors. J Biol Chem. 1999;274:23463–7. doi: 10.1074/jbc.274.33.23463. [DOI] [PubMed] [Google Scholar]
- 76.Aiello LP, Avery RL, Arrigg PG, Keyt BA, Jampel HD, Shah ST, et al. Vascular endothelial growth factor in ocular fluid of patients with diabetic retinopathy and other retinal disorders. N Engl J Med. 1994;331:1480–7. doi: 10.1056/NEJM199412013312203. [DOI] [PubMed] [Google Scholar]
- 77.Aiello LP, Bursell SE, Clermont A, Duh E, Ishii H, Takagi C, et al. Vascular endothelial growth factor-induced retinal permeability is mediated by protein kinase C in vivo and suppressed by an orally effective β-isoform-selective inhibitor. Diabetes. 1997;46:1473–80. doi: 10.2337/diab.46.9.1473. [DOI] [PubMed] [Google Scholar]
- 78.Elman MJ, Bressler NM, Qin H, Beck RW, Ferris FL, 3rd, Friedman SM, et al. Expanded 2-year follow-up of ranibizumab plus prompt or deferred laser or triamcinolone plus prompt laser for diabetic macular edema. Ophthalmology. 2011;118:609–14. doi: 10.1016/j.ophtha.2010.12.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Patel JI, Hykin PG, Gregor ZJ, Boulton M, Cree IA. Angiopoietin concentrations in diabetic retinopathy. Br J Ophthalmol. 2005;89:480–3. doi: 10.1136/bjo.2004.049940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Rangasamy S, Srinivasan R, Maestas J, McGuire PG, Das A. A potential role for angiopoietin-2 in the regulation of the blood-retinal barrier in diabetic retinopathy. Invest Ophthalmol Vis Sci. 2011;52:3784–91. doi: 10.1167/iovs.10-6386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Fiedler U, Reiss Y, Scharpfenecker M, Grunow V, Koidl S, Thurston G, et al. Angiopoietin-2 sensitizes endothelial cells to TNF-alpha and has a crucial role in the induction of inflammation. Nat Med. 2006;12:235–9. doi: 10.1038/nm1351. [DOI] [PubMed] [Google Scholar]
- 82.Huang H, Lai JY, Do J, Liu D, Li L, Del Rosario J, et al. Specifically targeting angiopoietin-2 inhibits angiogenesis, Tie2-expressing monocyte infiltration, and tumor growth. Clin Cancer Res. 2011;17:1001–11. doi: 10.1158/1078-0432.CCR-10-2317. [DOI] [PubMed] [Google Scholar]
- 83.Kessenbrock K, Plaks V, Werb Z. Matrix Metalloproteinases: regulators of the tumor microenvironment. Cell. 2010;141:52–67. doi: 10.1016/j.cell.2010.03.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Parks WC, Wilson CL, López-Boado YS. Matrix metalloproteinases as modulators of inflammation and innate immunity. Nat Rev Immunol. 2004;4:617–29. doi: 10.1038/nri1418. [DOI] [PubMed] [Google Scholar]
- 85.Jin M, Kashiwagi K, Iizuka Y, Tanaka Y, Imai M, Tsukahara S. Matrix metalloproteinases in human diabetic and nondiabetic vitreous. Retina. 2001;21:28–33. doi: 10.1097/00006982-200102000-00005. [DOI] [PubMed] [Google Scholar]
- 86.Giebel SJ, Menicucci G, McGuire PG, Das A. Matrix metalloproteinases in early diabetic retinopathy and their role in alteration of the blood-retinal barrier. Lab Invest. 2005;85:597–607. doi: 10.1038/labinvest.3700251. [DOI] [PubMed] [Google Scholar]
- 87.Allport JR, Muller WA, Luscinskas FW. Monocytes induce reversible focal changes in vascular endothelial cadherin complex during transendothelial migration under flow. J Cell Biol. 2000;148:203–16. doi: 10.1083/jcb.148.1.203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Elner SG, Elner VM, Jaffe GJ, Stuart A, Kunkel SL, Strieter RM. Cytokines in proliferative diabetic retinopathy and proliferative vitreoretinopathy. Curr Eye Res. 1995;14:1045–53. doi: 10.3109/02713689508998529. [DOI] [PubMed] [Google Scholar]
- 89.Abu el-Asrar AM, Van Damme J, Put W, Veckeneer M, Dralands L, Billiau A, et al. Monocyte chemotactic protein-1 in proliferative viteroretinal disorders. Am J Ophthalmol. 1997;123:599–606. doi: 10.1016/s0002-9394(14)71072-4. [DOI] [PubMed] [Google Scholar]
- 90.Bian ZM, Elner VM, Yoshida A, Kunkel SL, Elner SG. Signaling pathways for glycated human serum albumin-induced IL-8 and MCP-1 secretion in human RPE cells. Invest Ophthalmol Vis Sci. 2001;42:1660–8. [PubMed] [Google Scholar]
- 91.Capeans C, De Rojas MV, Lojo S, Salorio MS. C-C chemokines in the vitreous of patients with proliferative vitreoretinopathy and proliferative diabetic retinopathy. Retina. 1998;18:546–50. [PubMed] [Google Scholar]
- 92.Tashimo A, Mitamura Y, Nagai S, Nakamura Y, Ohtsuka K, Mizue Y, et al. Aqueous levels of macrophage migration inhibitory factor and monocyte chemotactic protein-1 in patients with diabetic retinopathy. Diabet Med. 2004;21:1292–7. doi: 10.1111/j.1464-5491.2004.01334.x. [DOI] [PubMed] [Google Scholar]
- 93.Campochiaro PA, Brown DM, Pearson A, Ciulla T, Boyer D, Holz FG, et al. Long-term benefit of sustained-delivery fluocinolone acetonide vitreous inserts for diabetic macular edema. Ophthalmology. 2011;118:626–35.e2. doi: 10.1016/j.ophtha.2010.12.028. [DOI] [PubMed] [Google Scholar]


