Abstract
Background
The rate of infection control for one-stage revision of infected knee arthroplasties is unclear as are the factors influencing infection control. Such factors include duration of infection and the type of infected prosthesis.
Questions/purposes
We therefore determined: (1) the rate of infection control with one-stage revision of septic knee prostheses, (2) the clinical knee scores that can be achieved, (3) whether the duration of infection or the type of prosthesis influence the level of infection control, and (4) whether different types of prostheses influence the knee scores.
Methods
We retrospectively reviewed prospectively collected data from 63 patients who underwent one-stage revisions of septic knee endoprostheses (six unicondylar, 37 primary total knee replacement prostheses, and 20 hinged knee endoprostheses) between 2004 and 2006. All were treated locally and systemically with microorganism-specific antibiotics. For this study we excluded patients with Methicillin-resistant Staphylococcus aureus and Methicillin-resistant Staphylococcus epidermidis or unknown microorganisms. The patients were examined for infection every 3 months and Oxford and Knee Society scores were assessed at the same time. The minimum followup was 24 months (mean followup, 36 months; range, 24–70 months).
Results
None of the patients with replacement unicondylar and primary total knee replacement prostheses had recurrence of infection. Three of the 20 patients with the hinged infected knee prostheses had recurrences; these three patients had chronic infections and had undergone two to three revision operations during at least a 5-year period. The likelihood of infection control was influenced by the duration of infection. The mean Knee Society knee score 24 months after surgery was 72 points (range, 20–98 points), the Knee Society function score was 71 points (range, 10–100 points), and the Oxford-12 knee score was 27 points (range, 13–44 points).
Conclusions
One-stage revision of septic knee prostheses achieved an infection control rate of 95% and higher knee scores than reported for two-stage revisions. Higher rates of recurrent infection appeared to be associated with long-term chronic infections of hinged prostheses.
Keywords: Medicine & Public Health; Conservative Orthopedics; Orthopedics; Sports Medicine; Surgery; Surgical Orthopedics; Medicine/Public Health, general
Introduction
Although relatively infrequent, a periprosthetic infection is a serious complication of primary knee arthroplasties, with an incidence of approximately 1% [34, 36, 45, 47]. In some studies, these infections have been cited as the most frequent cause of failure during the first 5 years after prosthesis implantation [15, 48, 50, 56]. Late infection occurring after 4 weeks of implantation usually requires complete revision [12, 32].
The two-stage revision of septic implants is the most common procedure for treating infected prostheses [16, 20, 25, 28, 32, 42] and involves temporary placement of an antibiotic-loaded cement spacer. In the case of infected total knee endoprostheses, this procedure can lead to a rate of infection control between 91% and 96% [28, 32, 42]. Some authors prefer a mobile spacer during the interim phase because the knee can remain mobilized, reimplantation of the prosthesis is easier than with static spacers, and the loss of bone compared with static spacers during the spacer period is less [7, 16, 20, 25, 28, 32, 42].
One-stage revisions of septic prostheses have been reported only from a few centers [6, 23, 40, 52, 56]. The arguments for this procedure focus on the fact that only one operation is necessary, with presumably less morbidity, lower cost, and faster restoration of functionality. von Foerster et al. [57] first reported on this procedure, with infection control of 73% in 104 cases. Other retrospective studies with eight to 21 patients had higher rates of infection control between 87% and 93% [6, 23, 40, 52] (Table 1). Patients with infections caused by methicillin-resistant Staphylococcus aureus (MRSA) or methicillin-resistant Staphylococcus epidermidis (MRSE) were not included in these studies; infections with these pathogens generally have lower rates of infection control than periprosthetic infections with other pathogens [10, 31, 38, 39]. None of these studies report function (as reflected by knee scores) or analyses of potential factors that influence the rate of infection control after one-stage revisions. According to published data [8, 9, 27, 49], factors that potentially influence the rate of infection control could be the duration of infection and the type of infected prostheses; hinged prostheses, for example, are associated with high infection rates [8, 27, 49].
Table 1.
Results of septic one-stage revisions
| Study | Number of patients | Followup (months) | Rate of infection control | Score | Flexion |
|---|---|---|---|---|---|
| von Foerster et al. [57] | 104 | 75.5 | 73.1% | – | – |
| Lu et al. [40] | 8 | 20.1 | 87% | – | |
| Göksan & Freeman [23] | 18 | 60 | 89% | – | 87° |
| Buechel et al. [6] | 21 | 122.4 | 91% | 79.5 points (New Jersey Orthopaedic Hospital knee score) | |
| Sofer et al. [54] | 15 | 18.4 | 93% | 116.4 points (Knee Society score) | |
| Current study | 63 | 35.9 | 95% | 144.1 points (Knee Society score) | 114° |
We therefore determined: (1) the rate of infection control with one-stage revision of septic knee prostheses, (2) the clinical knee scores that can be achieved, (3) whether the duration of infection or the type of prosthesis influences the level of infection control, and (4) whether different types of prostheses influence the knee scores.
Patients and Methods
We retrospectively reviewed the records of prospectively collected data for 63 patients who underwent one-stage revision surgery for infected knee prostheses between July 2004 and February 2006. During that time, we treated a total of 72 patients with infected prostheses. The indications for one-stage revision were: (1) known microorganism with an antibiotic susceptibility profile and (2) wounds that could be closed during surgery. The contraindications were: (1) an unknown microorganism (two cases), (2) wound defects that could not be closed during surgery (one case), and (3) patients with infections caused by MRSA or MRSE (six cases) because these were not considered in other studies of one-stage revisions, and the fact that these microorganisms generally were associated with a lower rate of infection control [6, 10, 23, 31, 38–40, 53, 57]. The nine excluded patients left 63 included in the study, 32 women and 31 men, with an average age of 70.7 ± 10.5 years (range, 31–89 years). The mean lifetime of their implants was 37.2 ± 36.5 months (range, 3–170 months). There were six infected unicondylar prostheses replaced with a primary total knee replacement prosthesis with retropatellar replacement (Innex; Zimmer GmbH, Winterthur, Switzerland) (Group 1), 37 infected primary total knee replacement prostheses replaced with rotating hinged prostheses (Roknep; Waldemar Link, Norderstedt, Germany) (Group 2), and 20 infected hinged prostheses replaced with hinged prostheses again or with modular rotating hinged prostheses (Roknep and modular Roknep; Waldemar Link) (Group 3). None of the patients in Groups 1 or 2 had undergone previous septic revision surgery. In Group 3, six of the 20 patients had undergone previous septic revision surgery (three had one revision, two had two revisions, and one had three two-stage revisions) (Table 2). The minimum followup was 24 months (mean, 36 months; range, 24–70 months). No patients were lost to followup before the minimum followup. All patients were recalled specifically for this study and analyses. All subjects gave informed consent to participate in the study and the protocol was approved by the research ethics board of our hospital.
Table 2.
Demographic data and potential risk factors for reinfection of the different groups
| Demographic data | Group 1 (unicompartmental prostheses) (n = 6) | Group 2 (primary total knee replacement prostheses) (n = 37) | Group 3 (rotating hinged prosthesis with and without reinfection) (n = 20) | Group 3 (rotating hinged prostheses without reinfection) (n = 17) | Group 3 (rotating hinged prostheses with reinfection) (n = 3) |
|---|---|---|---|---|---|
| Age (years) | 57 ± 15 | 71 ± 8.3 | 74 ± 9.6 | 75 ± 10 | 73 ± 7.6 |
| BMI | 26 ± 2.3 | 31 ± 4.0 | 28 ± 3. 6 | 28 ± 3.5 | 29 ± 4.4 |
| Men | 4 | 14 | 13 | 10 | 3 |
| Women | 2 | 23 | 7 | 7 | 0 |
| Septic revisions before (number of patients) | 0 | 0 | 6 | 3 | 3 |
| Two-stage revisions | 0 | 0 | 10 | 3 | 7 |
| Fistula | 0 | 2 | 7 | 4 | 3 |
| Anemia | 0 | 11 | 7 | 5 | 2 |
| Urinary tract infection | 0 | 2 | 1 | 1 | 0 |
| Smoking | 0 | 4 | 1 | 1 | 0 |
| Thyroid disease | 0 | 5 | 2 | 2 | 0 |
| Vascular disease | 0 | 3 | 5 | 4 | 1 |
| Diabetes mellitus | 1 | 6 | 6 | 4 | 2 |
| Insulin use | 0 | 4 | 1 | 1 | 0 |
| Coronary heart disease | 1 | 10 | 7 | 7 | 0 |
| Heart insufficiency | 0 | 13 | 7 | 7 | 0 |
| Hypertonus | 2 | 27 | 15 | 12 | 3 |
| Rheumatoid arthritis | 0 | 3 | 1 | 1 | 0 |
| Lung disease | 0 | 3 | 2 | 2 | 0 |
| Steroid/immunosuppressive therapy | 0 | 5 | 2 | 2 | 0 |
| Hyperuricemia | 0 | 4 | 2 | 1 | 1 |
| Hyperlipidemia | 0 | 7 | 6 | 6 | 0 |
| ASA I + II | 6 | 32 | 17 | 14 | 3 |
| ASA III + IV | 0 | 5 | 3 | 3 | 0 |
ASA = American Society of Anesthesiology.
The demographic data show some differences between the groups with respect to age and BMI (Table 2). Patients in Group 1 were younger than patients in Groups 2 and 3 (p = 0.001 and p = 0.002, Tukey’s Honestly Significant Difference [HSD] test). Patients of Groups 1 and 3 had lower BMI than patients in Group 3 (p = 0.006 and p = 0.022, Tukey’s HSD test).
The responsible bacteria were identified in presurgical biopsy specimens as described previously [18] (Table 3). Briefly, the biopsy specimens were obtained using arthroscopic biopsy forceps introduced via a small anterolateral approach, typical for arthroscopy, and were taken from the synovial lining and the periprosthetic tissue in different areas of the knee, close to the prosthesis. At least five samples were incubated for 14 days for microbiologic analysis and five samples were examined histologically. Infections involving two pathogens were identified in 11 cases (33%). Our microbiologist (LF) identified antibiotics specific for the microorganisms concerned. These antibiotics were mixed into the cement used to fix the prosthesis (Table 4). Previous studies have found antibiotics affect each other’s elution from the cement: the use of two antibiotics results in a synergistic effect in antibiotic release and the release of the individual components is greater than that of single antibiotics [1, 2, 14, 46, 53]. Therefore, we always used at least two antibiotics in the cement. Copal®G+C bone cement (Heraeus Medical, Wehrheim, Germany), which contains gentamicin and clindamycin, was used for the spacer if the microorganism was sensitive to gentamicin and clindamycin and because of the synergistic effect on the release of each antibiotic (28 cases) [14]. If the pathogens were resistant to one or both of these antibiotics, a supplement of 2 g vancomycin was added to 42.7 g Copal®G+C cement during the operation (27 cases). Copal®G+C cement was still used in these cases because release of both antibiotics from this bone cement is prolonged and Copal®G+C acts more effectively on biofilm formation as compared with gentamicin-loaded bone cement alone or other cement [14, 43]. In case of allergy to clindamycin, other antibiotics were added to the gentamicin-loaded bone cement chosen according to the specific susceptibility of the microorganisms (Table 4). Two antibiotics were used in the cement in 31 cases and three in 32 cases (Table 4); however, the total amount of antibiotic powder added to the cement was never more than 10% of the amount of cement powder because the mechanical properties of the cement otherwise would be compromised [30].
Table 3.
Identified pathogens
| Pathogen | Percentage (number of patients) |
|---|---|
| Staphylococcus epidermidis | 30% (19) |
| Staphylococcus aureus | 24% (15) |
| Staphylococcus hominis | 11% (7) |
| Enterococcus faecalis | 10% (6) |
| Staphylococcus capitis | 6% (4) |
| Staphylococcus haemolyticus | 6% (4) |
| Streptococcus | 5% (3) |
| Propionibacterium acnes | 8% (5) |
Table 4.
Antibiotic cocktails included in the bone cement
| Package size of bone cement | Antibiotic 1 | Antibiotic 2 | Antibiotic 3 | Number of patients |
|---|---|---|---|---|
| 42.7 g* | Gentamicin 1.0 g | Clindamycin 1.0 g | 28 | |
| 42.7 g* | Gentamicin 1.0 g | Clindamycin 1.0 g | Vancomycin 2.0 g | 27 |
| 40 g† | Gentamicin 0.5 g | Vancomycin 2.0 g | Ofloxacin 1.0 g | 2 |
| 40 g† | Gentamicin 0.5 g | Vancomycin 2.0 g | Ampicillin 1.0 g | 2 |
| 40 g† | Gentamicin 1.0 g | Vancomycin 3.0 g | 2 | |
| 42.7 g* | Gentamicin 1.0 g | Clindamycin 1.0 g | Ofloxacin 1.0 g | 1 |
| 40 g† | Gentamicin 1.0 g | Ofloxacin 2.0 g | 1 |
* Copal®G+C (Heraeus Medical GmbH, Wehrheim, Germany). The package size is 42.7 g cement containing 1.0 g gentamicin and 1.0 g clindamycin; †Palacos®R+G (Heraeus Medical GmbH, Wehrheim, Germany). The package size is 40.0 g cement containing 0.5 g gentamicin.
The revisions were performed by one of two experienced surgeons (JS, BF). During surgery, excision of the old wound and existing fistula (nine cases) was performed. A radical total synovectomy, débridement of all macroscopically suspicious tissue, and removal of all foreign material (implants and cement) were done. This was followed by lavage for approximately 3 minutes at least twice with 1 L antiseptic fluid for mucous membranes (Octinesept®, Schülke & Mayr GmbH, Norderstedt, Germany) each time. Thereafter 1 L of saline was used to clean the wound. Before reimplantation of the prosthesis, gloves and instruments were changed and the skin around the wound was disinfected again. Because we saw that radical débridement of knees with primary total knee replacement prostheses and hinged prostheses often resulted in ligamentous instability with mismatch of the flexion and extension gap, rotating hinged prostheses were chosen for reimplantation in these patients.
Physiotherapy began on the first postoperative day including mobilization using crutches with full weightbearing and isometric exercises of the muscles of the leg for antithrombotic prophylaxis and strengthening of the muscles. Wound drainage tubes were removed on the second postoperative day and thereafter physiotherapy started to move the joint with free movement and passive motion machines. Stair climbing was started on the fifth postoperative day and further exercises to strengthen the muscles during the second postoperative week. Additional systemic antibiotics were given for 6 weeks, intravenously for the first 2 weeks after surgery (Table 5), immediately followed by oral antibiotics for 4 weeks. Thirty-two patients (50.8%) were treated with two antibiotics. Linezolid was administered for infections (six) caused by multidrug-resistant Enterococcus.
Table 5.
Systemic antibiotics
| Antibiotic | Percentage (number of patients) |
|---|---|
| Flucloxacillin | 24% (15) |
| Rifampicin | 43% (27) |
| Vancomycin | 24% (15) |
| Ampicillin + sulbactam | 8% (5) |
| Ciprofloxacin | 29% (18) |
| Imipinem + cilastatin | 22% (14) |
| Cefuroxime | 17% (11) |
| Clindamycin | 3% (2) |
| Linezolid | 9% (6) |
| Penicillin G | 5% (3) |
One of us not involved in the surgery (AM) examined the patients clinically and radiographically every 3 months for the first 2 years after reimplantation surgery and thereafter every 6 months. Inflammatory parameters (C-reactive protein [CRP]) also were monitored. According to Haddad et al. [28] and Zimmerli et al. [58], a patient could be judged infection-free at followup if he or she was free of clinical signs for infection (fever, local pain, redness, warmth, sinus tract infection), had a CRP level less than 10 mg/dL, and showed no radiographic signs of osteolysis. The mean followup was 35.9 ± 8.1 months and the minimum followup was 24 months. We assessed functionality using the Knee Society (KS) scores (KS, 200 points possible) with the KS knee (100 points possible) and function (100 points possible) scores [35] and the Oxford knee score [13].
Two of us (JS, AM) independently evaluated all radiographs for osteolysis. We considered any osteolysis or any progressive radiolucencies observed during the followup period to be indicative of infection [28, 58]. The intraclass and interclass correlation coefficients for radiographic determination of progressive radiolucencies were 0.99 and 0.98, respectively.
The reinfection rate was given descriptively as the ratio of patients with any reinfection to all patients under study. The duration of the original infection was compared between patients who had recurrence of infection and patients who did not have reinfection, using the nonparametric Mann-Whitney test (a normal distribution was questionable). The times of functional indices were analyzed separately for the three groups of patients with different types of prostheses descriptively (mean, SD). Differences between these groups were tested with one-way ANOVA (omnibus test) for each time observed. Pair-wise differences between the three groups were tested with Tukey’s HSD test. The chi-square test was used for comparison of nominal variables between groups. Data were analyzed with the SPSS statistical package (Version 11.0; SPSS Inc, Chicago, IL, USA).
Results
None of the patients in Groups 1 and 2 experienced reinfection. Three of the 20 patients in Group 3 (infected hinged prostheses) had reinfections after revision surgery. These data calculate to a rate of infection control of 100% for Groups 1 and 2, 85% for Group 3, and 95% for all patients.
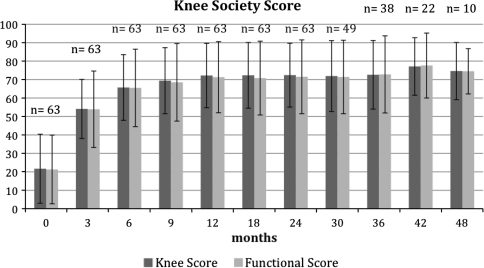
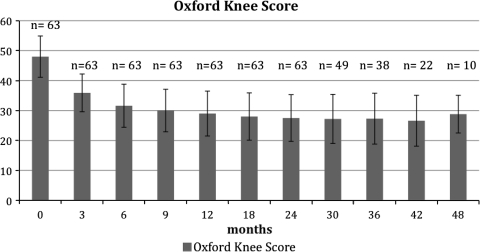
We observed improvement in the clinical scores in all 60 patients who did not have a reinfection. The mean preoperative KS knee score was 22, KS mean function score 21, and mean Oxford-12 score 48 (Table 6). Twenty-four months after surgery, the mean KS knee score was 72, mean KS function score 71 ± 20 (Fig. 1), and mean Oxford-12 knee score 27 (Fig. 2). The mean degree of flexion 2 years after surgery was 104° ± 11°.
Table 6.
Knee Society knee and function scores and Oxford scores
| Parameter | Number of patients | Group 1 | Group 2 | Group 3 | Group 1 ↔ Group 2 | Group 1 ↔ Group 3 | Group 2 ↔ Group 3 |
|---|---|---|---|---|---|---|---|
| Knee score preoperative | 63 | 47 ± 21 | 22 ± 17 | 14 ± 13 | p = 0.002 | p = 0.001 | p = 0.138 |
| Knee score 3 months postoperative | 63 | 67 ± 16 | 55 ± 15 | 47 ± 16 | p = 0.074 | p = 0.015 | p = 0.063 |
| Knee score 6 months postoperative | 63 | 78 ± 13 | 66 ± 17 | 69 ± 19 | p = 0.112 | p = 0.038 | p = 0.203 |
| Knee score 9 months postoperative | 63 | 79 ± 16 | 71 ± 18 | 62 ± 17 | p = 0.29 | p = 0.049 | p = 0.104 |
| Knee score 12 months postoperative | 63 | 84 ± 15 | 73 ± 7 | 67 ± 15 | p = 0.147 | p = 0.054 | p = 0.246 |
| Knee score 18 months postoperative | 63 | 83 ± 16 | 74 ± 17 | 66 ± 20 | p = 0.199 | p = 0.068 | p = 0.147 |
| Knee score 24 months postoperative | 63 | 85 ± 15 | 73 ± 16 | 67 ± 20 | p = 0.095 | p = 0.07 | p = 0.288 |
| Knee score 30 months postoperative | 49 | 83 ± 17 | 72 ± 18 | 66 ± 22 | p = 0.178 | p = 0.105 | p = 0.323 |
| Knee score 36 months postoperative | 38 | 84 ± 15 | 72 ± 17 | 66 ± 22 | p = 0.148 | p = 0.115 | p = 0.417 |
| Knee score 42 months postoperative | 22 | 94 ± 3 | 76 ± 14 | 72 ± 17 | p = 0.057 | p = 0.069 | p = 0.558 |
| Knee score 48 months postoperative | 10 | 90 ± 4 | 73 ± 12 | 73 ± 16 | p = 0.062 | p = 0.071 | p = 0.579 |
| Function score preoperative | 63 | 48 ± 16 | 21 ± 17 | 13 ± 14 | p = 0.001 | p < 0.001 | p = 0.088 |
| Function score 3 months postoperative | 63 | 73 ± 21 | 56 ± 18 | 43 ± 21 | p = 0.031 | p = 0.008 | p = 0.0296 |
| Function score 6 months postoperative | 63 | 83 ± 15 | 67 ± 18 | 56 ± 25 | p = 0.054 | p = 0.0246 | p = 0.0872 |
| Function score 9 months postoperative | 63 | 86 ± 19 | 70 ± 19 | 59 ± 23 | p = 0.058 | p = 0.0195 | p = 0.081 |
| Function score 12 months postoperative | 63 | 84 ± 18 | 73 ± 17 | 62 ± 22 | p = 0.158 | p = 0.0383 | p = 0.0474 |
| Function score 18 months postoperative | 63 | 84 ± 17 | 73 ± 17 | 62 ± 24 | p = 0.134 | p = 0.0508 | p = 0.079 |
| Function score 24 months postoperative | 63 | 85 ± 17 | 73 ± 17 | 64 ± 24 | p = 0.12 | p = 0.0612 | p = 0.1211 |
| Function score 30 months postoperative | 49 | 85 ± 19 | 72 ± 18 | 63 ± 23 | p = 0.123 | p = 0.0553 | p = 0.1554 |
| Function score 36 months postoperative | 38 | 87 ± 17 | 73 ± 18 | 62 ± 25 | p = 0.121 | p = 0.056 | p = 0.151 |
| Function score 42 months postopertive | 22 | 95 ± 9 | 78 ± 15 | 68 ± 20 | p = 0.09 | p = 0.07 | p = 0.275 |
| Function score 48 months postoperative | 10 | 85 ± 11 | 72 ± 16 | 77 ± 20 | p = 0.099 | p = 0.079 | p = 0.285 |
| Oxford score preoperative | 63 | 40 ± 4 | 48 ± 6 | 50 ± 7 | p = 0.004 | p = 0.003 | p = 0.387 |
| Oxford score 3 months postoperative | 63 | 30 ± 3 | 36 ± 6 | 38 ± 7 | p = 0.01 | p = 0.018 | p = 0.427 |
| Oxford score 6 months postoperative | 63 | 24 ± 5 | 32 ± 7 | 33 ± 7 | p = 0.009 | p = 0.017 | p = 0.781 |
| Oxford score 9 months postoperative | 63 | 22 ± 6 | 30 ± 7 | 32 ± 6 | p = 0.005 | p = 0.001 | p = 0.43 |
| Oxford score 12 months postoperative | 63 | 20 ± 7 | 30 ± 7 | 31 ± 7 | p = 0.003 | p = 0.0066 | p = 0.578 |
| Oxford score 18 months postoperative | 63 | 20 ± 7 | 28 ± 8 | 30 ± 7 | p = 0.021 | p = 0.007 | p = 0.309 |
| Oxford score 24 months postoperative | 63 | 20 ± 7 | 28 ± 7 | 29 ± 9 | p = 0.007 | p = 0.029 | p = 0.736 |
| Oxford score 30 months postoperative | 49 | 20 ± 8 | 28 ± 7 | 29 ± 8 | p = 0.024 | p = 0.024 | p = 0.433 |
| Oxford score 36 months postoperative | 38 | 20 ± 8 | 28 ± 8 | 32 ± 9 | p = 0.029 | p = 0.017 | p = 0.213 |
| Oxford score 42 months postoperative | 22 | 19 ± 10 | 27 ± 8 | 30 ± 9 | p = 0.131 | p = 0.128 | p = 0.474 |
| Oxford score 48 months postoperative | 10 | 20 ± 10 | 29 ± 8 | 29 ± 9 | p = 0.14 | p = 0.132 | p = 0.482 |
| Flexion 24 months postoperative (°) | 63 | 120 ± 4 | 106 ± 9 | 94 ± 10 | p = 0.001 | p < 0.001 | p < 0.001 |
↔ = Pair-wise comparison between the three groups with Tukey’s Honestly Significant Difference (HSD) test.
Fig. 1.
The Knee Society scores after one-stage revision of septic knee arthroplasties are shown for the complete collective (n = number of patients examined)
Fig. 2.
The Oxford knee scores after one-stage revision of septic knee arthroplasties are shown for the complete collective (n = number of patients examined)
All of the patients with infection recurrence had infected prostheses for at least 5 years and had undergone septic revision surgery several times at other institutions (two patients with two two-stage revision operations and one patient with three two-stage revision operations). Previous septic revision surgery occurred more often in patients with reinfected hinged prostheses than in patients with hinged prostheses without reinfection (p = 0.004). After extensive debridement and removal of the infected tibial tuberosity, all three reinfections underwent septic arthrodesis with an arthrodesis rod; as a result, no additional reinfections occurred. The duration of the original infection in the patients who had a recurrence of infection was 60 ± 8.1 months and was longer (p = 0.046) than the 32 ± 23 months for patients who did not have reinfections.
Group 1 had better scores at all times than Group 3 and better data than Group 2 at many of the followups (Table 6). Group 2 scores were only slightly better than those of Group 3 (Table 6). Twenty-four months after septic revision, flexion was best in Group 1, followed by Group 2, and then Group 3.
In Group 2, three more operations were performed: one patient had debridement of wound necrosis with secondary closure and two had manipulation under anesthesia owing to flexion less than 90° at the 6-week followup. In Group 3, apart from the three patients who had revision surgeries, an additional two patients had débridement for wound necrosis and three had manipulations under anesthesia.
Discussion
The rate of infection control for one-stage revision of infected knee arthroplasties is unclear: the only study with a reasonable number of cases [57] had a low rate of 73%, whereas other studies with higher rates between 87% and 91% were based on only eight to 21 patients [6, 23, 40, 52, 57]. Moreover, the function of these patients and potential factors influencing the rate of infection control are unknown. We therefore determined: (1) the rate of infection control with one-stage revision of septic knee prostheses, (2) the clinical knee scores that can be achieved, (3) whether the duration of infection or the type of prosthesis influence the level of infection control, and (4) whether different types of prostheses influence the knee scores.
This study has numerous limitations. First, we had a relatively short followup. Although reinfections have been described for single cases up to 5 years after implantation, in general they are observed within the first 2 postoperative years [24, 29, 31, 39, 57]. Second, Groups 1 to 3 differ in size, and for age and BMI of the patients, so they are not comparable for every parameter. However the treatment was the same for all patients, only the type of prosthesis differed between groups; therefore, we believe that separate descriptions of infection and clinical scores for these groups are appropriate. Third, this is a descriptive study and the number of reinfections was too small to perform multivariate analysis to analyze influencing factors on the rate of infection control and to obtain conclusive information; the statistical tests we used to compare groups do not control for confounding variables. However, the differences in age and BMI were between patients in Group 1 and the others but not between patients with and without reinfection. Because of these statistical limitations, the results should be interpreted with caution and other factors influencing the rate of reinfection might not necessarily have been identified.
The one-stage revision procedure led to an infection control rate of 95%, which is comparable to the rate resulting from two-stage revision. Rates of infection control for two-stage revisions have been reported between 89% and 96% [20, 22, 28, 32, 41, 42]. Bengtson and Knutson [4] analyzed data from the Swedish Register and found no difference between the infection control rates after one-stage or two-stage procedures. Similarly, Jämsen et al. [37] found no difference in control of infections after a search of relevant publications.
We found a mean KS score (200 points possible) of 144 points after 2 years followup and 149 points after 4 years followup. These data are comparable to the results reported by Buechel et al. [6], with a mean of 79 points (with the New Jersey Orthopaedic Hospital knee score, 100 points possible) and better than the mean results reported by Sofer et al. [54] with 116 points (KS score) for one-stage septic revision surgery (Table 1). They also are better than results reported for two-stage revision surgery in studies using the same clinical scoring that we used (Table 7). Freeman et al. [20] implanted mobile spacers and reported a mean KS pain score of 45 points of a possible 50 and a KS function score of 70 points after a followup of 71 months. Barrack et al. [3] reported a mean KS total score of 115 points and a KS function score of 33 points; Haalem et al. [29] reported a mean KS knee score of 89 points and a KS function score of 50 points after 7.2 years; and Meek et al. [42] reported a mean Oxford-12 score of 67.3 points after 2 years. The average flexion of the knee after two-stage revision surgery is reportedly between 87° and 106° and most of the time there was a slight extension deficit of 5° [3, 15, 28, 29, 32, 42] (Table 7). These data were slightly worse than those from our study: 104° ± 11° flexion (between 93° and 115°).
Table 7.
Results of septic two-stage revisions
| Study | Number of patients | Followup (months) | Rate of infection control | Score | Flexion |
|---|---|---|---|---|---|
| Freeman et al. [20] | 28 static | 86.6 | 92% | 50 (KS knee score) + 45 (KS function score) | – |
| 48 articulating spacers | 62.2 | 95% | 45 (KS knee score) + 70 (KS function score) | ||
| Barrack et al. [3] | 28 | 36 | 93% | 115 points (KS score) |
89° |
| Haalem et al. [29] | 94 | 86.4 | 91% | 139 points (KS score) |
90° |
| Meek et al. [42] | 47 | 24 | 96% | 67.3 points (Oxford-12 knee score) | 87° |
| Current study | 63 | 35.9 | 95% | 144.1 points (KS score) |
114° |
| 27.5 points (Oxford-12 knee score) |
KS = Knee Society.
We found the type of infected implant plays a role in the outcome of one-stage septic revision surgery. Thus, one-stage revision of infected unicondylar and primary total knee replacement prostheses did not lead to any reinfections, whereas infected hinged prostheses were associated with a reinfection rate of 15%. This is in agreement with other studies showing higher infection rates between 6.9% and 13% for hinged prostheses in primary and revision arthroplasties [8, 27, 49]. In accordance with Cierny and DiPasquale [9], we found that duration of the infection is an important influencing factor for control of infection. In the current study it was apparent that all patients who had recurring infections had experienced chronic infections for several years before, therefore it can be assumed that these patients had osteomyelitis of the long bones. In these patients, the infection could be eradicated only after radical debridement with removal of the tibial tuberosity required for attachment of the extensor musculature and subsequent arthrodesis. We believe arthrodesis was indicated in our patients because of involvement of the tibial tuberosity with osteomyelitis. We still perform a one-stage procedure for the first revision of infected hinged prostheses, with implantation of a rotating hinged prosthesis, but change to two-stage revisions if the periprosthetic infection of the hinged prostheses lasted longer than a year and more than one septic revision was done previously. Even though hinged prostheses are associated with higher reinfection rates, we use them for reimplantation after septic revision of primary total knee replacement prostheses because we observed that radical débridement led to ligamentous instability with mismatch of the flexion and extension gap and a high risk of dislocation if semiconstrained prostheses were used [8, 9, 27, 49].
Concerning the type of infected prosthesis, we found one-stage revision of a unicompartmental prosthesis to a total knee replacement prosthesis gives slightly better clinical scores than one-stage revision of an infected total knee replacement prosthesis to a hinged prosthesis, and substantially better clinical scores than one-stage exchange of a hinged prosthesis to another hinged prosthesis. The second group had only slightly higher clinical scores than the latter group.
We believe a prerequisite for the success of this one-stage procedure is isolation of the organism(s) from previously obtained aspirated fluid or biopsy material. This is followed by determination of their antibiotic susceptibility so that an organism-specific mixture of antibiotics can be added to the bone cement and specific local antibiotic treatment initiated [53, 56]. It is important for the fluid or tissue sample to be incubated for 14 days [17, 18, 26, 51]. This long incubation period is recommended because the pathogens causing the periprosthetic infection usually occur in very small numbers in the form of a biofilm and often are in a sessile state that is characterized by a slow rate of reproduction [11, 21, 26, 44, 47]. One analysis of 110 infected hip and knee endoprostheses showed the culture detection rate after 7 days, the longest incubation period reported in most studies, was 73.6%. To identify all infections, it was important to culture for at least 13 days [51]. Second, we believe rigorous removal of all foreign material and radical débridement of inflamed and necrotic tissues, including a total synovectomy during the operation, are essential for the success of any form of septic prosthesis revision [17, 57]. Third, a specific systemic therapy with an antibiotic of high bioavailability to which the bacteria are highly sensitive, coupled with a high dose of locally applied specific antibiotic in the antibiotic-loaded cement, seems decisive for effective treatment of periprosthetic infections [12, 54, 57]. Although there are a limited number of studies concerned with local release of antibiotics contained in the cement [5, 19, 33, 41 ], this is a procedure during one-stage and two-stage septic prosthesis revisions of the knee and hip that has become well accepted [30, 55, 56].
Mixing antibiotic into the cement affects the quality of the cement, which is why only antibiotic powder to a maximum of 10% of the total cement amount should be used [30]. Not all antibiotics can be used because they must be available in powder form, water soluble, and thermostable. The most commonly used are gentamicin, clindamycin, vancomycin, tobramycin, aztreonam, ampicillin, and ofloxacin [17, 30, 33]. Few data are available that address the release of antibiotics from cement in vivo during several weeks, although the level of released antibiotic has been suggested by some to be sufficient for at least 4 months [5, 19, 33, 41]. Furthermore, the antibiotics affect each other’s elution from the cement whereby the use of two antibiotics results in a synergistic effect and the release of the individual components is greater than that of the single antibiotics on their own [1, 2, 14, 46, 53]. Therefore, we always used at least two antibiotics in the cement and preferred COPAL®G+C cement to Palacos® R+G cement (Heraeus Medical, Wehrheim, Germany) whenever possible, because the former exhibits better release of gentamicin [14].
One-stage revision of infected knee prostheses leads to a high rate of infection control of 95% and reasonable patient function when the pathogen is identifiable and when MRSA and MRSE infections are excluded. This is particularly true for infected unicompartmental and primary total knee replacement prostheses. For patients with periprosthetic infections of hinged prostheses, in which the infection has been ongoing for several years, the probability of osteomyelitis is high: for these patients this method of revision surgery was associated with a rate of recurrence of 15%. The 100% rate of infection control in unicompartmental and primary total knee replacement prostheses with our study protocol suggests that this protocol also may be effective for infections with MRSA and MRSE in these types of prostheses, especially because infection with multidrug-resistant Enterococcus could be treated successfully. Because of the increasing numbers of periprosthetic infections with MRSA and MRSE bacteria, we will extend this treatment to periprosthetic infections of unicompartmental and primary total knee replacement prostheses caused by MRSA and MRSE.
Acknowledgments
We thank Martin S. Schulz MD and D. Labes, Biostatistics, Cooperative Clinical Drug Research and development, Neuenhagen, Germany, for support in performing the statistics. We thank Alexander Mittelstädt MD for assistance in assessing the data.
Footnotes
Each author certifies that he or she, or a member of their immediate family, has no commercial associations (eg, consultancies, stock ownership, equity interest, patent/licensing arrangements, etc) that might pose a conflict of interest in connection with the submitted article.
All ICMJE Conflict of Interest Forms for authors and Clinical Orthopaedics and Related Research editors and board members are on file with the publication and can be viewed on request.
Clinical Orthopaedics and Related Research neither advocates nor endorses the use of any treatment, drug, or device. Readers are encouraged to always seek additional information, including FDA-approval status, of any drug or device prior to clinical use.
Each author certifies that his or her institution approved the human protocol for this investigation, that all investigations were conducted in conformity with ethical principles of research, and that informed consent for participation in the study was obtained.
This work was performed at the Orthopaedic Clinic Markgröningen gGmbH, Markgröningen, Germany.
References
- 1.Anagnostakos K, Kelm J, Regitz T, Schmitt E, Jung W. In vitro evaluation of antibiotic release from and bacteria growth inhibition by antibiotic-loaded acrylic bone cement spacers. J Biomed Mater Res B Appl Biomater. 2005;72:373–378. doi: 10.1002/jbm.b.30171. [DOI] [PubMed] [Google Scholar]
- 2.Baleani M, Persson C, Zolezzi C, Andollina A, Borelli AM, Tigani D. Biological and biomechanical effects of vancomycin and meropenem in acrylic bone cement. J Arthroplasty. 2008;23:1232–1238. doi: 10.1016/j.arth.2007.10.010. [DOI] [PubMed] [Google Scholar]
- 3.Barrack RL, Engh G, Rorabeck C, Sawhney J, Woolfrey M. Patient satisfaction and outcome after septic versus aseptic revision total knee arthroplasty. J Arthroplasty. 2000;15:990–993. doi: 10.1054/arth.2000.16504. [DOI] [PubMed] [Google Scholar]
- 4.Bengtson S, Knutson K. The infected knee arthroplasty: a 6-year follow-up of 357 cases. Acta Orthop Scand. 1991;62:301–311. doi: 10.3109/17453679108994458. [DOI] [PubMed] [Google Scholar]
- 5.Bertazonni Minelli E, Benini A, Magnan B, Bartolozzi P. Release of gentamicin and vancomycin from temporary human hip spacers in two-stage revision of infected arthroplasty. J Antimicrob Chemother. 2004;53:329–334. doi: 10.1093/jac/dkh032. [DOI] [PubMed] [Google Scholar]
- 6.Buechel FF, Femino FP, D’Alessio J. Primary exchange revision arthroplasty for infected total knee replacement: a long-term study. Am J Orthop (Belle Mead NJ). 2004;33:190–198; discussion 198. [PubMed]
- 7.Calton TF, Fehring TK, Griffin WL. Bone loss associated with the use of spacer blocks in infected total knee arthroplasty. Clin Orthop Relat Res. 1997;345:148–154. doi: 10.1097/00003086-199712000-00020. [DOI] [PubMed] [Google Scholar]
- 8.Cameron HU, Hu C, Vyamont D. Hinge total knee replacement revisited. Can J Surg. 1997;40:278–283. [PMC free article] [PubMed] [Google Scholar]
- 9.Cierny G, 3rd, DiPasquale D. Periprosthetic total joint infections: staging, treatment, and outcomes. Clin Orthop Relat Res. 2002;403:23–28. doi: 10.1097/00003086-200210000-00005. [DOI] [PubMed] [Google Scholar]
- 10.Cordero-Ampuero J, Esteban J, Garcia-Rey E. Results after late polymicrobial, gram-negative, and methicillin-resistant infections in knee arthroplasty. Clin Orthop Relat Res. 2010;468:1229–1236. doi: 10.1007/s11999-009-1224-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Costerton JW. Biofilm theory can guide the treatment of device-related orthopaedic infections. Clin Orthop Relat Res. 2005;437:7–11. doi: 10.1097/00003086-200508000-00003. [DOI] [PubMed] [Google Scholar]
- 12.Cui Q, Mihalko WM, Shields JS, Ries M, Saleh KJ. Antibiotic-impregnated cement spacers for the treatment of infection associated with total hip or knee arthroplasty. J Bone Joint Surg Am. 2007;89:871–882. doi: 10.2106/JBJS.E.01070. [DOI] [PubMed] [Google Scholar]
- 13.Dawson J, Fitzpatrick R, Murray D, Carr A. Questionnaire on the perceptions of patients about total knee replacement. J Bone Joint Surg Br. 1998;80:63–69. doi: 10.1302/0301-620X.80B1.7859. [DOI] [PubMed] [Google Scholar]
- 14.Ensing GT, Horn JR, Mei HC, Busscher HJ, Neut D. Copal bone cement is more effective in preventing biofilm formation than Palacos R-G. Clin Orthop Relat Res. 2008;466:1492–1498. doi: 10.1007/s11999-008-0203-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Fehring TK, Griffin WL. Revision of failed cementless total knee implants with cement. Clin Orthop Relat Res. 1998;356:34–38. doi: 10.1097/00003086-199811000-00007. [DOI] [PubMed] [Google Scholar]
- 16.Fehring TK, Odum S, Calton TF, Mason JB. Articulating versus static spacers in revision total knee arthroplasty for sepsis: The Ranawat Award. Clin Orthop Relat Res. 2000;380:9–16. doi: 10.1097/00003086-200011000-00003. [DOI] [PubMed] [Google Scholar]
- 17.Fink B, Grossmann A, Fuerst M, Schäfer P, Frommelt L. Two-stage cementless revision of infected hip endoprostheses. Clin Orthop Relat Res. 2009;467:1848–1858. doi: 10.1007/s11999-008-0611-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Fink B, Makowiak C, Fuerst M, Berger I, Schäfer P, Frommelt L. The value of synovial biopsy, joint aspiration, and C-reactive protein in the diagnosis of late peri-prosthetic infection of total knee replacements. J Bone Joint Surg Br. 2008;90:874–878. doi: 10.1302/0301-620X.90B7.20417. [DOI] [PubMed] [Google Scholar]
- 19.Fink B, Vogt S, Reinsch M, Büchner H. Sufficient release of antibiotic by a spacer 6 weeks after implantation in two-stage revision of infected hip prostheses. Clin Orthop Relat Res. 2011;469:3141–3147. doi: 10.1007/s11999-011-1937-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Freeman MG, Fehring TK, Odum SM, Fehring K, Griffin WL, Mason JB. Functional advantage of articulating versus static spacers in 2-stage revision for total knee arthroplasty infection. J Arthroplasty. 2007;22:1116–1121. doi: 10.1016/j.arth.2007.04.009. [DOI] [PubMed] [Google Scholar]
- 21.Gallo J, Kolar M, Novotny R, Rihakova P, Ticha V. Pathogenesis of prosthesis-related infection. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2003;147:27–35. doi: 10.5507/bp.2003.004. [DOI] [PubMed] [Google Scholar]
- 22.Ghanem E, Azzam K, Seeley M, Joshi A, Parvizi J. Staged revision for knee arthroplasty infection: what is the role of serologic tests before reimplantation? Clin Orthop Relat Res. 2009;467:1699–1705. doi: 10.1007/s11999-009-0742-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Göksan SB, Freeman MA. One-stage reimplantation for infected total knee arthroplasty. J Bone Joint Surg Br. 1992;74:78–82. doi: 10.1302/0301-620X.74B1.1732271. [DOI] [PubMed] [Google Scholar]
- 24.Goldman RT, Scuderi GR, Insall JN. 2-stage reimplantation for infected total knee replacement. Clin Orthop Relat Res. 1996;331:118–124. doi: 10.1097/00003086-199610000-00016. [DOI] [PubMed] [Google Scholar]
- 25.Goldstein WM, Kopplin M, Wall R, Berland K. Temporary articulating methylmethacrylate antibiotic spacer (TAMMAS): a new method of intraoperative manufacturing of a custom articulating spacer. J Bone Joint Surg Am. 2001;83(suppl 2):92–97. [PubMed] [Google Scholar]
- 26.Gollwitzer H, Diehl P, Gerdesmeyer L, Mittelmeier W. [Diagnostic strategies in cases of suspected periprosthetic infection of the knee: a review of the literature and current recommendations][in German]. Orthopäde. 2006;35:904, 906–908, 910–916. [DOI] [PubMed]
- 27.Guenoun B, Latargez L, Freslon M, Defossez G, Salas N, Gayet LE. Complications following rotating hinge Endo-Modell (Link) knee arthroplasty. Orthop Traumatol Surg Res. 2009;95:529–536. doi: 10.1016/j.otsr.2009.07.013. [DOI] [PubMed] [Google Scholar]
- 28.Haddad FS, Masri BA, Campbell D, McGraw RW, Beauchamp CP, Duncan CP. The PROSTALAC functional spacer in two-stage revision for infected knee replacements: prosthesis of antibiotic-loaded acrylic cement. J Bone Joint Surg Br. 2000;82:807–812. doi: 10.1302/0301-620X.82B6.10486. [DOI] [PubMed] [Google Scholar]
- 29.Haleem AA, Berry DJ, Hanssen AD. Mid-term to long-term followup of two-stage reimplantation for infected total knee arthroplasty. Clin Orthop Relat Res. 2004;428:35–39. doi: 10.1097/01.blo.0000147713.64235.73. [DOI] [PubMed] [Google Scholar]
- 30.Hanssen AD, Spangehl MJ. Practical applications of antibiotic loaded bone cement for treatment of infected joint replacements. Clin Orthop Relat Res. 2004;427:79–85. doi: 10.1097/01.blo.0000143806.72379.7d. [DOI] [PubMed] [Google Scholar]
- 31.Hirakawa K, Stulberg BN, Wilde AH, Bauer TW, Secic M. Results of 2-stage reimplantation for infected total knee arthroplasty. J Arthroplasty. 1998;13:22–28. doi: 10.1016/S0883-5403(98)90071-7. [DOI] [PubMed] [Google Scholar]
- 32.Hofmann AA, Kane KR, Tkach TK, Plaster RL, Camargo MP. Treatment of infected total knee arthroplasty using an articulating spacer. Clin Orthop Relat Res. 1995;321:45–54. [PubMed] [Google Scholar]
- 33.Hsieh PH, Chang YH, Chen SH, Ueng SW, Shih CH. High concentration and bioactivity of vancomycin and aztreonam eluted from Simplex cement spacers in two-stage revision of infected hip implants: a study of 46 patients at an average follow-up of 107 days. J Orthop Res. 2006;24:1615–1621. doi: 10.1002/jor.20214. [DOI] [PubMed] [Google Scholar]
- 34.Huotari K, Lyytikäinen O, Hospital Infection Surveillance Team Impact of postdischarge surveillance on the rate of surgical site infection after orthopedic surgery. Infect Control Hosp Epidemiol. 2006;27:1324–1329. doi: 10.1086/509840. [DOI] [PubMed] [Google Scholar]
- 35.Insall JN, Dorr LD, Scott RD, Scott WN. Rationale of the Knee Society clinical rating system. Clin Orthop Relat Res. 1989;248:13–14. [PubMed] [Google Scholar]
- 36.Jämsen E, Stogiannidis I, Malmivaara A, Pajamäki J, Puolakka T, Konttinen YT. Outcome of prosthesis exchange for infected knee arthroplasty: the effect of treatment approach. Acta Orthop. 2009;80:67–77. doi: 10.1080/17453670902805064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Jämsen E, Varonen M, Huhtala H, Lehto MU, Lumio J, Konttinen YT, Moilanen T. Incidence of prosthetic joint infections after primary knee arthroplasty. J Arthroplasty. 2010;25:87–92. doi: 10.1016/j.arth.2008.10.013. [DOI] [PubMed] [Google Scholar]
- 38.Kordelle J, Frommelt L, Klüber D, Seemann K. Results of one-stage endoprosthesis revision in periprosthetic infection caused by methicillin-resistant Staphylococcus aureus][in German. Z Orthop Ihre Grenzgeb. 2000;138:240–244. doi: 10.1055/s-2000-10143. [DOI] [PubMed] [Google Scholar]
- 39.Kurd MF, Ghanem E, Steinbrecher J, Parvizi J. Two-stage exchange knee arthroplasty: does resistance of the infecting organism influence the outcome? Clin Orthop Relat Res. 2010;468:2060–2066. doi: 10.1007/s11999-010-1296-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Lu H, Kou B, Lin J. One-stage reimplantation for the salvage of total knee arthroplasty complicated by infection][in Chinese. Zhonghua Wai Ke Za Zhi. 1997;35:456–458. [PubMed] [Google Scholar]
- 41.Masri BA, Duncan CP, Beauchamp CP. Long-term elution of antibiotics from bone-cement: an in vivo study using the prosthesis of antibiotic-loaded acrylic cement (PROSTALAC) system. J Arthroplasty. 1998;13:331–338. doi: 10.1016/S0883-5403(98)90179-6. [DOI] [PubMed] [Google Scholar]
- 42.Meek RM, Masri BA, Dunlop D, Garbuz DS, Greidanus NV, McGraw R, Duncan CP. Patient satisfaction and functional status after treatment of infection at the site of a total knee arthroplasty with use of the PROSTALAC articulating spacer. J Bone Joint Surg Am. 2003;85:1888–1892. doi: 10.1302/0301-620X.85B8.14214. [DOI] [PubMed] [Google Scholar]
- 43.Neut D, Groot EP, Kowalski RS, Horn JR, Mei HC, Busscher HJ. Gentamicin-loaded bone cement with clindamycin or fusidic acid added: biofilm formation and antibiotic release. J Biomed Mater Res A. 2005;73:165–170. doi: 10.1002/jbm.a.30253. [DOI] [PubMed] [Google Scholar]
- 44.Neut D, Horn JR, Kooten TG, Mei HC, Busscher HJ. Detection of biomaterial-associated infections in orthopaedic joint implants. Clin Orthop Relat Res. 2003;413:261–268. doi: 10.1097/01.blo.0000073345.50837.84. [DOI] [PubMed] [Google Scholar]
- 45.Peersman G, Laskin R, Davis J, Peterson M. Infection in total knee replacement: a retrospective review of 6489 total knee replacements. Clin Orthop Relat Res. 2001;392:15–23. doi: 10.1097/00003086-200111000-00003. [DOI] [PubMed] [Google Scholar]
- 46.Penner MJ, Masri BA, Duncan CP. Elution characteristics of vancomycin and tobramycin combined in acrylic bone–cement. J Arthroplasty. 1996;11:939–944. doi: 10.1016/S0883-5403(96)80135-5. [DOI] [PubMed] [Google Scholar]
- 47.Peters G, Herrmann M, Eiff C. The changing pattern of coagulase–negative staphylococci as infectious pathogens. Curr Opin Infect Dis. 1995;8(suppl):S12–S19. doi: 10.1097/00001432-199503001-00004. [DOI] [Google Scholar]
- 48.Phillips JE, Crane TP, Noy M, Elliot TS, Grimer RJ. The incidence of deep prosthetic infections in a specialist orthopaedic hospital: a 15-year prospective survey. J Bone Joint Surg Br. 2006;88:943–948. doi: 10.1302/0301-620X.88B7.17150. [DOI] [PubMed] [Google Scholar]
- 49.Pour AE, Parvizi J, Slenker N, Purtill JJ, Sharkey PF. Rotating hinged total knee replacement: use with caution. J Bone Joint Surg Am. 2007;89:1735–1741. doi: 10.2106/JBJS.F.00893. [DOI] [PubMed] [Google Scholar]
- 50.Saleh KJ, Rand JA, McQueen DA. Current status of revision total knee arthroplasty: how do we assess results? J Bone Joint Surg Am. 2003;85(suppl 1):18–20. doi: 10.2106/00004623-200300001-00005. [DOI] [PubMed] [Google Scholar]
- 51.Schäfer P, Fink B, Sandow D, Margull A, Berger I, Frommelt L. Prolonged bacterial culture to identify late periprosthetic joint infection: a promising strategy. Clin Infect Dis. 2008;47:1403–1409. doi: 10.1086/592973. [DOI] [PubMed] [Google Scholar]
- 52.Scuderi GR, Insall JN, Windsor RE, Moran MC. Survivorship of cemented knee replacements. J Bone Joint Surg Br. 1989;71:798–803. doi: 10.1302/0301-620X.71B5.2584250. [DOI] [PubMed] [Google Scholar]
- 53.Simpson PM, Dall GF, Breusch SJ, Heisel C. In vitro elution and mechanical properties of antibiotic-loaded SmartSet HV and Palacos R acrylic bone cements][in German. Orthopäde. 2005;34:1255–1262. doi: 10.1007/s00132-005-0861-2. [DOI] [PubMed] [Google Scholar]
- 54.Sofer D, Regenbrecht B, Pfeil J. Early results of one-stage septic revision arthroplasties with antibiotic-laden cement: a clinical and statistical analysis][in German. Orthopäde. 2005;34:592–602. doi: 10.1007/s00132-005-0780-2. [DOI] [PubMed] [Google Scholar]
- 55.Steinbrink K, Frommelt L. [Treatment of periprosthetic infection of the hip using one-stage exchange surgery][in German] Orthopäde. 1995;24:335–343. [PubMed] [Google Scholar]
- 56.Stockley I, Mockford BJ, Hoad-Reddick A, Norman P. The use of two-stage exchange arthroplasty with depot antibiotics in the absence of long-term antibiotic therapy in infected total hip replacement. J Bone Joint Surg Br. 2008;90:145–148. doi: 10.1302/0301-620X.90B2.19855. [DOI] [PubMed] [Google Scholar]
- 57.Foerster G, Klüber D, Käbler U. [Mid- to long-term results after treatment of 118 cases of periprosthetic infections after knee joint replacement using one-stage exchange surgery][in German] Orthopäde. 1991;20:244–252. [PubMed] [Google Scholar]
- 58.Zimmerli W, Widmer AF, Blatter M, Frei R, Ochsner PE. Role of rifampin for treatment of orthopedic implant-related staphylococcal infections: a randomized controlled trial. Foreign-Body Infection (FBI) Study Group. JAMA. 1998;279:1537–1541. doi: 10.1001/jama.279.19.1537. [DOI] [PubMed] [Google Scholar]