Abstract
Purpose
Early identification of neonatal sepsis is a global issue because of limitations in diagnostic procedures. The objective of this study was to compare the diagnostic accuracy of neutrophil CD64 and C-reactive protein (CRP) as a single test for the early detection of neonatal sepsis.
Methods
A prospective study enrolled newborns with documented sepsis (n=11), clinical sepsis (n=12) and control newborns (n=14). CRP, neutrophil CD64, complete blood counts and blood culture were taken at the time of the suspected sepsis for the documented or clinical group and at the time of venipuncture for laboratory tests in control newborns. Neutrophil CD64 was analyzed by flow cytometry.
Results
CD64 was significantly elevated in the groups with documented or clinical sepsis, whereas CRP was not significantly increased compared with controls. For documented sepsis, CD64 and CRP had a sensitivity of 91% and 9%, a specificity of 83% and 83%, a positive predictive value of 83% and 33% and a negative predictive value of 91% and 50%, respectively, with a cutoff value of 3.0 mg/dL for CD64 and 1.0 mg/dL for CRP. The area under the receiver-operating characteristic curves for CD64 index and CRP were 0.955 and 0.527 (P<0.01), respectively.
Conclusion
These preliminary data show that diagnostic accuracy of CD64 is superior to CRP when measured at the time of suspected sepsis, which implies that CD64 is a more reliable marker for the early identification of neonatal sepsis as a single determination compared with CRP.
Keywords: Neutrophil CD64, C-reactive protein, Neonatal sepsis
Introduction
In spite of extensive research and development in the understanding and treatment of neonatal sepsis, it continues to be the one of the most common causes of neonatal morbidity and mortality1,2). Early identification of neonatal sepsis is difficult because of the nonspecific or minimal clinical presentations. However, the clinical course of neonatal sepsis can be fulminant within hours of onset3), and infection in the newborn period is associated with 10% of neonatal deaths4). Thus, it is extremely important to make an early and accurate identification of neonatal sepsis for prompt antimicrobial therapy and better outcomes.
The current gold standard for confirming diagnosis of neonatal sepsis is blood culture. However, blood culture results are not available for 48 hours after starting the culture, and if blood cultures are drawn after administration of antibiotics, growth of microorganisms can be suppressed5). Hence, a reliable inflammatory marker or set of markers is required for prompt and accurate identification of neonatal sepsis, so that delayed or unnecessary treatment can be avoided.
C-reactive protein (CRP) is a conventional inflammatory marker as a kind of acute phase protein. Concentrations of CRP increase at around 24 hours after onset of infection, peak between 36 and 50 hours and remain elevated throughout infection6). CRP necessarily has a limitation for the early identification of infection within 24 hours of onset; therefore, serial measures are required 24 hours later for accurate detection of infection3). Currently, in most neonatal intensive care units (NICUs) in Korea, CRP is practically used as an inflammatory marker, and a more accurate diagnostic maker is required on clinical base for better care of the newborns.
Recently, attention has been directed to leukocyte cell surface antigens as diagnostic markers of neonatal sepsis7). Neutrophil surface marker, CD64 has high affinity to the Fc portion of immunoglobulin G (IgG) and is expressed and upregulated by bacterial or endotoxin interaction8). Up until now, there have been several reports to show CD64's utility as an inflammatory marker for detection of neonatal infection including pneumonia, septicemia, necrotizing enterocolitis (NEC), meningitis and peritonitis in term or preterm newborns9-12). However, there have never been any reports to suggest that CD64 is a more superior diagnostic marker for neonatal sepsis compared to CRP as a single test regardless of the time of evaluation from onset of suspected sepsis in neonates and also there have never been any investigations to define CD64's utility for neonatal sepsis in Korea.
In this study, the authors investigated whether neutrophil CD64 is a more accurate diagnostic inflammatory marker compared to CRP as a single determination for the early detection of neonatal sepsis.
Materials and methods
1. Subjects
This prospective study included 37 neonates (20 males and 17 females) born at ≥30 weeks of gestation and admitted to the NICU of Kangwon National University Hospital from May to August 2010. Subjects were classified into one of the following three groups:
Documented sepsis (n=11): positive bacterial culture with the presence of two or more clinical symptoms or signs as below.
Clinical sepsis (n=12): negative bacterial culture with the presence of two or more clinical symptoms or signs of infection as below.
Control (n=14): newborns with no symptoms or signs of infection, but who took blood samples drawn because of suspicion of different diseases with normal findings and no illness subsequently detected.
Two or more of the following clinical manifestations were used to determine patients for sepsis evaluations: 1) respiratory compromise: respiratory rate of >60 breaths per minute, or cessation of respiration for ≥20 seconds, occurring at a rate of ≥2 times per hour, or pulse oxymeter readings of ≤85%; 2) cardiovascular compromise: heart rate of <100 beats per minute, pallor or hypotension; 3) metabolic changes: hypothermia (rectal temperature of <36℃), body temperature of >38℃, feeding intolerance (increased gastric residuals of >50% of milk volume in ≥2 feedings within 24 hours), glucose instability (blood glucose level of <45 mg/dL or >125 mg/dL), or metabolic acidosis (pH<7.25); or 4) neurologic changes: lethargy or decreased activity.
2. Methods
1) Blood samples
Samples of peripheral blood or through umbilical catheter (only one episode in the clinical group) for complete blood count (CBC), CRP, CD64 and blood culture were obtained when infection was suspected. Samples from group 3 were taken at the time of venipuncture for laboratory tests and within the first 24 hours of admission.
2) Hematologic criteria
The following hematologic criteria were used as indications for sepsis: 1) absolute neutrophil count (ANC) of <1,800 or >6,500/µL at birth, <7,000 or >12,000/µL at 3 to 24 hours of age, <4,000 or >9,000/µL at 25 to 48 hours of age, and <2,000 or >7,000/µL beyond 48 hours of age13); 2) absolute band count of >1,500/µL; 3) immature/total neutrophil ratio of >0.16; 4) white blood cells (WBC) count of >18,000/µL or <6,000/µL; or 5) platelet count of <150,000/µL14,15).
3) Blood culture and CRP
All blood cultures were collected using standard sterile techniques, and the Bactec Microbial Detection System (Becton Dickinson, Parsipanny, NJ, USA) was used to detect positive blood cultures. CRP was measured by high sensitive-CRP reagent (Synchron Clinical System, Beckman Coulter Inc., Brea, CA, USA) using the Automated Chemistry Analyzer (UniCel DxC 800, Beckman Coulter Inc.).
4) CD64 Index
Neutrophil CD64 expression was measured using the Leuko 64 assay (Leuko64 kit, Trillium Diagnostics, Scarborough, ME, USA). Briefly, 50 µL of well-mixed anticoagulated whole blood was incubated for 10 minutes at room temperature with saturating amounts of fluorescein isothiocyanate-conjugated anti CD64 murine monoclonal antibody or isotype control (Leuko64 kit; Trillium Diagnostics), followed by ammonium chloride-based red cell lysis. Samples were washed once and resuspended in 0.5 mL of phosphate-buffered saline with 0.1% bovine serum albumin. Flow cytometric analyses were performed using a Becton-Dickinson FACScan system to collect data on the logarithm of green (fluorescein isothiocyanate) and linear right-angle side and forward scatter for a minimum of 50,000 leukocytes. Interassay standardization and CD64 quantitation were performed by using fluorescein isothiocyanate calibration beads (Leuko64; Trillium Diagnostics). Data analysis was performed by using light scatter gating to define the neutrophil population, and the neutrophil CD64 index was quantified as mean equivalent soluble fluorescence units by using QuickCal for Winlist (Verity Software House, Topsham, ME, USA); correction for nonspecific antibody binding was performed by substracting values for the isotope control.
3. Statistical analysis
Data were expressed as mean±SE. Comparisons between two groups were analyzed by unpaired Student's t-test for normally distributed (parametric) data and Mann-Whitney test for non-parametric data, while proportions of patients were compared by χ2 test. For multiple-group comparisons, data were analyzed with analysis of variance. To determine a diagnostic cutoff value for neutrophil CD64 in newborns, a ROC curve was constructed for each sampling point. The optimal cutoff value for CD64 was then selected on the graph with sensitivity and specificity >80%. All data were analyzed using IBM SPSS ver. 18.0 (IBM, New York, NY, USA). A P value <0.05 was considered to be statistically significant.
4. Ethical approval
The study was approved by the Research Ethics Committee of Kangwon National University Hospital. Informed written consent was received from parents of all study patients.
Results
1. Characteristics of study population
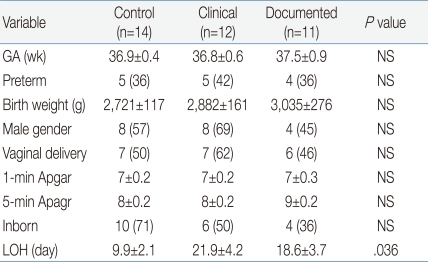
Thirty-seven neonates were enrolled in the study and classified into three groups (Table 1) as follows: 1) Documented sepsis (n=11); 2) Clinical sepsis (n=12), and 3) Control (n=14).
Table 1.
Characteristics of Study Grooups
Values are presented as mean±SE or number (%).
LOH, length of hospitalization; NS, not significant.
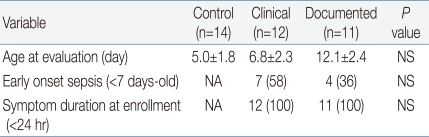
There were no significant differences in gestational age, birth weight, preterm birth, sex distribution, mode of delivery, Apgar scores or inborn births among the three groups (Table 1). However, hospital length-of-stay was significantly longer in sepsis groups compared to control group (Table 1). As shown in Table 2, enrollment age of subject was similar among the three groups, and all patients in groups 2 and 3 were enrolled within 24 hours of onset of suspected infection. The number of early onset sepsis cases, defined as presentation of symptoms or signs before 7 days of life, did not differ between the clinical and documented groups (Table 2).
Table 2.
Characteristics of Study Groups at Study Entry
Values are presented as mean±SE or number (%).
NA, not available; NS, not significant.
2. CRP, CD64 and hematologic profiles
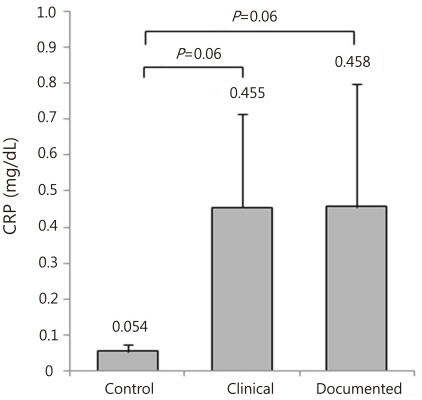
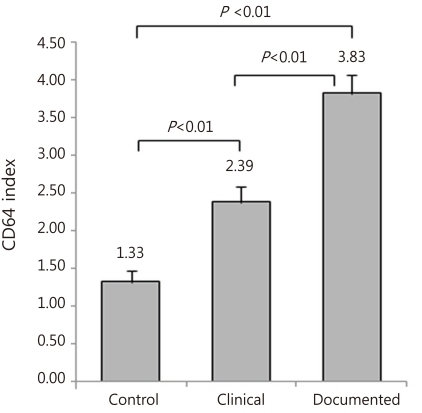
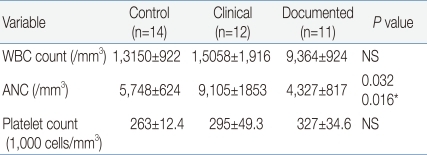
CRP concentrations were not significantly different among the three groups and generally were less than 1.0 mg/dL in the three groups (Fig. 1). Especially, CRP did not differ between the clinical and documented group (Fig. 1). However, CD64 index increased significantly in the clinical and documented groups compared to the control group and in particular, presented significantly higher in the documented group compared to the clinical group (Fig. 2). Hematologic profiles including WBC and platelet count were not different among the three groups (Table 3). However, ANC was significantly different among the three groups, and also was significantly lower in the documented group compared to the clinical group (Table 3).
Fig. 1.
C-reactive protein (CRP) concentrations in the control, clinical and documented group. The results are represented as the mean±SE.
Fig. 2.
CD64 index in the control, clinical and documented group. The results are represented as the mean±SE.
Table 3.
Hematologic Profiles of Study Groups
Values are presented as mean±SE.
WBC, white blood cells; ANC, absolute neutrophil count; NS, not significant.
*Analyzed between the clinical and documented group.
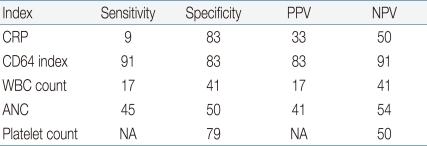
3. Sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV) of CD64 and CRP
Sensitivity, specificity, PPV and NPV of CD64 and CRP for the documented sepsis were assessed using the clinical group as reference (Table 4). For culture-positive sepsis episodes, CD64 and CRP had a sensitivity of 91% and 9%, a specificity of 83% and 83%, a PPV of 83% and 33% and an NPV of 9% and 50%, respectively, with a cutoff value of 3.0 for CD64 and 1.0 mg/dL for CRP (Table 4). Hematologic profiles including WBC, ANC and platelet count presented variable sensitivity and NPV ranging from 17 to 50% (Table 4).
Table 4.
Sensitivity, Specificity, PPV and NPV
Values are presented as (%).
CRP, C-reactive protein; WBC, white blood cell; ANC, absolute neutrophil count; PPV, positive predictive value; NPV, negative predictive value; NA, not available.
Additionally, we investigated the sensitivity and specificity of CD64/CRP combination, and addition of CRP to CD64 showed a sensitivity of 100% and a specificity of 66%.
4. ROC-curve analysis of CD64 and CRP
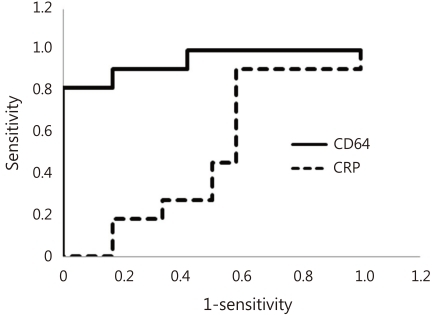
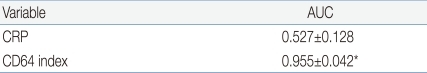
The ROC curves of CD64 and CRP for the documented sepsis are taken using the clinical group as reference (Fig. 3). There were 11 documented sepsis episodes with positive culture results: 6 involved coagulase-negative staphylococcal species, 1 Enterobacter cloacae, 2 Streptococcus anginosus, 1 Streptococcus agalactiae, 1 Streptococcus viridance. All these pathogens were identified within 24 hours from starting of the blood cultures. And out of six patients with coagulase-negative staphylococcal species grown, two patients were feeding parenterally for around 2 weeks, and another one patient had a rapidly enlarging and infected epidermal cyst at his back, where the same pathogen has grown, and another two patients were suspicious of NEC, and another one patient had aspiration pneumonia with severe leukocytosis at birth due to prolonged labour. For the documented sepsis episodes, CD64 index had a high area under curve (AUC, 0.955) in the ROC analysis, whereas CRP had an AUC of 0.527 (P<0.01) (Table 5, Fig. 3).
Fig. 3.
Receiver operating characteristic-curve analysis of CD64 and C-reactive protein (CRP) in documented sepsis episodes (n=11). CD64 had higher area under curve (0.955) compared with CRP (0.527).
Table 5.
Comparison of AUC Values from Receiver Operating Characteristic Curves
Values are presented as mean±SE.
CRP, C-reactive protein; AUC, area under curve.
*AUC value of CD64 is significantly higher when compared to CRP (P<0.01).
Discussion
An accurate inflammatory marker with high diagnostic sensitivity, specificity and NPV for neonatal sepsis would be a valuable tool for therapeutic decision-making and avoidance of unnecessary use of antibiotics10,16). In the current study, CD64 as a leukocyte surface antigen was compared to the conventional and routine inflammatory marker, CRP. Our investigation showed that CD64 was a more superior diagnostic marker for the early detection of neonatal sepsis compared to CRP as a single determination.
In the current study, we evaluated CD64 and CRP's diagnostic accuracy for only neonatal sepsis excluding local infections, unlikely to the previous studies12). The reason for this is that local infections such as pneumonia, urinary tract infection, meningitis or NEC can be identified easily by other procedures including chest X-ray, urinalysis, spinal tap or other X-rays. However sepsis can be confirmed by only blood culture, that takes at least 48 hours for the result to be available. Hence, neonatal systemic infection is the most important cause of neonatal mortality and morbidity, and consequently, early identification of sepsis is still a critical issue in neonatal area.
In terms of hematologic indices, our findings presented that only ANC significantly differed among the three groups. It has already been recognized that hematologic indices such as WBC and ratios alone cannot be confidently used as decision criteria for the identification of sepsis or for guiding antibiotic treatment17). Our observations regarding WBC are congruent with a previous study showing variable sensitivity and specificity ranging from 17 to 90% for WBC and ratios17). However, in general, an immature/total (I/T) ratio ≥0.2 is regarded as having high sensitivity, and leukopenia or neutropenia are considered to have high specificity17-19). Similarly, our observations showed low neutrophil counts (-4,500 cells/µL) in the documented group, which was a significantly lower value compared to the -9,000/µL observed in the clinical group. However, the sensitivity, specificity, PPV and NPV of ANC in our study ranged from 40 to 54%, which implicates that ANC alone cannot be a sensitive laboratory marker for neonatal sepsis. Our observation is partly supported by the previous study demonstrating WBC counts and ANCs can be affected by many factors besides infection, including age of the neonate, blood sampling method, mode of delivery, maternal hypertension and gender20). In the present study, it was not available to assess the accuracy of I/T ratio as an inflammatory marker, because the immature neutrophils (band count) were not counted in the patients enrolled in the current study. Additionally, in the present study, it was not feasible to evaluate sensitivity and PPV of platelets, because there were no culture-confirmed cases with platelet counts less than 150,000/µL. However, similar to other hematologic indices, the NPV of platelets was only 50%.
CRP goes up rapidly at around 24 hours after onset of infection. Consequently, CRP requires serial measures after 24 hours for accurate identification of sepsis, which is supported by a previous report showing that the sensitivity and NPV of CRP are enhanced higher when measured serially 24-hours later from admission, compared to a single measure taken at admission in premature infants21).
CD64 is normally expressed in very low concentrations by unstimulated neutrophils, whereas it is considerably upregulated on the trigger of bacterial invasion22). Namely, CD64 is the activation marker for neutrophils, and neutrophils react within an hour of acute inflammation23). And CD64 has already been identified as a high-affinity Fc-gamma receptor of IgG antibody in the process of phagocytosis and intracellular killing of opsonized microbes24,25). The pathogens that require opsonization are encapsulated bacteria26), so that CD64 expression is not be related to viral infection. Based on these unique properties, CD64 is presumed to increase within an hour of bacterial infection, differently from CRP. In our study, symptom or sign duration of enrolled neonates were all within 24 hours, and the sensitivity, the specificity, the PPV and the NPV of CD64 were shown to be 91%, 83%, 83% and 91%, respectively, which were higher than those observed with CRP. Based on the fact that CRP usually goes up after 24 hours from onset of infection, these findings may be explained by the nature of CD64 to be capable of going up within one hour of infection. Although diagnostic utilities such as sensitivity, specificity, PPV and NPV, eventually determine the accuracy and usefulness of a clinical test, a high sensitivity and NPV would be the most important in neonatal infection because all genuinely infected newborns should be identified and treated25). Additionally, acceptable specificity is considered to be >80% so that unnecessary use of antibiotics could be minimized25). In the current study, we chose CD64's cutoff value of 3.0 to be the optimal point for a high sensitivity, specificity, PPV and NPV >80%. The cutoff value, 3.0, chosen in the present study, was close to 4.0 selected in the previous study27), where CD64 index had a sensitivity of 80%, and a specificity of 79%, with a cutoff value of 4.02 for culture-positive sepsis episodes in neonates27).
Additionally, the sensitivity and specificity of the CD64/CRP combination showed 100% and 66%, respectively. Hence the sensitivity showed similar to single CD64's (91%), whereas the specificity showed lower than 80% and rather lower than single CRP's. These results imply that the CD64/CRP combination could be efficacious in guiding decisions to start empirical antibiotic therapy similarly to CD64, while not be efficacious in guide decisions to withhold antibiotic therapy.
According to the previous evidence, the diagnostic accuracy of CD64 for sepsis was achieved 24 hours later in term newborns10). However the recent study showed that CD64 index measured within 24 hours from onset of suspected sepsis was reliable with high sensitivity, specificity, PPV and NPV for the early determination of sepsis, which indicates that CD64 is a useful laboratory marker as a single value for neonatal sepsis, regardless of the time from onset of suspected sepsis. Additionally, our present data showed that CD64 index has a significantly higher AUC (0.955) compared to CRP (0.527), with a cutoff value of 3.0 of CD64 and 1.0 mg/dL of CRP.
In the current study, coagulase-negative staphylococci have grown in six patients, and all these pathogens were identified within 24 hours from starting of the blood cultures. Based on the previous evidences showing that pediatric BAC-TEC system found 97% positive within 48 hours from starting of the blood culture, and microorganisms grown beyond 48 hours from starting of the blood cultures should mostly be caused by contamination28), and in most circumstances, if blood culture results are not reported as positive by 48 hours, then empiric administration of antibiotics may be discontinued14,29,30), coagulase-negative staphylococci grown in six patients enrolled in the study should be considered to be pathogens. Additionally, their clinical backgrounds such as prolonged total parenteral nutrition, suspicious NEC and infected epidermal cyst so on forth mentioned in the results should support that they are pathogens, not contaminants.
CD64 index has some general favorable characteristics; The expression of CD64 does not differ with age, as its expression only occurs upon cell activation21) and is stable for more than 30 hours at room temperature27). In addition to accuracy, the laboratory test for CD64 is rapid (<60 minutes) with the use of flow cytometry and requires minimal blood volume (<100 µL). In fact, in our study, no extra blood was obtained to calculate the CD64 index, as CBC samples proved sufficient. Hence, it is practical to obtain the CD64 index in a clinical setting. However, the critical issues such as cost and clinical settings should be considered carefully. For the current study, it cost approximately $16 to obtain one CD64 index, which was more expensive than CRP's. For the recent study, CD64 index were measured using a FACscan system established at the department of clinical diagnosis.
In conclusion, our preliminary study suggests that the CD64 index is a more reliable diagnostic measure for confirmed neonatal sepsis as a single value compared to CRP, and the CD64 index may be promise for replacing CRP. However, the important issues of cost and availability are required to be evaluated in routine clinical setting.
Acknowledgements
We thank the nursing staffs of NICU, Kangwon National University Hospital, for their valuable cooperation.
References
- 1.Bizzarro MJ, Raskind C, Baltimore RS, Gallagher PG. Seventy-five years of neonatal sepsis at Yale: 1928-2003. Pediatrics. 2005;116:595–602. doi: 10.1542/peds.2005-0552. [DOI] [PubMed] [Google Scholar]
- 2.Lawn JE, Wilczynska-Ketende K, Cousens SN. Estimating the causes of 4 million neonatal deaths in the year 2000. Int J Epidemiol. 2006;35:706–718. doi: 10.1093/ije/dyl043. [DOI] [PubMed] [Google Scholar]
- 3.Ng PC, Cheng SH, Chui KM, Fok TF, Wong MY, Wong W, et al. Diagnosis of late onset neonatal sepsis with cytokines, adhesion molecule, and C-reactive protein in preterm very low birthweight infants. Arch Dis Child Fetal Neonatal Ed. 1997;77:F221–F227. doi: 10.1136/fn.77.3.f221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Stoll BJ, Holman RC, Schuchat A. Decline in sepsis-associated neonatal and infant deaths in the United States, 1979 through 1994. Pediatrics. 1998;102:e18. doi: 10.1542/peds.102.2.e18. [DOI] [PubMed] [Google Scholar]
- 5.Icardi M, Erickson Y, Kilborn S, Stewart B, Grief B, Scharnweber G. CD64 index provides simple and predictive testing for detection and monitoring of sepsis and bacterial infection in hospital patients. J Clin Microbiol. 2009;47:3914–3919. doi: 10.1128/JCM.00628-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Batlivala SP. Focus on diagnosis: the erythrocyte sedimentation rate and the C-reactive protein test. Pediatr Rev. 2009;30:72–74. doi: 10.1542/pir.30-2-72. [DOI] [PubMed] [Google Scholar]
- 7.Ng PC, Lam HS. Diagnostic markers for neonatal sepsis. Curr Opin Pediatr. 2006;18:125–131. doi: 10.1097/01.mop.0000193293.87022.4c. [DOI] [PubMed] [Google Scholar]
- 8.Hoffmeyer F, Witte K, Schmidt RE. The high-affinity Fc gamma RI on PMN: regulation of expression and signal transduction. Immunology. 1997;92:544–552. doi: 10.1046/j.1365-2567.1997.00381.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dilli D, Oğuz ŞS, Dilmen U, Köker MY, Kızılgün M. Predictive values of neutrophil CD64 expression compared with interleukin-6 and C-reactive protein in early diagnosis of neonatal sepsis. J Clin Lab Anal. 2010;24:363–370. doi: 10.1002/jcla.20370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Groselj-Grenc M, Ihan A, Pavcnik-Arnol M, Kopitar AN, Gmeiner-Stopar T, Derganc M. Neutrophil and monocyte CD64 indexes, lipopolysaccharide-binding protein, procalcitonin and C-reactive protein in sepsis of critically ill neonates and children. Intensive Care Med. 2009;35:1950–1958. doi: 10.1007/s00134-009-1637-7. [DOI] [PubMed] [Google Scholar]
- 11.Bhandari V, Wang C, Rinder C, Rinder H. Hematologic profile of sepsis in neonates: neutrophil CD64 as a diagnostic marker. Pediatrics. 2008;121:129–134. doi: 10.1542/peds.2007-1308. [DOI] [PubMed] [Google Scholar]
- 12.Ng PC, Li G, Chui KM, Chu WC, Li K, Wong RP, et al. Neutrophil CD64 is a sensitive diagnostic marker for early-onset neonatal infection. Pediatr Res. 2004;56:796–803. doi: 10.1203/01.PDR.0000142586.47798.5E. [DOI] [PubMed] [Google Scholar]
- 13.Manroe BL, Weinberg AG, Rosenfeld CR, Browne R. The neonatal blood count in health and disease. I. Reference values for neutrophilic cells. J Pediatr. 1979;95:89–98. doi: 10.1016/s0022-3476(79)80096-7. [DOI] [PubMed] [Google Scholar]
- 14.Gonzalez BE, Mercado CK, Johnson L, Brodsky NL, Bhandari V. Early markers of late-onset sepsis in premature neonates: clinical, hematological and cytokine profile. J Perinat Med. 2003;31:60–68. doi: 10.1515/JPM.2003.009. [DOI] [PubMed] [Google Scholar]
- 15.Smulian JC, Bhandari V, Campbell WA, Rodis JF, Vintzileos AM. Value of umbilical artery and vein levels of interleukin-6 and soluble intracellular adhesion molecule-1 as predictors of neonatal hematologic indices and suspected early sepsis. J Matern Fetal Med. 1997;6:254–259. doi: 10.1002/(SICI)1520-6661(199709/10)6:5<254::AID-MFM2>3.0.CO;2-F. [DOI] [PubMed] [Google Scholar]
- 16.Morsy AA, Elshall LY, Zaher MM, Abd Elsalam M, Nassr AE. CD64 cell surface expression on neutrophils for diagnosis of neonatal sepsis. Egypt J Immunol. 2008;15:53–61. [PubMed] [Google Scholar]
- 17.Da Silva O, Ohlsson A, Kenyon C. Accuracy of leukocyte indices and C-reactive protein for diagnosis of neonatal sepsis: a critical review. Pediatr Infect Dis J. 1995;14:362–366. doi: 10.1097/00006454-199505000-00005. [DOI] [PubMed] [Google Scholar]
- 18.Rodwell RL, Leslie AL, Tudehope DI. Early diagnosis of neonatal sepsis using a hematologic scoring system. J Pediatr. 1988;112:761–767. doi: 10.1016/s0022-3476(88)80699-1. [DOI] [PubMed] [Google Scholar]
- 19.Cornbleet PJ. Clinical utility of the band count. Clin Lab Med. 2002;22:101–136. doi: 10.1016/s0272-2712(03)00069-6. [DOI] [PubMed] [Google Scholar]
- 20.Newman TB, Puopolo KM, Wi S, Draper D, Escobar GJ. Interpreting complete blood counts soon after birth in newborns at risk for sepsis. Pediatrics. 2010;126:903–909. doi: 10.1542/peds.2010-0935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wagle S, Grauaug A, Kohan R, Evans SF. C-reactive protein as a diagnostic tool of sepsis in very immature babies. J Paediatr Child Health. 1994;30:40–44. doi: 10.1111/j.1440-1754.1994.tb00564.x. [DOI] [PubMed] [Google Scholar]
- 22.Fjaertoft G, Håkansson L, Ewald U, Foucard T, Venge P. Neutrophils from term and preterm newborn infants express the high affinity Fcgamma-receptor I (CD64) during bacterial infections. Pediatr Res. 1999;45:871–876. doi: 10.1203/00006450-199906000-00016. [DOI] [PubMed] [Google Scholar]
- 23.Cohen S, Burns RC. Pathways of the pulp. 8th ed. St. Louis: Mosby; 2002. p. 465. [Google Scholar]
- 24.de Haas M, Vossebeld PJ, von dem Borne AE, Roos D. Fc gamma receptors of phagocytes. J Lab Clin Med. 1995;126:330–341. [PubMed] [Google Scholar]
- 25.Sánchez-Mejorada G, Rosales C. Signal transduction by immunoglobulin Fc receptors. J Leukoc Biol. 1998;63:521–533. doi: 10.1002/jlb.63.5.521. [DOI] [PubMed] [Google Scholar]
- 26.Roitt I, Brostoff J, Male DK. Immunology. 6th ed. New York: Mosby; 2001. p. 5. [Google Scholar]
- 27.Davis BH, Olsen SH, Ahmad E, Bigelow NC. Neutrophil CD64 is an improved indicator of infection or sepsis in emergency department patients. Arch Pathol Lab Med. 2006;130:654–661. doi: 10.5858/2006-130-654-NCIAII. [DOI] [PubMed] [Google Scholar]
- 28.Rowley AH, Wald ER. Incubation period necessary to detect bacteremia in neonates. Pediatr Infect Dis. 1986;5:590–591. doi: 10.1097/00006454-198609000-00022. [DOI] [PubMed] [Google Scholar]
- 29.Janjindamai W, Phetpisal S. Time to positivity of blood culture in newborn infants. Southeast Asian J Trop Med Public Health. 2006;37:171–176. [PubMed] [Google Scholar]
- 30.Jardine L, Davies MW, Faoagali J. Incubation time required for neonatal blood cultures to become positive. J Paediatr Child Health. 2006;42:797–802. doi: 10.1111/j.1440-1754.2006.00980.x. [DOI] [PubMed] [Google Scholar]