Abstract
Ischemia of the myocardium and lower limbs is a common consequence of arterial disease and a major source of morbidity and mortality in modernized countries. Inducing neovascularization for the treatment of ischemia is an appealing therapeutic strategy for patients for whom traditional treatment modalities cannot be performed or are ineffective. In the past, the stimulation of blood vessel growth was pursued using direct delivery of growth factors, angiogenic gene therapy, or cellular therapy. Although therapeutic angiogenesis holds great promise for treating patients with ischemia, current methods have not found success in clinical trials. Fibroblast growth factor-2 (FGF-2) was one of the first growth factors to be tested for use in therapeutic angiogenesis. Here, we present a method for improving the biological activity of FGF-2 by codelivering the growth factor with a liposomally embedded coreceptor, syndecan-4. This technique was shown to increase FGF-2 cellular signaling, uptake, and nuclear localization in comparison with FGF-2 alone. Delivery of syndecan-4 proteoliposomes also increased endothelial proliferation, migration, and angiogenic tube formation in response to FGF-2. Using an animal model of limb ischemia, syndecan-4 proteoliposomes markedly improved the neovascularization following femoral artery ligation and recovery of perfusion of the ischemic limb. Taken together, these results support liposomal delivery of syndecan-4 as an effective means to improving the potential of using growth factors to achieve therapeutic neovascularization of ischemic tissue.
Keywords: heparan sulfate, proteoglycan, arteriogenesis, growth factor trafficking
Chronic myocardial and peripheral ischemic disease affects more than 27 million patients in the United States and is the leading cause of morbidity and mortality in developed countries. Current therapy for ischemia consists of pharmacological interventions to slow the progression of vascular disease or physical interventions such as endovascular stent placement and surgical bypass of stenosed arteries. A significant proportion of patients with ischemic heart disease either cannot be treated with traditional revascularization methods or do not respond to these treatments. For these patients, there are currently no effective treatments for restoring the blood supply to the myocardial tissue. In the continuing search for new therapies for both refractory myocardial and peripheral ischemia, the therapeutic delivery of angiogenic growth factors has received much attention in both basic and clinical studies (1).
Angiogenesis is a complex physiological process through which new blood vessels develop. This process requires the coordination of endothelial cells, vascular smooth muscle cells, pericytes and macrophages with intricate regulation through cytokines, signaling cascades, and extracellular matrix molecules. Fibroblast growth factor-2 (FGF-2) was one of the first growth factors to be tested for clinical efficacy in treating myocardial and peripheral ischemia (2). This growth factor has been demonstrated to be an important regulator of endothelial cell proliferation, migration, and angiogenic differentiation (3). Fibroblast growth factor receptors (FGFRs) are transmembrane proteins that dimerize and undergo autophosphorylation following FGF binding (4). Members of the FGF family have a high affinity for cell-surface heparan sulfate proteoglycans and heparin (5). Heparan sulfate proteoglycans are complex molecules consisting of a core protein with covalently attached heparan sulfate chains. Binding to heparan sulfate is an essential part of the formation of active FGF–FGF receptor complexes and a prerequisite for effective intracellular signaling (4).
The syndecans are a family of single-pass transmembrane proteins with three to five sites for posttranslational glyosylation with predominantly heparan sulfate chains (6, 7). All syndecans have a short cytoplasmic region containing sites for phosphorylation and interaction with signaling and protein-scaffolding molecules (6). Syndecan-4 is the most widely found syndecan and has a number of distinct molecular roles in comparison with other syndecan types. Syndecan-4 is recruited to focal adhesions (7), has both phosphatidylinositol 4,5-bisphosphate and PSD-95/Discs-large/ZO-1 homology domain binding regions and has been implicated in the activation of protein kinase C (8). Syndecan-4 is a powerful regulator of FGF-2 signaling and can modulate growth factor response in multiple cell types (9–11). In addition, syndecan-4 is capable of signaling in response to FGF independently of FGF receptor interactions (12).
Although therapeutic angiogenesis through the direct delivery of growth factors has shown promising results in animal studies, the implementation of this strategy in humans has not been successful (2, 13). Clinically prevalent diseases such as diabetes and hyperlipidemia are known to dramatically modulate the expression of syndecan-4 (14, 15) and may consequently disrupt effective growth factor signaling in the patient population most likely to need angiogenic therapies. In this work, we present a strategy for increasing the effectiveness of FGF-2 therapy by delivering the growth factor in combination with syndecan-4 proteoliposomes. Liposomes are lipid-based nanoparticles consisting of a vesicular lipid bilayer surrounding an inner aqueous phase. The therapeutic advantages of these entities include the ability to deliver a large amount of encapsulated drugs to a specific region in the body, thus resulting in improved drug pharmacokinetics, efficacy, and reduced drug toxicity. Proteoliposomes have specifically obtained application in vaccine development (16).
In our studies, codelivery of syndecan-4 proteoliposomes with FGF-2 increased the cellular uptake, trafficking, and nuclear localization of the growth factor. Furthermore, syndecan-4 proteoliposomes enhanced endothelial signaling through MAPK and p90RSK in response to FGF-2 delivery. These alterations in cellular signaling, trafficking, and nuclear localization led to increased proliferation, migration, and angiogenic differentiation in response to FGF-2 treatment. In an animal model of hind-limb ischemia, syndecan-4 proteoliposomes markedly enhanced the development of arterioles and small vessels following treatment with FGF-2. Together, our results demonstrate an approach for increasing the efficacy of growth factor-based therapeutics by combining them with liposomally associated coreceptors.
Results
Syndecan-4 Proteoliposomes Enhance Endothelial Cell Proliferation, Migration, and Intracellular Signaling in Response to FGF-2.
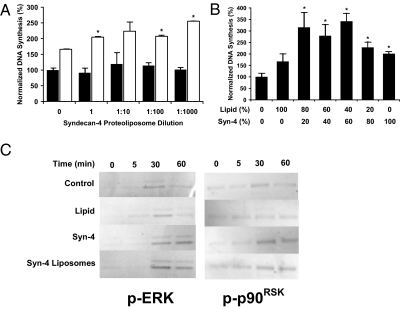
FGF-2 is a potent mediator of proliferation and migration in endothelial cells (4). Effective stimulation of cellular proliferation by FGF-2 requires signaling through the MAPK pathway (12). We treated endothelial cells with FGF-2 in combination with various doses of syndecan-4 proteoliposomes and measured cell proliferation. Syndecan-4 proteoliposomes were found to be effective at increasing endothelial cell proliferation in response to FGF-2 treatment over a broad range of concentrations (Fig. 1A). Syndecan-4 proteoliposomes did not enhance proliferation in the absence of FGF-2 and showed low toxicity at relatively high concentrations. To optimize the proteoliposome composition, we varied the lipid-to-protein ratio of the syndecan-4 proteoliposomes and measured the proliferative response of endothelial cells to FGF-2 (Fig. 1B). When optimized for both concentration and composition, syndecan-4 proteoliposomes increased proliferation of endothelial cells to about 3.4 times that of FGF-2 alone. A range of optimal liposomal compositions was found with lipid-to-protein ratios from 80:20 to 40:60 (lipid:protein) (Fig. 1B).
Fig. 1.
Syndecan-4 proteoliposomes enhance FGF-2–induced proliferation, migration, and intracellular signaling. (A) Syndecan-4 proteoliposomes induce enhanced endothelial proliferation in response to FGF-2 treatment. Black bars are samples without FGF-2 and white bars are samples treated with 10 ng/mL FGF-2. (B) Midrange composition (lipid-to-protein ratio) proteoliposomes were optimally active in inducing enhanced endothelial proliferation. (C) Western blotting analysis of cells treated with 10 ng/mL FGF-2 shows increased and prolonged phosphorylation of MAPK with syndecan-4 or syndecan-4 proteoliposome treatment. Phosphorylation of p90RSK was increased in endothelial cells treated with FGF-2 in combination with syndecan-4 or syndecan-4 proteoliposomes *Statistically different from FGF-2 alone treatment group (P < 0.05). All Western blots were performed in duplicate from separate experiments with similar results.
FGF-2 signaling can lead to the activation of members of the MAPK family (p42/44MAPK) through MEK1 and is often associated with the stimulation of cell proliferation (17). The activated p42/44MAPK, in turn, can phosphorylate an array of cellular substrates, including downstream effector kinases such as p90RSK (18). We examined activation of this pathway by FGF-2 and found increased MAPK and p90RSK phosphorylation in the presence of syndecan-4 proteoliposomes (Fig. 1C and Fig. S1). The mitogenic properties of FGF-2 were then examined by wounding monolayers of confluent endothelial cells in the presence of FGF-2 alone or in combination with syndecan-4 proteoliposomes of varying composition. This analysis demonstrated maximal enhancement of migration in endothelial cells with liposomes having higher concentrations of protein and with syndecan-4 protein alone (Fig. 2 A and B). Syndecan-4 proteoliposomes and syndecan-4 stimulated wound edge migration to a rate about twice as fast as that of FGF-2 alone.
Fig. 2.
In vitro migration and wound closure is increased by FGF-2 treatment in combination with syndecan-4 proteoliposomes. Confluent monolayers of endothelial cells were wounded by scraping with a plastic cell scraper. Wound closure and cell migration was examined at various times with phase-contrast microscopy. (A) Quantitative analysis of wound edge closure following wounding and in the presence of various treatments. (B) Wound-healing assay shows increased wound edge migration with syndecan-4 proteoliposome treatment. (Scale bar, 100 μm.) *Statistically significant difference between samples (P < 0.05).
FGF-2 Uptake and Nuclear Localization in Endothelial Cells Is Increased by Codelivery of Syndecan-4 Proteoliposomes.
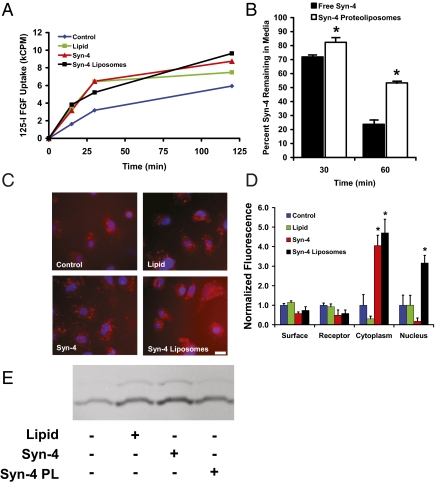
Cellular binding and uptake of FGF-2 is necessary for effective signaling and downstream alterations in cell behavior to take place (19). We performed an analysis of the uptake of 125I-labeled FGF-2 in endothelial cells and found that the presence of either liposomes or syndecan-4 protein alone enhanced FGF-2 uptake (Fig. 3A). Syndecan-4 proteoliposomes induced the greatest increase in FGF-2 uptake, leading to a 2.3-fold enhancement in uptake after 15 min and a 1.6-fold increase after 120 min. To examine the effect of liposomal encapsulation on the stability and cellular uptake of syndecan-4, we metabolically radiolabeled syndecan-4 and applied it to endothelial cells in the presence of FGF-2. Liposomal embedding was found to prolong the presence of syndecan-4 within the soluble medium in comparison with free syndecan-4 (Fig. 3B). Together, these results demonstrate that associating syndecan-4 with a liposomal carrier enhanced the uptake of FGF-2 and stabilized the presence of syndecan-4 in the soluble milieu.
Fig. 3.
Syndecan-4 proteoliposomes enhance FGF-2 uptake and intracellular trafficking. (A) Liposomally embedded syndecan-4 increases uptake of 125I-labeled FGF-2 in endothelial cells. FGF-2 alone (blue line), FGF-2 with liposomes (green line), FGF-2 with syndecan-4 (red line), and FGF-2 with syndecan-4 proteoliposomes (black line). (B) Liposome embedding prolongs 3H-labeled syndecan-4 presence in the media after 30 or 60 min. Syndecan-4 alone is shown with black bars, and syndecan-4 proteoliposomes are shown with white bars. (C) Uptake of fluorescently labeled FGF-2 is increased with syndecan-4 proteoliposomes. FGF-2 was labeled with Texas Red dye and added to cultures of endothelial cells for 4 h. (Scale bar, 20 μm.) (D) Nuclear localization of FGF-2 is increased in the presence of syndecan-4 proteoliposomes. Endothelial cells were exposed to fluorescently labeled FGF-2 for 4 h. The cells were then fractionated and the fluorescence in each fraction was measured and corrected for autofluorescence. (E) Lipids, syndecan-4, and syndecan-4 proteoliposomes enhance rac-1 activity. Cells were treated with various compounds and subjected to a pull-down assay for the active GTP-bound form of Rac-1.
At the initiation of FGF-2 signaling, the growth factor first binds to FGF receptors and induces dimerization and autophosphorylation. The FGF–FGFR complex is then internalized through both clatherin-dependent and -independent mechanisms. Once internalized, FGF-2 is capable of translocating through the endosomal membrane, entering the cytosol, and being translocated to the nucleus. Nuclear localization of FGF-2 is associated with enhanced cell proliferation and ribosomal DNA transcription (20). To examine the intracellular trafficking of FGF-2, we fluorescently labeled FGF-2 and applied it to endothelial cells in culture. Fluorescent microscopy revealed an increase in internalized FGF-2 with syndecan-4 proteoliposome treatment (Fig. 3C). Cellular fractionation demonstrated increased fluorescence in the cytoplasmic fraction with treatment with syndecan-4 protein or syndecan-4 proteoliposomes (Fig. 3D). Nuclear localization of FGF-2 was also markedly increased by the addition of syndecan-4 proteoliposomes (Fig. 3D). Tkachenko et al. demonstrated that syndecan-4 endocytosis requires activation of the Rho-GTPase, Rac-1 (10). We hypothesized that liposome embedding enhanced activation of Rac1 and thus enhanced the macropinocytotic pathway uptake of syndecan-4 and FGF-2. We used an activity assay to measure Rac-1 activity cells treated with liposomes, proteoliposomes, and syndecan-4 in the absence of FGF-2. This experiment demonstrated that the liposomes, free syndecan-4, and syndecan-4 proteoliposomes increased Rac-1 activity and could thus facilitate the endocytic uptake of FGF-2 (Fig. 3E). Notably, the liposomes themselves also activated Rac-1, although to a lesser extent than free syndecan-4 or syndecan-4 proteoliposomes.
Syndecan-4 Proteoliposomes Increase Angiogenic Differentiation in an in Vitro Tube Formation Assay.
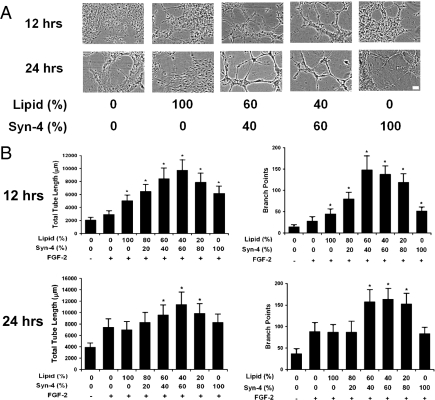
Endothelial cell differentiation and network formation are essential to obtaining effective neovascularization. We examined the effects of syndecan-4 proteoliposomes on FGF-2–mediated angiogenic differentiation in an in vitro tube formation assay. Endothelial cells were seeded on extracellular matrix-coated plates and exposed to FGF-2 with varying liposome/syndecan-4 formulations (Fig. 4A). Midrange composition syndecan-4 proteoliposomes were the most effective in enhancing FGF-2 activity in stimulating angiogenic differentiation, leading to a maximum of a 9.9-fold increase in tube length and a 4.7-fold increase in branch points after 12 h of FGF-2 exposure (Fig. 4B). This enhancement was maintained after 24 h, with a 2.9-fold increase in tube length and a 4.4-fold increase in branch points (Fig. 4B).
Fig. 4.
Syndecan-4 proteoliposomes enhance in vitro tube formation in combination with FGF-2. HUVECs were seeded on extracellular matrix and exposed to various treatments. In vitro tube formation was quantified by measuring total tube length and the number of branch points. All samples were treated with FGF-2 (10 ng/mL) unless labeled otherwise. (A) Phase-contrast micrographs of endothelial cells in Matrigel after 12 h of treatment. (Scale bar, 20 μm.) (B) Quantitative analysis of tube length formed by endothelial network after 12 h of treatment. Number of branch points in endothelial networks after 12 h. Length of tubes in the endothelial network following 24 h of incubation. Branch points in endothelial networks after 24 h of treatment. *Statistically different from FGF-2 group (P < 0.05).
Syndecan-4 Proteoliposomes Enhance FGF-2–Induced Angiogenesis and Arteriogenesis in Hind-Limb Ischemia.
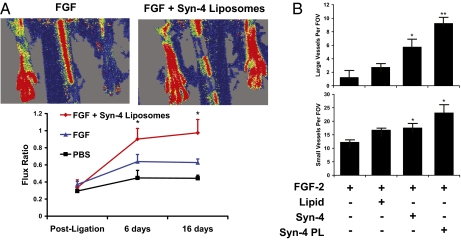
We created hind limb ischemia in rats by surgically ligating the femoral artery and then implanted them with osmotic pumps containing PBS, FGF-2, or FGF-2 in combination with the optimized syndecan-4 proteoliposomes. Using laser Doppler imaging, we tracked the recovery of perfusion in the hind limb over time. We found that proteoliposomes delivered with FGF-2 caused almost complete resolution of ischemia in rats in about 7 d whereas FGF-2 alone did not resolve the ischemia by 16 d of treatment (Fig. 5A). This result was confirmed by histological analysis (Fig. 5B and Fig. S2). Syndecan-4 proteoliposomes and syndecan-4 treatment groups showed qualitatively fewer ischemic changes to the hind-limb muscle (Fig. S2). Consistent with these findings, staining for the endothelial marker PECAM and morphometric analysis showed a greater than sevenfold increase in arterioles (Fig. 5B) and an approximately twofold increase in capillary density (Fig. 5B) in the syndecan-4 proteoliposome treatment group versus FGF-2 treatment alone. Together, these results support that syndecan-4 proteoliposomes can enhance the efficacy of FGF-2 therapy for ischemia in animal models.
Fig. 5.
Enhanced neovascularization of ischemic muscle after treatment with syndecan-4 proteoliposomes in combination with FGF-2. Hind-limb ischemia was created in rats through femoral artery ligation, and treatments were delivered over 7 d with an osmotic pump. (A) Routine histological staining revealed reduced ischemic changes following treatment with FGF-2 in combination with syndecan-4 alone or with liposomally embedded syndecan-4. (Scale bar, 100 μm.) (B) Quantification of large vessel number per field of view. Quantification of capillary number per field of view. *Statistically different from FGF group (P < 0.05). **Statistically different from all other groups (P < 0.05).
Discussion
Many studies have evaluated potential therapies in the pursuit of efficacious revascularization therapy (1). Treatment modalities have included delivery of growth factor proteins (2, 13), viral delivery of growth factor/transcription factor genes (21), induction/mobilization of endogenous endothelial progenitor cells, and implantation of bone marrow or progenitor cells (22). Each of these strategies has shown some promise in early phase experimental work and animal models, but to date none have shown efficacy in large, randomized clinical trials. The FIRST trial, a phase II, randomized, and double-blinded trial using delivery of FGF-2, found no improvement in myocardial perfusion or exercise treadmill testing (ETT), despite promising early studies (2). Similarly, a phase II/III trial for the delivery of VEGF (the VIVA trial) found no improvement in comparison with placebo (13). Clinical trials of adenovirally delivered DNA have also shown no improvement in ETT but a moderate increase in myocardial perfusion (21).
Although prior work has focused on delivering recombinant proteins, genes, or cells that can facilitate angiogenesis, we examined an approach to facilitate the effectiveness of growth factors by increasing the availability of coreceptors. Direct delivery of growth factors has potential advantages over other types of therapy in that protein therapeutics do not require modification of the host genome or delivery of exogenous cellular material (1). However, this method has not been found to have sufficient efficacy as a clinical therapy. It would therefore be highly advantageous to improve the effectiveness of this type of therapy without altering its inherent safety. Here we have demonstrated that syndecan-4 proteoliposomes can facilitate cellular uptake of FGF-2 and increase endothelial cell proliferation, migration, and angiogenic differentiation in vitro. Furthermore, syndecan-4 proteoliposomes markedly enhanced FGF-2 activity in the neovascularization of ischemic limbs. Together, these results support that this approach is an effective means for increasing the efficacy of growth factor therapy.
Heparin has been used to enhance the activity of FGF-2 in cells and in the treatment of ischemia (4, 23). Heparin is known to enhance FGF-2 signaling and to protect FGF-2 from proteolytic degradation (23). A major mechanistic theory of heparin's activity in FGF signaling is its ability to stabilize the ternary complex of dimerized FGF–FGFR pairs (4). In addition, recent studies suggest that heparin may increase FGF signaling through mechanisms in addition to receptor complex stabilization (24). Cell-surface heparan sulfates are also effective in facilitating FGF-2 signaling and can stabilize the ternary FGF-signaling complex (4). A major question that arises in this work is whether our delivery system contributes more than simply the benefit of added heparan sulfate chains. In our studies, we used syndecan-4 alone to control for these effects and examine whether there was a benefit from using liposomally embedded heparan sulfate-bearing proteins in comparison with free proteins. In both cell and animal studies, the proteoliposomal formulation was superior to free syndecan-4 in inducing cellular proliferation, angiogenic differentiation, or vessel formation in response to FGF-2 treatment. Our mechanistic studies revealed moderate increases in intracellular signaling and striking increases in cellular uptake and nuclear localization of FGF-2 in the presence of syndecan-4 proteoliposomes. Free syndecan-4 protein increased intracellular signaling and cellular uptake but did not increase nuclear localization of FGF-2. Thus, the enhancement of nuclear localization would be a potential mechanism leading to the superiority of liposomally associated syndecan-4 over free protein. These findings support the importance of the downstream processes for FGF signaling (after receptor binding and intracellular signaling) in determining ultimate biological response.
One of the fascinating properties of FGF-2 is its ability to transverse the endosomal membrane and eventually be transported into the nucleus. In the nucleus, FGF-2 promotes ribosomal DNA transcription, proliferation, and cell survival (25). This process enhances the ultimate effector mechanisms of FGF-2, including cell proliferation and metastasis in cancer cells (25, 26). Liposomes are known to facilitate the penetration of drugs through cellular membranes and tissue (27). Liposomal embedding of syndecan-4 could facilitate the passage of FGF-2 through the cytoplasmic, endosomal, and nuclear membranes of the cell. This enhancement would allow a faster and more robust entry of FGF-2 into the nucleus, as was observed in our trafficking studies. Syndecan-4 alone facilitated cytoplasmic entry of FGF-2 but did not increase FGF-2 in the nuclear fraction. It is likely that exogenously delivered syndecan-4 facilitated FGF-2 uptake by increasing FGFR binding and signaling complex internalization. However, once internalized, free syndecan-4 did not enhance endosomal membrane translocation and nuclear localization. Thus, the liposomal component of this delivery system provides an advantage over providing heparan sulfate chains alone.
The efficacy of syndecan-4 proteoliposomes was relatively insensitive to the concentration of the delivered compound. One might hypothesize that soluble syndecan-4 could serve to competitively inhibit receptor binding, a property that is exhibited by heparin at high concentrations. However, we did not observe this effect even at high concentrations of syndecan-4 proteoliposomes. Activity over a broad range of concentrations is highly advantageous for a drug delivery system, providing a robust biological response despite varying drug concentrations. In our studies, FGF-2–stimulated migration was enhanced by the presence of syndecan-4 proteoliposomes as well. In these experiments, the addition of syndecan-4, independently from incorporation into proteoliposomes, enhanced endothelial migration in the presence of FGF-2. These results are in contrast to our findings for the proliferation and in vitro tube formation studies in which the optimal reagent had a nearly equal ratio between lipid and protein. In addition to being a coreceptor for FGF-2, syndecan-4 also has a role in cell attachment. Syndecan-4 is an essential component for the activation of focal adhesion kinase and is capable of binding fibronectin with its heparan sulfate chains (28). Consequently, the results in the migration study may be a result of the combined effects of delivering both an enhancer of FGF-2 signaling and an exogenously delivered adhesion receptor.
Taken together, the results presented here support that the delivery of liposomally embedded surface receptors can markedly enhance the therapeutic potential of exogenously delivered growth factors. The conceptual paradigm of delivering a receptor or coreceptor to increase cellular response may be relevant to a wide variety of therapeutic applications that are amenable to growth factor and cytokine therapy. We used the syndecan-4/FGF-2 system to demonstrate this archetype of therapy and demonstrated enhancement of in vitro proliferation, migration, and differentiation in endothelial cells. This technique was also effective in enhancing the neovascularization of the ischemic hind limb in response to FGF-2. It is clear that the delivery of recombinant growth factors alone is not sufficient to achieve clinical efficacy and the strategy presented here may facilitate the development of more effective forms of therapeutic neovascularization.
Materials and Methods
Cell Culture and Measurement of DNA Synthesis.
Human umbilical vein endothelial cells (HUVECs) were cultured under standard conditions and used for measurements of endothelial response to various treatments. DNA synthesis was measured using 3H-thymidine incorporation. Detailed information for these methods can be found in SI Materials and Methods.
Production of Recombinant Syndecan Protein.
A constitutive expression vector containing the full-length syndecan-4 gene (Origene; National Center for Biotechnology Information Reference Sequence NM_002999.3) was transfected into HeLa cells using the FuGENE HD transfection reagent (Roche) per the manufacturer's specifications. Two days posttransfection, cell lysis was performed with a buffer containing the following: 20 mM Tris (pH 8.0), 150 mM NaCl, 1% Triton X-100, 0.1% SDS, 2 mM sodium orthovanadate, 2 mM PMSF, 50 mM NaF, and protease inhibitors (Roche). The lysates were clarified by centrifugation for 15 min at 15,000 × g, and the supernatant was collected. The pooled lysates were desalted and separated sequentially using ion exchange and affinity chromatography. The separations were done with an AKTA FPLC (GE Healthcare) using a Q anion exchange column or an affinity column with immobilized antibodies to syndecan-4 (Santa Cruz Biotechnology). The samples were then concentrated/separated using ion exchange chromatography, desalted using dialysis, and concentrated using membrane filtration. The final samples were analyzed for purity by SDS/PAGE and silver staining (shown in Fig. S3).
Preparation of Proteoliposomes.
Stock solutions of 10 mg/mL each of 1,2-dioleoyl-sn-glycero-3-phosphocholine, 1,2-dioleoyl-sn-glycero-3-phosphoethanolamine, cholesterol, and sphingomyelin (Avanti Polar Lipids) were dissolved in chloroform and mixed in a ratio of 40:20:20:20 by volume, respectively. The solution was placed in a round-bottom flask, and the solvent was removed under a stream of argon gas. The lipids were resuspended by mixing, sonicating, and freeze-thawing in a Hepes-buffered salt solution (10.0 mM Hepes and 150 mM NaCl in PBS, pH 7.4) to form a final solution of 13.2 mM total lipid. The lipid solution was then extruded through a 400-nm polycarbonate membrane (Avestin). A detergent, 1% n-octyl-β-d-glucopyranoside (OG), was added to both the 13.2-mM lipid and the 71-μg/mL syndecan-4 protein solutions, and these were combined in various ratios to form different formulations. Each of the proteoliposome solutions was incubated for 1 h at room temperature with mixing. The concentration of the solution was reduced to 40% of the original in 10% increments every 30 min through dilution with PBS. The detergent and free protein was removed by extensive dialysis in PBS at 4 °C. Residual OG was removed by repeated BioBead treatments (SM-2; Bio-Rad).
Measurement of FGF-2 Uptake and Intracellular Trafficking.
Uptake of FGF-2 was measured using radiolabeled 125I. Metabolically labeled syndecan-4 was created by adding 100 μCi/mL of a 3H-labeled amino acid mix (MP Biomedical) to HeLa cells and purifying the protein as described above. Fluorescently labeled FGF-2 was made by binding the FGF-2 to a heparin column (GE Healthcare) and by reacting with Texas Red-X succinimidyl ester (Invitrogen) for 1 h at room temperature. The cells were fractionated by ultracentrifugation, and fluorescence in the fractions was measured using a fluorescent plate reader (Thermo Fisher Scientific).
Cell Lysis and Western Blotting.
Following treatments, endothelial cells were lysed in a buffer containing the following: 20 mM Tris at pH 8, 150 mM NaCl, 1% triton, 0.1% SDS, 2 mM sodium orthovanadate, 2 mM PMSF, 50 mM NaF, and protease inhibitors (Roche). The samples were separated by gel electrophoresis and transferred to a nitrocellulose membrane using the iBlot system (Invitrogen). The membranes were then blocked for 1 h in 5% nonfat milk in PBS with 0.01% Tween-20 and exposed to anti–phospho-ERK (Thr202/Tyr204) and anti–phospho-p90RSK (Cell Signaling) primary antibodies overnight at 4 °C. The membranes were washed and incubated at room temperature for 2 h with secondary antibody and were detected using a chemiluminescence substrate (Thermo Fisher Scientific).
Wound Edge Migration Assay.
Plates of confluent HUVEC cells were wounded with the edge of a cell scraper, and the initial boundaries of the wound were marked on the underside of the plates using a hypodermic needle. The dishes were washed three times with serum-free media, and solutions of FGF-2 (10 ng/mL) with various liposome formulations were applied. The wounds were photographed using an inverted, phase-contrast microscope with digital camera, and the migration distance was quantified using Photoshop CS3 (Adobe).
In Vitro Angiogenesis Assay.
In vitro angiogenic differentiation was measured using a tube formation assay (Millipore). Briefly, six-well culture plates were coated with extracellular matrix allowed to gel overnight at 37 °C. To each well, 2 × 104 endothelial cells were added in the presence of the appropriate treatment (i.e., liposome/FGF formulation). At various time points, the cells were imaged using phase-contrast microscopy. Quantification of tube length and branch points was performed using MetaMorph software (Molecular Devices).
Rat Hind-Limb Ischemia Model.
Sprague–Dawley rats were anesthetized using isofluorane gas, and a 1-cm longitudinal incision was made over the inguinal region of the right hind limb. The femoral artery was separated from the femoral nerve and vein and ligated twice using surgical silk. Using blunt dissection, a pocket was created in the s.c. space, and a small osmotic pump (DURECT) was implanted containing 5 μg of FGF-2 in combination with various treatments. The pump was designed to deliver the entire volume of 100 μL over a period of 28 d. The incision was then sealed using surgical clips. The animals were imaged at various time points using laser Doppler imaging (Moor Instruments) after the ligation surgery, as well as at 6 and 16 d following the surgery. After 7–16 d, the animals were killed, and the hind-limb muscles were harvested and snap-frozen in liquid nitrogen. All animal experiments were performed in accordance with the Guide for the Care and Use of Laboratory Animals published by the National Institutes of Health (publication no. 85–23).
Histological and Immunohistochemical Analysis.
Harvested muscle samples were sectioned using a cryotome equipped with a steel knife. Before hematoxylin and eosin staining, the sections were dried and fixed in formalin for 10 min. Histological staining was then performed using standard methods. For immunohistochemical staining for PECAM, the sections were air dried and fixed in acetone at −20 °C for 2 min. The samples were then blocked with 20% FBS for 45 min and exposed to a 1:25 dilution of goat anti–PECAM-1 (Santa Cruz Biotechnology) for 2 h. The samples were washed three times with PBS and treated with a 1:100 dilution of secondary antibody conjugated to a fluorescent marker (Alexa Fluor 594; Invitrogen) for 1 h. The sections were then rinsed with PBS and coverslipped with DAPI containing anti-fade mounting medium (Vector Labs).
Statistical Analysis.
All results are shown as mean ± SEM. An ANOVA with Bonferroni's post hoc test was used to compare groups of continuous variables. P < 0.05 was defined as being statistically significant.
Supplementary Material
Acknowledgments
This work was supported by Philip Morris International through a postdoctoral research fellowship and Scientist Development Grant 10SDG2630139 from the American Heart Association (to A.B.B.).
Footnotes
Conflict of interest statement: The authors declare a conflict of interest. A.B.B. and E.R.E. have filed a patent on the technology discussed in this paper.
*This Direct Submission article had a prearranged editor.
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1117885109/-/DCSupplemental.
References
- 1.Tirziu D, Simons M. Angiogenesis in the human heart: Gene and cell therapy. Angiogenesis. 2005;8:241–251. doi: 10.1007/s10456-005-9011-z. [DOI] [PubMed] [Google Scholar]
- 2.Simons M, et al. Pharmacological treatment of coronary artery disease with recombinant fibroblast growth factor-2: Double-blind, randomized, controlled clinical trial. Circulation. 2002;105:788–793. doi: 10.1161/hc0802.104407. [DOI] [PubMed] [Google Scholar]
- 3.Bikfalvi A, Klein S, Pintucci G, Rifkin DB. Biological roles of fibroblast growth factor-2. Endocr Rev. 1997;18:26–45. doi: 10.1210/edrv.18.1.0292. [DOI] [PubMed] [Google Scholar]
- 4.Nugent MA, Iozzo RV. Fibroblast growth factor-2. Int J Biochem Cell Biol. 2000;32:115–120. doi: 10.1016/s1357-2725(99)00123-5. [DOI] [PubMed] [Google Scholar]
- 5.Rapraeger AC, Krufka A, Olwin BB. Requirement of heparan sulfate for bFGF-mediated fibroblast growth and myoblast differentiation. Science. 1991;252:1705–1708. doi: 10.1126/science.1646484. [DOI] [PubMed] [Google Scholar]
- 6.Tkachenko E, Rhodes JM, Simons M. Syndecans: New kids on the signaling block. Circ Res. 2005;96:488–500. doi: 10.1161/01.RES.0000159708.71142.c8. [DOI] [PubMed] [Google Scholar]
- 7.Alexopoulou AN, Multhaupt HA, Couchman JR. Syndecans in wound healing, inflammation and vascular biology. Int J Biochem Cell Biol. 2007;39:505–528. doi: 10.1016/j.biocel.2006.10.014. [DOI] [PubMed] [Google Scholar]
- 8.Horowitz A, Murakami M, Gao Y, Simons M. Phosphatidylinositol-4,5-bisphosphate mediates the interaction of syndecan-4 with protein kinase C. Biochemistry. 1999;38:15871–15877. doi: 10.1021/bi991363i. [DOI] [PubMed] [Google Scholar]
- 9.Horowitz A, Tkachenko E, Simons M. Fibroblast growth factor-specific modulation of cellular response by syndecan-4. J Cell Biol. 2002;157:715–725. doi: 10.1083/jcb.200112145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tkachenko E, Lutgens E, Stan RV, Simons M. Fibroblast growth factor 2 endocytosis in endothelial cells proceed via syndecan-4-dependent activation of Rac1 and a Cdc42-dependent macropinocytic pathway. Journal of Cell Science. 2004;117:3189–3199. doi: 10.1242/jcs.01190. [DOI] [PubMed] [Google Scholar]
- 11.Zhang Y, Li J, Partovian C, Sellke FW, Simons M. Syndecan-4 modulates basic fibroblast growth factor 2 signaling in vivo. Am J Physiol Heart Circ Physiol. 2003;284:H2078–H2082. doi: 10.1152/ajpheart.00942.2001. [DOI] [PubMed] [Google Scholar]
- 12.Chua CC, Rahimi N, Forsten-Williams K, Nugent MA. Heparan sulfate proteoglycans function as receptors for fibroblast growth factor-2 activation of extracellular signal-regulated kinases 1 and 2. Circ Res. 2004;94:316–323. doi: 10.1161/01.RES.0000112965.70691.AC. [DOI] [PubMed] [Google Scholar]
- 13.Henry TD, et al. VIVA Investigators The VIVA trial: Vascular endothelial growth factor in Ischemia for Vascular Angiogenesis. Circulation. 107:1359–1365. doi: 10.1161/01.cir.0000061911.47710.8a. [DOI] [PubMed] [Google Scholar]
- 14.Bortoff KD, Wagner WD. Reduced syndecan-4 expression in arterial smooth muscle cells with enhanced proliferation. Exp Mol Pathol. 2005;78:10–16. doi: 10.1016/j.yexmp.2004.08.010. [DOI] [PubMed] [Google Scholar]
- 15.Olsson U, Bondjers G, Camejo G. Fatty acids modulate the composition of extracellular matrix in cultured human arterial smooth muscle cells by altering the expression of genes for proteoglycan core proteins. Diabetes. 1999;48:616–622. doi: 10.2337/diabetes.48.3.616. [DOI] [PubMed] [Google Scholar]
- 16.Neelapu SS, et al. A novel proteoliposomal vaccine induces antitumor immunity against follicular lymphoma. Blood. 2007;109:5160–5163. doi: 10.1182/blood-2006-12-063594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Garrington TP, Johnson GL. Organization and regulation of mitogen-activated protein kinase signaling pathways. Curr Opin Cell Biol. 1999;11:211–218. doi: 10.1016/s0955-0674(99)80028-3. [DOI] [PubMed] [Google Scholar]
- 18.Thomson S, Mahadevan LC, Clayton AL. MAP kinase-mediated signalling to nucleosomes and immediate-early gene induction. Semin Cell Dev Biol. 1999;10:205–214. doi: 10.1006/scdb.1999.0302. [DOI] [PubMed] [Google Scholar]
- 19.Prudovsky I, et al. Intact and functional fibroblast growth factor (FGF) receptor-1 trafficks near the nucleus in response to FGF-1. J Biol Chem. 1994;269:31720–31724. [PubMed] [Google Scholar]
- 20.Reilly JF, Mizukoshi E, Maher PA. Ligand dependent and independent internalization and nuclear translocation of fibroblast growth factor (FGF) receptor 1. DNA Cell Biol. 2004;23:538–548. doi: 10.1089/dna.2004.23.538. [DOI] [PubMed] [Google Scholar]
- 21.Grines CL, et al. Angiogenic Gene Therapy (AGENT) trial in patients with stable angina pectoris. Circulation. 2002;105:1291–1297. doi: 10.1161/hc1102.105595. [DOI] [PubMed] [Google Scholar]
- 22.Meyer GP, et al. Intracoronary bone marrow cell transfer after myocardial infarction: Eighteen months’ follow-up data from the randomized, controlled BOOST (BOne marrOw transfer to enhance ST-elevation infarct regeneration) trial. Circulation. 2006;113:1287–1294. doi: 10.1161/CIRCULATIONAHA.105.575118. [DOI] [PubMed] [Google Scholar]
- 23.Fujita M. Heparin and angiogenic therapy. Eur Heart J. 2000;21:270–274. doi: 10.1053/euhj.1999.1700. [DOI] [PubMed] [Google Scholar]
- 24.Goodger SJ, et al. Evidence that heparin saccharides promote FGF2 mitogenesis through two distinct mechanisms. J Biol Chem. 2008;283:13001–13008. doi: 10.1074/jbc.M704531200. [DOI] [PubMed] [Google Scholar]
- 25.Bailly K, Soulet F, Leroy D, Amalric F, Bouche G. Uncoupling of cell proliferation and differentiation activities of basic fibroblast growth factor. FASEB J. 2000;14:333–344. [PubMed] [Google Scholar]
- 26.Thomas-Mudge RJ, et al. Nuclear FGF-2 facilitates cell survival in vitro and during establishment of metastases. Oncogene. 2004;23:4771–4779. doi: 10.1038/sj.onc.1207638. [DOI] [PubMed] [Google Scholar]
- 27.Kreilgaard M. Influence of microemulsions on cutaneous drug delivery. Adv Drug Deliv Rev. 2002;54(Suppl 1):S77–S98. doi: 10.1016/s0169-409x(02)00116-3. [DOI] [PubMed] [Google Scholar]
- 28.Wilcox-Adelman SA, et al. Syndecan-4: dispensable or indispensable? Glycoconj J. 2002;19:305–313. doi: 10.1023/A:1025304602057. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.