Abstract
Peptides that bind to fibrin but not to fibrinogen or serum albumin were selected from phage display libraries as targeting moieties for thrombus molecular imaging probes. Three classes of cyclic peptides (cyclized via disulfide bond between two Cys) were identified with consensus sequences XArXCPY(G/D)LCArIX (Ar=aromatic, Tn6), X2CXYYGTCLX (Tn7), and NHGCYNSYGVPYCDYS (Tn10). These peptides bound to fibrin at ~2 sites with Kd=4.1 μM, 4.0 μM, and 8.7 μM, respectively, whereas binding to fibrinogen was at least 100 – fold weaker. The peptides also bind to the fibrin degradation product DD(E) with similar affinity to that measured for fibrin. The Tn7 and Tn10 peptides bind to the same site on fibrin while the Tn6 peptides bind to a unique site. Alanine scanning identified the N- and C-terminal ends of the Tn6 and Tn7 peptides as most tolerant to modification. Peptide conjugates with either fluorescein or diethylenetriaminepentaaceto gadolinium(III) (GdDTPA) at the N-terminus were prepared for potential imaging applications and these retained fibrin binding affinity and specificity in plasma. Relaxivity and binding studies on the GdDTPA derivatives revealed that an N-terminal glycyl linker had modest effect on fibrin affinity but resulted in lower fibrin-bound relaxivity.
Keywords: Fibrin, MR Imaging, binding, peptide, phage display
Introduction
Thrombotic diseases, including atherothrombosis and stroke, are the leading cause of death in the Western world, and consequently there is considerable need for new diagnostic agents and therapies. Aberrant fibrin clot formation is associated with thrombotic disease and is initiated by the proteolysis of fibrinogen by thrombin and subsequent fibrin polymerization. Fibrin is well suited as a marker for diagnostic imaging and depot for targeted therapy as it is inexorably linked to thrombosis and is abundant in thrombi (20 – 200 μM). Several groups have explored fibrin specific antibodies coupled to radionuclides, nanoparticles, or incorporated into echogenic liposomes as potential contrast agents for nuclear, optical or ultrasound imaging(1). With respect to thrombolytic agents, anti-fibrin antibody conjugation has been investigated as a mechanism to enhance the specificity and reactivity of plasminogen activators(2). Additionally, conjugation of the thrombin inhibitor hirudin(3) or the Factor Xa inhibitor tick anticoagulant peptide(4) to anti-fibrin antibodies has improved the specificity and efficacy of these inhibitors, suggesting that a fibrin targeting strategy may also yield more potent antithrombotic agents.
In addition to monoclonal antibodies and the protein tissue plasminogen activator (t-PA), fibrin targeting has also been achieved through small peptides. MR contrast agents were reported that contain fibrin-specific peptide(s) conjugated to multiple gadolinium complexes. Such agents, like EP-2104R, have demonstrated efficacy in thrombus detection in a number of preclinical models of thrombosis (5–11) and recent data in human subjects has also proved promising (12). However, there has been little reported on the fibrin specific peptides themselves: their identification, specificity, cross reactivity for different fibrin binding sites, the effects of chelate conjugation, and sensitivity to amino acid substitution.
In this report we describe the strategy used to identify fibrin binding peptides with high specificity for fibrin over fibrinogen and other serum proteins. Three families of peptides were identified and the interaction of these peptides with fibrin, the soluble fibrin fragment DD(E), and fibrinogen is probed with four techniques: plate-based depletion assay, fluorescence polarization of fluorescent probes and competitive binding, isothermal calorimetry, and proton relaxation rate enhancement. The cross reactivity of these peptides for specific fibrin binding sites is also examined. The critical residues required for binding are probed using alanine and other amino acid substitutions, providing data on the critical binding motif while pointing to conjugation strategies compatible with maintaining binding affinity. The effect of fluorescein or GdDTPA conjugation on protein binding, relaxivity, and specificity is also explored. While aspects of the in vivo efficacy of some of these peptides as conjugates to GdDTPA have been reported, the discovery, identity and properties of these sequences have not been previously described in detail.
Experimental Procedures
Protein Preparation
Human fibrinogen was purchased from American Diagnostica. Human citrated plasma was obtained from the American Red Cross and used without further processing. The soluble fibrin fragment DD(E) was prepared as previously described(13). DD(E) used in this study contained subunits of 61 kD and 72kD, assigned to Fragments E1 and E2 and present in a roughly 1:1 ratio, and 180 kD (Fragment DD).
Phage Display
Tn6, Tn7, and Tn10 libraries were constructed as gene III coat protein fusions by Dyax Corporation and are described elsewhere(14). Phage selection was conducted through four rounds. Before each round, libraries were depleted of fibrinogen binders by incubating the phage with biotinylated fibrinogen bound to bead immobilized streptavidin. Fibrin binding phage were selected by contacting these non-binding phage with either fibrin (polymerized in a microtiter plate well) or to biotinylated DD(E), bound to bead immobilized streptavidin, and eluting the bound phage. Fibrin specific clones from round four were identified by phage ELISA assays as having high response against fibrin and DD(E), but low response against fibrinogen, and human serum albumin. Fibrin plates were prepared with fibrinogen (50 μL, 0.075 mg/mL) in 50 mM Tris, pH 7.4, 150 mM NaCl and 5 mM sodium citrate polymerized in the wells of a 96 well plate (Immulon-II®) by addition of CaCl2 (7 mM) and thrombin (1 U/mL) and dried overnight at 37°C).
Peptide synthesis
Peptides were synthesized by standard Fmoc solid phase protocols using rink amide or NovasynTGR-amide resin from EMD Biosciences, and prepared as C-terminal amides. Masses of all compounds were verified by electrospray mass spectrometry, and purities were >90% as assessed by HPLC. All peptides were prepared as cyclic cysteine disulfides, except where noted. Compounds are denoted by the phage library from which they were derived followed by a number to denote the sequence and then “Fl” or “Gd” if derivatized with fluorescein or GdDTPA, e.g. Tn7-3-Fl denotes a peptide derived from the Tn7 library, sequence 3, and derivatized with fluorescein. Specific sequences are listed in Tables 1 and 2. Conjugate Tn7-3b-Gd was reported previously (15) and in that publication is referred to as EP-782.
Table 1.
Peptides and Peptide conjugates discussed in this work. Equilibrium dissociation (Kd) and inhibition (Ki) constants are given in micromolar (μM). Values in parentheses represent the number of equivalent binding sites, Nbd, obtained from the fit to the data.
| Compound | Sequence | Kd (Nbd) Fibrina | Kd (Nbd) DD(E)b | Ki DD(E)c |
|---|---|---|---|---|
| Tn6-1 | WFHCPYDLCHIL | 4.1±0.4 (2.3±0.1) | n.d. | 3.1±0.4 |
| Tn6-2a | WECPYGLCWIQ | 2.9±0.2 (2.4±0.4) | 0.85±0.03 (2.0±0.1) | 0.9±0.2 |
| Tn6-2b | QWECPYGLCWIQ | n.d. | n.d. | 2.3±0.5 |
| Tn6-2b-Fl | Fluor-Aca-QWECPYGLCWIQ | n.d. | 0.057±0.006 | n.d. |
| Tn6-1-Gd | GdDTPA-GG-WFHCPYDLCHIL | 3.1±0.3 (2.3±0.1) | n.d. | 2.2±0.2 |
| Tn7-3 | LPCDYYGTCLD | 4.0±0.3 (2.1±0.05) | 2.2±0.06 (1.9±0.1) | 2.8±0.7 |
| Tn7-3-Fl | Fluor-Aca-LPCDYYGTCLD | n.d. | 1.3±0.4 | n.d. |
| Tn7-3a-Gd | GdDTPA-LPCDYYGTCLD | 6.9±0.8 (3.0±0.1) | n.d. | 2.9±0.2 |
| Tn7-3b-Gd | GdDTPA-GLPCDYYGTCLD | 8.4±0.5 (1.8±0.1) | n.d. | 3.9±0.3 |
| Tn10-4 | Ac-NHGCYNSYGVPYCDYS | 8.7±1.5 (2.3±0.1) | 3.0±0.1 (1.6±0.1) | 3.5±0.3 |
| Tn10-4-Fl | Fluor-AEGTGSNHGCYNSYGVPYCDYSAPG | n.d. | 2.7±0.6 | n.d |
direct binding to fibrin coated plate.
direct binding to DD(E) measured by fluorescence anisotropy for Tn6-2b-Fl, Tn7-3-Fl, Tn10-4-Fl and by isothermal calorimetry for the other compounds.
determined by inhibition of binding of the corresponding fluorescent probe, e.g. Tn6-1 inhibiting binding of Tn6-2b-Fl.
Table 2.
Structural requirements for Tn6, Tn7 and Tn10 peptides, as determined by alanine scanning and DD(E) binding. Binding of Tn7 - Tn10 hybrid peptides are also presented. Data obtained by FP probe displacement assay using Tn6-2b-Fl (Tn6) and Tn7-3-Fl (Tn7 and Tn10). Standard errors were ≤10%.
| Tn6-1 | Ki (μM) | Tn6-2b | Ki (μM) | Tn7-3 | Ki (μM) | Tn10-4 | Ki (μM) |
|---|---|---|---|---|---|---|---|
| WFHCPYDLCHIL | 3.1 | QWECPYGLCWIQ | 2.3 | LPCDYYGTCLD | 2.8 | NHGCYNSYGVPYCDYS | 3.5 |
| AFHCPYDLCHIL | 8.0 | AWECPYGLCWIQ | -- | ALCDYYGTCLD | 3.1 | NHGCYDSYGVPYCDYS | >100 |
| WAHCPYDLCHIL | 3.9 | QAECPYGLCWIQ | 4.3 | LACDYYGTCLD | 2.9 | NHGCYNYYGVPYCDYS | 5.9 |
| WFACPYDLCHIL | 3.3 | QWACPYGLCWIQ | 6.2 | LPCAYYGTCLD | 5.9 | NHGCYNYYGTPYCDYS | >500 |
| WFHCAYDLCHIL | 62 | QWECAYGLCWIQ | 32 | LPCDAYGTCLD | 4.5 | NHGCYDYYGTPYCDYS | >500 |
| WFHCPADLCHIL | 16 | QWECPAGLCWIQ | 42 | LPCDYAGTCLD | >100 | ||
| WFHCPYALCHIL | 16 | QWECPYALCWIQ | 7.7 | LPCDYYATCLD | 12 | ||
| WFHCPYDACHIL | 97 | QWECPYGACWIQ | 93 | LPCDYYGACLD | >100 | ||
| WFHCPYDLCAIL | 28 | QWECPYGLCAIQ | 4.3 | LPCDYYGTCAD | 15 | ||
| WFHCPYDLCHAL | 19 | QWECPYGLCWAQ | 17 | LPCDYYGTCLA | 7.3 | ||
| WFHCPYDLCHIA | 2.6 | QWECPYGLCWIA | -- | LPCDYYGVCLD | 28 |
Fibrin Binding
The fibrin binding assay has been described previously (16). For peptide binding experiments, fibrin plates were prepared as for phage display but at higher fibrin concentration (100 μL; 2.5 mg/mL). Peptide or conjugate (1 – 100 μM; 100 μL/well), dissolved in water, were incubated in duplicate fibrin plate wells for 2 hr at 37°C. Peptide concentrations of the peptide solution added to the wells, [Peptide]total, and in the well supernatant at equilibrium, [Peptide]free, were quantified by RP-HPLC (C4 column) interfaced to a mass spectrometer or fluorescence detector. Bound peptide concentration was calculated as the difference [Peptide]total - [Peptide]free. The dissociation constant, Kd, and number of binding sites, Nbd, were determined from a non-linear least-squares fit of the data, Nbd vs. [Peptide]free, to a single class of sites binding model where Nbd is the number of molecules bound per mol fibrin (fbn), and [fbn] is the fibrin concentration, eq 1.
| (1) |
Fluorescence polarization
Peptide and peptide conjugate binding to DD(E) was measured by a fluorescence polarization (FP) assay. The anisotropy (robs) of the fluorescein labeled peptides (Fluor•pep) binding to DD(E) (0 – 20 μM) or fibrinogen (0 – 40 μM) was measured in TBS•Ca at fixed Tn6-2b-Fl (0.05 μM), Tn7-3-Fl (0.1 μM), or Tn10-4-Fl (0.1 μM) concentrations, and the data were fit to a single site model(17) to obtain the dissociation constant for the DD(E) • (fluorescent peptide, “Fl”) complex, Kd, and reference values for rbd, the anisotropy of “Fl” when fully bound to DD(E) and rfr, the anisotropy in solution.
| (2) |
Non-fluorescent peptide binding to DD(E) was measured by fluorescent peptide displacement. Tn6 binding: DD(E) (150 nM), Tn6-2b-Fl (50 nM) and peptide (0.1 – 40 μM) were mixed in TBS•Ca containing 0.01% Triton X100. Tn7 binding: DD(E) (1.5 μM) and Tn7-3-Fl (1 μM) and competing peptide (0.1 – 50 μM) were mixed in TBS•Ca. Sample robs (100 μL, n = 3 wells) was measured in a 96 - well microplate (Costar Cat. No. 3915), using a Tecan Polarian 96 - well FP microplate reader (ex = 485 nm; em = 535 nm). In the presence of an inhibitor, an apparent dissociation constant for the fluorescent probe, Kdapp, is determined, eq 3. The inhibition constant, Ki, is related to Kdapp by eq 4 where Kd is the true dissociation constant of the fluorescent probe measured in the absence of inhibitor. Ki values were obtained by least squares fitting of the data as described elsewhere (18).
| (3) |
| (4) |
Isothermal Calorimetry
Peptide binding to fibrinogen or DD(E) was measured by isothermal calorimetry (ITC) carried out at MicroCal, LLC Northampton, MA. Heat changes were measured for titration of peptides Tn6-2a (721 μM), Tn7-3 (1.097 mM), or Tn10-4 (551 μM) into DD(E) (16 μM in TBS•Ca) or fibrinogen (18 μM in TBS containing 10 mM citrate). Integrated heats of binding data determined from the DD(E) titrations were fit to a single class of sites, non-cooperative binding model (19).
Relaxation rate measurements
Solvent water 1H relaxation rates (1/T1) were measured using a Bruker NMS 120 minispec operating at 0.47T and 37°C. Solutions (200 μL) containing 0 – 250 μM GdDTPA or Tn7-3a-Gd were prepared in pH 7.4 TBS with or without 30 μM fibrinogen. The fibrinogen solutions were converted to fibrin gels by addition of by addition of 4 μL of 2M CaCl2 and 2 μL of 100 U/mL thrombin solution, followed by rapid mixing and then equilibration for at least 30 min. Relaxivity of the fibrin-bound complex, r1bd, and the unbound complex, r1free, were determined by a nonlinear least squares fit of equations 5 and 6 where fbd and ffree are the mol fraction of the bound and unbound species, respectively and 1/T10 is the relaxation rate in the absence of Gd. In the absence of fibrin, fbd = 0, and r1free is determined from a linear fit of 1/T1 vs [Gd].
| (5) |
| (6) |
Supporting Information Available
Additional description of phage display screening and selection procedures (including phage binding and selectivity assays measured by ELISA), synthesis of peptides and conjugates, and fibrin, fibrinogen, and DD(E) binding assays are provided. This material is available free of charge via the internet at http://pubs.acs.org.
Results
Phage Display
Our strategy for isolating fibrin specific peptides by phage display relied upon three key elements: alternative target presentations, negative selection to remove fibrinogen binders, and selection from multiple phage libraries. The target fibrin was presented in two different forms, as polymerized, gelled fibrin and as the plasmin degradation product DD(E). DD(E) is a useful soluble model of the insoluble fibrin polymer and has been shown to retain many epitopes unique to fibrin, including the D-D and D–E domain interfaces, factor XIIIa crosslinks on the γ-chain, and a fibrin specific t-PA binding site(20). Fibrin was presented as a thin dried film, a format that minimized non-specific phage retention, while maintaining fibril structures similar to those of wet clots(21). The fibrin clots were stable to the phage selection procedures with negligible loss of protein from the plate upon repeated washing, and bound fibronectin (IC50 = 4.9 ± 1.9 nM, ELISA data not shown) with affinity similar to previously reported values(22). The high degree of structural similarity between fibrin and fibrinogen necessitated a negative screen to remove fibrinogen-binding phage which was repeated before each round of panning to promote removal of fibrinogen binders potentially carried over and amplified from the previous round.
Fibrin specific phage were obtained from three phage libraries, all containing cysteine disulfide cross-linked sequences: X3CX4CX3 (Tn6 library), X3CX5CX3 (Tn7) and X3CX8CX3 (Tn10) where X was variagated. These selectants were identified after four rounds of selection by phage ELISA, as yielding high responses on the immobilized DD(E) and fibrin, but low background signals with immobilized fibrinogen and HSA. Sequencing of these clones (>20/library) yielded a single consensus sequence for each library. The two unique sequences obtained from the Tn6 library, QWECPYGLCWIQ and WFHCPYDLCHIL together conformed to a XArXCPY(G/D)LCArIX (Ar = aromatic) consensus. The ten unique Tn7 sequences aligned to a X2CXYYGTCLX sequence where the YYGT sequence was strictly conserved along with a preference for polar amino acids (H, D, S, and N) at the lone variable position within the disulfide loop. A single sequence was obtained from the Tn10 selection, NHGCYNSYGVPYCDYS, which contained a ‘SYGV’ motif that appeared to be functionally homologous to the ‘YYGT’ motif of the Tn7 sequence. Sequences for the Tn6, Tn7 and Tn10 peptides were compared against the protein sequence database, but no proteins having a functional role related to fibrin(ogen) recognition or haemostasis were found. The Tn7 consensus sequence YYGT is similar to a surface exposed loop on the fibrin(ogen) gamma chain YYQGGT, γ(348–353), and to the sequence SYGT, α(257–261), but no functional role has been ascribed to these sequences.
Analysis of Peptide Binding to Fibrin, DD(E) and Fibrinogen
Fibrin Binding Measurements
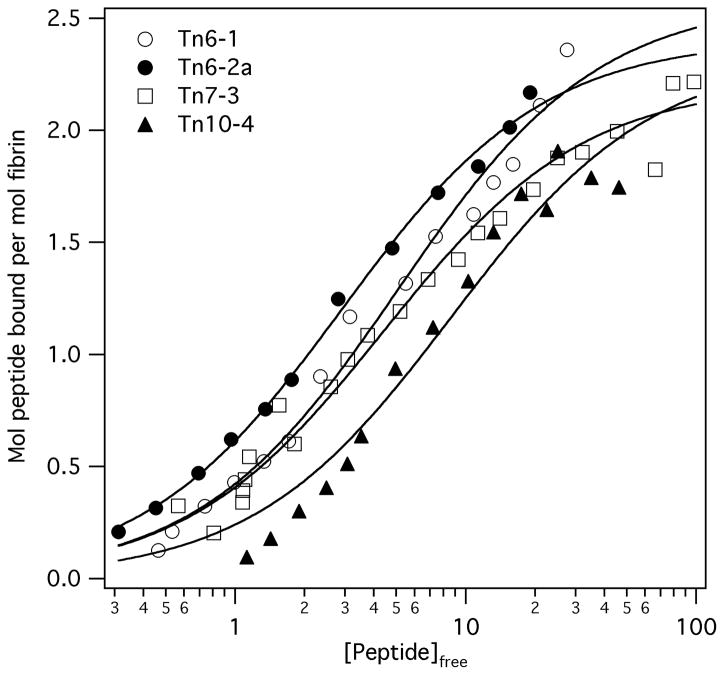
Peptides corresponding to representative selected sequences from the Tn6, Tn7 and Tn10 libraries were synthesized, and binding to fibrin films was measured in a microtiter plate equilibrium binding assay. This method allowed for rapid diffusion of the peptide into the fibrin gel and equilibrium was reached within 45 minutes, as assessed by time courses of peptide uptake into fibrin. Peptides derived from the two Tn6 sequences, Tn6-1 and Tn6-2a (truncation of the N-terminal glutamine from the phage derived sequence Tn6-2b increased affinity two fold and improved peptide solubility), bound to fibrin with similar affinities, Table 1 and Figure 1. A representative Tn7 peptide, Tn7-3, and the Tn10 peptide Tn10-4 also bound with affinities in the low micromolar range, Table 1. Notably, all peptides bound to approximately two binding sites per fibrin monomer, consistent with the dimeric structure of this protein. Peptide affinities, however, were considerably lower than expected based on phage ELISA where very tight binding was observed. This affinity difference was probably due to avidity effects arising from polyvalent peptide display (up to five peptides per phage), although some additional affinity due to the native phage protein cannot be ruled out. No fibrin binding was observed for a modified Tn7 peptide (sequence LPCDYYGACLD), where the single threonine residue was replaced with alanine. This observation highlighted the importance of this consensus residue in fibrin binding and also indicated that binding measurements were not complicated by peptide entrapment.
Figure 1.
Fibrin binding data for the Tn6-1, Tn6-2a, Tn7-3, and Tn10-4 peptides, and fits to single class-of-sites binding model demonstrating low micromolar affinity and binding to two sites on fibrin. Standard errors for individual points were <15% of the ordinate value.
Fluorescence Polarization
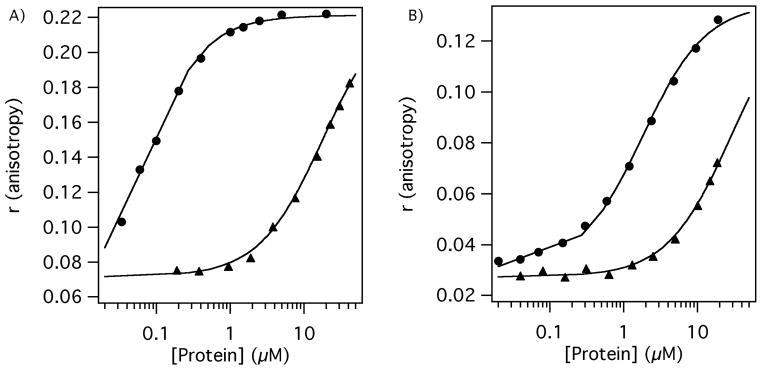
Selectivity and affinity of the Tn6, Tn7 and Tn10 peptides to DD(E) and fibrinogen was measured by a fluorescence polarization assay using N-terminal fluorescein conjugates of these peptides. Affinity of the fluorescein derivatives of the Tn7 and Tn10 peptides to DD(E) was low micromolar, similar to that of the parent peptides to fibrin or DD(E), Table 1 and Figure 2. On the other hand, affinity of Tn6-2b-Fl for DD(E) was higher with Kd = 0.057 μM, Figure 2. Binding was saturable for all 3 fluorescent probes.
Figure 2.
Binding of fluorescein labeled peptides Tn6-2b-Fl (0.050 μM, panel A) and Tn7-3-Fl (0.100 μM, panel B) to DD(E) (closed circles) and to fibrinogen (closed triangles) demonstrating high selectivity for DD(E) over fibrinogen.
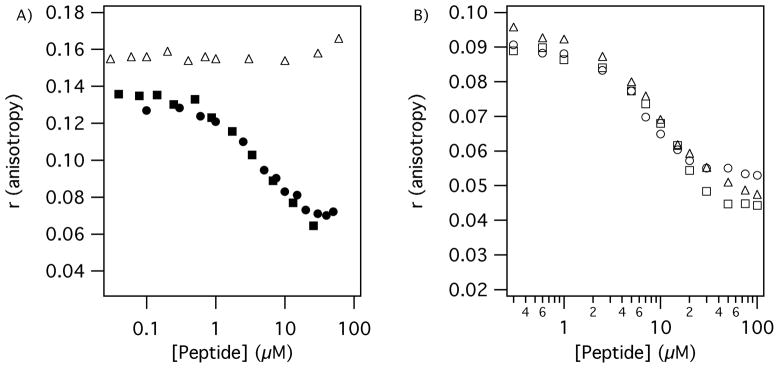
Binding of unlabeled Tn6 and Tn7 peptides to DD(E) was measured by a competition assay (Figure 3), and compared to the fluorescein labeled analogs. Tn7-3 displaced Tn7-3-Fl with Ki = 2.8 μM, an affinity similar to the fluorescein labeled probe. However, Tn6-2a displaced Tn6-2b-Fl with Ki = 0.9 μM, a 16-fold reduction in binding affinity compared to the fluorescent probe, indicating that the aromatic fluorescein moiety enhanced the affinity of this peptide (Figure 3). Likewise, Tn6-1, the other Tn6 sequence type, displaced Tn6-2b-Fl with Ki = 3.1 μM. The DD(E) competition assay also established that the Tn6 and Tn7 peptides bind to separate sites on DD(E), since Tn6-1 and Tn6-2a failed to reduce Tn7-3-Fl anisotropy, nor did the Tn7 peptide Tn7-3 displace Tn6-2b-Fl. However Tn10-4 did displace Tn7-3-Fl (Ki = 3.5 ± 0.3 μM) with affinity similar to Tn7-3 indicating that peptide Tn10-4 also binds to the Tn7 site. The Tn10 peptide could not displace the Tn6 probe at concentrations up to 100 μM.
Figure 3.
DD(E) (1.5 μM) binding measured by displacement of fluorescent probes. A) Tn6-2b-Fl (0.050 μM) is displaced by Tn6 compounds Tn6-1 (filled circles) and Tn6-1-Gd (filled squares) but not by Tn10 peptide Tn10-4 (open triangles). B) Tn7-3-Fl (1.0 μM) is displaced by Tn7 compounds Tn7-3 (open circles) and Tn7-3b-Gd (open squares) and by the Tn10 peptide Tn10-4 (open triangles) indicating the Tn7 and Tn10 binding site is the same.
Despite some functional sequence homology, in that the DYYGT sequence in Tn7 has similarity to the NSYGV sequence in Tn10, the DYYGT sequence cannot functionally substitute for the associated residues in the Tn10 sequence. Four hybrid peptides of the Tn10 peptide were prepared, focusing on the N6, S7 and V9 positions. Replacement of S7 with tyrosine had little effect, while substitution of either N6 with aspartate, V9 with threonine, or all three residues together sharply reduced binding (Table 2). Conversely, replacement of T8 in Tn7-3 with its β-branched analog valine reduced binding to DD(E) 10 – fold. Despite binding to the same site, the residue preference and orientation is exquisitely sensitive to constraints of the disulfide architecture.
The fluorescein labeled peptides exhibited strong selectivity for DD(E) over fibrinogen when anisotropy changes were examined. The data for Tn6-2b-Fl (Figure 2a) fit to a Kd = 22 μM, 400 – fold weaker than to DD(E). The fibrinogen affinity of the unlabeled peptide was much weaker since the fluorescein moiety enhanced fibrinogen binding, as observed with DD(E). The Tn6 data were shifted only slightly in the presence of a 100-fold excess of the unlabeled Tn6-1, see Supporting Information. When binding of Tn6-1 (0 – 50 μM) to fibrinogen was assessed at a fixed concentration of fibrinogen (7 μM) and Tn6-2b-Fl (0.1 μM), displacement was partial and did not change for Tn6-1 concentrations >20 μM. Overall, the data indicated that Tn6-1 competed poorly with Tn6-2b-Fl, and a Kd > 200 μM was estimated. For Tn7-3-Fl, the anisotropy increased in the presence of fibrinogen, but the change was small and nearly linear with increasing fibrinogen concentration, suggesting that the anisotropy effects were due largely to increasing viscosity at high fibrinogen concentrations (Figure 2b). Consistent with a non-specific effect, the anisotropy increase was not reversed by competition, as anisotropy data obtained in the presence of a 100-fold (100 μM) excess of unlabeled Tn7 peptide (Tn7-3) were nearly identical, see Supporting Information. In summary, both the Tn6 and Tn7 sequences have a ~100 – fold selectivity for DD(E) over fibrinogen, as measured by this method.
Isothermal Calorimetry
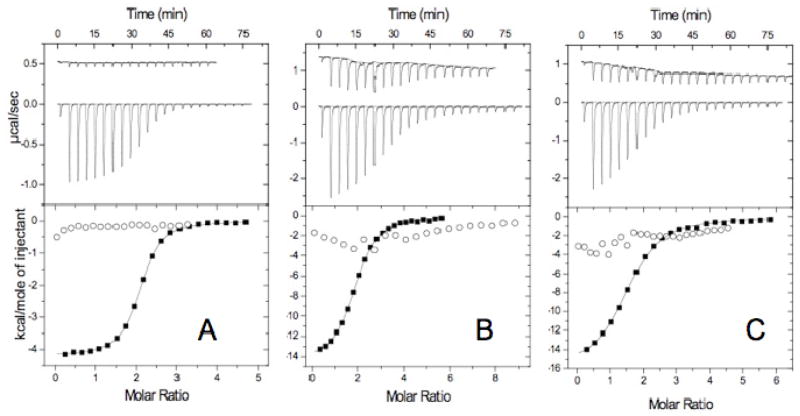
Isothermal calorimetry (ITC) data for binding of the Tn6, Tn7 and Tn10 peptides to DD(E) and fibrinogen confirmed the affinity and specificity measurements obtained by fluorescence polarization (Figure 4). Data for these peptides titrated into a DD(E) solution displayed exothermic binding heats and it appeared at the end of the titrations that nearly all DD(E) binding sites had been occupied by ligand. Binding constants for all three peptides were very close to the values obtained from the fluorescence polarization assay, Table 1. In addition, the data fit to stoichiometries of 2 binding events per DD(E), indicating that, like fibrin, the peptides bind independently to two binding sites on DD(E).
Figure 4.
Calorimetry data for peptide binding to fibrinogen and DD(E). Upper panels: raw heat data measured over a series of injections (Fibrinogen, top curves; DD(E) lower curves). Lower panels: integrated heat per mol injected peptide plotted against the molar ratio of added peptide to protein (Fibrinogen binding, open circles, no fit; DD(E) binding, closed boxes with fit). Conditions for titrations and data were as follows. Figure 4a: Tn6-2a (721 μM) into 28.5 μM fibrinogen, 10 μl/inj. and 24.7 μM DD(E), 5 μL/inj. Figure 4b: 1.099 mM Tn7-3 into 17.5 μM fibrinogen, 10 μl/inj. and 16 μM DD(E) 10 μl/inj. Figure 4c: 462 μM Tn10-4 into 17.5 μM fibrinogen, 10 μl/inj. and 551 μM Tn10-4 into 16 μM DD(E), 10 μl/inj.
Data for Tn6-2a titrated with fibrinogen displayed very small, constant heat effects that were comparable to the control, indicating that any binding interaction between the Tn6 peptide and fibrinogen is weak with Kd > 50 μM. The data for Tn7-3 and Tn10-4 titrated with fibrinogen were more complex. Addition of these peptides to concentrated fibrinogen solutions resulted in fibrinogen aggregation, and formation of a watery gel. However, the heat released with each Tn7-3 addition to fibrinogen was very small compared to those observed with DD(E) and probably arose from fibrinogen aggregation. In a separate experiment (see supplemental data), Tn7-3 was added to a fibrinogen solution (5 mg/mL), and the scattered light intensity, measured by absorbance at 350 nm, increased in 10 min from 0.04 to 0.32 in a sigmoidal transition, confirming that peptide addition induced aggregation of the fibrinogen. This phenomenon did not occur in citrated human plasma, presumably due to lower fibrinogen concentration, and an overall high protein concentration that would disfavor fibrinogen self-association. The titration calorimetry data did not fit to a standard binding model, but it can be inferred that Tn7-3 affinity to fibrinogen is very weak, with Kd ≫ 20 μM. Very similar results were obtained for Tn10-4, consistent with the prior observation that it binds to the same site on fibrin as Tn7-3.
Peptide Binding Determinants
To identify residues in the Tn6 and Tn7 sequences required for fibrin binding, each position (except the cysteines) was systematically changed to alanine (Table 2), and tested for binding to DD(E) in the fluorescence polarization assay. The two Tn6 peptides bind in a similar fashion with residues P5, Y6, and L8 within the PY(G/D)L consensus sequence exhibiting particular sensitivity to substitution and conferring most of the binding affinity. In addition, alanine substitutions at the residues flanking the C-terminal cysteine, especially the aromatic W10/H10 and hydrophobic I11 sites, led to much weaker binding than similar substitutions at the N-terminal flanking region, indicating that the C-terminus contributes more to the peptide-fibrin interaction. For the Tn7 sequence, a substantial reduction in binding was observed for changes at Y6 and T8, highlighting the critical role of these conserved residues for fibrin binding. Reduction of hydrophobicity at L10 also lowered binding affinity, indicating a secondary contribution from this residue. Substitution of the other exocyclic residues or at the D4 position had little effect on binding, consistent with the greater variability observed at these sites in the sequences obtained by phage display.
The conformation as maintained by the cyclic disulfide was also critical for high affinity binding. Reduction of the disulfide in Tn6-1 and Tn7-3 with excess tris(2-carboxyethyl)phosphine (TCEP), or substitution of the cysteines with serine in Tn7-3 abrogated binding to DD(E) and fibrin completely. Even modulation of the disulfide conformation reduced binding considerably. For example, in Tn6-1 either L-cysteine replaced with D-cysteine, or in Tn7-3, substitution of C3 with penicillamine (β, β′-dimethylcysteine) resulted in non-binding peptides.
Gd-DTPA Derivatives
Prototype fibrin targeted MRI contrast agents were synthesized by conjugating the paramagnetic gadolinium chelate Gd(DTPA) to the N-terminus of Tn6-1 and Tn7-3 via 0 – 2 glycyl linkers. Despite the addition of the large Gd(DTPA) moiety, the conjugates Tn6-1-Gd and Tn7-3b-Gd bound to DD(E) and to fibrin in TBS buffer with similar affinity to the parent peptides (Table 1). When binding to fibrin was measured in human citrated plasma, the fibrin affinity was only decreased 2–3-fold: Tn6-1-Gd bound to fibrin with Kd = 3.1 ± 0.3 μM and 2.3 ± 0.1 sites per fibrin monomer, while Tn7-3b-Gd bound with Kd = 8.4 ± 0.5 μM and 1.8 ± 0.1 sites/fibrin. For the Tn7 peptide, both peptide derivatization and plasma binding contributed roughly equally to the observed lower binding affinity, as the Kd values in buffer (Table 1) were intermediate between those measured for the peptide in TBS and for the conjugate in plasma. No measurable fibrin binding was observed for a scrambled version of Tn7-3b-Gd (sequence DTPA-GYLCGDYTLCPD-NH2) (15).
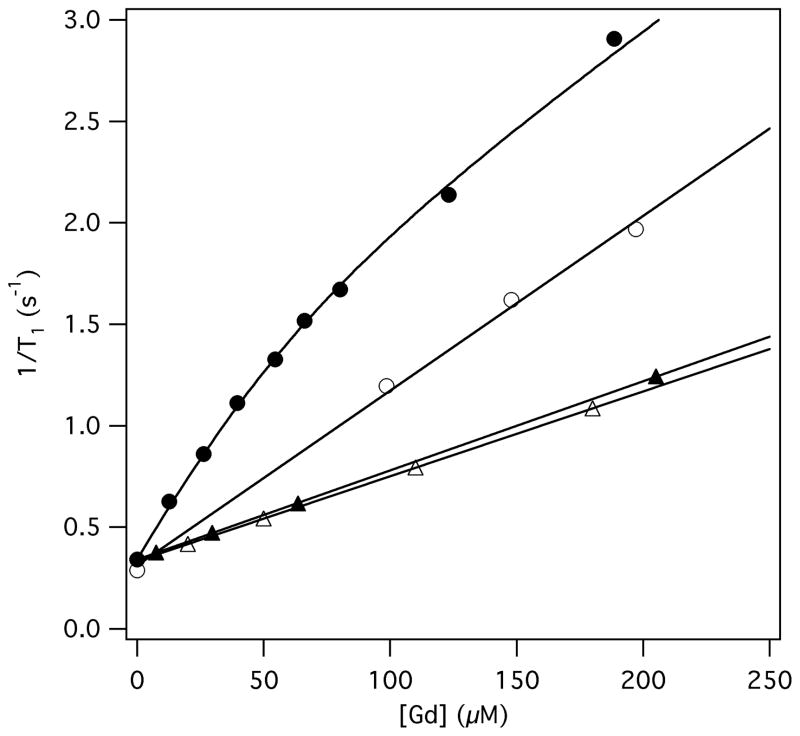
The presence of the paramagnetic Gd(III) moiety allows estimation of fibrin affinity using the proton relaxation enhancement (PRE) effect. The longitudinal relaxation rate (1/T1) of solvent water increases if the peptide conjugate binds to protein. Solutions containing varying concentrations of Tn7-3a-Gd were prepared either in TBS or in TBS containing 30 μM fibrinogen. The fibrinogen solutions were converted to fibrin gels by addition of CaCl2 and thrombin. Figure 5 shows that 1/T1 is higher for Tn7-3a-Gd in the presence of fibrin compared to buffer only. The 1/T1 data is nonlinear for the data in fibrin indicative of a saturable binding event. A control study showed no enhancement of 1/T1 for GdDTPA in fibrin gel compared to GdDTPA in buffer demonstrating a specific interaction between Tn7-3a-Gd and fibrin.
Figure 5.
Relaxation rate data for Tn7-3a-Gd (circles) and GdDTPA (triangles) in the presence (filled symbols) and absence (open symbols) of 30 μM fibrin. Solid lines are fits to the data as described in the text.
The relaxivity of Tn7-3a-Gd bound to fibrin was 25.5 ± 0.7 mM−1s−1 at 37 °C, 0.47T which is significantly higher than that for Tn7-3b-Gd under the same conditions (14.9 mM−1s−1), whereas the relaxivity in the absence of fibrin was similar for the two compounds: 8.6 ± 0.3 and 10.1 mM−1s−1 for Tn7-3a-Gd and Tn7-3b-Gd respectively (15). The increased fibrin-bound relaxivity of Tn7-3a-Gd can be attributed to the lack of the glycyl linker. The GdDTPA moiety is coupled directly to the leucine N-terminus resulting in restricted rotational motion when the compound binds fibrin. While placing the large GdDTPA group close to the conserved portion of the binding sequence could be expected to reduce affinity, in this case affinity was similar regardless of the spacer, while there was a clear advantage to minimizing the spacer length to obtain optimal relaxivity properties.
Discussion
We describe three families of small peptides that discriminate in their binding between two distinct conformational states of fibrinogen, the soluble protein central to haemostasis, and fibrin, the polymerized form that provides the structural network of thrombi. These cyclic peptide classes were obtained by phage display from three disulfide constrained libraries that exhibited high binding affinity to fibrin, in the low micromolar range, and >100-fold selectivity for fibrin over fibrinogen. Fibrin and DD(E) binding was assessed by four independent methods: plate-based depletion, fluorescence polarization and probe displacement, isothermal calorimetry, and proton relaxation enhancement assays. No evidence was observed for specific fibrinogen binding in these assays. Peptide binding to DD(E) was comparable to fibrin, which is evidence that DD(E) is a good structural model for fibrin, since the neo-epitopes recognized by these peptides are structurally maintained and accessible in both proteins. It is remarkable that this degree of specificity is conferred by such short peptides, especially given that much of the overall conformation is expected to be similar between fibrin and fibrinogen(23). A few residues on each peptide were responsible for fibrin recognition, and these hot spots corresponded to conserved residues highlighted in the phage display results. A peptide architecture rigidified by disulfide bonds also proved crucial to holding the peptides in a high affinity conformation.
All peptides bound to two sites per fibrin monomer, consistent with the dimeric structure of fibrin. These peptides potentially recognize new surfaces formed by interdomain contacts present in fibrin, but absent in fibrinogen, or they bind to surfaces exposed by domain movements induced by fibrin polymerization. Identification of the binding site location may help elucidate conformational differences between fibrin and fibrinogen, and is ongoing. The fibrin N-termini, well-known epitopes recognized by fibrin-specific antibodies(24) but absent in DD(E), as well as the ‘a’ and ‘b’ holes, fully or partially occupied in fibrin, can be ruled out. The Tn7/Tn10 binding site is of particular interest as it may be involved in fibrin polymerization, as suggested by these peptides’ ability to induce fibrinogen self-association and gelation. By binding at an interface, such as between the D-E or D-D domains, it may stabilize a complex of three fibrinogen monomers oriented to form a DD(E)-like structure that has similar surface interactions between the monomers, but is of course absent any covalent crosslinks or a′ and ‘b’ site occupation. Such peptide-fibrinogen complexes, even if present at low concentration, evidently speed the rate of protofibril nucleation, the rate determining step in (thrombin independent) fibrin gel assembly (25), seeding further aggregation.
The N-terminus of all three classes of peptide can be modified with either a fluorophore or a GdDTPA moiety without significant loss of affinity, provided a spacer (e.g. glycine) is introduced at the N-terminus. The GdDTPA conjugates of the Tn6 and Tn7 peptides both retained excellent in vitro selectivity and high fibrin binding affinity in plasma, despite the presence of fibrinogen at a concentration equimolar to fibrin and 660 μM HSA. The low μM binding constants are within the range required for fibrin targeting in vivo, since the high concentration of fibrin in human thrombi (25 – 200 μM) should promote binding of these compounds under physiological conditions. For the conjugates here, we conservatively expect a percent bound of >80% resulting in a fibrin:plasma ratio of >4:1. Moreover, the two peptide binding sites per fibrin molecule doubles the effective capacity, providing an added enhancement in imaging sensitivity.
The feasibility of this approach was recently demonstrated with imaging agents based on the Tn6 peptide termed EP-1873 and EP-2104R (16, 26), and on the Tn7 peptide (15, 27, 28). These compounds contain a modified form of the parent Tn6 (or Tn7) peptide and have four Gd chelates attached. The Gd-modified peptides, both Tn6 and Tn7, maintain >100-fold specificity for fibrin over fibrinogen and >1000-fold specificity for fibrin over serum albumin (16, 27). These agents were used in swine models of coronary thrombosis (5, 7), pulmonary embolism (7, 8, 10), atrial thrombus (8), and cerebral venous sinus thrombosis (11), rabbit models of ruptured atherosclerotic plaque (26) and carotid thrombosis (29), and rat models of ischemic stroke (30, 31). Recent clinical trial data with a fibrin-targeted gadolinium-based probe, EP-2104R, indicated that this probe can identify thrombi in the heart chambers, carotid arteries, or aortic arch (32). The positive image contrast persists for hours (12), indicating a slow off-rate of the peptide from the clot due to the high concentration of fibrin binding sites.
These peptides could also find applications in other modalities. Fluorescence imaging of fibrin is readily achieved with the fluorescein derivatives described herein and such probes may find use in optical imaging applications such as intravital microscopy (33). Alternatively, these peptides could be coupled to plasminogen activators to enhance the clot specificity of fibrinolytic treatments, or conjugated to antithrombotic compounds to inhibit active clotting factors associated with pathological thrombi while minimizing systemic side effects. These small peptides are easily synthesized and may be readily conjugated without loss of function, thus providing a new and more flexible approach to fibrin targeting than any previous strategies.
CONCLUSIONS
Phage display, with a preselection to remove fibrinogen and serum protein binders, is a powerful tool to identify fibrin specific peptides. Three classes of disulfide bridged cyclic peptides were identified that bind to two unique sites on fibrin and DD(E). The peptides show only weak nonspecific affinity to fibrinogen or plasma proteins. Alanine and other point substitutions identify the amino acids critical to fibrin binding within these short peptides. The peptides are readily modified at the N-terminus without affinity loss and represent useful building blocks to create conjugates for fibrin-targeted imaging or therapy.
Supplementary Material
Acknowledgments
This work was supported in part by Award Number R21NS075627 from the National Institute Of Neurological Disorders And Stroke to P.C.
Footnotes
Supporting Information Available: Additional details of the phage display study, protein preparations, binding assays, and peptide and conjugate syntheses. This material is available free of charge via the Internet at http://pubs.acs.org.
References
- 1.Morawski AM, Lanza GA, Wickline SA. Targeted contrast agents for magnetic resonance imaging and ultrasound. Curr Opin Biotechnol. 2005;16:89–92. doi: 10.1016/j.copbio.2004.11.001. [DOI] [PubMed] [Google Scholar]
- 2.Runge MS, Harker LA, Bode C, Ruef J, Kelly AB, Marzec UM, Allen E, Caban R, Shaw SY, Haber E, Hanson SR. Enhanced thrombolytic and antithrombotic potency of a fibrin-targeted plasminogen activator in baboons. Circulation. 1996;94:1412–22. doi: 10.1161/01.cir.94.6.1412. [DOI] [PubMed] [Google Scholar]
- 3.Peter K, Graeber J, Kipriyanov S, Zewe-Welschof M, Runge MS, Kubler W, Little M, Bode C. Construction and functional evaluation of a single-chain antibody fusion protein with fibrin targeting and thrombin inhibition after activation by factor Xa. Circulation. 2000;101:1158–64. doi: 10.1161/01.cir.101.10.1158. [DOI] [PubMed] [Google Scholar]
- 4.Hagemeyer CE, Tomic I, Jaminet P, Weirich U, Bassler N, Schwarz M, Runge MS, Bode C, Peter K. Fibrin-targeted direct factor Xa inhibition: construction and characterization of a recombinant factor Xa inhibitor composed of an anti-fibrin single-chain antibody and tick anticoagulant peptide. Thromb Haemost. 2004;92:47–53. doi: 10.1160/TH04-01-0047. [DOI] [PubMed] [Google Scholar]
- 5.Botnar RM, Buecker A, Wiethoff AJ, Parsons EC, Jr, Katoh M, Katsimaglis G, Weisskoff RM, Lauffer RB, Graham PB, Gunther RW, Manning WJ, Spuentrup E. In vivo magnetic resonance imaging of coronary thrombosis using a fibrin-binding molecular magnetic resonance contrast agent. Circulation. 2004;110:1463–6. doi: 10.1161/01.CIR.0000134960.31304.87. Epub 2004 Jul 6. [DOI] [PubMed] [Google Scholar]
- 6.Sirol M, Fuster V, Badimon JJ, Fallon JT, Moreno PR, Toussaint JF, Fayad ZA. Chronic thrombus detection with in vivo magnetic resonance imaging and a fibrin-targeted contrast agent. Circulation. 2005;112:1594–600. doi: 10.1161/CIRCULATIONAHA.104.522110. Epub 2005 Sep 6. [DOI] [PubMed] [Google Scholar]
- 7.Spuentrup E, Buecker A, Katoh M, Wiethoff AJ, Parsons EC, Botnar RM, Weisskoff RM, Graham PB, Manning WJ, Gunther RW. Molecular magnetic resonance imaging of coronary thrombosis and pulmonary emboli with a novel fibrin-targeted contrast agent. Circulation. 2005;111:1377–1382. doi: 10.1161/01.CIR.0000158478.29668.9B. [DOI] [PubMed] [Google Scholar]
- 8.Spuentrup E, Fausten B, Kinzel S, Wiethoff AJ, Botnar RM, Graham PB, Haller S, Katoh M, Parsons EC, Manning WJ, Busch T, Gunther RW, Buecker A. Molecular magnetic resonance imaging of atrial clots in a swine model. Circulation. 2005;112:396–399. doi: 10.1161/CIRCULATIONAHA.104.529941. [DOI] [PubMed] [Google Scholar]
- 9.Spuentrup E, Katoh M, Wiethoff AJ, Parsons EC, Botnar RM, Mahnken AH, Gunther RW, Buecker A. Molecular magnetic resonance imaging of pulmonary emboli with a fibrin-specific contrast agent. Am J Resp Crit Care Med. 2005;172:494–500. doi: 10.1164/rccm.200503-379OC. [DOI] [PubMed] [Google Scholar]
- 10.Spuentrup E, Katoh M, Buecker A, Fausten B, Wiethoff AJ, Wildberger JE, Haage P, Edward CP, Botnar RM, Graham PB, Vettelsehoss M, Gunther RW. Molecular MR imaging of human thrombi in a swine model of pulmonary embolism using a fibrin-specific contrast agent. Invest Radiol. 2007;42:586–595. doi: 10.1097/RLI.0b013e31804fa154. [DOI] [PubMed] [Google Scholar]
- 11.Stracke CP, Katoh M, Wiethoff AJ, Parsons EC, Spangenberg P, Spuntrup E. Molecular MRI of cerebral venous sinus thrombosis using a new fibrin-specific MR contrast agent. Stroke. 2007;38:1476–1481. doi: 10.1161/STROKEAHA.106.479998. [DOI] [PubMed] [Google Scholar]
- 12.Spuentrup E, Botnar RM, Wiethoff AJ, Ibrahim T, Kelle S, Katoh M, Oezgun M, Nagel E, Vymazal J, Graham PB, Gunther RW, Maintz D. MR imaging of thrombi using EP-2104R, a fibrin-specific contrast agent: initial results in patients. Eur Radiol. 2008;18:1995–2005. doi: 10.1007/s00330-008-0965-2. [DOI] [PubMed] [Google Scholar]
- 13.Moskowitz KA, Budzynski AZ. The (DD)E complex is maintained by a composite fibrin polymerization site. Biochemistry. 1994;33:12937–44. doi: 10.1021/bi00248a001. [DOI] [PubMed] [Google Scholar]
- 14.Sato AK, Sexton DJ, Morganelli LA, Cohen EH, Wu QL, Conley GP, Streltsova Z, Lee SW, Devlin M, DeOliveira DB, Enright J, Kent RB, Wescott CR, Ransohoff TC, Ley AC, Ladner RC. Development of mammalian serum albumin affinity purification media by peptide phage display. Biotechnol Prog. 2002;18:182–92. doi: 10.1021/bp010181o. [DOI] [PubMed] [Google Scholar]
- 15.Nair S, Kolodziej AF, Bhole G, Greenfield MT, McMurry TJ, Caravan P. Monovalent and Bivalent Fibrin-specific MRI Contrast Agents for Detection of Thrombus. Angew Chem Int Ed Engl. 2008;47:4918–21. doi: 10.1002/anie.200800563. [DOI] [PubMed] [Google Scholar]
- 16.Overoye-Chan K, Koerner S, Looby RJ, Kolodziej AF, Zech SG, Deng Q, Chasse JM, McMurry TJ, Caravan P. EP-2104R: a fibrin-specific gadolinium-based MRI contrast agent for detection of thrombus. J Am Chem Soc. 2008;130:6025–39. doi: 10.1021/ja800834y. [DOI] [PubMed] [Google Scholar]
- 17.Burke T, Bolger R, Checovich W, Lowery R. Measurement of Peptide Binding Affinities Using Fluorescence Polarization. In: Kay BK, Winter J, McCafferty J, editors. Phage Display of Peptides and Proteins: A Laboratory Manual. Academic Press; San Diego, CA: 1996. pp. 305–326. [Google Scholar]
- 18.Feng S, Kasahara C, Rickles RJ, Schreiber SL. Specific interactions outside the proline-rich core of two classes of Src homology 3 ligands. Proc Natl Acad Sci U S A. 1995;92:12408–15. doi: 10.1073/pnas.92.26.12408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Indyk L, Fisher HF. Theoretical aspects of isothermal titration calorimetry. Methods Enzymol. 1998;295:350–64. doi: 10.1016/s0076-6879(98)95048-0. [DOI] [PubMed] [Google Scholar]
- 20.Yakovlev S, Makogonenko E, Kurochkina N, Nieuwenhuizen W, Ingham K, Medved L. Conversion of fibrinogen to fibrin: mechanism of exposure of tPA- and plasminogen-binding sites. Biochemistry. 2000;39:15730–41. doi: 10.1021/bi001847a. [DOI] [PubMed] [Google Scholar]
- 21.Tymkewycz PM, Creighton-Kempsford LJ, Hockley D, Gaffney PJ. Screening for fibrin specific monoclonal antibodies: the development of a new procedure. Thromb Haemost. 1992;68:48–53. [PubMed] [Google Scholar]
- 22.Makogonenko E, Tsurupa G, Ingham K, Medved L. Interaction of fibrin(ogen) with fibronectin: further characterization and localization of the fibronectin-binding site. Biochemistry. 2002;41:7907–13. doi: 10.1021/bi025770x. [DOI] [PubMed] [Google Scholar]
- 23.Doolittle RF. Structural basis of the fibrinogen-fibrin transformation: contributions from X-ray crystallography. Blood Rev. 2003;17:33–41. doi: 10.1016/s0268-960x(02)00060-7. [DOI] [PubMed] [Google Scholar]
- 24.Hui KY, Haber E, Matsueda GR. Monoclonal antibodies to a synthetic fibrin-like peptide bind to human fibrin but not fibrinogen. Science. 1983;222:1129–32. doi: 10.1126/science.6648524. [DOI] [PubMed] [Google Scholar]
- 25.Hantgan R, McDonagh J, Hermans J. Fibrin assembly. Ann N Y Acad Sci. 1983;408:344–66. doi: 10.1111/j.1749-6632.1983.tb23256.x. [DOI] [PubMed] [Google Scholar]
- 26.Botnar RM, Perez AS, Witte S, Wiethoff AJ, Laredo J, Hamilton J, Quist W, Parsons EC, Jr, Vaidya A, Kolodziej A, Barrett JA, Graham PB, Weisskoff RM, Manning WJ, Johnstone MT. In vivo molecular imaging of acute and subacute thrombosis using a fibrin-binding magnetic resonance imaging contrast agent. Circulation. 2004;109:2023–9. doi: 10.1161/01.CIR.0000127034.50006.C0. Epub 2004 Apr 5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zhang Z, Kolodziej AF, Greenfield MT, Caravan P. Heteroditopic binding of magnetic resonance contrast agents for increased relaxivity. Angew Chem Int Ed Engl. 2011;50:2621–4. doi: 10.1002/anie.201007689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zhang Z, Kolodziej AF, Qi J, Nair SA, Wang X, Case AW, Greenfield MT, Graham PB, McMurry TJ, Caravan P. Effect of Peptide-Chelate Architecture on Metabolic Stability of Peptide-based MRI Contrast Agents. New J Chem. 2010;2010:611–616. doi: 10.1039/b9nj00787c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Sirol M, Aguinaldo JG, Graham PB, Weisskoff R, Lauffer R, Mizsei G, Chereshnev I, Fallon JT, Reis E, Fuster V, Toussaint JF, Fayad ZA. Fibrin-targeted contrast agent for improvement of in vivo acute thrombus detection with magnetic resonance imaging. Atherosclerosis. 2005;182:79–85. doi: 10.1016/j.atherosclerosis.2005.02.013. Epub 2005 Mar 25. [DOI] [PubMed] [Google Scholar]
- 30.Uppal R, Catana C, Ay I, Benner T, Sorensen AG, Caravan P. Bimodal Thrombus Imaging: Simultaneous PET/MR Imaging with a Fibrin-targeted Dual PET/MR Probe--Feasibility Study in Rat Model. Radiology. 2011;258:812–20. doi: 10.1148/radiol.10100881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Uppal R, Ay I, Dai G, Kim YR, Sorensen AG, Caravan P. Molecular MRI of intracranial thrombus in a rat ischemic stroke model. Stroke. 2010;41:1271–7. doi: 10.1161/STROKEAHA.109.575662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Vymazal J, Spuentrup E, Cardenas-Molina G, Wiethoff AJ, Hartmann MG, Caravan P, Parsons EC., Jr Thrombus imaging with fibrin-specific gadolinium-based MR contrast agent EP-2104R: results of a phase II clinical study of feasibility. Invest Radiol. 2009;44:697–704. doi: 10.1097/RLI.0b013e3181b092a7. [DOI] [PubMed] [Google Scholar]
- 33.Falati S, Gross P, Merrill-Skoloff G, Furie BC, Furie B. Real-time in vivo imaging of platelets, tissue factor and fibrin during arterial thrombus formation in the mouse. Nat Med. 2002;8:1175–81. doi: 10.1038/nm782. Epub 2002 Sep 16. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.