Abstract
Signaling from the human hematopoietic stem cell (HSC) niche formed by osteoblastic cells regulates hematopoiesis. We previously found that retinoic acid receptor alpha (RARα), a transcription factor activated by retinoic acid (RA), mediates both granulocytic and osteoblastic differentiation. This effect depends on decreased phosphorylation of serine 77 of RARα (RARαS77) by the cyclin-dependent kinase-activating kinase (CAK) complex, a key cell cycle regulator. Here we report that by suppressing CAK phosphorylation of RARα, RA induces FGF8f to mediate osteosarcoma U2OS cell differentiation in an autocrine manner. By contrast, paracrine FGF8f secreted into osteoblast-conditioned medium (OCM) by U2OS cells transduced with FGF8f or a phosphorylation-defective RARαS77 mutant, RARαS77A, bypasses RA stimuli to cross-mediate granulocytic differentiation of different types of human leukemic myeloblasts and normal primitive hematopoietic CD34+ cells, likely through modulating mitogen-activated protein kinase (MAPK) pathways. Further experiments using recombinant human FGF8f (rFGF8f) stimuli, antibody neutralization, and peptide blocking demonstrate that paracrine FGF8f is required for mediating terminal leukemic myeloblast differentiation. These studies suggest a novel regulatory mechanism of granulocytic differentiation instigated by RA from the HSC niche, which links loss of CAK phosphorylation of RARα with paracrine FGF8f-mediated MAPK signaling to mediate leukemic myeloblast differentiation in the absence of RA. Hence, these findings provide a compelling molecular rationale for further investigation of paracrine FGF8f regulation, with the intent of devising HSC niche-based FGF8f therapeutics for myeloid leukemia, with or without RA-resistance.
Keywords: Osteoblast-formed HSC niche, Leukemic myeloblasts differentiation, Normal human primitive CD34+ cells, RA-induced CAK-RARα signaling, MAPK pathway
Introduction
RA therapy for acute promyelocytic leukemia (APL) represents the most successful example of differentiation-induction therapy in current clinical oncology (1–3); however, this success has not extended to the remaining 85% of myeloid leukemia subtypes, largely because the mechanisms of RA-induced myeloid differentiation remain unclear. RA signaling is elicited through both classical genomic pathway and rapid non-genomic signal transduction. The role of the classical RA-induced genomic pathway (4) in mediating cancer cell differentiation (5, 6) is well recognized (7). On the other hand, RA can exert rapid non-genomic effects independently of RAR-mediated gene transcription (8) to induce cell differentiation or apoptosis (9, 10). Such non-genomic effects are mediated through cytoplasmic signaling kinases, e.g., MAPK pathways related to PI3K or ERK signal transduction (10, 11). Moreover, recent insights into the role of osteoblast-formed HSC niche cues in modulating HSC development (12, 13) and leukemogenesis (13, 14) have gained attention on RA-mediated epigenetic regulation of terminal differentiation of myeloid leukemia cells.
It is known that RA-activated RARα regulates myeloid differentiation by transcriptional regulation of differentiation target gene (4, 6, 7). RARα, a phosphoprotein and transcription factor, is a substrate of CAK complex (6, 15). Human CAK is an enzyme consisting of cyclin-dependent kinase 7 (CDK7) (16), cyclin H (17), and MAT1 (18). Ser-77 in the ligand independent AF-1 domain of RARα (RARαS77) is the main residue phosphorylated by CAK (6, 15). CAK exists in cells either as free CAK or as part of the general transcription factor IIH (TFIIH) complex. Both free CAK and TFIIH-containing CAK phosphorylate Ser-77 of RARα (6, 15). Free CAK controls G1 exit, a stage in which cells commit to proliferation or to differentiation (19–21). We have found that either RA-decreased CAK phosphorylation of RARα or expression of the phosphorylation-defective RARαS77 mutant, RARαS77A, not only mediates granulocytic differentiation of both malignant and normal hematopoietic precursors (20–22), but also regulates osteoblastic differentiation and inhibits osteosarcoma formation (23). These studies demonstrate that RA-induced CAK-RARα signaling is involved in regulating differentiation of both osteoblastic and hematopoietic precursors.
Osteoblastic cells represent a regulatory component of the bone marrow microenvironment that mediate myelopoiesis, B-lymphocyte commitment, and HSC plasticity (12, 13, 24, 25), whereas disruption of this niche signal induces leukemogenesis (13, 26). Osteoblasts that are derived from human mesenchymal stem cells (MSC) constitute the HSC niche (13, 24), and RA induces osteoblastic differentiation of both MSC and osteosarcoma cells (23, 27). Striking success in epigenetic reversion of the genetic malignant phenotypes, as exemplified by RA treatment of APL harboring a PML-RARα fusion gene (2), provides proof of concept that RA-mediated HSC niche signaling can effect changes in the differentiation state of myeloid leukemia cells, even as the genome remains malignant and unstable (28). Previous studies strongly suggest the existence of a reciprocal relationship between osteoblasts and hematopoietic cells (12, 29), but the dimension of these interactions has yet to be defined. Because RA-induced loss of CAK phosphorylation of RARα or phosphorylation-defective RARαS77A mediates osteoblastic differentiation pathway through induction of FGF8f (23), we sought to investigate whether this osteoblast-derived FGF8f mediates granulocytic differentiation in a paracrine manner. Our studies show that osteoblast-secretion of FGF8f induced by either RA or RARαS77A regulates terminal granulocytic differentiation of myeloid leukemic cells, revealing a novel CAK-RARα signaling induced by RA to coordinate granulocytic differentiation at the paracrine level.
Materials and Methods
Cell Culture
Human myeloid leukemic HL60, HL60R (RA-resistant), NB4 (APL), and human osteosarcoma U2OS cells were cultured as described (20, 21, 23). Cells within 5 to 15 passages of HL60 and U2OS cell lines, expanded immediately after receiving the cells from the American Type Culture Collection (Manassas, VA), were used for less than 5 months. HL60R (20) and NB4 cells (21) were tested to be mycoplasma free by PCR methods after receiving cells from our collaborators, and each of those 5 to 15 passages of HL60R and NB4 cells was used for less than 5 months. The cancer cells were authenticated by their ability to form cancers in NOD/SCID and/or nude mice. Normal human primitive hematopoietic CD34+ cells were from AllCells (Emeryville, CA) and maintained with myeloid medium (MM) as described (22). The MM adapted for inducing granulopoiesis (MM-G) is supplemented with hydrocortisone for blocking the growth of lymphoid cells, while eliminates erythropoietin for prohibiting the growth of erythroid cells (22). CD34 cells, certified to be HIV and mycoplasma free by AllCells, were cultured for maximum 12 days without passaging after their initial expansion by following the manufacturer’s instructions. ATRA (RA) was from Sigma (St. Louis, MO). 1 μm of RA was used in the experiments. Recombinant human FGF8f was from R&D Systems (Minneapolis, MN).
Characterization of Nuclear Segmentation
Granulocytic differentiation, as judged by morphology nuclear segmentation, was described before (20). Briefly, cells were cytocentrifuged for 5-min at 400-rpm in a Cytospin, fixed by using methanol, and stained with Wright-Giemsa (Sigma). The morphological indicators of differentiation (nuclear/cytoplasmic ratio, nuclear shape, and degree of nuclear segmentation) were evaluated under a Zeiss Axioplan microscope. Images were color balanced in Adobe Photoshop.
Osteogenic differentiation
U2OS cells treated with RA or transduced with lentiviral pCCL-FGF8f or vector (Supplemental Figure 1) were grown in 24-well plates. After reaching 70–80% confluence, the cells were washed and cultured for 21 days with bone differentiation medium (culture medium supplemented with 10 nM dexamethasone [Sigma, # D2915], 20 mM β-glycerolphosphate [Sigma, # G9891], 50 μM L-Ascorbic acid 2-phosphate [Sigma, # A8960]). Cells were then fixed with 10% buffered formalin, and bone differentiation was judged by matrix mineralization as described (30) using Alizarin Red S (ARS; Sigma) staining.
Cell proliferation analysis
Cell duplication was determined by cell count as described previously (31).
Lentiviral transduction
Transduction of U2OS cells with lentiviral human RARαS77A or FGF8f (23) was described before (22).
Western Blotting
Western blotting (WB) was performed as described previously (20). Antibodies for FGF8, P-p38-MAPK (Thr 180/Tyr 182), P-p42/44-ERK (Thr 202/Tyr 204), p38-MAPK, p42/44-ERK, and β-actin were from Santa Cruz Biotechnology (Santa Cruz, CA).
Measurement of RA levels in the medium
The retained RA in the RA-OCM was monitored by using F9 reporter cells containing a lacZ reporter gene as described (32). X-gal was from Promega (San Luis Obispo, CA).
Quantitative real-time PCR (qRT-PCR)
cDNAs were generated from DNase I–treated RNA (iScript; Bio-Rad, Hercules, CA), and qRT-PCR was performed with QuantiTect SYBR Green PCR kits (Qiagen, Valencia, CA) by following the manufacturer’s instructions, using 384-well optical plates on the 7900HT Fast qRT-PCR System (Applied Biosystems). The housekeeping gene GAPDH was used as an internal control. Two negative controls were carried in parallel through all steps of the experiments. Standard curves for cDNA were composed of three 10-fold dilutions of control cDNA. The primers sequences and amplification conditions are given as supplemental Table 1.
Analysis of FGF8f Protein in the OCM
To assay secreted FGF8f, media conditioned by RA treatment of U2OS cells and FGF8f (or RARαS77A) overexpression in U2OS cells were harvested. These different OCM were concentrated using Pierce microconcentrators (Pierce Biotechnology, Inc. Rockford, IL) and the protein content measured by the improved Lowry assay using Bio-Rad (Hercules, CA) DC Protein Assay (33). The presence of FGF8f in the OCM was confirmed by WB analysis.
Neutralization of FGF8f
Antibody neutralization of FGF8f was described before (23). Blocking peptide against FGF8 antibody (sc-27144P, Santa Cruz Biotechnology) was used as described (23).
Flow cytometric analysis
Flow cytometry using a direct immunofluorescence staining was performed as described (20) for analysis of myeloid differentiation marker CD11b. CD11b-APC and CD34-PE antibodies were from BD Biosciences (San Jose, CA). Corresponding isotypes conjugated to irrelevant antibodies, isotype APC and isotype PE (BD Biosciences, San Jose, CA), were used as controls
Statistical analysis
Student’s unpaired 2-tailed t test was used when appropriate.
Results
Osteoblast-secreted FGF8f in the presence of RA regulates osteoblastic differentiation in an autocrine manner
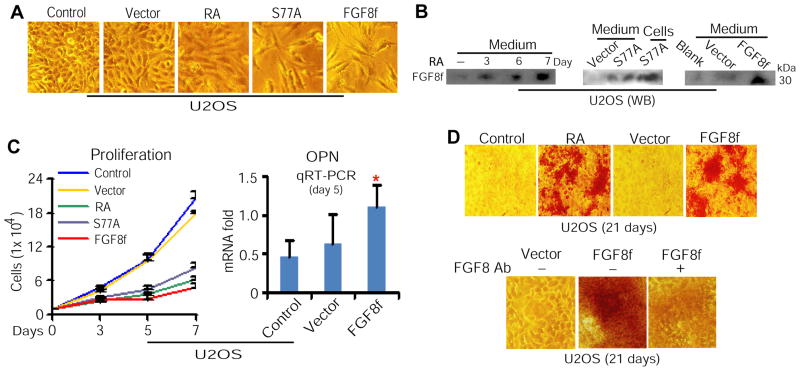
Because HSC niche formed by osteoblastic cells mediates HSC development (13, 24, 25) through secreting its regulatory signaling molecules (24, 25, 34), whereas RA stimuli or expression of a phosphorylation-defective RARαS77 mutant, RARαS77A, in osteosarcoma U2OS cells up-regulates FGF8f expression (23), we investigated whether such up-regulated FGF8f can be sufficiently secreted by U2OS cells and in turn directly mediates osteoblastic differentiation of U2OS cells. In a series of experiments using U2OS cells treated with RA or transduced with lentiviral RARαS77A or FGF8f (Supplementary Figure 1), we found that overexpression of RARαS77A or FGF8f mimicked the effect of RA or RARαS77A on inducing cellular morphological change (Figure 1A), up-regulating FGF8f secretion (Figure 1B), inhibiting proliferation, and enhancing expression of osteoblastic differentiation regulator osteopontin (OPN) (Figure 1C). Moreover, bone differentiation analysis demonstrated that similar to RA stimuli, overexpression of FGF8f induces bone differentiation (Figure 1D, top panel). Further to verify the effect of FGF8f on inducing bone differentiation, we chose to perform antibody neutralization of FGF8f. Because of the special genetic structure of FGF8f shared with its alternatively spliced products FGF8e, FGF8b, and FGF8a (23, 35), antibody specifically against FGF8f cannot be produced. However, in U2OS cells, FGF8f is the main isoform induced by RA or RARαS77A, and FGF8 antibodies only recognize a major 30-kDa protein, corresponding to the molecular mass of FGF8f (23). We therefore used FGF8 antibodies to neutralize the effect of FGF8f, as described (23), on inducing osteoblastic differentiation. We found that removal of FGF8f activities by FGF8 antibody neutralization significantly reduced bone matrix deposition of U2OS cells, as judged by ARS staining (Figure 1D, bottom panel). These data collectively show that RA-induced FGF8f is sufficiently secreted by U2OS cells, which possesses a direct regulatory effect on mediating osteoblastic differentiation at the autocrine level.
Figure 1. Osteoblast-secreted FGF8f in the presence of RA regulates osteoblastic differentiation in an autocrine manner.
(A) Morphology of U2OS cells treated with RA or transduced with lentiviral RARαS77A or FGF8f. (B) WB analysis of FGF8f in the conditioned medium collected from U2OS cells treated with RA (left) or transduced by RARαS77A (middle) or FGF8f (right). (C) Proliferation analysis by cell count (left) and qRT-PCR depiction of OPN (right). *P<0.05. (D) Bone differentiation induced by RA stimuli or FGF8f overexpression, as judged by ARS staining (top), and FGF8 antibody neutralization of FGF8f-induced bone differentiation (bottom). Ab, antibodies.
RA-mediated FGF8f secretion by differentiating U2OS cells inhibits proliferation and induces differentiation of different subtypes of leukemic myeloblasts at the paracrine level
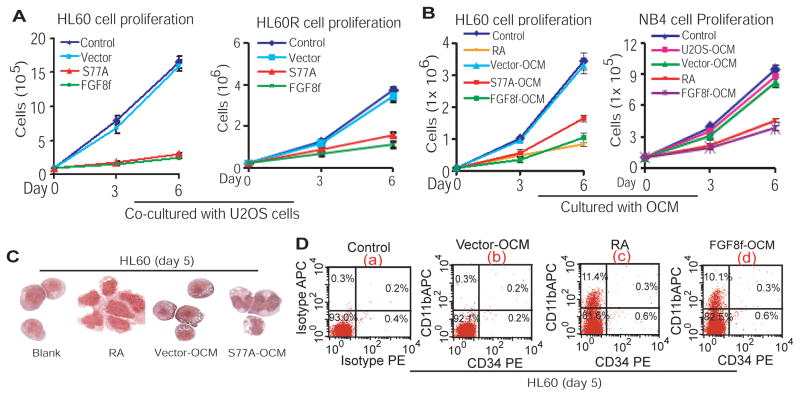
Osteoblast-formed HSC niche secretes signaling molecules to cross-mediate myelopoiesis (24, 25, 34), whereas FGF8f secreted by U2OS cells treated with RA or transduced with lentiviral FGF8f or RARαS77A (Figure 1) (23) is functionally active, as reflected by mediating osteoblastic differentiation in an autocrine manner (Figure 1D). We therefore tested whether such osteoblast-secreted FGF8f can function in the absence of RA at the paracrine level to inhibit proliferation of myeloid leukemia cells, an indispensable biological process required for induction of granulocytic differentiation. By co-culturing HL60 cells with U2OS cells overexpressing FGF8f or RARαS77A, we found that the proliferation of both RA-sensitive HL60 and RA-resistant HL60R cells was inhibited (Figure 2A). Since HL60R cells harbouring a truncated ligand-dependent AF-2 domain of RARα (36, 37), the results show that osteoblast-derived paracrine FGF8f can bypass the upstream defect occurring at the levels of RA:RARα interaction to inhibit proliferation of RA-resistant leukemic myeloblasts, similar to the effect of RA on inhibiting proliferation of RA-sensitive cells. Moreover, by culturing NB4 cells, which harbor a PML-RARα fusion gene (2), as well as HL60 cells in the presence of either RA or OCM collected from U2OS cells transduced with RARαS77A (S77A-OCM) or FGF8f (FGF8f-OCM), we found that cell proliferation in both HL60 and NB4 cells was inhibited (Figure 2B). These results suggest that in the absence of RA, osteoblast-secreted FGF8f, a downstream target of RA signaling, is capable of inhibiting proliferation of both RA-sensitive and RA-resistant leukemic myeloblasts at the paracrine level.
Figure 2. RA-mediated FGF8f secretion by differentiating U2OS cells inhibits proliferation and induces differentiation of different subtypes of leukemic myeloblasts at the paracrine level.
(A) Proliferation analysis of HL60 (left) and HL60R (right) cells co-cultured with U2OS cells expressing RARαS77A or FGF8f. (B) Proliferation analysis of HL60 (left) and NB4 (right) cells cultured with different OCM. (C) Granulocytic morphologic differentiation of HL60 cells. (D) Flow cytometric analysis of the expression of CD11b. The corresponding isotypes as well as CD34 antibodies were used as controls.
Because the effect of RA signaling is identified with the induction of granulocytic differentiation (1, 3) and because differentiation can only be induced from the cells where proliferation is arrested, we wondered whether RA-induced inhibitory effect mediated by paracrine FGF8f on cell proliferation (Figure 2A,B) leads to granulocytic differentiation of myeloid leukemic cells. To address this issue, HL60 cells were treated with RA or cultured with S77A-OCM or vector-OCM. Granulocytic morphology analysis indicates that FGF8f secreted into S77A-OCM (Figure 1B, middle panel) induced HL60 cell differentiation (Figure 2C). Correspondingly, the levels of CD11b, a myeloid differentiation marker expressed on myelocytes and more mature granulocytes, were induced by either RA or FGF8f-OCM in the absence of CD34+ expression, compared to blank or vector controls (Figure 2D, panels c, d vs. a, b). Hence, these data suggest that RA-mediated induction of paracrine FGF8f by osteoblasts instigates HSC niche signaling, in the absence of RA, to inhibit proliferation and induce differentiation of leukemic myeloblasts.
Paracrine FGF8f derived from osteoblastic niche cells is required for mediating granulocytic differentiation of myeloid leukemic cells
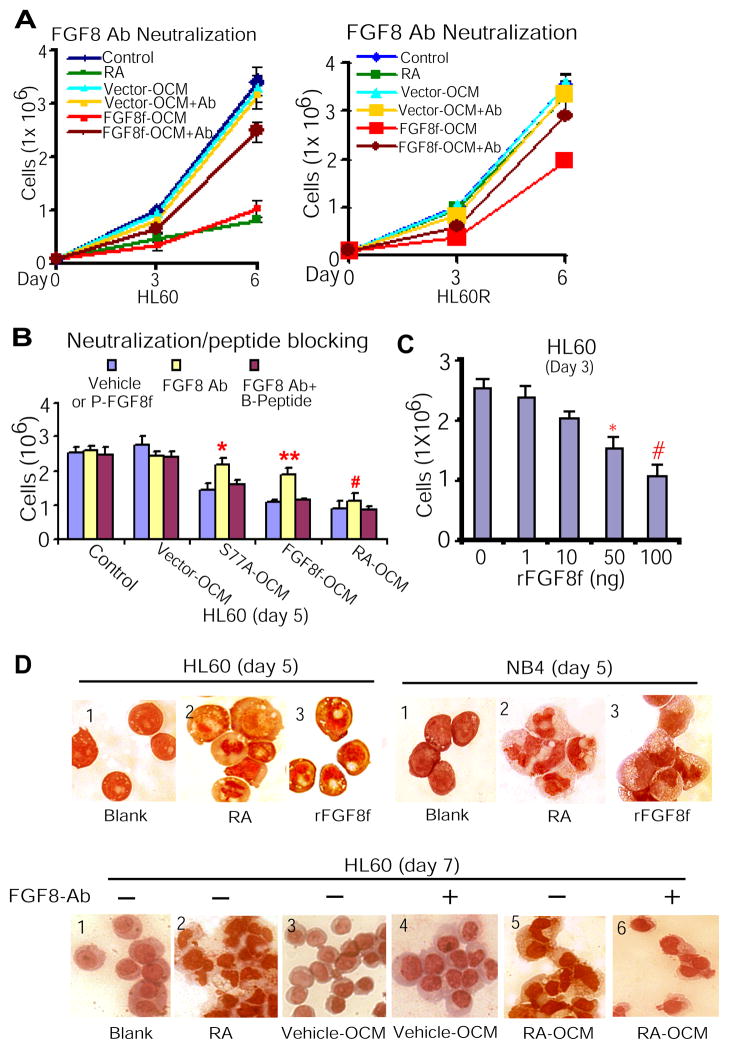
To verify the inhibitory effect of paracrine FGF8f on leukemic cell proliferation, we chose to use FGF8 antibody neutralization of paracrine FGF8f effect, as described above (Figure 1D). We cultured both HL60 and HL60R cells with FGF8f-OCM in the presence or absence of FGF8 antibodies as described (23). The results showed that whereas FGF8f-OCM inhibited cell proliferation in either HL60 or HL60R cells, this inhibitory effect of paracrine FGF8f was neutralized by FGF8 antibodies (Figure 3A). Moreover, since FGF8f is downstream target of RARαS77A (Figure 1B, middle panel) (23), we next verified whether the inhibitory effect of both RARαS77A- and FGF8f-OCM on cell proliferation is FGF8f-specific. We found that in the presence of negative (blank, vector) and positive (RA-OCM) controls, FGF8 antibodies neutralized the inhibitory effect of either RARαS77A-OCM or FGF8f-OCM containing FGF8f on cell proliferation, while the blocking peptides of FGF8 antibodies overrode such counteraction of FGF8 antibodies (Figure 3B). In parallel, the RA levels in RA-OCM were monitored for not to exceed physiological levels (10−9 M) by using F9 RA-reporter cells (Supplemental Figure 2). Hence, the above data show that OCM-containing paracrine FGF8f is required for inhibiting proliferation of both RA-sensitive and RA-resistant leukemic myeloblasts.
Figure 3. Paracrine FGF8f derived from osteoblastic niche cells is required for mediating granulocytic differentiation of myeloid leukemic cells.
(A) FGF8 antibodies (100 ng/ml) neutralized the inhibitory effect of FGF8f-OCM on HL60 (left) and HL60R cell proliferation (right). (B) The inhibitory effect of paracrine FGF8f in S77A-OCM or FGF8f-OCM or RA-OCM on HL60 cell proliferation (blue bar) was neutralized in the presence of FGF8f antibodies (yellow bar), whereas such neutralizing effect of FGF8f antibodies was reversed in the presence of blocking peptides (red bar). FGF8 Ab vs. FGF8 Ab + peptide: **P <0.002, *P<0.007, and #P<0.02. P-FGF8f, paracrine FGF8f that presents in S77A- or FGF8f- or RA-OCM; B-Peptide, blocking peptide for FGF8 antibodies. (C) HL60 cells treated with rFGF8f. P<0.0005; *P<0.002. (D) rFGF8f mimicked the effect of RA on inducing terminal granulocytic differentiation of HL60 and NB4 cells (top). The effect of RA-OCM containing FGF8f on inducing terminal granulocytic morphology differentiation was reversed by FGF8 antibodies (bottom).
To provide demonstration that the observed effects of paracrine FGF8f are not due to unknown cytokines co-secreted and present in the conditioned medium, we first cultured HL60 cells in the presence of human recombinant FGF8f (rFGF8f). The result showed that rFGF8f significantly inhibited HL60 cell proliferation in the absence of RA (Figure 3C). To examine that paracrine FGF8f specifically mimicked RA induction of granulocytic differentiation, we cultured HL60 and NB4 cells in the presence of RA or rFGF8f. The results showed that similar to RA stimuli, rFGF8f actually induces granulocytic morphology differentiation of HL60 and NB4 cells, an essential phenotype of terminal granulocytic differentiation (Figure 3D, top panel, sections 3 vs. 2, respectively). Furthermore, with the antibody neutralization approach to define that paracrine FGF8f in OCM is a key factor to induce granulocytic differentiation, HL60 cells were cultured with RA-OCM in parallel to different controls, including vector-OCM, RA stimuli, and blank. Distinctively, while FGF8 antibodies reversed the effect of RA-OCM containing FGF8f on inducing granulocytic morphologic differentiation of HL60 cells (Figure 3D, bottom panel, sections 5 vs. 6), vector-OCM with FGF8 antibodies showed no effect on cellular status, similar to blank and vector-OCM groups (Figure 3D, bottom panel, sections 4 vs. 1,3). Altogether, these data demonstrated that paracrine FGF8f secreted by osteoblastic niche cells specifically cross-regulates granulocytic differentiation of different subtypes of leukemic myeloblasts.
Paracrine FGF8f induces terminal granulocytic differentiation of CD34+ cells
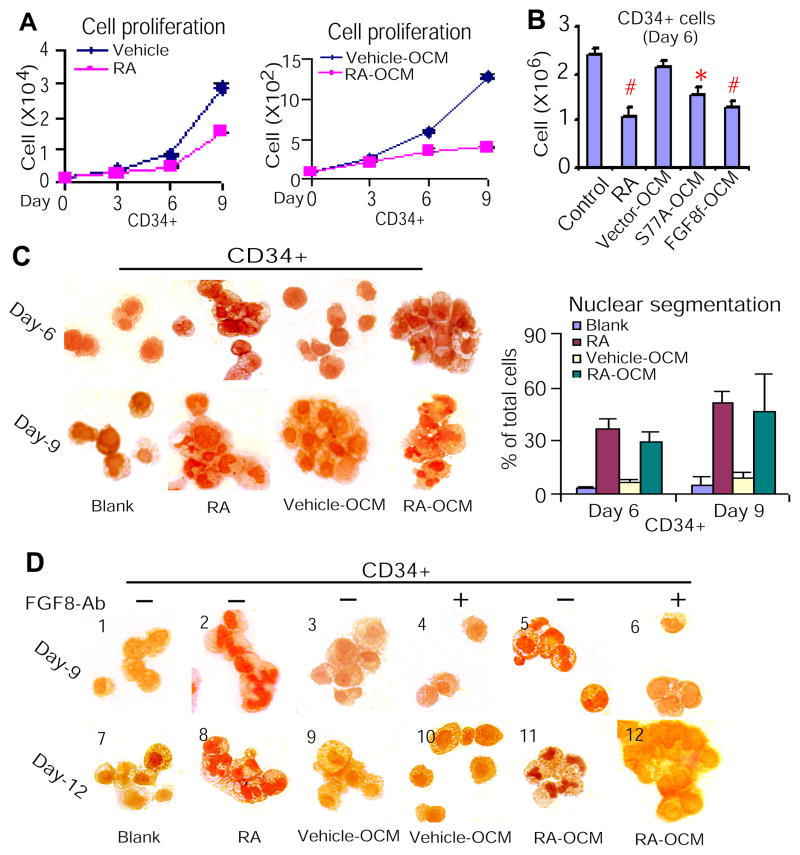
Hematopoietic development and granulopoiesis have been well characterized (38, 39). Although other groups and we have determined that RA mediates granulocytic differentiation of both normal and malignant hematopoietic precursors (1, 6, 20–22, 40), it remains unknown whether signaling instigated by RA from the HSC niche cross-regulates granulopoiesis. Given that paracrine FGF8f bypasses RA stimuli to induce terminal granulocytic differentiation of leukemic myeloblasts (Figures 2,3), and that many pathways associated with cancer normally regulate stem cell development (41), we investigated whether paracrine FGF8f has similar effect on mediating terminal granulocytic differentiation of normal primitive hematopoietic precursors. CD34+ cells were treated with RA or cultured with RA-OCM or S77A-OCM or FGF8f-OCM, in parallel to different controls. The results showed that both RA and OCM-containing FGF8f inhibited proliferation of CD34+ cells (Figure 4A,B). Since change in morphology is an essential phenotype for assessment of terminal granulocytic differentiation, we further to analyze granulocytic morphologic differentiation of CD34+ cells treated with RA or RA-OCM. The results showed that RA-OCM containing FGF8f mimics the effect of RA on inducing granulocytic differentiation (Figure 4C). To verify that the observed response in CD34+ cells is not due to unknown cytokines co-secreted in the RA-OCM, we treated CD34+ cells with RA-OCM in the presence and absence of FGF8 antibodies. The results demonstrated that while RA-OCM containing FGF8f mimics RA effect on inducing terminal granulocytic morphology differentiation of CD34+ cells (Figure 4D, sections 5,11 vs. 2,8), FGF8 antibodies spatiotemporally neutralized such effect of RA-OCM containing FGF8f (Figure 4D, sections 5,11 vs. 6,12). These data provide evidence that RA-mediated secretion of FGF8f by osteoblasts can bypasses RA stimuli to inhibit proliferation and induce granulocytic differentiation of normal hematopoietic precursors.
Figure 4. Paracrine FGF8f induces terminal granulocytic differentiation of CD34+ cells.
(A) Proliferation analysis of CD34+ cells treated with RA (left) or cultured with RA-OCM (right). (B) Proliferation analysis of CD34+ cells treated with RARαS77A- and FGF8f-OCM. P<0.001; *P<0.004. (C) Granulocytic morphologic differentiation of CD34+ cells treated with RA or RA-OCM at different time points (left). Quantification of granulocytic differentiation (right) of the left panel. (D) FGF8 antibodies neutralized the effect of RA-OCM containing FGF8f on inducing terminal granulocytic differentiation of CD34+ cells.
RA-mediated induction of FGF8f by osteoblasts modulates MAPK activities in both osteoblastic and hematopoietic precursors
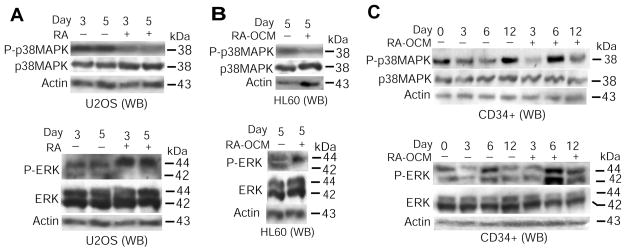
Above data show that osteoblast-secretion of FGF8f mediated by RA signaling coordinates osteoblastic differentiation (Figure 1) with granulocytic differentiation of hematopoietic precursors (Figures 2–4). These findings prompted us to ask a question: Which pathway is possibly involved in coupling FGF8f effect at both the autocrine and paracrine levels? Because FGF signaling is largely transduced through MAPK pathway (42) and because, in parallel to FGF8f induction, RA up-regulates expression of MAPK signaling molecules in osteoblasts (23), we investigated whether MAPK signaling lies downstream of RA-originated FGF8f regulation in those osteoblastic and hematopoietic precursors. WB analysis using specific antibodies against different phosphorylated MAP kinases showed that in FGF8f-mediated osteoblastic differentiation of U2OS cells (Figure 1), p38-MAPK phosphorylation was inhibited (Figure 5A, top panel) but p44-ERK phosphorylation was up-regulated in association with decreased p42-ERK phosphorylation (Figure 5A, bottom panel), in contrast to their even levels of total proteins. A similar dynamic pattern was also detected in HL60 cells cultured with RA-OCM, as shown by up-regulated p44-ERK phosphorylation (Figure 5B, bottom panel), accompanied with down-regulated phosphorylation of both p38-MAPK (Figure 5B, top panel) and p42-ERK (Figure 5B, bottom panel). Furthermore, in CD34+ cells cultured with RA-OCM, the phosphorylation of p42/44-ERK was periodically sustained (Figure 5C, bottom panel) while the p38-MAPK phosphorylation was inhibited in general (Figure 5C, top panel), during granulocytic differentiation induced by RA-OCM (Figure 4). Altogether, these results support the notion that likely through modulation of MAPK signaling at both the autocrine and paracrine levels, RA-induced FGF8f coordinates differentiation of osteoblastic and hematopoietic precursors.
Figure 5. RA-mediated induction of FGF8f by osteoblasts modulates MAPK activities in both osteoblastic and hematopoietic precursors.
(A) WB analysis of MAPK phosphorylation in U2OS cells using antibodies against phosphorylated p38-MAPK (P-p38MAPK) or phosphorylated p42/44-ERK (P-ERK). Antibodies against total p38-MAPK or ERK were used as additional controls. (B & C) WB analysis of MAPK phosphorylation in HL60 (B) and CD34+ cells (C), as described in panel A.
Discussion
Osteoblast-derived paracrine FGF8f mediates granulocytic differentiation of both malignant and normal hematopoietic precursors in an RA-independent manner
The difficulty of integrating signals instigated by the genetic programs of hematopoietic precursors with the developmental cues originated from osteoblast-constituted HSC niche, where myeloid leukemia cells appear to arise, is a major barrier to effectively manipulate RA therapy for diverse types of myeloid leukemia. Our studies demonstrate that RA-mediated FGF8f induction by osteoblasts coordinates osteoblastic maturation (Figure 1) with granulocytic differentiation of both leukemic myeloblasts and normal hematopoietic precursors (Figures 2–4). Moreover, such paracrine FGF8f is also capable of inhibiting proliferation of myeloid leukemic cells by overcoming RA-resistance (Figures 2A; 3A), which results from mutation deletion of ligand-dependent AF-2 domain of RARα (36, 37). This effect of paracrine FGF8f on cross-regulating granulocytic differentiation of hematopoietic precursors is likely through modulation of MAPK signaling (Figure 5) via FGF8f-mediated interaction with FGF receptor (FGF-R) (Figure 6). Hence, these findings provide evidence that in the absence of RA, the downstream regulatory target of RA signaling, HSC niche-derived FGF8f, is able to execute the RA-induced signaling effect on inducing granulocytic differentiation of both malignant and normal hematopoietic precursors (Figures 2–4). Success in identifying which FGF-R on hematopoietic precursors interacts with paracrine FGF8f is crucial to determine the mechanisms of HSC niche-mediated granulopoiesis through modulation of MAPK signaling.
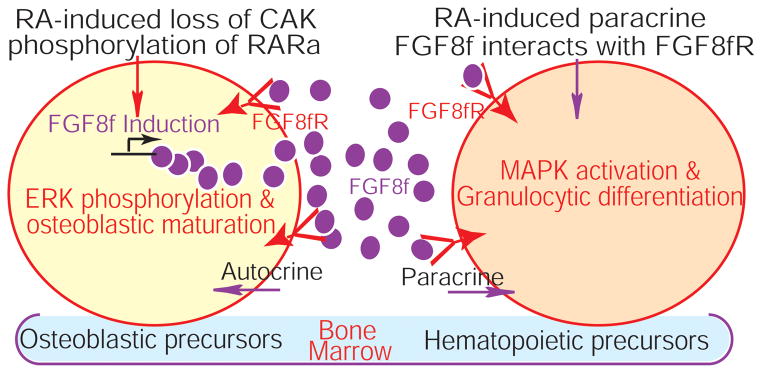
Figure 6.
RA-induced paracrine FGF8f from the osteoblastic niche cells bypasses RA stimuli to cross-regulates granulocytic differentiation of hematopoietic precursors. FGF8fR, FGF8f receptor.
RARα hypophosphorylation induces FGF8f to coordinate osteoblastic maturation with granulocytic differentiation
How does RA mediate osteoblast induction of FGF8f to coordinate the regulation of osteoblastic differentiation with granulocytic differentiation? The important role of RARα is identified with myeloid development (3) as well as its mutation-resistance to myeloid differentiation (36, 37). It has been demonstrated that RA-activated RARα regulates myeloid differentiation by transcription of differentiation target genes (1, 3, 4, 7), and that RA-suppressed CAK phosphorylation of RARα on RARαS77 of the AF-1 domain (6, 15) induces differentiation of both osteosarcoma cells (23) and leukemic myeloblasts (20, 21). Previous studies show that the human FGF8 promoter contains RARα binding sites, and that RA-mediated interaction of RARα with these sites induces CAT activity (43). We also demonstrated that RA-suppressed CAK phosphorylation of RARα remodels RARα-chromatin interaction to induce transcription of differentiation target genes (6), including FGF8f that is induced by RA stimuli or overexpression of RARαS77A (23). Moreover, whereas FGF-Rs are expressed on nearly every cell of hematopoietic origin, FGFs are basically produced by bone marrow stromal cells, and cells from some mature peripheral blood lineages (13, 44). These findings suggest that RA-mediated FGF8f induction by osteoblasts (Figure 1B) (23) cross-regulates granulocytic differentiation (Figures 2–4), likely through its interaction with specific FGF-R expressed ubiquitously on hematopoietic cells (13, 44) in the bone marrow (Figure 6). Hence, determining the mechanism of RA-mediated transactivation of FGF8f in the HSC niche as well as the interaction of FGF8f:FGF-R on hematopoietic precursors are crucial to establish the role of RA-instigated epigenetic regulation of granulocytic differentiation.
RA-mediated signaling interaction of FGF8f with MAPK
FGF signals are generally transduced through the MAPK pathways (42). Some FGFs can act at both the autocrine and paracrine levels to mediate the biological processes between blood and bone-forming cells by interacting with distinct FGF-Rs (44, 45). Data from embryonic stem cells show that rapid retinoid induction of FGF8 and downstream ERK activity ensures loss of self-renewal and initiates differentiation (46). Furthermore, recent studies indicate that RA activates RARα and induces granulocytic differentiation of leukemic myeloblasts through the MAPK pathway (47, 48). These findings indicate a mechanistic link of RA-mediated granulocytic differentiation among retinoid stimuli, RARα activation, FGF8 induction, and MAPK signaling. Our studies show that RA-decreased CAK phosphorylation of RARα induces FGF8f (23). The induced FGF8f (Figure 1B), in turn, mediates osteoblastic differentiation in an autocrine manner (Figure 1) and couples granulocytic differentiation of hematopoietic precursors at the paracrine level (Figures 2–4). Such bidirectional regulation of FGF8f is associated with a dynamic change in ERK phosphorylation in both osteoblastic and hematopoietic precursors (Figure 5). These findings suggest that RA coordinates CAK-RARα regulation with FGF8f-ERK signaling to couple osteoblastic maturation with granulocytic differentiation (Figure 6). It is therefore important, in future studies, to delineate the involvement of ERK signaling mediated by FGF8f-dependent epigenetic regulation, using combined loss-of-function strategies including antibody neutralization, shRNA silencing, and inhibition of ERK pathways. However, because FGF8f is also engaged in cross-talk with Wnt8B or WISP3 (23) through GSK3 or catenin signals (49) to activate the FGF-PI3K-AKT pathway in mediating granulocytic differentiation, the above mentioned loss-of-function approaches might encounter difficulty in modulating granulocytic differentiation through blocking FGF8f-ERK pathway. If so, it is necessary to alternatively test whether paracrine FGF8f mediates granulocytic differentiation through cross-talk with Wnt signaling to activate PI3K-AKT pathway, which has been implicated in the granulocytic differentiation of myeloid leukemia cells (50). Success in determining the mechanisms of this orchestration mediated by paracrine FGF8f should provide new insights into the niche-derived myeloid leukemogenesis.
In conclusion, our studies discover that FGF8f, a downstream targeting molecule induced by RA signaling in osteobastic niche cells, is capable of cross-mediating granulocytic differentiation of both malignant and normal hematopoietic precursors in the absence RA. These findings demonstrate that without RA stimuli, modulation of the downstream targeting molecule of RA signaling not only can efficiently implement the effect of RA on inducing granulocytic differentiation of myeloid leukemic cells, but also can bypass the upstream defect occurring at the levels of RA:RARα interaction in RA-resistant cells. These findings thereby present a compelling molecular rationale for developing effective differentiation therapies against different types of myeloid leukemia by targeting paracrine FGF8f and its potential cofactors.
Supplementary Material
Acknowledgments
This work was supported by grants from the National Institutes of Health (R01 CA120512 and 3R01CA120512-02S1 to L. Wu).
Footnotes
Conflict of interest: The authors declare no conflict of interest.
References
- 1.Evans T. Regulation of hematopoiesis by retinoid signaling. Exp Hematol. 2005;33:1055–61. doi: 10.1016/j.exphem.2005.06.007. [DOI] [PubMed] [Google Scholar]
- 2.Fenaux P, Chomienne C, Degos L. Acute promyelocytic leukemia: biology and treatment. Seminars in oncology. 1997;24:92–102. [PubMed] [Google Scholar]
- 3.Melnick A, Licht JD. Deconstructing a disease: RARalpha, its fusion partners, and their roles in the pathogenesis of acute promyelocytic leukemia. Blood. 1999;93:3167–215. [PubMed] [Google Scholar]
- 4.Chambon P. A decade of molecular biology of retinoic acid receptors. FASEB J. 1996;10:940–54. [PubMed] [Google Scholar]
- 5.Rochette-Egly C, Plassat JL, Taneja R, Chambon P. The AF-1 and AF-2 activating domains of retinoic acid receptor-alpha (RARalpha) and their phosphorylation are differentially involved in parietal endodermal differentiation of F9 cells and retinoid-induced expression of target genes. Mol Endocrinol. 2000;14:1398–410. doi: 10.1210/mend.14.9.0527. [DOI] [PubMed] [Google Scholar]
- 6.Wang A, Alimova IN, Luo P, Jong A, Triche TJ, Wu L. Loss of CAK phosphorylation of RAR{alpha} mediates transcriptional control of retinoid-induced cancer cell differentiation. FASEB J. 2010;24:833–43. doi: 10.1096/fj.09-142976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Soprano DR, Qin P, Soprano KJ. Retinoic acid receptors and cancers. Annu Rev Nutr. 2004;24:201–21. doi: 10.1146/annurev.nutr.24.012003.132407. [DOI] [PubMed] [Google Scholar]
- 8.Losel RM, Falkenstein E, Feuring M, Schultz A, Tillmann HC, Rossol-Haseroth K, et al. Nongenomic steroid action: controversies, questions, and answers. Physiol Rev. 2003;83:965–1016. doi: 10.1152/physrev.00003.2003. [DOI] [PubMed] [Google Scholar]
- 9.Liao YP, Ho SY, Liou JC. Non-genomic regulation of transmitter release by retinoic acid at developing motoneurons in Xenopus cell culture. J Cell Sci. 2004;15:2917–24. doi: 10.1242/jcs.01153. [DOI] [PubMed] [Google Scholar]
- 10.Zanotto-Filho A, Cammarota M, Gelain DP, Oliveira RB, Delgado-Canedo A, Dalmolin RJ, et al. Retinoic acid induces apoptosis by a non-classical mechanism of ERK1/2 activation. Toxicol In Vitro. 2008;22:1205–12. doi: 10.1016/j.tiv.2008.04.001. [DOI] [PubMed] [Google Scholar]
- 11.Masia S, Alvarez S, de Lera AR, Barettino D. Rapid, nongenomic actions of retinoic acid on phosphatidylinositol-3-kinase signaling pathway mediated by the retinoic acid receptor. Mol Endocrinol. 2007;21:2391–402. doi: 10.1210/me.2007-0062. [DOI] [PubMed] [Google Scholar]
- 12.Calvi LM, Adams GB, Weibrecht KW, Weber JM, Olson DP, Knight MC, et al. Osteoblastic cells regulate the haematopoietic stem cell niche. Nature. 2003;425:841–6. doi: 10.1038/nature02040. [DOI] [PubMed] [Google Scholar]
- 13.Li Z, Li L. Understanding hematopoietic stem-cell microenvironments. Trends Biochem Sci. 2006;31:589–95. doi: 10.1016/j.tibs.2006.08.001. [DOI] [PubMed] [Google Scholar]
- 14.Weaver VM, Gilbert P. Watch thy neighbor: cancer is a communal affair. Journal of cell science. 2004;117:1287–90. doi: 10.1242/jcs.01137. [DOI] [PubMed] [Google Scholar]
- 15.Rochette-Egly C, Adam S, Rossignol M, Egly JM, Chambon P. Stimulation of RAR alpha activation function AF-1 through binding to the general transcription factor TFIIH and phosphorylation by CDK7. Cell. 1997;90:97–107. doi: 10.1016/s0092-8674(00)80317-7. [DOI] [PubMed] [Google Scholar]
- 16.Wu L, Yee A, Liu L, Carbonaro-Hall D, Venkatesan N, Tolo VT, et al. Molecular cloning of the human CAK1 gene encoding a cyclin-dependent kinase-activating kinase. Oncogene. 1994;9:2089–96. [PubMed] [Google Scholar]
- 17.Fisher RP, Morgan DO. A novel cyclin associates with MO15/CDK7 to form the CDK-activating kinase. Cell. 1994;78:713–24. doi: 10.1016/0092-8674(94)90535-5. [DOI] [PubMed] [Google Scholar]
- 18.Tassan JP, Jaquenoud M, Fry AM, Frutiger S, Hughes GJ, Nigg EA. In vitro assembly of a functional human CDK7-cyclin H complex requires MAT1, a novel 36 kDa RING finger protein. The EMBO journal. 1995;14:5608–17. doi: 10.1002/j.1460-2075.1995.tb00248.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wu L, Chen P, Shum CH, Chen C, Barsky LW, Weinberg KI, et al. MAT1-modulated CAK activity regulates cell cycle G(1) exit. Molecular and cellular biology. 2001;21:260–70. doi: 10.1128/MCB.21.1.260-270.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wang J, Barsky LW, Shum CH, Jong A, Weinberg KI, Collins SJ, et al. Retinoid-induced G1 arrest and differentiation activation are associated with a switch to cyclin-dependent kinase-activating kinase hypophosphorylation of retinoic acid receptor alpha. The Journal of biological chemistry. 2002;277:43369–76. doi: 10.1074/jbc.M206792200. [DOI] [PubMed] [Google Scholar]
- 21.Wang JG, Barsky LW, Davicioni E, Weinberg KI, Triche TJ, Zhang XK, et al. Retinoic acid induces leukemia cell G1 arrest and transition into differentiation by inhibiting cyclin-dependent kinase-activating kinase binding and phosphorylation of PML/RARalpha. The FASEB journal. 2006;20:2142–4. doi: 10.1096/fj.06-5900fje. [DOI] [PubMed] [Google Scholar]
- 22.Luo P, Wang A, Payne KJ, Peng H, Wang JG, Parrish YK, et al. Intrinsic retinoic acid receptor alpha-cyclin-dependent kinase-activating kinase signaling involves coordination of the restricted proliferation and granulocytic differentiation of human hematopoietic stem cells. Stem Cells. 2007;25:2628–37. doi: 10.1634/stemcells.2007-0264. [DOI] [PubMed] [Google Scholar]
- 23.Luo P, Yang X, Ying M, Chaudhry P, Wang A, Shimada H, et al. Retinoid-suppressed phosphorylation of RARalpha mediates the differentiation pathway of osteosarcoma cells. Oncogene. 2010;29:2772–83. doi: 10.1038/onc.2010.50. [DOI] [PubMed] [Google Scholar]
- 24.Mayack SR, Wagers AJ. Osteolineage niche cells initiate hematopoietic stem cell mobilization. Blood. 2008;112:519–31. doi: 10.1182/blood-2008-01-133710. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 25.Zhu J, Garrett R, Jung Y, Zhang Y, Kim N, Wang J, et al. Osteoblasts support B-lymphocyte commitment and differentiation from hematopoietic stem cells. Blood. 2007;109:3706–12. doi: 10.1182/blood-2006-08-041384. [DOI] [PubMed] [Google Scholar]
- 26.Zhang J, Grindley JC, Yin T, Jayasinghe S, He XC, Ross JT, et al. PTEN maintains haematopoietic stem cells and acts in lineage choice and leukaemia prevention. Nature. 2006;441:518–22. doi: 10.1038/nature04747. [DOI] [PubMed] [Google Scholar]
- 27.Orimo H, Shimada T. Regulation of the human tissue-nonspecific alkaline phosphatase gene expression by all-trans-retinoic acid in SaOS-2 osteosarcoma cell line. Bone. 2005;36:866–76. doi: 10.1016/j.bone.2005.02.010. [DOI] [PubMed] [Google Scholar]
- 28.Kenny PA, Bissell MJ. Tumor reversion: correction of malignant behavior by microenvironmental cues. Int J Cancer. 2003;107:688–95. doi: 10.1002/ijc.11491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zhang J, Niu C, Ye L, Huang H, He X, Tong WG, et al. Identification of the haematopoietic stem cell niche and control of the niche size. Nature. 2003;425:836–41. doi: 10.1038/nature02041. [DOI] [PubMed] [Google Scholar]
- 30.Zhang W, Deng ZL, Chen L, Zuo GW, Luo Q, Shi Q, et al. Retinoic acids potentiate BMP9-induced osteogenic differentiation of mesenchymal progenitor cells. PLoS One. 2010;5:e11917. doi: 10.1371/journal.pone.0011917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wu L, Chen P, Hwang JJ, Barsky LW, Weinberg KI, Jong A, et al. RNA antisense abrogation of MAT1 induces G1 phase arrest and triggers apoptosis in aortic smooth muscle cells. The Journal of biological chemistry. 1999;274:5564–72. doi: 10.1074/jbc.274.9.5564. [DOI] [PubMed] [Google Scholar]
- 32.Wagner MA. Use of reporter cells to study endogenous retinoid sources in embryonic tissues. Methods Enzymol. 1997;282:98–107. doi: 10.1016/s0076-6879(97)82099-x. [DOI] [PubMed] [Google Scholar]
- 33.Gnanapragasam VJ, Robson CN, Neal DE, Leung HY. Regulation of FGF8 expression by the androgen receptor in human prostate cancer. Oncogene. 2002;21:5069–80. doi: 10.1038/sj.onc.1205663. [DOI] [PubMed] [Google Scholar]
- 34.Taichman RS, Emerson SG. Human osteoblasts support hematopoiesis through the production of granulocyte colony-stimulating factor. The Journal of experimental medicine. 1994;179:1677–82. doi: 10.1084/jem.179.5.1677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Gemel J, Gorry M, Ehrlich GD, MacArthur CA. Structure and sequence of human FGF8. Genomics. 1996;35:253–7. doi: 10.1006/geno.1996.0349. [DOI] [PubMed] [Google Scholar]
- 36.Collins SJ, Robertson KA, Mueller L. Retinoic acid-induced granulocytic differentiation of HL-60 myeloid leukemia cells is mediated directly through the retinoic acid receptor (RAR-alpha) Mol Cell Biol. 1990;10:2154–63. doi: 10.1128/mcb.10.5.2154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Robertson KA, Emami B, Collins SJ. Retinoic acid-resistant HL-60R cells harbor a point mutation in the retinoic acid receptor ligand-binding domain that confers dominant negative activity. Blood. 1992;80:1885–9. [PubMed] [Google Scholar]
- 38.Xiao M, Dooley DC. Cellular and molecular aspects of human CD34+ CD38− precursors: analysis of a primitive hematopoietic population. Leuk Lymphoma. 2000;38:489–97. doi: 10.3109/10428190009059267. [DOI] [PubMed] [Google Scholar]
- 39.Hoang T. The origin of hematopoietic cell type diversity. Oncogene. 2004;23:7188–98. doi: 10.1038/sj.onc.1207937. [DOI] [PubMed] [Google Scholar]
- 40.Collins SJ. The role of retinoids and retinoic acid receptors in normal hematopoiesis. Leukemia. 2002;16:1896–905. doi: 10.1038/sj.leu.2402718. [DOI] [PubMed] [Google Scholar]
- 41.Reya T, Morrison SJ, Clarke MF, Weissman IL. Stem cells, cancer, and cancer stem cells. Nature. 2001;414:105–11. doi: 10.1038/35102167. [DOI] [PubMed] [Google Scholar]
- 42.Jackson RA, Nurcombe V, Cool SM. Coordinated fibroblast growth factor and heparan sulfate regulation of osteogenesis. Gene. 2006;379:79–91. doi: 10.1016/j.gene.2006.04.028. [DOI] [PubMed] [Google Scholar]
- 43.Brondani V, Klimkait T, Egly JM, Hamy F. Promoter of FGF8 reveals a unique regulation by unliganded RARalpha. J Mol Biol. 2002;319:715–28. doi: 10.1016/S0022-2836(02)00376-5. [DOI] [PubMed] [Google Scholar]
- 44.Allouche M. Basic fibroblast growth factor and hematopoiesis. Leukemia. 1995;9:937–42. [PubMed] [Google Scholar]
- 45.Bikfalvi A, Han ZC. Angiogenic factors are hematopoietic growth factors and vice versa. Leukemia. 1994;8:523–9. [PubMed] [Google Scholar]
- 46.Stavridis MP, Collins BJ, Storey KG. Retinoic acid orchestrates fibroblast growth factor signaling to drive embryonic stem cell differentiation. Development. 2010;137:881–90. doi: 10.1242/dev.043117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Wang J, Yen A. A MAPK-positive feedback mechanism for BLR1 signaling propels retinoic acid-triggered differentiation and cell cycle arrest. J Biol Chem. 2008;283:4375–86. doi: 10.1074/jbc.M708471200. [DOI] [PubMed] [Google Scholar]
- 48.Glasow A, Prodromou N, Xu K, von Lindern M, Zelent A. Retinoids and myelomonocytic growth factors cooperatively activate RARA and induce human myeloid leukemia cell differentiation via MAP kinase pathways. Blood. 2005;105:341–9. doi: 10.1182/blood-2004-03-1074. [DOI] [PubMed] [Google Scholar]
- 49.Katoh M. Cross-talk of WNT and FGF signaling pathways at GSK3beta to regulate beta-catenin and SNAIL signaling cascades. Cancer Biol Ther. 2006;5:1059–64. doi: 10.4161/cbt.5.9.3151. [DOI] [PubMed] [Google Scholar]
- 50.Scholl S, Bondeva T, Liu Y, Clement JH, Hoffken K, Wetzker R. Additive effects of PI3-kinase and MAPK activities on NB4 cell granulocyte differentiation: potential role of phosphatidylinositol 3-kinase gamma. J Cancer Res Clin Oncol. 2008;134:861–72. doi: 10.1007/s00432-008-0356-8. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.