Abstract
Promoting ectopic development of pancreatic beta cells from other cell types is one of the strategies being pursued for the treatment of diabetes. To achieve this, a detailed outline of the molecular lineage that operates in pancreatic progenitor cells to generate beta cells over other endocrine cell types is necessary. Here, we demonstrate that early transient expression of the endocrine progenitor bHLH protein Neurogenin 3 (Ngn3) favors the promotion of pancreatic beta and delta cell fates over an alpha cell fate, while later transient expression promotes ectopic development of all three endocrine cell fates. We found that short-term activation of Ngn3 in Xenopus laevis endoderm just after gastrulation was sufficient to promote both early and ectopic development of beta and delta cells. By examining gene expression changes four hours after Ngn3 activation we identified several new downstream targets of Ngn3. We show that several of these are required for the promotion of ectopic beta cells by Ngn3 as well as for normal beta cell development. These results provide new detail regarding the Ngn3 transcriptional network operating in endocrine progenitor cells to specify a beta cell phenotype and should help define new approaches to promote ectopic development of beta cells for diabetes therapy.
Keywords: Diabetes, Reprogramming, Endoderm Progenitor, Microarray, Endocrine, Transcription Factor, Hormone inducible
Introduction
The pancreatic transcriptional program defining development of the various cell types is conserved from zebrafish to Xenopus to mammals (Gittes, 2009; Pearl et al., 2009; Tiso et al., 2009). In Xenopus laevis both early acting genes, such as Ptf1a (Pancreas Specific Transcription Factor, 1a) and Pdx1 (Pancreatic and Duodenal Homeobox 1), and later acting genes, such as Insm1 (Insulinoma-Associated 1) and Rfx6 (Regulatory Factor X, 6), have been shown to operate in a similar fashion as their mammalian counterparts (Afelik et al., 2006; Horb et al., 2009; Jarikji et al., 2007; Pearl et al., 2011). However, as expected there are some species specific characteristics of how the pancreas develops in Xenopus. Morphologically, the dorsal and ventral anlagen do not develop as buds off the gut tube as is seen in mammals. During early stages the endoderm in Xenopus is used as a nutrient source for the developing embryo and tadpole, and as such organization of the endoderm into a gut tube occurs relatively late in development (Chalmers and Slack, 1998, 2000). The dorsal pancreatic bud first emerges at stage 35/36, with the two ventral buds arising slightly later at stage 37/38; these buds fuse by stage 39. Molecular genetic differences between these buds has recently been elucidated and shown to define the migratory behavior of how they fuse and identify spatial differences in cell fate specification (Horb and Horb, 2010; Jarikji et al., 2009). Differentiation of the various endocrine cell types occurs in a precisely defined pattern (Kelly and Melton, 2000; Pearl et al., 2009). Beta cells differentiate first only in the dorsal pancreatic endoderm with insulin expression first detected at stage 32; by stage 46/47 insulin expression can be detected in the ventral portion of the pancreas as well. In contrast, expression of glucagon and somatostatin is not pancreas specific. They are first expressed in the stomach/duodenum beginning at stage 40, with expression in the pancreas not detected until stage 44/45 (Horb and Slack, 2002). The fact that the mammalian endocrine transcriptional program is identical in Xenopus, combined with the major embryological and molecular benefits of this model, make it an ideal system for elucidation of the transcriptional regulatory network controlling development of the various pancreatic cell types.
Recent advances in promoting ectopic development of beta cells using defined factors have contributed greatly towards a cell-based replacement therapy for diabetes (Borowiak and Melton, 2009; Horb et al., 2003; Kordowich et al., 2010; Murtaugh, 2007; Pearl and Horb, 2008; Yechoor and Chan, 2010). The goal has been to use pancreatic transcription factors to reprogram other cell types, whether differentiated cells or stem cells, into Insulin-expressing endocrine beta cells. Many of these studies however, did not result in the promotion of a single cell fate, but rather resulted in ectopic promotion of both exocrine and endocrine cell fates. In an effort to promote only endocrine cell fates, work has focused on using the endocrine progenitor bHLH protein Neurogenin 3 (Ngn3) (Rukstalis and Habener, 2009). This has not however resulted in the promotion of insulin producing beta cells, but rather in ectopic development of glucagon producing alpha cells (Apelqvist et al., 1999; Grapin-Botton et al., 2001; Schwitzgebel et al., 2000).
Recently, it was shown that the timing of Ngn3 function during embryonic development was essential in regulating its ability to promote one endocrine cell fate over another (Johansson et al., 2007), suggesting that it is the competence of the responding cell that is important. In agreement with this, it was found that a combination of Ngn3 and other pancreatic factors that sequentially contribute to beta cell development was sufficient to convert acinar cells to beta cells (Zhou et al., 2008). These results point to the fact that understanding how transcriptional information is integrated downstream of Ngn3 to specify the beta cell lineage over other endocrine lineages would help in the development of strategies for the directed differentiation of functional beta cells from other cell types.
Over the last decade much effort has been placed on elucidating the Ngn3 transcriptional regulatory network. Studies have focused on identifying gene expression changes in various contexts including, in pancreatic tissue lacking Ngn3 (Petri et al., 2006), in pancreatic tissue or stem cells overexpressing Ngn3 (Gasa et al., 2004; Serafimidis et al., 2008) and in endogenously isolated Ngn3+ cells (White et al., 2008). Though these studies have identified numerous potential targets, in each instance no separation was made to define one endocrine lineage over another, and the time points chosen for analysis were long after Ngn3 expression commenced. As such the Ngn3 transcriptional network that defines a single endocrine lineage (such as a beta cell) over another (such as an alpha cell) has yet to be resolved.
In this study we precisely define two different temporal windows in which Ngn3 promotes ectopic development of only beta and delta cells in the first instance and beta, delta and alpha cells in the second instance. By injecting a Dexamethasone inducible Ngn3 into Xenopus laevis embryos we were able to control its activity for a limited duration in the endoderm at specific times during development. Our results show that short-term activation (1–4 hours) immediately after gastrulation was sufficient to promote ectopic and premature development of pancreatic beta and delta cells, but not alpha cells. In contrast, continuous or short-term activation at late neurula stages resulted in ectopic development of all three endocrine lineages. Based on these phenotypic differences, we identified early downstream targets of Ngn3 involved in beta cell development by performing a microarray analysis of early endoderm four hours after Ngn3 activation. We identified several new genes required not only for Ngn3-induced ectopic beta cell development, but also for normal beta cell development. These results provide new information regarding the early transcriptional network of beta cells that will help define methods for the promotion of beta cells for diabetes therapies.
Results
Transient Ngn3 misexpression promotes a β cell fate over an α cell fate
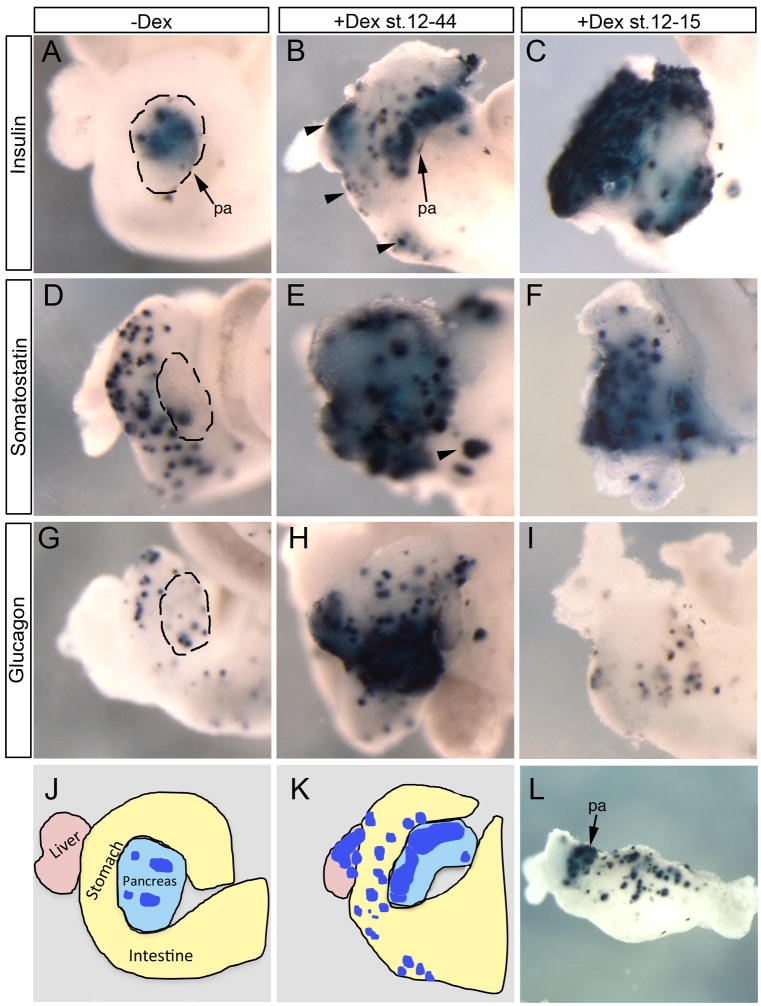
To define the competence window of ectopic beta cell creation by Ngn3, we sought to overexpress Ngn3 in naïve endoderm (prior to its endogenous expression) at different times and for limited duration. During normal development, endodermal expression of Ngn3 is not detected until late tail bud stages, similar to that seen with the zinc finger islet transcription factor Insulinoma Associated 1 (Insm1) (Horb et al., 2009). We constructed a Dexamethasone (Dex) inducible Ngn3 by fusing the glucocorticoid ligand binding domain (GR) to the C-terminus of Ngn3 (hence named Ngn3-GR) and injected it into vegetal blastomeres of Xenopus laevis embryos; mainly anterior endoderm was targeted. Continuous activation of Ngn3-GR after gastrulation (stages 12–44, 80 hours) promoted ectopic expression of insulin, somatostatin and glucagon (Fig. 1B,E,H). However, activation of Ngn3-GR for only four hours after gastrulation (stages 12–15) resulted in ectopic expression of only insulin and somatostatin and not glucagon (Fig. 1C,F,I). In both cases ectopic expression of endocrine markers was observed throughout the liver, stomach and duodenum (black arrowheads, Fig. 1B,E). In rare instances when more posterior endoderm was also targeted we observed ectopic expression of insulin in very posterior locations of the intestine (Fig. 1L).
Fig. 1. Differential effects of Ngn3-GR temporal activation.
Whole mount in situ hybridization of stage 44 whole guts from embryos injected with 1800pg of Xenopus Ngn3-GR mRNA at the eight-cell stage. (A,D,G) Control embryos injected with Ngn3-GR mRNA but not treated with dex. (B,E,H) Dex treatment from stages 12–44. Increased expression of glucagon (n=14/20), insulin (n=23/23) and somatostatin (n=10/10). Arrowheads indicate ectopic insulin expression in the liver, stomach and duodenum. (C,F,I) Dex treatment for 4 hours from stages 12–15. Increased insulin (n=55/55) and somatostatin (n=22/22) but not glucagon (n=24/24). (J,K) Schematics illustrating organs in the whole gut of panels a and b (blue-insulin). (L) Insulin expression was detected in rare instances (n=2/50) in posterior areas of the intestine. Pa: pancreas.
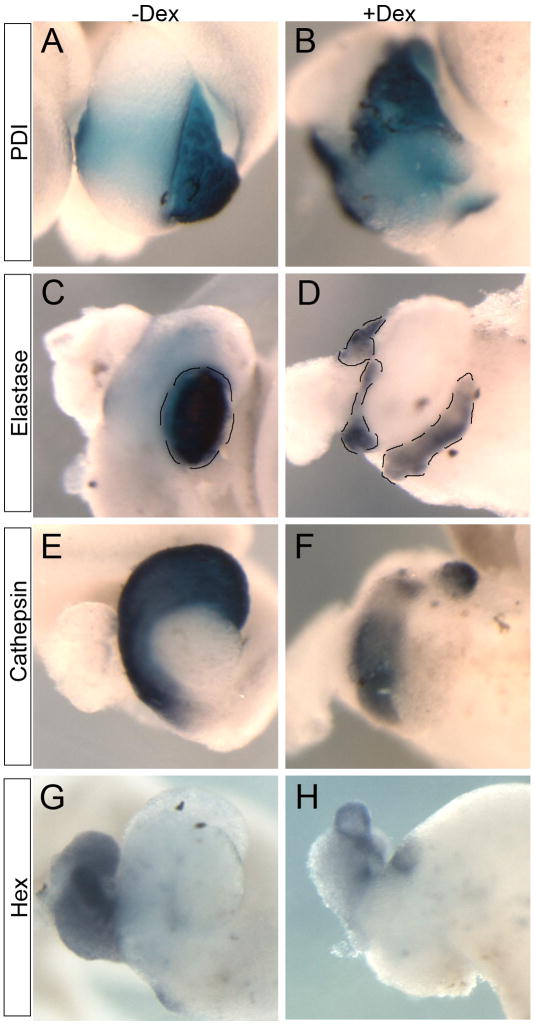
Although the morphology of the pancreas was disturbed, we did find expression of other acinar pancreatic markers, protein disulphide isomerase (XPDI) and elastase, in the tissue that remained (Fig. 2A–D). In contrast, expression of stomach and liver markers, cathepsin E and hex, were reduced (Fig. 2E–H). As we were expressing Ngn3-GR in the anterior endoderm (targeting stomach and liver in addition to pancreas) we believe the reduction in these markers was due to the fact that Ngn3 was converting prospective liver and stomach into pancreas. Unless specified, in all subsequent experiments we activated Ngn3-GR for four hours at stage 12.
Fig. 2. Ngn3-GR activation affects anterior gut markers at stage 44.
(A–D) Exocrine pancreatic markers PDI and elastase are slightly decreased (n=16/16 and n=14/15, respectively). (E,F) Stomach marker cathepsin E is significantly reduced (n=14/14). (G,H) Liver and duodenum marker hex is slightly decreased (n=7/7). Pancreas is outlined in panels C and D.
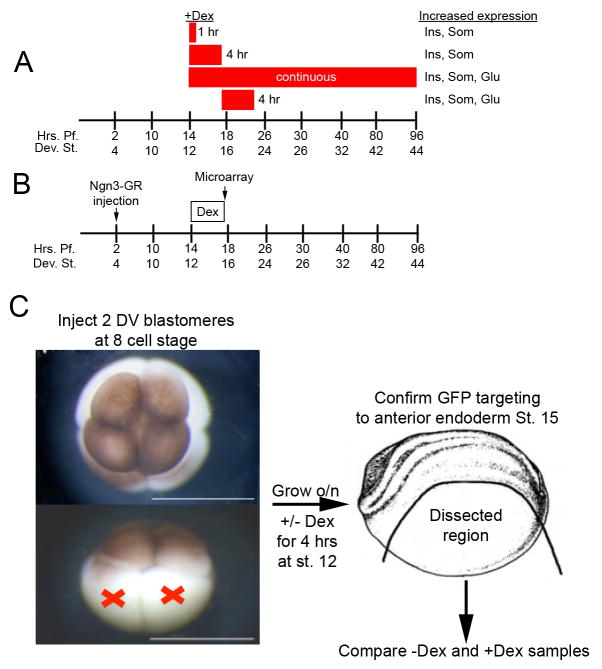
To ascertain whether it was the stage of Dex addition that was most critical in defining the different effects of Ngn3, we tested short-term activation of Ngn3-GR at later stages. Activation for 4 hours at stage 15 (until stage 20) resulted in increased expression of all the three hormones as previously seen with continuous activation (data not shown). To determine the minimal time of Ngn3 activity required for ectopic promotion of beta cells we activated Ngn3-GR for different time periods after gastrulation, and found that activation for as short as one hour (at either stage 12 or 14) was sufficient to promote ectopic insulin and somatostatin expression over glucagon expression (see Fig. 7A). These results show that the competence of endodermal cells is indeed important for the ability of Ngn3 to promote one endocrine cell type over another.
Fig. 7. Summary of results and schematic diagram of microarray experiment.
(A) Summary of ectopic expression of endocrine markers. Red boxes indicate time of Dex treatment. Activation of Ngn3-GR for 1 or 4 hours beginning at stage 12 resulted in increased insulin and somatostatin expression, whereas continuous activation beginning at stage 12 or a 4 hour activation beginning at stage 15 resulted in increased expression of insulin, somatostatin and glucagon. (B,C) Diagram of microarray experiment. Ngn3-GR mRNA was injected in the two dorsal vegetal blastomeres at the eight cell stage. Embryos were grown until stage 12 activated with dexamethasone for four hours and confirmed targeting to the anterior endoderm at stage 15 with GFP fluorescence. All dorsal structures were removed and RNA was extracted immediately after. Four replicates of ten embryos were used to hybridize to the Affymetrix Xenopus laevis GeneChip 2.0.
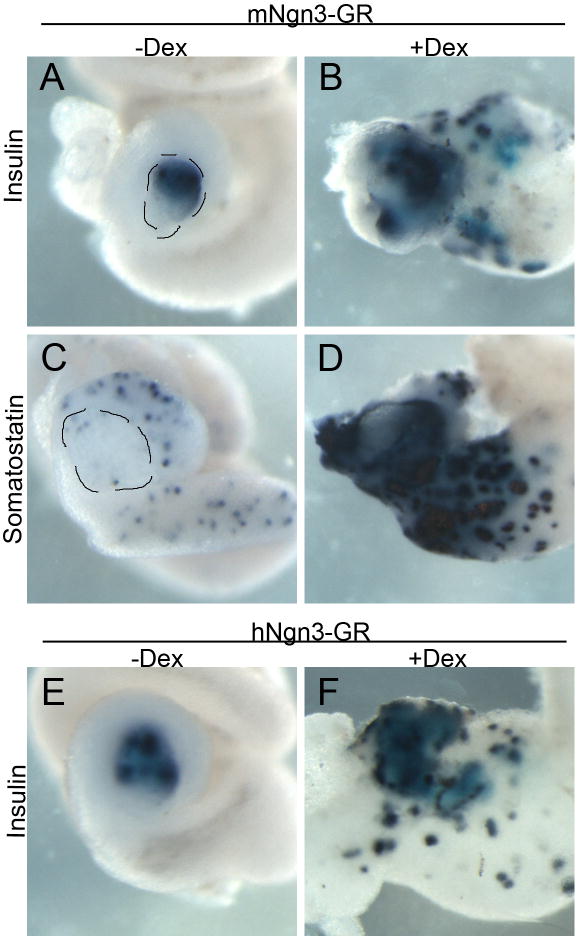
We believe the differential ability of Ngn3 to promote one cell fate over another is dependent on the protein complexes it forms, which is related to its amino acid sequence. To ascertain whether mouse and human Ngn3, which are only 55% identical to Xenopus Ngn3, were also able to act in a similar fashion to activate the same transcriptional network in promoting beta and delta fates over an alpha cell fate we created GR inducible versions of human and mouse Ngn3 and tested whether they also acted similarly to Xenopus Ngn3. We found that transient overexpression of either mouse or human Ngn3-GR was sufficient to specifically promote ectopic beta and delta cell fates and not alpha cell development similar to Xenopus Ngn3 (Fig. 3). The only difference we observed was in the amount of mRNA required to generate these phenotypes; human and Xenopus Ngn3 required the same amount of mRNA, whereas mouse Ngn3 was more active, requiring 50 times less mRNA; the reason for this difference is unclear. These results suggested that the important amino acids required for Ngn3 activity in promoting beta cell differentiation are found in the specific amino acids found in all three species.
Fig. 3. Transient activation of mouse Ngn3-GR and human Ngn3-GR also promoted increased insulin expression.
(A–D) Embryos injected with 15 pg of mouse Ngn3-GR and activated with dexamethasone for four hours at st.12 showed increased insulin (n=10/10) and somatostatin (n=7/7) expression. (E,F) Injection of 1800 pg of human Ngn3-GR also caused increased insulin expression (n=8/8). Pancreas is outlined in panels A and C.
Ngn3 misexpression prematurely activates insulin via Insm1 and Rfx6 function
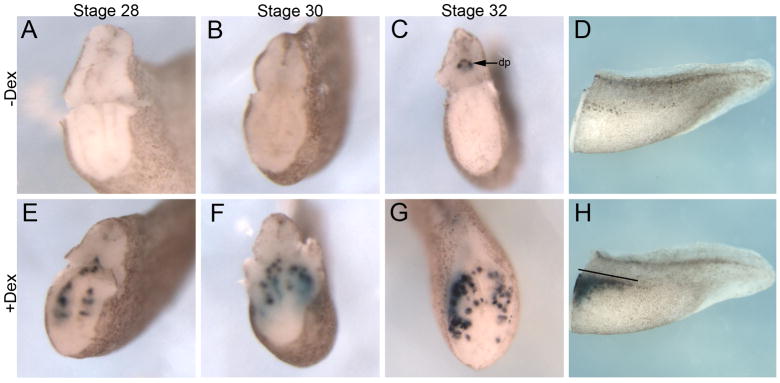
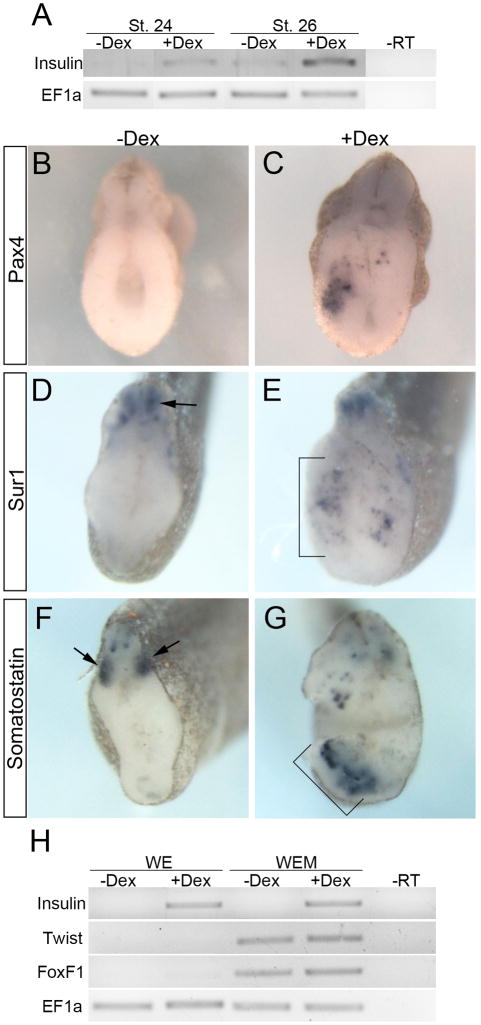
To establish the earliest point when ectopic insulin expression appeared upon Ngn3-GR misexpression, we determined at what stage ectopic insulin expression was first detected. Since endogenous insulin expression is initially detected in the dorsal pancreas at stage 32 (Pearl et al., 2009) we examined Ngn3-GR injected embryos at stages 28, 30 and 32 for ectopic expression of insulin. By in situ hybridization we found insulin to be expressed 8 hours earlier than endogenous (stage 28) in Ngn3-GR injected embryos treated with Dex (Fig. 4E). In addition to the timing differences, we also found insulin to be expressed more posteriorly and ventrally in the endoderm (Fig. 4H). Using RT-PCR we were able to detect ectopic insulin expression in Dex treated embryos as early as stage 24, which is about 12 hours after activation of Ngn3-GR (Fig. 5A). To eliminate the possibility that the results were due to ectopic transcription of the Insulin gene without true beta cell differentiation, we determined if other beta cell markers (Pax4 and Sur1) were also ectopically expressed. Pax4 (Paired Box 4) is the beta cell specific transcription factor, which has been shown to be both necessary and sufficient for proper differentiation of beta cells and not alpha cells (Collombat et al., 2003; Collombat et al., 2009; Sosa-Pineda et al., 1997). Sur1 (Sulfonylurea Receptor 1; also known as Abcc8, ATP-binding cassette sub-family C member 8) is the mature pancreatic beta cell KATP channel, which is critical for proper Insulin secretion (Bennett et al., 2010). In agreement with the fact that the ectopic insulin expressing cells were undergoing true differentiation into beta cells, we found ectopic and early expression of both pax4 and sur1 throughout the endoderm at stage 32 (Fig. 5B–E). In addition, we also found increased expression of the endocrine specific delta cell marker somatostatin at stage 32 (Fig. 5F,G). These results demonstrate that Ngn3-GR misexpression was sufficient to promote premature and ectopic differentiation of beta and delta cells within the early endoderm.
Fig. 4. Ectopic and early induction of beta cells by Ngn3-GR.
Insulin expression in tadpoles that have been sectioned through the dorsal pancreas, with the head removed. (A–D) Control injected embryos. Endogenous insulin expression is first detected at stage 32 in a small dorsal domain. Endogenous insulin expression in dorsal pancreas (dp) is labeled by arrow in panel C. (E–H) Ngn3-GR injected embryos treated with Dex for four hours at stage 12 showed increased insulin expression at stage 28 (n=14/18), stage 30 (n=24/30) and stage 32 (n=46/56). (H) Side view of stage 34 tadpole treated with Dex showing the extent of ectopic insulin expression (line).
Fig. 5. Ngn3-GR promotes beta cell differentiation.
(A) RT-PCR for insulin of whole embryos at stages 24 and 26. (B,C) Increased pax4 expression detected in Dex treated embryos at stage 32 (n=15/17). (D,E) Increase in the mature beta cell marker, sur1, at stage 32 (n=19/31) in Dex treated embryos. In panel D, arrow indicates nonendodermal expression of sur1. In panel E, bracket denotes ectopic expression in endoderm. (F,G) Increase in somatostatin at stage 32 (n=12/27) in Dex treated embryos. In panel F, arrow indicates expression of som outside the endoderm. In panel G, bracket denotes ectopic expression in endoderm. (H) RT-PCR at stage 26 of whole endoderm (WE) and whole endoderm plus mesoderm explants (WEM). Explants were dissected at stage 15 following four hours of Ngn3-GR activation, cultured until stage 26 and collected for RT-PCR. Twist and FoxF1 are mesoderm markers, which are detected only in the WEM explants.
Previous results in Xenopus have shown that endoderm-mesoderm interactions are critical for differentiation of pancreatic beta cells (Horb and Slack, 2001). We therefore examined whether Ngn3-GR was promoting ectopic beta cell differentiation independent of signals from the mesoderm. We isolated explants of either endoderm alone (WE) or endoderm plus mesoderm (WEM) from Ngn3-GR injected embryos at stage 15, grew them until stage 26 and examined for ectopic insulin expression by RT-PCR. In agreement with our hypothesis, ectopic insulin expression was detected in Dex treated explants of endoderm alone (Fig. 5H). These results show that Ngn3 misexpression in the early endoderm directly promoted beta cell differentiation independent of signals from the mesoderm, though we cannot exclude the remote possibility of earlier mesoderm interactions during the short Dex treatment.
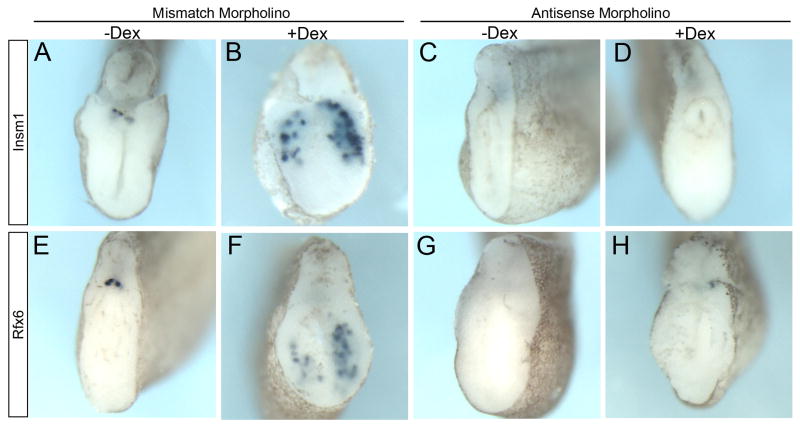
Since we were ectopically expressing Ngn3 (as well as using a hormone inducible version) at an earlier time and in ectopic locations than in normal development, it was possible that the pathway activated by Ngn3-GR was not the same as that utilized by endogenous Ngn3. To ascertain whether this promotion of early and ectopic beta cell differentiation by Ngn3 was indeed acting through the endogenous pathway of beta cell development, we examined whether this activity was dependent on the proper function of two endocrine specific transcription factors Insm1 and Rfx6, both of which have been shown to act downstream of Ngn3 in the endogenous pathway of endocrine cell development (Gierl et al., 2006; Horb et al., 2009; Mellitzer et al., 2006; Pearl et al., 2011; Smith et al., 2010; Soyer et al., 2010). Injection of the antisense Insm1 morpholinos abolished endogenous insulin expression in the −Dex controls as well as ectopic insulin expression in +Dex tadpoles (Fig. 6C,D). Similarly, knockdown of Rfx6 also inhibited the ability of Ngn3-GR to promote ectopic beta cell differentiation as well as endogenous insulin expression (Figs. 6G,H). Similar results were also observed for both the Insm1 and Rfx6 morpholinos when insulin expression was analyzed at stage 44 (data not shown). These results show that Ngn3 promotion of ectopic and early beta cell differentiation occurred through recognized pathways of beta cell differentiation.
Fig. 6. Insm1 and Rfx6 are necessary for ectopic beta cell promotion by Ngn3-GR.
In situ hybridizations for insulin expression in all panels. Ngn3-GR mRNA (1800pg) was coinjected with a control mismatch morpholino (mMO) or morpholinos targeting insm1 or rfx6 mRNA, activated with Dex for 4 hours and fixed at stage 32. (A,B) Insm1 mMO (20 ng) had no effect on endogenous insulin expression in either −Dex (n=8/8) or +Dex (n=12/17) treated embryos. (C,D) Injection of Insm1 MOs (2 morpholinos, 20 ng each) abolished endogenous insulin expression in −Dex embryos (n=7/7) as well as ectopic insulin expression in +Dex embryos (n=15/15). (E,F) Rfx6 mismatch morpholino (25ng) did not affect endogenous or ectopic insulin expression in −Dex (n=9/9) or +Dex embryos (n=19/41) (G,H) Antisense Rfx6 morpholino (25ng) inhibited endogenous insulin expression in -Dex embryos (n=33/33) as well as ectopic insulin expression in +Dex embryos (n=46/46).
Tbx2, Mtg8, Mtg16 and Mtgr1 are needed for ectopic beta cell development
To identify Ngn3 downstream targets involved in the formation of ectopic beta cells, we performed a microarray analysis immediately after activation of Ngn3-GR (Fig. 7B). Briefly, we activated Ngn3-GR injected embryos with Dexamethasone for four hours at stage 12 and collected embryos at stage 15 and removed all dorsal structures, which included neural tissue and ectoderm (Fig. 7C). Total RNA was extracted and hybridized to the Affymetrix Xenopus laevis GeneChip 2.0. Comparison of hybridization results between +Dex and −Dex samples (four replicates of ten embryos each) was done using the Robust Multi-Array Average and the algorithm EB (Wright and Simon) for statistical analysis. The results of this analysis yielded 162 genes upregulated by more than 1.5 fold in the +Dex samples (see Table 1 for a partial list). Several transcription factors that were previously identified as downstream of Ngn3 in other microarray studies were also identified in our screen, including Insm1 (2.51 fold), Neurogenic Differentiation 4 (NeuroD4; 2.12 fold), Zinc Finger Protein 238 (Zfp238; 2.65 fold), and Hairy and Enhancer of Split 5 (Hes5; 1.75 fold). In addition, we also found increased expression of several other transcription factors that have not been shown to function in beta cell development. These include the POU class V protein Oct-25 (Oct25; 2.48 fold), as well as all three Myeloid translocation gene family members (Mtg), Mtg8 (2.09 fold), Mtg16 (2.18 fold) and Mtgr1 (2.01 fold). MTG proteins are known to be important for neural and gut development and they act (to repress transcription) as mediators of multiprotein transcriptional networks to promote repression of target genes (Amann et al., 2005; Calabi et al., 2001; Koyano-Nakagawa and Kintner, 2005; Rossetti et al., 2004). We also found increased expression of other transcription factors, including several Hairy and enhancer of split related genes, Hes3 (2.2 fold), Hes5 (1.75 fold), Hes9 (1.6 fold) and Hey1 (1.69 fold), and the T-box gene Tbx2 (1.4 fold).
Table 1.
Partial list of genes upregulated in stage 15 endoderm after fours hours of Ngn3-GR activation
| Gene Symbol | Unigene ID | Gene Title | FC |
|---|---|---|---|
| MGC115198 | Xl.16704 | Hypothetical protein MGC115198 | 3.93 |
| Aif1l | Xl.10091 | Allograft inflammatory factor 1-like | 3.77 |
| Hba3 | Xl.1125 | Hemoglobin alpha 3 subunit | 3.02 |
| Slc16a3 | Xl.2852 | Solute carrier family 16, member 3 | 2.80 |
| Ankrd37 | Xl.8033 | Ankyrin repeat domain 37 | 2.67 |
| Btg-x | Xl.49497 | B-cell translocation protein x | 2.67 |
| Znf238.3 | Xl.47150 | Zinc finger protein 238.3 | 2.65 |
| LOC100158417 | Xl.17761 | Hypothetical protein LOC100158417 | 2.59 |
| Insm1 | Xl.14733 | Insulinoma-associated 1 | 2.51 |
| Oct-25 | Xl.48605 | POU class V protein oct-25 | 2.48 |
| Rfesd | Xl.29449 | Rieske (Fe-S) domain containing | 2.47 |
| Slc3a2 | Xl.3242 | Solute carrier family 3 member 2 | 2.47 |
| Pdk4 | Xl.1717 | Pyruvate dehydrogenase kinase, isozyme 4 | 2.42 |
| Brn3 | Xl.701 | POU class 4 homeobox 1 | 2.25 |
| Hes3.1 | Xl.585 | Hairy and enhancer of split 3, gene 1 | 2.23 |
| Twsg1 | Xl.502 | Twisted gastrulation homolog 1 | 2.21 |
| Hes3.3 | Xl.12067 | Hairy and enhancer of split 3, gene 3 | 2.18 |
| Mtg16 | Xl.14852 | Myeloid translocation gene 16 | 2.18 |
| Neurod4 | Xl.1263 | Neurogenic differentiation 4 | 2.12 |
| Ptgs2 | Xl.33329 | Prostaglandin-endoperoxide synthase 2 | 2.11 |
| Mtg8 | Xl.24705 | Myeloid translocation gene 8 | 2.09 |
| MGC68579 | Xl.34749 | Hypothetical protein MGC68579 | 2.08 |
| Cer1 | Xl.368 | Cerberus 1 | 2.07 |
| MGC131136 | Xl.14925 | Hypothetical protein MGC131136 | 2.05 |
| Dbndd1 | Xl.47316 | Dysbindin domain containing 1 | 2.02 |
| Tsc22d3 | Xl.12378 | TSC22 domain family, member 3 | 2.01 |
| Mtgr1 | Xl.4960 | Myeloid translocation gene Related 1 | 2.01 |
| Fgf13 | Xl.13859 | Fibroblast growth factor 13 | 1.98 |
| Chn1 | Xl.9113 | Chimerin | 1.92 |
| MGC68858 | Xl.16456 | Hypothetical protein MGC68858 | 1.90 |
| Fam101b | Xl.56544 | Family with sequence similarity 101, member B | 1.88 |
| Bmp7 | Xl.3326 | Bone morphogenetic protein 7 | 1.87 |
| Dll1 | Xl.14759 | Delta-like 1 | 1.85 |
| Elavl3 | Xl.1035 | ELAV (embryonic lethal, abnormal vision)-like 3 | 1.84 |
| Zfand2a | Xl.48357 | Zinc finger, AN1-type domain 2A | 1.82 |
| Stmn3 | Xl.21810 | Stathmin-like 3 | 1.81 |
| Ndrg4-b | Xl.9099 | Protein NDRG4-B | 1.81 |
| Hoxd1 | Xl.53491 | Homeobox D1 | 1.81 |
| Esr10-A | Xl.9271 | Enhancer of split related 10 | 1.80 |
| Chst10 | Xl.8239 | Carbohydrate sulfotransferase 10 | 1.80 |
| Atoh7-a | Xl.176 | Atonal homolog 5a | 1.79 |
| Dlc1 | Xl.47179 | Deleted in liver cancer 1 | 1.79 |
| Rgs1 | Xl.31883 | Regulator of G-protein signaling 1 | 1.76 |
| Irf-6 | Xl.1273 | Interferon regulatory factor 6 | 1.76 |
| Sgk223 | Xl.3085 | Tyrosine-protein kinase SgK223 | 1.76 |
| Hes5.1 | Xl.48575 | Hairy and enhancer of split 5, gene 1 | 1.75 |
| Foxa4-a | Xl.441 | Forkhead box A4 | 1.75 |
| Sat1 | Xl.24275 | Spermidine/spermine N1-acetyltransferase 1 | 1.74 |
| Pik3ip1 | Xl.49469 | Phosphoinositide-3-kinase interacting protein 1 | 1.73 |
| Igf2 | Xl.47110 | Insulin-like growth factor 2 | 1.72 |
| LOC100158359 | Xl.55698 | Hypothetical protein LOC100158359 | 1.71 |
| Pgam1 | Xl.11442 | Phosphoglycerate mutase 1 | 1.70 |
| Hey1 | Xl.469 | Hairy/enhancer-of-split related with YRPW motif 1 | 1.69 |
| Gadd45g | Xl.12125 | Growth arrest and DNA-damage-inducible, gamma | 1.69 |
| Stripy | Xl.9206 | Stripy | 1.69 |
| Unc119b-A | Xl.16769 | Protein unc-119 homolog B-A | 1.68 |
| LOC496170 | Xl.6950 | Hypothetical LOC496170 | 1.68 |
| MGC53782 | Xl.8440 | Similar to enhancer of split related | 1.68 |
| Ccnb1 | Xl.4297 | Cyclin B1 | 1.68 |
| LOC100036864 | Xl.50224 | Hypothetical protein LOC100036864 | 1.68 |
| Mospd1 | Xl.26524 | Motile sperm domain containing 1 | 1.67 |
| Kcnk5 | Xl.16485 | Potassium channel, subfamily K, member | 1.63 |
| Egln3 | Xl.12606 | Egl nine homolog 3 | 1.61 |
| Unc93a | Xl.48437 | Unc-93 homolog A | 1.60 |
| Hes9.1-b | Xl.12444 | hes9.1-b hairy and enhancer of split 9, gene 1 | 1.60 |
| Frzb | Xl.55584 | Frizzled-related protein | 1.60 |
| Dbn1 | Xl.49084 | Drebrin 1 | 1.60 |
| Ppp1r14a | Xl.24383 | Protein phosphatase 1, regulatory (inhibitor) subunit 14A | 1.60 |
| Tob1 | Xl.6421 | Transducer of ERBB2, 1 | 1.59 |
| Slc7a8 | Xl.15844 | Solute carrier family 7, member 8 | 1.59 |
| Dlc | Xl.68 | Putative ortholog of delta-like protein C precursor | 1.58 |
| Mespb | Xl.53943 | Mesoderm posterior homolog B | 1.58 |
| Gramd3 | Xl.7613 | GRAM domain containing 3 | 1.57 |
| Hspc159-a | Xl.13396 | Galectin-related protein A | 1.56 |
| Ggt1 | Xl.18682 | Gamma-glutamyltransferase 1 | 1.56 |
| Mxi1 | Xl.50498 | Max Interactor 1 | 1.56 |
| LOC100036879 | Xl.16140 | Hypothetical protein LOC100036879 | 1.55 |
| Ppm1k | Xl.17302 | Protein phosphatase, Mg2+/Mn2+ dependent, 1K | 1.53 |
| Asb5 | Xl.48897 | Ankyrin repeat and SOCS box-containing 5 | 1.52 |
| Ventx1.1 | Xl.1420 | VENT homeobox 1, gene 1 | 1.51 |
| Mcoln2 | Xl.1707 | Mucolipin 2 | 1.50 |
| Nudt22 | Xl.6817 | Nudix -type motif 22 | 1.50 |
| LOC100158350 | Xl.54372 | Hypothetical protein LOC100158350 | 1.50 |
| MGC115510 | Xl.53440 | Hypothetical protein MGC115510 | 1.50 |
| Arl5b-b | Xl.7620 | ADP-ribosylation factor-like 5B | 1.50 |
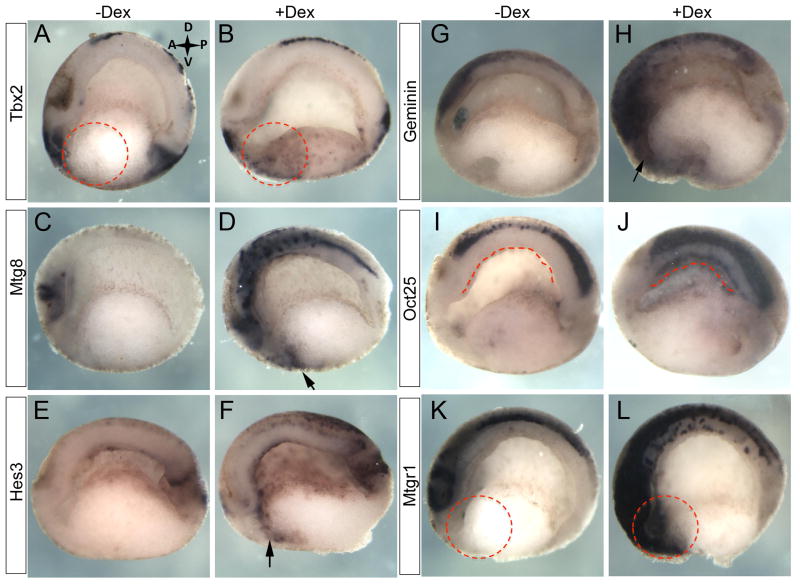
We next sought to confirm differential expression of selected target genes. We examined the spatial expression of specific candidate genes in bisected embryos following four hours of Ngn3-GR activation at two developmental time points, one immediately after activation for four hours at stage 15 and the other at stage 20. At stage 15, we confirmed increased expression of tbx2, mtg8, hes3 and geminin in the anterior ventral endoderm (Fig. 8A–H dashed circles and arrows). Geminin was chosen as a candidate representative of genes that were found increased in the microarray, but below 1.5 fold. For oct25 we detected increased expression in the dorsal endoderm of the roof of the archenteron (Fig. 8I,J dashed line). We also confirmed increased expression of the second MTG family member, Mtgr1 at stage 20 (Fig. 8K,L dashed circles).
Fig. 8. Validation of microarray data.
Following 4 hours of Ngn3-GR activation at stage 12 candidate genes from the microarray analysis were increased in the endoderm at stage 15: (A,B) tbx2 (n=24/24). (C,D) mtg8 (n=21/21). (E,F) hes3 (n=14/42). (G,H) geminin (n=13/44). (I,J) oct25 (n=13/13). And at stage 20: (K,L) mtgr1 (n=9/13). Dashed circles and arrows indicate ectopic expression in the anterior endoderm. Dashed line indicates ectopic expression in the dorsal endoderm.
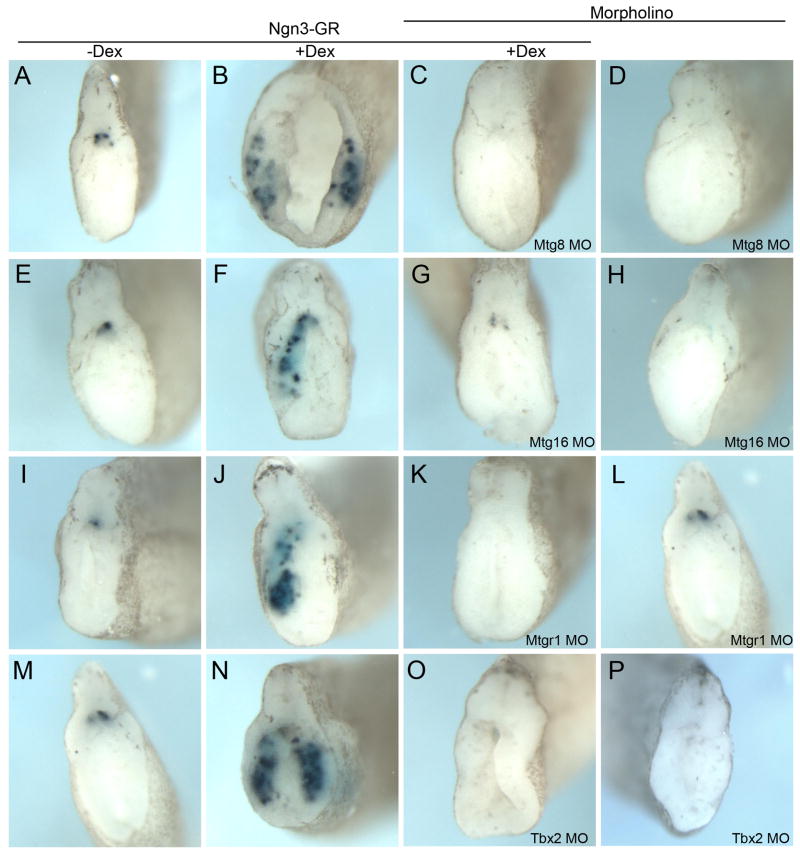
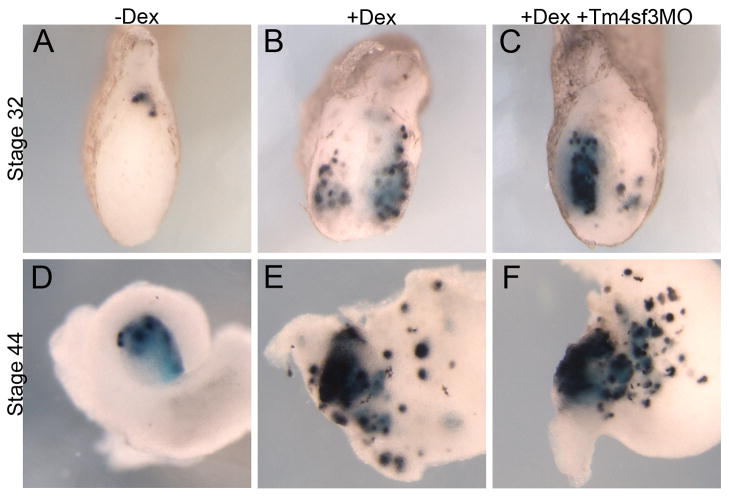
We then chose four of these genes (Mtg8, Mtg16, Mtgr1 and Tbx2) and examined whether they do indeed function downstream of Ngn3 in the promotion of ectopic beta cells using antisense morpholinos. Knockdown of Tbx2, Mtg8, Mtgr1 or Mtg16 in the presence of Ngn3-GR completely abolished promotion of ectopic beta cells by Ngn3 at stage 32 (Fig. 9C,G,K,O). Further, injection of Tbx2, Mtg8 or Mtg16 morpholinos alone significantly reduced endogenous insulin expression at stage 32, whereas the Mtgr1 morpholino alone had no effect (Fig. 9D,H,L,P). Analysis at the later stage 44 showed similar results (data not shown). To ensure that the results with these morpholinos were specific and not found with unrelated morpholinos we examined whether knockdown of Tm4sf3, which we previously showed affected pancreatic bud fusion (Jarikji et al., 2009), blocked Ngn3 promotion of ectopic beta cell development. We found that the use of the antisense Tm4sf3 morpholino did not affect Ngn3-GR promotion of ectopic beta cells (Fig. 10). These data demonstrate that all four candidates are required for the ectopic promotion of beta cells by Ngn3, whereas only Tbx2, Mtg8 and Mtg16 are required for endogenous beta cell development.
Fig. 9. Tbx2, Mtg8, Mtg16 and Mtgr1 function is required for ectopic and endogenous beta cell development at stage 32.
Tbx2 or Mtg morpholinos (40 ng each) were injected alone or with Ngn3-GR mRNA (1800 pg) and activated with Dex for four hours at stage 12 and the embryos grown to stage 32 and analyzed for insulin expression. (A–D) Injection of Mtg8 morpholino blocked ectopic insulin expression by Ngn3-GR (n=20/27). Mtg8 morpholino alone inhibited endogenous insulin expression (n=40/77). (E–H) Mtg16 morpholino blocked ectopic insulin expression by Ngn3-GR (n=16/20). Mtg16 morpholino alone inhibited endogenous insulin expression (n=27/52). (I–L) Mtgr1 morpholino blocked ectopic insulin expression by Ngn3-GR (n=28/28). Mtgr1 morpholino alone did not inhibit endogenous insulin expression (n=46/59). (M–P) Tbx2 morpholino blocked ectopic insulin expression by Ngn3-GR (n=27/27). Tbx2 morpholino alone inhibited endogenous insulin expression (n=20/37).
Fig. 10. Knockdown of Tm4sf3 does not affect Ngn3 promotion of ectopic insulin expression.
Ngn3-GR was injected alone or in combination with Tm4sf3 morpholino. Insulin expression at stage 32 in (A) control tadpoles; (B) +dex treated tadpoles and (C) in tadpoles injected with Tm4sf3 morpholino. (D–F) Insulin expression at stage 44.
Discussion
Our study demonstrates that transient misexpression of Ngn3 alone shortly after gastrulation can direct differentiation of endoderm towards the beta cell lineage, and suggests that Ngn3 control of specific endocrine lineages is dependent on cell competence. By activating Ngn3 for a few hours at different times we were able to pinpoint specific temporal windows for the ectopic creation of beta cells over alpha cells. The observation that activation at stage 12 for four hours promoted insulin and somatostatin expression while activation at stage 15 for four hours promoted expression of insulin, somatostatin and glucagon argues against an Ngn3 dosage effect in the promotion of each endocrine lineage. In addition, we show for the first time that Ngn3 misexpression is able to promote ectopic formation of beta cells in the stomach and duodenum. However, we cannot rule out the possibility that these cells were generated from pancreatic progenitors, which then migrated throughout the endoderm. Taken together, our results suggest that the spatio-temporal context is crucial for directed and ectopic differentiation of beta cells by Ngn3 and possibly for other transcription factors throughout development. Our results agree with previous data that had identified different competency windows for Ngn3 function in mice (Johansson et al., 2007). However, in that paper temporal windows were evaluated in days rather than hours as we show here, and their results mirrored the endogenous temporal expression profiles for endocrine hormones. We are the first to demonstrate that transient Ngn3 can be used to directly promote ectopic development specifically of beta and delta cells over alpha cells, and earlier than endogenous expression.
The ability to tightly control Ngn3 activity allowed us to isolate immediately early targets of Ngn3 and has provided a specific list of genes that function in the beta cell lineage. In particular, we have for the first time implicated Mtg8 and Mtg16 in normal pancreas development. Although Mtg8 was found to be essential for development of enteroendocrine cells, whether it was involved in pancreas development was not reported (Calabi et al., 2001). Previous studies have defined a relationship between Mtg proteins and Ngn1 and Ngn2 proteins in Xenopus ectoderm and in the chick developing neuronal system whereby Mtg proteins are downstream of Ngn proteins, and act as negative feedback regulators of Ngn function (Aaker et al., 2009; Aaker et al., 2010; Cao et al., 2002). In relation to pancreas development this would suggest that the pathway to beta cells requires short activation by Ngn3 and subsequently its inhibition by Mtg proteins. If so, then this would explain why previous studies promoted only alpha cell development upon Ngn3 overexpression, since they maintained continuous activation of Ngn3, whereas we blocked Ngn3 function shortly after its activation.
Alternatively, it is possible that the interaction between Ngn3 and Mtg proteins in beta cell development is not simply to block Ngn3 function, but to promote repression of genes required for other endocrine lineages. In agreement with this, a recent study showed that beta cell identity is maintained by DNA methylation-mediated repression of Arx by histone modifying enzymes (Dhawan et al., 2011). Since the Mtg proteins have been shown to interact with histone modifying enzymes and transcriptional corepressors orchestrating protein-protein interactions that might cause epigenetic changes for gene downregulation and silencing (Rossetti et al., 2004), it is possible that Ngn3 both activates transcription of Mtg genes and interacts with the Mtg proteins to form complexes that repress key genes like Arx to promote a beta cell fate over other endocrine cell fates.
Although Tbx genes are generally known for their role in mesoderm and ectoderm development, previous studies have shown that Tbx3, which is thought to act redundantly with Tbx2, acts as a transcriptional repressor and plays a role in liver progenitor proliferation and cell-fate determination (Ludtke et al., 2009; Suzuki et al., 2008). Given the proposed model of a common pancreas and liver progenitor, the involvement of Tbx2 in early pancreas development would not be unexpected. Similar to Tbx3 in liver development, Tbx2 could work downstream of Ngn3 acting to inhibit expression and/or activity of Arx and promoting proliferation of beta cell progenitors. It will be interesting to see if any key pancreatic genes are silenced following overexpression of Tbx2 and Mtg genes.
In comparison to previous Ngn3 microarray studies, we found remarkably little overlap with our gene list (Gasa et al., 2004; Gasa et al., 2008; Petri et al., 2006; Serafimidis et al., 2008; Treff et al., 2006; White et al., 2008). Of the 85 genes listed in Table 1, we found only 8 in common with other lists: Insm1, NeuroD4, Irf6, Znf38, Hes5, Stmn3, Hey1 and Mxi1. It is at present unclear why there was so little overlap. However, there were two main aspects of our study that were different from previous studies. First, we targeted naïve endoderm in vivo at an early stage, while previous studies targeted pancreatic tissue in vivo after it had been specified, pancreatic cells in vitro or in ES cells in vitro that had been artificially pushed down the endoderm lineage. Second, we performed our analysis at a much earlier stage. We isolated and compared endoderm tissue 4 hours after Ngn3 activation, whereas the earliest time point previously examined was 12 hours. Because of these differences we chose to begin our analysis of targets on those genes not previously identified. Our results with Mtg8, Mtg16, MtgR1 and Tbx2 are the first to demonstrate a role for these genes in pancreas development.
In summary, transient misexpression of Ngn3 in early Xenopus endoderm was sufficient to promote ectopic and early development of pancreatic beta cells. The use of Xenopus to define the transcriptional network of genes involved in the ectopic creation of beta cells will allow for future discovery and elucidation of transcriptional pathways that promote direct beta cell lineage development.
METHODS
Antisense morpholino and mRNA injections
Antisense morpholino oligonucleotides were designed and produced by Gene Tools LLC to the translation start. Antisense morpholino sequences are tbx2 5′-GGTGCATACACAAATCCAGCAGGGA-3′, mtg8 5′-GGTGCTTGAGATTCCCACCATTCGC-3′, mtgr1 5′-GAT AGGGTCCTGGAATCCCAACCAT-3 ′ and mtg16 5′-TGAGTCTGGCATTGTAAACGCTGTA-3′. For mRNA injections, all mRNA was synthesized in vitro (Ambion mMessage) and coinjected with 400pg gfp mRNA to verify targeting by observing fluorescence in whole embryos or dissected whole guts. For both mRNA and morpholinos we injected the two dorsal vegetal blastomeres at the eight cell stage, thus targeting the anterior endoderm in most instances. To activate Ngn3-GR protein, embryos were cultured in dexamethasone in a final concentration of 10μM in 0.1X MMR.
Microarray Analysis
Embryos injected with Ngn3-GR mRNA were incubated in dexamethasone for four hours at stage 12, and then all dorsal structures were removed, leaving only the ventral portion of the embryo. Sample sets (4 sets, 10 embryos/set) were kept in RNAlater (Ambion) until RNA extraction with TRIzol (Invitrogen) followed by RNeasy Kit (Qiagen). RNA quality analysis, cDNA synthesis and hybridization to the Affymetrix GeneChip Xenopus laevis Genome 2.0 Array were performed by Genome Quebec. Microarray results were analyzed using the expression analysis software FlexArray V1.4.1 (Genome Quebec, Montreal, Canada, URL http://genomequebec.mcgill.ca/FlexArray). Data were normalized with Robust Multi-Array Average (RMA) and the algorithm EB (Wright and Simon) was used for statistical analysis. Microarray data has been deposited in NCBI’s Gene Expression Omnibus under accession number GSE29017. All data entries for transcribed locus have been removed from Table 1.
Nucleic Acids
The full-length Xenopus laevis ngn3 ORF (NM_001134785) was cloned from wild type stage 42 cDNA into the pCS2+ vector. The ORF was then subcloned into a pCS2+ vector containing the human glucocorticoid receptor ligand-binding domain (GR) with primers SP6 5′-GATTTAGGTGACACTATAG-3′ and Ngn3GrRevClaI 5′-AAGCTATAGGCAT CGATACAAGAACTCTA-3′. The GR was fused to the C-terminus of the Xenopus Ngn3 ORF and referred to as Ngn3-GR. GenBank Accession Numbers for mtg8, mtgr1, tbx2, geminin, hes3.1 and oct25 are NM_001095596, NM_001086057, NM_001086520, NM_001088403, NM_001088503 and NM_001093992, respectively. Whole mount in situ hybridizations were performed as described (Horb et al., 2003).
Acknowledgments
We are grateful to Zeina Jarikji for her valuable assistance in the microinjections and in situ hybridizations and to Frédéric Bourque for his care of the frogs.
Grant: National Institutes of Health (DK077197)
References
- Aaker JD, Patineau AL, Yang HJ, Ewart DT, Gong W, Li T, Nakagawa Y, McLoon SC, Koyano-Nakagawa N. Feedback regulation of NEUROG2 activity by MTGR1 is required for progression of neurogenesis. Mol Cell Neurosci. 2009;42:267–277. doi: 10.1016/j.mcn.2009.07.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aaker JD, Patineau AL, Yang HJ, Ewart DT, Nakagawa Y, McLoon SC, Koyano-Nakagawa N. Interaction of MTG family proteins with NEUROG2 and ASCL1 in the developing nervous system. Neurosci Lett. 2010;474:46–51. doi: 10.1016/j.neulet.2010.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Afelik S, Chen Y, Pieler T. Combined ectopic expression of Pdx1 and Ptf1a/p48 results in the stable conversion of posterior endoderm into endocrine and exocrine pancreatic tissue. Genes Dev. 2006;20:1441–1446. doi: 10.1101/gad.378706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amann JM, Chyla BJ, Ellis TC, Martinez A, Moore AC, Franklin JL, McGhee L, Meyers S, Ohm JE, Luce KS, Ouelette AJ, Washington MK, Thompson MA, King D, Gautam S, Coffey RJ, Whitehead RH, Hiebert SW. Mtgr1 is a transcriptional corepressor that is required for maintenance of the secretory cell lineage in the small intestine. Mol Cell Biol. 2005;25:9576–9585. doi: 10.1128/MCB.25.21.9576-9585.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Apelqvist A, Li H, Sommer L, Beatus P, Anderson DJ, Honjo T, Hrabe dA, Lendahl U, Edlund H. Notch signalling controls pancreatic cell differentiation. Nature. 1999;400:877–881. doi: 10.1038/23716. [DOI] [PubMed] [Google Scholar]
- Bennett K, James C, Hussain K. Pancreatic beta-cell KATP channels: Hypoglycaemia and hyperglycaemia. Rev Endocr Metab Disord. 2010;11:157–163. doi: 10.1007/s11154-010-9144-2. [DOI] [PubMed] [Google Scholar]
- Borowiak M, Melton DA. How to make beta cells? Curr Opin Cell Biol. 2009;21:727–732. doi: 10.1016/j.ceb.2009.08.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calabi F, Pannell R, Pavloska G. Gene targeting reveals a crucial role for MTG8 in the gut. Mol Cell Biol. 2001;21:5658–5666. doi: 10.1128/MCB.21.16.5658-5666.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cao Y, Zhao H, Grunz H. XETOR regulates the size of the proneural domain during primary neurogenesis in Xenopus laevis. Mech Dev. 2002;119:35–44. doi: 10.1016/s0925-4773(02)00285-x. [DOI] [PubMed] [Google Scholar]
- Chalmers AD, Slack JM. Development of the gut in Xenopus laevis. Dev Dyn. 1998;212:509–521. doi: 10.1002/(SICI)1097-0177(199808)212:4<509::AID-AJA4>3.0.CO;2-L. [DOI] [PubMed] [Google Scholar]
- Chalmers AD, Slack JM. The Xenopus tadpole gut: fate maps and morphogenetic movements. Development. 2000;127:381–392. doi: 10.1242/dev.127.2.381. [DOI] [PubMed] [Google Scholar]
- Collombat P, Mansouri A, Hecksher-Sorensen J, Serup P, Krull J, Gradwohl G, Gruss P. Opposing actions of Arx and Pax4 in endocrine pancreas development. Genes Dev. 2003;17:2591–2603. doi: 10.1101/gad.269003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collombat P, Xu X, Ravassard P, Sosa-Pineda B, Dussaud S, Billestrup N, Madsen OD, Serup P, Heimberg H, Mansouri A. The ectopic expression of Pax4 in the mouse pancreas converts progenitor cells into alpha and subsequently beta cells. Cell. 2009;138:449–462. doi: 10.1016/j.cell.2009.05.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dhawan S, Georgia S, Tschen SI, Fan G, Bhushan A. Pancreatic beta Cell Identity Is Maintained by DNA Methylation-Mediated Repression of Arx. Dev Cell. 2011;20:419–429. doi: 10.1016/j.devcel.2011.03.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gasa R, Mrejen C, Leachman N, Otten M, Barnes M, Wang J, Chakrabarti S, Mirmira R, German M. Proendocrine genes coordinate the pancreatic islet differentiation program in vitro. Proc Natl Acad Sci U S A. 2004;101:13245–13250. doi: 10.1073/pnas.0405301101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gasa R, Mrejen C, Lynn FC, Skewes-Cox P, Sanchez L, Yang KY, Lin CH, Gomis R, German MS. Induction of pancreatic islet cell differentiation by the neurogenin-neuroD cascade. Differentiation. 2008;76:381–391. doi: 10.1111/j.1432-0436.2007.00228.x. [DOI] [PubMed] [Google Scholar]
- Gierl MS, Karoulias N, Wende H, Strehle M, Birchmeier C. The zinc-finger factor Insm1 (IA-1) is essential for the development of pancreatic beta cells and intestinal endocrine cells. Genes Dev. 2006;20:2465–2478. doi: 10.1101/gad.381806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gittes GK. Developmental biology of the pancreas: a comprehensive review. Dev Biol. 2009;326:4–35. doi: 10.1016/j.ydbio.2008.10.024. [DOI] [PubMed] [Google Scholar]
- Grapin-Botton A, Majithia AR, Melton DA. Key events of pancreas formation are triggered in gut endoderm by ectopic expression of pancreatic regulatory genes. Genes & Development. 2001;15:444–454. doi: 10.1101/gad.846001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horb LD, Horb ME. BrunoL1 regulates endoderm proliferation through translational enhancement of cyclin A2 mRNA. Dev Biol. 2010;345:156–169. doi: 10.1016/j.ydbio.2010.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horb LD, Jarkji ZH, Horb ME. Xenopus Insm1 is essential for gastrointestinal and pancreatic endocrine cell development. Dev Dyn. 2009;238:2505–2510. doi: 10.1002/dvdy.22071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horb ME, Shen CN, Tosh D, Slack JM. Experimental conversion of liver to pancreas. Curr Biol. 2003;13:105–115. doi: 10.1016/s0960-9822(02)01434-3. [DOI] [PubMed] [Google Scholar]
- Horb ME, Slack JM. Endoderm specification and differentiation in Xenopus embryos. Dev Biol. 2001;236:330–343. doi: 10.1006/dbio.2001.0347. [DOI] [PubMed] [Google Scholar]
- Horb ME, Slack JM. Expression of amylase and other pancreatic genes in Xenopus. Mech Dev. 2002;113:153–157. doi: 10.1016/s0925-4773(02)00019-9. [DOI] [PubMed] [Google Scholar]
- Jarikji Z, Horb LD, Shariff F, Mandato CA, Cho KW, Horb ME. The tetraspanin Tm4sf3 is localized to the ventral pancreas and regulates fusion of the dorsal and ventral pancreatic buds. Development. 2009;136:1791–1800. doi: 10.1242/dev.032235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jarikji ZH, Vanamala S, Beck CW, Wright CV, Leach SD, Horb ME. Differential ability of Ptf1a and Ptf1a-VP16 to convert stomach, duodenum and liver to pancreas. Dev Biol. 2007;304:786–799. doi: 10.1016/j.ydbio.2007.01.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johansson KA, Dursun U, Jordan N, Gu G, Beermann F, Gradwohl G, Grapin-Botton A. Temporal control of neurogenin3 activity in pancreas progenitors reveals competence windows for the generation of different endocrine cell types. Dev Cell. 2007;12:457–465. doi: 10.1016/j.devcel.2007.02.010. [DOI] [PubMed] [Google Scholar]
- Kelly OG, Melton DA. Development of the pancreas in Xenopus laevis. Dev Dyn. 2000;218:615–627. doi: 10.1002/1097-0177(2000)9999:9999<::AID-DVDY1027>3.0.CO;2-8. [DOI] [PubMed] [Google Scholar]
- Kordowich S, Mansouri A, Collombat P. Reprogramming into pancreatic endocrine cells based on developmental cues. Mol Cell Endocrinol. 2010;323:62–69. doi: 10.1016/j.mce.2009.12.016. [DOI] [PubMed] [Google Scholar]
- Koyano-Nakagawa N, Kintner C. The expression and function of MTG/ETO family proteins during neurogenesis. Dev Biol. 2005;278:22–34. doi: 10.1016/j.ydbio.2004.10.010. [DOI] [PubMed] [Google Scholar]
- Ludtke TH, Christoffels VM, Petry M, Kispert A. Tbx3 promotes liver bud expansion during mouse development by suppression of cholangiocyte differentiation. Hepatology. 2009;49:969–978. doi: 10.1002/hep.22700. [DOI] [PubMed] [Google Scholar]
- Mellitzer G, Bonne S, Luco RF, Van De CM, Lenne-Samuel N, Collombat P, Mansouri A, Lee J, Lan M, Pipeleers D, Nielsen FC, Ferrer J, Gradwohl G, Heimberg H. IA1 is NGN3-dependent and essential for differentiation of the endocrine pancreas. EMBO J. 2006;25:1344–1352. doi: 10.1038/sj.emboj.7601011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murtaugh LC. Pancreas and beta-cell development: from the actual to the possible. Development. 2007;134:427–438. doi: 10.1242/dev.02770. [DOI] [PubMed] [Google Scholar]
- Pearl EJ, Bilogan CK, Mukhi S, Brown DD, Horb ME. Xenopus pancreas development. Dev Dyn. 2009;238:1271–1286. doi: 10.1002/dvdy.21935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pearl EJ, Horb ME. Promoting ectopic pancreatic fates: pancreas development and future diabetes therapies. Clin Genet. 2008;74:316–324. doi: 10.1111/j.1399-0004.2008.01081.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pearl EJ, Jarikji Z, Horb ME. Functional analysis of Rfx6 and mutant variants associated with neonatal diabetes. Dev Biol. 2011;351:135–145. doi: 10.1016/j.ydbio.2010.12.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petri A, Ahnfelt-Ronne J, Frederiksen KS, Edwards DG, Madsen D, Serup P, Fleckner J, Heller RS. The effect of neurogenin3 deficiency on pancreatic gene expression in embryonic mice. J Mol Endocrinol. 2006;37:301–316. doi: 10.1677/jme.1.02096. [DOI] [PubMed] [Google Scholar]
- Rossetti S, Hoogeveen AT, Sacchi N. The MTG proteins: chromatin repression players with a passion for networking. Genomics. 2004;84:1–9. doi: 10.1016/j.ygeno.2004.02.011. [DOI] [PubMed] [Google Scholar]
- Rukstalis JM, Habener JF. Neurogenin3: a master regulator of pancreatic islet differentiation and regeneration. Islets. 2009;1:177–184. doi: 10.4161/isl.1.3.9877. [DOI] [PubMed] [Google Scholar]
- Schwitzgebel VM, Scheel DW, Conners JR, Kalamaras J, Lee JE, Anderson DJ, Sussel L, Johnson JD, German MS. Expression of neurogenin3 reveals an islet cell precursor population in the pancreas. Development. 2000;127:3533–3542. doi: 10.1242/dev.127.16.3533. [DOI] [PubMed] [Google Scholar]
- Serafimidis I, Rakatzi I, Episkopou V, Gouti M, Gavalas A. Novel effectors of directed and Ngn3-mediated differentiation of mouse embryonic stem cells into endocrine pancreas progenitors. Stem Cells. 2008;26:3–16. doi: 10.1634/stemcells.2007-0194. [DOI] [PubMed] [Google Scholar]
- Smith SB, Qu HQ, Taleb N, Kishimoto NY, Scheel DW, Lu Y, Patch AM, Grabs R, Wang J, Lynn FC, Miyatsuka T, Mitchell J, Seerke R, Desir J, Eijnden SV, Abramowicz M, Kacet N, Weill J, Renard ME, Gentile M, Hansen I, Dewar K, Hattersley AT, Wang R, Wilson ME, Johnson JD, Polychronakos C, German MS. Rfx6 directs islet formation and insulin production in mice and humans. Nature. 2010;463:775–780. doi: 10.1038/nature08748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sosa-Pineda B, Chowdhury K, Torres M, Oliver G, Gruss P. The Pax4 gene is essential for differentiation of insulin-producing beta cells in the mammalian pancreas. Nature. 1997;386:399–402. doi: 10.1038/386399a0. [DOI] [PubMed] [Google Scholar]
- Soyer J, Flasse L, Raffelsberger W, Beucher A, Orvain C, Peers B, Ravassard P, Vermot J, Voz ML, Mellitzer G, Gradwohl G. Rfx6 is an Ngn3-dependent winged helix transcription factor required for pancreatic islet cell development. Development. 2010;137:203–212. doi: 10.1242/dev.041673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suzuki A, Sekiya S, Buscher D, Izpisua Belmonte JC, Taniguchi H. Tbx3 controls the fate of hepatic progenitor cells in liver development by suppressing p19ARF expression. Development. 2008;135:1589–1595. doi: 10.1242/dev.016634. [DOI] [PubMed] [Google Scholar]
- Tiso N, Moro E, Argenton F. Zebrafish pancreas development. Mol Cell Endocrinol. 2009;312:24–30. doi: 10.1016/j.mce.2009.04.018. [DOI] [PubMed] [Google Scholar]
- Treff NR, Vincent RK, Budde ML, Browning VL, Magliocca JF, Kapur V, Odorico JS. Differentiation of embryonic stem cells conditionally expressing neurogenin 3. Stem Cells. 2006;24:2529–2537. doi: 10.1634/stemcells.2006-0082. [DOI] [PubMed] [Google Scholar]
- White P, May CL, Lamounier RN, Brestelli JE, Kaestner KH. Defining pancreatic endocrine precursors and their descendants. Diabetes. 2008;57:654–668. doi: 10.2337/db07-1362. [DOI] [PubMed] [Google Scholar]
- Yechoor V, Chan L. Minireview: beta-cell replacement therapy for diabetes in the 21st century: manipulation of cell fate by directed differentiation. Mol Endocrinol. 2010;24:1501–1511. doi: 10.1210/me.2009-0311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou Q, Brown J, Kanarek A, Rajagopal J, Melton DA. In vivo reprogramming of adult pancreatic exocrine cells to beta-cells. Nature. 2008;455:627–632. doi: 10.1038/nature07314. [DOI] [PMC free article] [PubMed] [Google Scholar]