Abstract
Mucopolysaccharidosis VII (MPS VII) is due to mutations within the gene encoding the lysosomal enzyme β-glucuronidase, and results in the accumulation of glycosaminoglycans. MPS VII causes aortic dilatation and elastin fragmentation, which is associated with upregulation of the elastases cathepsin S (CtsS) and matrix metalloproteinase 12 (MMP12). To test the role of these enzymes, MPS VII mice were crossed with mice deficient in CtsS or MMP12, and the effect upon aortic dilatation was determined. CtsS deficiency did not protect against aortic dilatation in MPS VII mice, but also failed to prevent an upregulation of cathepsin enzyme activity. Further analysis with substrates and inhibitors specific for particular cathepsins suggests that this enzyme activity was due to CtsB, which could contribute to elastin fragmentation. Similarly, MMP12 deficiency and deficiency of both MMP12 and CtsS could not prevent aortic dilatation in MPS VII mice. Microarray and reverse-transcriptase real-time PCR were performed to look for upregulation of other elastases. This demonstrated that mRNA for complement component D was elevated in MPS VII mice, while immunostaining demonstrated high levels of complement component C3 on surfaces within the aortic media. Finally, we demonstrate that neonatal intravenous injection of a retroviral vector encoding β-glucuronidase reduced aortic dilatation. We conclude that neither CtsS nor MMP12 are necessary for elastin fragmentation in MPS VII mouse aorta, and propose that CtsB and/or complement component D may be involved. Complement may be activated by the GAGs that accumulate, and may play a role in signal transduction pathways that upregulate elastases.
Keywords: Mucopolysaccharidosis VII, Cathepsin S, Matrix metalloproteinase 12, Complement system, Aortic dilatation, Gene therapy
1. Introduction
Mucopolysaccharidoses (MPS) are a group of 11 diseases caused by mutations in genes that encode lysosomal enzymes that degrade glycosaminoglycans (GAGs) [1]. MPS is associated with accumulation of GAGs throughout the body and multisystemic disease. The focus of this project was to better understand the pathogenesis of aortic disease in MPS using the murine model of MPS VII, which is an autosomal recessive disease due to β-glucuronidase (GUSB) deficiency (Online Mendelian Inheritance in Man #253220). An elegant model for the pathogenesis of MPS involves the binding of GAGs to Toll-like Receptor 4 (TLR4), which upregulates cytokines such as tumor necrosis factor α (TNFα), Ccl4 (MIP-1β), and interleukin 6 (IL-6), which in turn upregulate destructive proteases [2–4]. Adult humans with attenuated MPS I have aortas that are 122% of normal diameter [5] and reduced elasticity [6], while one patient with MPS VII required an aortic graft [7]. Mice with MPS I [8–10] and MPS VII [11], cats with MPS I and MPS VI [12], and dogs with MPS I [13–15] and MPS VII [14,16,17] also have aortic dilatation.
Elastin represents 30% of the dry weight of the aorta [18]. Tropoelastin monomers are secreted and then crosslinked into elastic fibers in a process that involves elastin binding protein, extracellular matrix microfibrils, and crosslinking enzymes [19]. Elastin was fragmented in the ascending aorta of humans, mice, and dogs with MPS I, and in humans and dogs with MPS VII [10,13,20–22]. Hinek et al. demonstrated that exogenous administration of dermatan sulfate, a GAG that accumulates in many types of MPS, reduced elastin binding protein levels and inhibited elastin assembly in vitro, and proposed that reduced assembly caused elastin defects in MPS I [23]. Alternatively, we reported in MPS I mice and dogs and in MPS VII dogs [10,14] that elastin fragmentation was temporally associated with increases in RNA and enzyme activity for two elastases, cathepsin S (CtsS) and matrix metalloproteinase 12 (MMP12), which contribute to aortic aneurisms in mouse models [24,25], and proposed that degradation was the major factor leading to elastin fragmentation. Although collagen is another important extracellular matrix protein of the aorta, collagen fibrils appeared to be relatively intact with histochemical stains in MPS I and MPS VII dogs [14].
Hematopoietic stem cell transplantation can reduce clinical manifestations of MPS, as hematopoietic cells migrate into tissues and secrete mannose 6-phosphate (M6P)-modified enzyme that can be taken up via the M6P receptor by nearby cells [26]. This has reduced, but not prevented, accumulation of GAGs, elastin fragmentation, and/or dilatation of the aorta in MPS VII mice [27] and dogs [16], MPS VI rats [28], and MPS I dogs [15] and cats [29]. Enzyme replacement therapy (ERT) involves intravenous (IV) injection of M6P-modified enzyme that can diffuse to organs and be taken up via the M6P receptor [30,31], although ERT is not available for MPS VII. ERT had little effect on lysosomal storage accumulation in aortic smooth muscle cells in MPS VI cats [32]. Gene therapy is also being tested in animal models [33]. One approach involves neonatal IV injection of a retroviral vector (RV) expressing the appropriate enzyme, which results in transduction of liver cells and secretion of enzyme into blood [33–35]. This reduced aortic dilatation, but MPS I mice required very high expression for a full therapeutic effect [8–10], and MPS VII dogs developed aortic dilatation after 5 years [14].
The data that CtsS and MMP12 are upregulated in MPS aortas led us to hypothesize that deletion of CtsS and/or MMP12 might reduce elastin fragmentation. To test this hypothesis, CtsS−/− and MMP12−/− mice were crossed with MPS VII (GUSB−/−) mice and the effect upon the aorta diameters was determined. In addition, microarray analysis was performed to determine if other genes that could contribute to aortic dilatation were upregulated. The results demonstrate that CtsS and MMP12 are not essential for aortic dilatation, but a related cathepsin, CtsB, may contribute. These studies also demonstrate that the complement system may directly result in elastin fragmentation, or may indirectly contribute by induction of signal transduction pathways that result in upregulation of elastases.
2. Materials and methods
Reagents were from Sigma-Aldrich Chemical (St. Louis, MO) unless otherwise stated.
2.1. Animals
National Institutes of Health (NIH) guidelines for the care and use of animals in research were followed. MPS VII [27], CtsS-deficient [36], and MMP12 deficient [37] mice were all in a C57BL/6 background. Some MPS VII mice were injected IV with 1 × 1010 transducing units/kg of the RV designated hAAT-cGUSB-WPRE [38] at 2 to 3 days after birth to enable them to survive and breed. Genotyping for MPS VII mice used a SNP assay on tail DNA with a forward primer (5′ CCATAGTCATGATACCAAGAAAAGTAGCT-3′), a reverse primer (5′-TGACTATTCTGACCTCAGTGTGTGA-3′), a wild-type minor-groove binding probe labeled with Vic (5′-TTGTCTTAGGCCCCGTACGT-3′; the underlined C represents the position deleted in the MPS VII mouse), a mutant minor groove binding probe labeled with FAM (5′-TTGTCTTAGGCCC-GTACGT-3′) and a One-Step Plus PCR machine (Applied Biosystems; Foster City, CA). PCR for the wild-type CtsS allele used the primers 5′-CTTGAAGGGCAGCTGAAGCTG-3′ (forwards) and 5′-GTAGGAAGCGTCTGCCTCTAT-3′ (reverse), and PCR for the mutant CtsS allele used the primers 5′-CTCTGTGTAGCCTGGAATTC-3′ (forwards) and 5′-CTAAAGCGCATGCTCCAGACTGCC-3′ (reverse) [36] with analysis of the CT using SYBR green real-time PCR of DNA. MMP12 mice genotyping PCR used a forward primer common to the wild-type and mutant MMP12 alleles (5′-CCCTCGATGTGGAGTGCCCG-3′), a reverse primer specific for the PGK-neo cassette (5′-AAGAACGGAGCCGGTTGGCG-3′), and a reverse primer specific for the MMP12 wild-type allele (5′-ACTTGCCCTGAGCACCCCCT-3′), with gel electrophoresis used to identify wild-type (337 bp) or mutant (460 bp) alleles.
2.2. Measurement of aortic diameter and histopathology
Mice were anesthetized with 120 mg/kg ketamine/40 mg/kg xylazine in phosphate buffered saline (PBS) at pH 7.4. For some animals, aortic compliance was assessed, in which the outer diameter of isolated aortas was determined at different internal pressures [39]. For histopathology, ascending aortas obtained from 1 to 2 mm from the aortic valve were fixed with buffered formalin, embedded in paraffin, and 6 µm sections were stained with Verhoeff Van Gieson (VVG) stain. For biochemical analyses, animals were perfused with 20 ml of PBS, and the aorta from just above the aortic valve to just before the first branch was cleaned of surrounding fat. To test the effect of gene therapy, the width of dissected ascending aortas was measured after gently pressing it against a surface.
2.3. RNA analysis
Frozen ascending aortas were homogenized in Trizol for 30 s with a hand-held homogenizer (Kimble-Kontes; Vineland, NJ), and RNA was isolated using a Qiagen column. Reverse transcription (RT) was performed on 1 µg of DNase I-treated RNA with an oligo (dT) 20 primer using a Superscript III kit (Invitrogen Corp., Carlsbad, CA) in 20 µl, followed by real-time PCR on 0.4 µl of each cDNA sample using SYBR green reagents from Applied Biosystems [10]. Primer sequences are in our previous publication [10] or in Supplementary Table 1. The percentage of a test RNA to that of β-actin was calculated by subtracting the cycle to reach the threshold (CT) for a gene from the CT for a separate assay using β-actin primers to determine the ΔCT, and the formula: percent β-actin = (100) × 2ΔCT. The percent β-actin for MPS animals was divided by the percent β-actin in normal animals to determine the ratio of the gene in MPS to normal mice.
For microarray, RNA was reverse transcribed with primers with a T7 RNA polymerase binding site, amplified with T7 RNA polymerase with fluorescently-labeled deoxynucleotides, and hybridized to an Illumina bead microarray (Mouse8, version 2). Expression analysis was performed with ParTek software (St. Louis, MO). Pathway analysis was performed with GeneGo interactions software (https://portal.genego.com/cgi/data_manager.cgi; St. Joseph, MI).
2.4. Enzyme and GAG assays
For the GUSB, α-l-iduronidase (IDUA), and cathepsin assays, frozen aortas were homogenized with the hand-held homogenizer in 100 mM sodium acetate pH 5.5 containing 2.5 mM ethylenediaminetetraacetic acid, 0.1% Triton X-100, and 2.5 mM dithiothreitol, and centrifuged at 10,000 g for 5 min at 4 °C. The protein concentration was determined with the Bradford assay (BioRad Laboratories, Hercules CA). For the MMP12 and GAG assays, extracts were homogenized in the neutral buffer provided with the MMP12 kit with 0.1% Triton-X.
GUSB and IDUA assays were performed with the extracts prepared at pH 5.5 using the fluorogenic substrates 4-methylumbelliferyl-β-l-glucuronide (Sigma-Aldrich, St. Louis, MO) for GUSB and 4-methylumbelliferyl-α-l-iduronide (Toronto Research Chemicals, North York, Canada) for IDUA and a Fluoroskan Ascent microplate fluorometer (Thermo Electron, Milford, MA) as previously described [9]. One unit of enzyme converts 1 nmol of substrate to product per hour at 37 °C. GAG content was determined in the samples obtained at neutral pH using the commercial kit Blyscan (Biocolor, Carrickfergus, UK) using 30 µg of protein from each sample as described [10].
For the general cathepsin assay, 1 µg or less of the supernatant was incubated with 100 µM benzyloxycarbonyl-l-phenylalanyl-l-arginine-7-amido-4-methylcoumarin (Z-Phe-Arg-AMC) from Anaspec (San Jose, CA) at pH 7.5 in 100 mM sodium acetate with 2.5 mM ethylenediaminetetraacetic acid, 0.01% Triton X-100, and 2.5 mM dithiothreitol in a microtiter plate at 37 °C for 1 h [10]. The amount of product was determined by excitation at 355 nm and emission at 460 nm using kinetic readings and comparison with 7-amino-4-methylcoumarin (AMC) standards from Anaspec. One unit (U) of enzyme released 1 nmol of product per hour at 37 °C. The CtsB assay used the same extracts, the substrate Z-Arg-Arg-AMC (Bachem, Torrance, CA) at pH 7.5, and the same wavelengths as for the general cathepsin assay. CtsK activity was measured at pH 7.5 with 10 µM of the substrate 2-aminobenzoic acid-HPGGPQ-N-(2,4-dinitrophenyl)-ethylenediamine (Abz-HPGGPQ-EDDnp) from Anaspec, which is cleaved by CtsK but not other cathepsins, and 2-aminobenzoic acid was the standard. The CtsD assay was performed at pH 4 with 10 µM of the substrate 7-methoxycoumarin-4-acetyl (Mca)-Gly-Lys-Pro-Ile-Leu-Phe-Phe-Arg-Leu-Lys-2,4 nitrophenyl (Dnp)-D-Arg-NH2, which can also be cleaved by CtsE, with Mca-Pro-Leu-OH (Enzo Life Sciences) as the standard. CtsK and CtsD assays were read at 320 nm for excitation and 420 nm for emission. Inhibitors were from Calbiochem (San Diego, CA) and included the CtsS inhibitor Z-FL-COCHO (#219393), the CtsK inhibitor I [1,3-Bis (N-carbobenzoyloxy-l-leucyl) amino acetone; #219377] and the CtsB inhibitor Ac-Leu-Val-Lysinal (#219385). Samples were incubated with the inhibitor for 10 min prior to starting the assay. Additional assays were performed with human recombinant purified CtsB [R&D systems, Minneapolis, MN; specific activity 150 nmol of substrate cleaved per hour (U)/µg protein], CtsK (Enzo Life Sciences, Farmington, NY; 90 U/µg protein), CtsL (R&D systems; 900 U/µg protein), CtsS (R&D systems; 18 U/µg protein) and with CtsH purified from human liver (Enzo Life Sciences; 61 U/µg protein). An MMP12 assay kit (SensolyteTM 490 MMP12) was obtained from Anaspec for which the substrate can also be cleaved by MMP1, 2, 3, 8, and 13 and was performed as described previously with ~5 µg of extract in 100 µl reactions [10].
2.5. Immunostaining
Immunostaining for STAT3 that was phosphorylated at tyrosine 705 was performed as described previously [10]. For C3 immunostaining, frozen sections of aorta in optimal cutting temperature compound were fixed with formalin for 10 min at room temperature, and washed 3 times with TBS (Tris-buffered saline; 50 mM Tris pH 7.6, 150 mM NaCl, 0.1% Triton X-100). Endogenous peroxidase was inhibited with 0.6% H2O2 in water for 30 min. Samples were washed 3 times with TBS, and then preincubated with blocking buffer (TBS with 10% horse serum) for 30 min at room temperature. A goat-anti-mouse antibody specific for C3 (MP Biomedicals #55474, Solon, OH) was incubated overnight at 4 °C at a 1:100 dilution in blocking buffer, and then washed 3 times with TBS. A horse-radish peroxidase-conjugated horse anti-goat IgG (Vector Laboratories, Burlingame CA) was incubated at a 1:100 dilution for 1 h at RT in blocking buffer, and samples were washed 3 times with TBS. Samples were developed with 0.7 mg/ml 3,3′-diaminobenzidine with 0.7 mg/ml urea for 5 min.
2.6. Statistics
The Student's t test compared values between 2 groups, and ANOVA with Tukey post-hoc analysis compared values between 3 or more groups using Sigma Stat software (Systat Software, Inc., Point Richmond, CA).
3. Results
3.1. Aortic dilatation in MPS VII mice
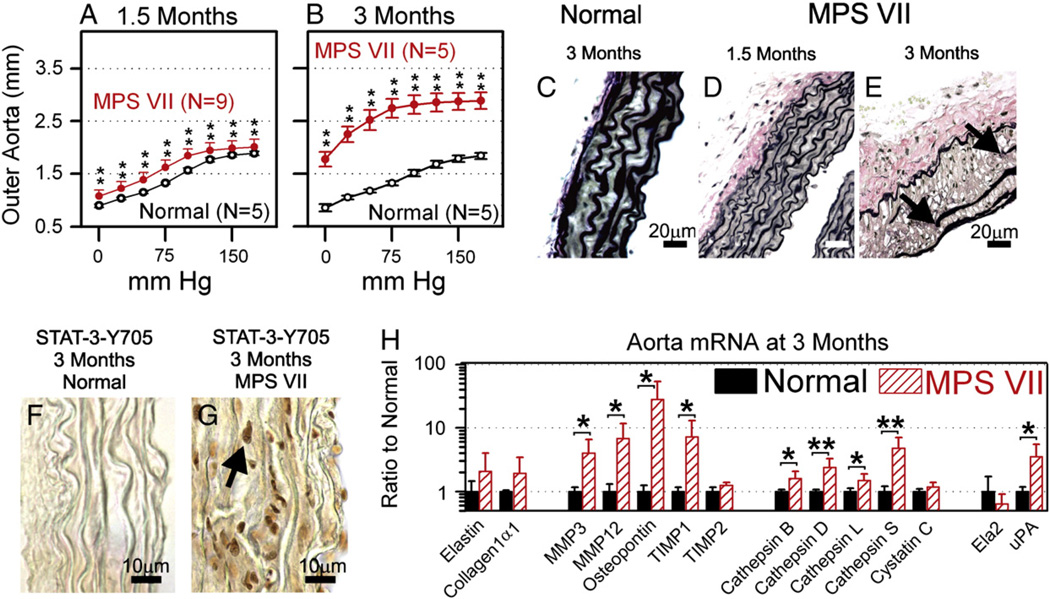
A goal of this study was to determine if deficiency in other genes could reduce aortic dilatation in MPS animals. We chose to study mice, as animals with deficiency of our candidate genes, CtsS and MMP12, were available. We had previously demonstrated in MPS I mice that aortic dilatation was severe at 6 months, but was only mild at 3 months [10]. Since aortic disease was more severe in MPS VII dogs than in MPS I dogs [14], we postulated that disease might similarly develop more quickly in MPS VII mice than in MPS I mice, which would allow for earlier analysis for a protective effect of other genes. Others had demonstrated with echocardiography that MPS VII mice had ascending aorta internal diameters that were 158% of normal at 5 months [11], but evaluation at earlier ages was not performed. As shown in Fig. 1A, isolated aortas from male MPS VII mice had mild dilatation at 1.5 months of age, when the outer diameter was 1.6 ± 0.1 mm at 75 mm Hg, which was 122% of the value of 1.3 ± 0.02 mm Hg found in normal mice (p<0.001). At 3 months (Fig. 1B), the outer diameter of male MPS VII mouse aortas was markedly dilated at 2.7 ± 0.2 mm at 75 mm Hg, which was 208% of the value of 1.3 ± 0.04 mm Hg found in normal mice at the same age (p<0.001). In contrast, the left carotid diameter was only slightly dilated at 109% of normal (p = 0.02) and the abdominal aorta was 98% of normal (not significant) at 3 months in MPS VII mice (data not shown), demonstrating that arterial disease was more severe in the ascending aorta than in other blood vessels. At 3 months, normal mice had systolic and diastolic blood pressures (BP) of 106 ± 3 and 75 ± 3 mm Hg, respectively, while MPS VII mice had an elevated systolic BP of 146 ± 14 mm Hg, and a reduced diastolic BP of 60 ± 4 mm Hg (p<0.001 for MPS VII vs. normal mice for both systolic and diastolic BP; data not shown).
Fig. 1.
Evaluation of MPS VII aortas. A and B. Compliance curves. Aortas were isolated from normal (GUSB+/−, +/+ for other genes) or MPS VII (GUSB−/−, +/+ for other genes) males at 1.5 or 3 months of age, and the average outer diameter of the aorta at the indicated mm Hg ± SD was determined. The Student's t-test was used to compare values in the 2 groups at each pressure, and ** indicates a p valueb0.01. C–E. Elastin stain. Sections of fixed ascending aortas were stained with VVG, which stains elastin a dark color, at the indicated age. The arrows indicate the edges of fragmented elastin fibers in the MPS VII mouse, size bars indicate 20 µm. For all panels, the intima is located on the right side, and the adventitia is on the left side. F and G. Immunostain for tyrosine-phosphorylated STAT3. Frozen sections were stained for the presence of STAT3 that was phosphorylated at tyrosine 705. The arrow identifies the dark nucleus of a positive cell in the MPS VII mouse. Bars indicate 10 µm. H. mRNA levels at 3 months. RNA was isolated from aortas at 3 months of age, and real-time PCR was used to compare levels of genes in normal (N = 6) and MPS VII (N = 5) mice after normalization to β-actin levels. Abbreviations are TIMP for tissue inhibitor of metalloprotease, ELA2 for neutrophil elastase, and uPA for urokinase plasminogen activator.
Histological evaluation of the ascending aorta demonstrated normal elastin structure at 3 months in normal mice (Fig. 1C), near-normal structure at 1.5 months in MPS VII mice (Fig. 1D), and marked elastin fragmentation at 3 months in MPS VII mice (Fig. 1E). In addition, there was increased phosphorylation at tyrosine 705 of STAT3 at 3 months in the nuclei of MPS VII mice (Fig. 1G) as compared with normal mice (Fig. 1F). Furthermore, analysis of RNA levels at 3 months (Fig. 1H) demonstrated that elastin levels were near-normal at 2.1 ± 1.9-fold normal (not significant vs. normal), collagen Iα1 levels were near-normal at 1.9 ± 1.5-fold normal (not significant), MMP12 levels were elevated at 6.8 ± 4.9-fold normal (p = 0.02), and CtsS mRNA was elevated at 4.8 ± 2.3-fold normal (p = 0.004). mRNA for osteopontin (SPP1), a protein that can activate MMPs by non-proteolytic mechanisms and plays a role in signal transduction, was markedly elevated at 28 ± 25.6-fold normal (p = 0.03). In addition, mRNA for MMP3, an inhibitor of MMPs (TIMP1), cathepsin B (CtsB), cathepsin D (CtsD), cathepsin L (CtsL), and the MMP-activating enzyme urokinase plasminogen activator (uPA) were also significantly elevated, although the magnitude was not as high. These data demonstrate that aortic dilatation is due to elastin fragmentation in the MPS VII mouse model, which in turn is associated with upregulation of CtsS and MMP12 mRNA. In addition, there was upregulation of other genes and phosphorylation of the transcription factor STAT3 that mimic closely what occurs in the MPS I mouse model [10], suggesting that the pathogenesis is similar. Since aortic dilatation was severe by 3 months in MPS VII mice, analysis at 3 months or later should be informative.
3.2. Effect of CtsS or MMP12 deficiency on aortic dilatation in MPS VII mice
MPS VII (GUSB−/−) mice on a C57BL/6 background were crossed with CtsS−/− or MMP12−/− mice on C57BL/6 backgrounds, and the F1 offspring were then crossed to generate mice that were deficient in one or both genes. During breeding, some GUSB−/− mice were injected with 1 × 1010 transducing units/kg of the RV designated hAAT-cGUSB-WPRE, which contains the canine GUSB cDNA, and was previously demonstrated to result in high expression of GUSB in liver cells, high serum GUSB activity, and the ability to survive and breed long-term [38]. Aortas from RV-treated MPS VII mice were not included in analyses for mice of a particular genotype, as treatment reduced aortic dilatation. Mice from the GUSB−/− CtsS−/− colony were then crossed with mice from the GUSB−/− MMP12−/− colony to generate mice that were deficient in GUSB, CtsS, and MMP12.
Compliance studies were performed on aortas from 3 month-old male mice. Fig. 2A shows that none of the aortas were dilated in normal mice, while all of the aortas from purebred MPS VII mice (black lines in Fig. 2B) were dilated. Interestingly, some of the GUSB−/− mice that were CtsS+/+ and MMP12+/+ that had been crossed through the Cts−/− colony (red lines in panel 2B) had relatively normal diameters, while others were dilated. This suggests the presence of an independently-segregating gene in the CtsS colony that can provide protection from aortic dilatation. Similarly, some of the MPS VII mice derived from the triple colony (cross of GUSB−/− CtsS−/− mice or related genotypes with GUSB−/− MMP12−/− mice or related genotypes) that were CtsS+/+ and MMP12+/+ (blue lines in Fig. 2B) had near-normal diameters, while others were dilated to a varying extent.
Fig. 2.
Effect of CtsS and/or MMP12 deficiency on aortic dilatation in MPS VII mice. Aortas were isolated from male mice at 3 months of age, and the outer diameter was determined at the indicated mm Hg of internal pressure. Each line represents a single animal. Black lines represent animals that were never crossed through the CtsS colony, which were derived from either the original MPS VII colony or MPS VII mice that were crossed with MMP12-deficient mice. Red lines represent animals derived from a cross of the MPS VII and the CtsS−/− mice. Blue lines represent animals that were derived from a triple cross of GUSB−/−, CtsS−/−, and MMP12−/− mice. A. Normal. Phenotypically normal mice (GUSB+/− CtsS+/+ MMP12+/+) were analyzed. B. MPS VII mice. GUSB−/− CtsS+/+ MMP12+/+ mice were analyzed C. MPS VII mice with CtsS deficiency. MPS VII mice that were also deficient in CtsS (GUSB−/−, CtsS−/−, MMP12+/+) were analyzed. D. MPS VII mice with MMP12 deficiency. MPS VII mice that also deficient in MMP12 (GUSB−/−, CtsS+/+, MMP12−/−) were analyzed. E. MPS VII mice with CtsS and MMP12 deficiency. MPS VII mice that were also deficient in CtsS and MMP12 (GUSB−/−, CtsS−/−, MMP12−/−) were analyzed. F. Average aortic diameter. The average outer aortic diameter ± SD at 75 mm Hg was determined from the compliance curves in panels A to E. Statistical comparisons were performed between normal mice and other groups; ** indicates a p value<0.01.
GUSB−/− CtsS−/− mice (Fig. 2C) showed marked variability between individual mice, with some mice exhibiting near-normal diameters, and others showing marked dilatation. GUSB−/− CtsS−/− mice with near-normal aortic diameters tended to be found early in the breeding strategy when the representation of the CtsS colony was high and derived from particular parents, while animals of the same genotype with dilated aortas were usually found later in breeding and to derive from different parents. The genetic data are consistent with the presence of an independently-segregating gene in the CtsS colony that conferred protection from aortic dilatation when present in a homozygous recessive state, although the identification of this putative gene remains unknown. GUSB−/− MMP12−/− mice aortas were consistently dilated, although one animal was less severe than the others (Fig. 2D). GUSB−/− CtsS−/− MMP12−/− mice (Fig. 2E) all had dilated aortas.
Average aortic diameters ± SD at 75 mm Hg for each of these groups are shown in Fig. 2F. Aortic diameters from GUSB−/− CtsS−/− mice, GUSB−/− MMP12−/− mice, or GUSB−/− CtsS−/− MMP12−/− mice were not statistically different from GUSB−/− mice without additional deficiencies, and all MPS VII groups were statistically different from normal mice. We conclude that deficiency of CtsS, MMP12, or both cannot prevent aortic dilatation in MPS VII mice.
3.3. Effect of gene therapy on functional and biochemical abnormalities in the aorta
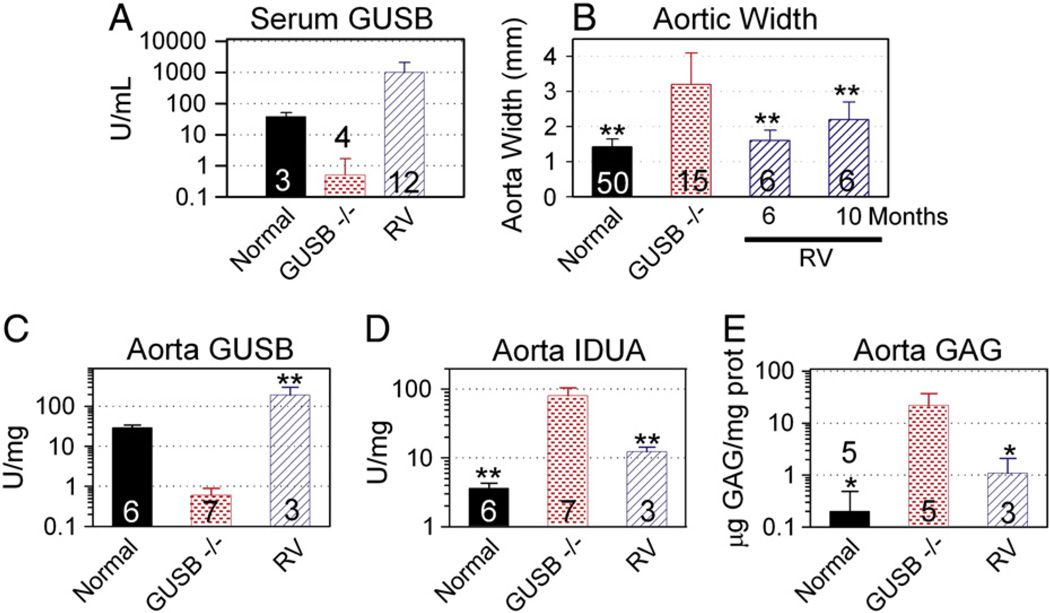
The effect of gene therapy on aortic dilatation in MPS VII mice was determined. Some MPS VII mice received IV injection of an RV expressing GUSB at 2–3 days after birth. This resulted in high GUSB activity in serum, as shown in Fig. 3A. Values in individual animals varied from 182 to 4042 U/ml, which is consistent with our previous results showing marked variation in individual RV-treated mice [38]. Aorta diameters were measured after exsanguination with no internal application of pressure. RV-treated MPS VII mice had improvement of aortic diameters to 1.6 ± 0.3 mm at 6 months of age (Fig. 3B; 113% normal but not significant vs. normal). Aorta GUSB activity increased to 195 ± 108 U/mg in RV-treated mice (Fig. 3C), which was 5.0-fold the value in normal mice (p<0.001), and was 325-fold the value in untreated MPS VII mice (p<0.001). Elevation of other lysosomal enzymes generally occurs in lysosomal storage diseases, and normalization of this elevation is a good biochemical marker of correction of disease. Indeed, IDUA activity (Fig. 3D) was 22-fold normal in MPS VII mice (p<0.001), and RV-treated mice had a reduction in IDUA activity to 15% of that found in untreated MPS VII mice (p<0.001), although it remained elevated at 3-fold normal (not significant vs. normal). Similarly, GAGs (Fig. 3E) were elevated in MPS VII mouse aortas to 111-fold normal (p = 0.01), and were reduced in RV-treated aortas to 5% of the value in untreated MPS VII mice (p = 0.04), although they remained 6-fold normal. The failure to achieve complete biochemical correction is likely responsible for the fact that RV-treated MPS VII mice had modest dilatation of the aorta at 10 months at 2.2 ± 0.5 mm, which was 155% (p = 0.01) of the value of 1.4 ± .02 mm in the normals (Fig. 3B), demonstrating that gene therapy was not fully effective at preventing aortic disease long term.
Fig. 3.
Effects of gene therapy on aortic abnormalities. Some MPS VII mice were injected IV with 1 × 1010 transducing units/kg of the RV designated hAAT-cGUSB-WPRE at 2–3 days after birth, while other mice were untreated. A. Serum GUSB activity. GUSB activity was determined at 2 to 6 months of age for normal, untreated MPS VII, and RV-treated MPS VII mice for the indicated number of animals. B. Aortic width. Aortas were dissected and the width was measured at 6 months of age for normal and MPS VII mice, and at 6 months or 10 months as indicated for RV-treated MPS VII mice. C and D. Aorta GUSB and IDUA activity. Extracts prepared at pH 5.5 were tested for GUSB and IDUA activity. E. GAG levels. Extracts prepared at pH 7.5 were tested for GAG levels. Values were compared using ANOVA with Tukey post-hoc analysis, and * represents p ≤ 0.05 and ** represents p ≤ 0.01 as indicated above the bars for comparison with values in untreated MPS VII mice.
3.4. Cathepsin enzyme activity in aortas
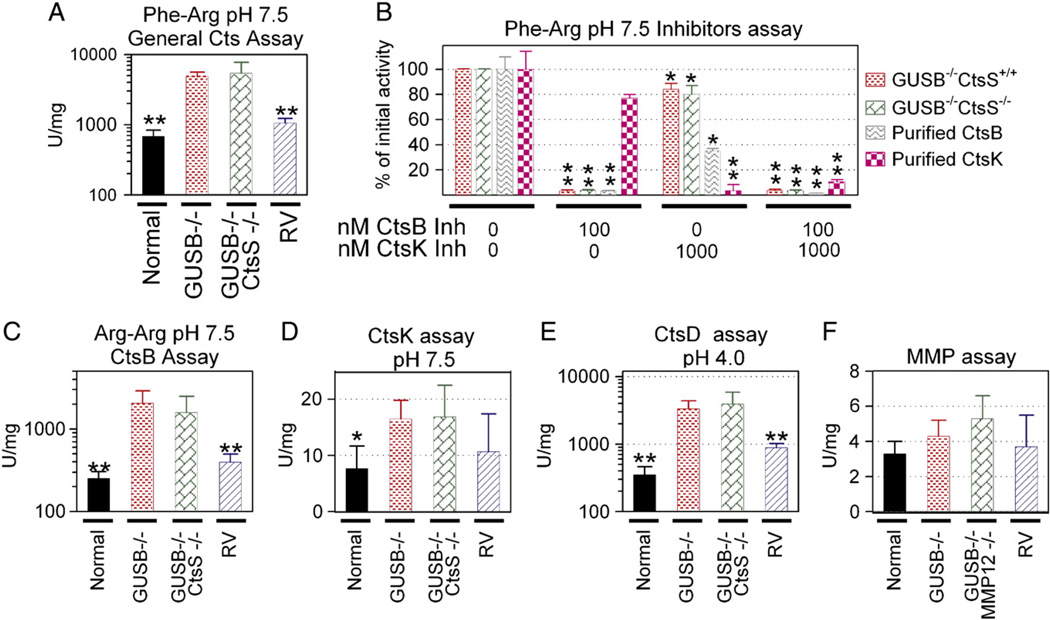
We focused on the role of CtsS in aortic dilatation despite the fact that mRNAs for other cysteine cathepsins were elevated in MPS VII aortas. This was because CtsS was reported to be the only cysteine cathepsin that was active at neutral pH [40,41], which is the pH expected outside of the cell where elastin resides, and a CtsS inhibitor markedly reduced enzyme activity in MPS I mice [10]. The failure of CtsS deficiency to protect MPS VII mice from aortic dilatation led us to test the assumption that other cathepsins are inactive at neutral pH. Supplementary Fig. 1 shows the activity of purified cathepsins against different substrates and inhibitors at pH 7.5. CtsB, CtsH, CtsK, and CtsS maintained activity at pH 7.5 against the fluorogenic peptide Z-Phe-Arg-AMC that can be cleaved by all enzymes at lower pH, although CtsL was inactive at pH 7.5. This result differs from the literature, which suggested that CtsB and CtsK were inactive at neutral pH [40,41]. The CtsB inhibitor was reasonably specific for CtsB, as 96% of the activity was inhibited at 100 nM, while CtsH and CtsK activity were at most modestly reduced at this concentration. The CtsK-specific inhibitor was relatively specific for CtsK, although it had modest activity against CtsB (50% inhibition) at 1000 nM. However, the CtsS inhibitor was quite promiscuous, inhibiting CtsB, CtsK, and CtsL almost as effectively as CtsS.
The ability of extracts from aortas of MPS VII mice with or without deficiency of CtsS to cleave cathepsin substrates is shown in Fig. 4. Extracts from GUSB−/− CtsS+/+ aortas had high activity against Z-Phe-Arg-AMC at pH 7.5 (4838 ± 777 U/mg), which was 7-fold the value in normal mice of 678 ± 159 U/mg (p = 0.01), as shown in Fig. 4A. Surprisingly, extracts from GUSB−/− CtsS−/− mice also had very high activity against Z-Phe-Arg-AMC at pH 7.5 at 5377 ± 2354 U/mg, which was 111% of the value in purebred MPS VII mice.
Fig. 4.
Cathepsins and MMP assays. Aortas from normal mice, GUSB−/− CtsS+/+ mice (labeled GUSB−/− on the figure), GUSB−/− CtsS−/− mice, or RV-treated MPS VII mice (RV) were homogenized at pH 5.5 for cathepsin assays, while aortas from normal mice, GUSB−/− MMP12+/+ mice (labeled GUSB−/− on figure), GUSB−/− MMP12−/− mice, or RV-treated MPS VII mice (RV) were homogenized at pH 7.5 for the MMP assay. A. Total cathepsin activity at pH 7.5. Samples were incubated with the non-specific substrate Z-Phe-Arg-AMC (Phe-Arg) at pH 7.5, and the activity in U/mg determined. B. Effect of inhibitors on activity against Z-Phe-Arg at pH 7.5. Samples from GUSB−/− CtsS+/+ mice, samples from GUSB−/− CtsS−/− mice with dilated aortas, purified CtsB (0.15 U), or purified CtsK (0.15 U) were incubated with the non-specific Z-Phe-Arg-AMC substrate with and without inhibitors of CtsB and CtsK at the indicated concentration, and the activity relative to that present in samples that did not receive an inhibitor was determined. C. CtsB activity at pH 7.5. Samples were incubated with the CtsB-specific substrate Z-Arg-Arg-AMC (Arg-Arg) at pH 7.5. D. CtsK activity at pH 7.5. Samples were incubated with a CtsK-specific substrate. E. CtsD activity at pH 4.0. Activity for CtsD was determined using a CtsD substrate. F. MMP activity. MMP activity was determined using a commercial kit. For panel B, values in samples that were treated with an inhibitor were compared with values for samples that did not receive the inhibitor using the Student's t test. For other panels, values in other groups were compared with values in untreated MPS VII mice using ANOVA with Tukey post-hoc analysis. * represents p ≤ 0.05 and ** represents p ≤ 0.01.
To further investigate the identity of the cathepsin responsible for the activity in extracts from GUSB−/− CtsS−/− mice, specific inhibitors of cathepsins and other substrates were used. Fig. 4B demonstrates that the CtsB inhibitor reduced overall activity of both GUSB−/−Cts+/+ and GUSB−/−CtsS−/− extracts vs. Z-Phe-Arg-AMC by 95%, suggesting that CtsB was the major cathepsin responsible for activity, while the CtsK inhibitor had a modest effect, which could be due to some activity against CtsB (see Supplementary Fig. 1). Z-Arg-Arg-AMC is a substrate that is specific for CtsB, as shown in Supplementary Fig. 1. As shown in Fig. 4C, aortic extracts from GUSB−/− Cts+/+ and GUSB−/− CtsS−/− had 2049 ± 847 and 1580 ± 898 U/mg vs. Z-Arg-Arg-AMC, which were 8-fold and 6-fold, respectively, the value of 251 ± 54 in normal aortas. The lower activity of these extracts against Z-Arg-Arg-AMC as compared with Z-Phe-Arg-AMC is consistent with the fact that purified CtsB has only 61% as much activity against Z-Arg-Arg-AMC as against Z-Phe-Arg-AMC.
The data shown above suggest that CtsB is the major enzyme responsible for cathepsin activity in MPS VII mice. It is possible, however, that some other cathepsin is active at neutral pH, and that this is difficult to detect due to the high CtsB activity. CtsK is a very important enzyme, as it maintains activity at pH 7.5 (Supplementary Fig. 1), it has elastin-degrading activity [42], and is activated by chondroitin sulfate [43], which is one of the GAGs that accumulates in MPS VII. A CtsK substrate was obtained that was not cleaved by CtsB or CtsS to an appreciable extent, although it was cleaved by CtsL 1% as efficiently as CtsK (data not shown). Activity with this CtsK substrate was elevated in GUSB−/−Cts+/+ or in GUSB−/− CtsS−/− mice to 16.5 ± 3.3 U/mg and 16.9 ± 5.6 U/mg, respectively, which was 2.2-fold the value of 7.4 ± 4 U/mg in normal mice (Fig. 4D). Although activity was significantly elevated in the MPS VII mice (p = 0.019 vs. normal), the CtsK activity was <0.1% of the CtsB activity on a U/mg basis.
CtsD is an aspartyl protease that can activate other cathepsins such as CtsB [44], although it has no activity against substrates that are cleaved by cysteine cathepsins. CtsD activity at pH 4.0 (Fig. 4E) was 9.5- and 11.2-fold normal in GUSB−/−CtsS+/+ and GUSB−/− CtsS−/− mice, respectively, suggesting a mechanism for activation of CtsB.
We conclude that CtsS does not play an important role in aortic dilatation in MPS VII, and that the major cysteine cathepsin that is upregulated is CtsB, which is generally believed to have low, albeit detectable, activity against elastin [50]. Although there was some increase in CtsK activity, which is a known elastase, this activity was very low. It was difficult to assess CtsH activity, as no specific assay was identified.
3.5. MMP12 activity
MMP12 was a good candidate for an enzyme that could degrade elastin in MPS VII aortas, as it is a known elastase, its mRNA was 7-fold normal at 3 months in MPS VII mice, its enzyme activity was elevated in MPS I mouse aortas [10] and in MPS I and MPS VII dog aortas [14], and its deficiency reduced aortic dilatation in one model [24]. Ascending aorta extracts from MPS VII mice were tested for MMP12 activity using a fluorogenic substrate that can be cleaved by MMP12. This demonstrated that MMP12 activity was not elevated in MPS VII mice at 6 months of age (Fig. 4F). This substrate can be cleaved by other MMPs, as demonstrated by the fact that extracts from MMP12-deficient mice still had activity in this assay. The failure of MMP12 deficiency to prevent dilatation of MPS VII aortas demonstrates that this enzyme does not play a major role in this model.
3.6. RNA analysis in MPS VII mice
The above studies demonstrate that deficiency of both CtsS and MMP12 cannot prevent aortic dilatation in MPS VII mice. We therefore used microarray analysis of RNA from the aorta of normal mice (GUSB+/− CtsS+/+ MMP12+/+) and MPS VII mice with aortic dilatation (GUSB−/− CtsS+/+ MMP12+/+) to attempt to identify other genes whose protein products might contribute to aortic dilatation. Samples were isolated at 6 months of age, when aortic disease was well-established. Table 1 summarizes the genes in various categories whose expression was upregulated (≥2.5-fold normal) or down-regulated (≤0.4-fold normal) in a statistically significant fashion (p value ≤0.01) after comparison of values in normal with those in MPS VII mice. Supplementary Table 2 shows values for all up- and down-regulated genes, which are sorted into different categories. In addition, all microarray data were uploaded to the GEO microarray data base (GSE30657), and values for genes whose expression was not significantly altered can be found there. Some categories of genes are discussed below, while others simply appear in Table 1 or Supplementary Table 2. Finally, a list of all elastases that were interrogated is in Supplementary Table 3, while a list of all complement components that were interrogated is in Supplementary Table 4.
Table 1.
Genes differentially expressed in the MPS VII aorta. RNA was extracted from normal (N = 3) or MPS VII (N = 3) aortas at 6 months of age and a microarray was performed. Genes were considered significantly upregulated (≥2.5-fold Normal) or downregulated (≤0.4-fold Normal) when p was <0.01. The gene symbol is shown here. Supplementary Table 2 shows the gene symbol, the ratio of the signal in MPS VII to normal mice, the signal for each probe for normal mice, and the p value for comparison of MPS VII with normal mice.
| Classification | Upregulated (>2.5-fold normal) | Downregulated (<0.4-fold normal) |
|---|---|---|
| Extracellular matrix proteins | Spp1, Reln, Fbln2, Tnc | Vit, Tnxb, Cyr61, Npnt, Pcolce2, Omd, Optc |
| Proteases | Ctsl, Dpep2, Mmp3, Mmp12, Lgmn, Napsa, Ctsk, Ctsa, Cpxm2 | Dpp10, Phex |
| Protease Inhibitors | Fetub, Timp4 | Serpine2, Wfdc1 |
| Complement system | C1qc, C1qb, C1qa, CFD, C4, Fcna, C2 | None |
| Lectins | Clec4d, Clec4n, Lgals3, CD83, Mrc1 | Clec3b |
| Toll-like receptor and TNFα signaling | Trem2, Tlr13, Tnfaip2, Tyrobp, CD14, Lcp1, Tnfaip8l2, Litaf | None |
| Fc Immunoglobulin receptors | Ms4a7, Fcgr4, Fcgr3, Fcgr2b, Fcer1g, Msr2, Lilrb4 | None |
| Chemokines or growth factors | Ccl21c, Igfbp2, Ccl4, Igfbp3, Cxcr4, Pdgfb, Ngfb, Cx3cl1, Agtr1a, Il10ra, Cxcl16, Igf1 | Igf2, Ltbl4, Bmp3, Cytl1, Bmpr1b |
| Kinases or kinase binders | Rps6ka1, Tec, Btk, Pbk, Fes, Ptk2b | Prkaa2 |
| Phosphatases | Ptpre, Inpp5d | Pitpnm3, Ppm1e, Dusp8, Dusp1, Ppp1r1b |
| G protein-associated proteins | Gpr176, Gpr109a, Ednrb, Gpr34, Gpr81, Gng2, Cnr2, Gpr97 | Ngef, Rgs4 |
| Cytoskeletal proteins, integrins, cell adhesion | Lpxn, Myo1f, Nckap1l, Myo7a, Emcn, Vcam1, Icam1, Unc93b1, Cotl1, Arhgdib, Rai14, Svep1, Gja4, Arhgap25, Vill, Itgb7, Pcdh1, Lasp1, Tpm3, Cldn5, Thbs1, Arhgap22, Was, Arhgap30 | Cnn1, Pdlim4, Pdlim3, Slmap, Scin, Wasf1, Mcam, Tmem47, Myoz2, Thsd4, Unc5c, Sema5a, Dtna, Tppp3, Sgca, Xirp1, Des, Amigo2, Bves, Actg2, Tcap |
| Wnt pathway | Sprf1, Wisp1, Wisp2 | Wif1, Cxxc4 |
| Lipid metabolism | Pld3, Ptgds, Pld4, Agpat2, | Dgkg |
| Transcription factors | Cebpa, Elk3 | Crtc3, Creb5, Zfp281, Id1, Myocd, Atf3, Fos |
| Miscellaneous signal transduction proteins | Gpnmb, Lat2, Ccnd1, Rerg, Xlr4a, Emr1, Cd84, Ly9, Havcr2, Nrn1, Ly86, Sirpa, CD52, H2-Ab1, Ncf4, Insig1, CDC20, Cd74, H2-Eb1, Irs3, Fst | Gucy1a3, Retnla, Rab9b |
| Lysosomal or peroxisomal proteins | Cd68, Lyz2, Lyz, Laptm5, Atp6ap2 | Hyal1, Gusb |
| Proteoglycans | Sdc3 | Acan |
| Glycosynthesis-related enzymes or proteins in ER or Golgi | Chst1, Ugt1a10, Renbp, Mfng, Uap1l1, Man2a1, Gal3st1 | Pigz |
| Vesicle transport | Scg2, Stab1, Rftn1, Syngr1 | Syngr3, Rtn2, Cav3, Stx1a, Sorl1, Kalrn |
| Ubiquitination | None | Fbxl13, Fbxo30, Hspa1a |
| Neurodegeneration | Sncg | Npy1r |
| Apoptosis | Arl11, Cidec, Bid | None |
| DNA/RNA metabolism | Apobec1, Fus | Rbm8a, Hdac5, Brip1 |
| Metabolism | Cyp2e1, Car3, Ddah1, Cyp2f2, Hbb-b2, Trf, Apoe, Acaa2, Car5b, Htatip2, Mup2, Mgst1, Aldh3b1, Akr1c12, Hk3, Eno1, Cp, Chpt1, Cyp1b1, Cyp4f18, Tcn2, Pdhb, Idh1, Cryz | Bdh1, Stc2, Agpat5, Pgm5, Vldlr, Adh7, Noxo1 |
| Solute or water transporters | Slc15a3, Slc40a1, Slc11a1, Slco2a1, Abcc3, Slc7a10, Slc27a1, Slc13a4, Aqp7, Slc16a4 | None |
| Ion channels | Ttyh2, Hvcn1, Atp1b1, Fxyd6 | Atp1b2, Kcnk1, Kcnab1 |
| Miscellaneous function | None | Hspa2, Olfml2b, Hspa1l |
| Unclear function | MsA46d, Evi2a, Tmem176b, Art4, Klhl6, Mlana, Lrrc33, | Tspan11, Stbd1, Crispld2, Fam171b, Vstm2b, Fam107a, Scube3 |
GeneGo software was used to determine if the changes in gene expression observed in MPS VII aortas resemble those that occur in other processes, networks, or diseases, using a level of upregulation of 2.5-fold normal, and a p value ≤0.01. The most upregulated process was the immune system process, with 62 genes upregulated out of 1617 genes that were put into this category by the software (not all of which are regulated at the transcriptional level), with a p value for significant upregulation of 2 × 10−27. The most upregulated process network was inflammation of the complement system, with 12 genes upregulated out of 73 genes that were put into this pathway, for a p value of 2 × 10−11. The most upregulated disease was rheumatoid arthritis, with 58 genes upregulated out of 941 that were placed in this category, for a p value of 6 × 10−25.
3.7. RNA for extracellular matrix proteins
Extracellular matrix protein mRNAs that were evaluated include elastin and collagen, genes that are necessary for their assembly, and miscellaneous extracellular matrix genes. Elastin mRNA was not altered, while the elastin-associated protein fibulin 2 (Fbln2) mRNA was modestly elevated at 3.1-fold normal in the MPS VII aorta, as shown in Table 1 and in Supplementary Table 2. Some extracellular matrix proteins were reduced. For example, mRNA for procollagen C-endopeptidase enhancer 2 (Pcolce2), which enhances processing of types I and II procollagens, was 0.3-fold normal. Finally, mRNA for optican (Optc), a protein that affects collagen fibrils in the eye, was markedly reduced at 0.15-fold normal. We conclude that elastin fragmentation was probably not due to downregulation of mRNA for elastin or elastin-associated proteins, but there were a few abnormalities in genes associated with collagen assembly.
3.8. RNA for elastases
A major hypothesis of this project was that a protease that can degrade elastin was upregulated in the aorta of MPS VII mice. According to GeneGo interactions software and the national library of medicine (http://www.ncbi.nlm.nih.gov/pubmed/) gene category, elastin can be cleaved by at least 28 different proteases. Supplementary Table 3 lists all known elastases that were interrogated on the microarray regardless of their expression level. mRNA was significantly higher in MPS VII than in normal mice for cathepsin L (6.2-fold), complement component D (CFD, 4.0-fold normal; 16,645 fluorescent units (FU/) spot in normal mice; p = 0.006), MMP12 (3.6-fold normal) and cathepsin K (CtsK; 2.8-fold normal). In addition, mRNAs for some other elastases were elevated on the microarray, but were not ≥2.5-fold normal or did not achieve a p value ≤ 0.01 for comparison of the 2 groups. These include MMP2, CtsS, CtsB, CtsH, and legumain (Lgmn), which is a cysteine protease that is induced by LPS whose substrates are poorly characterized. mRNA for several other elastases were not significantly altered, as summarized in Supplementary Table 3.
3.9. RNA for genes of the complement system
Complement components play important roles in the innate and the acquired immune response, and activation of the complement pathway can result in upregulation of destructive proteases. As noted above, the process network that most resembles the changes in gene expression observed in MPS VII aortas was the complement system, with a p value of 2 × 10−11. In addition, there is one report that CFD (also called adipsin or endogenous vascular elastase) can cleave elastin [45], and CFD were very abundant in the normal aorta and elevated to 4.0-fold normal in MPS VII mice (p = 0.006). Genes of the complement system that were upregulated on the microarray are shown on Table 1 and on page 2 of Supplementary Table 2. In addition, values for all complement genes that were interrogated on the microarray are shown in Supplementary Table 4.
Activation of the complement system may occur by 3 different pathways [46]. The classical pathway involves binding of IgG or IgM to a complex containing the C1 complex (containing C1q, C1r, and C1s), which can then cleave C4 and then C2 to generate C3 convertase, which in turn can activate C3 to C3a and C3b by proteolytic cleavage. C1 (6.1-fold normal for the most upregulated component), C4 (3.4-fold normal), and C2 (2.5-fold normal) were all upregulated in MPS VII aortas. A second pathway of complement activation is the alternative pathway, which can be initiated by spontaneous decay of C3 to C3a and C3b, or by generation of C3b from other pathways. This involves cleavage of complement component B of a C3bB complex by CFD, after which the C3bBb complex can cleave additional C3, while properdin (CFP) stabilizes the C3bBb complex and protects it from regulation by complement inhibitors. CFD was elevated to 4-fold normal, while CFP was 1.9-fold normal. A third pathway for complement activation involves the lectin pathway, in which mannose-binding lectin (MBL) or a ficolin binds to carbohydrates such as mannose or N-acetyl glucosamine that results in activation of MBL-associated serine proteinase 1 (MASP1) or MASP2, which in turn activate complement. Ficolin A (FcnA) was 2.7-fold normal. Cleavage of C3 can result from all three pathways of complement, and generates an anaphylatoxin (C3a) and C3b. C3b can initiate formation of the membrane attack complex as well as generate another anaphylatoxin, C5a. Although mRNA for none of these downstream components or receptors for anaphylatoxins were upregulated in the MPS VII aorta, they were expressed, and thus poised to respond to upstream events. Of the complement inhibitors, complement factor H was reduced to 0.64-fold in MPS VII mice, while others were not significantly affected, and were generally expressed at fairly low levels except for CD59, which is an inhibitor of the late stages of complement.
3.10. Signal transduction molecules
As one hypothesis for the mechanism of disease in MPS VII is that GAGs bind to the TLR4 and induces inflammatory signals, genes of the TLR pathway or downstream signaling molecules are shown in Supplementary Table 2. Although TLR4 was not elevated in MPS VII mice, it was moderately abundant, and thus could respond to GAGs. In addition, mRNA for proteins CD14, which associates with the TLR4 was 4.0-fold normal. Finally, genes that are upregulated by TLR signaling such as TREM2 (10.8-fold normal) and its associated protein, Tyrobp (DAP12 = 4.1-fold normal) were elevated, as were several other proteins that are known to be induced by TLR or TNFα signaling.
Receptors for immunoglobulins can act synergistically with TLR receptors or with complement in signal transduction, and can activate many of the same downstream molecules [47]. Many mRNAs for genes that encode Fc receptors were markedly elevated, as shown in Supplementary Table 2. Fc receptors were also elevated in MPS VI synovial cells [3].
Complement receptors, TLRs, and Fc receptors can all result in alteration of mRNA for cytokines, growth factors, intracellular signal transduction proteins, or proteins that bind to these proteins. Genes for such proteins whose expression was altered in MPS VII mouse aorta are shown in Supplementary Table 2. Ccl21 is the most highly upregulated cytokine at 35.5-fold normal, while the cytokine Cxcl4 (MIP-1β) was 10.3-fold normal, and is a gene that is upregulated in many models of MPS. The increase in angiotensin II receptor, type 1, a receptor for angiotensin II, to 2.9-fold normal, was of interest, as inhibitors of this receptor such as losartan can reduce elastin fragmentation in Marfan Syndrome mice [48]. Genes that were downregulated included latent transforming growth factor beta binding protein 4 (0.4-fold normal), bone morphogenetic protein 3 (BMP3; 0.3-fold normal), and BMP receptor, type IB (0.2-fold normal).
3.11. Immunohistochemsitry for C3
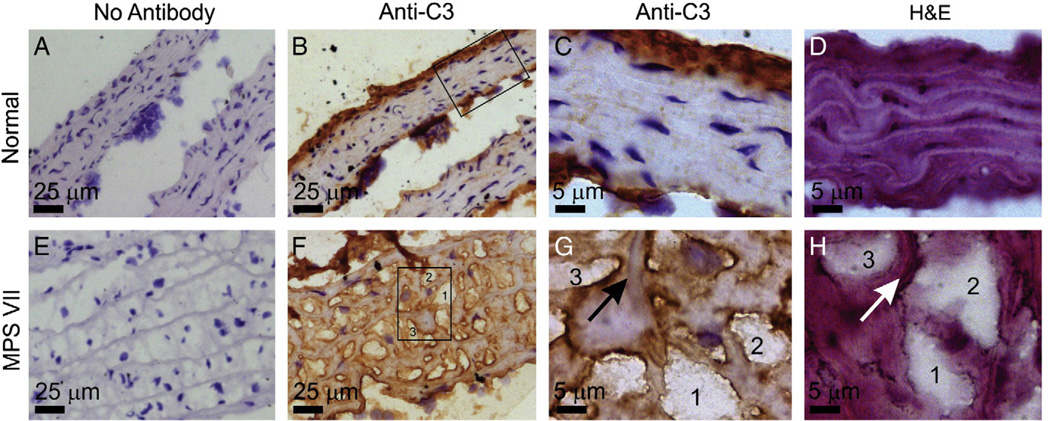
Aortas were tested for deposition of C3 (Fig. 5), since several genes of the complement system were upregulated in MPS VII aortas on the microarray. MPS VII aortas had a strong positive signal in the media, which was localized at the edge of GAG deposits and to a lesser extent along the edge of elastin fibers. Although normal mice had some C3 deposition in the intima and adventitia, there was little signal in the media. These data confirmed that the complement system was activated in the MPS VII aortas, and suggest that it occurs at sites of GAG deposits.
Fig. 5.
Immunostain for C3. Aortas were from a normal (panels A–D) and an MPS VII (panels E–H) aorta isolated at 6 months. Frozen sections underwent immunostaining without the addition of the anti-C3 first antibody (panels A and E; No Antibody), or with the addition of the anti-C3 first antibody (panels B–C and F–G; Anti-C3) as detailed in the Materials and methods section, and were counterstained with hematoxylin. Panels D and H received hematoxylin and eosin staining only (H&E). The boxes in panels B and F are the regions that are shown at higher power in panels C and G, respectively. Panels G and H represent adjacent sections of the same region of the MPS VII aorta, where 3 separate deposits of GAGs are numbered 1, 2, or 3, and the arrow identifies an elastin fiber. The brown color indicates a positive signal for C3, and nuclei appear blue. Size bars are indicated in each panel. The adventitia is at the top of all low magnification panels.
3.12. Real-time PCR for complement genes
Real-time reverse transcriptase analysis of expression of complement genes (Fig. 6) confirmed elevation of genes in MPS VII aortas that were found on the microarray. For example, CFD was elevated at 34.6 ± 27.3-fold normal and was very abundant at 4.5-fold the level of β-actin, while properdin was 3.7 ± 2.4-fold normal. In addition, there was upregulation of mRNA for genes related to the classical pathway such as C1qa (11.5 ± 2.4-fold normal), C2 (6.4 ± 2.7-fold normal), C4 (11.1 ± 8.2-fold normal), and the lectin pathway such as FcnA (9.5 ± 5.4-fold normal), MASP1 (0.01% β-actin; not detectable in normal) and MASP2 (0.3% β-actin; not detectable in normal). Furthermore, genes related to downstream events of complement pathways were also elevated in MPS VII aortas, including C3 (4.0 ± 2.5-fold normal) and C5 (0.005% β-actin signal; undetectable in normal). Finally, regulators of complement were either significantly reduced (CFH) or moderately increased (CFI, CD55) in MPS VII as compared with normal mice.
Fig. 6.
mRNA for complement genes. RNA was extracted from normal (N = 4) and MPS VII (N = 6) mice at 6 months of age and used for reverse transcriptase real-time PCR to assess complement gene mRNA levels. Results are shown as the percentage of β-actin. Genes with expression lower than 0.002% of β-actin were considered not present and are shown as 0.002% in the figure. *p<0.05 or **p<0.01 for comparison of values in MPS VII with those in normal mice using Student's t test. Abbreviations include Fcna for ficolin A, Fcnb for ficolin B, MBL for mannose-binding lectin, MASP for mannan-binding lectin associated serine protease, CD55 for decay accelerating factor, and CD59 for protectin.
4. Discussion
Aortic dilatation in MPS is important, as it will likely result in aortic dissection and possibly death as patients live longer after treatment with HSCT or ERT. Identification of the pathogenesis of elastin fragmentation might lead to the identification of a drug that would prevent this from occurring in patients. We favor the hypothesis that degradation of elastin is the most important mechanism responsible for elastin fragmentation, as MPS VII aortas had minimal amounts of lysosomal storage material, relatively normal elastin, and only minimal dilatation at 6 weeks of age, when elastin formation is believed to be largely completed [19]. Elastin fragmentation then developed in conjunction with progressive accumulation of lysosomal storage material, suggesting that degradation was involved. It remains possible that elastin assembly contributes to abnormal elastin structure, as proposed by Hinek et al. for MPS I [23]. The present work focused on the role of elastin, as collagen fibers were not overtly abnormal in MPS VII aortas (data not shown) or in MPS I or MPS VII aortas [14].
4.1. CtsS and MMP12 deficiency do not prevent elastin fragmentation in MPS VII aorta
A hypothesis of this project was that CtsS and/or MMP12 played pivotal roles in the elastin fragmentation that is likely responsible for the dilatation that occurs in MPS VII aortas. This hypothesis was clearly wrong, as deficiency of CtsS, MMP12, or both could not prevent aortic dilatation in MPS VII mice. In the case of CtsS deficiency, biochemical analyses demonstrated that the elevation in cysteine proteinase activity that we had previously attributed to CtsS in MPS I mice [10] and in MPS I and MPS VII dogs [14] was still present in GUSB−/− CtsS−/− mice, suggesting that another cathepsin was responsible. There are 11 lysosomal cysteine cathepsins, all of which are primarily destined for the lysosome but can also be secreted [49]. Further biochemical analyses demonstrated that this cathepsin activity in MPS VII aortas was primarily due to CtsB activity based upon efficient cleavage of a CtsB-specific substrate (Z-Arg-Arg-AMC) and inhibition with a CtsB-specific inhibitor at 100 nM. However, CtsB is believed to have relatively low activity against elastin [50], although it remains possible that the very high CtsB activity observed could have sufficient elastin-degrading activity to result in damage over time. One interesting feature was the fact that CtsB activity was markedly elevated at 8-fold normal, while CtsB mRNA was only 1.5-fold normal. It is possible that an activator of CtsB was upregulated, and indeed, the enzyme activity for the aspartyl protease CtsD that can activate CtsB by cleavage [44] was elevated to 10-fold normal. It is also possible that CtsK activity contributed to elastin fragmentation, as CtsK RNA and enzyme activity were 2.8- and 2.2-fold normal, respectively, and CtsK is known to be a potent elastase, although the activity appeared to be very low. Although CtsL mRNA was elevated at 6.2-fold normal, it is unlikely to be important, as the levels of RNA were very low, and CtsL is inactive at neutral pH. Although it is possible that CtsH contributes to elastin degradation, as its mRNA was 1.5-fold normal, we were unable to test its activity due to the absence of a specific assay. Finally, legumain is a poorly characterized cysteine protease whose mRNA was elevated to 2.9-fold normal.
MMP12 was clearly not necessary for aortic dilatation, as deficiency of MMP12 did not prevent aortic dilatation in MPS VII mice. This was likely due to the fact that MMP12 activity was only 1.5-fold normal in GUSB−/− mice at 6 months, and was not statistically different from values in normal mice, or from values in GUSB−/− MMP12−/− mice despite the fact that MMP12 mRNA was 6.8 ± 4.9-fold normal at 3 months, and was 3.6-fold normal at 6 months. This discrepancy may reflect the fact that MMP12 needs to activated by proteolytic cleavage. The matrix metalloproteinase (MMP) family has at least 20 members, of which MMP-2, -7, -8, -9, -10, -12, and -14 have elastase activity [51], and of which MMP2 and MMP8 can cleave the peptide that was used in our MMP12 assay. Thus, although MMP2 mRNA was modestly elevated at 2-fold normal in the microarray, it is unlikely that MMP2 contributes to elastin fragmentation, as upregulation of MMP2 enzyme activity should have been detected in this enzyme assay.
4.2. Possible role of another gene involved in aortic dilatation in MPS VII
An interesting feature of this study was the fact that some GUSB−/− CtsS+/+ MMP12+/+ mice that derived from breeding through the CtsS colony did not have dilated aortas. We hypothesize that this was due to an independently-segregating gene that initiated from the CtsS−/− colony that conferred protection from aortic dilatation when present in an autosomal recessive state. The CtsS−/− mice were generated in 129 mice whose subtype was not specified, and then backcrossed with C57BL/6 mice. Interestingly, 129/SvEv mice are less susceptible to formation of aortic aneurisms in one model of disease than are C57BL/6 mice [52], which is consistent with the presence of a gene that confers protection from aortic dilatation in 129/SvEv mice. Although there were no differences in cathepsin activity between GUSB−/−Cts+/+MMP12+/+ mice with dilated aortas and GUSB−/−Cts+/+MMP12+/+ with non-dilated aortas, MPS VII mice from the CtsS colony with unexpectedly-small aortas had reduced mRNA levels of cytokines as compared with MPS VII mice with dilated aortas (data not shown). We are currently trying to map the gene that reduces aortic dilatation in MPS VII mice and derives from the CtsS colony.
4.3. Possible role of complement in aortic dilatation
The absence of a major effect of CtsS and/or MMP12 deficiency on aortic dilatation in MPS VII mice led us to use microarray to look for other elastases that might play a role. CFD was intriguing, as it was very abundant in the microarray at 16,645 FU/spot in normal mice, and was elevated to 4.0-fold normal (60,202 FU/spot) in MPS VII aortas. Real-time reverse transcriptase PCR confirmed it to be elevated compared to normal (28-fold) and to be very abundant at 3.7-fold the level of β-actin. CFD was originally cloned as endogenous vascular elastase, a factor present in lung that could degrade elastin in a model of lung damage [45], and has also been cloned as adipsin, a gene expressed in fat cells. Although it was puzzling at the time that recombinant CFD did not cleave elastin, it is now clear that CFD needs to be activated by cleavage of 5 amino acids from the N-terminus, and that this cleavage is absolutely dependent on MASP1/3, an enzyme of the lectin pathway of complement [53,54]. CFD is very low in serum and abundant in adipocytes, but was not previously known to be expressed in aorta. Interestingly, we found that complement was strongly activated in aortas of MPS VII mice, as C3 was present at high levels on surfaces of the aorta media, although it is unclear if this occurs via the lectin, alternative, or classical pathway of complement. Analysis of mRNA with real-time RT-PCR demonstrates that several components were expressed in the aorta of MPS VII mice, many of which were increased as compared with normal mice. A role for complement proteins has previously been reported for the development of aneurisms in an elastase-injury model [55], while mRNA for complement genes was upregulated in the brains of MPS I and MPS III mice [56] and synovial cells of MPS VI rats [3]. Complement can be activated via the classical or the lectin pathways by carbohydrates [46], raising the possibility that the GAGs that accumulate in MPS VII directly activate complement.
4.4. Role of signal transduction in MPS VII aorta
This paper identifies several signal transduction pathways that are upregulated in MPS VII aortas, and may be potential targets for inhibition in the future. First, the JAK–STAT pathway appears to be activated by phosphorylation, as shown in Fig. 1, where STAT3 was phosphorylated at tyrosine 705 in MPS VII aortas. That could be due to a variety of pathways including the TLR4 pathway. Evidence for activation of the TLR4 pathway include the marked upregulation of osteopontin, TREM2 and its binding partner Tyrobp, as well as numerous other genes. There was a marked upregulation of several Fc receptors, and these are known to interact with TLR to augment signaling [47]. Finally, the complement pathway was clearly activated, as C3 was very abundant on the surface of cells in the MPS VII aorta. C3a and C5a, which are degradation products of C3 and C5, respectively, and are known to synergize with TLR4 in signal transduction [57].
4.5. Effects of gene therapy
MPS VII mice that received neonatal gene therapy with an RV vector expressing canine GUSB had normal aortic diameters and marked, but not complete, improvements in biochemical abnormalities at 6 months. However, some aortic dilatation was observed at 10 months, suggesting that gene therapy was not fully corrective, which likely reflects poor diffusion of GUSB in the interior of the relatively avascular aorta. We observed a similar outcome in the dog MPS VII model [14], which developed aortic dilatation at 5 years after neonatal gene therapy. These results highlight the importance of searching for the mechanisms responsible for the pathogenesis of aortic disease, as some ancillary therapy may be needed to prevent this manifestation.
4.6. Implications and further directions
These data demonstrate that CtsS and MMP12 are not essential for elastin fragmentation, and hence would not be good targets for drug inhibition in attempts to prevent aortic dilatation. A candidate for another elastase is CFD, as this is a known elastase, is very abundant, and there is evidence of complement activation in the MPS VII aortas. CtsB and CtsK are also candidates, although CtsB has low elastase activity, while CtsK levels were relatively low. It is also possible that there are other enzymes with elastase activity that are upregulated in the aorta. These studies also illustrate the activation of complement as well as other signal transduction pathways that are almost certainly critical for the upregulation and/or activation of destructive proteases, and may be targets for drug inhibition.
Supplementary Material
Acknowledgments
This work was supported by the MPS Society and the National Institutes of Health (HD061879 awarded to KPP, and R01 AI041592 and U19 AI070489 awarded to JPA). Histology was supported by P30 DK52574 from the NIH. GB received support from the Conselho Nacional de Desenvolvimento Cientifico-CNPg Brazil, grant number 200584/2010-3. We thank Robert Thompson and John Curci (Washington University St. Louis) for the generous gift of CtsS and MMP12 deficient mice, Paul W. Bigg for helpful comments on the manuscript, and Doug Tollefsen for help with enzyme assays.
Abbreviations
- MPS
mucopolysaccharidosis
- Cts
cathepsin
- MMP
matrix metalloproteinase
- GUSB
β-glucuronidase
- GAGs
glycosaminoglycans
- TLR4
toll-like receptor 4
- TNFα
tumor necrosis factor alpha
- IL-6
interleukin 6
- M6P
mannose 6-phosphate
- ERT
enzyme replacement therapy
- RV
retroviral vector
- PBS
phosphate buffered saline
- VVG
Verhoeff Van Gieson
- AMC
7-amino-4-methylcoumarin
- IDUA
α-l-iduronidase
- TBS
tris-buffered saline
- BP
blood pressure
- STAT3
signal transducer and activator of transcription 3
Footnotes
Supplementary materials related to this article can be found online at doi:10.1016/j.ymgme.2011.08.018.
References
- 1.Neufeld EF, Muenzer J. The mucopolysaccharidoses. In: Scriver CR, Beaudet AL, Sly WS, Valle D, editors. Metabolic and Molecular Basis of Inherited Disease. New York: McGraw Hill; 2001. pp. 3421–3452. [Google Scholar]
- 2.Ausseil J, Desmaris N, Bigou S, Attali R, Corbineau S, Vitry S, Parent M, Cheillan D, Fuller M, Maire I, Vanier MT, Heard JM. Early neurodegeneration progresses independently of microglial activation by heparan sulfate in the brain of mucopolysaccharidosis IIIB mice. PLoS One. 2008;3:1–11. doi: 10.1371/journal.pone.0002296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Simonaro CM, D'Angelo M, He X, Eliyahu E, Shtraizent N, Haskins ME, Schuchman EH. Mechanism of glycosaminoglycan-mediated bone and joint disease: implications for the mucopolysaccharidoses and other connective tissue diseases. Am. J. Pathol. 2008;172:112–122. doi: 10.2353/ajpath.2008.070564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Simonaro CM, Ge Y, Eliyahu E, He X, Jepsen KJ, Schuchman EH. Involvement of the Toll-like receptor 4 pathway and use of TNF-alpha antagonists for treatment of the mucopolysaccharidoses. Proc. Natl. Acad. Sci. U. S. A. 2010;107:222–227. doi: 10.1073/pnas.0912937107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Soliman OI, Timmermans RG, Nemes A, Vletter WB, Wilson JH, ten Cate FJ, Geleijnse ML. Cardiac abnormalities in adults with the attenuated form of mucopolysaccharidosis type I. J. Inherit. Metab. Dis. 2007;30:750–757. doi: 10.1007/s10545-007-0586-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Nemes A, Timmermans RG, Wilson JH, Soliman OI, Krenning BJ, ten Cate FJ, Geleijnse ML. The mild form of mucopolysaccharidosis type I (Scheie syndrome) is associated with increased ascending aortic stiffness. Hear. Vessel. 2008;23:108–111. doi: 10.1007/s00380-007-1013-x. [DOI] [PubMed] [Google Scholar]
- 7.Beaudet AL, Ferrante NM, Ferry GD, Nichols BL, Mullins CE. Variation in the phenotypic expression of beta-glucuronidase deficiency. J. Pediatr. 1975;86:388–394. doi: 10.1016/s0022-3476(75)80968-1. [DOI] [PubMed] [Google Scholar]
- 8.Liu Y, Xu L, Hennig AK, Kovacs A, Fu A, Chung S, Lee D, Wang B, Herati RS, Mosinger OJ, Cai S-R, Ponder KP. Liver-directed neonatal gene therapy prevents cardiac, bone, ear, and eye disease in mucopolysaccharidosis I mice. Mol. Ther. 2005;11:35–47. doi: 10.1016/j.ymthe.2004.08.027. [DOI] [PubMed] [Google Scholar]
- 9.Metcalf JA, Ma X, Linders B, Wu S, Schambach A, Ohlemiller KK, Kovacs A, Bigg M, He L, Tollefsen DM, Ponder KP. A self-inactivating gamma retroviral vector reduces manifestations of mucopolysaccharidosis I in mice. Mol. Ther. 2010;18:334–342. doi: 10.1038/mt.2009.236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ma X, Tittiger M, Knutsen RH, Kovacs A, Schaller L, Mecham RP, Ponder KP. Upregulation of elastase proteins results in aortic dilatation in mucopolysaccharidosis I mice. Mol. Genet. Metab. 2008;94:298–304. doi: 10.1016/j.ymgme.2008.03.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Woloszynek JC, Kovacs A, Ohlemiller KK, Roberts M, Sands MS. Metabolic adaptations to interrupted glycosaminoglycan recycling. J. Biol. Chem. 2009;23:29684–29691. doi: 10.1074/jbc.M109.020818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sleeper MM, Kusiak CM, Shofer FS, O'Donnell P, Bryan C, Ponder KP, Haskins ME. Clinical characterization of cardiovascular abnormalities associated with feline mucopolysaccharidosis I and VI. J. Inherit. Metab. Dis. 2008;31:424–431. doi: 10.1007/s10545-008-0821-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Traas AM, Wang P, Ma X, Tittiger M, Schaller L, O'donnell P, Sleeper MM, Vite C, Herati R, Aguirre GD, Haskins M, Ponder KP. Correction of clinical manifestations of canine mucopolysaccharidosis I with neonatal retroviral vector gene therapy. Mol. Ther. 2007;15:1423–1431. doi: 10.1038/sj.mt.6300201. [DOI] [PubMed] [Google Scholar]
- 14.Metcalf JA, Linders B, Wu S, Bigg P, O'Donnell P, Sleeper MM, Whyte MP, Haskins M, Ponder KP. Upregulation of elastase activity in aorta in mucopolysaccharidosis I and VII dogs may be due to increased cytokine expression. Mol. Genet. Metab. 2010;99:396–407. doi: 10.1016/j.ymgme.2009.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gompf RE, Shull RM, Breider MA, Scott JA, Constantopoulos GC. Cardiovascular changes after bone marrow transplantation in dogs with mucopolysaccharidosis I. Am. J. Vet. Res. 1990;51:2054–2060. [PubMed] [Google Scholar]
- 16.Sammarco C, Weil M, Just C, Weimelt S, Hasson C, O'Malley T, Evans SM, Wang P, Casal ML, Wolfe J, Haskins M. Effects of bone marrow transplantation on the cardiovascular abnormalities in canine mucopolysaccharidosis VII. Bone Marrow Transplant. 2000;25:1289–1297. doi: 10.1038/sj.bmt.1702448. [DOI] [PubMed] [Google Scholar]
- 17.Sleeper MM, Fornasari B, Ellinwood NM, Weil MA, Melniczek J, O'Malley TM, Sammarco CD, Xu L, Ponder KP, Haskins ME. Gene therapy ameliorates cardiovascular disease in dogs with mucopolysaccharidosis VII. Circulation. 2004;110:815–820. doi: 10.1161/01.CIR.0000138747.82487.4B. [DOI] [PubMed] [Google Scholar]
- 18.Cattell MA, Anderson JC, Hasleton PS. Age-related changes in amounts and concentrations of collagen and elastin in normotensive human thoracic aorta. Clin. Chim. Acta. 1996;245:73–84. doi: 10.1016/0009-8981(95)06174-6. [DOI] [PubMed] [Google Scholar]
- 19.Mithieux SM, Weiss AS. Elastin. Adv. Protein Chem. 2005;70:437–461. doi: 10.1016/S0065-3233(05)70013-9. [DOI] [PubMed] [Google Scholar]
- 20.Jordan MD, Zheng Y, Ryazantsev S, Rozengurt N, Roos KP, Neufeld EF. Cardiac manifestations in the mouse model of mucopolysaccharidosis I. Mol. Genet. Metab. 2005;86:233–243. doi: 10.1016/j.ymgme.2005.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Renteria VG, Ferrans VJ, Roberts WC. The heart in the hurler syndrome: gross, histologic and ultrastructural observations in five necropsy cases. Am. J. Cardiol. 1976;38:487–501. doi: 10.1016/0002-9149(76)90468-9. [DOI] [PubMed] [Google Scholar]
- 22.Braunlin E, Mackey-Bojack S, Panoskaltsis-Mortari A, Berry JM, McElmurry RT, Riddle M, Sun LY, Clarke LA, Tolar J, Blazar BR. Cardiac functional and histopathologic findings in humans and mice with mucopolysaccharidosis type I. Pediatr. Res. 2006;59:27–32. doi: 10.1203/01.pdr.0000190579.24054.39. [DOI] [PubMed] [Google Scholar]
- 23.Hinek A, Wilson SE. Impaired elastogenesis in Hurler disease: dermatan sulfate accumulation linked to deficiency in elastin-binding protein and elastic fiber assembly. Am. J. Pathol. 2000;156:925–938. doi: 10.1016/S0002-9440(10)64961-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Longo GM, Buda SJ, Fiotta N, Xiong W, Griener T, Shapiro S, Baxter BT. MMP-12 has a role in abdominal aortic aneurysms in mice. Surgery. 2005;137:457–462. doi: 10.1016/j.surg.2004.12.004. [DOI] [PubMed] [Google Scholar]
- 25.Sukhova Y-O, Zhang J-H, Pan GK, Wada Y, Yamamoto T, Naito M, Kodama T, Tsimikas S, Witztum JL, Lu ML, Sakara Y, Chin MT, Libby P, Shi GP. Deficiency of cathepsin S reduces atherosclerosis in LDL receptor-deficient mice. J. Clin. Invest. 2003;111:897–906. doi: 10.1172/JCI14915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Braunlin EA, Stauffer NR, Peters CH, Bass JL, Berry JM, Hopwood JJ, Krivit W. Usefulness of bone marrow transplantation in the Hurler syndrome. Am. J. Cardiol. 2003;92:882–886. doi: 10.1016/s0002-9149(03)00909-3. [DOI] [PubMed] [Google Scholar]
- 27.Birkenmeier EH, Barker JE, Vogler CA, Kyle JW, Sly WS, Gwynn B, Levy B, Pegors C. Increased life span and correction of metabolic defects in murine mucopolysaccharidosis type VII after syngeneic bone marrow transplantation. Blood. 1991;78:3081–3092. [PubMed] [Google Scholar]
- 28.Simonaro CM, Haskins ME, Kunieda T, Evans SM, Visser JW, Schuchman EH. Bone marrow transplantation in newborn rats with mucopolysaccharidosis type VI: biochemical, pathological, and clinical findings. Transplantation. 1997;63:1386–1393. doi: 10.1097/00007890-199705270-00003. [DOI] [PubMed] [Google Scholar]
- 29.Ellinwood NM, Colle MA, Weil MA, Casal ML, Vite CH, Wiemelt S, Hasson CW, O'Malley TM, He X, Prociuk U, Verot L, Melniczek JR, Lannon A, Aguirre GD, Knox VW, Evans SM, Vanier MT, Schuchman EH, Walkley SU, Haskins ME. Bone marrow transplantation for feline mucopolysaccharidosis I. Mol. Genet. Metab. 2007;91:239–250. doi: 10.1016/j.ymgme.2007.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sifuentes M, Doroshow R, Hoft R, Mason G, Walot I, Diament M, Okazaki S, Huff K, Cox GF, Swiedler SJ, Kakkis ED. A follow-up study of MPS I patients treated with laronidase enzyme replacement therapy for 6 years. Mol. Genet. Metab. 2007;90:171–180. doi: 10.1016/j.ymgme.2006.08.007. [DOI] [PubMed] [Google Scholar]
- 31.Braunlin EA, Berry JM, Whitley CB. Cardiac findings after enzyme replacement therapy for mucopolysaccharidosis type I. Am. J. Cardiol. 2006;98:416–418. doi: 10.1016/j.amjcard.2006.02.047. [DOI] [PubMed] [Google Scholar]
- 32.Auclair D, Hopwood JJ, Brooks DA, Lemontt JF, Crawley AC. Replacement therapy in mucopolysaccharidosis type VI: advantages of early onset of therapy. Mol. Genet. Metab. 2003;78:163–174. doi: 10.1016/s1096-7192(03)00007-6. [DOI] [PubMed] [Google Scholar]
- 33.Ponder KP, Haskins ME. Gene therapy for mucopolysaccharidosis. Expert. Opin. Biol. Ther. 2007;7:1333–1345. doi: 10.1517/14712598.7.9.1333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ponder KP, Melniczek JR, Xu L, Weil MA, O'Malley TM, O'Donnell PA, Knox VW, Aguirre GD, Mazrier H, Ellinwood NM, Sleeper M, Maguire AM, Volk SW, Mango RL, Zweigle J, Wolfe JH, Haskins ME. Therapeutic neonatal hepatic gene therapy in mucopolysaccharidosis VII dogs. Proc. Natl. Acad. Sci. U. S. A. 2002;99:13102–13107. doi: 10.1073/pnas.192353499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Xu L, Haskins ME, Melniczek JR, Gao C, Weil MA, O'Malley TM, O'Donnell PA, Mazrier H, Ellinwood NM, Zweigle J, Wolfe JH, Ponder KP. Transduction of hepatocytes after neonatal delivery of a Moloney murine leukemia virus based retroviral vector results in long-term expression of beta-glucuronidase in mucopolysaccharidosis VII dogs. Mol. Ther. 2002;5:141–153. doi: 10.1006/mthe.2002.0527. [DOI] [PubMed] [Google Scholar]
- 36.Shi GP, Sukhova GK, Kuzuya M, Ye Q, Du J, Zhang Y, Pan ML, Lu JH, Cheng XW, Iguchi A, Perrey S, Lee AM, Chapman HA, Libby P. Deficiency of the cysteine protease cathepsin S impairs microvessel growth. Circ. Res. 2003;92:493–500. doi: 10.1161/01.RES.0000060485.20318.96. [DOI] [PubMed] [Google Scholar]
- 37.Shipley JP, Wesselschmidt RL, Kobayashi DK, Ley TJ, Shapiro Sd. Metaloelastase is required for macrophage-mediated proteolysis and matrix invasion in mice. Proc. Natl. Acad. Sci. U. S. A. 1996;93:3942–3946. doi: 10.1073/pnas.93.9.3942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Xu L, Mango RL, Sands MS, Haskins ME, Ellinwood NM, Ponder KP. Evaluation of pathological manifestations of disease in mucopolysaccharidosis VII mice after neonatal hepatic gene therapy. Mol. Ther. 2002;6:745–758. doi: 10.1006/mthe.2002.0809. [DOI] [PubMed] [Google Scholar]
- 39.Wagenseil JE, Nerurkar NL, Knutsen RH, Okamoto RJ, Li DY, Mecham RP. Effects of elastin haploinsufficiency on the mechanical behavior of mouse arteries. Am. J. Physiol. Heart Circ. Physiol. 2005;289:H1209–H1217. doi: 10.1152/ajpheart.00046.2005. [DOI] [PubMed] [Google Scholar]
- 40.Jordans S, Jenko-Kokalj S, Kühl NM, Tedelind S, Sendt W, Brömme D, Turk D, Brix K. Monitoring compartment-specific substrate cleavage by cathepsins B, K, L, and S at physiological pH and redox conditions. BMC Biochem. 2009;10:23. doi: 10.1186/1471-2091-10-23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Vasiljeva O, Dolinar M, Pungercar JR, Turk V, Turk B. Recombinant human procathepsin S is capable of autocatalytic processing at neutral pH in the presence of glycosaminoglycans. FEBS Lett. 2005;579:1285–1290. doi: 10.1016/j.febslet.2004.12.093. [DOI] [PubMed] [Google Scholar]
- 42.Novinec M, Grass RN, Stark WJ, Turk V, Baici A, Lenarcic B. Interaction between human cathepsins K, L, and S and elastins: mechanism of elastinolysis and inhibition by macromolecular inhibitors. J. Biol. Chem. 2007;282:7893–7902. doi: 10.1074/jbc.M610107200. [DOI] [PubMed] [Google Scholar]
- 43.Li Z, Yasuda Y, Li W, Bogyo M, Katz N, Gordon RE, Fields GB, Brömme D. Regulation of collagenase activities of human cathepsins by glycosaminoglycans. J. Biol. Chem. 2004;279:5470–5479. doi: 10.1074/jbc.M310349200. [DOI] [PubMed] [Google Scholar]
- 44.van der Stappen JW, Williams AC, Maciewicz RA, Paraskeva C. Activation of cathepsin B, secreted by a colorectal cancer cell line requires low pH and is mediated by cathepsin D. Int. J. Cancer. 1996;67:547–554. doi: 10.1002/(SICI)1097-0215(19960807)67:4<547::AID-IJC14>3.0.CO;2-4. [DOI] [PubMed] [Google Scholar]
- 45.Zhu L, Wigle D, Hinek A, Kobayashi J, Ye C, Zuker M, Dodo H, Keeley FW, Rabinovitch M. The endogenous vascular elastase that governs development and progression of monocrotaline-induced pulmonary hypertension in rats is a novel enzyme related to the serine proteinase adipsin. J. Clin. Invest. 1994;94:1163–1171. doi: 10.1172/JCI117432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Speth C, Prodinger WM, Wurzner R, Stoiber H, Dierich MP. Complement. In: Paul William E., editor. Fundamental Immunology (0-7817-6519-6, 978-0-7817-6519-0) Wolters Kluwer/Lippincott Williams & Wilkins; 2008. pp. 1048–1107. [Google Scholar]
- 47.Ivashkiv LB. Cross-regulation of signaling by ITAM-associated receptors. Nat. Immunol. 2009;10:340–347. doi: 10.1038/ni.1706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Habashi JP, Doyle JJ, Holm TM, Aziz H, Schoenhoff F, Bedja D, Chen Y, Modiri AN, Judge DP, Dietz HC. Angiotensin II type 2 receptor signaling attenuates aortic aneurysm in mice through ERK antagonism. Science. 2011;332:361–365. doi: 10.1126/science.1192152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Bromme D, Wilson S. Role of cysteine cathepsins in extracellular proteolysis. In: Parks WC, Mecham RP, editors. Extracellular Matrix Degradation (Biology of Extracellular Matrix) New York: Springer; 2011. pp. 23–51. [Google Scholar]
- 50.Mason RW, Johnson DA, Barrett AJ, Chapman HA. Elastinolytic activity of human cathepsin L. Biochem. J. 1986;233:925–927. doi: 10.1042/bj2330925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Fingleton B. MMPs as therapeutic targets—still a viable option? Semin. Cell Dev. Biol. 2008;19:61–68. doi: 10.1016/j.semcdb.2007.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Thompson RW, Curci JA, Ennis D, Mao MB, Pagano TL, Pham CT. Pathophysiology of abdominal aortic aneurysms: insights from the elastase-induced model in mice with different genetic backgrounds. Ann. N. Y. Acad. Sci. 2006;1085:59–73. doi: 10.1196/annals.1383.029. [DOI] [PubMed] [Google Scholar]
- 53.Banda NK, Takahashi M, Levitt B, Glogowska M, Nicholas J, Takahashi K, Stahl GL, Fujita T, Arend WP, Holers VM. Essential role of complement mannose-binding lectin-associated serine proteases-1/3 in the murine collagen antibody-induced model of inflammatory arthritis. J. Immunol. 2010;185:5598–5606. doi: 10.4049/jimmunol.1001564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Takahashi M, Ishida Y, Iwaki D, Kanno K, Suzuki T, Endo Y, Homma Y, Fujita T. Essential role of mannose-binding lectin-associated serine protease-1 in activation of the complement factor D. J. Exp. Med. 2010;207:29–37. doi: 10.1084/jem.20090633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Pagano MB, Zhou HF, Ennis TL, Wu X, Lambris JD, Atkinson JP, Thompson RW, Hourcade DE, Pham CT. Complement-dependent neutrophil recruitment is critical for the development of elastase-induced abdominal aortic aneurysm. Circulation. 2009;119:1805–1813. doi: 10.1161/CIRCULATIONAHA.108.832972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Ohmi K, Greenberg DS, Rajavel KS, Ryazantsev S, Li HH, Neufeld EF. Activated microglia in cortex of mouse models of mucopolysaccharidoses I and IIIB. Proc. Natl. Acad. Sci. U. S. A. 2003;100:1902–1907. doi: 10.1073/pnas.252784899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Hajishengallis G, Lambris JD. Crosstalk pathways between Toll-like receptors and the complement system. Trends Immunol. 2010;31:154–163. doi: 10.1016/j.it.2010.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.