Abstract
Objective: The aim of this study was to evaluate associations between pregnancy outcomes and prepregnancy body mass index and gestational weight gain among pregnant women who regularly attended health centers of Kazerun, Fars, Iran.
Methods: In this descriptive study records from 5172 pregnant women were considered in this study, based on the methodology criteria. Women were distributed across 4 prepregnancy categories according to the Institute of Medicine (IOM) (1990) classification of body mass index, and to 4 end-of-pregnancy categories according to median weekly gestational weight gain. Results: The risks for gestational diabetes, gestational hypertension, pre-eclampsia, and preterm premature rupture of membranes were higher for those who were overweight or obese before becoming pregnant (P < 0.05). Moreover, a gestational weight gain of 0.50 kg per week or greater was associated with a higher risk for gestational hypertension, preterm premature rupture of membranes, and fetal macrosomia (P < 0.05). Women in the highest quartile for weight gain (≥ 0.59 kg per week) were at higher risk for pre-eclampsia (P < 0.05).
Discussion: The results seems to indicate that excessive gestational weight gain and high prepregnancy body mass index were associated with increased risks for adverse pregnancy outcomes.
Keywords: Pregnancy, Gestational weight gain, Obesity, Body mass index
Introduction
Maternal weight gain is positively and significantly associated with birth weight and exerts a direct influence on pregnancy outcome. The previous studies suggest that maternal age, pre-pregnancy body mass, parity, race, ethnicity, hypertension and diabetes also influence the pattern of weight gain (1) and (2).
The prevalence of obesity is increasing steadily among women of reproductive age. In the United States, 25% of women aged 20 to 39 years are overweight and 29% are obese (3). In a study conducted from 1989 through 1997 in the United Kingdom, the respective prevalence rates of overweight and obesity in the adult female population were 27.5% and 10.9% (4). In 2002, the rates of overweight and obesity were 38% and 11%, respectively, among women in Sweden (5). Overweight or obesity are associated with increased risks of maternal and neonatal complications, such as gestational hypertension, pre-eclampsia, gestational diabetes, premature rupture of membranes, prematurity, macrosomia, and shoulder dystocia. However, there is a paucity of information about the maternal and fetal effects of excessive prepregnancy body mass index (BMI) or of excessive gestational weight gain in low-resource countries. The first objective of the present study was to determine the rates of prepregnancy overweight and obesity and assess gestational weight gain among pregnant women who regularly attended health care centers of Kazerun, Fars, Iran. The second objective was to look for associations between pregnancy outcomes and different categories of both prepregnancy BMI (calculated as weight in kilograms divided by height in meters squared) and gestational weight gain among the same population.
Materials and methods
A total of 5928 women were considered for this retrospective cohort study conducted from January 2007 through January 2010 who had received prenatal care in the whole urban prenatal care centers of Kazerun, south of Iran, (six health institutions). They were all delivered in Valiasr hospital in Kazerun that is alone hospital in this city. Exclusion criteria were incomplete prenatal care information (n = 294); no routine glycemic screening for gestational diabetes mellitus (GDM) (n = 247); multiple gestation (n = 54); a history of hypertension, diabetes, heart disease, hepatitis, chronic renal disease, or other systemic disease (n = 149); pregnancy termination for fetal malformations (n = 12). The final sample consisted of 5172 women.
Prepregnancy weight was self-reported and recorded at our institutions during the first prenatal care visit, usually between the 8th and 12th gestational weeks, and height and weight were measured on hospital admission for delivery. Prepregnancy BMI was categorized according to the BMI classification proposed by the Institute of Medicine (IOM) (1990), American Academy of Pediatrics and American College of Obstetricians and Gynecologists (2002). The 4 BMI categories were underweight (< 19.8), normal weight (19.8-26), overweight (26-29), and obesity (> 29). We used the median weekly weight gain during pregnancy as an indicator of gestational weight gain (6). It was calculated as the total weight gain during pregnancy divided by the number of gestational weeks, and categorized according to quartile distribution (< 0.41 kg; 0.41-0.49 kg; 0.50-0.58 kg; and ≥ 0.59 kg per week).
Trained female investigators administered questionnaires inquiring about the following: age, parity, education level (< 12 years, 12 years, > 12 years), and employment (relatively stable jobs were categorized as “yes” and no stable job or housework at home was classified as “no”); family history of diabetes or hypertension; and height, prepregnancy weight, weight before delivery, and mode of delivery. Obstetric outcomes included the following: preterm delivery (< 37 weeks of gestation); preterm premature rupture of membranes (PPROM, i.e., rupture of membranes before 37 weeks); hypertensive disorders of pregnancy (i.e., gestational hypertension and pre-eclampsia); and GDM or gestational impaired glucose tolerance (GIGT), which was defined according to the clinical diagnosis and treatment guidelines of pregnancy with diabetes recommended by health of ministry in Iran. Neonatal outcomes included gestational age at delivery, birth weight, Apgar scores at 1 minute, and admission to the neonatal intensive care unit. Macrosomia was defined as a birth weight greater than 4000 g. The questionnaires were completed by accessing prenatal care and medical records during the period of hospitalization for delivery, and by more interviews with the study participants.
All participants gave written informed consent. The survey results were kept confidential.
Statistical analysis was performed using SPSS software, version 13.0 (SPSS, Chicago, IL, USA). A descriptive analysis assessed the distribution of all studied variables. Statistical significance was calculated using the χ2 test for differences between qualitative variables and analysis of variance for differences between continuous variables. Multiple logistic regression was performed to compare the effects of prepregnancy BMI and gestational weight gain on pregnancy outcomes. The risks of maternal and neonatal complications are presented as odds ratios (ORs) with their 95% confidence intervals (CIs) after adjusting for possible confounding variables. P < 0.05 was considered significant.
Results
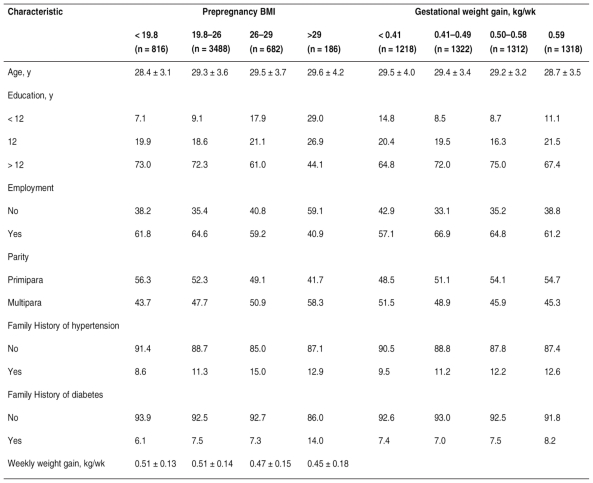
Maternal characteristics by prepregnancy BMI and gestational weight gain are shown in Table 1. The median age of the 5172 participants at delivery was 29 years (range, 16-46 years); 4766 (92.2%) of the participants were primiparas and 406(7.8%) were multiparas. According to prepregnancy BMI classification, 816 participants (15.8%) were underweight, 3488 (67.4%) were of normal weight, 682 (13.2%) were overweight, and 186 (3.6%) were obese. The median weekly weight gain was 0.50 kg per week (range, 0.04-1.35 kg per week). Prepregnancy BMI and age at delivery were positively correlated, (F = 9.65, P < 0.001). The proportion of overweight or obesity was lower among women with the highest level of education (> 12 years) than those with the lowest (< 12 years) (χ2 = 73.42, P < 0.001).
Table 1.
- Maternal characteristics by prepregnancy BMI and gestational weight gaina.
Abbreviations: BMI, body mass index, calculated as weight in kilograms divided by height in meters squared.
a Values are given as mean ± SD or percentage.
The prevalence of overweight and obesity was higher for unemployed women (χ2 = 23.59, P < 0.001) and for those who were multiparas (χ2 = 28.12, P < 0.001). The mean age at delivery was lower for the group with the greatest gestational weight gain than for the other 3 groups (F = 6.33, P < 0.001). Gestational weight gain was less among women with less than 12 years of education (χ2 = 26.03), among those who were unemployed (χ2 = 14.89), and among multiparas (χ2 = 21.44) (P < 0.001). As prepregnancy BMI increased, the amount of weekly weight gain decreased (F = 15.27, P < 0.001).
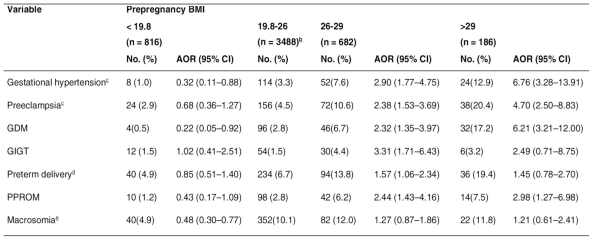
Table 2 shows the rates of adverse pregnancy outcomes in the different prepregnancy BMI groups as well as the adjusted ORs associated with each outcome in each BMI group. The reference group was the group of women of normal weight (19.8-26). Regarding maternal and neonatal complications for different categories of weekly weight gain, the adjusted ORs were calculated taking the group with the lowest weekly weight gain as the reference group (< 0.41 kg) (Table 3). All ORs were mutually adjusted by prepregnancy BMI and gestational weight gain, and additionally adjusted for maternal age, education level, occupation, family history of hypertension, family history of diabetes, and parity. The ORs for hypertensive disorder of pregnancy were also adjusted for gestational diabetes, and those for preterm delivery were also adjusted for gestational hypertension, pre-eclampsia, and GDM. The ORs for macrosomia were also adjusted for height and GDM. The incidence rates of maternal and neonatal complications increased with increasing prepregnancy BMI. Compared with the women with normal prepregnancy weight, those who were overweight before pregnancy had a significantly increased risk of gestational hypertension, pre-eclampsia, GDM, GIGT, and PPROM. Compared with the women who had a normal prepregnancy BMI, those who were obese before becoming pregnant were significantly more likely to have gestational hypertension, pre-eclampsia, GDM, and PPROM. Being underweight before pregnancy was protective against gestational hypertension, GDM, and fetal macrosomia. For women who were overweight or obese before pregnancy, the crude ORs for preterm delivery were 2.22 (95% CI, 1.55-3.19) and 3.33 (95% CI, 1.93-5.77), respectively. But after adjusting for potential confounding factors, especially after adjusting for hypertensive disorder of pregnancy, the ORs for preterm delivery were significantly less in the groups with a prepregnancy BMI greater than normal (Table 2).
Table 2.
- Adjusted odds ratios for associations between prepregnancy BMI and pregnancy outcome.a.
Abbreviations: AOR, adjusted odds ratio; BMI, body mass index; CI, confidence interval; GDM, gestational diabetes mellitus; GIGT, gestational impaired glucose tolerance; PPROM, preterm premature rupture of membranes.
a Adjusted for gestational weight gain, maternal age, education level, occupation, family history of hypertension, family history of diabetes, and parity.
b Reference group.
c Also adjusted for gestational diabetes.
d Also adjusted for gestational hypertension, preeclampsia. and gestational diabetes.
e Also adjusted for height and GDM.
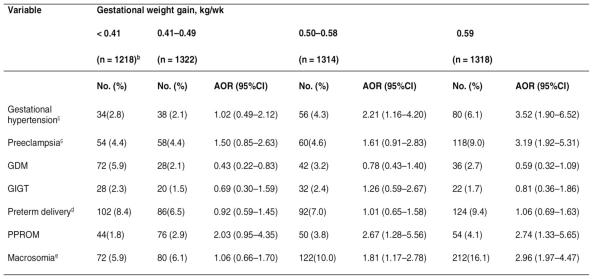
Table 3.
- Maternal characteristics by prepregnancy BMI and gestational weight gaina.
a Adjusted for prepregnancy BMI, maternal age, education level, occupation, family history of hypertension, family history of diabetes, and parity. For abbreviations and notes b, c , d, e, see Table 2.
Compared with the group with the lowest gestational weight gain (< 0.41 kg per week), the women whose gestational weight gain was in the third quartile (0.50-0.58 kg per week) were at increased risks for gestational hypertension, PPROM, and fetal macrosomia, and those with the greatest weight gain (≥ 0.59 kg per week) were at increased risks for gestational hypertension, pre-eclampsia, PPROM, and fetal macrosomia. Gestational diabetes was less likely to develop in women whose gestational weight gain was in the second quartile (0.41-0.49 kg per week) (Table 3).
Discussion
Maternal obesity, and its adverse maternal and neonatal effects, is common in the United States (7, 8). In our study, 13.2% of the women were overweight, 3.6% were obese, and 15.8% were underweight before they became pregnant. The first 2 rates are much lower and the third is higher than the corresponding rates reported for high-income countries (3-5).
The new guidelines the Institute of Medicine (IOM) recently published for gestational weight gain differ from those issued in 1990 in 2 ways. First, the guidelines are now based on World Health Organization (WHO) BMI categories. Second, they recommend a specific, relatively narrow range of weight gain for obese women (9). However, neither the WHO BMI categories nor the IOM recommendations about gestational weight gain are optimal for Iranian women. Considering the lack of clear guidelines about weight gain during pregnancy in Iran, we used the median weekly weight gain during pregnancy as an indicator of gestational weight gain, thus circumventing the effect of a shorter pregnancy in cases of preterm births.
As other authors (10-15), we found the risks of gestational hypertension and pre-eclampsia to be higher for women who are overweight or obese before pregnancy. Fortner et al. (12) reported that, compared with women who gained weight within the 1990 IOM guidelines, women from Latin America who gained excessive gestational weight had an almost 3-fold risk of hypertensive disorders of pregnancy and a 4-fold risk of pre-eclampsia. Our findings also indicate that hypertensive disorders of pregnancy are more likely to develop in women who gain excessive weight during pregnancy. Those with the highest weight gain had a 3-fold risk of pre-eclampsia in our study.
We found that the risk of GDM was increased for overweight and obese women, which is consistent with similar studies (10,14-17). Herring et al. (18) reported that a greater gestational weight gain prior to glycemic screening predicted GIGT, but not GDM. Since adiposity-generated insulin resistance promotes GDM, we speculated that an excessive weight gain during pregnancy would increase the risk of GDM. However, excessive gestational weight gain did not increase the risks of GDM or GIGT in our study. It is now recommended that women be screened for GDM between the 24th and 28th weeks of pregnancy, and pregnant women are advised to restrict their energy intake and perform moderate exercise if they are diagnosed as having GDM or GIGT. But a lesser weight gain over the course of the pregnancy, and a slower weight gain during the third trimester, resulted in the false conclusion that a lesser gestational weight gain was associated with a higher risk of GDM. Having considered the effect of different factors such as race, heredity, diet, height, age, smoking, alcohol consumption, psychosocial status, nutritional counseling, and the prenatal care more research on the causes of these results seems necessary.
Before adjustments were made for confounding factors in our study, preterm deliveries were associated with overweight or obesity and the ORs were 2.22 and 3.33, respectively. After adjusting for gestational hypertension and pre-eclampsia, the effect of prepregnancy BMI on preterm delivery was significantly weaker, however. This finding suggests that, for an obese woman, hypertensive disorders of pregnancy and an ensuing iatrogenic delivery are the major contributing factors to a preterm delivery. Dietz et al. (19) found that women with a very high gestational weight gain (> 0.79 kg per week) had approximately twice the odds of experiencing a very preterm delivery (between 20 and 31 weeks), regardless of their prepregnancy BMI. No relationship between gestational weight gain and preterm delivery was found in our study. The interesting thing is the difference between developed and developing Country, such as Iran, in the risk of pregnancy adverse outcomes correlated to prepregnancy BMI and weight gain. The researcher feels the need for more studies on cases in which no relationship was seen, whereas a research concerning more samples may indicate different results.
Few data exist regarding a possible relationship between obesity and PPROM. In our study, as in a study by Nohr et al. (20), being overweight or obese before pregnancy, or gaining excessive weight during pregnancy, increased the risk of PPROM. Obese women are also prone to infections of the genitourinary tract (4), and during pregnancy the proteases, collagenases, and elastases produced by bacteria can degrade the matrix and collagen of fetal membrane cells, and lead to membrane rupture. Additionally, obesity is associated with low-grade inflammation and slightly elevated levels of cytokines such as IL-6, IL-8, and TNF-α in body fluids and tissues. These proinflammatory cytokines could harm fetal membranes in pregnant women (21), and lead to PPROM by activation of lysosomal enzymes.
Most studies on the effect of prepregnancy obesity have suggested an association between obesity and an increased risk of macrosomia (14) and (22). In a report by Kabali et al. (23), the risk of fetal macrosomia was significantly elevated for women who were overweight before pregnancy and for those who gained excessive gestatinal weight. The risk was not increased, however, for women of normal weight before pregnancy who gained excessive gestational weight, or for those who were overweight before pregnancy but gained a normal or low gestational weight. Our findings indicate that, in this Iranian population, gestational weight gain may exert a more significant influence on the risk of macrosomia than prepregnancy BMI. The risks of macrosomia were increased almost 2and 3-fold, respectively, for women who gained 0.50 kg per week or more during pregnancy, and for those whose weekly weight gain was greater than 0.59 kg per week.
Generally speaking, continue to ask critical questions about weight gain and pregnancy to ensure that we are providing the best guidance and care for pregnant women. We need to address the criticisms that have been leveled at the clinical use of weight-gain recommendations, and weight monitoring during pregnancy.
Given the sensitivity of iranian women to weight and bodyimage issues, we need to discover and validate experimentally effective and thoughtful interventions to support women’s nutritional and other needs during pregnancy. Consequently, health care providers should have access to easy-to-use instruments for setting desirable weight gain goals for each individual mother and for monitoring weight gain during the course of pregnancy and must have an educational program in order to explain the risk of an excessive pregnancy weight gain for the pregnancy and the neonatal outcomes.Therefore, the investigation of their practice is very important and is a way to improve the mother and child’s health.
This study has some limitations,It did not have enough statistical power to detect relatively rare adverse outcomes, such as intrauterine fetal death and neonatal death. Moreover, because it is impossible to have every woman’s weight objectively measured just before she becomes pregnant, we relied on self-reported prepregnancy weight to calculate prepregnancy BMI and gestational weight gain. Thus, it is likely that prepregnancy BMI was underestimated and gestational weight gain overestimated, which may be one of the reasons why our population had a high proportion of women who were underweight before they became pregnant. However, several recent studies have reported an overall correlation coefficient of 0.99 between self-reported and measured prepregnancy weight, and concluded that self-reported weight reflected actual weight (24). A large number of studies have shown that obesity increased the risk of undergoing a cesarean or instrumental delivery (4, 10, 14). The reason why we did not look for relations between delivery mode and prepregnancy obesity or excessive weight gain was that the rate of selective cesarean delivery was high in our population for quite other reasons.
References
- 1.Coulfield L, Witter F, Stolzhis R. Determinants of gestational weight gain outside the recommended ranges among black and white women. Obstet Gynecol. 1996;87:760–6. doi: 10.1016/0029-7844(96)00023-3. [DOI] [PubMed] [Google Scholar]
- 2.Hickey C, Cliver S, Goldenberg R, McNeal S, Hoffman H. Relationship of psychosocial status to low prenatal weight gain among non obese black and white women delivering at term. Obstet Gyneco. 1995;86:177–83. doi: 10.1016/0029-7844(95)00161-j. [DOI] [PubMed] [Google Scholar]
- 3.Hedley A.A, Ogden C.L, Johnson C.L, Carroll M.D., Curtin L.R., Flegal K.M. Prevalence of overweight and obesity among US children, adolescents, and adults 1999-2002. JAMA. 2004;291(23):2847–2850. doi: 10.1001/jama.291.23.2847. [DOI] [PubMed] [Google Scholar]
- 4.Sebire N.J, Jolly M, Harris J.P, Wadsworth J, Joffe M., Beard R.W., et al. Maternal obesity and pregnancy outcome: a study of 287, 213 pregnancies in London. Int J Obes Relat Metab Disord. 2001;25(8):1175–1182. doi: 10.1038/sj.ijo.0801670. [DOI] [PubMed] [Google Scholar]
- 5.Berg C, Rosengren A, Aires N., Lappas G, Torén K., Thelle D., et al. Trends in overweight and obesity from 1985 to 2002 in Göteborg, West Sweden. Int J Obes (Lond) 2005;29(8):916–924. doi: 10.1038/sj.ijo.0802964. [DOI] [PubMed] [Google Scholar]
- 6.Chen A., Feresu S.A, Fernandez C., Rogan W.J. Maternal obesity and the risk of infant death in the United States. Epidemiology. 2009;20(1):74–81. doi: 10.1097/EDE.0b013e3181878645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Chu S.Y, Kim S.Y, Bish C.L. Prepregnancy obesity prevalence in the United States 2004-2005. Matern Child Health J. 2009;13(5):614–620. doi: 10.1007/s10995-008-0388-3. [DOI] [PubMed] [Google Scholar]
- 8.American College of Obstetricians and Gynecologists , ACOG Committee Opinion number 315, September 2005. Obesity in pregnancy. Obstet Gynecol. 2005;106(3):671–675. doi: 10.1097/00006250-200509000-00054. [DOI] [PubMed] [Google Scholar]
- 9.Institute of Medicine, Weight Gain During Pregnancy. Reexamining the Guidelines; The National Academies Press; Washington DC. [PubMed] [Google Scholar]
- 10.Doherty D.A, Magann E.F, Francis J, Morrison J.C., Newnham J.P. Pre-pregnancy body mass index and pregnancy outcomes. Int J Gynecol Obstet. 2006;95(3):242–247. doi: 10.1016/j.ijgo.2006.06.021. [DOI] [PubMed] [Google Scholar]
- 11.Leenersx B, Rath W, Kuse S, Irawan C, Imthurn B., Neumaier P. BMI: new aspects of a classical risk factor for hypertensive disorders in pregnancy. Clin Sci ( Lond ) 2006;111(1):81–86. doi: 10.1042/CS20060015. [DOI] [PubMed] [Google Scholar]
- 12.Fortner R.T, Pekow P, Solomon C.G, Markenson G., Chasan L. Prepregnancy body mass index, gestational weight gain, and risk of hypertensive pregnancy among Latina women. Am J Obstet Gynecol. 2009;200(2):167.e1–167.e7. doi: 10.1016/j.ajog.2008.08.021. [DOI] [PubMed] [Google Scholar]
- 13.Bodnar L.M, Ness R.B, Markovic N., Roberts J. The risk of preeclampsia rises with increasing prepregnancy body mass index. Ann Epidemiol. 2005;15(7):475–482. doi: 10.1016/j.annepidem.2004.12.008. [DOI] [PubMed] [Google Scholar]
- 14.Weiss J.L., Malone F.D, Emig D, Ball R.H, Nyberg D.A., Comstock C.H., et al. Obesity, obstetric complications and cesarean delivery rate: a population-based screening study. Am J Obstet Gynecol. 2004;190(4):1091–1097. doi: 10.1016/j.ajog.2003.09.058. [DOI] [PubMed] [Google Scholar]
- 15.Leung T.Y, Leung T.N, Sahota D.S, Chan O.K, Chan L.W., Fung T.Y., et al. Trends in maternal obesity and associated risks of adverse pregnancy outcomes in a population of Chinese women. BJOG. 2008;115(12):1529–1537. doi: 10.1111/j.1471-0528.2008.01931.x. [DOI] [PubMed] [Google Scholar]
- 16.Chu S.Y, Callaghan W.M, Kim S.Y, Schmid C.H, Lau J., England L.J., et al. Maternal obesity and risk of gestational diabetes mellitus. Diabetes Care. 2007;30(8):2070–2076. doi: 10.2337/dc06-2559a. [DOI] [PubMed] [Google Scholar]
- 17.Siega-Riz B., Laraia B. The implications of maternal overweight and obesity on the course of pregnancy and birth outcomes. Matern Child Health J. 2006;10(5 Suppl):S153–S156. doi: 10.1007/s10995-006-0115-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Herring S.J, Oken E, Rifas-Shiman S.L, Rich-Edwards J.W, Stuebe A.M., Kleinman K.P., et al. Weight gain in pregnancy and risk of maternal hyperglycemia. Am J Obstet Gynecol. 2009;201(1):61.e1–61.e7. doi: 10.1016/j.ajog.2009.01.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Dietz P.M, Callaghan W.M, Cogswell M.E, Morrow B, Ferre C., Schieve L.A. Combined effects of prepregnancy body mass index and weight gain during pregnancy on the risk of preterm delivery. Epidemiology. 2006;17(2):170–177. doi: 10.1097/01.ede.0000198470.26932.9a. [DOI] [PubMed] [Google Scholar]
- 20.Nohr E.A, Bech B.H, Vaeth M, Rasmussen K.M, Henriksen T.B., Olsen J. Obesity, gestational weight gain and preterm birth: a study within the Danish National Birth Cohort. Paediatr Perinat Epidemiol. 2007;21(1):5–14. doi: 10.1111/j.1365-3016.2007.00762.x. [DOI] [PubMed] [Google Scholar]
- 21.Ramsay J.E, Ferrell W.R, Crawford L, Wallace A.M, Greer I.A., Sattar N. Maternal obesity is associated with dysregulation of metabolic, vascular, and inflammatory pathways. J Clin Endocrinol Metab. 2002;87(9):4231–4237. doi: 10.1210/jc.2002-020311. [DOI] [PubMed] [Google Scholar]
- 22.Sahu M.T, Agarwal A, Das V., Pandey A. Impact of maternal body mass index on obstetric outcome. J Obstet Gynaecol Res. 2007;33(5):655–659. doi: 10.1111/j.1447-0756.2007.00646.x. [DOI] [PubMed] [Google Scholar]
- 23.Kabali C., Werler M.M. Pre-pregnant body mass index, weight gain and the risk of delivering large babies among non-diabetic mothers. Int J Gynecol Obstet. 2007;97(2):100–104. doi: 10.1016/j.ijgo.2007.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Oken E, Taveras E.M, Kleinman K.P, Rich-Edwards J.W., Gillman M.W. Gestational weight gain and child adiposity at age 3 years. Am J Obstet Gynecol. 2007;96(4):322.e1–322.e8. doi: 10.1016/j.ajog.2006.11.027. [DOI] [PMC free article] [PubMed] [Google Scholar]