Abstract
Rejuvenation of telomeres with various lengths has been found in induced pluripotent stem cells (iPSCs). Mechanisms of telomere length regulation during induction and proliferation of iPSCs remain elusive. We show that telomere dynamics are variable in mouse iPSCs during reprogramming and passage, and suggest that these differences likely result from multiple potential factors, including the telomerase machinery, telomerase-independent mechanisms and clonal influences including reexpression of exogenous reprogramming factors. Using a genetic model of telomerase-deficient (Terc−/− and Terc+/−) cells for derivation and passages of iPSCs, we found that telomerase plays a critical role in reprogramming and self-renewal of iPSCs. Further, telomerase maintenance of telomeres is necessary for induction of true pluripotency while the alternative pathway of elongation and maintenance by recombination is also required, but not sufficient. Together, several aspects of telomere biology may account for the variable telomere dynamics in iPSCs. Notably, the mechanisms employed to maintain telomeres during iPSC reprogramming are very similar to those of embryonic stem cells. These findings may also relate to the cloning field where these mechanisms could be responsible for telomere heterogeneity after nuclear reprogramming by somatic cell nuclear transfer.
Keywords: telomere, telomerase, recombination, iPSCs, reprogramming
Introduction
Reprogramming of somatic cells to induced pluripotent stem cells (iPSCs) with an ESC-like state has been successfully achieved by overexpression of defined transcription factors 1, 2, 3. Like mouse embryonic stem cells (ESCs), iPSCs can self-renew indefinitely and generate all cell types in the body, exhibiting true pluripotency 4, 5, 6. Notably, iPSC lines differ in their pluripotency and are not equally successful in producing viable iPSC pups, and some show defective fetal development at embryonic stages in the tetraploid embryo complementation (TEC) test 5.
Telomeres consist of repeated guanine-rich sequences and associated protein complexes known as shelterin that cap the end of chromosomes to maintain genomic stability 7, 8. Telomere length is primarily maintained by the ribonucleoprotein telomerase, a complex of a reverse transcriptase encoded by two core components: Tert (telomerase reverse transcriptase) and template RNA Terc (essential RNA component), stabilized by dyskerin 8. Telomerase and telomeres are important for proliferation of ESCs and other types of stem cells 9, 10. Telomerase activity decreases during cell differentiation of fetal and adult development 11, 12. Telomere length counts the number of times the cells have divided, and predicts replicative capacity 13. Reduced telomerase activity in most somatic cells eventually leads to telomere shortening, cellular senescence or replicative aging 14, 15. Telomere length also can be regulated by alternative lengthening of telomeres (ALT), in association with epigenetic modification at subtelomeres by DNA methytransferases (Dnmt3a and Dnmt3b) and histone methylation by Suv39h1 and Suv39h2 16, 17, 18, and Zscan4-mediated telomere recombination specifically in ESCs 19.
Remarkably, telomeres of somatic cells are reprogrammed to elongate in at least some of iPSC lines, which acquire telomere length similar to that of ESCs 20, 21, 22, 23, 24, and iPSCs also exhibit high telomerase activity like ESCs 25, 26. However, variations in telomere lengths have also been found in iPSCs 20, 21, 22, 23, 24, 27, 28. While human iPSCs exhibit elongated telomeres at early passages (P5) 23, prematurely aged (shortened) telomeres also are a common feature of human iPSCs, coincident with reduced telomerase activity during passages 22. Telomere shortening inevitably would limit the scalability and safety, and thus therapeutic efficacy in the potential application of iPSCs. We attempted to understand mechanisms of telomere elongation and length variations during induction and clonal expansion of iPSCs, and explored telomerase-independent mechanisms possibly involved in telomere elongation and genomic stability of iPSCs, using isogenic ESCs as controls, and by taking advantages of the first generation (G1) telomerase-deficient (Terc−/−) and telomerase haplo-insufficient (Terc+/−) mouse cells.
Results
Activation of telomerase genes during iPSC induction
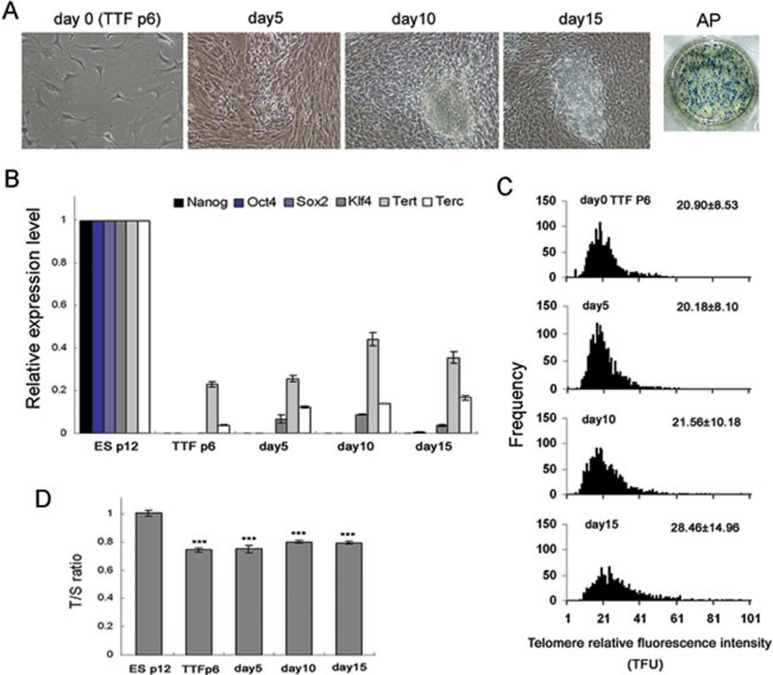
Primary iPSC colonies consistently formed from day 10 to 15 after transfection of tail-tip fibroblasts (TTF) with Yamanaka factors Oct4, Sox2, Klf4 and c-Myc. The primary colonies exhibited positive alkaline phosphatase (AP) activity (Figure 1A). Prior to induction of iPSCs, TTF (passage 6) at day 0 expressed very low levels of the two major components of telomerase, Tert and Terc (Supplementary information, Figure S1), compared to ESCs of the same genetic background C57BL/6J (N3329). During the reprogramming of TTF, expression of Tert and particularly Terc gradually increased from day 5 to 15, though at a lower level than that of ESCs, while the endogenous pluripotent genes Nanog and Sox2 did not exhibit noticeable expression, and Klf4 was expressed at low levels from day 5 (Figure 1B). Expression of Oct4 was increased by day 15 compared with progenitor fibroblasts at day 0, but at levels considerably lower than those of ESCs. Telomeres mildly elongated from day 10 to 15 when primary iPSC-like colonies formed (Figure 1C), as estimated by the telomere quantitative fluorescent in situ hybridization (QFISH) assay. This was confirmed by the real-time PCR assay, but the telomere lengths (T/S ratio) were significantly shorter than in ESCs (Figure 1D). These data show that exogenous expression of the four Yamanaka factors activates Tert and Terc earlier than endogenous pluripotent genes. Activation of telomerase could be important for proliferation and telomere elongation of primary iPSC colonies.
Figure 1.
Dynamics of expression of telomerase genes and telomeres during iPSC induction. (A) Morphology of TTF and primary iPSC clones following transfection of the four Yamanaka factors. AP, alkaline phosphatase activity. (B) Expression of Tert and Terc and endogenous genes. ESCs at P12 served as controls were arbitrarily designated as 1, and others were compared with ESCs. (C) Histogram shows distribution of relative telomere length expressed as TFU measured by QFISH method. Medium telomere length is shown as Mean TFU ± SD. (D) Relative telomere length shown as T/S ratio measured by quantitative real-time PCR. ***P < 0.001, compared to ES controls.
Telomere elongation during passages of iPSCs
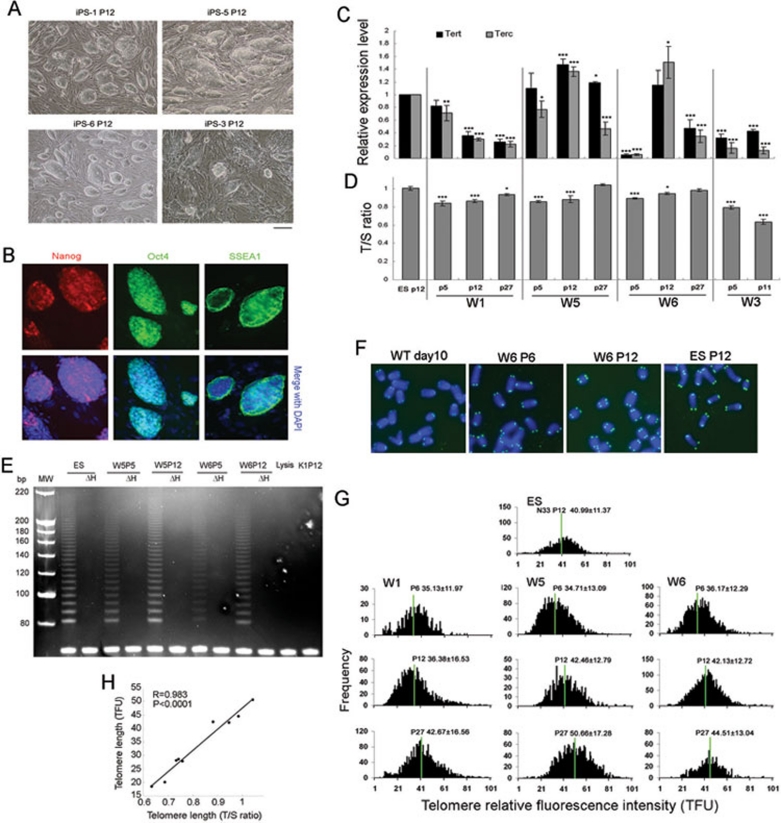
Continuous passages of iPSC-like primary colonies led to stable iPSC lines resembling typical ESC colonies in morphology, with large nuclei and nucleoli and clear compact clonal boundaries, distinct from feeder fibroblasts (Figure 2A). These iPSC clones expressed common ESC markers, SSEA1 on the cell surface, and Oct4 and Nanog in the nuclei (Figure 2B). However, iPSCs proliferated differently and some iPSC lines, e.g. iPSC-3 (also named as wild-type iPSC-3 or W3) proliferated slowly after passages, and formed fewer clones by P12 (Figure 2A). We asked whether these differences in proliferation were associated with limited activity of telomerase and telomere function. Indeed, Tert and particularly Terc were expressed at very low levels in W3 iPSCs at early-to-mid-passages, in contrast to their higher expression in ESCs at P12, and iPSC W5 and W6 (Figure 2C). Interestingly, W1 iPSCs at P5 expressed Tert comparable to ESCs but lower levels of Terc, and expression of Tert and Terc was reduced at P12 and P27. Moreover, W5 or W6 iPSCs at P12 expressed higher levels of Tert and Terc than those of ESCs. However, Terc expression decreased over more passages by P27. These data show that telomerase expression is unstable in various iPSC lines and reduced especially by late passages. Reduced expression of both Tert and Terc was associated with telomere shortening in iPSC W3 (Figure 2D), which could not be passaged further due to significantly reduced cell proliferation. No colonies were available for telomere QFISH analysis after P12. Expression levels of Tert and Terc correlated with telomerase activity (Figure 2E). Increased telomerase activity in iPSCs W5 and W6 coincided with telomere elongation at P12, while reduced telomerase activity in iPSC W1 corresponded to minimal telomere length change (Figure 2D, 2F-2H). Interestingly, variability in telomerase component activation and telomere lengths was not specific to iPSC cell clones during passages, but also was found in ES cells (Supplementary information, Figure S1B).
Figure 2.
Telomerase activity and maintenance of telomeres during passages of iPSCs. (A) Morphology under bright field with phase contrast optics of various iPSC clones at P12. Bar = 100 μm. (B) Expression of ESC markers Oct4, Nanog and SSEA1 by immunofluorescence microscopy. Nuclei stained with DAPI (blue). (C) Expression of Tert and Terc in various iPSC lines (W1, W5, W6 and W3) at various passages (P), compared to ESCs as controls (arbitrarily designated as 1). *P < 0.05; **P < 0.01; ***P < 0.001. (D) Relative telomere length expressed as T/S ratio by qPCR. (E) Telomerase activity by TRAP assay. ΔH, heat-inactivated. White bands at the bottom of the gel indicate internal loading control. Lysis buffer and K1P12 (Terc−/− cells) both served as negative controls. (F) Telomere FISH images of partial chromosome spreads from iPSCs compared with ESCs and cells at day 10 during iPS induction. WT, wild type. Blue, DAPI-stained chromosomes; Green dots, telomeres; P, passage. (G) Histogram shows distribution of relative telomere length shown as TFU by QFISH. The medium telomere length (green bars) also is shown as Mean ± SD above each panel. (H) Relative telomere length shown as T/S ratio is closely correlated (P < 0.0001) with relative telomere length expressed as TFU estimated by QFISH method.
Telomerase is dispensable for iPSC induction but required for telomere maintenance during passages of iPSCs
The above data suggest that telomerase activation may not be required for induction and maintenance of iPSCs at least for early passages. To test whether telomerase is essential for iPSC induction and/or telomere shortening affects iPSC induction, we isolated TTF from the first generation (G1) telomerase RNA null (Terc−/−) mice with a pure C57BL/6 background 30 to generate iPSCs, using the same method for derivation of WT iPSCs described above. Terc−/− (KO) TTF generated iPSC primary colonies at high efficiency, with characteristic morphology and AP activity similar to iPSCs from WT TTF (Supplementary information, Figure S2A; 3.43% ± 0.46% versus 3.68% ± 0.34%, P > 0.05). As expected, no Terc expression was observed. Tert showed increased expression, but at significantly lower levels than in ESCs. Klf4 also was expressed, similar to that of WT TTF, despite minimal expression of endogenous Nanog, Oct4 and Sox2 (Supplementary information, Figure S2B). Even in the absence of telomerase, telomere lengths remained stable or even slightly increased during iPSC induction and clonal formation of iPSC-like cells (Supplementary information, Figure S2C and S2D). Moreover, telomere signal-free ends, indicative of telomere loss in the progenitor Terc−/− TTF, were substantially reduced during induction of iPSCs. Telomeres were much shorter in Terc−/− TTF and in formed primary iPSC colonies, compared with ESCs (Supplementary information, Figure S2D). By contrast, WT and Terc−/− TTF without being reprogrammed by transfection of the four factors showed signs consistent with senescence over culture for 15 days, and their telomere lengths decreased slightly (Supplementary information, Figure S2E and S2F), suggesting that telomeres are being maintained by some mechanisms specific to the process of reprogramming.
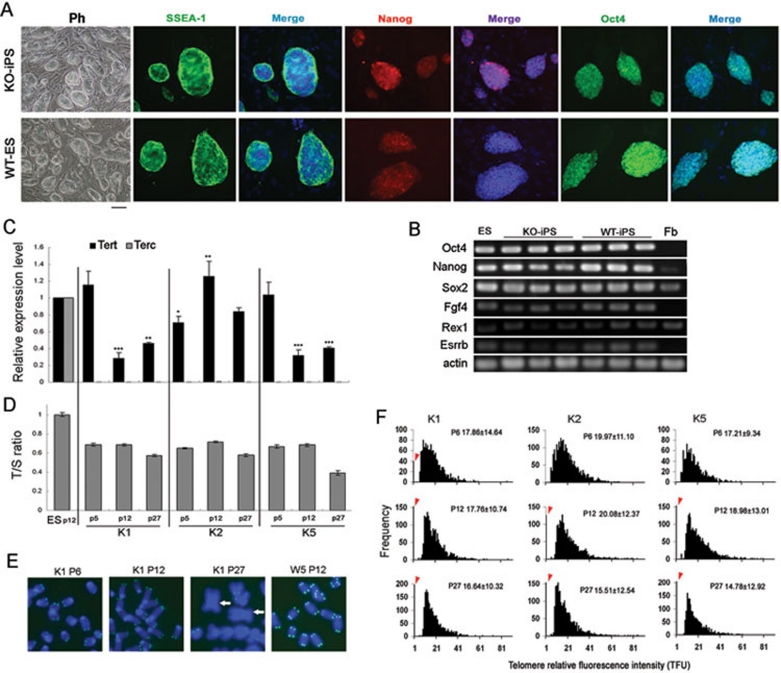
Despite short telomeres in primary Terc−/− iPSC-like colonies, Terc−/− iPSCs showed morphology typical of ESCs, and expressed Oct4, Nanog, SSEA1, like WT iPSCs and ESCs even after several passages (Figure 3A and 3B). Tert was expressed at high levels during early passage (P5), but declined after additional passages (from P12 to P27) in K1 and K5 Terc−/− iPSCs. K2 Terc−/− iPSCs expressed higher levels of Tert from early to late passages. Expectedly, no Terc was found in any of the three Terc−/− iPSC lines studied (Figure 3C), and the telomerase activity was undetectable in Terc−/− iPSCs (K1 shown here) despite the expression of Tert (Figure 2E).
Figure 3.
Telomere shortening during passages of Terc−/− iPSCs in the absence of telomerase. (A) Clonal morphology by phase contrast optics (Ph) of KO (Terc−/−) iPSC lines at P12 (Bar = 100 μm), which also express ESC markers Oct4, Nanog and SSEA1 by immunofluorescence. Nuclei stained with DAPI (blue). ES cells from the same genetic background (C57BL/6) of wild-type (WT) mice served as controls. (B) Expression of pluripotent genes by conventional RT-PCR analysis of KO (Terc−/−) iPSC lines, compared with ESCs and WT iPSC lines. Fb, fibroblasts. (C) Expression of Tert and Terc in various Terc−/− iPSC lines (K1, K2 and K5) at various passages (P), compared to ESCs. *P < 0.05; **P < 0.01; ***P < 0.001. (D) Relative telomere length of various iPSC lines at various passages shown as T/S ratio by qPCR. (E) Telomere FISH images of partial chromosome spreads from Terc−/− iPSCs K1 compared with WT iPSCs W5. Blue, DAPI-stained chromosomes; Green dots, telomeres. White arrows indicate telomere loss/chromosome fusion. (F) Histogram shows distribution of relative telomere length as TFU by QFISH. Heavy black bars on Y-axis indicated by red arrowheads show frequency of telomere signal-free ends. Medium telomere length is shown as Mean TFU ± SD.
Telomere lengths were maintained during early passages of Terc−/− iPSCs, but shortened (about 4.5 telomere fluorescence intensity unit (TFU) for K2 and K5 Terc−/− iPSCs) over more passages (P12-P27) (Figures 3D-3F). Some Terc−/− iPSCs (K2 and K5) exhibited normal karyotypes at early passages, but telomere loss and chromosome fusion were increased in Terc−/− iPSCs following additional passages (Figure 3E and 3F, Supplementary information, Tables S1 and S2). Thus, telomerase is essential for self-renewal, telomere maintenance and chromosomal stability of iPSCs during long-term passages, although telomerase deficiency does not prevent induction of iPSCs, and telomerase-deficient iPSCs exhibit ESCs-like morphology and express Oct4, Nanog and SSEA1 markers routinely used for characterization of ESCs/iPSCs.
Haploinsufficiency of telomerase limits telomere elongation of iPSCs
To associate the levels of telomerase gene expression with telomere lengths following passages, we generated iPSCs from Terc+/− TTF and assessed their telomere length and genomic stability. Terc showed markedly reduced expression, but Tert of various Terc+/− iPSCs (H1, H2 and H3) was expressed at high levels, compared with ESCs (Supplementary information, Figure S3A). Telomere lengths of Terc+/− iPSCs elongated (about 5-7 TFU) from passage 6 to 22, but were shorter than those of WT iPSCs and ESCs (Supplementary information, Figure S3B and S3C), indicating reduced elongation of telomeres in Terc+/− iPSCs.
Telomerase-independent mechanism for telomere elongation of iPSCs
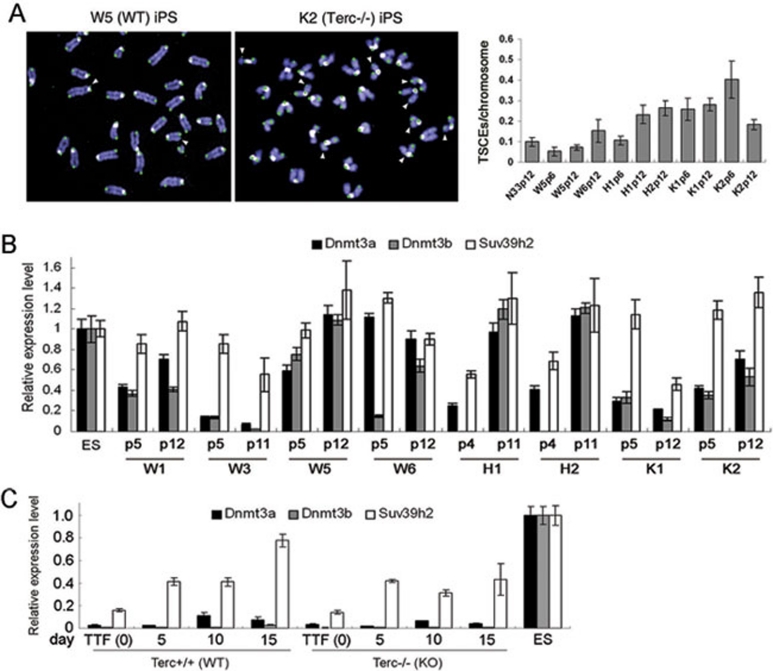
In the absence of telomerase activity, telomeres are expectedly shortened in cultured TTFs, prior to iPSC induction and in subsequent passages of iPSCs. Telomerase haploinsufficiency also could not elongate telomeres sufficiently in iPSCs. Yet, telomeres were maintained or even elongated to some extents in Terc−/− or Terc+/− iPSCs. Further, although telomerase Terc was reduced by later passage (e.g., P27), telomeres of WT iPSCs actually elongated (Figure 2C, 2D and 2G). These data suggest that telomerase-independent mechanisms might compensate for telomere attrition during long-term passages. We initially analyzed in iPSCs the telomere sister chromatid exchange (TSCE) as a recombination-based mechanism 31, 32, 33. Both WT ESCs and iPSCs exhibited low rates of TSCE. By contrast, rates of TSCE were elevated in general in Terc−/− (K1 and K2) and Terc+/− (H1 and H2) iPSCs at various passages, compared to WT iPSCs (Figure 4A), perhaps suggesting that the Terc+/− and Terc−/− iPSCs display higher levels of recombination based on selection or adaptation. Increased rates of TSCE were associated with shorter telomeres in those iPSCs. Consistently, TSCEs also were significantly increased in mTert−/− ESCs possessing critically short telomeres 9; moreover, short telomeres initiate telomere recombination 34. Telomere recombination generally requires relaxed chromatin structures and epigenetic modifications, such as reduced DNA methylation by reduced DNA methyltransferases (Dnmts) 17, 18. Compared to ESCs at P12, WT iPSCs W6 and W5 at P12 exhibited similar expression of Dnmt3a and Dnmt3b, but W3 showed much reduced expression of Dnmt3a and Dnmt3b. Notably, Terc+/− (H1 and H2 at P4) iPSCs and Terc−/− iPSCs at early passages (K1 and K2 at P5 and P12) expressed markedly reduced Dnmt3a and Dnmt3b (Figure 4B). Terc+/− (H1 and H2 at P11) iPSCs expressed Dnmt3a and Dnmt3b at levels similar to those of WT iPSCs and ESCs at P12, yet showed higher rates of TSCEs than WT iPSCs, suggesting that mechanisms other than DNA methylation also might be involved in TSCEs for lengthening of telomeres, which await further investigation. Moreover, expression levels of Dnmt3a and Dnmt3b, as well as Suv39h2, were much lower during induction of iPSCs than in ESCs, regardless of telomerase status (Figure 4C).
Figure 4.
Telomerase-independent telomere maintenance of iPSCs. (A) Representative CO-FISH images showing TSCEs (TSCEs, indicated by white arrowheads) and frequency of TSCE in various iPSCs. (B) qPCR analysis of expression levels of Dnmts (Dnmt3a and Dnmt3b) and histone methyltransferase (Suv39h2) in WT W1, W3, W5 and W6 iPSCs, Terc+/− H1 and H2 iPSCs and Terc−/− K1 and K2 iPSCs. (C) Relative expression of Dnmt3a, Dnmt3b and Suv39h2 in WT and Terc−/− TTFs during induction of iPSC clonal formation.
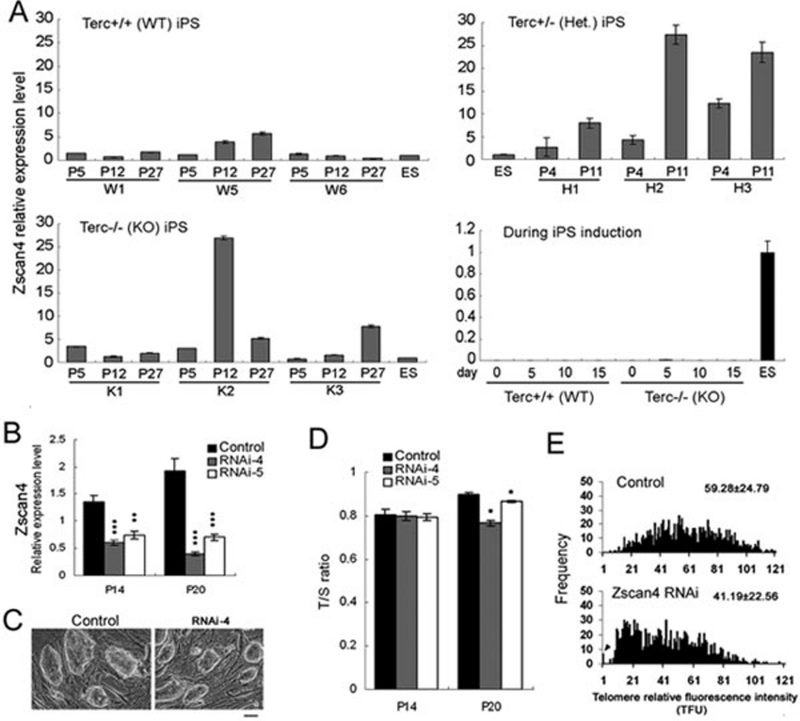
Recently, a specific gene, Zscan4, has been shown to be involved in telomere recombination and elongation in ESCs 19. We show that Zscan4 was highly expressed in Terc+/− iPSCs (H2 and H3) and also in Terc−/− iPSCs K2, and slightly increased in K3 compared to the WT iPSCs W5 and W6, and ESCs (Figure 5A). In contrast, expression of Zscan4 was quite low during induction of iPSCs. To determine whether Zscan4 was involved in telomere lengthening of iPSCs, we employed small hairpin RNA (shRNA) method to knock down Zscan4 mRNA levels and estimated their telomere length. Indeed, Zscan4 RNAi caused telomere shortening during passages of iPSCs. Telomeres elongated over passages of control iPSCs in association with increased Zscan4 expression (Figure 5B-5D), whereas iPSCs with reduced Zscan4 by shRNA exhibited telomere shortening and loss with increased passages (Figure 5D and 5E). The combined evidence of TSCE, alteration of DNA methylation, and/or Zscan4 expression and knockdown data supports the notion that iPSCs also likely utilize ALT, presumably involving telomere recombination to elongate telomeres. Moreover, increased alternative telomere lengthening may contribute to the reduced telomere shortening in Terc−/− iPSCs and partial telomere elongation of Terc+/− iPSCs. Collectively, the above results suggest that telomerase-independent mechanisms of telomere lengthening may operate in WT iPSCs as an adjunct to telomerase activity and can be over-activated during telomere dysfunction.
Figure 5.
Zscan4 is involved in telomere elongation of iPSCs. (A) Relative expression of Zscan4 determined by qPCR analysis in various iPSC lines and during induction of iPSC clonal formation. (B) Reduced Zscan4 mRNA by shRNA knockdown of W5 iPSCs at immediate passage (P14) and more passages (P20). *P < 0.05; **P < 0.01; ***P < 0.001, compared with control iPSCs. (C) Morphology of iPSC clones after Zscan4 knockdown. Bar = 50 μm. (D) Relative telomere length expressed as T/S ratio by qPCR. (E) Histogram shows distribution of relative telomere length of iPSCs at P20 by Zscan4 knockdown expressed as TFU measured by QFISH. Heavy black bar on Y-axis indicated by arrowheads show frequency of telomere signal-free ends, indicative of telomere loss. Medium telomere length is shown as Mean TFU ± SD on the upper right hand corner.
Fully reprogrammed telomere length is critical for iPSC pluripotency
In association with telomere reprogramming of iPSCs, endogenous genes Oct4, Sox2, Klf4 and c-Myc were usually expressed at high levels comparable to ESCs, but at lower levels in some iPSCs at early passages (e.g., Oct4 and Klf4 in WT iPSC-6 and K2 iPSC at P5). Yet, Terc−/− K1 iPSCs at P12 expressed very low levels of both Oct4 and Sox2 (Supplementary information, Figure S4A). Surprisingly, exogenous Oct4, Klf4 or c-Myc were reactivated in Terc−/− iPSCs (K1, K2 and K5), while all four exogenous genes remained silenced in WT iPSCs (Supplementary information, Figure S4B). Particularly, the levels for exogenous Klf4 and Myc were significantly increased in some iPSCs (Supplementary information, Figure S5). Some Terc+/− iPSCs exhibited lower expression of endogenous Oct4 and reactivation of some exogenous genes (e.g., Klf4) (Supplementary information, Figure S4C). Telomerase-deficient iPSCs at early passages showed the potential to differentiate into three embryonic layer cells by induction of embryoid body formation, a standard in vitrodifferentiation test. However, the differentiation potential was significantly reduced at late passages compared to early passsges (Supplementary information, Figure S6). Telomerase-deficient iPSCs could be induced with high efficiency and expressed the typical ESC markers (Figure 3), and also could differentiate into three embryonic germ layers by the teratoma test (Supplementary information, Figure S7A).
To test the authentic pluripotency, we randomly selected several iPSC lines with various telomere lengths, injected them into eight-cell embryos and obtained chimeras and complete iPSC pups from WT iPSC W6 and W5 with pure black coats (Supplementary information, Figure S7B), consistent with iPSCs of C57BL/6 origin and also confirmed by microsatellite genotyping. WT iPSCs W5 and W6 with sufficient telomere length produced pups (40%-70%) with black or black and albino coat. Terc+/− iPSCs with intermediate telomere length generated colored pups at reduced rate (17%-33%, Supplementary information, Table S3). Terc−/− iPSCs with shortened telomeres almost failed to generate pups with colored coat. Likewise, WT iPSCs W3 with shortened telomeres over extended passages also produced no chimeras (0%). Thus, iPSCs with longer telomeres generated chimeras with higher efficiency than iPSCs with shorter telomeres. Contributions of iPSCs to various tissues or organs of chimeras were confirmed by microsatellite genotyping (Supplementary information, Figure S7C, Tables S5 and S6). Linear regression analysis showed a clear correlation between telomere length and rates of chimeras generated from iPSCs (Supplementary information, Figure S7D and Table S4). These data suggest that telomere length and function might indicate the extent to which the iPSCs are reprogrammed to true pluripotency.
Discussion
We report that telomere lengths and stability of iPSCs are influenced by activation of telomerase genes, passage numbers, appropriate silencing of exogenous genes and telomerase-independent mechanism. Reprogramming of telomerase and telomeres during induction of iPSCs occurs gradually, but prior to activation of endogenous pluripotent genes. Notably, Tert and Terc are activated prior to Nanog, Oct4 and Sox2 in the iPSC-like colonies with AP activity. Klf4 is upregulated relative to progenitor TTFs, coincident with activation of Tert in WT and Terc−/− iPSCs. Klf4 might directly activate Tert 35. c-Myc is dispensable for telomerase activation in mouse iPSCs 20. Exogenously expressed Oct4 can activate Terc 21. Also, Tert expression is activated earlier than Terc during induction of adult TTFs. The dynamics of Tert and Terc activation may depend on the cell types used for induction of iPSCs as well as the method for selection. Fibroblasts isolated from tail-tip biopsies of newborn mice and reprogrammed with a tet-inducible lentivirus called LV-tetO system show faster dynamics, as evidenced by earlier activation of Oct4, Sox2 and Tert 26. Basal levels of Tert expression in MEFs are higher than those in adult TTFs (Supplementary information, Figure S1). Further studies are required to determine whether this contributes to earlier activation of Tert and other pluripotent factors in the derivation of iPSCs from MEFs compared to adult fibroblasts.
TTFs from Terc−/− mice or Terc+/− mice generated iPSC lines with high efficiency, at rates similar to WT TTFs, indicating that telomerase is not essential for the induction of iPSCs. It is also noteworthy that telomeres undergo mild elongation, late in primary WT iPSC-like colonies, when telomerase is only mildly activated. Telomeres are maintained or even slightly elongated in primary Terc−/− iPSC-like colonies at day 15 when otherwise they would expectedly have shortened in the absence of telomerase. Telomere maintenance during iPSC induction presumably results from some telomerase-independent mechanisms involving chromatin remodeling initiated by reprogramming factors 36, as shown by reduced expression of Dnmts3a and 3b and Suv39h2, which also is important for global demethylation and reprogramming required for iPSC induction.
Telomere length differs in various iPSCs; while some exhibit telomere lengthening, others show shorter telomeres over more passages, consistent with previous reports 20, 21, 22, 23, 24. Moreover, some WT iPSCs fail to elongate telomeres, in association with inadequate activation of telomerase genes, particularly at early passages, despite their expression of Oct4 and Nanog and the ability to form teratomas. When telomerase genes are insufficiently activated, telomeres elongate less even over extended passages. Moreover, telomeres are shorter in Terc+/− iPSCs with haploinsufficient telomerase than in WT iPSCs, and even more shortened in Terc−/− iPSCs without telomerase. However, telomere loss is found in Terc−/− iPSCs but not in Terc+/− iPSCs. This is consistent with earlier findings in ESCs heterozygous for mTert on dosage-dependent function of telomerase in average telomere-length maintenance and the selective maintenance of critically short telomeres 37. These data are consistent with the notion that activated telomerase is required for telomere maintenance and self-renewal of iPSCs.
Besides telomerase activity, passage number also explains heterogeneity of telomere reprogramming and lengths, and pluripotency of various iPSC lines. The number of cell divisions drives epigenetic reprogramming to pluripotency 38; not surprisingly, sufficient cell divisions are required for telomere elongation during propagation of iPSCs, as shown here and in previous reports 20, 21. Interestingly, early-passage iPSCs retain a transient epigenetic memory of their somatic cells of origin, which manifests as differential gene expression and altered differentiation capacity, but continuous passaging of iPSCs largely attenuates these differences 39. Extended culture and passaging of human and mouse iPSCs enhance reprogramming of global epigenomes and transcriptomes, to approximate ESCs 39, 40, 41, consistent with the observation that continuous telomere reprogramming and lengthening occur during propagation of iPSCs.
Retroviral silencing appears to be important for full reprogramming of somatic cells into iPSCs 26. Appropriate expression of the de novomethyltransferases Dnmt3a and Dnmt3b might be required for different stages of retroviral silencing. It appears that there is a trend toward higher levels of exogenous transgene reactivation in the telomerase-deficient iPSCs (Supplementary information, Figure S5). The amount of exogenous transcript for Oct4 and Sox2 is very little compared to that expressed endogenously. For Klf4 and Myc, however, the exogenous levels may be significant in some iPSCs. Klf4 reverses developmentally programmed restriction of ground state pluripotency 42. Reactivation of exogenous genes, particularly Klf4, in telomerase-deficient or haploinsufficient iPSCs might partially compensate for reduced self-renewal of iPSCs that otherwise would result due to insufficient telomerase and telomeres. Also, Klf4 and c-Myc have been shown to activate telomerase expression 35, 43, 44. Reactivation of transgenes Klf4 and c-Myc is associated with relatively elevated levels of Tert in some of Terc+/− and Terc−/− iPSCs. Interestingly, reactivation of exogenous transcription factors in telomerase-deficient iPSCs and telomerase haploinsufficient iPSCs at early passages coincides with reduced expression levels of Dnmt3a or Dnmt3b and increased rates of TSCE, indicative of a telomerase-independent mechanism that likely contributes to telomere maintenance despite to limited extent.
Although activation of telomerase is important for telomere reprogramming, telomerase-independent mechanisms also may underlie telomere elongation of iPSCs, as evidenced by maintenance of telomere length in Terc−/− iPSCs and moderate telomere lengthening of Terc+/− iPSCs compared to their progenitor TTFs, coincident with increased frequency of TSCE, downregulation of Dnmts and upregulation of Zscan4, which regulates telomere elongation by recombination in ESCs 19. Indeed, reducing Zscan4 expression by RNAi results in telomere shortening of iPSCs. Also, reduced expression of Dnmt3a and Dnmt3b that leads to reduced DNA methylation promotes telomere elongation by recombination 18, which could be indirectly marked by TSCE 31, 32. Consistently, epigenetic reprogramming and DNA demethylation are required for induction of iPSCs, and DNA methyltransferase inhibitors can improve the efficiency of the reprogramming process 36, 45. iPSCs with haploinsufficiency of telomerase (Terc+/−) show an increase in mean telomere length between P6 and P22, which coincides with increased heterogeneity (Supplementary information, Figure S3B), yet have much reduced expression levels of Dnmt3a, Dnmt3b and Suv39h2 (Figure 4B) at early passage and increased TSCE (Figure 4A) at early-to-mid-passage, likely suggesting that recombination appears to be a response to the lack of active telomerase. However, excess telomere recombination by TSCE in telomerase-deficient cells also can cause accelerated cellular senescence 46. Aberrant frequent telomere sister chromatid recombination, telomere loss and instability, together with insufficient reprogramming of iPSCs due to telomerase deficiency and abnormal epigenetic changes eventually lead to cell senescence as seen in Terc−/− iPSCs at late passages.
Together, telomerase is essential but not sufficient, while telomere length is critical for chromosomal stability and pluripotency of iPSCs. Appropriate ALT, involving epigenetic modifications and telomere recombination, also contributes to telomere function of iPSCs. Direct reprogramming of somatic cells to authentic pluripotent stem cells by overexpression of transcription factors is a gradual process that achieves partial reprogramming at very early passages, but iPSCs become fully reprogrammed over passages. Telomere dysfunction and chromosomal instability reduce self-renewal and limit developmental pluripotency of iPSCs. Selection of iPSCs with fully reprogrammed telomeres could have implications in large-scale production of the pluripotent stem cells for therapeutic applications.
Materials and Methods
iPSC generation
Derivation of iPSCs from fibroblasts of adult mouse tail-tips (TTFs) has been described previously 29. iPSCs were induced by transduction with four Yamanaka factors using standard protocol 3, without using selection markers. Reprogrammed pluripotent cells can be isolated from genetically unmodified somatic donor cells solely based upon morphological criteria 47. The day before transduction, Plat-E cells were seeded at 5 × 106 cells per 100 mm dish. Next day, pMXs-based retroviral vectors (pMXs-Sox2, Klf4, Oct4 and c-Myc) were introduced into Plat-E cells using lipo-2000 transfection reagents. The cells were replated in 10 ml ESC medium containing knockout Dulbecco's modified Eagle medium added with 20% fetal bovine serum, 1 000 U/ml LIF, 0.1 mM β-mercaptoethanol, 1 mM L-glutamine, 0.1 mM nonessential amino acids, 100 units/ml penicillin and 100 μg/ml streptomycin. Three days after infection, the cells were passaged on MEF feeders and the medium was changed every day. ESC-like colonies were picked and passaged using standard protocols. We chose and collected only cultures that show clear cell aggregates and colonies during reprogramming for analysis of transcripts and telomeres.
Telomerase activity assay
Telomerase activity was determined by the Stretch PCR method according to manufacturer's instruction using TeloChaser Telomerase assay kit (TLK-101, TOYOBO, Osaka, Japan). About 2.5 × 104 cells from each sample were lysed, and lysed cells heated at 70 °C for 10 min served as negative control. PCR products of cell lysates were separated on nondenaturing TBE-based 10% polyacrylamide gel electrophoresis and visualized by ethidium bromide staining.
Telomere QFISH
Telomere length and function (telomere integrity and chromosome stability) was estimated by telomere QFISH 29, 30. Telomeres were denatured at 80 °C and hybridized with telomere PNA probe (0.5 μg/ml) (Panagene, Korea). Chromosomes were stained with 0.5 μg/ml 4',6-diamidino-2-phenylindole (DAPI). Fluorescence from chromosomes and telomeres was digitally imaged on a Zeiss microscope with fluorescein isothiocyanate (FITC)/DAPI filters, using AxioCam and AxioVision software 4.6. Telomere length shown as telomere fluorescence intensity was integrated using the TFL-TELO program (a gift kindly provided by P Lansdorp, Terry Fox Laboratory), and calibrated using standard fluorescence beads.
CO-FISH (Chromatid orientation-FISH)
Strand-specific CO-FISH was performed according to Bailey et al. 31, with minor modification. Subconfluent cell monolayers were incubated with 5'-bromo-2'-deoxyuridine (BrdU). Nocodazole was added for 1 h prior to cell harvest, and metaphase spreads were prepared by standard cytogenetic method as described above. Chromosome preparations were stained with Hoechst 33258 (0.5 μg/ml), incubated in 2× SSC (Invitrogen) for 20 min at room temerature and exposed to 365 nm UV light (Stratalinker 1800 UV irradiator) for 40 min. The BrdU-substituted DNA was digested with Exonuclease III (Promega). The slides were then dehydrated through cold ethanol series and air-dried. PNA-FISH was performed with Fluorescein-OO-(CCCTAA)3 (Bio-Synthesis Inc, Texas, USA). Slides were hybridized, washed, dehydrated, mounted and counterstained with Vectashield antifade medium (Vector) containing 0.1 μg/ml DAPI. Digital images were captured using a CCD camera on a Zeiss Axio-Imager Z1 (Zeiss, Germany) microscope equipped with a Metasystems Isis Software.
Zscan4 RNAi
For stable knockdown of Zscan4, shRNA sequence 19 was synthesized and cloned into pSIREN-retroQ, according to manufacturer's instructions. The negative control shRNA without sequence homology to mouse genes served as a negative control. Cells (2 × 105) were transfected with 2 μg plasmid using lipofectamineTM 2000 (Invitrogen) and selected by 1.5 μg/ml puromycin, and clones were picked.
Immunofluorescence microscopy
Cells were washed twice in phosphate-buffered saline (PBS), then fixed in freshly prepared 3.7% paraformaldehyde in PBS (pH 7.4), permeabilized in 0.1% Triton X-100 in blocking solution (3% goat serum in PBS) for 30 min, washed and left in blocking solution for 1 h. Cells were incubated overnight at 4 °C with primary antibodies against Oct4 (sc5279, Santa Cruz, CA, USA), Nanog (Abcam, ab10626) and SSEA-1 (DSHB, MC-480), washed and incubated for 1 h with secondary antibodies, Texas red conjugated anti-mouse IgG (Vector, TI-2000, CA, USA), or FITC-labeled goat anti-rabbit IgG (BD Biosciences Pharmingen) or Alexa fluor 488 goat anti-mouse IgM (Molecular Probes, Invitrogen) diluted with blocking solution. Samples were counterstained with 0.5 μg/ml DAPI in Vectashield mounting medium. Fluorescence was imaged using a Zeiss or Leica fluorescence microscope. AP assay was performed using the Vector blue kit from Vector Laboratories (DAKO, Carpinteria, CA, USA).
iPSC mouse and chimera generation
Microinjection of ESCs into four- or eight-cell embryos facilitated by a Piezo injector can generate not only germline competent chimeras, but also complete ESC mice with high efficiency 48, thus providing an attractive one-step assay alternative to both the TEC method and injection of diploid blastocysts to test developmental pluripotency 29. Approximately, 10-15 iPSCs were injected into eight-cell embryos as hosts from a different genetic background using a Piezo injector as described 48. Injected embryos were cultured overnight in KSOMAA medium and developed blastocysts were transferred into uterine horns of 2.5 dpc surrogate mice. Pregnant females delivered pups naturally at about 19.5 dpc. Pups were identified initially by coat color. The contribution of iPSCs to various tissues in chimeras was confirmed by standard DNA microsatellite genotyping analysis 48, using specific D12Mit136 primer (http://www.jaxmice.jax.org/strain/004132.html) (Supplementary information, Table S5).
Teratoma test
Approximately 2 × 106 cells were injected subcutaneously into 4-week-old immunodeficient nude mice. At 1 month after injection, the mice were humanely sacrificed, the teratomas excised, fixed in 10% neutral-buffered formalin, washed in 70% ethanol, embedded in paraffin and sectioned for histological examination by haematoxylin and eosin staining.
Gene expression by real-time PCR
For gene expression analysis during iPSC induction, the cells at day 5, 10 and 15 were cultured without feeder in normal ES medium without affecting iPSC induction 49. iPSCs were removed off feeder cells twice based on their differences in the adherence to the bottom of dish, and relatively pure iPSCs were used for gene expression analysis or telomere measurement. Total RNA was isolated using RNeasy mini kit (Qiagen). RNA was subjected to cDNA synthesis using M-MLV Reverse Transcriptase (Invitrogen). PCR reaction was set up in duplicates using the FastStart Universal SYBR Green Master (ROX, Roche) and run on the iCycler real-time PCR detection system (Bio-Rad). Each sample was repeated three times and analyzed with glyceraldehyde 3-phosphate dehydrogenase as the internal control. Most primers (Supplementary information, Table S5) were designed using IDT DNA website or Oligo6 software.
Statistics
Data were analyzed by analysis of variance and means compared by Fisher's protected least-significant difference using the StatView software from SAS Institute Inc. (Cary, NC). Linear relation analysis was performed using SigmaPlot 8.0. Significant differences were defined as P < 0.05, 0.01 or lower.
Acknowledgments
We thank Dr Susan Bailey's lab for help and advice on CO-FISH experiments. This work was supported by MOST National Major Basic Research Program (2009CB941000), the Ministry of Agriculture of China Transgenic Special Program (2009ZX08006-010B) and NIH 2 R01 DK054369 (XP and HZ).
Footnotes
(Supplementary information is linked to the online version of the paper on the Cell Research website.)
Supplementary Information
(A) Expression of Tert and Terc by qPCR analysis of N33 (ESCs), MEF(mouse embryonic fibroblasts), WTfb (Tail-tip fibroblasts (TTF) from adult wild-type mice at the age of 8 weeks, and Kofb (Tail-tips fibroblasts from adult telomerase Terc deficient mice).
Dynamics of telomerase and telomeres during induction of iPSCs from telomerase Terc knockout (KO, Terc−/−) TTF.
Telomerase activity and maintenance of telomeres during passages of telomerase haplo-insufficient heterozygous (Het., Terc+/−) iPSCs.
Expression of endogenous and exogenous genes of iPSCs determined by real-time PCR analysis.
Relative amount of exogenous transcript as a proportion of total transcripts in iPSCs determined by real-time PCR analysis.
Differentiation tests in vitro of Terc-deficient iPSCs at early and late passages.
Association of telomere shortening and dysfunction with reduced developmental pluripotency of iPSCs.
Karyotyping of iPSC lines
Telomere loss and chromosome fusion of iPSCs at early passage (P6)
Chimeras of iPSCs generated by injection into 8-cell albino ICR embryos
Comparison of telomere length and efficiency (%) of chimeras
Primers for PCR analysis
Ratio of contribution of iPSCs in the chimeras by microsatellite analysis
References
- Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126:663–676. doi: 10.1016/j.cell.2006.07.024. [DOI] [PubMed] [Google Scholar]
- Wernig M, Meissner A, Foreman R, et al. In vitro reprogramming of fibroblasts into a pluripotent ES-cell-like state. Nature. 2007;448:318–324. doi: 10.1038/nature05944. [DOI] [PubMed] [Google Scholar]
- Okita K, Ichisaka T, Yamanaka S. Generation of germline-competent induced pluripotent stem cells. Nature. 2007;448:313–317. doi: 10.1038/nature05934. [DOI] [PubMed] [Google Scholar]
- Jaenisch R, Young R. Stem cells, the molecular circuitry of pluripotency and nuclear reprogramming. Cell. 2008;132:567–582. doi: 10.1016/j.cell.2008.01.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao XY, Li W, Lv Z, et al. iPS cells produce viable mice through tetraploid complementation. Nature. 2009;461:86–90. doi: 10.1038/nature08267. [DOI] [PubMed] [Google Scholar]
- Boland MJ, Hazen JL, Nazor KL, et al. Adult mice generated from induced pluripotent stem cells. Nature. 2009;461:91–94. doi: 10.1038/nature08310. [DOI] [PubMed] [Google Scholar]
- Blackburn EH. Switching and signaling at the telomere. Cell. 2001;106:661–673. doi: 10.1016/s0092-8674(01)00492-5. [DOI] [PubMed] [Google Scholar]
- Palm W, de Lange T. How shelterin protects mammalian telomeres. Annu Rev Genet. 2008;42:301–334. doi: 10.1146/annurev.genet.41.110306.130350. [DOI] [PubMed] [Google Scholar]
- Wang Y, Erdmann N, Giannone RJ, et al. An increase in telomere sister chromatid exchange in murine embryonic stem cells possessing critically shortened telomeres. Proc Natl Acad Sci USA. 2005;102:10256–10260. doi: 10.1073/pnas.0504635102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hiyama E, Hiyama K. Telomere and telomerase in stem cells. Br J Cancer. 2007;96:1020–1024. doi: 10.1038/sj.bjc.6603671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wright WE, Piatyszek MA, Rainey WE, Byrd W, Shay JW. Telomerase activity in human germline and embryonic tissues and cells. Dev Genet. 1996;18:173–179. doi: 10.1002/(SICI)1520-6408(1996)18:2<173::AID-DVG10>3.0.CO;2-3. [DOI] [PubMed] [Google Scholar]
- Forsyth NR, Wright WE, Shay JW. Telomerase and differentiation in multicellular organisms: turn it off, turn it on, and turn it off again. Differentiation. 2002;69:188–197. doi: 10.1046/j.1432-0436.2002.690412.x. [DOI] [PubMed] [Google Scholar]
- Wright WE, Shay JW. Historical claims and current interpretations of replicative aging. Nat Biotechnol. 2002;20:682–688. doi: 10.1038/nbt0702-682. [DOI] [PubMed] [Google Scholar]
- Blackburn EH. Telomere states and cell fates. Nature. 2000;408:53–56. doi: 10.1038/35040500. [DOI] [PubMed] [Google Scholar]
- Gomes NM, Shay JW, Wright WE. Telomere biology in Metazoa. FEBS Lett. 2010;584:3741–3751. doi: 10.1016/j.febslet.2010.07.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blasco MA. The epigenetic regulation of mammalian telomeres. Nat Rev Genet. 2007;8:299–309. doi: 10.1038/nrg2047. [DOI] [PubMed] [Google Scholar]
- Benetti R, Gonzalo S, Jaco I, et al. Suv4-20h deficiency results in telomere elongation and derepression of telomere recombination. J Cell Biol. 2007;178:925–936. doi: 10.1083/jcb.200703081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gonzalo S, Jaco I, Fraga MF, et al. DNA methyltransferases control telomere length and telomere recombination in mammalian cells. Nat Cell Biol. 2006;8:416–424. doi: 10.1038/ncb1386. [DOI] [PubMed] [Google Scholar]
- Zalzman M, Falco G, Sharova LV, et al. Zscan4 regulates telomere elongation and genomic stability in ES cells. Nature. 2010;464:858–863. doi: 10.1038/nature08882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marion RM, Strati K, Li H, et al. Telomeres acquire embryonic stem cell characteristics in induced pluripotent stem cells. Cell Stem Cell. 2009;4:141–154. doi: 10.1016/j.stem.2008.12.010. [DOI] [PubMed] [Google Scholar]
- Agarwal S, Loh YH, McLoughlin EM, et al. Telomere elongation in induced pluripotent stem cells from dyskeratosis congenita patients. Nature. 2010;464:292–296. doi: 10.1038/nature08792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaziri H, Chapman KB, Guigova A, et al. Spontaneous reversal of the developmental aging of normal human cells following transcriptional reprogramming. Regen Med. 2010;5:345–363. doi: 10.2217/rme.10.21. [DOI] [PubMed] [Google Scholar]
- Suhr ST, Chang EA, Rodriguez RM, et al. Telomere dynamics in human cells reprogrammed to pluripotency. PLoS One. 2009;4:e8124. doi: 10.1371/journal.pone.0008124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mathew R, Jia W, Sharma A, et al. Robust activation of the human but not mouse telomerase gene during the induction of pluripotency. FASEB J. 2010;24:2702–2715. doi: 10.1096/fj.09-148973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takahashi K, Tanabe K, Ohnuki M, et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007;131:861–872. doi: 10.1016/j.cell.2007.11.019. [DOI] [PubMed] [Google Scholar]
- Stadtfeld M, Maherali N, Breault DT, Hochedlinger K. Defining molecular cornerstones during fibroblast to iPS cell reprogramming in mouse. Cell Stem Cell. 2008;2:230–240. doi: 10.1016/j.stem.2008.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Batista LF, Pech MF, Zhong FL, et al. Telomere shortening and loss of self-renewal in dyskeratosis congenita induced pluripotent stem cells. Nature. 2011;474:399–402. doi: 10.1038/nature10084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Agarwal S, Daley GQ. Telomere dynamics in dyskeratosis congenita: the long and the short of iPS. Cell Res. 2011;21:1157–1160. doi: 10.1038/cr.2011.120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang J, Wang F, Okuka M, et al. Association of telomere length with authentic pluripotency of ES/iPS cells. Cell Res. 2011;21:779–792. doi: 10.1038/cr.2011.16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herrera E, Samper E, Martin-Caballero J, et al. Disease states associated with telomerase deficiency appear earlier in mice with short telomeres. EMBO J. 1999;18:2950–2960. doi: 10.1093/emboj/18.11.2950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bailey SM, Brenneman MA, Goodwin EH. Frequent recombination in telomeric DNA may extend the proliferative life of telomerase-negative cells. Nucleic Acids Res. 2004;32:3743–3751. doi: 10.1093/nar/gkh691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Londono-Vallejo JA, Der-Sarkissian H, Cazes L, Bacchetti S, Reddel RR. Alternative lengthening of telomeres is characterized by high rates of telomeric exchange. Cancer Res. 2004;64:2324–2327. doi: 10.1158/0008-5472.can-03-4035. [DOI] [PubMed] [Google Scholar]
- Liu L, Bailey SM, Okuka M, et al. Telomere lengthening early in development. Nat Cell Biol. 2007;9:1436–1441. doi: 10.1038/ncb1664. [DOI] [PubMed] [Google Scholar]
- Morrish TA, Greider CW. Short telomeres initiate telomere recombination in primary and tumor cells. PLoS Genet. 2009;5:e 1000357. doi: 10.1371/journal.pgen.1000357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong CW, Hou PS, Tseng SF, et al. Kruppel-like transcription factor 4 contributes to maintenance of telomerase activity in stem cells. Stem Cells. 2010;28:1510–1517. doi: 10.1002/stem.477. [DOI] [PubMed] [Google Scholar]
- Koche RP, Smith ZD, Adli M, et al. Reprogramming factor expression initiates widespread targeted chromatin remodeling. Cell Stem Cell. 2011;8:96–105. doi: 10.1016/j.stem.2010.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Y, Kha H, Ungrin M, Robinson MO, Harrington L. Preferential maintenance of critically short telomeres in mammalian cells heterozygous for mTert. Proc Natl Acad Sci USA. 2002;99:3597–3602. doi: 10.1073/pnas.062549199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hanna J, Saha K, Pando B, et al. Direct cell reprogramming is a stochastic process amenable to acceleration. Nature. 2009;462:595–601. doi: 10.1038/nature08592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Polo JM, Liu S, Figueroa ME, et al. Cell type of origin influences the molecular and functional properties of mouse induced pluripotent stem cells. Nat Biotechnol. 2010;28:848–855. doi: 10.1038/nbt.1667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chin MH, Pellegrini M, Plath K, Lowry WE. Molecular analyses of human induced pluripotent stem cells and embryonic stem cells. Cell Stem Cell. 2010;7:263–269. doi: 10.1016/j.stem.2010.06.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim K, Doi A, Wen B, et al. Epigenetic memory in induced pluripotent stem cells. Nature. 2010;467:285–290. doi: 10.1038/nature09342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo G, Yang J, Nichols J, et al. Klf4 reverts developmentally programmed restriction of ground state pluripotency. Development. 2009;136:1063–1069. doi: 10.1242/dev.030957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu KJ, Grandori C, Amacker M, et al. Direct activation of TERT transcription by c-MYC. Nat Genet. 1999;21:220–224. doi: 10.1038/6010. [DOI] [PubMed] [Google Scholar]
- Flores I, Evan G, Blasco MA. Genetic analysis of myc and telomerase interactions in vivo. Mol Cell Biol. 2006;26:6130–6138. doi: 10.1128/MCB.00543-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mikkelsen TS, Hanna J, Zhang X, et al. Dissecting direct reprogramming through integrative genomic analysis. Nature. 2008;454:49–55. doi: 10.1038/nature07056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hagelstrom RT, Blagoev KB, Niedernhofer LJ, Goodwin EH, Bailey SM. Hyper telomere recombination accelerates replicative senescence and may promote premature aging. Proc Natl Acad Sci USA. 2010;107:15768–15773. doi: 10.1073/pnas.1006338107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meissner A, Wernig M, Jaenisch R. Direct reprogramming of genetically unmodified fibroblasts into pluripotent stem cells. Nat Biotechnol. 2007;25:1177–1181. doi: 10.1038/nbt1335. [DOI] [PubMed] [Google Scholar]
- Huang J, Deng K, Wu H, et al. Efficient production of mice from embryonic stem cells injected into four- or eight-cell embryos by piezo micromanipulation. Stem Cells. 2008;26:1883–1890. doi: 10.1634/stemcells.2008-0164. [DOI] [PubMed] [Google Scholar]
- Huangfu D, Maehr R, Guo W, et al. Induction of pluripotent stem cells by defined factors is greatly improved by small-molecule compounds. Nat Biotechnol. 2008;26:795–797. doi: 10.1038/nbt1418. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(A) Expression of Tert and Terc by qPCR analysis of N33 (ESCs), MEF(mouse embryonic fibroblasts), WTfb (Tail-tip fibroblasts (TTF) from adult wild-type mice at the age of 8 weeks, and Kofb (Tail-tips fibroblasts from adult telomerase Terc deficient mice).
Dynamics of telomerase and telomeres during induction of iPSCs from telomerase Terc knockout (KO, Terc−/−) TTF.
Telomerase activity and maintenance of telomeres during passages of telomerase haplo-insufficient heterozygous (Het., Terc+/−) iPSCs.
Expression of endogenous and exogenous genes of iPSCs determined by real-time PCR analysis.
Relative amount of exogenous transcript as a proportion of total transcripts in iPSCs determined by real-time PCR analysis.
Differentiation tests in vitro of Terc-deficient iPSCs at early and late passages.
Association of telomere shortening and dysfunction with reduced developmental pluripotency of iPSCs.
Karyotyping of iPSC lines
Telomere loss and chromosome fusion of iPSCs at early passage (P6)
Chimeras of iPSCs generated by injection into 8-cell albino ICR embryos
Comparison of telomere length and efficiency (%) of chimeras
Primers for PCR analysis
Ratio of contribution of iPSCs in the chimeras by microsatellite analysis