Abstract
Debate still surrounds the physiological roles of the alternative respiratory enzymes found in many fungi and plants. It has been proposed that alternative NADH:ubiquinone oxidoreductases (NADH dehydrogenases) may protect against oxidative stress, conversely, elevated activity of these enzymes has been linked to senescence. Here we show that inhibition of these enzymes in a fungal protein expression system (Aspergillus niger) leads to significantly enhanced specific growth rate, substrate uptake, carbon dioxide evolution, higher protein content, and more efficient use of substrates. These findings are consistent with a protective role of the NADH dehydrogenases against oxidative stress, thus, when electron flow via these enzymes is blocked, flux through the main respiratory pathway rises, leading to enhanced ATP generation. We anticipate that our findings will stimulate further studies in fungal and plant cultures leading to significant improvements in these expression systems, and to deeper insights into the cellular roles of alternative respiration.
The electron transport chain (ETC) of many filamentous fungi is highly branched and contains, in addition to the ‘core' electron transfer complexes, numerous alternative respiratory enzymes1. The alternative enzymes include at least three alternative NADH:ubiquinone oxidoreductases (NADH dehydrogenases), as characterised in Neurospora crassa, which can act in place of Complex I2,3, and an alternative oxidase (AOX), which furnishes the organism with the ability to effectively bypass Complexes III and IV4. At present, much work has been carried out into the physiological role of AOX, as this has been implicated in numerous industrially important bioprocesses, for example the production of citric acid by A. niger5,6, whereas little is known as regards the alternative NADH dehydrogenases. Currently, it has been demonstrated that alternative NADH dehydrogenases in filamentous fungi show enhanced activity under conditions of oxidative stress, primarily that of elevated dissolved oxygen (DOT) in bioprocesses, brought about by sparging submerged cultures with oxygen-enriched air7. This corresponded to a decreased reactive oxygen species (ROS) generation in these cultures, an observation which has been previously described in other cultures8,9.
Most, if not all of the research relating to the physiological role of alternative NADH dehydrogenases in filamentous fungi has been performed by measuring the activity of these enzymes in the presence of rotenone, a known inhibitor of Complex I1. However, to fully understand the relevance of the alternative NADH dehydrogenases, to submerged culture fermentation processes it would seem sensible to examine the result of removing the fungus’ ability to utilise these enzymes, both in control and oxidatively stressful situations. One method by which this could be achieved is by generation of a knockout mutant of the organism, in which the genes encoding these alternative enzymes were missing. However, this approach could be challenging, so a simpler, yet effective method was used in the current work: inhibition of the alternative NADH dehydrogenases.
Inhibitors exist for each of the ‘core’ proton-pumping electron transfer enzymes, these being rotenone and piericidin (Complex I), antimycin A and myxothiazol (Complex III), and cyanide (Complex IV). Similarly, AOX can be inhibited by salicylhydroxamic acid (SHAM) and n-propyl gallate1. However, few authors have reported inhibitors of the alternative NADH dehydrogenases of fungi. A literature search for potential inhibitors of plant alternative NADH dehydrogenases was similarly lacking in definitive answers, although two compounds were commonly associated with such studies, 6-dimethylallyl-3,5,7,8-tetrahydroxyflavone (platanetin) and 7-iodoacridone 4-carboxylic acid (IACA)10,11. IACA was also found to strongly inhibit both the external and internal alternative NADH dehydrogenase from S.cerevisiae12 even though the exact mechanism of inhibition remains elusive to date. Therefore, IACA seemed to be the most efficient of these compounds as described by the literature, and so the current work used this to inhibit the alternative NADH dehydrogenases in A. niger B1-D, and to attempt to further determine the physiological relevance of these enzymes in filamentous fungi and their impact on bioprocesses.
Filamentous fungi have been used to produce a broad range of compounds in large quantities. The basic advantages of theses organisms lie in their ability to grow in high biomass levels, carry out post-translational modification and glycosylation, and the existence of an effective protein secretion system capable of producing enzymes at levels up to 100 g/L13. However, these organisms have some drawbacks including their problematic secretion of heterologous proteins14 and their low growth rates with doubling times ranging from 2 to 7 hours15.
It would be of particular interest to further optimise the fermentations of these industrially interesting organisms, and by inhibiting the energy wasting alternative NADH dehydrogenase may be one way. Assuming that all electrons derived from the substrate oxidation go through the energy generating complex I, an increase in cellular metabolism would be expected which may lead to higher biomass levels, growth rates and intracellular protein concentrations. It is also very important to determine all the negative consequences associated with such inhibition (most notable of which would possibly be the high ROS formation). Therefore, this study, aims at shedding light on the complex role of alternative NADH dehydrogenase and to weigh the advantages and disadvantages of a potential inhibition of these enzymes for the optimisation of the fermentation process.
Results
Determination of IACA concentration
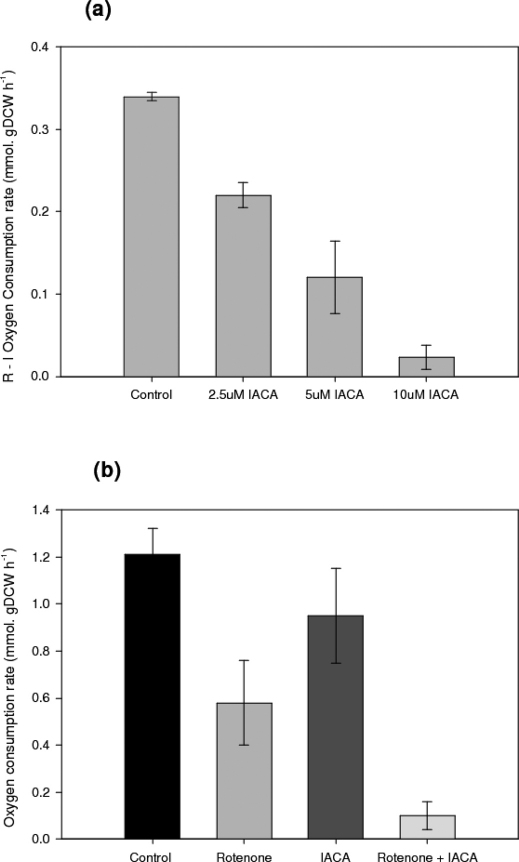
In order to determine the appropriate concentration of IACA to be used, shake flask batch cultures were carried out where different concentrations of the inhibitor were used. The oxygen consumption rate was measured in the presence of the complex I inhibitor, rotenone, and therefore the rotenone insensitive (i.e. electron transfer via alternative NADH dehydrogenases as opposed to Complex I) respiration was determined as can be seen in Figure 1.a.
Figure 1.
(a) The effect of different IACA concentration on the rotenone – insentive oxygen consumption rate in submerged batch cultures of A. niger B1-D. (b) The effect of 3μM rotenone and 10μM IACA upon oxygen consumption rates in submerged batch cultures of A. niger B1-D.
Finally, to ensure IACA was indeed an inhibitor of the alternative respiratory enzymes of filamentous fungi, as it is of plants and yeast, the oxygen consumption rates of cultures were measured in the presence of each of IACA and rotenone independently and also simultaneously. The results are presented in Figure 1.b. Concentrations of 3μM rotenone and 10μM IACA were used to achieve this.
Biomass production and specific growth rates
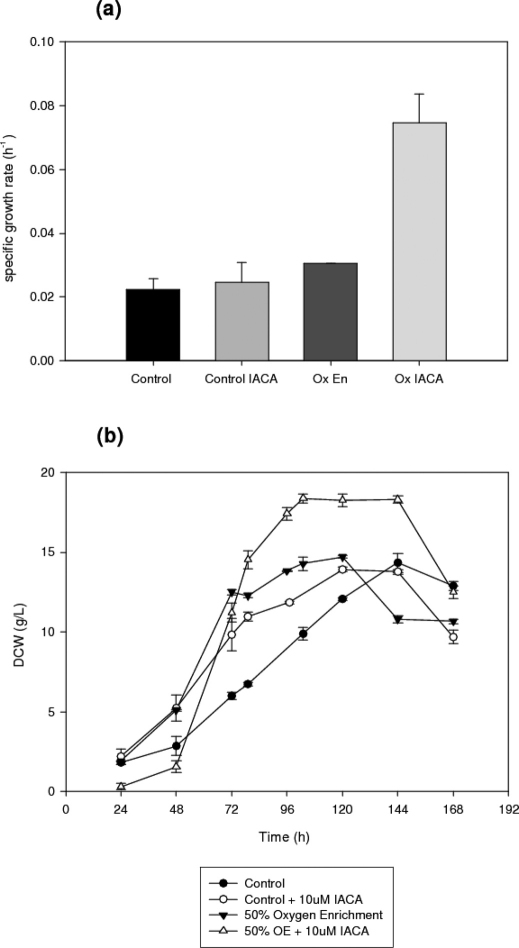
Figure 2 shows the dry cell weight (DCW) for each of the four culture types. In control cultures, the addition of IACA causes a significant increase in DCW (t-test, n = 3, a = 0.05, p = 0.011). A shorter lag phase is also noted in control cultures pretreated with IACA, whilst the specific growth rate is increased. In oxygen enriched cultures, the DCW values are markedly higher overall than control cultures, either with or without inhibitor, although the addition of IACA to the culture results in a further significant increase in both DCW and specific growth rate. Moreover, the respective DCW maxima are 14.7 and 18.3 g/l for oxygen enriched and IACA treated oxygen enriched cultures. The IACA treated oxygen enriched process has a significantly higher average DCW throughout the total duration of stationary phase, containing up to 4 g/l more biomass at its maximum. Interestingly, in IACA pre-treated oxygen enriched cultures, the lag phase was not decreased as in the control, but was in fact increased from 24 to 48 h. After 48 h, however, growth progressed at a very rapid rate, much faster in fact than any other culture. The specific growth rate was increased by the addition of IACA, the inhibitor increased the air sparged culture (9%), while there was a 2.5 fold increase for the oxygen enriched culture when IACA was added (Figure 2.a).
Figure 2. (a) Specific growth rates and (b) time profiles of the dry cell weight concentrations in submerged cultures of A. niger B1-D using either control or 10 μM IACA treated cultures.
Total intracellular protein concentrations
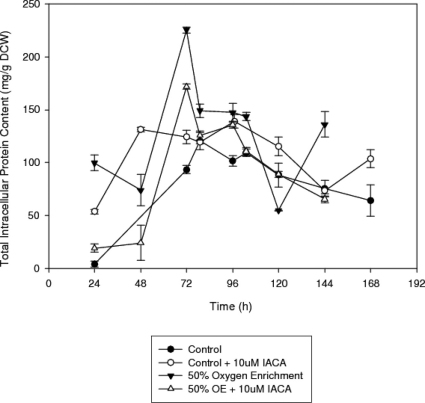
Figure 3 outlines the total intracellular protein content for each culture, and clearly shows that the addition of IACA to cultures results in a significant increase in intracellular protein concentrations (t-test, n = 3, a = 0.05, p = 0.007) relative to the control. Conversely, in oxygen enriched cultures the addition of inhibitor significantly decreased the intracellular protein content (t-test, n = 3, a = 0.05, p = 0.002). After 103 h, there is a sharp decrease in the protein concentrations of the oxygen enriched process to a significantly lower concentration than the IACA pretreated oxygen enriched culture. In both control and oxygen enriched cultures the addition of IACA did not result in any changes in the general intracellular protein concentration trends, but it radically influenced the concentrations themselves. The IACA treated control culture was increased in comparison to the control, but was greatly decreased compared to the IACA-treated oxygen enriched culture. Moreover, the intracellular protein concentrations of the oxygen enriched process containing inhibitor were significantly lower than those of the IACA-pretreated control cultures. Therefore, for cultures containing inhibitor, the application of oxygen enriched air significantly decreases the total intracellular protein content.
Figure 3. Time profiles of the intracellular protein concentrations in submerged cultures of A. niger B1-D using either control or 10 μM IACA treated cultures.
ATP concentrations
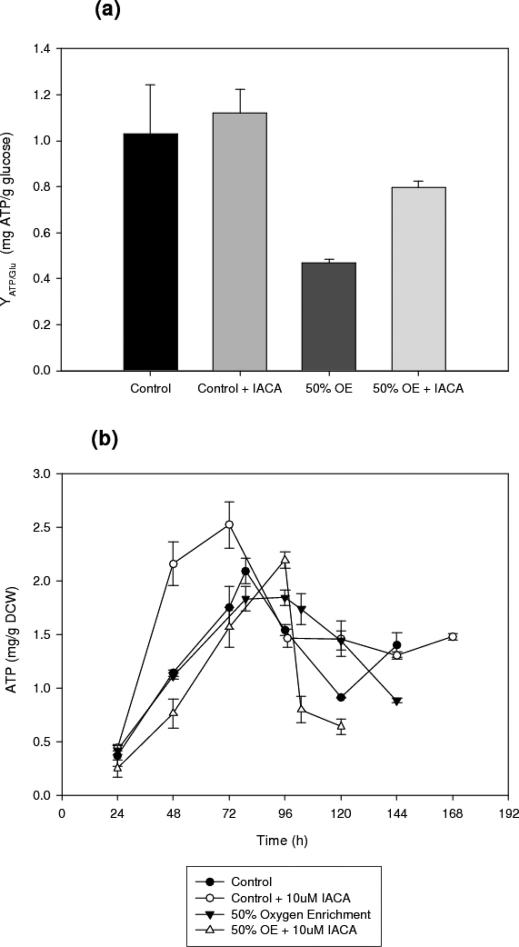
The intracellular ATP concentrations of each culture (Figure 4) are also significantly affected by the addition of IACA. ATP concentrations were significantly higher in the culture containing IACA, especially until 79 h (unpaired, two tailed t-test of the slopes, n = 3, a = 0.05, p = 0.010), than in the control. The maximum ATP concentrations were obtained at 79 h (2.09 mg/g DCW) and 72 h (2.52 mg/g DCW) for the control and the control + IACA respectively. In the case of the oxygen enriched processes, the ATP concentrations for the process containing IACA were lower during the first 48 hours. However, as time progressed, ATP concentrations become relatively stable in both processes, and in this instance the IACA treated culture has significantly higher ATP concentrations than the uninhibited culture (t-test, n = 3, a = 0.05, p = 0.011). There was however a further sharp decrease in the inhibited culture after 96 h, eventually causing a much decreased ATP pool as compared with the uninhibited cultures. A comparison between the two cultures containing IACA clearly demonstrates that oxygen enrichment leads to a substantial decrease in the average ATP concentrations throughout the entire duration of the process.
Figure 4. (a) ATP generation per glucose consumed and (b) time profiles of the intracellular ATP concentrations in submerged cultures of A. niger B1-D using either control or 10 μM IACA treated cultures.
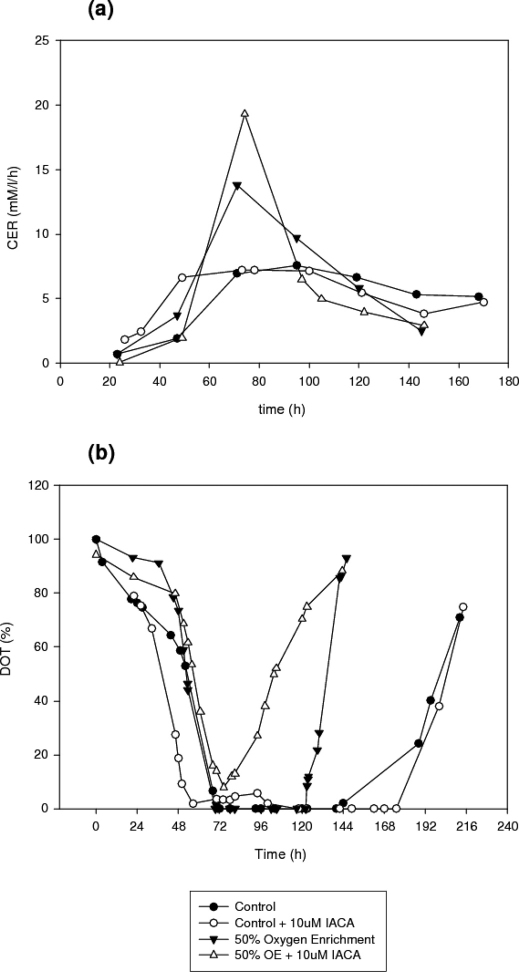
Dissolved oxygen and Carbon dioxide evolution rate
Figure 5.b. shows the dissolved oxygen tension values of the four cultures versus time. It can be clearly noted that in the case of the control cultures, the culture containing inhibitor showed an earlier onset of high metabolic activity and remained metabolically active for a longer period than the control cultures. As regards oxygen enrichment, the DOT values of the two bioprocesses show that the culture with IACA was slightly more metabolically active during the lag phase, which in the present culture lasted from 0 h to 46 h. During the growth phase it is evident that the culture containing IACA could not fully utilise all the available oxygen as the lowest value was 7.9% at 74 h. There was then a very abrupt, rapid increase in the dissolved oxygen tension values from 75 h onwards, signifying earlier culture degeneration.
Figure 5. Time profiles of (a) carbon dioxide evolution rates and (b) dissolved oxygen tensions in submerged cultures of A. niger B1-D using either control or 10 μM IACA treated cultures.
Further metabolic data can be gleaned from the carbon dioxide evolution rate (CER) of the cultures, as outlined in Figure 5.a. The addition of IACA in control cultures increases the CER values during the first 50 hours of the process, again demonstrating that the addition of IACA increases the respiratory activity of the culture. However, the CER decreases much quicker and also earlier in the IACA pretreated cultures, further showing earlier culture degeneration. For oxygen enrichment, the addition of inhibitor causes an initial decrease in the CER values, but after the 50 h this culture exhibits a much sharper increase in CER, accompanied by a much greater CER overall (19.3 mM/l/h compared with 13.8 mM/l/h). In contrast to uninhibited cultures, inhibited cultures show much more abrupt decreases in CER values after the maximum respiratory activity.
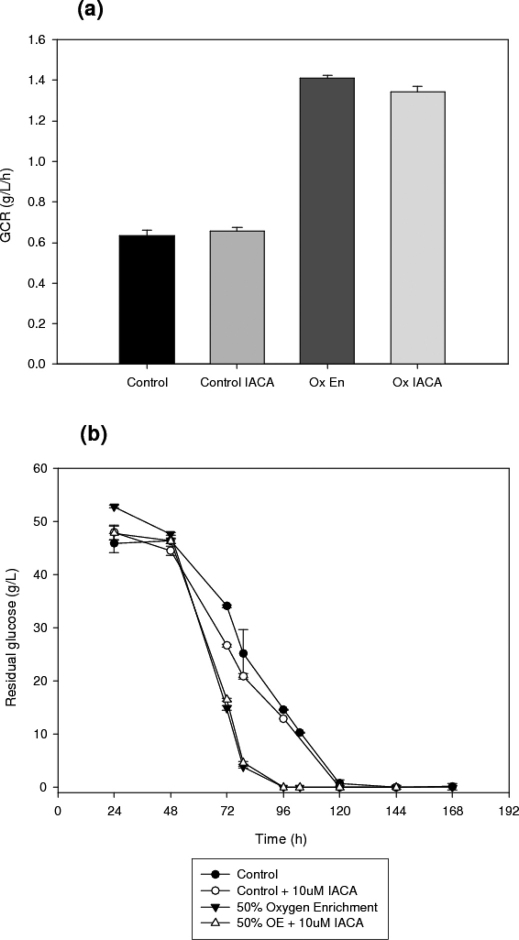
Glucose and ammonium concentrations and consumption rates
Figure 6. outlines the residual glucose concentrations and glucose consumption rates (GCRs) of each process. Although there are no significant differences in residual glucose or GCRs between inhibited and non-inhibited cultures treatments, it is worth noting that oxygen enrichment leads to a significantly increased GCR when compared to aerated cultures (t-test, n = 3, a = 0.05, p = 0.0). These data also allow the determination of the yield of biomass on glucose, in terms of g DCW/g glucose consumed. In this instance, not only does a basic oxygen enrichment increase the GCR in comparison to the aerated control, but IACA addition can be seen to clearly elicit an increase in yield in each of the pretreated cultures, i.e. control and oxygen enriched cultures minus IACA have a yield of 0.25 g/g and 0.31 g/g respectively, whereas the IACA treated equivalents demonstrate increased yields of 0.32 g/g and 0.51 g/g, for control and oxygen enriched batches respectively.
Figure 6. (a) Glucose consumption rates and (b) time profiles of residual glucose concentrations in submerged cultures of A. niger B1-D using either control or 10 μM IACA treated cultures.
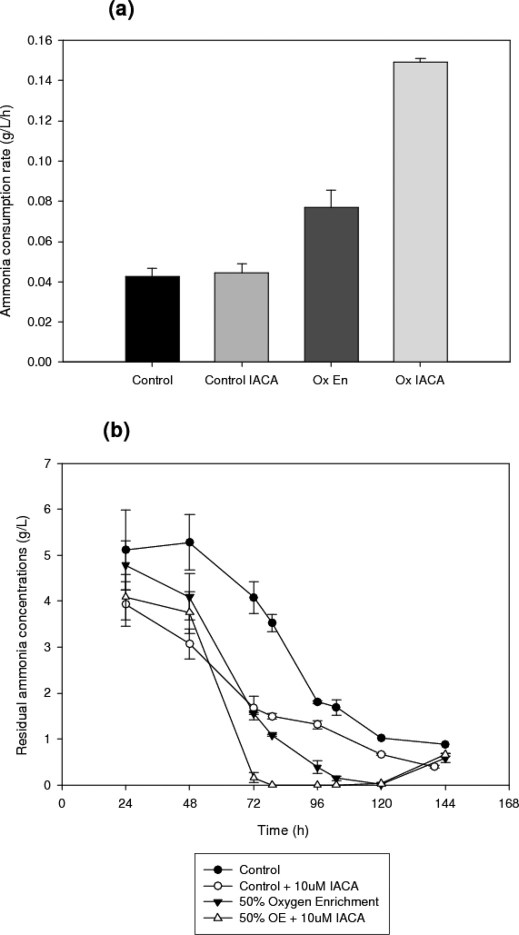
In addition to changes in glucose consumption rates, IACA also elicits distinct changes in ammonia uptake, as detailed in Figure 7. It can be seen from this figure that oxygen enrichment alone causes a decrease in the concentrations of residual ammonia salts as compared to control cultures, eventually reaching a concentration of 0.01 g/L at 120 h, whereas the control only drops as low as 0.85 g/L at 144 h. IACA treated control cultures on the other hand reach a minimum residual ammonia concentration of 0.40 g/L by 140 h, and the ammonia consumption rate (0.05 g/L/h) can be seen to be slightly increased in these cultures. By comparison, the oxygen enriched cultures show massive differences in ammonia consumption rates, from 0.08 g/L/h untreated to 0.15 g/L/h in the presence of IACA. Moreover, nitrogen starvation is a bigger risk in the IACA treated oxygenated batches, as concentrations of residual ammonia in these cultures reach zero from 80 h onwards, only increasing again from 120 h onwards, at which point autolysis is likely to be occurring in the culture (as it also coincides with a decrease in biomass concentration at the same time).
Figure 7. (a) Ammonia consumption rates and (b) time profiles residual ammonia concentrations in submerged cultures of A. niger B1-D using either control or 10 μM IACA treated cultures.
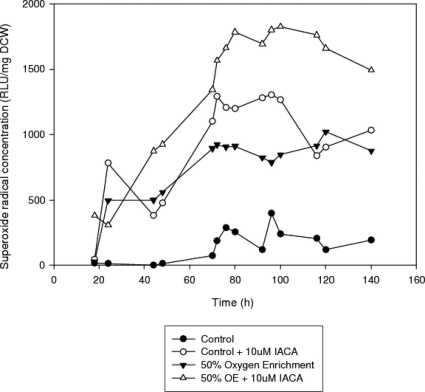
Superoxide anion concentrations
Superoxide anion concentrations in the IACA containing cultures are increased, as can be seen in Figure 8. Oxygen enrichment + IACA in particular can be seen to produce excessive ROS concentrations in comparison with any other treatment, although oxygen enrichment alone is lower than the IACA treated control cultures.
Figure 8. Time profiles of the superoxide anion concentrations in submerged cultures of A. niger B1-D using either control or 10μM IACA treated cultures.
Discussion
IACA clearly inhibits the rotenone-insensitive alternative NADH dehydrogenases of filamentous fungi, as it does those of plants. A search of the published reports indicates that 10μM of IACA is sufficient to inhibit the alternative NADH dehydrogenases of S.cerevisiae12. By contrast, in isolated mitochondria of Solanum tuberosum the concentration needed to achieve full inhibition of the alternative NADH dehydrogenases reached 150μM. Higher concentrations of IACA have been found to lead to inhibition of noth succinate dehydrogenase and complex I16.
Since IACA inhibits the alternative NADH dehydrogenases, it should therefore force the organism to theoretically use only the core electron transport chain, or potentially Complex I in combination with alternative oxidase (AOX). However, no cyanide-insensitive respiration has been found in the present culture17, thus it is likely that only the core electron transport chain is being used for respiration when the alternative NADH dehydrogenases are blocked. Furthermore, no significant increase in fermentative metabolism was detected in inhibited cultures, thus negating the possibility that ethanol production is a major physiological influence in these processes. As such, respiration solely via the core electron transport chain should result in a theoretical increase in ATP generation; inhibition of the alternative NADH dehydrogenases prevents uncoupled respiration, thus all respiratory activity will create a proton-motive force for driving the F1F0 ATP synthase1,18.
O’Donnell et al.17 proposed that this would probably lead to increases in cellular metabolism and viability, in that it would increase growth rates, intracellular protein contents, and most likely also nutrient consumption rates.
In cultures of A. niger B1-D, sparged with 1 vvm air, IACA did cause exactly this general increase in metabolic activity. Regarding cell growth, for example, the dry cell weight (DCW) and µ were increased, while the lag phase before growth was decreased. The same is also true of intracellular protein content in these processes; the total intracellular protein contents in inhibited cultures are significantly increased in comparison to the control cultures. Almost all metabolic indices studied (DOT, CER, ammonia uptake) pointed towards an increase in culture metabolism, the sole exception being glucose, in which no significant difference was noted in GCR though overall yield on glucose rose. All of these data correlate well with a significantly increased intracellular ATP concentration in control cultures.
Considering the results obtained in normally aerated cultures, it would be reasonable to assume that IACA might also serve to increase cellular metabolism in oxygen enriched processes. Oxygen enrichment has been previously shown to increase µ and DCW in cultures of A. niger19 and has been used in several industrial processes to increase product formation20,21,22.
The addition of the IACA to the oxygen enriched culture led to a dramatic increase in maximum biomass (29% higher) and specific growth rate (143.8% higher). From the results of the oxygen enriched culture it is clear that the addition of the inhibitor caused a significant increase of the ammonia consumption rate; however, there was no significant difference in the glucose consumption rate. Those indices clearly show enhanced metabolic activity after the addition of the inhibitor of the alternative NADH dehydrogenase. Regarding the specific growth rate, the slight difference between the air sparged cultures and the vast difference between the inhibited and uninhibited oxygen enriched cultures suggests that in the oxygen enriched culture there is great loss of energy which could otherwise be used for biomass production in the uninhibited oxygen enriched culture. It is worth noting that the oxygen enrichment only slightly increased the specific growth rate of the culture, while the inhibited oxygen enriched culture presented a remarkable increase. This can be explained by the ability of the non inhibited cultures to adjust the relative flux between the conventional and alternative respiratory pathways according to the catabolic flux (NADH and FADH concentration) and available oxygen. In case of catabolic overflow, oxygen overflow or both, the flux through the non energy-generating alternative NADH dehydrogenase reduces the risk of ROS formation and also reduces the amount of the produced ATP. Therefore, it can be assumed that the difference in the specific growth rate correspond to the amount of energy lost as heat23 in the non inhibited oxygen enriched culture.
Regarding the ROS generations, recent studies suggested that the iron-sulphur cluster N2 of Complex I is the main site of electron leak. Electron leaks at Complex I can release single electrons to oxygen and convert it into superoxide anion, a progenitor ROS24,25. While 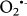 can be generated at two different sites, Qo and Q1, within complex III during the auto-oxidation of ubisemiquinone (
can be generated at two different sites, Qo and Q1, within complex III during the auto-oxidation of ubisemiquinone (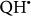 )26. On the other hand the alternative NADH dehydrogenase enzymes do not contain the characteristic electron leaking iron sulphur clusters and, like AOX, are unable to translocate protons across the membrane resulting in lower membrane potential27. In addition, it can be assumed that their co-existence with complex I helps keeping the NADH/NAD+ ratio lower and as a result contribute to a reduction of the electron leak and generation of reactive oxygen species. Therefore, alternative NADH dehydrogenases have a lower electron leak propensity than the proton pumping main respiratory complexes.
)26. On the other hand the alternative NADH dehydrogenase enzymes do not contain the characteristic electron leaking iron sulphur clusters and, like AOX, are unable to translocate protons across the membrane resulting in lower membrane potential27. In addition, it can be assumed that their co-existence with complex I helps keeping the NADH/NAD+ ratio lower and as a result contribute to a reduction of the electron leak and generation of reactive oxygen species. Therefore, alternative NADH dehydrogenases have a lower electron leak propensity than the proton pumping main respiratory complexes.
Even though the ATP pool is very dynamic, as the produced ATP molecules are immediately used to drive cellular processes, Figure 4.a. confirms that the inhibition of the alternative NADH dehydrogenases increased the efficiency with which ATP is generated per glucose consumed. Especially in the oxygen enriched cultures the IACA addition almost doubled the amount of the ATP produced per glucose consumed. Therefore, as expected from the other indices, blocking the energy wasting alternative NADH dehydrogenase significantly increased the ability of the organism to harness energy from the same amount of substrate.
Since ammonium uptake proceeds via an active transport mechanisms, in order to both maintain the ionic balance of the cell intact and drive the intermembrane transport of the ammonium, through the extrusion of hydrogen ions, it is entirely likely that some of the ATP being produced is being used to drive nutrient uptake28. Ammonium uptake is known to be affected by respiratory activity, and as such the increased respiration in these cultures may be increasing ammonium uptake simply due to the elevated ATP concentrations. However, in the case of glucose, fungi use an active transport system only when glucose concentration is low, while in the case of high glucose concentrations this system is repressed and glucose enters the cell by a constitutive facilitated diffusion system29.
Therefore, the uptake of glucose can also be influenced by the respiration but only when glucose is becoming limited, and this is possibly the reason why the glucose consumption rates do not exhibit any significant differences between the inhibited and uninhibited cultures, as transfer rates into the cells are solely dependent upon concentration gradient, although yields are significantly different.
Another seemingly paradoxical phenomenon in the oxygen enriched cultures is the lower intracellular protein concentration of the inhibited culture. However, a close look at Figure 8 may reveal a possible explanation for this, as the expectedly very high concentration of the very reactive superoxide anions in the IACA treated culture may have caused extensive damage to intracellular protein. The exceptionally high superoxide anion levels are due to electron leakage from Complex I, which would naturally be increased in the absence of functioning alternative NADH dehydrogenases1. Therefore it is possible that the intracellular protein turnover is much more intense in the IACA treated oxygen enriched culture and this can also be inferred from the significantly higher ammonia uptake rate (implying higher protein production). These data are consistent with findings of previous studies where under oxidatively stressful conditions the high ROS production causes excessive carbonylation of the intracellular proteins30,31. The irreversible and unrepairable nature of these modifications leads to the activation of the proteasome enhancing protein turnover31,32. Moreover, even though evidence from different organisms show that the protein degradation proceeds through an ATP independent mechanism33,34,35, the de novo synthesis of the replacement protein molecules is a very energy demanding process, which explains the lower ATP concentrations in the oxygen enriched inhibited culture compared to the air sparged culture (Figure 4.b.).
The excessive ROS levels found in the oxygen enriched IACA treated culture and the much higher ROS concentration of the control IACA treated culture in comparison with both the control and oxygen enriched culture alone shows that the alternative NADH dehydrogenases probably do have a role in the protection from elevated ROS generation. Alternatively, another explanation could be that the excessive growth and ATP synthesis seen on IACA addition is consistent with excessive ROS formation. All IACA is doing then is upping the metabolism of these fungal cultures. One way or the other, the effect of the alternative NADH dehydrogenase inhibition appear to both benefit the cell physiology by boosting the metabolism (increased growth and biomass, higher ATP production) and at the same to be damaging (higher ROS concentration, reduced protein concentration only under oxygen enriched conditions though for the latter).
Although the effects of IACA are complex, there are several key points which are clear from this work. First of all, IACA addition leads to increased DCW and specific growth rates in all cultures. This is associated with an increase in the respiratory activity of IACA treated cultures, and specifically the ability of these cultures to use oxygen more effectively than the untreated counterparts. Since the alternative NADH dehydrogenases are inhibited by IACA, Complex I must be used for all initial electron transfer in inhibited cultures. Since Complex I produces one mole of ATP for every four electrons transported, ATP concentrations are increased in IACA treated cultures. This energy increase in turn produces a greater rate of ammonia uptake, which is linked to increased protein concentrations.
However, the inhibition of the alternative NADH dehydrogenases also causes numerous deleterious effects. In particular, ROS concentrations are significantly increased in the presence of IACA, due to the high frequency of electron leakage from Complex I onto molecular oxygen. This then leads to a decrease in culture viability as a whole, and causes an earlier and much more rapid culture decline than in untreated processes. Protein concentrations, in the inhibited oxygen enriched culture, are perhaps not as high as would be expected here; this is most likely caused by extensive protein carbonylation by the excess ROS30. Thus, as far as the physiological role is concerned, it can be stated that these enzymes can act to maintain ROS concentrations below a threshold in the cell, and are useful in limiting ATP production during periods in which there is no highly-energy requiring process operating.
Regarding the industrial implications, inhibition or genetic deletion of the alternative NADH dehydrogenases may lead to strains with the ability to use oxygen more effectively and grow faster and to higher biomass levels than the parent strains. It is obvious that there will be disadvantages which can however be faced with the application of the right fermentation techniques.
To conclude, even though the physiological role of such ubiquitous enzymes as the alternative NADH dehydrogenases is still a matter of some debate, the current work sheds some light on the reason why these enzymes have remained present in so many organisms during the process of evolution. Moreover, the findings of the present study show that inhibition of the A.niger NADH dehydrogenases has significant effects on the productivity of a bioprocess. Moderate oxygenation of an inhibited culture can have a high increase of the intracellular protein concentration and energy levels, while when oxygen levels become very high due to the application of oxygen enrichment, both intracellular protein and the energy levels fell to very low levels. Therefore, the use of an alternative NADH dehydrogenase deficient strain along with the application of the appropriate fermentation conditions, could be a route to significant increasing the productivity of an industrial fungal bioprocess by increasing the growth rate and consequently reducing fermentation times, while at the same time increasing the energetic efficiency.
Methods
Microorganism and Culture Conditions
The microorganism used was a recombinant Aspergillus niger B1-D, kindly gifted by Professor David Archer of the University of Nottingham, in which the hen egg white lysozyme (HEWL) cDNA gene is placed under the control of the Aspergillus awamori glucoamylase promoter (glaA)36. Master cultures were maintained on agar slopes, stored at 4°C and plated at regular intervals to check for mutation. Working cultures were grown on potato dextrose agar (PDA), obtained from Oxoid Ltd. (Hampshire, UK), from which spore suspensions were prepared by washing a 7 day old culture, grown at 30°C, with 0.1 % (w/v) Tween 80. The spore concentration was determined by use of a haemocytometer and a spore inoculum of 2–4 mL from a 105 spores/mL stock solution was used. The medium used for the cultivation of the innoculum (preculture) was described by Wongwicharn et al.19 with 50 g/l glucose as carbon and energy source.
Bioreactor Batch Processes
Batch cultivations were performed in parallel using BioFlo 110 bioreactors (New Brunswick Scientific, Edison, USA). The working volume of the bioreactors was 12 L but a volume of 8 L was used. The reactors were inoculated with a (5% v/v) 48 h-old shake flask culture grown at 25°C and 200 rpm. The pH was kept constant at 4.0 by automatic titrant addition (1.5 M NaOH, 1.5 M H2SO4) and the temperature was 25°C throughout. Air flow rate was controlled at 1.0 volume of air per volume of culture per minute (vvm). In cultures sparged with oxygen-enriched air, air and pure oxygen were blended at flow rates of 0.5 vvm air:0.5 vvm O2 prior to entering the bioreactor. The flow rate of air and oxygen was controlled by using a gas mixer by Braun Biotech International. Agitation was maintained at 400 rpm for the duration of the process. In the case of the cultures containing inhibitor, IACA was added to the inoculum just prior to introduction into the reactor so that the final concentration of IACA in the reactor was 10μM.
Shake Flask Cultivation
Shake flask cultures were performed in 1000 mL conical flasks containing 400 mL sterile medium. These were inoculated with a concentrated spore suspension to give an inoculum concentration of 100 spores/mL, and were subsequently incubated at 25°C in an orbital incubator set at 200 rpm. The spore concentrations were enumerated using a haemocytometer and appropriate dilutions were made accordingly.
Analytical Methods
Biomass was estimated by means of dry weight measurement as described by Wongwicharn et al.19. Culture fluid aliquots (5 mL) were filtered through predried and weighed GF/C filter papers, with a particle retention size of approximately 1.2 mm (Whatman Ltd.,Maidstone, UK). The mycelium was washed with 10 mL distilled water, dried in a microwave oven on the defrost setting for 20 min, and weighed. All samples were analysed in triplicate. Ammonium ion and glucose concentrations were measured using commercial kits (Ammonia:Kit No. AA0100; Glucose:Kit No. 315–500; both from Sigma Ltd., Dorset, UK).
Intracellular protein concentrations were measured by the method of Bradford37. 5 mL aliquots of fungal culture were withdrawn and were filtered through a 4.25 cm diameter GF/C filter. The filter cake was washed twice with 5 mL of distilled water before resuspending in the same volume of a sodium phosphate buffer (50 mM, pH = 7.5). The cell suspensions were stored in ice before being assayed. Before measurement, fungal cells were disrupted in a high-pressure cell homogenizer (Constant Systems Ltd., Warwick, UK) at 30 kpsi and centrifuged at 14,000 rpm for 30 min to obtain a clear supernatant.
Intracellular ATP concentrations were measured using a commercial kit (Sigma-Aldrich cat no. FL-AA), and results were obtained as relative light units (RLU) counted on a luminometer. Prior to measurement, ATP had to be extracted. This was achieved by boiling fresh samples from the bioreactor in preheated Tris-EDTA buffer, pH 7.4. After boiling for 5 minutes, samples were placed on ice for 10 mins, and subsequently centrifuged at 10,000 rpm for 10 mins to provide a clear supernatant. The supernatant was diluted 100-fold before measurement of RLU. ATP standards were in the range of 1 – 50 mg/L. All samples were analysed in triplicate.
The superoxide radical concentration in any given sample was determined by the chemiluminescent response in the presence of lucigenin (bis-N-methylacridinium nitrate), as described by Bai et al.38. The chemiluminescence of each sample was recorded as the relative light units (RLU) emitted every second over a period of ten minutes, using a Lumac Biocounter M2500 (Lumac BV, Landgraat, Netherlands). Exhaust gas readings were collected by a Servomex Xentra 4100 gas analyzer.
The total and rotenone-insensitive oxygen consumption rates were determined as described by Bai et al.39, using a Clark-type oxygen electrode (Model 970, Jenway, Essex, UK). The change in dissolved oxygen tension was monitored for 2–4 minutes, and oxygen consumption rate (OCR) was determined by the following equation:
where DOTend is the final dissolved oxygen tension (DOT), as a percentage; Vc is the volume of reactant solution (in mL); t is the period of measurement, in minutes; Gb is the dry weight of biomass (g); and 2.4x10−1 is the concentration of oxygen in saturated water at 25°C (mmole.mL−1). All measurements were carried out at 25°C. All samples were analysed in triplicate.
Author Contributions
IV carried out the experiments and analysis and wrote part of the manuscript. AOD helped with the experimental design and wrote part of the manuscripts, he also carried out the shake flasks experiment and the superoxide anion assay. LMH and BMcN supervised the whole work and helped in the preparation and writing of the manuscripts. All authors reviewed the manuscript.
References
- Joseph-Horne T., Hollomon D. W. & Wood P. M. Fungal respiration: a fusion of standard and alternative components. Biochim. Biophys. Acta 1504, 179–195 (2001). [DOI] [PubMed] [Google Scholar]
- Carneiro P., Duarte M. & Videira A. The main external alternative NAD(P)H dehydrogenase of Neurospora crassa mitochondria. Biochim. Biophys. Acta 1608, 45–52 (2004). [DOI] [PubMed] [Google Scholar]
- Videira A. & Duarte M. From NADH to ubiquinone in Neurospora mitochondria. Biochim. Biophys. Acta 1555, 187–191 (2002). [DOI] [PubMed] [Google Scholar]
- Medentsev A. G., Arinbasarova A. Y. & Akimenko V. K. Regulation and physiological role of cyanide-resistant oxidases in fungi and plants. Biochemistry (Mosc) 64, 1230–1243 (1999). [PubMed] [Google Scholar]
- Kirimura K. et al. Contribution of cyanide-insensitive respiratory pathway, catalyzed by the alternative oxidase, to citric acid production in Aspergillus niger. Biosci. Biotechnol. Biochem 64, 2034–2039 (2000). [DOI] [PubMed] [Google Scholar]
- Li Q., Harvey L. M. & McNeil B. Oxidative stress in industrial fungi. Crit Rev Biotechnol 29, 199–213 (2009). [DOI] [PubMed] [Google Scholar]
- Bai Z., Harvey L. M. & McNeil B. Oxidative Stress in Submerged Cultures of Fungi. Crit Rev Biotechnol 23, 267–302 (2003). [DOI] [PubMed] [Google Scholar]
- Guerrero-Castillo S., Vázquez-Acevedo M., González-Halphen D. & Uribe-Carvajal S. In Yarrowia lipolytica mitochondria, the alternative NADH dehydrogenase interacts specifically with the cytochrome complexes of the classic respiratory pathway. Biochim. Biophys. Acta 1787, 75–85 (2009). [DOI] [PubMed] [Google Scholar]
- Li Y., Zhu H. & Trush M. A. Detection of mitochondria-derived reactive oxygen species production by the chemilumigenic probes lucigenin and luminol. Biochim. Biophys. Acta 1428, 1–12 (1999). [DOI] [PubMed] [Google Scholar]
- Kerscher S. J. Diversity and origin of alternative NADH:ubiquinone oxidoreductases. Biochim. Biophys. Acta 1459, 274–283 (2000). [DOI] [PubMed] [Google Scholar]
- Rasmusson A. G., Geisler D. A. & Møller I. M. The multiplicity of dehydrogenases in the electron transport chain of plant mitochondria. Mitochondrion 8, 47–60 (2008). [DOI] [PubMed] [Google Scholar]
- Oettmeier W., Masson K., Soll M. & Reil E. Acridones and quinolones as inhibitors of ubiquinone functions in the mitochondrial respiratory chain. Biochem. Soc. Trans. 213–216 (1994). [DOI] [PubMed] [Google Scholar]
- Cherry J. R. & Fidantsef A. L. Directed evolution of industrial enzymes: an update. Curr Opin Biotechnol 14, 438–443 (2003). [DOI] [PubMed] [Google Scholar]
- Gellissen G., Strasser A. W. M. & Suckow M. in Production of Recombinant Proteins: Novel Microbial and Eukaryotic Expression Systems, ed Gerd Gellissen, Weinheim, Germany: Wiley pp 1–6 (2005). [Google Scholar]
- Kavanagh K. in Fungi: Biology and Applications, ed Kevin Kavanagh, West Sussex, UK: Wiley pp 89–111 (2005). [Google Scholar]
- Roberts T. H., Rasmusson A. G. & Møller I. M. Platanetin and 7-iodo-acridone-4-carboxylic acid are not specific inhibitors of respiratory NAD(P)H dehydrogenases in potato tuber mitochondria. Physiol. Plant. 96, 263–267 (1996). [Google Scholar]
- O'Donnell A., Harvey L. M. & McNeil B. The roles of the alternative NADH dehydrogenases during oxidative stress in cultures of the filamentous fungus Aspergillus niger. Fungal Biol 115, 359–369 (2011). [DOI] [PubMed] [Google Scholar]
- Li Q. et al. Oxidative stress in fungal fermentation processes: the roles of alternative respiration. Biotechnol Lett 33, 457–467 (2011). [DOI] [PubMed] [Google Scholar]
- Wongwicharn A., McNeil B. & Harvey L. M. Effect of oxygen enrichment on morphology, growth, and heterologous protein production in chemostat cultures of Aspergillus niger B1-D. Biotechnol Bioeng 65, 416–424 (1999). [DOI] [PubMed] [Google Scholar]
- Choi J. H., Keum K. C. & Lee S. Y. Production of recombinant proteins by high cell density culture of Escherichia coli. Chem Eng Sci 61, 876–885 (2006). [Google Scholar]
- Lee C. Y., Lee S. J., Jung K. H., Katoh S. & Lee E. K. High dissolved oxygen tension enhances heterologous protein expression by recombinant Pichia pastoris. Process Biochem 38, 1147–1154 (2003). [Google Scholar]
- Wang L., Ridgway D., Gu T. & Moo-Young M. Bioprocessing strategies to improve heterologous protein production in filamentous fungal fermentations. Biotechnol Adv 23, 115–129 (2005). [DOI] [PubMed] [Google Scholar]
- Siedow J. N. & Umbach A. L. Plant mitochondrial electron transfer and molecular biology. Plant Cell 7, 821–831 (1995). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fato R. et al. Differential effects of mitochondrial Complex I inhibitors on production of reactive oxygen species. Biochim. Biophys. Acta 1787, 384–392 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lenaz G. et al. Mitochondrial Complex I: Structural and functional aspects. Biochim. Biophys. Acta 1757, 1406–1420 (2006). [DOI] [PubMed] [Google Scholar]
- Poyton R. O., Ball K. A. & Castello P. R. Mitochondrial generation of free radicals and hypoxic signaling. Trends Endocrinol Metab 20, 332–340 (2009). [DOI] [PubMed] [Google Scholar]
- Murphy M. P. How mitochondria produce reactive oxygen species. Biochem J 417, 1–13 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garrill A. in The Growing Fungus, eds Gow, N. A., & Gadd, G. M. London, UK: Chapman & Hall pp 163–181 (1995). [Google Scholar]
- Carlile M. J., Gooday G. W. & Watkinson S. C. in The Fungi, London UK: Academic press pp 85–184 (2001). [Google Scholar]
- Li Q., Harvey L. M. & McNeil B. Oxygen enrichment effects on protein oxidation, proteolytic activity and the energy status of submerged batch cultures of Aspergillus niger B1-D. Process Biochem 43, 238–243 (2008). [Google Scholar]
- Nystrom T. Role of oxidative carbonylation in protein quality control and senescence. EMBO J 24, 1311–1317 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inai Y. & Nishikimi M. Increased degradation of oxidized proteins in yeast defective in 26 S proteasome assembly. Arch Biochem Biophys 404, 279–284 (2002). [DOI] [PubMed] [Google Scholar]
- Davies K. J. A. Degradation of oxidized proteins by the 20S proteasome. Biochimie 83, 301–310 (2001). [DOI] [PubMed] [Google Scholar]
- Groll M. et al. Structure of 20S proteasome from yeast at 2.4A resolution. Nature 386, 463–471 (1997). [DOI] [PubMed] [Google Scholar]
- Grune T., Merker K., Sandig G. & Davies K. J. A. Selective degradation of oxidatively modified protein substrates by the proteasome. Biochem Biophys Res Commun 305, 709–718 (2003). [DOI] [PubMed] [Google Scholar]
- Archer D. B. et al. Hen egg white lysozyme expressed in, and secreted from, Aspergillus niger is correctly processed and folded. Nat Biotech 8, 741–745 (1990). [DOI] [PubMed] [Google Scholar]
- Bradford M. A rapid and sensitive method for the quantitation of microgram quantities of proteins utilizing the principle of protein-dye binding. Anal Biochem 72, 248 – 254 (1976). [DOI] [PubMed] [Google Scholar]
- Bai Z., Harvey L. M. & McNeil B. Use of the chemiluminescent probe lucigenin to monitor the production of the superoxide anion radical in a recombinant Aspergillus niger (B1-D). Biotechnol Bioeng 75, 204–211 (2001). [DOI] [PubMed] [Google Scholar]
- Bai Z., Harvey L. M., White S. & McNeil B. Effects of oxidative stress on production of heterologous and native protein, and culture morphology in batch and chemostat cultures of Aspergillus niger (B1-D). Enzyme Microb Technol 34, 10–21 (2004). [Google Scholar]