Abstract
Amateur choral singing is a common pastime and worthy of study, possibly conferring benefits to health and social behaviour. Participants might be expected to possess musical ability and share some behavioural characteristics. Polymorphisms in genes concerned with serotonergic neurotransmission are associated with both behaviour and musical aptitude. Those investigated previously include the variable number tandem repeats RS1, RS3 and AVR in the AVPR1A (arginine vasopressin receptor 1a) gene and STin2 in the SLC6A4 (solute carrier family 6 [neurotransmitter transporter, serotonin], member 4) gene, as well as the SLC6A4 promoter region polymorphism, 5-HTTLPR. We conducted a genetic association study on 523 participants to establish whether alleles at these polymorphisms occur more commonly in choral singers than in those not regularly participating in organised musical activity (non-musicians). We also analysed tagging single nucleotide polymorphisms (SNPs) for AVPR1A and SLC6A4 to determine whether other variants in these genes were associated with singer/non-musician status. At the STin2 polymorphism, overall association with singer/non-musician status was evident at P = 0.006. The 9-repeat (P = 0.04) and 12-repeat (P = 0.04) alleles were more common in singers and the 10-repeat allele less so (P = 0.009). Odds ratios were 0.73 (95% CI 0.57–0.94) for the 10-repeat allele and 2.47 (95% CI 0.88–6.94) for the rarer 9-repeat allele. No overall association was detected at P<0.05 between any other polymorphism and singer/non-musician status. Our null findings with respect to RS3, RS1 and AVR, polymorphisms associated with musical ability by other authors, suggest that choir membership may depend partly on factors other than musical ability. In a related musical project involving one participating choir, a new 40-part unaccompanied choral work, “Allele”, was composed and broadcast on national radio. In the piece, each singer's part incorporated their personal RS3 genotype.
Introduction
Choral singing is a common pastime which merits serious study. Singers in amateur choirs subjectively perceive physical, emotional, social and spiritual benefits [1]. Teenage American students involved in choirs or other non-sporting extracurricular activities have lower odds for alcohol use, binge drinking, marijuana use and vandalism than their counterparts whose activities are solely sport-related [2]. There is also evidence that singing may have biochemical effects. After singing, but not after listening to music, both positive affect and salivary secretory immunoglobulin A (S-IgA) are increased in choral singers [3]. In other contexts, S-IgA increases with relaxation [4] and decreases with stressful events [5].
Choral singing is a highly sociable activity. The genetics of social behaviour have long been the subject of extensive research and it now appears that some of the relevant genes and polymorphisms may influence musical ability as well. In a recent study, 298 individuals from 19 families were genotyped at pre-selected polymorphisms in five gene regions – AVPR1A, SLC6A4, TPH1, COMT and DRD2. All five genes code for proteins concerned with neurotransmission. The first three of these are involved in, or influenced by, the serotonergic system. Association was sought with the Karma Music Test and with the Seashore measures of musical perception, both formal tests of musical ability.
The associations with smallest P values were found between music test scores and AVPR1A haplotypes incorporating combinations of the highly variable promoter region VNTRs RS1, RS3 and the intronic AVR. A weak association was also detected between the Karma Music Test score and an SLC6A4 haplotype combining the intronic VNTR, STin2, and the promoter region 5-HTTLPR [6]. In another study, RS1 and RS3 polymorphisms also influenced the likelihood of an individual's involvement in creative dance - an association strengthened when conditional on polymorphisms in the regulatory regions of SLC6A4 [7].
The AVPR1A RS3 polymorphism has been studied for its associations with social, behavioural and personality traits [8]. Its length appears to influence pair-bonding [9] and altruism [10]. Polymorphisms in the SLC6A4 gene have been the subject of sustained interest in psychiatry, in particular the 5-HTTLPR polymorphism. This has been associated with depression [11], autism [12], obsessive compulsive disorder [13] and antidepressant response [14].
Given that AVPR1A and SLC6A4 polymorphisms apparently influence both social and musical traits, we believed choral singers represented a group which might usefully be studied further. Our hypothesis was that allelic variants at complex polymorphisms in AVPR1A and SLC6A4, associated with musical phenotypes by Ukkola et al. [6] and Bachner-Melman et al. [7], would be more common in choral singers than in non-musicians. We aimed to determine this with a gene association study. To capture genetic variability more fully than the earlier studies, we also compared genotypes between the two groups at a set of tagging polymorphisms for AVPR1A and SLC6A4.
Methods
Ethics statement
This study was approved by St. Thomas' Research Ethics Committee and written informed consent was obtained from all participants.
Data collection
We recruited 523 individuals, aged 16–90 years and of white ethnicity. Potential participants determined their ethnic eligibility by referring to our institution's diversity monitoring list. This includes 70 individual designations in five broader categories – “White”, “Black or black British”, “Asian or Asian British”, “Mixed background” or “Other ethnic groups”. Self identified ethnicity correlates well with ancient geographic ancestry, a major determinant of genetic structure [15].
The choral group (n = 262) comprised regular singers from nine different amateur choirs. All choirs required singers to audition for membership and were deemed by one of the authors (AM) to be of a moderate to high standard on the basis of repertoire, performance history and reputation. The non-musician group (n = 261) was recruited from among staff at Guy's and St. Thomas' Hospitals and the MRC Social, Genetic and Developmental Psychiatry Centre, as well as patients attending for outpatient surgery and their relatives. Additional participants in the non-musician group were recruited from doctors attending academic meetings in London and the south of England. Each participant in the non-musician group was required to confirm that they did not “participate regularly in any organised musical activity, including a choir, dance class, orchestra or rock band”.
For each participant recruited to the study, gender, date of birth and musical status (choral singer or non-musician) was recorded. Each participant provided cheek swabs for DNA analysis.
Genotyping
For SLC6A4 and its 1000 bp margins, we genotyped the same panel of polymorphisms as tested in a recent paper by our group [16] with one omission. The panel included a set of tagging SNPs, selected using the SNPTagger program (www.broad.mit.edu/mpg/tagger) run in Windows XP and the HAPMAP data on the CEPH CEU population with European ancestry (CEPH NCBI Build 35/UCSC hg17/May 2004 coordinates) [17]. Selection criteria were a minimal allele frequency of 5% in the white population and pairwise r2 = 0.8. The ten selected SNPs provided 92% coverage of the DNA sequence variation in the SLC6A4 gene. In the current study, as before, we genotyped the SLC6A4 promoter region 5-HTTLPR and the VNTR STin2 (intron 2). We omitted the VNTR StIn4 (intron 4), which we previously found to be the only other polymorphic SLC6A4 VNTR. This is because our earlier study showed it to be in tight linkage disequilibrium with neighbouring markers [16].
For the current study, we also applied the same tagging criteria to AVPR1A and its 1000 bp margins, generating a set of three SNPs. We also genotyped those AVPR1A repeat polymorphisms investigated by others in the context of musical phenotypes [6], [7], namely RS1, RS3 and AVR.
DNA was extracted from buccal swabs provided by participants as previously described [18]. SNPs in the AVPR1A and SLC6A4 genes were determined using Taqman SNP Genotyping Assays (Applied Biosystems) and analysed on a 7900HT Sequence Detection System (Applied Biosystems). The 5-HTTLPR and the rs25531 SNP were genotyped together using a two-stage method described previously [16]. The regions containing the other VNTRs were amplified by PCR using the following flanking oligonucleotide primer pairs: Stin2 forward 5′-FAM-GTCAGTATCACAGGCTGCGAG-3′ and reverse 5′-GTTTCTTTGTTCCTAGTCTTACGCCAGTG-3′; RS1 forward 5′-FAM-AGGGACTGGTTCTACAATCTG-3′ and reverse 5′-GTTTCTTACCTCTCAAGTTATGTT GGTG-3′; RS3 forward 5′-FAM-CCTGTAGAGATGTAAGTGCT-3′ and reverse 5′-GTTTCTTTCTGGAAGAGACTTAGATGG-3′; AVR forward 5′-FAM-ATCCCATGTCCGTCTGGAC-3′ and reverse 5′-FAM-ATCCCATGTCCGTCTGGAC-3′. The product sizes were determined by running on a 3130xl Genetic Analyzer (Applied Biosystems).
Statistical analysis
Statistical analysis was performed using UNPHASED [19] to test for association between choral singer/control status and SNPs, VNTRs and 5-HTTLPR. PLINK was used for quality control for SNPs, assessing completeness of genotyping, allele frequency and departures from Hardy-Weinberg equilibrium [20]. Testing for relatedness between study participants was not possible with the limited genotyping performed. Markers were analysed as single polymorphisms and in two-locus haplotypes within each gene. Across genes, specific tests of association of AVPR1A polymorphisms and two-locus haplotypes with singer/non-musician status, conditional on the 5-HTTLPR and STin2 VNTRs, were performed in an attempt to replicate previous findings [7]. At each VNTR, rare alleles (<5% frequency) were pooled with alleles of similar length but higher frequency to increase power to detect association (Results section, table 1). This pragmatic strategy is standard in analysing highly polymorphic VNTRs and predicated on the hypothesis that alleles with similar number of repeats at a given VNTR have common ancestry.
Table 1. Association of AVPR1A and SLC6A4 VNTRs with singer/non-musician status.
| VNTR/pooled alleles or repeats | Individual alleles | Choral singers (n = 261) Number (%) of alleles | Non-musicians (n = 258) Number (%) of alleles | Odds ratio (95% CI) | P value* |
| AVPR1A RS3 | |||||
| 1 | 320, 327,329, 331 | 47 (9) | 35 (7) | 1.35 (0.82–2.23) | 0.20 |
| 2 | 333 | 40 (8) | 43 (8) | 0.94 (0.57–1.54) | 0.65 |
| 3 | 335 | 128 (25) | 129 (25) | 1 | 0.77 |
| 4 | 337 | 128 (25) | 113 (22) | 1.14 (0.80–1.62) | 0.37 |
| 5 | 339 | 39 (7) | 57 (11) | 0.69 (0.43–1.11) | 0.04 |
| 6 | 341, 343 | 82 (16) | 79 (15) | 1.05 (0.71–1.55) | 0.92 |
| 7 | 345,347,349,350,352 | 54 (10) | 50 (10) | 1.09 (0.08–0.77) | 0.77 |
| Overall significance | 0.39 | ||||
| AVPR1A RS1 | |||||
| 1 | 309,313 | 64 (12) | 72 (14) | 0.88 (0.60–1.30) | 0.43 |
| 2 | 317 | 215 (41) | 214 (41) | 1 | 0.97 |
| 3 | 321 | 120 (23) | 128 (25) | 0.93 (0.68–1.28) | 0.51 |
| 4 | 325 | 44 (8) | 39 (8) | 1.12 (0.70–1.80) | 0.59 |
| 5 | 329, 333, 337, 341, 345 | 77 (15) | 63 (12) | 1.22 (0.83–1.78) | 0.22 |
| Overall significance | 0.66 | ||||
| AVPR1A AVR | |||||
| 1 | 211, 214, 215, 217 | 86 (17) | 82 (16) | 1.05 (0.74–1.50) | 0.76 |
| 2 | 218 | 61 (12) | 56 (11) | 1.09 (0.73–1.63) | 0.64 |
| 3 | 219 | 75 (15) | 75 (15) | 1.00 (0.70–1.44) | 0.98 |
| 4 | 221 | 255 (49) | 256 (50) | 1 | 0.90 |
| 5 | 223, 225, 226, 227 | 39 (8) | 45 (9) | 0.87 (0.55–1.38) | 0.48 |
| Overall significance | 0.95 | ||||
| SLC6A4 STin2 | |||||
| 9 | 14 (3) | 5 (1) | 2.47 (0.88–6.94) | 0.04 | |
| 10 | 176 (34) | 212 (41) | 0.73 (0.57–0.94) | 0.009 | |
| 12 | 330 (63) | 291 (56) | 1 | 0.04 | |
| Overall significance | 0.006 |
*The P value for each allele is derived from the test of association between choral singers/controls for that allele against all other alleles pooled.
In this exploratory study, we avoided the highly conservative Bonferroni correction [21], instead correcting for multiple testing within genes with SNPSpD. This method uses linkage disequilibrium between a set of SNPs to determine the number of independent tests [22]. For SLC6A4, 10 genotyped SNPs were equivalent to 5 independent tests. Adding the 2 VNTRs genotyped gives 7 tests and a p-value threshold for gene-wide significance of 0.05/7, or 0.0071. For AVPR1A, 3 SNPs contribute two independent tests. When these are combined with the 3 VNTRs, the p-value threshold for gene-wide significance is 0.05/5 = 0.01. Only those P values reaching these gene-wide thresholds can be interpreted as statistically significant. No correction for multiple testing across genes has been applied.
Power calculations [23] indicated that a sample size of 250 choral singers and 250 controls would provide power of 78% to detect a difference in allele frequency assuming each allele confers a two-fold increased chance of being a choral singer, for an allele frequency of 0.05 (at a significance level of 0.05). With a higher allele frequency of 0.1, power is 96%. At 0.2, power rises to 99% for the same two-fold increase, and is sufficient, at 84%, to detect a much lower odds ratio – 1.5 and 2.25 for hetero- and homozygotes respectively. A sample size of 500 also means that genotyping an SNP with an allele frequency of 0.1 in LD (r2 = 0.9) with the causal variant provides power of 92%. The range of theoretical allele frequencies in these calculations are broadly in keeping with actual frequencies previously reported for our genotyped polymorphisms [6], [7], [16]. The calculations suggest that the study was sufficiently well-powered to detect association in the proposed sample size, across a wide range of marker properties.
Results
After extraction, adequate DNA samples were available for 261 singers and 258 non-musicians. Of these participants, three failed to state full age and gender information. From the available data, mean age (SD) of the singers was 42 (15) years and 122∶139 (47%∶53%) were male∶female. Corresponding figures for the non-musicians were 40 (12) years and 117∶139 (45∶54%). Genotyping failure did not exceed 2% at any polymorphism except 5-HTTLPR (19%). For AVPR1A and SLC6A4, the designated gene-wide P value thresholds for significance were 0.01 and 0.0071 respectively (see Methods).
AVPR1A
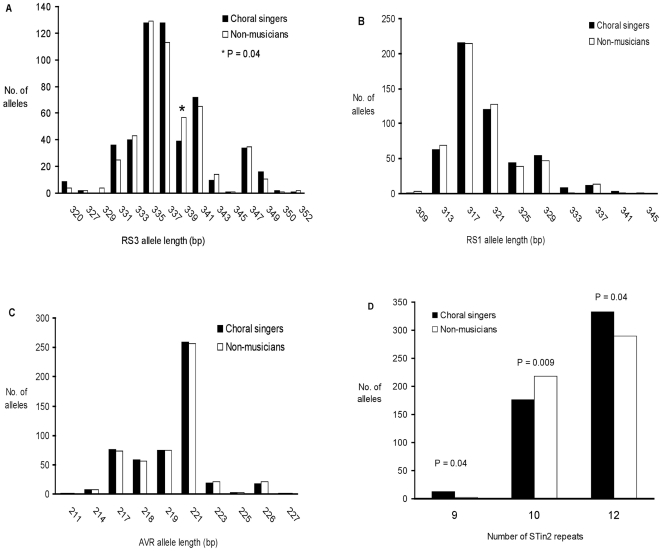
Allele frequencies at the RS3, RS1 and AVR VNTRs are illustrated in figure 1. Having pooled alleles of frequency <5%, no significant overall association of any VNTR with musical status was seen. Pooled allele frequencies for AVPR1A VNTRs, with estimates of individual allelic effects, are given in table 1. No significant effect of carrier status was observed for any allele. None of the VNTRs demonstrated a significant effect in logistic regression models incorporating a sex/musical status interaction term.
Figure 1. Allele frequencies in choral singers (n = 261) and non musicians (n = 258) for AVPR1A and SLC6A4 VNTRs. (A) RS3 (B) RS1 (C) AVR and (D) STin2.
All AVPR1A SNPs were in Hardy-Weinberg equilibrium (HWE). No SNP was significantly associated with musical status (table 2) and this finding was unaffected by the introduction of age in a logistic regression model.
Table 2. Association of AVPR1A and SLC6A4 SNPs, and 5 -HTTLPR, with singer/non-musician status.
| Gene/polymorphism | Location (basepairs) | Individual alleles (minor/major) | Minor allele frequency in choral singers (n = 261) | Minor allele frequency in non-musicians (n = 258) | Odds ratio (95% CI) | P value |
| AVPR1A | ||||||
| rs1042615 | 63544209 | A/G | 0.42 | 0.42 | 1.01 (0.79–1.29) | 0.93 |
| rs10877969 | 63547239 | C/T | 0.14 | 0.14 | 1.05 (0.74–1.50) | 0.78 |
| rs11174811 | 63540476 | A/C | 0.14 | 0.15 | 0.94 (0.67–1.33) | 0.73 |
| SLC6A4 | ||||||
| 5-HTTLPR | 28564498+ | (La+Lg)/(Sa+Sg)* | 0.42 | 0.40 | 1.10 (0.83–1.45) | 0.50 |
| rs2020933 | 28561755 | T/A | 0.05 | 0.06 | 0.81 (0.47–1.41) | 0.46 |
| rs2066713 | 28551665 | A/G | 0.37 | 0.42 | 0.81 (0.63–1.04) | 0.09 |
| rs2020939 | 28550732 | A/G | 0.44 | 0.39 | 1.24 (0.97–1.59) | 0.08 |
| rs8076005 | 28547210 | G/A | 0.20 | 0.20 | 1.02 (0.75–1.39) | 0.89 |
| rs2020942 | 28546914 | T/C | 0.37 | 0.42 | 0.81 (0.63–1.04) | 0.11 |
| rs140700 | 28543389 | T/C | 0.10 | 0.10 | 1.02 (0.69–1.53) | 0.90 |
| rs4583306 | 28538715 | G/A | 0.42 | 0.38 | 1.18 (0.92–1.51) | 0.19 |
| rs140701 | 28538532 | T/C | 0.42 | 0.38 | 1.19 (0.93–1.52) | 0.17 |
| rs4325622 | 28526475 | C/T | 0.47 | 0.43 | 1.15 (0.90–1.47) | 0.25 |
| rs3813034 | 28524804 | C/A | 0.46 | 0.43 | 1.14 (0.90–1.46) | 0.28 |
*At 5-HTTLPR long alleles La and Lg have been combined, as have short alleles Sa and Sg, to permit analysis as a bi-allelic marker.
Two-marker AVPR1A haplotypes, incorporating SNPs, VNTRs or one of each, showed no significant overall association with musical status, the lowest P value being 0.018 (for both RS3-rs2066713 and RS3-2020942 haplotypes).
SLC6A4
The rs25531 SNP (A/G) is contained within 5-HTTLPR (long/short) so that the combined assay used to characterise the region identifies four alleles; La, Lg, Sa, Sg. In our study, frequencies were low for Sg (0.2%) and Lg (6.6%). Consequently, statistical analysis was conducted only with respect to allele length, pooling La with Lg, and Sa with Sg to create a bi-allelic marker (L/S). Allele length was not associated with musical status (table 2).
The STin2 VNTR allele frequencies differed between the two groups. These, and the estimates of allele effects, are illustrated in figure 1 and table 1 respectively. There was an overall association with singer/non-musician status at this locus (P = 0.006). The 9-repeat (STin2.9, P = 0.04) and 12-repeat (STin2.12, P = 0.04) alleles were more common in choral singers and the 10-repeat allele (STin2.10) more common in non-musicians (P = 0.009). Odds ratios were 0.73 (95% CI 0.57–0.94) for the 10-repeat allele and 2.47 (95% CI 0.88–6.94) for the rarer 9-repeat allele compared to the baseline 12-repeat allele. For no haplotype incorporating STin2 and another locus, whether SNP or VNTR, did the additional locus decrease the P value for association below that of STin2 alone.
All ten SLC6A4 SNPs were in HWE in the non-musician group, though three deviated from HWE in the choral singer group (rs3813034, P = 0.025; rs4325622, P = 0.046; rs2020939, P = 0.024). None were significantly associated with musical status, either alone (table 2) or with age in a logistic regression model. Of all possible two-locus haplotypes within the SLC6A4 gene, incorporating SNPs alone or with 5-HTTLPR, none were significantly associated with musical status, the lowest P value being 0.038 for rs2020933-rs8076005.
Conditional associations
Of all AVPR1A markers and two-locus haplotypes, none showed an overall association with singer/non-musician status whether conditional on 5-HTTLPR or STin2, the lowest P value being 0.03 for RS1-RS3 conditional on 5-HTTLPR.
Discussion
Our hypothesis in this study was that allelic variants in SLC6A4 and AVPR1A genes, known to be associated with musical aptitude, would also be associated with a related behavioural outcome, namely choir membership. A priori power calculations indicated that our sample size would be adequate to detect effects on this outcome of our chosen polymorphisms, assuming broadly similar allele frequencies in our participants to those reported by other groups. This assumption proved correct. After statistical correction for multiple testing, we found that STin2.9 and STin2.12 alleles, in the SLC6A4 gene, were more common in choral singers than in non-musicians, and the STin2.10 allele less common.
In the same two groups, no overall difference was detected for allele frequencies in the AVPR1A gene at RSI, RS3 and AVR polymorphisms. These AVPR1A polymorphisms have been associated with performance in musical tests in a study which, to our knowledge, is the only one other than our own to address a possible association between STin2 and phenotypes related to music. In that study, Karma Music Test scores were associated with the haplotype 5-HTTLPR.La/STin2.12 [6].
Our allele frequencies for STin2 VNTR, a repeating 17-bp motif, were broadly in keeping with those in similar populations, with STin2.12 and STin2.10 being common alleles and STin2.9 a rare allele. We also found a single 11-repeat allele, a finding consistent with other studies [24]. We found no differences between groups in allele frequencies at 5-HTTLPR, the other SLC6A4 VNTR we studied. At 19%, the genotyping failure rate at this polymorphism was high but this may reflect the complex nature of the assay, including both the repeat and associated SNP.
Validation of our findings will require replication in an independent sample. Furthermore, while we attempted to select a study population of reasonable ethnic homogeneity, we made no correction for cryptic population structure in the association analysis. Failure to apply such correction is believed to be a factor contributing to false positive results in candidate gene studies like our own [25]. It was not feasible for us to conduct the additional genotyping typically employed for correction purposes in genome-wide association studies. Our results, then, must be regarded as preliminary ones that must be tested more robustly with the appropriate controls for population structure.
STin2 has been widely investigated in connection with psychiatric or neurological disease. Conflicting studies report associations between all three major STin2 alleles and depression-related phenotypes [26], [27], [28], [29]. Findings relating to psychotic symptoms in Alzheimer's patients are similarly inconsistent, with some studies implicating STin2.12 [30] and others STin2.10 [31]. STin2 alleles may [32] or may not [33] be associated with obsessive-compulsive disorders. The STin2.12 homozygote genotype has been associated with increased risk of schizophrenia [34], post stroke depression [35], migraine [36], [37], [38], a better response to drug treatment in depression [39], [40] and premature ejaculation [41] but a more inconsistent one to triptan therapy in migraine [42]. Haplotypes incorporating STin2 and the SLC6A4 5-HTTLPR polymorphism have been associated with sleep apnoea [43], postpartum depression [44] and attention deficit hyperactivity disorder [45].
The literature hints at some causative mechanisms behind these statistical associations. In a variety of experimental contexts, Stin2 appears to affect transcriptional regulation, the STin2.12 allele enhancing gene expression more than Stin2.10 [46], [47], [48]. Some haplotypes combining STin2 and 5-HTTLPR alleles also appear to influence transcriptional regulation by the transcription factor, CCTC-binding factor [49].
Relationships between these in vitro phenomena and clinical biochemical effects are also suggested by recent studies. In autistic individuals, for example, the STin2.10 allele is found more commonly in those with hyperserotonemia than in those with normal 5-HT [50]. Among male Han Chinese patients with obstructive sleep apnoea syndrome, those with a specific haplotype incorporating STin2.12 have lower plasma 5-HT level and 5-hydroxyindolacetic acid (5-HIAA) levels compared with non- STin2.12 carriers [43]. 5-HT availability, assessed by single photon emission computed tomography, is reduced in suicide attempters with a STin2.12 allele [51].
The influence of STin2 on personality in healthy individuals, such as the participants in this study, has also been investigated. There is some evidence of an association between the polymorphism and reward dependence or cooperation [52]. Individuals with the STin2.10 allele appear to be more risk tolerant to financial losses than those with the Stin2.12 allele [53]. The STin2.10 allele is also associated with lower neuroticism scores, as measured by the Temperament and Character Inventory, and lower harm avoidance scores in the Eysenck Personality Inventory [54].
Choir membership represents a complex behavioural phenotype and the two genes we investigated affect both musical ability and behaviour. As a single response from each participant in our study determined phenotype designation, “choir member” or “non-musician”, we cannot say whether the difference in allele frequencies between the groups reflects STin2 effects on musical ability or on some other behavioural trait.
Regarding musical ability, one would expect choral singers to share many genetic characteristics with those scoring highly in objective tests of musical aptitude. The fact that AVPR1A “musical genotypes”, identified with more certainty than STin2 by other authors, were similarly distributed in our choral singers and non-musicians may relate to our phenotype selection. Practical considerations mean that we were obliged to study amateur choral singers, albeit those at a very high level. Had we recruited professional singers for our choral singer group, they might have had a higher frequency of musical genotypes. Similarly, non-musicians identified themselves solely by reference to non-participation in musical activities. Some individuals in this group may merely have been deprived of relevant opportunities and were, in fact, musical but ignorant of the fact. If we had used formal music tests to select our two groups, the phenotypic distribution for musical ability in our study might have been closer to that of other studies.
As far as behavioural traits are concerned, membership of a successful choir entails many qualities in addition to, and perhaps for some individuals instead of, those assessed by formal musical tests. It is possible that these qualities are the ones associated with the STin2 polymorphism in our singers, rather than musical ability. Leaving aside a good voice, behavioural characteristics required of choir members include enthusiasm, concentration and attention to detail. A choral singer must also be able to modulate his/her own voice in response to the overall sound of the choir and to understand non-verbal cues from the conductor. The STin2 effect we observed may relate to one of these characteristics, rather than musical ability. If so, one might expect the STin2 allele frequency distribution to differ between choral singers and other musicians for whom some of these skills are arguably less important, such as conductors or solo instrumentalists.
Social behaviour that is not choir specific may also account for the STin2 effect we observed. The polymorphism may contribute to a “predisposed to group activity” phenotype. Our non-musicians were recruited individually at one-off educational meetings, where delegates could not be said to represent an ongoing social unit like a choir. In this respect, it might have been instructive to study STin2 allele frequencies in a third cohort comprising individuals in another established social group unrelated to music - cricket club members, for example.
Finally, the effect we identified might relate to behavioural traits in the non-musician group and not the choral singers. While very few of our singers were hospital workers, the majority of subjects in our non-musician group were. Of these many were doctors, particularly anaesthetists. It is possible that STin2-related personality traits are more common among hospital staff than those employed elsewhere.
This study was conducted in conjunction with a musical project involving one participating choir. A new 40-part unaccompanied choral work, “Allele”, was composed and broadcast on national radio. In the piece, each singer's part incorporated their personal RS3 genotype. Further details are provided in Text S1.
In conclusion, the STin2 VNTR in the SLC6A4 gene is associated with choir membership in this study. Allele frequencies at other AVPR1A and SLC6A4 polymorphisms, more strongly associated with musical phenotypes than STin2 by other authors [6], were similar in choral singers and non-musicians. Our study therefore failed to confirm the hypothesis that these polymorphisms are associated with musical ability, as assessed by choir membership. One possible interpretation of our results is that genetic factors other than those affecting musical ability alone may influence whether an individual belongs to a choir or not.
Supporting Information
The composition and performance of “Allele”.
(DOC)
Acknowledgments
Our thanks to the following for their assistance in subject recruitment and sample collection: staff of the anaesthetic department and day surgery units at Guy's and St Thomas' Hospitals; administrators, musical directors and members of the New London Chamber Choir, Merbecke Choir, BBC Symphony Chorus, Southwark Cathedral Choir, Elysian Singers, Hampton Court Palace Choir, St Alfege's Parish Church Choir, Clare College Choir, Renaissance Singers; organisers, chairmen and delegates at meetings or study days of the following - South East Thames Society of Anaesthetists, Norfolk and Norwich Hospital anaesthetic department; Royal College of Anaesthetists; Anglia Ruskin University anaesthetic department, MRC Social, Genetic and Developmental Psychiatry Centre, King's College London and the orthopaedic, urology and perioperative medicine departments of Guy's and St. Thomas' Hospitals'.
Further information about Allele and its composition are available on the Music from the Genome website (www.musicfromthegenome.org.uk. Accessed 2011 Jun 20), where Allele can also be heard in full, free of charge.
Footnotes
Competing Interests: The authors have declared that no competing interests exist.
Funding: The work was funded by a Wellcome Trust Arts Award and a related Extension Award, respectively WT089550A and WT089550Z (http://www.wellcome.ac.uk). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Clift SM, Hancox G. The perceived benefits of singing: findings from preliminary surveys of a university college choral society. J R Soc Promot Health. 2001;121:248–256. doi: 10.1177/146642400112100409. [DOI] [PubMed] [Google Scholar]
- 2.Harrison PA, Narayan G. Differences in behavior, psychological factors, and environmental factors associated with participation in school sports and other activities in adolescence. J Sch Health. 2003;73:113–120. doi: 10.1111/j.1746-1561.2003.tb03585.x. [DOI] [PubMed] [Google Scholar]
- 3.Kreutz G, Bongard S, Rohrmann S, Hodapp V, Grebe D. Effects of choir singing or listening on secretory immunoglobulin A, cortisol, and emotional state. J Behav Med. 2004;27:623–635. doi: 10.1007/s10865-004-0006-9. [DOI] [PubMed] [Google Scholar]
- 4.Green RG, Green ML. Relaxation increases salivary immunoglobulin A1. Psychol Rep. 1987;61:623–629. doi: 10.2466/pr0.1987.61.2.623. [DOI] [PubMed] [Google Scholar]
- 5.Martin RA, Dobbin JP. Sense of humor, hassles, and immunoglobulin A: evidence for a stress-moderating effect of humor. Int J Psychiatry Med. 1988;18:93–105. doi: 10.2190/724b-3v06-qc5n-6587. [DOI] [PubMed] [Google Scholar]
- 6.Ukkola LT, Onkamo P, Raijas P, Karma K, Jarvela I. Musical aptitude is associated with AVPR1A-haplotypes. PLoS One. 2009;4:e5534-. doi: 10.1371/journal.pone.0005534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bachner-Melman R, Dina C, Zohar AH, Constantini N, Lerer E, et al. AVPR1a and SLC6A4 gene polymorphisms are associated with creative dance performance. PLoS Genet. 2005;1:e42-. doi: 10.1371/journal.pgen.0010042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Israel S, Lerer E, Shalev I, Uzefovsky F, Reibold M, et al. Molecular genetic studies of the arginine vasopressin 1a receptor (AVPR1a) and the oxytocin receptor (OXTR) in human behaviour: from autism to altruism with some notes in between. Prog Brain Res. 2008;170:435–449. doi: 10.1016/S0079-6123(08)00434-2. [DOI] [PubMed] [Google Scholar]
- 9.Walum H, Westberg L, Henningsson S, Neiderhiser JM, Reiss D, et al. Genetic variation in the vasopressin receptor 1a gene (AVPR1A) associates with pair-bonding behavior in humans. Proc Natl Acad Sci USA. 2008;105:14153–14156. doi: 10.1073/pnas.0803081105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Knafo A, Israel S, Darvasi A, Bachner-Melman R, Uzefovsky F, et al. Individual differences in allocation of funds in the dictator game associated with length of the arginine vasopressin 1a receptor RS3 promoter region and correlation between RS3 length and hippocampal mRNA. Genes Brain Behav. 2008;7:266–275. doi: 10.1111/j.1601-183X.2007.00341.x. [DOI] [PubMed] [Google Scholar]
- 11.Kato M, Serretti A. Review and meta-analysis of antidepressant pharmacogenetic findings in major depressive disorder. Mol Psychiatry. 2010;15:473–500. doi: 10.1038/mp.2008.116. [DOI] [PubMed] [Google Scholar]
- 12.Huang CH, Santangelo SL. Autism and serotonin transporter gene polymorphisms: a systematic review and meta-analysis. Am J Med Genet B Neuropsychiatr Genet. 2008;147B:903–913. doi: 10.1002/ajmg.b.30720. [DOI] [PubMed] [Google Scholar]
- 13.Bloch MH, Landeros-Weisenberger A, Sen S, Dombrowski P, Kelmendi B, et al. Association of the serotonin transporter polymorphism and obsessive-compulsive disorder: systematic review. Am J Med Genet B Neuropsychiatr Genet. 2008;147B:850–858. doi: 10.1002/ajmg.b.30699. [DOI] [PubMed] [Google Scholar]
- 14.Drago A, De Ronchi D, Serretti A. Pharmacogenetics of antidepressant response: an update. Hum Genomics. 2009;3:257–274. doi: 10.1186/1479-7364-3-3-257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tang H, Quertermous T, Rodriguez B, Kardia SL, Zhu X, et al. Genetic structure, self-identified race/ethnicity, and confounding in case-control association studies. Am J Hum Genet. 2005;76:268–275. doi: 10.1086/427888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Huezo-Diaz P, Uher R, Smith R, Rietschel M, Henigsberg N, et al. Moderation of antidepressant response by the serotonin transporter gene. Br J Psychiatry. 2009;195:30–38. doi: 10.1192/bjp.bp.108.062521. [DOI] [PubMed] [Google Scholar]
- 17.de Bakker PI, Yelensky R, Pe'er I, Gabriel SB, Daly MJ, et al. Efficiency and power in genetic association studies. Nat Genet. 2005;37:1217–1223. doi: 10.1038/ng1669. [DOI] [PubMed] [Google Scholar]
- 18.Freeman B, Smith N, Curtis C, Huckett L, Mill J, et al. DNA from buccal swabs recruited by mail: evaluation of storage effects on long-term stability and suitability for multiplex polymerase chain reaction genotyping. Behav Genet. 2003;33:67–72. doi: 10.1023/a:1021055617738. [DOI] [PubMed] [Google Scholar]
- 19.Dudbridge F. Likelihood-based association analysis for nuclear families and unrelated subjects with missing genotype data. Hum Hered. 2008;66:87–98. doi: 10.1159/000119108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Purcell S, Neale B, Todd-Brown K, Thomas L, Ferreira MA, et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet. 2007;81:559–575. doi: 10.1086/519795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Perneger TV. What's wrong with Bonferroni adjustments. BMJ. 1998;316:1236–1238. doi: 10.1136/bmj.316.7139.1236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Nyholt DR. A simple correction for multiple testing for single-nucleotide polymorphisms in linkage disequilibrium with each other. Am J Hum Genet. 2004;74:765–769. doi: 10.1086/383251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Purcell S, Cherny SS, Sham PC. Genetic Power Calculator: design of linkage and association genetic mapping studies of complex traits. Bioinformatics. 2003;19:149–150. doi: 10.1093/bioinformatics/19.1.149. [DOI] [PubMed] [Google Scholar]
- 24.Niesler B, Kapeller J, Fell C, Atkinson W, Moller D, et al. 5-HTTLPR and STin2 polymorphisms in the serotonin transporter gene and irritable bowel syndrome: effect of bowel habit and sex. Eur J Gastroenterol Hepatol. 2010;22:856–861. doi: 10.1097/MEG.0b013e32832e9d6b. [DOI] [PubMed] [Google Scholar]
- 25.Newton-Cheh C, Hirschhorn JN. Genetic association studies of complex traits: design and analysis issues. Mutat Res. 2005;573:54–69. doi: 10.1016/j.mrfmmm.2005.01.006. [DOI] [PubMed] [Google Scholar]
- 26.Ogilvie AD, Battersby S, Bubb VJ, Fink G, Harmar AJ, et al. Polymorphism in serotonin transporter gene associated with susceptibility to major depression. Lancet. 1996;347:731–733. doi: 10.1016/s0140-6736(96)90079-3. [DOI] [PubMed] [Google Scholar]
- 27.Sarosi A, Gonda X, Balogh G, Domotor E, Szekely A, et al. Association of the STin2 polymorphism of the serotonin transporter gene with a neurocognitive endophenotype in major depressive disorder. Prog Neuropsychopharmacol Biol Psychiatry. 2008;32:1667–1672. doi: 10.1016/j.pnpbp.2008.06.014. [DOI] [PubMed] [Google Scholar]
- 28.Lopez de LC, Dumais A, Rouleau G, Lesage A, Dumont M, et al. STin2 variant and family history of suicide as significant predictors of suicide completion in major depression. Biol Psychiatry. 2006;59:114–120. doi: 10.1016/j.biopsych.2005.06.021. [DOI] [PubMed] [Google Scholar]
- 29.Bellivier F, Leroux M, Henry C, Rayah F, Rouillon F, et al. Serotonin transporter gene polymorphism influences age at onset in patients with bipolar affective disorder. Neurosci Lett. 2002;334:17–20. doi: 10.1016/s0304-3940(02)01029-7. [DOI] [PubMed] [Google Scholar]
- 30.Proitsi P, Lupton MK, Reeves SJ, Hamilton G, Archer N, et al. Association of serotonin and dopamine gene pathways with behavioral subphenotypes in dementia. Neurobiol Aging. 2012 doi: 10.1016/j.neurobiolaging.2010.06.011. In Press. [DOI] [PubMed] [Google Scholar]
- 31.Pritchard AL, Pritchard CW, Bentham P, Lendon CL. Role of serotonin transporter polymorphisms in the behavioural and psychological symptoms in probable Alzheimer disease patients. Dement Geriatr Cogn Disord. 2007;24:201–206. doi: 10.1159/000107081. [DOI] [PubMed] [Google Scholar]
- 32.Saiz PA, Garcia-Portilla MP, Arango C, Morales B, Bascaran MT, et al. Association study between obsessive-compulsive disorder and serotonergic candidate genes. Prog Neuropsychopharmacol Biol Psychiatry. 2008;32:765–770. doi: 10.1016/j.pnpbp.2007.12.005. [DOI] [PubMed] [Google Scholar]
- 33.Wendland JR, Kruse MR, Cromer KR, Murphy DL. A large case-control study of common functional SLC6A4 and BDNF variants in obsessive-compulsive disorder. Neuropsychopharmacology. 2007;32:2543–2551. doi: 10.1038/sj.npp.1301394. [DOI] [PubMed] [Google Scholar]
- 34.Fan JB, Sklar P. Meta-analysis reveals association between serotonin transporter gene STin2 VNTR polymorphism and schizophrenia. Mol Psychiatry. 2005;10:928–38, 891. doi: 10.1038/sj.mp.4001690. [DOI] [PubMed] [Google Scholar]
- 35.Kohen R, Cain KC, Mitchell PH, Becker K, Buzaitis A, et al. Association of serotonin transporter gene polymorphisms with poststroke depression. Arch Gen Psychiatry. 2008;65:1296–1302. doi: 10.1001/archpsyc.65.11.1296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Schurks M, Rist PM, Kurth T. STin2 VNTR polymorphism in the serotonin transporter gene and migraine: pooled and meta-analyses. J Headache Pain. 2010;11:317–326. doi: 10.1007/s10194-010-0230-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Park JW, Han SR, Yang DW, Kim YI, Lee KS. Serotonin transporter protein polymorphism and harm avoidance personality in migraine without aura. Headache. 2006;46:991–996. doi: 10.1111/j.1526-4610.2006.00439.x. [DOI] [PubMed] [Google Scholar]
- 38.Szilagyi A, Boor K, Orosz I, Szantai E, Szekely A, et al. Contribution of serotonin transporter gene polymorphisms to pediatric migraine. Headache. 2006;46:478–485. doi: 10.1111/j.1526-4610.2006.00379.x. [DOI] [PubMed] [Google Scholar]
- 39.Min W, Li T, Ma X, Li Z, Yu T, et al. Monoamine transporter gene polymorphisms affect susceptibility to depression and predict antidepressant response. Psychopharmacology (Berl) 2009;205:409–417. doi: 10.1007/s00213-009-1550-3. [DOI] [PubMed] [Google Scholar]
- 40.Smits KM, Smits LJ, Schouten JS, Stelma FF, Nelemans P, et al. Influence of SERTPR and STin2 in the serotonin transporter gene on the effect of selective serotonin reuptake inhibitors in depression: a systematic review. Mol Psychiatry. 2004;9:433–441. doi: 10.1038/sj.mp.4001488. [DOI] [PubMed] [Google Scholar]
- 41.Safarinejad MR. Analysis of association between the 5-HTTLPR and STin2 polymorphisms in the serotonin-transporter gene and clinical response to a selective serotonin reuptake inhibitor (sertraline) in patients with premature ejaculation. BJU Int. 2010;105:73–78. doi: 10.1111/j.1464-410X.2009.08714.x. [DOI] [PubMed] [Google Scholar]
- 42.Terrazzino S, Viana M, Floriddia E, Monaco F, Mittino D, et al. The serotonin transporter gene polymorphism STin2 VNTR confers an increased risk of inconsistent response to triptans in migraine patients. Eur J Pharmacol. 2010;641:82–87. doi: 10.1016/j.ejphar.2010.04.049. [DOI] [PubMed] [Google Scholar]
- 43.Yue W, Liu H, Zhang J, Zhang X, Wang X, et al. Association study of serotonin transporter gene polymorphisms with obstructive sleep apnea syndrome in Chinese Han population. Sleep. 2008;31:1535–1541. doi: 10.1093/sleep/31.11.1535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Sanjuan J, Martin-Santos R, Garcia-Esteve L, Carot JM, Guillamat R, et al. Mood changes after delivery: role of the serotonin transporter gene. Br J Psychiatry. 2008;193:383–388. doi: 10.1192/bjp.bp.107.045427. [DOI] [PubMed] [Google Scholar]
- 45.Li J, Wang Y, Zhou R, Zhang H, Yang L, et al. Association between polymorphisms in serotonin transporter gene and attention deficit hyperactivity disorder in Chinese Han subjects. Am J Med Genet B Neuropsychiatr Genet. 2007;144B:14–19. doi: 10.1002/ajmg.b.30373. [DOI] [PubMed] [Google Scholar]
- 46.MacKenzie A, Quinn J. A serotonin transporter gene intron 2 polymorphic region, correlated with affective disorders, has allele-dependent differential enhancer-like properties in the mouse embryo. Proc Natl Acad Sci U S A. 1999;96:15251–15255. doi: 10.1073/pnas.96.26.15251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Fiskerstrand CE, Lovejoy EA, Quinn JP. An intronic polymorphic domain often associated with susceptibility to affective disorders has allele dependent differential enhancer activity in embryonic stem cells. FEBS Lett. 1999;458:171–174. doi: 10.1016/s0014-5793(99)01150-3. [DOI] [PubMed] [Google Scholar]
- 48.Hranilovic D, Stefulj J, Schwab S, Borrmann-Hassenbach M, Albus M, et al. Serotonin transporter promoter and intron 2 polymorphisms: relationship between allelic variants and gene expression. Biol Psychiatry. 2004;55:1090–1094. doi: 10.1016/j.biopsych.2004.01.029. [DOI] [PubMed] [Google Scholar]
- 49.Ali FR, Vasiliou SA, Haddley K, Paredes UM, Roberts JC, et al. Combinatorial interaction between two human serotonin transporter gene variable number tandem repeats and their regulation by CTCF. J Neurochem. 2010;112:296–306. doi: 10.1111/j.1471-4159.2009.06453.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Coutinho AM, Oliveira G, Morgadinho T, Fesel C, Macedo TR, et al. Variants of the serotonin transporter gene (SLC6A4) significantly contribute to hyperserotonemia in autism. Mol Psychiatry. 2004;9:264–271. doi: 10.1038/sj.mp.4001409. [DOI] [PubMed] [Google Scholar]
- 51.Bah J, Lindstrom M, Westberg L, Manneras L, Ryding E, et al. Serotonin transporter gene polymorphisms: effect on serotonin transporter availability in the brain of suicide attempters. Psychiatry Res. 2008;162:221–229. doi: 10.1016/j.pscychresns.2007.07.004. [DOI] [PubMed] [Google Scholar]
- 52.Saiz PA, Garcia-Portilla MP, Herrero R, Arango C, Corcoran P, et al. Interactions between functional serotonergic polymorphisms and demographic factors influence personality traits in healthy Spanish Caucasians. Psychiatr Genet. 2010;20:171–178. doi: 10.1097/YPG.0b013e32833a20b9. [DOI] [PubMed] [Google Scholar]
- 53.Zhong S, Israel S, Xue H, Sham PC, Ebstein RP, et al. A neurochemical approach to valuation sensitivity over gains and losses. Proc Biol Sci. 2009;276:4181–4188. doi: 10.1098/rspb.2009.1312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Kazantseva AV, Gaysina DA, Faskhutdinova GG, Noskova T, Malykh SB, et al. Polymorphisms of the serotonin transporter gene (5-HTTLPR, A/G SNP in 5-HTTLPR, and STin2 VNTR) and their relation to personality traits in healthy individuals from Russia. Psychiatr Genet. 2008;18:167–176. doi: 10.1097/YPG.0b013e328304deb8. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
The composition and performance of “Allele”.
(DOC)