Abstract
Prenatal exposure to smoking and alcohol increases the risk for Sudden Infant Death Syndrome (SIDS). Physiological changes associated with these exposures are not well studied. Full-term infants were tested within the first 3 days of life. We hypothesized that maternal alcohol consumption and/or smoking during pregnancy would alter autonomic nervous system function. Newborns whose mothers smoked during pregnancy had lower beat-to-beat heart rate variability in quiet sleep. Infants whose mothers consumed alcohol had lower global heart rate variability, but only in active sleep. Unexposed infants demonstrated increases in heart rate with head-up tilt and decreases in heart rate with head-down tilt, but smoking and alcohol-exposed infants showed no significant responses. These results indicate that autonomic function is altered by prenatal exposure to alcohol and smoking. Such markers may provide early identification of infants at greatest risk for SIDS.
Keywords: SIDS, pregnancy, neonate, alcohol, smoking
INTRODUCTION
Sudden Infant Death Syndrome (SIDS) is defined as a sudden unexpected death during the first year of life, in which the cause remains unexplained even after careful examination including autopsy, death scene investigation, and review of prior symptoms (Willinger, James, & Catz, 1991). Although the rate of SIDS in several countries has shown a marked decline in recent years coincident with efforts to promote supine sleeping (de Jonge et al., 1993; Dwyer, Ponsonby, Blizzard, Newman, & Cochrane, 1995; Fleming & Blair, 1997; Mitchell, Brunt, & Everard, 1994), rates appear to have stabilized in most countries (Hauck & Tanabe, 2008) and this syndrome remains the most common cause of postneonatal deaths (Martin, Kochanek, Strobino, Guyer, & MacDorman, 2005). Moreover, in Native American infants in the Northern Plains, the subjects in this current study, the rate of SIDS has historically been among the highest in the nation (Tomashek et al., 2006).
Much of the evidence for specific prenatal factors that predispose infants to SIDS comes from extensive epidemiological studies and postmortem pathology. Maternal smoking stands out as a primary risk factor. There are at least 60 case–control investigations, and several more cohort studies from around the world, documenting a strong relationship between maternal smoking and SIDS (Chong, Yip, & Karlberg, 2004; Fleming & Blair, 1997; Mitchell, 1995; Mitchell & Milerad, 2006; Sullivan & Barlow, 2001). Children of smoking mothers have lower birth weights, are more likely to have been delivered prematurely, are smaller in size, display deficits in wakening to hypoxia, have higher BP at birth and at four months of age, and reduced arousability (Bardy et al., 1993; Beratis, Panagoulias, & Varvarigou, 1996; Bulterys, Greenland, & Kraus, 1990; Franco et al., 1999; Horne, Franco, Adamson, Groswasser, & Kahn, 2004; Land & Stockbauer, 1993; Malloy, Hoffman, & Peterson, 1992; O’Sullivan, Kearney, & Crowley, 1996; Wilcox, 1993). Although prenatal alcohol exposure is not consistently associated with increased risk for SIDS (Alm et al., 1999; Blair et al., 1996) evidence from a study in the Northern Plains suggests that alcohol consumption just before and/or during pregnancy increased risk for SIDS in Native Americans from this region (Iyasu et al., 2002). Smoking was not a significant risk factor in that study, but smoking was highly prevalent in both the cases and the controls.
Many studies have attempted to assess the ability of infants to respond to respiratory, thermal, and autonomic challenges (Fox & Matthews, 1989; Franco et al., 2003; Galland et al., 2000; Gootman, 1991; Lagercrantz, Edwards, Henderson-Smart, Hertzberg, & Jeffery, 1990; Thach, Davies, & Koenig, 1988). Particularly relevant for this current study, baroreceptor reflexes are functioning during the neonatal period in many species (Gootman, 1991). The heart rate (HR) response to changes in blood pressure following postural adjustment, a reflex mediated by the baroreceptors, has also been demonstrated in several studies with newborns in the first few days after birth. By carefully monitoring motor and respiratory activity following postural manipulation in neonates, reliable increases in HR and decreases in blood pressure have been found after head-up tilts (Andrasyova & Kellerova, 1996; Chen, Tsai, & Lan, 1995; Fifer, Greene, Hurtado, & Myers, 1999; Finley, Hamilton, & MacKenzie, 1984; Myers, Gomez-Gribben, Smith, Tseng, & Fifer, 2006; Thoresen, Cowan, & Walloe, 1991). This homeostatic response provides a measure of the competence of neural mechanisms to process and respond to a frequently encountered stimulus.
There are no reports on the possible effects of prenatal alcohol exposure on cardiorespiratory responses to tilting in infants, but two studies have investigated the effects of maternal smoking on these responses. In one study, HR and blood pressure responses to 70° head-up tilting were measured at 2–3 days and at 3 months of age (Browne, Colditz, & Dunster, 2000). No significant effects of maternal smoking were found for HR; however, in newborns, blood pressure fell with head-up tilting in infants of smoking mothers but remained unchanged in unexposed infants. Interestingly, at 3 months of age, blood pressure did not change with tilting in exposed infants but increased in unexposed infants. In the second study, infants were studied at 1 and 3 months of age and no differences between unexposed infants and infants of smoking mothers were found in HR responses to 60° head-up tilting (Galland et al., 2000). In this current study, we have investigated the potential influence of maternal smoking on newborn physiological responses to tilting within the first 2 days of life and have included infants whose mothers acknowledge consumption of alcohol during pregnancy. The study was conducted in Northern Plains Native Americans.
METHODS
Subjects
The subjects in this study were 271 healthy full-term infants born at the Indian Health Service Hospital in the Northern Plains. All high-risk births and newborns with any medical complications are transferred to an urban hospital and were not enrolled. Data from three newborns who subsequently died during infancy were excluded, though data from one case of confirmed SIDS within this cohort is reported elsewhere (Kinney et al., 2005). All procedures used were approved by the IRBs at the New York State Psychiatric Institute, the College of Physicians and Surgeons at Columbia University, and by the Aberdeen Area Indian Health Service.
Procedures
Babies were tested 10–60 min in the prone position after a normal feeding between 9 am and 3 pm. After attaching cardiorespiratory leads, they were lightly swaddled and placed on their stomachs in a specially designed bassinet that could be manually tilted 30° in either the head-up or head-down position. ECG leads were attached to a standard clinical monitor (Hewlett Packard, Palo Alto, CA 78202A). A bedside computer equipped with an analog-to-digital converter acquired all data. The amplified ECG signal was processed through a special-purpose RR-interval preprocessor that measures RR intervals with ±1 ms accuracy. In addition to the RR intervals, the respiration impedance signal from the monitor was simultaneously digitized at 50 Hz. For data collected during the tilting sequence, peaks in the respiratory tracing were marked with interactive software and the times between peaks were computed.
After the infant was judged to be asleep by behavioral criteria, data were collected during a baseline period of approximately 5 min. After this, the tilting protocol was initiated. Babies were first tilted to a 30° head-up angle over a period of 30 s. This slow rate of tilting was chosen to minimize arousal from sleep. After remaining in the head-up position for 1 min, they were tilted back to the flat position. Following 1 min in the flat position, infants were then tilted to a 30° head-down position where they remained for 1 min before being returned to the flat position for another minute. This entire sequence was then repeated up to four times.
Data Scoring
Prior to the tilting sequence, baseline data for HR and HR variability were analyzed in 1-min epochs. For the tilting portion of the protocol, respiratory rates were also computed. Differences between the 30 s periods immediately before tilting, and the second of two 30 s blocks after achieving the tilted position were analyzed. The impedance respiratory tracings were also examined visually for large excursions that were more than double the amplitude of normal breaths. These were noted as movements and the epochs were coded as to whether movements had occurred.
From the visual examination of the impedance tracing a sleep state code was assigned (Fifer et al., 1999). If the tracing was very rhythmic with minimal amplitude and rate variability, it was coded as quiet sleep (QS). If the epoch was characterized as having obvious rate and amplitude variation, it was coded as active sleep (AS). These codings of the respiratory tracings were augmented with behavioral observations taken at the time of data acquisition.
Physiological Variables Recorded
From the R-to-R intervals (RRi), mean HR, and the interquartile range of HR (IQR) were computed for each epoch. The ratio of the IQR over HR was also computed as a measure of rate-adjusted variability. Three measures of RRi variability were also computed: standard deviation of RRi (SD), root mean square of successive changes in RRi (RMSSD), and the percentage of successive changes in RRi that were sustained over two consecutive increases or two consecutive decreases (SUS). Using peak-to-peak intervals from the respiratory tracing, mean respiratory rate (RSP) was computed for each epoch of interest during the tilting sequence.
Ascertainment of Smoking and Alcohol Use
At the time of enrollment, mothers were asked by the research assistant if they had smoked at any time during pregnancy and if they had consumed alcohol at any time during pregnancy. In addition, about half of the women enrolled in the study were given a substance abuse questionnaire, which was administered by a member of the medical staff (Bull, Kvigne, Leonardson, Lacina, & Welty, 1999). Answers to this questionnaire are included in the mothers’ medical records. Acknowledgement of smoking or alcohol use from either source was taken as evidence for exposure.
RESULTS
Maternal and Infant Characteristics
Table 1 shows the characteristics of the 271 subjects included in these analyses. Subjects are divided into four groups with regard to whether mothers acknowledged consumption of alcohol at any time during pregnancy and/or acknowledged smoking at any time during pregnancy. Of the 115 women in this latter group, 3 acknowledged using a form of smokeless tobacco, but all others smoked cigarettes. These data were analyzed using two-way ANOVAs. Results from these analyses showed that there was a main effect of smoking on birth weight and hematocrit, and significant smoking by alcohol interactions for both of these variables. Birth weight was lower in infants of mothers who smoked, but this effect was increased in mothers who also drank during pregnancy. Hematocrit was lower in mothers who smoked but, in this case, the interaction appears to be due to a somewhat elevated level in the alcohol-alone group. A marginally significant effect of alcohol was seen for maternal age (alcohol consuming mothers tended to be younger).
Table 1.
Characteristics of Study Infants in Four Groups Based on Exposure to Maternal Smoking or Alcohol Consumption at Any Time During Pregnancy: No Smoking/No Alcohol (S−/A−), Smoking/No Alcohol (S+/A−), No Smoking/Alcohol (S−/A+), Smoking/Alcohol (S+/A+)
|
p-Values From ANOVA |
|||||||
|---|---|---|---|---|---|---|---|
| Variable | S−/A− | S+/A− | S−/A+ | S+/A+ | S | A | S × A |
| Birth weight (g) ± SE (n) | 3,659 ± 46 (112) | 3,552 ± 68 (50) | 3,756 ± 79 (37) | 3,273 ± 62 (61) | <.001 | NS | .004 |
| Baby test age (hr) ± SE (n) | 42.9 ± 1.5 (111) | 41.4 ± 2.3 (49) | 38.4 ± 2.7 (37) | 39.5 ± 2.1 (60) | NS | NS | NS |
| Maternal age (years) ± SE (n) | 24.9 ± .6 (111) | 24.8 ± .8 (52) | 24.1 ± 1.0 (37) | 22.7 ± .8 (60) | NS | .061 | NS |
| Gravida ± SE (n) | 3.3 ± .2 (111) | 3.3 ± .3 (50) | 2.8 ± .4 (38) | 2.9 ± .3 (59) | NS | NS | NS |
| Parity ± SE (n) | 1.9 ± .2 (111) | 2.1 ± .3 (50) | 1.5 ± .3 (38) | 1.6 ± .3 (59) | NS | NS | NS |
| Third trimester hematocrit (%) ± SE (n) | 35.2 ± .3 (90) | 34.6 ± .5 (39) | 37.3 ± .6 (29) | 34.3 ± .5 (50) | <.001 | .073 | .018 |
NS signifies p > .10.
In a subset of women administered a substance abuse questionnaire (Bull et al., 1999), estimates of number of cigarettes smoked and number of alcoholic drinks (1 drink = 1 beer or its equivalent) were obtained. These data were obtained for the period “just before this pregnancy” and “during this current pregnancy.” As can be seen in Table 2, women who smoked, but did not drink during pregnancy, reported a median of 3 cigarettes per day during pregnancy. These same women smoked a median of 6 cigarettes per day just before the pregnancy. Women who acknowledged drinking during pregnancy consumed a median of 3 drinks per month during pregnancy and a median of 12 drinks per month just before pregnancy. Women who acknowledge both smoking and drinking during pregnancy consumed a median of 6 drinks per month and 3 cigarettes per day during pregnancy. Just before pregnancy these same women consumed a median of 15 drinks per month and smoked 4 cigarettes per day.
Table 2.
Self-Reports of Amounts of Smoking and Drinking “Just Before” and “During” Pregnancy From Women Administered the Substance Abuse Questionnaire at One of Their Prenatal Visits (Average Months Pregnant = 3.4 Months)
| S−/A− (n = 41) | S+/A− (n = 24) | S−/A+ (n = 37) | S+/A+ (n = 67) | |
|---|---|---|---|---|
| Just before this pregnancy | ||||
| Cigarettes/day (median/range) | 0 (0–0) | 4 (1–60) | 0 (0–0) | 5 (1–40) |
| Drinking (times/month) (median/range) | 0 (0–0) | 0 (0–0) | 1 (1–20) | 2 (1–12) |
| Drinking (usual # drinks) (median/range) | 0 (0–0) | 0 (0–0) | 4 (1–13) | 6 (1–36) |
| Total drinks/month (median/range) | 0 (0–0) | 0 (0–0) | 6 (1–240) | 12 (1–144) |
| S−/A− (n = 88) | S+/A− (n = 42) | S−/A+ (n = 15) | S+ /A+ (n = 20) | |
| During this current pregnancy | ||||
| Cigarettes/day (median/range) | 0 (0–0) | 3 (0–60) | 0 (0–0) | 3.5 (1–20) |
| Drinking (times/month) (median/range) | 0 (0–0) | 0 (0–0) | 2 (1–20) | 1.5 (1–4) |
| Drinking (usual # drinks) (median/range) | 0 (0–0) | 0 (0–0) | 3 (1–13) | 3 (1–41) |
| Total drinks/month (median/range) | 0 (0–0) | 0 (0–0) | 6 (1–240) | 4 (1–41) |
The top section shows median and ranges for 169 mothers who reported that “just before this pregnancy” they did not smoke or consume alcohol (S−/A−), smoked but not consume (S+/A−), consumed alcohol but did not smoke (S−/A+), or both smoked and consumed alcohol (S+/A+). The bottom section shows median and ranges for 165 mothers’ reports “during this current pregnancy.”
Baseline Cardiorespiratory Data
Baseline data obtained in AS and QS are shown in Table 3. These data were analyzed using two-way ANOVAs. In QS, the only significant effect seen was for smoking on RMSSD. Overall, infants of mothers who smoked during pregnancy had lower levels of this measure of beat-to-beat heart period variability. In AS, there were no significant effects of smoking or interactions between smoking and alcohol for any of the baseline variables. In contrast, HR was higher and several indices of HR variability were lower in alcohol-exposed infants. In particular, measures of overall variability (i.e., SD and IQR), rather than beat-to-beat variability, were more sensitive indicators of effects of alcohol exposure.
Table 3.
Baseline Heart Rate and Heart Period Variability Measures in Active and Quiet Sleep of 198 Infants in Four Groups Based on Exposure to Maternal Smoking or Alcohol Consumption at Any Time During Pregnancy: No Smoking/No Alcohol (S−/A−), Smoking/No Alcohol (S+/A−), No Smoking/Alcohol (S−/A+), Smoking/Alcohol (S+/A+)
| p-Values From ANOVA | |||||||
|---|---|---|---|---|---|---|---|
| Variable | S−/A− | S+/A− | S−/A+ | S+/A+ | S | A | S × A |
| Quiet sleep | |||||||
| n (total = 141) | 64 | 24 | 22 | 31 | |||
| # minutes | 5.0 ± .4 | 4.2 ± .6 | 5.9 ± .7 | 5.1 ± .6 | |||
| HR (bpm) | 124 ± 1 | 125 ± 4 | 122 ± 2 | 125 ± 2 | NS | NS | NS |
| IQR (bpm) | 7.7 ± .5 | 7.9 ± .8 | 8.5 ± .8 | 8.6 ± .7 | NS | NS | NS |
| IQR/HR (%) | 6.3 ± .4 | 6.4 ± .7 | 7.0 ± .7 | 6.8 ± .6 | NS | NS | NS |
| SD (ms) | 22.4 ± 1.3 | 21.9 ± 2.0 | 23.6 ± 2.1 | 23.1 ± 1.8 | NS | NS | NS |
| RMSSD (ms) | 10.3 ± .4 | 9.6 ± .7 | 11.7 ± .7 | 9.0 ± .6 | .009 | NS | NS |
| SUS (%) | 35.3 ± 1.0 | 33.9 ± 1.6 | 34.5 ± 1.7 | 34.4 ± 1.4 | NS | NS | NS |
| Active sleep | |||||||
| n (total = 141) | 61 | 33 | 16 | 31 | |||
| # minutes | 3.2 ± .4 | 3.2 ± .5 | 3.0 ± .7 | 3.7 ± .5 | |||
| HR (bpm) | 128 ± 1 | 125 ± 2 | 133 ± 3 | 129 ± 2 | NS | .057 | NS |
| IQR (bpm) | 11.1 ± .5 | 10.0 ± .7 | 8.9 ± 1 | 8.5 ± .8 | NS | .022 | NS |
| IQR/HR (%) | 8.8 ± .5 | 8.1 ± .6 | 6.9 ± .9 | 6.7 ± .6 | NS | .014 | NS |
| SD (ms) | 28.4 ± 1.4 | 27.1 ± 1.9 | 24.5 ± 2.7 | 22.7 ± 2.0 | NS | .043 | NS |
| RMSSD (ms) | 9.5 ± .4 | 9.8 ± .5 | 8.9 ± .8 | 8.7 ± .6 | NS | NS | NS |
| SUS (%) | 38.1 ± 1.1 | 38.1 ± 1.5 | 33.4 ± 2.0 | 35.9 ± 1.5 | NS | .030 | NS |
p-Values for main effects of smoking and drinking, and their interactions are from two-way ANOVAs: “NS” signifies p > .10. See Methods Section for further explanation and variable definitions.
Although pseudo-rhythmic sympathetic and parasympathetic activity can influence measures of global HR variability, these indices are also affected by nonstationary events such as body movements. Thus, lower levels of HR variability in alcohol-exposed infants could be due to fewer or smaller body movements or smaller HR changes associated with movement. To explore this hypothesis, we reanalyzed the data after dividing epochs into those in which significant body movements occurred versus those epochs without obvious movement. Table 4 shows the results from these analyses. In epochs with no large body movements, there were no significant effects of alcohol exposure or maternal smoking on any of the HR and variability measures. In contrast, analysis of epochs containing movements revealed significant or nearly significant effects of alcohol exposure on all variables; HR was higher and all measures of HR variability were lower in alcohol-exposed infants. In these epochs, infants whose mothers smoked during pregnancy had significantly lower baseline HRs. A similar set of analyses was performed on data collected in QS. There were no significant differences in any of the indices of HR variability in epochs either with or without movement.
Table 4.
Baseline Heart Rate and Heart Period Variability Measures in Active Sleep in Epochs In Which No Body Movements Were Noted (Top) or In Which One or More Body Movements Were Noted (Bottom)
| p-Values From ANOVA | |||||||
|---|---|---|---|---|---|---|---|
| Variable | S−/A− | S+/A− | S−/A+ | S+/A+ | S | A | S × A |
| Epochs with no movement | |||||||
| n (total = 94) | 38 | 24 | 10 | 22 | |||
| HR (bpm) | 126 ± 2 | 124 ± 2 | 127 ± 3 | 127 ± 2 | NS | NS | NS |
| IQR (bpm) | 10.0 ± .8 | 9.4 ± 1.1 | 8.2 ± 1.6 | 8.5 ± 1.1 | NS | NS | NS |
| IQR/HR (%) | 8.0 ± .6 | 7.7 ± .8 | 6.6 ± 1.2 | 6.8 ± .8 | NS | NS | NS |
| SD (ms) | 27.0 ± 1.7 | 26.8 ± 2.1 | 23.5 ± 3.2 | 22.7 ± 2.2 | NS | NS | NS |
| RMSSD (ms) | 10.0 ± .5 | 10.3 ± .6 | 9.7 ± 1.0 | 8.7 ± .7 | NS | NS | NS |
| SUS (%) | 36.5 ± 1.3 | 36.9 ± 1.7 | 34.8 ± 2.6 | 35.5 ± 1.7 | NS | NS | NS |
| Epochs with movement | |||||||
| n (total = 114) | 51 | 26 | 12 | 25 | |||
| HR (bpm) | 130 ± 2 | 127 ± 2 | 138 ± 3 | 129 ± 2 | .035 | .050 | NS |
| IQR (bpm) | 12.2 ± .6 | 10.6 ± .9 | 9.6 ± 1.4 | 9.7 ± .9 | NS | .081 | NS |
| IQR/HR (%) | 9.6 ± .5 | 8.3 ± .7 | 7.2 ± 1.1 | 7.5 ± .8 | NS | .053 | NS |
| SD (ms) | 29.9 ± 1.6 | 27.0 ± 2.3 | 23.8 ± 3.3 | 25.0 ± 2.3 | NS | .096 | NS |
| RMSSD (ms) | 9.4 ± .4 | 9.2 ± .6 | 8.1 ± .9 | 8.5 ± .6 | NS | NS | NS |
| SUS (%) | 39.4 ± 1.3 | 38.3 ± 1.9 | 32.5 ± 2.7 | 37.8 ± 1.9 | NS | .073 | NS |
Infants are divided into four groups formed based on exposure to maternal smoking or alcohol consumption at any time during pregnancy: no smoking/ no alcohol (S−/A−), smoking/no alcohol (S+/A−), no smoking/alcohol (S−/A+), smoking/alcohol (S+/A+). Degrees of freedom for effects in two-way ANOVAs for no movement are 1and 97 and for movement 1 and 115 (“NS” signifies p > .10).
Responses to Head-Up and Head-Down Tilting
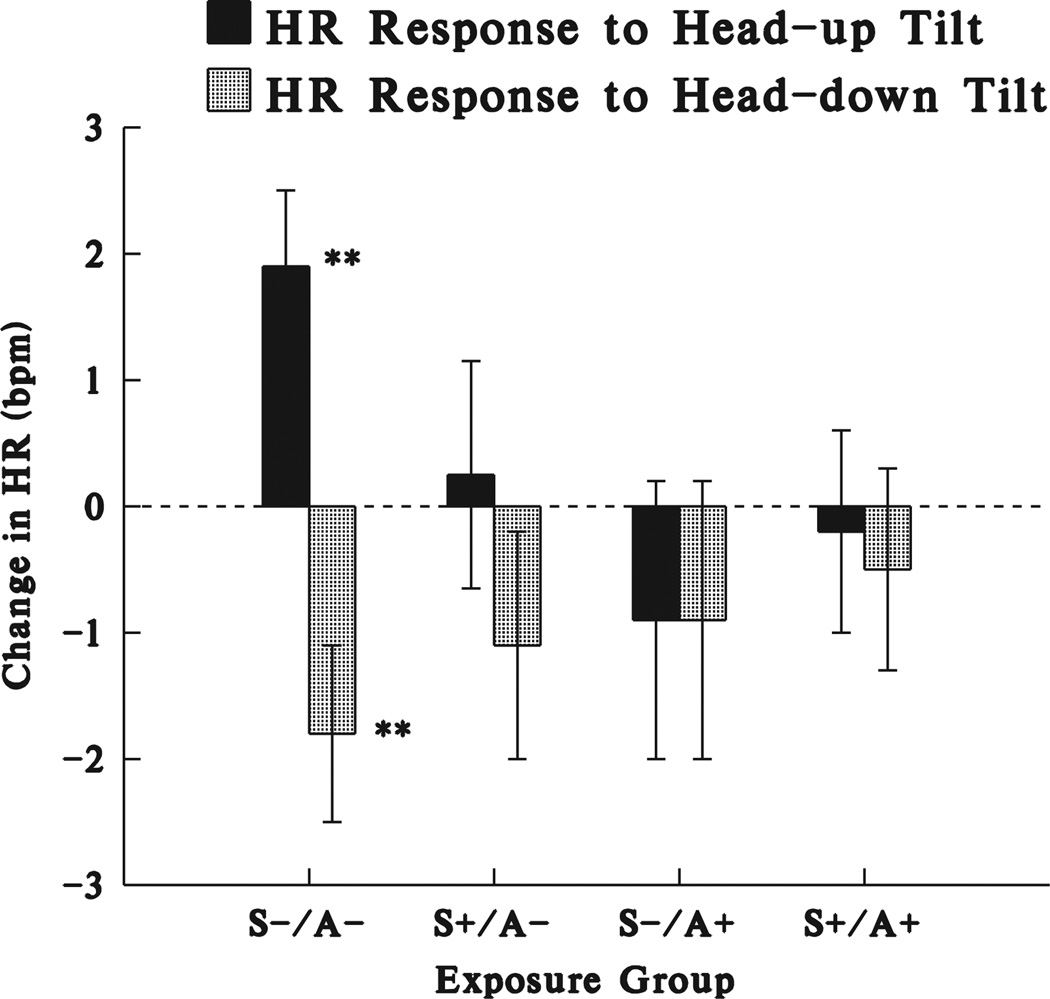
The average HR responses to head-up and head-down tilting for the 170 infants completing the tilt protocol and for which there was artifact-free ECG data are shown in Figure 1. Since in a prior study we did not find a significant effect of sleep state on tilt responses (Myers et al., 2006) data were averaged across sleep states. Consistent with our prior work (Fifer et al., 1999; Myers et al., 2006), infants of mothers who did not smoke or consume alcohol during pregnancy show small, but highly significant (p < .01) increases in HR following head-up tilting and decreases in HR following head-down tilting. In contrast, changes in HR were not significant for either direction of tilting in any of the three exposure groups.
FIGURE 1.
This figure shows changes in heart rate (mean ± E) from the 30 s baseline period just before tilting to the average of the two 30 s blocks in the tilted position. These infants completed at least one head-up and one head-down tilt. The four groups were infants whose mothers denied smoking and consumption of alcohol at any time during pregnancy (S−/A−, n = 49), acknowledged smoking but denied consumption of alcohol during pregnancy (S+/A−, n = 24), denied smoking but acknowledged consumption of alcohol during pregnancy (S−/A+, n = 17), or acknowledged both smoking and alcohol consumption during pregnancy (S+/A+, n = 32). ** indicates the changes were significant at the .01 level.
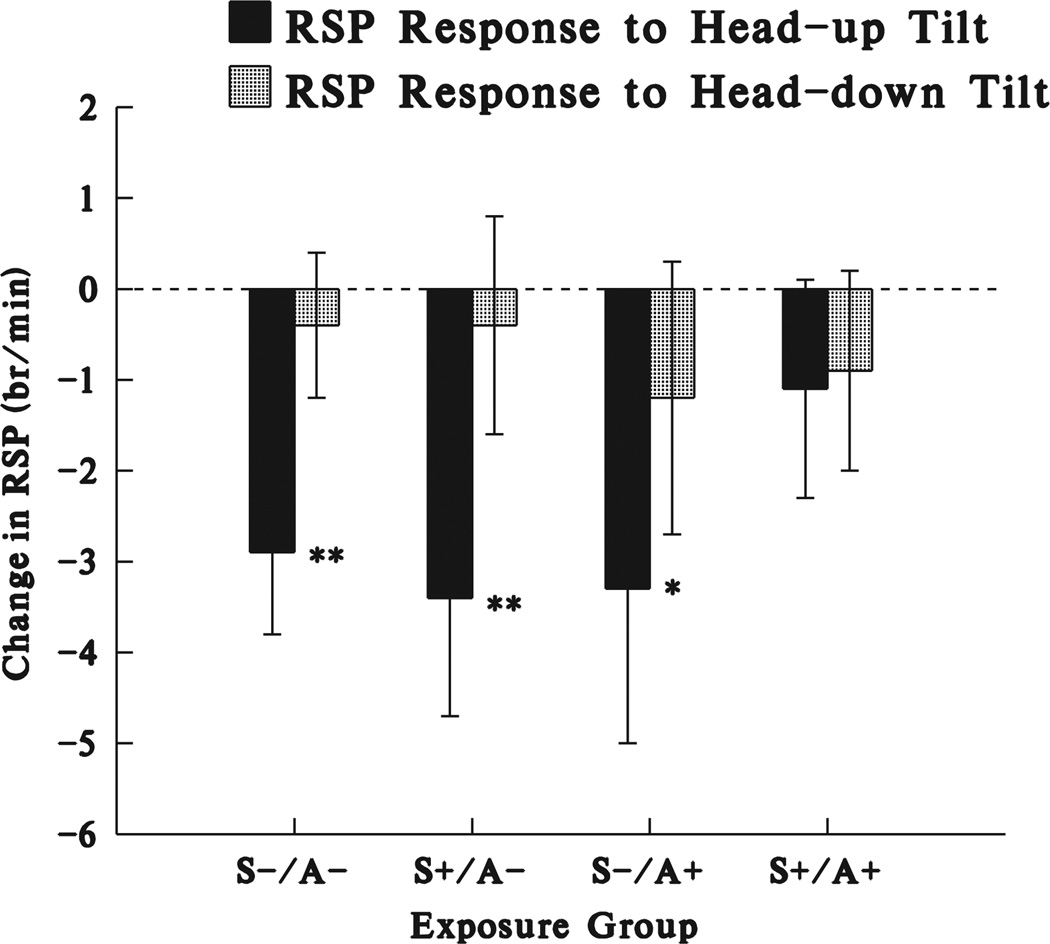
Changes in respiratory rate following head-up and head-down tilting for the 168 infants completing the tilt protocol and for which there was artifact-free respiratory wave-form data are shown in Figure 2. As found in a prior study (Fifer et al., 1999), there were no significant changes in respiratory rate following head-down tilts in any of the exposure groups. As expected from our prior study, respiratory rate exhibited robust decreases with head-up tilting in the unexposed infants (p < .001). Significant decreases were also seen in the smoking-alone group (p < .001), and somewhat smaller decreases in the alcohol-alone group (p < .05). No significant changes were seen in the dual-exposed group.
FIGURE 2.
This figure shows changes in breathing rate (mean ± SE) from the 30 s baseline period just before tilting to the average of the two 30 s blocks in the tilted position. These infants completed at least one head-up and one head-down tilt. The four groups were infants whose mothers denied smoking and consumption of alcohol at any time during pregnancy (S−/A−, n = 57), acknowledged smoking but denied consumption of alcohol during pregnancy (S+/A−, n = 28), denied smoking but acknowledged consumption of alcohol during pregnancy (S−/A+, n = 17), or acknowledged both smoking and alcohol consumption during pregnancy (S+/A+, n = 35). * and *** indicate the changes were significant at the .05 and .001 levels, respectively.
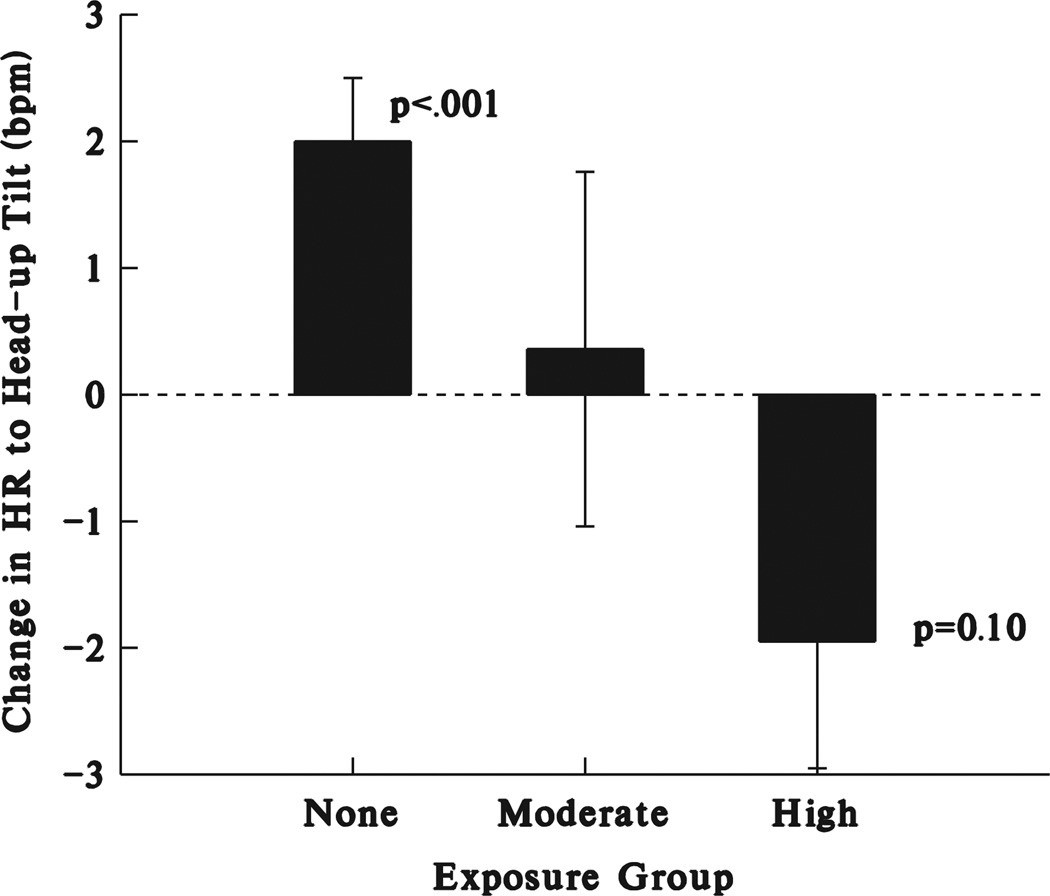
To determine if differences in head-up tilt responses could be related to degree of exposure to alcohol, we used the substance abuse questionnaire data to form three groups: no exposure to alcohol during pregnancy (n = 81), moderate exposure (less than 4 drinks/month during pregnancy, n = 11), or high exposure (4 or more drinks/month during pregnancy, n = 16). Systematic effects of exposure were seen only for HR responses to head-up tilting (see Fig. 3). There was a highly significant difference in response among the alcohol amount groups (F(2,105) = 6.62, p = .002), with a clear indication of a dose response. In the no-exposure group HR increased following tilting (p < .001), with moderate exposure there was virtually no change in HR, and in the high exposure group, HR showed a marginally significant decrease.
FIGURE 3.
This figure shows changes in heart rate (mean ± SE) from the 30 s baseline period just before tilting to the average of the two 30 s blocks in the head-up tilted position. Based on the Substance Abuse Questionnaires, three groups were formed: infants whose mothers denied any consumption of alcohol during pregnancy (None, n = 74), Moderate exposure (greater than 0 but less than 4 drinks/month during pregnancy, n = 11), or High exposure (4 or more drinks/month during pregnancy, n = 14).
DISCUSSION
One strategy employed in SIDS research is to assess cardiorespiratory function in infants known to be at increased risk. The siblings of SIDS victims, low birth weight infants, and infants recovered from apparent life-threatening events (ALTE), all are populations studied in attempts to identify mechanisms involved in SIDS. Results from this current study indicate that both alcohol and smoking exposure in utero are associated with altered autonomic control soon after birth in a population of infants at very high risk for SIDS, namely Native American infants of the Northern Plains. On the reservations the majority of the inhabitants live in rural, nonagricultural dwellings. Poverty is very common, with a large percentage of the population unemployed. One obvious explanation for the extremely high SIDS rate on the Northern Plains Reservations is the high incidence of risk factors associated with impoverished prenatal conditions as well as an increased probability of postnatal stressors. These include smoking during pregnancy, maternal anemia, young maternal age, low education attainment, high parity, and crowded living and sleeping conditions (Bulterys, 1990; Oyen, Bulterys, Welty, & Kraus, 1990; Petersen et al., 1984). Maternal alcohol consumption during pregnancy is prevalent and has emerged as a SIDS risk on the Northern Plains Reservations (Oyen et al., 1990). Furthermore, due to a high prevalence of postnatal risk factors, including exposure to passive smoke, opportunities for overheating, and impoverished environments, the window of vulnerability may in fact be wider than in other populations.
We report that prenatal smoking and/or alcohol exposure is associated with specific alterations in autonomic control during sleep that can be observed in the first few days of postnatal life. Smoking was associated with diminished beat-to-beat variability in QS. This effect on parasympathetic influence on HR was not observed in AS and not associated with alcohol exposure. In contrast, during periods of movement in AS, HR was higher and HR variability was markedly decreased in those neonates exposed to alcohol during pregnancy. For smoking-exposed infants, HR variability was not altered during epochs of movement, but average HR was lower. Although there were significant effects of smoking and smoking plus alcohol on birth weight and mothers’ hematocrit, in analyses not shown, these factors were themselves not significantly associated with the autonomic variables analyzed in this study. The typical HR increase to head-up tilt was diminished in all exposure groups, though the expected respiratory change was altered only in the infants with both exposures. The exposure effects most clearly emerged when amount of alcohol exposure was analyzed. Even moderate exposure resulted in virtually no change in HR following head-up tilt, and the highest exposed had a response in the opposite direction from nonexposed infants. This altered homeostatic response to a modest challenge of 30° during sleep suggests that exposed infants may not have the capacity to respond to cardiovascular challenges during sleep which ordinarily should not be life-threatening. This diminished response may become particularly problematic during the peak period of vulnerability for SIDS, often characterized as a developmental period of homeostatic instability (Harper, Kinney, Fleming, & Thach, 2000a).
These results suggest that the lower levels of HR variability found in infants exposed prenatally to alcohol may not be due to an underlying tonic difference in autonomic control of the heart. Rather this effect of alcohol may be related to differences in the amount of movement or the coupling between HR and movement. More detailed measures of body movements and timing and magnitude of coupled changes in HR are required to understand this relationship. Measurement of beat-to-beat changes in blood pressure would significantly add to our understanding of the deficits in cardiovascular responses to tilt, but, to date, the technology is unsuitable for long recordings with neonates. The possible effects of smoking and alcohol exposures on the vestibular component of a tilt response also warrant further examination, as deficits in vestibular mediated cardiovascular control may play a role in the pathophysiology of SIDS (Harper, Woo, & Alger, 2000b).
A limitation of this current study is the reliance on maternal report for smoking and alcohol exposure and a lack of information about the timing and duration of exposure. However, data obtained from the substance abuse questionnaire, validated for this population (Bull et al., 1999), show three important points. As expected, birth weight was lower in infants of mothers who smoked, and this effect was exacerbated by maternal drinking during pregnancy. Secondly, although the percentage of women who smoke during pregnancy is quite high, the actual number of cigarettes smoked is low. Finally, the number of drinks consumed each month dropped considerably during pregnancy. This is encouraging and shows the positive impact of the many efforts to improve infant mortality in this region. Our program of research is attempting to develop more precise and reliable assessments of prenatal environmental exposures which, when paired with physiologic characteristics of newborn infants, will allow identification of infants at increased risk prior to the vulnerable period for SIDS.
Acknowledgments
Contract grant sponsor: NICHD
Contract grant number: R37 HD032774
REFERENCES
- Alm B, Wennergren G, Norvenius G, Skjaerven R, Oyen N, Helweg-Larsen K, et al. Caffeine and alcohol as risk factors for sudden infant death syndrome. Nordic Epidemiological SIDS Study. Arch Dis Child. 1999;81(2):107–111. doi: 10.1136/adc.81.2.107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andrasyova D, Kellerova E. Blood pressure and heart rate response to head-up position in full-term newborns. Early Hum Dev. 1996;44(3):169–178. doi: 10.1016/0378-3782(95)01706-2. [DOI] [PubMed] [Google Scholar]
- Bardy AH, Seppala T, Lillsunde P, Kataja JM, Koskela P, Pikkarainen J, et al. Objectively measured tobacco exposure during pregnancy: Neonatal effects and relation to maternal smoking. Br J Obstet Gynaecol. 1993;100(8):721–726. doi: 10.1111/j.1471-0528.1993.tb14262.x. [DOI] [PubMed] [Google Scholar]
- Beratis NG, Panagoulias D, Varvarigou A. Increased blood pressure in neonates and infants whose mothers smoked during pregnancy. J Pediatr. 1996;128(6):806–812. doi: 10.1016/s0022-3476(96)70333-5. [DOI] [PubMed] [Google Scholar]
- Blair PS, Fleming PJ, Bensley D, Smith I, Bacon C, Taylor E, et al. Smoking and the sudden infant death syndrome: Results from 1993–5 case-control study for confidential inquiry into stillbirths and deaths in infancy. Confidential Enquiry into Stillbirths and Deaths Regional Coordinators and Researchers. Br Med J. 1996;313(7051):195–198. doi: 10.1136/bmj.313.7051.195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Browne CA, Colditz PB, Dunster KR. Infant autonomic function is altered by maternal smoking during pregnancy. Early Hum Dev. 2000;59(3):209–218. doi: 10.1016/s0378-3782(00)00098-0. [DOI] [PubMed] [Google Scholar]
- Bull LB, Kvigne VL, Leonardson GR, Lacina L, Welty TK. Validation of a self-administered questionnaire to screen for prenatal alcohol use in Northern Plains Indian women. Am J Prev Med. 1999;16(3):240–243. [PubMed] [Google Scholar]
- Bulterys M. High incidence of sudden infant death syndrome among northern Indians and Alaska natives compared with southwestern Indians: Possible role of smoking. J Community Health. 1990;15(3):185–194. doi: 10.1007/BF01350256. [DOI] [PubMed] [Google Scholar]
- Bulterys MG, Greenland S, Kraus JF. Chronic fetal hypoxia and sudden infant death syndrome: Interaction between maternal smoking and low hematocrit during pregnancy. Pediatrics. 1990;86(4):535–540. [PubMed] [Google Scholar]
- Chen CM, Tsai TC, Lan MC. Effect of body tilting on physiological functions in healthy term neonates. Acta Paediatr. 1995;84(5):474–477. doi: 10.1111/j.1651-2227.1995.tb13677.x. [DOI] [PubMed] [Google Scholar]
- Chong DS, Yip PS, Karlberg J. Maternal smoking: An increasing unique risk factor for sudden infant death syndrome in Sweden. Acta Paediatr. 2004;93(4):471–478. doi: 10.1080/08035250310023495. [DOI] [PubMed] [Google Scholar]
- de Jonge GA, Burgmeijer RJ, Engelberts AC, Hoogenboezem J, Kostense PJ, Sprij AJ. Sleeping position for infants and cot death in The Netherlands 1985–91. Arch Dis Child. 1993;69(6):660–663. doi: 10.1136/adc.69.6.660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dwyer T, Ponsonby AL, Blizzard L, Newman NM, Cochrane JA. The contribution of changes in the prevalence of prone sleeping position to the decline in sudden infant death syndrome in Tasmania. J Am Med Assoc. 1995;273(10):783–789. [PubMed] [Google Scholar]
- Fifer WP, Greene M, Hurtado A, Myers MM. Cardiorespiratory responses to bidirectional tilts in infants. Early Hum Dev. 1999;55(3):265–279. doi: 10.1016/s0378-3782(99)00026-2. [DOI] [PubMed] [Google Scholar]
- Finley JP, Hamilton R, MacKenzie MG. Heart rate response to tilting in newborns in quiet and active sleep. Biol Neonate. 1984;45(1):1–10. doi: 10.1159/000241756. [DOI] [PubMed] [Google Scholar]
- Fleming PJ, Blair PS. The role of sleeping position in the aetiology of the sudden infant death syndrome. London: Saunders; 1997. [Google Scholar]
- Fox GP, Matthews TG. Autonomic dysfunction at different ambient temperatures in infants at risk of sudden infant death syndrome. Lancet. 1989;2(8671):1065–1067. doi: 10.1016/s0140-6736(89)91080-5. [DOI] [PubMed] [Google Scholar]
- Franco P, Groswasser J, Hassid S, Lanquart JP, Scaillet S, Kahn A. Prenatal exposure to cigarette smoking is associated with a decrease in arousal in infants. J Pediatr. 1999;135(1):34–38. doi: 10.1016/s0022-3476(99)70324-0. [DOI] [PubMed] [Google Scholar]
- Franco P, Lipshut W, Valente F, Adams S, Groswasser J, Kahn A. Cardiac autonomic characteristics in infants sleeping with their head covered by bedclothes. J Sleep Res. 2003;12(2):125–132. doi: 10.1046/j.1365-2869.2003.00340.x. [DOI] [PubMed] [Google Scholar]
- Galland BC, Hayman RM, Taylor BJ, Bolton DP, Sayers RM, Williams SM. Factors affecting heart rate variability and heart rate responses to tilting in infants aged 1 and 3 months. Pediatr Res. 2000;48(3):360–368. doi: 10.1203/00006450-200009000-00017. [DOI] [PubMed] [Google Scholar]
- Gootman PM. Developmental aspects of reflex control of the circulation. In: Zucker IH, Gilmore JP, editors. Reflex control of the circulation. Boca Raton: CRC Press, Inc.; 1991. pp. 965–1027. [Google Scholar]
- Harper RM, Kinney HC, Fleming PJ, Thach BT. Sleep influences on homeostatic functions: Implications for sudden infant death syndrome. Respir Physiol. 2000a;119(2–3):123–132. doi: 10.1016/s0034-5687(99)00107-3. [DOI] [PubMed] [Google Scholar]
- Harper RM, Woo MA, Alger JR. Visualization of sleep influences on cerebellar and brainstem cardiac and respiratory control mechanisms. Brain Res Bull. 2000b;53(1):125–131. doi: 10.1016/s0361-9230(00)00317-8. [DOI] [PubMed] [Google Scholar]
- Hauck FR, Tanabe KO. International trends in sudden infant death syndrome: Stabilization of rates requires further action. Pediatrics. 2008;122(3):660–666. doi: 10.1542/peds.2007-0135. [DOI] [PubMed] [Google Scholar]
- Horne RS, Franco P, Adamson TM, Groswasser J, Kahn A. Influences of maternal cigarette smoking on infant arousability. Early Hum Dev. 2004;79(1):49–58. doi: 10.1016/j.earlhumdev.2004.04.005. [DOI] [PubMed] [Google Scholar]
- Iyasu S, Randall LL, Welty TK, Hsia J, Kinney HC, Mandell F, et al. Risk factors for sudden infant death syndrome among northern plains Indians. J Am Med Assoc. 2002;288(21):2717–2723. doi: 10.1001/jama.288.21.2717. [DOI] [PubMed] [Google Scholar]
- Kinney HC, Myers MM, Belliveau RA, Randall LL, Trachtenberg FL, Fingers ST, et al. Subtle autonomic and respiratory dysfunction in sudden infant death syndrome associated with serotonergic brainstem abnormalities: A case report. J Neuropathol Exp Neurol. 2005;64(8):689–694. doi: 10.1097/01.jnen.0000174334.27708.43. [DOI] [PubMed] [Google Scholar]
- Lagercrantz H, Edwards D, Henderson-Smart D, Hertzberg T, Jeffery H. Autonomic reflexes in preterm infants. Acta Paediatr Scand. 1990;79(8–9):721–728. doi: 10.1111/j.1651-2227.1990.tb11546.x. [DOI] [PubMed] [Google Scholar]
- Land GH, Stockbauer JW. Smoking and pregnancy outcome: Trends among black teenage mothers in Missouri. Am J Public Health. 1993;83(8):1121–1124. doi: 10.2105/ajph.83.8.1121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malloy MH, Hoffman HJ, Peterson DR. Sudden infant death syndrome and maternal smoking. Am J Public Health. 1992;82(10):1380–1382. doi: 10.2105/ajph.82.10.1380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin JA, Kochanek KD, Strobino DM, Guyer B, MacDorman MF. Annual summary of vital statistics—2003. Pediatrics. 2005;115(3):619–634. doi: 10.1542/peds.2004-2695. [DOI] [PubMed] [Google Scholar]
- Mitchell EA. Smoking: The next major and modifiable risk factor. In: Rognum TO, editor. Sudden infant death syndrome: New trends in the nineties. Oslo: Scandinavian University Press; 1995. pp. 114–118. [Google Scholar]
- Mitchell EA, Milerad J. Smoking and the sudden infant death syndrome. Rev Environ Health. 2006;21(2):81–103. doi: 10.1515/reveh.2006.21.2.81. [DOI] [PubMed] [Google Scholar]
- Mitchell EA, Brunt JM, Everard C. Reduction in mortality from sudden infant death syndrome in New Zealand: 1986–92. Arch Dis Child. 1994;70(4):291–294. doi: 10.1136/adc.70.4.291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Myers MM, Gomez-Gribben E, Smith KS, Tseng A, Fifer WP. Developmental changes in infant heart rate responses to head-up tilting. Acta Paediatr. 2006;95(1):77–81. doi: 10.1080/08035250500325074. [DOI] [PubMed] [Google Scholar]
- O’Sullivan MJ, Kearney PJ, Crowley MJ. The influence of some perinatal variables on neonatal blood pressure. Acta Paediatr. 1996;85(7):849–853. doi: 10.1111/j.1651-2227.1996.tb14166.x. [DOI] [PubMed] [Google Scholar]
- Oyen N, Bulterys M, Welty TK, Kraus JF. Sudden unexplained infant deaths among American Indians and whites in North and South Dakota. Paediatr Perinat Epidemiol. 1990;4(2):175–183. doi: 10.1111/j.1365-3016.1990.tb00636.x. [DOI] [PubMed] [Google Scholar]
- Petersen LP, Leonardson G, Wingert RI, Stanage W, Gergen J, Gilmore HT. Pregnancy complications in Sioux Indians. Obstet Gynecol. 1984;64(4):519–523. [PubMed] [Google Scholar]
- Sullivan FM, Barlow SM. Review of risk factors for sudden infant death syndrome. Paediatr Perinat Epidemiol. 2001;15(2):144–200. doi: 10.1046/j.1365-3016.2001.00330.x. [DOI] [PubMed] [Google Scholar]
- Thach BT, Davies AM, Koenig JS. Pathophysiology of sudden upper airway obstruction in sleeping infants and its relevance for SIDS. Ann N Y Acad Sci. 1988;533:314–328. doi: 10.1111/j.1749-6632.1988.tb37261.x. [DOI] [PubMed] [Google Scholar]
- Thoresen M, Cowan F, Walloe L. Cardiovascular responses to tilting in healthy newborn babies. Early Hum Dev. 1991;26(3):213–222. doi: 10.1016/0378-3782(91)90161-u. [DOI] [PubMed] [Google Scholar]
- Tomashek KM, Qin C, Hsia J, Iyasu S, Barfield WD, Flowers LM. Infant mortality trends and differences between American Indian/Alaska Native infants and white infants in the United States, 1989–1991 and 1998–2000. Am J Public Health. 2006;96(12):2222–2227. doi: 10.2105/AJPH.2004.053744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilcox AJ. Birth weight and perinatal mortality: The effect of maternal smoking. Am J Epidemiol. 1993;137(10):1098–1104. doi: 10.1093/oxfordjournals.aje.a116613. [DOI] [PubMed] [Google Scholar]
- Willinger M, James LS, Catz C. Defining the sudden infant death syndrome (SIDS): Deliberations of an expert panel convened by the National Institute of Child Health and Human Development. Pediatr Pathol. 1991;11(5):677–684. doi: 10.3109/15513819109065465. [DOI] [PubMed] [Google Scholar]