Abstract
Background
Impaired fibrinolysis is found in impaired glucose tolerance and type 2 diabetes, associated with components of the metabolic syndrome. There are no data concerning fibrinolysis in subjects with normal glucose tolerance that convert to diabetes.
Methods
We studied the activities of tissue plasminogen activator (tPA) and plasminogen activator inhibitor-1 (PAI-1) and the levels of tPA antigen (a marker of endothelial dysfunction) in 551 subjects with normal glucose tolerance in 1990 in relation to incident diabetes during nine years of follow-up.
Results
Subjects with diabetes at follow-up (n = 15) had significantly lower baseline tPA activity and higher PAI-1 activity and tPA antigen than non-converters. The risk of diabetes increased linearly across quartiles of PAI-activity (p = 0.007) and tPA antigen (p < 0.001) and decreased across quartiles of tPA activity (p = 0.026). The risk of diabetes with low tPA activity or high PAI-1 activity persisted after adjustment for age and sex but diminished to a non-significant level after further adjustments. The odds ratio of diabetes for high tPA antigen was 10.4 (95% confidence interval 2.7–40) adjusted for age and sex. After further adjustment for diastolic blood pressure, waist circumference, insulin, triglycerides, fasting and post load glucose the odds ratio was 6.5 (1.3–33, p = 0.024).
Conclusions
Impaired fibrinolysis and endothelial dysfunction are evident in subjects with normal glucose tolerance who later develop diabetes. High tPA antigen is predictive of future diabetes independent from the metabolic syndrome.
Background
The risk of stroke and myocardial infarction is considerably increased in subjects with diabetes [1]. Already at the time of diagnosis of type 2 diabetes, many patients have manifest cardiovascular disease (CVD) [2]. This could be due to a long presymtomatic period with increased glucose level as evident by the increased CVD risk already present with impaired glucose tolerance (IGT) [3,4]. Thus, interest has focused on factors, not only glucose levels, in the prediabetic state that would increase the atherothrombotic process [5]. A common, possibly genetic, antecedent of both type 2 diabetes and CVD has been proposed as the "common soil" hypotheses [6,7].
Recently, markers of inflammation, such as highly sensitive C-reactive protein, have been found to predict type 2 diabetes in long-time follow up of population samples, although adjusting for measurements of obesity attenuates the relationship [8-12]. Such markers are weakly related to variables reflecting endothelial function such as adhesion molecules and von Willebrant factor [13].
In a large population-based study, also increased plasma levels of plasminogen activator inhibitor-1 (PAI-1) were strongly related to the development of diabetes independent from insulin resistance and obesity [8]. The possibility of elevated PAI-1 being a very early risk marker of the insulin resistance syndrome and diabetes was raised. Furthermore, haemostatic variables related to endothelial function, such as von Willebrant factor and factor VIII, also predicted diabetes, especially in women [14]. Data are lacking regarding other aspects of the fibrinolytic system and the development of diabetes, such as the activities of PAI-1 and tissue plasminogen activator (tPA) or the mass concentration of the endothelial-derived tPA ("tPA antigen"), factors related to CVD [15-19]
Thus, there are two questions related to fibrinolytic variables and the development of diabetes. Firstly, do subjects with normal glucose tolerance, who later convert to diabetes, have disturbances in the fibrinolytic system and endothelial function that could explain their increased risk of CVD even before diabetes ensues? If so, do these changes predict future diabetes, independent of the influence of the insulin resistance syndrome, thereby pointing to other pathways to the diabetic state such as endothelial dysfunction?
We studied fibrinolytic activity, as measured by tPA and PAI-1 activity, and a marker of endothelial dysfunction, as measured by tPA antigen, in a population sample with normal glucose tolerance at baseline and analysed data according to conversion to diabetes or not over nine years of follow-up.
Methods
This study was performed within the framework of the Northern Sweden MONICA study [20]. In 1990 a population sample was screened for cardiovascular risk factors. A total of 2,000 randomly selected subjects aged 25 to 64 years were invited. In all, 1,583 persons participated (79.2 %). A 75-gram oral glucose tolerance test (OGTT) was performed after an overnight fast in a randomly selected subset of subjects without known diabetes [21,22]. Venous plasma glucose samples were analysed by the hexokinase method (Boehringer Mannheim Automated Analysis for BM/Hitachi System 717). Glucose tolerance was classified according to WHO criteria from 1999 [23]:
Normal glucose tolerance (NGT) – fasting glucose <7 mmol/L and post load glucose <7.8 mmol/L,
Impaired glucose tolerance (IGT) – fasting glucose <7 mmol/L and post load glucose 7.8–11.0 mmol/L, and
Diabetes – fasting glucose ≥7.0 mmol/L and/or post load glucose ≥11.1 mmol/L.
Weight was measured with a balance scale to the nearest 0.2 kg, and height was measured to the nearest centimetre. BMI was calculated as total body weight (kg) / height (m2). Waist circumference was measured midway between the lower rib margin and the iliac crest, which in most occasions was identical with the level of the umbilicus. All measurements were done in a standing position while breathing normally, and participants were asked to wear light underwear and remove shoes. For the measurement of waist circumference, the recording was done after a gentle breath-out.
Blood sampling and OGTT took place before noon, after an overnight fast. tPA and PAI-1 activities were determined by chromogenic assays, Spectrolyse/fibrin and Spectrolyse/pl kit (Biopool AB, Umeå, Sweden), respectively. tPA antigen was measured by an enzyme-linked immunosorbent assay method (TintElize tPA, Biopool AB). Details regarding assay imprecision and analytical sensitivity have been published [24]. Serum insulin was determined by radio immunoassay with a double-antibody solid-phase technique (Phadaseph Insulin RIA, Pharmacia Diagnostics AB, Uppsala, Sweden)
In 1999, all participants, alive and still living in the area, were recalled for a follow-up examination. The same questionnaire and anthropometrical measurements as in the baseline survey were used. Altogether 1,148 (72.5 %) subjects turned up for re-examination. Subjects who previously had performed an OGTT with normal or impaired glucose tolerance, and answered no to the question "Do you have diabetes mellitus?" were once more offered an OGTT.
Participants, who denied having diabetes in the baseline survey and answered affirmative to the question "Do you have diabetes mellitus?" at follow up, were defined as incident cases of known diabetes mellitus according tho the WHO criteria from 1999 [23]. In November 2002, a questionnaire exploring details related to the diagnosis of diabetes, and to the type and duration of current hypoglycaemic treatment was sent to them. Case records were scrutinised to confirm diabetes diagnosis and time for start of insulin therapy. An algorithm from AusDiab was used to classify cases in subtypes [25]. Subjects that started their insulin treatment within two years of diagnosis were classified as type 1. However, for subjects fulfilling these criteria but with a age of 40 or above at onset, current BMI had to be <27 to be classified as type 1. All other cases were classified as type 2.
The Research Ethics Committee of Umeå University and the National Computer Data Inspection Board have approved the Northern Sweden MONICA Study.
Statistics
Means or medians are given for those who remained non-diabetic and those who converted to diabetes. Because the distributions for serum triglycerides, insulin and the fibrinolytic variables are highly skewed, we used logarithmically transformed values (i.e. geometric means). Differences between the two groups were compared by t-test. The risk of incident diabetes was compared across quartiles of the fibrinolytic variables, calculated from the total sample. Chi-squares analysis with linear-to-linear association was used. Logistic regression was used for calculation of odds ratios and 95% confidence intervals (CI) for the development of diabetes comparing the quartile with highest risk with the other three pooled quartiles. Stepwise adjustment for age and sex, waist, serum insulin, triglycerides and diastolic blood pressure was used.
Results
At baseline, 754 subjects had an OGTT performed. Five hundred and fifty-one subjects who initially had normal glucose tolerance returned for re-examination. A new OGTT was done in 477 of those with normal glucose tolerance and without clinically diagnosed diabetes. Thus, there was no repeated OGTT in 13 %, mostly due to logistic reasons such as not being able to turn up in the morning after an over night fast.
Among persons with initially normal glucose tolerance, in all 15 subjects (2.7 %) had type 2 diabetes diagnosed, either clinically (n = 4) or by OGTT (n = 11) after a 9-year follow-up or 4,959 person years. The rough incidence was 3.0 cases/1000 person years. Fifty-one subjects (10,7 %) worsened from normal glucose tolerance to impaired glucose tolerance.
Baseline characteristics are given in Table 1. Converters from NGT to diabetes were 7 years older and had higher BMI and waist circumference. Diastolic blood pressure did not differ but both fasting triglycerides, insulin, fasting and post load plasma glucose were higher in subjects with subsequent diabetes. Plasma levels of tPA antigen and PAI-1 activity were higher and tPA activity was lower.
Table 1.
Baseline characteristics of subjects with normal glucose tolerance according to diabetes or not during 9 years of follow-up
| Non-DM | DM | p-value | |
| n | 536 | 15 | |
| Age (years) | 44.9 (10.9) | 51.9 (8.7) | 0.015 |
| Sex (% men) | 60 | 46 | 0.3 |
| BMI (kg/m2) | 25.0 (3.5) | 28.4 (3.3) | <0.001 |
| Waist circumference (cm) | 84.2 (11.0) | 95.3 (9.8) | <0.001 |
| Diastolic blood pressure (mm Hg) | 79.8 (10.7) | 81.3 (10.8) | 0.6 |
| Triglycerides (mmol/l) | 1.2 (0.9–1.7) [1.25] | 2.1 (1.3–2.8) [1.95] | < 0.001 |
| Fasting plasma glucose (mmol/l) | 5.16 (0.50) | 5.89 (0.59) | <0.001 |
| Post load plasma glucose (mmol/l) | 5.13 (1.12) | 5.77 (1.04) | 0.028 |
| Fasting insulin (mU/ml) | 5.0 (4.0–7.0) [5.7] | 7.0 (8.0–10.0) [8.5] | 0.002 |
| tPA activity (IU/ml) | 0.79 (0.56–1.0) [0.74] | 0.59 (0.33–0.85) [0.54] | 0.020 |
| tPA antigen (μg/l) | 5.7 (4.0 – 8.4) [5.7] | 10.4 (9.0 – 13.7) [10.7] | <0.001 |
| PAI-1 activity (IU/ml) | 6.1 (2.9 – 11.2) [6.2] | 16.3 (7.1 – 18.8) [11.5] | 0.003 |
Data are means (SD), median (interquartile range) and [geometric means]. P values are for t test, or X2 test as appropriate.
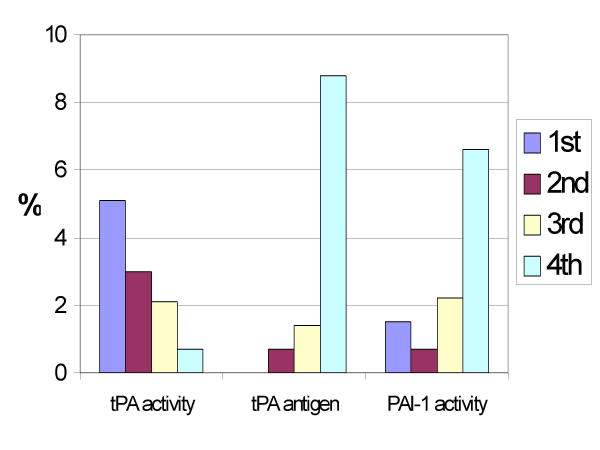
The risk of developing diabetes increased with increasing quartiles of tPA antigen and PAI-1 activity and decreased in a similar way for tPA activity (Figure 1). In the highest quartile of tPA antigen (above 8.7 μg/l) nearly 9 % had incident diabetes compared to none in the lowest quartile.
Figure 1.
Incidence of type 2 diabetes over nine years in subjects with normal glucose tolerance according to baseline quartiles of fibrinolytic variables. The number of cases in each quartile for tPA activity was 7, 4, 3 and 1. Corresponding numbers for tPA antigen was 0, 1, 2 and 12 and for PAI-1 activity 2, 1, 3 and 9, respectively. P values for X2 (linear by linear association) were 0.026 for tPA activity, <0,001 for tPA antigen and 0,007 for PAI-1 activity.
Logistic regression was performed with an initial model adjusting for age and sex. In a second model, waist circumference was added and in a third model, also diastolic blood pressure, serum insulin and triglycerides were added. Quartiles 1–3 of tPA antigen and PAI-1 activity were contrasted with the fourth quartile and, for tPA activity, the first quartiles with quartile 2–4 (Table 2).
Table 2.
Logistic regression analysis for fibrinolytic variables at baseline with incident type 2 diabetes as the dependent variable.
| OR | 95%CI | p value | |
| tPA activity Q2-4 vs Q1 | 2.7 | (0.96–7.6) | 0.059 |
| Adjusted for age and sex | 3.0 | (1.03–8.6) | 0.044 |
| Adjusted for also for waist | 1.7 | (0.5–5.5) | 0.4 |
| Adjusted also for diastolic BP, fasting insulin and triglycerides | 1.3 | (0.4–4.6) | 0.7 |
| tPA antigen Q1-3, vs 4 | 13.0 | (3.6–47) | <0.001 |
| Adjusted for age and sex | 10.4 | (2.7–40) | 0.001 |
| Adjusted for also for waist | 6.7 | (1.6–28) | 0.009 |
| Adjusted also for diastolic BP, fasting insulin and triglycerides | 5.5 | (1.2–25) | 0.026 |
| Adjusted also for fasting and postload glucose | 6.5 | (1.3–33) | 0.024 |
| PAI-1 activity, Q1-3, vs 4 | 4.8 | (1.7–14) | 0.004 |
| Adjusted for age and sex | 4.4 | (1.5–13) | 0.006 |
| Adjusted for also for waist | 2.8 | (0.9–8.5) | 0.08 |
| Adjusted also for diastolic BP, fasting insulin and triglycerides | 2.3 | (0.6–8.7) | 0.24 |
Q = quartile.
The lowest quartile of tPA activity was associated with a 3-fold increase in the risk of diabetes, which was attenuated after adjustment for waist circumference. After further adjustment for diastolic blood pressure, fasting insulin and triglycerides, no prediction from low tPA activity on diabetes development was seen.
Subjects in the highest quartile of tPA antigen experienced more than a ten-fold increase in risk, although the confidence intervals were wide due to few cases. Taking waist circumference into account, the odds ratio diminished to 6.7 and diminished further to 5.5 when adjusting for blood pressure, triglycerides and insulin. However, additional adjustment for baseline fasting and post load glucose increased the OR. The analysis was repeated using BMI instead of waist circumference. This lead to somewhat higher age- and gender adjusted odds ratio (7.4; CI 1.8–30, p value 0.005). Exchanging diastolic with systolic blood pressure did not change the findings. Furter adjustment for leisure time physical activity reduced the odds ratio from 5.5 to 5.1 (p value 0.03).
High PAI-1 activity increased the risk of diabetes four-fold, although the result was attenuated to an insignificant OR of 2.0 when all factors were adjusted for simultaneously.
The risk of developing diabetes or IGT during follow up (n = 66) increased stepwise across quartiles of tPA antigen, from 7.8% to 19.8% (p = 0,003, test for linear trend). When adjusted for age and sex, the odds ratio for the highest vs. the three lower quartiles of tPA antigen was 1.9 (CI 1.01–1.6, p = 0.04) but diminished further when also waist circumference was adjusted for, OR = 1.3 (CI 0.63–6.5, p = 0.5).
Discussion
Subjects with normal glucose tolerance, who subsequently develop type 2 diabetes over the ensuing nine years, are already at baseline characterised by impaired fibrinolysis and endothelial dysfunction. Low tPA activity and high PAI-1 activity is mainly explained by the presence of abdominal obesity but also by other metabolic disturbances characteristic of the insulin resistance syndrome, such as high serum levels of triglycerides and insulin. A novel finding, in this context, is that a high plasma level of the endothelial-derived tPA is only slightly explained by such factors. Thus, the development of type 2 diabetes is preceded by many years of perturbations in fibrinolytic and endothelial function, which increases the risk of atherothrombotic disease long before overt diabetes is present.
PA1-1 is partly synthesised in fat cells and its activity is strongly related to abdominal obesity as mirrored by high waist circumference or WHR [26]. High circulating levels of PAI-1 inhibit tPA released from the vessel walls and lead to low levels of free tPA, i.e. low tPA activity and impaired capability of thrombolysis. The only report of an association between fibrinolysis and the development of diabetes is a recent study where PAI-1 antigen was an independent predictor of diabetes in American-Mexicans that were followed up to 5 years [10]. There is no known pathway whereby impaired fibrinolysis would increase insulin resistance or decrease insulin release and our finding that high PAI-1 and low tPA activity precedes diabetes probably mirrors the strong relationship with intraabdominal fat and circulating levels of free fatty acids.
A recent experimental study show an almost three fold increase in the level of PAI-1 antigen with an infusion of triacylglycerol in healthy subjects without any impact on tPA [27]. This effect was noted with constant levels of insulin and glucose and also increased levels of soluble vascular cell adhesion molecules-1 was found. This support our findings that adjustment for baseline triglyceride levels, which were increased in converters, did not significantly change the relationships noted for tPA antigen but did decrease those for PAI-1.
tPA antigen has not previously been studied in prospective diabetes studies. A clear distinction is not possible between markers of endothelial dysfunction and inflammatory markers [13]. It is thus possible that also tPA concentration is an inflammatory marker although we found low correlations between tPA antigen and fibrinogen [24] and highly sensitive CRP (own unpublished data). Recently, it was described in type 2 diabetes patients, that increased levels of tPA antigen, among other endothelial markers, predicted the development of urinary albumin excretion, a strong marker for CVD risk [28]. These findings should be related to recent reports where inflammatory markers predict the development of diabetes in longitudinal studies. In most cases, this association was diminished or vanished after indices of obesity were taken into account. Pro-inflammatory substances derived from abdominal fat tissue may be the common background for these findings.
We therefore find it plausible that the relation between high tPA and diabetes is a marker of some early disturbance in the vascular wall that operates independent of obesity and inflammation. Already borderline hypertension is associated with high concentrations of tPA antigen, even after adjustment for abdominal obesity, insulin and triglycerides [29]. Thus, tPA mass may mirror an endothelial dysfunction that is not causally related to the insulin resistance syndrome but acts by a parallel pathogenetic mechanism leading to atherothrombosis [30-32]. Insulin resistance, on the other hand, is associated with endothelial dysfunction and impaired vasodilatation due to defects in endothelium-derived nitric oxide [33,34]. Taken together, these findings put focus on the role of the vessel walls in the pathogenesis of type 2 diabetes.
The relevance of impaired fibrinolysis, i.e. low activity of tissue plasminogen activator (tPA) or high activity of its inhibitor (PAI-1), as independent risk factors for CVD is unclear, as these variables seem to mirror most components of the insulin resistance syndrome [21,26]. In some studies, decreased fibrinolysis, as measured by high PAI-1 activity, has been associated with cardiovascular events [15,16]. High levels of tPA antigen independently predict cardiovascular events both in a healthy population [16,17] and in patients with prevalent coronary disease [18,19].
The major shortcoming of our study is the small number of incident cases of diabetes leading to wide confidence intervals. On the other hand, the odds ratios for tPA antigen are high and consistent and based on a truly representative population sample where basal normal glucose tolerance was well defined, as were the incident cases of diabetes. Thus, the risk of bias due to selection or case ascertainment should be low. Admittedly, the lack of a renewed OGTT in 13 % of the subjects could contribute to some misclassifiaction and dilute the results somewhat. An ongoing analysis of two more cohorts will serve to extend our observations.
Conclusions
The early prediabetic state, with normal glucose tolerance, is characterised by both impaired fibrinolysis and endothelial dysfunction years before glucose levels increase. Changes in endothelial function are independent from the insulin resistance syndrome and point towards a distinctly different pathway both for cardiovascular disease and for the development of diabetes. To answer a classical question from 1990 [5] – Yes, the clock for coronary heart disease seems to start ticking before the onset of clinical diabetes!
Abbreviations
BMI = body mass index
CI = confidence interval
CRP = C-reactive protein
CVD = cardiovascular disease
IGT = impaired glucose tolerance
MONICA = MONItoring of trends and determinants of CArdiovascular disease
NGT = normal glucose tolerance
OGTT = oral glucose tolerance test
PAI-1 = plasminogen activator inhibitor-1
tPA = tissue plasminogen activator
Competing interests
None declared.
Authors' contributions
ME drafted the research on fibrinolysis within the MONICA project, performed the statistical analysis and drafted the manuscript. BS and BL participated in the design of Northern Sweden MONICA study and participated in its design and coordination. JHJ participated in the analysis. All authors read and approved the final manuscript.
Acknowledgments
Acknowledgements
Supported by grants from Norrbotten and Västerbotten counties, by the Joint Committee of Northern Sweden Health Care Region, the Swedish Public Health Institute and the Swedish Medical Research Council.
Contributor Information
Mats CE Eliasson, Email: mats.eliasson@nll.se.
Jan-Håkan Jansson, Email: janhakan.jansson@vll.se.
Bernt Lindahl, Email: bernt.lindahl@medicin.umu.se.
Birgitta Stegmayr, Email: birgitta.stegmayr@medicin.umu.se.
References
- Eliasson M, Lindahl B, Lundberg V, Stegmayr B. Diabetes and obesity in northern Sweden - occurence and risk factor for stroke and myocardial infarction. Scand J Publ Health. 2003;31:70–77. doi: 10.1080/14034950310001360. [DOI] [PubMed] [Google Scholar]
- UK Prospective Diabetes Study 6. Complications in newly diagnosed type 2 diabetic patients and their association with different clinical and biochemical risk factors. Diabetes Res. 1990;13:1–11. [PubMed] [Google Scholar]
- DECODE Study Group Glucose tolerance and cardiovascular mortality: comparison of fasting and 2-hour diagnostic criteria. Arch Intern Med. 2001;161:397–405. doi: 10.1001/archinte.161.3.397. [DOI] [PubMed] [Google Scholar]
- Lundblad D, Eliasson M. Silent myocardial infarction in women with impaired glucose tolerance: The Northern Sweden MONICA study. Cardiovasc Diabetol. 2003;2:9. doi: 10.1186/1475-2840-2-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haffner SM, Stern MP, Hazuda HP, Mitchell BD, Patterson JK. Cardiovascular risk factors in confirmed prediabetic individuals. Does the clock for coronary heart disease start ticking before the onset of clinical diabetes? JAMA. 1990;263:2893–2898. doi: 10.1001/jama.263.21.2893. [DOI] [PubMed] [Google Scholar]
- Jarrett RJ, Shipley MJ. Type 2 (non-insulin-dependent) diabetes mellitus and cardiovascular disease--putative association via common antecedents; further evidence from the Whitehall Study. Diabetologia. 1988;31:737–740. doi: 10.1007/BF00274775. [DOI] [PubMed] [Google Scholar]
- Stern MP. Diabetes and cardiovascular disease. The "common soil" hypothesis. Diabetes. 1995;44:369–374. doi: 10.2337/diab.44.4.369. [DOI] [PubMed] [Google Scholar]
- Festa A, D'Agostino R., Jr., Tracy RP, Haffner SM. Elevated levels of acute-phase proteins and plasminogen activator inhibitor-1 predict the development of type 2 diabetes: the insulin resistance atherosclerosis study. Diabetes. 2002;51:1131–1137. doi: 10.2337/diabetes.51.4.1131. [DOI] [PubMed] [Google Scholar]
- Freeman DJ, Norrie J, Caslake MJ, Gaw A, Ford I, Lowe GD, O'Reilly DS, Packard CJ, Sattar N. C-reactive protein is an independent predictor of risk for the development of diabetes in the West of Scotland Coronary Prevention Study. Diabetes. 2002;51:1596–1600. doi: 10.2337/diabetes.51.5.1596. [DOI] [PubMed] [Google Scholar]
- Han TS, Sattar N, Williams K, Gonzalez-Villalpando C, Lean ME, Haffner SM. Prospective study of C-reactive protein in relation to the development of diabetes and metabolic syndrome in the Mexico City Diabetes Study. Diabetes Care. 2002;25:2016–2021. doi: 10.2337/diacare.25.11.2016. [DOI] [PubMed] [Google Scholar]
- Thorand B, Lowel H, Schneider A, Kolb H, Meisinger C, Frohlich M, Koenig W. C-reactive protein as a predictor for incident diabetes mellitus among middle-aged men: results from the MONICA Augsburg cohort study, 1984-1998. Arch Intern Med. 2003;163:93–99. doi: 10.1001/archinte.163.1.93. [DOI] [PubMed] [Google Scholar]
- Nakanishi S, Yamane K, Kamei N, Okubo M, Kohno N. Elevated C-reactive protein is a risk factor for the development of type 2 diabetes in Japanese americans. Diabetes Care. 2003;26:2754–2757. doi: 10.2337/diacare.26.10.2754. [DOI] [PubMed] [Google Scholar]
- Krakoff J, Funahashi T, Stehouwer CD, Schalkwijk CG, Tanaka S, Matsuzawa Y, Kobes S, Tataranni PA, Hanson RL, Knowler WC, Lindsay RS. Inflammatory markers, adiponectin, and risk of type 2 diabetes in the Pima Indian. Diabetes Care. 2003;26:1745–1751. doi: 10.2337/diacare.26.6.1745. [DOI] [PubMed] [Google Scholar]
- Duncan BB, Schmidt MI, Offenbacher S, Wu KK, Savage PJ, Heiss G. Factor VIII and other hemostasis variables are related to incident diabetes in adults. The Atherosclerosis Risk in Communities (ARIC) Study. Diabetes Care. 1999;22:767–772. doi: 10.2337/diacare.22.5.767. [DOI] [PubMed] [Google Scholar]
- Hamsten A, Wiman B, de Faire U, Blomback M. Increased plasma levels of a rapid inhibitor of tissue plasminogen activator in young survivors of myocardial infarction. N Engl J Med. 1985;313:1557–1563. doi: 10.1056/NEJM198512193132501. [DOI] [PubMed] [Google Scholar]
- Thogersen AM, Jansson JH, Boman K, Nilsson TK, Weinehall L, Huhtasaari F, Hallmans G. High plasminogen activator inhibitor and tissue plasminogen activator levels in plasma precede a first acute myocardial infarction in both men and women: evidence for the fibrinolytic system as an independent primary risk factor. Circulation. 1998;98:2241–2247. doi: 10.1161/01.cir.98.21.2241. [DOI] [PubMed] [Google Scholar]
- Ladenvall P, Johansson L, Jansson JH, Jern S, Nilsson TK, Tjarnlund A, Jern C, Boman K. Tissue-type plasminogen activator -7,351C/T enhancer polymorphism is associated with a first myocardial infarction. Thromb Haemost. 2002;87:105–109. [PubMed] [Google Scholar]
- Jansson JH, Nilsson TK, Olofsson BO. Tissue plasminogen activator and other risk factors as predictors of cardiovascular events in patients with severe angina pectoris. Eur Heart J. 1991;12:157–161. doi: 10.1093/oxfordjournals.eurheartj.a059862. [DOI] [PubMed] [Google Scholar]
- Jansson JH, Olofsson BO, Nilsson TK. Predictive value of tissue plasminogen activator mass concentration on long-term mortality in patients with coronary artery disease. A 7-year follow-up. Circulation. 1993;88:2030–2034. doi: 10.1161/01.cir.88.5.2030. [DOI] [PubMed] [Google Scholar]
- Stegmayr B, Lundberg V, Asplund K. The event registration and survey procedures in the Northern Sweden MONICA Project. Scand J Publ Health. 2003;31:9–17. doi: 10.1080/14034950310001441. [DOI] [PubMed] [Google Scholar]
- Eliasson M, Asplund K, Evrin PE, Lindahl B, Lundblad D. Hyperinsulinemia predicts low tissue plasminogen activator activity in a healthy population: the Northern Sweden MONICA Study. Metabolism. 1994;43:1579–1586. doi: 10.1016/0026-0495(94)90020-5. [DOI] [PubMed] [Google Scholar]
- Eliasson M, Lindahl B, Lundberg V, Stegmayr B. No increase in the prevalence of known diabetes between 1986 and 1999 in subjects 25-64 years of age in northern Sweden. Diabet Med. 2002;19:874–880. doi: 10.1046/j.1464-5491.2002.00789.x. [DOI] [PubMed] [Google Scholar]
- World Health Organisation . Definition, diagnosis and classification of diabetes mellitus and its complications. Report of a WHO consultation Part 1: Diagnosis and classification of diabetes mellitus. Geneva, WHO/NCD/NCS/99.2; 1999. [Google Scholar]
- Eliasson M, Evrin PE, Lundblad D, Asplund K, Rånby M. Influence of gender, age and sampling time on plasma fibrinolytic variables and fibrinogen. Fibrinolysis. 1993;7:316–323. [Google Scholar]
- Welborn TA, Garcia-Webb P, Bonser A, McCann V, Constable I. Clinical criteria that reflect C-peptide status in idiopathic diabetes. Diabetes Care. 1983;6:315–316. doi: 10.2337/diacare.6.3.315. [DOI] [PubMed] [Google Scholar]
- Eliasson M, Evrin PE, Lundblad D. Fibrinogen and fibrinolytic variables in relation to anthropometry, lipids and blood pressure. The Northern Sweden MONICA Study. J Clin Epidemiol. 1994;47:513–524. doi: 10.1016/0895-4356(94)90298-4. [DOI] [PubMed] [Google Scholar]
- Krebs M, Geiger M, Polak K, Vales A, Schmetterer L, Wagner OF, Waldhausl W, Binder BR, Roden M. Increased plasma levels of plasminogen activator inhibitor-1 and soluble vascular cell adhesion molecule after triacylglycerol infusion in man. Thromb Haemost. 2003;90:422–428. doi: 10.1160/TH03-07-0457. [DOI] [PubMed] [Google Scholar]
- Stehouwer CD, Gall MA, Twisk JW, Knudsen E, Emeis JJ, Parving HH. Increased urinary albumin excretion, endothelial dysfunction, and chronic low-grade inflammation in type 2 diabetes: progressive, interrelated, and independently associated with risk of death. Diabetes. 2002;51:1157–1165. doi: 10.2337/diabetes.51.4.1157. [DOI] [PubMed] [Google Scholar]
- Eliasson M, Jansson JH, Nilsson P, Asplund K. Increased levels of tissue plasminogen activator antigen in essential hypertension. A population-based study in Sweden. J Hypertens. 1997;15:349–356. doi: 10.1097/00004872-199715040-00005. [DOI] [PubMed] [Google Scholar]
- Diamond SL, Eskin SG, McIntire LV. Fluid flow stimulates tissue plasminogen activator secretion by cultured human endothelial cells. Science. 1989;243:1483–1485. doi: 10.1126/science.2467379. [DOI] [PubMed] [Google Scholar]
- Iba T, Sumpio BE. Tissue plasminogen activator expression in endothelial cells exposed to cyclic strain in vitro. Cell Transplant. 1992;1:43–50. doi: 10.1177/096368979200100108. [DOI] [PubMed] [Google Scholar]
- Salame MY, Samani NJ, Masood I, deBono DP. Expression of the plasminogen activator system in the human vascular wall. Atherosclerosis. 2000;152:19–28. doi: 10.1016/S0021-9150(99)00441-4. [DOI] [PubMed] [Google Scholar]
- Steinberg HO, Chaker H, Leaming R, Johnson A, Brechtel G, Baron AD. Obesity/insulin resistance is associated with endothelial dysfunction. Implications for the syndrome of insulin resistance. J Clin Invest. 1996;97:2601–2610. doi: 10.1172/JCI118709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baron AD. The coupling of glucose metabolism and perfusion in human skeletal muscle. The potential role of endothelium-derived nitric oxide. Diabetes. 1996;45:S105–9. doi: 10.2337/diab.45.1.s105. [DOI] [PubMed] [Google Scholar]