Abstract
Background. Many studies have reported an association between tea drinking and Parkinson's disease (PD). Our purpose is to summarize the available information and evaluate the risk of PD associated with tea drinking. Methods. We searched all publications in English language on the association of tea drinking and PD risk published up to December 2010. The pooled analysis was performed with Review Manager 5.0. Results. In total, eight articles including 1418 cases and 4250 controls were included in the meta-analysis. The pooled odds ratio (95% CI) was 0.85 (0.74–0.98), which suggests the protective effect of tea drinking in PD risks. Moreover, the summary OR (OR: 0.83, 95% CI = 0.69–0.99) for drinkers of ≤1 cup of tea per day versus nonconsumers and that (OR: 0.96, 95% CI = 0.73–1.27) for drinkers of >1 cups of tea per day versus nonconsumers showed that there was not an apparent dose-response relationship. No indication for publication bias was found. Conclusions. This meta-analysis showed that tea drinking can lower the risk of PD, while no apparent dose-response relationship was found. Further effort is needed to fully understand the mechanism underlying the beneficial effect of tea consumption in lowering PD risk.
1. Introduction
Parkinson's disease (PD) is a common neurodegenerative disorder, and, with the accelerated aging of human society, the prevalence of PD is expected to rise steadily in future [1, 2]. Despite the pathogenesis of PD is not fully elucidated, many epidemiological factors, such as coffee drinking and smoking, have been demonstrated to lower the risk of PD [3–5]. Tea is one of the most popular beverages worldwide, and the effects of tea consumption on PD risk have attracted much attention in recent years [6–17], as the tea components, such as flavonoids, caffeine, and theanine, have been proven to be neuroprotective in animal models of PD. Many studies have found that tea drinking can lower the risk of PD [6–10, 12–15], while this beneficial effect of tea is not observed in other reports [11, 16]. The purpose of the present study was to perform a meta-analysis to provide a comprehensive conclusion on the association between tea drinking and PD risk, which has important implications for the prevention and treatment of this disease.
2. Methods
2.1. Information Retrieval
We identified procurable published studies in English through a computerized MEDLINE search from 1966 to December 2010 and also the relational document links. We used the following medical subject headings and free text words to search the PubMed database: (TEA) AND (PARKINSON OR PD) AND (CASE-CONTROL OR CASE-REFERENT OR RETROSPECTIVE OR COHORT OR FOLLOW-UP OR INCIDENCE OR PROSPECTIVE OR EPIDEMIOLOG). We also examined conference proceedings; the references in the articles retrieved through the computerized search.
2.2. Selection Criteria
The identified studies must meet the following criteria: (i) the original data includes the number of PD cases studied and the odds ratio (OR) or relative risk (RR), (ii) its corresponding 95% confidence interval (CI) is for highest versus non-/lowest level of tea intake, and (iii) these studies must define the outcome of interest as incident PD based on clearly stated diagnostic criteria or identified through diagnostic codes with additional confirmation. In total, eight published articles were included in the meta-analysis [8–15]. The details of search strategy and study selection processes were shown in Figure 1.
Figure 1.
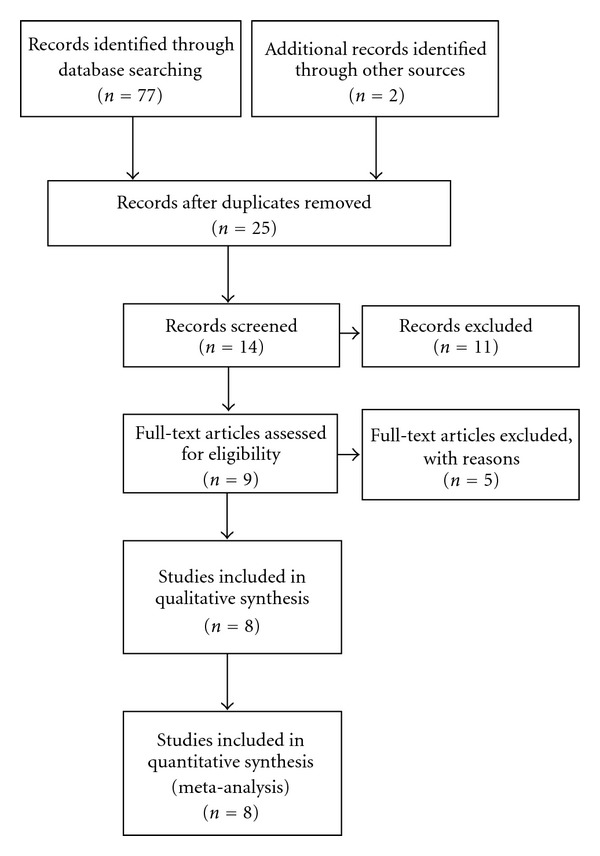
Selection of studies for inclusion in the present meta-analysis.
2.3. Data Extraction and Meta-Analysis
We extracted information from each paper including first author, year, and country of publication, the number of cases and controls, the adjusted RRs or ORs, and 95% CI. We examined possible heterogeneity in the identified result. If the P value >0.10, it means that the study does not have statistical significance. We used the fixed effect model to calculate the summary OR and its 95% CI across homogeneous studies and used the random effect model to calculate the summary OR and its 95% CI across heterogeneous studies. Egger's weighted regression method was employed to evaluate P value for publication bias. Finally, stratified analyses were conducted to examine the differences by cups of drinking. Statistical computation was performed using the Review Manager 5.0 statistical software.
3. Results
3.1. Tea Drinking and PD Risks
The eight case-control studies (including 1418 cases and 4250 controls) which met the eligibility criteria were conducted in 5 countries between 1966 and 2010. Table 1 with the extracted information present the relative risk for each of studies along with their summary OR derived from the meta-analysis. Figure 2 presents the forest plot of comparison of tea drinkers versus non-consumers. There was statistically heterogeneity among the eight results (P = 0.04). The analyses compared exposed with unexposed for tea drinking, and the overall summary OR based on all studies indicated that tea drinking can protect PD (summary OR = 0.85, 95% CI = 0.85–0.98) with eight cases.
Table 1.
Tea drinking and PD risk.
| Author | Year of publication | Country | Cases | Controls | Relative risk | 95% CI | ||
|---|---|---|---|---|---|---|---|---|
| N | n | N | n | |||||
| Haack et al. [8] | 1981 | USA | 237 | 223 | 474 | 456 | 0.63 | [0.31, 1.29] |
| Ho et al. [9] | 1989 | China | 34 | 21 | 105 | 70 | 0.81 | [0.36, 1.80] |
| Fall et al. [10] | 1999 | Sweden | 113 | 64 | 263 | 174 | 0.67 | [0.43, 1.05] |
| Preux et al. [11] | 2000 | France | 140 | 40 | 280 | 54 | 1.67 | [1.04, 2.68] |
| Paganini-Hill [12] | 2001 | USA | 395 | 182 | 2320 | 1126 | 0.91 | [0.73, 1.12] |
| Morano et al. [13] | 1994 | Spain | 74 | 6 | 148 | 6 | 2.09 | [0.65, 6.71] |
| Chan et al. [14] | 1998 | China | 215 | 113 | 313 | 196 | 0.66 | [0.46, 0.94] |
| Checkoway et al. [15] | 2002 | USA | 210 | 72 | 347 | 145 | 0.73 | [0.51, 1.04] |
| Total | 1418 | 4250 | 0.85 | [0.74, 0.98] | ||||
Test for heterogeneity among all studies: Q = 15.03 based on 7 degrees of freedom P = 0.04. Summary OR was based on fixed effect models. N: number of cases/controls, n : number of tea drinkers in cases/controls.
Figure 2.
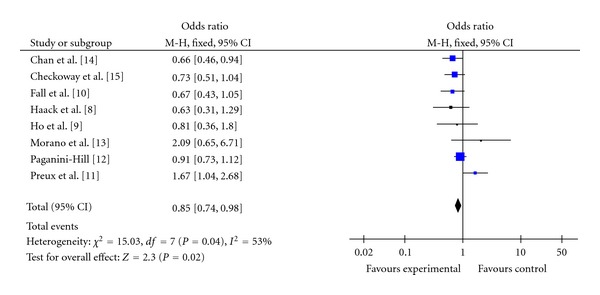
Pooled estimate of OR and 95% CI of PD risk for tea drinking. Blue diamonds indicate adjusted OR in each study. Open diamonds are pooled OR. Horizontal line represents 95% CI. Studies are ordered by alphabetical order of last name of first author.
3.2. The Dose Effect Relationship of Tea Drinking and PD Risks
Among the eight studies that included in our present analysis, three reported the results for drinkers of ≤1 and >1 cup of tea per day versus non-consumers. [14, 16, 18]. The information according to the number of cups of tea drinking daily was included in Table 2. It can be seen that, for drinkers of ≤1 cup of tea per day versus non-consumers, the summary OR (OR: 0.83, 95% CI = 0.69–0.99) showed a protective effect against PD (Figure 3). For drinkers of >1 cup of tea per day versus non-consumers, the summary OR (OR: 0.96, 95% CI = 0.73–1.27) also showed a protective effect (Figure 4). According to the present results, we concluded that there was not an apparent dose-response relationship.
Table 2.
The dose effect relationship of tea drinking and PD risk.
| Author | Year of publication | Country | Cases | Controls | Relative risk | 95% CI | ||
|---|---|---|---|---|---|---|---|---|
| N | n | N | n | |||||
| Drinkers of ≤1 cup per day of tea versus non-consumers studies | ||||||||
| Fall et al. [10] | 1999 | Sweden | 113 | 57 | 263 | 143 | 0.85 | [0.55, 1.33] |
| Paganini-Hill [12] | 2001 | USA | 395 | 132 | 2320 | 892 | 0.80 | [0.64, 1.01] |
| Checkoway et al. [15] | 2002 | USA | 210 | 61 | 347 | 110 | 0.88 | [0.61, 1.28] |
| Total | 718 | 2930 | 0.83 | [0.69, 0.99] | ||||
|
| ||||||||
| Drinkers of >1 cups per day of tea versus non-consumers studies | ||||||||
| Fall et al. [10] | 1999 | Sweden | 113 | 7 | 263 | 31 | 0.49 | [0.21, 1.16] |
| Paganini-Hill [12] | 2001 | USA | 395 | 50 | 2320 | 234 | 1.29 | [0.93, 1.79] |
| Checkoway et al. [15] | 2002 | USA | 210 | 11 | 347 | 35 | 0.49 | [0.24, 0.99] |
| Total | 718 | 2930 | 0.96 | [0.73, 1.27] | ||||
N: number of cases/controls, n: number of tea drinkers in cases/controls.
Figure 3.
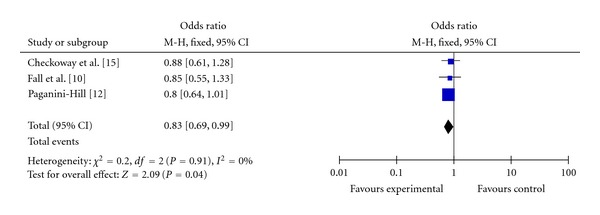
Pooled estimate of OR and 95% CI of PD risk for tea drinking of ≤1 cup per day. Blue diamonds indicate adjusted OR in each study. Open diamonds are pooled OR. Horizontal line represents 95% CI. Studies are ordered by alphabetical order of the last name of first author.
Figure 4.
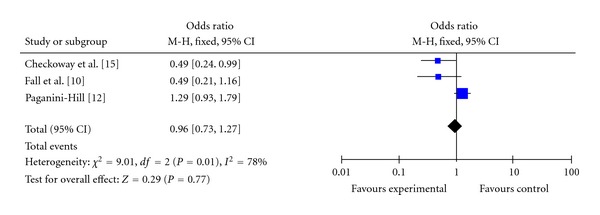
Pooled estimate of OR and 95% CI of PD risk for tea drinking of >1 cups per day. Blue diamonds indicate adjusted OR in each study. Open diamonds are pooled OR. Horizontal line represents 95% CI. Studies are ordered by alphabetical order of the last name of first author.
3.3. Publication Bias
Figure 5 shows the funnel plot of tea drinking and the risk of PD, in which, the logOR from each study is plotted on the horizontal axis, and its standard error is plotted on the vertical axis. Inspection of the Begg funnel plot did not find the presence of publication bias because the logORs plotted against their standard errors yielded a symmetric distribution.
Figure 5.
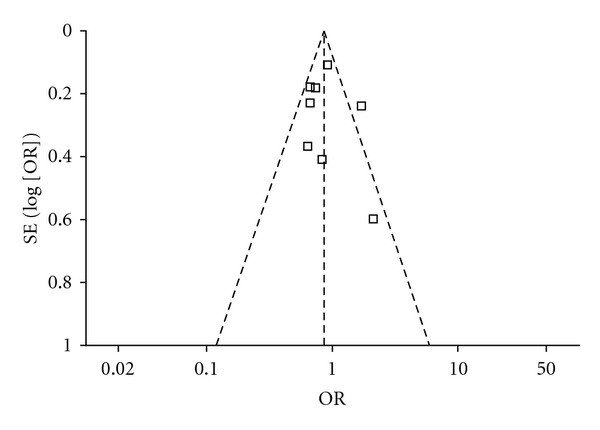
Funnel plot of tea drinking and the risk of PD. The log OR from each study is plotted on the horizontal axis, and its standard error is plotted on the vertical axis.
4. Discussion
As we know, PD is a common neurodegenerative disorder in middle- and old-aged crowd. The etiology of PD has long been thought to involve many factors, and there were five major hypotheses regarding the pathogenesis of PD, such as the theory of genetic factors, the theory of environmental factors, immunity doctrine, apoptosis theory, and the oxidative stress theory [18–22]. In oxidative stress theory, oxidative stress contributes to the cascade leading to dopamine cell degeneration in PD. Oxidative stress can impair altered ubiquitination and degradation of proteins directly, which have been implicated as key to dopaminergic cell death in PD, and products of oxidative damage (e.g., 4-hydroxynonenal) can damage the 26S proteasome [23].
Tea is one of the most ancient and popular beverages consumed around the world. There are many important substances in tea leaves, such as polyphenols, methylxanthine, caffeine, different lipids, amino acids, mineral substances, and volatile compounds. The demonstrated health benefits of tea include weight loss, preventing heart disease and cancer, and so forth [24]. For example, epigallocatechin gallate (EGCG), one of the most important antioxidant ingredients of green tea, is responsible for green tea's weight loss effect by increasing metabolism and the body's ability to burn fat [25]. Polyphenols in tea have been proven to inhibit tumor cell proliferation and induce apoptosis [26, 27]. Moreover, in in vitro study and experimental animal model, it has been identified that tea drinking can obviously reduce the formation of molecular products of various small lipid peroxidation and induce antioxidative enzyme activities [28].
In recent years, there were more and more studies devoted to exploring the effects of tea consumption on PD risk. Many studies reported that tea drinking can lower the risk of PD [6–10, 12–15], while this protective effect of tea is not demonstrated by other groups [11, 16]. With the aim to evaluate the association between tea drinking and PD risk, a meta-analysis was conducted based on published studies. The estimated summary OR is 0.85, which donates that tea drinking can decrease the risk of PD. This is consistent with the results of the similar study by Quintana et al. [6] and, thus, further confirms the relation of tea consumption and PD. The mechanisms underlying the protective effects of tea drinking against PD risk should be multiple. In recent years, many natural products have been proposed to be multipotent agent to combat neurodegenerative diseases [29–32]. Firstly, through synthesizing the studies in vitro and experimental animal model and the epidemiological studies we collected, we can infer that the antioxidant effect of tea components, especially polyphones, should be the predominant mechanism of tea against PD. Tea polyphones, such as EGCG, have been found to exhibit neuroprotective effect and the mechanism of the neuroprotection may involve three aspects: antioxidant, anti-inflammatory and iron-chelating activities [29, 33, 34]. Secondly, many other biological activities of tea components may contribute to the beneficial effect of tea in lowering PD risk. For instance, it was found that theanine can protect the brain by promoting the secretion of dopamine [35]. Caffeine has been demonstrated to be an adenosine A2A receptor antagonist that may enhance locomotor activity in animal models of PD and improve motor function in patients with PD [36]. In addition, Tan et al. proposed that flavonoids in tea with their anti-inflammatory effects on the cardiovascular system may help ward off PD by increasing circulation to the brain [16].
Moreover, according to the present meta-analysis, we did not find an apparent dose-response relationship as expected. It may arise from the following factors: (i) black tea and green tea differ markedly in the nature of their polyphenols [37, 38], while there were only few studies reporting stratified results according to the types of tea and (ii) the contents of the bioactive compounds in tea may fluctuate because of differences in producing areas, materials, and manufacturing [37, 38].
There were heterogeneity and limitations in our study. Firstly, the population studied in the included references was small and their location was not widespread, which make us unable to perform analysis on race. Secondly, other factors with possible influence on disease progression such as anti-Parkinsonian drugs, food supplements, or alcohol consumption were not considered. Thirdly, there were only few studies reporting stratified results according to specific types of tea (black or green) and the gender of the population studied. If more data is available in future for performing stratified analyses according to the types of tea, through comparing the components of the specific types of tea, we can identify the most important compounds, which will be of significance to the prevention and treatment of PD.
To summarize, our findings indicate that tea drinking can protect against PD, which leads human to value the impact of lifestyle on health. Despite the mechanisms behind this association should be multiple, in view of the widely accepted pathogenic role of oxidative stress in PD and the well-documented antioxidant potential of bioactive tea components, the antioxidant activity is expected to be an important aspect. Further effort is encouraged to fully understand the comprehensive mechanism underlying the beneficial effect of tea consumption in lowering PD risk.
Acknowledgment
This work was supported by the National Natural Science Foundation of China (Grant nos. 30800184 and 30700113).
References
- 1.Polymeropoulos MH, Lavedan C, Leroy E, et al. Mutation in the alphasynuclein gene identified in families with Parkinson’s disease. Science. 1997;276(5321):2045–2047. doi: 10.1126/science.276.5321.2045. [DOI] [PubMed] [Google Scholar]
- 2.Lang AE, Lozano AM. Parkinson’s disease. The New England Journal of Medicine. 1998;339(15):1044–1053. doi: 10.1056/NEJM199810083391506. [DOI] [PubMed] [Google Scholar]
- 3.Wirdefeldt K, Adami HO, Cole P, Trichopoulos D, Mandel J. Epidemiology and etiology of Parkinson’s disease: a review of the evidence. European Journal of Epidemiology. 2011;26(supplement 1):S1–S58. doi: 10.1007/s10654-011-9581-6. [DOI] [PubMed] [Google Scholar]
- 4.Hernán MA, Takkouche B, Gestal-Otero JJ, Caamaño-Isorna F. A meta-analysis of coffee drinking, cigarette smoking, and the risk of Parkinson’s disease. Annals of Neurology. 2002;52(3):276–284. doi: 10.1002/ana.10277. [DOI] [PubMed] [Google Scholar]
- 5.Cao GH, Sofic E, Prior RL. Antioxidant capacity of tea and common vegetables. Journal of Agricultural and Food Chemistry. 1996;44(11):3426–3431. [Google Scholar]
- 6.Quintana JLB, Allam MF, del Castillo AS, Navajas RFC. Parkinson’s disease and tea: a quantitative review. Journal of the American College of Nutrition. 2009;28(1):1–6. doi: 10.1080/07315724.2009.10719754. [DOI] [PubMed] [Google Scholar]
- 7.Ascherio A, Zhang SM, Hernán MA, et al. Prospective study of caffeine consumption and risk of Parkinson’s disease in men and women. Annals of Neurology. 2001;50(1):56–63. doi: 10.1002/ana.1052. [DOI] [PubMed] [Google Scholar]
- 8.Haack DG, Baumann RJ, McKean HE. Nicotine exposure and parkinson disease. American Journal of Epidemiology. 1981;114(2):191–200. doi: 10.1093/oxfordjournals.aje.a113182. [DOI] [PubMed] [Google Scholar]
- 9.Ho SC, Woo J, Lee CM. Epidemiologic study of Parkinson’s disease in Hong Kong. Neurology. 1989;39(10):1314–1318. doi: 10.1212/wnl.39.10.1314. [DOI] [PubMed] [Google Scholar]
- 10.Fall PA, Fredrikson M, Axelson O, Granérus AK. Nutritional and occupational factors influencing the risk of Parkinson’s disease: a case-control study in southeastern Sweden. Movement Disorders. 1999;14(1):28–37. doi: 10.1002/1531-8257(199901)14:1<28::aid-mds1007>3.0.co;2-o. [DOI] [PubMed] [Google Scholar]
- 11.Preux PM, Condet A, Anglade C, et al. Parkinson’s disease and environmental factors: matched case-control study in the Limousin region, France. Neuroepidemiology. 2000;19(6):333–337. doi: 10.1159/000026273. [DOI] [PubMed] [Google Scholar]
- 12.Paganini-Hill A. Risk factors for Parkinson’s disease: the Leisure World cohort study. Neuroepidemiology. 2001;20(2):118–124. doi: 10.1159/000054770. [DOI] [PubMed] [Google Scholar]
- 13.Morano A, Jiménez-Jiménez FJ, Molina JA, Antolin MA. Risk-factors for Parkinson’s disease: case-control study in the province of Caceres, Spain. Acta Neurologica Scandinavica. 1994;89(3):164–170. doi: 10.1111/j.1600-0404.1994.tb01655.x. [DOI] [PubMed] [Google Scholar]
- 14.Chan DK, Woo J, Ho SC, et al. Genetic and environmental risk factors for Parkinson’s disease in a Chinese population. Journal of Neurology Neurosurgery and Psychiatry. 1998;65(5):781–784. doi: 10.1136/jnnp.65.5.781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Checkoway H, Powers K, Smith-Weller T, Franklin GM, Longstreth WT, Swanson PD. Parkinson’s disease risks associated with cigarette smoking, alcohol consumption, and caffeine intake. American Journal of Epidemiology. 2002;155(8):732–738. doi: 10.1093/aje/155.8.732. [DOI] [PubMed] [Google Scholar]
- 16.Tan LC, Koh WP, Yuan JM, et al. Differential effects of black versus green tea on risk of Parkinson’s disease in the Singapore Chinese health study. American Journal of Epidemiology. 2008;167(5):553–560. doi: 10.1093/aje/kwm338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zayed J, Ducic S, Campanella G, et al. Environmental factors in the etiology of Parkinson’s disease. Canadian Journal of Neurological Sciences. 1990;17(3):286–291. [PubMed] [Google Scholar]
- 18.Volles MJ, Lee SJ, Rochet JC, et al. Vesicle permeabilization by protofibrillar α-synuclein: implications for the pathogenesis and treatment of Parkinson’s disease. Biochemistry. 2001;40(26):7812–7819. doi: 10.1021/bi0102398. [DOI] [PubMed] [Google Scholar]
- 19.Liou HH, Tsai MC, Chen CJ, et al. Environmental risk factors and Parkinson’s disease: a case-control study in Taiwan. Neurology. 1997;48(6):1583–1588. doi: 10.1212/wnl.48.6.1583. [DOI] [PubMed] [Google Scholar]
- 20.Le WD, Rowe DB, Jankovic J, Xie W, Appel SH. Effects of cerebrospinal fluid from patients with Parkinson disease on dopaminergic cells. Archives of Neurology. 1999;56(2):194–200. doi: 10.1001/archneur.56.2.194. [DOI] [PubMed] [Google Scholar]
- 21.Viswanath V, Wu Y, Boonplueang R, et al. Caspase-9 activation results in downstream caspase-8 activation and bid cleavage in 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced Parkinson’s disease. Journal of Neuroscience. 2001;21(24):9519–9528. doi: 10.1523/JNEUROSCI.21-24-09519.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bharath S, Hsu M, Kaur D, Rajagopalan S, Andersen JK. Glutathione, iron and Parkinson’s disease. Biochemical Pharmacology. 2002;64(5-6):1037–1048. doi: 10.1016/s0006-2952(02)01174-7. [DOI] [PubMed] [Google Scholar]
- 23.Jenner P. Oxidative stress in Parkinson’s disease. Annals of Neurology. 2003;53(3):S26–S36. doi: 10.1002/ana.10483. [DOI] [PubMed] [Google Scholar]
- 24.Yang CS, Landau JM. Effects of tea consumption nutrition health. Journal of Nutrition. 2000;130(10):2409–2412. doi: 10.1093/jn/130.10.2409. [DOI] [PubMed] [Google Scholar]
- 25.Kao YH, Chang HH, Lee MJ, Chen CL. Tea, obesity, and diabetes. Molecular Nutrition and Food Research. 2006;50(2):188–210. doi: 10.1002/mnfr.200500109. [DOI] [PubMed] [Google Scholar]
- 26.Seeram NP, Henning SM, Niu Y, Lee R, Scheuller HS, Heber D. Catechin and caffeine content of green tea dietary supplements and correlation with antioxidant capacity. Journal of Agricultural and Food Chemistry. 2006;54(5):1599–1603. doi: 10.1021/jf052857r. [DOI] [PubMed] [Google Scholar]
- 27.Lambert JD, Yang CS. Mechanisms of cancer prevention by tea constituents. Journal of Nutrition. 2003;133(supplement 10):3262S–3267S. doi: 10.1093/jn/133.10.3262S. [DOI] [PubMed] [Google Scholar]
- 28.Yen GC, Chen HY. Antioxidant activity of various tea extracts in relation to their antimutagenicity. Journal of Agricultural and Food Chemistry. 1995;43(1):27–32. [Google Scholar]
- 29.Ji HF, Zhang HY. Multipotent natural agents to combat Alzheimer’s disease. Functional spectrum and structural features. Acta Pharmacologica Sinica. 2008;29(2):143–151. doi: 10.1111/j.1745-7254.2008.00752.x. [DOI] [PubMed] [Google Scholar]
- 30.Ji HF, Shen L. Berberine: a potential multipotent natural product to combat Alzheimer’s disease. Molecules. 2011;16(8):6732–6740. doi: 10.3390/molecules16086732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ji HF, Li XJ, Zhang HY. Natural products and drug discovery: can thousands of years of ancient medical knowledge lead us to new and powerful drug combinations in the fight against cancer and dementia? EMBO Reports. 2009;10(3):194–200. doi: 10.1038/embor.2009.12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Shen L, Zhang HY, Ji HF. A theoretical study on Cu(II)-chelating properties of curcumin and its implications for curcumin as a multipotent agent to combat Alzheimer’s disease. Journal of Molecular Structure: THEOCHEM. 2005;757(1–3):199–202. [Google Scholar]
- 33.Weinreb O, Mandel S, Amit T, Youdim MBH. Neurological mechanisms of green tea polyphenols in Alzheimer’s and Parkinson’s diseases. Journal of Nutritional Biochemistry. 2004;15(9):506–516. doi: 10.1016/j.jnutbio.2004.05.002. [DOI] [PubMed] [Google Scholar]
- 34.Ji HF, Zhang HY. Theoretical evaluation of flavonoids as multipotent agents to combat Alzheimer’s disease. Journal of Molecular Structure: THEOCHEM. 2006;767(1–3):3–9. [Google Scholar]
- 35.Cho HS, Kim S, Lee SY, Park JA, Kim SJ, Chun HS. Protective effect of the green tea component, L-theanine on environmental toxins-induced neuronal cell death. NeuroToxicology. 2008;29(4):656–662. doi: 10.1016/j.neuro.2008.03.004. [DOI] [PubMed] [Google Scholar]
- 36.Kachroo A, Orlando LR, Grandy DK, Chen JF, Young AB, Schwarzschild MA. Interactions between metabotropic glutamate 5 and adenosine A2A receptors in normal and parkinsonian mice. Journal of Neuroscience. 2005;25(45):10414–10419. doi: 10.1523/JNEUROSCI.3660-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Shen L. Tea intake and liver diseases. Hepatology. 2011;53(1):373–374. doi: 10.1002/hep.23872. [DOI] [PubMed] [Google Scholar]
- 38.Crozier A, Jaganath IB, Clifford MN. Dietary phenolics: chemistry, bioavailability and effects on health. Natural Product Reports. 2009;26(8):1001–1043. doi: 10.1039/b802662a. [DOI] [PubMed] [Google Scholar]


