Abstract
The interaction between the stress axis and endogenous opioid systems has gained substantial attention as it is increasingly recognized that stress alters individual sensitivity to opiates. One site at which opiates and stress substrates may interact to have global effects on behavior is within the locus coeruleus (LC). We have previously described interactions of several opioid peptides (e.g. pro-opiomelanocortin, enkephalin) with the stress-related peptide, corticotropin-releasing factor (CRF) in the LC. To further examine interactions between DYN, ENK and CRF in the LC, sections were processed for detection of DYN, CRF or DYN and ENK in rat brain. DYN- and CRF-containing axon terminals overlapped noradrenergic dendrites in this region. Dual immunoelectron microscopy showed coexistence of DYN and CRF with 35% of axon terminals containing DYN that were also immunoreactive for CRF. In contrast, few axon terminals contained both DYN and ENK. A potential DYN/CRF afferent is the central nucleus of the amygdala (CeA). Dual in situ hybridization showed that in CeA neurons, 31% of DYN mRNA-positive cells co-localized with CRF mRNA while 53% of CRF mRNAcontaining cells co-localized with DYN mRNA. Finally, to determine whether limbic DYN afferents target the LC, the CeA was electrolytically lesioned. Light-level densitometry of DYN labeling in the LC showed a significant decrease in immunoreactivity on the side of the lesion. Taken together, these data indicate that DYN-and CRF-labeled axon terminals, most likely arising from amygdalar sources are positioned to dually impact LC function whereas DYN and ENK function in parallel.
Keywords: dynorphin, corticotropin-releasing factor, tyrosine hydroxylase, amygdala, electron microscopy, in situ hybridization
INTRODUCTION
Containing the largest cluster of noradrenergic neurons in the brain, the locus coeruleus (LC) has been implicated in a wide variety of autonomic functions (Lechner et al., 1997; Miyawaki et al., 1991; Morilak et al., 1987; Murase et al., 1994). LC neurons are involved in stress related disorders such as anxiety, depression and post-traumatic stress disorder (Bremner et al., 1996; Lechner et al., 1997; Page and Valentino, 1994; Svensson, 1987; Valentino et al., 1993). The LC innervates the entire neuraxis through a divergent efferent system. Single LC neurons collateralize to distant and functionally diverse brain regions. It is the sole source of norepinephrine in many forebrain regions that have been implicated in cognition (e.g., cortex and hippocampus). LC neurons are spontaneously active and discharge in a synchronous manner that is linked to oscillations in membrane potential (Aston-Jones and Bloom, 1981a; Aston-Jones and Bloom, 1981b; Ishimatsu and Williams, 1996). In addition to their synchronous spontaneous activity, LC neurons are homogenous in their polymodal response to stimuli (Aston-Jones and Bloom, 1981b; Foote et al., 1983). Coupled with a highly divergent, collateralized efferent system, this synchronous activity provides a mechanism for global norepinephrine release throughout the neuraxis in response to stimuli.
During stress, corticotropin-releasing factor (CRF) serves as a neurotransmitter that regulates diverse extrahypophyseal systems (Vale et al., 1981). Neurochemical and electrophysiological studies support direct actions of CRF on LC neuronal activity (Curtis et al., 1997; Valentino et al., 1983) and norepinephrine release in LC targets (Page and Abercrombie, 1999) following stress. In contrast to excitatory actions of CRF, potent inhibitory effects of μ-OR activation on LC neurons in vivo and in vitro have been well documented (Alreja and Aghajanian, 1993; Williams et al., 1982). Our laboratory previously demonstrated that enkephalin (ENK)-containing axon terminals densely innervate the LC (Van Bockstaele et al., 1995) and that CRF and ENK axon terminals converge onto common LC dendrites (Tjoumakaris et al., 2003). We also provided evidence for opioid-mediated post-stress inhibition of the LC system that may serve as a counterregulatory mechanism to balance or limit this cognitive limb of the stress response, much in the same vein that glucocorticoids counter-regulate the hypothalamo-pituitary limb (Xu et al., 2003).
The dynorphin (DYN) system has been recently highlighted as a link between stress and drug abuse. For example, stress, which promotes relapse and can facilitate place preference for drugs of abuse, increases prodynorphin gene expression in the limbic system (Shirayama et al., 2004). Genetic deletion of prodynorphin or pharmacological antagonism of kappa receptors prevented stress-induced preference, implicating the DYN system in stress-induced facilitation of drug abuse (Shirayama et al., 2004). Additionally, kappa opioid receptor (κ–OR) antagonists prevent stress-elicited behaviors that are endpoints of depression such as immobility in the forced swim test and passive behavior in learned helplessness (Mague et al., 2003; McLaughlin et al., 2003; Shirayama et al., 2004). Our recent evidence for DYN-immunoreactive terminals targeting tyrosine hydroxylase (TH)-immunoreactive perikarya and processes in the LC (Reyes et al., 2007) showed that the majority of synaptic specializations observed were asymmetric synapses suggesting co-existing excitatory transmitters such as glutamate or CRF. These data have compelled us to consider the impact of this additional opioid peptide family in our model of co-regulation of the LC system by endogenous opioids and the stress peptide, CRF. In addition to considering opposing postsynaptic effects on LC neurons, the working model includes potential κ-OR-mediated presynaptic regulation, which could directly impact on phasic activity of LC neurons by inhibiting afferent signaling.
LC neurons are enriched with DYN-immunoreactive fibers and processes (Elde and Hokfelt, 1993; Fallon and Leslie, 1986; Khachaturian et al., 1982) and express mRNA for DYN (Mansour et al., 1994; Zamir et al., 1983). DYN is implicated in stress regulation (McLaughlin et al., 2003; Shirayama et al., 2004), analgesia (Basbaum and Fields, 1984; Piercey et al., 1982) and a myriad of neuroendocrine functions (Akil et al., 1984). In an attempt to further examine the cellular substrates underlying interactions of DYN with other stress related peptides in the LC, immunofluorescence and immunoelectron microscopic analysis were carried out. We investigated the co-localization of DYN with CRF or ENK and observed a prominent co-localization of DYN and CRF in the LC. A potential candidate for sources of DYN and CRF afferents is the central nucleus of the amygdala (CeA), since CeA is enriched with DYN and CRF cell bodies (Fallon and Leslie, 1986; Khachaturian et al., 1982; Watson et al., 1981; Watson et al., 1982; Watson et al., 1983). We therefore performed dual fluorescence in situ hybridization to quantify the degree of CRF and DYN co-localization in neurons within the CeA. In addition, retrograde and anterograde tracing studies have shown that CeA provides CRF afferents to the noradrenergic processes in the LC (Van Bockstaele et al., 1998; Tjoumakaris et al., 2003). Thus, in the present study the CeA was electrolytically lesioned to determine whether DYN arising from the CeA targets the LC.
MATERIALS AND METHODS
The procedures used in the present study were approved by the Institutional Animal Care and Use Committee of Thomas Jefferson University and conformed to NIH guidelines. All efforts were made to utilize only the minimum number of animals necessary to produce reliable scientific data and attempts were made to minimize animal suffering.
Tissue preparation
Fourteen adult male Sprague-Dawley rats (225–249 g; Harlan Sprague-Dawley, Inc., Indianapolis, IN) were used in this study. Six rats were used for examining the cellular associations of DYN with CRF-containing cellular profiles in the dorsal pontine tegmentum. Two (n=2) rats received injections of colchicine (50 μg; Sigma Immunochemical, St. Louis, MO) into each lateral ventricle, 24 h prior to transcardial perfusion and were used to study DYN/CRF study in the CeA. Four rats received electroylytic lesions of the CeA where DYN and CRF immunoreactivities were assessed in the LC. Lastly, two rats were used to examine the localization of DYN and CRF mRNA in the CeA. The rats were housed 2–3 per cage on a 12-h light schedule in a temperature controlled (20 °C) colony room. They were given free access to standard rat chow and water.
Immunofluorescence
Rats were deeply anesthetized with sodium pentobarbital (60 mg/kg) and transcardially perfused through the ascending aorta with 500 ml of 4% formaldehyde in 0.1 M phosphate buffer (PB; pH 7.4). Brains were then removed and post fixed in 4% formaldehyde overnight at 4°C. Forty micrometer thick sections through the LC were cut in the coronal plane with a Vibratome (Technical Product International, St Louis, MO, USA) and rinsed extensively in 0.1 M PB and 0.1 M tris-buffered saline (TBS; pH 7.6). Sections were then incubated in 0.5% bovine serum albumin (BSA) and 0.25% Triton X-100 in 0.1M TBS for 30 min and rinsed extensively in 0.1 M TBS. Tissue sections were incubated in a cocktail containing guinea-pig anti-preprodynorphin (ppDYN) at 1:16,000, CRF at 1:2,000 and mouse TH at 1:1,000 or guinea-pig anti-ppDYN at 1:16,000 and rabbit anti-methionine5-ENK at 1:4,000 in 0.1% BSA and 0.25% Triton X-100 in 0.1M TBS for 15–18 h at room temperature. ppDYN antiserum was kindly provided by Dr. R. Elde, University of Minnesota, MN (Shuster et al., 2000; Arvidsson et al., 1995, see discussion), CRF antiserum was kindly provided by Dr. W. Vale of the Salk Institute; lot c70) directed against the rat/human form of CRF (Rivier et al., 1983; Vale et al., 1981) Mouse anti-TH (Immunostar Inc., Hudson, WI, USA; catalog number: 22941 and lot number: 148003) and rabbit anti-methionine5-ENK (Incstar Incorporation, Stilwater, MN; catalog number: 20066 and lot number: 550214) were obtained from commercial sources. Incubation time was 15–18 h on a rotary shaker at room temperature. Sections were then washed in 0.1 M TBS and incubated in a secondary antibody cocktail containing flourescein isothiocyanate (FITC) donkey anti-rabbit (1:100; Jackson ImmunoResearch Laboratories Inc., West Grove, PA, USA), tetramethyl rhodamine isothiocyanate (TRITC) donkey anti-guinea pig (1:100; Jackson ImmunoResearch) and aminomethylcoumarin acetate (AMCA) donkey anti-mouse (1:200; Jackson ImmunoResearch) antibodies prepared in 0.1 % BSA and 0.25% Triton X-100 in 0.1 M TBS for 2 h in the dark on a rotary shaker.
Following incubation with the secondary antibodies, the tissues were washed thoroughly in 0.1 M TBS. The tissue sections were then mounted on slides and allowed to dry in complete darkness. The slides were dehydrated in a series of alcohols, soaked in xylene and coverslipped using DPX (Sigma-Aldrich Inc., St. Louis, MO, USA). Sections were visualized using a Leica DMRBE microscope (Leica Microsystems, Wetzlar, Germany) equipped for fluorescence with rhodamine and fluorescein filters. Images were captured using Spot Advance software (Diagnostic Instruments Inc., Sterling Heights, MI). Figures were assembled, pseudocolored and adjusted for brightness and contrast in Adobe Photoshop.
Immunoelectron microscopy
Six rats were deeply anesthetized with sodium pentobarbital (60 mg/kg) and perfused transcardially through the ascending aorta with 10 ml heparinized saline followed with 50 ml of 3.75% acrolein (Electron Microscopy Sciences, Fort Washington, PA, USA), and 200 ml of 2% formaldehyde in 0.1 M PB (pH 7.4). The brains were removed immediately after perfusion fixation, sectioned into 1–3 mm coronal slices and postfixed in the same fixative overnight at 4°C.
Alternate 40-μm-thick coronal sections through the rostrocaudal extent of the LC were processed for electron microscopic analysis of DYN and CRF or DYN and ENK. In both tissue sections, immunoperoxidase labeling was used to identify DYN immunoreactivity while immunogold-silver labeling was used to identify CRF or ENK immunoreactivity. The dilutions of primary antibodies used in immunofluorescence protocol were followed except that for DYN and ENK immunolabeling, a cocktail of antibody contained pig anti-ppDYN at 1:16,000 and rabbit anti-methionine5-ENK (Incstar Incorporation) at 1:4,000. Sections were placed for 30 min in 1% sodium borohydride in 0.1 M PB to remove reactive aldehydes and incubated in 0.5% bovine serum albumin (BSA) in 0.1M TBS for 30 min. Following thorough rinses in 0.1 M TBS, sections processed for DYN and CRF were incubated in a cocktail containing guinea pig anti-ppDYN (1:16,000) and CRF antiserum (1:2,000). The following day, tissue sections were rinsed three times in 0.1 M TBS and incubated in biotinylated donkey anti-guinea pig (1:400; Vector Laboratories, Burlingame, CA) for 30 min followed by rinses in 0.1 M TBS. Subsequently, a 30-minute incubation of avidin-biotin complex (Vector Laboratories) followed. For all incubations and washes, sections were continuously agitated with a rotary shaker. DYN was visualized by a 4-minute reaction in 22 mg of 3,3′-diaminobenzidine (Sigma-Aldrich Inc., St. Louis, MO) and 10 μl of 30% hydrogen peroxide in 100 ml of 0.1 M TBS. Some sections were collected, dehydrated and coverslipped for light microscopic analysis of DYN immunoreactivity.
For gold-silver localization of CRF (in sections processed for DYN and CRF) and ENK (in sections processed for DYN and ENK), sections were rinsed three times with 0.1 M TBS, followed by rinses with 0.1 M PB and 0.01 phosphate buffered saline (PBS; pH 7.4). Sections were then incubated in a 0.2% gelatin-PBS and 0.8% BSA buffer for 10 min. This was followed by incubation in goat anti-rabbit IgG conjugate in 1 nm gold particles (Amersham Bioscience Corp., Piscataway, NJ, USA) at room temperature for 2 h. Sections were then rinsed in buffer containing the same concentration of gelatin and BSA as above and subsequently rinsed with 0.01 M PBS. Sections were then incubated in 2% glutaraldehyde (Electron Microscopy Sciences) in 0.01 M PBS for 10 min followed by washes in 0.01 M PBS and 0.2 M sodium citrate buffer (pH 7.4). A silver enhancement kit (Amersham Bioscience Corp.) was used for silver intensification of the gold particles. The optimal times for silver enhancement time were determined by empirical observation for each experiment and ranged 7–8 min. Following intensification, tissue sections were rinsed in 0.2 M citrate buffer and 0.1 M PB, and incubated in 2% osmium tetroxide (Electron Microscopy Sciences) in 0.1 M PB for 1 h, washed in 0.1 M PB, dehydrated in an ascending series of ethanol followed by propylene oxide and flat embedded in Epon 812 (Electron Microscopy Sciences; Leranth and Pickel, 1989). Thin sections of approximately 50–100 nm in thickness were cut with a diamond knife (Diatome-US, Fort Washington, PA, USA) using a Leica Ultracut (Leica Microsystems, Wetzlar, Germany). Sections were collected on copper mesh grids and counterstained stained with 5% uranyl acetate followed by Reynold’s lead citrate. Captured images of selected sections were compared with captured light microscopic images of the block face before sectioning. Sections were examined with a Hitachi electron microscope. Figures were assembled and adjusted for brightness and contrast in Adobe Photoshop.
Specificity of antisera
The antiserum against residues 235–248 (SQENPNTYSEDLDV) of rat ppDYN was generated in guinea pig (Arvidsson et al., 1995). The specificity of ppDYN antiserum was tested by absorption controls with the cognate peptides (Arvidsson et al., 1995; Reyes et al., 2007). Preabsorption of ppDYN with the antigenic peptide at 1 μM blocked the immunoreactivity in regions known to express DYN (hippocampus, spinal cord). This is in agreement with others showing that DYN staining in rat brain tissue was blocked at peptide concentrations of 0.1 to 1 μM (Arvidsson et al., 1995). The distribution of ppDYN immunoreactivity was consistent with the localization of DYN immunoreactivity seen in other studies that examined LC (Elde and Hokfelt, 1993). Tissue sections processed in the absence of primary antibody did not exhibit any detectable immunoreactivity (Reyes et al., 2007).
The rabbit antiserum raised against methionine5-ENK has been previously characterized for cross-reactivity against several members of the opioid family of peptides (Velley et al., 1991; Pickel and Chan, 1994). Immunodot blots showed that the antiserum principally recognized methionine5-ENK except at high concentrations (1:2,000) where there was cross-reactivity with Leu5-ENK (Larsson 1981; Velley et al., 1991). A lower concentration (1:4,000) was used in the present study. The immunodot blot also showed that there was no cross-reactivity with alpha and gamma endorphin; however, a limited cross-reaction with beta endorphin was detected at higher concentrations that were not used in the present study. In previous studies, omission of the primary antibody eliminated any detectable staining (Van Bockstaele et al., 1995).
The CRF serum (lot C70) was raised in rabbit and was generated against the human/rat CRF peptide (SEEPPISLDLTFHLLREVLEMARAEQLAQQAHSNRKLMEIINH2), conjugated to human α-globulins. The antibody has been found in radioimmunoassay to recognize the NH2 terminus (residues 4–20) of ovine hypothalamic CRF (Rivier et al., 1983). Based on Edman degradation, peptide mapping, and amino acid analysis, the primary structure of the rat CRF was established to be: H-Ser-Glu-Glu-Pro-Pro-Ile-Ser-Leu-Asp_leu-Thr-Phe-His-Leu-Leu-Arg-Glu-Val-Leu-Glu-Met-Ala-Glu-Gln-Leu-Ala-Gln-Gln-Ala-His-Ser-Asn-Arg-Lys-Leu-Met-Glu-Ile-Ile-Nh2 (Rivier et al., 1983). The specificity of the CRF serum was tested by incubating sections in primary CRF antiserum preabsorbed for 24 hours with CRF at 1 mg/ml. Specific immunoreactivity was eliminated by prior absorption with its immunogen at 1 mg/ml and 10 mg/ml (Sawchenko et al., 1984; Van Bockstaele et al., 1996). An immunodot blot showed that the CRF antiserum used in the present study exhibited intense recognition of the parent antigen CRF and no cross-reactivity with any of the dilutions of other peptides including melanin-concentrating hormone or α-melanocyte-stimulating hormone (VanBockstaele et al., 1996). Additionally, sections processed in the absence of the primary CRF antiserum did not exhibit CRF immunoreactivity (Van Bockstaele et al., 1996).
The immunogen for mouse monoclonal antiserum was raised against denatured TH from rat pheochromocytoma, labels a single band at approximately 62kD corresponding to TH, and does not cross-react with dopamine-β-hydroxylase, dihydropterdine reductase, phenylethanolamine-N-methyltransferase, phenylalanine hydroxylase or tryptophan hydroxylase. The antibody has a wide species cross-reactivity. The specificity of the TH antibody has been examined by preabsorption of the antibody with a high concentration of TH (Van Bockstaele and Pickel, 1993). Omission of the primary antibody abolished any detectable immunoreactivity (Reyes et al., 2007).
Controls and Data analysis
Some sections were used for control experiments and were processed in the absence of the primary antibodies. These sections were processed in parallel with tissue sections incubated with the primary antibodies. Sections processed in the absence of DYN, CRF or ENK primary antibody did not exhibit any detectable immunoreactivity. To evaluate cross-reactivity of the primary antisera by secondary antisera, some sections were processed for dual labeling with omission of one of the primary antisera. Tissue sections from rats with optimal preservation of ultrastructural morphology and with both markers clearly apparent were used for the analysis. For quantification of labeled profiles in 40 μm-thick sections immunolabeled before embedding for electron microscopy, we have observed that the collection of sections only from the surface of the section minimizes artifacts that may be associated with incomplete penetration of antisera. The analysis of tissue sections collected at the plastic-tissue interface ensured that both markers were detectable in all sections used for analysis (Chan et al., 1990). At least 15 grids containing 4–8 ultrathin sections each were collected from at least 3 plastic embedded sections of the LC-periLC from each animal. All axon and axon terminals containing immunoperoxidase labeling and immunogold-silver labeling for DYN and CRF, respectively, in the same fields of at least 11,000X magnification were photographed and later classified. This approach resulted in 300 DYN-labeled axon profiles.
The cellular elements were identified based on the description of Peters and colleagues (Peters and Palay, 1996; Peters et al., 1991). Somata contained a nucleus, Golgi apparatus and smooth endoplasmic reticulum. Proximal dendrites contained endoplasmic reticulum, were postsynaptic to axon terminals and were larger than 0.7 μm in diameter. A terminal was considered to form a synapse if it showed a junctional complex, a restricted zone of parallel membranes with slight enlargement of the intercellular space, and/or associated with postsynaptic thickening. Asymmetric synapses were identified by thick postsynaptic densities (Gray’s type I; Gray, 1959), in contrast, symmetric synapses had thin densities (Gray’s type II; Gray, 1959) both pre- and postsynaptically. A non-synaptic contact or apposition was defined as an axon terminal plasma membrane juxtaposed to that of a dendrite or soma devoid of recognizable membrane specializations and no intervening glial processes.
All potential neuronal targets of DYN-labeled axon terminals throughout the analysis were examined by defining their total associations with other profiles regardless of whether a membrane specialization was seen within the plane of section. These associations consisted of close appositions of neuronal plasma membranes not separated by glial processes. These profiles were considered since opioids as well as other peptides are released by exocytosis from nonsynaptic portions of plasmalemma (Karhunen et al., 2001; Thureson-Klein et al., 1986; Zhu et al., 1986). DYN-labeled profiles forming clear synaptic specializations were classified as symmetric (Gray’s Type II) or asymmetric (Gray’s Type I). On the other hand, undefined contacts were characterized by a junctional complex that was not readily identifiable.
Identification of gold-silver labeling in profiles
Selective gold-silver labeled profiles were identified by the presence, in single thin sections, of at least three gold particles within a cellular compartment. Whenever possible, the more lightly labeled axonal labeling for CRF was confirmed by detection in at least two serial sections. A profile containing only two gold particles in adjacent thin sections was designated as lacking detectable immunoreactivity. As observed in low magnification electron micrographs, background labeling in the neuropil, deemed spurious, was not commonly encountered. The criterion of three gold particles as indicative of CRF labeling is conservative and may have led to an underestimation of the number of CRF-labeled profiles. Another factor that may have led to the underestimation of labeled profiles is the limitation of immunocytochemical methods to detect trace amounts of CRF. Moreover, unbiased stereological methods were not used for counting labeled profiles, and the results of the numerical analysis can only be considered to be an estimate of the numbers of synapses and labeled profiles.
Lesion Experiments
Four rats were initially anesthetized with a combination of ketamine hydrochloride (100 mg/kg) and xylazine (2mg/kg) in saline and placed in a stereotaxic apparatus for surgery. Anesthesia was supplemented with isoflurane (Abbott Laboratories, North Chicago, IL; 0.5–1.0%, in air) via a specialized nose cone affixed to the sterotaxic frame (Stoelting Corp., Wood Dale, IL). The temperature of the animal was monitored throughout the surgical procedure and kept at 37°C by using a feedback-controlled heating device. The electrode consisted of a fine steel wire encased in a glass micropipette with an exposed steel tip. The ventral placement of the electrode was made into the CeA following the coordinates obtained from the Paxinos and Watson rat brain atlas (Paxinos and Watson, 1986), −3.2 mm posterior from bregma, −4.8 mm medial/lateral, 8.1 mm ventral from the top of the skull. Once the electrode was in place, unilateral lesions were made by passing 0.5 mA constant anodal current for 30s through the glass lined steel tip electrode. Following the procedure, the animals were allowed free access to food and water. After a five day survival period, the animals were transcardially perfused and the brains were collected for immunocytochemical labeling of DYN.
Light level densitometry
For light microscopic experiments, every third section cut in the coronal plane through the rostro-caudal segment of LC was processed for immunocytochemical visualization of DYN using standard immunoperoxidase labeling techniques. Images of immunostained sections were captured using a using a Leica DMRBE microscope (Leica Microsystems, Wetzlar, Germany). Sections were acquired between −9.19 and −10.04 from the bregma (Paxinos and Watson, 1986). Images were analyzed using the NIH Image J software. The average mean densities were determined from 7 sections from each rat. Data collected were compared within groups to allow for variations in staining intensities that occur across different immunocytochemical processing runs. Data were expressed as mean ± SEM. Significant decreases were statistically analyzed using the Student’s t-test. Statistical analysis was carried out using GraphPad Prism (GraphPad Software, Inc., San Diego, CA, USA).
Double fluorescent in situ hybridization
Protocols for riboprobe synthesis and in situ hybridization were adapted from previous investigations in this and other laboratories (Poulin et al., 2006; Simmons et al., 1989). The plasmids were generously provided by Dr Douglass (DYN; NCBI Accession M10088; (Civelli et al., 1985) and by Dr Mayo (CRF; NCBI Accession X03036; (Jingami et al., 1985). Sections were mounted onto positively charged slides (Surgipath) and dried in vacuum. The pre-hybridization treatment included permeabilization in proteinase K (10 μg/mL at 37°C; Roche), acetylation in 0.25% acetic anhydride in 100 mM TEA. Riboprobes were synthesized using the Fluorescein RNA labeling mix (Roche; CRF) and Digoxigenin RNA Labeling mix (Roche; DYN). Fluorescent-labeled antisense RNA copies were synthesized by incubating 1 μg of linearized plasmid in transcription buffer (1X), 10 mM dithiothreitol, 40 U of RNase inhibitor (Roche), and 20 U appropriate RNA polymerase for 120 min at 37°C. The riboprobes were purified with a RNA column (Roche Diagnostics). The probe (100–500 ng) was mixed into 1 ml of hybridization solution (518 μl of formamide, 62 μl of 5 M NaCl, 10 μl of 1 M Tris (pH 8.0), 2 μl of 0.5 M EDTA (pH 8.0), 20 μl of 50X Denhardt’s solution, 207 μl of 50% dextran sulfate, 50 μl of 10 mg/ml transfer RNA, 10μl of 1 M dithiothreitol, and completed to final volume with RNAse-free water). Hybridization was completed overnight on slide warmers set at 60°C and 100μL of the hybridization solution was spotted on each slide. Post-hybridization washes included 4X standard saline citrate (SSC), RNase A (20 μg/mL, 37°C), 2X SSC, 1X SSC, 0.5X SSC, 30 min 0.1 X SSC at 60°C, and 0.1 X SSC at room temperature. The final step consisted of a 30min blocking step (0.3% Triton-X and 0.5% Blocking Reagent (Roche) in PBS), followed by a 1h incubation in 1/200 Anti-FITC-POD antibody (Roche) diluted in PBS with 0.3% Triton-X and 1% Bovine Albumin Serum. We then washed the slides in PBS + 0.05% Tween 20 (4 × 5min) and revealed the antibody with FITC-Tyramide as instructed by the manufacturer (Perkin Elmer, TSA kit). After additional washes, the peroxidase was inactivated in 0.02N HCl for 20min (Liu et al., 2006) and the preceding steps were repeated to reveal the DIG-labeled probe, but an anti-DIG-POD (Roche) antibody and Cy3-Tyramide were used instead (Perkin Elmer, TSA kit). The slides were washed and coverslipped with an antifading solution (Vector). To validate the specificity of fluorescent in situ hybridization protocol (FISH), a slide was hybridized with sense probes was included in each experiment. To ensure the complete inactivation of the horseradish peroxidase (POD) after the first revelation with FITC-tyramide, we omitted the second antibody and incubated the slides with Cy3-Tyramide. In the absence of 0.02N HCl treatment, all FITC-positive cells were double labeled as expected. However, following the 20min 0.02N HCl incubation none of the FITC-positive cells were labeled with Cy3 and therefore showing the complete inactivation of the POD coupled with the first antibody. In addition, the extent of labeling observed following FISH is similar to the labeling observed with radioactive ISH, confirming the sensitivity of our protocol.
Data analysis
Data were collected on a Leica DM4200B microscope equipped with a Retiga 2000R digital camera. A series of high-resolution micrographs for each fluorochrome was taken using Openlab 5.0.1 (Improvision), and the co-localization of each fluorochrome was quantified. Two sections were analyzed per structure for a total of 2 animals. Neuroanatomical boundaries of CeA were identified using the adjacent thionin stained sections. The nomenclature used is derived from Swanson (Swanson, 2003). The photomicrographs were adjusted for brightness and contrast in Adobe Photoshop and the figures were assembled in Adobe Illustrator.
RESULTS
Immunofluorescence labeling of DYN, CRF, and TH in the LC
Immunofluorescence labeling using three distinct fluorophore-tagged secondary antibodies to identify DYN, CRF, and TH was conducted in the same section of tissue through the dorsal pontine tegmentum (Fig. 1A–F). Processes exhibiting immunolabeling for DYN (magenta labeling) or CRF (green labeling) overlapped in the LC core (Fig. 1A–B, 1D, 1F) and peri-LC (Fig. 1C–E) areas. The core of the LC is defined as the region enriched with noradrenergic cell bodies (Bajic et al., 2000), while the peri-LC is defined as the region of the dorsal pontine tegmentum medial and lateral to the LC that contains noradrenergic dendrites (Shipley et al., 1996). Numerous fibers, however, contained both DYN and CRF immunoreactivities (yellow labeling) in the core (Fig. 1A–B, 1D, 1F) and the peri-LC (Fig. 1C–E).
Figure 1.
Confocal immunofluorescence photomicrographs showing triple-labeling for DYN, CRF and TH in the coronal section through the LC. DYN immunoreactivity was labeled with rhodamine isothiocyanate (pseudocolored in magenta), CRF was labeled with fluorescein isothiocyanate (green) and TH was labeled with aminomethylcoumarin acetate (blue). A–C. High magnification photomicrographs of D showing DYN and CRF varicose processes overlapping TH neurons in the core LC (B–C) or peri-LC (A). D. Low magnification photomicrograph of A–C. Arrows indicate dorsal (D) and lateral (L) orientation of the tissue section. IV: fourth ventricle; scp: superior cerebellar peduncle. E–F. High magnification immunofluorescence images in a more rostral section through the LC showing processes containing DYN and CRF in close proximity to TH labeled perikarya. Straight arrows indicate processes containing both DYN and CRF (yellow). Scale bar, 250 μm.
Ultrastructural analysis of DYN and CRF
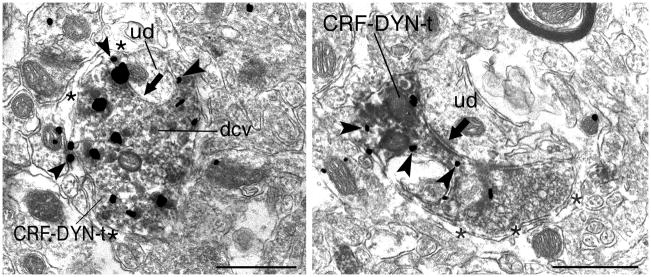
By electron microscopy, peroxidase and gold-silver labeling for DYN and CRF, respectively, were primarily identified in axon terminals and were abundant in similar portions of the neuropil (Fig. 2). Peroxidase labeling for DYN could be identified as a diffuse reaction product primarily within vesicle-filled axon terminals (Fig. 2). Silver-intensified gold labeling for CRF appeared as large, irregularly contoured black deposits that were often distributed throughout, as well as along the perimeter, of axon terminals (Fig. 2). The gold-silver deposits indicative of CRF immunolabeling were clearly distinguishable from the peroxidase reaction product indicative of DYN in the LC neuropil.
Figure 2.
Ultrastructural evidence for co-localization of DYN and CRF in the LC. A. An immunoperoxidase labeled axon terminal contains diffuse immunoreactivity for DYN, some of which is associated with dense core vesicles (dcv) and immunogold-silver labeling (arrowheads) for CRF. This terminal is surrounded by an astrocytic process (asterisks) and forms a symmetric synapse with an unlabeled dendrite (ud). B. A dually labeled CRF-DYN-axon terminal (CRF-DYN-t) forms an asymmetric synapse (black arrow) with an unlabeled dendrite (ud). Myelinated axons (ma) and unlabeled terminals (ut) can be seen in the neuropil. Scale bars, 0.5 μm.
Both DYN and CRF immunoreactivities were abundant in the neuropil at the ultrastructural level (Fig. 2). The targets of DYN and CRF were often small to medium dendritic processes. Single axon terminals were identified that contained both peroxidase labeling for DYN as well as gold-silver labeling for CRF (Fig. 2). Dually labeled axon terminals contained small clear synaptic vesicles and large dense core vesicles (Fig. 2A, 2C). Semi-quantitative analysis revealed that of the 277 axon terminals exhibiting DYN immunoreactivity, 35% (n = 97) also exhibited CRF immunoreactivity. Meanwhile, of the 243 axon terminals exhibiting CRF immunoreactivity, 38% (n = 97) also exhibited DYN immunoreactivity. Dual DYN and CRF-labeled axon terminals frequently synapsed on somatodendritic processes (Fig. 2A–C). Of the 97 DYN and CRF dual-labeled axon terminals, 46 (47%) did not form a recognizable synaptic contact in the plane of section analyzed. Approximately, 10% (9/83) of DYN and CRF-labeled axon terminals were enveloped by astrocytic processes (Fig. 2A–B). When synaptic specializations were identifiable, of the 97 DYN- and CRF-labeled axon terminals, 33% (32/97) were asymmetric type (Fig. 2B) while only 10% (10/97) were symmetric (Fig. 2A, 2C).
Single dendrites in the LC occasionally received convergent input from DYN and CRF-labeled axon terminals. Convergent input was defined as synaptic contacts formed by multiple axon terminals on the same cellular element.
Co-existence of DYN and CRF in the CeA
Our previous tract-tracing studies have provided anatomical evidence that amygdalar CRF targets noradrenergic LC dendrites (Van Bockstaele et al., 1998) and that lesions of the CeA reduce CRF immunoreactivity in the LC region (Tjoumakaris et al., 2003). As the CeA contains abundant DYN-positive cell bodies, we hypothesized that the CeA may provide DYN afferents to the LC. Peroxidase labeling for DYN was identified in the perikarya and dendritic processes in the CeA (Fig. 3A). Dual fluorescence in situ hybridization labeling showed DYN (magenta, Fig. 3B) and CRF (green, Fig. 3C) distributed in the CeA. Co-localization of DYN and CRF is evident in the CeA (Fig. 3D). Of the 35 neurons exhibiting DYN immunoreactivity, 31% co-localized with CRF. Of the 21 neurons exhibiting CRF, 53% colocalized with DYN.
Figure 3.

DYN and CRF co-localized in amygdalar neurons innervate LC neurons. A. Brightfield micrograph showing labeling for DYN in the coronal section through the CeA. DYN immunolabeling can be identified in neurons and processes. Small arrows indicate individual DYN-labeled perikarya and processes. Arrows point dorsally (D) and laterally (L). B–C. Fluorescence photomicrographs illustrating CRF mRNA (green, B) and DYN mRNA (magenta, C) in the CeA. Arrows point dorsally (D) and laterally (L). D. A merged image of B and C showing coexistence of CRF mRNA and DYN mRNA (some examples are pointed by arrowheads) in the CeA. Scale bars, 100 μm.
Lesions of the CeA result in decreases in DYN and CRF in the LC
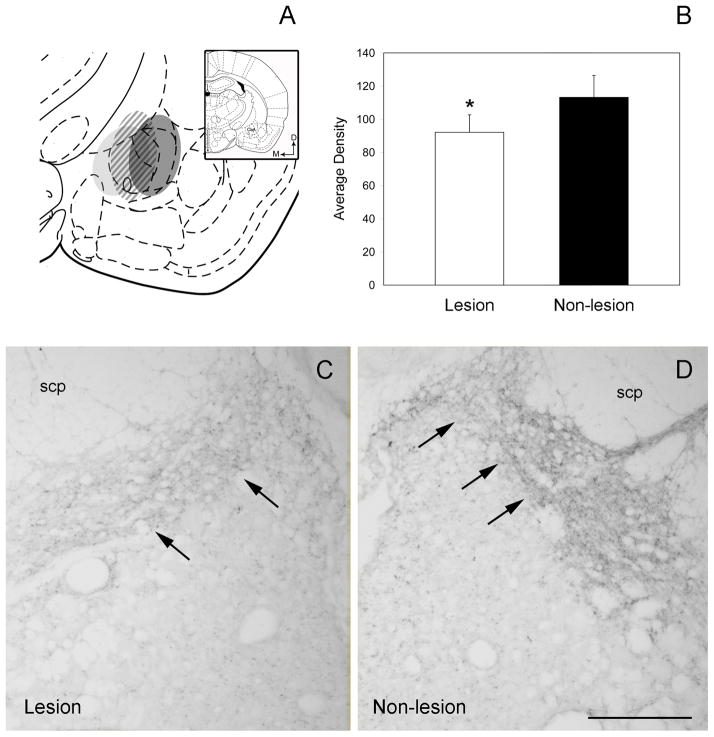
Every second section through the rostro-caudal segment of the dorsal pontine tegmentum from rats receiving an electrolytic lesion of the CeA was processed for the immunoperoxidase identification of DYN. A figure showing three examples of unilateral electrolytic lesions that were targeted to the CeA can be seen (Figure 4A). Lesion sites were identified using the Perls iron reaction as described in our previous study (Tjoumakaris et al., 2003). The lesions included medial (light gray), lateral (dark gray) and midline (hatched area) portions of the CeA (Figure 4A). Panels 4C–D of Figure 4 show DYN immunoreactivity arising from the lesion in Figure 4A that is represented by the dark gray oval. By light microscopy, on the side contralateral to the lesion site, peroxidase labeling for DYN was abundant within the LC/peri-LC area (Fig. 4D). In contrast, DYN immunoreactivity was significantly reduced on the side ipsilateral to the lesion as compared to the contralateral side (Fig. 4C). Light level densitometric analysis of peroxidase labeling for DYN was conducted in LC sections taken from the lesion and non-lesion side. A significant decrease in DYN immunoreactivity was noted on the lesion side as compared to the non-lesion side when comparisons of pixel intensity values were conducted (Fig. 4B).
Figure 4.
Electrolytic lesions of the CeA yield decreases in DYN immunoreactivity in the LC. A. Schematic representation of three cases showing the boundaries of electrolytic lesions that were centered in the CeA. The extent of the individual lesions can be identified by ovals containing either light or dark gray shading or diagonal lines. An inset shows a schematic diagram adapted from the rat brain atlas of Swanson (1992) showing the level where the CeA was lesioned. Arrows indicate dorsal (D) and medial (M) orientation of the section illustrated. CeA, central nucleus of the amygdala B. Light level densitometry measurements of coronal sections through the LC taken from rats with unilateral electrolytic lesions of the CeA. Sections were processed for immunoperoxidase localization of DYN. Following an electrolytic lesion of the CeA, a significant decrease in the average density of DYN immunoreactivity was observed on the ipsilateral side of the lesion compared to the contralateral side (n=5). *, P < 0.05 vs contralateral side. C. DYN immunoreactivity (arrows) is shown on the ipsilateral side to the lesion. A significant decrease in DYN immunolabeling in the LC on the ipsilateral side to the lesion is noted. D. DYN immunoreactivity (arrows) is shown on the side contralateral to the lesion. scp, superior cerebellar peduncle. Scale bar Scale bars = 100 μm.
DYN and ENK co-existence is not frequent in the LC
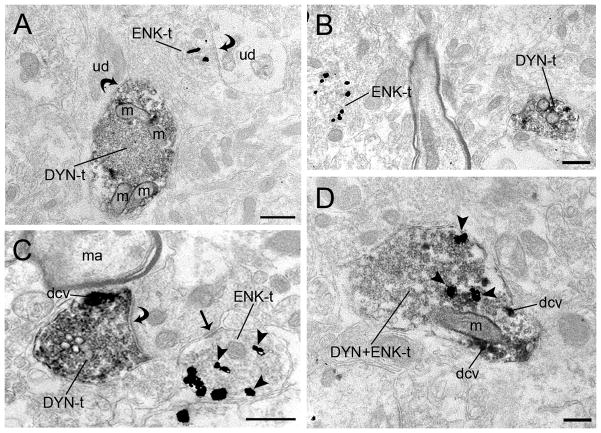
Immunohistochemical labeling for DYN and ENK was conducted in the same section of tissues through the LC (Fig. 5A–F). Processes that were labeled for DYN and ENK were distributed throughout the peri-LC (Fig. 5A–C) and the LC core (Fig. 5D–F). Although not abundant, DYN and ENK dually labeled punctate structures could be visualized in the peri-LC and LC core. Using electron microscopy, alternate sections through the rostro-caudal segment of the dorsal pontine tegmentum were processed for the immunoperoxidase labeling of DYN and immunogold-silver labeling of ENK. DYN-and ENK-labeled axon terminals were abundantly distributed in separate dendritic profiles in the LC neurons (Fig. 6A–B). Though dual-labeled DYN and ENK axon terminals were present (Fig. 6C–D), their co-existence was less frequent. Semi-quantitative analysis revealed that of the 311 axon terminals exhibiting DYN immunoreactivity, 12% (n = 36) also exhibited ENK immunoreactivity. Of the 342 axon terminals exhibiting ENK immunoreactivity, 11% (n = 36) also exhibited DYN immunoreactivity. Axon terminals showing dual labeling for DYN and ENK formed synaptic specializations with somatodendritic processes (not shown). Of the 36 DYN and ENK dual-labeled axon terminals, approximately 50% (n = 18) did not form a recognizable synaptic contact in the plane of section analyzed. When synaptic specializations were identifiable, of the 36 DYN- and ENK-labeled axon terminals, 42% (15/36) were of the symmetric type while only 10% (3/36) were of the asymmetric type. Interestingly, individual DYN- and ENK-labeled axon terminals frequently converged on a common dendrite (Fig. 5C).
Figure 5.
Confocal fluorescence photomicrographs showing DYN and ENK immunoreactivities in the coronal section through the peri LC (A–C) and LC core (D–F). A and D. DYN immunolabeling was detected by rhodamine isothiocyanate (pseudocolored in magenta). Arrows point to individual DYN-varicose processes that contain DYN. B and E. CRF was detected by fluorescein isothiocyanate (green). Arrows point to individual CRF-varicose processes that contain CRF. C and F. Merged images. Arrows point to DYN- and CRF-dual labeled varicose processes. Insets in panels C and F are high magnification images of the boxed regions. Scale bar, 100 μm.
Figure 6.
Electron photomicrographs showing immunoperoxidase and immunogold-silver labeling for DYN and ENK, respectively, in the LC. A. A DYN-labeled axon terminal (DYN-t) enriched with mitochondria (m) is contacting (curved arrow) an unlabeled dendrite (ud). In the same field, an ENK-labeled axon terminal (ENK-t) forms a symmetric synapse (curved arrow) with an unlabeled dendrite (ud). B. A DYN-t and ENK-t are shown in the same field. C. A DYN-t contains dense core vesicles (dcv) and an ENK-t converge in a common unlabeled dendrite (ud). Also shown is an asymmetric synapse (curved arrow) and symmetric synapse (arrow) formed by DYN-t and ENK-t in a common dendrite (ud). D. An axon terminal exhibit both DYN and ENK (DYN+ENK-t) immunolabeling containing dense core vesicles (dcv). m, mitochodria; ma, myelinated axon. Scale bar, 0.5 μm.
DISCUSSION
The results of this study provide ultrastructural evidence for the prominent coexistence of DYN and CRF in afferents to the LC of rat brain. These axons provide a potential mechanism for the co-release of these peptides in response to certain stimuli. These data are interesting in light of our previous study showing that coexistence of ENK and CRF is not frequent. The finding that the CeA provides robust DYN and CRF innervation to the LC supports the involvement of limbic pathways in the regulation of the LC in response to stress-related stimuli.
Methodological considerations
A caveat inherent in electron microscopic dual immunohistochemical labeling is related to penetration of primary and secondary antisera in thick tissue sections. Consequently, the limited penetration of DYN, CRF or ENK antiserum, especially in axon terminals with low concentrations of the peptide, may result in an underestimation of the relative frequency of peptide-immunoreactive axon terminals. To mitigate this possible limitation, tissue sections were collected near the tissue-Epon interface and profiles were sampled only when both markers were clearly present in the fields included in the analysis.
Evidence for opioid-CRF interactions in the LC
Previous ultrastructural studies have shown that axon terminals containing ENK or CRF synaptically contact LC dendrites, but that they do not frequently coexist in the same processes. Physiological studies support the idea that during stress or physiological challenges, endogenous CRF is released in the LC region to activate noradrenergic neurons. In fact, direct intracoerulear microinfusion of CRF antagonists prevents LC activation by hypotensive stress and colonic distention (Curtis et al., 1994; Lechner et al., 1997) and handling-induced increase in norepinephrine terminal release (Kawahara et al., 2000). Correspondingly, naloxone administration systemically potentiated LC discharge rates of restraint stressed rats supporting the idea that endogenous opioids are also released during stress to regulate LC activity (Abercrombie and Jacobs, 1988). Furthermore, Curtis and colleagues (Curtis et al., 2001) demonstrated that intracoerulear microinfusion of naloxone prevented LC inhibition associated with the termination of hypotensive stress. This result provides evidence for the functional release of an endogenous opioid within the LC (Curtis et al., 2001). Besides, it also demonstrates that while CRF release within the LC mediates LC activation during stress, endogenous opioid release within the LC mediates LC inhibition following stress. In light of this finding, it is possible that a common stimulus may elicit the release of both neuropeptides within the LC to modulate activity of this system in an opposing fashion. It is hypothesized that the action of endogenous opioid at the later onset may serve to counterbalance the excitatory effects of CRF on the LC-norepinephrine system when the stressor is terminated.
Co-localization of DYN and CRF in axon terminals in the LC
Immunofluorescence miscroscopy revealed prominent co-existence between DYN and CRF terminal fields in the LC region. Specifically, DYN and CRF co-localization was evident in the core of the LC as well as within the peri-LC, medial and ventral to the superior cerebellar peduncle and dorsolateral to the core. The anatomical distribution of DYN and CRF co-localization is similar with the distribution of enkephalin and CRF in the LC (Tjoumakaris et al., 2003); these fibers were more often distributed in the LC core with some fibers observed dorsolateral to the LC core. Conversely, the pro-opiomelanocortin and CRF co-localization showed prominent distribution in the ventromedial aspect of the LC and little evidence in the dorsolateral aspect of the LC (Reyes et al., 2006). The DYN and CRF co-localization was confirmed at the ultrastructural level where dually-labeled DYN and CRF axon terminals were concentrated in the LC core and dorsolateral to it. Relative to the degree of co-localization with GABA (38% of ENK terminals; Van Bockstaele and Chan, 1997) or glutamate (28% of ENK terminals; Tjoumakaris et al., 2003; Van Bockstaele et al., 2000) the percentage of DYN-immunolabeled terminals that exhibited CRF immunolabeling in this study was significant (35%). However, with respect to ENK, the frequency of coexistence was higher (16% of CRF terminals; Tjoumakaris et al., 2003). CRF-immunolabeling is infrequently observed in GABA-immunolabeled terminals in the peri-LC, although it is frequently co-localized with glutamate immunoreactivity (Valentino et al., 2001). This is interesting in light of the prominent distribution of DYN and CRF in axon terminals forming asymmetric synapses. By specifically activating neurons that give rise to these terminals, stimuli may modulate LC activity via a co-release of DYN and CRF.
Sources of DYN/CRF terminals
Previous anatomical studies have shown that CeA is enriched with DYN cell bodies (Khachaturian et al., 1982; Watson et al., 1983). Likewise, retrograde and anterograde tracing studies revealed that CeA is the source of the CRF innervation in the LC (Sakanaka et al., 1986; Van Bockstaele et al., 1998). Accordingly, the CeA is considered a potential source of DYN/CRF terminals in the LC. In the present study, unilateral lesions of the CeA substantially decreased CRF-immunolabeling in the LC (data not shown), consistent with our previous reports (Van Bockstaele et al., 1998; Tjoumakaris et al., 2003) as well as others (Sakanaka et al., 1986). Evidence of reduced DYN innervation in the same cases indicates that DYN innervation of this region and the dually labeled axon terminals are derived from common limbic sources. Additional sources of potential afferent inputs to the LC that colocalize DYN and CRF include the bed nucleus of stria terminalis, the nucleus of the solitary tract and hypothalamic regions including the paraventricular nucleus of the hypothalamus (Reyes et al., 2005; Van Bockstaele et al., 1999a; Van Bockstaele et al., 1999b).
Mechanisms underlying co-release of CRF and DYN
Electrophysiological evidence implicated CeA as the source of CRF underlying LC activation during hypotensive challenge (Curtis et al., 2001). Taken together with the present findings that the CeA considerably contributes to DYN innervation of the LC/peri-LC region, suggest that in response to hypotensive challenge CRF and DYN are co-released from the same terminals. Therefore, it is likely that hypotensive challenge may engage CRF and DYN originating from common neurons from the CeA that innervate LC processes. As such, the termination of hypotensive challenge may be mediated by DYN. Future studies are required to determine what stimuli elicit co-release of DYN and CRF in the LC.
Co-localization of DYN and ENK in the LC
Immunofluorescence and ultrastructural studies revealed evidence of co-localization of DYN- and ENK-labeled axon terminals in the LC, however, it was less frequent. More often, the cellular interactions between individual DYN- and ENK-labeled axon terminals showed a convergence on a common LC dendrite, and specifically with presynaptic associations. This result suggests that co-release of DYN and ENK in the LC by distinct afferents can impact common targets. Potential sources of DYN is the CeA (Khachaturian et al., 1982; Watson et al., 1983), and this is supported by the electrolytic lesions of the CeA in the present study. However, CeA does not provide robust ENK innervation to the LC (Tjoumakaris et al., 2003). Therefore, while the CeA serves as a source of DYN in the LC, it is not the source of axon terminals that co-localize DYN and ENK. A possible mechanism by which a common stimulus could result in DYN and ENK release to impact the same LC neuron is via parallel stimulation of separate populations of DYN and ENK neurons that converge on common targets in the noradrenergic LC. It has been established that the PGi provides a robust enkephalinergic projections to the core and peri-LC regions (Drolet et al., 1992). Hence, it is likely that two important autonomic brain regions, the CeA and the PGi, converge on the LC to influence the activity of noradrenergic LC neurons under certain conditions.
Acknowledgments
Grant sponsor: National Institutes of Health; Grant number: DA 09082 and DA 15395 (E. J. V. B.)
Canadian Institute of Health Research; MOP-62921 (G.D.)
The authors would like to thank Ms. Sylvie Laforest and Geneviève Pleau for their excellent technical assistance with FISH techniques, and Ms. Azuree Johnson and Julia Glaser for their excellent technical assistance with electron microscopy.
References
- Abercrombie ED, Jacobs BL. Systemic naloxone administration potentiates locus coeruleus noradrenergic neuronal activity under stressful but not non-stressful conditions. Brain Res. 1988;441:362–366. doi: 10.1016/0006-8993(88)91415-1. [DOI] [PubMed] [Google Scholar]
- Akil H, Watson SJ, Young E, Lewis ME, Khachaturian H, Walker JM. Endogenous opioids: biology and function. Annu Rev Neurosci. 1984;7:223–255. doi: 10.1146/annurev.ne.07.030184.001255. [DOI] [PubMed] [Google Scholar]
- Alreja M, Aghajanian GK. Opiates suppress a resting sodium-dependent inward current and activate an outward potassium current in locus coeruleus neurons. J Neurosci. 1993;13:3525–3532. doi: 10.1523/JNEUROSCI.13-08-03525.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arvidsson U, Riedl M, Chakrabarti S, Vulchanova L, Lee JH, Nakano AH, Lin X, Loh HH, Law PY, Wessendorf MW, Elde R. The kappa-opioid receptor is primarily postsynaptic: combined immunohistochemical localization of the receptor and endogenous opioids. Proc Natl Acad Sci USA. 1995;92:5062–5066. doi: 10.1073/pnas.92.11.5062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aston-Jones G, Bloom FE. Activity of norepinephrine-containing locus coeruleus neurons in behaving rats anticipates fluctuations in the sleep-waking cycle. J Neurosci. 1981a;1:876–886. doi: 10.1523/JNEUROSCI.01-08-00876.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aston-Jones G, Bloom FE. Norepinephrine-containing locus coeruleus neurons in behaving rats exhibit pronounced responses to non-noxious environmental stimuli. J Neurosci. 1981b;1:887–900. doi: 10.1523/JNEUROSCI.01-08-00887.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bajic D, Proudfit HK, Van Bockstaele EJ. Periaqueductal gray neurons monosynaptically innervate extranuclear noradrenergic dendrites in the rat pericoerulear region. J Comp Neurol. 2000;427:649–662. doi: 10.1002/1096-9861(20001127)427:4<649::aid-cne11>3.0.co;2-m. [DOI] [PubMed] [Google Scholar]
- Basbaum AI, Fields HL. Endogenous pain control systems: brainstem spinal pathways and endorphin circuitry. Annu Rev Neurosci. 1984;7:309–338. doi: 10.1146/annurev.ne.07.030184.001521. [DOI] [PubMed] [Google Scholar]
- Bremner JD, Krystal JH, Southwick SM, Charney DS. Noradrenergic mechanisms in stress and anxiety: II. Clinical studies. Synapse. 1996;23:39–51. doi: 10.1002/(SICI)1098-2396(199605)23:1<39::AID-SYN5>3.0.CO;2-I. [DOI] [PubMed] [Google Scholar]
- Chan J, Aoki C, Pickel VM. Optimization of differential immunogold-silver and peroxidase labeling with maintenance of ultrastructure in brain sections before plastic embedding. J Neurosci Methods. 1990;33:113–127. doi: 10.1016/0165-0270(90)90015-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Civelli O, Douglass J, Goldstein A, Herbert E. Sequence and expression of the rat prodynorphin gene. Proc Natl Acad Sci USA. 1985;82:4291–4295. doi: 10.1073/pnas.82.12.4291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Commons KG, Milner TA. Ultrastructural heterogeneity of enkephalin-containing terminals in the rat hippocampal formation. J Comp Neurol. 1995;358:324–342. doi: 10.1002/cne.903580303. [DOI] [PubMed] [Google Scholar]
- Curtis AL, Bello NT, Valentino RJ. Evidence for functional release of endogenous opioids in the locus coeruleus during stress termination. J Neurosci. 2001;21:RC152. doi: 10.1523/JNEUROSCI.21-13-j0001.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Curtis AL, Grigoriadis DE, Page ME, Rivier J, Valentino RJ. Pharmacological comparison of two corticotropin-releasing factor antagonists: in vivo and in vitro studies. J Pharmacol Exp Ther. 1994;268:359–365. [PubMed] [Google Scholar]
- Curtis AL, Lechner SM, Pavcovich LA, Valentino RJ. Activation of the locus coeruleus noradrenergic system by intracoerulear microinfusion of corticotropin-releasing factor: effects on discharge rate, cortical norepinephrine levels and cortical electroencephalographic activity. J Pharmacol Exp Ther. 1997;281(1):163–172. [PubMed] [Google Scholar]
- Drolet G, Van Bockstaele EJ, Aston-Jones G. Robust enkephalin innervation of the locus coeruleus from the rostral medulla. J Neurosci. 1992;12:3162–3174. doi: 10.1523/JNEUROSCI.12-08-03162.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elde R, Hokfelt T. Coexistence of opioid peptides with other neurotransmitters. In: Herz A, editor. Handbook of Experimental Pharmacology, Opioids I. Berlin: Springer; 1993. pp. 585–624. [Google Scholar]
- Fallon JH, Leslie FM. Distribution of dynorphin and enkephalin peptides in the rat brain. J Comp Neurol. 1986;249:293–336. doi: 10.1002/cne.902490302. [DOI] [PubMed] [Google Scholar]
- Foote SL, Bloom FE, Aston-Jones G. Nucleus locus ceruleus: new evidence of anatomical and physiological specificity. Physiol Rev. 1983;63:844–914. doi: 10.1152/physrev.1983.63.3.844. [DOI] [PubMed] [Google Scholar]
- Gray EG. Axosomatic and axo-dendritic synapses of the cerebral cortex: an electron microscopic study. J Anat. 1959;93:420–433. [PMC free article] [PubMed] [Google Scholar]
- Ishimatsu M, Williams JT. Synchronous activity in locus coeruleus results from dendritic interactions in pericoerulear regions. J Neurosci. 1996;16:5196–5204. doi: 10.1523/JNEUROSCI.16-16-05196.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jingami H, Mizuno N, Takahashi H, Shibahara S, Furutani Y, Imura H, Numa S. Cloning and sequence analysis of cDNA for rat corticotropin-releasing factor precursor. FEBS Letters. 1985;191:63–66. doi: 10.1016/0014-5793(85)80994-7. [DOI] [PubMed] [Google Scholar]
- Karhunen T, Vilim FS, Alexeeva V, Weiss KR, Church PJ. Targeting of peptidergic vesicles in cotransmitting terminals. J Neurosci. 2001;21:RC127. doi: 10.1523/JNEUROSCI.21-03-j0005.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawahara H, Kawahara Y, Westerink BH. The role of afferents to the locus coeruleus in the handling stress-induced increase in the release of noradrenaline in the medial prefrontal cortex: a dual-probe microdialysis study in the rat brain. Eur J Pharmacol. 2000;387:279–286. doi: 10.1016/s0014-2999(99)00793-1. [DOI] [PubMed] [Google Scholar]
- Khachaturian H, Watson SJ, Lewis ME, Coy D, Goldstein A, Akil H. Dynorphin immunocytochemistry in the rat central nervous system. Peptides. 1982;3:941–954. doi: 10.1016/0196-9781(82)90063-8. [DOI] [PubMed] [Google Scholar]
- Larsson LI. A novel immunocytochemical model system for specificity and sensitivity screening of antisera against multiple antigens. J Histochem Cytochem. 1981;29:408–410. doi: 10.1177/29.3.7016977. [DOI] [PubMed] [Google Scholar]
- Lechner SM, Curtis AL, Brons R, Valentino RJ. Locus coeruleus activation by colon distention: role of corticotropin-releasing factor and excitatory amino acids. Brain Res. 1997;756:114–124. doi: 10.1016/s0006-8993(97)00116-9. [DOI] [PubMed] [Google Scholar]
- Leranth C, Pickel VM. Electron microscopic preembedding double-labeling methods. In: Heimer L, Zaborszky L, editors. Neuroanatomical tracing methods. 1. Vol. 2. New York: Plenum Press; 1989. pp. 129–172. [Google Scholar]
- Liu G, Amin S, Okuhama NN, Liao G, Mingle LA. A quantitative evaluation of peroxidase inhibitors for tyramide signal amplification mediated cytochemistry and histochemistry. Histochem Cell Biol. 2006;126:283–291. doi: 10.1007/s00418-006-0161-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mague SD, Pliakas AM, Todtenkopf MS, Tomasiewicz HC, Zhang Y, Stevens WC, Jr, Jones RM, Portoghese PS, Carlezon WA., Jr Antidepressant-like effects of kappa-opioid receptor antagonists in the forced swim test in rats. J Pharmacol Exp Ther. 2003;305:323–330. doi: 10.1124/jpet.102.046433. [DOI] [PubMed] [Google Scholar]
- Mansour A, Fox CA, Meng F, Akil H, Watson SJ. Kappa 1 receptor mRNA distribution in the rat CNS: comparison to kappa receptor binding and prodynorphin mRNA. Mol Cell Neurosci. 1994;5:124–144. doi: 10.1006/mcne.1994.1015. [DOI] [PubMed] [Google Scholar]
- McLaughlin JP, Marton-Popovici M, Chavkin C. Kappa opioid receptor antagonism and prodynorphin gene disruption block stress-induced behavioral responses. J Neurosci. 2003;23:5674–5683. doi: 10.1523/JNEUROSCI.23-13-05674.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miyawaki T, Kawamura H, Komatsu K, Yasugi T. Chemical stimulation of the locus coeruleus: inhibitory effects on hemodynamics and renal sympathetic nerve activity. Brain Res. 1991;568:101–108. doi: 10.1016/0006-8993(91)91384-d. [DOI] [PubMed] [Google Scholar]
- Morilak DA, Fornal CA, Jacobs BL. Effects of physiological manipulations on locus coeruleus neuronal activity in freely moving cats. II. Cardiovascular challenge. Brain Res. 1987;422:24–31. doi: 10.1016/0006-8993(87)90536-1. [DOI] [PubMed] [Google Scholar]
- Murase S, Inui K, Nosaka S. Baroreceptor inhibition of the locus coeruleus noradrenergic neurons. Neuroscience. 1994;61:635–643. doi: 10.1016/0306-4522(94)90440-5. [DOI] [PubMed] [Google Scholar]
- Page ME, Abercrombie ED. Discrete local application of corticotropin-releasing factor increases locus coeruleus discharge and extracellular norepinephrine in rat hippocampus. Synapse. 1999;33:304–313. doi: 10.1002/(SICI)1098-2396(19990915)33:4<304::AID-SYN7>3.0.CO;2-Z. [DOI] [PubMed] [Google Scholar]
- Page ME, Valentino RJ. Locus coeruleus activation by physiological challenges. Brain Res Bull. 1994;35:557–560. doi: 10.1016/0361-9230(94)90169-4. [DOI] [PubMed] [Google Scholar]
- Paxinos G, Watson C. The rat brain in stereotaxic coordinates. New York: Academic Press; 1986. [DOI] [PubMed] [Google Scholar]
- Peters A, Palay SL. The morphology of synapses. J Neurocytol. 1996;25:687–700. doi: 10.1007/BF02284835. [DOI] [PubMed] [Google Scholar]
- Peters A, Palay SL, Webster Hd. The Fine Structure of the Nervous System. New York: Oxford University Press; 1991. [Google Scholar]
- Pickel VM, Chan J. Met5-enkephalin is localized within axon terminals in the subfornical organ: vascular contacts and interactions with neurons containing gamma-aminobutyric acid. J Neurosci Res. 1994;37:735–749. doi: 10.1002/jnr.490370608. [DOI] [PubMed] [Google Scholar]
- Piercey MF, Varner K, Schroeder LA. Analgesic activity of intraspinally administered dynorphin and ethylketocyclazocine. Eur J Pharmacol. 1982;80:283–284. doi: 10.1016/0014-2999(82)90072-3. [DOI] [PubMed] [Google Scholar]
- Poulin JF, Chevalier B, Laforest S, Drolet G. Enkephalinergic afferents of the centromedial amygdala in the rat. J Comp Neurol. 2006;496:859–876. doi: 10.1002/cne.20956. [DOI] [PubMed] [Google Scholar]
- Reyes BAS, Glaser JD, Magtoto R, Van Bockstaele EJ. Proopiomelanocortin co-localizes with corticotropin-releasing factor in axon terminals of the noradrenergic nucleus locus coeruleus. Eur J Neurosci. 2006;23:2067–2077. doi: 10.1111/j.1460-9568.2006.04744.x. [DOI] [PubMed] [Google Scholar]
- Reyes BAS, Valentino RJ, Xu G, Van Bockstaele EJ. Hypothalamic projections to locus coeruleus neurons in rat brain. Eur J Neurosci. 2005;22:93–106. doi: 10.1111/j.1460-9568.2005.04197.x. [DOI] [PubMed] [Google Scholar]
- Reyes BAS, Johnson AD, Glaser JD, Commons KG, Van Bockstaele EJ. Dynorphin-containing axons directly innervate noradrenergic neurons in the rat nucleus locus coeruleus. Neuroscience. 2007;145:1077–1086. doi: 10.1016/j.neuroscience.2006.12.056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rivier J, Spiess J, Vale W. Characterization of rat hypothalamic corticotropin-releasing factor. Proc Natl Acad Sci USA. 1983;80:4851–4855. doi: 10.1073/pnas.80.15.4851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sakanaka M, Shibasaki T, Lederis K. Distribution and efferent projections of corticotropin-releasing factor-like immunoreactivity in the rat amygdaloid complex. Brain Res. 1986;382:213–238. doi: 10.1016/0006-8993(86)91332-6. [DOI] [PubMed] [Google Scholar]
- Sawchenko PE, Swanson LW, Vale WW. Co-expression of corticotropin-releasing factor and vasopressin immunoreactivity in parvocellular neurosecretory neurons of the adrenalectomized rat. Proc Natl Acad Sci USA. 1984;81:1883–1887. doi: 10.1073/pnas.81.6.1883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shipley MT, Fu L, Ennis M, Liu WL, Aston-Jones G. Dendrites of locus coeruleus neurons extend preferentially into two pericoerulear zones. J Comp Neurol. 1996;365:56–68. doi: 10.1002/(SICI)1096-9861(19960129)365:1<56::AID-CNE5>3.0.CO;2-I. [DOI] [PubMed] [Google Scholar]
- Shirayama Y, Ishida H, Iwata M, Hazama GI, Kawahara R, Duman RS. Stress increases dynorphin immunoreactivity in limbic brain regions and dynorphin antagonism produces antidepressant-like effects. J Neurochem. 2004;90:1258–1268. doi: 10.1111/j.1471-4159.2004.02589.x. [DOI] [PubMed] [Google Scholar]
- Shuster SJ, Riedl M, Li X, Vulchanova L, Elde R. The kappa opioid receptor and dynorphin co-localize in vasopressin magnocellular neurosecretory neurons in guinea-pig hypothalamus. Neuroscience. 2000;96:373–383. doi: 10.1016/s0306-4522(99)00472-8. [DOI] [PubMed] [Google Scholar]
- Simmons DM, Arriza JL, Swanson LW. A complete protocol for in situ hybridization of messenger RNAs in brain and other tissues with radiolabeled single-stranded RNA probes. J Histotechnology. 1989;12:169–181. [Google Scholar]
- Svensson TH. Peripheral, autonomic regulation of locus coeruleus noradrenergic neurons in brain: putative implications for psychiatry and psychopharmacology. Psychopharmacology (Berl) 1987;92:1–7. doi: 10.1007/BF00215471. [DOI] [PubMed] [Google Scholar]
- Thureson-Klein A, Klein RL, Zhu PC. Exocytosis from large dense cored vesicles as a mechanism for neuropeptide release in the peripheral and central nervous system. Scan Electron Microsc. 1986;(Pt 1):179–187. [PubMed] [Google Scholar]
- Tjoumakaris SI, Rudoy C, Peoples J, Valentino RJ, Van Bockstaele EJ. Cellular interactions between axon terminals containing endogenous opioid peptides or corticotropin-releasing factor in the rat locus coeruleus and surrounding dorsal pontine tegmentum. J Comp Neurol. 2003;466:445–456. doi: 10.1002/cne.10893. [DOI] [PubMed] [Google Scholar]
- Vale W, Spiess J, Rivier C, Rivier J. Characterization of a 41-residue ovine hypothalamic peptide that stimulates secretion of corticotropin and beta-endorphin. Science. 1981;213:1394–1397. doi: 10.1126/science.6267699. [DOI] [PubMed] [Google Scholar]
- Valentino RJ, Foote SL, Aston-Jones G. Corticotropin-releasing factor activates noradrenergic neurons of the locus coeruleus. Brain Res. 1983;270:363–367. doi: 10.1016/0006-8993(83)90615-7. [DOI] [PubMed] [Google Scholar]
- Valentino RJ, Foote SL, Page ME. The locus coeruleus as a site for integrating corticotropin-releasing factor and noradrenergic mediation of stress responses. Ann N Y Acad Sci. 1993;697:173–188. doi: 10.1111/j.1749-6632.1993.tb49931.x. [DOI] [PubMed] [Google Scholar]
- Valentino RJ, Rudoy C, Saunders A, Liu XB, Van Bockstaele EJ. Corticotropin-releasing factor is preferentially colocalized with excitatory rather than inhibitory amino acids in axon terminals in the peri-locus coeruleus region. Neuroscience. 2001;106:375–384. doi: 10.1016/s0306-4522(01)00279-2. [DOI] [PubMed] [Google Scholar]
- Van Bockstaele EJ, Branchereau P, Pickel VM. Morphologically heterogeneous met-enkephalin terminals form synapses with tyrosine hydroxylase-containing dendrites in the rat nucleus locus coeruleus. J Comp Neurol. 1995;363:423–438. doi: 10.1002/cne.903630307. [DOI] [PubMed] [Google Scholar]
- Van Bockstaele EJ, Chan J. Electron microscopic evidence for coexistence of leucine5-enkephalin and gamma-aminobutyric acid in a subpopulation of axon terminals in the rat locus coeruleus region. Brain Res. 1997;746:171–182. doi: 10.1016/s0006-8993(96)01194-8. [DOI] [PubMed] [Google Scholar]
- Van Bockstaele EJ, Colago EEO, Valentino RJ. Corticotropin-releasing factor-containing axon terminals synapse onto catecholamine dendrites and may presynaptically modulate other afferents int he rostral pole of the nucleus locus coeruleus in the rat brain. J Comp Neurol. 1996;364:523–534. doi: 10.1002/(SICI)1096-9861(19960115)364:3<523::AID-CNE10>3.0.CO;2-Q. [DOI] [PubMed] [Google Scholar]
- Van Bockstaele EJ, Colago EEO, Valentino RJ. Amygdaloid corticotropin-releasing factor targets locus coeruleus dendrites: substrate for the co-ordination of emotional and cognitive limbs of the stress response. J Neuroendocrin. 1998;10:743–757. doi: 10.1046/j.1365-2826.1998.00254.x. [DOI] [PubMed] [Google Scholar]
- Van Bockstaele EJ, Peoples J, Telegan P. Efferent projections of the nucleus of the solitary tract to peri-locus coeruleus dendrites in rat brain: evidence for a monosynaptic pathway. J Comp Neurol. 1999a;412:410–428. doi: 10.1002/(sici)1096-9861(19990927)412:3<410::aid-cne3>3.0.co;2-f. [DOI] [PubMed] [Google Scholar]
- Van Bockstaele EJ, Peoples J, Valentino RJ. A.E. Bennett Research Award. Anatomic basis for differential regulation of the rostrolateral peri-locus coeruleus region by limbic afferents. Biol Psychiatry. 1999b;46:1352–1363. doi: 10.1016/s0006-3223(99)00213-9. [DOI] [PubMed] [Google Scholar]
- Van Bockstaele EJ, Pickel VM. Ultrastructure of serotonin-immunoreactive terminals in the core and shell of the rat nucleus accumbens: cellular substrates for interactions with catecholamine afferents. J Comp Neurol. 1993;334:603–617. doi: 10.1002/cne.903340408. [DOI] [PubMed] [Google Scholar]
- Van Bockstaele EJ, Saunders A, Commons K, Liu X-B, Peoples J. Evidence for co-existence of enkephalin and glutamate in axon terminals and cellular sites for functional interactions of their receptors in the rat locus coeruleus. J Comp Neurol. 2000;417:103–114. doi: 10.1002/(sici)1096-9861(20000131)417:1<103::aid-cne8>3.0.co;2-l. [DOI] [PubMed] [Google Scholar]
- Velley L, Milner TA, Chan J, Morrison SF, Pickel VM. Relationship of Met-enkephalin-like immunoreactivity to vagal afferents and motor dendrites in the nucleus of the solitary tract: a light and electron microscopic dual labeling study. Brain Res. 1991;550:298–312. doi: 10.1016/0006-8993(91)91332-u. [DOI] [PubMed] [Google Scholar]
- Watson SJ, Akil H, Ghazarossian VE, Goldstein A. Dynorphin immunocytochemical localization in brain and peripheral nervous system: preliminary studies. Proc Natl Acad Sci USA. 1981;78:1260–1263. doi: 10.1073/pnas.78.2.1260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watson SJ, Khachaturian H, Akil H, Coy DH, Goldstein A. Comparison of the distribution of dynorphin systems and enkephalin systems in brain. Science. 1982;218:1134–1136. doi: 10.1126/science.6128790. [DOI] [PubMed] [Google Scholar]
- Watson SJ, Khachaturian H, Taylor L, Fischli W, Goldstein A, Akil H. Pro-dynorphin peptides are found in the same neurons throughout rat brain: immunocytochemical study. Proc Natl Acad Sci USA. 1983;80:891–894. doi: 10.1073/pnas.80.3.891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams JT, Egan TM, North RA. Enkephalin opens potassium channels on mammalian central neurones. Nature. 1982;299(5878):74–77. doi: 10.1038/299074a0. [DOI] [PubMed] [Google Scholar]
- Xu G, Van Bockstaele EJ, Reyes B, Bethea T, Valentino RJ. Chronic morphine sensitizes the brain norepinephrine system to stress. J Neurosci. 2004;24:8193–8197. doi: 10.1523/JNEUROSCI.1657-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zamir N, Palkovits M, Brownstein MJ. Distribution of immunoreactive dynorphin in the central nervous system of the rat. Brain Res. 1983;280:81–93. doi: 10.1016/0006-8993(83)91176-9. [DOI] [PubMed] [Google Scholar]
- Zhu PC, Thureson-Klein A, Klein RL. Exocytosis from large dense cored vesicles outside the active synaptic zones of terminals within the trigeminal subnucleus caudalis: a possible mechanism for neuropeptide release. Neuroscience. 1986;19:43–54. doi: 10.1016/0306-4522(86)90004-7. [DOI] [PubMed] [Google Scholar]