Abstract
Non–AIDS-defining cancers are a rising health concern among HIV-infected patients. Cancer screening is now an important component of health maintenance in HIV clinical practice. The decision to screen an HIV-infected patient for cancer should include an assessment of individualized risk for the particular cancer, life expectancy, and the harms and benefits associated with the screening test and its potential outcome. HIV-infected patients are at enhanced risk of several cancers compared to the general population; anal cancer, hepatocellular carcinoma, Hodgkin’s lymphoma, and lung cancer all have good evidence demonstrating an enhanced risk in HIV-infected persons. A number of cancer screening interventions have shown benefit for specific cancers in the general population, but data on the application of these tests to HIV-infected persons are limited. Here we review the epidemiology and background literature relating to cancer screening interventions in HIV-infected persons. We then use these data to inform a conceptual model for evaluating HIV-infected patients for cancer screening.
Keywords: Human immunodeficiency virus (HIV), Cancer screening, Non-AIDS defining cancers (NADCs), Combination antiretroviral therapy(cART), Cervical cancer, Anal cancer, Colorectal cancer, Prostate cancer, Breast cancer, Hepatocellular cancer, Lung cancer, Pap testing, Payoff time, Life-expectancy, High-resolution anoscopy (HRA), Anal cytology, Digital rectal examination (DRE), Mammography, Fecal occult blood testing (FOBT), Ultrasonography, Colonoscopy, Sigmoidoscopy, Alpha-fetoprotein (AFP), Low-dose CT
Introduction
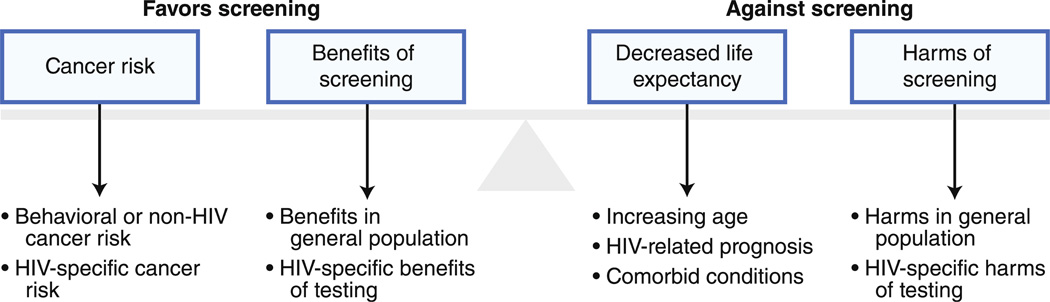
Non–AIDS-related causes of morbidity and mortality are becoming increasingly prevalent in HIV-infected patients and non–AIDS-defining cancers (NADCs) are emerging as a significant source of mortality [1, 2]. Cancer screening, an important consideration in the general population, has become more relevant in an aging HIV-infected population. Although the approach to screening is similar in HIV-infected patients, several important issues should be considered in adapting screening strategies from the general population to those with HIV infection (Fig. 1). Here we describe a general approach to screening and review the data regarding epidemiology and prevention strategies relating to screening for NADCs in HIV-infected patients. This topic was last reviewed in 2009 by Phillips and Justman [3], and we provide a critical evaluation of the updated literature as well as a conceptual framework for screening.
Fig. 1.
Conceptual model for consideration of cancer screening in asymptomatic HIV-infected patients
Conceptual Framework
Screening, as with any diagnostic or therapeutic intervention in a chronic disease management paradigm, should be approached with consideration of the benefits and harms associated with the screening procedure and the outcome in question (Fig. 1) [4]. The assessment of the minimum time until benefits exceed harms for a procedure, or “payoff time,” can help personalize existing screening guidelines for individual patients [5•, 6]. Assessment of three contributing factors is essential for such individualized screening considerations: the patient’s life expectancy based on his or her age and comorbid conditions, an estimate of the patient’s risk of the particular cancer, and an estimate of the benefits and harms of the screening intervention [4].
Life Expectancy
Although combination antiretroviral therapy (cART) has provided significant life expectancy gains for HIV-infected persons, disparities between the life expectancy of HIV-infected persons and the life expectancy of the general population remain [2, 7•, 8, 9]. In a collaborative analysis of persons on cART in 14 cohorts in high-income countries, the average number of years remaining to be lived at age 20 among these persons was about two-thirds that in the general population [7•]. It is important to note that in addition to being at increased risk of death due to HIV-induced immunosuppression, HIV-infected persons are also at increased risk of death due to high prevalence of behavioral risk factors and comorbidities, including drug and alcohol abuse, cigarette smoking, and hepatitis C virus (HCV) infection [8, 9]. Further, those with HIV infection appear to be more likely to continue substance use into older age [10]. Nevertheless, there is considerable variability in life expectancy among subgroups of patients, defined by factors such as CD4 count, HIV viral load, demographic factors, behavioral risk factors, comorbidities, and retention in care [8, 11, 12].
Validated models have been derived to assess prognosis in HIV-infected persons using HIV-related biomarkers [11]. Reflecting the increasing impact of non-AIDS outcomes on prognosis in HIV-infected persons, the significance of other markers representative of long-term sequelae of infection and comorbid illness is now increasingly important [13•, 14, 15]. The ubiquity of electronic medical records and personal data assistants will allow for easier use of these models for rapid clinical decision support.
Incidence of NADCs
From the beginning of the AIDS epidemic, cancer has been closely associated with HIV infection, but the pattern of cancer types found in HIV-infected individuals has shifted in the cART era [16]. AIDS-defining cancers (Kaposi’s sarcoma, non-Hodgkin’s lymphoma, and cervical cancer) with strong links to immunosuppression have declined in incidence with widespread use of cART [17]. Overall cancer risk, however, remains elevated in HIV-infected persons, with estimates of overall NADC risk being twice that of the general population [18••]. The use of cART appears to have had little impact on overall rates of NADCs, and there is growing recognition that an elevated risk of several specific NADCs is responsible for the overall increased cancer risk noted in HIV-infected persons [18••, 19]. This enhanced risk ranges from modest twofold to fivefold elevations in risk for cancers such as melanoma, lung cancer, and hepatocellular carcinoma, to larger tenfold to 30-fold risk elevations for anal cancer and Hodgkin’s lymphoma (Table 1). Some of the increased risk may be explained by a high prevalence of cancer risk factors, such as smoking, alcohol consumption, human papilloma virus (HPV) infection, and HCV infection, among HIV-infected persons [20••].
Table 1.
Cancer risk in HIV-infected persons compared to general population, estimated cancer incidence rates, proposed cancer screening modalities, and benefits and harms of respective screening tests
| Cancer | cART era RR in HIV+ |
Estimated IR in HIV+ (per 100,000 p-y)a |
Screening modalities |
Benefit of screening test |
Harms of screening test |
|---|---|---|---|---|---|
| Cervical | 3.0–13 [18••, 20••] | 24–293 [18••, 20••] | Pap testing | Sensitive, and detects treatable pre-cursor lesions and early cancers; mortality benefit inferred from ecological studies | Minimally invasive, low specificity, leading to false-positive tests |
| HPV testing | Noninvasive but benefits unclear in HIV-infected patients | Low specificity, likely leading to false-positive tests | |||
| Anal | 15–47 [18••, 37•] | 111–130 [18••, 20••] | Anal Pap testing | Sensitive for premalignant lesions, impact on mortality unknown | Minimally invasive, low specificity, leading to false-positive tests |
| HRA | Sensitive and specific for premalignant and malignant lesions, impact on mortality unknown. | Moderately invasive, not widely available | |||
| DRE | Inexpensive, minimally invasive but diagnostic utility not studied | Sensitivity and specificity unknown, harms unquantified | |||
| Breast | 0.64–0.90b [20••, 37•] | 18 [20••] | Mammography | Mortality benefit from RCTs | False positives lead to unnecessary biopsies, psychological distress |
| Colorectal | 1.2b [20••, 37•] | 41 [20••] | FOBT | Mortality benefit from RCTs | False positives lead to moderately invasive testing |
| Sigmoidoscopy | Mortality benefit in recent RCT | Moderately invasive, procedural complications | |||
| Colonoscopy | Mortality benefit from observational data only, but highly likely | Moderately invasive, procedural complications | |||
| Liver | 2.8–7.5 [18••, 37•] | 26–98 [18••, 20••] | Ultrasonography | Mortality benefit shown in HBV mono-infection, inferred benefit in hepatitis co-infection with HIV | False-positive tests may lead to invasive biopsy, treatment options limited in non-transplant candidates |
| AFP testing | Limited benefits | Limited sensitivity and specificity leading to false positives and false negatives | |||
| Prostate | 0.56–1.0b [18••, 37•] | 97–260 [18••, 20••] | PSA testing | Noninvasive with small or no mortality benefits seen in recent RCTs | Low specificity leading to excessive invasive testing and excess treatment |
| Lung | 2.0–3.5 [18••, 37•] | 64–288 [18••, 20••] | Low-dose chest CT | Early RCT data suggesting mortality benefit | Confirmatory testing is invasive, false positives may experience significant harms |
Unadjusted incidence rates
Not statistically significant
AFP alpha-fetoprotein; cART combination antiretroviral therapy; DRE digital rectal examination; FOBT fecal occult blood testing; HBV hepatitis B virus; HPV human papilloma virus; HRA high-resolution anoscopy; IR incidence rate; PSA prostate-specific antigen; p-y person-year; RCT randomized control trial; RR relative risk
Harms/Benefits of Cancer Screening
Recommendations supporting screening interventions must carefully consider the potential benefits and harms of a cancer screening test; the test must identify cancers at a stage at which an intervention can provide increased life expectancy, but also minimize morbidity from false-positive results [4]. Optimally, a screening test is studied in a randomized control trial (RCT) with death as the end point to establish a clear mortality benefit and quantify harms. Because this type of evaluation can be difficult, often screening tests are evaluated using data from nonrandomized or observational studies, which are susceptible to various biases or confounding [4]. Guidelines exist for cancer screening in the general population, which are derived via expert opinion regarding the best available scientific evidence. The cancer screening recommendations of the Unites States Preventive Services Taskforce (USPSTF) are updated regularly and incorporate a rigorous evaluation of the harms and benefits of screening procedures [21]. Screening guidelines are also published by the American Cancer Society and various professional organizations [22].
Limited guidelines have been developed for cancer screening in HIV-infected persons, as very little primary data exist regarding screening interventions specifically for HIV-infected persons (Table 2) [3]. Instead, the benefits of screening among HIV-infected populations have largely been estimated based on extrapolations of data from the HIV-uninfected population. While this extrapolation is likely generally true, there may be important exceptions that would impact the contributing factors that are integral to assessing benefits and harms of screening. For example, in the case of anal and cervical cancer, HIV-infected persons have a greater prevalence of HPV carriage than HIV-uninfected persons, and thus HPV testing as an initial screen may yield unacceptably high rates of false-positive results. Similarly, in the case of hepatocellular cancer, eligibility for cancer treatment (liver transplantation) may differ by HIV status, also potentially altering the benefit and harm profile of the screening modality.
Table 2.
Cancer screening guidelines for HIV-infected individuals [3]
| Cancer type | Screening strategy | Populationa | Frequencya |
|---|---|---|---|
| Anal cancer | Visual inspection of the perianal region and DRE | All HIV-infected adults [25, 26] | Annual [25, 26] |
| Anal cytology (Pap test) | Recommended by some experts in all HIV-infected adults [25] vs recommended for those HIV-infected adults with HPV infection risk [26]: | Baseline and annual; repeat every 3–6 months if abnormal [26] | |
| (Refer for high-resolution anoscopy and/or biopsy for cytology abnormalities including ASCUS and ASC-H) |
|
||
| Cervical cancer | Pap testb | Women ≥18 or when sexually active, whichever comes first | Baseline, 6 months after baseline, then annually [13•, 25, 26, 37•] |
| Colposcopy | Any female with abnormal Pap test result | Case by case basis | |
| Hepatocellular cancer | Abdominal ultrasound and AFP | HBV/HCV co-infected HIV-infected adults (if candidate for liver transplantation) [44] | Every 6–12 months |
| Other non–AIDS-defining malignancies | Clinicians should promote risk-reduction behaviors (eg, smoking cessation) and should adhere to standard, age-appropriate screening recommendations that apply to the non–HIV-infected population [26] |
Reference [25]: US Department of Health and Human Services; reference 26: New York State Department of Health AIDS Institute
There is insufficient evidence for HPV testing; however, there may be a future role of HPV testing to triage need for colposcopy [13•]
AFP alpha-fetoprotein; ASCUS atypical squamous cells of undetermined significance; ASC-H atypical squamous cells, high-grade squamous intraepithelial lesion cannot be excluded; DRE digital rectal examination
Cervical Cancer
Epidemiology of Cervical Cancer in HIV
While invasive cervical cancer (ICC) is an AIDS-defining cancer, we include it in this review because screening approaches for this condition have continued to evolve. ICC is relatively uncommon in the general population with rates of 7 cases per 100,000 person-years (p-y), but was classified an AIDS-defining malignancy early in the AIDS epidemic after recognition of high rates of precursor lesions in women with AIDS [23•, 24]. Large registry linkage studies have estimated standardized incidence rates for ICC in HIV-infected women to be five to six times that of the general population, but these estimates are heavily influenced by pre-cART era data [23•, 25, 26]. The Women’s Interagency Health Study (WIHS) has recently reported no increased risk of ICC in HIV-infected compared with HIV-uninfected women, potentially reflecting improved screening practices or a protective effect associated with cART use [27•, 28]. In contrast, a large cART era study in the Kaiser system found continued elevated risk [20••].
Benefits/Harms of Screening for Cervical Cancer
Papanicolaou (Pap) cytology testing, the primary method of screening for cervical cancer, has not been evaluated in the general population in a prospective RCT, although strong observational data support its impact on both cervical cancer incidence rates and cervical cancer–related mortality [29]. A large European and South African cohort study of HIV-infected women evaluated the efficacy of Pap testing compared to a gold standard of biopsy and found the former to be highly sensitive for high-grade cervical intraepithelial neoplasia (CIN) [30], which is known to progress to ICC. Recently published results from a retrospective natural history study of high-risk lesions in a general population of women showed a median time of progression of 27 years [31]. It is unknown if this transition is accelerated in HIV-infected women. Premalignant cervical lesions as well as early ICC are treatable, often with local excision, and are associated with markedly improved prognosis when compared to late disease [32]. Harms associated with Pap testing are limited, and are primarily associated with complications and discomfort from colposcopy and biopsy as well as the psychosocial distress associated with false-positive results [33].
Cervical HPV testing has been proposed to augment or potentially substitute for cytology as a primary method of screening, as high-risk HPV (hrHPV) strains are associated with the development of ICC [34]. Although strategies utilizing HPV testing have shown merit in the general population, there is little evidence to suggest any advantage in HIV-infected women, as baseline prevalences of hrHPV strains are high, limiting the discriminatory power of testing [30].
Current recommendations published by the USPSTF for the general population are that all women be screened with Pap testing within 3 years of onset of sexual activity or age 21, and that follow-up screening occur every 3 years [33]. HIV-specific guidelines, proposed by the Centers for Disease Control and Prevention, Department of Health and Human Services, and New York State, suggest more frequent Pap testing: upon HIV diagnosis, 6 months later, and then annually [3, 35]. It is likely, however, that HIV-infected women on a stable cART regimen with viral suppression can be safely monitored in a fashion similar to HIV-uninfected women [28, 36].
Anal Cancer
Epidemiology of Anal Cancer in HIV
Anal squamous cell carcinoma (SCC) is an uncommon cancer in the general population, but is now increasingly recognized as an important source of morbidity and mortality in HIV-infected persons. Two meta-analyses have reported an approximately 30-fold increased risk of the disease in HIV-infected persons [37•, 38]. Additionally, incidence of anal cancer appears to have initially increased in the cART era, possibly due to increased survival of at-risk patients, but later appears to have plateaued [39, 40]. HIV-infected men who have sex with men (MSM) appear to be at highest risk [39, 40], although there is evidence of an independent association of anal cancer with HIV infection [41].
Anal cancer is clearly linked to infection with hrHPV [42, 43]. It is also suspected that immunosuppression augments the development of anal squamous cell dysplasia. This is supported by the observation of higher rates of precursor lesions as well as anal SCC in HIV-infected persons in lower CD4 strata in several cohorts [18••, 44, 45].
The pathogenesis of anal cancer is an important consideration in the approach to screening for this disease. Cancers arise from precursor high-grade anal intraepithelial lesions (AIN) within the anal canal [46]. Time of progression to cancer is controversial, but one observational cohort of HIV-infected MSM noted a median time of progression of high-grade AIN to cancer of 8.6 months in participants who refused high-resolution anoscopy (HRA) [47•]. The natural history of low-grade AIN is less clear.
Benefits/Harms of Screening for Anal Cancer
No RCTs have been performed to evaluate screening methods for anal cancer; thus much of the rationale behind screening regimens are inferred from the cervical cancer screening literature. The commonly proposed screening methods for detecting high-grade AIN include anal cytology testing and HRA. Digital rectal examination (DRE), which detects condylomata and palpable cancers, has also been proposed as a screening modality for anal cancer [48].
The performance of anal cytology has been evaluated in a number of observational studies. Cytology testing in which cytologic abnormalities of atypical squamous cells of uncertain significance and higher are referred for HRA is 69%–93% sensitive for high-grade AIN [48, 49]. Using a threshold of higher-grade cytologic abnormalities allows for a greater specificity but progressively poorer sensitivity [48]. Alternatively, preliminary data have suggested that in HIV-infected MSM, the highest anal cancer risk group, the most cost-effective screening modality may be primary screening with HRA [50•]. This is the most sensitive and specific test available for high-grade AIN [51]; however, the limited availability of practitioners makes this strategy difficult to implement widely.
As with cervical cancer screening for women, testing for hrHPVas a screening modality for anal cancer appears to have limited benefit in HIV-infected men and women. The presence of hrHPV has a very high sensitivity but low specificity for high-grade AIN because of high prevalence of hrHPV carriage in HIV-infected persons [49]. It is therefore minimally useful in the decision-making process for triage of cytology findings or HRA referral.
No guidelines exist for anal cancer screening in the general population. The New York State AIDS Institute has recommended baseline and annual screening with anal cytology in HIV-infected MSM, women, and persons with a history of anal condylomata. Additionally, DRE is recommended in all persons with HIV on an annual basis [52].
The benefits of anal cancer screening in HIV-infected persons have not been well established. When high-grade AIN is detected, lesions can be treated by a variety of methods, such as imiquimod (a topical immune response modulator with known activity in HPV-related disease) or ablative therapy, which appear to mitigate development of invasive disease [53, 54]. Identification of early-stage carcinoma also appears to be beneficial, as it carries a 5-year relative survival rate of 80% compared to 30% in late-stage disease [55]. Harms related to screening include anxiety related to false-positive cytology testing [56] and procedural discomfort from HRA and biopsy. Nonetheless, the incidence of anal cancer in HIV-infected persons is significant, and screening of high-risk groups with cytology or HRA should be considered while further studies of the harms and benefits are underway.
Breast Cancer
Epidemiology of Breast Cancer in HIV
Breast cancer is the most common malignancy (apart from non-melanoma skin cancer) in women in the general population [57]. Data from several sources in the pre-cART era suggested that HIV-infected women might be at lower risk of breast cancer [26, 58]. A large registry linkage study of cART-era data has found that breast cancer risk in HIV-infected women now mirrors that of the general population [59]. Additionally, a recent nested case-control from the WIHS and HIV Epidemiology Research Study cohorts has shown that low breast cancer risk may be linked to infection with CXCR-4 tropic virus [60]. This could explain why lower risk was observed in the pre-cART era, but not in the cART era, when virus, including CXCR-4 tropic virus, is better controlled.
Benefits/Harms of Screening for Breast Cancer
The USPSTF published an update to their breast cancer screening recommendations in 2009 for the general population, recommending biennial mammography for women ages 50–74 years based on a survival benefit noted in their meta-analysis of RCTs, and discouraging routine screening of younger women [61]. They reported that evidence regarding benefit of other screening methodologies including MRI and self breast-examination was either inconclusive or nonsupportive. The recommendation regarding women ages 40–49 years was controversial, as evidence exists demonstrating a mortality benefit of screening in this population. However, it was felt that the harms associated with screening were excessive due to the high rate of false-positive mammograms in this age group, which can lead to additional testing, unnecessary biopsies, and substantial anxiety for the patients involved [62]. Application of USPSTF or other national breast cancer screening guidelines to HIV-infected women seems appropriate, provided prognosis conferred by HIV or other comorbidities is considered in the decision-making process.
Colorectal Cancer
Epidemiology of Colorectal Cancer in HIV
Colorectal cancer is the third leading cause of cancer death among both men and women in the general population [57]. In persons with HIV, there is no evidence of an increased risk of colorectal cancers in either cART-era cohort studies or registry linkage studies [18••, 20••, 38]. A single-institution cohort of more than 1000 HIV-infected and uninfected persons undergoing colonoscopies found no differences in the prevalence of adenomatous polyps or cancers [63•]; however, little is known regarding rates of colorectal cancer screening among HIV-infected persons.
Benefits/Harms of Screening for Colorectal Cancer
Colorectal cancer is especially amenable to screening, as premalignant adenomas exhibit a slow progression to malignancy, and are often visibly identifiable and treatable via colonoscopy. Despite these advantages, half of colorectal cancers present at advanced stage, likely due to inadequate adherence to screening and barriers to health care utilization present in some populations [64]. Fecal occult blood testing (FOBT) and sigmoidoscopy, with evidence from RCTs [65•], and colonoscopy, with evidence from its integral role in FOBT trials and extrapolation from studies supporting sigmoidoscopy, are all recommended in patients ages 50–75 years by the USPSTF [21]. Patients older than 75 years do not benefit from screening given the long latency period associated with the development of colorectal cancer. Other screening modalities including CT colonography and fecal DNA testing do not have adequate evidence documenting harms and benefits and are not supported by the USPSTF [21]. Harms of colorectal cancer screening by colonoscopy, sigmoidoscopy, and FOBT are primarily related to morbidity associated with complications of endoscopy, including intestinal perforation.
Colorectal cancer screening guidelines specific to HIV-infected persons echo recommendations for the general population [66]. The long latency period associated with colorectal cancer confers a relatively lengthy payoff time of approximately 6–7 years [5•]. Life expectancy and presence of severe comorbid illnesses should therefore be considered in the decision to screen HIV-infected persons.
Prostate Cancer
Epidemiology of Prostate Cancer in HIV
Prostate cancer has the highest incidence of any cancer (except non-melanoma skin cancer) and is the second leading cause of cancer death in men [57]. There has been consistent reporting of a decreased risk of prostate cancer in cART-era HIV-infected men [18••, 20••, 37•]. Shiels et al. [67•] found that in the Johns Hopkins HIV Clinical Cohort (JHHCC) this decreased incidence could largely be explained by a lower rate of prostate-specific antigen (PSA) testing among HIV-infected men. However, the lower prostate cancer incidence rate in HIV-infected men in the Kaiser Permanente Northern California cohort was not explained by lower PSA testing rates, with higher testing rates observed in the HIV-infected men in this cohort [68].
Benefits/Harms of Screening for Prostate Cancer
Although PSA screening for prostate cancer is highly prevalent [67•], there is little consensus among national recommendations regarding screening for prostate cancer in the general population. The most recent USPSTF recommendations, published in 2008, concluded that current evidence was insufficient to assess the balance of benefits and harms of PSA screening in men younger than age 75 years and recommended against PSA screening in men age 75 years or older [69]. Subsequently, two large RCTs of prostate cancer screening, the European Randomized Study of Screening for Prostate Cancer (ERSSCP) and the Prostate, Lung, Colorectal, and Ovarian (PLCO) Cancer Screening Trial, have reported results. The PLCO reported no benefit to prostate cancer screening and the ERSSCP found a small mortality benefit that was achieved at the cost of significant over-diagnosis and over-treatment resulting in appreciable morbidity, including impotence and incontinence [70••, 71••].
There are little data to suggest different clinical characteristics of prostate cancer in HIV-infected men; data from the JHHCC have demonstrated no difference in prognosis between HIV-infected prostate cancer patients and the general population, despite lower rates of screening in the former [67•]. In sum, neither data from the general population nor data from those with HIV infection currently support widespread screening for prostate cancer.
Hepatocellular Carcinoma
Hepatocellular carcinoma (HCC) is an uncommon cancer in the general population, occurring primarily in persons with underlying liver disease [57]. HCC occurs most commonly in patients with advanced liver fibrosis, of which the most common etiologies are chronic viral hepatitis B or C (HBV, HCV), or alcohol-related liver injury [72]. In patients with chronic viral hepatitis, coinfection with HIV appears to accelerate fibrosis, exacerbating HCC risk [73]. Pooled cART-era incidence data suggest an approximately fivefold to sixfold risk increase for HCC in HIV-infected persons compared to the general population [37•]. This is explained in part by higher rates of viral hepatitis (HBV and/or HCV) among HIV-infected persons, but HIV-induced immunosuppression may be involved as well [18••, 44].
Benefits/Harms of Screening for Hepatocellular Cancer
There is limited guidance regarding screening for HCC in the general population. The American Association for the Study of Liver Diseases is the only national organization to have published HCC screening guidelines, with recommendations issued in 2005, advocating liver ultrasonography every 6 months in groups at high risk for HCC [72]. The strongest evidence supporting these guidelines is a large Chinese RCT of HCC screening in patients with chronic HBV infection. This trial demonstrated a 37% mortality reduction with the use of biennial liver ultrasonography and alpha-fetoprotein (AFP) testing [74]. Application of this result to HIV patients co-infected with viral hepatitis in developed countries is difficult, however, as the patients in the Chinese trial were much less likely to exhibit advanced liver disease, and were HBV co-infected, whereas HCV infection predominates in the US. Harms associated with ultrasonography as a screening test include excess radiation and contrast dye exposure associated with follow-up imaging in false-positive tests, as well as potential complications of subsequent biopsy [75].
Testing for AFP, a tumor marker associated with HCC, has been studied as an adjunct to radiographic screening. However, the AFP test has been found to lack sufficient sensitivity or specificity to be useful as a screening test [76]. It is therefore not included in more recent screening protocols [72].
Screening interventions for HCC may be useful when the disease is at an early stage, and although non-cirrhotic patients may be eligible for limited surgical resections, most patients require liver transplantation for cure [72]. Incident HCC in HIV patients occurs primarily in the setting of cirrhosis (95%), and therefore patients who are considered for screening should also be eligible for liver transplantation [77]. A survival benefit for HIV-uninfected persons has been demonstrated in the use of liver transplantation for early HCC [72], and because there is limited treatment available for later-stage disease, eligible HIV-infected persons should likely be considered for screening with biennial liver ultrasonography.
Lung Cancer
Epidemiology of Lung Cancer in HIV
Lung cancer will likely become the leading cause of cancer-related mortality in HIV-infected persons [1], as it is in the general population [37•]. HIV-infected persons are at greater risk of lung cancer than HIV-uninfected persons, and several adjusted analyses suggest that this risk is only partially explained by excess smoking observed in HIV-infected persons [78, 79]. As in HIV-uninfected populations, HIV-infected current smokers and those who have quit in the recent past have the highest risk of lung cancer [78].
Benefits/Harms of Screening for Lung Cancer
Early-stage lung cancers are treatable with reasonable prognosis, whereas late-stage lung cancers have an extremely poor prognosis [80]. To decrease harm associated with screening, lung cancer screening tests must have high positive predictive value, as positive testing leads to invasive biopsy via surgery or bronchoscopy. Thus, screening is most likely to benefit high-risk groups, primarily heavy smokers. Studies have consistently documented a higher prevalence of smoking among persons with HIV than in the general population [78, 81].
Lung cancer screening is not currently recommended in the general population by the USPSTF [21]. Evaluations of chest radiographs and sputum cytology as lung cancer screening modalities in RCTs have not shown adequate benefits to justify harms [21]. Lung cancer screening of smokers with low-dose CT (ldCT) scanning has been evaluated in several studies and appears to be highly sensitive and specific [82]. The National Lung Screening Trial (NLST), a large RCT comparing ldCT scanning to chest radiographs in heavy smokers, was recently stopped early because of a 20% mortality benefit in the ldCT arm [83]. The full description of these results has not yet been published, but this suggests a potential benefit of lung cancer screening.
HIV-infected persons, many of whom are heavy smokers, have a modestly increased risk of lung cancer compared to HIV-uninfected persons. Recommendations regarding any benefits of screening among HIV-infected patients at this time are limited, given a lack of data on the rate of false-positive findings that may be found on ldCT scanning in this population. Pending the final results of the NLST, periodic screening with ldCT scans may be a beneficial screening intervention for heavy smokers; whether this approach will be beneficial for HIV-infected smokers remains to be determined.
Conclusions
Limited primary data exist regarding harms and benefits of cancer screening interventions specific to HIV-infected persons. Current data suggest that the approach to screening for three common cancers (anal, liver, and lung cancer) may require adaptation for those with HIV infection because of increased risk associated with HIV as well as behaviors and conditions common among those with HIV. Anal cancer screening with anal cytology, HRA, and DRE as well as HCC screening with ultrasonography are currently supported by HIV-specific screening guidelines [66], although harms and benefits associated with these tests in HIV-infected patients are not clear. Liver cancer screening is potentially indicated among HIV-infected individuals with evidence of liver injury that are eligible for liver transplant. Lung cancer screening is not widely supported, although emerging data may show potential benefits in certain risk groups such as those with HIV infection.
Screening recommendations for the general population can be applied to HIV-infected patients for certain cancers, including Pap testing in cervical cancer screening, mammography in breast cancer screening, and FOBT, sigmoidoscopy, and colonoscopy in colorectal cancer screening. Finally, harms may outweigh the benefits of prostate cancer screening in the general population, and this is also likely true in HIV-infected men.
NADCs represent a significant source of morbidity and mortality in the aging HIV-infected population. However, the decision to screen HIV-infected persons for cancer is complex and should include consideration of the risk of the particular cancer, the life expectancy of the patient, and the specific benefits and harms that may stem from the screening intervention. Further studies are therefore needed to quantify the harms and benefits of cancer screening interventions in HIV-infected persons.
Acknowledgments
This work was supported by the following National Institutes of Health grants supporting Drs. Justice, Dubrow, and Crothers: NIAAA (U10-AA13566), NHLBI (R01-HL090342; RCI-HL100347), and NIAID (U01-A1069918). Dr. Silverberg's effort was supported in part by NIAID (K01AI071725).
Footnotes
Disclosure No potential conflicts of interest relevant to this article were reported.
Contributor Information
Keith Sigel, Email: keith.sigel@mssm.edu, Division of Infectious Diseases, Mount Sinai School of Medicine, 1 Gustave Levy Place, New York, NY 10029, USA.
Robert Dubrow, Yale School of Public Health, Yale School of Medicine, New Haven, CT, USA.
Michael Silverberg, Kaiser Permanente Northern California, Division of Research, Oakland, CA, USA.
Kristina Crothers, University of Washington School of Medicine, Seattle, WA, USA.
Scott Braithwaite, New York University Langone Medical Center, New York, NY, USA.
Amy Justice, VA Connecticut Healthcare System and Yale University Schools of Medicine and Public Health, New Haven, CT, USA.
References
Papers of particular interest, published recently, have been highlighted as:
• Of importance
•• Of major importance
- 1.Simard EP, Engels EA. Cancer as a cause of death among people with AIDS in the United States. Clin Infect Dis. 2010;51:957–962. doi: 10.1086/656416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lewden C, May T, Rosenthal E, et al. Changes in causes of death among adults infected by HIV between 2000 and 2005: The "Mortalite 2000 and 2005" surveys (ANRS EN19 and Mortavic) J Acquir Immune Defic Syndr. 2008;48:590–598. doi: 10.1097/QAI.0b013e31817efb54. [DOI] [PubMed] [Google Scholar]
- 3.Phillips AA, Justman JE. Screening HIV-infected patients for non-AIDS-defining malignancies. Curr HIV/AIDS Rep. 2009;6:83–92. doi: 10.1007/s11904-009-0013-0. [DOI] [PubMed] [Google Scholar]
- 4.Croswell JM, Ransohoff DF, Kramer BS. Principles of cancer screening: lessons from history and study design issues. Semin Oncol. 2010;37:202–215. doi: 10.1053/j.seminoncol.2010.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Braithwaite RS, Fiellin D, Justice AC. The payoff time: a flexible framework to help clinicians decide when patients with comorbid disease are not likely to benefit from practice guidelines. Med Care. 2009;47:610–617. doi: 10.1097/MLR.0b013e31819748d5. This article describes "payoff time," a conceptual approach to tailoring cancer screening guidelines to patients with significant comorbid illness.
- 6.Justice AC. Prioritizing primary care in HIV: comorbidity, toxicity, and demography. Top HIV Med. 2006;14:159–163. [PubMed] [Google Scholar]
- 7. Life expectancy of individuals on combination antiretroviral therapy in high-income countries: a collaborative analysis of 14 cohort studies. Lancet. 2008;372:293–299. doi: 10.1016/S0140-6736(08)61113-7. This analysis provides information on life expectancy in HIV patients on cART depending on age and CD4 count at treatment initiation.
- 8.Losina E, Schackman BR, Sadownik SN, et al. Racial and sex disparities in life expectancy losses among HIV-infected persons in the united states: impact of risk behavior, late initiation, and early discontinuation of antiretroviral therapy. Clin Infect Dis. 2009;49:1570–1578. doi: 10.1086/644772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Harrison KM, Song R, Zhang X. Life expectancy after HIV diagnosis based on national HIV surveillance data from 25 states, United States. J Acquir Immune Defic Syndr. 2010;53:124–130. doi: 10.1097/QAI.0b013e3181b563e7. [DOI] [PubMed] [Google Scholar]
- 10.Green TC, Kershaw T, Lin H, et al. Patterns of drug use and abuse among aging adults with and without HIV: a latent class analysis of a US Veteran cohort. Drug Alcohol Depend. 2010;110:208–220. doi: 10.1016/j.drugalcdep.2010.02.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.May M, Sterne JA, Sabin C, et al. Prognosis of HIV-1-infected patients up to 5 years after initiation of HAART: collaborative analysis of prospective studies. AIDS. 2007;21:1185–1197. doi: 10.1097/QAD.0b013e328133f285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lewden C, Chene G, Morlat P, et al. HIV-infected adults with a CD4 cell count greater than 500 cells/mm3 on long-term combination antiretroviral therapy reach same mortality rates as the general population. J Acquir Immune Defic Syndr. 2007;46:72–77. doi: 10.1097/QAI.0b013e318134257a. [DOI] [PubMed] [Google Scholar]
- 13. Justice AC, McGinnis KA, Skanderson M, et al. Towards a combined prognostic index for survival in HIV infection: the role of 'non-HIV' biomarkers. HIV Med. 2010;11:143–151. doi: 10.1111/j.1468-1293.2009.00757.x. This article describes the derivation and validation of a prognostic index strongly correlated with survival for HIV-infected patients utilizing both HIV-specific and non–HIV-related biomarkers.
- 14.Justice A, Modur S, Tate J, et al. A Prognostic Index for those Aging with HIV: Extension of the VACS Index to those on Combination Anti-Retroviral Therapy (cART) [abstract #793]. Presented at the 18th Conference on Retroviruses and Opportunistic Infections; Boston, MA. 2011. [Google Scholar]
- 15.Brown ST, Kyriakides T, Kirkwood K, et al. The VACS Risk Index responds to treatment interventions and is highly correlated with and previctive of mortality events in the OPTIMA study [Poster 16436]. Presented at the 14th International Workshop on HIV Observational Databases.2010. [Google Scholar]
- 16.Engels EA, Biggar RJ, Hall HI, et al. Cancer risk in people infected with human immunodeficiency virus in the United States. Int J Cancer. 2008;123:187–194. doi: 10.1002/ijc.23487. [DOI] [PubMed] [Google Scholar]
- 17.Seaberg EC, Wiley D, Martinez-Maza O, et al. Cancer incidence in the multicenter aids cohort study before and during the HAART era, 1984 to 2007. Cancer. 2010;116:5507–5516. doi: 10.1002/cncr.25530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Bedimo RJ, McGinnis KA, Dunlap M, et al. Incidence of non-AIDS-defining malignancies in HIV-infected versus noninfected patients in the HAART era: impact of immunosuppression. J Acquir Immune Defic Syndr. 2009;52:203–228. doi: 10.1097/QAI.0b013e3181b033ab. This article provides excellent estimates of cART era NADC risk and incidence rates using data from a large US HIV cohort.
- 19.Silverberg MJ, Abrams DI. AIDS-defining and non-AIDS-defining malignancies: cancer occurrence in the antiretroviral therapy era. Curr Opin Oncol. 2007;19:446–451. doi: 10.1097/CCO.0b013e3282c8c90d. [DOI] [PubMed] [Google Scholar]
- 20. Silverberg MJ, Chao C, Leyden WA, et al. HIV infection and the risk of cancers with and without a known infectious cause. AIDS. 2009;23:2337–2345. doi: 10.1097/QAD.0b013e3283319184. This study provides excellent estimates of cARTera NADC risk and incidence rates using data from a large US HIV cohort.
- 21.Recommendations of the United States Preventive Services Task Force. [Accessed March 2011]; Available at Available at http://www.uspreventiveservicestaskforce.org. Accessed. [Google Scholar]
- 22.Smith RA, Cokkinides V, Brooks D, et al. Cancer screening in the United States, 2010: a review of current American Cancer Society guidelines and issues in cancer screening. CA Cancer J Clin. 2010;60:99–119. doi: 10.3322/caac.20063. [DOI] [PubMed] [Google Scholar]
- 23. Chaturvedi AK, Madeleine MM, Biggar RJ, Engels EA. Risk of human papillomavirus-associated cancers among persons with AIDS. J Natl Cancer Inst. 2009;101:1120–1130. doi: 10.1093/jnci/djp205. This article describes the results of a large registry linkage study of HPV-related cancer incidence (1980–2004) in persons with AIDS.
- 24.1993 revised classification system for HIV infection and expanded surveillance case definition for AIDS among adolescents and adults. MMWR Recomm Rep. 1993;41:1–19. [PubMed] [Google Scholar]
- 25.Serraino D, Carrieri P, Pradier C, et al. Risk of invasive cervical cancer among women with, or at risk for, HIV infection. Int J Cancer. 1999;82:334–337. doi: 10.1002/(sici)1097-0215(19990730)82:3<334::aid-ijc5>3.0.co;2-c. [DOI] [PubMed] [Google Scholar]
- 26.Frisch M, Biggar RJ, Engels EA, Goedert JJ. Association of cancer with AIDS-related immunosuppression in adults. JAMA. 2001;285:1736–1745. doi: 10.1001/jama.285.13.1736. [DOI] [PubMed] [Google Scholar]
- 27. Massad LS, Seaberg EC, Watts DH, et al. Long-term incidence of cervical cancer in women with human immunodeficiency virus. Cancer. 2009;115:524–530. doi: 10.1002/cncr.24067. This article describes cervical cancer rates in HIV-infected and uninfected participants in the WIHS study, finding no increased risk in the HIV-infected participants.
- 28.Minkoff H, Zhong Y, Burk RD, et al. Influence of adherent and effective antiretroviral therapy use on human papillomavirus infection and squamous intraepithelial lesions in human immunodeficiency virus-positive women. J Infect Dis. 2010;201:681–690. doi: 10.1086/650467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Screening for squamous cervical cancer: duration of low risk after negative results of cervical cytology and its implication for screening policies. IARC Working Group on evaluation of cervical cancer screening programmes. Br Med J (Clin Res Ed) 1986;293:659–664. doi: 10.1136/bmj.293.6548.659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kitchener H, Nelson L, Adams J, et al. Colposcopy is not necessary to assess the risk to the cervix in HIV-positive women: an international cohort study of cervical pathology in HIV-1 positive women. Int J Cancer. 2007;121:2484–2491. doi: 10.1002/ijc.22947. [DOI] [PubMed] [Google Scholar]
- 31.McCredie MR, Sharples KJ, Paul C, et al. Natural history of cervical neoplasia and risk of invasive cancer in women with cervical intraepithelial neoplasia 3: a retrospective cohort study. Lancet Oncol. 2008;9:425–434. doi: 10.1016/S1470-2045(08)70103-7. [DOI] [PubMed] [Google Scholar]
- 32.Ries LAKC, Hankey BF, Miller BA, editors. SEER cancer statistics review, 1973–1996: tables and graphs: M.N.C.I.N.P.N. 1999. [Google Scholar]
- 33.Recommendations of the United States Preventive Services Task Force: Cervical cancer screening. [Accessed March 2011]; Available at http://www.uspreventiveservicestaskforce.org. [Google Scholar]
- 34.Clifford GM, Smith JS, Plummer M, et al. Human papillomavirus types in invasive cervical cancer worldwide: a meta-analysis. Br J Cancer. 2003;88:63–73. doi: 10.1038/sj.bjc.6600688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.AIDS Institute of New York State Guidelines: Neoplastic complications of HIV: Cervical Cancer. [Accessed March 2011]; Available at http://www.hivguidelines.org/clinical-guidelines/adults/neoplastic-complications-of-hiv-infection/. [Google Scholar]
- 36.Harris TG, Burk RD, Palefsky JM, et al. Incidence of cervical squamous intraepithelial lesions associated with HIV serostatus, CD4 cell counts, and human papillomavirus test results. JAMA. 2005;293:1471–1476. doi: 10.1001/jama.293.12.1471. [DOI] [PubMed] [Google Scholar]
- 37. Shiels MS, Cole SR, Kirk GD, Poole C. A meta-analysis of the incidence of non-AIDS cancers in HIV-infected individuals. J Acquir Immune Defic Syndr. 2009;52:611–622. doi: 10.1097/QAI.0b013e3181b327ca. This article describes the results of a large registry linkage study of age at cancer diagnosis in persons with AIDS, finding younger ages at presentation with lung cancer and anal cancer, and similar ages to the general population at presentation for other cancers.
- 38.Grulich AE, van Leeuwen MT, Falster MO, Vajdic CM. Incidence of cancers in people with HIV/AIDS compared with immunosuppressed transplant recipients: a meta-analysis. Lancet. 2007;370:59–67. doi: 10.1016/S0140-6736(07)61050-2. [DOI] [PubMed] [Google Scholar]
- 39.Silverberg MJ, Lau B, Y J, et al. Risk of anal cancer in HIV-infected patients and HIV-uninfected controls in North America. Presented at the 12th International Conference on Malignancies in AIDS and Other Acquired Immunodeficiencies; April 2010; Bethesda, MD. [Google Scholar]
- 40.Piketty C, Selinger-Leneman H, Grabar S, et al. Marked increase in the incidence of invasive anal cancer among HIV-infected patients despite treatment with combination antiretroviral therapy. AIDS. 2008;22:1203–1211. doi: 10.1097/QAD.0b013e3283023f78. [DOI] [PubMed] [Google Scholar]
- 41.D'Souza G, Wiley DJ, Li X, et al. Incidence and epidemiology of anal cancer in the multicenter AIDS cohort study. J Acquir Immune Defic Syndr. 2008;48:491–499. doi: 10.1097/QAI.0b013e31817aebfe. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Frisch M, Fenger C, van den Brule AJ, et al. Variants of squamous cell carcinoma of the anal canal and perianal skin and their relation to human papillomaviruses. Cancer Res. 1999;59:753–757. [PubMed] [Google Scholar]
- 43.Hoots BE, Palefsky JM, Pimenta JM, Smith JS. Human papillomavirus type distribution in anal cancer and anal intraepithelial lesions. Int J Cancer. 2009;124:2375–2383. doi: 10.1002/ijc.24215. [DOI] [PubMed] [Google Scholar]
- 44.Guiguet M, Boue F, Cadranel J, et al. Effect of immunodeficiency, HIV viral load, and antiretroviral therapy on the risk of individual malignancies (FHDH-ANRS CO4): a prospective cohort study. Lancet Oncol. 2009;10:1152–1159. doi: 10.1016/S1470-2045(09)70282-7. [DOI] [PubMed] [Google Scholar]
- 45.de Pokomandy A, Rouleau D, Ghattas G, et al. HAART and Progression to High-Grade Anal Intraepithelial Neoplasia in Men Who Have Sex with Men and Are Infected with HIV. Clin Infect Dis. 2011 doi: 10.1093/cid/cir064. Epub ahead of print. [DOI] [PubMed] [Google Scholar]
- 46.Watson AJ, Smith BB, Whitehead MR, et al. Malignant progression of anal intra-epithelial neoplasia. ANZ J Surg. 2006;76:715–717. doi: 10.1111/j.1445-2197.2006.03837.x. [DOI] [PubMed] [Google Scholar]
- 47. Kreuter A, Potthoff A, Brockmeyer NH, et al. Anal carcinoma in human immunodeficiency virus-positive men: results of a prospective study from Germany. Br J Dermatol. 2010;162:1269–1277. doi: 10.1111/j.1365-2133.2010.09712.x. This is a prospective study of HIV-infected MSM screened for anal cancer with anal cytology and subsequent HRA when indicated. It provides an estimate of rates of AIN and the correlation of these findings with cytology and HPV testing, as well as an estimate of the progression time of AIN to invasive cancer.
- 48.Darragh TM, Winkler B. Anal cancer and cervical cancer screening: Key differences. Cancer Cytopathol. 2011;119:5–19. doi: 10.1002/cncy.20126. [DOI] [PubMed] [Google Scholar]
- 49.Berry JM, Palefsky JM, Jay N, et al. Performance characteristics of anal cytology and human papillomavirus testing in patients with high-resolution anoscopy-guided biopsy of high-grade anal intraepithelial neoplasia. Dis Colon Rectum. 2009;52:239–247. doi: 10.1007/DCR.0b013e31819793d9. [DOI] [PubMed] [Google Scholar]
- 50. Lam JM, Hoch JS, Tinmouth J, et al. Cost-effectiveness of screening for anal precancers in HIV-positive men. AIDS. 2011;25:635–642. doi: 10.1097/QAD.0b013e3283434594. This article describes a cost-effectiveness analysis using data from a cross-sectional study of HIV-infected MSM who received concurrent screening with anal cytology, HRA, and HPV testing, finding HRA to be the most cost-effective modality.
- 51.Salit IE, Lytwyn A, Raboud J, et al. The role of cytology (Pap tests) and human papillomavirus testing in anal cancer screening. AIDS. 2010;24:1307–1313. doi: 10.1097/QAD.0b013e328339e592. [DOI] [PubMed] [Google Scholar]
- 52.Neoplastic complications of HIV: Anal Cancer. [Accessed March 15, 2011]; Available at http://www.hivguidelines.org/clinical-guidelines/adults/neoplastic-complications-of-hiv-infection/. [Google Scholar]
- 53.Park IU, Palefsky JM. Evaluation and Management of Anal Intraepithelial Neoplasia in HIV-Negative and HIV-Positive Men Who Have Sex with Men. Curr Infect Dis Rep. 2010;12:126–133. doi: 10.1007/s11908-010-0090-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Fox PA, Nathan M, Francis N, et al. A double-blind, randomized controlled trial of the use of imiquimod cream for the treatment of anal canal high-grade anal intraepithelial neoplasia in HIV-positive MSM on HAART, with long-term follow-up data including the use of open-label imiquimod. AIDS. 2010;24:2331–2335. doi: 10.1097/QAD.0b013e32833d466c. [DOI] [PubMed] [Google Scholar]
- 55.Surveillance, Epidemiology and End Results Cancer Stat Fact Sheets. [Accessed Accessed March 15, 2011]; Available at http://seer.cancer.gov/statfacts/html/anus.html.
- 56.Tinmouth J, Raboud J, Ali M, et al. The psychological impact of being screened for anal cancer in HIV-infected men who have sex with men. Dis Colon Rectum. 54:352–359. doi: 10.1007/DCR.0b013e31820349c1. [DOI] [PubMed] [Google Scholar]
- 57.Jemal A, Siegel R, Xu J, Ward E. Cancer statistics, 2010. CA Cancer J Clin. 2010;60:277–300. doi: 10.3322/caac.20073. [DOI] [PubMed] [Google Scholar]
- 58.Goedert JJ, Cote TR, Virgo P, et al. Spectrum of AIDS-associated malignant disorders. Lancet. 1998;351:1833–1839. doi: 10.1016/s0140-6736(97)09028-4. [DOI] [PubMed] [Google Scholar]
- 59.Goedert JJ, Schairer C, McNeel TS, et al. Risk of breast, ovary, and uterine corpus cancers among 85,268 women with AIDS. Br J Cancer. 2006;95:642–648. doi: 10.1038/sj.bjc.6603282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Hessol NA, Napolitano LA, Smith D, et al. HIV tropism and decreased risk of breast cancer. PLoS One. 2010;5:e14349. doi: 10.1371/journal.pone.0014349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Recommendations of the United States Preventive Services Task Force: Breast Cancer Screening. [Accessed Accessed March 2011]; Available at Available at http://www.uspreventiveservicestaskforce.org. [Google Scholar]
- 62.Nelson HD, Tyne K, Naik A, et al. Screening for breast cancer: an update for the U.S. Preventive Services Task Force. Ann Intern Med. 2009;151:727–737. W237–W742. doi: 10.1059/0003-4819-151-10-200911170-00009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Kothari ND, Engelson ES, Drake V, et al. Effect of HIV infection on the prevalence of colorectal adenomas during screening colonoscopy. J Clin Gastroenterol. 2010;44:77–78. doi: 10.1097/MCG.0b013e3181a1f5b5. This is a cross sectional study of a large group of HIV-infected and uninfected patients undergoing colonoscopy; no difference in rates of adenomas was observed in either group.
- 64.Henley SJ, King JB, German RR, et al. Surveillance of screening-detected cancers (colon and rectum, breast, and cervix) - United States, 2004–2006. MMWR Surveill Summ. 2010;59:1–25. [PubMed] [Google Scholar]
- 65. Atkin WS, Edwards R, Kralj-Hans I, et al. Once-only flexible sigmoidoscopy screening in prevention of colorectal cancer: a multicentre randomised controlled trial. Lancet. 2010;375:1624–1633. doi: 10.1016/S0140-6736(10)60551-X. This is a recently published RCT showing a mortality benefit associated with colorectal cancer screening by sigmoidoscopy.
- 66.AIDS Institute of New York State Guidelines: Neoplastic complications of HIV - Colorectal cancer screening. [Accessed March 2011]; Available at http://www.hivguidelines.org/clinical-guidelines/adults/neoplastic-complications-of-hiv-infection/. [Google Scholar]
- 67. Shiels MS, Goedert JJ, Moore RD, et al. Reduced risk of prostate cancer in U.S. Men with AIDS. Cancer Epidemiol Biomarkers Prev. 2010;19:2910–2915. doi: 10.1158/1055-9965.EPI-10-0741. This article estimates prostate cancer risk in men with AIDS in a large registry-linkage study, as well as PSA testing rates from a clinical cohort, finding a decreased risk of prostate cancer in men with AIDS as well as low PSA testing rates.
- 68.Silverberg MJ, Klein D, Leyden WA, et al. Prostate cancer risk and use of prostate specific antigen tests among HIV-infected and HIV-uninfected men [abstract#1097]. Presented at the 48th Infectious Disease Society of America Annual Meeting; October 2010; Vancouver, Canada. [Google Scholar]
- 69.Screening for prostate cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2008;149:185–191. doi: 10.7326/0003-4819-149-3-200808050-00008. [DOI] [PubMed] [Google Scholar]
- 70. Andriole GL, Crawford ED, Grubb RL, 3rd, et al. Mortality results from a randomized prostate-cancer screening trial. N Engl J Med. 2009;360:1310–1319. doi: 10.1056/NEJMoa0810696. This article describes the initial results of a large RCT of prostate cancer screening, finding no mortality difference among screened and unscreened patients.
- 71. Schroder FH, Hugosson J, Roobol MJ, et al. Screening and prostate-cancer mortality in a randomized European study. N Engl J Med. 2009;360:1320–1328. doi: 10.1056/NEJMoa0810084. These are the results of a large RCT of prostate cancer screening, finding a small mortality benefit with screening at the cost of significant overtreatment.
- 72.Bruix J, Sherman M. Management of hepatocellular carcinoma. Hepatology. 2005;42:1208–1236. doi: 10.1002/hep.20933. [DOI] [PubMed] [Google Scholar]
- 73.Brau N, Fox RK, Xiao P, et al. Presentation and outcome of hepatocellular carcinoma in HIV-infected patients: a U.S.-Canadian multicenter study. J Hepatol. 2007;47:527–537. doi: 10.1016/j.jhep.2007.06.010. [DOI] [PubMed] [Google Scholar]
- 74.Zhang BH, Yang BH, Tang ZY. Randomized controlled trial of screening for hepatocellular carcinoma. J Cancer Res Clin Oncol. 2004;130:417–422. doi: 10.1007/s00432-004-0552-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Hepatocellular carcinoma: Rationale for screening. [Accessed July 2010]; Available at http://www.cancer.gov/cancertopics/pdq/screening/hepatocellular/HealthProfessional/page3.
- 76.Forner A, Reig M, Bruix J. Alpha-fetoprotein for hepatocellular carcinoma diagnosis: the demise of a brilliant star. Gastroenterology. 2009;137:26–29. doi: 10.1053/j.gastro.2009.05.014. [DOI] [PubMed] [Google Scholar]
- 77.Vibert E, Duclos-Vallee JC, Ghigna MR, et al. Liver transplantation for hepatocellular carcinoma: The impact of human immunodeficiency virus infection. Hepatology. 2011 doi: 10.1002/hep.24062. Epub ahead of print. [DOI] [PubMed] [Google Scholar]
- 78.Sigel K, Wisnivesky J, Justice AC, et al. HIV is an Independent Risk Factor for Lung Cancer Incidence [abstract#30]. Presented at the 17th Conference on Retroviruses and Opportunistic Infections; San Francisco, CA. 2010. [Google Scholar]
- 79.Shiels MS, Cole SR, Mehta SH, Kirk GD. Lung cancer incidence and mortality among HIV-infected and HIV-uninfected injection drug users. J Acquir Immune Defic Syndr. 2010;55:510–515. doi: 10.1097/QAI.0b013e3181f53783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Ries LAG YJ, Keel GE, Eisner MP, Lin YD, Horner M-J, editors. SEER Survival Monograph: Cancer Survival Among Adults: U.S. SEER Program, 1988–2001, Patient and Tumor Characteristics.: National Cancer Institute, SEER Program, NIH Pub. No. 07-6215. Bethesda, MD: 2007. [Google Scholar]
- 81.Crothers K, Goulet JL, Rodriguez-Barradas MC, et al. Impact of cigarette smoking on mortality in HIV-positive and HIV-negative veterans. AIDS Educ Prev. 2009;21:40–53. doi: 10.1521/aeap.2009.21.3_supp.40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Henschke CI, Naidich DP, Yankelevitz DF, et al. Early lung cancer action project: initial findings on repeat screenings. Cancer. 2001;92:153–159. doi: 10.1002/1097-0142(20010701)92:1<153::aid-cncr1303>3.0.co;2-s. [DOI] [PubMed] [Google Scholar]
- 83.National Lung Screening Trial (NLST) Initial Results. [Accessed 2010]; Available at http://www.cancer.gov/newscenter/pressreleases/NLSTFastFacts.