Abstract
OBJECTIVE
To investigate the association between prostate cancer, diabetes, and long-term general and cancer-specific health-related quality of life (HRQL) in a cohort of men with non-metastatic prostate cancer.
PATIENTS AND METHODS
We used data from self-administered surveys to assess the HRQL of men with localized or locally advanced disease at 6 (baseline), 12, 24, and 60 months after initial diagnosis.
We examined changes in general and cancer-specific HRQL with repeated measures analyses using a mixed-model approach.
RESULTS
In total, we evaluated 1811 men, including 13% with prevalent (pre-prostate cancer diagnosis) diabetes, 12% with incident (post-prostate cancer diagnosis) diabetes, and 75% who never reported being diagnosed with diabetes.
Generally, men with prevalent diabetes had the poorest scores on general HRQL and non-diabetic men the best scores, independent of treatment.
Similarly, men with prevalent diabetes had the lowest urinary control and sexual function scores over time, while men without diabetes had the highest scores. Men with incident diabetes reported intermediate scores.
CONCLUSIONS
Prostate cancer survivors with comorbid diabetes have poorer general and cancer-specific HRQL than those without diabetes.
Keywords: diabetes mellitus, health-related quality of life, prostate cancer, symptoms
INTRODUCTION
Type 2 diabetes mellitus and prostate cancer often occur concurrently in men over 60 years [1]. The comorbidity of diabetes can affect prostate cancer treatment decision-making [1,2] and influence clinical outcomes such as survival [3] and health-related quality of life (HRQL).
Men with diabetes at the time of their prostate cancer diagnosis are less likely to receive aggressive treatment [3,4]. Previous studies have shown that prostate cancer survivors with diabetes report worse urinary and sexual function, more symptom bother [5,6], and poorer general health and vitality [7]. However, these reports were limited either by their cross-sectional study design or singular focus on a specific treatment method for localized prostate cancer. Furthermore, no distinction was made between prevalent diabetes (present when prostate cancer was diagnosed) or incident diabetes (diagnosed after the prostate cancer diagnosis, with or without treatment). This is an important limitation because androgen deprivation therapy (ADT), which is commonly used as a treatment for prostate cancer, may lead to diabetes. Specifically, a large population-based study of 73 196 men reported that prostate cancer survivors who received ADT increased their risk of developing diabetes by 40% [8].
Few longitudinal and population-based studies have evaluated the effect of comorbid conditions on general and cancer-specific HRQL in men with prostate cancer. Previous studies have shown that prostate cancer survivors with moderate or severe cardiovascular disease (CVD) had a poorer HRQL compared with those without CVD over 4 years of follow-up [9,10], and that having diabetes and a greater body mass index were more likely to result in significant declines in urinary function over time than having either diabetes or greater, BMI alone [11]. We used data from the longitudinal, population-based Prostate Cancer Outcomes Study (PCOS) [12] to assess the relationship of prevalent and incident diabetes to general and cancer-specific HRQL in men with non-metastatic prostate cancer, followed up for 5 years from the time of diagnosis.
PATIENTS AND METHODS
SETTING AND PARTICIPANTS
This is a secondary analysis of data from the PCOS, investigating urinary, sexual function, bowel function and HRQL in patients with prostate cancer. A more detailed description of the study methodology has been reported elsewhere [13]. Briefly, subjects were identified through six sites participating in the National Cancer Institute’s Surveillance, Epidemiology and End Results (SEER) program. African-American, Caucasian and Hispanic men diagnosed with prostate cancer from 1 October 1994 to 31 October 1995 who resided in an area covered by one of six SEER cancer registries (the states of Connecticut, New Mexico and Utah, and the metropolitan areas of Atlanta, GA, Los Angeles County, CA, and King County, WA) were eligible for inclusion in the PCOS. All men aged 39 to 89 years were eligible except in King County, where inclusion was limited to men aged 60 to 89 years. Subjects were identified within 6 months of diagnosis using a rapid case ascertainment system. This study was approved by the local institutional review boards.
DATA COLLECTION AND HRQL QUESTIONNAIRES
Eligible patients were contacted by post (90%) or telephone (10%) up to 6 months after the date of diagnosis and were asked to complete a self-administered survey and provide consent for access to medical records. This survey included information on demographics, treatment of prostate cancer and medical history. General HRQL was assessed using selected domains from the Medical Outcomes Study short form (SF)-36 [14], including general health status, role limitations attributable to physical health problems, role limitations attributable to mental health problems, bodily pain, general mental health, and vitality. The domains were linearly converted to a 0–100 scale according to standard scoring procedures, with higher scores indicating better HRQL. For the SF-36, differences of ≥5 points (the general health domain) [14] and 7.9 points (the mental health domain) were considered clinically meaningful [15]. Clinically meaningful differences for other subscales were determined with Norman’s ‘rule of thumb’, whereby a difference of ≈0.5 SD indicates a threshold of discriminant change in HRQL scores for a chronic illness [16].
In addition, information regarding urinary, sexual and bowel function in the past month before being surveyed, as well as patients’ functions just before prostate cancer diagnosis, was collected using modified versions of previously validated and reliable instruments [17–19]. Subjects were contacted again at 12, 24 and 60 months after their diagnosis date. Each time, they were asked to complete a survey that contained questions on HRQL and functional status in the past month. Outpatient and inpatient medical record abstraction were completed at ≈1 and 5 years after diagnosis in order to obtain details of the clinical presentation, diagnostic and staging evaluation, tumour characteristics and treatment. These records, combined with SEER records, were used to define the stage of disease, tumour grade, primary treatment and dates of therapy.
Baseline diabetes status was determined from the participants’ 6-month self-report, or at 12 months if the 6-month assessment was not available. Patients were considered to have prevalent diabetes if they reported this as a comorbid condition, if they were taking diabetes medication, or if diabetes was reported in the medical record. Other comorbid conditions at baseline were similarly determined at either the 6- or 12-month assessment. Incident (after prostate cancer diagnosis) diabetes was defined as present if a patient who did not report being diagnosed with, or treated for, diabetes on the 6- and/or 12-month surveys subsequently reported being diagnosed with diabetes or receiving hypoglycaemic medication at either the 24- or 60-month follow-up. The primary cancer management strategy was categorized as follows: radical prostatectomy, pelvic radiotherapy of any type, hormone therapy (medical or surgical), or watchful waiting. For men who received multiple treatments, the primary treatment was categorized as the more aggressive treatment.
STUDY COHORT
Of the 5672 eligible patients with prostate cancer initially contacted for inclusion in the PCOS, 3533 (62.3%) gave informed consent and completed a 6- and/or 12-month HRQL survey. For this analysis, we included all men with non-metastatic cancer, either localized or locally advanced disease (clinical stage ≤ T3). This sample of 1811 (51%) men had available baseline information on diabetes status and had returned at least one follow-up questionnaire. Of these men, 219 returned one, 442 returned two, and 1150 returned three follow-up questionnaires.
STATISTICAL ANALYSES
Differences in demographic, clinical characteristics, HRQL scores, and symptom function/bother scores between the three diabetes status groups were compared using a chi-squared test for categorical variables and ANOVA or Kruskall-Wallis tests for continuous variables, where appropriate.
Linear regression analysis was used to evaluate the association between baseline demographic and clinical characteristics and HRQL. Repeated measures analyses, using a mixed-model approach, were conducted to examine changes in general HRQL and symptom burden from baseline up to 60 months after initial diagnosis. We evaluated the positive regression slope, assessing HRQL at baseline, 12, 24, and 60 months. From previous longitudinal studies of prostate cancer survivors, the pretreatment HRQL rating was known to be associated with outcome over time [20,21]. We, therefore, included the pretreatment urinary, sexual and bowel functioning scores as a covariate for adjustment in disease-specific HRQL. Covariates included for adjustment were selected a priori or had shown an independent association (P < 0.05) with HRQL. We also included the interaction term for diabetes status and time in our analyses to evaluate whether diabetes status was associated with differences in HRQL over time. All statistical analyses were performed using SAS (version 9.1 for Windows, SAS institute Inc., Cary, NC, USA). Statistical differences were considered significant if P < 0.01 and reported P-values are two-sided. As the risk of Type II errors could increase with multiple testing, we used a more conservative P-value of <0.01 to reduce the possibility that some of the results reported were attributable to chance.
RESULTS
Of the current sample, 239 (13.2%) men had prevalent diabetes, 215 (11.9%) had incident diabetes, and 1357 (75.0%) did not report diabetes on any surveys. Compared with prostate cancer survivors without prevalent diabetes, those with prevalent diabetes were more likely: (i) to be African-American; (ii) to be unmarried at baseline; (iii) to be less educated; (iv) to report a total household income of ≤$30,000 per year; (v) to be retired; (vi) to have lower stage disease (cT2 or less); (vii) to be managed with watchful waiting or receive radiotherapy as initial treatment; and (viii) to have >1 comorbid conditions (see Table 1).
TABLE 1.
Demographic and clinical characteristics of patients according to diabetes mellitus status
| Characteristics | No DM, % (n = 1357) | DM, % (n = 239) | Post-DM, % (n = 215) | P |
|---|---|---|---|---|
| Age at time of survey | ||||
| 39–49 | 2.7 | 1.3 | 2.3 | |
| 50–64 | 43.1 | 36.0 | 38.1 | |
| 65–74 | 39.8 | 47.7 | 45.6 | |
| ≥75 | 14.4 | 15.1 | 13.9 | n.s. |
| Race/ethnicity | ||||
| Non-hispanic white | 72.7 | 51.9 | 58.6 | |
| Non-hispanic black | 12.3 | 28.4 | 22.8 | |
| Hispanic | 13.0 | 16.3 | 17.2 | |
| Other | 1.9 | 3.3 | 1.4 | <0.001 |
| SEER registry (geographic location) | ||||
| CT | 21.9 | 21.3 | 20.9 | |
| NM | 12.9 | 10.9 | 10.7 | |
| Seattle | 11.4 | 7.1 | 7.4 | |
| UT | 15.8 | 15.5 | 13.0 | |
| Atlanta | 9.3 | 11.7 | 11.2 | |
| Los Angeles | 28.7 | 33.5 | 36.7 | n.s. |
| Married at first interview | ||||
| Yes | 82.1 | 72.8 | 79.5 | |
| No | 17.2 | 25.9 | 18.6 | 0.009 |
| Education | ||||
| ≤8 grade | 7.3 | 12.5 | 10.7 | |
| Some high school | 8.8 | 15.9 | 18.1 | |
| High-school graduate | 20.0 | 24.3 | 21.4 | |
| Some college | 25.5 | 25.1 | 21.4 | |
| College graduate | 15.2 | 10.9 | 10.7 | |
| Graduate school | 22.0 | 10.5 | 16.3 | <0.001 |
| Income | ||||
| <$10 000 | 5.5 | 13.0 | 10.7 | |
| $10 000–20 000 | 13.3 | 23.4 | 19.1 | |
| $20 000–30 000 | 14.1 | 17.6 | 15.8 | |
| $30 000–40 000 | 14.4 | 13.4 | 10.7 | |
| $40 000–50 000 | 12.0 | 7.9 | 9.8 | |
| $50 000–75 000 | 14.1 | 7.5 | 14.4 | |
| >$75 000 | 18.6 | 8.8 | 10.2 | |
| Not sure | 2.1 | 4.6 | 4.6 | |
| Unanswered | 5.7 | 3.8 | 4.6 | <0.001 |
| Employment status | ||||
| Full-time | 30.7 | 16.3 | 22.8 | |
| Part-time | 9.8 | 10.5 | 7.4 | |
| Retired | 55.0 | 68.6 | 65.6 | |
| Other | 3.8 | 3.8 | 3.3 | |
| Unknown | 0.7 | 0.8 | 0.9 | 0.001 |
| Clinical stage | ||||
| ≤2 | 57.4 | 63.2 | 69.8 | |
| 3 | 35.9 | 32.2 | 22.3 | |
| 4 | 6.6 | 4.6 | 7.9 | 0.001 |
| Gleason score | ||||
| 2–4 | 7.5 | 6.7 | 4.7 | |
| 5 | 14.2 | 9.2 | 14.4 | |
| 6 | 22.5 | 20.9 | 18.1 | |
| 7 | 18.3 | 16.3 | 22.3 | |
| 8–10 | 8.1 | 9.2 | 7.0 | n.s. |
| Initial treatment | ||||
| Radical prostatectomy | 58.7 | 45.2 | 50.2 | |
| Radiation therapy | 22.0 | 26.4 | 21.4 | |
| Hormonal therapy | 9.2 | 8.8 | 12.1 | |
| Watchful waiting | 10.1 | 19.7 | 16.3 | <0.001 |
| Other comorbid conditions* | ||||
| None | 49.1 | 23.4 | 20.5 | |
| 1 | 32.6 | 37.2 | 39.5 | |
| >1 | 18.3 | 39.3 | 40.0 | <0.001 |
| Progression or recurrence of cancer | ||||
| Yes | 25.6 | 27.2 | 23.7 | |
| No | 74.4 | 72.8 | 76.3 | n.s. |
Comorbid conditions assessed include arthritis, inflammatory bowel disease, ulcers, chronic lung disease, heart failure, stroke, hypertension, heart attack, chest pain, depression and liver disease.
Table 2 outlines the mean scores of the general HRQL stratified by treatment. In general, men with prevalent diabetes reported the poorest scores, and men without diabetes the best. Men who developed incident diabetes had scores that were intermediate between the other two groups. Separate analyses by management strategy showed that men without diabetes and men with prevalent diabetes who were treated with prostatectomy, radiotherapy or managed with watchful waiting had significantly different scores on various HRQL scales. A clinically significant difference was noted in general health status scores between the men without diabetes and men with diabetes treated with prostatectomy or managed with watchful waiting. No statistical differences in scores were noted among men who received hormone treatment.
TABLE 2.
General HRQL at 6 months after diagnosis according to diabetes status and primary treatment/management strategy
| Primary treatment/management strategy | HRQL score ± SD |
||
|---|---|---|---|
| No diabetes diagnosis | Prevalent diabetes | Incident diabetes | |
| Prostatectomy | n = 796 | n = 108 | n = 108 |
| General health status†‡ | 78.2 ± 21.0 | 62.5 ± 26.8*** | 72.5 ± 22.1** |
| Role function: physical | 65.2 ± 40.5 | 54.4 ± 43.4 | 57.2 ± 43.4 |
| Role function: emotional | 78.5 ± 35.5 | 66.7 ± 41.2** | 63.2 ± 42.7** |
| Mental health | 78.9 ± 17.5 | 74.8 ± 17.5 | 75.1 ± 17.8 |
| Bodily pain | 82.0 ± 22.6 | 74.6 ± 27.2** | 76.6 ± 26.0 |
| Vitality | 65.2 ± 23.3 | 58.3 ± 20.7** | 58.9 ± 22.1 |
| Radiation therapy | n = 299 | n = 63 | n = 46 |
| General health status | 70.6 ± 24.4 | 58.6 ± 26.5** | 64.3 ± 23.6 |
| Role function: physical | 62.1 ± 42.2 | 50.8 ± 44.0 | 61.4 ± 40.7 |
| Role function: emotional | 75.3 ± 37.4 | 64.0 ± 42.4 | 70.3 ± 37.4 |
| Mental health | 79.4 ± 17.5 | 73.6 ± 22.5 | 76.9 ± 17.0 |
| Bodily pain | 79.3 ± 23.5 | 73.1 ± 28.0 | 80.6 ± 19.5 |
| Vitality | 60.3 ± 23.3 | 51.5 ± 24.6 | 58.4 ± 22.0 |
| Hormone therapy | n = 125 | n = 21 | n = 26 |
| General health status | 60.9 ± 28.3 | 50.5 ± 28.4 | 57.2 ± 24.4 |
| Role function: physical | 49.2 ± 42.5 | 52.5 ± 43.6 | 53.8 ± 44.0 |
| Role function: emotional | 67.2 ± 40.5 | 78.3 ± 39.4 | 60.2 ± 42.2 |
| Mental health | 78.6 ± 17.1 | 76.8 ± 16.9 | 72.6 ± 18.0 |
| Bodily pain | 73.7 ± 25.8 | 76.2 ± 21.8 | 69.4 ± 29.1 |
| Vitality | 57.8 ± 21.8 | 51.5 ± 24.6 | 57.3 ± 23.5 |
| Watchful waiting | n = 137 | n = 47 | n = 35 |
| General health status† | 72.1 ± 26.6 | 54.1 ± 25.1*** | 63.2 ± 27.7 |
| Role function: physical | 70.7 ± 38.9 | 56.5 ± 44.5 | 59.5 ± 41.7 |
| Role function: emotional | 81.2 ± 33.7 | 70.3 ± 43.5 | 72.5 ± 38.9 |
| Mental health | 79.4 ± 24.1 | 68.8 ± 26.3 | 72.8 ± 21.5 |
| Bodily pain | 79.9 ± 17.3 | 73.8 ± 16.7 | 75.3 ± 17.0 |
| Vitality | 64.2 ± 21.7 | 55.1 ± 20.3** | 58.5 ± 20.3 |
Significantly different from men without diabetes:
P < 0.001;
P < 0.01.
Clinically significant different: men without diabetes vs. prevalent diabetes.
Prevalent diabetes vs. incident diabetes: P < 0.01.
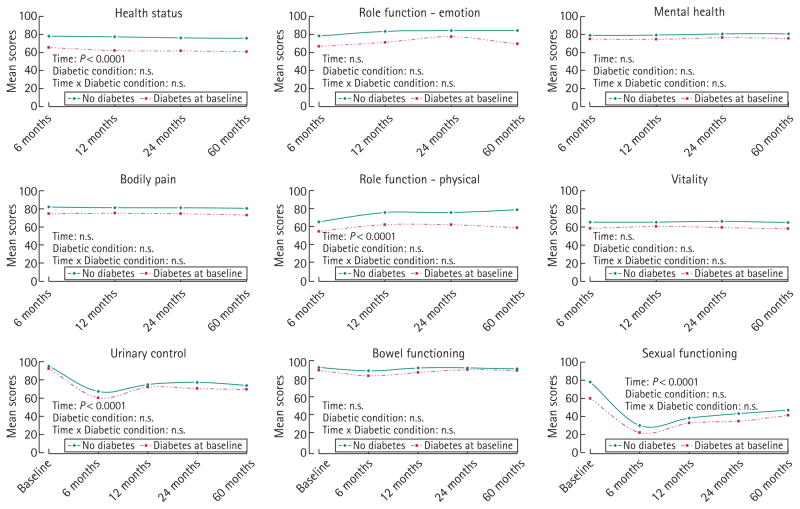
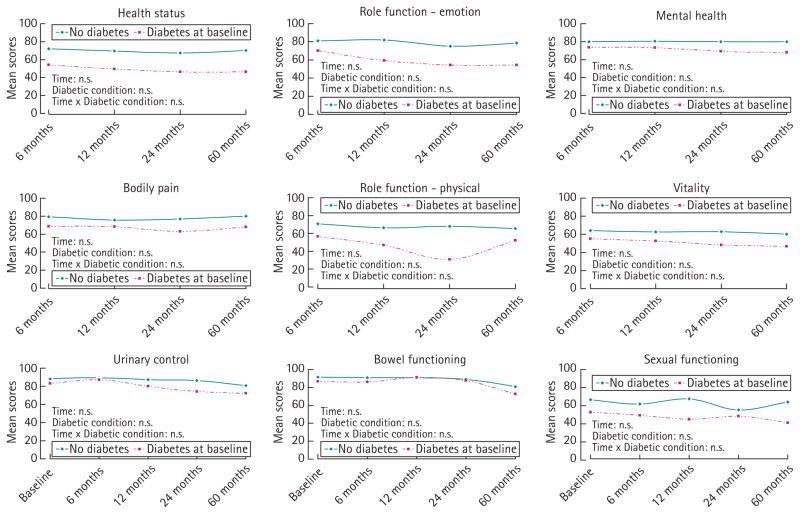
During follow-up, unadjusted results showed that having prevalent diabetes was associated with poorer general HRQL scores for men treated with prostatectomy or managed with watchful waiting (Figs 1,2). After adjustments across a range of demographic and clinical variables, no significant interaction effect was found between diabetes status and time for general HRQL in the prostatectomy group. General HRQL in men with incident diabetes was mostly lower than that of men without diabetes over time, but better or equal to that of men with prevalent diabetes (data not shown). Similar results of HRQL over time were found for men treated with either radiotherapy or hormone therapy (data not shown).
FIG. 1.
Longitudinal changes in cancer-specific HRQL over time after prostatectomy according to diabetes status. P-values adjusted for age at diagnosis, marital status, education level, annual income, employment status, baseline cancer-specific HRQL score, cancer stage, baseline PSA, baseline Gleason score.
FIG. 2.
Longitudinal changes in cancer-specific HRQL over time for men managed with watchful waiting according to diabetes status. P-values adjusted for age at diagnosis, marital status, education level, annual income, employment status, baseline cancer-specific HRQL score, cancer stage, primary treatment, baseline PSA, baseline Gleason score.
For the most part, cancer-specific HRQL scores showed patterns similar to those seen in general HRQL, with men with diabetes reporting the poorest scores and men without diabetes the best. The only significant difference noted between men with prevalent diabetes and men without diabetes treated with prostatectomy was in baseline sexual function (59.5 ± 41.4 vs. 77.9 ± 32.7, P < 0.001) (Table 3). Non-diabetic men treated with prostatectomy reported the greatest change in sexual function at 6 months from baseline (−48.4 points), which was significantly different from the change score of −37.6 points reported by men with diabetes.
TABLE 3.
Disease-specific HRQL at baseline and change within 6 months after diagnosis according to diabetes status and treatment
| HRQL score ± SD |
|||
|---|---|---|---|
| No diabetes diagnosis | Prevalent diabetes | Incident diabetes | |
| Prostatectomy | n = 796 | n = 108 | n = 108 |
| Urinary control | |||
| Baseline | 95.1 ± 14.9 | 92.3 ± 18.6 | 93.8 ± 15.3 |
| Change 6 months | −27.8 ± 25.7 | −32.2 ± 31.6 | −33.5 ± 28.8 |
| Bowel function | |||
| Baseline | 92.5 ± 17.5 | 89.2 ± 19.8* | 90.7 ± 20.9 |
| Change 6 months | −3.9 ± 20.9 | −6.2 ± 26.7 | −5.0 ± 23.6 |
| Sexual function | |||
| Baseline | 77.9 ± 32.7 | 59.5 ± 41.4*** | 70.3 ± 37.9 |
| Change 6 months | −48.4 ± 35.6 | −37.6 ± 34.6† | −45.9 ± 34.5 |
| Radiation therapy | n = 299 | n = 63 | n = 46 |
| Urinary control | |||
| Baseline | 87.9 ± 20.3 | 85.7 ± 25.2 | 87.5 ± 19.1 |
| Change 6 months | −2.7 ± 20.3 | −2.4 ± 25.2 | 0.6 ± 19.2 |
| Bowel function | |||
| Baseline | 87.2 ± 23.2 | 84.1 ± 26.3 | 83.4 ± 24.7 |
| Change 6 months | −13.3 ± 27.7 | −9.1 ± 33.0 | −6.2 ± 27.6 |
| Sexual function | |||
| Baseline | 68.8 ± 35.9 | 64.2 ± 36.3 | 66.3 ± 34.4 |
| Change 6 months | −11.0 ± 38.8 | −14.6 ± 41.1 | −14.5 ± 38.4 |
| Hormone therapy | n = 125 | n = 21 | n = 26 |
| Urinary control | |||
| Baseline | 84.8 ± 23.9 | 80.9 ± 27.0 | 80.1 ± 29.8 |
| Change 6 months | 1.3 ± 23.2 | 2.4 ± 27.6 | −3.8 ± 27.1 |
| Bowel function | |||
| Baseline | 83.1 ± 28.2 | 78.7 ± 28.4 | 90.4 ± 17.4 |
| Change 6 months | −0.8 ± 27.9 | 3.8 ± 28.2 | −1.0 ± 17.6 |
| Sexual function | |||
| Baseline | 61.9 ± 40.0 | 50.0 ± 38.0 | 54.0 ± 38.0 |
| Change 6 months | −13.9 ± 43.0 | −5.3 ± 43.8 | −14.4 ± 38.9 |
| Watchful waiting | n = 137 | n = 47 | n = 35 |
| Urinary control | |||
| baseline | 88.3 ± 22.8 | 83.3 ± 24.1 | 88.2 ± 21.2 |
| Change 6 months | 0.9 ± 19.0 | 4.4 ± 24.2 | 2.1 ± 21.0 |
| Bowel function | |||
| Baseline | 91.6 ± 18.5 | 86.7 ± 25.4* | 87.8 ± 18.6 |
| Change 6 months | −0.7 ± 16.0 | −0.6 ± 23.8 | 3.0 ± 18.5 |
| Sexual function | |||
| Baseline | 66.7 ± 37.1 | 52.7 ± 38.8 | 56.4 ± 37.0 |
| Change 6 months | −4.8 ± 20.4 | −4.9 ± 25.1 | −10.1 ± 26.1 |
Significantly different from men without diabetes:
P < 0.001;
P < 0.05.
Change scores (men without diabetes vs. prevalent diabetes): P < 0.05 according to Tukey’s test.
In longitudinally adjusted analyses of disease-specific HRQL by treatment, no interaction effect of time and diabetes status was found in the prostatectomy and watchful waiting groups (Figs 1,2). Similar results were found for the radiotherapy and hormone treatment group (data not shown). The disease-specific HRQL scores of men with incident diabetes ranged between those of men without diabetes and men with diabetes (data not shown).
DISCUSSION
Our study assessed the longitudinal effect of diabetes and prostate cancer, with or without treatment, on general- and cancer-specific HRQL in a population-based cohort of men followed up for 5 years after prostate cancer diagnosis. Overall, patients with prostate cancer and diabetes at baseline had a poorer general HRQL than men without diabetes and men subsequently diagnosed with diabetes during 60 months of follow-up, independent of treatment. Similarly, men with prevalent diabetes had the lowest, and men without diabetes had the highest, urinary control and sexual function scores over time, with those noted to have incident diabetes falling between the two other groups.
As hypothesized, we found that, in general, men with prevalent diabetes had the poorest general HRQL during follow-up and men without diabetes had the best. This difference in HRQL remained constant over time. In another cross-sectional study of a large group of long-term cancer survivors (including genitourinary malignancies), with demographics comparable with our sample, survivors without diabetes reported better overall health and fewer problems with other comorbid conditions than those with diabetes [22]. In a Dutch study, long-term prostate cancer survivors with diabetes also reported poorer general health and vitality scores on the SF-36 [7].
Among the men with baseline diabetes, those who underwent prostatectomy had generally better baseline HRQL scores when compared with those in other management strategy groups. A possible explanation is that healthier patients, regardless of diabetes status, were more likely to undergo prostatectomy.
Prostate cancer-specific HRQL was the lowest among men with prevalent diabetes when compared with men without diabetes and those with incident diabetes. Similarly, another longitudinal study of men with prostate cancer treated with radiotherapy reported that diabetes predicted erectile dysfunction at 1 year of follow-up [5]. In the CaPSURE study [11], a longitudinal disease registry of men with prostate cancer, men with diabetes had poorer urinary control but not sexual or bowel function over a 24-month follow-up. In that study, men without diabetes actually reported a greater decline in sexual function than those with diabetes, although men with diabetes had worse function at baseline. Similarly, our study found the steepest decline in sexual function from pre-diagnosis to 6-month post-diagnosis among men without diabetes treated with prostatectomy. This floor effect, whereby men with diabetes already have a lower pre-treatment level of sexual functioning and therefore cannot score lower than the scale limit, also seemed to be common with other comorbid conditions such as CVD [10].
Our results show that men with incident diabetes reported general and disease-specific HRQL scores lower than those of men without diabetes, but similar or better than those with diabetes at baseline. The observed lower HRQL when compared with men without diabetes could represent the effects of either undiagnosed diabetes or the predisposing risk factors for diabetes.
Over the course of our study, men with prevalent diabetes had poorer general and disease-specific HRQL scores than those never diagnosed with diabetes. This suggests that prostate cancer survivors with diabetes could benefit from targeted interventions to improve decision-making at the time of diagnosis and during the course of the disease, as different prostate cancer treatments have variable impact on HRQL scores in men with diabetes. To this end, prostate cancer survivors with prevalent or incident diabetes could benefit from educational interventions that promote better self-management of diabetes, as this may positively affect HRQL outcomes. Previous studies have shown that individuals with newly diagnosed diabetes, who followed a structured educational program that addressed their illness beliefs, reported a better understanding and control of the disease, a lesser impact from diabetes on their daily activities, and a better HRQL after 3 months of following the program [23].
Prostate cancer survivors, in particular those at risk of developing diabetes mellitus could benefit from lifestyle changes. Studies have shown that lifestyle interventions such as exercise and dietary changes could prevent progression to diabetes mellitus among high-risk individuals. [24–26]. As such, clinical management of high-risk prostate cancer survivors such as those overweight, with family history of diabetes mellitus, with impaired glucose tolerance or who are receiving ADT could include recommendations for exercise and dietary changes. Assessment with a simple diabetes mellitus screening instrument could be incorporated as part of clinical management [27].
Possible limitations of the present study include, first, the use of self-report of comorbid conditions such as diabetes, which could influence the reliability of the information provided over time. However, an earlier study from the PCOS reported that the participants were able to provide reliable information on comorbid conditions over time [28,29]. Second, there was no pre-diagnosis assessment of general HRQL against which to assess post-diagnosis general HRQL. Moreover, the pre-diagnosis scores on cancer-specific HRQL were assessed retrospectively after initial diagnosis and treatment in the majority (roughly 80%) of patients. As such, recall bias could result in overestimation of pre-diagnosis HRQL [29]. The small numbers in samples, such as men with baseline diabetes who received hormone therapy, suggest that the results of subgroup analyses should be interpreted with caution. Although we report on the HRQL of men with incident diabetes, these results should be interpreted cautiously as we can only imprecisely estimate when the self-reported diabetes was actually diagnosed (between 12–24 or 24–60 months). This makes it difficult to assess the burden of diabetic disease and treatment on HRQL.
The strengths of this study include the use of longitudinal data with a follow-up of 60 months. Furthermore, we believe that this is the first analysis to measure the effects of prevalent and incident diabetes on the long-term HRQL of patients with prostate cancer. Our results suggest that prostate cancer survivors with diabetes have poorer HRQL than those without diabetes and this may have implications for prostate cancer treatment decisions.
Acknowledgments
We thank the men who, by their participation in the PCOS, have contributed to a better understanding of the effects of prostate cancer on men’s lives. We also thank the physicians in the six SEER areas who assisted us in the collection of data from their patients and from medical records. The work was supported by National Institutes of Health (NIH) grant R01CA114524 and data collection was supported by NIH contracts to each of the six participating institutions: NO1-PC-67000, NO1-PC-67005, NO1-PC-67006, NO1-PC-67007, NO1-PC-67009, and NO1-PC-67010.
Abbreviations
- HRQL
health-related quality of life
- ADT
androgen deprivation therapy
- CVD
cardiovascular disease
- PCOS
Prostate Cancer Outcomes Study
- SEER
Surveillance, Epidemiology and End Results
- SF
short form
Footnotes
CONFLICT OF INTEREST
None declared.
References
- 1.National Institute of Diabetes and Digestive and Kidney Diseases. National Diabetes Statistics, 2007 Fact Sheet. Bethesda, MD: U.S. Department of Health and Human Services, National Institutes of Health; 2008. [Google Scholar]
- 2.Houterman S, Janssen-Heijnen ML, Hendrikx AJ, van den Berg HA, Coebergh JW. Impact of comorbidity on treatment and prognosis of prostate cancer patients: a population-based study. Crit Rev Oncol Hematol. 2006;58:60–7. doi: 10.1016/j.critrevonc.2005.08.003. [DOI] [PubMed] [Google Scholar]
- 3.van de Poll-Franse LV, Houterman S, Janssen-Heijnen MLG, Dercksen MW, Coebergh JWW, Haak HR. Less aggressive treatment and worse overall survival in cancer patients with diabetes: a large population based analysis. Int J Cancer. 2007;120:1986–92. doi: 10.1002/ijc.22532. [DOI] [PubMed] [Google Scholar]
- 4.Hall HI, Satariano WA, Thompson T, Ragland KE, Van Den Eeden SK, Selvin S. Initial treatment for prostate carcinoma in relation to comorbidity and symptoms. Cancer. 2002;95:2308–15. doi: 10.1002/cncr.10926. [DOI] [PubMed] [Google Scholar]
- 5.Pinkawa M, Gagel B, Piroth MD, et al. Erectile dysfunction after external beam radiotherapy for prostate cancer. Eur Urol. 2009;55:227–34. doi: 10.1016/j.eururo.2008.03.026. [DOI] [PubMed] [Google Scholar]
- 6.Karakiewicz PI, Bhojani N, Neugut A, et al. The effect of comorbidity and socioeconomic status on sexual and urinary function and on general health-related quality of life in men treated with radical prostatectomy for localized prostate cancer. J Sex Med. 2008;5:919–27. doi: 10.1111/j.1743-6109.2007.00741.x. [DOI] [PubMed] [Google Scholar]
- 7.Mols F, Aquarius AE, Essink-Bot ML, Aaronson NK, Kil PJ, van de Poll-Franse LV. Does diabetes mellitus as a comorbid condition affect the health-related quality of life in prostate cancer survivors? Results of a population-based observational study. BJU Int. 2008;102:1594–600. doi: 10.1111/j.1464-410X.2008.07812.x. [DOI] [PubMed] [Google Scholar]
- 8.Keating NL, O’Malley AJ, Smith MR. Diabetes and cardiovascular disease during androgen deprivation therapy for prostate cancer. J Clin Oncol. 2006;24:4448–56. doi: 10.1200/JCO.2006.06.2497. [DOI] [PubMed] [Google Scholar]
- 9.van de Poll-Franse LV, Sadetsky N, Kwan L, Litwin MS. Severity of cardiovascular disease and health-related quality of life in men with prostate cancer: a longitudinal analysis from CaPSURE. Qual Life Res. 2008;17:845–55. doi: 10.1007/s11136-008-9356-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.van de Poll-Franse LV, Kwan L, Reiter RE, Lee SP, Litwin MS. The influence of cardiovascular disease on health related quality of life in men with prostate cancer: a 4-year followup study. J Urol. 2008;179:1362–7. doi: 10.1016/j.juro.2007.11.086. [DOI] [PubMed] [Google Scholar]
- 11.Latini DM, Chan JM, Cowan JE, et al. Health-related quality of life for men with prostate cancer and diabetes: a longitudinal analysis from CaPSURE. Urology. 2006;68:1242–7. doi: 10.1016/j.urology.2006.08.1096. [DOI] [PubMed] [Google Scholar]
- 12.Potosky AL, Davis WW, Hoffman RM, et al. Five-year outcomes after prostatectomy or radiotherapy for prostate cancer: the prostate cancer outcomes study. J Natl Cancer Inst. 2004;96:1358–67. doi: 10.1093/jnci/djh259. [DOI] [PubMed] [Google Scholar]
- 13.Potosky AL, Harlan LC, Stanford JL, et al. Prostate cancer practice patterns and quality of life: the Prostate Cancer Outcomes Study. J Natl Cancer Inst. 1999;91:1719–24. doi: 10.1093/jnci/91.20.1719. [DOI] [PubMed] [Google Scholar]
- 14.Ware J., Jr . SF-36 Health Survey: Manual and Interpretation Guide. Boston, MA: The Health Institute, New England Medical Center; 1993. [Google Scholar]
- 15.Norman GR, Sridhar FG, Guyatt GH, Walter SD. Relation of distribution- and anchor-based approaches in interpretation of changes in health-related quality of life. Med Care. 2001;39:1039–47. doi: 10.1097/00005650-200110000-00002. [DOI] [PubMed] [Google Scholar]
- 16.Norman GR, Sloan JA, Wyrwich KW. Interpretation of changes in health-related quality of life: the remarkable universality of half a standard deviation. Med Care. 2003;41:582–92. doi: 10.1097/01.MLR.0000062554.74615.4C. [DOI] [PubMed] [Google Scholar]
- 17.Fowler FJ, Jr, Barry MJ, Lu-Yao G, Roman A, Wasson J, Wennberg JE. Patient-reported complications and follow-up treatment after radical prostatectomy. The National Medicare Experience: 1988–1990 (updated June 1993) Urology. 1993;42:622–9. doi: 10.1016/0090-4295(93)90524-e. [DOI] [PubMed] [Google Scholar]
- 18.Litwin MS, Hays RD, Fink A, Ganz PA, Leake B, Brook RH. The UCLA Prostate Cancer Index: development, reliability, and validity of a health-related quality of life measure. Med Care. 1998;36:1002–12. doi: 10.1097/00005650-199807000-00007. [DOI] [PubMed] [Google Scholar]
- 19.Talcott JA, Rieker P, Clark JA, et al. Patient-reported symptoms after primary therapy for early prostate cancer: results of a prospective cohort study. J Clin Oncol. 1998;16:275–83. doi: 10.1200/JCO.1998.16.1.275. [DOI] [PubMed] [Google Scholar]
- 20.Stanford JL, Feng Z, Hamilton AS, et al. Urinary and sexual function after radical prostatectomy for clinically localized prostate cancer: the Prostate Cancer Outcomes Study. JAMA. 2000;283:354–60. doi: 10.1001/jama.283.3.354. [DOI] [PubMed] [Google Scholar]
- 21.Talcott JA, Manola J, Clark JA, et al. Time course and predictors of symptoms after primary prostate cancer therapy. J Clin Oncol. 2003;21:3979–86. doi: 10.1200/JCO.2003.01.199. [DOI] [PubMed] [Google Scholar]
- 22.Stava CJ, Beck ML, Feng L, Lopez A, Busaidy N, Vassilopoulou-Sellin R. Diabetes mellitus among cancer survivors. J Cancer Surviv. 2007;1:108–15. doi: 10.1007/s11764-007-0016-z. [DOI] [PubMed] [Google Scholar]
- 23.Skinner TC, Carey ME, Cradock S, et al. Diabetes education and self-management for ongoing and newly diagnosed (DESMOND): process modelling of pilot study. Patient Educ Couns. 2006;64:369–77. doi: 10.1016/j.pec.2006.04.007. [DOI] [PubMed] [Google Scholar]
- 24.Absetz P, Oldenburg B, Hankonen N, et al. Type 2 diabetes prevention in the real world: three year results of the GOAL lifestyle implementation trial. Diabetes Care. 2009;32:1418–20. doi: 10.2337/dc09-0039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Galvao DA, Spry N, Taaffe DR, et al. A randomized controlled trial of an exercise intervention targeting cardiovascular and metabolic risk factors for prostate cancer patients from the RADAR trial. BMC Cancer. 2009;9:419. doi: 10.1186/1471-2407-9-419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Tuomilehto J, Lindstrom J, Eriksson JG, et al. Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance. N Engl J Med. 2001;344:1343–50. doi: 10.1056/NEJM200105033441801. [DOI] [PubMed] [Google Scholar]
- 27.Schwarz PEH, Li J, Lindstrom J, Tuomilehto J. Tools for predicting the risk of type 2 diabetes in daily practice. Horm Metab Res. 2009;41:86–97. doi: 10.1055/s-0028-1087203. [DOI] [PubMed] [Google Scholar]
- 28.Klabunde CN, Reeve BB, Harlan LC, Davis WW, Potosky AL. Do patients consistently report comorbid conditions over time? results from the prostate cancer outcomes study. Med Care. 2005;43:391–400. doi: 10.1097/01.mlr.0000156851.80900.d1. [DOI] [PubMed] [Google Scholar]
- 29.Litwin MS, McGuigan KA. Accuracy of recall in health-related quality-of-life assessment among men treated for prostate cancer. J Clin Oncol. 1999;17:2882–8. doi: 10.1200/JCO.1999.17.9.2882. [DOI] [PubMed] [Google Scholar]