Abstract
Intestinal barrier breakdown following traumatic brain injury (TBI) is characterized by increased intestinal permeability, leading to bacterial translocation, and inflammation. The hormone ghrelin may prevent intestinal injury and have anti-inflammatory properties. We hypothesized that exogenous ghrelin prevents intestinal injury following TBI. A weight-drop model created severe TBI in three groups of anesthetized Balb/c mice. Group TBI: animals underwent TBI only; Group TBI/ghrelin: animals were given 10 μg of ghrelin intraperitoneally prior and 1 h following TBI; Group sham: no TBI or ghrelin injection. Intestinal permeability was measured 6 h following TBI by detecting serum levels of FITC-Dextran after injection into the intact ileum. The terminal ileum was harvested for histology, expression of the tight junction protein MLCK and inflammatory cytokine TNF-α. Permeability increased in the TBI group compared to the sham group (109.7 ± 21.8 μg/mL vs. 32.2 ± 10.1 μg/mL; p < 0.002). Ghrelin prevented TBI-induced permeability (28.3 ± 4.2 μg/mL vs. 109.7 ± 21.8 μg/mL; p < 0.001). The intestines of the TBI group showed blunting and necrosis of villi compared to the sham group, while ghrelin injection preserved intestinal architecture. Intestinal MLCK increased 73% compared to the sham group (p < 0.03). Ghrelin prevented TBI-induced MLCK expression to sham levels. Intestinal TNF-α increased following TBI compared to the sham group (46.2 ± 7.1 pg/mL vs. 24.4 ± 2.2 pg/mL p < 0.001). Ghrelin reduced TNF-α to sham levels (29.2 ± 5.0 pg/mL; p = NS). We therefore conclude that ghrelin prevents TBI-induced injury, as determined by intestinal permeability, histology, and intestinal levels of TNF-α. The mechanism for ghrelin mediating intestinal protection is likely multifactorial, and further studies are needed to delineate these possibilities.
Key words: ghrelin, intestinal permeability, tight junctions, traumatic brain injury
Introduction
Non-neurologic physiologic alterations are known to occur following traumatic brain injury (TBI), including systemic inflammation, autonomic dysfunction, and end organ dysfunction, primarily involving the cardiovascular, respiratory, and gastrointestinal (GI) systems (Baguley et al., 2008; Kao et al., 1998). Patients manifesting post-TBI GI dysfunction have increased morbidity and longer periods of hospitalization; therefore, treatment modalities targeting prevention of GI dysfunction have important clinical implications (Cook et al., 2008; Kemp et al., 2008). Previously, we, along with others, have shown, in a mouse model, that TBI causes GI dysfunction characterized by increased intestinal permeability and marked mucosal injury, which was confirmed by intestinal histology (Bansal et al., 2009; Feighery et al., 2008; Hang et al., 2003). Furthermore, expression of the intestinal tight junction proteins, ZO-1 and occludin, decrease following TBI, which may explain, at least in part, the observed increase in intestinal permeability (Bansal et al., 2009).
The orexigenic hormone ghrelin, described in 1999, has been shown to have several anti-inflammatory properties in addition to its known effect on pituitary regulation, hunger, and satiety (Kojima et al., 1999; Waseem et al., 2008). Wu and colleagues (2007) have shown that exogenous ghrelin decreases levels of the pro-inflammatory cytokines IL-1 and IL-6 following sepsis. In another study, exogenous ghrelin has also been shown to ameliorate intestinal barrier dysfunction induced in a murine sepsis model (Wu et al., 2009). The ghrelin receptor (GHR) has been localized to the dorsal motor nucleus of the vagus (DMNV), hypothalamus, midbrain, and brainstem of the rat, indicating that ghrelin may regulate autonomic function such as cardiopulmonary and GI homeostasis (Qiu et al., 2008; van der Lely, 2009). The effects of ghrelin on GI dysfunction following TBI have never been reported. We hypothesize that exogenous ghrelin prevents intestinal injury and inflammation, reduces intestinal permeability, and restores tight junction protein alterations following TBI.
Methods
Mouse TBI model
Animal experiments, including anesthesia, TBI, and recuperation, were approved through the university Institutional Animal Care and Use Committee (Approval #S08110). Male Balb/c mice (20–24 g) were obtained commercially (The Jackson Laboratory, Sacramento, CA) and placed under a 12-h light/dark cycle. A weight-drop TBI model, as previously described, was used to cause a right-sided cerebral contusion (Bansal et al., 2009; Stahel et al., 2000). Briefly, animals were anesthetized with 3% inhaled isoflurane. Each animal (n = 6–8 per group) was manually secured and its head shaved with an electric clipper, after which a vertical incision was made over the cranium and, using a surgical drill, a burr hole, 4 mm in diameter, 1 mm lateral and 1 mm posterior to the bregma, was created to expose the dura mater. A 250-g metal rod was dropped from a height of 2 cm onto the exposed dura mater. The incision was closed with vet bond, and buprenorphine (100 μL) was injected subcutaneously for analgesia in all animal groups. Food and water were provided ad libitum.
Ghrelin administration
Animals in the ghrelin group (n = 8) received two doses of ghrelin (10 μg) intraperitoneally (i.p.) immediately prior to and 1 h after TBI. The dosing and timing of ghrelin administration was determined from previous experiments showing that i.p. ghrelin has a rapid onset of action and its response is potentiated by a second i.p. injection within 4 h (Qiu et al., 2008; Wren et al., 2000).
Histological evaluation
Segments of terminal ileum previously stored in formalin were embedded in paraffin blocks. Sections were cut, placed onto glass slides, and stained with hematoxylin-eosin (Thermo Scientific, Portsmouth, NH). Images were later obtained using Q-imaging software and an Olympus IX70 light microscope at varying magnifications. A pathologist, blinded to the groups, examined each terminal ileum specimen and scored each specimen using a modified histopathologic score by Cuzzocrea and colleagues (2002). A scale of 0–3 was used to assess intestinal damage: 0 = normal, no damage; 1 = mild, focal epithelial edema and necrosis; 2 = moderate, diffuse swelling or necrosis of the villi; 3 = severe, diffuse necrosis of the villi, with evidence of neutrophil infiltration in the submucosa/hemorrhage.
In vivo intestinal permeability assay
Animals underwent an in vivo intestinal permeability assay at 6 h following TBI according to methods previously described (Bansal et al., 2009; Costantini et al., 2008). At 6 h following TBI ± ghrelin or sham operation, animals were anesthetized by inhaled isoflurane. A midline laparotomy was performed, the cecum was located, and a 5-cm segment of terminal ileum was isolated between silk ties. Previously prepared FITC-Dextran (25 mg 4.4 kDa FITC-Dextran in 200 μL PBS) was injected into the lumen of the isolated ileum. The eviscerated intestine was returned into the abdominal cavity, and the abdominal wall was closed with 3-0 silk stitches. At 30 min following the injection, blood was collected by cardiac puncture. Blood samples were placed into heparinized Eppendorf® tubes and centrifuged at 10,000g for 10 min. Plasma was removed and subsequently assayed using a SpectraMax M5 fluorescence spectrophotometer (Molecular Devices, Sunnyvale, CA) to determine the concentration of FITC-Dextran. A standard curve for the assay was obtained through the serial dilution of FITC-Dextran in mouse serum.
Immunoblotting
Samples of terminal ileum in the sham group or 6 h following TBI ± ghrelin were homogenized in a 500-μL solution containing ice-cold tissue protein extraction reagent (TPER) with 1% protease inhibitor and 1% phosphatase inhibitor (Pierce Biotechnology, Rockford, IL). The homogenates were centrifuged at 10,000g for 5 min, and the supernatant was collected. The protein concentration of each sample was determined using the BCA assay kit (Pierce Biotechnology) and Western blots were performed by separating proteins with SDS-polyacrylamide gel electrophoresis using 8–16% Tris-Glycine polyacrylamide gels (Invitrogen, Carlsbad, CA). Proteins were transferred onto nitrocellulose membranes (Invitrogen) and blocked with 5% bovine serum albumin (BSA) in Tris-buffered saline (TBS)/Tween 20. Membranes were incubated in primary anti-mouse antibody prepared in 5% BSA (1:500) for MLCK (Sigma-Aldrich, St. Louis, MO). Membranes were incubated with secondary antibody, horseradish peroxidase-linked anti-mouse IgG (1:2000; Cell Signaling, Danvers, MA) prepared in 5% BSA blocking solution. The selection and concentration of antibodies were optimized from previous studies (Costantini et al., 2008; Costantini, Loomis, et al., 2009). Membranes were incubated with Pierce Supersignal West Pico Chemiluminescent Kit for 3 min prior to detection of luminescence using the Xenogen IVIS Lumina imaging system (Caliper Life Sciences, Mountain View, CA). The mean pixel density of each gel was estimated using UN-SCAN-IT Gel Digitizing software (Silk Scientific, Orem, UT). The relative band density of each band was calculated by dividing the pixel density by the mean pixel density of the sham samples.
Measuring intestinal TNF-α
Protein was extracted from the terminal ileum by homogenizing tissue in 500 μL of ice-cold tissue protein extraction reagent (T-PER) containing 1% protease inhibitor and 1% phosphatase inhibitor (Pierce Biotechnology). Homogenates were centrifuged at 10,000g for 5 min. The supernatant was obtained and stored at −70°C. Intestinal TNF-α were measured in the sham group and at 2, 4, and 6 h following TBI ± ghrelin administration using a commercially available ELISA assay (R&D system, Minneapolis, MN). Values are reported as pg/mL.
Statistical analysis
Values are expressed as mean ± standard error of the mean (SEM). The statistical significance among groups was determined by a two-tailed t test, and a p value < 0.05 was considered statistically significant. Comparison of intestinal histopathologic scores and Western blot band densities were determined by the Mann–Whitney U test.
Results
Histopathologic evaluation
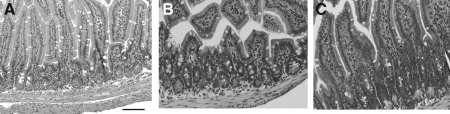
The terminal ileum was harvested 6 h following sham, TBI, and TBI + ghrelin for histologic analysis using H&E staining. Sham animals had normal-appearing villi with consistent villous height, whereas intestinal specimens from TBI animals were notable for marked blunting of intestinal villi with architectural deformity, villi necrosis, and the presence of neutrophils at the base of the villi (Fig. 1). Ghrelin-treated TBI animals did not have significant evidence of intestinal injury nor architectural deformity, with an appearance consistent to that of the sham group. The mean intestinal injury was markedly increased in the TBI group (2.8 ± 0.4) when compared to the TBI + ghrelin group (1.3 ± 0.2; p = 0.01) and the sham group (1.0 ± 0.0; p = 0.001).
FIG. 1.
Terminal ileum H&E staining (all images are 60 × magnification, bar = 100 μm). (A) Sham terminal ileum showing normal villi and consistent villous height (mean injury = 1.0 ± 0.0). (B) TBI terminal ileum specimens were notable for marked intestinal villi blunting, villi necrosis, and neutrophil infiltration (mean injury = 2.8 ± 0.4). (C) Ghrelin-injected TBI animals showed no evidence of intestinal necrosis nor architectural deformity, with an appearance consistent to that of the sham group (mean injury = 1.3 ± 0.2; p = 0.01).
Intestinal permeability
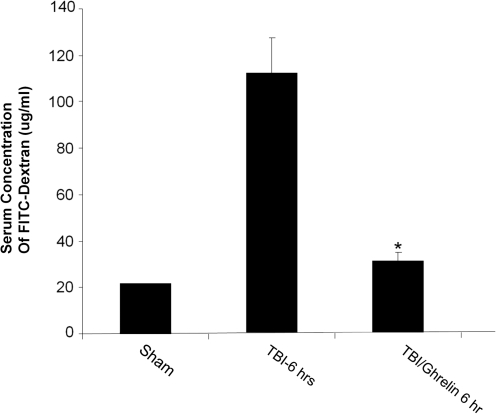
In vivo intestinal permeability was determined 6 h following sham, TBI, or TBI + ghrelin by spectrophotometric measurement of plasma following intraluminal injection of 4.4 kDa FITC-Dextran (Fig. 2). Serum FITC-Dextran was markedly increased in TBI compared to the sham group (109.7 ± 21.8 μg/mL vs. 22.2 ± 0.3 μg/mL; p < 0.002). Ghrelin prevented TBI-induced intestinal permeability (28.3 ± 4.2 μg/mL vs. 109.7 ± 21.8 μg/mL; p < 0.001).
FIG. 2.
In vivo intestinal permeability determined by intraluminal injection of 4.4 kDa FITC-Dextran and plasma spectrophotometric detection 6 h after injury (n = 6 in each group). Intestinal permeability was increased in the TBI group compared to the sham group (109.7 ± 21.8 μg/mL vs. 22.2 ± 0.3 μg/mL; p < 0.002). Ghrelin prevented TBI-induced intestinal permeability to that of sham levels (28.3 ± 4.2 μg/mL vs. 109.7 ± 21.8 μg/mL; *p < 0.001).
MLCK expression
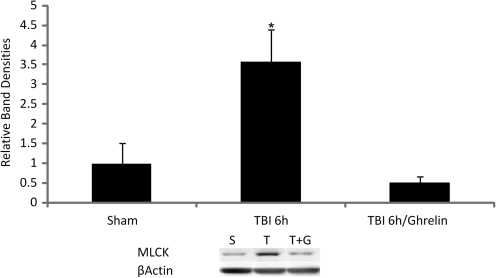
Intestinal MLCK was measured 6 h following sham, TBI, or TBI + ghrelin by Western immunoblotting. At 6 h following TBI, intestinal MLCK increased by 73% when compared to sham animals (p < 0.03). Ghrelin administration prevented TBI-induced MLCK expression to levels of the sham group. Graphical representation of relative band densities and representative Western blots are shown in Figure 3.
FIG. 3.
Intestinal MLCK expression was measured by Western immunoblotting and quantified by relative band densities (n = 8 in each group). Following TBI, intestinal MLCK increased by 73% when compared to sham animals (*p < 0.03). Ghrelin administration prevented TBI-induced MLCK expression to levels of the sham group. A representative Western blot shows relative MLCK expression accordingly (S = sham, T = TBI, T + G = TBI + ghrelin).
Levels of intestinal TNF-α
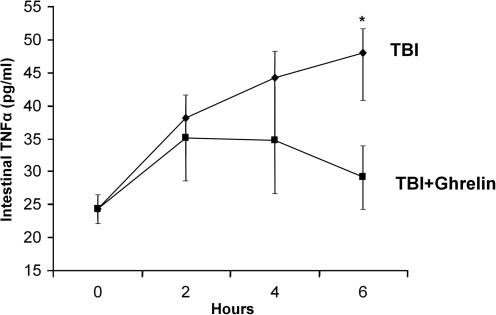
In order to quantify intestinal inflammation, levels of the cytokine TNF-α were measured by ELISA in intestinal extracts at 2, 4, and 6 h following sham and TBI ± ghrelin (Fig. 4). Compared to the sham group (24.4 ± 2.2 pg/mL), animals undergoing TBI had a consistent increase in intestinal TNF-α at each time point (2 h: 38.1 ± 4.3 pg/mL; 4 h: 42.7 ± 1.6 pg/mL; 6 h: 46.2 ± 7.1 pg/mL; p < 0.001). For animals receiving i.p. ghrelin following TBI, intestinal TNF-α initially increased at 2 h (35.1 ± 6.6 pg/mL) and 4 h (34.8 ± 8.2 pg/mL), but levels were restored to that of the sham group at 6 h (29.2 ± 5.0 pg/mL; p = NS).
FIG. 4.
TNF-α as measured by ELISA in intestinal extracts at 2-, 4-, and 6-h intervals following injury (n = 6 in each group). Intestinal TNF-α increased in TBI animals at each hourly interval (2 h: 38.1 ± 4.3 pg/mL; 4 h: 42.7 ± 1.6 pg/mL; 6 h: 46.2 ± 7.1 pg/mL; *p < 0.001). For animals receiving i.p. ghrelin following TBI, intestinal TNF-α initially increased at 2 h (35.1 ± 6.6 pg/mL) and 4 h (34.8 ± 8.2 pg/mL), but levels were restored to that of the sham group at 6 h (29.2 ± 5.0 pg/mL; p = NS).
Discussion
Ghrelin, a 28 amino-acid peptide, was originally purified from the rat stomach in 1999 and was subsequently found in humans (Kojima et al., 1999). Initially, the function of ghrelin was thought to be limited only as an endocrine mediator, initiating and regulating hunger and food intake. This led several investigators to postulate that antagonizing the GR would promote weight loss and control obesity by hunger suppression (Gura, 2003). Later research indicated that ghrelin has an important role in pituitary hormone regulation and GI homeostasis. Given the widespread anatomic and cellular distribution of the GR, a variety of endocrine and paracrine functions likely exist (Wu et al., 2009). For example, ghrelin has been shown to improve intestinal motility and gastric emptying following abdominal operations and large body surface area burns (Sallam et al., 2007). More recently, ghrelin has been shown to have potent anti-inflammatory properties. In a study by Wu and colleagues (2009), intravenous ghrelin injections in septic rats decreased intestinal permeability and bacterial intestinal translocation. The authors also demonstrated that High-mobility group box 1 (HMGB-1), a potent mediator of sepsis lethality, decreased in ghrelin-treated animals. To our knowledge, the specific systemic and GI effects of ghrelin following TBI have not been previously studied. Therefore, it was our objective to test whether exogenous ghrelin administration would prevent TBI-induced GI dysfunction.
TBI and torso trauma cause significant alterations and impairment to the GI system, illustrated by Cushing's ulceration, intestinal paralysis (ileus), and the breakdown of intestinal epithelium and cellular tight junction integrity (Lenz et al., 2007). The molecular basis explaining increased gut permeability and alteration in intestinal architecture following TBI is now being investigated. Intestinal tight junctions form the barrier between intestinal epithelial cells, which protects neighboring tissue from the bacterial and toxic contents located within the intestinal lumen (Schneeberger and Lynch, 2004). Tight junction proteins regulate the permeability of an intact intestinal barrier. We have previously reported that TBI decreases the expression of the tight junction proteins, ZO-1 and occludin (Bansal et al., 2009). In this study, we chose to analyze the expression of MLCK, since injury to the intestinal epithelial cell leads to the activation of MLCK, a key upstream regulator of tight junction permeability. Activation and increased expression of MLCK leads to cytoskeletal contraction of peri-junctional actin, which consequently leads to increased intestinal permeability. Previous investigators have reported that TNF-α increases transcription and activation of MLCK, in cultured intestinal epithelial cells, with a similar increase in magnitude as detailed in our study (Graham et al., 2006). Using a severe burn model, Costantini, Loomis, and colleagues (2009) and Costantini, Peterson, and colleagues (2009) have shown that modulating inflammation by the drug pentoxifylline, which has been shown to decrease TNF-α, attenuates MLCK expression and prevents burn-induced intestinal permeability. The authors also showed that decreased MLCK expression correlated with a decrease in the inflammatory cytokine IL-6. Systemic inflammation, measured by an increase in inflammatory cytokines, may play a significant role in physiologic homeostasis. Pro-inflammatory cytokines (TNF α, IL-1, IL-6, IL-12) are increased, often immediately following either TBI, burns, or severe torso trauma (Hang et al., 2005; Kemp et al., 2008; Lenz et al., 2007). Unlike other studies that examine TBI-induced intestinal and inflammatory changes at later time points, we studied the early effects of TBI on intestinal injury. For this reason, TNF-α is the ideal inflammatory cytokine, since it rises acutely following TBI and tends to plateau at 6 h (Bansal et al., 2010). This increased inflammatory milieu may manifest as the set of physiologic changes coined the systemic inflammatory response (SIRS), which can lead to micro-thrombosis, decreased tissue perfusion, tissue injury, and eventually sepsis and death (Swank and Deitch, 1996). Our current investigative efforts focus on how TBI acutely disrupts the neuroenteric axis, rapidly leading to intestinal barrier dysfunction. Future studies are being designed to elucidate TBI-induced brain edema and inflammation, and how specific changes may lead to gastrointestinal dysfunction. The intestine is important in the cycle of organ hypoperfusion, and in particular may serve as a “window” into global body homeostasis. In an analysis of post-TBI intestines, Hang and colleagues (2005) have shown the importance of a localized inflammatory response mediated by an increase in nuclear factor kappa B activation, and intestinal concentrations of TNF-α and IL-6. We have also shown an increase in intestinal TNF-α within hours following TBI (Bansal et al., 2010). Experiments targeted to decrease post-TBI inflammation have been shown to prevent intestinal injury. Recently, we have demonstrated that electric vagal stimulation can decrease TNF-α following TBI, similar to the decrease in sepsis first demonstrated by Tracey and colleagues (2007). Similarly, other investigators have demonstrated that exogenous glutamine and progesterone, administered following TBI, decrease IL-1, TNF-α, and IL-6 levels in both intestinal and brain tissue (Chen, Shi, Jin, et al., 2008; Chen, Shi, Qi, et al., 2008). Since ghrelin has been shown to have anti-inflammatory properties and ghrelin mediates intestinal homeostasis, it was our hypothesis that ghrelin would protect against GI dysfunction caused by TBI.
In this study, we report that exogenous ghrelin prevents intestinal injury, as determined by histology, conserves intestinal barrier function, as measured by preserving intestinal permeability and decreased MLCK expression, and decreases local inflammation, as evidenced by decreased intestinal TNF-α. These findings are consistent with other models of intestinal injury. In a mouse model of colitis, Gonzalez-Rey and colleagues (2006) have shown that i.p. ghrelin restored intestinal epithelium, improved survival, and increased food intake and mouse body weight. The mechanism behind these observations may be explained by either ghrelin's endocrine effects on the central nervous system or by the direct anti-inflammatory effects via GRs located on resident monocytes and T-cells of either intestinal tissue or circulating in the blood. In a rat sepsis model, Wu and colleagues (2007) reported that exogenous ghrelin decreased inflammatory cytokines likely by centrally stimulating the parasympathetic nervous system. Consequently, vagotomized animals did not have a ghrelin mediated decrease in TNF-α or IL-6 following sepsis. Similarly, Zhang and colleagues (2004) have reported that ghrelin administration increases neurogenesis and neuron proliferation of the DMNV, suggesting that ghrelin may have potent regulatory effects on DMNV function. Ammori and colleagues (2008) have shown that chemically induced colitis decreased DMNV proliferation and that exogenous ghrelin administration ameliorates these effects. If ghrelin effectively promotes neuron proliferation following experimentally induced intestinal inflammation, it is interesting to demonstrate conversely that ghrelin prevents intestinal dysfunction following TBI. Under the neuroenteric axis paradigm, communication between the brain and the gut (or vice versa) may be modulated by the enteric nervous system, or chemical signaling through neuroendocrine hormones such as ghrelin (Camilleri and Chang, 2008). Ghrelin readily passes through the blood–brain barrier, and GRs have been localized throughout brain tissue, including the hypothalamus, substantia nigra, pituitary, and other regions (Chung et al., 2007). Miao and colleagues (2007) have shown that exogenous ghrelin has a neuroprotective effect following cerebral ischemia and reperfusion, decreasing neouronal apoptosis and reducing total infarct volume after the ischemic insult. Similar to our study, Ersahin and colleagues (2010) have shown that intraperitoneally injected ghrelin reduced serum TNF-α and IL-1, brain edema, and cerebral myeloperoxidase activity following subarrachnoid hemorrhage in a rat. We recognize that our current study design administers ghrelin both preceding and post TBI, thus mitigating the potential relevance as a pharmacologic treatment. However, the preexisting endocrine profile of a patient (or experimental animal model for that matter) before physiologic stress likely plays an important role in outcomes. Recently, Koch and colleagues (2010) demonstrated that higher endogenous ghrelin levels correlated with increased survival in critically ill patients. This is in addition to several publications showing the importance of endocrine and metabolic profiles as a prognosticator of outcomes following trauma or sepsis (Grimble, 2010). Given these studies, it is plausible that ghrelin may be neuroprotective following TBI, which consequently prevents intestinal dysfunction through a preserved neuroenteric axis. Future studies including the use of ghrelin receptor knockout mice, ghrelin receptor antagonists, and perhaps abdominal vagotomy preceding TBI and ghrelin administration need to be conducted to delineate this intriguing possibility.
In summary, we have observed that exogenous ghrelin prevents TBI-induced intestinal permeability, intestinal injury, and decreased intestinal inflammation. The mechanism for ghrelin mediating intestinal protection after TBI is complex and underscores the need for further research.
Acknowledgments
This research was supported in part by the 2009 Inje Research and Scholarship Foundation.
Author Disclosure Statement
No competing financial interests exist.
References
- Ammori J.B. Zhang W.Z. Li J.Y. Chai B.X. Mulholland M.W. Effects of ghrelin on neuronal survival in cells derived from dorsal motor nucleus of the vagus. Surgery. 2008;144:159–167. doi: 10.1016/j.surg.2008.03.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baguley I.J. Heriseanu R.E. Cameron I.D. Nott M.T. Slewa-Younan S. A critical review of the pathophysiology of dysautonomia following traumatic brain injury. Neurocrit. Care. 2008;8:293–300. doi: 10.1007/s12028-007-9021-3. [DOI] [PubMed] [Google Scholar]
- Bansal V. Costantini T. Kroll L. Peterson C. Loomis W. Eliceiri B. Baird A. Wolf P. Coimbra R. Traumatic brain injury and intestinal dysfunction: Uncovering the neuro-enteric axis. J. Neurotrauma. 2009;26:1353–1359. doi: 10.1089/neu.2008.0858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bansal V. Costantini T. Ryu S.Y. Peterson C. Loomis W. Putnam J. Elicieri B. Baird A. Coimbra R. Stimulating the central nervous system to prevent intestinal dysfunction after traumatic brain injury. J. Trauma. 2010;68:1059–1064. doi: 10.1097/TA.0b013e3181d87373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Camilleri M. Chang L. Challenges to the therapeutic pipeline for irritable bowel syndrome: end points and regulatory hurdles. Gastroenterology. 2008;135:1877–1891. doi: 10.1053/j.gastro.2008.09.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen G. Shi J. Jin W. Wang L. Xie W. Sun J. Hang C. Progesterone administration modulates TLRs/NF-kappaB signaling pathway in rat brain after cortical contusion. Ann. Clin. Lab. Sci. 2008;38:65–74. [PubMed] [Google Scholar]
- Chen G. Shi J. Qi M. Yin H. Hang C. Glutamine decreases intestinal nuclear factor kappa B activity and pro-inflammatory cytokine expression after traumatic brain injury in rats. Inflamm. Res. 2008;57:57–64. doi: 10.1007/s00011-007-7101-7. [DOI] [PubMed] [Google Scholar]
- Chung H. Kim E. Lee D.H. Seo S. Ju S. Lee D. Kim H. Park S. Ghrelin inhibits apoptosis in hypothalamic neuronal cells during oxygen-glucose deprivation. Endocrinology. 2007;148:148–159. doi: 10.1210/en.2006-0991. [DOI] [PubMed] [Google Scholar]
- Cook A.M. Peppard A. Magnuson B. Nutrition considerations in traumatic brain injury. Nutr. Clin. Pract. 2008;23:608–620. doi: 10.1177/0884533608326060. [DOI] [PubMed] [Google Scholar]
- Costantini T.W. Loomis W.H. Putnam J.G. Drusinsky D. Deree J. Choi S. Wolf P. Baird A. Eliceiri B. Bansal V. Coimbra R. Burn-induced gut barrier injury is attenuated by phosphodiesterase inhibition: effects on tight junction structural proteins. Shock. 2008;4:416–422. doi: 10.1097/SHK.0b013e3181863080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Costantini T.W. Loomis W.H. Putnam J.G. Kroll L. Eliceiri B.P. Baird A. Bansal V. Coimbra R. Pentoxifylline modulates intestinal tight junction signaling after burn injury: effects on myosin light chain kinase. J. Trauma. 2009;66:17–24. doi: 10.1097/TA.0b013e318191bb1f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Costantini T.W. Peterson C.Y. Kroll L. Loomis W.H. Putnam J.G. Wolf P. Eliceiri B.P. Baird A. Bansal V. Coimbra R. Burns, inflammation, and intestinal injury: protective effects of an anti-inflammatory resuscitation strategy. J. Trauma. 2009;67:1162–1168. doi: 10.1097/TA.0b013e3181ba3577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cuzzocrea S. Chatterjee P.K. Mazzon E. Dugo L. De Sarro A. Van de Loo F.A. Caputi A.P. Thiemermann C. Role of induced nitric oxide in the initiation of the inflammatory response after postischemic injury. Shock. 2002;18:169–176. doi: 10.1097/00024382-200208000-00014. [DOI] [PubMed] [Google Scholar]
- Ersahin M. Toklu H.Z. Erzik C. Cetinel S. Bangir D. Ogunc A.V. Tetik S. Ozdemir Z.N. Sener G. Yegen B.C. The anti-inflammatory and neuroprotective effects of ghrelin in subarachnoid hemorrhage-induced oxidative brain damage in rats. J. Neurotrauma. 2010;27:1142–1155. doi: 10.1089/neu.2009.1210. [DOI] [PubMed] [Google Scholar]
- Feighery L. Smyth A. Keely S. Baird A.W. O'Connor W.T. Callanan J.J. Brayden D.J. Increased intestinal permeability in rats subjected to traumatic frontal lobe percussion brain injury. J. Trauma. 2008;64:131–137. doi: 10.1097/TA.0b013e3181568d9f. [DOI] [PubMed] [Google Scholar]
- Gonzalez-Rey E. Chorny A. Delgado M. Therapeutic action of ghrelin in a mouse model of colitis. Gastroenterology. 2006;130:1707–1720. doi: 10.1053/j.gastro.2006.01.041. [DOI] [PubMed] [Google Scholar]
- Graham W.V. Wang F. Clayburgh D.R. Cheng J.X. Yoon B. Wang Y. Lin A. Turner J.R. Tumor necrosis factor-induced long myosin light chain kinase transcription is regulated by differentiation-dependent signaling events. Characterization of the human long myosin light chain kinase promoter. J. Biol. Chem. 2006;281:26205–26215. doi: 10.1074/jbc.M602164200. [DOI] [PubMed] [Google Scholar]
- Grimble R.F. The true cost of in-patient obesity: impact of obesity on inflammatory stress and morbidity. Proc. Nutr. Soc. 2010:1–7. doi: 10.1017/S0029665110001709. (epub ahead of print). [DOI] [PubMed] [Google Scholar]
- Gura T. Obesity drug pipeline not so fat. Science. 2003;299:849–852. doi: 10.1126/science.299.5608.849. [DOI] [PubMed] [Google Scholar]
- Hang C.H. Shi J.X. Li J.S. Li W.Q. Wu W. Expressions of intestinal NF-kappaB, TNF-alpha, and IL-6 following traumatic brain injury in rats. J. Surg. Res. 2005;123:188–193. doi: 10.1016/j.jss.2004.08.002. [DOI] [PubMed] [Google Scholar]
- Hang C.H. Shi J.X. Li J.S. Wu W. Yin H.X. Alterations of intestinal mucosa structure and barrier function following traumatic brain injury in rats. World J. Gastroenterol. 2003;9:2776–2781. doi: 10.3748/wjg.v9.i12.2776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kao C.H. ChangLai S.P. Chieng P.U. Yen T.C. Gastric emptying in head-injured patients. Am. J. Gastroenterol. 1998;93:1108–1112. doi: 10.1111/j.1572-0241.1998.00338.x. [DOI] [PubMed] [Google Scholar]
- Kemp C.D. Johnson J.C. Riordan W.P. Cotton B.A. How we die: the impact of nonneurologic organ dysfunction after severe traumatic brain injury. Am. Surg. 2008;74:866–872. [PubMed] [Google Scholar]
- Koch A. Sanson E. Helm A. Voigt S. Trautwein C. Tacke F. Regulation and prognostic relevance of serum ghrelin concentrations in critical illness and sepsis. Crit. Care. 2010;14 doi: 10.1186/cc9029. (epub ahead of print). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kojima M. Hosoda H. Date Y. Nakazato M. Matsuo H. Kangawa K. Ghrelin is a growth-hormone-releasing acylated peptide from stomach. Nature. 1999;402:656–660. doi: 10.1038/45230. [DOI] [PubMed] [Google Scholar]
- Lenz A. Franklin G.A. Cheadle W.G. Systemic inflammation after trauma. Injury. 2007;38:1336–1345. doi: 10.1016/j.injury.2007.10.003. [DOI] [PubMed] [Google Scholar]
- Miao Y. Xia Q. Hou Z. Zheng Y. Pan H. Zhu S. Ghrelin protects cortical neuron against focal ischemia/reperfusion in rats. Biochem. Biophys. Res. Commun. 2007;359:795–800. doi: 10.1016/j.bbrc.2007.05.192. [DOI] [PubMed] [Google Scholar]
- Qiu W.C. Wang Z.G. Lv R. Wang W.G. Han X.D. Yan J. Wang Y. Zheng Q. Ai K.X. Ghrelin improves delayed gastrointestinal transit in alloxan-induced diabetic mice. World J. Gastroenterol. 2008;14:2572–2577. doi: 10.3748/wjg.14.2572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sallam H.S. Oliveira H.M. Gan H.T. Herndon D.N. Chen J.D. Ghrelin improves burn-induced delayed gastrointestinal transit in rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2007;292:R253–257. doi: 10.1152/ajpregu.00100.2006. [DOI] [PubMed] [Google Scholar]
- Schneeberger E.E. Lynch R.D. The tight junction: a multifunctional complex. Am. J. Physiol. Cell. Physiol. 2004;286:C1213–1228. doi: 10.1152/ajpcell.00558.2003. [DOI] [PubMed] [Google Scholar]
- Stahel P.F. Shohami E. Younis F.M. Kariya K. Otto V.I. Lenzlinger P.M. Grosjean M.B. Eugster H.P. Trentz O. Kossmann T. Morganti-Kossmann M.C. Experimental closed head injury: analysis of neurological outcome, blood–brain barrier dysfunction, intracranial neutrophil infiltration, and neuronal cell death in mice deficient in genes for pro-inflammatory cytokines. J. Cereb. Blood Flow Metab. 2000;20:369–380. doi: 10.1097/00004647-200002000-00019. [DOI] [PubMed] [Google Scholar]
- Swank G.M. Deitch E.A. Role of the gut in multiple organ failure: bacterial translocation and permeability changes. World J. Surg. 1996;20:411–417. doi: 10.1007/s002689900065. [DOI] [PubMed] [Google Scholar]
- Tracey K.J. Physiology and immunology of the cholinergic anti-inflammatory pathway. J. Clin. Invest. 2007;117:289–296. doi: 10.1172/JCI30555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van der Lely A.J. Ghrelin and new metabolic frontiers. Horm. Res. 2009;71(Suppl 1):129–133. doi: 10.1159/000178055. [DOI] [PubMed] [Google Scholar]
- Waseem T. Duxbury M. Ito H. Ashley S.W. Robinson M.K. Exogenous ghrelin modulates release of pro-inflammatory and anti-inflammatory cytokines in LPS-stimulated macrophages through distinct signaling pathways. Surgery. 2008;143:334–342. doi: 10.1016/j.surg.2007.09.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wren A.M. Small C.J. Ward H.L. Murphy K.G. Dakin C.L. Taheri S. Kennedy A.R. Roberts G.H. Morgan D.G. Ghatei M.A. Bloom S.R. The novel hypothalamic peptide ghrelin stimulates food intake and growth hormone secretion. Endocrinology. 2000;141:4325–4328. doi: 10.1210/endo.141.11.7873. [DOI] [PubMed] [Google Scholar]
- Wu R. Dong W. Cui X. Zhou M. Simms H.H. Ravikumar T.S. Wang P. Ghrelin down-regulates proinflammatory cytokines in sepsis through activation of the vagus nerve. Ann. Surg. 2007;245:480–486. doi: 10.1097/01.sla.0000251614.42290.ed. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu R. Dong W. Qiang X. Wang H. Blau S.A. Ravikumar T.S. Wang P. Orexigenic hormone ghrelin ameliorates gut barrier dysfunction in sepsis in rats. Crit. Care Med. 2009;37:2421–2426. doi: 10.1097/CCM.0b013e3181a557a2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang W. Lin T.R. Hu Y. Fan Y. Zhao L. Stuenkel E.L. Mulholland M.W. Ghrelin stimulates neurogenesis in the dorsal motor nucleus of the vagus. J. Physiol. 2004;559:729–737. doi: 10.1113/jphysiol.2004.064121. [DOI] [PMC free article] [PubMed] [Google Scholar]