Abstract
In response to environmental stresses, the mammalian serine threonine kinases PERK, GCN2, HRI, and PKR phosphorylate the regulatory serine 51 of the eukaryotic translation initiation factor 2α (eIF2α) to inhibit global protein synthesis. Plasmodium, the protozoan that causes malaria, expresses three eIF2α kinases: IK1, IK2, and PK4. Like GCN2, IK1 regulates stress response to amino acid starvation. IK2 inhibits development of malaria sporozoites present in the mosquito salivary glands. Here we show that the phosphorylation by PK4 of the regulatory serine 59 of Plasmodium eIF2α is essential for the completion of the parasite's erythrocytic cycle that causes disease in humans. PK4 activity leads to the arrest of global protein synthesis in schizonts, where ontogeny of daughter merozoites takes place, and in gametocytes that infect Anopheles mosquitoes. The implication of these findings is that drugs that reduce PK4 activity should alleviate disease and inhibit malaria transmission.
Malaria is among the most deadly and devastating infectious diseases. Eradication of malaria requires a vaccine that protects at least 80% of individuals for several years. Such a vaccine is still not available (1). Other measures such as pyrethroid-treated bed nets, indoor spraying to kill mosquitoes, and artemisinin-based combination therapies have contributed to contain the disease in some areas. Nevertheless, every year over a million people still die of malaria and hundreds of millions are incapacitated by the disease. The emergence of artemisinin-resistant Plasmodium falciparum parasites in Southeast Asia and Africa (2, 3) is a cause of particular concern considering the speed with which drug resistance has spread in the past. Furthermore, Plasmodium vivax malaria that is endemic in parts of Asia, South America, and Central America is becoming resistant to the standard chloroquine treatment (4). Therefore, new anti-malarial drugs are urgently needed, in particular of drugs that are chemically distinct from those in current use. Still not exploited is the development of drugs against essential kinases for parasite development. The kinome of Plasmodium includes about 100 serine–threonine (S/T) kinases and no tyrosine kinase (5). Among the S/T enzymes, about 40 are considered likely to be necessary for the completion of the Plasmodium erythrocyic cycle that causes disease (6, 7). One of them, PK4, is part of the family of kinases predicted to phosphorylate the eukaryotic translation initiation factor 2α(eIF2α). PK4 is the subject of this article.
When human cells are subjected to stressful conditions, the PERK, GCN2, HRI, and PKR kinases phosphorylate the regulatory serine 51 of eIF2α. This leads to global inhibition of protein synthesis and provides the stressed cells with time to recover (8). Each of the human kinases is activated during different stress conditions: PERK by endoplasmic reticulum (ER) stress (9), GCN2 during amino acid starvation (10), HRI by heme deficiency in reticulocytes (11), and PKR following viral invasion (12).
In Plasmodium, the parasitic protozoon that causes malaria in humans, three eIF2α kinases have been identified: IK1, IK2, and PK4 (5). IK1 and PK4 are mostly transcribed in the asexual blood stages, and IK2 is predominantly transcribed in salivary gland sporozoites (13). The disruption of IK1 or IK2 does not affect the development of the parasites in the blood of the mammalian host or in the mosquito vector. IK1 function is similar to that of GCN2; it regulates the stress response of blood-stage parasites to amino acid starvation (14). IK2 controls the latency of malaria parasites in mosquito salivary glands. After injection in the mammalian host, eIF2α-P is dephosphorylated to permit the continuation of the life cycle (13). PK4 is transcribed in blood stages, but its function is not understood (13, 15). Nevertheless, it likely plays an essential role in the parasite's erythryotic stage of development because several attempts to inactivate PK4 in Plasmodium berghei (13) and in P. falciparum (7) have failed. Because PK4's only known function is phosphorylation of eIF2α of Plasmodium, the implication is that lack of phosphorylation is likely to be lethal for the parasite's blood stages. Here we provide direct evidence for this conclusion and show that eIF2α of schizonts and gametocytes is the target of PK4 activity. Becasue there are PK4 orthologs in all human Plasmodium species, the practical implication of our findings is that a drug that inhibits PK4 kinase activity is likely to cure infections with any species of malaria parasites.
Results
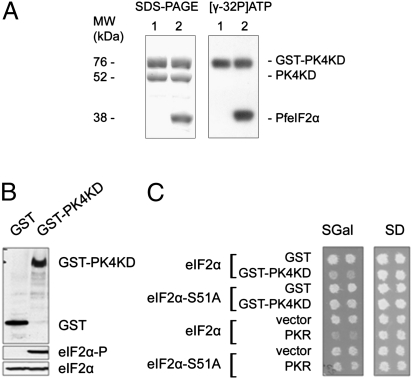
PK4 (PFF1370w) is a S/T kinase transcribed in Plasmodium blood stages (13, 15). In vitro PK4's kinase domain autophosphorylates and phosphorylates eIF2α (Fig. 1A). Similar to the effect of human PKR and Toxoplasma TgIF2K-A (16), expression of the Plasmodium PK4 kinase domain fused to the dimer glutathione S-transferase (GST) inhibited yeast growth (Fig. 1 B and C). This was most likely due to inhibition of global protein synthesis as it was not observed in yeast expressing nonphosphorylatable eIF2α-S51A (Fig. 1C).
Fig. 1.
PK4 phosphorylates Plasmodium eIF2α in vitro and yeast eIF2α. (A) In vitro kinase assay. The purified GST-PfPK4KD with (lane 2) or without (lane 1) substrate PfeIF2α was incubated with [γ-32P]ATP at 37 ºC for 30 min. Radiolabeled proteins were separated by SDS/PAGE and visualized by Coomassie staining and autoradiography. As shown, the PK4 kinase autophosphorylates and phosphorylates eIF2α. (B) PK4 phosphorylates eIF2α in yeast. Plasmids expressing GST or GST fused to the intact PK4KD were transformed into yeast strain H2557 expressing wild-type eIF2α. Whole-cell extracts were subjected to SDS/PAGE followed by immunoblot analysis using antibodies to GST, to phosphorylated Ser51 (eIF2α-P), or to total yeast eIF2α. (C) PK4 regulates yeast cell growth. An empty vector or plasmids expressing the positive control human PKR, GST, or GST fused to the PK4KD under the control of a galactose-inducible promoter were transformed into isogenic yeast strains expressing wild-type eIF2α (eIF2α; H2557) or nonphosphorylatable eIF2α-S51A (J223). Patches of two independent transformants for each plasmid were grown to confluence on synthetic dextrose (SD) plates, where the galactose-inducible promoter is repressed, and then replica-plated to synthetic galactose (SGal) medium, where the galactose-regulated promoter is induced. Plates were incubated at 30 ºC for 2 d.
Attempts to study the function of PK4 by generating a direct knockout in P. berghei or in P. falciparum blood stages [including a recent one (Fig. S1)] were not successful (7, 13). To prove that PK4 is required for development of blood stages, we generated in P. berghei a PK4 conditional mutant using the Flp/FRT recombination system. We targeted PK4 in a clone named TRAP/FlpL whose behavior throughout the life cycle is indistinguishable from that of WT parasites (17). In the conditional mutant, the Flp recombinase is expressed under the control of the TRAP promoter (Fig. S2A). The TRAP promoter is not active in blood stages where the absence of PK4 would be lethal; activation of the TRAP promoter starts in the mosquito cycle in midgut oocysts. Furthermore, PK4 is under the control of FRT-flanked TRAP 3′ regulatory sequences. Therefore, in the conditional parasites (named PbPK4cKO), the excision of the TRAP 3′ sequence and the selection cassette human dihydrofolate reductase (hDHFR) by Flp recombinase starts only in mosquito midgut sporozoites. TRAP continues to be expressed in salivary gland sporozoites. Therefore, there is ample time for silencing of PK4 in most parasites during traffic from the midgut to the salivary gland.
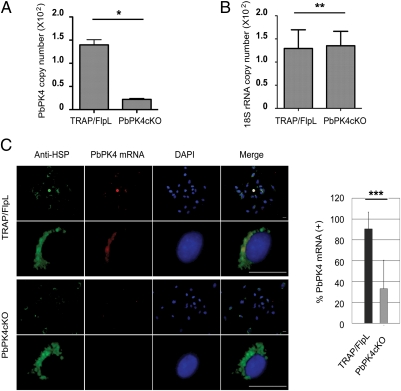
The integration of the targeting plasmid at the PK4 locus was confirmed by PCR and by Southern blot (Fig. S2 B and C). It did not impair development of PbPK4cKO in the blood of mice (Fig. S2D) or in the mosquito (Fig. S3A). The efficacy of PK4 excision, evaluated in the salivary gland sporozoites, was 84 ± 0.5% as measured by quantitative PCR (qPCR) of the cDNA (Fig. 2A). The mutant salivary gland sporozoites were then used to infect HepG2 cells in vitro and to infect mice. The absence of PK4 mRNA did not affect liver-stage development of PbPK4cKO as measured by in situ hybridization, qPCR, and immunofluorescence assay (IFA) (Fig. 2 B and C and Fig. S3 B–D). In sharp contrast, the ability of PbPK4cKO sporozoites to infect mice was dramatically reduced (Table 1). When mice were injected i.v. with 104 sporozoites, fewer mice were infected with PbPK4cKO sporozoites. Importantly, all parasites circulating in the blood were in fact PK4(+) as determined by PCR of genomic DNA (gDNA) and cDNA (Fig. S4). When the dose was reduced to 103 sporozoites, only the control mice injected with TRAP/FlpL developed blood infection. We could not prove that the PK4(−) liver-stage merozoites are released to the blood. It is possible that PK4 is also required in late liver-stage schizogony, similar to its role in the schizogony of the erythrocytic cycle. On the basis of the present findings and the multiple previous failures to target PK4 in blood stages of P. berghei and P. falciparum, we conclude that PK4 expression is required for the completion of the erythrocytic cycle of Plasmodium.
Fig. 2.
PbPK4cKO development into liver stages. (A) Excision of PbPk4 in PbPK4cKO sporozoites was quantitated by qPCR. Shown are the means ± SD of three independent experiments. *P < 0.0001 (two-tailed Student's t test). (B) C57BL/6 mice (six mice per group) were i.v. injected with 104 TRAP/FlpL or PbPK4cKO sporozoites. Liver-stage parasite burden was measured by qPCR, and the means ± SD (**P = 0.8353) are shown. This experiment was performed two times with similar results. (C) HepG2 cells infected with TRAP/FlpL or PbPK4cKO sporozoites were subjected to in situ hybridization followed by immunofluorescence with anti-HSP antibody to reveal the infected hepatocytes. A Cy3-probe was used to label the PK4 mRNA. Infected cells that presented PK4 mRNA were counted in both wild-type and mutant parasites, and results are expressed as a percentage. Only 33 ± 27% exoerythrocytic forms contained PK4 transcripts. (Scale bars, 20 μm.) ***P < 0.0001.
Table 1.
Comparision of infectivities of TRAP/FlpL and PbPK4cKO sporozoites
| Parasite line | No. of sporozoites injected | Mice positive/mice injected | Genotype of parasites in infected mice |
| Experiment 1 | |||
| TRAP/FlpL | 104 | 5/5 | PK4(+) |
| PbPK4cKO | 104 | 2/5 | PK4(+) |
| Experiment 2 | |||
| TRAP/FlpL | 104 | 5/5 | PK4(+) |
| PbPK4cKO | 104 | 3/5 | PK4(+) |
| Experiment 3 | |||
| TRAP/FlpL | 103 | 9/9 | PK4(+) |
| PbPK4cKO | 103 | 0/8 |
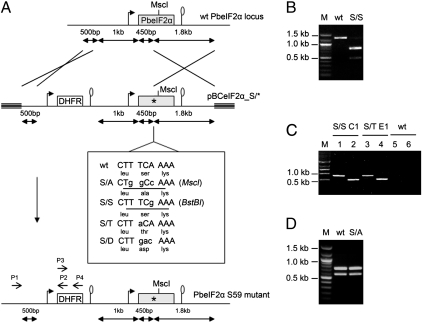
Serine 59 (ser59) of eIF2α is conserved in all Plasmodium species and corresponds to the regulatory ser51 of human eIF2α (Fig. S5). Because the function of PK4 is to phosphorylate ser59 of Plasmodium eIF2α, we tested whether eIF2α was important for parasite development in erythrocytes. For this, we replaced the WT eIF2α in P. berghei by alleles encoding eIF2α in which ser59 was substituted with nonphosphorylatable alanine, or with threonine, which is structurally similar to serine, or with aspartic acid, which mimics a phosphorylated serine (18), using replacement plasmids pBCeIF2α_S/* (A, T, D) (Fig. 3A). As a control, we used plasmid pBCeIF2α_S/S, which contained a synonymous mutation in the ser59 codon (TCA to TCg) tagged by a unique BstBI restriction site, for evaluating the frequency of mutation correction during double cross-over recombination of the plasmid series. The plasmids were independently transfected in P. berghei blood-stage parasites, and then the recombinant parasites were selected under pyrimethamine pressure in mice (19) and analyzed by PCR analysis of the eIF2α gene using primers that amplify a 1,280-bp eIF2α fragment encompassing the ser59 codon from gDNA. In three independent experiments analyzing populations obtained after transfection of the control plasmid pBCeIF2α_S/S (S/S populations), the majority of the PCR fragments contained the BstBI-tagged S/S mutation. This was shown by the relative amounts of the 480- and 800-bp products after BstBI digestion (Fig. 3B). Therefore, a mutation in ser59 codon in the plasmid was frequently transferred to the endogenous eIF2α during plasmid recombination as long as it did not affect eIF2α function. Similar experiments performed on parasite clones obtained from S/S populations revealed the presence of only the 480-and 800-bp bands (Fig. S6), demonstrating the actual replacement of WT eIF2α by the S/S allele. Likewise, eIF2α S/T mutant parasites were obtained in three independent transfections of plasmid pBCeIF2α_S/T, indicating that threonine functionally replaced serine at position 59 in eIF2α. The presence of PbeIF2α S/T and S/S mutations was also verified by DNA sequencing and by replacement-specific gPCR analysis (Fig. 3C). The S/S and S/T gametocytes were able to infect mosquitoes and generated normal numbers of sporozoites. The salivary gland sporozoites normally infected mice after a mosquito bite or i.v. injection.
Fig. 3.
Generation of parasites with PbeIF2α Ser59 substitutions. (A) Diagram depicting the PbeIF2α genomic locus, the pBCeIF2α_S/* (A, T, D, or S) allelic replacement vectors, and the PbeIF2α Ser59 mutant parasite locus. Triple lines indicate the replacement vector backbone. The mutated nucleotides are presented in lowercase. The DHFR cassette is under the control of the 5′ UTR of EF1α and the 3′UTR of DHFR-TS. The restriction sites MscI and BstBI (underlined) were introduced with the S/A and S/S mutations, respectively. (B) BstBI digestion analysis of the PbeIF2α PCR product from S/S population gDNA. (C) Replacement-specific PCR analysis of PbeIF2α genomic locus. Lanes 1, 3, and 5: primers P1+P2; lanes 2, 4, and 6: primers P3+P4. Confirmation of integration is shown by primers P1+P2 and P3+P4 that amplify an 820- and a 550-bp fragment, respectively, from the gDNA of PbeIF2α S/S clone C1 and S/T clone E1; note that these bands were absent in the wild-type parasites. (D) MscI digestion analysis of the PbeIF2α PCR product from the pBCeIF2α_S/A-transfected and pyrimethamine-resistant parasite population gDNA. MscI digestion of the PbeIF2α PCR product from wild-type parasites produced two bands of 720 and 560 bp. Although three bands of 450, 270, and 560 bp were expected from digestion of the S/A mutant population, only the wild-type pattern was observed.
In contrast, when plasmid pBCeIF2α_S/A was transfected into WT P. berghei, only one resistant population was obtained in four independent transfection experiments. However, PCR analysis of the resistant parasite population showed that the MscI-tagged S/A mutation had been corrected during plasmid integration (Fig. 3D) (20). In agreement with this finding, parasite clones from the resistant population contained only the recombinant eIF2α locus lacking the S/A mutation. We conclude that the eIF2α S/A mutation is deleterious to parasite viability. Similarly, transfection of plasmid pBCeIF2α_S/D into WT P. berghei failed to generate any resistant parasite population in three independent experiments. Presumably, the eIF2α S/D mutation inhibited protein synthesis, but this inhibition could not be reversed as normally occurs by removal of PO4 from eIF2α-P, which is required for the continuation of the life cycle (13). We cannot exclude, however, that the S/D mutation inactivates the central function of eIF2α in translation. In conclusion, the parasite blood stages bearing eIF2α S/A or S/D mutations were not viable, whereas the S/S or S/T mutant parasites were able to complete their life cycles successfully.
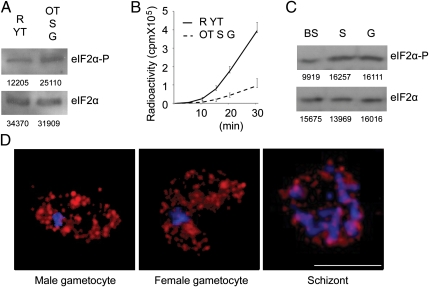
The overall conclusion from the PK4 and eIF2α studies is that the phosphorylation of ser59 by PK4 is required for the completion of the erythrocytic cycle of Plasmodium. This cycle starts when merozoites invade erythrocytes and transform into ring stages; these grow into trophozoites and schizonts where they undergo DNA replication and generate new merozoites. Some merozoites transform into gametocytes that circulate for days in the blood until picked up by mosquitoes. Next, we tried to identify the parasite stage at which ser59 is phosphorylated and protein synthesis is attenuated. We subjected the blood of P. berghei-infected mice to Percoll density gradient centrifugation and separated two fractions: the top fraction contained schizonts, old trophozoites, and gametocytes, and the bottom fraction contained ring stages and young trophozoites. We found that protein synthesis was inhibited in the top fraction (Fig. 4B) and that their eIF2α was highly phosphorylated (Fig. 4A). In addition, we found heavily phosphorylated eIF2α in P. berghei schizonts and gametocytes obtained directly from blood cultures (Fig. 4C). In cells under stress, the untranslated messages and binding proteins are known to accumulate in the cytoplasm as granules (22). Indeed, the accumulation of stress granules containing eIF2α-P was documented in cultured P. falciparum gametocytes and schizonts (Fig. 4D). We conclude that PK4 is active in gametocytes and schizonts.
Fig. 4.
Phosphorylation of eIF2α in gametocytes and schizonts. (A) P. berghei ANKA-infected mouse blood was separated by 70% percoll gradient centrifugation. (Upper) Old trophozoites (OT), schizonts (S), and gametocytes (G). (Lower) Ring stage (R) and young trophozoites (Y). Upper and Lower bands were collected and uninfected erythrocytes were removed by 0.2% saponin treatment. PbeIF2α-P and total PbeIF2α were revealed by Western blots using specific antibodies (21). Densitometry results are presented below each band. This experiment was repeated with identical results. (B) Kinetic incorporation of [35S] Met/Cys in parasites from percoll separated Upper and Lower bands. (C) Mature schizonts were synchronized in an in vitro culture of blood from a P. berghei ANKA-infected mouse. Gametocytes were collected from the infected mouse treated with pyrimethamine for 2 d. Blood stages, BS. PbeIF2α-P and total PbeIF2α were revealed by Western blots. Densitometry results are presented below each band. (D) Stress granule-associated protein phosphorylated eIF2α (22) was revealed by immunofluorescence in P. falciparum male and female gametocytes and in schizonts. (Scale bar, 10 μm.)
Discussion
The complex life cycle of apicomplexan parasites involves drastic morphological and physiological changes that require precise control of transcription (23, 24) and posttranscriptional modifications (25, 26). One of these, the regulation of translation of mRNAs, has a prominent role in Plasmodium development. Translation can be selectively repressed: before fertilization of female gametocytes present in the mosquito midgut, a RNA helicase inhibits specific mRNAs, and they accumulate in the cytoplasm as P granules. Translation is derepressed only in the zygotes (27–29). Another striking example is the selective repression of translation by the RNA-binding protein Puf-2, a member of the Pumilio family. Although the protein encoded by the arrested mRNA is unknown, its absence leads to the premature transformation of salivary sporozoites into early liver stages (30). In addition to the inhibition of translation of selected mRNAs, translation can be globally repressed in Plasmodium. This was first documented in salivary gland sporozoites (13) where translation is inhibited by the kinase IK2 that phosphorylates eIF2α. The phosphorylated eIF2α is dephosphorylated only after sporozoites are injected into the mammalian host leading to relief of the translation repression and transformation of the parasite into its liver stages.
We show directly that PK4 phosphorylates eIF2α (Fig. 4A) and that protein synthesis is inhibited in old trophozoites, schizonts, and gametocytes (Fig. 4B). Importantly, PK4 activity is required for the development of Plasmodium blood stages. This was documented both by targeting PK4, and by preventing the phosphorylation of the regulatory serine 59 of eIF2α. The targets of PK4 are gametocytes and schizonts. Mature gametocytes can remain in the blood circulation for many days before being taken up by mosquitoes. Similarly to salivary sporozoites, the global inhibition of protein synthesis leads to a dormant or latent condition in gametocytes, which is overcome only when gametocytes are taken up by mosquitoes. The down-regulation of protein synthesis in schizonts was not surprising; the energy requirements in those end stages need to be focused on DNA replication and on the profound remodeling during assembly and sequestration of organelles in the newly formed merozoites.
Still unresolved is the nature of the physiological signal that activates the PK4. An attractive possibility is the involvement of a nutritional stress as suggested for IK1 (14). Indeed, in schizonts and in mature gametocytes, the red cell hemoglobin is likely expended. It is also plausible that ER stress is involved. PK4, like mammalian PERK, has a classical signal sequence and a long hydrophobic amino acid sequence closer to the C terminus that might serve to anchor PK4 in the ER membrane.
Global translational inhibition is important for the development of another apicomplexan parasite, Toxoplasma gondii. During its cycle in mammals, the rapidly proliferating Toxoplasma tachyzoites develop into dormant bradyzoites, which encyst in several tissues. Bradyzoite development can be mimicked in vitro by applying a variety of stress agents to tachyzoites cultures, and this process promotes eIF2α phosphorylation (21, 31). In Leishmania, the lack of eIF2α phosphorylation delays the differentiation process to the intracellular amastigote stage (32). The phosphorylation of eIF2α is essential for the development of Trypanosoma cruzi in the insect vector (33). An eIF2α kinase has been found in Trypanosoma brucei, but its function is not known (34).
This paper highlights the importance of PK4 and the phosphorylation of eIF2α in two important life cycle transitions: (i) the route from the merozoite-containing schizonts to the invasion of new erythrocytes and (ii) the infection of mosquitoes by gametocytes. During all of these life-cycle conversions, the parasite undergoes an obligatory “latency” stage that is surmounted only by the activity of specific phosphatases. To date, these transitions have not been exploited for developing therapies for malaria (7). We suggest that PK4 inhibitors will alleviate malaria disease and inhibit transmission.
Materials and Methods
In Vitro Kinase Assay.
A “codon-optimized” DNA fragment encoding a segment from the C terminus of PfPK4 that includes the kinase domain (residues 2,109–3,073) was obtained from F. Sicheri (Samuel Lunenfeld Research Institute, Toronto, Canada). To reduce the degradation of PfPK4, a 521-residues region (residues 2,237–2,757) predicted to be sensitive to Escherichia coli proteases was deleted. The PfPK4 kinase domain bearing the deletion (PfPK4KD) was expressed and purified as a GST fusion in E. coli. The purified GST-PfPK4KD and PfeIF2α were incubated with 20 μCi [γ-32P]ATP in a final concentration of 50 μM and kinase buffer solution [50 mM Hepes (pH 7.5), 50 mM NaCl, 0.2 mM tris(2-carboxyethyl)phosphine, 5 mM MgCl2] in a volume of 50 μL at 37 °C for 30 min.
Generation of PbeIF2α Ser59 Mutant Parasites.
The site-directed mutagenesis of PbeIF2α Ser59 was engineered by using the overlap extension PCR method. To generate the allelic replacement construct for PbeIF2α Ser59 substitutions, two fragments (−1,500 bp to −1,000 bp and −1,000 bp to +1,000 bp with mutations) were amplified and inserted on oppositing ends of the hDHFR cassette of the pBC_hDHFR vector, as illustrated in Fig. 3A. The hDHFR cassette is under the control of the 5′UTR of EF1α and 3′ UTR of dihydrofolate reductase-thymidylate synthase (DHFR-TS). Sequences for primers used for this study are in Table S1. The allelic replacement vectors pBCeIF2α_S/* (A, T, D, or S) were linearized with XholI and AscI restriction endonucleases, transfected into P. berghei ANKA merozoites, and then selected and single-cloned in mice (19).
PbPK4 Knockout Attempts.
For disruption of PK4 by double homologous recombination, two independent constructs were generated (Fig. S1). In the first construct, two 500-bp fragments of the PK4 N terminus and C terminus were cloned into the pBC_GFP_DHFR plasmid, and the targeting construct was named pBC_PbPK4KO construct (1). GFP CDS is flanked by 5′UTR and 3′UTR of Hsp70, and DHFR CDS is controlled by 5′UTR of EF1α and 3′UTR of DHFR-TS. GFP fluorescent parasites were examined under a fluorescence microscope. In the second knockout construct, two 500-bp fragments were amplified from the N terminus and 3′ UTR of the PK4 gene. Both fragments were cloned into the pBC_GFP_DHFR plasmid, and the targeting construct was named pBC-PbPK4KO (2). Primers are described in Table S1.
Construction of PbPK4cKO Parasites.
The conditional knockout targeting vector was constructed by cloning two PCR products into a vector that carries the two Flp recognition target sequences (FRT sites) flanking 3′ UTR of TRAP gene and the hDHFR expression cassette (17). Primers PK4-F8 and PK4-R8 were used to amplify a 650-bp fragment encompassing the extreme C terminus of the gene. Primers PK4-F9 and PK4-R9 were used to amplify a 500-bp fragment encompassing the 3′ UTR of PbPK4. Both fragments were sequentially cloned into plasmid p3′TRAP-hDHFRflirte′3 to generate the targeting construct pUC-PbPK4cKO (Fig. S2A). The SphI/KpnI-linearized plasmid was transfected into P. berghei merozoites (19). After pyrimethamine selection, the resistant parasites were single-cloned.
Southern Hybridization and PCR.
Parasite gDNA was digested with PstI before transfer to a nylon membrane. The membrane was probed with a dioxygenin-labeled 650-bp fragment (DIG High Prime DNA labeling and detection kit; Roche Applied Sciences). DNA hybridization was visualized using a chemiluminescent substrate following the manufacturer's instructions. PCR was performed using gDNA extracted from erythrocytic-stage parasites.
Mosquito Infection with Parasites.
Animal care was in accordance with New York University School of Medicine laboratory animal protocol 010202–01. Anopheles stephensi mosquitoes were fed on anesthetized Swiss Webster mice infected with parasites. Mosquitoes were maintained at 20 ºC until day 16 post blood meal and then transferred to 25 ºC. Sporozoites were dissected from their salivary glands at day 25 post feeding.
Mouse Infection with Parasites.
For transmission of sporozoites to mice, naive C57BL/6 mice (6 wk old) were bitten by infected A. stephensi mosquitoes (25 d post infectious blood meal, 10 mosquitoes per mouse) or injected i.v. (103 or 104 sporozoites). Then infection was monitored daily by microscopic examination of Giemsa-stained thick blood smears.
qPCR Analysis of PK4 Transcription in PbPK4cKO Sporozoites.
RNA was extracted from PbPK4cKO and TRAP/FlpL sporozoites using TRIzol (Invitrogen). RNA was isolated according to the manufacturer's instructions and then reverse-transcribed using a RT-PCR kit (Applied Biosystems) with random hexamers. The real-time PCR was performed as described earlier (35). Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was set as internal control.
IFAs.
HepG2 cells (2.0 × 105/well) were seeded in Permanox eight-chambered Laboratory-Tek wells. A total of 2 × 104 sporozoites was added to the cells and then centrifuged at 1,500 × g for 3 min. Two days later, cells were fixed with methanol and stained with mAb 2E6 (36) followed by goat anti-mouse IgG conjugated to FITC. IFA was performed in triplicate, and at least 50 fields per well were counted.
qPCR Determination of Sporozoite Infectivity.
Two days post sporozoite infection of HepG2 cells in vitro, RNA was isolated and cDNA was made, followed by qPCR (35). In vivo, C57BL/6 mice were i.v. injected with 104 sporozoites, and 40 h post infection the parasite load within each mouse liver was determined. The normalized liver-stage parasite burden was determined as the following ratio: P. berghei 18S RNA copy number to mouse GAPDH copy number.
In Situ Hybridization and IFA.
In situ hybridization and immunofluorescence assay were perform as previously described except that 0.1% saponin was used instead of Triton X-100 to permeabilize infected HepG2 cells and a Cy3-labeled probe to detect PbPK4 mRNA (7.5μg/mL) [sequence: (Cy3)GGTACCCTGCGTTCCGGTTTCGTCACTTATAGTA] was used instead of a biotinylated oligo(dT) probe (37, 38). Liver stages were immunostained with mouse anti–heat-shock protein (anti-heat shock protein 70) (2 μg/mL) followed by FITC donkey anti-mouse IgG (1:100).
Statistical Analysis.
All results are presented as mean values ± SD. Student's t tests were used to determine the statistical significance of differences observed between indicated groups.
Supplementary Material
Acknowledgments
This work was supported by National Institutes of Health Grants AI084031 and AI077502 (to W.J.S.); the Intramural Research Program of the National Institutes of Health (T.E.D.); and National Institutes of Health Grant R01 GM067159 (to B.M.A.F.).
Footnotes
The authors declare no conflict of interest.
*This Direct Submission article had a prearranged editor.
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1121567109/-/DCSupplemental.
References
- 1.Hill AV. Vaccines against malaria. Philos Trans R Soc Lond B Biol Sci. 2011;366:2806–2814. doi: 10.1098/rstb.2011.0091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Noedl H, et al. Artemisinin resistance in Cambodia: A clinical trial designed to address an emerging problem in Southeast Asia. Clin Infect Dis. 2010;51:e82–e89. doi: 10.1086/657120. [DOI] [PubMed] [Google Scholar]
- 3.Borrmann S, et al. Declining responsiveness of Plasmodium falciparum infections to artemisinin-based combination treatments on the Kenyan coast. PLoS ONE. 2011;6:e26005. doi: 10.1371/journal.pone.0026005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Baird JK. Resistance to therapies for infection by Plasmodium vivax. Clin Microbiol Rev. 2009;22:508–534. doi: 10.1128/CMR.00008-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ward P, Equinet L, Packer J, Doerig C. Protein kinases of the human malaria parasite Plasmodium falciparum: The kinome of a divergent eukaryote. BMC Genomics. 2004;5:79. doi: 10.1186/1471-2164-5-79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Tewari R, et al. The systematic functional analysis of Plasmodium protein kinases identifies essential regulators of mosquito transmission. Cell Host Microbe. 2010;8:377–387. doi: 10.1016/j.chom.2010.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.L Solyakov, et al. Global kinomic and phospho-proteomic analyses of the human malaria parasite Plasmodium falciparum. Nature Commun. 2011;2:565. doi: 10.1038/ncomms1558. [DOI] [PubMed] [Google Scholar]
- 8.Proud CG. eIF2 and the control of cell physiology. Semin Cell Dev Biol. 2005;16:3–12. doi: 10.1016/j.semcdb.2004.11.004. [DOI] [PubMed] [Google Scholar]
- 9.Harding HP, Ron D. Endoplasmic reticulum stress and the development of diabetes: A review. Diabetes. 2002;51(Suppl 3):S455–S461. doi: 10.2337/diabetes.51.2007.s455. [DOI] [PubMed] [Google Scholar]
- 10.Dever TE, et al. Phosphorylation of initiation factor 2 alpha by protein kinase GCN2 mediates gene-specific translational control of GCN4 in yeast. Cell. 1992;68:585–596. doi: 10.1016/0092-8674(92)90193-g. [DOI] [PubMed] [Google Scholar]
- 11.Chen JJ. Regulation of protein synthesis by the heme-regulated eIF2alpha kinase: Relevance to anemias. Blood. 2007;109:2693–2699. doi: 10.1182/blood-2006-08-041830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sadler AJ, Williams BR. Structure and function of the protein kinase R. Curr Top Microbiol Immunol. 2007;316:253–292. doi: 10.1007/978-3-540-71329-6_13. [DOI] [PubMed] [Google Scholar]
- 13.Zhang M, et al. The Plasmodium eukaryotic initiation factor-2α kinase IK2 controls the latency of sporozoites in the mosquito salivary glands. J Exp Med. 2010;207:1465–1474. doi: 10.1084/jem.20091975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fennell C, et al. PfeIK1, a eukaryotic initiation factor 2alpha kinase of the human malaria parasite Plasmodium falciparum, regulates stress-response to amino-acid starvation. Malar J. 2009;8:99. doi: 10.1186/1475-2875-8-99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Möhrle JJ, Zhao Y, Wernli B, Franklin RM, Kappes B. Molecular cloning, characterization and localization of PfPK4, an eIF-2alpha kinase-related enzyme from the malarial parasite Plasmodium falciparum. Biochem J. 1997;328:677–687. doi: 10.1042/bj3280677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sullivan WJ, Jr, Narasimhan J, Bhatti MM, Wek RC. Parasite-specific eIF2 (eukaryotic initiation factor-2) kinase required for stress-induced translation control. Biochem J. 2004;380:523–531. doi: 10.1042/BJ20040262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Combe A, et al. Clonal conditional mutagenesis in malaria parasites. Cell Host Microbe. 2009;5:386–396. doi: 10.1016/j.chom.2009.03.008. [DOI] [PubMed] [Google Scholar]
- 18.Kedersha N, et al. Evidence that ternary complex (eIF2-GTP-tRNA(i)(Met))-deficient preinitiation complexes are core constituents of mammalian stress granules. Mol Biol Cell. 2002;13:195–210. doi: 10.1091/mbc.01-05-0221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Janse CJ, Ramesar J, Waters AP. High-efficiency transfection and drug selection of genetically transformed blood stages of the rodent malaria parasite Plasmodium berghei. Nat Protoc. 2006;1:346–356. doi: 10.1038/nprot.2006.53. [DOI] [PubMed] [Google Scholar]
- 20.Nunes A, et al. Subtle mutagenesis by ends-in recombination in malaria parasites. Mol Cell Biol. 1999;19:2895–2902. doi: 10.1128/mcb.19.4.2895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Narasimhan J, et al. Translation regulation by eukaryotic initiation factor-2 kinases in the development of latent cysts in Toxoplasma gondii. J Biol Chem. 2008;283:16591–16601. doi: 10.1074/jbc.M800681200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kedersha N, Anderson P. Mammalian stress granules and processing bodies. Methods Enzymol. 2007;431:61–81. doi: 10.1016/S0076-6879(07)31005-7. [DOI] [PubMed] [Google Scholar]
- 23.Balaji S, Babu MM, Iyer LM, Aravind L. Discovery of the principal specific transcription factors of Apicomplexa and their implication for the evolution of the AP2-integrase DNA binding domains. Nucleic Acids Res. 2005;33:3994–4006. doi: 10.1093/nar/gki709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Painter HJ, Campbell TL, Llinás M. The Apicomplexan AP2 family: Integral factors regulating Plasmodium development. Mol Biochem Parasitol. 2011;176:1–7. doi: 10.1016/j.molbiopara.2010.11.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Chung DW, Ponts N, Cervantes S, Le Roch KG. Post-translational modifications in Plasmodium: More than you think! Mol Biochem Parasitol. 2009;168:123–134. doi: 10.1016/j.molbiopara.2009.08.001. [DOI] [PubMed] [Google Scholar]
- 26.Lopez-Rubio JJ, Mancio-Silva L, Scherf A. Genome-wide analysis of heterochromatin associates clonally variant gene regulation with perinuclear repressive centers in malaria parasites. Cell Host Microbe. 2009;5:179–190. doi: 10.1016/j.chom.2008.12.012. [DOI] [PubMed] [Google Scholar]
- 27.Shaw MK, Thompson J, Sinden RE. Localization of ribosomal RNA and Pbs21-mRNA in the sexual stages of Plasmodium berghei using electron microscope in situ hybridization. Eur J Cell Biol. 1996;71:270–276. [PubMed] [Google Scholar]
- 28.Mair GR, et al. Regulation of sexual development of Plasmodium by translational repression. Science. 2006;313:667–669. doi: 10.1126/science.1125129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Mair GR, et al. Universal features of post-transcriptional gene regulation are critical for Plasmodium zygote development. PLoS Pathog. 2010;6:e1000767. doi: 10.1371/journal.ppat.1000767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Gomes-Santos CS, et al. Transition of Plasmodium sporozoites into liver stage-like forms is regulated by the RNA binding protein Pumilio. PLoS Pathog. 2011;7:e1002046. doi: 10.1371/journal.ppat.1002046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Joyce BR, Queener SF, Wek RC, Sullivan WJ., Jr Phosphorylation of eukaryotic initiation factor-2α promotes the extracellular survival of obligate intracellular parasite Toxoplasma gondii. Proc Natl Acad Sci USA. 2010;107:17200–17205. doi: 10.1073/pnas.1007610107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Chow C, Cloutier S, Dumas C, Chou MN, Papadopoulou B. Promastigote to amastigote differentiation of Leishmania is markedly delayed in the absence of PERK eIF2alpha kinase-dependent eIF2alpha phosphorylation. Cell Microbiol. 2011;13:1059–1077. doi: 10.1111/j.1462-5822.2011.01602.x. [DOI] [PubMed] [Google Scholar]
- 33.Tonelli RR, Augusto LdaS, Castilho BA, Schenkman S. Protein synthesis attenuation by phosphorylation of eIF2α is required for the differentiation of Trypanosoma cruzi into infective forms. PLoS ONE. 2011;6:e27904. doi: 10.1371/journal.pone.0027904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Moraes MC, et al. Novel membrane-bound eIF2alpha kinase in the flagellar pocket of Trypanosoma brucei. Eukaryot Cell. 2007;6:1979–1991. doi: 10.1128/EC.00249-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Bruña-Romero O, et al. Detection of malaria liver-stages in mice infected through the bite of a single Anopheles mosquito using a highly sensitive real-time PCR. Int J Parasitol. 2001;31:1499–1502. doi: 10.1016/s0020-7519(01)00265-x. [DOI] [PubMed] [Google Scholar]
- 36.Tsuji M, Mattei D, Nussenzweig RS, Eichinger D, Zavala F. Demonstration of heat-shock protein 70 in the sporozoite stage of malaria parasites. Parasitol Res. 1994;80:16–21. doi: 10.1007/BF00932618. [DOI] [PubMed] [Google Scholar]
- 37.Chakraborty P, Satterly N, Fontoura BM. Nuclear export assays for poly(A) RNAs. Methods. 2006;39:363–369. doi: 10.1016/j.ymeth.2006.07.002. [DOI] [PubMed] [Google Scholar]
- 38.Chakraborty P, et al. Nucleoporin levels regulate cell cycle progression and phase-specific gene expression. Dev Cell. 2008;15:657–667. doi: 10.1016/j.devcel.2008.08.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.