Abstract
Chronic pain is a frequent component of many neurological disorders, affecting 20–40% of patients for many primary neurological diseases. These diseases result from a wide range of pathophysiologies including traumatic injury to the central nervous system, neurodegeneration and neuroinflammation, and exploring the aetiology of pain in these disorders is an opportunity to achieve new insight into pain processing. Whether pain originates in the central or peripheral nervous system, it frequently becomes centralized through maladaptive responses within the central nervous system that can profoundly alter brain systems and thereby behaviour (e.g. depression). Chronic pain should thus be considered a brain disease in which alterations in neural networks affect multiple aspects of brain function, structure and chemistry. The study and treatment of this disease is greatly complicated by the lack of objective measures for either the symptoms or the underlying mechanisms of chronic pain. In pain associated with neurological disease, it is sometimes difficult to obtain even a subjective evaluation of pain, as is the case for patients in a vegetative state or end-stage Alzheimer's disease. It is critical that neurologists become more involved in chronic pain treatment and research (already significant in the fields of migraine and peripheral neuropathies). To achieve this goal, greater efforts are needed to enhance training for neurologists in pain treatment and promote greater interest in the field. This review describes examples of pain in different neurological diseases including primary neurological pain conditions, discusses the therapeutic potential of brain-targeted therapies and highlights the need for objective measures of pain.
Keywords: brain imaging, Parkinson's disease, complex regional pain syndrome, migraine, brain trauma
Introduction
Recent advances in basic and clinical neuroscience suggest the brain plays a pivotal role in the chronic pain state. Recent advances in pain research, fuelled by neuroimaging studies, have engendered a transformation in our understanding of how pain affects the brain. As a result, the notion that changes in sensory systems are the predominant process in chronic pain has been replaced by a conceptualization of chronic pain as a very complex CNS state in which patterns of sensory system activation are integrated aberrantly with activity in other brain systems, including emotional, cognitive and modulatory processes. Obvious causes such as peripheral nerve injury-induced pain (neuropathic pain) affect a large number of brain regions with a wide range of other functions such as the anterior cingulate cortex, insular cortex, ventrolateral orbitofrontal area, amygdala, striatum, thalamus, hypothalamus, rostral ventromedial medulla, periaqueductal grey, pons (locus coeruleus), red nucleus, medulla oblongata and other less obvious causes of pain including those associated with primary depression where there is no injury or prior pain condition. More recently, clinicians and researchers have come to the conclusion that, in many cases, chronic pain is a direct result of the neurological disease, or may even be considered an integral part of the underlying disease. Perhaps the best example of this is Parkinson's disease, where 40–60% of patients report chronic pain (Simuni and Sethi, 2008; Ford, 2010). Table 1 shows the prevalence of pain in many neurological diseases, including some that are primary pain disorders commonly seen by neurologists, and the rates are striking. As noted in Fig. 1, pain produces changes throughout the CNS with particular effects on emotional processing. The latter interaction is complex, for example, pain causes depression and depression causes pain (Lepine and Briley, 2004; Borsook et al., 2007; Husain et al., 2007; Maletic and Raison, 2009; Elman et al., 2011). The prevalence of co-morbid depression is high in many chronic neurological diseases (Table 1).
Table 1.
Pain and neurological diseases
| Disease | Prevalence of pain | Pain symptoms | Co-morbid depression |
|---|---|---|---|
| Degenerative | |||
| Parkinson's disease | 40–60% (Simuni and Sethi, 2008; Ford, 2010) |
|
|
| Huntington's disease | Unknown |
|
40% (Paulsen et al., 2005) |
| Alzheimer's disease | 57% (Pautex et al., 2006) | Musculoskeletal Other |
|
| CNS damage | |||
| Post-stroke (thalamic) | 8–14% of all stroke patients (Kumar et al., 2009) |
|
36% (Bour et al., 2010) |
| Multiple sclerosis | 50–86% (O'Connor et al., 2008; Bermejo et al., 2010) | Extremity pain, trigeminal neuralgia, Lhermitte's sign, painful tonic spasms, back pain and headache. | 50% (Siegert and Abernethy, 2005) |
| Syringomyelia |
|
|
Unknown |
| Spinal cord injurya | 64.9% (Modirian et al., 2010) |
|
|
| Traumatic brain injury |
|
|
|
| Metabolic | |||
| Fabry's disease | 81% (males); 65% (females) (Hoffmann et al., 2007) | Hands > feet > whole body |
|
| Diabetic neuropathya | 63% (Davies et al., 2006) |
|
41% with diabetes (Raval et al., 2010) |
| Neuromuscular | |||
| Amyotrophic lateral sclerosis |
|
Arms > other parts (de Castro-Costa et al., 1999) |
|
| Guillaine-Barrea |
|
|
18% Extreme depression (Khan et al., 2010) |
| Tumours | |||
| NF2 Schwannomatosis Schwannomas | >60% pain (MacCollin et al., 2005) |
|
Unknown |
| Peripheral nerve | |||
| Complex regional pain syndrome |
|
|
41% (Ciccone et al., 1997) |
| Post-herpetic neuralgiaa | PHN in 20% of HZ patients; 90% of these have pain (Johnson et al., 2010) |
|
44% (Atkinson et al., 1986) |
| Back paina |
|
|
5–54% (Sullivan et al., 1992) |
| Post-surgical neuropathic paina | 10–50% of surgeries (Kehlet et al., 2006) | Classic neuropathic symptoms | 2% (Kalliomaki et al., 2009) |
| Migraine (episodic) |
|
|
|
| HIV/AIDS | 30–90% (Hewitt et al., 1997) |
|
8–45% (Perkins et al., 1994) |
| Idiopathic/unknown aetiology | |||
| Fibromyalgiaa |
|
Widespread pain Extremity dysaesthesias | 20–80% (Fietta and Manganelli, 2007) |
a Not discussed in this article. HZ = herpes zoster; PHN = post-herpetic neuralgia.
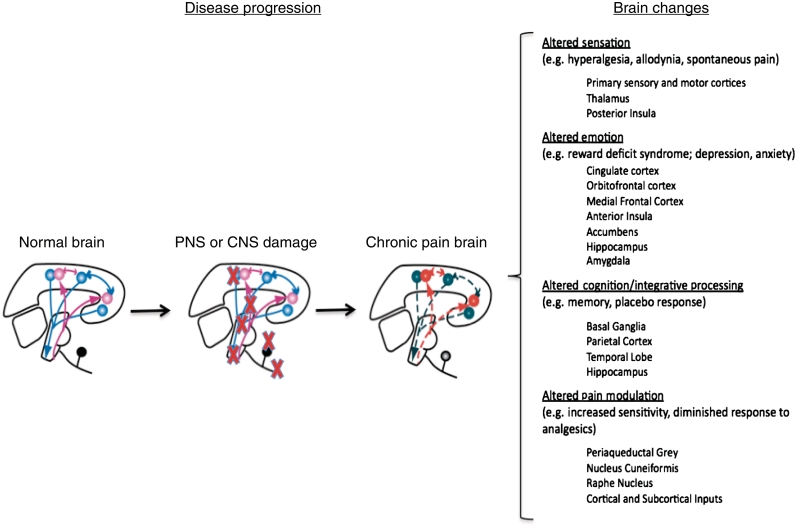
Figure 1.
Brain changes in chronic pain. The figure summarizes two concepts that relate to the development of chronic pain following damage to either peripheral or CNS pathways involved in pain. (i) The first is that following injury (red crosses) progressive changes take place in the brain: the normal brain is altered in a manner that produces changes in function and structure in the chronic pain state; (ii) the second, noted in the text on the right, indicates that multiple brain regions involved in sensation, emotion, cognition and pain modulation may manifest in varied behavioural symptoms from ongoing pain to anxiety and depression. While it is easy to conceptualize altered systems, altered circuitry is best considered in the context of interactive brain processes that are disrupted in chronic pain. Both central and peripheral origins of pain are noted; the peripheral sources are clearly important in producing or maintaining central changes whether these are part of a disease affecting both central and peripheral systems or not.
Damage to either the peripheral or CNS is a well-defined cause of neuropathic pain. Considering the altered patterns of brain activity in neurological disease with pain may provide insight into pain processing in the brain in chronic disease. In contrast to the many neurological diseases with associated pain symptoms, some neurological conditions are associated with diminished pain or no pain (i.e. congenital insensitivity to pain). The underlying pathology and regional changes in brain systems are well described for some of these disorders, and examining them may also shed light on how alterations in the central circuitry of the brain produce chronic pain. In this article, we define chronic pain as a brain disease based on significant changes in function, anatomy (see discussion below on morphological changes in chronic pain) and chemistry, which occur following pathophysiological alterations in pain pathways. These changes occur in areas of the brain involved in sensory, emotional and modulatory systems and are ‘brain-wide’ [e.g. including regions not normally associated with pain such as the cerebellum (Moulton et al., 2010)]. These changes are a direct consequence of pain or secondary to comorbid changes such as depression or anxiety (Elman et al., 2011).
The changes in brain activity that underlie chronic pain may result in changes in central circuits that manifest as pain in the absence of the peripheral trigger. ‘Centralization’ of pain, here defined as ‘the persistent static or dynamic brain functional state that contributes to or causes the behavioural responses to pain (e.g. depression increased sensitivity to stimuli, ongoing pain)’, occurs as a result of altered brain dynamics not only in specific sensory systems, but also in other brain systems including emotional, cognitive and motor systems. This altered state results in a cognitive, sensory and emotional experience of pain, whether the initial instigating process is in the peripheral (perhaps including muscle) or CNS, as a result of primary brain disease, or secondary to afferent input as a result of nerve or spinal cord damage. Figure 2 summarizes known alterations in brain systems in chronic pain, including functional, anatomical and chemical changes. Specific examples are referenced.
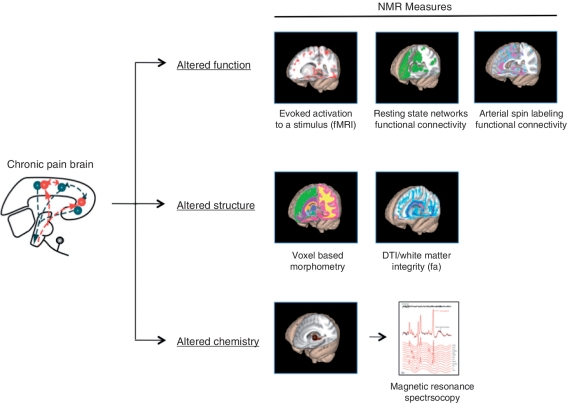
Figure 2.
Changes in function, structure and chemistry in chronic pain. Chronic pain alters the brain (left) and produces alterations in function [e.g. increased activation as in central sensitization (Lee et al., 2008)]; altered resting state networks (Apkarian et al., 2004); and altered chemistry [e.g. changes in excitatory and inhibitory amino acids, (Gussew et al., 2011)]. While the example of nuclear magnetic resonance (NMR) approaches for evaluating alterations in the brain in pain conditions (Borsook et al., 2007; Borsook and Becerra, 2011) is provided, other approaches have been employed to measure pain-related alterations in brain systems including electroencephalography (Brinkmeyer et al., 2010), magnetoencephalography (Maihofner et al., 2010) and near infrared spectroscopy (Slater et al., 2006; Becerra et al., 2008). fa = fractional anisotropy; fMRI = functional MRI.
While a number of recent reviews cover neuropathic pain (Baron et al., 2010), the primary focus here is on specific neurological conditions that have pain as a co-morbid condition (defined as the presence of pain in addition to the primary neurological disorder), with a discussion of the potential insights into pain neurobiology provided by what we know about each disease state (Fig. 3). The section `Neurological disease and pain' summarizes links between neurological disease, disease markers and genetic traits that may contribute to the pathophysiology of pain in neurological diseases. In section, ‘Brain-based restorative approaches for chronic pain’, novel therapies that target brain systems are discussed as opportunities for neurologists to take the lead role in chronic pain treatment and clinical research. In the final section, ‘Smarter tools for objective diagnosis of pain’ section, we briefly discuss the need for objective markers for pain.
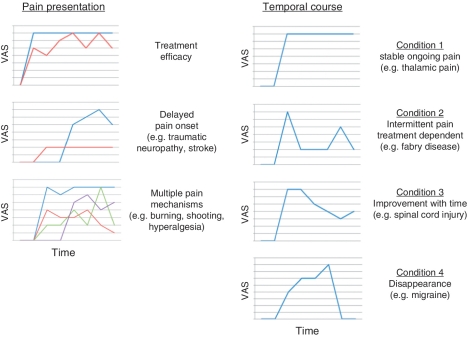
Figure 3.
Chronic pain symptoms and variations in temporal course. Chronobiological effects of pain, treatment effects, environmental changes (e.g. barometric pressure) activity all may contribute to a variation in pain over time (Auvil-Novak, 1999; Lake, 2005; Odrcich et al., 2006; Dworkin et al., 2007; Kloss-Brandstatter et al., 2011). Left: Examples of features of pain presentation. (i) Relatively small effect of therapy (red line) versus pain (blue line) [pain is rated by patients on an 11-point (0–10) Likert's scale (VAS)] usually decreases by just two points on this scale; on average most pharmacological treatments decrease pain by 20–30% in population placebo controlled studies; (ii) the onset of pain may be variable and even be delayed following disease onset (red line)—this is more easily observed with specific PNS or CNS damage (i.e. traumatic neuropathy or thalamic stroke); (iii) most conditions have more than one pain (lines in different colours representing different pains) that can be defined mechanistically—e.g. shooting pain due to ectopic activity; burning pain due to inhibitory neuron dropout (Baron et al., 2010). Right: Representative examples of different temporal courses for pain from constant (Condition 1) to intermittent or dependent on specific treatment (Condition 2) to a natural course of improvement (Condition 3) to complete remission (Condition 4). The colours are used to differentiate time courses where multiple plots are used.
Understanding potential links between pain pathophysiology and neurological disease
Recent advances have greatly increased our understanding of pain mechanisms. Although much of this work has been in peripheral systems and lower CNS areas—peripheral nerve, the spinal cord and brainstem (Dubner, 2004), research focused on higher brain centres is increasing rapidly (Woolf, 2011), including neuroimaging of pain in humans (Tracey and Mantyh, 2007). Pain may be a sentinel marker of disease (e.g. acute back pain, acute shingles) or a consequence of disease (e.g. post-herpetic neuralgia, thalamic stroke, spinal cord injury). Neuroimaging has identified states of activation that associate with pain; however, it is unclear if the initial pain-related state markers (usually transient) transform into trait markers (usually enduring) of a neurological disease. In some primary neurological diseases, pain contributes to the course of the condition; this is perhaps most obvious in back pain, but is significantly less clear in other neurological conditions with pain (e.g. Parkinson's disease). Where pain contributes to the course of disease, this may be a direct result of pain-related CNS changes or may be the result of associated processes such as immune response. Conversely, the immune response may modulate pain, thereby affecting disease course (Ren and Dubner, 2010; Austin and Moalem-Taylor, 2010).
Only a few studies have examined genetic, neurobiological and behavioural factors in specific neurological diseases with a focus on pain. However, some of these have identified potential genetic markers of the risk of developing chronic pain, including the GTP-cyclohydrolase 1, encoded by GCH1 pain-protective gene haplotype, which decreases pain levels (Tegeder et al., 2006); the potassium channel alpha subunit KCNS1 that associates with several chronic pain conditions e.g. back pain, amputation (Costigan et al., 2010); and the calcium channel gamma subunit gene CACNG2, a protein intimately involved in the trafficking of glutamatergic α-amino-3-hydroxy-5-methyl-4 isoxazolepropionic acid receptors that is implicated in susceptibility to chronic pain (Nissenbaum et al., 2010). We are unaware of any twin studies that have evaluated trait markers that may contribute to the relationship between pain and primary neurological conditions. Clearly, genome-wide association studies are the way forward to define clinically relevant genetic markers that predict pain susceptibility, severity and treatment responses in neurological conditions. Insights into genetic mutations that prevent pain may open new opportunities to understanding pain in neurological conditions and their treatment (Oertel and Lotsch, 2008). Characterization of pain endophenotypes through measures including functional brain imaging may allow us to connect genetic findings with defined biomarkers underlying pain-related processing (Tracey, 2011).
Neurological diseases and pain
This section discusses examples of neurological diseases that have pain as a co-existing or co-morbid process. This is not intended to be an exhaustive review of pain in neurological disease but to illustrate how commonly pain presents with neurological disease across a spectrum of underlying pathologies and to provide some insight into how the pathophysiologies of each disease may facilitate or synergize with brain mechanisms involved in chronic pain. For many of these diseases, the underlying pathophysiology is not clear, so our discussion of the potential interaction is necessarily brief and is intended to spark interest rather than present definitive connections. For most neurological conditions not discussed in this article, there are already excellent reviews of the literature on pain as related to the condition: fibromyalgia (Brederson et al., 2011; Smith et al., 2011); phantom pain occurring in missing limbs (Flor, 2002; Grüsser et al., 2003; Ketz, 2008) but also many other body regions (Holland et al., 1994); post-traumatic painful neuropathy occurring in significant numbers (10–50%) of patients following surgery (Kehlet et al., 2006); chronic back pain (Manchikanti et al., 2009) including failed back surgery (Chan and Peng, 2011); peripheral neuropathic pain such as painful diabetic neuropathy and small fibre neuropathies (Vaillancourt and Langevin, 1999; Sommer, 2003); and post-herpetic neuralgia (Hempenstall et al., 2005; Philip and Thakur, 2011).
Central nervous system degenerative diseases
Parkinson's disease and pain
Parkinson's disease is perhaps the best example of co-morbid pain as an integral part of a neurodegenerative disease. Between 40 and 60% of patients with Parkinson's disease have chronic pain that frequently includes more than one type of pain (Giuffrida et al., 2005; Simuni and Sethi, 2008; Ford, 2010). Using the Brief Pain Inventory to assess average pain over a 24-h period, patients with Parkinson's disease reported an average pain level of 2.85, significantly greater than the general population, with >50% of patients reporting one, 24% reporting two and 5% reporting three pain types. Of these, musculoskeletal pain was noted in 70%, dystonic pain in 40%, radicular–neuropathic pain in 20% and central neuropathic pain in 10%. Around 34% of patients in this study were on analgesic medication. Pain was significantly higher (83%) in those with dystonic symptoms (Beiske et al., 2009). Pain in Parkinson's disease correlates with age, disease duration and severity, and, as with many pain conditions, female gender is a significant predictor of pain. In addition, chronic use of analgesic prescription drugs is highly prevalent in Parkinson's disease (Brefel-Courbon et al., 2009): patients with Parkinson's disease received more prescriptions for analgesics than the general population (82% versus 77%), but fewer than patients with osteoarthritis (82% versus 90%). As a comparison, no significant difference in analgesic use was found between Parkinson's disease and diabetic patients, a patient group traditionally used to evaluate analgesics in clinical trials. Clearly, pain is a problem in Parkinson's disease and the growing interest in Parkinson's disease and pain over the past two decades is reflected by the increase in publications related to pain and Parkinson's disease and by increased efforts to manage pain symptoms (Fig. 4).
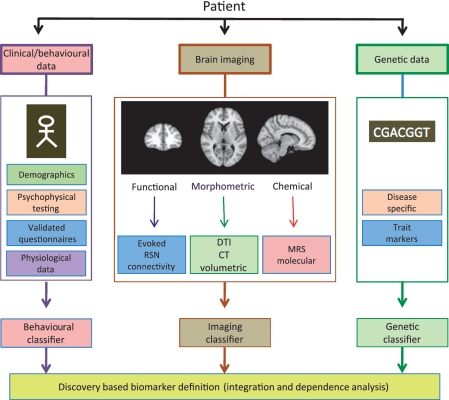
Figure 4.
Objective imaging assays. Current research for pain biomarkers includes brain imaging (Borsook et al., 2011a, b), clinical/behavioural approaches (Freynhagen et al., 2006; Scholz et al., 2009) and genetic markers (Tegeder et al., 2006). Each of these approaches has yielded some promising results, suggesting that specific classifiers can be defined that may be used alone or, more likely, in combination, as pain biomarkers. DTI = diffusion tensor imaging; RSN = resting state network; MRS = magnetic resonance spectroscopy.
Specific changes in psychophysical measures of pain have been detected clinically in Parkinson's disease. For example, using heat and laser pinprick paradigms in patients with Parkinson's disease in the OFF condition, patients with Parkinson's disease with central pain had lower thresholds for heat pain and laser pinprick than patients with Parkinson's disease with no central pain or control subjects. These effects were attenuated with l-DOPA treatment (Schestatsky et al., 2007). Similarly, another report showed that l-DOPA increases the pain threshold in Parkinson's disease as assessed by the RIII nociceptive flexion reflex (Gerdelat-Mas et al., 2007). Patients with Parkinson's disease also exhibited facilitation of temporal summation, a process in which the response to repeated painful stimuli is greater than to a single stimulus of the same intensity. Temporal summation is frequently enhanced in chronic pain, and is often used as an indicator of central sensitization. Patients with Parkinson's disease are more sensitive than normal controls to repeated painful stimuli, suggesting alterations in supraspinal inputs to pain modulatory systems (Perrotta et al., 2010).
Dopaminergic systems are involved in the modulation and integration of sensory information and the response to pain (Wolters, 2009; Juri et al., 2010). Pain symptoms increase and decrease with dopaminergic fluctuation. For example, in a Parkinson's disease patient cohort of >200, 47% reported pain and 45% therapy patients reported decrease (sensitivity 59.5% and specificity of 65% and a negative predictive value of 75%) in pain level with dopaminergic therapy (Stacy et al., 2010). Patients with Parkinson's disease reported increased unpleasantness in response to heat pain but only while ON medication, compared with the OFF state (Nandhagopal et al., 2010). Significantly, pain may be alleviated by adjustments of l-DOPA medications (Letro et al., 2009; Nebe and Ebersbach, 2009). Central pain may also be a result of dopaminergic loss, as is reported during dopamine agonist withdrawal: patients with Parkinson's disease who have their dopamine agonist stopped exhibit a withdrawal syndrome that is dose-dependent and includes pain (Rabinak and Nirenberg, 2010). Studies looking at other pain syndromes also support the role of dopaminergic systems in pain. The dopamine agonist pramipexole, a dopamine 3 receptor agonist, improved symptoms of patients with fibromyalgia (Holman and Myers, 2005). In addition, genetic variants of catechol-O-methyltransferase (COMT), which inactivates catecholamines including dopamine, are associated with different responses to acute and chronic pain (Andersen and Skorpen, 2009; Belfer and Segall, 2011).
Dopamine is centrally involved in CNS reward systems and reward and pain are at opposite ends of a behavioural motivational spectrum. Depression is common in pain conditions and vice versa. Depression has been reported to affect ∼45% of patients with Parkinson's disease, but prevalence varies across studies (Burn, 2002; Lemke, 2008). Thus, depression may also be a contributing factor to the pain symptoms reported in patients with Parkinson's disease (Lohle et al., 2009). A complex link exists between pain and emotions, and there is an overlap in pathways involved in reward and pain (Zubieta et al., 2001; Scott et al., 2006; Borsook et al., 2007). Dopamine regulated neurons have been implicated in motivational control both in reward and aversion and in defining motivational salience as it relates to rapid detection of important sensory information (Martin et al., 2010). Dopaminergic systems are implicated in both reward and pain (Zubieta and Stohler, 2009). The neuroanatomical and functional overlap between pain and brain circuitry involved in emotion/reward/motivation brain suggests integration and mutual modulation of these systems (Elman et al., 2011). As noted above, catechol-O-methyltransferase polymorphisms contribute to the interindividual variability in human pain phenotypes. Such genes may alter the brain's mesolimbic reward circuitry (Chen et al., 2009). Alterations in dopamine levels may contribute to alterations in reward function in chronic pain resulting in a ‘reward deficit state’ (Comings and Blum, 2000).
Alzheimer's disease and pain
Pain processing may be altered in dementias (Scherder et al., 2003; Schmidt et al., 2010) including Alzheimer's disease (Pickering et al., 2000). The issue is complicated by patients’ diminished ability to report pain because of cognitive deficits (Schmidt et al., 2010). A recent brain imaging study reports that pain perception and processing are not diminished in Alzheimer's disease (Cole et al., 2006). This is consistent with reports that sensory-discriminative components of pain are preserved even in advanced stages of Alzheimer's disease (Benedetti et al., 2004), while pain tolerance increases with disease severity (Benedetti et al., 1999). Other studies report similar findings: in studies comparing patients with Alzheimer's disease to age-matched subjects without dementia, patients with Alzheimer's disease consistently report lower pain intensity in response to painful stimuli, and have diminished pain affect (Scherder et al., 2001). Although there are data suggesting that the threshold for pain tolerance is markedly increased and the autonomic pain reaction is diminished in Alzheimer's disease (Rainero et al., 2000; Kunz and Lautenbacher, 2004), these results need to be evaluated in the context of altered autonomic function (Toledo and Junqueira, 2010) and delayed ability to respond. The prevalence of pain in the elderly in nursing homes is estimated to be between 40% and 80%; however, pain prevalence is difficult to assess in Alzheimer's disease because of the difficulty in measuring pain when there is cognitive impairment.
Alzheimer's disease is a double-edged sword when it comes to pain assessment. Pain affects cognitive function (Lee et al., 2010; Moriarty et al., 2011) and cognitive function also affects pain assessment and pain treatment because the primary method for pain assessment is still patient reporting (Licht et al., 2009). Thus, it is difficult, if not impossible, to obtain reliable subjective measures of pain in subjects with advanced Alzheimer's disease. As discussed in the section ‘Pain in the unconscious or non-communicative patient’ below, the same difficulty is present in treating or studying pain in patients who are neurologically compromised for other reasons (i.e. non-communicative brain injured patients, vegetative state) (Owen et al., 2006; Boly et al., 2007) and in preverbal infants. However, as noted above, new data obtained from imaging studies of experimental pain in patients with Alzheimer's disease show that presumed pain perception (involvement of sensory and emotional systems) is not diminished despite morphological and functional changes in cortical regions. Indeed, there is a greater amplitude and duration of pain-related activity in sensory, affective and cognitive processing regions in patients with early onset Alzheimer's disease compared with age matched controls (Cole et al., 2006). Regions of the brain like the thalamic nuclei (Rudelli et al., 1984) and the overall sensory/discriminative cortex appear relatively unaffected by Alzheimer's disease (Farrell et al., 1996). However, loss in limbic structures, hippocampus and prefrontal cortex (Hyman et al., 1984) may explain the deterioration of affective aspects and emotions in cognitively impaired elderly adults suffering from Alzheimer's disease (Pickering et al., 2000; Greicius et al., 2003). Thus, assessing pain at different stages of Alzheimer's disease progression may provide an opportunity to differentiate between pain-related activation in sensory systems (i.e. intensity) and emotional processing (i.e. affect, unpleasantness) of pain. Altered pain processing may also reflect diminished modulation of pain at the level of regions such as the periaqueductal grey (Parvizi et al., 2000), which could explain the perseveration of brain responses reported in imaging studies (Cole et al., 2006). Thus, patients with Alzheimer's disease may not be able to remember, interpret, respond to, or report pain in a normal fashion and may exhibit abnormal behaviours as a result, including agitation, aggression and other affective changes (Benedetti et al., 2004; Shega et al., 2007).
Without objective tests or biomarkers, evaluating pain in any population is complex and variable. In the Alzheimer's disease group, because of the added complication of diminished cognitive processing, pain assessment is even more difficult, with about a third of subjects unable to complete a number of pain assessment tools (Krulewitch et al., 2000). A number of instruments have been developed to measure pain in patients with decreased cognitive function including evaluating facial grimacing (Shega et al., 2008), pain intensity scale (Krulewitch et al., 2000); Mahoney Pain Scale (Mahoney and Peters, 2008) and Pain Assessment in Advanced Dementia (Zwakhalen et al., 2011). These are all subjective measures.
Huntington's disease and pain
The prevalence of pain in Huntington's disease is unknown. An initial case report describes severe pain in two patients with this condition (Albin and Young, 1988). In a more recent study, 11 of 19 patients with Huntington's disease had pain, with altered pain perception to pinprick, touch and temperature in some subjects (Scherder and Statema, 2010). In Huntington's disease, alterations in peripheral tissue (muscle) may be due to alterations in mitochondrial dysfunction and energy metabolism (Sassone et al., 2009). Exercise-induced muscle pain has been described in a marathon runner who subsequently developed Huntington's disease (Kosinski et al., 2007), suggesting that it may be an early unrecognized symptom of the disease. Significantly, there is a 10–25% prevalence of diabetes in patients with Huntington's disease (Farrer, 1985); diabetes is a relatively common cause of neuropathic pain in a subset of diabetics, but we are unaware of any reports related to how this may impact patients with Huntington's disease. As discussed above, pain and depression are frequently co-morbid conditions; the prevalence of severe depression is twice as high in Huntington's disease as in the general population, reportedly as high as 40% (Folstein et al., 1985; Paulsen et al., 2005).
Huntington's disease is an autosomal dominant progressive neurodegenerative disorder (Ross and Tabrizi, 2011) that affects the brain, primarily the basal ganglia where there is extensive atrophy of the caudate, globus pallidus and putamen, with consequent changes in motor, cognitive, and emotional functioning (Grove et al., 2003). The disease also affects the thalamus (particularly the ventrolateral nucleus), a region considered to play a role in sensory perception (Ro et al., 2007). The basal ganglia are involved in both acute and chronic pain processing (Borsook et al., 2010) and have a prominent role in sensorimotor integration, which is altered in Huntington's disease, in which these regions may actually become deafferented (Abbruzzese and Berardelli, 2003). Basal ganglia activations are common in functional imaging studies of pain (Borsook et al., 2010), thus it would not be surprising if the compromised function of the basal ganglia in Huntington's disease led to alterations in pain processing. Abnormal cortical and subcortical activation in patients with Huntington's disease following passive sensory stimulation as evaluated by functional PET studies (Boecker et al., 1999). Although there are no published studies of pain processing in Huntington's disease, it is known that experimental lesions of the caudate impair pain avoidance, indicating impaired pain processing (Koyama et al., 2000).
Ataxia and pain
Machado–Joseph disease is the most common spinocerebellar ataxia, also known as spinocerebellar ataxia type 3 (Rub et al., 2008) and is a neurodegenerative disease that includes ataxia, opthalmoplegia and peripheral neuropathy (D'Abreu et al., 2010). Although classically described as affecting the cerebellum, it affects a number of other brain regions including brainstem, basal ganglia, thalamus and cerebral cortex (D'Abreu et al., 2010). In a small study, nearly 50% of patients reported chronic pain including muscle cramps (Franca et al., 2007). Muscle excitability abnormalities occur in >80% of these patients and peripheral nerve damage correlates with the extent of muscle fasciculations (Franca et al., 2008). In addition, widespread neurodegeneration is observed in somatosensory (although pain is not specifically delineated) as well as primary sensory systems with alterations in dopaminergic and cholinergic systems (Rub et al., 2008). Sensory symptoms including pain are observed across subtypes of spinocerebellar ataxia, with 48% of subjects complaining of pain or discomfort (Schmitz-Hubsch et al., 2008).
One emerging concept is that the cerebellum may play a role in chronic pain (Moulton et al., 2010), based on its complex role in cognitive and affective processing (Stoodley and Schmahmann, 2010). Current data suggest that the cerebellum is an integrator of multiple effector systems including affective processing, pain modulation, as well as sensorimotor processing.
Neuromuscular diseases
Amyotrophic lateral sclerosis
Chronic pain is common in neuromuscular diseases, including amyotrophic lateral sclerosis (Wijesekera and Leigh, 2009), where the prevalence is 15–20% (Franca et al., 2007). In a small case series of amyotrophic lateral sclerosis, pain was the first symptom manifested in >20% of patients (de Castro-Costa et al., 1999), with the arms as the primary affected region. Cases of chronic central pain have also been reported in patients with amyotrophic lateral sclerosis (Drake, 1983). One study reported mild (29% of patients) and severe (6%) depression in amyotrophic lateral sclerosis (Atassi et al., 2010), which may be a major contributor to the pain manifestations in this disease.
Although amyotrophic lateral sclerosis predominantly affects the motor system, sensory changes are present, including paraesthesias. A multi-centre European study reported generalized sensory changes in amyotrophic lateral sclerosis (Pugdahl et al., 2007), but made no specific reference to pain. Quantitative sensory testing performed to evaluate function in unmyelinated fibre systems using heat stimuli for thresholds of cold and warm sensation found no differences between amyotrophic lateral sclerosis and control subjects in cold or warm thresholds (Deepika et al., 2006); however, no suprathreshold testing was performed.
Is the underlying disease in amyotrophic lateral sclerosis a cause of pain? Both the animal (Chen et al., 2010) and human (Gerber et al., 2011) literature suggest that a muscle neuropathic-like pain syndrome exists in amyotrophic lateral sclerosis. Imaging studies have reported functional deficits in secondary/higher order sensory processing areas in amyotrophic lateral sclerosis (Lule et al., 2010), but the authors do not describe changes in response to pain.
Central nervous system damage
Stroke
It is well-documented that strokes affecting the CNS, particularly the structures along the spino-thalamocortico-tract (spinothalamic tract, lateral thalamus, thalamic–parietal projections), produce central pain syndromes (central post-stroke pain) (Bowsher et al., 1998). Despite the fact that the classic description of thalamic stroke producing pain was published >100 years ago (Dejerine and Roussy, 1906), the mechanisms underlying the severe, spontaneous, burning pain that occurs with thalamic stroke remain unclear. However, it is clear that damage to specific regions of the brain produces central pain. In operculo-insular pain, a central pain syndrome resulting from posterior parasylvian lesions, thermal and pain sensations are altered and laser-evoked potentials to thermo-nociceptive stimuli are abnormal (Garcia-Larrea et al., 2010). A pseudothalamic syndrome, producing pain asymbolia (absent or inadequate emotional responses to painful stimuli) (Berthier et al., 1988), results from a stroke producing damage to the posterior insula region (Masson et al., 1991). This is consistent with evidence indicating a significant role of the posterior insula in processing of thalamic pain (Craig, 2000).
A number of important findings related to central pain shed light on pain processing: (i) damage to the classic pain sensory systems (spinothalamic tract) seems to be pivotal in producing central pain syndromes resulting from stroke (Hong et al., 2010). Loss of grey matter in chronic pain has been well described and the altered connectivity resulting from either direct damage or indirect changes may contribute to a central pain syndrome. In contrast, most patients with a loss of cortical sensory evoked potentials and a CT scan finding of ischaemic lesion of the posterolateral thalamus do not exhibit central pain (Wessel et al., 1994); (ii) in thalamic pain, there is increased excitability of thalamic regions. Although there may be diminished activation in the thalamus at rest, hyperactivity (including bursting activity) is found in central post-stroke pain, suggesting derangement of an oscillatory pattern inside a sensory corticothalamocortical reverberatory loop (Lumer et al., 1997); and (iii) other changes including alterations in neural connectivity (deafferentation) (Boivie et al., 1989), decreases in opioid receptor concentrations (Jones et al., 2004; Maarrawi et al., 2007), damage to lateral nociceptive thalamoparietal fibres (Schmahmann and Leifer, 1992), and altered chemistry (evaluated by magnetic resonance spectroscopy, Veldhuijzen et al., 2007) are present in central pain. Functional imaging studies of a patient with thalamic pain suggest that the release of activity in anterior cingulate and posterior parietal regions is a plausible mechanism for central pain (Seghier et al., 2005). The article provides evidence for specific damage to the thalamoparietal fibres (using 3D diffusion tensor imaging), consistent with clinical studies of thalamic pain (Demasles et al., 2008) and with increased responses to pain in the insula, putamen and parietal lobe on the affected side. The latter have been shown to be involved in analgesia (Borsook et al., 2010; Mhuircheartaigh et al., 2010).
Syringomyelia
Syringomyelia and its sister disorder, syringobulbia, are disorders associated with Arnold–Chiari malformation (Koyanagi and Houkin, 2010) or spinal cord trauma (Schurch et al., 1996). Syringomyelia and syringobulbia frequently produce pain (Todor et al., 2000; Greitz, 2006; Hatem et al., 2010) and result in an increasing cavity of CSF within the spinal cord tissue (i.e. not involving the central canal). Differential alterations in CSF flow and pressure seem to be important components of an underlying mechanism that initiates or potentiates the evolution of the syrinx (Greitz, 2006; Martin et al., 2010). Pain is present when the syrinx presses on the crossing fibres of spinothalamic tracts.
As in thalamic stroke, the disease affects the spinothalamic tract at the level of the spinal cord in the dorsolateral quadrant (or brainstem in syringobulbia). In patients with neuropathic pain, higher average daily pain intensity is correlated with greater structural damage to the spinal cord (Hatem et al., 2010). Pain descriptors (describing spontaneous pain or paraesthesias) were negatively correlated with fibre reconstruction and were clearly different in patients with both spontaneous and evoked pain compared to patients with spontaneous pain only. Patients with spontaneous pain only had more severe spinal cord damage.
Traumatic brain injury
Multiple pain syndromes have been described in different patients following traumatic brain injury with diffuse axonal injury (Raghupathi and Margulies, 2002), including neuropathic pain, central pain, and thalamic pain (Formisano et al., 2009). Pain after traumatic brain injury is common (Walker, 2004). Headache following head trauma is observed in >50% of patients and has received increasing attention as it relates to combat blast-induced injuries (Vargas, 2009; Risdall and Menon, 2011). The prevalence of chronic pain following mild traumatic brain injury (75%) is higher than following severe traumatic brain injury (32%) (Nampiaparampil, 2008). Significantly, in traumatic brain injury, chronic pain is independent of post-traumatic stress disorder and depression, but is frequently associated with other brain changes including auditory and visual deficits (Nampiaparampil, 2008), autonomic dysfunction (Kanjwal et al., 2010), insomnia (Zeitzer et al., 2009) and psychiatric disease (Halbauer et al., 2009). Traumatic brain injury produces diffuse axonal injury. Diffusion tensor imaging of mild blast injury veterans has not shown any differences compared with veterans without a history of traumatic brain injury (Levin et al., 2010). Little information is available on assessing and treating pain in traumatic brain injury (Dobscha et al., 2009).
The aetiology of headache is unknown. Its immediate trigger may be the tearing of trigeminal fibres that innervate the meninges. Small fibre (calcitonin gene related peptide positive) meningeal nerves infiltrate the calvarial bones of the skull and the nerves may be tethered predominantly in the calvarial sutures (Kosaras et al., 2009). With impact producing movement of the meningeal surface, these nerves are stretched or torn. In traumatic brain injury, disturbances in brain perfusion in the frontal and temporal lobes correlate with the headache (Lyczak and Lyczak-Rucinska, 2005).
Multiple sclerosis
Chronic pain is experienced in 40–75% of patients with multiple sclerosis (Kenner et al., 2007; Bermejo et al., 2010; Solaro and Messmer Uccelli, 2010). Multiple sclerosis, an inflammatory, demyelinating autoimmune disease of the CNS, has been associated with multiple pain syndromes including extremity pain, trigeminal neuralgia, Lhermitte's sign, painful tonic spasms, back pain and headache (O'Connor et al., 2008). While it seems as though the presentation of pain should be correlated with sites of demyelination, a study evaluating CNS pathways in patients with multiple sclerosis with and without pain found no association between chronic pain and the site of demyelination. Increased pain in multiple sclerosis correlates with depression, spinal cord involvement at the onset and the presence of spinal cord lesions (Grau-Lopez et al., 2011). Thus the aetiology of pain may involve a more complex phenomenon including local cytokine processes or alterations in white and grey matter integrity of networks that produce pain.
Damage to pain pathways in multiple sclerosis may result from inflammatory processes involving glia and cytokines (Merson et al., 2010), which are also a potential mechanism of central pain (Graeber, 2010; Nakagawa and Kaneko, 2010). Approaches using anti-cytokine gene therapy reportedly decrease sensory dysfunction in multiple sclerosis (Sloane et al., 2009). Disease modifying medications targeted at the immune system do not seem to provide significant pain relief.
Metabolic diseases
Fabry's disease and pain
Fabry's disease is an X-linked recessive lysosomal disease caused by α-galactosidase A deficiency. Although Fabry's disease-related pain syndromes include migraine (Albano et al., 2010), distal limb pain is a common presenting feature (Pagnini et al., 2011) and is the most common feature in childhood. An important observation is that the gender effect on pain prevalence in Fabry's disease is opposite to that in most kinds of pain: pain is more prevalent in males (80%) than females (65%) with Fabry's disease, although it interfered more with daily activities in females (Hoffmann et al., 2007). In adults, Fabry's disease-associated alterations in brain structure are well established (Nill et al., 2006), including cerebrovascular events relating to accumulation of lysosomes in several tissues, particularly vascular endothelium and smooth muscle cells (Fellgiebel et al., 2006). These vascular events primarily affect the posterior circulation, resulting in damage to periventricular white matter, brainstem, cerebellum, and basal ganglia in particular (Clavelou et al., 2006). In addition, T1-weighted MRI studies have identified pulvinar calcification in particular as a structure with abnormalities in Fabry's disease (Moore et al., 2003).
The mechanisms by which α-galactosidase A deficiency causes these physiological abnormalities are poorly understood. Small fibre reductions are noted in skin biopsies in symptomatic and asymptomatic individuals (Liguori et al., 2010) as are abnormalities in autonomic function (Moller et al., 2009). Similarly, a mouse model of Fabry's disease exhibits decreased density of both non-myelinated and thinly myelinated fibres (Onishi and Dyck, 1974; Rodrigues et al., 2009). Glycolipid accumulation in the dorsal root ganglion or nerves may explain phenomena such as shooting pains (Gadoth and Sandbank, 1983). Enzyme replacement therapy with α-galactosidase A significantly reduces pain but can take a few years to be effective (Schiffmann et al., 2001; Hoffmann et al., 2007).
Tumours and pain
Neurofibromatosis
Neurofibromatosis is an autosomal dominant neurocutaneous disorder subdivided into neurofibromatosis 1 (NF1), neurofibromatosis 2 (NF2) and schwannomatosis (Lu-Emerson and Plotkin, 2009). NF1 is the most common neurogenetic disorder (Lu-Emerson and Plotkin, 2009) where the most common lesion is a benign tumour—the neurofibroma. NF1 tumours may develop anywhere in the nervous system including the skin and PNS and usually produce ‘unmanageable pain’ (Huson et al., 2011). NF2 tumours occur in the CNS, including bilateral vestibular schwannomas and meningiomas. Schwannomatosis is characterized by multiple non-vestibular, non-intradermal schwannomas and chronic pain. Pain is the most common presenting feature of schwannnomatosis (MacCollin et al., 2005) and paediatric plexiform (51%) neurofibromas (Serletis et al., 2007). There is an increased incidence of itch in NF1, which may be related to increases in mast cells in the skin (Nurnberger and Moll, 1994). Common sites for neurofibromatosis-related tumours include intraspinal, paraspinal, brachial plexus, femoral nerve and sciatic nerve. Pain may become manifest as a result of compression (e.g. with foraminal tumours). These tumours are distinct from benign schwannomas, which are common tumours of peripheral and cranial nerves, also presenting with pain, neurological deficits and enlargement of a pre-existing peripheral nerve sheath tumour in NF1 (Valeyrie-Allanore et al., 2005).
These tumours illustrate two processes involved in pain: (i) compressive neuropathy (Corey, 2006), where there is nerve sheath involvement (Wang et al., 2005); and (ii) the contribution of inflammatory mediators (through mast cells) to pain (Staser et al., 2010). In neurofibromatosis-1, these inflammatory mediators induce vascular changes that may lead to vasculo-occlusive disease (Lasater et al., 2010), producing microinfarcts in the vasa vasorum that may contribute to painful symptoms.
Peripherally initiated changes in central nervous system pain processing
Each of the three clinical examples presented in this section provides insight into how changes in peripheral pain pathways impact CNS pain processing. Complex regional pain syndrome illustrates how peripheral nerve damage may transform brain systems; congenital insensitivity to pain provides a dramatic example of the profound consequences of loss of peripheral pain sensation; and migraine demonstrates that a common neurological disease that involves the trigeminal system is not simply an intermittent manifestation of pain and associated symptoms, but may alter brain systems.
Complex regional pain syndrome
Perhaps no pain condition represents the centralization of pain more clearly than complex regional pain syndrome (CRPS; Janig and Baron, 2002; Bruehl, 2010). Following a peripheral nerve injury, usually trivial, a series of progressive changes may take place that include some or all of the following: (i) spreading pain that may cross the midline and involve the whole body, suggestive of centralization of sensory processing at the thalamus or higher centres (Maleki et al., 2000); (ii) autonomic changes suggestive of hypothalamic changes (Gradl and Schurmann, 2005); (iii) neglect-like symptoms suggestive of parietal lobe dysfunction (Galer and Jensen, 1999); and (iv) in some cases, dystonias or other motor changes suggestive of potential basal ganglia involvement [see review by Maihofner et al. (2010)]. Taken together, these findings implicate alterations in CNS processing, a notion supported by functional brain imaging studies that demonstrate reproducible alterations in adult (Geha et al., 2008; Maihofner et al., 2010) and paediatric (Lebel et al., 2008) patients with CRPS.
In CPRS, persistent pain and subsequent progressive changes in the brain (namely automonomic, cognitive, central sensitization, hemineglect) are observed following mild peripheral nerve injury. This and other brain imaging studies suggest that all patients with neuropathic pain have alterations in brain systems that may result in cognitive and other behavioural changes, which may go unrecognized because they are not as prominent as the pain symptoms. Longitudinal MRI studies of paediatric patients with CRPS indicate that the brains of these children continue to exhibit significant differences from normal controls after symptoms resolve (Lebel et al., 2008).
Congenital insensitivity to pain
This is a rare and severe autosomal recessive condition (Rosemberg et al., 1994) that leads to self-mutilation in early life. The underlying pathophysiology is alteration of pain and temperature perception due to involvement of the autonomic and sensory nervous system involving small-calibre (A-delta and C) nerve fibres (Danziger and Willer, 2009). In addition, bone fractures, scars, osteomyelitis, joint deformities, limb amputation and mental retardation are common in individuals with congenital insensitivity to pain (Thrush, 1973). These patients lack nerve growth factor-dependent unmyelinated (C-) and thinly myelinated (Aδ-) fibres (Indo, 2010). Clinically, congenital insensitivity to pain is characterized by insensitivity to all modalities of pain, with the possible exception of neuropathic pain, since some case reports describe patients with congenital insensitivity to pain with burning ‘pain’ following post-herpetic neuralgia (Tomioka et al., 2002). The disorder results from mutation (deletion) in the SNC9A gene, which encodes the Na1.7 channel (Cox et al., 2010; Kurban et al., 2010). This is of interest since activating mutations in SNC9A produces severe pain due to gain of channel function in paroxysmal extreme pain disorder and primary erythermalgia (Choi et al., 2011).
Congenital insensitivity to pain is an important human model for pain genetics, representing a functional ‘knockout’. However, the ethics of studying pain in individuals with congenital insensitivity to pain are complex, since profound mental retardation frequently makes it difficult to determine whether or not they suffer even without a behavioural response to pain per se (Borsook and Becerra, 2009a, 2009b). In a report on neural correlates of empathy in congenital insensitivity to pain, brain activations to observed pain were reported in the anterior mid-cingulate cortex and anterior insula (regions of the so-called ‘shared circuits’ for self and other pain).
Migraine/headache
Episodic migraine is one of the common primary headache disorders (Robbins and Lipton, 2010). Until relatively recently, it was considered an episodic pain syndrome without effects on CNS processing. Current evidence suggests that the brains of patients with episodic migraine are significantly different from healthy controls. For example, even during the interictal period, these brains exhibit increased cortical excitability to pain (Moulton et al., 2011), to light (Denuelle et al., 2011), and to smell (Demarquay et al., 2008), as well as altered brainstem processing (Moulton et al., 2008) and associated changes in grey matter volume (May, 2009). Thus, migraine is now considered a brain disease and not simply a recurrent acute pain syndrome. Like many chronic pain syndromes, migraine predominantly affects females.
Migraine provides an interesting pain model: (i) it provides a clear-cut model of gender-related pain issues, generally uncomplicated by prior history that contributes to chronic pain (e.g. post-traumatic stress disorder); (ii) migraine is a disease associated with progression or chronification (Bigal and Lipton, 2011), a process that has an unknown basis except that it may be produced by medication overuse (Jonsson et al., 2011); although chronic daily headache affects a relatively small per cent of the migraine population, it is these patients that most frequently seek medical attention (Manack et al., 2011); (iii) migraine is an ideal model of pain systems involved in central sensitization since with the onset of the headache, progressive central sensitization is associated with allodynia in the face, body and limbs (Burstein et al., 2010) and for understanding altered (ineffective) modulatory circuits in pain (Moulton et al., 2008) that is observed in pain processing in humans (Becerra et al., 2006; Seifert et al., 2009); (iv) diffuse brain systems are involved including the temporal pole (Moulton et al., 2010) and areas involved in modulation of sensory systems that may produce pain [e.g. light (Noseda et al., 2010)]; and (v) cortical spreading depression, a process involved in both stroke and migraine is now understood to drive trigeminovascular pain neurons (Zhang et al., 2011).
Pain in the unconscious or non-communicative patient
Altered states of consciousness pose a huge dilemma in diagnosis (misdiagnosed in 43% of cases) and in determining whether a patient is experiencing pain and suffering (Coleman et al., 2009). Assessing acute and chronic pain in unconscious (comatose) patients or those who cannot communicate is a critical problem. In the USA, between 100 000 and 280 000 patients are believed to be in a minimally conscious state (Strauss et al., 2000). Recent neuroimaging studies have reported conscious awareness and potential cognition in patients in a vegetative state (Owen et al., 2006, 2007; Monti et al., 2010). Less is known about the ability of such patients to perceive pain (as opposed to nociception), and studying this issue presents a clinical and ethical dilemma. In a functional brain imaging study of pain in response to median nerve stimulation in patients in a minimally conscious state, patterns of activation similar to controls were reported (thalamus, S1 and the secondary somatosensory or insular, frontoparietal and anterior cingulate cortices), suggesting that pain perception may be intact in these patients (Boly et al., 2008). Newer technologies may be able to determine patterns of activation in pain networks at the bedside (Becerra et al., 2008, 2009a). Recommendations on how to best evaluate and treat such patients have been drawn up by nursing groups (Herr et al., 2006; Pudas-Tahka et al., 2009), anaesthesia/critical care (Gelinas et al., 2006) and neonatologist (Duhn and Medves, 2004) services. However, none of these approaches include objective measures of pain, and rates of misdiagnosis of patients in the minimally conscious state (motor cortex stimulation; some evidence of awareness of self and environment) or vegetative state (wakefulness without awareness) (Fins et al., 2007). Attempts have been made to provide objective measures of cognition and pain using neuroimaging. PET studies show activation in regions of the pain network (thalamus, S1 and the secondary somatosensory or insular, frontoparietal, and anterior cingulate cortices) in patients with motor cortex stimulation and vegetative state (at a lower level) and demonstrate loss of functional connectivity between S1 and frontoparietal regions in both conditions (Boly et al., 2008). In an earlier study, pain stimulation activated the midbrain (in the same regions activated by somatosensory stimulation), contralateral thalamus, and primary somatosensory cortex in all vegetative state patients, even in the absence of cortical evoked potentials (Laureys et al., 2002). The activation in the primary cortex seems to be isolated and dissociated from higher order associative cortices in vegetative state.
What do these functional MRI studies tell us about pain processing in unconscious/non-communicating patients? Measures of pain processing in the brain across the spectrum from awake/conscious processing to pain processing in unconscious anaesthetized states provide some insight (Brown et al., 2010). As noted in the reports above, in comatose patients (vegetative or minimally conscious state), pain stimuli activate well-defined pain pathways in the brain. What is unclear is if the patient comprehends or suffers from pain. Behavioural scales have been used to try to evaluate pain in comatose patients, including the Nociceptive Coma Scale (Schnakers et al., 2010). While neuroimaging studies may provide data relating to brain regions involved in aversion and affective dimensions of pain, currently we can utilize functional MRI methods to define nociceptive processing but not pain itself. These data indicate that a better understanding of pain perception in the minimally conscious or vegetative state is a clinical and ethical imperative. Further studies are required, as well as the development of new neuroimaging modalities that can be applied easily at the bedside.
Pain and strange/unexplained symptoms
There is an unfortunate tendency to define patients as ‘crazy’ if their pain symptoms are unusual or clinical evaluation yields a non-classical finding. Examples of these atypical presentations include non-dermatomal sensory deficit in patients who have pain (Mailis-Gagnon et al., 2003; Egloff et al., 2009; Mailis-Gagnon and Nicholson, 2010) and altered manifestations of phantom sensations with pain (Borsook et al., 1998). Other more complex phenomena include neglect-like symptoms observed in patients with CRPS and non-CRPS neuropathic pain (Galer and Jensen, 1999; Frettloh et al., 2006). In addition, the evolution of pain in depressed patients with no prior history of pain or non-traumatic post-traumatic stress disorder provides further insight into the complexity of alterations in brain circuits producing pain. Some patients display a recognizable illness without any pathology, making it difficult to determine if the illness is real or simulated. Similarly puzzling are instances of delayed onset of pain following an insult and spontaneous resolution of pain, which may take place over a short time period even if pain has been present for years (Schott, 2001). Finally, the effect of opioids in chronic pain is often counter-intuitive. For example, they have relatively limited efficacy (a decrease of 13 points on a 100 point scale) (Eisenberg et al., 2006), and, in some neurological conditions, opioids either exacerbate pain or diminish future treatment efficacy. For example, opioids induce migraine chronification (Bigal and Lipton, 2009). Opioids clearly alter brain systems (Upadhyay et al., 2010); however, the impact of these changes on chronic pain is not understood. Objective measures of pain would help clarify many of these issues and aid in the assessment of clinical efficacy of analgesics.
Brain-based restorative approaches for chronic pain
Despite trials of a number of approaches, chronic pain is largely refractory to treatment. Chronic pain associated with neurological disease has varied presentations and temporal profiles (Fig. 5) that add to the complexity of treatment approaches. Below, I discuss therapeutic approaches that may be more logical in light of the new understanding that chronic pain is a disease of the brain. The over-riding goal of these approaches is to restore brain networks to states that are adaptive. This discussion is not meant to suggest specific therapies for pain in particular neurological diseases, but rather to spur further exploration of new chronic pain treatment options that represent a fairly radical departure from our previous therapeutic approach.
Figure 5.
Schematic of altered pain processing in neurological disease. The figure summarizes altered pain processing in examples of neurological disease, as well as underlying mechanisms that contribute to chronic disease-related pain. In most diseases, multiple regions are affected (as opposed to secondary effects such as centralization of pain following a peripheral nerve injury). Whether or not the disease is a primary or secondary cause of pain, pain itself drives an altered brain state (Fig. 1). Damage anywhere along the pain pathway from peripheral nerve to spinothalamic tracts and more central pathways including thalamus and thalamocortical projections may result in neuropathic pain. In the figure, damage as indicated by red crosses or abnormal activation in pain pathways (circles), contributes to other changes in brain systems.
Motor training
Motor and sensory systems are well integrated (Flor and Diers, 2009). In the setting of chronic pain, a number of ‘motor’ treatments have been used, including physical therapy, mirror movement in patients with chronic pain including phantom limb pain (Ramachandran and Altschuler, 2009; Ramachandran et al., 2010) and complex regional pain syndrome (Selles et al., 2008), motor cortex stimulation (discussed below) and motor imagery (Moseley, 2006). It has been suggested that phantom limb pain is caused by motor cortex dysfunction that is the result of dissociation between motor and sensory representations (Karl et al., 2001; Sumitani et al., 2010). Through motor training, phantom-limb patients may decrease their pain, presumably by resetting these altered sensory–motor dissociations. Recent work has indicated that mirror movement differentially activates the sensory cortex in amputees with and without phantom pain, further implicating altered functional connectivity (Diers et al., 2010). Related to this is the use of smart limb prosthetics (Marasco et al., 2011) or sensory feedback that may utilize sensory motor feedback loops to decrease phantom limb pain (Weiss et al., 1999; Flor, 2002).
Brain ‘shocks’—insights into pain control
Electroconvulsive therapy has been suggested to be of use in chronic neurological diseases such as Parkinson's disease—on both psychiatric manifestations and motor systems (Popeo and Kellner, 2009). Electroconvulsive therapy has also been recommended for chronic pain (Fukui et al., 2002; Wasan et al., 2004; Usui et al., 2006; Suda et al., 2008; Suzuki et al., 2009). Thalamic blood flow is reportedly normalized following electroconvulsive therapy in patients with CRPS (Fukui et al., 2002). The interesting application in many of the neurological orders discussed relates to electroconvulsive therapy's strong antidepressant effects (Merkl et al., 2009), mitigating the use of medications that may produce side-effects.
A possible chemical ‘equivalent’ of electroconvulsive therapy is the N-Methyl-D-aspartic acid (NMDA) antagonist ketamine. Ketamine has been used to treat chronic pain and depression (Berman et al., 2000; Zarate et al., 2006). In chronic pain, higher doses seem to be more effective and reports have suggested that very high or continued dosing (anaesthetic levels) may reverse conditions such as CRPS (Kiefer et al., 2007; Becerra et al., 2009b). Indeed in uncontrolled trials, anaesthetic levels of ketamine reversed pain in 20 patients with chronic pain, producing complete relief in all patients at 1 month; pain relief persisted in 17 of these patients at 3 months, and in 16 at 6 months (Kiefer et al., 2008). Clearly, controlled trials are required to verify these findings, but they raise the exciting possibility that ketamine alters brain systems in a significant manner in a highly resistant population. It remains unclear if the changes are the result of ‘reordering’ or resetting neural networks or of ketamine-induced lesions at a cellular level that somehow result in recovery. This approach is clearly not recommended in patients with neurodegenerative disease, particularly those with hyperexcitability-related pathogenesis, in which ketamine could potentially exacerbate ongoing neurotoxicity.
Central nervous system lesions—do they inform us more than they provide effective pain control?
Neurosurgical approaches have included stereotaxic surgery (Weigel and Krauss, 2004) and high intensity focused ultrasound of various brain structures including the thalamus (Martin et al., 2009). Cingulotomy is perhaps the classic neurosurgical ablative technique reported to provide pain control (Wilkinson et al., 1999), but like other neurosurgical procedures for pain, claims for its efficacy are based on class III evidence (i.e. possibly effective) (Cetas et al., 2008). Subjects report ‘the pain is the same but they don't care’, consistent with the proposed role for the cingulate cortex in rank ordering the salience of incoming stimuli in animal (LaGraize et al., 2006) and human studies (Williams et al., 2004). Following cingulotomy (for a non-pain disorder), pain affect in response to noxious heat and cold was altered (Davis et al., 1994), suggesting that either this area is involved in suppression of pain affect or involved in rank ordering stimulus salience (De Martino et al., 2009). In patients undergoing cingulotomy, microelectode single unit measures of cingulate neurons following reward-based stimuli revealed decrease in neuronal activity predicting movement. However, following ablation, patients made more errors (Williams et al., 2004). Morphine has been postulated to have a similar effect on modulation of affect by the cingulate cortex (LaGraize et al., 2006). Clearly any lesion of the brain will alter the dynamics of brain function both at a local (brain region) and at a systems level (through afferent and efferent connections)
Brain stimulation
Stimulation techniques, whether extracranial (e.g. transcranial magnetic stimulation) or intracranial (deep brain stimulation or motor cortex stimulation), have the benefit of being reversible. Although small case studies of deep brain stimulation reported successful outcomes (Owen et al., 2006), these were not prospective. A meta-analysis of deep brain stimulation for pain (Bittar et al., 2005) reported that the long-term pain alleviation rate was highest with deep brain stimulation of the periventricular/periaqueductal grey matter (79%), or the periventricular/periaqueductal grey matter plus sensory thalamus/internal capsule (87%). Stimulation of the sensory thalamus alone was less effective (58% long-term success) (Bittar et al., 2005). In the same meta-analysis, stimulation was successful in ∼50% of those with post-stroke pain, and 58% of patients permanently implanted with deep brain stimulation devices achieved ongoing pain relief. Even higher rates of success were seen with phantom limb pain and neuropathies. To date, stimulation sites have included the ventroposterolateral thalamus and the periaqueductal grey region. Motor cortex stimulation has been proposed for the treatment of chronic pain (Lefaucheur et al., 2009). Deep brain stimulation of the subthalamic nucleus has had a landmark effect in Parkinson's disease, but its effects on pain processing also seem potentially useful. In one study, chronic pain was reduced following deep brain stimulation, but pain sensitivity to quantitative sensory testing was unaltered (Gierthmuhlen et al., 2010). In another study, early pain relief was observed in 20 of 23 patients (Kim et al., 2008). Neuroimaging studies have suggested that deep brain stimulation targeting the ipsilateral posterior inferior hypothalamus might be effective for chronic cluster headache; this is now an established treatment for intractable cases (Leone, 2006; Leone et al., 2008).
Motor cortex stimulation is another intracranial approach that has been used to treat refractory neuropathic pain (Lefaucheur et al., 2009) with substantial effects (48% of subjects reported decreased pain intensity). In a 4-year outcome evaluation, 10% of subjects rated the benefit as excellent (>70% pain relief), 42% as good (40–69% relief), 35% as poor (10–39%) and 13% as negligible (0–9%). Intake of analgesic drugs was decreased in 52% of patients in this study (Nuti et al., 2005). Given the interactions between sensory and motor systems and the fact that pain may inhibit motor cortex function (patients may limit their movement) (Farina et al., 2003), such motor function-based approaches may ‘unglue’ the status quo via mechanisms that may include activation of corticobasal ganglia thalamic loops or modulation of the pain.
A relatively recent approach is the use of transcranial magnetic stimulation for chronic pain (Lefaucheur, 2008), migraine (Lipton and Pearlman, 2010), spinal cord injury pain (Defrin et al., 2007) and traumatic neuropathic pain (Schwenkreis et al., 2010). Transcranial magnetic stimulation is currently US Food and Drug Administration (FDA) approved for depression (Schonfeldt-Lecuona et al., 2010). Experimental use in neurological diseases including Alzheimer's disease (Bentwich et al., 2011) and Huntington's disease (Medina and Tunez, 2010) suggests transcranial magnetic stimulation can induce improvement in some features of these diseases, including depression. In Parkinson's disease (Baumer et al., 2009), cerebellar stimulation resets or improves tremor (Ni et al., 2010), suggesting that transcranial magnetic stimulation may have long-lasting effects on cerebellar outputs/cortical excitability (Popa et al., 2010). Further studies are needed to determine its potential clinical utility in neurological diseases with pain.
Centrally active drugs developed for neurological disease and their potential role in pain therapeutics
Specific pharmacotherapies developed for many of these neurological conditions have been assessed for their ability to treat chronic pain (Finnerup et al., 2010; Haanpaa et al., 2011). The most notably successful are the anti-epilepsy drugs, but antidepressants, membrane stabilizers and opioids have also been used to treat chronic pain, with varying levels of success. Given our new understanding that alterations in grey matter volume correlate with chronic pain, recent trials have investigated drugs with the potential to modify putative underlying disease mechanisms. The major categories of drugs that have been assayed are neuroprotectants, including the excitatory neurotransmitter antagonists (e.g. NMDA), and agonists of inhibitory neurotransmitter systems (e.g. γ-aminobutyric acid, glycine). Examples of cross-use of therapies for neurological disease in chronic/neuropathic pain are provided below.
Amantidine is a drug originally used in Parkinson's disease. A derivative, memantine, an NMDA antagonist, has been marketed for treatment of Alzheimer's disease and other neurodegenerative disorders (Sonkusare et al., 2005) and has been in trials for use in chronic neuropathic pain. Another example, d-cycloserine, an antibiotic, is a partial agonist of the NMDA receptor. It has been used in preclinical models of CNS degeneration (Ogawa et al., 2003) as well as neuropathic pain, and affects limbic circuitry in rats (Millecamps et al., 2007). Riluzole, an FDA-approved drug for the treatment of amyotrophic lateral sclerosis, can reverse pain behaviour in spinal cord injured rats (Hama and Sagen, 2010). It is thought to act by inhibiting glutamate release. Other examples of therapies directed at primary neurological disease that may be useful for pain include: (i) minocycline, a microglial inhibitor that may be a neuroprotective agent. It has been used in preclinical models of Huntington's disease (Smith et al., 2003) but has some effects in pain models as well (Chang and Waxman, 2010); (ii) propentofylline, a unique methylxanthine with clear cyclic alpha-amino-3-hydroxy-5-methyl-4-isoxazoleproprionic acid, phosphodiesterase and adenosine actions, which have neuroprotective and anti-inflammatory effects. It has effects in a number of preclinical CNS-related disease models including chronic pain (Sweitzer and De Leo, 2011).
Smart treatments—targeting through localized delivery
The use of CT or MRI to direct delivery of small amounts of drugs to specific regions or nerves for pain control is gaining increased attention in preclinical models. The amount of drug delivered is small enough that it has no systemic effect. For example, when injected into a nerve in very small quantities, adriamycin can provide pain relief through retrograde transport and killing of the dorsal root ganglion cells of the specific nerves affected (Grant et al., 2008). Another example is the use of small volumes of agents that specifically knock out C-pain fibres. One such agent is resniferatoxin, a capsaicin analogue that inactivates sensory neurons by binding to the vanilloid (TRPV1) receptor and producing a calcium influx (Bates et al., 2010). Although clearly not a preferred solution since it destroys the sensory neurons, targeted delivery of such an agent may be used to control pain in certain conditions, such as cancer affecting the face, when other efforts have failed. Another possible future application of this kind of approach is the targeted treatment of schwannomas based on newly defined preclinical developments (Saydam et al., 2011).
Smarter tools for objective diagnosis of pain
A major issue in pain diagnosis and research is the lack of an objective measurement of pain. Even in patients able to report subjective pain ratings, these are clearly insufficient (Victor et al., 2008; Lin et al., 2011). In cases where patients cannot communicate, the problem is even more complex. There is an urgent need to develop biomarkers for pain. The search for reliable markers of chronic pain has focused on a number of approaches. These include clinical questionnaires (see below) and screening tools such as painDETECT (Freynhagen et al., 2006; Scholz et al., 2009), psychophysical measures (Jaaskelainen et al., 2005; Arendt-Nielsen and Yarnitsky, 2009; Serra, 2010), and potentially, imaging (Borsook et al., 2011a, b). Questionnaires that have been used in the evaluation of chronic pain attempt to also determine changes that occur in addition to changes in pain intensity [e.g. Leeds Assessment of Neuropathic Symptoms and Signs (LANSS); Neuropathic Pain Questionnaire (NPQ)] as summarized by Bennett et al. (2007). Tools adopted from the psychiatric literature, which can be used to evaluate other dimensions of chronic pain, including quality of life, depression, anxiety, catastrophizing and drug-abuse potential, are being included in chronic pain evaluation because of the multi-dimensional nature of the condition (Haythornthwaite, 2010). In addition, psychophysical/neurophysiological measures such as quantitative sensory testing continue to provide additional information on a subject's pain (Jaaskelainen, 2004). These neurophysiological approaches aim to provide a differentiated assessment based on underlying pathophysiology that may include measures of sensitization, abnormal fibre type, sensory loss etc. Given the multidimensional effects of pain on behaviour, inputs on psychological/psychiatric alterations in patients with chronic pain (Borsook et al., 2007; Elman et al., 2011) would probably improve outcomes. Few of these assessment tools are routinely used in current clinical settings.
Aside from diffusion tensor imaging that is currently employed in many institutions to evaluate alterations in white matter integrity, two current imaging technologies may soon be in the clinic for evaluation of patients with pain. The first is measures of grey matter and the second is resting state networks. A defining article by Apkarian et al. (2004) reported loss of grey matter in the thalamus and dorsolateral prefrontal cortex in patients with chronic back pain. Since then a number of groups have reported such changes in various neurological conditions including trigeminal neuropathy (DaSilva et al., 2008) and migraine (May, 2009). While it is not yet well-understood, the finding that the grey matter changes revert towards normal with treatment is highly intriguing (Rodriguez-Raecke et al., 2009; Seminowicz et al., 2011). One potential mechanism that has been evaluated in pain models relates to dendritic sprouting/branching and loss (Metz et al., 2009). A second approach with potential utility in the clinic is evaluation of resting state networks (Greicius et al., 2009; Uddin et al., 2009). Such networks can differ in disease states (Chen et al., 2011) or drug effects (Boveroux et al., 2010) and may provide a signature for specific pain syndromes such as back pain (Balenzuela et al., 2010) or diabetic neuropathy (Cauda et al., 2010), fibromyalgia (Napadow et al., 2010) or may at least objectively differentiate pain from non-painful conditions. If methodological issues can be ironed out in terms of how best to evaluate the multiple resting state networks of health and disease, the approach is potentially of high value in the clinic as it does not require any intervention with patients during scanning procedures.
Figure 5 summarizes integrative approaches for evaluating pain. Of these, imaging has taken the stage in its ability to evaluate functional, morphological and chemical changes in disease states and provide a new window of understanding disease neurobiology related to chronic pain. Having markers for chronic pain would allow for a specific diagnosis of pain and for measures of clinical efficacy (given that the current success rates of analgesics for chronic pain are ∼30%). The successful development of drug (analgesic) and disease (neuropathic pain) brain signatures and subsequently the validation as biomarkers would allow for objective indices for clinical drug development and for clinical practice. Since brain action provides a basis for behaviour (pain or analgesia) brain imaging holds the promise of defining potential makers that would need to then be shown to be sensitive, reproducible, validated and subsequently adopted by clinicians and regulatory agencies. A number of recent reviews have addressed the current state of imaging the brain state in pain and its potential in providing objective measures of drug and other therapeutic measures (Tracey and Mantyh, 2007; Apkarian, 2008; Borsook and Becerra, 2010).
Conclusions
Pain is common in neurological diseases affecting diverse parts of the PNS and the CNS (Fig. 5) that may have subsequent effects on altered brain circuitry and result in pain. Most neurologists treat patients with chronic pain, but few specialize in the discipline. In fact, when neurologists rate their preference for treating diseases, chronic pain ranks low (with the exception of migraine, which ranks high) (Evans and Evans, 2010). The reasons for this may include: (i) lack of formal training in pain management (Galer et al., 1999); (ii) the fact that pain clinics treat such a broad spectrum of syndromes (with the exception of those treating conditions such as migraine); (iii) a sense that there are few viable treatment options; (iv) the notion that patients with chronic pain are complex to deal with; or (v) the lack of financial incentive.
Thus, there is a high unmet need for chronic pain treatment and for more research into the underlying mechanisms of pain diseases. With current technological advances and investigation into mechanism-based treatment approaches (Finnerup, 2008), we are at a critical juncture in pain research. However, progress will remain slow unless we fully recognize pain as a brain disease and increase the involvement of neurologists in the treatment of and research into chronic pain. The first step towards this goal is to include a comprehensive survey of pain conditions, management and research as part of the standard training of new neurologists.
Funding
NINDS (to D.B.). Support for this work is from National Institutes of Health (NIH) – National Institute of Neurological Disorders and Stroke (NINDS; Grants K24NS64050, R01NS065051 and R01NS056195) and the Herlands Fund for Pain Research.
Glossary
Abbreviations
- CRPS
complex regional pain syndrome
References
- Abbruzzese G, Berardelli A. Sensorimotor integration in movement disorders. Mov Disord. 2003;18:231–40. doi: 10.1002/mds.10327. [DOI] [PubMed] [Google Scholar]
- Aghakhani N, Parker F, David P, Morar S, Lacroix C, Benoudiba F, et al. Long-term follow-up of Chiari-related syringomyelia in adults: analysis of 157 surgically treated cases. Neurosurgery. 2009;64:308–15. doi: 10.1227/01.NEU.0000336768.95044.80. [DOI] [PubMed] [Google Scholar]
- Albano B, Dinia L, Del Sette M, Gandolfo C, Sivori G, Finocchi C. Fabry disease in patients with migraine with aura. Neurol Sci. 2010;31(Suppl 1):S167–9. doi: 10.1007/s10072-010-0314-5. [DOI] [PubMed] [Google Scholar]
- Albin RL, Young AB. Somatosensory phenomena in Huntington's disease. Mov Disord. 1988;3:343–6. doi: 10.1002/mds.870030411. [DOI] [PubMed] [Google Scholar]
- Alderfer BS, Arciniegas DB, Silver JM. Treatment of depression following traumatic brain injury. J Head Trauma Rehabil. 2005;20:544–62. doi: 10.1097/00001199-200511000-00006. [DOI] [PubMed] [Google Scholar]
- Andersen S, Skorpen F. Variation in the COMT gene: implications for pain perception and pain treatment. Pharmacogenomics. 2009;10:669–84. doi: 10.2217/pgs.09.13. [DOI] [PubMed] [Google Scholar]
- Antonaci F, Nappi G, Galli F, Manzoni GC, Calabresi P, Costa A. Migraine and psychiatric comorbidity: a review of clinical findings. J Headache Pain. 2011;12:115–25. doi: 10.1007/s10194-010-0282-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Apkarian AV. Pain perception in relation to emotional learning. Curr Opin Neurobiol. 2008;18:464–8. doi: 10.1016/j.conb.2008.09.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Apkarian AV, Sosa Y, Sonty S, Levy RM, Harden RN, Parrish TB, et al. Chronic back pain is associated with decreased prefrontal and thalamic gray matter density. J Neurosci. 2004;24:10410–5. doi: 10.1523/JNEUROSCI.2541-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arbus C, Gardette V, Bui E, Cantet C, Andrieu S, Nourhashemi F, et al. Antidepressant use in Alzheimer's disease patients: results of the REAL.FR cohort. Int Psychogeriatr. 2010;22:120–8. doi: 10.1017/S1041610209990780. [DOI] [PubMed] [Google Scholar]
- Arendt-Nielsen L, Yarnitsky D. Experimental and clinical applications of quantitative sensory testing applied to skin, muscles and viscera. J Pain. 2009;10:556–72. doi: 10.1016/j.jpain.2009.02.002. [DOI] [PubMed] [Google Scholar]
- Atassi N, Cook A, Pineda CM, Yerramilli-Rao P, Pulley D, Cudkowicz M. Depression in amyotrophic lateral sclerosis. Amyotroph Lateral Scler. 2010;12:109–12. doi: 10.3109/17482968.2010.536839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Atkinson JH, Jr, Ingram RE, Kremer EF, Saccuzzo DP. MMPI subgroups and affective disorder in chronic pain patients. J Nerv Ment Dis. 1986;174:408–13. doi: 10.1097/00005053-198607000-00004. [DOI] [PubMed] [Google Scholar]
- Austin PJ, Moalem-Taylor G. The neuro-immune balance in neuropathic pain: involvement of inflammatory immune cells, immune-like glial cells and cytokines. J Neuroimmunol. 2010;229:26–50. doi: 10.1016/j.jneuroim.2010.08.013. [DOI] [PubMed] [Google Scholar]
- Auvil-Novak SE. The chronobiology, chronopharmacology, and chronotherapeutics of pain. Annu Rev Nurs Res. 1999;17:133–53. [PubMed] [Google Scholar]
- Balenzuela P, Chernomoretz A, Fraiman D, Cifre I, Sitges C, Montoya P, et al. Modular organization of brain resting state networks in chronic back pain patients. Front Neuroinform. 2010;4:116. doi: 10.3389/fninf.2010.00116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baron R, Binder A, Wasner G. Neuropathic pain: diagnosis, pathophysiological mechanisms, and treatment. Lancet Neurol. 2010;9:807–19. doi: 10.1016/S1474-4422(10)70143-5. [DOI] [PubMed] [Google Scholar]
- Bates BD, Mitchell K, Keller JM, Chan CC, Swaim WD, Yaskovich R, et al. Prolonged analgesic response of cornea to topical resiniferatoxin, a potent TRPV1 agonist. Pain. 2010;149:522–8. doi: 10.1016/j.pain.2010.03.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baumer T, Hidding U, Hamel W, Buhmann C, Moll CK, Gerloff C, et al. Effects of DBS, premotor rTMS, and levodopa on motor function and silent period in advanced Parkinson's disease. Mov Disord. 2009;24:672–6. doi: 10.1002/mds.22417. [DOI] [PubMed] [Google Scholar]
- Becerra L, Harter K, Gonzalez RG, Borsook D. Functional magnetic resonance imaging measures of the effects of morphine on central nervous system circuitry in opioid-naive healthy volunteers. Anesth Analg. 2006;103:208–16. doi: 10.1213/01.ane.0000221457.71536.e0. [DOI] [PubMed] [Google Scholar]
- Becerra L, Harris W, Joseph D, Huppert T, Boas DA, Borsook D. Diffuse optical tomography of pain and tactile stimulation: activation in cortical sensory and emotional systems. Neuroimage. 2008;41:252–9. doi: 10.1016/j.neuroimage.2008.01.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Becerra L, Harris W, Grant M, George E, Boas D, Borsook D. Diffuse optical tomography activation in the somatosensory cortex: specific activation by painful vs. non-painful thermal stimuli. PLoS One. 2009a;4:e8016. doi: 10.1371/journal.pone.0008016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Becerra L, Schwartzman RJ, Kiefer RT, Rohr P, Moulton EA, Wallin D, et al. CNS measures of pain responses pre- and post-anesthetic ketamine in a patient with complex regional pain syndrome. Pain Med. 2009b doi: 10.1111/pme.12939. [DOI] [PubMed] [Google Scholar]
- Beiske AG, Loge JH, Ronningen A, Svensson E. Pain in Parkinson's disease: Prevalence and characteristics. Pain. 2009;141:173–7. doi: 10.1016/j.pain.2008.12.004. [DOI] [PubMed] [Google Scholar]
- Belfer I, Segall S. COMT genetic variants and pain. Drugs Today. 2011;47:457–67. doi: 10.1358/dot.2011.47.6.1611895. [DOI] [PubMed] [Google Scholar]
- Benedetti F, Arduino C, Vighetti S, Asteggiano G, Tarenzi L, Rainero I. Pain reactivity in Alzheimer patients with different degrees of cognitive impairment and brain electrical activity deterioration. Pain. 2004;111:22–9. doi: 10.1016/j.pain.2004.05.015. [DOI] [PubMed] [Google Scholar]
- Benedetti F, Vighetti S, Ricco C, Lagna E, Bergamasco B, Pinessi L, et al. Pain threshold and tolerance in Alzheimer's disease. Pain. 1999;80:377–82. doi: 10.1016/s0304-3959(98)00228-0. [DOI] [PubMed] [Google Scholar]
- Bennett MI, Attal N, Backonja MM, Baron R, Bouhassira D, Freynhagen R, et al. Using screening tools to identify neuropathic pain. Pain. 2007;127:199–203. doi: 10.1016/j.pain.2006.10.034. [DOI] [PubMed] [Google Scholar]
- Bentwich J, Dobronevsky E, Aichenbaum S, Shorer R, Peretz R, Khaigrekht M, et al. Beneficial effect of repetitive transcranial magnetic stimulation combined with cognitive training for the treatment of Alzheimer's disease: a proof of concept study. J Neural Transm. 2011;118:463–71. doi: 10.1007/s00702-010-0578-1. [DOI] [PubMed] [Google Scholar]
- Berman RM, Cappiello A, Anand A, Oren DA, Heninger GR, Charney DS, et al. Antidepressant effects of ketamine in depressed patients. Biol Psychiatry. 2000;47:351–4. doi: 10.1016/s0006-3223(99)00230-9. [DOI] [PubMed] [Google Scholar]
- Bermejo PE, Oreja-Guevara C, Diez-Tejedor E. Pain in multiple sclerosis: prevalence, mechanisms, types and treatment. Rev Neurol. 2010;50:101–8. [PubMed] [Google Scholar]
- Berthier M, Starkstein S, Leiguarda R. Asymbolia for pain: a sensory-limbic disconnection syndrome. Ann Neurol. 1988;24:41–9. doi: 10.1002/ana.410240109. [DOI] [PubMed] [Google Scholar]
- Bigal ME, Lipton RB. Overuse of acute migraine medications and migraine chronification. Curr Pain Headache Rep. 2009;13:301–7. doi: 10.1007/s11916-009-0048-3. [DOI] [PubMed] [Google Scholar]
- Bigal ME, Lipton RB. Migraine chronification. Curr Neurol Neurosci Rep. 2011;11:139–48. doi: 10.1007/s11910-010-0175-6. [DOI] [PubMed] [Google Scholar]
- Bittar RG, Kar-Purkayastha I, Owen SL, Bear RE, Green A, Wang S, et al. Deep brain stimulation for pain relief: a meta-analysis. J Clin Neurosci. 2005;12:515–9. doi: 10.1016/j.jocn.2004.10.005. [DOI] [PubMed] [Google Scholar]
- Boecker H, Ceballos-Baumann A, Bartenstein P, Weindl A, Siebner HR, Fassbender T, et al. Sensory processing in Parkinson's and Huntington's disease: investigations with 3D H(2)(15)O-PET. Brain. 1999;122(Pt 9):1651–65. doi: 10.1093/brain/122.9.1651. [DOI] [PubMed] [Google Scholar]
- Boivie J, Leijon G, Johansson I. Central post-stroke pain–a study of the mechanisms through analyses of the sensory abnormalities. Pain. 1989;37:173–85. doi: 10.1016/0304-3959(89)90128-0. [DOI] [PubMed] [Google Scholar]
- Boly M, Coleman MR, Davis MH, Hampshire A, Bor D, Moonen G, et al. When thoughts become action: an fMRI paradigm to study volitional brain activity in non-communicative brain injured patients. Neuroimage. 2007;36:979–92. doi: 10.1016/j.neuroimage.2007.02.047. [DOI] [PubMed] [Google Scholar]
- Boly M, Faymonville ME, Schnakers C, Peigneux P, Lambermont B, Phillips C, et al. Perception of pain in the minimally conscious state with PET activation: an observational study. Lancet Neurol. 2008;7:1013–20. doi: 10.1016/S1474-4422(08)70219-9. [DOI] [PubMed] [Google Scholar]
- Bombardier CH, Richards JS, Krause JS, Tulsky D, Tate DG. Symptoms of major depression in people with spinal cord injury: implications for screening. Arch Phys Med Rehabil. 2004;85:1749–56. doi: 10.1016/j.apmr.2004.07.348. [DOI] [PubMed] [Google Scholar]
- Borsook D, Becerra L. Emotional pain without sensory pain–dream on? Neuron. 2009;61:153–5. doi: 10.1016/j.neuron.2009.01.003. [DOI] [PubMed] [Google Scholar]
- Borsook D, Becerra L. Using NMR approaches to drive the search for new CNS therapeutics. Curr Opin Investig Drugs. 2010;11:771–8. [PMC free article] [PubMed] [Google Scholar]
- Borsook D, Becerra L. How close are we in utilizing functional neuroimaging in routine clinical diagnosis of neuropathic pain? Curr Pain Headache Rep. 2011;15:223–9. doi: 10.1007/s11916-011-0187-1. [DOI] [PubMed] [Google Scholar]
- Borsook D, Becerra L, Hargreaves R. Biomarkers for chronic pain and analgesia part 1: the need, reality, challenges, and solutions. Discovery Med. 2011a;11:197–207. [PubMed] [Google Scholar]
- Borsook D, Becerra L, Hargreaves R. Biomarkers for chronic pain and analgesia part 2: how, where and what to look for using functional imaging. Discovery Med. 2011b;11:209–19. [PubMed] [Google Scholar]
- Borsook D, Upadhyay J, Chudler EH, Becerra L. A key role of the basal ganglia in pain and analgesia–insights gained through human functional imaging. Mol Pain. 2010;6:27. doi: 10.1186/1744-8069-6-27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borsook D, Becerra L, Carlezon WA, Jr, Shaw M, Renshaw P, Elman I, et al. Reward-aversion circuitry in analgesia and pain: implications for psychiatric disorders. Eur J Pain. 2007;11:7–20. doi: 10.1016/j.ejpain.2005.12.005. [DOI] [PubMed] [Google Scholar]
- Borsook D, Becerra L, Fishman S, Edwards A, Jennings CL, Stojanovic M, et al. Acute plasticity in the human somatosensory cortex following amputation. Neuroreport. 1998;9:1013–7. doi: 10.1097/00001756-199804200-00011. [DOI] [PubMed] [Google Scholar]
- Bour A, Rasquin S, Aben I, Boreas A, Limburg M, Verhey F. A one-year follow-up study into the course of depression after stroke. J Nutr Health Aging. 2010;14:488–93. doi: 10.1007/s12603-010-0033-x. [DOI] [PubMed] [Google Scholar]
- Boveroux P, Vanhaudenhuyse A, Bruno MA, Noirhomme Q, Lauwick S, Luxen A, et al. Breakdown of within- and between-network resting state functional magnetic resonance imaging connectivity during propofol-induced loss of consciousness. Anesthesiology. 2010;113:1038–53. doi: 10.1097/ALN.0b013e3181f697f5. [DOI] [PubMed] [Google Scholar]
- Bowsher D, Leijon G, Thuomas KA. Central poststroke pain: correlation of MRI with clinical pain characteristics and sensory abnormalities. Neurology. 1998;51:1352–8. doi: 10.1212/wnl.51.5.1352. [DOI] [PubMed] [Google Scholar]
- Brederson JD, Jarvis MF, Honore P, Surowy CS. Fibromyalgia: Mechanisms, Current Treatment, and Animal Models. Curr Pharm Biotechnol. 2011 doi: 10.2174/138920111798357258. Advance Access published on April 5, 2011. [DOI] [PubMed] [Google Scholar]
- Brefel-Courbon C, Grolleau S, Thalamas C, Bourrel R, Allaria-Lapierre V, Loi R, et al. Comparison of chronic analgesic drugs prevalence in Parkinson's disease, other chronic diseases and the general population. Pain. 2009;141:14–8. doi: 10.1016/j.pain.2008.04.026. [DOI] [PubMed] [Google Scholar]
- Brinkmeyer J, Mobascher A, Warbrick T, Musso F, Wittsack HJ, Saleh A, et al. Dynamic EEG-informed fMRI modeling of the pain matrix using 20-ms root mean square segments. Hum Brain Mapp. 2010;31:1702–12. doi: 10.1002/hbm.20967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bromberg-Martin ES, Matsumoto M, Hikosaka O. Dopamine in motivational control: rewarding, aversive, and alerting. Neuron. 2010;68:815–34. doi: 10.1016/j.neuron.2010.11.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown EN, Lydic R, Schiff ND. General anesthesia, sleep, and coma. N Engl J Med. 2010;363:2638–50. doi: 10.1056/NEJMra0808281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bruehl S. An update on the pathophysiology of complex regional pain syndrome. Anesthesiology. 2010;113:713–25. doi: 10.1097/ALN.0b013e3181e3db38. [DOI] [PubMed] [Google Scholar]
- Burn DJ. Depression in Parkinson's disease. Eur J Neurol. 2002;9(Suppl 3):44–54. doi: 10.1046/j.1468-1331.9.s3.6.x. [DOI] [PubMed] [Google Scholar]
- Burstein R, Jakubowski M, Garcia-Nicas E, Kainz V, Bajwa Z, Hargreaves R, et al. Thalamic sensitization transforms localized pain into widespread allodynia. Ann Neurol. 2010;68:81–91. doi: 10.1002/ana.21994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Busch CR, Alpern HP. Depression after mild traumatic brain injury: a review of current research. Neuropsychol Rev. 1998;8:95–108. doi: 10.1023/a:1025661200911. [DOI] [PubMed] [Google Scholar]
- Cauda F, D'Agata F, Sacco K, Duca S, Cocito D, Paolasso I, et al. Altered resting state attentional networks in diabetic neuropathic pain. J Neurol Neurosurg Psychiatry. 2010;81:806–11. doi: 10.1136/jnnp.2009.188631. [DOI] [PubMed] [Google Scholar]
- Cetas JS, Saedi T, Burchiel KJ. Destructive procedures for the treatment of nonmalignant pain: a structured literature review. J Neurosurg. 2008;109:389–404. doi: 10.3171/JNS/2008/109/9/0389. [DOI] [PubMed] [Google Scholar]
- Chan CW, Peng P. Failed back surgery syndrome. Pain Med. 2011;12:577–606. doi: 10.1111/j.1526-4637.2011.01089.x. [DOI] [PubMed] [Google Scholar]
- Chang YW, Waxman SG. Minocycline attenuates mechanical allodynia and central sensitization following peripheral second-degree burn injury. J Pain. 2010;11:1146–54. doi: 10.1016/j.jpain.2010.02.010. [DOI] [PubMed] [Google Scholar]
- Chen AL, Chen TJ, Waite RL, Reinking J, Tung HL, Rhoades P, et al. Hypothesizing that brain reward circuitry genes are genetic antecedents of pain sensitivity and critical diagnostic and pharmacogenomic treatment targets for chronic pain conditions. Med Hypotheses. 2009;72:14–22. doi: 10.1016/j.mehy.2008.07.059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen X, Green PG, Levine JD. Neuropathic pain-like alterations in muscle nociceptor function associated with vibration-induced muscle pain. Pain. 2010;151:460–6. doi: 10.1016/j.pain.2010.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choi JS, Boralevi F, Brissaud O, Sanchez-Martin J, Te Morsche RH, Dib-Hajj SD, et al. Paroxysmal extreme pain disorder: a molecular lesion of peripheral neurons. Nat Rev Neurol. 2011;7:51–5. doi: 10.1038/nrneurol.2010.162. [DOI] [PubMed] [Google Scholar]
- Ciccone DS, Bandilla EB, Wu W. Psychological dysfunction in patients with reflex sympathetic dystrophy. Pain. 1997;71:323–33. doi: 10.1016/s0304-3959(97)00009-2. [DOI] [PubMed] [Google Scholar]
- Clavelou P, Besson G, Elziere C, Ferrier A, Pinard JM, Hermier M, et al. Neurological aspects of Fabry's disease. Rev Neurol. 2006;162:569–80. doi: 10.1016/s0035-3787(06)75051-2. [DOI] [PubMed] [Google Scholar]
- Cole AL, Lee PJ, Hughes DA, Deegan PB, Waldek S, Lachmann RH. Depression in adults with Fabry disease: a common and under-diagnosed problem. J Inherit Metab Dis. 2007;30:943–51. doi: 10.1007/s10545-007-0708-6. [DOI] [PubMed] [Google Scholar]
- Cole LJ, Farrell MJ, Duff EP, Barber JB, Egan GF, Gibson SJ. Pain sensitivity and fMRI pain-related brain activity in Alzheimer's disease. Brain. 2006;129:2957–65. doi: 10.1093/brain/awl228. [DOI] [PubMed] [Google Scholar]
- Coleman MR, Davis MH, Rodd JM, Robson T, Ali A, Owen AM, et al. Towards the routine use of brain imaging to aid the clinical diagnosis of disorders of consciousness. Brain. 2009;132:2541–52. doi: 10.1093/brain/awp183. [DOI] [PubMed] [Google Scholar]
- Comings DE, Blum K. Reward deficiency syndrome: genetic aspects of behavioral disorders. Prog Brain Res. 2000;126:325–41. doi: 10.1016/S0079-6123(00)26022-6. [DOI] [PubMed] [Google Scholar]
- Cooke LJ, Becker WJ. Migraine prevalence, treatment and impact: the canadian women and migraine study. Can J Neurol Sci. 2010;37:580–7. doi: 10.1017/s0317167100010738. [DOI] [PubMed] [Google Scholar]
- Corey JM. Genetic disorders producing compressive radiculopathy. Semin Neurol. 2006;26:515–22. doi: 10.1055/s-2006-951624. [DOI] [PubMed] [Google Scholar]
- Costigan M, Belfer I, Griffin RS, Dai F, Barrett LB, Coppola G, et al. Multiple chronic pain states are associated with a common amino acid-changing allele in KCNS1. Brain. 2010;133:2519–27. doi: 10.1093/brain/awq195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cox JJ, Sheynin J, Shorer Z, Reimann F, Nicholas AK, Zubovic L, et al. Congenital insensitivity to pain: novel SCN9A missense and in-frame deletion mutations. Hum Mutat. 2010;31:E1670–86. doi: 10.1002/humu.21325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Craig AD. The functional anatomy of lamina I and its role in post-stroke central pain. Prog Brain Res. 2000;129:137–51. doi: 10.1016/S0079-6123(00)29010-9. [DOI] [PubMed] [Google Scholar]
- D'Abreu A, Franca MC, Jr, Paulson HL, Lopes-Cendes I. Caring for Machado-Joseph disease: current understanding and how to help patients. Parkinsonism Relat Disord. 2010;16:2–7. doi: 10.1016/j.parkreldis.2009.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Danziger N, Willer JC. [Congenital insensitivity to pain] Rev Neurol. 2009;165:129–36. doi: 10.1016/j.neurol.2008.05.003. [DOI] [PubMed] [Google Scholar]
- DaSilva AF, Becerra L, Pendse G, Chizh B, Tully S, Borsook D. Colocalized structural and functional changes in the cortex of patients with trigeminal neuropathic pain. PLoS One. 2008;3:e3396. doi: 10.1371/journal.pone.0003396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davies M, Brophy S, Williams R, Taylor A. The prevalence, severity, and impact of painful diabetic peripheral neuropathy in type 2 diabetes. Diabetes Care. 2006;29:1518–22. doi: 10.2337/dc05-2228. [DOI] [PubMed] [Google Scholar]
- Davis KD, Hutchison WD, Lozano AM, Dostrovsky JO. Altered pain and temperature perception following cingulotomy and capsulotomy in a patient with schizoaffective disorder. Pain. 1994;59:189–99. doi: 10.1016/0304-3959(94)90071-X. [DOI] [PubMed] [Google Scholar]
- de Castro-Costa CM, Oria RB, Machado-Filho JA, Franco MT, Diniz DL, Giffoni SD, et al. Amyotrophic lateral sclerosis. Clinical analysis of 78 cases from Fortaleza (northeastern Brazil) Arq Neuropsiquiatr. 1999;57:761–74. doi: 10.1590/s0004-282x1999000500006. [DOI] [PubMed] [Google Scholar]
- De Martino B, Kalisch R, Rees G, Dolan RJ. Enhanced processing of threat stimuli under limited attentional resources. Cereb Cortex. 2009;19:127–33. doi: 10.1093/cercor/bhn062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deepika J, Manvir B, Sumit S, Vinay G, Trilochan S, Garima S, et al. Quantitative thermal sensory testing in patients with amyotrophic lateral sclerosis using reaction time exclusive method of levels (MLE) Electromyogr Clin Neurophysiol. 2006;46:145–8. [PubMed] [Google Scholar]
- Defrin R, Grunhaus L, Zamir D, Zeilig G. The effect of a series of repetitive transcranial magnetic stimulations of the motor cortex on central pain after spinal cord injury. Arch Phys Med Rehabil. 2007;88:1574–80. doi: 10.1016/j.apmr.2007.07.025. [DOI] [PubMed] [Google Scholar]
- Dejerine J, Roussy G. Le syndrome thalamique. Revue Neurologique. 1906;14:521–32. [Google Scholar]
- Demarquay G, Royet JP, Mick G, Ryvlin P. Olfactory hypersensitivity in migraineurs: a H(2)(15)O-PET study. Cephalalgia. 2008;28:1069–80. doi: 10.1111/j.1468-2982.2008.01672.x. [DOI] [PubMed] [Google Scholar]
- Demasles S, Peyron R, Garcia Larrea L, Laurent B. [Central post-stroke pain] Rev Neurol. 2008;164:825–31. doi: 10.1016/j.neurol.2008.07.016. [DOI] [PubMed] [Google Scholar]
- Denuelle M, Boulloche N, Payoux P, Fabre N, Trotter Y, Geraud G. A PET study of photophobia during spontaneous migraine attacks. Neurology. 2011;76:213–8. doi: 10.1212/WNL.0b013e3182074a57. [DOI] [PubMed] [Google Scholar]
- Diers M, Christmann C, Koeppe C, Ruf M, Flor H. Mirrored, imagined and executed movements differentially activate sensorimotor cortex in amputees with and without phantom limb pain. Pain. 2010;149:296–304. doi: 10.1016/j.pain.2010.02.020. [DOI] [PubMed] [Google Scholar]
- Dobscha SK, Clark ME, Morasco BJ, Freeman M, Campbell R, Helfand M. Systematic review of the literature on pain in patients with polytrauma including traumatic brain injury. Pain Med. 2009;10:1200–17. doi: 10.1111/j.1526-4637.2009.00721.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Drake ME., Jr Chronic pain syndrome in amyotrophic lateral sclerosis. Arch Neurol. 1983;40:453–4. doi: 10.1001/archneur.1983.04050070083025. [DOI] [PubMed] [Google Scholar]
- Dubner R. The neurobiology of persistent pain and its clinical implications. Suppl Clin Neurophysiol. 2004;57:3–7. doi: 10.1016/s1567-424x(09)70337-x. [DOI] [PubMed] [Google Scholar]
- Duhn LJ, Medves JM. A systematic integrative review of infant pain assessment tools. Adv Neonatal Care. 2004;4:126–40. doi: 10.1016/j.adnc.2004.04.005. [DOI] [PubMed] [Google Scholar]
- Dworkin RH, Jensen MP, Gammaitoni AR, Olaleye DO, Galer BS. Symptom profiles differ in patients with neuropathic versus non-neuropathic pain. J Pain. 2007;8:118–26. doi: 10.1016/j.jpain.2006.06.005. [DOI] [PubMed] [Google Scholar]
- Egloff N, Sabbioni ME, Salathe C, Wiest R, Juengling FD. Nondermatomal somatosensory deficits in patients with chronic pain disorder: clinical findings and hypometabolic pattern in FDG-PET. Pain. 2009;145:252–8. doi: 10.1016/j.pain.2009.04.016. [DOI] [PubMed] [Google Scholar]
- Eisenberg E, McNicol E, Carr DB. Opioids for neuropathic pain. Cochrane Database Syst Rev. 2006;3:CD006146. doi: 10.1002/14651858.CD006146. [DOI] [PubMed] [Google Scholar]
- Elman I, Zubieta JK, Borsook D. The missing p in psychiatric training: why it is important to teach pain to psychiatrists. Arch Gen Psychiatry. 2011;68:12–20. doi: 10.1001/archgenpsychiatry.2010.174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evans RW, Evans RE. A survey of neurologists on the likeability of headaches and other neurological disorders. Headache. 2010;50:1126–9. doi: 10.1111/j.1526-4610.2010.01708.x. [DOI] [PubMed] [Google Scholar]
- Farina S, Tinazzi M, Le Pera D, Valeriani M. Pain-related modulation of the human motor cortex. Neurol Res. 2003;25:130–42. doi: 10.1179/016164103101201283. [DOI] [PubMed] [Google Scholar]
- Farrell MJ, Katz B, Helme RD. The impact of dementia on the pain experience. Pain. 1996;67:7–15. doi: 10.1016/0304-3959(96)03041-2. [DOI] [PubMed] [Google Scholar]
- Farrer LA. Diabetes mellitus in Huntington disease. Clin Genet. 1985;27:62–7. doi: 10.1111/j.1399-0004.1985.tb00185.x. [DOI] [PubMed] [Google Scholar]
- Fellgiebel A, Muller MJ, Ginsberg L. CNS manifestations of Fabry's disease. Lancet Neurol. 2006;5:791–5. doi: 10.1016/S1474-4422(06)70548-8. [DOI] [PubMed] [Google Scholar]
- Fietta P, Manganelli P. Fibromyalgia and psychiatric disorders. Acta Biomed. 2007;78:88–95. [PubMed] [Google Scholar]
- Finnerup NB. A review of central neuropathic pain states. Curr Opin Anaesthesiol. 2008;21:586–9. doi: 10.1097/ACO.0b013e32830a4c11. [DOI] [PubMed] [Google Scholar]
- Finnerup NB, Sindrup SH, Jensen TS. The evidence for pharmacological treatment of neuropathic pain. Pain. 2010;150:573–81. doi: 10.1016/j.pain.2010.06.019. [DOI] [PubMed] [Google Scholar]
- Fins JJ, Master MG, Gerber LM, Giacino JT. The minimally conscious state: a diagnosis in search of an epidemiology. Arch Neurol. 2007;64:1400–5. doi: 10.1001/archneur.64.10.1400. [DOI] [PubMed] [Google Scholar]
- Flor H. The modification of cortical reorganization and chronic pain by sensory feedback. Appl Psychophysiol Biofeedback. 2002;27:215–27. doi: 10.1023/a:1016204029162. [DOI] [PubMed] [Google Scholar]
- Flor H, Diers M. Sensorimotor training and cortical reorganization. NeuroRehabilitation. 2009;25:19–27. doi: 10.3233/NRE-2009-0496. [DOI] [PubMed] [Google Scholar]
- Folstein MF, Robinson R, Folstein S, McHugh PR. Depression and neurological disorders. New treatment opportunities for elderly depressed patients. J Affect Disord. 1985;19(Suppl 1):S11–4. doi: 10.1016/0165-0327(85)90083-7. [DOI] [PubMed] [Google Scholar]
- Ford B. Pain in Parkinson's disease. Mov Disord. 2010;25(Suppl 1):S98–103. doi: 10.1002/mds.22716. [DOI] [PubMed] [Google Scholar]
- Formisano R, Bivona U, Catani S, D'Ippolito M, Buzzi MG. Post-traumatic headache: facts and doubts. J Headache Pain. 2009;10:145–52. doi: 10.1007/s10194-009-0108-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Franca MC, Jr, D'Abreu A, Nucci A, Lopes-Cendes I. Muscle excitability abnormalities in Machado-Joseph disease. Arch Neurol. 2008;65:525–9. doi: 10.1001/archneur.65.4.525. [DOI] [PubMed] [Google Scholar]
- Franca MC, Jr, D'Abreu A, Friedman JH, Nucci A, Lopes-Cendes I. Chronic pain in Machado-Joseph disease: a frequent and disabling symptom. Arch Neurol. 2007;64:1767–70. doi: 10.1001/archneur.64.12.1767. [DOI] [PubMed] [Google Scholar]
- Frettloh J, Huppe M, Maier C. Severity and specificity of neglect-like symptoms in patients with complex regional pain syndrome (CRPS) compared to chronic limb pain of other origins. Pain. 2006;124:184–9. doi: 10.1016/j.pain.2006.04.010. [DOI] [PubMed] [Google Scholar]
- Freynhagen R, Baron R, Gockel U, Tolle TR. painDETECT: a new screening questionnaire to identify neuropathic components in patients with back pain. Curr Med Res Opin. 2006;22:1911–20. doi: 10.1185/030079906X132488. [DOI] [PubMed] [Google Scholar]
- Fukui S, Shigemori S, Nosaka S. Changes in regional cerebral blood flow in the thalamus after electroconvulsive therapy for patients with complex regional pain syndrome type 1 (preliminary case series) Reg Anesth Pain Med. 2002;27:529–32. doi: 10.1053/rapm.2002.34330. [DOI] [PubMed] [Google Scholar]
- Gadoth N, Sandbank U. Involvement of dorsal root ganglia in Fabry's disease. J Med Genet. 1983;20:309–12. doi: 10.1136/jmg.20.4.309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galer BS, Jensen M. Neglect-like symptoms in complex regional pain syndrome: results of a self-administered survey. J Pain Symptom Manage. 1999;18:213–7. doi: 10.1016/s0885-3924(99)00076-7. [DOI] [PubMed] [Google Scholar]
- Galer BS, Keran C, Frisinger M. Pain medicine education among American neurologists: a need for improvement. Neurology. 1999;52:1710–2. doi: 10.1212/wnl.52.8.1710. [DOI] [PubMed] [Google Scholar]
- Garcia-Larrea L, Perchet C, Creac'h C, Convers P, Peyron R, Laurent B, et al. Operculo-insular pain (parasylvian pain): a distinct central pain syndrome. Brain. 2010;133:2528–39. doi: 10.1093/brain/awq220. [DOI] [PubMed] [Google Scholar]
- Geha PY, Baliki MN, Harden RN, Bauer WR, Parrish TB, Apkarian AV. The brain in chronic CRPS pain: abnormal gray-white matter interactions in emotional and autonomic regions. Neuron. 2008;60:570–81. doi: 10.1016/j.neuron.2008.08.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gelinas C, Fillion L, Puntillo KA, Viens C, Fortier M. Validation of the critical-care pain observation tool in adult patients. Am J Crit Care. 2006;15:420–7. [PubMed] [Google Scholar]
- Gerber RK, Nie H, Arendt-Nielsen L, Curatolo M, Graven-Nielsen T. Local pain and spreading hyperalgesia induced by intramuscular injection of nerve growth factor are not reduced by local anesthesia of the muscle. Clin J Pain. 2011;27:240–7. doi: 10.1097/AJP.0b013e3182048481. [DOI] [PubMed] [Google Scholar]
- Gerdelat-Mas A, Simonetta-Moreau M, Thalamas C, Ory-Magne F, Slaoui T, Rascol O, et al. Levodopa raises objective pain threshold in Parkinson's disease: a RIII reflex study. J Neurol Neurosurg Psychiatry. 2007;78:1140–2. doi: 10.1136/jnnp.2007.120212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghai B, Dureja GP. Complex regional pain syndrome: a review. J Postgrad Med. 2004;50:300–7. [PubMed] [Google Scholar]
- Gierthmuhlen J, Arning P, Binder A, Herzog J, Deuschl G, Wasner G, et al. Influence of deep brain stimulation and levodopa on sensory signs in Parkinson's disease. Mov Disord. 2010;25:1195–202. doi: 10.1002/mds.23128. [DOI] [PubMed] [Google Scholar]
- Giuffrida R, Vingerhoets FJ, Bogousslavsky J, Ghika J. Pain in Parkinson's disease. Rev Neurol. 2005;161:407–18. doi: 10.1016/s0035-3787(05)85070-2. [DOI] [PubMed] [Google Scholar]
- Gradl G, Schurmann M. Sympathetic dysfunction as a temporary phenomenon in acute posttraumatic CRPS I. Clin Auton Res. 2005;15:29–34. doi: 10.1007/s10286-005-0237-z. [DOI] [PubMed] [Google Scholar]
- Graeber MB. Changing face of microglia. Science. 2010;330:783–8. doi: 10.1126/science.1190929. [DOI] [PubMed] [Google Scholar]
- Grant PE, Gallagher J, Gonzalez RG, Borsook D. Adriamycin injection into the medial cord of the brachial plexus: computed tomography-guided targeted pain therapy. Pain Med. 2008;9:83–7. doi: 10.1111/j.1526-4637.2006.00139.x. [DOI] [PubMed] [Google Scholar]
- Grau-Lopez L, Sierra S, Martinez-Caceres E, Ramo-Tello C. Analysis of the pain in multiple sclerosis patients. Neurologia. 2011;26:208–13. doi: 10.1016/j.nrl.2010.07.014. [DOI] [PubMed] [Google Scholar]
- Greicius MD, Krasnow B, Reiss AL, Menon V. Functional connectivity in the resting brain: a network analysis of the default mode hypothesis. Proc Natl Acad Sci USA. 2003;100:253–8. doi: 10.1073/pnas.0135058100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greicius MD, Supekar K, Menon V, Dougherty RF. Resting-state functional connectivity reflects structural connectivity in the default mode network. Cereb Cortex. 2009;19:72–8. doi: 10.1093/cercor/bhn059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greitz D. Unraveling the riddle of syringomyelia. Neurosurg Rev. 2006;29:251–63. doi: 10.1007/s10143-006-0029-5. [DOI] [PubMed] [Google Scholar]
- Grove M, Vonsattel JP, Mazzoni P, Marder K. Huntington's disease. Sci Aging Knowledge Environ. 2003;2003:dn3. doi: 10.1126/sageke.2003.43.dn3. [DOI] [PubMed] [Google Scholar]
- Grüsser SM, Diers M, Flor H. Phantom limb pain: aspects of neuroplasticity and intervention. Anasthesiol Intensivmed Notfallmed Schmerzther. 2003;38:762–6. doi: 10.1055/s-2003-45403. [DOI] [PubMed] [Google Scholar]
- Gussew A, Rzanny R, Gullmar D, Scholle HC, Reichenbach JR. 1H-MR spectroscopic detection of metabolic changes in pain processing brain regions in the presence of non-specific chronic low back pain. Neuroimage. 2011;54:1315–23. doi: 10.1016/j.neuroimage.2010.09.039. [DOI] [PubMed] [Google Scholar]
- Haanpaa M, Attal N, Backonja M, Baron R, Bennett M, Bouhassira D, et al. NeuPSIG guidelines on neuropathic pain assessment. Pain. 2011;152:14–27. doi: 10.1016/j.pain.2010.07.031. [DOI] [PubMed] [Google Scholar]
- Halbauer JD, Ashford JW, Zeitzer JM, Adamson MM, Lew HL, Yesavage JA. Neuropsychiatric diagnosis and management of chronic sequelae of war-related mild to moderate traumatic brain injury. J Rehabil Res Dev. 2009;46:757–96. doi: 10.1682/jrrd.2008.08.0119. [DOI] [PubMed] [Google Scholar]
- Hama A, Sagen J. Antinociceptive effect of riluzole in rats with neuropathic spinal cord injury pain. J Neurotrauma. 2010;28:127–34. doi: 10.1089/neu.2010.1539. [DOI] [PubMed] [Google Scholar]
- Hatem SM, Attal N, Ducreux D, Gautron M, Parker F, Plaghki L, et al. Clinical, functional and structural determinants of central pain in syringomyelia. Brain. 2010;133:3409–22. doi: 10.1093/brain/awq244. [DOI] [PubMed] [Google Scholar]
- Haythornthwaite JA. IMMPACT recommendations for clinical trials: opportunities for the RDC/TMD. J Oral Rehabil. 2010;37:799–806. doi: 10.1111/j.1365-2842.2010.02152.x. [DOI] [PubMed] [Google Scholar]
- Hempenstall K, Nurmikko TJ, Johnson RW, A'Hern RP, Rice AS. Analgesic therapy in postherpetic neuralgia: a quantitative systematic review. PLoS Med. 2005;2:e164. doi: 10.1371/journal.pmed.0020164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herr K, Coyne PJ, Key T, Manworren R, McCaffery M, Merkel S, et al. Pain assessment in the nonverbal patient: position statement with clinical practice recommendations. Pain Manag Nurs. 2006;7:44–52. doi: 10.1016/j.pmn.2006.02.003. [DOI] [PubMed] [Google Scholar]
- Hewitt DJ, McDonald M, Portenoy RK, Rosenfeld B, Passik S, Breitbart W. Pain syndromes and etiologies in ambulatory AIDS patients. Pain. 1997;70:117–23. doi: 10.1016/s0304-3959(96)03281-2. [DOI] [PubMed] [Google Scholar]
- Hoffmann B, Beck M, Sunder-Plassmann G, Borsini W, Ricci R, Mehta A. Nature and prevalence of pain in Fabry disease and its response to enzyme replacement therapy–a retrospective analysis from the Fabry outcome survey. Clin J Pain. 2007;23:535–42. doi: 10.1097/AJP.0b013e318074c986. [DOI] [PubMed] [Google Scholar]
- Holland JM, Feldman JL, Gilbert HC. Phantom orchalgia. J Urol. 1994;152:2291–3. doi: 10.1016/s0022-5347(17)31660-9. [DOI] [PubMed] [Google Scholar]
- Holman AJ, Myers RR. A randomized, double-blind, placebo-controlled trial of pramipexole, a dopamine agonist, in patients with fibromyalgia receiving concomitant medications. Arthritis Rheum. 2005;52:2495–505. doi: 10.1002/art.21191. [DOI] [PubMed] [Google Scholar]
- Hong JH, Bai DS, Jeong JY, Choi BY, Chang CH, Kim SH, et al. Injury of the spino-thalamo-cortical pathway is necessary for central post-stroke pain. Eur Neurol. 2010;64:163–8. doi: 10.1159/000319040. [DOI] [PubMed] [Google Scholar]
- Husain MM, Rush AJ, Trivedi MH, McClintock SM, Wisniewski SR, Davis L, et al. Pain in depression: STAR*D study findings. J Psychosom Res. 2007;63:113–22. doi: 10.1016/j.jpsychores.2007.02.009. [DOI] [PubMed] [Google Scholar]
- Huson SM, Acosta MT, Belzberg AJ, Bernards A, Chernoff J, Cichowski K, et al. Back to the future: proceedings from the 2010 NF Conference. Am J Med Genet A. 2011;155A:307–21. doi: 10.1002/ajmg.a.33804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hyman BT, Van Hoesen GW, Damasio AR, Barnes CL. Alzheimer's disease: cell-specific pathology isolates the hippocampal formation. Science. 1984;225:1168–70. doi: 10.1126/science.6474172. [DOI] [PubMed] [Google Scholar]
- Indo Y. Nerve growth factor, pain, itch and inflammation: lessons from congenital insensitivity to pain with anhidrosis. Expert Rev Neurother. 2010;10:1707–24. doi: 10.1586/ern.10.154. [DOI] [PubMed] [Google Scholar]
- Jaaskelainen SK. Clinical neurophysiology and quantitative sensory testing in the investigation of orofacial pain and sensory function. J Orofac Pain. 2004;18:85–107. [PubMed] [Google Scholar]
- Jaaskelainen SK, Teerijoki-Oksa T, Forssell H. Neurophysiologic and quantitative sensory testing in the diagnosis of trigeminal neuropathy and neuropathic pain. Pain. 2005;117:349–57. doi: 10.1016/j.pain.2005.06.028. [DOI] [PubMed] [Google Scholar]
- Janig W, Baron R. Complex regional pain syndrome is a disease of the central nervous system. Clin Auton Res. 2002;12:150–64. doi: 10.1007/s10286-002-0022-1. [DOI] [PubMed] [Google Scholar]
- Johnson RW, Bouhassira D, Kassianos G, Leplege A, Schmader KE, Weinke T. The impact of herpes zoster and post-herpetic neuralgia on quality-of-life. BMC Med. 2010;8:37. doi: 10.1186/1741-7015-8-37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones AK, Watabe H, Cunningham VJ, Jones T. Cerebral decreases in opioid receptor binding in patients with central neuropathic pain measured by [11C]diprenorphine binding and PET. Eur J Pain. 2004;8:479–85. doi: 10.1016/j.ejpain.2003.11.017. [DOI] [PubMed] [Google Scholar]
- Jonsson P, Hedenrud T, Linde M. Epidemiology of medication overuse headache in the general Swedish population. Cephalalgia. 2011;31:1015–22. doi: 10.1177/0333102411410082. [DOI] [PubMed] [Google Scholar]
- Juri C, Rodriguez-Oroz M, Obeso JA. The pathophysiological basis of sensory disturbances in Parkinson's disease. J Neurol Sci. 2010;289:60–5. doi: 10.1016/j.jns.2009.08.018. [DOI] [PubMed] [Google Scholar]
- Kalliomaki ML, Sandblom G, Gunnarsson U, Gordh T. Persistent pain after groin hernia surgery: a qualitative analysis of pain and its consequences for quality of life. Acta Anaesthesiol Scand. 2009;53:236–46. doi: 10.1111/j.1399-6576.2008.01840.x. [DOI] [PubMed] [Google Scholar]
- Kanjwal K, Karabin B, Kanjwal Y, Grubb BP. Autonomic dysfunction presenting as postural tachycardia syndrome following traumatic brain injury. Cardiol J. 2010;17:482–7. [PubMed] [Google Scholar]
- Karl A, Birbaumer N, Lutzenberger W, Cohen LG, Flor H. Reorganization of motor and somatosensory cortex in upper extremity amputees with phantom limb pain. J Neurosci. 2001;21:3609–18. doi: 10.1523/JNEUROSCI.21-10-03609.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kehlet H, Jensen TS, Woolf CJ. Persistent postsurgical pain: risk factors and prevention. Lancet. 2006;367:1618–25. doi: 10.1016/S0140-6736(06)68700-X. [DOI] [PubMed] [Google Scholar]
- Kenner M, Menon U, Elliott DG. Multiple sclerosis as a painful disease. Int Rev Neurobiol. 2007;79:303–21. doi: 10.1016/S0074-7742(07)79013-X. [DOI] [PubMed] [Google Scholar]
- Ketz AK. The experience of phantom limb pain in patients with combat-related traumatic amputations. Arch Phys Med Rehabil. 2008;89:1127–32. doi: 10.1016/j.apmr.2007.11.037. [DOI] [PubMed] [Google Scholar]
- Khan F, Pallant JF, Ng L, Bhasker A. Factors associated with long-term functional outcomes and psychological sequelae in Guillain-Barre syndrome. J Neurol. 2010;257:2024–31. doi: 10.1007/s00415-010-5653-x. [DOI] [PubMed] [Google Scholar]
- Kiefer RT, Rohr P, Ploppa A, Altemeyer KH, Schwartzman RJ. Complete recovery from intractable complex regional pain syndrome, CRPS-type I, following anesthetic ketamine and midazolam. Pain Pract. 2007;7:147–50. doi: 10.1111/j.1533-2500.2007.00123.x. [DOI] [PubMed] [Google Scholar]
- Kiefer RT, Rohr P, Ploppa A, Dieterich HJ, Grothusen J, Koffler S, et al. Efficacy of ketamine in anesthetic dosage for the treatment of refractory complex regional pain syndrome: an open-label phase II study. Pain Med. 2008;9:1173–201. doi: 10.1111/j.1526-4637.2007.00402.x. [DOI] [PubMed] [Google Scholar]
- Kim HJ, Paek SH, Kim JY, Lee JY, Lim YH, Kim MR, et al. Chronic subthalamic deep brain stimulation improves pain in Parkinson disease. J Neurol. 2008;255:1889–94. doi: 10.1007/s00415-009-0908-0. [DOI] [PubMed] [Google Scholar]
- Klit H, Finnerup NB, Jensen TS. Central post-stroke pain: clinical characteristics, pathophysiology, and management. Lancet Neurol. 2009;8:857–68. doi: 10.1016/S1474-4422(09)70176-0. [DOI] [PubMed] [Google Scholar]
- Kloss-Brandstatter A, Hachl O, Leitgeb PC, Buchner A, Coassin S, Rasse M, et al. Epidemiologic evidence of barometric pressure changes inducing increased reporting of oral pain. Eur J Pain. 2011;15:880–4. doi: 10.1016/j.ejpain.2011.01.013. [DOI] [PubMed] [Google Scholar]
- Kosaras B, Jakubowski M, Kainz V, Burstein R. Sensory innervation of the calvarial bones of the mouse. J Comp Neurol. 2009;515:331–48. doi: 10.1002/cne.22049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kosinski CM, Schlangen C, Gellerich FN, Gizatullina Z, Deschauer M, Schiefer J, et al. Myopathy as a first symptom of Huntington's disease in a Marathon runner. Mov Disord. 2007;22:1637–40. doi: 10.1002/mds.21550. [DOI] [PubMed] [Google Scholar]
- Koyama T, Kato K, Mikami A. During pain-avoidance neurons activated in the macaque anterior cingulate and caudate. Neurosci Lett. 2000;283:17–20. doi: 10.1016/s0304-3940(00)00894-6. [DOI] [PubMed] [Google Scholar]
- Koyanagi I, Houkin K. Pathogenesis of syringomyelia associated with Chiari type 1 malformation: review of evidences and proposal of a new hypothesis. Neurosurg Rev. 2010;33:271–84. doi: 10.1007/s10143-010-0266-5. discussion 84–5. [DOI] [PubMed] [Google Scholar]
- Krause JS, Saunders LL, Reed KS, Coker J, Zhai Y, Johnson E. Comparison of the patient health questionnaire and the older adult health and mood questionnaire for self-reported depressive symptoms after spinal cord injury. Rehabil Psychol. 2009;54:440–8. doi: 10.1037/a0017402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krulewitch H, London MR, Skakel VJ, Lundstedt GJ, Thomason H, Brummel-Smith K. Assessment of pain in cognitively impaired older adults: a comparison of pain assessment tools and their use by nonprofessional caregivers. J Am Geriatr Soc. 2000;48:1607–11. doi: 10.1111/j.1532-5415.2000.tb03871.x. [DOI] [PubMed] [Google Scholar]
- Kumar B, Kalita J, Kumar G, Misra UK. Central poststroke pain: a review of pathophysiology and treatment. Anesth Analg. 2009;108:1645–57. doi: 10.1213/ane.0b013e31819d644c. [DOI] [PubMed] [Google Scholar]
- Kunz M, Lautenbacher S. The impact of Alzheimer's disease on the pain processing. Fortschr Neurol Psychiatr. 2004;72:375–82. doi: 10.1055/s-2004-818389. [DOI] [PubMed] [Google Scholar]
- Kurban M, Wajid M, Shimomura Y, Christiano AM. A nonsense mutation in the SCN9A gene in congenital insensitivity to pain. Dermatology. 2010;221:179–83. doi: 10.1159/000314692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kurt A, Nijboer F, Matuz T, Kubler A. Depression and anxiety in individuals with amyotrophic lateral sclerosis: epidemiology and management. CNS Drugs. 2007;21:279–91. doi: 10.2165/00023210-200721040-00003. [DOI] [PubMed] [Google Scholar]
- LaGraize SC, Borzan J, Peng YB, Fuchs PN. Selective regulation of pain affect following activation of the opioid anterior cingulate cortex system. Exp Neurol. 2006;197:22–30. doi: 10.1016/j.expneurol.2005.05.008. [DOI] [PubMed] [Google Scholar]
- Lake AE., 3rd Chronic daily headache in the absence of medication overuse: is daily or continuous pain more treatment-resistant than chronic daily headache with pain-free days? Curr Pain Headache Rep. 2005;9:4–6. doi: 10.1007/s11916-005-0067-7. [DOI] [PubMed] [Google Scholar]
- Lasater EA, Li F, Bessler WK, Estes ML, Vemula S, Hingtgen CM, et al. Genetic and cellular evidence of vascular inflammation in neurofibromin-deficient mice and humans. J Clin Invest. 2010;120:859–70. doi: 10.1172/JCI41443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laureys S, Faymonville ME, Peigneux P, Damas P, Lambermont B, Del Fiore G, et al. Cortical processing of noxious somatosensory stimuli in the persistent vegetative state. Neuroimage. 2002;17:732–41. [PubMed] [Google Scholar]
- Lebel A, Becerra L, Wallin D, Moulton EA, Morris S, Pendse G, et al. fMRI reveals distinct CNS processing during symptomatic and recovered complex regional pain syndrome in children. Brain. 2008;131:1854–79. doi: 10.1093/brain/awn123. [DOI] [PubMed] [Google Scholar]
- Lee DM, Pendleton N, Tajar A, O'Neill TW, O'Connor DB, Bartfai G, et al. Chronic widespread pain is associated with slower cognitive processing speed in middle-aged and older European men. Pain. 2010;151:30–6. doi: 10.1016/j.pain.2010.04.024. [DOI] [PubMed] [Google Scholar]
- Lee MC, Zambreanu L, Menon DK, Tracey I. Identifying brain activity specifically related to the maintenance and perceptual consequence of central sensitization in humans. J Neurosci. 2008;28:11642–9. doi: 10.1523/JNEUROSCI.2638-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lefaucheur JP. Use of repetitive transcranial magnetic stimulation in pain relief. Expert Rev Neurother. 2008;8:799–808. doi: 10.1586/14737175.8.5.799. [DOI] [PubMed] [Google Scholar]
- Lefaucheur JP, Drouot X, Cunin P, Bruckert R, Lepetit H, Creange A, et al. Motor cortex stimulation for the treatment of refractory peripheral neuropathic pain. Brain. 2009;132:1463–71. doi: 10.1093/brain/awp035. [DOI] [PubMed] [Google Scholar]
- Lemke MR. Depressive symptoms in Parkinson's disease. Eur J Neurol. 2008;15(Suppl 1):21–5. doi: 10.1111/j.1468-1331.2008.02058.x. [DOI] [PubMed] [Google Scholar]
- Leone M. Deep brain stimulation in headache. Lancet Neurol. 2006;5:873–7. doi: 10.1016/S1474-4422(06)70575-0. [DOI] [PubMed] [Google Scholar]
- Leone M, Proietti Cecchini A, Franzini A, Broggi G, Cortelli P, Montagna P, et al. Lessons from 8 years' experience of hypothalamic stimulation in cluster headache. Cephalalgia. 2008;28:787–97. doi: 10.1111/j.1468-2982.2008.01627.x. [DOI] [PubMed] [Google Scholar]
- Lepine JP, Briley M. The epidemiology of pain in depression. Hum Psychopharmacol. 2004;19(Suppl 1):S3–7. doi: 10.1002/hup.618. [DOI] [PubMed] [Google Scholar]
- Letro GH, Quagliato EM, Viana MA. Pain in Parkinson's disease. Arq Neuropsiquiatr. 2009;67:591–4. doi: 10.1590/s0004-282x2009000400003. [DOI] [PubMed] [Google Scholar]
- Levin HS, Wilde E, Troyanskaya M, Petersen NJ, Scheibel R, Newsome M, et al. Diffusion tensor imaging of mild to moderate blast-related traumatic brain injury and its sequelae. J Neurotrauma. 2010;27:683–94. doi: 10.1089/neu.2009.1073. [DOI] [PubMed] [Google Scholar]
- Licht E, Siegler EL, Reid MC. Can the cognitively impaired safely use patient-controlled analgesia? J Opioid Manag. 2009;5:307–12. doi: 10.5055/jom.2009.0031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liguori R, Di Stasi V, Bugiardini E, Mignani R, Burlina A, Borsini W, et al. Small fiber neuropathy in female patients with fabry disease. Muscle Nerve. 2010;41:409–12. doi: 10.1002/mus.21606. [DOI] [PubMed] [Google Scholar]
- Lin CP, Kupper AE, Gammaitoni AR, Galer BS, Jensen MP. Frequency of chronic pain descriptors: Implications for assessment of pain quality. Eur J Pain. 2011;15:628–33. doi: 10.1016/j.ejpain.2010.11.006. [DOI] [PubMed] [Google Scholar]
- Lipton RB, Pearlman SH. Transcranial magnetic simulation in the treatment of migraine. Neurotherapeutics. 2010;7:204–12. doi: 10.1016/j.nurt.2010.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lipton RB, Bigal ME, Diamond M, Freitag F, Reed ML, Stewart WF. Migraine prevalence, disease burden, and the need for preventive therapy. Neurology. 2007;68:343–9. doi: 10.1212/01.wnl.0000252808.97649.21. [DOI] [PubMed] [Google Scholar]
- Lohle M, Storch A, Reichmann H. Beyond tremor and rigidity: non-motor features of Parkinson's disease. J Neural Transm. 2009;116:1483–92. doi: 10.1007/s00702-009-0274-1. [DOI] [PubMed] [Google Scholar]
- Lu-Emerson C, Plotkin SR. The neurofibromatoses. Part 2: NF2 and schwannomatosis. Rev Neurol Dis. 2009;6:E81–6. [PubMed] [Google Scholar]
- Lu-Emerson C, Plotkin SR. The Neurofibromatoses. Part 1: NF1. Rev Neurol Dis. 2009;6:E47–53. [PubMed] [Google Scholar]
- Lule D, Diekmann V, Muller HP, Kassubek J, Ludolph AC, Birbaumer N. Neuroimaging of multimodal sensory stimulation in amyotrophic lateral sclerosis. J Neurol Neurosurg Psychiatry. 2010;81:899–906. doi: 10.1136/jnnp.2009.192260. [DOI] [PubMed] [Google Scholar]
- Lumer ED, Edelman GM, Tononi G. Neural dynamics in a model of the thalamocortical system. I. Layers, loops and the emergence of fast synchronous rhythms. Cereb Cortex. 1997;7:207–27. doi: 10.1093/cercor/7.3.207. [DOI] [PubMed] [Google Scholar]
- Lyczak P, Lyczak-Rucinska M. Chronic post-traumatic headache and brain perfusion changes assessed using magnetic resonance imaging. Neurol Neurochir Pol. 2005;39:S49–54. [PubMed] [Google Scholar]
- Maarrawi J, Peyron R, Mertens P, Costes N, Magnin M, Sindou M, et al. Differential brain opioid receptor availability in central and peripheral neuropathic pain. Pain. 2007;127:183–94. doi: 10.1016/j.pain.2006.10.013. [DOI] [PubMed] [Google Scholar]
- MacCollin M, Chiocca EA, Evans DG, Friedman JM, Horvitz R, Jaramillo D, et al. Diagnostic criteria for schwannomatosis. Neurology. 2005;64:1838–45. doi: 10.1212/01.WNL.0000163982.78900.AD. [DOI] [PubMed] [Google Scholar]
- Mahoney AE, Peters L. The Mahoney Pain Scale: examining pain and agitation in advanced dementia. Am J Alzheimers Dis Other Demen. 2008;23:250–61. doi: 10.1177/1533317508317524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maihofner C, Seifert F, Markovic K. Complex regional pain syndromes: new pathophysiological concepts and therapies. Eur J Neurol. 2010;17:649–60. doi: 10.1111/j.1468-1331.2010.02947.x. [DOI] [PubMed] [Google Scholar]
- Maihofner C, Jesberger F, Seifert F, Kaltenhauser M. Cortical processing of mechanical hyperalgesia: a MEG study. Eur J Pain. 2010;14:64–70. doi: 10.1016/j.ejpain.2009.02.007. [DOI] [PubMed] [Google Scholar]
- Mailis-Gagnon A, Nicholson K. Nondermatomal somatosensory deficits: overview of unexplainable negative sensory phenomena in chronic pain patients. Curr Opin Anaesthesiol. 2010;23:593–7. doi: 10.1097/ACO.0b013e32833dd01c. [DOI] [PubMed] [Google Scholar]
- Mailis-Gagnon A, Giannoylis I, Downar J, Kwan CL, Mikulis DJ, Crawley AP, et al. Altered central somatosensory processing in chronic pain patients with “hysterical” anesthesia. Neurology. 2003;60:1501–7. doi: 10.1212/wnl.60.9.1501. [DOI] [PubMed] [Google Scholar]
- Maleki J, LeBel AA, Bennett GJ, Schwartzman RJ. Patterns of spread in complex regional pain syndrome, type I (reflex sympathetic dystrophy) Pain. 2000;88:259–66. doi: 10.1016/S0304-3959(00)00332-8. [DOI] [PubMed] [Google Scholar]
- Maletic V, Raison CL. Neurobiology of depression, fibromyalgia and neuropathic pain. Front Biosci. 2009;14:5291–338. doi: 10.2741/3598. [DOI] [PubMed] [Google Scholar]
- Mallen C, Peat G, Thomas E, Croft P. Severely disabling chronic pain in young adults: prevalence from a population-based postal survey in North Staffordshire. BMC Musculoskelet Disord. 2005;6:42. doi: 10.1186/1471-2474-6-42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manack AN, Buse DC, Lipton RB. Chronic migraine: epidemiology and disease burden. Curr Pain Headache Rep. 2011;15:70–8. doi: 10.1007/s11916-010-0157-z. [DOI] [PubMed] [Google Scholar]
- Manchikanti L, Singh V, Datta S, Cohen SP, Hirsch JA. Comprehensive review of epidemiology, scope, and impact of spinal pain. Pain Physician. 2009;12:E35–70. [PubMed] [Google Scholar]
- Marasco PD, Kim K, Colgate JE, Peshkin MA, Kuiken TA. Robotic touch shifts perception of embodiment to a prosthesis in targeted reinnervation amputees. Brain. 2011;134:747–58. doi: 10.1093/brain/awq361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin BA, Labuda R, Royston TJ, Oshinski JN, Iskandar B, Loth F. Spinal subarachnoid space pressure measurements in an in vitro spinal stenosis model: implications on syringomyelia theories. J Biomech Eng. 2010;132:111007. doi: 10.1115/1.4000089. [DOI] [PubMed] [Google Scholar]
- Martin E, Jeanmonod D, Morel A, Zadicario E, Werner B. High-intensity focused ultrasound for noninvasive functional neurosurgery. Ann Neurol. 2009;66:858–61. doi: 10.1002/ana.21801. [DOI] [PubMed] [Google Scholar]
- Masson C, Koskas P, Cambier J, Masson M. Left pseudothalamic cortical syndrome and pain asymbolia. Rev Neurol. 1991;147:668–70. [PubMed] [Google Scholar]
- May A. Morphing voxels: the hype around structural imaging of headache patients. Brain. 2009;132:1419–25. doi: 10.1093/brain/awp116. [DOI] [PubMed] [Google Scholar]
- May A. New insights into headache: an update on functional and structural imaging findings. Nat Rev Neurol. 2009;5:199–209. doi: 10.1038/nrneurol.2009.28. [DOI] [PubMed] [Google Scholar]
- McWilliams LA, Goodwin RD, Cox BJ. Depression and anxiety associated with three pain conditions: results from a nationally representative sample. Pain. 2004;111:77–83. doi: 10.1016/j.pain.2004.06.002. [DOI] [PubMed] [Google Scholar]
- Medina FJ, Tunez I. Huntington's disease: the value of transcranial meganetic stimulation. Curr Med Chem. 2010;17:2482–91. doi: 10.2174/092986710791556078. [DOI] [PubMed] [Google Scholar]
- Merkl A, Heuser I, Bajbouj M. Antidepressant electroconvulsive therapy: mechanism of action, recent advances and limitations. Exp Neurol. 2009;219:20–6. doi: 10.1016/j.expneurol.2009.04.027. [DOI] [PubMed] [Google Scholar]
- Merson TD, Binder MD, Kilpatrick TJ. Role of cytokines as mediators and regulators of microglial activity in inflammatory demyelination of the CNS. Neuromolecular Med. 2010;12:99–132. doi: 10.1007/s12017-010-8112-z. [DOI] [PubMed] [Google Scholar]
- Metz AE, Yau HJ, Centeno MV, Apkarian AV, Martina M. Morphological and functional reorganization of rat medial prefrontal cortex in neuropathic pain. Proc Natl Acad Sci USA. 2009;106:2423–8. doi: 10.1073/pnas.0809897106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mhuircheartaigh RN, Rosenorn-Lanng D, Wise R, Jbabdi S, Rogers R, Tracey I. Cortical and subcortical connectivity changes during decreasing levels of consciousness in humans: a functional magnetic resonance imaging study using propofol. J Neurosci. 2010;30:9095–102. doi: 10.1523/JNEUROSCI.5516-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milhorat TH, Kotzen RM, Mu HT, Capocelli AL, Jr, Milhorat RH. Dysesthetic pain in patients with syringomyelia. Neurosurgery. 1996;38:940–6. doi: 10.1097/00006123-199605000-00017. discussion 6–7. [DOI] [PubMed] [Google Scholar]
- Millecamps M, Centeno MV, Berra HH, Rudick CN, Lavarello S, Tkatch T, et al. D-cycloserine reduces neuropathic pain behavior through limbic NMDA-mediated circuitry. Pain. 2007;132:108–23. doi: 10.1016/j.pain.2007.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Modirian E, Pirouzi P, Soroush M, Karbalaei-Esmaeili S, Shojaei H, Zamani H. Chronic pain after spinal cord injury: results of a long-term study. Pain Med. 2010;11:1037–43. doi: 10.1111/j.1526-4637.2010.00865.x. [DOI] [PubMed] [Google Scholar]
- Moller AT, Bach FW, Feldt-Rasmussen U, Rasmussen AK, Hasholt L, Sommer C, et al. Autonomic skin responses in females with Fabry disease. J Peripher Nerv Syst. 2009;14:159–64. doi: 10.1111/j.1529-8027.2009.00227.x. [DOI] [PubMed] [Google Scholar]
- Monti MM, Vanhaudenhuyse A, Coleman MR, Boly M, Pickard JD, Tshibanda L, et al. Willful modulation of brain activity in disorders of consciousness. N Engl J Med. 2010;362:579–89. doi: 10.1056/NEJMoa0905370. [DOI] [PubMed] [Google Scholar]
- Moore DF, Ye F, Schiffmann R, Butman JA. Increased signal intensity in the pulvinar on T1-weighted images: a pathognomonic MR imaging sign of Fabry disease. AJNR Am J Neuroradiol. 2003;24:1096–101. [PMC free article] [PubMed] [Google Scholar]
- Moriarty O, McGuire BE, Finn DP. The effect of pain on cognitive function: a review of clinical and preclinical research. Prog Neurobiol. 2011;93:385–404. doi: 10.1016/j.pneurobio.2011.01.002. [DOI] [PubMed] [Google Scholar]
- Moseley GL. Graded motor imagery for pathologic pain: a randomized controlled trial. Neurology. 2006;67:2129–34. doi: 10.1212/01.wnl.0000249112.56935.32. [DOI] [PubMed] [Google Scholar]
- Moulin DE, Hagen N, Feasby TE, Amireh R, Hahn A. Pain in Guillain-Barre syndrome. Neurology. 1997;48:328–31. doi: 10.1212/wnl.48.2.328. [DOI] [PubMed] [Google Scholar]
- Moulton EA, Schmahmann JD, Becerra L, Borsook D. The cerebellum and pain: passive integrator or active participator? Brain Res Rev. 2010;65:14–27. doi: 10.1016/j.brainresrev.2010.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moulton EA, Burstein R, Tully S, Hargreaves R, Becerra L, Borsook D. Interictal dysfunction of a brainstem descending modulatory center in migraine patients. PLoS One. 2008;3:e3799. doi: 10.1371/journal.pone.0003799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moulton EA, Becerra L, Maleki N, Pendse G, Tully S, Hargreaves R, et al. Painful heat reveals hyperexcitability of the temporal pole in interictal and ictal migraine States. Cereb Cortex. 2010;21:435–48. doi: 10.1093/cercor/bhq109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moulton EA, Becerra L, Maleki N, Pendse G, Tully S, Hargreaves R, et al. Painful heat reveals hyperexcitability of the temporal pole in interictal and ictal migraine States. Cereb Cortex. 2011;21:435–48. doi: 10.1093/cercor/bhq109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakagawa T, Kaneko S. Spinal astrocytes as therapeutic targets for pathological pain. J Pharmacol Sci. 2010;114:347–53. doi: 10.1254/jphs.10r04cp. [DOI] [PubMed] [Google Scholar]
- Nampiaparampil DE. Prevalence of chronic pain after traumatic brain injury: a systematic review. JAMA. 2008;300:711–9. doi: 10.1001/jama.300.6.711. [DOI] [PubMed] [Google Scholar]
- Nandhagopal R, Troiano AR, Mak E, Schulzer M, Bushnell MC, Stoessl AJ. Response to heat pain stimulation in idiopathic Parkinson's disease. Pain Med. 2010;11:834–40. doi: 10.1111/j.1526-4637.2010.00866.x. [DOI] [PubMed] [Google Scholar]
- Napadow V, LaCount L, Park K, As-Sanie S, Clauw DJ, Harris RE. Intrinsic brain connectivity in fibromyalgia is associated with chronic pain intensity. Arthritis Rheum. 2010;62:2545–55. doi: 10.1002/art.27497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nebe A, Ebersbach G. Pain intensity on and off levodopa in patients with Parkinson's disease. Mov Disord. 2009;24:1233–7. doi: 10.1002/mds.22546. [DOI] [PubMed] [Google Scholar]
- Ni Z, Pinto AD, Lang AE, Chen R. Involvement of the cerebellothalamocortical pathway in Parkinson disease. Ann Neurol. 2010;68:816–24. doi: 10.1002/ana.22221. [DOI] [PubMed] [Google Scholar]
- Nill M, Muller MJ, Beck M, Stoeter P, Fellgiebel A. Pathophysiological aspects of brain structural disturbances in patients with Fabry disease: literature review. Fortschr Neurol Psychiatr. 2006;74:687–95. doi: 10.1055/s-2006-932190. [DOI] [PubMed] [Google Scholar]
- Noseda R, Kainz V, Jakubowski M, Gooley JJ, Saper CB, Digre K, et al. A neural mechanism for exacerbation of headache by light. Nat Neurosci. 2010;13:239–45. doi: 10.1038/nn.2475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nurnberger M, Moll I. Semiquantitative aspects of mast cells in normal skin and in neurofibromas of neurofibromatosis types 1 and 5. Dermatology. 1994;188:296–9. doi: 10.1159/000247170. [DOI] [PubMed] [Google Scholar]
- Nuti C, Peyron R, Garcia-Larrea L, Brunon J, Laurent B, Sindou M, et al. Motor cortex stimulation for refractory neuropathic pain: four year outcome and predictors of efficacy. Pain. 2005;118:43–52. doi: 10.1016/j.pain.2005.07.020. [DOI] [PubMed] [Google Scholar]
- O'Connor AB, Schwid SR, Herrmann DN, Markman JD, Dworkin RH. Pain associated with multiple sclerosis: systematic review and proposed classification. Pain. 2008;137:96–111. doi: 10.1016/j.pain.2007.08.024. [DOI] [PubMed] [Google Scholar]
- Odrcich M, Bailey JM, Cahill CM, Gilron I. Chronobiological characteristics of painful diabetic neuropathy and postherpetic neuralgia: diurnal pain variation and effects of analgesic therapy. Pain. 2006;120:207–12. doi: 10.1016/j.pain.2005.10.007. [DOI] [PubMed] [Google Scholar]
- Oertel B, Lotsch J. Genetic mutations that prevent pain: implications for future pain medication. Pharmacogenomics. 2008;9:179–94. doi: 10.2217/14622416.9.2.179. [DOI] [PubMed] [Google Scholar]
- Office of Communications and Public Liaison NIoNDaS, National Institutes of Health. Complex Regional Pain Syndrome Fact Sheet 2003. Bethesda, MD: National Institutes of Health; 2003. Report No.: NIH publication 04-4173. [Google Scholar]
- Ogawa M, Shigeto H, Yamamoto T, Oya Y, Wada K, Nishikawa T, et al. D-cycloserine for the treatment of ataxia in spinocerebellar degeneration. J Neurol Sci. 2003;210:53–6. doi: 10.1016/s0022-510x(03)00009-1. [DOI] [PubMed] [Google Scholar]
- Onishi A, Dyck PJ. Loss of small peripheral sensory neurons in Fabry disease. Histologic and morphometric evaluation of cutaneous nerves, spinal ganglia, and posterior columns. Arch Neurol. 1974;31:120–7. doi: 10.1001/archneur.1974.00490380068009. [DOI] [PubMed] [Google Scholar]
- Owen AM, Coleman MR, Boly M, Davis MH, Laureys S, Pickard JD. Detecting awareness in the vegetative state. Science. 2006;313:1402. doi: 10.1126/science.1130197. [DOI] [PubMed] [Google Scholar]
- Owen AM, Coleman MR, Boly M, Davis MH, Laureys S, Pickard JD. Using functional magnetic resonance imaging to detect covert awareness in the vegetative state. Arch Neurol. 2007;64:1098–102. doi: 10.1001/archneur.64.8.1098. [DOI] [PubMed] [Google Scholar]
- Owen SL, Green AL, Stein JF, Aziz TZ. Deep brain stimulation for the alleviation of post-stroke neuropathic pain. Pain. 2006;120:202–6. doi: 10.1016/j.pain.2005.09.035. [DOI] [PubMed] [Google Scholar]
- Pagnini I, Borsini W, Cecchi F, Sgalambro A, Olivotto I, Frullini A, et al. Distal limb pain as presenting feature of fabry disease. Arthritis Care Res. 2011;63:390–5. doi: 10.1002/acr.20385. [DOI] [PubMed] [Google Scholar]
- Parvizi J, Van Hoesen GW, Damasio A. Selective pathological changes of the periaqueductal gray matter in Alzheimer's disease. Ann Neurol. 2000;48:344–53. [PubMed] [Google Scholar]
- Paulsen JS, Nehl C, Hoth KF, Kanz JE, Benjamin M, Conybeare R, et al. Depression and stages of Huntington's disease. J Neuropsychiatry Clin Neurosci. 2005;17:496–502. doi: 10.1176/jnp.17.4.496. [DOI] [PubMed] [Google Scholar]
- Pautex S, Michon A, Guedira M, Emond H, Le Lous P, Samaras D, et al. Pain in severe dementia: self-assessment or observational scales? J Am Geriatr Soc. 2006;54:1040–5. doi: 10.1111/j.1532-5415.2006.00766.x. [DOI] [PubMed] [Google Scholar]
- Perkins DO, Stern RA, Golden RN, Murphy C, Naftolowitz D, Evans DL. Mood disorders in HIV infection: prevalence and risk factors in a nonepicenter of the AIDS epidemic. Am J Psychiatry. 1994;151:233–6. doi: 10.1176/ajp.151.2.233. [DOI] [PubMed] [Google Scholar]
- Perrotta A, Sandrini G, Serrao M, Buscone S, Tassorelli C, Tinazzi M, et al. Facilitated temporal summation of pain at spinal level in Parkinson's disease. Mov Disord. 2010;26:442–8. doi: 10.1002/mds.23458. [DOI] [PubMed] [Google Scholar]
- Philip A, Thakur R. Post herpetic neuralgia. J Palliat Med. 2011;14:765–73. doi: 10.1089/jpm.2011.9685. [DOI] [PubMed] [Google Scholar]
- Pickering G, Eschalier A, Dubray C. Pain and Alzheimer's disease. Gerontology. 2000;46:235–41. doi: 10.1159/000022166. [DOI] [PubMed] [Google Scholar]
- Popa T, Russo M, Meunier S. Long-lasting inhibition of cerebellar output. Brain Stimul. 2010;3:161–9. doi: 10.1016/j.brs.2009.10.001. [DOI] [PubMed] [Google Scholar]
- Popeo D, Kellner CH. ECT for Parkinson's disease. Med Hypotheses. 2009;73:468–9. doi: 10.1016/j.mehy.2009.06.053. [DOI] [PubMed] [Google Scholar]
- Pudas-Tahka SM, Axelin A, Aantaa R, Lund V, Salantera S. Pain assessment tools for unconscious or sedated intensive care patients: a systematic review. J Adv Nurs. 2009;65:946–56. doi: 10.1111/j.1365-2648.2008.04947.x. [DOI] [PubMed] [Google Scholar]
- Pugdahl K, Fuglsang-Frederiksen A, de Carvalho M, Johnsen B, Fawcett PR, Labarre-Vila A, et al. Generalised sensory system abnormalities in amyotrophic lateral sclerosis: a European multicentre study. J Neurol Neurosurg Psychiatry. 2007;78:746–9. doi: 10.1136/jnnp.2006.098533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rabinak CA, Nirenberg MJ. Dopamine agonist withdrawal syndrome in Parkinson disease. Arch Neurol. 2010;67:58–63. doi: 10.1001/archneurol.2009.294. [DOI] [PubMed] [Google Scholar]
- Raghupathi R, Margulies SS. Traumatic axonal injury after closed head injury in the neonatal pig. J Neurotrauma. 2002;19:843–53. doi: 10.1089/08977150260190438. [DOI] [PubMed] [Google Scholar]
- Rainero I, Vighetti S, Bergamasco B, Pinessi L, Benedetti F. Autonomic responses and pain perception in Alzheimer's disease. Eur J Pain. 2000;4:267–74. doi: 10.1053/eujp.2000.0185. [DOI] [PubMed] [Google Scholar]
- Ramachandran VS, Altschuler EL. The use of visual feedback, in particular mirror visual feedback, in restoring brain function. Brain. 2009;132:1693–710. doi: 10.1093/brain/awp135. [DOI] [PubMed] [Google Scholar]
- Ramachandran VS, Brang D, McGeoch PD. Dynamic reorganization of referred sensations by movements of phantom limbs. Neuroreport. 2010;21:727–30. doi: 10.1097/WNR.0b013e32833be9ab. [DOI] [PubMed] [Google Scholar]
- Raval A, Dhanaraj E, Bhansali A, Grover S, Tiwari P. Prevalence & determinants of depression in type 2 diabetes patients in a tertiary care centre. Indian J Med Res. 2010;132:195–200. [PubMed] [Google Scholar]
- Ren K, Dubner R. Interactions between the immune and nervous systems in pain. Nat Med. 2010;6:1267–76. doi: 10.1038/nm.2234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Risdall JE, Menon DK. Traumatic brain injury. Philos Trans R Soc Lond B Biol Sci. 2011;366:241–50. doi: 10.1098/rstb.2010.0230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ro T, Farne A, Johnson RM, Wedeen V, Chu Z, Wang ZJ, et al. Feeling sounds after a thalamic lesion. Ann Neurol. 2007;62:433241. doi: 10.1002/ana.21219. [DOI] [PubMed] [Google Scholar]
- Robbins MS, Lipton RB. The epidemiology of primary headache disorders. Semin Neurol. 2010;30:107–19. doi: 10.1055/s-0030-1249220. [DOI] [PubMed] [Google Scholar]
- Rodrigues LG, Ferraz MJ, Rodrigues D, Pais-Vieira M, Lima D, Brady RO, et al. Neurophysiological, behavioral and morphological abnormalities in the Fabry knockout mice. Neurobiol Dis. 2009;33:48256. doi: 10.1016/j.nbd.2008.09.001. [DOI] [PubMed] [Google Scholar]
- Rodriguez-Raecke R, Niemeier A, Ihle K, Ruether W, May A. Brain gray matter decrease in chronic pain is the consequence and not the cause of pain. J Neurosci. 2009;29:13746–50. doi: 10.1523/JNEUROSCI.3687-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosemberg S, Marie SK, Kliemann S. Congenital insensitivity to pain with anhidrosis (hereditary sensory and autonomic neuropathy type IV) Pediatr Neurol. 1994;11:50–6. doi: 10.1016/0887-8994(94)90091-4. [DOI] [PubMed] [Google Scholar]
- Ross CA, Tabrizi SJ. Huntington's disease: from molecular pathogenesis to clinical treatment. Lancet Neurol. 2011;10:83–98. doi: 10.1016/S1474-4422(10)70245-3. [DOI] [PubMed] [Google Scholar]
- Rub U, Brunt ER, Deller T. New insights into the pathoanatomy of spinocerebellar ataxia type 3 (Machado-Joseph disease) Curr Opin Neurol. 2008;21:111–6. doi: 10.1097/WCO.0b013e3282f7673d. [DOI] [PubMed] [Google Scholar]
- Rudelli RD, Ambler MW, Wisniewski HM. Morphology and distribution of Alzheimer neuritic (senile) and amyloid plaques in striatum and diencephalon. Acta Neuropathol. 1984;64:273–81. doi: 10.1007/BF00690393. [DOI] [PubMed] [Google Scholar]
- Russell IJ, Raphael KG. Fibromyalgia syndrome: presentation, diagnosis, differential diagnosis, and vulnerability. CNS Spectr. 2008;13:6–11. doi: 10.1017/s1092852900026778. [DOI] [PubMed] [Google Scholar]
- Ruts L, Drenthen J, Jongen JL, Hop WC, Visser GH, Jacobs BC, et al. Pain in Guillain-Barre syndrome: a long-term follow-up study. Neurology. 2010;75:143947. doi: 10.1212/WNL.0b013e3181f88345. [DOI] [PubMed] [Google Scholar]
- Sassone J, Colciago C, Cislaghi G, Silani V, Ciammola A. Huntington's disease: the current state of research with peripheral tissues. Exp Neurol. 2009;219:385–97. doi: 10.1016/j.expneurol.2009.05.012. [DOI] [PubMed] [Google Scholar]
- Saydam O, Ozdener GB, Senol O, Mizrak A, Prabhakar S, Stemmer-Rachamimov AO, et al. A novel imaging-compatible sciatic nerve schwannoma model. J Neurosci Methods. 2011;195:75–7. doi: 10.1016/j.jneumeth.2010.10.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scherder E, Statema M. Huntington's disease. Lancet. 2010;376:1464. doi: 10.1016/S0140-6736(10)61990-3. [DOI] [PubMed] [Google Scholar]
- Scherder E, Bouma A, Slaets J, Ooms M, Ribbe M, Blok A, et al. Repeated pain assessment in Alzheimer's disease. Dement Geriatr Cogn Disord. 2001;12:400–7. doi: 10.1159/000051287. [DOI] [PubMed] [Google Scholar]
- Scherder EJ, Sergeant JA, Swaab DF. Pain processing in dementia and its relation to neuropathology. Lancet Neurol. 2003;2:677–86. doi: 10.1016/s1474-4422(03)00556-8. [DOI] [PubMed] [Google Scholar]
- Schermuly I, Muller MJ, Muller KM, Albrecht J, Keller I, Yakushev I, et al. Neuropsychiatric symptoms and brain structural alterations in Fabry disease. Eur J Neurol. 2010;18:347–53. doi: 10.1111/j.1468-1331.2010.03155.x. [DOI] [PubMed] [Google Scholar]
- Schestatsky P, Kumru H, Valls-Sole J, Valldeoriola F, Marti MJ, Tolosa E, et al. Neurophysiologic study of central pain in patients with Parkinson disease. Neurology. 2007;69:2162–9. doi: 10.1212/01.wnl.0000295669.12443.d3. [DOI] [PubMed] [Google Scholar]
- Schiffmann R, Kopp JB, Austin HA, 3rd, Sabnis S, Moore DF, Weibel T, et al. Enzyme replacement therapy in Fabry disease: a randomized controlled trial. JAMA. 2001;285:2743–9. doi: 10.1001/jama.285.21.2743. [DOI] [PubMed] [Google Scholar]
- Schmahmann JD, Leifer D. Parietal pseudothalamic pain syndrome. Clinical features and anatomic correlates. Arch Neurol. 1992;49:1032–7. doi: 10.1001/archneur.1992.00530340048017. [DOI] [PubMed] [Google Scholar]
- Schmidt R, Bach M, Dal-Bianco P, Holzer P, Pluta-Fuerst A, Assem-Hilger E, et al. Dementia and pain. Neuropsychiatr. 2010;24:1–13. [PubMed] [Google Scholar]
- Schmitz-Hubsch T, Coudert M, Bauer P, Giunti P, Globas C, Baliko L, et al. Spinocerebellar ataxia types 1, 2, 3, and 6: disease severity and nonataxia symptoms. Neurology. 2008;71:982–9. doi: 10.1212/01.wnl.0000325057.33666.72. [DOI] [PubMed] [Google Scholar]
- Schnakers C, Chatelle C, Vanhaudenhuyse A, Majerus S, Ledoux D, Boly M, et al. The Nociception Coma Scale: a new tool to assess nociception in disorders of consciousness. Pain. 2010;148:215–9. doi: 10.1016/j.pain.2009.09.028. [DOI] [PubMed] [Google Scholar]
- Scholz J, Mannion RJ, Hord DE, Griffin RS, Rawal B, Zheng H, et al. A novel tool for the assessment of pain: validation in low back pain. PLoS Med. 2009;6:e1000047. doi: 10.1371/journal.pmed.1000047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schonfeldt-Lecuona C, Cardenas-Morales L, Freudenmann RW, Kammer T, Herwig U. Transcranial magnetic stimulation in depression–lessons from the multicentre trials. Restor Neurol Neurosci. 2010;28:569–76. doi: 10.3233/RNN-2010-0561. [DOI] [PubMed] [Google Scholar]
- Schott GD. Delayed onset and resolution of pain: some observations and implications. Brain. 2001;124:1067–76. doi: 10.1093/brain/124.6.1067. [DOI] [PubMed] [Google Scholar]
- Schurch B, Wichmann W, Rossier AB. Post-traumatic syringomyelia (cystic myelopathy): a prospective study of 449 patients with spinal cord injury. J Neurol Neurosurg Psychiatry. 1996;60:61–7. doi: 10.1136/jnnp.60.1.61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwenkreis P, Scherens A, Ronnau AK, Hoffken O, Tegenthoff M, Maier C. Cortical disinhibition occurs in chronic neuropathic, but not in chronic nociceptive pain. BMC Neurosci. 2010;11:73. doi: 10.1186/1471-2202-11-73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scott DJ, Heitzeg MM, Koeppe RA, Stohler CS, Zubieta JK. Variations in the human pain stress experience mediated by ventral and dorsal basal ganglia dopamine activity. J Neurosci. 2006;26:10789–95. doi: 10.1523/JNEUROSCI.2577-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seghier ML, Lazeyras F, Vuilleumier P, Schnider A, Carota A. Functional magnetic resonance imaging and diffusion tensor imaging in a case of central poststroke pain. J Pain. 2005;6:208–12. doi: 10.1016/j.jpain.2004.11.004. [DOI] [PubMed] [Google Scholar]
- Seifert F, Kiefer G, DeCol R, Schmelz M, Maihofner C. Differential endogenous pain modulation in complex-regional pain syndrome. Brain. 2009;132:788–800. doi: 10.1093/brain/awn346. [DOI] [PubMed] [Google Scholar]
- Selles RW, Schreuders TA, Stam HJ. Mirror therapy in patients with causalgia (complex regional pain syndrome type II) following peripheral nerve injury: two cases. J Rehabil Med. 2008;40:312–4. doi: 10.2340/16501977-0158. [DOI] [PubMed] [Google Scholar]
- Seminowicz DA, Wideman TH, Naso L, Hatami-Khoroushahi Z, Fallatah S, Ware MA, et al. Effective treatment of chronic low back pain in humans reverses abnormal brain anatomy and function. J Neurosci. 2011;31:7540–50. doi: 10.1523/JNEUROSCI.5280-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Serletis D, Parkin P, Bouffet E, Shroff M, Drake JM, Rutka JT. Massive plexiform neurofibromas in childhood: natural history and management issues. J Neurosurg. 2007;106:363–7. doi: 10.3171/ped.2007.106.5.363. [DOI] [PubMed] [Google Scholar]
- Serra J. Microneurography: an opportunity for translational drug development in neuropathic pain. Neurosci Lett. 2010;470:155–7. doi: 10.1016/j.neulet.2009.12.065. [DOI] [PubMed] [Google Scholar]
- Shega J, Emanuel L, Vargish L, Levine SK, Bursch H, Herr K, et al. Pain in persons with dementia: complex, common, and challenging. J Pain. 2007;8:373–8. doi: 10.1016/j.jpain.2007.03.003. [DOI] [PubMed] [Google Scholar]
- Shega JW, Rudy T, Keefe FJ, Perri LC, Mengin OT, Weiner DK. Validity of pain behaviors in persons with mild to moderate cognitive impairment. J Am Geriatr Soc. 2008;56:1631–7. doi: 10.1111/j.1532-5415.2008.01831.x. [DOI] [PubMed] [Google Scholar]
- Siegert RJ, Abernethy DA. Depression in multiple sclerosis: a review. J Neurol Neurosurg Psychiatry. 2005;76:469–75. doi: 10.1136/jnnp.2004.054635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simuni T, Sethi K. Nonmotor manifestations of Parkinson's disease. Ann Neurol. 2008;64(Suppl 2):S65–80. doi: 10.1002/ana.21472. [DOI] [PubMed] [Google Scholar]
- Slater R, Cantarella A, Gallella S, Worley A, Boyd S, Meek J, et al. Cortical pain responses in human infants. J Neurosci. 2006;26:3662–6. doi: 10.1523/JNEUROSCI.0348-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sloane E, Ledeboer A, Seibert W, Coats B, van Strien M, Maier SF, et al. Anti-inflammatory cytokine gene therapy decreases sensory and motor dysfunction in experimental multiple sclerosis: MOG-EAE behavioral and anatomical symptom treatment with cytokine gene therapy. Brain Behav Immun. 2009;23:92–100. doi: 10.1016/j.bbi.2008.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith DL, Woodman B, Mahal A, Sathasivam K, Ghazi-Noori S, Lowden PA, et al. Minocycline and doxycycline are not beneficial in a model of Huntington's disease. Ann Neurol. 2003;54:186–96. doi: 10.1002/ana.10614. [DOI] [PubMed] [Google Scholar]
- Smith HS, Harris R, Clauw D. Fibromyalgia: an afferent processing disorder leading to a complex pain generalized syndrome. Pain Physician. 2011;14:E217–45. [PubMed] [Google Scholar]
- Solaro C, Messmer Uccelli M. Pharmacological management of pain in patients with multiple sclerosis. Drugs. 2010;70:1245–54. doi: 10.2165/11537930-000000000-00000. [DOI] [PubMed] [Google Scholar]
- Sommer C. Painful neuropathies. Curr Opin Neurol. 2003;16:623–8. doi: 10.1097/01.wco.0000093106.34793.06. [DOI] [PubMed] [Google Scholar]
- Sonkusare SK, Kaul CL, Ramarao P. Dementia of Alzheimer's disease and other neurodegenerative disorders–memantine, a new hope. Pharmacol Res. 2005;51:1–17. doi: 10.1016/j.phrs.2004.05.005. [DOI] [PubMed] [Google Scholar]
- Stacy MA, Murck H, Kroenke K. Responsiveness of motor and nonmotor symptoms of Parkinson disease to dopaminergic therapy. Prog Neuropsychopharmacol Biol Psychiatry. 2010;34:57–61. doi: 10.1016/j.pnpbp.2009.09.023. [DOI] [PubMed] [Google Scholar]
- Staser K, Yang FC, Clapp DW. Plexiform neurofibroma genesis: questions of Nf1 gene dose and hyperactive mast cells. Curr Opin Hematol. 2010;17:287–93. doi: 10.1097/MOH.0b013e328339511b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Staud R. Chronic widespread pain and fibromyalgia: two sides of the same coin? Curr Rheumatol Rep. 2009;11:433–6. doi: 10.1007/s11926-009-0063-8. [DOI] [PubMed] [Google Scholar]
- Stoodley CJ, Schmahmann JD. Evidence for topographic organization in the cerebellum of motor control versus cognitive and affective processing. Cortex. 2010;46:831–44. doi: 10.1016/j.cortex.2009.11.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strauss DJ, Ashwal S, Day SM, Shavelle RM. Life expectancy of children in vegetative and minimally conscious states. Pediatr Neurol. 2000;23:312–9. doi: 10.1016/s0887-8994(00)00194-6. [DOI] [PubMed] [Google Scholar]
- Strober LB, Arnett PA. Assessment of depression in three medically ill, elderly populations: Alzheimer's disease, Parkinson's disease, and stroke. Clin Neuropsychol. 2009;23:205–30. doi: 10.1080/13854040802003299. [DOI] [PubMed] [Google Scholar]
- Suda S, Takagai S, Inoshima-Takahashi K, Sugihara G, Mori N, Takei N. Electroconvulsive therapy for burning mouth syndrome. Acta Psychiatr Scand. 2008;118:503–4. doi: 10.1111/j.1600-0447.2008.01261.x. [DOI] [PubMed] [Google Scholar]
- Sullivan MJ, Reesor K, Mikail S, Fisher R. The treatment of depression in chronic low back pain: review and recommendations. Pain. 1992;50:5–13. doi: 10.1016/0304-3959(92)90107-M. [DOI] [PubMed] [Google Scholar]
- Sumitani M, Miyauchi S, Uematsu H, Yozu A, Otake Y, Yamada Y. Phantom limb pain originates from dysfunction of the primary motor cortex. Masui. 2010;59:1364–9. [PubMed] [Google Scholar]
- Suzuki K, Ebina Y, Shindo T, Takano T, Awata S, Matsuoka H. Repeated electroconvulsive therapy courses improved chronic regional pain with depression caused by failed back syndrome. Med Sci Monit. 2009;15:CS77–9. [PubMed] [Google Scholar]
- Sweitzer S, De Leo J. Propentofylline: glial modulation, neuroprotection, and alleviation of chronic pain. Handb Exp Pharmacol. 2011;200:235–50. doi: 10.1007/978-3-642-13443-2_8. [DOI] [PubMed] [Google Scholar]
- Tegeder I, Costigan M, Griffin RS, Abele A, Belfer I, Schmidt H, et al. GTP cyclohydrolase and tetrahydrobiopterin regulate pain sensitivity and persistence. Nat Med. 2006;12:1269–77. doi: 10.1038/nm1490. [DOI] [PubMed] [Google Scholar]
- Thrush DC. Congenital insensitivity to pain. A clinical, genetic and neurophysiological study of four children from the same family. Brain. 1973;96:369–86. doi: 10.1093/brain/96.2.369. [DOI] [PubMed] [Google Scholar]
- Todor DR, Mu HT, Milhorat TH. Pain and syringomyelia: a review. Neurosurg Focus. 2000;8:E11. doi: 10.3171/foc.2000.8.3.11. [DOI] [PubMed] [Google Scholar]
- Toledo MA, Junqueira LF., Jr Cardiac autonomic modulation and cognitive status in Alzheimer's disease. Clin Auton Res. 2010;20:11–7. doi: 10.1007/s10286-009-0035-0. [DOI] [PubMed] [Google Scholar]
- Tomioka T, Awaya Y, Nihei K, Hanaoka K. Post-herpetic neuralgia in a patient with congenital insensitivity to pain and anhidrosis. J Anesth. 2002;16:84–6. doi: 10.1007/s540-002-8100-y. [DOI] [PubMed] [Google Scholar]
- Tracey I. Can neuroimaging studies identify pain endophenotypes in humans? Nat Rev Neurol. 2011;7:173–81. doi: 10.1038/nrneurol.2011.4. [DOI] [PubMed] [Google Scholar]
- Tracey I, Mantyh PW. The cerebral signature for pain perception and its modulation. Neuron. 2007;55:377–91. doi: 10.1016/j.neuron.2007.07.012. [DOI] [PubMed] [Google Scholar]
- Uddin LQ, Kelly AM, Biswal BB, Xavier Castellanos F, Milham MP. Functional connectivity of default mode network components: correlation, anticorrelation, and causality. Hum Brain Mapp. 2009;30:625–37. doi: 10.1002/hbm.20531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Upadhyay J, Maleki N, Potter J, Elman I, Rudrauf D, Knudsen J, et al. Alterations in brain structure and functional connectivity in prescription opioid-dependent patients. Brain. 2010;133:2098–114. doi: 10.1093/brain/awq138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Usui C, Doi N, Nishioka M, Komatsu H, Yamamoto R, Ohkubo T, et al. Electroconvulsive therapy improves severe pain associated with fibromyalgia. Pain. 2006;121:276–80. doi: 10.1016/j.pain.2005.12.025. [DOI] [PubMed] [Google Scholar]
- Vaillancourt PD, Langevin HM. Painful peripheral neuropathies. Med Clin North Am. 1999;83:627–42. doi: 10.1016/s0025-7125(05)70127-9. [DOI] [PubMed] [Google Scholar]
- Valeyrie-Allanore L, Ismaili N, Bastuji-Garin S, Zeller J, Wechsler J, Revuz J, et al. Symptoms associated with malignancy of peripheral nerve sheath tumours: a retrospective study of 69 patients with neurofibromatosis 1. Br J Dermatol. 2005;153:79–82. doi: 10.1111/j.1365-2133.2005.06558.x. [DOI] [PubMed] [Google Scholar]
- van Doorn PA, Ruts L, Jacobs BC. Clinical features, pathogenesis, and treatment of Guillain-Barre syndrome. Lancet Neurol. 2008;7:939250. doi: 10.1016/S1474-4422(08)70215-1. [DOI] [PubMed] [Google Scholar]
- Vanderheyden JE, Gonce M, Bourgeois P, Cras P, De Nayer AR, Flamez A, et al. Epidemiology of major depression in Belgian parkinsonian patients. Acta Neurol Belg. 2010;110:148–56. [PubMed] [Google Scholar]
- Vargas BB. Posttraumatic headache in combat soldiers and civilians: what factors influence the expression of tension-type versus migraine headache? Curr Pain Headache Rep. 2009;13:470–3. doi: 10.1007/s11916-009-0082-1. [DOI] [PubMed] [Google Scholar]
- Veldhuijzen DS, Greenspan JD, Kim JH, Coghill RC, Treede RD, Ohara S, et al. Imaging central pain syndromes. Curr Pain Headache Rep. 2007;11:183–9. doi: 10.1007/s11916-007-0189-1. [DOI] [PubMed] [Google Scholar]
- Verhaak PF, Kerssens JJ, Dekker J, Sorbi MJ, Bensing JM. Prevalence of chronic benign pain disorder among adults: a review of the literature. Pain. 1998;77:231–9. doi: 10.1016/S0304-3959(98)00117-1. [DOI] [PubMed] [Google Scholar]
- Verma A. Epidemiology and clinical features of HIV-1 associated neuropathies. J Peripher Nerv Syst. 2001;6:8–13. doi: 10.1046/j.1529-8027.2001.006001008.x. [DOI] [PubMed] [Google Scholar]
- Vestergaard K, Nielsen J, Andersen G, Ingeman-Nielsen M, Arendt-Nielsen L, Jensen TS. Sensory abnormalities in consecutive, unselected patients with central post-stroke pain. Pain. 1995;61:177–86. doi: 10.1016/0304-3959(94)00140-A. [DOI] [PubMed] [Google Scholar]
- Victor TW, Jensen MP, Gammaitoni AR, Gould EM, White RE, Galer BS. The dimensions of pain quality: factor analysis of the Pain Quality Assessment Scale. Clin J Pain. 2008;24:550–5. doi: 10.1097/AJP.0b013e31816b1058. [DOI] [PubMed] [Google Scholar]
- Walker WC. Pain pathoetiology after TBI: neural and nonneural mechanisms. J Head Trauma Rehabil. 2004;19:72–81. doi: 10.1097/00001199-200401000-00007. [DOI] [PubMed] [Google Scholar]
- Wang Y, Nicol GD, Clapp DW, Hingtgen CM. Sensory neurons from Nf1 haploinsufficient mice exhibit increased excitability. J Neurophysiol. 2005;94:3670–6. doi: 10.1152/jn.00489.2005. [DOI] [PubMed] [Google Scholar]
- Wasan AD, Artin K, Clark MR. A case-matching study of the analgesic properties of electroconvulsive therapy. Pain Med. 2004;5:50–8. doi: 10.1111/j.1526-4637.2004.04006.x. [DOI] [PubMed] [Google Scholar]
- Weigel R, Krauss JK. Center median-parafascicular complex and pain control. Review from a neurosurgical perspective. Stereotact Funct Neurosurg. 2004;82:115–26. doi: 10.1159/000079843. [DOI] [PubMed] [Google Scholar]
- Weiss T, Miltner WH, Adler T, Bruckner L, Taub E. Decrease in phantom limb pain associated with prosthesis-induced increased use of an amputation stump in humans. Neurosci Lett. 1999;272:13124. doi: 10.1016/s0304-3940(99)00595-9. [DOI] [PubMed] [Google Scholar]
- Wessel K, Vieregge P, Kessler C, Kompf D. Thalamic stroke: correlation of clinical symptoms, somatosensory evoked potentials, and CT findings. Acta Neurol Scand. 1994;90:167–73. doi: 10.1111/j.1600-0404.1994.tb02700.x. [DOI] [PubMed] [Google Scholar]
- Wijesekera LC, Leigh PN. Amyotrophic lateral sclerosis. Orphanet J Rare Dis. 2009;4:3. doi: 10.1186/1750-1172-4-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilkinson HA, Davidson KM, Davidson RI. Bilateral anterior cingulotomy for chronic noncancer pain. Neurosurgery. 1999;45:1129–34. doi: 10.1097/00006123-199911000-00023. discussion 34–6. [DOI] [PubMed] [Google Scholar]
- Williams ZM, Bush G, Rauch SL, Cosgrove GR, Eskandar EN. Human anterior cingulate neurons and the integration of monetary reward with motor responses. Nat Neurosci. 2004;7:1370–5. doi: 10.1038/nn1354. [DOI] [PubMed] [Google Scholar]
- Wolters E. Non-motor extranigral signs and symptoms in Parkinson's disease. Parkinsonism Relat Disord. 2009;15(Suppl 3):S6–12. doi: 10.1016/S1353-8020(09)70770-9. [DOI] [PubMed] [Google Scholar]
- Woolf CJ. Central sensitization: implications for the diagnosis and treatment of pain. Pain. 2011;152:S2–15. doi: 10.1016/j.pain.2010.09.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu EQ, Borton J, Said G, Le TK, Monz B, Rosilio M, et al. Estimated prevalence of peripheral neuropathy and associated pain in adults with diabetes in France. Curr Med Res Opin. 2007;23:2035–42. doi: 10.1185/030079907X210516. [DOI] [PubMed] [Google Scholar]
- Zarate CA, Jr, Singh JB, Carlson PJ, Brutsche NE, Ameli R, Luckenbaugh DA, et al. A randomized trial of an N-methyl-D-aspartate antagonist in treatment-resistant major depression. Arch Gen Psychiatry. 2006;63:856–64. doi: 10.1001/archpsyc.63.8.856. [DOI] [PubMed] [Google Scholar]
- Zeitzer JM, Friedman L, O'Hara R. Insomnia in the context of traumatic brain injury. J Rehabil Res Dev. 2009;46:827–36. doi: 10.1682/jrrd.2008.08.0099. [DOI] [PubMed] [Google Scholar]
- Zhang X, Levy D, Kainz V, Noseda R, Jakubowski M, Burstein R. Activation of central trigeminovascular neurons by cortical spreading depression. Ann Neurol. 2011;69:855–65. doi: 10.1002/ana.22329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zubieta JK, Stohler CS. Neurobiological mechanisms of placebo responses. Ann N Y Acad Sci. 2009;1156:198–210. doi: 10.1111/j.1749-6632.2009.04424.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zubieta JK, Smith YR, Bueller JA, Xu Y, Kilbourn MR, Jewett DM, et al. Regional mu opioid receptor regulation of sensory and affective dimensions of pain. Science. 2001;293:311–5. doi: 10.1126/science.1060952. [DOI] [PubMed] [Google Scholar]
- Zwakhalen SM, van der Steen JT, Najim MD. Which score most likely represents pain on the observational PAINAD pain scale for patients with Dementia? J Am Med Dir Assoc. 2011 doi: 10.1016/j.jamda.2011.04.002. Advance Access published on June 3, 2011, doi:10.1016/j.jamda.2011.04.002. [DOI] [PubMed] [Google Scholar]