Abstract
The proteasome is a multicatalytic protease complex whose activity is required for the growth of normal or tumor cells. It has been shown that human cancer cells are more sensitive to proteasome inhibition than normal cells, indicating that the proteasome could be a target of chemotherapy. Studies suggest that traditional Chinese medicine (TCM) is an effective approach for cancer treatment. Here we reviewed several TCMs for their potential in treatment of cancer. This short review focuses mainly on the TCMs that potentially target the tumor cellular proteasome and NF-κB pathway whose activation is dependent on the proteasome activity.
Keywords: Proteasome inhibitors, natural products, medicinal compounds, cancer, prevention, treatment
1. INTRODUCTION
The use of traditional Chinese medicine (TCM) for medicinal applications has a long and distinguished track record. Dating back to 1500’s, Li Shizhen, one of the greatest physicians and pharmacologists in Chinese history described more than one thousand herbs and their application toward the treatment of diseases. TCM continues to be practiced worldwide, and its continued impact on human health and disease may serve as a useful model for modern drug discovery. Achieving a better understanding of the mechanisms of these antiquated medicines may help expedite their use in the clinical setting. Here we attempted to discuss the role of several TCMs in the treatment of human cancer, with special emphasis on tumor cellular proteasome and NF-kappaB (NF-κB) as putative molecular targets; however we sincerely apologize to those authors whose work has not been cited in this short review because of space limitation.
2. CANCER, UBIQUITIN-PROTEASOME SYSTEM AND NF-κB PATHWAY
The ubiqutin-proteasome pathway is responsible for the timely degradation of most intercellular proteins. First, the targeted protein is marked with a chain of small polypeptides called ubiquitin. Ubiquitination is mediated by a series of enzymes, called E1 ubiquitin activating enzyme, E2 ubiquitin-conjugating enzyme and E3 ubiquitin-ligating enzyme [1]. Ubiquitin is activated by E1 ubiquitin enzyme through adenylation and formation of high-energy thiol ester bond, and then linked to ubiquitin-conjugating enzyme E2 in an ATP-dependent manner. The E3 ubiquitin ligase links the ubiquitin molecule to a reactive lysine residue of a target protein [2]. These polyubiquitinated proteins can then be recognized and targeted by the proteasome for degradation.
The proteasome is a large proteolytic complex with a molecular weight of ~2400 kDa. The proteasome is often referred to as the 26S proteasome, and is expressed in both the nucleus and the cytoplasm in all eukaryotic cells. It consists of one or two 19S regulatory particles and a 20S catalytic core. The 20S proteasome consists of four stacked rings: two inner beta rings and two outer alpha rings, each of which consists of seven subunits [3].
The 19S particle (700 kDa) consists of both a lid and a base, which is responsible for controlling the access of substrates into the 20S proteasome. Furthermore, the 19S particle is responsible for unfolding and threading the ubiquitinated proteins into the catalytic site of the 20S proteasome [4]. These diverse actions are fulfilled by multiple subcomplexes of the 19S regulatory particle. For example, in S. cerevisae, the base subcomplex usually contains 8 subunits and six of them are ATPases that provide ATP for protein substrate unfolding [5]. At least nine subunits composing the lid are non-ATPases that are important for 19S assembly and deubiquitination of ubiquitinated proteasome target proteins [5].
The 20S proteasome hydrolyzes the target protein through three proteolytic activities, caspase or peptidyl-glutamyl peptide-hydrolyzing (PGPH)-like, trypsin-like and chymotrypsin-like activities. These activities are mediated through three β subunits, β1 (PGPH-like), β2 (trypsin-like), and β5 (chymotrypsin-like), respectively [6]. In accordance with enzymes with similar hydrolytic activities, the β1 subunit cuts peptides after acidic residues, the β2 subunit prefers to cleave after basic amino acids, and the β5 subunit hydrolyzes after hydrophobic residues. All the three proteolytic activities depend on the presence of the threonine residue at the amino terminal (Thr1) [7].
The higher level of proteasome activity was observed in tumor cells compared to normal cells, suggesting the importance of the proteasome in the tumor growth. Indeed, several antiapoptotic signaling pathways require proteasomal activity for their function. This is especially true for the pro-oncogenic NF-κB pathway. NF-κB plays an important role in tumorigenesis via transactivation of genes involved in cell proliferation, apoptosis, tumor cell invasion, metastasis, and angiogenesis [8]. Constitutive activation of NF-κB is commonly observed in many types of cancer [8].
Five members, that are p65 (RelA), RelB, c-Rel, p50, and p52 subunits, have been identified in the NF-κB family, all of which contain a REL-homology domain that is responsible for DNA binding, dimerization, nuclear translocation and endogenous inhibitor of NF-κB (IκB) binding [9]. They form homo- and heterodimers through combinatorial assembly, but p50 and p52 must dimerize with a transactivation domain-containing member such as p65, c-REL or REL-B to activate gene transcription [10]. The classic form of NF-κB is p50/p65 heterodimer. The p50 and p52 subunits are processed from their precursors, p105 and p100, respectively, through ubiquitin-dependent proteasome degradation pathway. IκB bind to the REL-homology domain of NF-κB through ankyrin repeats, covering the nuclear localization sequence of NF-κB [9]. Therefore NF-κB is normally sequestered in the cytoplasm through association with its endogenous inhibitor IκB.
Upon stimulation, IκB-α is rapidly phosphorylated by kinase IKK (IκB kinase) composed of two catalytic subunits, IKK-α and IKK-β, and one regulatory subunit IKK-γ [11]. Phosphorylated IκB-α is specifically recognized by SCFβ-TrCP ubiquitin ligase at the 19-amino-acid destruction motif in residues 21-41 [12] and transferred to the proteasome for degradation while NF-κB is allowed to be translocated to the nucleus and mediate transcription of anti-apoptotic genes. However, accumulation of the IκB-α protein via proteasome inhibition prevents the activation of anti-apoptotic NF-κB [13], resulting in tumor growth inhibition and/or apoptosis.
Patients with multiple myeloma (MM) have elevated circulating proteasome, supporting the idea of targeting the proteasome for MM therapy [14]. Bortezomib is the first proteasome inhibitor that entered clinical trials and has been extensively investigated in MM [15]. Remarkable efficacy of bortezomib as a single agent or in combination with other chemotherapeutic drugs was observed in newly diagnosed or refractory myeloma patients. Bortezomib also demonstrated effectiveness against lymphoma [15]. Due to the space limit of this succinct review, we can not refer extensive literature on this topic and we sincerely apologize to those authors whose work has not been cited here.
3. LEI GONG TENG (THUNDER GOD VINE)
Lei gong teng (Thunder God Vine) is ivy like shrub, named Tripterygium wilfordii Hook F, belonging to the family Celastraceae. It is indigenous to southern China and has been used as folk medicine for autoimmune and inflammatory diseases over centuries [16]. This creeping plant is also called “Seven-step vine”, indicating the high toxicity. Indeed, although most of the active components are isolated from the root of the plant [17], ingestion of leaves, flowers, or even the pollens is capable of causing death [18]. Triterpenes and diterpenes and a number of other bioactive compounds have been isolated from lei gong teng Lei Gong Teng [19]. Most research was conduced with triptolide and celastrol, the two major components of lei gong teng.
3.1. Triptolide
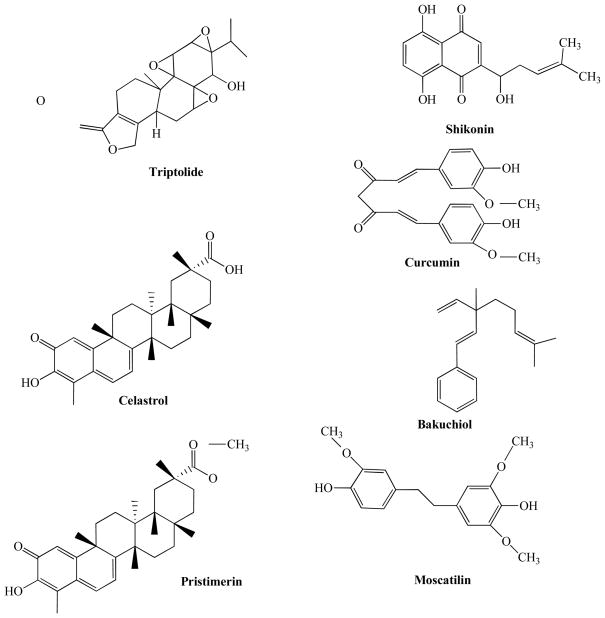
Triptolide is predominantly extracted from the tender root of lei gong teng [17]. Triptolide (PG490) is a diterpene triepoxide (Fig. 1), which is highly oxygenated at the 7,8; 9,11; and 12,13 positions of epoxides. The hydrogen-bonded 9,11-epoxy-14-beta-hydroxy system may be involved in triptolide’s antineoplastic activity [20]. A study using a 3H-labeled triptolide showed that triptolide binding was saturable, reversible, and primarily localized to cell membranes [21]. It inhibited cell growth at low concentrations and induced apoptosis at high concentrations [22]. Triptolide has not been shown to act as an inhibitor of the proteasome, but several reports demonstrated that it affected the NF-κB pathway, whose activation is highly dependent on the ubiquitin-proteasome pathway.
Fig. (1).
Chemical structures of compounds isolated from TCMs.
Triptolide inhibited transactivation of NF-κB induced by Tumor Necrosis Factor-α (TNF-α) and further blocked NF-κB-mediated induction of the inhibitor of apoptosis c- Inhibitor of Apoptosis Protein 1 (IAP1) and c-IAP2 [23]. Trip-tolide inhibited NF-κB transactivation without inhibiting nuclear NF-κB DNA binding activity [23, 24] or even with increased nuclear NF-κB DNA binding activity [25]. Further study demonstrated that triptolide inhibited the binding of p65 to transcriptional coactivator CBP/p300 [26]. Triptolide also suppressed the phorbol myristyl acetate (PMA)-induced activation of NF-κB, in turn, inhibited the overexpression of urokinase-type plasminogen activator receptor (uPAR), which is required for tumor cell invasion [27]. In addition, Triptolide inhibited up to 80% mRNA de novo synthesis in various tumor cells by inhibiting RNA polymerase I and II [28], indicating that it hits a wide spectrum of targets.
F60008 is a semi-synthetic derivative of triptolide that is highly water soluble, and acts as a prodrug of triptolide. F60008 is converted to triptolide in vivo following intravenous administration. A phase I and pharmacological trial was conducted using F60008 given intravenously as a weekly infusion for 2 weeks every 3 weeks in patients with advanced solid tumors. F60008 was converted into triptolide, but the conversion was not complete. Two patients encountered lethal side effects. High inter-individual variability was observed [29]. All these suggested that F60008 was not an optimal derivative of triptolide for clinical use.
3.2. Celastrol
Celastrol is a quinone methide triterpene (Fig. 1) isolated from lei gong teng. It is also extracted from Tripterygium regeli, a substitute of lei gong teng found in northeast of China [30]. We have predicted that celastrol is a proteasome inhibitor by molecular modeling and later confirmed its proteasome-inhibitory activity by in vitro and in vivo experiments [31]. Analysis of atomic orbital energy on celastrol showed that C2 on A-ring and C6 on B-ring of celastrol had a high susceptibility toward a nucleophilic attack. Celastrol could be docked into the S1 pocket of β5 subunit of the proteasome in a conformation to interact with Thr1, the active site of β5 subunit at 92% possibility. To provide direct evidence for proteasome inhibition by celastrol, we performed a cell-free proteasome activity assay using a purified rabbit 20S proteasome in the presence of celastrol at various concentrations. Celastrol potently and preferentially inhibits the chymotrypsin-like activity of a purified 20S proteasome with an IC50 value 2.5 μmol/L. In intact human prostate cancer cells, cellular 26S proteasome was inhibited by celastrol at 1–5 μmol/L. The inhibition of the proteasome activity by celastrol results in accumulation of ubiquitinated proteins and three natural proteasome substrates (IκB-α, Bax and p27), and induction of apoptosis in androgen receptor-positive or androgen receptor-negative prostate cancer cells. Treatment of human prostate tumor-bearing nude mice with celastrol (1–3 mg/kg/day, i.p., for 1 to 31 days) resulted in significant inhibition (65–93%) of the tumor growth. Multiple assays using the animal tumor tissue samples from both early and end time-points demonstrated in vivo inhibition of the proteasomal activity and induction of apoptosis after celastrol treatment. These results demonstrate that celastrol is a natural proteasome inhibitor that has a great potential to be used for cancer prevention and treatment [31].
Antitumor activity of celastrol was also observed in other types of cancer. Celastrol inhibited ~ 60% tumor growth in breast cancer xenograft through NF-κB inhibition [32]. The NF-κB inhibition by celastrol includes inhibition of its DNA-binding activity and inhibition of proteasome-mediated IκB-α degradation which was induced by TNF-α or phorbol myristyl acetate (PMA) [32]. Further investigation showed that celastrol suppressed the NF-κB activation by targeting cysteine 179 in the IκB-α kinase (IKK) [32]. Celastrol was able to potentiate apoptotic effects induced by TNF and chemotherapeutic agents, both regulated by NF-κB activation. It suppressed both inducible and constitutive NF-κB activation. Celastrol was also found to inhibit the TNF-induced activation of IκB-α kinase, IκB-α phosphorylation, IκB-α degradation, p65 nuclear translocation and phosphorylation, and NF-κB-mediated reporter gene expression. TNF induced the expression of gene products involved in anti-apoptosis, proliferation, invasion, and angiogenesis and that celastrol treatment suppressed their expression [33].
Inhibition of NF-κB was observed in concurrence with of activation of heat shock response, indicating a potential link between the two pathways [34]. Indeed, celastrol induced activation of Hsp70, Hsp40 and Hsp27, which was similar as observed in heat shock treatment [34]. Up-regulation of Hsp70A is a well established cell response to Hsp90 inhibition [35]. Two groups independently reported that celastrol is an inhibitor of Hsp90 [36]. Both groups confirmed that celastrol is different with classic Hsp90 inhibitor such as geldanamycin. Hieronymus et al. reported that celastrol inhibits ATP-binding activity of Hsp90 through mechanisms outside the N-terminal ATP-binding pocket of Hsp90 [36] while Zhang et al. reported that celastrol did not interfere with ATP binding to Hsp90 [37]. The difference might be from the different experimental conditions. Celastrol disrupted the interaction between Hsp90 and its cochaperone such as Cdc37 or p23 [36, 37]. Inhibition of Hsp90 by celastrol resulted in suppression of its client proteins that play an important role in tumor initiation and progression. Celastrol also suppressed androgen receptor (AR) that mediates signaling important for prostate cancer progression [36]. The aryl hydrocarbon receptor (AhR) is a client protein of Hsp 90, which plays a significant role in polycyclic aromatic hydrocarbon (PAH)-induced carcinogenesis. Celastrol suppressed AhR expression, resulting in decreased transctivation of CYP1A1 and CYP1B1, both of which encode proteins that convert PAH to genotoxic metabolites [38]. Celastrol also inhibited antiangiogenesis by suppressing vascular endothelial growth factor (VEGF) receptors expression [39].
4. FEN BEI NAN SHE TENG [CELASTRUS HYPOLEUCUS (OLIV) WARB F ARGUTIOR LOES]
Fen bei nan she teng or Celastrus hypoleucus (Oliv) Warb f argutior Loes is another genus that belongs to Celastraceae family. It is widely distributed in China and has been used in the treatment of inflammation and detumescence. Celastrol and methyl celastrol, prestimerin were isolated from this plant [40]. They are also found in other plants of the Celastraceae family that are well distributed throughout tropical America [41].
Pristimerin is a natural analog of celastrol (Fig. 1). Not surprisingly, it also targets the proteasome [42]. Nucleophilic susceptibility and in silico docking studies show that C6 of pristimerin is highly susceptible towards a nucleophilic attack by the hydroxyl group of Thr1 of the proteasomal chymotrypsin subunit. Consistently, pristimerin potently inhibits the chymotrypsin-like activity of a purified rabbit 20S proteasome (IC50 2.2 μmol/L) and human prostate cancer 26S proteasome (IC50 3.0 μmol/L). The accumulation of ubiquitinated proteins and three proteasome target proteins, Bax, p27 and IκB-α, in AR-negative PC-3 prostate cancer cells supports the conclusion that proteasome inhibition by pristimerin is physiologically functional. This observed proteasome inhibition subsequently led to the induction of apoptotic cell death in a dose- and kinetic-dependent manner. Furthermore, in AR-positive, androgen-dependent LNCaP and AR-positive, androgen-independent C4-2B prostate cancer cells, proteasome inhibition by pristimerin results in suppression of AR protein prior to apoptosis. These data demonstrate that the proteasome is a target of pristimerin in prostate cancer cells and inhibition of the proteasomal chymotrypsin-like activity by pristimerin is responsible for its cancer cell death-inducing properties [42]. Inhibition of the chymotryptic activity of the proteasome by pristimerin was confirmed by another group [43].
Currently, it is still not completely clear how pritimerin induces proteasome inhibition and apoptosis. Pristimerin-triggered caspase activation was confirmed in human breast cancer cells [44]. MDA-MB-231 cells treated with pristimerin showed rapid induction of apoptosis through caspase activation, which could be completely prevented by pre-treatment with a pancaspase inhibitor benzyloxycarbonyl-Val-Ala-Asp-fluoromethyl ketone (z-VAD-fmk). Treatment of breast tumor cells with pristimerin resulted in a rapid release of cytochrome c from mitochondria, which preceded caspase activation and the decrease of mitochondrial membrane potential. This process did not depend on Bcl-2 family (Bcl-2, Bcl-XL and Bax) protein levels and did not require translocation of Bax to the mitochondria. Moreover, the generation of reactive oxygen species (ROS) in MDA-MB-231 cells was also not affected by pristimerin [44]. However, pristimerin-induced mitochondrial cell death in cervical cancer cells was reported through ROS generation [45]. It activated JNK and PARP-1 pathways, resulting loss of mitochondrial membrane potential, and apoptotic cell death. JNK phosphorylation caused Bax mitochondrial translocation, cytochrome c release and caspase 3 or 9 activation. Using NAC to inhibit ROS generation prevents pristimerin-induced cell death involved in both pathways [45].
5. ZI CAO (PURPLE GROMWELL)
Zi cao or purple gromwell is the dried root of Lithospermum erythrorhizon Sieb. et Zucc, Arnebia euchroma (Royle) Johnst, or Arnebia guttata Bunge. It is a commonly used herbal medicine in China and other countries [46]. Dating back to the 5th century, zi cao had been used for the treatment of throat sore, burns, cut and skin diseases such as macular eruption, measles and carbuncles in China [47].
Naphthoquinone shikonin (Fig. 1) is a main component isolated from zi cao. Our computational modeling predicts that the carbonyl carbons C1 and C4 of shikonin potentially interact with the catalytic site of β5 chymotryptic subunit of the proteasome [48]. Indeed, shikonin potently inhibits the chymotrypsin-like activity of purified 20S proteasome (IC50 12.5 μmol/L) and tumor cellular 26S proteasome (IC50 between 2–16 μmol/L). Inhibition of the proteasome by shikonin in murine hepatoma H22, leukemia P388 and human prostate cancer PC-3 cultures resulted in accumulation of ubiquitinated proteins and several proteasome target proapoptotic proteins (IκB-α, Bax and p27), followed by induction of cell death. Shikonin treatment resulted in tumor growth inhibition in both H22 allografts and PC-3 xenografts, associated with suppression of the proteasomal activity and induction of cell death in vivo. Finally, shikonin treatment significantly prolonged the survival period of mice bearing P388 leukemia. These results indicate that the tumor proteasome is one of the cellular targets of shikonin and inhibition of the proteasome activity by shikonin contributes to its antitumor properties [48].
Other studies revealed that shikonin induced different modes of cell death depending on the concentrations applied [49]. In leukemia cell line HL60, shikonin induced a dominant apoptosis at ≤2.5 μM, a dominant necroptosis ≥10 μM, and a mixed type at 5 μM. When HL60 cells were treated with shikonin (≥10 μM) in the presence of Nec-1, an specific necroptosis inhibitor, necroptosis was switched to apoptosis [49]. The death mode switch was at least partially due to the conversion from mitochondrial inner membrane permeability to outer membrane permeability [49]. In some drug-resistant tumor cell lines, such as MCF-7 and HEK293 that overexpressed P-gp, Bcl-2 or Bcl-XL, shikonin induced a dominant necroptosis, which could circumvent cancer drug resistance [50]. Shiikonin also down-regulated expression of steroid sulfatase mRNA and decreased enzymatic activity in breast cancer [51].
So far, only one clinical trial using shikonin-containing mixture was found in the literature. In 19 cases of late-stage lung cancer that were not suitable for surgery and irradiation, a shikonin-containing mixture reduced tumor diameter radiographically in 63.3% of patients, with a reported remission rate of 36.9%, and 1-year survival rate of 47.3% [52]. Furthermore, administration of the shikonin-containing mixture increased body weight and appetite of patients. No harmful effects on the peripheral system, heart, kidney and liver were observed after shikonin treatment [52].
6. JIANGHUANG (TUMERIC)
Spice tumeric (Curcuma logma Linn) has been used in China, India and Southeast Asia for centuries as food coloring and flavoring agent. In addition, it has a well-documented history in medicine in China and India [53]. Tumeric is used to treat various diseases, including respiratory conditions, inflammation, liver disorders, diabetic wounds, cough, and certain tumors [54].
Curcumin (diferuloylmethane) (Fig. 1) is the yellow pigment derived from the rhizome of Curcuma longa. It inhibits tumor cell proliferation, invasion, angiogenesis and metastasis, involved in multiple cellular targets [55]. The tumor cellular proteasome is most likely one of important targets of curcumin. Nucleophilic susceptibility and in silico docking studies show that both carbonyl carbons of the curcumin molecule are highly susceptible to a nucleophilic attack by the hydroxyl group of the N-terminal threonine of the proteasomal chymotrypsin-like subunit [56]. Curcumin could inhibit all three proteasomal activities but with the highest potency to the chymotrypsin-like activity. Furthermore, inhibition of proteasome activity by curcumin in human colon cancer HCT-116 and SW480 cell lines leads to accumulation of ubiquitinated proteins and several proteasome target proteins, and subsequent induction of apoptosis. Finally, treatment of HCT-116 colon tumor-bearing ICR SCID mice with curcumin resulted in decreased tumor growth, associated with proteasome inhibition, proliferation suppression, and apoptosis induction in tumor tissues [56].
Many studies have shown that curcumin also targets the NF-κB pathway. Curcumin suppresses constitutive activation of NF-κB which is often observed in leukemia cells such as multiple myeloma, mantle cell lymphoma and Burkitt’s lymphoma [57–59] and solid tumors such as pancreatic cancer [60]. Curcumin also inhibits drug-induced NF-κB activation that is triggered by many currently used chemotherapeutic drugs. Paclitaxel induced proteasome-mediated degradation of IκB-α, which led to activation of NF-κB in human MDA-MB-231 breast cancer cells. This process could be inhibited by curcumin, thus resulting in enhanced efficacy of paclitaxel for inhibiting breast cancer growth in vitro and in vivo [61]. Curcumin reduced gemcitabine-induced NF-κB activation in cultured human bladder cancer 253JBV cells and abolished the constitutive activation of NF-κB in the tumor tissue of orthotopic mouse model, suggesting that curcumin potentiates the effects of gemictabine, possibly through the modulation of NF-κB signaling pathway [62].
The major problems that limit the clinical applications of curcumin are its poor water solubility and bioavailability. Indeed, curcumin exhibited poor bioavailability after oral administration with low nanomolar levels of the parent compound and large amount of its glucuronide and sulphate conjugates found in the peripheral or portal circulation [63, 64]. Chemotherapeutic effects of curcumin were observed in colon cancer. Administration of curcumin-containing extract demonstrated radiologically stable disease in 5 out 15 patients for 2–4 months [65]. A daily dose of 3.6 g curcumin could significantly decrease levels of inducible Prostaglandin E2 (PGE2) production and DNA adduct M1G formed by malondialdehyde, confirming that curcumin could be achieved at pharmacologically active levels in the colorectum of humans [66, 67]. Trails to determine effects of curcumin against other types of cancers were conducted, showing curcumin was not efficient against the tumors distant from the locus of absorption [63]. Trials currently underway include an evaluation of colon cancer patients treated with gemcitabine in combination with curcumin and celecoxib (NCT00295035; Tel-Aviv Sourasky Medical Center) and pancreatic cancer patients treated with curcumin combined with gemcitabine (NCT00192842; Rambam Health Care Campus).
7. BU GU ZHI (PSORALEA CORYLIFOLIA L.)
Bu gu zhi, also called Psoralea corylifolia L. is a plant belonging to the Fabaceae family. This plant has been used in Chinese and Indian traditional medicine for the treatment of leukoderma, leprosy, psoriasis, vitiligo, and inflammatory diseases of the skin [68]. Recent animal studies suggest it potential use to prevent brain aging [69].
More than 35 components have been isolated from bu gu zhi, such as psoralen, isopsoralen, psoralidin, corylifolin, corylin, corylifolinin, isobavachalcone, corylifol A, and bakuchiol [70–73], which are abundant in the seed [73]. No monomer of Psoralea corylifolia L. has been reported to target the proteasome, while the whole extract of this medicinal plant was found to inhibit the proteasome [68]. Incubation of SH-SY5Y cells with 20 μg/mL of Psoralea corylifolia for 24 h significantly inhibited all the three proteasome activities to a similar level (about 20% lower compared to control). Accumulation of poly-ubiquitinated proteins was observed in Psoralea corylifolia extract-treated SH-SY5Y cells, supporting the inhibition of proteasome activities by Psoralea corylifolia [68]. In addition, bakuchiol (Fig. 1), one monomer of Psoralea corylifolia was found to inhibit NF-κB binding activity that was induced by pro-inflammatory cytokines, interferon γ and lipopolysaccharide (LPS) [74].
8. ORCHID DENDROBIUM LODDIGESII ROLFE
Dendrobium loddigesii Rolfe has been commonly used in traditional Chinese medicine as a tonic to nourish the stomach, replenish body fluid, and reduce fever [75]. Dendrophenol (4,4′-dihydroxy-3,3′,5-trimethoxybibenzyl), also named Moscatilin (Fig. 1) was isolated from the stem of Dendrobium loddigesii Rolfe [76] and was shown to act as a NF-κB inhibitor [77]. It inhibited phosphorylation of IκB-α, suppressed nuclear translocation of NF-κB subunits, p65 and p50, and NF-κB activity. Mosatilin also showed antiproliferative effects against a panel of tumor cell lines including liver, lung and stomach cancers [75]. Moscatilin induced apoptosis in colon cancer HCT 116 cells through tubulin depolymerization and DNA damage stress, which lead to JNK activation and mitochondria-involved apoptosis [78], suggesting the potential clinical anticancer application of moscatilin.
CONCLUSION
In this review we attempted to focus mainly on the TCMs that potentially target the tumor cellular proteasome and NF-κB pathway. Medicinal plants have been used as folk medicines in China for over centuries. Separation techniques allowed researchers to analyze monomer component or purified compounds from the plant extract of TCM. Development on molecular biology allowed researchers to dissect the complex network which aberrant lead to malfunction of normal cell or cancer. To better understand how TCM hit the cellular targets for disease treatment, scientists tried to identify the molecules that are involved in the action of TCM.
In tumor cells, high level of the proteasome activity is often observed, suggesting that the proteasome is essential in tumor growth. Indeed, the proteasome is required for NF-κB activation which is required for cell proliferation, tumor cell invasion, metastasis, and angiogenesis. The first proteasome inhibitor Bortezomib has been tested in clinical trials for several years and significant efficacies against multiple myeloma have been reported in different setting of trials. The results in some cases have been impressive. There is less evidence of botezomib’s efficacy against solid tumors. There is therefore interest in developing the natural proteasome inhibitors from TCMs. Whether TCM target the proteasome and/or NF-κB pathway has been investigated by using TCM pure monomers or isolated compounds. Isolated celastrol, shikonin and curcumin have been shown to inhibit purified 20S proteasome and 26S proteasome in cultured tumor cells and tumor tissues. Triptolide, celastrol and moscatilin have also been shown to suppress NF-κB pathway. They inhibited IκB-α degradation, nuclear translocation of NF-κB subunits p65 and p50, and NF-κB binding activity. These properties entitle these TCM compounds with ability to suppress tumor growth. Future studies should focus on the molecularly targeted clinical trails using TCMs.
Acknowledgments
The research was partially supported by grants from National Cancer Institute (1R01CA120009 and 3R01CA120009-04S1 to Q.P. Dou) and a grant from The National High Technology Research and Development Program of China (2006AA02Z4B5 to J. Liu). The authors thank Mr. Michal Frezza for critical reading of the manuscript.
ABBREVIATIONS
- AR
Androgen receptor
- AhR
Hydrocarbon receptor
- EGFR
Epidermal growth factor receptor
- IAP
Inhibitor of apoptosis protein
- IκB
Inhibitor of NF-κB
- LPS
Lipopolysaccharide
- MM
Multiple myeloma
- NF-κB
Nuclear factor kappa B
- PAH
Polycyclic aromatic hydrocarbon
- PGPH
Peptidyl-glutamyl peptide-hydrolyzing-like
- PMA
Phorbol myristyl acetate
- PGE2
Prostaglandin E2
- ROS
Reactive oxygen species
- Thr1
The threonine residue at the amino terminal
- TNF
Tumor necrosis factor
- uPAR
Urokinase-type plasminogen activator receptor
- VEGF
Vascular endothelial growth factor
References
- 1.Ciechanover A. The ubiquitin-proteasome proteolytic pathway. Cell. 1994;79(1):13–21. doi: 10.1016/0092-8674(94)90396-4. [DOI] [PubMed] [Google Scholar]
- 2.Ciechanover A. The ubiquitin-proteasome pathway: on protein death and cell life. EMBO J. 1998;17(24):7151–60. doi: 10.1093/emboj/17.24.7151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Goldberg AL. Protein degradation and protection against misfolded or damaged proteins. Nature. 2003;426(6968):895–99. doi: 10.1038/nature02263. [DOI] [PubMed] [Google Scholar]
- 4.Dou QP, Smith DM, Daniel KG, Kazi A. Interruption of tumor cell cycle progression through proteasome inhibition: implications for cancer therapy. Prog Cell Cycle Res. 2003;5:441–6. [PubMed] [Google Scholar]
- 5.Pickart CM, Cohen RE. Proteasomes and their kin: proteases in the machine age. Nat Rev Mol Cell Biol. 2004;5(3):177–87. doi: 10.1038/nrm1336. [DOI] [PubMed] [Google Scholar]
- 6.Orlowski RZ, Kuhn DJ. Proteasome inhibitors in cancer therapy: Lessons from the first decade. Clin Cancer Res. 2008;14(6):1649–57. doi: 10.1158/1078-0432.CCR-07-2218. [DOI] [PubMed] [Google Scholar]
- 7.Groll M, Ditzel L, Lowe J, et al. Structure of 20S proteasome from yeast at 2.4 A resolution. Nature. 1997;386(6624):463–71. doi: 10.1038/386463a0. [DOI] [PubMed] [Google Scholar]
- 8.Orlowski RZ, Baldwin AS., Jr NF-kappaB as a therapeutic target in cancer. Trends Mol Med. 2002;8(8):385–9. doi: 10.1016/s1471-4914(02)02375-4. [DOI] [PubMed] [Google Scholar]
- 9.Baud V, Karin M. Is NF-kappaB a good target for cancer therapy? Hopes and pitfalls. Nat Rev Drug Discov. 2009;8(1):33–40. doi: 10.1038/nrd2781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bhoj VG, Chen ZJ. Ubiquitylation in innate and adaptive immunity. Nature. 2009;458(7237):430–37. doi: 10.1038/nature07959. [DOI] [PubMed] [Google Scholar]
- 11.Solt LA, May MJ. The IkappaB kinase complex: master regulator of NF-kappaB signaling. Immunol Res. 2008;42(1–3):3–18. doi: 10.1007/s12026-008-8025-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Winston JT, Strack P, Beer-Romero P, Chu CY, Elledge SJ, Harper JW. The SCFbeta-TRCP-ubiquitin ligase complex associates specifically with phosphorylated destruction motifs in IkappaBalpha and beta-catenin and stimulates IkappaBalpha ubiquitination in vitro. Genes Dev. 1999;13(3):270–83. doi: 10.1101/gad.13.3.270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Biswas DK, Iglehart JD. Linkage between EGFR family receptors and nuclear factor kappaB (NF-kappaB) signaling in breast cancer. J Cell Physiol. 2006;209(3):645–52. doi: 10.1002/jcp.20785. [DOI] [PubMed] [Google Scholar]
- 14.Jakob C, Egerer K, Liebisch P, et al. Circulating proteasome levels are an independent prognostic factor for survival in multiple myeloma. Blood. 2007;109(5):2100–5. doi: 10.1182/blood-2006-04-016360. [DOI] [PubMed] [Google Scholar]
- 15.Yang H, Zonder JA, Dou QP. Clinical development of novel proteasome inhibitors for cancer treatment. Expert Opin Investig Drugs. 2009;18(7):957–71. doi: 10.1517/13543780903002074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Xu G, Tu W, Jiang D, Xu C. Tripterygium wilfordii Hook F treatment for idiopathic refractory nephrotic syndrome in adults: A meta-analysis. Nephron. 2009;111(4):c223–28. doi: 10.1159/000208990. [DOI] [PubMed] [Google Scholar]
- 17.Luo XL, Shao Q, Qu HB, Cheng YY. Simple method for determination of five terpenoids from different parts of Tripterygium wilfordii and its preparations by HPLC coupled with evaporative light scattering detection. J Sep Sci. 2007;30(9):1284–91. doi: 10.1002/jssc.200600450. [DOI] [PubMed] [Google Scholar]
- 18.Deng W. Analysis of the Toxicities and Side Effects of Leigongteng. Pharmacology and Clinics of Chinese Materia Medica. 2001;17(3):42–47. [Google Scholar]
- 19.Zhou BN. Some progress on the chemistry of natural bioactive terpenoids from Chinese medicinal plants. Mem Inst Oswaldo Cruz. 1991;86(Suppl 2):219–26. doi: 10.1590/s0074-02761991000600049. [DOI] [PubMed] [Google Scholar]
- 20.Kupchan SM, Schubert RM. Selective alkylation: A biomimetic reaction of the antileukemic triptolides? Science. 1974;185(153):791–3. doi: 10.1126/science.185.4153.791. [DOI] [PubMed] [Google Scholar]
- 21.Leuenroth SJ, Crews CM. Studies on calcium dependence reveal multiple modes of action for triptolide. Chem Biol. 2005;12(12):1259–68. doi: 10.1016/j.chembiol.2005.09.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kiviharju TM, Lecane PS, Sellers RG, Peehl DM. Antiproliferative and proapoptotic activities of triptolide (PG490), a natural product entering clinical trials, on primary cultures of human prostatic epithelial cells. Clin Cancer Res. 2002;8(8):2666–74. [PubMed] [Google Scholar]
- 23.Lee KY, Chang W, Qiu D, Kao PN, Rosen GD. PG490 (triptolide) cooperates with tumor necrosis factor-alpha to induce apoptosis in tumor cells. J Biol Chem. 1999;274(19):13451–55. doi: 10.1074/jbc.274.19.13451. [DOI] [PubMed] [Google Scholar]
- 24.Jiang XH, Wong BC, Lin MC, et al. Functional p53 is required for triptolide-induced apoptosis and AP-1 and nuclear factor-kappaB activation in gastric cancer cells. Oncogene. 2001;20(55):8009–18. doi: 10.1038/sj.onc.1204981. [DOI] [PubMed] [Google Scholar]
- 25.Qiu D, Zhao G, Aoki Y, et al. Immunosuppressant PG490 (triptolide) inhibits T-cell interleukin-2 expression at the level of purine-box/nuclear factor of activated T-cells and NF-kappaB transcriptional activation. J Biol Chem. 1999;274(19):13443–50. doi: 10.1074/jbc.274.19.13443. [DOI] [PubMed] [Google Scholar]
- 26.Zhu W, Ou Y, Li Y, et al. A small-molecule triptolide suppresses angiogenesis and invasion of human anaplastic thyroid carcinoma cells via down-regulation of the nuclear factor-kappa B pathway. Mol Pharmacol. 2009;75(4):812–9. doi: 10.1124/mol.108.052605. [DOI] [PubMed] [Google Scholar]
- 27.Chang HJ, Kim MH, Baek MK, et al. Triptolide inhibits tumor promoter-induced uPAR expression via blocking NF-kappaB signaling in human gastric AGS cells. Anticancer Res. 2007;27(5A):3411–17. [PubMed] [Google Scholar]
- 28.Vispe S, Devries L, Creancier L, et al. Triptolide is an inhibitor of RNA polymerase I and II-dependent transcription leading predominantly to down-regulation of short-lived mRNA. Mol Cancer Ther. 2009;8(10):2780–90. doi: 10.1158/1535-7163.MCT-09-0549. [DOI] [PubMed] [Google Scholar]
- 29.Kitzen JJ, de Jonge MJ, Lamers CH, et al. Phase I dose-escalation study of F60008, a novel apoptosis inducer, in patients with advanced solid tumours. Eur J Cancer. 2009;45(10):1764–72. doi: 10.1016/j.ejca.2009.01.026. [DOI] [PubMed] [Google Scholar]
- 30.Chen D, Cui QC, Yang H, Dou QP. Disulfiram, a clinically used anti-alcoholism drug and copper-binding agent, induces apoptotic cell death in breast cancer cultures and xenografts via inhibition of the proteasome activity. Cancer Res. 2006;66(21):10425–33. doi: 10.1158/0008-5472.CAN-06-2126. [DOI] [PubMed] [Google Scholar]
- 31.Yang H, Chen D, Cui QC, Yuan X, Dou QP. Celastrol, a triterpene extracted from the Chinese “Thunder of God Vine,” is a potent proteasome inhibitor and suppresses human prostate cancer growth in nude mice. Cancer Res. 2006;66(9):4758–65. doi: 10.1158/0008-5472.CAN-05-4529. [DOI] [PubMed] [Google Scholar]
- 32.Lee JH, Koo TH, Yoon H, et al. Inhibition of NF-kappa B activation through targeting I kappa B kinase by celastrol, a quinone methide triterpenoid. Biochem Pharmacol. 2006;72(10):1311–21. doi: 10.1016/j.bcp.2006.08.014. [DOI] [PubMed] [Google Scholar]
- 33.Sethi G, Ahn KS, Pandey MK, Aggarwal BB. Celastrol, a novel triterpene, potentiates TNF-induced apoptosis and suppresses invasion of tumor cells by inhibiting NF-kappaB-regulated gene products and TAK1-mediated NF-kappaB activation. Blood. 2007;109(7):2727–35. doi: 10.1182/blood-2006-10-050807. [DOI] [PubMed] [Google Scholar]
- 34.Westerheide SD, Bosman JD, Mbadugha BN, et al. Celastrols as inducers of the heat shock response and cytoprotection. J Biol Chem. 2004;279(53):56053–60. doi: 10.1074/jbc.M409267200. [DOI] [PubMed] [Google Scholar]
- 35.Cullinan SB, Whitesell L. Heat shock protein 90: A unique chemotherapeutic target. Semin Oncol. 2006;33(4):457–65. doi: 10.1053/j.seminoncol.2006.04.001. [DOI] [PubMed] [Google Scholar]
- 36.Hieronymus H, Lamb J, Ross KN, et al. Gene expression signature-based chemical genomic prediction identifies a novel class of HSP90 pathway modulators. Cancer Cell. 2006;10(4):321–30. doi: 10.1016/j.ccr.2006.09.005. [DOI] [PubMed] [Google Scholar]
- 37.Zhang T, Hamza A, Cao X, et al. A novel Hsp90 inhibitor to disrupt Hsp90/Cdc37 complex against pancreatic cancer cells. Mol Cancer Ther. 2008;7(1):162–70. doi: 10.1158/1535-7163.MCT-07-0484. [DOI] [PubMed] [Google Scholar]
- 38.Hughes D, Guttenplan JB, Marcus CB, Subbaramaiah K, Dannenberg AJ. Heat shock protein 90 inhibitors suppress aryl hydrocarbon receptor-mediated activation of CYP1A1 and CYP1B1 transcription and DNA adduct formation. Cancer Prev Res. 2008;1(6):485–93. doi: 10.1158/1940-6207.CAPR-08-0149. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 39.Huang Y, Zhou Y, Fan Y, Zhou D. Celastrol inhibits the growth of human glioma xenografts in nude mice through suppressing VEGFR expression. Cancer Lett. 2008;264(1):101–6. doi: 10.1016/j.canlet.2008.01.043. [DOI] [PubMed] [Google Scholar]
- 40.Luo DQ, Wang H, Tian X, Shao HJ, Liu JK. Antifungal properties of pristimerin and celastrol isolated from Celastrus hypoleucus. Pest Manag Sci. 2005;61(1):85–90. doi: 10.1002/ps.953. [DOI] [PubMed] [Google Scholar]
- 41.Buffa Filho W, Corsino J, Bolzani da SV, Furlan M, Pereira AM, Franca SC. Quantitative determination for cytotoxic Friedo-noroleanane derivatives from five morphological types of Maytenus ilicifolia (Celastraceae) by reverse-phase high-performance liquid chromatography. Phytochem Anal. 2002;13(2):75–8. doi: 10.1002/pca.626. [DOI] [PubMed] [Google Scholar]
- 42.Yang H, Landis-Piwowar KR, Lu D, et al. Pristimerin induces apoptosis by targeting the proteasome in prostate cancer cells. J Cell Biochem. 2008;103(1):234–44. doi: 10.1002/jcb.21399. [DOI] [PubMed] [Google Scholar]
- 43.Tiedemann RE, Schmidt J, Keats JJ, et al. Identification of a potent natural triterpenoid inhibitor of proteosome chymotrypsin-like activity and NF-kappaB with antimyeloma activity in vitro and in vivo. Blood. 2009;113(17):4027–37. doi: 10.1182/blood-2008-09-179796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wu CC, Chan ML, Chen WY, Tsai CY, Chang FR, Wu YC. Pristimerin induces caspase-dependent apoptosis in MDA-MB-231 cells via direct effects on mitochondria. Mol Cancer Ther. 2005;4(8):1277–85. doi: 10.1158/1535-7163.MCT-05-0027. [DOI] [PubMed] [Google Scholar]
- 45.Byun JY, Kim MJ, Eum DY, et al. Reactive oxygen species-dependent activation of Bax and PARP-1 is required for mitochondrial cell death induced by triterpenoid pristimerin in human cervical cancer cells. Mol Pharmacol. 2009;76(4):734–44. doi: 10.1124/mol.109.056259. [DOI] [PubMed] [Google Scholar]
- 46.Chen X, Yang L, Zhang N, et al. Shikonin, a component of chinese herbal medicine, inhibits chemokine receptor function and suppresses human immunodeficiency virus type 1. Antimicrob Agents Chemother. 2003;47(9):2810–16. doi: 10.1128/AAC.47.9.2810-2816.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Chen X, Yang L, Oppenheim JJ, Howard MZ. Cellular pharmacology studies of shikonin derivatives. Phytother Res. 2002;16(3):199–209. doi: 10.1002/ptr.1100. [DOI] [PubMed] [Google Scholar]
- 48.Yang H, Zhou P, Huang H, et al. Shikonin exerts antitumor activity via proteasome inhibition and cell death induction in vitro and in vivo. Intl J Cancer. 2009;124(10):2450–59. doi: 10.1002/ijc.24195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Han W, Xie J, Li L, Liu Z, Hu X. Necrostatin-1 reverts shikonin-induced necroptosis to apoptosis. Apoptosis. 2009;14(5):674–86. doi: 10.1007/s10495-009-0334-x. [DOI] [PubMed] [Google Scholar]
- 50.Han W, Li L, Qiu S, et al. Shikonin circumvents cancer drug resistance by induction of a necroptotic death. Mol Cancer Ther. 2007;6(5):1641–9. doi: 10.1158/1535-7163.MCT-06-0511. [DOI] [PubMed] [Google Scholar]
- 51.Zhang Y, Qian RQ, Li PP. Shikonin, an ingredient of Lithospermum erythrorhizon, down-regulates the expression of steroid sulfatase genes in breast cancer cells. Cancer Lett. 2009;284(1):47–54. doi: 10.1016/j.canlet.2009.04.008. [DOI] [PubMed] [Google Scholar]
- 52.Guo XP, Zhang XY, Zhang SD. Clinical trial on the effects of shikonin mixture on later stage lung cancer. Zhong Xi Yi Jie He Za Zhi. 1991;11(10):598–99. 580. [PubMed] [Google Scholar]
- 53.Hatcher H, Planalp R, Cho J, Torti FM, Torti SV. Curcumin: From ancient medicine to current clinical trials. Cell Mol Life Sci. 2008;65(11):1631–52. doi: 10.1007/s00018-008-7452-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Aggarwal BB, Sung B. Pharmacological basis for the role of curcumin in chronic diseases: An age-old spice with modern targets. Trends Pharmacol Sci. 2009;30(2):85–94. doi: 10.1016/j.tips.2008.11.002. [DOI] [PubMed] [Google Scholar]
- 55.Kunnumakkara AB, Anand P, Aggarwal BB. Curcumin inhibits proliferation, invasion, angiogenesis and metastasis of different cancers through interaction with multiple cell signaling proteins. Cancer Lett. 2008;269(2):199–225. doi: 10.1016/j.canlet.2008.03.009. [DOI] [PubMed] [Google Scholar]
- 56.Milacic V, Banerjee S, Landis-Piwowar KR, Sarkar FH, Majumdar AP, Dou QP. Curcumin inhibits the proteasome activity in human colon cancer cells in vitro and in vivo. Cancer Res. 2008;68(18):7283–92. doi: 10.1158/0008-5472.CAN-07-6246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Bharti AC, Shishodia S, Reuben JM, et al. Nuclear factor-kappaB and STAT3 are constitutively active in CD138+ cells derived from multiple myeloma patients, and suppression of these transcription factors leads to apoptosis. Blood. 2004;103(8):3175–84. doi: 10.1182/blood-2003-06-2151. [DOI] [PubMed] [Google Scholar]
- 58.Hussain AR, Ahmed M, Al-Jomah NA, et al. Curcumin suppresses constitutive activation of nuclear factor-kappa B and requires functional Bax to induce apoptosis in Burkitt’s lymphoma cell lines. Mol Cancer Ther. 2008;7(10):3318–29. doi: 10.1158/1535-7163.MCT-08-0541. [DOI] [PubMed] [Google Scholar]
- 59.Shishodia S, Amin HM, Lai R, Aggarwal BB. Curcumin (diferuloylmethane) inhibits constitutive NF-kappaB activation, induces G1/S arrest, suppresses proliferation, and induces apoptosis in mantle cell lymphoma. Biochem Pharmacol. 2005;70(5):700–713. doi: 10.1016/j.bcp.2005.04.043. [DOI] [PubMed] [Google Scholar]
- 60.Wang W, Abbruzzese JL, Evans DB, Larry L, Cleary KR, Chiao PJ. The nuclear factor-kappa B RelA transcription factor is constitutively activated in human pancreatic adenocarcinoma cells. Clin Cancer Res. 1999;5(1):119–27. [PubMed] [Google Scholar]
- 61.Kang HJ, Lee SH, Price JE, Kim LS. Curcumin suppresses the paclitaxel-induced nuclear factor-kappaB in breast cancer cells and potentiates the growth inhibitory effect of paclitaxel in a breast cancer nude mice model. Breast J. 2009;15(3):223–9. doi: 10.1111/j.1524-4741.2009.00709.x. [DOI] [PubMed] [Google Scholar]
- 62.Tharakan ST, Inamoto T, Sung B, Aggarwal BB, Kamat AM. Curcumin potentiates the antitumor effects of gemcitabine in an orthotopic model of human bladder cancer through suppression of proliferative and angiogenic biomarkers. Biochem Parmacol. 2010;79(2):218–28. doi: 10.1016/j.bcp.2009.08.007. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 63.Garcea G, Jones DJ, Singh R, et al. Detection of curcumin and its metabolites in hepatic tissue and portal blood of patients following oral administration. Br J Cancer. 2004;90(5):1011–5. doi: 10.1038/sj.bjc.6601623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Dhillon N, Aggarwal BB, Newman RA, et al. Phase II trial of curcumin in patients with advanced pancreatic cancer. Clin Cancer Res. 2008;14(14):4491–99. doi: 10.1158/1078-0432.CCR-08-0024. [DOI] [PubMed] [Google Scholar]
- 65.Sharma RA, McLelland HR, Hill KA, et al. Pharmacodynamic and pharmacokinetic study of oral Curcuma extract in patients with colorectal cancer. Clin Cancer Res. 2001;7(7):1894–900. [PubMed] [Google Scholar]
- 66.Sharma RA, Euden SA, Platton SL, et al. Phase I clinical trial of oral curcumin: Biomarkers of systemic activity and compliance. Clin Cancer Res. 2004;10(20):6847–54. doi: 10.1158/1078-0432.CCR-04-0744. [DOI] [PubMed] [Google Scholar]
- 67.Garcea G, Berry DP, Jones DJ, et al. Consumption of the putative chemopreventive agent curcumin by cancer patients: Assessment of curcumin levels in the colorectum and their pharmacodynamic consequences. Cancer Epidemiol Biomarkers Prev. 2005;14(1):120–25. [PubMed] [Google Scholar]
- 68.Tang SY, Gruber J, Wong KP, Halliwell B. Psoralea corylifolia L. inhibits mitochondrial complex I and proteasome activities in SH-SY5Y cells. Ann N Y Acad Sci. 2007;1100:486–96. doi: 10.1196/annals.1395.053. [DOI] [PubMed] [Google Scholar]
- 69.Zhao G, Zheng XW, Qin GW, Gai Y, Jiang ZH, Guo LH. In vitro dopaminergic neuroprotective and in vivo antiparkinsonian-like effects of Delta 3,2-hydroxybakuchiol isolated from Psoralea corylifolia (L) Cell Mol Life Sci. 2009;66(9):1617–29. doi: 10.1007/s00018-009-9030-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Ji L, Xu Z. Review of constituents in fruits of Psoralea corylifolia L. Zhongguo Zhong Yao Za Zhi. 1995;20(2):120–22. 128. [PubMed] [Google Scholar]
- 71.Yadava RN, Verma V. A new biologically active flavonol glycoside from Psoralea corylifolia (Linn) J Asian Nat Prod Res. 2005;7(4):671–75. doi: 10.1080/10286020310001608921. [DOI] [PubMed] [Google Scholar]
- 72.Chen B, Liu LL, Zhai ZX, Chen W, Fang HS, Wu YT. Determination of major components in Psoralea corylifolia L. by using supercritical fluid extraction (SFE) Se Pu. 2000;18(1):61–3. [PubMed] [Google Scholar]
- 73.Qiao CF, Han QB, Mo SF, et al. Psoralenoside and isopsoralenoside, two new benzofuran glycosides from Psoralea corylifolia. Chem Pharm Bull. 2006;54(5):714–716. doi: 10.1248/cpb.54.714. [DOI] [PubMed] [Google Scholar]
- 74.Pae HO, Cho H, Oh GS, et al. Bakuchiol from Psoralea corylifolia inhibits the expression of inducible nitric oxide synthase gene via the inactivation of nuclear transcription factor-kappaB in RAW 264.7 macrophages. Int Immunopharmacol. 2001;1(9–10):1849–55. doi: 10.1016/s1567-5769(01)00110-2. [DOI] [PubMed] [Google Scholar]
- 75.Ho CK, Chen CC. Moscatilin from the orchid Dendrobrium loddigesii is a potential anticancer agent. Cancer Invest. 2003;21(5):729–36. doi: 10.1081/cnv-120023771. [DOI] [PubMed] [Google Scholar]
- 76.Li MF, Hirata Y, Xu GJ, Niwa M, Wu HM. Studies on the chemical constituents of Dendrobium loddigesii rolfe. Yao Xue Xue Bao. 1991;26(4):307–710. [PubMed] [Google Scholar]
- 77.Liu YN, Pan SL, Peng CY, et al. Moscatilin repressed lipopolysaccaride-induced HIF-1alpha accumulation and NF-kappaB activation in murine RAW264.7 cells. Shock. 2009 May 18; doi: 10.1097/SHK.0b013e3181a7ff4a. [DOI] [PubMed] [Google Scholar]
- 78.Chen TH, Pan SL, Guh JH, et al. Moscatilin induces apoptosis in human colorectal cancer cells: a crucial role of c-Jun NH2-terminal protein kinase activation caused by tubulin depolymerization and DNA damage. Clin Cancer Res. 2008;14(13):4250–58. doi: 10.1158/1078-0432.CCR-07-4578. [DOI] [PubMed] [Google Scholar]