Abstract
Background
Dermatofibrosarcoma protuberans (DFSP) is a rare malignant skin tumor associated with a characteristic chromosomal translocation (t[17;22][q22;q13]) resulting in the COL1A1-platelet-derived growth factor β (PDGFB) fusion gene. This malignancy is rarely diagnosed in childhood. We observed a high incidence of this tumor in children affected with adenosine deaminase–deficient severe combined immunodeficiency (ADA-SCID).
Methods
Twelve patients with ADA-SCID were evaluated with a complete dermatologic examination and skin biopsy when indicated. Conventional cytogenetic and molecular analyses (fluorescence in situ hybridization, RT-PCR, or both) were performed when possible.
Results
Eight patients were found to have DFSP. Six patients had multicentric involvement (4-15 lesions), primarily of the trunk and extremities. Most lesions presented as 2- to 15-mm, round atrophic plaques. Nodular lesions were present in 3 patients. In all cases CD34 expression was diffusely positive, and diagnosis was confirmed either by means of cytogenetic analysis, molecular testing, or both. The characteristic DFSP-associated translocation, t(17;22)(q22;q13), was identified in 6 patients; results of fluorescence in situ hybridization were positive for fusion of the COL1A1 and PDGFB loci in 7 patients; and RT-PCR showed the COL1A1-PDGFB fusion transcript in 6 patients.
Conclusions
We describe a previously unrecognized association between ADA-SCID and DFSP with unique features, such as multicentricity and occurrence in early age. We hypothesize that the t(17;22)(q22;q13) translocation that results in dermal overexpression of PDGFB and favors the development of fibrotic tumors might arise because of the known DNA repair defect in patients with ADA-SCID. Although the natural course of DFSP in the setting of ADA-SCID is unknown, this observation should prompt regular screening for DFSP in patients with ADA-SCID.
Keywords: Severe combined immunodeficiency, adenosine deaminase, dermatofibrosarcoma, adenosine, fibrosis
Adenosine deaminase–deficient severe combined immunodeficiency (ADA-SCID) is a rare genetic disorder with an estimated incidence between 1:200,000 and 1:1,000,000. Both humoral and cellular immunity are affected, which results in increased risk of recurrent and life-threatening infections. Unlike most other forms of severe combined immunodeficiency, adenosine deaminase (ADA) deficiency is a metabolic disorder with clinical complications beyond the immune system that include skeletal, endocrine, and central nervous system abnormalities.1,2 Several pathophysiologic mechanisms have been implicated in the disease, including DNA repair defects caused by cellular nucleotide pool imbalance and inhibition of DNA polymerase activity. In addition, deoxyadenosine, one of the substrates that accumulate in ADA-deficient cells, has been reported to result in chromosome breakage.1,2 Treatment options include enzyme replacement therapy with pegylated bovine adenosine deaminase (PEG-ADA), hematopoietic cell transplantation (HCT), and gene therapy.3
Dermatofibrosarcoma protuberans (DFSP) is an uncommon mesenchymal tumor of the skin and subcutaneous tissues with low-to-intermediate malignant potential and low risk for metastasis (<2% of cases).4,5 The incidence of DFSP is 4.2 per million, accounting for approximately 0.1% of all cancers.6 Because of its slow growing characteristics, the clinical diagnosis of DFSP can be delayed for 2 to 3 decades.7,8 The occurrence of DFSP is even more uncommon in the pediatric setting, with fewer than 200 cases reported.9-13 Although DFSP typically presents as a solitary subcutaneous nodule, other forms exist, including atrophic, anetoderma-like, and morpheaform variants. The characteristic histologic finding is a spindle cell tumor with a storiform pattern. These lesions are CD34+ and are negative for Factor XIIIa and S100. The giant cell fibroblastoma (GCF) variant, which is more commonly seen in children, demonstrates characteristic pseudovascular spaces and giant cell florets. DFSP grows in an infiltrative manner, requiring wide local excision or microscopic margin control to achieve tumor clearance. Depending on margin widths and surgical techniques, local recurrence has been reported in 0% to 50% of cases.8,14,15 In the spectrum of fibrous tumors of the skin, DFSP is considered more worrisome than benign fibrous histiocytoma and less aggressive than malignant fibrous histiocytoma. At the cytogenetic level, DFSP is characterized by abnormal rearrangement of chromosomes 17 and 22, often leading to ring chromosomes or unbalanced t(17;22)(q22;q13).16,17 This characteristic rearrangement is described in more than 95% of cases when tested by using karyo-type, fluorescence in situ hybridization (FISH), or RT-PCR.18 The resulting chimeric COL1A1–platelet-derived growth factor β (PDGFB) gene produces a functional and constitutively active PDGFβ protein.
Here we present a cohort of 12 unselected cases of ADA-SCID, 8 of whom had atrophic, nodular, or both DFSP lesions. This remarkable increased risk for DFSP in patients with ADA-SCID has not previously been reported, to our knowledge, and suggests an important pathophysiologic link between these 2 rare diseases.
METHODS
Patients
All human subject research procedures were approved by the Institutional Review Board of the National Human Genome Research Institute (clinical research protocols registered with ClinicalTrials.gov as NCT00018018 and NCT00006319).
Twelve patients with ADA-SCID were followed at our institution between 2006 and 2009. The diagnosis of DFSP in 2 consecutive subjects prompted us to routinely screen for this rare malignancy. At the time of screening, patients were between 2 and 27 years old. In each case ADA-SCID was confirmed by sequencing of the ADA gene. Patients’ clinical characteristics are reported in Table I. All patients underwent comprehensive physical, immunologic, and dermatologic examinations. When medically indicated, punch skin biopsy, surgical excision, or both were performed.
TABLE I.
Patients’ characteristics and skin findings
| UPN | Age at screening/diagnosis of DFSP; sex |
Prior therapies | Treatment at screening or diagnosis of skin lesions |
Skin lesions (no.) | Size range (mm) |
Lesion(s) biopsied |
Location, size | Histologic findings |
Karyotype | FISH | RT-PCR | Final diagnosis |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ADA1 | 22 y; F | PEG-ADA Genetically corrected autologous lymphocytes |
PEG-ADA | Slightly raised right lower leg plaque (1) | 11 | 1 | Right lower leg, slightly raised plaque, 10 × 11 mm | CD34+ spindle cell lesion | Not done | COL1A1-PGFB fusion signal | COL1A1-PDGFB fusion transcript | DFSP |
| ADA3 | 12 y; M | T cell–depleted haploidentical BMT | PEG-ADA | Atrophic plaques (4) | 4-11 | 3 | Right lower leg, depressed plaque, 11 × 6 mm | CD34+ spindle cell lesion | Not done | Not done | Not informative | |
| Left upper abdomen, depressed plaque, 6 × 4 mm | CD34+ spindle cell lesion | t(17;22)(q11;q13) | COL1A1-PGFB fusion signal | Not informative | DFSP | |||||||
| Right gluteal, 5 × 3 mm | CD34+ spindle cell lesion | Not done | COL1A1-PGFB fusion signal | Not informative | ||||||||
| ADA4 | 9 y; M | T cell–depleted haploidentical BMT | PEG-ADA | Atrophic plaques (5) | 3-10 | 1 | Right anterior thigh, depressed plaque, 8 × 4 mm | CD34+ spindle cell lesion | Normal | Negative | Not informative | |
| Left axillary depressed plaque, 7 × 5 mm | CD34+ spindle cell lesion | t(17;22)(q11;q13) | COL1A1-PGFB fusion signal | COL1A1-PDGFB fusion transcript | DFSP | |||||||
| 3 depressed plaques, 3-10 mm | Clinically suspicious for DFSP. No biopsy | NA | NA | NA | ||||||||
| ADA5 | 7 y; M | PEG-ADA | PEG-ADA | Atrophic plaques (2) | 5-7 | 1 | Right inner arm plaque, 5 × 5 mm | CD34+ spindle cell lesion | t(17;22)(q11;q13) | COL1A1-PGFB fusion signal | Negative | DFSP |
| Right lower abdomen, slightly depressed plaque, 7 × 4 mm | Clinically suspicious for DFSP; no biopsy | NA | NA | NA | ||||||||
| ADA7 | 5 y; M | T cell–depleted haploidentical BMT | PEG-ADA | Atrophic plaques (12) | 3-16 | 1 | Right lateral abdomen, depressed plaque, 13 × 10 mm | CD34+ spindle cell lesion | Not done | Equivocal | COL1A1-PDGFB fusion transcript | DFSP |
| 11 depressed plaques from 3-16 mm | Clinically suspicious for DFSP. No biopsy | NA | NA | NA | ||||||||
| ADA12 | 12 y; F | Genetically corrected autologous BMT | PEG-ADA | Left groin subcutaneous lump/nodule (1) | NA | 1 | Left inguinal subcutaneous fibrotic lump, size not available | GCF | Not done | Not informative | Not informative | GCF |
| Atrophic plaques (7) | 0.5-20 | 2 | Right lateral chest wall, depressed plaque, 10 × 12 mm | CD34+ spindle cell lesion | Not done | Not done | Not done | |||||
| Right anterior shoulder, 0.6 × 0.5 mm | CD34+ spindle cell lesion | t(17;22)(q11;q13 | COL1A1-PGFB fusion signal | COL1A1-PDGFB fusion transcript | DFSP | |||||||
| 5 flat to depressed plaques, 3-20 mm | Clinically suspicious for DFSP; no biopsy | NA | NA | NA | ||||||||
| ADA14 | 2 y; F | PEG-ADA | Genetically corrected autologous BMT | Atrophic plaques (14) | 2-14 | 6 | Right chest wall nodule within a depressed plaque, 5 × 5 mm | CD34+ spindle cell lesion | t(17;22)(q11;q13) | COL1A1-PGFB fusion signal | Not informative | DFSP |
| Medial right buttock, flat to slightly raised plaque, 13 × 8 mm | CD34+ spindle cell lesion | t(17;22)(q11;q13) | COL1A1-PGFB fusion signal | COL1A1-PDGFB fusion transcript | ||||||||
| Right upper abdomen, depressed plaque, 10 × 5 mm | CD34+ spindle cell lesion | Not done | COL1A1-PGFB fusion signal | Not informative | ||||||||
| Right lower abdomen, depressed, 8 × 3 mm | CD34+ spindle cell lesion | Not done | Equivocal | Not informative | ||||||||
| Left abdomen (flank), depressed plaque, 8 × 3 mm | CD34+ spindle cell lesion | Not done | COL1A1-PGFB fusion signal | COL1A1-PDGFB fusion transcript | ||||||||
| Left calf, depressed plaque, 10 × 10 mm | CD34+ spindle cell lesion | Not done | COL1A1-PGFB fusion signal | COL1A1-PDGFB fusion transcript | ||||||||
| Left waistband, violaceous soft flat plaque, site of BM, 14 × 5 mm | Clinically suspicious for DFSP; no biopsy | NA | NA | NA | ||||||||
| 8 depressed plaques from 2-8 mm | Clinically suspicious for DFSP; no biopsy | NA | NA | NA | ||||||||
| ADA16 | 2 y; M | Genetically corrected autologous BMT | Genetically corrected autologous BMT | Left groin lump (1) | 10 | 1 | Left thigh, subcutaneous lump, 10 × 10 mm | GCF | Not done | COL1A1-PGFB fusion signal | COL1A1-PDGFB fusion transcript | GCF |
| Atrophic plaques (5) | 3-11 | 3 | Right inner arm, depressed plaque, 5 × 3 mm | CD34+ spindle cell lesion | t(17;22)(q11;q13) | COL1A1-PDGFB fusion signal | Not informative | DFSP | ||||
| Right abdominal wall, depressed plaque, 5 × 4 mm | CD34+ spindle cell lesion | Not done | Not informative | Not informative | ||||||||
| Left supraclavicular depressed plaque, 9 × 3 mm | CD34+ spindle cell lesion | Not done | COL1A1-PDGFB fusion signal | COL1A1-PDGFB fusion transcript | ||||||||
| 2 depressed plaques, right lateral upper thigh, 6 × 4 mm and Midsuprapubic, 11 × 5 mm | Clinically suspicious for DFSP; no biopsy done | NA | NA | NA | ||||||||
| ADA6 | 7 y; M | PEG-ADA T cell–depleted haploidentical BMT |
PEG-ADA | Atrophic plaques (2) | 5-6 | 1 | Paucicellular spindle cell lesion | |||||
| ADA9 | 4 y; M | PEG-ADA | PEG-ADA | Slightly raised plaque | 8 | 1 | Scar | |||||
| ADA10 | 3 y; M | PEG-ADA | Genetically corrected autologous BMT | Papules (2) | 3 | 2 | Mastocytoma | |||||
| ADA13 | 28 y; F | PEG-ADA Genetically corrected autologous lymphocytes Genetically corrected autologous BMT |
PEG-ADA | None | NA | NA | NA |
BM, Bone marrow; BMT, bone marrow transplantation; NA, not applicable.
Histologic and immunohistochemical evaluation
Formalin-fixed, paraffin-embedded biopsy specimens were sectioned and stained with hematoxylin and eosin. Immunohistochemical analysis was performed with antibodies against CD34, Factor XIII, and S100 protein by using standard procedures.
See additional information in the Methods section in this article's Online Repository for cytogenetics, FISH analyses, and RT-PCR.
RESULTS
Clinical findings
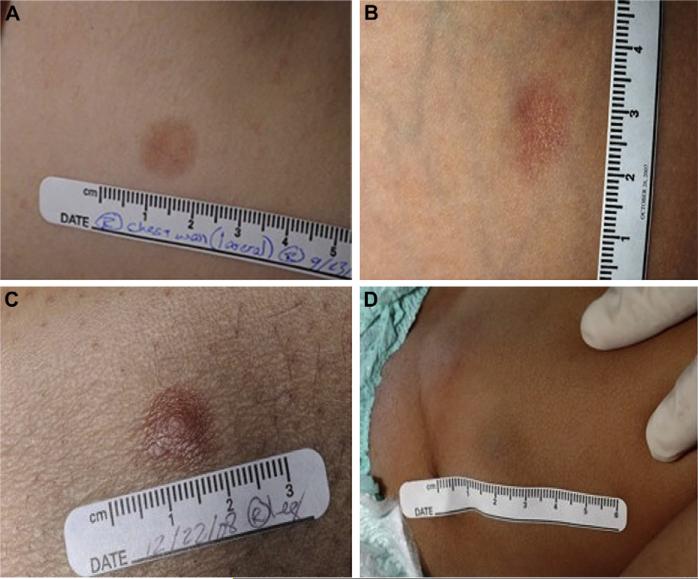
On examination, 11 of 12 patients presented with 1 or more skin lesions that warranted biopsy. The majority of lesions were small, atrophic, red-brown plaques less than 1 cm in greatest dimension. Plaques were either raised, flat, or depressed. One lesion presented as a slowly healing ulceration in the context of diaper dermatitis. One other lesion was detected as a violaceous, painless, subcutaneous mass (Fig 1). Only a minority of lesions had a nodular appearance. In one case a nodule developed within a previously identified plaque. In another case the lesion was nodular at diagnosis. Lesions were distributed on the trunk and extremities and ranged in number from 1 to 15 per patient. No facial or scalp lesions were observed.
FIG 1.
Clinical presentation of DFSP in patients with ADA-SCID. A, Flat plaque (ADA12). B, Depressed plaque (ADA7). C, Slightly raised plaque (ADA1; see also Fig 2, B, E, and H). D, Subcutaneous nodule (ADA16; see also Fig 2, C, F, and I).
Typically, patients, their parents, or both were unable to define the time of onset of the lesions that, in some cases, were reported as present from birth. In patients in whom serial examinations were performed, growth of some lesions and appearance of new lesions within 1 to 2 years of the initial examination could be documented. In 1 patient (ADA14) a nodular lesion was only ascertained after computed tomographic radiologic staging.
At diagnosis, patients were typically lymphopenic (range, 184-478 lymphocytes/μL) but showed conserved lymphocyte proliferation responses to PHA at levels varying between 91% and 581% of the lower limit of normal, with the exception of patient ADA7, who showed PHA responses at only 14% of the lower limit of normal.
Histology and immunohistochemistry
Eight of 11 patients in whom a biopsy of at least 1 lesion was performed showed a uniformly CD34+ spindle cell proliferation consistent with a diagnosis of DFSP. As reported in Table I and Fig 2, various histologic features were appreciated. Lesions from 2 patients (ADA12 and ADA16) showed classic findings of giant cell fibroblastoma with evidence of floret cells, multinucleated giant cells, loose myxoid stroma, and pseudovascular spaces (Fig 2, C and F). The classic DFSP storiform pattern was described only in 1 patient (ADA1; Fig 2, B and E). All other CD34+ lesions showed a nonspecific pattern of spindle cell proliferation. Cellularity ranged from paucicellular to densely cellular, and mitotic activity was low (<2 per high-powered field). In all cases tumor borders were indistinct, often infiltrating into the sub-cutaneous tissue. In several lesions this infiltration was best appreciated by using immunohistochemistry evaluation for CD34 (Fig 1, G). All CD34+ lesions were negative for Factor XIII and S100. DFSP was ruled out from biopsy material in 3 patients (Table I).
FIG 2.
Histologic features of DFSP in patients with ADA-SCID. A and D, Low cellularity from a depressed plaque (ADA14). B and E, Storiform pattern from a raised plaque (ADA1). C and F, Vascular spindle cell lesion in a myxoid background with floret cells characteristic of GCF (ADA16). G-I, CD34 stain decorating spindle cells in all 3 lesions.
Cytogenetic and molecular findings
Eight lesions from 6 patients with a histologic diagnosis consistent with DFSP yielded sufficient fresh tissue for conventional cytogenetics. An abnormal karyotype revealing the characteristic t(17;22)(q22;q13) translocation was evident in all but 1 lesion from 1 patient (ADA4; Table I and Fig 3, A). However, another lesion from the same patient showed the characteristic translocation t(17;22)(q22;q13). No ring chromosomes were identified. Biopsy specimens from 2 patients (ADA6 and ADA9) with inconclusive histologic findings showed normal karyotype.
FIG 3.
Molecular characteristics of DFSP in patients with ADA-SCID. A, Karyotyping from a lesion (ADA14) showing t(17;22)(q22;q13). B, FISH of a lesion (ADA16) revealing 2 fused red/green signals consistent with the presence of the COL1A1-PDGFB fusion. C, Sequence of the RT-PCR product from a lesion (ADA7) showing the junction of the COL1A1 and PDGFB transcripts.
Nineteen lesions from the 8 patients with histologic findings consistent with DFSP were studied by using FISH. In 14 lesions from 7 patients, fusion of the COL1A1 and PDGFB loci was identified (Table I). FISH analysis of 4 lesions yielded equivocal results or failed because of insufficient cellularity of the samples, exhaustion of the lesional tissue, or possibly prolonged formalin exposure. In 1 patient (ADA7) no additional samples were available to study. In the other 3 cases (patients ADA12, ADA14, and ADA16) additional biopsy specimens were found to be positive for the COL1A1-PDGF fusion signal. In patient ADA4, 2 CD34+ spindle cell lesions were studied by using FISH, only one of which was found to harbor fusion of the COL1A1 and PDGFB loci. As expected, FISH results were negative for a COL1A1-PDGFB fusion in the 2 lesions studied from patients ADA6 and ADA9, in whom histologic findings did not support the diagnosis of DFSP (Table I).
Twenty lesions from the 8 patients with histologic diagnosis consistent with DFSP were studied by using RT-PCR for the presence of COL1A1-PDGFB fusion transcripts. Nine lesions from 6 patients returned positive results, including 1 lesion that had resulted in equivocal FISH findings (patient ADA7). On the contrary, RT-PCR returned negative results in 1 lesion from patient ADA5, who was given a diagnosis of being positive for the presence of fusion of the COL1A1 and PDGFB loci by means of FISH. Noninformative RT-PCR results were obtained from 10 lesions of 5 patients. Of the 10 lesions, 4 had been found to harbor a COL1A1-PDGFB fusion by using FISH, and 1 was found to be negative by using FISH (ADA4, Table I). Discordant FISH and RT-PCR findings can be explained by differences in sampling, sensitivity, or both between the 2 technical approaches or possibly unusual molecular variants not detectable with the primer set or sets used.
In summary, 26 skin biopsy specimens were performed in 11 patients with ADA-SCID. A total of 22 lesions from 8 patients showed CD34+ spindle cell proliferation consistent with DFSP. The diagnosis of DFSP was unequivocally established in all 8 patients by means of cytogenetic testing, molecular testing, or both. Eight lesions were positive for the presence of the COL1A1-PDGFB fusion by using both FISH and RT-PCR, 6 were determined to be positive by using FISH and not informative or negative by using RT-PCR, and 1 was determined to be positive by using RT-PCR but equivocal by using FISH. Importantly, in those cases in which multiple lesions from a single patient were studied, different breakpoints were identified by means of sequence analysis, thus indicating independently originating events (data not shown).
DISCUSSION
We report here the association of 2 rare conditions: ADA-SCID and DFSP. Of 12 patients studied, 8 had DFSP, and in 7 patients the tumor had multicentric presentation.
The occurrence of DFSP in patients with ADA-SCID is remarkable in several ways. At diagnosis, our cohort of affected patients had an average age of 8.9 years (range, 2-22 years), which points to ADA-SCID as a condition that predisposes to childhood presentation of DFSP, a rare occurrence in itself. More remarkable still is the finding of multiple DFSP lesions in 7 (87.5%) of 8 patients. In addition, most DFSP lesions presented in the preprotuberant morpheaform plaque stage, which might be overlooked by parents and clinicians alike. Finally, the histology of lesions differed from that of classic DFSP by the absence of the classic storiform histologic pattern. Only in one of the 8 patients in whom DFSP was diagnosed did the lesion present as a protuberant nodule and show the typical microscopic storiform features (Figs 1, C, and 2, B and E).
Two case reports have described single DFSP and GCF lesions in isolated cases of ADA-SCID.19,20 A third case has been reported in abstract form.21 However, our observations are unprecedented because they establish a marked increase in the incidence of such tumors in patients with ADA-SCID. The previous lack of reporting of this finding might reflect underdiagnosis because of the benign appearance of the plaques, the very slow growth of the tumors, and the limited life expectancy of children with ADA-SCID. In contrast to the previous reported cases, our series also demonstrates a remarkable phenomenon of multiple DFSP tumors (up to 15) in patients with ADA-SCID. The finding of different COL1A1-PDGFB junction breakpoints in different tumors argues that these lesions are multiple primary malignant events and not cutaneous metastases. No correlations were noted between locations of the breakpoints and the clinical/pathologic features of the tumors.
The natural history of DFSP tumors in the setting of ADA-SCID is unknown. Most of the lesions found in our patients were atrophic plaques rather than nodular tumors at the time of diagnosis and lacked the classic storiform histology. One potential explanation is that lesions were detected earlier than typical DFSP because of the heightened scrutiny of our patient population.
Our findings are relevant to the clinical management of ADA-SCID and should prompt physicians to have a high clinical suspicion for DFSP. Importantly, small DFSP lesions were difficult to distinguish from small scars and areas of benign macular pigmentation. Furthermore, the atypical histology of DFSP in the setting of ADA-SCID might lead to histologic misdiagnosis in the absence of clinical correlation. Our observations that not all DFSP lesions were positive by using conventional cytogenetics, FISH, and RT-PCR suggest that a combination of immunohistochemical, cytogenetic, and/or molecular studies should be considered to rule out DFSP in any spindle cell lesion identified in patients with ADA-SCID.
The high frequency of a rare tumor in patients with a separate rare disease argues for the existence of a common pathophysiologic mechanism. Given the underlying immunodeficient state of patients with ADA-SCID, one possible hypothesis would be an infectious cause of DFSP tumors. Although we cannot formally rule out this possibility, results of preliminary investigations of in situ hybridization using degenerate primers targeting herpes virus sequences were negative. Similarly, electron microscopic studies of biopsy samples did not detect viral particles (data not shown). In addition, we have not observed any correlation between the immunologic and metabolic status of our patients at the time of screening or the type of therapeutic intervention used to treat their underlying ADA deficiency and the occurrence of DFSP. However, it is worth noting that because it was not possible to clearly define the time of occurrence of the tumors in our patients, the immunologic and metabolic details of the subjects at the time of tumor appearance remain unknown. Of note, occurrence and relapse of DFSP have been reported after successful HCT and immunologic correction with PEG-ADA,19-21 which allows us to postulate that DFSP can develop in ADA-deficient patients despite successful therapeutic intervention.
An alternative explanation is that the presence of DFSP in our patients is related to the metabolic consequences of ADA deficiency. ADA deficiency causes increased concentrations of adenosine and deoxyadenosine in the plasma of affected patients.1,2 Detailed information regarding adenosine/deoxyadeno-sine levels in tissues of patients with ADA-SCID is not available; however, studies in ADA-deficient mice have shown increased adenosine and deoxyadenosine levels in multiple organs, including the thymus, spleen, liver kidney, lung, and skin.22,23 In these models increased tissue adenosine levels have been associated with pulmonary and dermal fibrosis.22,24,25 Interestingly, a number of profibrotic mediators, including TGF-b1, IL-16, IL-13, connective tissue growth factor, and PDGF-AA, are significantly increased in skin homogenates from ADA-deficient mice.22 Although there are no reports of a general increase in dermal fibrosis in patients with ADA deficiency, it can be postulated that patients with ADA-SCID might be exposed to the fibrogenic effects of adenosine in the skin, as well as in other organs. Therapeutic interventions that replace ADA activity, such as HCT, enzyme replacement therapy, and gene therapy, are expected to decrease the concentrations of ADA substrates. In fact, administration of PEG-ADA has been shown to prevent and reverse lung fibrosis in mice.25 Data on adenosine tissue levels in human subjects treated with PEG-ADA are not available. However, plasma adenosine levels have been reported to remain high after HCT,1,26 which could lead to the presence of a profibrotic milieu in patients with ADA-SCID both before and after the implementation of measures aimed at the correction of their immune function.
It is also important to note that deoxyadenosine is known to cause single-strand DNA breaks and that deoxyadenosine triphosphate accumulation can interfere with DNA repair mechanisms.27 Therefore it is possible that the increased propensity for DNA strand breaks in ADA deficiency coupled with a fibrogenic dermal environment creates unique conditions amenable to the development of fibrous tumors, such as DFSP.
In conclusion, we show that DFSP is commonly found in patients with ADA-SCID. The association of the 2 disorders and the frequent multicentricity of DFSP are previously unreported observations and have important implications for further studies of the pathogenesis and pathophysiology of both diseases.
Acknowledgments
We thank Drs Linda M. Muul and Michael S. Hershfield for biochemical assays and helpful discussions, Dr Richard Sherry for surgical care and expertise, and Dr Jeffrey Cohen and his laboratory for performing EBV PCR assays. We thank the patients and their families for volunteering to participate in the clinical research studies.
Supported by the Intramural Research Program of the National Institutes of Health, National Human Genome Research Institute and National Cancer Institute.
Abbreviations used
- ADA
Adenosine deaminase
- ADA-SCID
Adenosine deaminase–deficient severe combined immune deficiency
- DFSP
Dermatofibrosarcoma protuberans
- FISH
Fluorescence in situ hybridization
- GCF
Giant cell fibroblastoma
- HCT
Hematopoietic cell transplantation
- PDGFβ
Platelet-derived growth factor β
- PEG-ADA
Pegylated bovine adenosine deaminase
METHODS
Cytogenetic and FISH analyses
Conventional cytogenetic analysis was performed on representative fresh tissue according to standard procedures of chromosomal preparations. For each case, 15 to 25 Giemsa-trypsin-Giemsa–banded metaphase cells were analyzed. FISH analysis was performed either on cytologic touch preparations or on formalin-fixed, paraffin-embedded tissue sections by using a homebrew dual-color (a spectrum orange-labeled COL1A1 probe and a spectrum green-labeled PDGFB), dual-fusion probe set that spans the COL1A1 and PDGFB loci and has been validated for use in the clinical laboratory of the University of Nebraska Medical Center.E1 Hybridization signals were assessed in 100 to 200 interphase nuclei; strong well-delineated signals were independently scored by 2 subjects. An interphase cell specimen was interpreted as abnormal if juxtaposed COL1A1-PDGFB probe set signals were detected in greater than 10% of cells (>2 SDs greater than the average false-positive rate). Images were acquired with the CytoVision Image Analysis System (Applied Imaging, Santa Clara, Calif).
RNA extraction and RT-PCR
RNA extraction from either two 20-μm paraffin-embedded representative tumor tissue sections or from 3 to 6 unstained formalin-fixed, paraffin-embedded tissue sections was carried out with the Optimum FFPE RNA Isolation Kit (Ambion Diagnostics, Austin, Tex), according to the manufacturer's instructions. RT-PCR to detect COL1A1-PDGFB fusion transcripts was performed by using sets of specific COL1A1 forward primers and a single PDGFB reverse primer, as previously described.E2 PCR products were sequenced with the ABI PRISM Dye Terminator Cycle Sequencing Ready Reaction kit and the ABI PRISM 3100-Avant Genetic Analyzer (Applied Biosystems, Foster City, Calif). Sequence and breakpoint analyses were performed by using BLAST sequence similarity searches.
Footnotes
Disclosure of potential conflict of interest: The authors declare that they have no relevant conflicts of interest.
Clinical implications: Patients affected by ADA deficiency should be screened for the presence and followed for the possible occurrence of DFSP.
REFERENCES
- 1.Hirschorn R, Candotti F. Immunodeficiency due to defects of purine metabolism. In: Ochs H, Smith C, Puck J, editors. Primary immunodeficiency diseases. Oxford University Press; Oxford (United Kingdom): 2006. pp. 169–96. [Google Scholar]
- 2.Mitchell BS, Hershfield MS. Immunodeficiency diseases caused by adenosine deaminase deficiency and purine nucleoside phosphorylase deficiency. In: Scriver CR, editor. Metabolic and molecular bases of inherited disease. 8th ed. McGraw-Hill; New York: 2001. [Google Scholar]
- 3.Gaspar HB, Aiuti A, Porta F, Candotti F, Hershfield MS, Notarangelo LD. How I treat ADA deficiency. Blood. 2009;114:3524–32. doi: 10.1182/blood-2009-06-189209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bowne WB, Antonescu CR, Leung DH, Katz SC, Hawkins WG, Woodruff JM, et al. Dermatofibrosarcoma protuberans: a clinicopathologic analysis of patients treated and followed at a single institution. Cancer. 2000;88:2711–20. [PubMed] [Google Scholar]
- 5.Fletcher CD. Pathology and genetics of tumours of soft tissue and bone (WHO classification of tumours) World Health Organization; Geneva (Switzerland): [Google Scholar]
- 6.Criscione VD, Weinstock MA. Descriptive epidemiology of dermatofibrosarcoma protuberans in the United States, 1973 to 2002. J Am Acad Dermatol. 2007;56:968–73. doi: 10.1016/j.jaad.2006.09.006. [DOI] [PubMed] [Google Scholar]
- 7.Topar G, Honlinger B, Eisendle K, Zelger B. An unusual congenital lesion in a 26-year-old woman. Clin Exp Dermatol. 2008;33:373–4. doi: 10.1111/j.1365-2230.2007.02501.x. [DOI] [PubMed] [Google Scholar]
- 8.Monnier D, Vidal C, Martin L, Danzon A, Pelletier F, Puzenat E, et al. Dermatofibrosarcoma protuberans: a population-based cancer registry descriptive study of 66 consecutive cases diagnosed between 1982 and 2002. J Eur Acad Dermatol Venereol. 2006;20:1237–42. doi: 10.1111/j.1468-3083.2006.01780.x. [DOI] [PubMed] [Google Scholar]
- 9.Weinstein JM, Drolet BA, Esterly NB, Rogers M, Bauer BS, Wagner AM, et al. Congenital dermatofibrosarcoma protuberans: variability in presentation. Arch Dermatol. 2003;139:207–11. doi: 10.1001/archderm.139.2.207. [DOI] [PubMed] [Google Scholar]
- 10.Reddy C, Hayward P, Thompson P, Kan A. Dermatofibrosarcoma protuberans in children. J Plast Reconstr Aesthet Surg. 2009;62:819–23. doi: 10.1016/j.bjps.2007.11.009. [DOI] [PubMed] [Google Scholar]
- 11.Terrier-Lacombe MJ, Guillou L, Maire G, Terrier P, Vince DR, de Saint Aubain Somerhausen N, et al. Dermatofibrosarcoma protuberans, giant cell fibroblastoma, and hybrid lesions in children: clinicopathologic comparative analysis of 28 cases with molecular data—a study from the French Federation of Cancer Centers Sarcoma Group. Am J Surg Pathol. 2003;27:27–39. doi: 10.1097/00000478-200301000-00004. [DOI] [PubMed] [Google Scholar]
- 12.Jafarian F, McCuaig C, Kokta V, Laberge L, Ben Nejma B. Dermatofibrosarcoma protuberans in childhood and adolescence: report of eight patients. Pediatr Dermatol. 2008;25:317–25. doi: 10.1111/j.1525-1470.2008.00674.x. [DOI] [PubMed] [Google Scholar]
- 13.Checketts SR, Hamilton TK, Baughman RD. Congenital and childhood dermatofibrosarcoma protuberans: a case report and review of the literature. J Am Acad Dermatol. 2000;42(suppl):907–13. doi: 10.1016/s0190-9622(00)90270-6. [DOI] [PubMed] [Google Scholar]
- 14.DuBay D, Cimmino V, Lowe L, Johnson TM, Sondak VK. Low recurrence rate after surgery for dermatofibrosarcoma protuberans: a multidisciplinary approach from a single institution. Cancer. 2004;100:1008–16. doi: 10.1002/cncr.20051. [DOI] [PubMed] [Google Scholar]
- 15.Kimmel Z, Ratner D, Kim JY, Wayne JD, Rademaker AW, Alam M. Peripheral excision margins for dermatofibrosarcoma protuberans: a meta-analysis of spatial data. Ann Surg Oncol. 2007;14:2113–20. doi: 10.1245/s10434-006-9233-3. [DOI] [PubMed] [Google Scholar]
- 16.Sandberg AA, Bridge JA. Updates on the cytogenetics and molecular genetics of bone and soft tissue tumors. Dermatofibrosarcoma protuberans and giant cell fibroblastoma. Cancer Genet Cytogenet. 2003;140:1–12. doi: 10.1016/s0165-4608(02)00848-8. [DOI] [PubMed] [Google Scholar]
- 17.Sirvent N, Maire G, Pedeutour F. Genetics of dermatofibrosarcoma protuberans family of tumors: from ring chromosomes to tyrosine kinase inhibitor treatment. Genes Chromosomes Cancer. 2003;37:1–19. doi: 10.1002/gcc.10202. [DOI] [PubMed] [Google Scholar]
- 18.Patel KU, Szabo SS, Hernandez VS, Prieto VG, Abruzzo LV, Lazar AJ, et al. Dermatofibrosarcoma protuberans COL1A1-PDGFB fusion is identified in virtually all dermatofibrosarcoma protuberans cases when investigated by newly developed multiplex reverse transcription polymerase chain reaction and fluorescence in situ hybridization assays. Hum Pathol. 2008;39:184–93. doi: 10.1016/j.humpath.2007.06.009. [DOI] [PubMed] [Google Scholar]
- 19.Carroll D, Ramani P, Lander AD. Giant-cell fibroblastoma in a patient with a bone-marrow transplant. Pediatr Surg Int. 2003;19:495–6. doi: 10.1007/s00383-002-0827-y. [DOI] [PubMed] [Google Scholar]
- 20.Rubocki RJ, Parsa JR, Hershfield MS, Sanger WG, Pirruccello SJ, Santisteban I, et al. Full hematopoietic engraftment after allogeneic bone marrow transplantation without cytoreduction in a child with severe combined immunodeficiency. Blood. 2001;97:809–11. doi: 10.1182/blood.v97.3.809. [DOI] [PubMed] [Google Scholar]
- 21.Nelson R, Shmalo J, Williams D, Hershfield M. Longterm survival and pegylated adenosine deaminase (ADA) therapy in a patient with ADA deficiency. Clin Immunol. 2006;119(suppl):S138. [Google Scholar]
- 22.Fernandez P, Trzaska S, Wilder T, Chiriboga L, Blackburn MR, Cronstein BN, et al. Pharmacological blockade of A2A receptors prevents dermal fibrosis in a model of elevated tissue adenosine. Am J Pathol. 2008;172:1675–82. doi: 10.2353/ajpath.2008.070952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Migchielsen AA, Breuer ML, van Roon MA, te Riele H, Zurcher C, Ossendorp F, et al. Adenosine-deaminase-deficient mice die perinatally and exhibit liver-cell degeneration, atelectasis and small intestinal cell death. Nat Genet. 1995;10:279–87. doi: 10.1038/ng0795-279. [DOI] [PubMed] [Google Scholar]
- 24.Blackburn MR, Volmer JB, Thrasher JL, Zhong H, Crosby JR, Lee JJ, et al. Metabolic consequences of adenosine deaminase deficiency in mice are associated with defects in alveogenesis, pulmonary inflammation, and airway obstruction. J Exp Med. 2000;192:159–70. doi: 10.1084/jem.192.2.159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Chunn JL, Molina JG, Mi T, Xia Y, Kellems RE, Blackburn MR. Adenosine-dependent pulmonary fibrosis in adenosine deaminase-deficient mice. J Immunol. 2005;175:1937–46. doi: 10.4049/jimmunol.175.3.1937. [DOI] [PubMed] [Google Scholar]
- 26.Hirschhorn R, Roegner-Maniscalco V, Kuritsky L, Rosen FS. Bone marrow transplantation only partially restores purine metabolites to normal in adenosine deaminase-deficient patients. J Clin Invest. 1981;68:1387–93. doi: 10.1172/JCI110389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Johnstone AP, Williams GT. Role of DNA breaks and ADP-ribosyl transferase activity in eukaryotic differentiation demonstrated in human lymphocytes. Nature. 1982;300:368–70. doi: 10.1038/300368a0. [DOI] [PubMed] [Google Scholar]
- E1.Huang D, Sumegi J, Dal Cin P, Reith JD, Yasuda T, Nelson M, et al. C11orf95-MKL2 is the resulting fusion oncogene of t(11;16)(q13;p13) in chondroid lipoma. Genes Chromosomes Cancer. 2010;49:810–8. doi: 10.1002/gcc.20788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- E2.Patel KU, Szabo SS, Hernandez VS, Prieto VG, Abruzzo LV, Lazar AJ, et al. Dermatofibrosarcoma protuberans COL1A1-PDGFB fusion is identified in virtually all dermatofibrosarcoma protuberans cases when investigated by newly developed multiplex reverse transcription polymerase chain reaction and fluorescence in situ hybridization assays. Hum Pathol. 2008;39:184–93. doi: 10.1016/j.humpath.2007.06.009. [DOI] [PubMed] [Google Scholar]