Abstract
Following cocaine self-administration and extinction training, activity in the infralimbic cortex (IL) suppresses cocaine-seeking behavior. IL inactivation induces cocaine-seeking, whereas activation suppresses cocaine-reinstated drug-seeking. We asked how the suppression of cocaine-seeking induced by IL activation integrates with the circuitry promoting reinstated cocaine-seeking. Following cocaine self-administration and extinction training, rats underwent cue-induced reinstatement. In order to activate IL projections, microinjections of PEPA, a positive allosteric modulator of AMPA receptors, were made into the IL in combination with microinjections into a variety of nuclei known to regulate cocaine-seeking. Intra-IL PEPA administration suppressed cue-induced reinstatement without affecting locomotor activity. The suppression of cocaine-seeking was reversed by activating dopamine neurons in the ventral tegmental area with microinjections of the μ-opioid receptor agonist DAMGO and partially reversed by dopamine microinjections into the prelimbic cortex or basolateral amygdala. Previous evidence suggests that the nucleus accumbens shell both promotes and suppresses cocaine-seeking. The suppression of cue-induced cocaine seeking by PEPA in the IL was reversed by intra-shell microinjections of either dopamine or the AMPA receptor antagonist CNQX, suggesting that the accumbens shell bidirectionally regulates cocaine-seeking depending on whether dopamine input is mimicked or glutamate input is inhibited. Together, these findings indicate that the IL acts “upstream” from structures promoting cocaine-seeking, including from the mesolimbic dopamine projections to the prelimbic cortex and basolateral amygdala and that the accumbens shell may be a crucial point of integration between the circuits that promote (ventral tegmental area) and inhibit (IL) reinstated cocaine-seeking.
Keywords: basolateral amygdala, ventral tegmental area, prelimbic cortex, reinstatement, rat
Inactivation studies reveal that reinstatement of cocaine-seeking requires activity in a variety of structures including the ventral tegmental area (VTA), the basolateral amygdala (BLA), the dorsal portion of the medial prefrontal cortex (prelimbic cortex, PL), and the nucleus accumbens (NA) core (McFarland & Kalivas, 2001; McLaughlin & See, 2003; Di Ciano & Everitt, 2004). Reinstated drug-seeking is a widely employed model of drug relapse (Shaham et al., 2003), and typically involves drug self-administration followed by extinction training, after which drug-seeking is reinstated. As extinction involves new learning rather than erasure of the original memory (Bouton, 2002), extinction learning and its expression involves a neural circuit that suppresses drug-seeking. Akin to findings with the extinction of fear conditioning (Quirk et al., 2006), recent work identified the ventral portion of the medial prefrontal cortex (infralimbic cortex, IL), as well as the IL’s efferent target, the NAshell, as critically involved in the suppression of reinstated cocaine-seeking (Peters et al., 2008). Inactivation of the IL or NAshell immediately prior to an extinction session induces cocaine-seeking without any other reinstatement trigger. Moreover, recent findings suggest that the IL is involved in the consolidation of memories for the extinction of cocaine-seeking behavior, confirming a role for this structure in the extinction of cocaine-seeking (LaLumiere et al., 2010).
Cocaine-seeking induced by IL inactivation is reversed by concurrent inactivation of the PL or BLA, suggesting that during the expression of extinction, activity in the IL actively competes with the circuitry underlying cocaine-seeking (Peters et al., 2008). In support, intra-IL microinjections of AMPA suppress cocaine-reinstated drug-seeking, though it is not known whether such activation suppresses cue-induced reinstatement. Although the NAshell is also involved in the suppression of cocaine-seeking behavior (Di Ciano et al., 2008; Peters et al., 2008), other work suggests that the NAshell promotes cocaine-seeking behavior. NAshell blockade of either D1 or D2 receptors or AMPA receptors prevents cocaine-induced reinstatement (Anderson et al., 2003; Anderson et al., 2006; Famous et al., 2008). One possible explanation for the discrepant results is that the NAshell is a site of competition between the IL and dopaminergic afferents in regulating cocaine-seeking. Thus, depending on the activated inputs, the NAshell can bidirectionally determine whether rats engage in cocaine-seeking. Therefore, the present experiments examined whether IL activation suppresses cue-induced cocaine-seeking and whether this could be reversed by increased dopaminergic tone in mesolimbic dopamine terminal fields important in reinstated cocaine-seeking. Additionally, we evaluated whether the NAshell could bidirectionally regulate cocaine-seeking.
Materials and Methods
Subjects
Male Sprague-Dawley rats (approximately 300 grams at the time of surgery, Charles River, n = 135) were used in this study. They were individually housed, maintained in a temperature-controlled environment (22 °C) on a 12-h light/dark cycle (lights on at 07:00 h) with food and water ad libitum, and given 6–7 days to acclimatize to the vivarium before undergoing surgery. Behavioral procedures began 5–6 days after surgery. All methods used were in compliance with NIH guidelines for care of laboratory animals and were approved by the Medical University of South Carolina Institutional Animal Care and Use Committee or the University of Iowa Institutional Animal Care and Use Committee.
Surgery
The rats were anesthetized with ketamine HCl (87.5 mg/kg, i.m.) and xylazine (5 mg/kg, i.m.). Ketorolac (3 mg/kg, i.p.) was administered prior to surgery to provide analgesia and again 1 d after surgery. For catheter implantation, a 13 cm piece of Silastic tubing (0.51 mm inner diameter, 0.94 mm outer diameter; Dow Corning) with a silicone ball placed 3 cm from the end was threaded under the skin from the back to the ventral side of the rat. The catheter was inserted into the jugular vein and secured with sutures. The other end was externalized through a small hole in the skin between the shoulder blades. The externalized end was connected to a 22 gauge guide cannula (22 ga, C313G; Plastics One, Inc., Wallingford, CT) that was secured in the middle of a harness (Strategic Applications, Inc., USA) that looped around the forelegs of the rat and thus remained securely attached to the rat. Rats were given daily infusions of cefazolin and heparin (0.1 ml of 100U/ml) for one week after surgeries and then infusions of heparin alone throughout the remainder of the self-administration.
The rats were then placed in a small animal stereotaxic instrument (Kopf Instruments, Tujunga, CA, USA). Four surgical screws were implanted into the skull as anchors, and guide cannulae were implanted and secured with dental cement. The nose bar was maintained at −3.5 mm relative to the interaural line. All AP and ML coordinates were calculated from Bregma, whereas DV coordinates were calculated from skull surface. The surgical coordinates were as follows: PL/IL (double-barreled cannulae), AP: +3.1 mm, DV: −2.6 mm; BLA, AP: −2.8 mm, ML: +/−5.0 mm, DV: −6.5 mm; VTA, AP: −5.2 mm, ML (10° angle): +/−2.2 mm, DV: −7.1 mm; NAshell, AP: +1.7 mm, ML (17° angle): +/−3.0 mm, DV: −5.8 mm. After the surgery, the rats were retained in an incubation chamber until they recovered from the anesthesia. Obdurators were placed in all cannulae and maintained through the reinstatement testing. The rats were then returned to their home cages and checked on the days following surgery to ensure that their wounds healed.
Self-administration, extinction, and reinstatement procedures
All self-administration experiments occurred in standard operant chambers (Med Associates, Fairfield, VT) with two retractable levers, a house light, and a cue light and tone-generator (2900 Hz). Prior to drug self-administration, all rats were food-deprived for 24 h and then underwent a single 15-h food-training session in which the rats were trained to press the active lever for a single food pellet (45 mg, Noyes, Lancaster, NH) on a fixed-ratio 1 (FR1) schedule. Following the food training, the rats were given a limited quantity of food (~ 20 g) immediately after every self-administration session, and this food restriction was maintained through extinction and reinstatement testing. One day after the food training, the rats began cocaine self-administration and each session lasted 2 h or until the rats had taken a maximum of 200 infusions. The self-administration program was an FR1 schedule with a 20 s timeout to prevent overdose. Each active lever press produced a 0.05 ml infusion of 200μg of cocaine (dissolved in 0.9% sterile saline; cocaine-HCl kindly provided by NIDA). Concurrent with the drug infusion, a cue tone (2900 hz) and cue light immediately above the active lever turned on for 5 s. Rats underwent self-administration 6 d/week, excluding Sundays, for at least 2 weeks (i.e. 12 days). In order to move into extinction, rats were required to take at least 10 days of at least 10 infusions of cocaine, including the last 3 days of self-administration, as well as discrimination between the active and inactive lever. If rats had not met these criteria by 4 weeks (i.e. 24 days), they were excluded from the study.
After self-administration, rats began extinction training. Active lever presses produced no drug infusion or light/tone cues. For all experiments, rats underwent at least 10 d of extinction and, in order to undergo reinstatement, rats had to have 25 or fewer active lever presses for at least 2 consecutive days immediately prior to the reinstatement session. The 2 days of extinction prior to reinstatement testing served as the extinction baseline. Rats were excluded if they did not reach extinction criterion within 40 days. For cue-induced reinstatement, the session program was identical to the self-administration session program (i.e. cue tone and light turned on with an active lever press). In one experiment, rats also underwent cocaine-induce reinstatement, in which they received a single i.p. injection of cocaine (10 mg/kg) immediately prior to the reinstatement session. Rats did not receive intravenous drug infusions during either reinstatement. Rats’ lever-pressing was re-extinguished between each reinstatement session with the same criteria described above and a minimum of 3 days of extinction between reinstatements.
Microinjection procedures
Microinjectors (33 ga) were connected to PE20 tubing, which was attached to 10-μl Hamilton syringes controlled by an infusion pump. For the BLA, VTA, and NAshell, the microinjectors extended 2 mm beyond the end of the cannula and into the structure of interest. For the PL and IL, the microinjectors extended 1 mm and 3 mm, respectively, beyond the end of the cannula. Rats were not anesthetized during microinjections. All microinjections (0.3 μl) occurred over 60 s, and the injectors were left in place for an additional 60 s to allow for diffusion from the site of the injection. Following the microinjection, rats then underwent their appropriate reinstatement testing. The following drugs were used in the microinjections: the positive modulator of AMPA receptors 2-[2,6-Difluoro-4-[[2-[(phenylsulfonyl)amino]ethyl]thio]phenoxy]acetamide (PEPA) (0.075 nmol/side), CNQX (1 nmol/side), dopamine (25 nmol/side for PL, 75 nmol/side for BLA, 100 nmol /side for NAshell), and [D-Ala2, NMe-Phe4, Gly-ol5]-enkephalin (DAMGO, 0.1 nmol/side). All drugs were dissolved in artificial cerebrospinal fluid (aCSF) except PEPA, which was dissolved initially in 50% DMSO/50% aCSF and was then diluted to 10% DMSO / 90% aCSF immediately prior to use. Doses of all four drugs were chosen based on prior work (Hooks & Kalivas, 1994; McFarland & Kalivas, 2001; LaLumiere et al., 2005; Zushida et al., 2007; LaLumiere & Kalivas, 2008).
Experiments
In all experiments, rats underwent two cue-induced reinstatements in a counterbalanced manner with respect to microinjection. In the first experiments, rats received microinjections of PEPA or vehicle into the IL immediately prior to the cue-induced reinstatement session. In all subsequent experiments, rats received PEPA/vehicle into the IL as well as microinjections into an additional structure. In the second and third experiments, rats received dopamine into the PL or BLA, respectively. In the fourth experiment, rats received the μ-opioid receptor agonist DAMGO into the VTA. In the fifth and sixth experiments, rats received dopamine or CNQX into the NAshell. In addition, a subset of rats (n = 11) was used in the locomotor activity test to determine whether PEPA administration into the IL affects locomotor activity.
Locomotor activity
Following completion of the reinstatement studies, a subset of rats underwent locomotor activity testing in order to determine whether intra-IL microinjections of PEPA affect locomotor activity. The behavioral chambers contained 16 photobeams (AccuScan Instruments). Rats were given microinjections of PEPA or its vehicle into the IL immediately prior to being placed into a chamber for a 2 h locomotor activity test.
Histology and statistics
Rats were overdosed with sodium pentobarbital (100 mg/kg, i.p.) and intracardially perfused with 0.9% saline. The brains were removed and stored in 10% formalin for at least 24 h. Coronal sections (100μm thick) were made using a vibratome. Sections were mounted on gel-coated slides and then stained with cresyl violet. Sites of injector needles were verified with a light microscope, according to the atlas of Paxinos and Watson (2005). Lever pressing data were analyzed using one-way analyses of variance (ANOVA). For the 1st experiment, Bonferroni post-hoc tests were conducted, while the remaining experiments used Bonferroni adjustments to the overall alpha level of 0.05 for planned comparisons between extinction baseline and reinstatement and between the reinstatements of the group receiving PEPA into the IL and vehicle into the other structure and the group receiving PEPA into the IL and a drug into the other structure. The locomotor activity data were analyzed using a two-way repeated-measures ANOVA.
Results
A total of 89 rats were included in the final data, while another 46 were excluded due to failure to acquire self-administration (17 rats), failure to extinguish lever pressing (3 rats), incorrect cannula placements (15 rats), higher inactive lever pressing than active lever pressing at the end of self-administration (2 rats), death prior to the end of the experiment (7 rats) and clogged cannulas (2 rats). Figure 1A shows active and inactive lever presses during self-administration from all animals included in the final data. Within each experiment there was no significant difference between microinjection treatment groups in average active lever pressing during the last 3 days of self-administration (data not shown).
Figure 1.
Suppression of cue-induced cocaine-seeking by intra-IL microinjections of PEPA. A, Active and inactive lever presses during last 12 days of cocaine self-administration. B, Active lever presses (mean +/− SEM) during extinction and cue-induce reinstatement. Intra-IL microinjections of PEPA immediately prior to a cue-induced reinstatement session suppressed cocaine-seeking behavior. C, Horizontal activity counts (mean +/− SEM) measured in an open-field test. Intra-IL microinjections of PEPA immediately prior to the open-field test did not affect the rats’ locomotor activity. *, p < 0.0001, compared to extinction baseline. +, p < 0.05 compared to extinction baseline and to vehicle group.
PEPA microinjections into the IL suppressed cue-induced cocaine-seeking
Figure 1B shows that intra-IL microinjections of PEPA immediately prior to a cue-induced reinstatement session significantly reduced active lever-pressing compared with that found in vehicle-control rats. A one-way ANOVA identified a significant main effect (F(2,23) = 15.87, p < 0.001). Post-hoc tests revealed that rats receiving vehicle had significantly more active lever presses during the cue-induced reinstatement compared to the extinction baseline (p < 0.0001). Those rats receiving PEPA had significantly more active lever presses compared to the extinction baseline but significantly fewer active lever presses compared to those of the vehicle-treated rats (p < 0.05). Thus, intra-IL microinjections of PEPA reduced, but did not completely abolish, cue-induced cocaine-seeking. There were no significant differences in active lever presses between rats receiving PEPA as the first or second microinjection (n = 4 for each, mean +/− SEM: first 23.25 +/− 6.61 and second 32.25 +/−1.97). Similarly, when data were pooled for all rats receiving PEPA alone across all experiments, there were no significant differences in active lever presses between rats receiving PEPA as the first or second microinjection (n = 19 for each; mean +/−SEM: first 23.84 +/− 4.60 and second 16.37 +/− 2.9). For rats shown in Figure 1B, there were no significant differences among the groups for inactive lever presses during reinstatement (data not shown). PEPA microinjections into the IL alone before being placed into an activity chamber did not affect the rats’ horizontal activity compared to vehicle-control rats (Figure 1C). A two-way repeated-measures ANOVA revealed there was a significant effect of time (F(11,99) = 26.82, p < 0.001) but no significant effect of drug (F(1,99) = 0.2836, p > 0.05) or interaction between the drug and time (F(11,99) = 0.3507, p > 0.05).
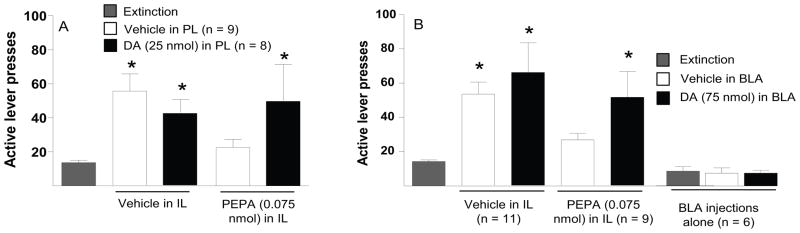
We then examined whether the suppression of cocaine-seeking was reversed by microinjections of dopamine into the PL (Figure 2A) or into the BLA (Figure 2B). For the PL experiment, a one-way ANOVA revealed a significant main effect (F(4,50) = 4.478, p < 0.01). Planned comparisons found that all groups significantly reinstated compared to their extinction baseline except those rats receiving PEPA into the IL and vehicle into the PL (p < 0.05). However, active lever pressing for rats receiving PEPA into the IL did not significantly differ from those receiving PEPA into the IL and dopamine into the PL. For the BLA experiment shown in figure 2B, a one-way ANOVA revealed a significant main effect (F(4,56) = 6.295, p < 0.001). Planned comparisons found that all groups significantly reinstated compared to their extinction baseline except those rats receiving PEPA into the IL and vehicle into the BLA (p < 0.05). As with the PL, active lever pressing for rats receiving PEPA into the IL did not significantly differ from those receiving PEPA into the IL and dopamine into the BLA. While it was previously shown that dopamine alone into the PL reinstates cocaine-seeking (McFarland et al., 2001), the effect of dopamine in the BLA is not known, although microinjection of a D1 antagonist into the BLA has been shown to inhibit cue-induced reinstatement (See et al., 2001). Figure 2B shows that dopamine given alone into the BLA immediately before an extinction session did not significantly reinstate cocaine seeking. Inactive lever pressing during the reinstatement trials did not differ between treatment groups (data not shown). These findings suggest that the IL’s ability to suppress cue-induced cocaine-seeking is reversed by mimicking activation of the mesolimbic dopaminergic afferents to the BLA and PL.
Figure 2.
Partial reversal of PEPA-induced suppression of cue-induced cocaine-seeking by dopaminergic activation of the PL or BLA. A, Active lever presses (mean +/− SEM) during extinction and cue-induced reinstatement for the IL-PL experiments. Intra-IL microinjections of PEPA suppressed cue-induced reinstatement, whereas rats receiving PEPA into the IL and dopamine into the PL showed significant reinstatement. Rats were counterbalanced across IL microinjections. B, Active lever presses (mean +/− SEM) during extinction and cue-induced reinstatement for the IL-BLA experiments. Intra-IL microinjections of PEPA suppressed cue-induced reinstatement, whereas rats receiving PEPA into the IL and dopamine into the PL showed significant reinstatement. For the IL-BLA experiment, intra-BLA microinjections of dopamine alone immediately prior to an extinction session did not significantly affect cocaine-seeking behavior. Rats were counterbalanced across BLA micrionjections. *, p < 0.05 compared to extinction baseline.
Therefore, in the next experiment illustrated in Figure 3, we determined whether broad activation of the mesocorticolimbic dopamine projection by VTA dopamine neurons by microinjecting the μ-opioid receptor agonist DAMGO into the VTA could reverse the suppression of cocaine-seeking (Kalivas, 1993). A one-way ANOVA revealed a significant main effect (F(4,32) = 4.562, p< 0.01). Planned comparisons revealed significant differences in active lever pressing between the extinction baseline and all groups except the group receiving PEPA into the IL and vehicle into the VTA (p < 0.05). Active lever pressing in rats receiving PEPA was significantly lower than that found in rats receiving PEPA into the IL and DAMGO into the VTA (p < 0.05), indicating that DAMGO microinjections into the VTA reversed the suppression induced by PEPA microinjections into the IL. When microinjected alone into the VTA to a subset of rats (n= 5) immediately prior to an extinction session with no cues, this dose of DAMGO did not significantly increase active lever presses. There were no significant differences in inactive lever presses during reinstatement (data not shown).
Figure 3.
Reversal of PEPA-induced suppression of cue-induced cocaine-seeking by VTA disinhibition, as shown by active lever presses (mean +/− SEM) during extinction and cue-induced reinstatement. Intra-IL microinjections of PEPA suppressed cue-induced reinstatement, an effect that was reversed by intra-VTA microinjections of the μ-opioid receptor agonist DAMGO. Intra-VTA microinjections of DAMGO did not significantly induce reinstatement when given alone prior to an extinction session. Rats were counterbalanced across IL microinjections. *, p < 0.05 compared to extinction baseline. +, p < 0.05 compared to PEPA-DAMGO group.
The NAshell regulates cue-induced cocaine-seeking
The NAshell receives glutamatergic input from the IL and dopaminergic input from the VTA (Fallon & Moore, 1978; Sesack et al., 1989), and previous evidence suggests that the NAshell can bidirectionally regulate reward-seeking (Schmidt et al., 2006; Peters et al., 2009). We therefore investigated whether stimulating dopamine receptors or blocking AMPA in the NAshell might differentially affect the capacity of PEPA activation of glutamatergic afferents to inhibit cue-reinstated cocaine seeking. In the first experiment, intra-NAshell microinjections of dopamine reversed the suppression of cue-induced cocaine-seeking by PEPA (Figure 4A). A one-way ANOVA revealed a significant main effect (F(4,41) = 5.666, p < 0.01). Planned comparisons revealed significant differences in active lever presses between the extinction baseline and all groups except those rats receiving PEPA into the IL and vehicle into the NAshell. Active lever pressing in rats receiving PEPA was significantly lower than that found in rats receiving PEPA into the IL and dopamine into the NAshell (p < 0.05). A one-way ANOVA also revealed a significant main effect for dopamine microinjected alone into the NAshell immediately prior to an extinction session (F(2,37) = 8.833, p < 0.001). Planned comparisons revealed that those rats receiving dopamine into the NAshell had significantly higher active lever presses compared to their extinction baseline (p < 0.05). Vehicle given alone into the NAshell had no effect. Figure 4B shows the inactive lever presses for the experiment. A one-way ANOVA found a significant effect for the inactive lever presses during cue-induced reinstatement (F(4,41) = 3.215, p < 0.05). Planned comparisons revealed that those rats receiving dopamine into the NAshell and vehicle into the IL had significantly higher inactive lever presses compared to their extinction baseline (p < 0.05), while inactive lever presses for those receiving dopamine into the NAshell and PEPA into the IL showed a trend toward an increase compared to the extinction baseline (p < 0.08). For microinjections given into the NAshell alone prior to an extinction session, a one-way ANOVA revealed a significant effect for inactive lever presses (F(2,37) = 5.419, p < 0.01), but planned comparisons did not reveal any significant differences between the inactive lever presses of the extinction baseline and those of either microinjection group. Because dopamine microinjections into the NAshell appeared to increase active and inactive lever presses, we created a difference score for both active and inactive lever presses, in which we subtracted the lever presses for rats receiving PEPA into the IL and vehicle into the shell from the lever presses for rats receiving PEPA into the IL and dopamine into the shell. These difference scores showed no significant difference between the increase in active lever presses (mean +/− SEM: 58.7 +/− 29.83) compared with the increase in inactive lever presses (36.5 +/− 21.31).
Figure 4.
Regulation of cue-induce cocaine-seeking behavior by the NAshell. A and B, Active and inactive lever presses (mean +/− SEM), respectively, during extinction and cue-induced reinstatement for rats receiving dopamine microinjections into the NAshell. Intra-IL microinjections of PEPA suppressed cue-induced reinstatement, an effect that was reversed by dopamine microinjections into the NAshell. Intra-NAshell microinjections of dopamine alone prior to an extinction session induced a significant increase in active lever presses. Rats were counterbalanced across NAshell microinjections. C, Active lever presses (mean +/− SEM) during extinction and cue- and cocaine prime-induced reinstatement for rats receiving intra-NAshell microinjections of the AMPA receptor antagonist CNQX. Intra-NAshell microinjections of PEPA suppressed cue-induced reinstatement, an effect that was reversed by intra-NAshell microinjections of CNQX. Rats then underwent a cocaine prime-induced reinstatement session in which CNQX or vehicle was administered into the NAshell prior to the session. CNQX administration did not prevent the cocaine prime-induced reinstatement. Rats were counterbalanced across NAshell microinjections. *, p < 0.05 compared to extinction baseline. #, p < 0.08 compared to extinction baseline. +, p < 0.05 compared to PEPA-dopamine or PEPA-CNQX group.
Figure 4C shows that intra-NAshell microinjections of the AMPA receptor antagonist CNQX reversed the suppression of cue-induced cocaine-seeking. A one-way ANOVA revealed a significant main effect (F(4,59) = 3.576, p < 0.05). Planned comparisons revealed significant differences in active lever presses between the extinction baseline and all groups except those rats receiving PEPA into the IL and vehicle into the NAshell (p < 0.05). Active lever pressing in rats receiving PEPA was not significantly different than that found in rats receiving PEPA into the IL and CNQX into the NAshell, but as the data in the PEPA alone group were non-normal in their distribution, a non-parametric analysis (Mann-Whitney U) was employed. A Mann-Whitney U Test revealed a significant difference between the two groups (p < 0.05). Because previous work indicated that AMPA receptor blockade in the NAshell prevents cocaine-prime induced reinstatement (Famous et al., 2008), the rats then underwent an additional experiment in which an injection of cocaine (10 mg/kg, i.p.) was administered immediately prior to the extinction session. A one-way ANOVA revealed a significant main effect of active lever presses (F(2,47) = 3.900, p < 0.05). Planned comparisons revealed that those rats receiving CNQX into the NAshell had significantly higher active lever presses than the extinction baseline (p < 0.05), whereas there was only a trend toward a difference in active lever presses between the extinction baseline and vehicle-treated rats (p < 0.13). The lack of a significant reinstatement in the vehicle-control rats may have been due to the repeated reinstatements the rats had received prior to this. There were no significant differences in inactive lever presses for any of the groups in Figure 4C (data not shown).
Histology
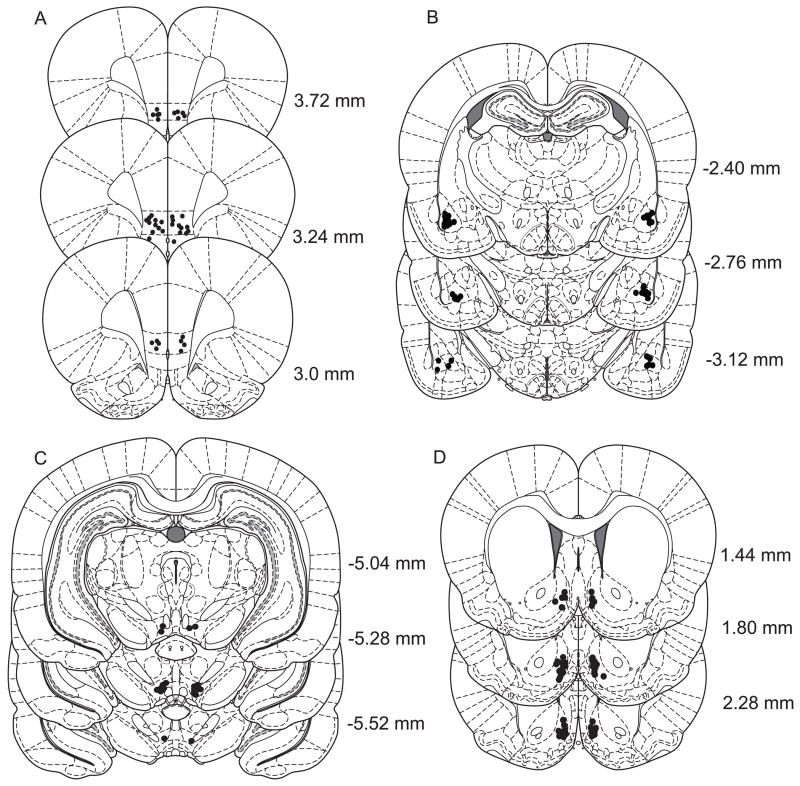
Figure 5 shows the location of the microinjector tips for the animals whose data were used in the study. Figure 5A shows the location of 20 randomly selected microinjector tips in the IL. Data from rats whose microinjectors terminated ventral to the IL in the dorsal peduncular cortex were included (n = 6), as evidence suggests that this structure is also part of the ventromedial prefrontal cortex that regulates extinction learning (Peters et al., 2009). Indeed, those rats with microinjectors in the dorsal peduncular nucleus who received PEPA into the IL and vehicle into the other brain structure (n = 4) showed similar reinstatement levels (21.75 +/−7.49), as compared with the values shown in the figures. Because the same cannulae were used for IL and PL microinjections, there are no separate tip locations for PL injections. Correct PL placement was assumed if the IL placement was correct. Figures 5B, 5C, and 5D show the location of the microinjector tips in the BLA, the VTA, and the NAshell, respectively.
Figure 5.
Diagrams showing the microinjector tracks. Figure 5 shows the location of the microinjector tips for the animals whose data were used in the study. A, Diagram showing the location of 20 randomly selected microinjector tips in the IL. Because the same cannulae were used for IL and PL microinjections, there are no separate tip locations for PL injections. Correct PL placement was assumed if the IL placement was correct. B, Diagram showing the location of the microinjector tips in the BLA. C, Diagram showing the location of the microinjector tips in the VTA. D, Diagram showing the location of the microinjector tips in the NAshell. Figures adapted from Paxinos and Watson (2005) and A/P coordinates (in mm) are given relative to Bregma.
Discussion
The present findings indicate that IL activation, via the AMPA receptor potentiator PEPA, reduces cue-induced cocaine-seeking, consistent with previous cocaine-seeking (Peters et al., 2008; LaLumiere et al., 2010) and fear conditioning studies (Quirk et al., 2000; Morgan et al., 2003) that support a critical role for the IL in the consolidation and expression of extinction in these paradigms. The findings regarding the IL suggest that cue-induce reinstatement “overrides” the normal signaling in the IL that suppresses cocaine-seeking. Although previous experiments used AMPA microinjections to activate the IL and suppress cocaine-prime reinstatement (Peters et al., 2008), PEPA provides a significant advantage as it is an AMPA receptor potentiator that reduces the rate of, or completely abolishes, desensitization of the AMPA receptor (Sekiguchi et al., 1997) and therefore requires the presence of endogenous glutamate to produce effects. Accordingly, the present results suggest that PEPA administration into the IL enhances the activation of glutamatergic receptors and, thereby, increases the activation of IL neurons and their output. This idea is supported by the fact that the NAshell is a major recipient of glutamatergic projections from the IL (Sesack et al., 1989) and blocking AMPA receptors in the NAshell reversed the capacity of PEPA in the IL to inhibit cue-reinstated cocaine seeking.
Previous findings indicate that reinstated cocaine-seeking depends on dopaminergic activation of the BLA and PL, which is believed to facilitate activity in neurons projecting from these nuclei to the NAcore and thereby reinstate cocaine seeking (McFarland & Kalivas, 2001; See et al., 2001; McFarland et al., 2003; Di Ciano & Everitt, 2004). Our findings that indirectly stimulating VTA dopamine neurons with DAMGO or directly stimulating dopamine receptors in the NAshell reversed the inhibition of reinstated cocaine seeking by PEPA in the IL suggests that the IL is “upstream” from activity in the circuit that promotes cocaine-seeking and that downstream activation of the circuit promoting cocaine-seeking is a prepotent regulator of behavior. In the PL and BLA, the effects were more mixed. Although rats receiving PEPA into the IL and dopamine into the PL and BLA showed significant reinstatement, they did not significantly differ from those rats receiving PEPA alone in their active lever presses, suggesting that the reversal of the inhibition was not as strong in these groups. Most likely, this finding reflects the necessary involvement of both the PL and the BLA in cue-induced cocaine-seeking and the fact that both structures independently project to the NAcore. Although not experimentally tested, it is likely that activation of the NAcore AMPA receptors would override the suppression signal, as previous findings suggest that PL and/or BLA projections to the NAcore are the critical pathways mediating reinstatement (McFarland et al., 2003; Di Ciano & Everitt, 2004). The idea that the IL is upstream from these other structures in the circuitry regulating cocaine-seeking is also consistent with the finding that cocaine-seeking induced by IL inactivation is reversed by simultaneous BLA or PL inactivation by microinjected GABA agonists (Peters et al., 2008). That the cocaine-seeking circuit is downstream from the suppression circuit may explain why, after weeks of extinction training, reinstatement is easily triggered by exposure to a variety of stimuli. Although the purpose of the present experiments was to determine whether the cocaine-seeking circuit was downstream from the suppression circuit, it is likely that there are also structures upstream from the IL that signal critical information to the IL to initiate the suppression signal. Future work will address this issue.
In apparent contrast with the present findings, Koya et al (2009) found that IL activation one day after self-administration induced cocaine-seeking whereas IL inactivation after 30 days of withdrawal inhibited cocaine-seeking. However, in both cases in that study, the IL manipulations were performed on the first day of extinction, either 1 day or 30 days after self-administration. Our previous work suggests that prior extinction training is required for the IL to be involved in suppressing cocaine-seeking (Peters et al., 2008). Thus, akin to what has been observed for fear conditioning (see below), the IL may be involved in the learning and expression of the most recent contingencies, and only after extensive extinction training and the associated new learning does the IL begin to suppress cocaine-seeking. It is critical to note that studies using inactivation and activation may not always produce opposite behavioral outcomes, especially as the mechanisms of inactivation or activation may be rather different and alter neural activity in different manners, and this may account for some discrepancies in the literature.
In contrast to studies with cocaine-seeking and fear conditioning, the evidence regarding the role of the IL in heroin- and morphine-seeking behavior has been conflicting. Previous work found that PL or IL inactivation prevents reinstatement of heroin-seeking (Rogers et al., 2008; Bossert et al., 2011), whereas studies with cocaine-seeking have shown that PL, but not IL, inactivation prevents cocaine-seeking (McFarland & Kalivas, 2001). However, other work suggests that, under at least some circumstances, the medial prefrontal cortex in general or the IL specifically is involved in the extinction of opiate-seeking or opiate conditioned place preference (Ovari & Leri, 2008; Van den Oever et al., 2008; Van den Oever et al., 2010; He et al., 2011). While the present data do not directly address the apparently distinct role of the IL in cocaine-seeking and fear conditioning versus heroin-seeking, it is interesting to consider that whereas cocaine is characterized as having aversive qualities in rats, heroin does not (Ettenberg & Geist, 1993; Ettenberg, 2004), and perhaps the aversive qualities of cocaine and fear conditioning contribute to the involvement of the IL in extinction and regulating motivated behavior.
Apparent contradictions also exist in the literature describing the role of the NAshell in cocaine-seeking. In the present study we found that AMPA receptor blockade of the NAshell restored cocaine-seeking that was inhibited by PEPA activation of IL afferents to the NAshell. This is consistent with previous studies showing that pharmacological inactivation of the NAshell induces cocaine-seeking (Peters et al., 2008), alcohol-seeking (Millan et al., 2010), second-order schedules of cocaine-seeking (Di Ciano et al., 2008), and food-seeking (Stratford & Kelley, 1997; Floresco et al., 2008). However, another study found that AMPA receptor blockade in the NAshell prevents reinstatement by a systemic cocaine priming injection (Famous et al., 2008). To determine whether the present data resulted from differences between cue- and cocaine-induced reinstatement, we conducted an additional experiment in which NAshell AMPA receptors were blocked during a cocaine prime-induced reinstatement and found a partial potentiation of the reinstatement, suggesting that AMPA receptors in the NAshell are not involved in promoting cocaine-seeking. It is not clear why the present results do not agree with those of Famous et al (2008), though it should be noted that Famous et al used a larger volume (0.5 μl) for microinjection compared to the current study (0.3 μl), posing the possibility that some of the drug may have diffused to nearby regions of the NAcore, and AMPA receptor blockade in the NAcore prevents cue-induced reinstatement of cocaine-seeking (Backstrom & Hyytia, 2007).
A number of studies find that NAshell blockade of dopamine receptors prevents cocaine prime-induced reinstatement (Anderson et al., 2003; Bachtell et al., 2005; Anderson et al., 2006). In the present study, intra-NAshell microinjections of dopamine reversed the suppression of cocaine-seeking by PEPA administration into the IL. Moreover, dopamine alone induced cocaine-seeking, in agreement with previous studies that dopaminergic input to the NAshell promotes drug-seeking (Schmidt et al., 2006; Anderson et al., 2008). Recent studies indicate that the NAshell plays a crucial role in suppressing inappropriate behaviors that the animal has learned not to engage in, such as responding to a nonrewarded cue or responding in the absence of a reward-related cue (Ambroggi et al., 2011; Ghazizadeh et al., In Press). Because inactive lever pressing or pressing on the active lever after extinction training are similar types of inappropriate behavior, it is not surprising that dopamine microinjections into the NAshell also increased inactive lever pressing. As previous work indicates that NAshell inactivation, via GABAergic agonists, increases inappropriate behaviors, including cocaine-seeking after extinction training (Peters et al., 2008) or responding during the nonrewarded cue (Ambroggi et al., 2011), it appears that NAshell inactivation via GABAergic agonists produces similar effects as dopamine in the NAshell. This conclusion is in agreement with evidence that dopamine receptor blockade in the NAshell prevents the increase in responding to the nonrewarded cue induced by IL inactivation (Ghazizadeh et al., In Press) and that dopamine inhibits reward-related firing of accumbens neurons (Carlezon & Thomas, 2009). If this hypothesis is correct, it would help to resolve the apparent contradiction in the literature, in which findings indicate that NAshell inactivation has no effect on (McFarland & Kalivas, 2001), whereas dopamine receptor blockade prevents, reinstatement of cocaine-seeking (Anderson et al., 2006).
In contrast to the dopamine signaling, IL signaling appears to suppress these inappropriate behaviors, as suggested by the present findings and previous work (Ishikawa et al., 2008; Ghazizadeh et al., In Press). Recent evidence indicates that reinstatement of cocaine conditioned place preference reduces the strength of excitatory inputs to the NAshell, suggesting that a mechanism within the NAshell permits reinstatement triggers to reduce IL inputs that normally inhibit drug-seeking (Rothwell et al., 2011). Indeed, dopamine acts as a modulator of glutamatergic transmission onto medium spiny neurons in the NAshell (Nicola et al., 2000). In countermanding PEPA activation of IL glutamatergic input to the NAshell, dopamine may be acting presynaptically on D1 receptors to inhibit glutamate or postsynaptically via D2 to inhibit the efficacy of glutamate signaling (Nicola et al., 2000). Because dopaminergic inputs to the NAcore produce similar effects as those in the NAshell yet prefrontal glutamatergic inputs to the NAcore induce, rather than inhibit, cocaine-seeking, it is unlikely that this mechanism fully accounts for the differences in the NAshell. As analysis of the NAcore and shell indicates subpopulations of neurons, rather than the entire structure, guide motivated behavior (Taha et al., 2007), understanding the interplay of glutamatergic and dopaminergic inputs in each structure with specific behavior will require future work focused on the distinct neuronal ensembles within the NAshell and core, rather than individual neurons, activated by specific inputs.
The present findings are consistent with human studies on the ventromedial prefrontal cortex (vmPFC or subgenual anterior cingulate cortex, including the medial orbitofrontal cortex), which appears to be functionally homologous to the IL (Phelps et al., 2004; Bechara, 2005; Milad et al., 2009). Activity in the vmPFC correlates with retention of extinction for fear conditioning (Phelps et al., 2004; Milad et al., 2009). Moreover, cue-induced cocaine craving is associated with decreased vmPFC activity (Bonson et al., 2002), and greater vmPFC activity correlates with longer durations of abstinence from cocaine use (Brewer et al., 2008). Other evidence indicates that low impulse control in humans correlates with reduced vmPFC volume (Boes et al., 2009) and that vmPFC-damaged patients make poor choices in the Iowa Gambling Task, showing a preference for high immediate rewards with long-term negative consequences (Bechara, 2005). Thus, the IL in rats or vmPFC in humans is likely involved in learning to suppress inappropriate behaviors, including cocaine-seeking, and is consequently critical for extinction learning and expression. It is tempting to speculate that differences in vmPFC activity in humans may contribute to an inability to suppress cocaine-seeking behavior, rendering the vmPFC a potential pharmacotherapeutic target for addiction treatment. In fact, PEPA appears to activate the medial prefrontal cortex relatively selectively over the BLA or hippocampus due to the increased presence in the medial prefrontal cortex of AMPA receptor subunits and splice variants that preferentially bind PEPA (Zushida et al., 2007). Intriguingly, in mice, systemic PEPA administration, as well as intra-medial prefrontal cortex microinjections of PEPA, enhance the rate of extinction of fear conditioning (Zushida et al., 2007; Yamada et al., 2009; Yamada et al., 2011), suggesting that it may prove to be a fruitful pharmacotherapy for promoting the inhibition of unwanted behaviors through extinction training.
Acknowledgments
This research was supported by NIH grants DA027055 (RTL) and DA015369 (PWK). We thank Rachel Miller, Kate Niehoff, Karen Corson, Ana Weiland, Mary Shaw, Dil Patel, and Giana Ravita for their excellent technical assistance.
Footnotes
The authors report no conflicts of interest.
References
- Ambroggi F, Ghazizadeh A, Nicola SM, Fields HL. Roles of nucleus accumbens core and shell in incentive-cue responding and behavioral inhibition. J Neurosci. 2011;31:6820–6830. doi: 10.1523/JNEUROSCI.6491-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anderson SM, Bari AA, Pierce RC. Administration of the D1-like dopamine receptor antagonist SCH-23390 into the medial nucleus accumbens shell attenuates cocaine priming-induced reinstatement of drug-seeking behavior in rats. Psychopharmacology (Berl) 2003;168:132–138. doi: 10.1007/s00213-002-1298-5. [DOI] [PubMed] [Google Scholar]
- Anderson SM, Famous KR, Sadri-Vakili G, Kumaresan V, Schmidt HD, Bass CE, Terwilliger EF, Cha JH, Pierce RC. CaMKII: a biochemical bridge linking accumbens dopamine and glutamate systems in cocaine seeking. Nat Neurosci. 2008;11:344–353. doi: 10.1038/nn2054. [DOI] [PubMed] [Google Scholar]
- Anderson SM, Schmidt HD, Pierce RC. Administration of the D2 dopamine receptor antagonist sulpiride into the shell, but not the core, of the nucleus accumbens attenuates cocaine priming-induced reinstatement of drug seeking. Neuropsychopharmacology. 2006;31:1452–1461. doi: 10.1038/sj.npp.1300922. [DOI] [PubMed] [Google Scholar]
- Bachtell RK, Whisler K, Karanian D, Self DW. Effects of intra-nucleus accumbens shell administration of dopamine agonists and antagonists on cocaine-taking and cocaine-seeking behaviors in the rat. Psychopharmacology (Berl) 2005;183:41–53. doi: 10.1007/s00213-005-0133-1. [DOI] [PubMed] [Google Scholar]
- Backstrom P, Hyytia P. Involvement of AMPA/kainate, NMDA, and mGlu5 receptors in the nucleus accumbens core in cue-induced reinstatement of cocaine seeking in rats. Psychopharmacology (Berl) 2007;192:571–580. doi: 10.1007/s00213-007-0753-8. [DOI] [PubMed] [Google Scholar]
- Bechara A. Decision making, impulse control and loss of willpower to resist drugs: a neurocognitive perspective. Nat Neurosci. 2005;8:1458–1463. doi: 10.1038/nn1584. [DOI] [PubMed] [Google Scholar]
- Boes AD, Bechara A, Tranel D, Anderson SW, Richman L, Nopoulos P. Right ventromedial prefrontal cortex: a neuroanatomical correlate of impulse control in boys. Soc Cogn Affect Neurosci. 2009;4:1–9. doi: 10.1093/scan/nsn035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bonson KR, Grant SJ, Contoreggi CS, Links JM, Metcalfe J, Weyl HL, Kurian V, Ernst M, London ED. Neural systems and cue-induced cocaine craving. Neuropsychopharmacology. 2002;26:376–386. doi: 10.1016/S0893-133X(01)00371-2. [DOI] [PubMed] [Google Scholar]
- Bossert JM, Stern AL, Theberge FR, Cifani C, Koya E, Hope BT, Shaham Y. Ventral medial prefrontal cortex neuronal ensembles mediate context-induced relapse to heroin. Nat Neurosci. 2011;14:420–422. doi: 10.1038/nn.2758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bouton ME. Context, ambiguity, and unlearning: sources of relapse after behavioral extinction. Biol Psychiatry. 2002;52:976–986. doi: 10.1016/s0006-3223(02)01546-9. [DOI] [PubMed] [Google Scholar]
- Brewer JA, Worhunsky PD, Carroll KM, Rounsaville BJ, Potenza MN. Pretreatment brain activation during stroop task is associated with outcomes in cocaine-dependent patients. Biol Psychiatry. 2008;64:998–1004. doi: 10.1016/j.biopsych.2008.05.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carlezon WA, Jr, Thomas MJ. Biological substrates of reward and aversion: a nucleus accumbens activity hypothesis. Neuropharmacology. 2009;56(Suppl 1):122–132. doi: 10.1016/j.neuropharm.2008.06.075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Di Ciano P, Everitt BJ. Direct interactions between the basolateral amygdala and nucleus accumbens core underlie cocaine-seeking behavior by rats. J Neurosci. 2004;24:7167–7173. doi: 10.1523/JNEUROSCI.1581-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Di Ciano P, Robbins TW, Everitt BJ. Differential effects of nucleus accumbens core, shell, or dorsal striatal inactivations on the persistence, reacquisition, or reinstatement of responding for a drug-paired conditioned reinforcer. Neuropsychopharmacology. 2008;33:1413–1425. doi: 10.1038/sj.npp.1301522. [DOI] [PubMed] [Google Scholar]
- Ettenberg A. Opponent process properties of self-administered cocaine. Neurosci Biobehav Rev. 2004;27:721–728. doi: 10.1016/j.neubiorev.2003.11.009. [DOI] [PubMed] [Google Scholar]
- Ettenberg A, Geist TD. Qualitative and quantitative differences in the operant runway behavior of rats working for cocaine and heroin reinforcement. Pharmacol Biochem Behav. 1993;44:191–198. doi: 10.1016/0091-3057(93)90298-8. [DOI] [PubMed] [Google Scholar]
- Fallon JH, Moore RY. Catecholamine innervation of the basal forebrain. IV. Topography of the dopamine projection to the basal forebrain and neostriatum. J Comp Neurol. 1978;180:545–580. doi: 10.1002/cne.901800310. [DOI] [PubMed] [Google Scholar]
- Famous KR, Kumaresan V, Sadri-Vakili G, Schmidt HD, Mierke DF, Cha JH, Pierce RC. Phosphorylation-dependent trafficking of GluR2-containing AMPA receptors in the nucleus accumbens plays a critical role in the reinstatement of cocaine seeking. J Neurosci. 2008;28:11061–11070. doi: 10.1523/JNEUROSCI.1221-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Floresco SB, McLaughlin RJ, Haluk DM. Opposing roles for the nucleus accumbens core and shell in cue-induced reinstatement of food-seeking behavior. Neuroscience. 2008;154:877–884. doi: 10.1016/j.neuroscience.2008.04.004. [DOI] [PubMed] [Google Scholar]
- Ghazizadeh A, Ambroggi F, Odean N, Fields HL. Prefrontal cortex mediates extinction of responding by a dual neural mechanism in accumbens shell. J Neurosci. doi: 10.1523/JNEUROSCI.3891-11.2012. (In Press) [DOI] [PMC free article] [PubMed] [Google Scholar]
- He YY, Xue YX, Wang JS, Fang Q, Liu JF, Xue LF, Lu L. PKMzeta Maintains Drug Reward and Aversion Memory in the Basolateral Amygdala and Extinction Memory in the Infralimbic Cortex. Neuropsychopharmacology. 2011 doi: 10.1038/npp.2011.63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hooks MS, Kalivas PW. Involvement of dopamine and excitatory amino acid transmission in novelty-induced motor activity. J Pharmacol Exp Ther. 1994;269:976–988. [PubMed] [Google Scholar]
- Ishikawa A, Ambroggi F, Nicola SM, Fields HL. Contributions of the amygdala and medial prefrontal cortex to incentive cue responding. Neuroscience. 2008;155:573–584. doi: 10.1016/j.neuroscience.2008.06.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalivas PW. Neurotransmitter regulation of dopamine neurons in the ventral tegmental area. Brain Res Brain Res Rev. 1993;18:75–113. doi: 10.1016/0165-0173(93)90008-n. [DOI] [PubMed] [Google Scholar]
- Koya E, Uejima JL, Wihbey KA, Bossert JM, Hope BT, Shaham Y. Role of ventral medial prefrontal cortex in incubation of cocaine craving. Neuropharmacology. 2009;56(Suppl 1):177–185. doi: 10.1016/j.neuropharm.2008.04.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LaLumiere RT, Kalivas PW. Glutamate release in the nucleus accumbens core is necessary for heroin seeking. J Neurosci. 2008;28:3170–3177. doi: 10.1523/JNEUROSCI.5129-07.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LaLumiere RT, Nawar EM, McGaugh JL. Modulation of memory consolidation by the basolateral amygdala or nucleus accumbens shell requires concurrent dopamine receptor activation in both brain regions. Learn Mem. 2005;12:296–301. doi: 10.1101/lm.93205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LaLumiere RT, Niehoff KE, Kalivas PW. The infralimbic cortex regulates the consolidation of extinction after cocaine self-administration. Learn Mem. 2010;17:168–175. doi: 10.1101/lm.1576810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McFarland K, Kalivas PW. The circuitry mediating cocaine-induced reinstatement of drug-seeking behavior. J Neurosci. 2001;21:8655–8663. doi: 10.1523/JNEUROSCI.21-21-08655.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McFarland K, Lapish CC, Kalivas PW. Prefrontal glutamate release into the core of the nucleus accumbens mediates cocaine-induced reinstatement of drug-seeking behavior. J Neurosci. 2003;23:3531–3537. doi: 10.1523/JNEUROSCI.23-08-03531.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McLaughlin J, See RE. Selective inactivation of the dorsomedial prefrontal cortex and the basolateral amygdala attenuates conditioned-cued reinstatement of extinguished cocaine-seeking behavior in rats. Psychopharmacology (Berl) 2003;168:57–65. doi: 10.1007/s00213-002-1196-x. [DOI] [PubMed] [Google Scholar]
- Milad MR, Pitman RK, Ellis CB, Gold AL, Shin LM, Lasko NB, Zeidan MA, Handwerger K, Orr SP, Rauch SL. Neurobiological basis of failure to recall extinction memory in posttraumatic stress disorder. Biol Psychiatry. 2009;66:1075–1082. doi: 10.1016/j.biopsych.2009.06.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Millan EZ, Furlong TM, McNally GP. Accumbens shell-hypothalamus interactions mediate extinction of alcohol seeking. J Neurosci. 2010;30:4626–4635. doi: 10.1523/JNEUROSCI.4933-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morgan MA, Schulkin J, LeDoux JE. Ventral medial prefrontal cortex and emotional perseveration: the memory for prior extinction training. Behav Brain Res. 2003;146:121–130. doi: 10.1016/j.bbr.2003.09.021. [DOI] [PubMed] [Google Scholar]
- Nicola SM, Surmeier J, Malenka RC. Dopaminergic modulation of neuronal excitability in the striatum and nucleus accumbens. Annu Rev Neurosci. 2000;23:185–215. doi: 10.1146/annurev.neuro.23.1.185. [DOI] [PubMed] [Google Scholar]
- Ovari J, Leri F. Inactivation of the ventromedial prefrontal cortex mimics re-emergence of heroin seeking caused by heroin reconditioning. Neurosci Lett. 2008;444:52–55. doi: 10.1016/j.neulet.2008.08.015. [DOI] [PubMed] [Google Scholar]
- Paxinos G, Watson C. The Rat Brain in Stereotaxic Coordinates. Elsevier Academic Press; Amsterdam: 2005. [Google Scholar]
- Peters J, Kalivas PW, Quirk GJ. Extinction circuits for fear and addiction overlap in prefrontal cortex. Learn Mem. 2009;16:279–288. doi: 10.1101/lm.1041309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peters J, LaLumiere RT, Kalivas PW. Infralimbic prefrontal cortex is responsible for inhibiting cocaine seeking in extinguished rats. J Neurosci. 2008;28:6046–6053. doi: 10.1523/JNEUROSCI.1045-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Phelps EA, Delgado MR, Nearing KI, LeDoux JE. Extinction learning in humans: role of the amygdala and vmPFC. Neuron. 2004;43:897–905. doi: 10.1016/j.neuron.2004.08.042. [DOI] [PubMed] [Google Scholar]
- Quirk GJ, Garcia R, Gonzalez-Lima F. Prefrontal mechanisms in extinction of conditioned fear. Biol Psychiatry. 2006;60:337–343. doi: 10.1016/j.biopsych.2006.03.010. [DOI] [PubMed] [Google Scholar]
- Quirk GJ, Russo GK, Barron JL, Lebron K. The role of ventromedial prefrontal cortex in the recovery of extinguished fear. J Neurosci. 2000;20:6225–6231. doi: 10.1523/JNEUROSCI.20-16-06225.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rogers JL, Ghee S, See RE. The neural circuitry underlying reinstatement of heroin-seeking behavior in an animal model of relapse. Neuroscience. 2008;151:579–588. doi: 10.1016/j.neuroscience.2007.10.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rothwell PE, Kourrich S, Thomas MJ. Synaptic adaptations in the nucleus accumbens caused by experiences linked to relapse. Biol Psychiatry. 2011;69:1124–1126. doi: 10.1016/j.biopsych.2010.12.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmidt HD, Anderson SM, Pierce RC. Stimulation of D1-like or D2 dopamine receptors in the shell, but not the core, of the nucleus accumbens reinstates cocaine-seeking behaviour in the rat. Eur J Neurosci. 2006;23:219–228. doi: 10.1111/j.1460-9568.2005.04524.x. [DOI] [PubMed] [Google Scholar]
- See RE, Kruzich PJ, Grimm JW. Dopamine, but not glutamate, receptor blockade in the basolateral amygdala attenuates conditioned reward in a rat model of relapse to cocaine-seeking behavior. Psychopharmacology (Berl) 2001;154:301–310. doi: 10.1007/s002130000636. [DOI] [PubMed] [Google Scholar]
- Sekiguchi M, Fleck MW, Mayer ML, Takeo J, Chiba Y, Yamashita S, Wada K. A novel allosteric potentiator of AMPA receptors: 4--2-(phenylsulfonylamino)ethylthio--2,6-difluoro-phenoxyaceta mide. J Neurosci. 1997;17:5760–5771. doi: 10.1523/JNEUROSCI.17-15-05760.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sesack SR, Deutch AY, Roth RH, Bunney BS. Topographical Organization of the Efferent Projections of the Medial Prefrontal Cortex in the Rat - an Anterograde Tract-Tracing Study with Phaseolus-Vulgaris Leucoagglutinin. Journal of Comparative Neurology. 1989;290:213–242. doi: 10.1002/cne.902900205. [DOI] [PubMed] [Google Scholar]
- Shaham Y, Shalev U, Lu L, De Wit H, Stewart J. The reinstatement model of drug relapse: history, methodology and major findings. Psychopharmacology (Berl) 2003;168:3–20. doi: 10.1007/s00213-002-1224-x. [DOI] [PubMed] [Google Scholar]
- Stratford TR, Kelley AE. GABA in the nucleus accumbens shell participates in the central regulation of feeding behavior. J Neurosci. 1997;17:4434–4440. doi: 10.1523/JNEUROSCI.17-11-04434.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taha SA, Nicola SM, Fields HL. Cue-evoked encoding of movement planning and execution in the rat nucleus accumbens. J Physiol. 2007;584:801–818. doi: 10.1113/jphysiol.2007.140236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van den Oever MC, Goriounova NA, Li KW, Van der Schors RC, Binnekade R, Schoffelmeer AN, Mansvelder HD, Smit AB, Spijker S, De Vries TJ. Prefrontal cortex AMPA receptor plasticity is crucial for cue-induced relapse to heroin-seeking. Nat Neurosci. 2008;11:1053–1058. doi: 10.1038/nn.2165. [DOI] [PubMed] [Google Scholar]
- Van den Oever MC, Lubbers BR, Goriounova NA, Li KW, Van der Schors RC, Loos M, Riga D, Wiskerke J, Binnekade R, Stegeman M, Schoffelmeer AN, Mansvelder HD, Smit AB, De Vries TJ, Spijker S. Extracellular matrix plasticity and GABAergic inhibition of prefrontal cortex pyramidal cells facilitates relapse to heroin seeking. Neuropsychopharmacology. 2010;35:2120–2133. doi: 10.1038/npp.2010.90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamada D, Wada K, Sekiguchi M. Facilitating actions of an AMPA receptor potentiator upon extinction of contextually conditioned fear response in stressed mice. Neurosci Lett. 2011;488:242–246. doi: 10.1016/j.neulet.2010.11.038. [DOI] [PubMed] [Google Scholar]
- Yamada D, Zushida K, Wada K, Sekiguchi M. Pharmacological discrimination of extinction and reconsolidation of contextual fear memory by a potentiator of AMPA receptors. Neuropsychopharmacology. 2009;34:2574–2584. doi: 10.1038/npp.2009.86. [DOI] [PubMed] [Google Scholar]
- Zushida K, Sakurai M, Wada K, Sekiguchi M. Facilitation of extinction learning for contextual fear memory by PEPA: a potentiator of AMPA receptors. J Neurosci. 2007;27:158–166. doi: 10.1523/JNEUROSCI.3842-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]