Abstract
Natural killer (NK) cells and CD8+ T cells play a prominent role in the clearance of mouse cytomegalovirus (MCMV) infection. The role of NK cells in modulating the CD8+ T-cell response to MCMV infection is still the subject of intensive research. For analyzing the impact of NK cells on mounting of a CD8+ T-cell response and the contribution of these cells to virus control during the first days postinfection (p.i.), we used C57BL/6 mice in which NK cells are specifically activated through the Ly49H receptor engaged by the MCMV-encoded ligand m157. Our results indicate that the requirement for CD8+ T cells in early MCMV control inversely correlates with the engagement of Ly49H. While depletion of CD8+ T cells has only a minor effect on the early control of wild-type MCMV, CD8+ T cells are essential in the control of Δm157 virus. The frequencies of virus epitope-specific CD8+ T cells and their activation status were higher in mice infected with Δm157 virus. In addition, these mice showed elevated levels of alpha interferon (IFN-α) and several other proinflammatory cytokines as early as 1.5 days p.i. Although the numbers of conventional dendritic cells (cDCs) were reduced later during infection, particularly in Δm157-infected mice, they were not significantly affected at the peak of the cytokine response. Altogether, we concluded that increased antigen load, preservation of early cDCs' function, and higher levels of innate cytokines collectively account for an enhanced CD8+ T-cell response in C57BL/6 mice infected with a virus unable to activate NK cells via the Ly49H–m157 interaction.
INTRODUCTION
Mouse cytomegalovirus (MCMV) has been extensively used as a model for studying the role of NK cells in virus control. NK cells play a crucial role in the early stage of MCMV infection, prior to the induction of the adaptive immune response (20). However, the contribution of NK cells in the control of early MCMV infection varies among mouse strains (38; reviewed in reference 43). In C57BL/6 mice, the activating NK cell receptor Ly49H mediates resistance to MCMV infection due to the specific binding of m157, a virally encoded protein (6, 46). NK cell activation through Ly49H–m157 interaction is characterized by perforin-mediated cytotoxicity and specific proliferation of Ly49H+ NK cells (13, 26, 49). Unlike C57BL/6 mice, MCMV-susceptible mouse strains are unable to mount an effective NK cell response to this virus (reviewed in reference 42).
In addition to their direct function, which results in the containment of viral infection, a large body of accumulated data suggests that NK cells play a role also in the shaping of the specific immune response (reviewed in reference 50). However, this topic is still controversial and the subject of intense studies. Robbins and colleagues (40) have shown that NK cell activation via the Ly49H–m157 pathway accelerates the CD8+ T-cell response in vivo. According to the proposed scenario, the activation of NK cells via this axis limits alpha/beta interferon (IFN-α/β) production by plasmacytoid dendritic cells (pDCs) and consequently prevents depletion of splenic conventional DCs (cDCs) and causes a prompt induction of a CD8+ T-cell response. However, Andrews et al. (3) have shown that an NK cell response mediated through Ly49H–m157 interaction limits the long-term efficacy of specific CD8+ and CD4+ T cells and that this is a consequence of an altered frequency and duration of infection of antigen-presenting cells. More recently, Stadnisky and colleagues (47) have demonstrated a role for inhibitory NK receptors in the CD8+ T-cell response to MCMV. Although these authors did not exclude a role for MCMV-specific activating NK receptors in their model, they provided first evidence to propose that the recognition of infected cells by licensed Ly49G2+ NK cells results in a faster recovery of splenic cDCs and an enhanced antigen-specific CD8+ T-cell response. In BALB/c mice, which do not express Ly49H and thus constitutively lack NK cell activation through Ly49H–m157 signaling, virus replication within the first 4 days postinfection (p.i.) was not notably controlled by either NK cells or CD8+ T cells, unless deletion of viral immune evasion genes, of gene m152 in particular, attenuated virus replication by recruiting NK cells and CD8+ T cells due to elevated expression of the NK cell-activating ligand RAE-1 and enhanced major histocompatibility complex class I (MHC-I) peptide presentation, respectively (7). In addition, we have recently shown that recombinant virus expressing activating NKG2D ligand RAE-1γ is able to activate a strong CD8+ T-cell response in spite of a significant NK cell- and NKG2D-dependent early attenuation (45).
In the present study, we have assessed the CD8+ T-cell response in C57BL/6 mice during the first few days of infection with wild-type (WT) MCMV able to engage the Ly49H receptor, or with Δm157 virus, which fails to activate NK cells via this receptor. In essence, our results have revealed that Ly49H–m157 signaling drives the NK cell response, whereas the inability of virus control via NK cells in the absence of Ly49H–m157 signaling results in a stronger CD8+ T-cell response. Specifically, the data demonstrate that NK cell engagement through the Ly49H–m157 interaction limits the contribution of CD8+ T cells to the control of MCMV. In contrast, in Ly49H+ mice infected with Δm157 MCMV, the CD8+ T-cell response proved to be not only enhanced but also indispensable for virus control. Notably, CD8+ T cells became essential even in the control of WT MCMV at high doses of infection. A more efficient CD8+ T-cell response is most likely supported by elevated levels of proinflammatory cytokines able to drive the expansion of antiviral CD8+ T cells. Altogether, our data suggest that an increased antigen load available for CD8+ T-cell priming in concert with the preservation of cDCs' function early after infection and with elevated levels of proinflammatory cytokines explains the enhanced CD8+ T-cell response in mice lacking early NK cell antiviral control.
MATERIALS AND METHODS
Mice.
Ly49H+ mice (C57BL/6, BALB.B6-Cmv1r) and Ly49H− mice (B6.Ly49h−/− [15], BALB/c, and 129/SvJ) were housed and bred under specific-pathogen-free conditions at the Central Animal Facility of the Medical Faculty, University of Rijeka, in accordance with the guidelines contained in the International Guiding Principles for Biomedical Research Involving Animals. The Ethical Committee at the University of Rijeka approved all animal experiments described in this paper. Eight- to 12-week-old mice were used in all experiments.
Viruses.
Unless otherwise indicated, mice were injected intravenously (i.v.) with 2 × 105 PFU of the tissue culture (TC)-grown virus in a volume of 500 μl of diluents. Bacterial artificial chromosome (BAC)-derived MCMV strain MW97.01 has previously been shown to be biologically equivalent to MCMV strain Smith (VR-1399) and is hereafter referred to as wild-type (WT) MCMV (51). MCMV recombinant virus expressing green fluorescent protein (MCMV-GFP), a derivative of the MCMV strain MW97.01, is hereafter referred to as WT MCMV-GFP (34). In addition, the mutant virus Δm157 MCMV, which was previously described (8), was used.
Depletion of lymphocyte subsets and quantitation of viral gene expression and infectivity.
The depletion of NK cells in vivo was performed by intraperitoneal (i.p.) injection of 300 μg of purified monoclonal antibody (MAb) PK136 (23) 1 day before infection, which was repeated on day 1 p.i. The depletion of the CD8+ T-lymphocyte subset was done by i.p. injection of 300 μg of MAb to CD8 (YTS 169.4) (10) on days 1 and 5 p.i. The efficacy of depletion was assessed by flow cytometric analysis of splenic leukocytes stained with allophycocyanin (APC)-labeled anti-NK1.1 (eBioscience) and phycoerythrin (PE)-labeled anti-CD8 (BD Pharmingen).
Viral transcription in draining popliteal lymph nodes (PLNs) was measured by absolute quantitation of spliced IE1 transcripts using a real-time one-step reverse transcription-PCR (RT-PCR) as described in greater detail previously (7, 44). For quantitating viral infectivity in organs, virus titers were determined by standard plaque assay, as described previously (21).
Flow cytometry and enzyme-linked immunospot (ELISpot) assay.
Splenic leukocytes were prepared as previously described and, in order to reduce nonspecific staining, Fc receptors were blocked with 2.4G2 MAbs (54). The following Abs were purchased from eBioscience or BD Pharmingen, and cell surface staining was performed specifically for the following antigens: anti-CD3ε (145-2C11), anti-NK1.1 (PK136), anti-Ly49H (3D10), anti-CD69 (H1.2F3), anti-CD27 (LG.7F9), anti-CD11b (M1/70), anti-CD8α (53–6.7), anti-IFN-γ (XMG1.2), anti-CD19 (1D3), anti-MHC-II (M5/114.15.2), anti-CD11c (N418), and PE-labeled streptavidin (SA-PE). For the in vivo cell proliferation assay, mice were i.p. injected with 2 mg of bromodeoxyuridine (BrdU; Sigma) and sacrificed 3 h later. To detect incorporated BrdU, splenic leukocytes were first stained for surface antigens and then fixed, permeabilized, refixed, treated with DNase I, and intracellularly stained according to the manufacturer's protocol (BrdU flow kit; BD Pharmingen). For the detection of IFN-γ expression by NK cells, incubation was performed in RPMI medium supplemented with 10% of fetal calf serum (FCS; Gibco) for 5 h in the presence of 500 IU/ml of interleukin-2 (IL-2) at 37°C with 1 μg/ml of brefeldin A (eBioscience) added for the last 4 h of incubation. For detection of IFN-γ expression by CD8+ T cells, incubation was performed in RPMI medium supplemented with 10% of FCS (Gibco) and 1 μg of H-2Db-restricted M45-derived peptide (985HGIRNASFI993), H-2Kb-restricted m139 derived peptide (419TVYGFCLL426), or H-2Kb-restricted M57 derived peptide (816SCLEFWQRV824) for 5 h at 37°C with 1 μg/ml of brefeldin A (eBioscience) added for the last 4 h of incubation (35). Custom MCMV-specific H-2b class I-restricted antigenic peptide synthesis to a purity of greater than 80% was performed by Jerini Peptide Technologies. After incubation, cells were first surface stained and then fixed and permeabilized using Cytofix/Cytoperm solutions (BD Pharmingen) followed by intracellular IFN-γ staining according to the manufacturer's protocol. For DC isolation, splenocytes were digested using collagenase D (Roche) as described previously (40). Flow cytometry was performed on FACSCalibur and FASCAria (BD Bioscience; San Jose, CA), and data were analyzed using the FlowJo software (Tree Star).
Frequencies of IFN-γ-secreting CD8+ T cells were determined by an ELISpot assay essentially as described in greater detail previously (reference 7 and references therein). Briefly, CD8+ T cells were isolated from lymph node cells by immunomagnetic cell sorting using the autoMACS system (Miltenyi Biotec Systems, Bergisch Gladbach, Germany) as described previously (27), and graded cell numbers in triplicate cultures were stimulated for 18 h with 105 EL-4 (H-2b) thymoma cells per assay culture and 10−6 M synthetic peptides M45, m139, or M57 (see above). After staining for IFN-γ and enzymatic color development, frequencies of responding cells and the corresponding 95% confidence intervals were determined from the spot counts by intercept-free linear regression analysis.
Quantification of serum cytokine levels.
Serum levels of IFN-α were determined by an enzyme-linked immunosorbent assay (ELISA) kit for IFN-α (PBL Biomedical Laboratories) according to the manufacturer's instructions. Serum levels of IL-12, IFN-γ, IL-6, tumor necrosis factor alpha (TNF-α), IL-15, and IL-10 were determined by the Bio-Rad mouse cytokine multiplex assay according to the manufacturer's instructions (Bio-Rad Laboratories, Hercules, CA).
Statistical analysis.
Statistical analysis was carried out using Prism 5 (GraphPad Software, La Jolla, CA). Statistically significant differences between two data sets in cytokine assays, phenotype analyses, and quantitation of viral transcripts (log-transformed numbers) were determined by the unpaired two-tailed Student's t test, and P values of <0.05 were considered significant. Differences in viral titers were determined by two-tailed Mann-Whitney U test.
RESULTS
Impact of NK cell control on the dynamics of an early antiviral CD8+ T-cell response.
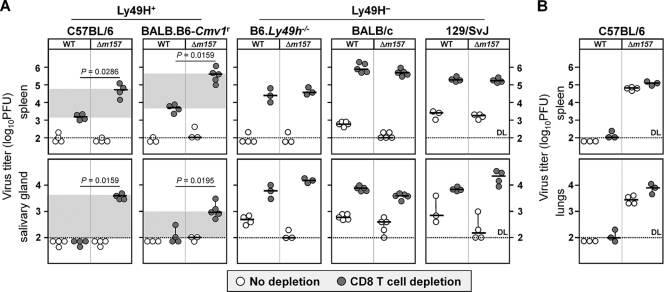
MCMV subverts NK cell control by encoding four proteins involved in the downmodulation of ligands for the activating NK cell receptor NKG2D. Specifically, m145, m152, and m155 interfere with cell surface expression of MULT-1, of RAE-1 family members, and of H60, respectively (19, 30). In addition, m138 simultaneously targets MULT-1, H60, and RAE-1ε (5, 29). However, NK cells can be alternatively engaged by the viral protein m157, a ligand of the activating NK cell receptor Ly49H expressed differentially in mouse strains (6, 46). Accordingly, for studying the impact of NK cells on the early CD8+ T-cell response, we here used Ly49H+ mouse strains capable of mounting an NK cell response regardless of the presence of viral inhibitors of NK cells. The effect of CD8+ T-cell depletion on virus titers in different organs 7 days p.i. in Ly49H+ and, for comparison, in Ly49H− mice infected with either WT MCMV or mutant virus lacking the m157 gene (Δm157 virus) was tested (Fig. 1A). On day 7 p.i., in both C57BL/6 and BALB.B6-Cmv1r mice (both Ly49H+) depleted of CD8+ T cells, a significant increase of Δm157 virus titer was observed, whereas only a minor effect could be seen in WT infected mice (Fig. 1A, left). In contrast, in B6.Ly49h−/− mice, as well as in BALB/c and 129/SvJ mice (all three Ly49H−), the depletion of CD8+ T cells significantly increased the viral titers irrespective of the virus used (Fig. 1A, right). However, contrary to the findings on day 7 p.i., the depletion of CD8+ T cells did not notably affect viral titers in organs of C57BL/6 mice on day 3.5 p.i., thus suggesting that in an early stage of infection CD8+ T cells do not compensate for the absence of NK cell activity that is caused by lack of Ly49H activation in Ly49H+ mice infected with the Δm157 virus (Fig. 1B). This is in agreement with the interpretation that expression of m152 simultaneously prevents early NK as well as CD8+ T-cell control by downmodulating RAE-1 and MHC-I, respectively (24, 31, 55). Accordingly, at the same time, NK as well as CD8+ T cells were found to control Δm152 virus (7). Notably, although it is well established that MCMV infection within the salivary glands is controlled primarily by CD4+ T cells (22; reviewed in reference 9), Δm157 virus titers in Ly49H+ mice as well as titers for both viruses in Ly49H− mice indicated that salivary gland colonization in the first 7 days is under CD8+ T cell control (Fig. 1A).
Fig 1.
NK cell-specific recognition of MCMV affects the CD8+ T-cell response. (A) Indicated strains of mice were i.v. injected with 2 × 105 PFU of either WT or Δm157 MCMV. On days 1 and 5 p.i., mice were i.p. injected with phosphate-buffered saline (PBS) or depleting anti-CD8 MAb. On day 7 p.i., viral titers in spleens and salivary glands were determined by plaque assay. (B) C57BL/6 mice were i.v. injected with 2 × 105 PFU of either WT or Δm157 MCMV. On day 1 p.i., mice were i.p. injected with PBS or depleting anti-CD8 MAb. On day 3.5 p.i., viral titers in spleens and lungs were determined by plaque assay. Titers of individual mice (circles) and median values (horizontal bars) are shown. Representative data of at least two independent experiments with three to five mice per group are shown. Differences are regarded as significant for P values of <0.05. Dotted line, detection limit.
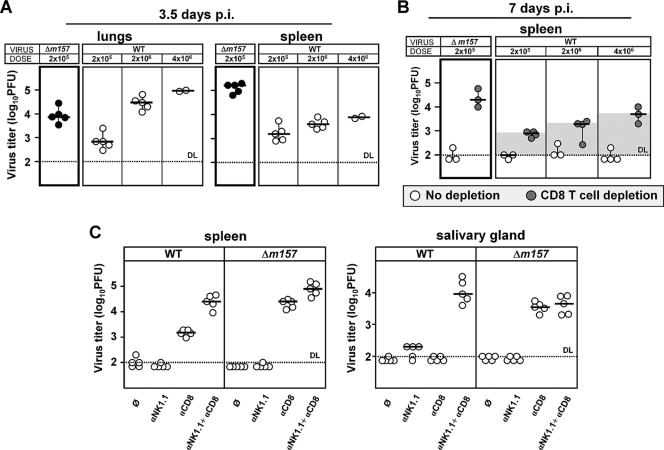
Next, we tested whether the differential requirements for CD8+ T cells on day 7 p.i. in control of WT MCMV and Δm157 virus (Fig. 1A, left) could be explained by a different virus load at early times when NK cells play a decisive role. To that aim, C57BL/6 mice were i.v. injected with different doses of WT MCMV (2 × 105 PFU, 2 × 106 PFU, or 4 × 106 PFU) to match the virus titers in mice that received a single dose of Δm157 virus (2 × 105 PFU). On day 3.5 p.i., only in mice that received 10- to 20-fold more WT MCMV, virus titers in organs reached those observed in mice infected with 2 × 105 PFU of Δm157 virus (Fig. 2A). Even the highest dose of WT MCMV failed to reach the titers observed for Δm157 virus in the spleen, thus illustrating the robustness of the Ly49H–m157 pathway in the control of the virus in this organ. In addition, we tested the requirement for CD8+ T cells on day 7 p.i. in mice that received different doses of WT MCMV and compared this to mice injected with only a single dose of Δm157 virus (Fig. 2B). In agreement with results shown in Fig. 1A, the depletion of CD8+ T cells had only a minor effect on the virus titer in mice infected with a lower dose of WT MCMV (Fig. 2B). However, by increasing the dose of WT MCMV (2 × 106 PFU or 4 × 106 PFU), CD8+ T cells became increasingly more important. Yet, the depletion of CD8+ T cells in mice infected with a 20-fold higher dose of WT MCMV still failed to reach the titer found in the spleen of the CD8+ T-cell depleted group infected with 2 × 105 PFU of Δm157 virus, thus suggesting that in addition to influencing viral load, NK cell signaling via Ly49H–m157 interaction may play also a qualitative role in the regulation of the CD8+ T-cell response. To test this assumption, we performed combined depletion of NK cells and CD8+ T cells in MCMV-infected mice (Fig. 2C). On day 7 p.i. in WT MCMV-infected mice depleted of both NK cells and CD8+ T cells, viral titers in spleen and salivary glands reached those observed in CD8+ T-cell depleted mice infected with Δm157 virus. Thus, this result indicates that enhanced CD8+ T-cell response in mice infected with Δm157 virus positively correlates with the virus load.
Fig 2.
Virus load affects the CD8+ T-cell-mediated control of MCMV infection in C57BL/6 mice. (A) C57BL/6 mice were i.v. injected with different doses of WT MCMV (2 × 105 PFU, 2 × 106 PFU, or 4 × 106 PFU) or with a single dose of 2 × 105 PFU of Δm157 MCMV. On day 3.5 p.i., viral titers in spleens and lungs were determined by plaque assay. (B) C57BL/6 mice were injected as described above, and on days 1 and 5 p.i. they were i.p. injected with PBS or depleting anti-CD8 MAb. On day 7 p.i., viral titers in spleens were determined by plaque assay. (C) C57BL/6 mice were i.v. injected with 2 × 105 PFU of either WT or Δm157 MCMV. Indicated groups were depleted of NK cells, CD8+ T cells, or both NK cells and CD8+ T cells. On day 7 p.i., viral titers in spleen and salivary glands were determined by plaque assay. Ø, untreated C57BL/6 mice. Data are compiled from two independent experiments with two to five mice per group. Titers of individual mice (circles) and median values (horizontal bars) are shown. Dotted line, detection limit.
The impact of Ly49H–m157 interaction on NK cell activation, maturation, and capacity to control virus replication.
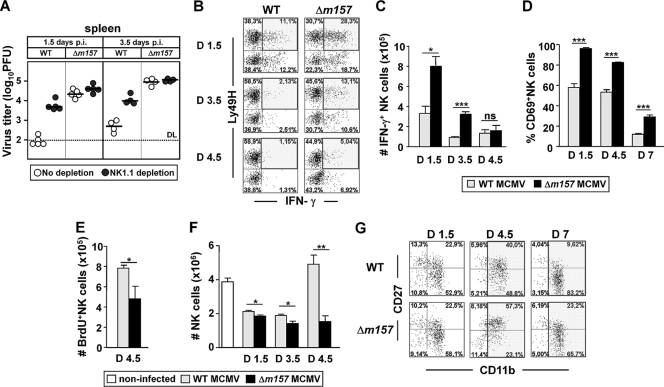
There has been great progress in understanding of how the innate immune response influences adaptive immunity (reviewed in reference 50). The results presented above suggest that an early CD8+ T-cell response in C57BL/6 mice is shaped by the Ly49H–m157 interaction. Next, we decided to test whether the enhanced CD8+ T-cell response in C57BL/6 mice infected with Δm157 MCMV can be related to a differential response of NK cells. In accordance with previous work (8), early virus control in C57BL/6 mice was largely dependent on the Ly49H–m157 interaction. Specifically, deletion of gene m157 from the virus genome abolished the antiviral control mediated by NK cells during the first 4 days p.i. (Fig. 3A). For characterizing the NK cells, C57BL/6 mice were injected i.v. with 2 × 105 PFU of the indicated viruses, and NK cells in the spleen were analyzed for IFN-γ production, proliferation, and expression of various markers. A high frequency of Ly49H+ IFN-γ+ NK cells was observed in mice infected with Δm157 MCMV compared to WT MCMV-infected mice as early as day 1.5 p.i. (Fig. 3B and C). This difference was present throughout the observed period. In accordance with the above, the absolute numbers of IFN-γ + NK cells were significantly higher in mice infected with Δm157 on days 1.5 and 3.5 p.i. (Fig. 3C). Similarly to IFN-γ, the expression of the early activation marker CD69 on NK cells was higher in mice infected with the Δm157 virus (Fig. 3D). At first glance, these findings were surprising, as one rather might have expected lower levels of NK cell activation and IFN-γ production in the absence of Ly49H–m157 signaling. It thus appears that low viral load after efficient virus control in WT MCMV infection limits the numbers of activated NK cells, possibly due to generally lower cytokine levels. Contrary to IFN-γ production, however, the proliferation of NK cells was significantly high in mice infected with WT MCMV (13, 16, 26) compared to Δm157-infected mice, indicating a role for Ly49H–m157 signaling in triggering NK cell proliferation (Fig. 3E). This is in line with the absolute numbers of NK cells in the spleen of infected mice (Fig. 3F). Specifically, whereas in WT MCMV-infected mice, after an initial drop, the NK cell number in the spleen returned to the control level on day 4.5 p.i., in Δm157-infected mice the numbers of splenic NK cells remained low. The kinetics of NK cell maturation was analyzed by the expression of CD27 and CD11b markers (17). The maturation of NK cells, characterized by a high frequency of NK cells expressing the most mature phenotype CD27low CD11bhigh, was enhanced in WT MCMV-infected mice (Fig. 3G). Compared to the situation in WT MCMV-infected mice, the frequency of CD27low CD11bhigh NK cells was lower on day 4.5 p.i. in mice infected with Δm157 MCMV, whereas the frequency of CD27high CD11bhigh NK cells was higher. By day 7 p.i., the majority of NK cells also had matured to the phenotype CD27low CD11bhigh, but, compared to WT MCMV-infected mice, a notable frequency of NK cells in mice infected with Δm157 MCMV still remained in the double-positive stage. Thus, these results showed that Ly49H–m157 signaling drives NK cell proliferation and phenotypic maturation, whereas IFN-γ secretion and CD69 expression were higher in Δm157-infected mice, putatively as a consequence of a higher virus load in organs.
Fig 3.
Impact of Ly49H–m157 signaling on the NK cell response during early MCMV infection. (A) C57BL/6 mice, either NK cell depleted with anti-NK1.1 MAb or left undepleted, were i.v. injected with 2 × 105 PFU of either WT or Δm157 MCMV. On days 1.5 and 3.5 p.i., viral titers in spleens were determined by plaque assay. Titers of individual mice (circles) and median values (horizontal bars) are shown. (B) On days 1.5, 3.5, and 4.5 p.i., splenocytes were assessed for IFN-γ production by Ly49H+ and Ly49H− subsets of CD3ε− NK1.1+ NK cells. (C to F) The absolute numbers of IFN-γ+ CD3ε− NK1.1+ NK cells (C), the percentages of CD69+ CD3ε− NK1.1+ NK cells (D), the absolute numbers of proliferating bromodeoxyuridine-positive (BrdU+) CD3ε− NK1.1+ NK cells (E), and the absolute numbers of CD3ε− NK1.1+ NK cells (F) in the total spleen cell population at the indicated time points are shown. (G) The maturation pattern of NK cells on days 1.5, 4.5, and 7 p.i. was determined by expression of cell surface markers CD27 and CD11b. Representative dot plots of splenic NK cells (CD3ε− NK1.1+) from two independent experiments with three or four mice per group are shown. (C to F) Data are presented as means ± standard error of the mean (SEM) of at least three mice per group from one of two independent experiments. Asterisks denote significant values as calculated by unpaired, two-tailed Student's t test: ns, nonsignificant; *, P < 0.05; **, P < 0.01; ***, P < 0.001. Dotted line, detection limit.
Enhanced CD8+ T-cell response in mice infected with the Δm157 virus.
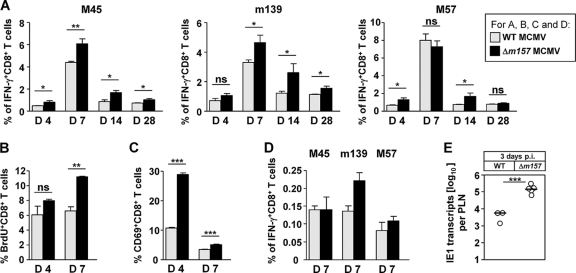
To further evaluate the impact of early NK cell activation on the CD8+ T-cell response, we measured the frequency of IFN-γ+ CD8+ T cells in MCMV-infected mice upon stimulation with M45, m139, and M57 peptides in vitro. As early as on day 4 p.i., we observed a slight increase in the frequency of CD8+ T cells specific for the above-mentioned viral epitopes in mice infected with Δm157 virus compared to WT MCMV-infected mice. The differences were more pronounced on day 7 p.i. for CD8+ T cells directed against the epitopes M45 and m139, whereas no significant difference was revealed for the M57 epitope (Fig. 4A). The high frequency of MCMV-specific CD8+ T cells in mice infected with Δm157 virus, as compared to WT MCMV-infected mice, was present throughout the observed period of 4 weeks (Fig. 4A). The proliferation capacity of CD8+ T cells was also enhanced on day 7 p.i. in mice infected with Δm157 MCMV, compared to the WT MCMV-infected group (Fig. 4B). In addition, on day 4 p.i., the frequency of activated CD8+ T cells expressing CD69 was significantly higher in mice infected with Δm157 MCMV (Fig. 4C) and was still somewhat increased on day 7.
Fig 4.
Absence of Ly49H–m157 signaling enhances the CD8+ T-cell response in MCMV-infected mice. C57BL/6 mice were infected intravenously (A to C) or at the left hind footpad (D and E) with 2 × 105 PFU of either WT or Δm157 MCMV. (A to D) On the indicated days p.i., splenic leukocytes (A to C) or immunomagnetically purified CD8+ T cells from the draining popliteal lymph node (PLN) (D) were stimulated in culture with the indicated peptides followed by intracellular IFN-γ staining in cytofluorometric analysis (A to C) or IFN-γ secretion in the ELISpot assay (D). (A) The percentages of IFN-γ+ CD8+ T cells specific for M45, m139, and M57 MCMV peptides among gated CD3ε+ CD8+ T cells are shown. (B and C) The percentages of proliferating BrdU+ CD8+ T cells (B) and CD69+ CD8+ T cells (C) among gated CD3ε+ CD8+ T cells are shown. (A to C) Data are presented as means ± SEM of at least three mice per group from one of at least three independent experiments. (D) Immunomagnetically purified CD8+ T cells from pools of PLN of five mice per group were stimulated for an IFN-γ-based ELISpot assay with EL-4 (H-2b) thymoma cells and a saturating concentration (10−6 M) of synthetic peptides corresponding to the epitopes indicated. Bars represent frequencies of responding CD8+ T cells; error bars indicate the 95% confidence intervals. (E) IE1 transcripts quantitated by RT-qPCR in draining PLN on day 3 after intraplantar infection. Symbols represent PLN of individual mice with the median value indicated. Throughout, asterisks denote significance values as calculated by unpaired, two-tailed Student's t test: ns, nonsignificant; *, P < 0.05; **, P < 0.01; ***, P < 0.001.
We next investigated if the enhanced CD8+ T-cell response against Δm157 virus is restricted to i.v. infection or applies to other routes of infection as well. To that aim, the frequencies of virus-specific CD8+ T cells present in the draining popliteal lymph node (PLN) on day 7 after footpad infection were assessed. The results shown in Fig. 4D essentially support the results discussed above for i.v. infection, although the pattern of epitopes that profit from missing NK cell activation was not absolutely congruent with that after i.v. infection. However, taking both routes of infection together, m139 showed consistent results, and all three epitopes had the potential to profit under either of the infection routes. One may speculate that epitope-specific differences depend on the relative contributions of direct antigen presentation and cross-presentation to CD8+ T-cell priming and that this might differ between viral antigenic proteins and routes of infection. Notably, enhanced priming in the PLN in the absence of Ly49H–m157 interaction correlated with an elevated intranodal viral gene expression (Fig. 4E) and thus with a higher viral antigen load.
Altogether, these results indicate that NK cell activation through Ly49H–m157 interaction negatively regulates the CD8+ T-cell response, which likely relates to a reduced virus replication and thus a reduced antigen load available for CD8+ T-cell priming.
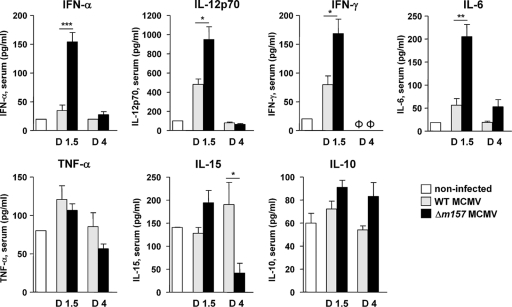
Secretion of IFN-α and other proinflammatory cytokines correlates with the CD8+ T-cell response.
A number of studies emphasized the role of IFN-α and other innate cytokines in supporting CD8+ T-cell priming through effects on both antigen-presenting cells and T cells (reviewed in reference 18). It has been shown previously that MCMV infection induces high serum levels of IFN-α, IL-12, and IFN-γ in B6.Ly49h−/− mice on day 1.5 p.i. (15). Based on this finding and in an attempt to explain the enhanced CD8+ T-cell response in mice infected with Δm157 MCMV, we assessed the levels of proinflammatory cytokines in sera of infected mice on days 1.5 and 4 p.i. At the early time point, the serum level of IFN-α was ∼4-fold higher in the group of mice infected with Δm157 virus than in WT MCMV-infected mice (Fig. 5). A similar pattern was observed for the other three proinflammatory cytokines analyzed, namely, IL-12, IFN-γ, and IL-6 (Fig. 5). Yet, the level of all four of these cytokines dropped almost completely by day 4 p.i., suggesting a tight feedback regulation. Contrary to those of the above-mentioned cytokines, the serum levels of TNF-α were similarly induced with both viruses and only partially downregulated by day 4. The IL-15 level was elevated on day 1.5 p.i. with Δm157 virus, compared to WT infected mice, but was strikingly reduced on day 4 p.i. specifically in the group of mice infected with the Δm157 virus. Of note is that this correlates with an impaired NK cell proliferation upon Δm157 MCMV infection (Fig. 3E). In contrast to proinflammatory cytokines, the concentration of IL-10 was only modestly upregulated in Δm157-infected C57BL/6 mice, but this upregulation lasted until day 4. These results suggest that an early antiviral response, characterized by a high level of proinflammatory cytokines, may be a parameter in determining the outcome of the CD8+ T-cell response.
Fig 5.
Impact of Ly49H–m157 signaling on cytokine production during early MCMV infection. C57BL/6 mice were i.v. injected with 2 × 105 PFU of either WT or Δm157 MCMV or left uninfected. On days 1.5 and 4 p.i., serum levels of the indicated cytokines were determined. Data are presented as means ± SEM of at least three mice per group from one of two independent experiments. Asterisks denote significant values as calculated by unpaired, two-tailed Student's t test: *, P < 0.05; **, P < 0.01; ***, P < 0.001. Φ, not detected.
To further evaluate the impact of cytokine-mediated CD8+ T-cell response, we analyzed the requirements for CD8+ T cells in control of MCMV infection of B6.IFNα/βR−/− and TNF-Rp55−/− mice. Unlike normal C57BL/6 mice, which control Δm157 virus on day 7 p.i. via a CD8+ T-cell-dependent mechanism, B6.IFNα/βR−/− mice failed to do so, suggesting that type I IFNs play a role in enhancing CD8+ T-cell response. Contrary to B6.IFNα/βR−/− mice, TNF-Rp55−/− mice efficiently controlled Δm157 infection by CD8+ T cells, indicating that TNF receptor type I signaling does not contribute to enhanced CD8+ T-cell control of Δm157 virus (data not shown).
The dynamics of splenic DCs correlates with the level of proinflammatory cytokines.
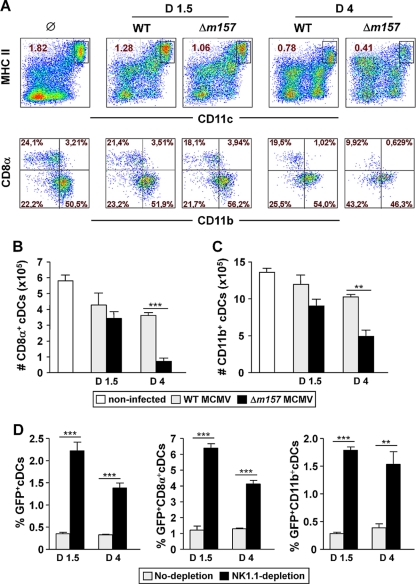
Both the life span and the survival of DCs in vivo are critical for the priming of the T-cell immune response. NK cells cross talk with DCs and thus shape the specific immune response (reviewed in reference 2). This includes NK cell killing of immature DCs and the promotion of DC maturation by NK cell-derived IFN-γ and TNF-α, which can eventually enhance antigen presentation to T cells (reviewed in reference 52). Conversely, DCs are important in NK cell activation and the initiation of the specific immune response (11). Upon MCMV infection in vivo, antigen-presenting cells such as conventional dendritic cells (cDCs), plasmacytoid dendritic cells (pDCs), and macrophages are key producers of type I IFN and other proinflammatory cytokines (1, 12, 25, 48, 57). However, pDCs seem to play a central role in mounting a protective type I IFN response (25, 57). In order to address the impact of DCs on virus susceptibility to NK cell control and the level of proinflammatory cytokines, we analyzed splenic cDCs during the early phase of MCMV infection (Fig. 6). On day 1.5 p.i., we observed no differences in the frequencies of splenic cDCs (MHC-II+ CD11c+) (Fig. 6A, top) or their CD8α+ and CD11b+ subsets (Fig. 6A, bottom) in the comparison between the two viruses. In addition, at this time point p.i., the absolute numbers of CD8α+ (Fig. 6B) and CD11b+ (Fig. 6C) cDC subsets were similar between the two infected groups of mice. However, on day 4 p.i., a dramatic reduction of splenic cDCs was observed specifically in mice infected with Δm157 virus, which may be secondary to the increased level of infection in the absence of an efficient NK cell control. To determine the extent of cDC infection, we infected C57BL/6 mice with recombinant WT MCMV expressing green fluorescent protein (WT MCMV-GFP). Depletion of NK cells significantly increased the frequency of MCMV-infected cDCs and CD8α+ and CD11b+ cDC subsets on days 1.5 and 4 p.i. (Fig. 6D). Therefore, although the frequency of splenic cDCs decreased on day 4 p.i. in mice lacking NK cell control, the high frequency of infected cDCs among the residual population might explain the improved priming of CD8+ T cells. To conclude, the preservation of cDC subsets on day 1.5 p.i. and the higher frequency of infected cDCs at early time points p.i., as well as the higher level of proinflammatory cytokines, might contribute to the enhanced CD8+ T-cell response observed in mice devoid of Ly49H signaling. In addition, our findings imply that the reduction of splenic cDCs observed on day 4 p.i. does not compromise the CD8+ T-cell response.
Fig 6.
Ly49H–m157 signaling preserves splenic cDCs during MCMV infection. C57BL/6 mice were i.v. injected with 2 × 105 PFU of either WT or Δm157 MCMV or left uninfected. On days 1.5 and 4 p.i., splenic leukocytes were stained to assess DC subsets cytofluorometrically. (A) Representative dot plots of splenic cDCs (CD3− CD19− MHC-II+ CD11c+) and CD8α+ and CD11b+ subsets gated on cDCs are shown. (B and C) The absolute numbers of CD8α+ cDCs (B) and CD11b+ cDCs (C) in total splenic leukocytes are shown. (D) C57BL/6 mice, either NK cell depleted or left undepleted, were i.v. injected with 106 PFU of WT MCMV-GFP. The frequency of infected splenic cDCs (CD3− CD19− MHC-II+ CD11c+ GFP+) and infected CD8α+ and CD11b+ subsets gated on cDCs on days 1 and 4 p.i. are shown. (B to D) Data are presented as means ± SEM of at least four mice per group from one of three independent experiments. Asterisks denote significant values as calculated by unpaired, two-tailed Student's t test: **, P < 0.01; ***, P < 0.001.
DISCUSSION
CD8+ T cells play a key role in the control of human cytomegalovirus (HCMV) infections (53). Therefore, the understanding of the cellular and molecular mechanisms involved in priming and shaping of CD8+ T-cell response is of high relevance. Both host and viral factors contribute to its nature and quality (reviewed in reference 28). There exists evidence to suggest that innate immunity mediated by NK cells is a critical parameter in determining the strength and quality of the primary CD8+ T-cell response to MCMV (3, 40). MCMV can activate NK cells by encoding protein m157, a ligand of the activating NK cell receptor Ly49H. Here we have specifically investigated the impact of NK cell activation through Ly49H-m157 signaling on CD8+ T-cell priming and antiviral control. Our results in C57BL/6 mice demonstrated that the requirement for CD8+ T cells in the virus control during the first week p.i. is determined by the engagement of the Ly49H receptor and the capacity of NK cells to control virus replication during the first 2 days. While CD8+ T cells proved to be dispensable for early virus control in mice infected with WT MCMV, they became essential in mice infected with a virus strain unable to activate NK cells through the Ly49H receptor, due to deletion of the ligand-coding gene m157. Moreover, depletion of NK cells in WT MCMV-infected mice also increased the requirements for CD8+ T-cell control to a level comparable with the one observed in mice infected with Δm157 virus. An enhanced CD8+ T-cell response after infection with Δm157 virus could at least in part be explained by a higher antigenic load, as suggested previously from a related model of CD8+ T-cell priming to MCMV (7; reviewed in reference 28). Here we provide evidence to conclude that, besides antigenic load, elevated levels of proinflammatory cytokines and preservation of cDCs at early times of infection contribute to the enhanced CD8+ T-cell response in mice in which the lack of Ly49H–m157 signaling precludes NK cell-dependent early virus control.
Our data show that Ly49H–m157 interaction affects the NK cell activation pattern early on. NK cell expression of both IFN-γ and CD69 were higher in mice that failed to control virus replication via the Ly49H–m157 pathway (Fig. 3B to D). This finding also correlates with the level of proinflammatory cytokines, known to induce CD69 upregulation and IFN-γ production by NK cells (14, 36). Although NK cells in mice infected with Δm157 virus were stimulated to produce more IFN-γ as early as 1.5 days p.i., this was obviously not sufficient to limit virus replication in the absence of a specific engagement of the Ly49H NK cell receptor. By the same token, NK cells derived from Δm157-infected mice failed to proliferate, thus confirming that a specific engagement of Ly49H receptor is required for this function (13, 16). This is in accordance with previous reports showing that ligation of Ly49H receptor to m157 is necessary for induction of NK cell proliferation (6, 46). Furthermore, reduced proliferation capacity of NK cells and their delayed maturation in mice infected with Δm157 virus may also be related to the observed sudden decrease in IL-15 secretion, which was not the case in mice infected with WT MCMV (Fig. 3E and G and 5).
Although interesting on their own, the different NK cell maturation and activation patterns in mice infected with WT MCMV or Δm157 virus could not directly explain the differences in the CD8+ T-cell response. Several studies have highlighted the role of proinflammatory cytokines in the activation of the cells of the innate and adaptive immune response (37; reviewed in reference 18). Cytokines produced by NK cells can prime DCs to produce IL-12 and promote their maturation, thereby enabling them to become potent antigen-presenting cells (1; reviewed in references 2 and 56). In addition, IFN-γ produced by NK cells promotes antigen processing and presentation to T cells (33; reviewed in reference 39). Here we showed a dramatic rise in proinflammatory cytokines, including IFN-α, IL-12, IFN-γ, and IL-6, which may drive a subsequent CD8+ T-cell response. Furthermore, using mice lacking IFNα/β receptor or TNF-Rp55 receptor, we demonstrated that type I IFNs play a role in enhancing CD8+ T-cell response to the virus unable to activate NK cells, whereas TNF signaling does not contribute to this function. Elevated levels of proinflammatory cytokines on day 1.5 p.i. are in agreement with a previous finding made in MCMV-infected B6.Ly49h−/− mice (15). The present study also demonstrated a tight regulation of proinflammatory cytokines, since on day 4 p.i. most of these were close to the levels observed in control groups. We speculate that this tight feedback control is aimed at preventing unleashed proliferation of NK cells and CD8+ T cells and possibly also immunopathology. At this time point, the serum levels of IL-10 were still high, suggesting its role in negative feedback regulation of immune response (26; reviewed in reference 41).
MCMV infection affects DC maturation, frequently resulting in their depletion from the spleen (4). In agreement with this work, we have shown here that on day 1.5 p.i., cDC subsets were not affected, irrespective of the virus used, and that a dramatic drop was observed 4 days p.i. only if the engagement of the Ly49H receptor was missing (4). Interestingly, in another work, the drop in splenic cDCs in Ly49H− mice was observed already on day 1.5 p.i. (40). These differences might be related to different mouse strains or the different source of virus used. In recent work, Stadnisky et al. (47) showed that NK cell activation by the missing-self mechanism also resulted in faster recovery of splenic cDCs after their initial drop and in an improved CD8+ T-cell response. Thus, the more robust CD8+ T-cell response in mice infected with Δm157 virus looks like a paradox since it is accompanied by a depletion of splenic cDCs. This could be explained by a higher level of virus replication and expression of epitope-encoding viral genes for antigen cross-presentation in Δm157 infection, analogous to the model proposed by Böhm et al., which demonstrated that a virus with a more efficient replication gives better priming (7; reviewed in reference 28). Specifically, in that model, WT MCMV was superior to an attenuated immune evasion gene deletion mutant in the priming of CD8+ T cells due to higher replication and antigen delivery. Our results represent the mirror image of this model by showing that in C57BL/6 mice, more efficiently replicating Δm157 virus primes CD8+ T cells better than WT MCMV does. In addition, the frequency of infected cDCs among residual population on day 4 p.i. was still significantly higher in mice lacking NK cell control. Thus, it is likely that these cells might serve as a source for antigen presentation and cross-presentation. Therefore, we believe that CD8+ T-cell response is determined by preserved cDCs, higher frequency of infected cDCs, and high levels of proinflammatory cytokines during the first 2 days p.i. and that the subsequent reduction of cDCs has no negative effect on the strength of ongoing CD8+ T-cell response. Alternatively, in Δm157-infected mice, cDCs might not be physically deleted from the spleen but might rather be recruited to other sites of infection due to a higher level of virus replication. It was suggested that the depletion of cDCs is primarily mediated by NK cell cytotoxicity in a perforin-dependent manner (3). However, we have found a dramatic reduction of the splenic cDC pool during an acute MCMV infection also in mice deficient in perforin (data not shown), suggesting that the depletion of cDCs is not based on this cytolytic mechanism.
The impact of NK cells on the subsequent dynamics and the overall quality of the adaptive immune response may also differ with respect to the activation signaling pathway in NK cells involved in the initial virus control. Indeed, in a recent study we have demonstrated that NK cell activation via the activating NK cell receptor NKG2D after infection of mice with a recombinant virus expressing the NKG2D ligand RAE-1γ resulted in a profound virus attenuation during the first days p.i., yet, notably, without affecting the magnitude of the MCMV-specific CD8+ T-cell response (45). Challenge experiments indicated that protection after immunization with RAE-1γMCMV was even superior to the protection observed after immunization with WT MCMV. Even more striking results were observed in C57BL/6 mice infected with RAE-1γMCMV. As this virus also expresses the m157 protein, the Ly49H NK receptor is engaged and controls the virus, which, as shown here, ought to reduce CD8+ T cell priming. Surprisingly, however, the response was enhanced at early times after infection with the recombinant virus expressing RAE-1γ in addition to m157. These findings might suggest that different NK cell signaling pathways have different impacts on the subsequent CD8+ T-cell response. Alternatively, enhancement of priming by expression of an NKG2D ligand on infected cells may involve costimulation by engagement of NKG2D expressed on CD8+ T cells (32).
Collectively, the currently available body of data clearly demonstrates that the impact of NK cells on the priming of a CD8+ T-cell response depends on the context of infection conditions and a complex set of host and viral factors, arguing against an easily predictable single mode and fixed direction of NK cell regulation of the primary CD8+ T cell response. This complexity of parameters and outcomes may explain some of the apparent discrepancies in the literature.
ACKNOWLEDGMENTS
We thank Luka Traven, Irena Slavuljica, and Alexej Schmidt for critical readings of the manuscript. We thank Edvard Ražić, Renata Grgurić, Dijana Rumora, and Karmela Miklić for technical help, as well as Milka Fritz Boukhatem for taking care of the mice.
This work was supported by NIH grant 1R01AI083201-01 (S.J.), the Croatian Science Foundation project 04/16 (S. Jonjić), and Croatian Ministry of Science and Technology project 0621261-1268 (A.K.) and the Canadian Institutes of Health Research MOP-7781 and the Canada Research Chairs Program (S.M.V.), as well as by the German Research Foundation, collaborative research grant SFB490, individual projects E3 (S.E.) and E4 (M.J.R.).
Footnotes
Published ahead of print 7 December 2011
REFERENCES
- 1. Andoniou CE, et al. 2005. Interaction between conventional dendritic cells and natural killer cells is integral to the activation of effective antiviral immunity. Nat. Immunol. 6:1011–1019 [DOI] [PubMed] [Google Scholar]
- 2. Andrews DM, et al. 2005. Cross-talk between dendritic cells and natural killer cells in viral infection. Mol. Immunol. 42:547–555 [DOI] [PubMed] [Google Scholar]
- 3. Andrews DM, et al. 2010. Innate immunity defines the capacity of antiviral T cells to limit persistent infection. J. Exp. Med. 207:1333–1343 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Andrews DM, Scalzo AA, Yokoyama WM, Smyth MJ, Degli-Esposti MA. 2003. Functional interactions between dendritic cells and NK cells during viral infection. Nat. Immunol. 4:175–181 [DOI] [PubMed] [Google Scholar]
- 5. Arapovic J, Lenac Rovis T, Reddy AB, Krmpotic A, Jonjic S. 2009. Promiscuity of MCMV immunoevasin of NKG2D: m138/fcr-1 down-modulates RAE-1epsilon in addition to MULT-1 and H60. Mol. Immunol. 47:114–122 [DOI] [PubMed] [Google Scholar]
- 6. Arase H, Mocarski ES, Campbell AE, Hill AB, Lanier LL. 2002. Direct recognition of cytomegalovirus by activating and inhibitory NK cell receptors. Science 296:1323–1326 [DOI] [PubMed] [Google Scholar]
- 7. Böhm V, et al. 2008. The immune evasion paradox: immunoevasins of murine cytomegalovirus enhance priming of CD8 T cells by preventing negative feedback regulation. J. Virol. 82:11637–11650 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Bubic I, et al. 2004. Gain of virulence caused by loss of a gene in murine cytomegalovirus. J. Virol. 78:7536–7544 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Campbell AE, Cavanaugh VJ, Slater JS. 2008. The salivary glands as a privileged site of cytomegalovirus immune evasion and persistence. Med. Microbiol. Immunol. 197:205–213 [DOI] [PubMed] [Google Scholar]
- 10. Cobbold SP, Jayasuriya A, Nash A, Prospero TD, Waldmann H. 1984. Therapy with monoclonal antibodies by elimination of T-cell subsets in vivo. Nature 312:548–551 [DOI] [PubMed] [Google Scholar]
- 11. Dalod M, et al. 2003. Dendritic cell responses to early murine cytomegalovirus infection: subset functional specialization and differential regulation by interferon alpha/beta. J. Exp. Med. 197:885–898 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Dalod M, et al. 2002. Interferon alpha/beta and interleukin 12 responses to viral infections: pathways regulating dendritic cell cytokine expression in vivo. J. Exp. Med. 195:517–528 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Dokun AO, et al. 2001. Specific and nonspecific NK cell activation during virus infection. Nat. Immunol. 2:951–956 [DOI] [PubMed] [Google Scholar]
- 14. Draghi M, et al. 2007. NKp46 and NKG2D recognition of infected dendritic cells is necessary for NK cell activation in the human response to influenza infection. J. Immunol. 178:2688–2698 [DOI] [PubMed] [Google Scholar]
- 15. Fodil-Cornu N, et al. 2008. Ly49h-deficient C57BL/6 mice: a new mouse cytomegalovirus-susceptible model remains resistant to unrelated pathogens controlled by the NK gene complex. J. Immunol. 181:6394–6405 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. French AR, et al. 2006. DAP12 signaling directly augments proproliferative cytokine stimulation of NK cells during viral infections. J. Immunol. 177:4981–4990 [DOI] [PubMed] [Google Scholar]
- 17. Hayakawa Y, Smyth MJ. 2006. CD27 dissects mature NK cells into two subsets with distinct responsiveness and migratory capacity. J. Immunol. 176:1517–1524 [DOI] [PubMed] [Google Scholar]
- 18. Huber JP, David Farrar J. 2011. Regulation of effector and memory T-cell functions by type I interferon. Immunology 132:466–474 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Jonjic S, Babic M, Polic B, Krmpotic A. 2008. Immune evasion of natural killer cells by viruses. Curr. Opin. Immunol. 20:30–38 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Jonjic S, Bubic I, Krmpotic A. 2006. Innate immunity to cytomegaloviruses, p 285–319 In Reddehase MJ. (ed), Cytomegaloviruses: molecular biology and immunology. Caister Academic Press, Wymondham, Norfolk, United Kingdom [Google Scholar]
- 21. Jonjic S, Krmpotic A, Arapovic J, Koszinowski UH. 2008. Dissection of the antiviral NK cell response by MCMV mutants. Methods Mol. Biol. 415:127–149 [DOI] [PubMed] [Google Scholar]
- 22. Jonjic S, Mutter W, Weiland F, Reddehase MJ, Koszinowski UH. 1989. Site-restricted persistent cytomegalovirus infection after selective long-term depletion of CD4+ T lymphocytes. J. Exp. Med. 169:1199–1212 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Koo GC, Peppard JR. 1984. Establishment of monoclonal anti-Nk-1.1 antibody. Hybridoma 3:301–303 [DOI] [PubMed] [Google Scholar]
- 24. Krmpotic A, et al. 1999. The immunoevasive function encoded by the mouse cytomegalovirus gene m152 protects the virus against T cell control in vivo. J. Exp. Med. 190:1285–1296 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Krug A, et al. 2004. TLR9-dependent recognition of MCMV by IPC and DC generates coordinated cytokine responses that activate antiviral NK cell function. Immunity 21:107–119 [DOI] [PubMed] [Google Scholar]
- 26. Lee SH, Kim KS, Fodil-Cornu N, Vidal SM, Biron CA. 2009. Activating receptors promote NK cell expansion for maintenance, IL-10 production, and CD8 T cell regulation during viral infection. J. Exp. Med. 206:2235–2251 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Lemmermann NAW, et al. 2010. CD8 T-cell immunotherapy of cytomegalovirus disease in the murine model. Methods Microbiol. 37:369–420 [Google Scholar]
- 28. Lemmermann NA, Bohm V, Holtappels R, Reddehase MJ. 2011. In vivo impact of cytomegalovirus evasion of CD8 T-cell immunity: facts and thoughts based on murine models. Virus Res. 157:161–174 [DOI] [PubMed] [Google Scholar]
- 29. Lenac T, et al. 2006. The herpesviral Fc receptor fcr-1 down-regulates the NKG2D ligands MULT-1 and H60. J. Exp. Med. 203:1843–1850 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Lisnic VJ, Krmpotic A, Jonjic S. 2010. Modulation of natural killer cell activity by viruses. Curr. Opin. Microbiol. 13:530–539 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Lodoen M, et al. 2003. NKG2D-mediated natural killer cell protection against cytomegalovirus is impaired by viral gp40 modulation of retinoic acid early inducible 1 gene molecules. J. Exp. Med. 197:1245–1253 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Markiewicz MA, et al. 2005. Costimulation through NKG2D enhances murine CD8+ CTL function: similarities and differences between NKG2D and CD28 costimulation. J. Immunol. 175:2825–2833 [DOI] [PubMed] [Google Scholar]
- 33. Martin-Fontecha A, et al. 2004. Induced recruitment of NK cells to lymph nodes provides IFN-gamma for T(H)1 priming. Nat. Immunol. 5:1260–1265 [DOI] [PubMed] [Google Scholar]
- 34. Mathys S, et al. 2003. Dendritic cells under influence of mouse cytomegalovirus have a physiologic dual role: to initiate and to restrict T cell activation. J. Infect. Dis. 187:988–999 [DOI] [PubMed] [Google Scholar]
- 35. Munks MW, et al. 2006. Genome-wide analysis reveals a highly diverse CD8 T cell response to murine cytomegalovirus. J. Immunol. 176:3760–3766 [DOI] [PubMed] [Google Scholar]
- 36. Orange JS, Biron CA. 1996. An absolute and restricted requirement for IL-12 in natural killer cell IFN-gamma production and antiviral defense. Studies of natural killer and T cell responses in contrasting viral infections. J. Immunol. 156:1138–1142 [PubMed] [Google Scholar]
- 37. Orange JS, Biron CA. 1996. Characterization of early IL-12, IFN-alphabeta, and TNF effects on antiviral state and NK cell responses during murine cytomegalovirus infection. J. Immunol. 156:4746–4756 [PubMed] [Google Scholar]
- 38. Pyzik M, Kielczewska A, Vidal SM. 2008. NK cell receptors and their MHC class I ligands in host response to cytomegalovirus: insights from the mouse genome. Semin. Immunol. 20:331–342 [DOI] [PubMed] [Google Scholar]
- 39. Raulet DH. 2004. Interplay of natural killer cells and their receptors with the adaptive immune response. Nat. Immunol. 5:996–1002 [DOI] [PubMed] [Google Scholar]
- 40. Robbins SH, et al. 2007. Natural killer cells promote early CD8 T cell responses against cytomegalovirus. PLoS Pathog. 3:1152–1164 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Saraiva M, O'Garra A. 2010. The regulation of IL-10 production by immune cells. Nat. Rev. Immunol. 10:170–181 [DOI] [PubMed] [Google Scholar]
- 42. Scalzo AA, Corbett AJ, Rawlinson WD, Scott GM, Degli-Esposti MA. 2007. The interplay between host and viral factors in shaping the outcome of cytomegalovirus infection. Immunol. Cell Biol. 85:46–54 [DOI] [PubMed] [Google Scholar]
- 43. Scalzo AA, Yokoyama WM. 2008. Cmv1 and natural killer cell responses to murine cytomegalovirus infection. Curr. Top. Microbiol. Immunol. 321:101–122 [DOI] [PubMed] [Google Scholar]
- 44. Simon CO, et al. 2006. CD8 T cells control cytomegalovirus latency by epitope-specific sensing of transcriptional reactivation. J. Virol. 80:10436–10456 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Slavuljica I, et al. 2010. Recombinant mouse cytomegalovirus expressing a ligand for the NKG2D receptor is attenuated and has improved vaccine properties. J. Clin. Invest. 120:4532–4545 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Smith HR, et al. 2002. Recognition of a virus-encoded ligand by a natural killer cell activation receptor. Proc. Natl. Acad. Sci. U. S. A. 99:8826–8831 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Stadnisky MD, Xie X, Coats ER, Bullock TN, Brown MG. 2011. Self MHC class I-licensed NK cells enhance adaptive CD8 T-cell viral immunity. Blood 117:5133–5141 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Strobl B, et al. 2005. Novel functions of tyrosine kinase 2 in the antiviral defense against murine cytomegalovirus. J. Immunol. 175:4000–4008 [DOI] [PubMed] [Google Scholar]
- 49. Sumaria N, et al. 2009. The roles of interferon-gamma and perforin in antiviral immunity in mice that differ in genetically determined NK-cell-mediated antiviral activity. Immunol. Cell Biol. 87:559–566 [DOI] [PubMed] [Google Scholar]
- 50. Vivier E, et al. 2011. Innate or adaptive immunity? The example of natural killer cells. Science 331:44–49 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Wagner M, Jonjic S, Koszinowski UH, Messerle M. 1999. Systematic excision of vector sequences from the BAC-cloned herpesvirus genome during virus reconstitution. J. Virol. 73:7056–7060 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Walzer T, Dalod M, Robbins SH, Zitvogel L, Vivier E. 2005. Natural-killer cells and dendritic cells: “l'union fait la force.” Blood 106:2252–2258 [DOI] [PubMed] [Google Scholar]
- 53. Wills MR, Carmichael AJ, Sissons PJG. 2006. Adaptive cellular immunity to human cytomegalovirus, p 341–365 In Reddehase MJ. (ed), Cytomegaloviruses: molecular biology and immunology. Caister Academic Press, Wymondham, Norfolk, United Kingdom [Google Scholar]
- 54. Yokoyama WM, Kim S. 2008. Analysis of individual natural killer cell responses. Methods Mol. Biol. 415:179–196 [DOI] [PubMed] [Google Scholar]
- 55. Ziegler H, et al. 1997. A mouse cytomegalovirus glycoprotein retains MHC class I complexes in the ERGIC/cis-Golgi compartments. Immunity 6:57–66 [DOI] [PubMed] [Google Scholar]
- 56. Zitvogel L, Terme M, Borg C, Trinchieri G. 2006. Dendritic cell-NK cell cross-talk: regulation and physiopathology. Curr. Top. Microbiol. Immunol. 298:157–174 [DOI] [PubMed] [Google Scholar]
- 57. Zucchini N, et al. 2008. Individual plasmacytoid dendritic cells are major contributors to the production of multiple innate cytokines in an organ-specific manner during viral infection. Int. Immunol. 20:45–56 [DOI] [PMC free article] [PubMed] [Google Scholar]