Abstract
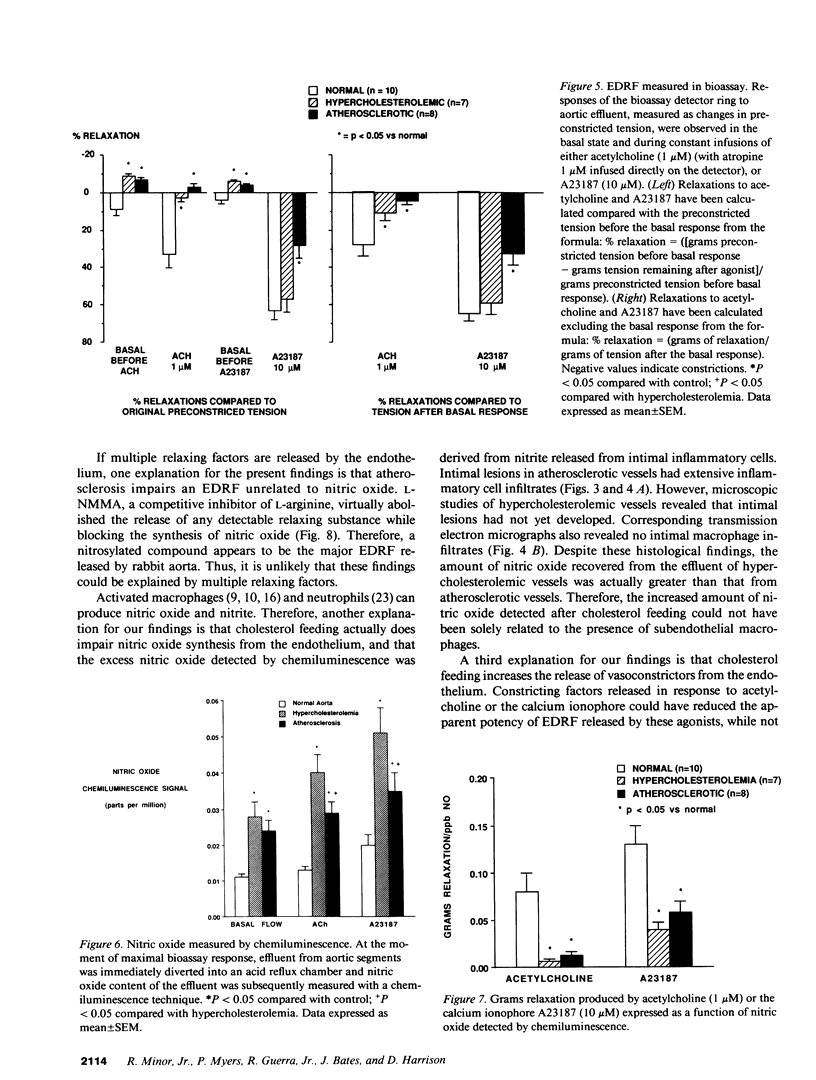
We examined the hypothesis that impaired endothelium-dependent vasodilation in atherosclerosis is associated with decreased synthesis of nitrogen oxides by the vascular endothelium. The descending thoracic aortae of rabbits fed either normal diet, a high cholesterol diet for 2-5 wk (hypercholesterolemic, HC), or a high cholesterol diet for 6 mo (atherosclerotic, AS) were perfused in a bioassay organ chamber with physiologic buffer containing indomethacin. Despite a dramatic impairment in the vasodilator activity of endothelium-dependent relaxing factor (EDRF) released from both HC and AS aortae (assessed by bioassay), the release of nitrogen oxides (measured by chemiluminescence) from these vessels was not reduced, but markedly increased compared to NL. Thus, impaired endothelium-dependent relaxation in atherosclerosis is neither due to decreased activity of the enzyme responsible for the production of nitrogen oxides from arginine nor to arginine deficiency. Because the production of nitrogen oxides increased in response to acetylcholine in both hypercholesterolemic and atherosclerotic vessels, impairments in signal transduction are not responsible for abnormal endothelium-dependent relaxations. Impaired vasodilator activity of EDRF by cholesterol feeding may result from loss of incorporation of nitric oxide into a more potent parent compound, or accelerated degradation of EDRF.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bossaller C., Habib G. B., Yamamoto H., Williams C., Wells S., Henry P. D. Impaired muscarinic endothelium-dependent relaxation and cyclic guanosine 5'-monophosphate formation in atherosclerotic human coronary artery and rabbit aorta. J Clin Invest. 1987 Jan;79(1):170–174. doi: 10.1172/JCI112779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bredt D. S., Snyder S. H. Isolation of nitric oxide synthetase, a calmodulin-requiring enzyme. Proc Natl Acad Sci U S A. 1990 Jan;87(2):682–685. doi: 10.1073/pnas.87.2.682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chappell S. P., Lewis M. J., Henderson A. H. Effect of lipid feeding on endothelium dependent relaxation in rabbit aortic preparations. Cardiovasc Res. 1987 Jan;21(1):34–38. doi: 10.1093/cvr/21.1.34. [DOI] [PubMed] [Google Scholar]
- Curran R. D., Billiar T. R., Stuehr D. J., Hofmann K., Simmons R. L. Hepatocytes produce nitrogen oxides from L-arginine in response to inflammatory products of Kupffer cells. J Exp Med. 1989 Nov 1;170(5):1769–1774. doi: 10.1084/jem.170.5.1769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freiman P. C., Mitchell G. G., Heistad D. D., Armstrong M. L., Harrison D. G. Atherosclerosis impairs endothelium-dependent vascular relaxation to acetylcholine and thrombin in primates. Circ Res. 1986 Jun;58(6):783–789. doi: 10.1161/01.res.58.6.783. [DOI] [PubMed] [Google Scholar]
- Förstermann U., Mügge A., Alheid U., Haverich A., Frölich J. C. Selective attenuation of endothelium-mediated vasodilation in atherosclerotic human coronary arteries. Circ Res. 1988 Feb;62(2):185–190. doi: 10.1161/01.res.62.2.185. [DOI] [PubMed] [Google Scholar]
- Garthwaite J., Southam E., Anderton M. A kainate receptor linked to nitric oxide synthesis from arginine. J Neurochem. 1989 Dec;53(6):1952–1954. doi: 10.1111/j.1471-4159.1989.tb09266.x. [DOI] [PubMed] [Google Scholar]
- Gryglewski R. J., Palmer R. M., Moncada S. Superoxide anion is involved in the breakdown of endothelium-derived vascular relaxing factor. Nature. 1986 Apr 3;320(6061):454–456. doi: 10.1038/320454a0. [DOI] [PubMed] [Google Scholar]
- Guerra R., Jr, Brotherton A. F., Goodwin P. J., Clark C. R., Armstrong M. L., Harrison D. G. Mechanisms of abnormal endothelium-dependent vascular relaxation in atherosclerosis: implications for altered autocrine and paracrine functions of EDRF. Blood Vessels. 1989;26(5):300–314. doi: 10.1159/000158779. [DOI] [PubMed] [Google Scholar]
- Harrison D. G., Armstrong M. L., Freiman P. C., Heistad D. D. Restoration of endothelium-dependent relaxation by dietary treatment of atherosclerosis. J Clin Invest. 1987 Dec;80(6):1808–1811. doi: 10.1172/JCI113276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hibbs J. B., Jr, Taintor R. R., Vavrin Z. Macrophage cytotoxicity: role for L-arginine deiminase and imino nitrogen oxidation to nitrite. Science. 1987 Jan 23;235(4787):473–476. doi: 10.1126/science.2432665. [DOI] [PubMed] [Google Scholar]
- Ignarro L. J. Biological actions and properties of endothelium-derived nitric oxide formed and released from artery and vein. Circ Res. 1989 Jul;65(1):1–21. doi: 10.1161/01.res.65.1.1. [DOI] [PubMed] [Google Scholar]
- Jayakody R. L., Senaratne M. P., Thomson A. B., Kappagoda C. T. Cholesterol feeding impairs endothelium-dependent relaxation of rabbit aorta. Can J Physiol Pharmacol. 1985 Sep;63(9):1206–1209. doi: 10.1139/y85-199. [DOI] [PubMed] [Google Scholar]
- Marletta M. A., Yoon P. S., Iyengar R., Leaf C. D., Wishnok J. S. Macrophage oxidation of L-arginine to nitrite and nitrate: nitric oxide is an intermediate. Biochemistry. 1988 Nov 29;27(24):8706–8711. doi: 10.1021/bi00424a003. [DOI] [PubMed] [Google Scholar]
- Mayer B., Böhme E. Ca2+-dependent formation of an L-arginine-derived activator of soluble guanylyl cyclase in bovine lung. FEBS Lett. 1989 Oct 9;256(1-2):211–214. doi: 10.1016/0014-5793(89)81750-8. [DOI] [PubMed] [Google Scholar]
- Mayer B., Schmidt K., Humbert P., Böhme E. Biosynthesis of endothelium-derived relaxing factor: a cytosolic enzyme in porcine aortic endothelial cells Ca2+-dependently converts L-arginine into an activator of soluble guanylyl cyclase. Biochem Biophys Res Commun. 1989 Oct 31;164(2):678–685. doi: 10.1016/0006-291x(89)91513-1. [DOI] [PubMed] [Google Scholar]
- Myers P. R., Guerra R., Jr, Harrison D. G. Release of NO and EDRF from cultured bovine aortic endothelial cells. Am J Physiol. 1989 Apr;256(4 Pt 2):H1030–H1037. doi: 10.1152/ajpheart.1989.256.4.H1030. [DOI] [PubMed] [Google Scholar]
- Myers P. R., Minor R. L., Jr, Guerra R., Jr, Bates J. N., Harrison D. G. Vasorelaxant properties of the endothelium-derived relaxing factor more closely resemble S-nitrosocysteine than nitric oxide. Nature. 1990 May 10;345(6271):161–163. doi: 10.1038/345161a0. [DOI] [PubMed] [Google Scholar]
- Palmer R. M., Ashton D. S., Moncada S. Vascular endothelial cells synthesize nitric oxide from L-arginine. Nature. 1988 Jun 16;333(6174):664–666. doi: 10.1038/333664a0. [DOI] [PubMed] [Google Scholar]
- Palmer R. M., Ferrige A. G., Moncada S. Nitric oxide release accounts for the biological activity of endothelium-derived relaxing factor. Nature. 1987 Jun 11;327(6122):524–526. doi: 10.1038/327524a0. [DOI] [PubMed] [Google Scholar]
- Palmer R. M., Rees D. D., Ashton D. S., Moncada S. L-arginine is the physiological precursor for the formation of nitric oxide in endothelium-dependent relaxation. Biochem Biophys Res Commun. 1988 Jun 30;153(3):1251–1256. doi: 10.1016/s0006-291x(88)81362-7. [DOI] [PubMed] [Google Scholar]
- Rimele T. J., Sturm R. J., Adams L. M., Henry D. E., Heaslip R. J., Weichman B. M., Grimes D. Interaction of neutrophils with vascular smooth muscle: identification of a neutrophil-derived relaxing factor. J Pharmacol Exp Ther. 1988 Apr;245(1):102–111. [PubMed] [Google Scholar]
- Rubanyi G. M., Vanhoutte P. M. Superoxide anions and hyperoxia inactivate endothelium-derived relaxing factor. Am J Physiol. 1986 May;250(5 Pt 2):H822–H827. doi: 10.1152/ajpheart.1986.250.5.H822. [DOI] [PubMed] [Google Scholar]
- Schmidt H. H., Wilke P., Evers B., Böhme E. Enzymatic formation of nitrogen oxides from L-arginine in bovine brain cytosol. Biochem Biophys Res Commun. 1989 Nov 30;165(1):284–291. doi: 10.1016/0006-291x(89)91067-x. [DOI] [PubMed] [Google Scholar]
- Shirahase H., Usui H., Kurahashi K., Fujiwara M., Fukui K. Possible role of endothelial thromboxane A2 in the resting tone and contractile responses to acetylcholine and arachidonic acid in canine cerebral arteries. J Cardiovasc Pharmacol. 1987 Nov;10(5):517–522. doi: 10.1097/00005344-198711000-00004. [DOI] [PubMed] [Google Scholar]
- Sreeharan N., Jayakody R. L., Senaratne M. P., Thomson A. B., Kappagoda C. T. Endothelium-dependent relaxation and experimental atherosclerosis in the rabbit aorta. Can J Physiol Pharmacol. 1986 Nov;64(11):1451–1453. doi: 10.1139/y86-246. [DOI] [PubMed] [Google Scholar]
- Tayeh M. A., Marletta M. A. Macrophage oxidation of L-arginine to nitric oxide, nitrite, and nitrate. Tetrahydrobiopterin is required as a cofactor. J Biol Chem. 1989 Nov 25;264(33):19654–19658. [PubMed] [Google Scholar]
- Verbeuren T. J., Jordaens F. H., Zonnekeyn L. L., Van Hove C. E., Coene M. C., Herman A. G. Effect of hypercholesterolemia on vascular reactivity in the rabbit. I. Endothelium-dependent and endothelium-independent contractions and relaxations in isolated arteries of control and hypercholesterolemic rabbits. Circ Res. 1986 Apr;58(4):552–564. doi: 10.1161/01.res.58.4.552. [DOI] [PubMed] [Google Scholar]
- Yanagisawa M., Kurihara H., Kimura S., Tomobe Y., Kobayashi M., Mitsui Y., Yazaki Y., Goto K., Masaki T. A novel potent vasoconstrictor peptide produced by vascular endothelial cells. Nature. 1988 Mar 31;332(6163):411–415. doi: 10.1038/332411a0. [DOI] [PubMed] [Google Scholar]