Abstract
Each year, tens of thousands of children undergo cardiopulmonary bypass (CPB) to correct congenital heart defects. Although necessary for surgery, CPB involves stopping the heart and exposing it to ischemic conditions. On reoxygenation, the heart can experience effects similar to that of acute myocardial infarction. Although much is known about adult injury, little is known about the effects of global ischemia on newborn ventricles. We studied newborn (2 to 4 days old) and adult (>8 weeks old) rabbit hearts subjected to ischemia–reperfusion (30 min of ischemia and 60 min of reperfusion). Our data demonstrated chamber- and age-specific changes in oxidative stress. During ischemia, hydrogen peroxide (H2O2) increased in both right-ventricular (RV) and left-ventricular (LV) myocytes of the newborn, although only the RV change was significant. In contrast, there was no significant increase in H2O2 in either RV or LV myocytes of adults. There was a fivefold increase in H2O2 formation in newborn RV myocytes compared with adults (P = 0.006). In whole-heart tissue, superoxide dismutase activity increased from sham versus ischemia in the left ventricle of both adult and newborn hearts, but it was increased only in the right ventricle of the newborn heart. Catalase activity was significantly increased after ischemia in both adult ventricles, whereas no increase was seen in newborn compared with sham hearts. In addition, catalase levels in newborns were significantly lower, indicating less scavenging potential. Nanoparticle-encapsulated ebselen, given as an intracardiac injection into the right or left ventricle of newborn hearts, significantly increased functional recovery of developed pressure only in the right ventricle, indicating the potential for localized antioxidant therapy during and after pediatric surgical procedures.
Keywords: Ischemia–reperfusion, Oxidative stress, Catalase, Hydrogen peroxide
Introduction
Congenital heart defects are the leading birth defect in the pediatric population. In most cases, surgical correction involving cardiopulmonary bypass (CPB) is the most successful treatment for these conditions. Successful repair during the first year of life may eliminate the need for a second repair, but it does not preclude the possibility of future reinterventions. As with any other surgical procedure, postoperative complications exist. One such complication is low cardiac output syndrome (LCOS), which is commonly (25% of patients) seen 6 to 18 h after CPB and results in inadequate organ perfusion and, ultimately, organ dysfunction [24]. Studies show that an increase in afterload without a corresponding improvement in contractility may aid in the deterioration of cardiac output after CPB [18, 22, 33, 43]. Ischemic and reperfusion injuries are other complications of CPB and result in an inflammatory process that causes damage to both ventricles of the myocardium. Various studies have proposed and employed certain strategies—such as modified ultrafiltration, which eliminates inflammatory mediators from the circulation and myocardial apoptosis prevention by radical scavenging—to prevent or attenuate the inflammatory effects of CPB [8]. Advanced surgical techniques have also been used to decrease the possibility of LCOS. These include the Norwood procedure, which causes hemodynamic improvement after surgery [21, 31].
Although ischemia and reperfusion are widely studied in the adult population, much less attention has been focused on the pediatric population. Several studies have shown that the pediatric heart is more sensitive to ischemia–reperfusion (IR) injury compared with the adult heart [28, 34]. Younger age and prolonged CPB time contribute to the degree of myocardial dysfunction after surgery. The mechanisms of the dysfunction caused by IR injury in the immature heart are not clear, but evidence suggests that reactive oxygen species (ROS), calcium overload, inflammation, accumulation of metabolic end products, and apoptosis may contribute to this injury [4, 5]. Still others argue that tissue pH plays a role in the newborn heart’s greater susceptibility to ischemic injury [28]. Several studies have demonstrated significant differences in anti-oxidant protein levels in newborns compared with adults, yielding a consensus that newborn hearts have diminished scavenging capacity [23]. In small amounts, ROS can act as important second messengers that initiate a cascade of events that regulate important normal cellular functions. In excess, however, these ROS lead to damaging sequelae, including apoptosis, necrosis, abnormal growth and migration, and recruitment of damaging inflammatory cells.
Oxygen intermediates, such as hydrogen peroxide (H2O2), superoxide anion ( ), and the hydroxyl (OH•) radical are some of the key players involved in cardiotoxicity occurring during IR. The sources of these radicals vary depending on the cell type and injury. Myocytes contain nicotinamide adenine dinucleotide phosphate oxidase (NADPH-oxidase), which may catalyze the production of both H2O2 and [15]. Moreover, ROS can be formed from breakdowns in the electron transport chain, thus leading to mitochondrial ROS [25]. Finally, infiltrating inflammatory cells may also contribute to the excessive ROS levels seen after IR. This newborn vulnerability to IR injury may be attributed to newborn hearts containing immature and inadequate number of sarcoplasmic reticula compared with adult hearts [28]. In such cases, the processing and storage of calcium appears to be impaired. In addition, underdeveloped sarcolemma contains high amounts of polyunsaturated fatty acids, which may predispose to membrane injury by ROS [2].
To date, little is known about the production of oxidative stress in newborns during ischemic injury, especially in the local setting. In the current study, to determine chamber- and age-specific differences in oxidative stress between adult and newborn hearts, we measured changes in antioxidant protein activity in control and IR hearts. Moreover, real-time H2O2 and superoxide generation were measured in isolated left-ventricular (LV) and right-ventricular (RV) myocytes from newborn and adult rabbits using a novel microscopy protocol. Our results demonstrate a failure of RV myocytes to increase catalase activity in response to IR as well as a significant increase in H2O2 production. With this localized increased production, we used ebselen-loaded nanoparticles (PK3-ebselen; Cayman Chemical) and observed an increased recovery in RV function, suggesting a potential new therapeutic strategy for improving cardiac function during and after surgery.
Materials and Methods
Langendorff Perfusion Studies
Newborn (2 to 5 days old) and adult New Zealand white rabbits (≥8 weeks old) were anesthesized with pentobarbital [Nembutal] (Lundbeck Inc., Deerfield, IL) administered intraperitoneally (newborn) or intravenously (adult) at a dose of 50 mg/kg 10 min after heparin sulfate (Hospira Inc., Lake Forest, IL) injection (500 IU/kg). A midline incision was performed to expose the chest cavity, and the heart great vessels were excised. Isolation and cannulation of the aorta (with the pulmonary artery left open) on a Langendorff perfusion system (EMKA Technologies, France), using normal Tyrode’s solution (NaCl, Glucose, Hepes, KCl, MgCl2, and CaCl2 [pH 7.4, 37°C]), was performed in a retrograde fashion. After excess blood was washed out, the right atrium was incised to allow entry of a water-filled latex balloon attached to a pressure transducer to measure RV-developed pressure. In some hearts, LV-developed pressure was measured (not concurrently) in the same manner as RV-developed pressure. Electrocardiogram electrodes were positioned along the right and left ventricles, and the whole heart was encased in a cylindrical water bath heated to 37°C. Perfusate was allowed to flow for 20 min for stabilization followed by 30 min of no flow (ischemia) and finally 60 min of reflow (reperfusion). The right and left ventricles were then separated and frozen for biochemical and immunohistochemical analysis. No volumetric measurements were made. All studies were approved by the Emory University Institutional Animal Care and Use Committee.
Myocyte Isolation and Confocal Microscopy
Cardiac myocytes were isolated using calcium-free Tyrode solution (NaCl, glucose, HEPES, KCl, MgSO4, Na2HPO4, bovine serum albumin, and collagenase), as described previously, from both newborn and adult rabbits and separated into RV and LV portions [6]. The cells were then incubated with deuterated dihydroethidium (DHE) and dichlorodihydrofluorescein diacetate (DCFDA) to allow for the visualization of superoxide and H2O2, respectively, by way of confocal microscopy (Olympus Fluoview FV1000). In addition, PK3-ebselen (300 μl) was incorporated into the cells, along with DHE and DCFDA, to determine its effect on H2O2 production. After 1 h of incubation, the cells were plated onto a plastic spherical cover slip. This was encased in a metal chamber (FCS2; Bioptechs) with ports at both ends, thus allowing for the entry and exit of solution gassed with either nitrogen or ambient oxygen, and positioned onto the confocal microscope. Real-time measurements of H2O2 and superoxide were measured during hypoxic (30 min at 25°C) and reoxygenation (60-min) periods.
Measurement of Antioxidant Activity
Catalase levels were measured in tissue homogenates using decomposition of H2O2 at 240 nm using a plate reader as described [30]. Measurements in the presence and absence of 3-aminotriazole (specific catalase inhibitor) were taken, and the inhibitable portion was deemed as catalase activity. Superoxide dismutase (SOD) activity was measured with a commercially available kit (Dojindo Molecular Technologies). Both activities were normalized to protein content.
H2O2-Scavenging Nanoparticles
Nanoparticles were synthesized from the polymer poly-(cyclohexane-1,4diyl acetone dimethylene ketal [PCADK]). Five hundred micrograms of ebselen was incubated with 50 mg of PCADK, and nanoparticles were formed by immersion in polyvinyl alcohol as described previously [39]. Ebselen loading was determined by hydrolyzing the particles and measuring absorbance at 270 nm using pure ebselen to generate a standard curve. Activity was confirmed by measuring decomposition of exogenous H2O2 at 240 nm.
Results
Recovery of Developed Pressure Was Similar in Right and Left Ventricles of Newborn Heart
To determine the extent of IR injury, developed pressure was measured in both ventricles before and after IR injury (30 min of ischemia and 60 min of reperfusion) and expressed as a percent recovery (RDPfinal/RDPinitial × 100; 100% = max). Percent recovery of developed pressure (RDP) in the right ventricle of the newborn rabbit heart after IR was 49.5 ± 5.4% (Fig. 1). RDP in the left ventricle was 43.4 ± 17.6%, and there was no significant difference between the two chambers.
Fig. 1.
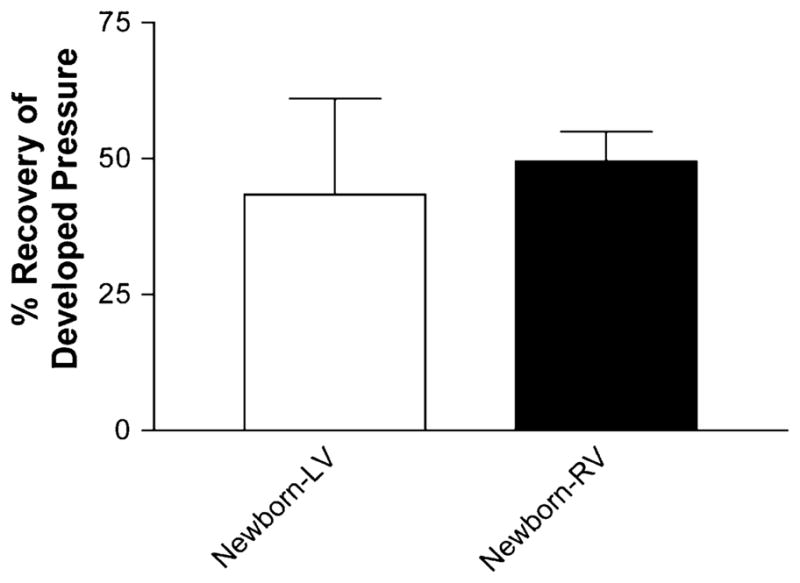
Similar recovery of developed pressure seen in both ventricles of newborn rabbits. Percent recovery of developed pressure in newborn left and right ventricles after IR. Calculated as developed pressurefinal (after 90 min of IR)/developed pressureinitial (after 20 min of stabilization) × 100. Values are mean ± SE; n = 5 to 9 hearts/group
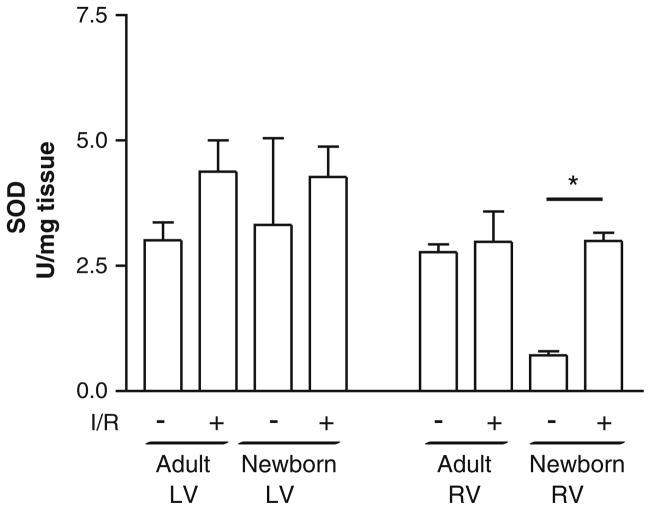
Differential SOD Activity in Left and Right Ventricles in Newborns and Adults
To determine superoxide-scavenging ability after IR, SOD activity was measured in both ventricles of adult and newborn rabbits (Fig. 2) subject to sham or IR surgery. In sham-operated adults, the left ventricle had 3.0 ± 0.4 U/mg tissue of SOD activity and 2.8 ± 0.2 U/mg tissue in the right ventricle. Although there was a trend for increased activity after IR in the left ventricle (4.4 ± 0.6 U/mg tissue [P = 0.06]), there was no change in the right ventricle after injury (2.9 ± 0.6 U/mg tissue). In newborn left ventricles, there was a similar level of SOD activity in sham-operated hearts (3.3 ± 1.7 U/mg tissue), but there was no significant change after IR (4.2 ± 0.5 U/mg tissue). In contrast, newborn RV tissue in sham-operated hearts had significantly less SOD activity (0.6 ± 0.1 U/mg tissue [P < 0.01 vs. adult sham right ventricle]), which was increased nearly fivefold in response to IR injury (3.0 ± 0.1 U/mg tissue [P < 0.05 vs. newborn sham right ventricle]). Two-way analysis of variance (ANOVA) demonstrated no significant interaction between age and chamber in either basal or IR-induced SOD activity. These data demonstrate that newborn right ventricle has decreased baseline SOD activity compared with adult, although it is able to increase activity to adult levels in response to IR. In addition, the newborn left ventricle has levels similar to those in adult tissue, although these do not increase activity in response to IR. Taken together, these data demonstrate distinct age-and chamber-specific differences in superoxide scavenging after IR injury.
Fig. 2.
SOD activity shows ventricle-specific regulation in adults and increased SOD activity in the right ventricle of newborns after IR. SOD activity increases in adult and newborn LV and RV homogenates after IR surgery. There was no significant difference after IR in the adult and newborn LV. A fourfold increase was observed in the newborn right ventricle after IR (*P < 0.05 [ANOVA followed by Bonferroni posttest]), whereas no significant difference was observed in the adult right ventricle. SOD activity was measured using a commercially available kit and normalized to tissue weight. Values are mean ± SE; n = 3 to 6 hearts/group
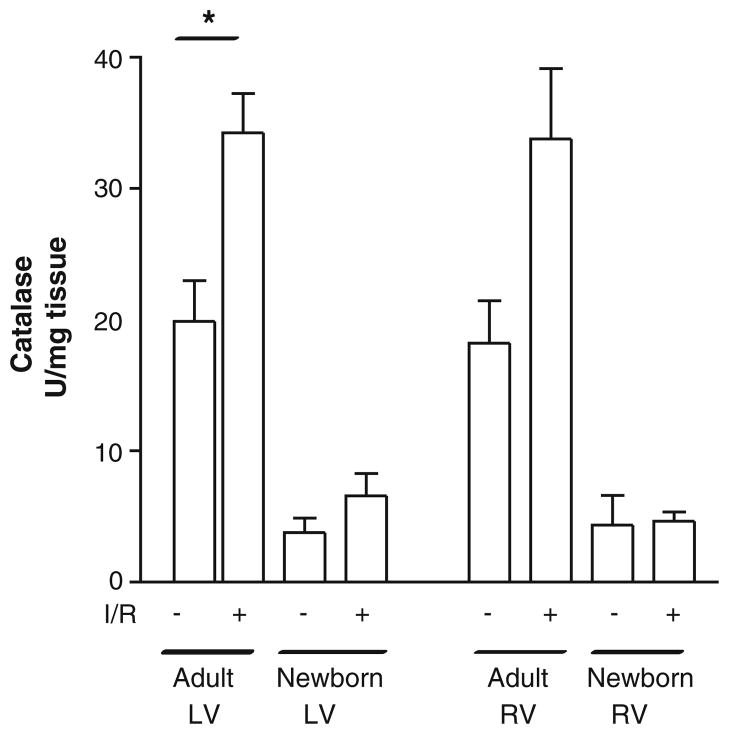
Differential Catalase Activity in Left and Right Ventricles in Newborns and Adults
To determine if a similar pattern was seen with H2O2-scavenging ability, catalase activity was also measured in adult and newborn (Fig. 3) rabbit hearts subjected to sham or IR surgery. Sham-operated adult hearts had baseline catalase levels of 19.9 ± 4.56 U/mg tissue in the left ventricle and 20.5 ± 2.84 U/mg tissue in the right ventricle. After IR, there was an increase in both ventricles to 34.3 ± 4.4 U/mg tissue (P < 0.05) and 33.8 ± 5.4 U/mg tissue (P = 0.06), respectively. Surprisingly, both the left and right ventricles in sham-operated newborn hearts had approximately 30% of the catalase activity of those in adults (3.8 ± 1.1 U/mg tissue in the left ventricle and 4.3 ± 2.3 U/mg tissue in the right ventricle). After IR, there was no significant increase in either chamber (5.0 ± 0.7 U/mg tissue in the left ventricle and 4.6 ± 1.7 U/mg tissue in the right ventricle). Analysis using two-way ANOVA demonstrated a significant difference due to age (P = 0.019), but there were no chamber differences. Moreover, there was no interaction between age and chamber under either sham or IR conditions. These data suggest the adult heart has a significantly greater capacity to scavenge H2O2, as well as increase catalase in response to IR injury, than the newborn heart.
Fig. 3.
Catalase levels are increased in both ventricles of adult rabbits, but newborns hearts have diminished activity after IR. Catalase activity increased in adult LV and RV tissue homogenates after IR surgery. Catalase activity was measured in ventricular homogenates, in the presence and absence of 3-AT, using decomposition of H2O2 at 240 nm and normalized to tissue weight. There was a significant difference in the adult LV whereas a non-significant increase was found in the adult RV after IR (*P < 0.05 [ANOVA followed by Bonferroni posttest]). No significant difference was observed in both newborn left and right ventricles after IR, and baseline levels were 50% of adult values (P < 0.05 [two-way ANOVA]). Values are mean ± SE; n = 3 to 6 hearts/group
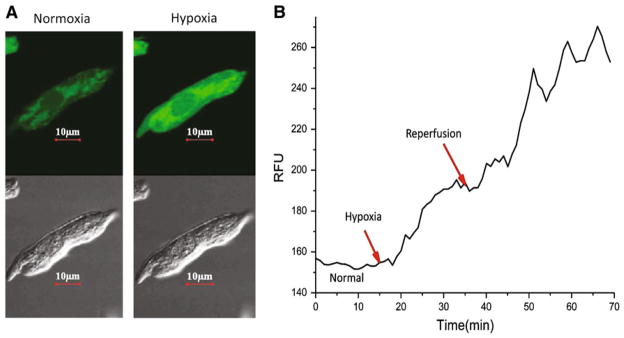
Confocal Microscopy Can be Used to Measure Real-Time ROS Production
Because tissue samples were too small to accurately measure levels of H2O2 and during the time frame of ischemia–reperfusion, chamber-specific cardiomyocytes were isolated and subject to hypoxia–reoxygenation to measure real-time H2O2 and levels. Cells were pre-loaded with DCFDA and DHE to measure H2O2 and , respectively, and imaged for 20 min of stabilization, 30 min of hypoxia, and 30 min of reoxygenation. Sample DCFDA images are shown in Fig. 4a. Fluorescent measurements were taken every minute and plotted during the duration of hypoxia and reoxygenation to determine rate of formation (relative fluorescent units [RFU]; Fig. 4b).
Fig. 4.
Method for measuring both real-time H2O2 and superoxide production in cultured myocytes. H2O2 generation after IR in newborn myocytes using confocal microscopy is shown. a Sample DCFDA image obtained before (left panel) and 20 min into (right panel) ischemia. Shown are both green fluorescence (top of both panels) for DCFDA and brightfield images (bottom of both panels). b Sample tracing demonstrating real-time fluorescence readings at all points during the experiment. Plotted values were used to determine slope or rate of production. Similar plots were made for superoxide using dihydroethidium (red) fluorescence
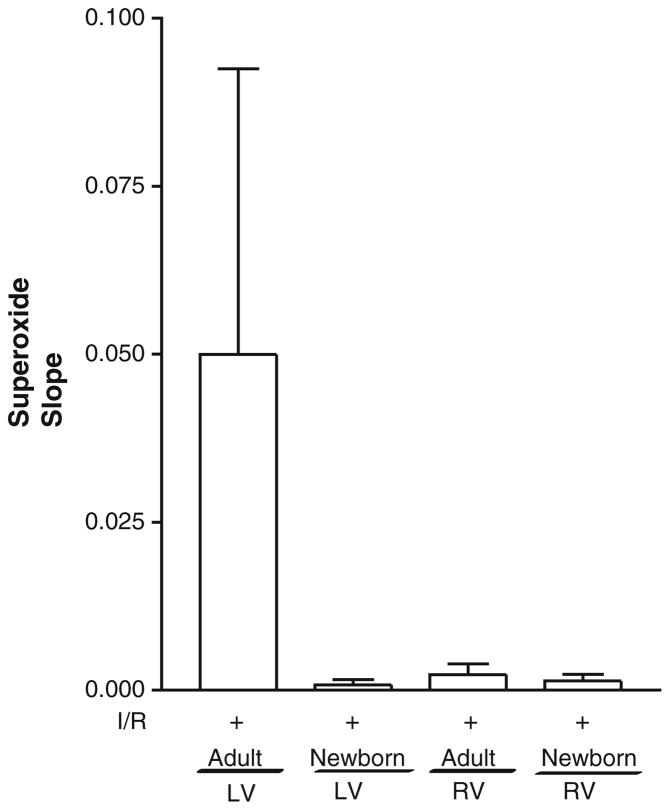
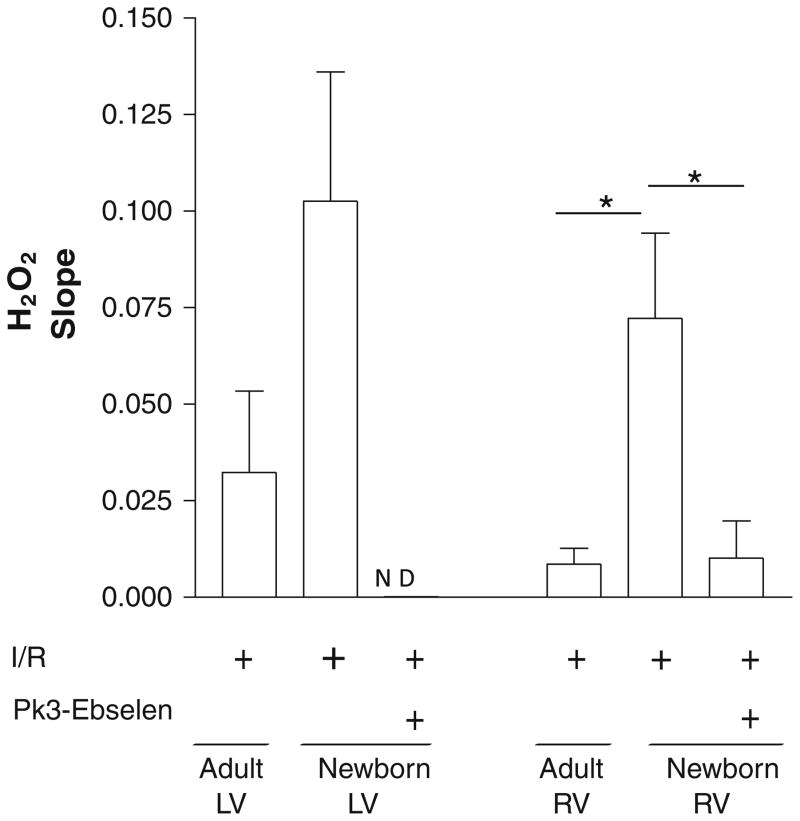
There was an expected increase in in the LV myocytes of adult compared with newborn rabbits, although no significant differences were seen in the RV myocytes of adults and newborns (Fig. 5). In contrast, there was a greater than fivefold increase in H2O2 formation in newborn RV myocytes compared with those of adults (0.072 ± 0.002 vs. 0.014 ± 0.006 RFU/min [P = 0.006]) (Fig. 6). Although there was a trend toward increased H2O2 formation in the LV myocytes of newborn compared with those of adults, the data were not significant (Fig. 6). Taken together, these results demonstrate that H2O2 generation is significantly greater in RV myocytes of newborn compared with those of adults, whereas no significant changes are seen in the LV myocytes of newborns for H2O2 or . When newborn LV and RV myocytes were incubated with PK3-ebselen and subjected to hypoxia, H2O2 was undetectable in the left ventricle and significantly diminished in the right ventricle (Fig. 6), suggesting effective decrease of H2O2 by the encapsulated scavenger.
Fig. 5.
Superoxide levels are increased in adult LV myocytes. Superoxide production in newborn and adult LV and RV myocytes was measured using real-time confocal microscopy on isolated myocytes (after incubation with dihydroethidium) after 90 min of IR. No significant difference was reported. Values are mean ± SE; n = 6 to 8 cells/group
Fig. 6.
H2O2 is increased in the right ventricle of newborns compared with that of adults after hypoxia. H2O2 generation in newborn and adult LV and RV myocytes was measured using real-time confocal microscopy on isolated myocytes (after incubation with DCFDA) after 90 min of IR. A fivefold increase was observed in the newborn right ventricle compared with adult RV cells (*P < 0.05 [ANOVA followed by Bonferroni posttest]), whereas no significant change was found in the newborn left ventricle compared with that of the adult. A threefold decrease was observed in the newborn right ventricle, whereas no signal was detected in the newborn left ventricle after PK3-ebselen treatment. Values are mean ± SE; n = 6 to 8 cells per group. ND not detected
Cardioprotection With Local Ebselen Delivery
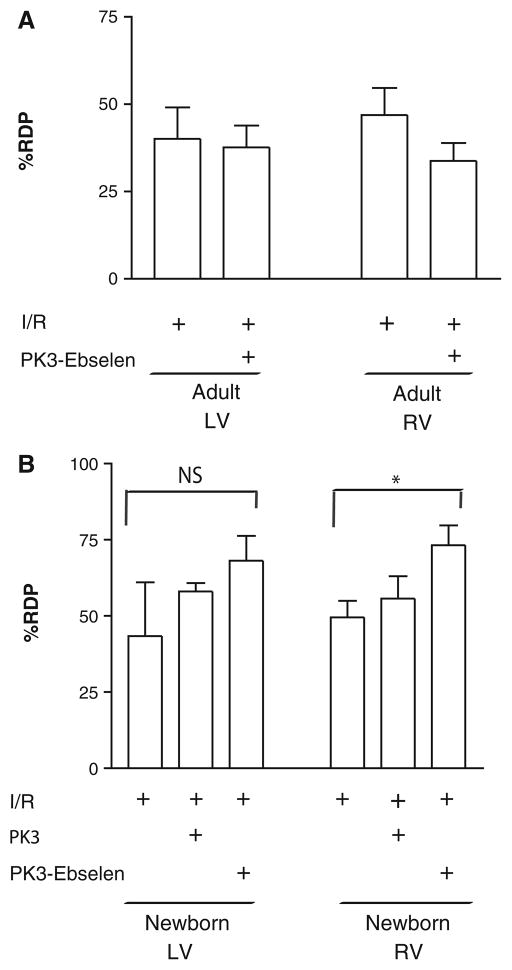
Because the production of ROS and activity of antioxidants demonstrated chamber-specific characteristics, local delivery of an H2O2 scavenger (ebselen) using polyketal nanoparticles (PK3) was performed [50]. In preliminary studies, there was no effect of free ebselen injected directly into the tissue (data not shown). Delivery of a single dose of ebselen within polyketal particles (PK3-ebselen) had no significant effect on adult rabbit LV or RV function (12 μg, 400 μl [Fig. 7a]). In contrast, when a similar dose of PK3-ebselen was administered as an intracardiac injection into the newborn right ventricle 1 minute before reperfusion, RDP increased significantly to 73.2 ± 6.4% (Fig. 7b [P < 0.05]). In the left ventricle, a slight increase was also observed (68.1 ± 8.2%) after intracardiac injection, but this was not significant. Similar doses of empty particles (PK3) had no significant effect on RDP in the right or left ventricle (55.7 ± 7.3% and 58.1 ± 2.7%, respectively). Finally, PK3-ebselen at similar concentrations in perfusate had no significant effect on RDP (data not shown), thus indicating the need for local delivery.
Fig. 7.
Local H2O2 scavenging with nanoparticles improves functional recovery in newborn right ventricles (but not left ventricles) after IR. RDP in both left and right ventricles after IR was measured in adult (a) and newborn (b) rabbit hearts. PK3-ebselen, administered as an intracardiac injection into the right ventricle of the newborn heart, resulted in a significant increase in RDP of saline-treated hearts (*P <0.05 [ANOVA followed by Bonferroni posttest]). No significant increase in recovery was observed in either adult ventricles or the newborn left ventricle. Values are mean ± SE; n = 5 to 9 hearts/group
Discussion
Although much is known about adult myocardial IR injury, little is known about injury to newborn hearts, specifically the right ventricle, because the literature reports findings observed mostly in the left ventricle. Previous studies have implicated ROS as the cause of the increased susceptibility of the newborn heart to ischemic injury [15, 34]. Although these ROS are continuously being produced in normal tissue, their overproduction during IR overwhelms endogenous scavengers (antioxidants), thus leading to cell death and diminished ventricular function, as was observed in our study (50% RDP in both newborn ventricles) and by others [3, 9]. This diminished function occurs in part due to increased local H2O2 production because local delivery of nanoparticles containing ebselen partly restored function only in the right ventricle.
Initially, we examined SOD activity because superoxide production plays an important role in the function of adult hearts. We saw increased activity of SOD in the adult left ventricle in response to IR but no changes in the right ventricle. This would be expected because most studies performed in adult animals demonstrate an acute increase in , which contributes to death of cardiomyocytes. Increasing SOD levels in the left ventricle has a positive effect on cardiac function and remodeling, underscoring the importance that scavenging has on attenuation of IR injury. Interestingly, newborn LV tissue demonstrated similar basal levels of SOD activity. Although our study demonstrates similarities between adults and newborn rabbits for SOD, most published studies show important species-dependent changes. In pigs, for example, SOD and catalase increase during the first few weeks of life before stabilizing [32], whereas rats generally demonstrate an age-dependent decrease in antioxidant levels [35]. In addition, we found that in newborns, the right ventricle had significantly decreased SOD activity compared with the left ventricle. Although this would be a cause for concern, there was a significant increase in response to IR, suggesting that the right ventricle has potential defense mechanisms for scavenging excessive ROS.
Although the mechanism for this increase is unclear, studies show that certain cell types respond to by increasing SOD in a compensatory manner. It has been reported that the mechanism involved in overexpression of SOD1 in murine cardiac grafts is decreased apoptosis and inflammation during IR, thereby preventing the development of graft coronary artery disease [41]. Decreased SOD has also been known to play a role in the exaggerated sympathetic activity seen in rabbits with congestive heart failure [7, 38]. Thus, there is a link between inadequate levels of SOD and progression of heart disease in adults.
Because catalase represents an important scavenging mechanism for H2O2, we next examined catalase activity in adults and newborn rabbits by examining the 3-aminotriazole inhibitable portion of peroxidase activity. Studies suggest that catalase is responsible for 80% of all peroxidase activity in mature myocytes, but few studies have been performed to compare age-related changes. Our data clearly demonstrate significant changes in basal catalase levels in newborns compared with adults. Previous studies from Maulik et al. [23] showed that catalase levels are quite low in the first 2 weeks of life before significantly increasing to a peak at 14 days, then significantly decreasing during the next 2 to 4 weeks. Although our studies would agree with these early findings, our data demonstrate that the adult heart is able to increase activity in response to injury. In adult left and right ventricles, there was a twofold to threefold increase in catalase activity after IR. This would suggest that there may be little benefit of catalase therapy in the acute setting, as supported by our published studies of inducible catalase overexpression [30]. When catalase overexpression was induced in adult myocytes just before or immediately after infarction, there was no significant increase in function. In addition, our unpublished data in adults suggest no major increase in H2O2 levels in the first 24 hours after IR.
In keeping with the previously mentioned studies, we observed chamber- and age-specific differences in oxidative stress in adult versus newborn rabbit hearts. In our preliminary studies, we were unable to detect ROS in such small tissue samples using conventional assays. Moreover, for these studies, the ischemic period was 30 min followed by 60 min of reperfusion; therefore, the optimal time to measure ROS in vivo was not clear. For these reasons, we devised a novel setup to measure real-time ROS production in isolated myocytes. This would enable simultaneous comparisons of left and right ventricle cells as well as both and H2O2. Although we observed no significant changes in LV H2O2 levels between adults and newborns, H2O2 production was increased in the right ventricle of newborns during hypoxia–reoxygenation treatment. This study did not examine potential sources of ROS because the Langendorff model is an isolated heart model and therefore excludes inflammatory cell infiltration as a source of ROS. Thus, most ROS are likely from cardiomyocytes or other cardiovascular cells. Cardiomyocytes contain both Nox2 and Nox4 subunits of the NADPH-oxidase, and both play a role in ROS production after IR [16, 19]. Another potential source is mitochondrial ROS because this has been shown to be important in the pathogenesis of IR injury [37]. In adults, IR increased catalase activity, reinforcing our in vitro experiments showing low levels of H2O2 during hypoxia–reoxygenation. Interestingly, it is unknown whether the increase in H2O2 seen in the newborn heart is a result of the low catalase levels or the fact that there is a significant increase in SOD in the right ventricle without a subsequent increase in catalase. In addition, the mechanism for increased catalase activity seen in adult hearts subjected to injury but not in newborn hearts is unknown. Numerous studies have delineated the developmental differences in newborn and adult hearts with respect to accumulation of metabolic end-products, hydrogen ion-buffering capacity, calcium handling [44–49], and catalase cofactors, such as selenium and cysteine, which are needed to upregulate glutathione-related enzymes [42]. These cofactors may play a role in ventricle-dependent responses and will be examined in future studies.
Although our data suggest that the extent of damage in the left and right ventricle was similar (as measured functionally), some studies have suggested ventricle-specific differences in growth factors, transcription factors, and metabolism. For example, previous studies demonstrated that during IR injury in newborn pigs, the left ventricle had significantly less ATP than the right ventricle as well as increased hydrogen ion formation [32, 48]. Although there were no data to determine whether this was the cause of diminished function, it does outline that the right and left ventricles experience different responses to the same injury.
In a recent study after CPB surgery in children [29], it was determined by way of Doppler analysis that RV myocardial mechanics were more abnormal than those of the left ventricle and that regional differences became more apparent by 24 h. Similar studies performed in sheep [36] demonstrated that after CPB, the right ventricle experiences more damage than the left ventricle and hence benefits more from cardioplegic protection. Moreover, differences have been seen in beta-adrenergic receptors in the left and right ventricles of newborn pigs [47]. In addition, their response to inotropes varied significantly compared with adults, suggesting both age- and chamber-specific differences in potential contractile pathways [26, 27]. Although we did not examine beta-adrenergic responses in these studies, this could be an area for future research because adult mouse hearts overexpressing Gaq demonstrate changes in SERCA and cardiac function that are reversed by catalase overexpression [17]. In addition, although H2O2 may have many effects on signaling, our unpublished data suggest that H2O2 added directly to myocytes decreases sarcomeric shortening only in newborn cells, whereas no effect is seen in adults. These data agree with previous studies demonstrating that H2O2 interferes with calcium handling and force-frequency relationships by way of alterations in SERCA and phospholamban activities [1, 20]. There is also evidence that intracellular calcium overload may be implicated in H2O2-induced injury, with subsequent action potential shortening, because decreased intracellular calcium levels (by way of adenosine triphosphate–activated potassium channel activation) reversed the cardiac dysfunction previously observed [10]. Last, another major aspect of H2O2-induced injury is the inhibition of oxidative phosphorylation in the mitochondria, thus leading to decreased glycogen stores [12]. These explanations highlight the role that H2O2 scavenging plays in reversing cardiac dysfunction.
The increase in H2O2 and diminished scavenging capability in newborn ventricles led us to explore strategies to improve the manner in which the newborn heart responds to IR injury. Ebselen, a glutathione peroxidase mimetic and organoselenium compound, was encapsulated in a polyketal nanoparticle to decrease diffusion away from the injury zone. Because the oxidative stress response we saw in our studies was localized in nature, we hypothesized that local therapy may improve function. Because cardiac tissue is highly vascularized and convective forces may rapidly remove compounds from the cardiomyocytes, nanoparticles represent an excellent vehicle for small-molecule retention. We have published several studies using these biocompatible nanoparticles for sustained local delivery [35, 39, 40]. In preliminary studies, local injection of free ebselen had no effect, most likely due to the small size and large diffusion area of the myocardium. When encapsulated in nanoparticles, ebselen retained activity as measured by the decomposition of exogenous H2O2. Thus, the scavenging ability of ebselen nanoparticles was independent of ebselen being released because H2O2 is small and freely diffusible; it is likely that the encapsulated ebselen acted as an H2O2-scavenging sink. Moreover, in our hypoxia–reoxygenation experiments in isolated myocytes, encapsulated ebselen significantly decreased H2O2 generation in all cells. Although the directly injected particles did not spread throughout the entire area of the RV wall, the encapsulated ebselen was likely able to decrease the local burst of H2O2 from the myocytes, thus preventing damaging effects. In addition, when delivered in perfusate, the particles were unlikely to extravasate out of the vasculature to the area of damage. Along with the direct oxidant-scavenging properties of ebselen [11, 13], another mechanism that may have facilitated postischemic recovery of developed pressure is through the prevention of calcium overload by way of decreasing intracellular inositol 1,4,5-triphosphate-induced calcium release [28]. Furthermore, many studies have demonstrated a positive correlation between coronary flow and recovery of developed pressure. Although we did not measure this parameter, it is possible that encapsulated ebselen increased recovery by improving coronary flow [14]. Future studies will determine the effects of PK3-ebselen on coronary flow to determine if there is indeed a relationship. Neither the addition of encapsulated ebselen to the perfusate alone nor its administration before the ischemic period afforded the same protection as administering it by way of intracardiac injection directly into the walls of the right and left ventricles just before the reperfusion period, underscoring the need for local, temporal-dependent scavenging. Although the encapsulated ebselen decreased H2O2 in LV myocytes as well, there was no significant effect on cardiac function. Although there was a trend for an increase, our data would suggest that there are clear ventricle-specific differences in newborn response to oxidative stress, with the right ventricle possibly being more susceptible. Future studies will be performed to determine this interesting mechanism by which H2O2 increases in both ventricles but only significantly damages one. Taken together, our data imply that a single dose may be sufficient and that local scavenging of ROS during reperfusion contributes largely to improving newborn RV cardiac function after IR injury.
Conclusion
In conclusion, we observed an increase in SOD activity in both adult and newborn rabbit left ventricles as well as a fivefold increase in the newborn right ventricle after IR injury, thus demonstrating that these ventricles have the potential to defend themselves against excessive superoxide production. Catalase activity showed the same trend in adult ventricles. In the newborn, however, not only were the baseline values half the adult values, there was also no change in activity after IR despite a fivefold increase in H2O2 production compared with the adult right ventricle. Although there was no increase in catalase in the left ventricle, H2O2 levels were not significantly different from those in adults. After intracardiac injection with nano-particle-encapsulated ebselen, our functional results demonstrated an increase in the newborn’s recovery of RV- and LV-developed pressure by approximately 48 and 25%, respectively. Although more studies must be performed to delineate the mechanisms involved in these differential responses to IR, it is quite clear that local therapy must be considered in the context of newborn hearts after injury. These studies may be helpful in minimizing injury both during and immediately after surgical procedures, such as CPB, that adversely affects the pediatric population.
Acknowledgments
This work was supported in part by an Emory-Children’s Center for Cardiovascular Biology Pilot Grant from Children’s Healthcare of Atlanta (M. E. D., G. D.) as well as Grant Nos. HL094527 and HL090601 to M. E. D. The authors thank Pao Lin Che for help in nanoparticle synthesis.
Footnotes
Conflict of interest M. E. Davis, as well as Emory University, are entitled to equity and royalties derived from Ketal Biomedical, Incorporated, which is developing products related to the nanoparticle technology described in this article. This study could affect their personal financial status. The terms of this arrangement have been reviewed and approved by Emory University in accordance with its conflict-of-interest policies.
Contributor Information
E. Bernadette Cabigas, Wallace H. Coulter Department of Biomedical Engineering at Emory University, Georgia Institute of Technology, Atlanta, GA 30322, USA. Division of Cardiology, Emory University School of Medicine, Atlanta, GA 30322, USA.
Guoliang Ding, Department of Pediatrics, Emory University School of Medicine, Atlanta, GA 30322, USA. Emory-Children’s Center for Cardiovascular Biology, Children’s Healthcare of Atlanta, Atlanta, GA 30322, USA.
Tao Chen, Department of Pediatrics, Emory University School of Medicine, Atlanta, GA 30322, USA. Emory-Children’s Center for Cardiovascular Biology, Children’s Healthcare of Atlanta, Atlanta, GA 30322, USA.
Talib B. Saafir, Department of Pediatrics, Emory University School of Medicine, Atlanta, GA 30322, USA. Emory-Children’s Center for Cardiovascular Biology, Children’s Healthcare of Atlanta, Atlanta, GA 30322, USA
Karl D. Pendergrass, Wallace H. Coulter Department of Biomedical Engineering at Emory University, Georgia Institute of Technology, Atlanta, GA 30322, USA. Division of Cardiology, Emory University School of Medicine, Atlanta, GA 30322, USA
Mary B. Wagner, Department of Pediatrics, Emory University School of Medicine, Atlanta, GA 30322, USA. Emory-Children’s Center for Cardiovascular Biology, Children’s Healthcare of Atlanta, Atlanta, GA 30322, USA
Michael E. Davis, Email: michael.davis@bme.emory.edu, Wallace H. Coulter Department of Biomedical Engineering at Emory University, Georgia Institute of Technology, Atlanta, GA 30322, USA. Division of Cardiology, Emory University School of Medicine, Atlanta, GA 30322, USA. Emory-Children’s Center for Cardiovascular Biology, Children’s Healthcare of Atlanta, Atlanta, GA 30322, USA
References
- 1.Armoundas AA, Rose J, Aggarwal R, Stuyvers BD, O’Rourke B, Kass DA, et al. Cellular and molecular determinants of altered Ca2+ handling in the failing rabbit heart: primary defects in SR Ca2+ uptake and release mechanisms. Am J Physiol Heart Circ Physiol. 2007;292(3):H1607–H1618. doi: 10.1152/ajpheart.00525.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Awad AB, Clay SW. Age-dependent alterations in lipids and function of rat heart sarcolemma. Mech Ageing Dev. 1982;19(4):333–342. doi: 10.1016/0047-6374(82)90017-3. [DOI] [PubMed] [Google Scholar]
- 3.Baker JE, Curry BD, Olinger GN, Gross GJ. Increased tolerance of the chronically hypoxic immature heart to ischemia. Contribution of the KATP channel. Circulation. 1997;95(5):1278–1285. doi: 10.1161/01.cir.95.5.1278. [DOI] [PubMed] [Google Scholar]
- 4.Chambers DJ, Braimbridge MV, Hearse DJ. Free radicals and cardioplegia. Free radical scavengers improve postischemic function of rat myocardium. Eur J Cardiothorac Surg. 1987;1(1):37–45. doi: 10.1016/s1010-7940(87)80012-x. [DOI] [PubMed] [Google Scholar]
- 5.de Jong JW, van der Meer P, Nieukoop AS, Huizer T, Stroeve RJ, Bos E. Xanthine oxidoreductase activity in perfused hearts of various species, including humans. Circ Res. 1990;67(3):770–773. doi: 10.1161/01.res.67.3.770. [DOI] [PubMed] [Google Scholar]
- 6.Ding G, Wiegerinck RF, Shen M, Cojoc A, Zeidenweber CM, Wagner MB. Dopamine increases L-type calcium current more in newborn than adult rabbit cardiomyocytes via D1 and beta2 receptors. Am J Physiol Heart Circ Physiol. 2008;294(5):H2327–H2335. doi: 10.1152/ajpheart.00993.2007. [DOI] [PubMed] [Google Scholar]
- 7.Ding Y, Li YL, Zimmerman MC, Davisson RL, Schultz HD. Role of CuZn superoxide dismutase on carotid body function in heart failure rabbits. Cardiovasc Res. 2009;81(4):678–685. doi: 10.1093/cvr/cvn350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Fischer UM, Tossios P, Huebner A, Geissler HJ, Bloch W, Mehlhorn U. Myocardial apoptosis prevention by radical scavenging in patients undergoing cardiac surgery. J Thorac Cardiovasc Surg. 2004;128(1):103–108. doi: 10.1016/j.jtcvs.2003.11.034. [DOI] [PubMed] [Google Scholar]
- 9.Fitzpatrick CM, Shi Y, Hutchins WC, Su J, Gross GJ, et al. Cardioprotection in chronically hypoxic rabbits persists on exposure to normoxia: role of NOS and KATP channels. Am J Physiol Heart Circ Physiol. 2005;288(1):H62–H68. doi: 10.1152/ajpheart.00701.2004. [DOI] [PubMed] [Google Scholar]
- 10.Gan XT, Cook MA, Moffat MP, Karmazyn M. Protective effects against hydrogen peroxide-induced toxicity by activators of the ATP-sensitive potassium channel in isolated rat hearts. J Mol Cell Cardiol. 1998;30(1):33–41. doi: 10.1006/jmcc.1997.0569. [DOI] [PubMed] [Google Scholar]
- 11.Hoshida S, Aoki K, Nishida M, Yamashita N, Igarashi J, Hori M, et al. Effects of preconditioning with ebselen on glutathione metabolism and stress protein expression. J Pharmacol Exp Ther. 1997;281(3):1471–1475. [PubMed] [Google Scholar]
- 12.Hyslop PA, Hinshaw DB, Halsey WA, Jr, Schraufstatter IU, Sauerheber RD, Spragg RG, et al. Mechanisms of oxidant-mediated cell injury. The glycolytic and mitochondrial pathways of ADP phosphorylation are major intracellular targets inactivated by hydrogen peroxide. J Biol Chem. 1988;263(4):1665–1675. [PubMed] [Google Scholar]
- 13.Ishikawa T, Yamamoto F, Ohashi T, Shimada Y, Kagisaki K, Kumada Y, et al. The effects of Ebselen upon post-ischemic functional recovery in rat heart. Nippon Kyobu Geka Gakkai Zasshi. 1995;43(4):458–465. [PubMed] [Google Scholar]
- 14.Ismail JA, McDonough KH. The role of coronary flow and adenosine in postischemic recovery of septic rat hearts. Am J Physiol. 1998;275(1 Pt 2):H8–H14. doi: 10.1152/ajpheart.1998.275.1.H8. [DOI] [PubMed] [Google Scholar]
- 15.Kloner RA, Przyklenk K, Whittaker P. Deleterious effects of oxygen radicals in ischemia/reperfusion. Resolved and unresolved issues. Circulation. 1989;80(5):1115–1127. doi: 10.1161/01.cir.80.5.1115. [DOI] [PubMed] [Google Scholar]
- 16.Kuroda J, Ago T, Matsushima S, Zhai P, Schneider MD, Sadoshima J. NADPH oxidase 4 (Nox4) is a major source of oxidative stress in the failing heart. Proc Natl Acad Sci USA. 2010;107(35):15565–15570. doi: 10.1073/pnas.1002178107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lancel S, Qin F, Lennon SL, Zhang J, Tong X, Mazzini MJ, et al. Oxidative posttranslational modifications mediate decreased SERCA activity and myocyte dysfunction in Galphaq-overexpressing mice. Circ Res. 2010;107(2):228–232. doi: 10.1161/CIRCRESAHA.110.217570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ley SJ. Myocardial depression after cardiac surgery: pharmacologic and mechanical support. AACN Clin Issues Crit Care Nurs. 1993;4(2):293–308. [PubMed] [Google Scholar]
- 19.Looi YH, Grieve DJ, Siva A, Walker SJ, Anilkumar N, Cave AC, et al. Involvement of Nox2 NADPH oxidase in adverse cardiac remodeling after myocardial infarction. Hypertension. 2008;51(2):319–325. doi: 10.1161/HYPERTENSIONAHA.107.101980. [DOI] [PubMed] [Google Scholar]
- 20.Luo J, Xuan YT, Gu Y, Prabhu SD. Prolonged oxidative stress inverts the cardiac force-frequency relation: role of altered calcium handling and myofilament calcium responsiveness. J Mol Cell Cardiol. 2006;40(1):64–75. doi: 10.1016/j.yjmcc.2005.09.013. [DOI] [PubMed] [Google Scholar]
- 21.Maher KO, Pizarro C, Gidding SS, Januszewska K, Malec E, Norwood WI, Jr, et al. Hemodynamic profile after the Norwood procedure with right ventricle to pulmonary artery conduit. Circulation. 2003;108(7):782–784. doi: 10.1161/01.CIR.0000087338.09589.21. [DOI] [PubMed] [Google Scholar]
- 22.Masse L, Antonacci M. Low cardiac output syndrome: identification and management. Crit Care Nurs Clin North Am. 2005;17(4):375–383. doi: 10.1016/j.ccell.2005.07.005. [DOI] [PubMed] [Google Scholar]
- 23.Maulik N, Baker JE, Engelman RM, Das DK. Postnatal developmental profiles of antioxidant enzymes in heart. Ann N Y Acad Sci. 1996;793:439–448. doi: 10.1111/j.1749-6632.1996.tb33538.x. [DOI] [PubMed] [Google Scholar]
- 24.Milano CA, White WD, Smith LR, Jones RH, Lowe JE, Smith PK, et al. Coronary artery bypass in patients with severely depressed ventricular function. Ann Thorac Surg. 1993;56(3):487–493. doi: 10.1016/0003-4975(93)90884-k. [DOI] [PubMed] [Google Scholar]
- 25.Mozaffari MS, Baban B, Liu JY, Abebe W, Sullivan JC, El-Marakby A. Mitochondrial complex I and NAD(P)H oxidase are major sources of exacerbated oxidative stress in pressure-overloaded ischemic-reperfused hearts. Basic Res Cardiol. 2011;106(2):287–297. doi: 10.1007/s00395-011-0150-7. [DOI] [PubMed] [Google Scholar]
- 26.Osaka T, Joyner RW. Developmental changes in the beta-adrenergic modulation of calcium currents in rabbit ventricular cells. Circ Res. 1992;70(1):104–115. doi: 10.1161/01.res.70.1.104. [DOI] [PubMed] [Google Scholar]
- 27.Osaka T, Joyner RW, Kumar R. Postnatal decrease in muscarinic cholinergic influence on Ca2+ currents of rabbit ventricular cells. Am J Physiol. 1993;264(6 Pt 2):H1916–H1925. doi: 10.1152/ajpheart.1993.264.6.H1916. [DOI] [PubMed] [Google Scholar]
- 28.Parrish MD, Payne A, Fixler DE. Global myocardial ischemia in the newborn, juvenile, and adult isolated isovolumic rabbit heart. Age-related differences in systolic function, diastolic stiffness, coronary resistance, myocardial oxygen consumption, and extracellular pH. Circ Res. 1987;61(5):609–615. doi: 10.1161/01.res.61.5.609. [DOI] [PubMed] [Google Scholar]
- 29.Pauliks LB, Undar A, Clark JB, Myers JL. Segmental differences of impaired diastolic relaxation following cardiopulmonary bypass surgery in children: a tissue Doppler study. Artif Organs. 2009;33(11):904–908. doi: 10.1111/j.1525-1594.2009.00923.x. [DOI] [PubMed] [Google Scholar]
- 30.Pendergrass KD, Varghese ST, Maiellaro-Rafferty K, Brown ME, Taylor WR, Davis ME. Temporal effects of catalase overexpression on healing after myocardial infarction. Circ Heart Fail. 2011;4(1):98–106. doi: 10.1161/CIRCHEARTFAILURE.110.957712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Pizarro C, Malec E, Maher KO, Januszewska K, Gidding SS, Murdison KA, et al. Right ventricle to pulmonary artery conduit improves outcome after stage I Norwood for hypoplastic left heart syndrome. Circulation. 2003;108(1):II155–II160. doi: 10.1161/01.cir.0000087390.94142.1d. [DOI] [PubMed] [Google Scholar]
- 32.Quaglietta D, Belanger MP, Wittnich C. Ventricle-specific metabolic differences in the newborn piglet myocardium in vivo and during arrested global ischemia. Pediatr Res. 2008;63(1):15–19. doi: 10.1203/PDR.0b013e31815b4842. [DOI] [PubMed] [Google Scholar]
- 33.Ravishankar C, Tabbutt S, Wernovsky G. Critical care in cardiovascular medicine. Curr Opin Pediatr. 2003;15(5):443–453. doi: 10.1097/00008480-200310000-00001. [DOI] [PubMed] [Google Scholar]
- 34.Rowland RT, Meng X, Ao L, Terada LS, Harken AH, Brown JM. Mechanisms of immature myocardial tolerance to ischemia: phenotypic differences in antioxidants, stress proteins, and oxidases. Surgery. 1995;118(2):446–452. doi: 10.1016/s0039-6060(05)80357-5. [DOI] [PubMed] [Google Scholar]
- 35.Seshadri G, Sy JC, Brown M, Dikalov S, Yang SC, Murthy N, et al. The delivery of superoxide dismutase encapsulated in polyketal microparticles to rat myocardium and protection from myocardial ischemia-reperfusion injury. Biomaterials. 2010;31(6):1372–1379. doi: 10.1016/j.biomaterials.2009.10.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Shuhaiber HJ, Juggi JS, John V, Yousof AM, Braveny P. Differences in the recovery of right and left ventricular function after ischaemic arrest and cardioplegia. Eur J Cardiothorac Surg. 1990;4(8):435–440. doi: 10.1016/1010-7940(90)90074-a. [DOI] [PubMed] [Google Scholar]
- 37.Sorescu D, Griendling KK. Reactive oxygen species, mitochondria, and NAD(P)H oxidases in the development and progression of heart failure. Congest Heart Fail. 2002;8(3):132–140. doi: 10.1111/j.1527-5299.2002.00717.x. [DOI] [PubMed] [Google Scholar]
- 38.Srivastava S, Chandrasekar B, Gu Y, Luo J, Hamid T, Hill BG, et al. Downregulation of CuZn-superoxide dismutase contributes to beta-adrenergic receptor-mediated oxidative stress in the heart. Cardiovasc Res. 2007;74(3):445–455. doi: 10.1016/j.cardiores.2007.02.016. [DOI] [PubMed] [Google Scholar]
- 39.Sy JC, Seshadri G, Yang SC, Brown M, Oh T, Dikalov S, et al. Sustained release of a p38 inhibitor from non-inflammatory microspheres inhibits cardiac dysfunction. Nat Mater. 2008;7(11):863–868. doi: 10.1038/nmat2299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Sy JC, Phelps EA, Garcia AJ, Murthy N, Davis ME. Surface functionalization of polyketal microparticles with nitrilotriacetic acid-nickel complexes for efficient protein capture and delivery. Biomaterials. 2010;31(18):4987–4994. doi: 10.1016/j.biomaterials.2010.02.063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Tanaka M, Mokhtari GK, Terry RD, Balsam LB, Lee KH, Kofidis T, et al. Overexpression of human copper/zinc superoxide dismutase (SOD1) suppresses ischemia–reperfusion injury and subsequent development of graft coronary artery disease in murine cardiac grafts. Circulation. 2004;110(11 Suppl 1):II200–II206. doi: 10.1161/01.CIR.0000138390.81640.54. [DOI] [PubMed] [Google Scholar]
- 42.Venardos K, Ashton K, Headrick J, Perkins A. Effects of dietary selenium on post-ischemic expression of antioxidant mRNA. Mol Cell Biochem. 2005;270(1–2):131–138. doi: 10.1007/s11010-005-5279-y. [DOI] [PubMed] [Google Scholar]
- 43.Wessel DL. Managing low cardiac output syndrome after congenital heart surgery. Crit Care Med. 2001;29(10 Suppl):S220–S230. doi: 10.1097/00003246-200110001-00004. [DOI] [PubMed] [Google Scholar]
- 44.Wittnich C. Age-related differences in myocardial metabolism affects response to ischemia. Age in heart tolerance to ischemia. Am J Cardiovasc Pathol. 1992;4(2):175–180. [PubMed] [Google Scholar]
- 45.Wittnich C, Peniston C, Ianuzzo D, Abel JG, Salerno TA. Relative vulnerability of neonatal and adult hearts to ischemic injury. Circulation. 1987;76(5 Pt 2):V156–V160. [PubMed] [Google Scholar]
- 46.Wittnich C, Wallen WJ, Belanger MP, Ikonomidis JS. Extracellular calcium concentration affects susceptibility to global ischemic injury in newborn but not adult hearts. J Heart Lung Transpl. 1999;18(7):675–683. doi: 10.1016/s1053-2498(99)00026-1. [DOI] [PubMed] [Google Scholar]
- 47.Wittnich C, Belanger MP, Bandali K. Are there ventricle-specific postnatal maturational differences in myocardial beta-adrenoceptors? Can J Physiol Pharmacol. 2006;84(8–9):859–865. doi: 10.1139/y06-026. [DOI] [PubMed] [Google Scholar]
- 48.Wittnich C, Su J, Boscarino C, Belanger M. Age-related differences in myocardial hydrogen ion buffering during ischemia. Mol Cell Biochem. 2006;285(1–2):61–67. doi: 10.1007/s11010-005-9055-9. [DOI] [PubMed] [Google Scholar]
- 49.Wittnich C, Belanger MP, Bandali KS. Newborn hearts are at greater ‘metabolic risk’ during global ischemia—advantages of continuous coronary washout. Can J Cardiol. 2007;23(3):195–200. doi: 10.1016/s0828-282x(07)70743-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Yang SC, Bhide M, Crispe IN, Pierce RH, Murthy N. Polyketal copolymers: a new acid-sensitive delivery vehicle for treating acute inflammatory diseases. Bioconjug Chem. 2008;19(6):1164–1169. doi: 10.1021/bc700442g. [DOI] [PMC free article] [PubMed] [Google Scholar]