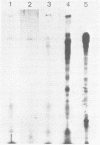
Abstract
Australian grapevine viroid (AGV, 369 residues) is a novel viroid with less than 50% sequence similarity with any known viroid. Nevertheless its entire sequence can be divided into regions, each with a high sequence similarity with segments from one of citrus exocortis, potato spindle tuber, apple scar skin, and grapevine yellow speckle viroids. AGV contains the entire central conserved region of the apple scar skin viroid group and is proposed as a member of this group. AGV appears to have originated from extensive RNA recombination involving other viroids. The vegetatively propagated grapevines which have been exposed to multiple viroid infections during their long history of cultivation may have allowed such recombination.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Diener T. O. Viroid processing: a model involving the central conserved region and hairpin I. Proc Natl Acad Sci U S A. 1986 Jan;83(1):58–62. doi: 10.1073/pnas.83.1.58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dinter-Gottlieb G. Viroids and virusoids are related to group I introns. Proc Natl Acad Sci U S A. 1986 Sep;83(17):6250–6254. doi: 10.1073/pnas.83.17.6250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- García-Arenal F., Pallás V., Flores R. The sequence of a viroid from grapevine closely related to severe isolates of citrus exocortis viroid. Nucleic Acids Res. 1987 May 26;15(10):4203–4210. doi: 10.1093/nar/15.10.4203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haas B., Klanner A., Ramm K., Sänger H. L. The 7S RNA from tomato leaf tissue resembles a signal recognition particle RNA and exhibits a remarkable sequence complementarity to viroids. EMBO J. 1988 Dec 20;7(13):4063–4074. doi: 10.1002/j.1460-2075.1988.tb03300.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hammond R. W., Diener T. O., Owens R. A. Infectivity of chimeric viroid transcripts reveals the presence of alternative processing sites in potato spindle tuber viroid. Virology. 1989 Jun;170(2):486–495. doi: 10.1016/0042-6822(89)90440-6. [DOI] [PubMed] [Google Scholar]
- Hammond R. W., Owens R. A. Mutational analysis of potato spindle tuber viroid reveals complex relationships between structure and infectivity. Proc Natl Acad Sci U S A. 1987 Jun;84(12):3967–3971. doi: 10.1073/pnas.84.12.3967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haseloff J., Symons R. H. Chrysanthemum stunt viroid: primary sequence and secondary structure. Nucleic Acids Res. 1981 Jun 25;9(12):2741–2752. doi: 10.1093/nar/9.12.2741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hashimoto J., Koganezawa H. Nucleotide sequence and secondary structure of apple scar skin viroid. Nucleic Acids Res. 1987 Sep 11;15(17):7045–7052. doi: 10.1093/nar/15.17.7045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ishikawa M., Meshi T., Okada Y., Sano T., Shikata E. In vitro mutagenesis of infectious viroid cDNA clone. J Biochem. 1985 Dec;98(6):1615–1620. doi: 10.1093/oxfordjournals.jbchem.a135431. [DOI] [PubMed] [Google Scholar]
- Keese P., Symons R. H. Domains in viroids: evidence of intermolecular RNA rearrangements and their contribution to viroid evolution. Proc Natl Acad Sci U S A. 1985 Jul;82(14):4582–4586. doi: 10.1073/pnas.82.14.4582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koltunow A. M., Rezaian M. A. A scheme for viroid classification. Intervirology. 1989;30(4):194–201. doi: 10.1159/000150093. [DOI] [PubMed] [Google Scholar]
- Koltunow A. M., Rezaian M. A. Grapevine viroid 1B, a new member of the apple scar skin viroid group contains the left terminal region of tomato planta macho viroid. Virology. 1989 Jun;170(2):575–578. doi: 10.1016/0042-6822(89)90450-9. [DOI] [PubMed] [Google Scholar]
- Koltunow A. M., Rezaian M. A. Grapevine yellow speckle viroid: structural features of a new viroid group. Nucleic Acids Res. 1988 Feb 11;16(3):849–864. doi: 10.1093/nar/16.3.849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kolunow A. M., Krake L. R., Johnson S. D., Rezaian M. A. Two related viroids cause grapevine yellow speckle disease independently. J Gen Virol. 1989 Dec;70(Pt 12):3411–3419. doi: 10.1099/0022-1317-70-12-3411. [DOI] [PubMed] [Google Scholar]
- Rezaian M. A., Koltunow A. M., Krake L. R. Isolation of three viroids and a circular RNA from grapevines. J Gen Virol. 1988 Feb;69(Pt 2):413–422. doi: 10.1099/0022-1317-69-2-413. [DOI] [PubMed] [Google Scholar]
- Rezaian M. A., Williams R. H., Gordon K. H., Gould A. R., Symons R. H. Nucleotide sequence of cucumber-mosaic-virus RNA 2 reveals a translation product significantly homologous to corresponding proteins of other viruses. Eur J Biochem. 1984 Sep 3;143(2):277–284. doi: 10.1111/j.1432-1033.1984.tb08370.x. [DOI] [PubMed] [Google Scholar]
- Sanger F., Coulson A. R., Barrell B. G., Smith A. J., Roe B. A. Cloning in single-stranded bacteriophage as an aid to rapid DNA sequencing. J Mol Biol. 1980 Oct 25;143(2):161–178. doi: 10.1016/0022-2836(80)90196-5. [DOI] [PubMed] [Google Scholar]
- Schnölzer M., Haas B., Raam K., Hofmann H., Sänger H. L. Correlation between structure and pathogenicity of potato spindle tuber viroid (PSTV). EMBO J. 1985 Sep;4(9):2181–2190. doi: 10.1002/j.1460-2075.1985.tb03913.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Symons B. Viroids. Pathogenesis by antisense. Nature. 1989 Apr 13;338(6216):542–543. doi: 10.1038/338542a0. [DOI] [PubMed] [Google Scholar]
- Visvader J. E., Forster A. C., Symons R. H. Infectivity and in vitro mutagenesis of monomeric cDNA clones of citrus exocortis viroid indicates the site of processing of viroid precursors. Nucleic Acids Res. 1985 Aug 26;13(16):5843–5856. doi: 10.1093/nar/13.16.5843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Visvader Jane E., Symons Robert H. Replication of in vitro constructed viroid mutants: location of the pathogenicity-modulating domain of citrus exocortis viroid. EMBO J. 1986 Sep;5(9):2051–2055. doi: 10.1002/j.1460-2075.1986.tb04465.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang K. S., Choo Q. L., Weiner A. J., Ou J. H., Najarian R. C., Thayer R. M., Mullenbach G. T., Denniston K. J., Gerin J. L., Houghton M. Structure, sequence and expression of the hepatitis delta (delta) viral genome. Nature. 1986 Oct 9;323(6088):508–514. doi: 10.1038/323508a0. [DOI] [PubMed] [Google Scholar]
- Wilbur W. J., Lipman D. J. Rapid similarity searches of nucleic acid and protein data banks. Proc Natl Acad Sci U S A. 1983 Feb;80(3):726–730. doi: 10.1073/pnas.80.3.726. [DOI] [PMC free article] [PubMed] [Google Scholar]