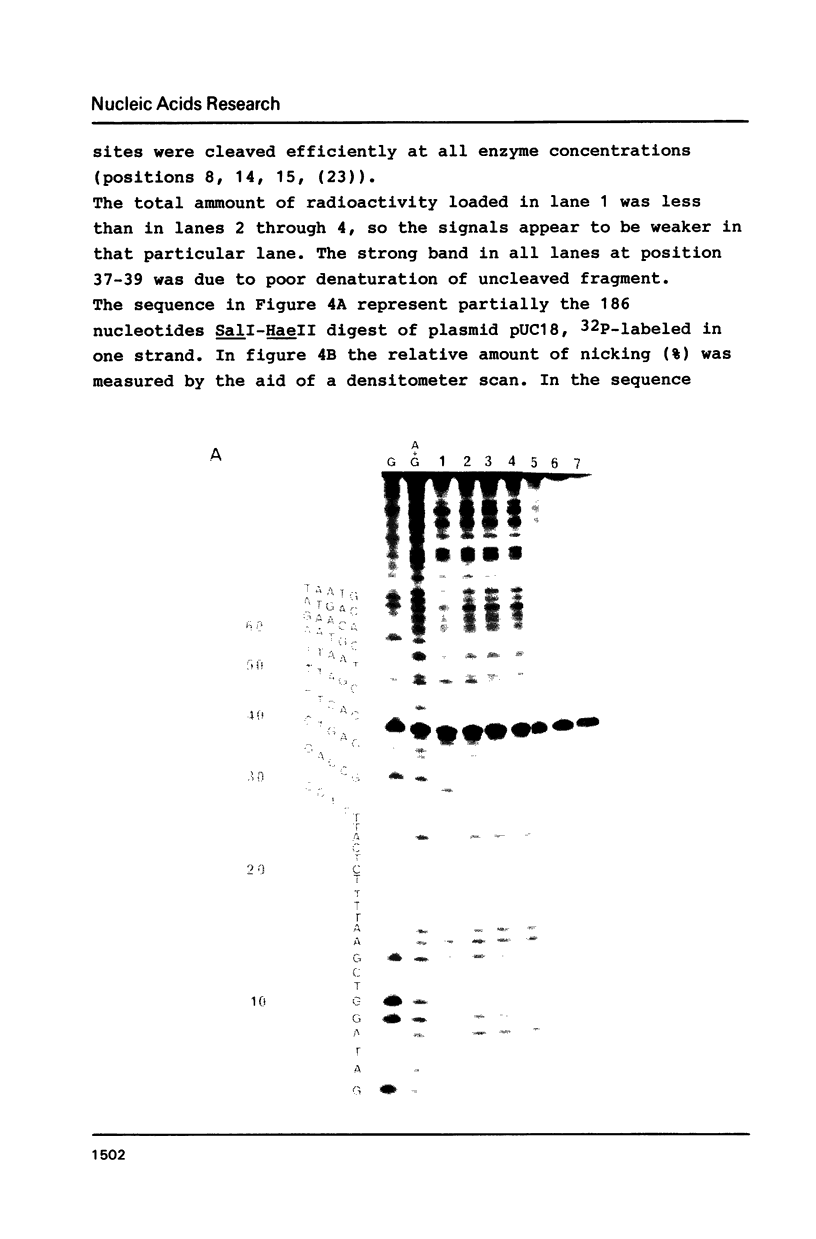
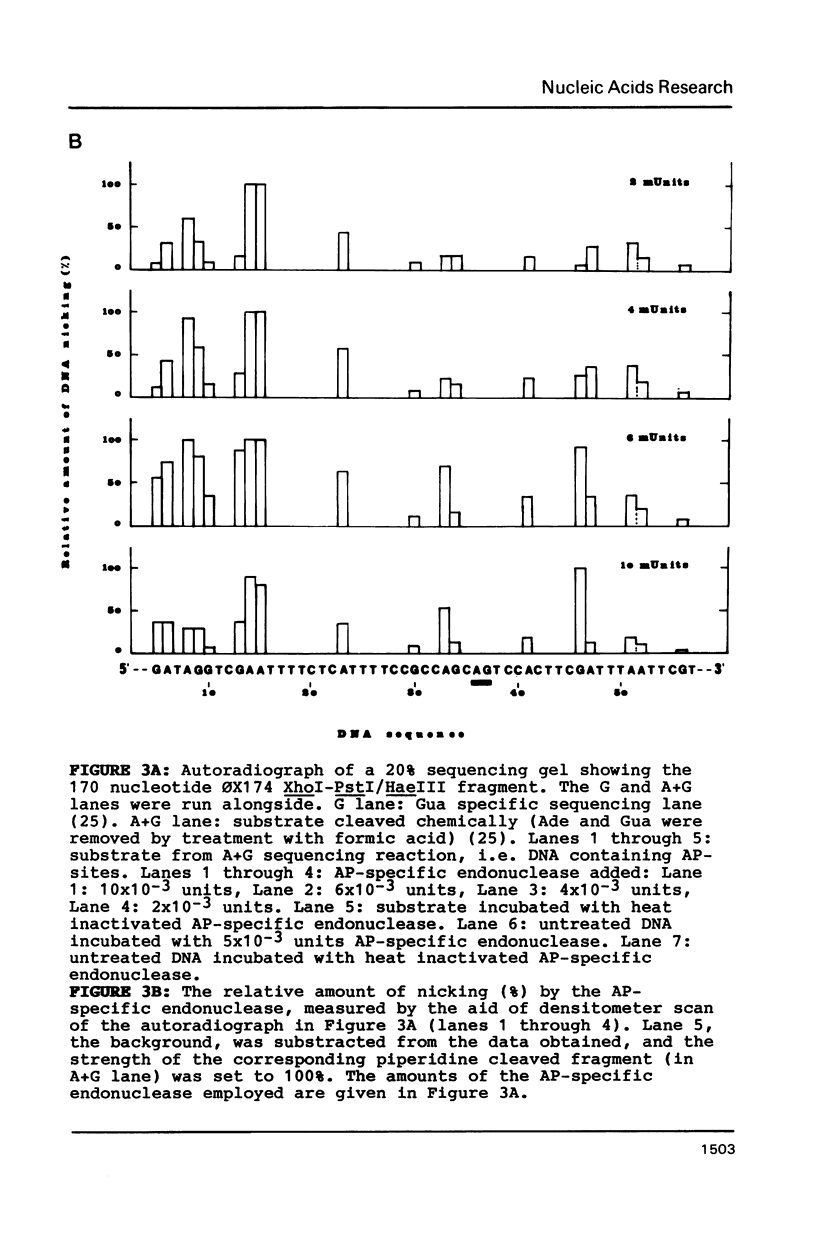
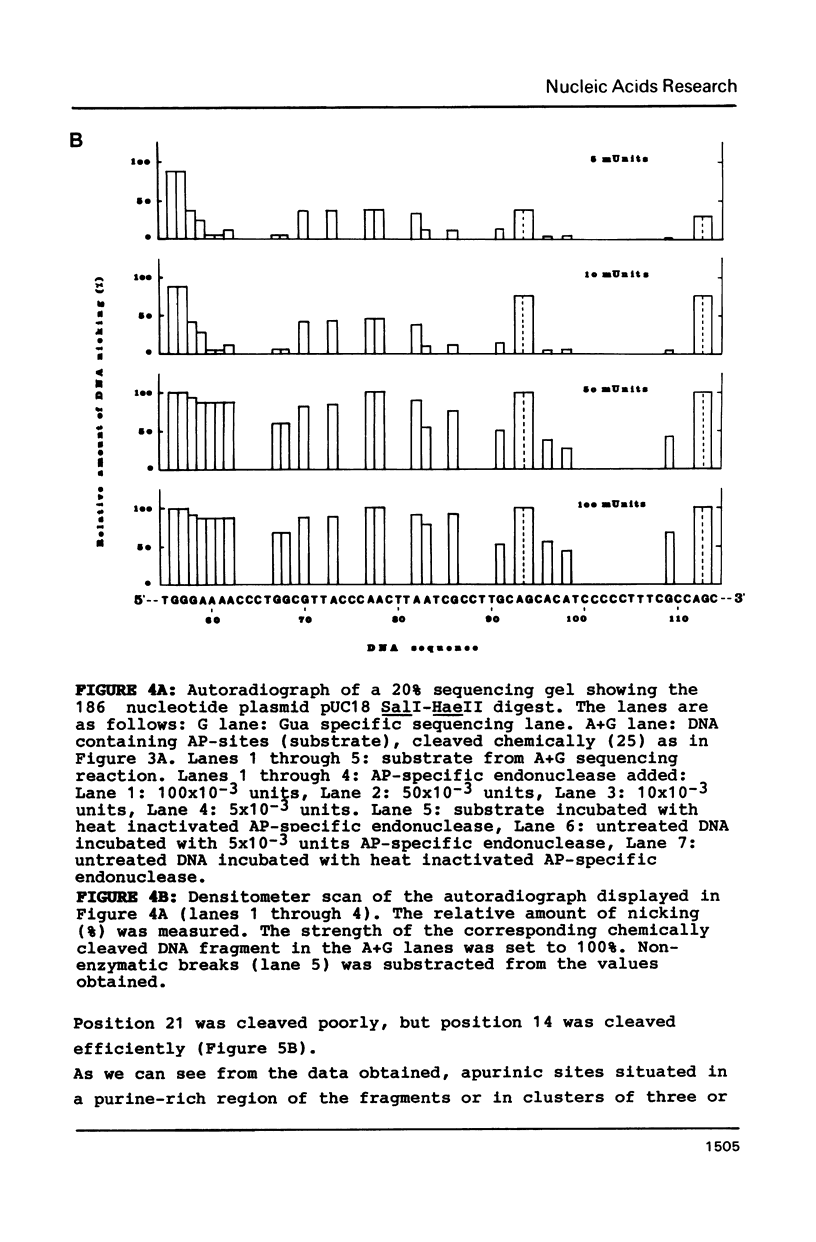
Abstract
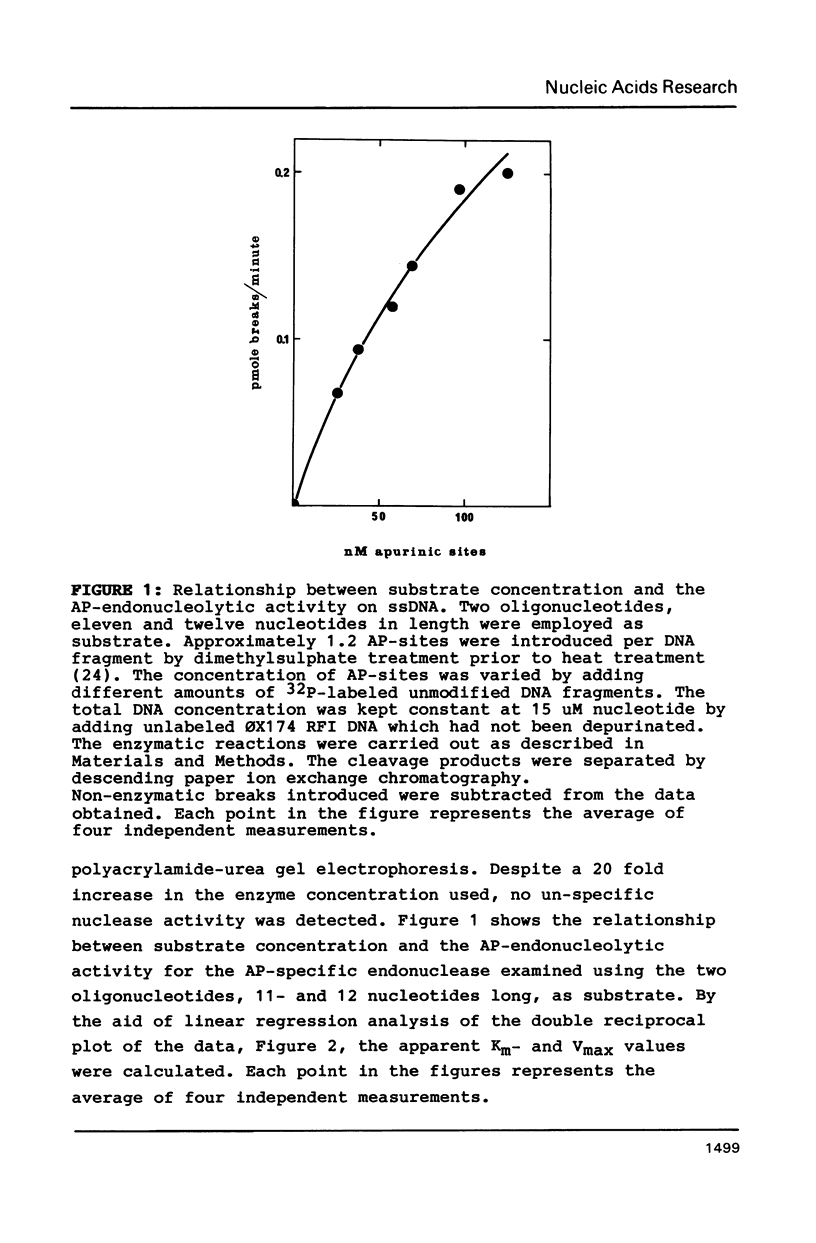
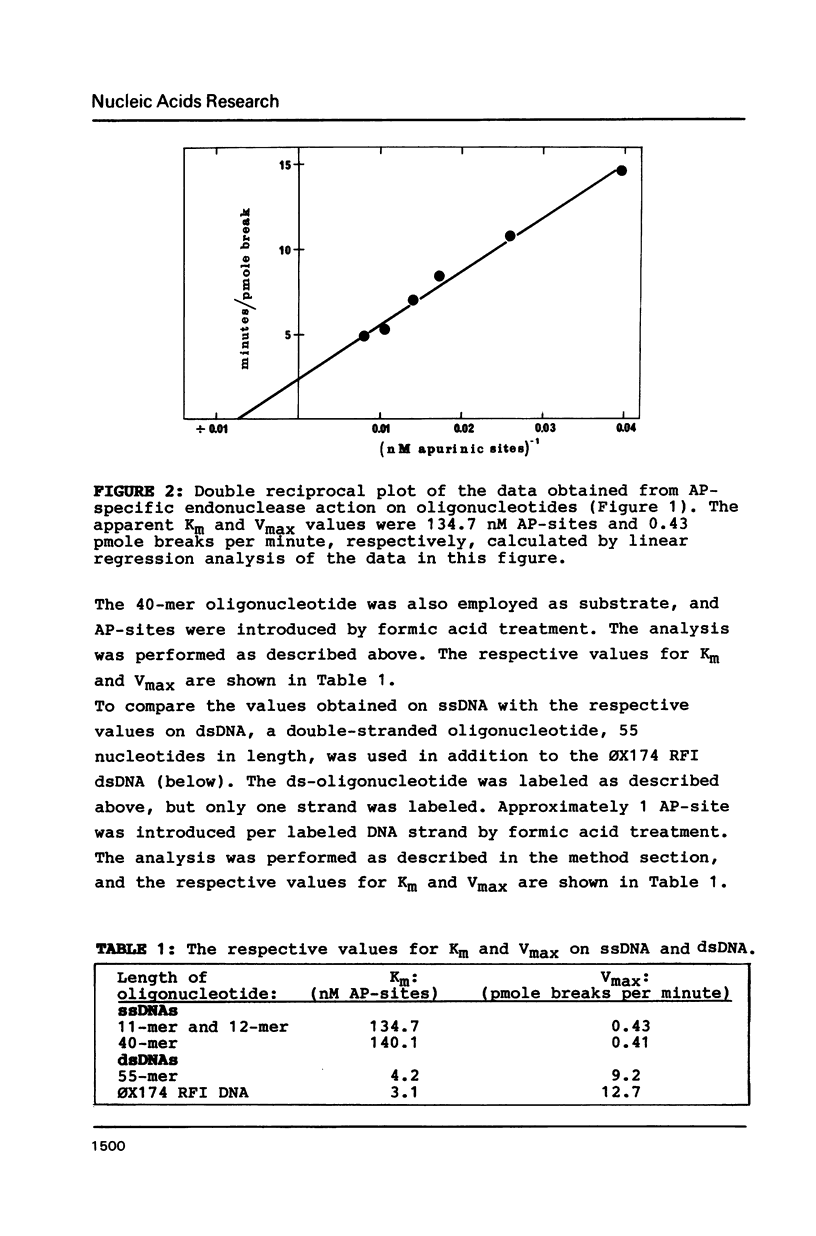
An apurinic/apyrimidinic (AP) specific endonuclease from mouse plasmacytoma cells (line MPC-11), was observed to cleave apurinic sites in oligonucleotides 9, 11, 12, 39 and 40 nucleotides in length. However, the enzyme failed to cleave AP-sites in two oligonucleotides 7 nucleotides in length. The maximum rates of digestion observed on these short single-stranded DNA (ssDNA) fragments were approximately 1/30 of the rates observed on double-stranded DNA (dsDNA). In studies using the Maxam-Gilbert DNA sequencing analysis, apurinic sites in purine-rich regions were preferentially cleaved in dsDNA but not in ssDNA, indicating that the enzyme has a sequence preference on dsDNA. These results suggest that some sites on DNA might be more efficiently repaired than others.
Full text
PDF











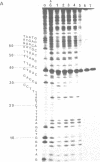
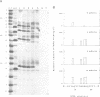
Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bailly V., Verly W. G. Escherichia coli endonuclease III is not an endonuclease but a beta-elimination catalyst. Biochem J. 1987 Mar 1;242(2):565–572. doi: 10.1042/bj2420565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boiteux S., Laval J. Coding properties of poly(deoxycytidylic acid) templates containing uracil or apyrimidinic sites: in vitro modulation of mutagenesis by deoxyribonucleic acid repair enzymes. Biochemistry. 1982 Dec 21;21(26):6746–6751. doi: 10.1021/bi00269a020. [DOI] [PubMed] [Google Scholar]
- Braun A., Grossman L. An endonuclease from Escherichia coli that acts preferentially on UV-irradiated DNA and is absent from the uvrA and uvrB mutants. Proc Natl Acad Sci U S A. 1974 May;71(5):1838–1842. doi: 10.1073/pnas.71.5.1838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Center M. S., Richardson C. C. An endonuclease induced after infection of Escherichia coli with bacteriophage T7. I. Purification and properties of the enzyme. J Biol Chem. 1970 Dec 10;245(23):6285–6291. [PubMed] [Google Scholar]
- Duker N. J., Teebor G. W. Different ultraviolet DNA endonuclease activity in human cells. Nature. 1975 May 1;255(5503):82–84. doi: 10.1038/255082a0. [DOI] [PubMed] [Google Scholar]
- Dunlap B., Cerutti P. Apyrimidinic sites in gamma-irradiated DNA. FEBS Lett. 1975 Mar 1;51(1):188–190. doi: 10.1016/0014-5793(75)80884-2. [DOI] [PubMed] [Google Scholar]
- Godson G. N., Vapnek D. A simple method of preparing large amounts of phiX174 RF 1 supercoiled DNA. Biochim Biophys Acta. 1973 Apr 11;299(4):516–520. doi: 10.1016/0005-2787(73)90223-2. [DOI] [PubMed] [Google Scholar]
- Hanawalt P. C., Cooper P. K., Ganesan A. K., Smith C. A. DNA repair in bacteria and mammalian cells. Annu Rev Biochem. 1979;48:783–836. doi: 10.1146/annurev.bi.48.070179.004031. [DOI] [PubMed] [Google Scholar]
- Haukanes B. I., Helland D. E., Kleppe K. Analysis of cleavage products of DNA repair enzymes and other nucleases. Characterization of an apurinic/apyrimidinic specific endonuclease from mouse plasmacytoma cells. Nucleic Acids Res. 1988 Jul 25;16(14B):6871–6882. doi: 10.1093/nar/16.14.6871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim J., Linn S. The mechanisms of action of E. coli endonuclease III and T4 UV endonuclease (endonuclease V) at AP sites. Nucleic Acids Res. 1988 Feb 11;16(3):1135–1141. doi: 10.1093/nar/16.3.1135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kunkel T. A. Mutational specificity of depurination. Proc Natl Acad Sci U S A. 1984 Mar;81(5):1494–1498. doi: 10.1073/pnas.81.5.1494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lindahl T. DNA glycosylases, endonucleases for apurinic/apyrimidinic sites, and base excision-repair. Prog Nucleic Acid Res Mol Biol. 1979;22:135–192. doi: 10.1016/s0079-6603(08)60800-4. [DOI] [PubMed] [Google Scholar]
- Lindahl T. DNA repair enzymes. Annu Rev Biochem. 1982;51:61–87. doi: 10.1146/annurev.bi.51.070182.000425. [DOI] [PubMed] [Google Scholar]
- Lindahl T., Nyberg B. Rate of depurination of native deoxyribonucleic acid. Biochemistry. 1972 Sep 12;11(19):3610–3618. doi: 10.1021/bi00769a018. [DOI] [PubMed] [Google Scholar]
- Male R., Haukanes B. I., Helland D. E., Kleppe K. Substrate specificity of 3-methyladenine-DNA glycosylase from calf thymus. Eur J Biochem. 1987 May 15;165(1):13–19. doi: 10.1111/j.1432-1033.1987.tb11188.x. [DOI] [PubMed] [Google Scholar]
- Maxam A. M., Gilbert W. A new method for sequencing DNA. Proc Natl Acad Sci U S A. 1977 Feb;74(2):560–564. doi: 10.1073/pnas.74.2.560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maxam A. M., Gilbert W. Sequencing end-labeled DNA with base-specific chemical cleavages. Methods Enzymol. 1980;65(1):499–560. doi: 10.1016/s0076-6879(80)65059-9. [DOI] [PubMed] [Google Scholar]
- Mosbaugh D. W., Linn S. Further characterization of human fibroblast apurinic/apyrimidinic DNA endonucleases. The definition of two mechanistic classes of enzyme. J Biol Chem. 1980 Dec 25;255(24):11743–11752. [PubMed] [Google Scholar]
- Nes I. F., Nissen-Meyer J. Endonuclease activities from a permanently established mouse cell line that act upon DNA damaged by ultraviolet light, acid and osmium tetroxide. Biochim Biophys Acta. 1978 Aug 23;520(1):111–121. doi: 10.1016/0005-2787(78)90012-6. [DOI] [PubMed] [Google Scholar]
- Nes I. F. Purification and characterization of an endonuclease specific for apurinic sites in DNA from a permanently established mouse plasmacytoma cell line. Nucleic Acids Res. 1980 Apr 11;8(7):1575–1589. doi: 10.1093/nar/8.7.1575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nes I. F. Purification and properties of a mouse-cell DNA-repair endonuclease, which recognizes lesions in DNA induced by ultraviolet light, depurination, gamma-rays, and OsO4 treatment. Eur J Biochem. 1980 Nov;112(1):161–168. doi: 10.1111/j.1432-1033.1980.tb04997.x. [DOI] [PubMed] [Google Scholar]
- Randall S. K., Eritja R., Kaplan B. E., Petruska J., Goodman M. F. Nucleotide insertion kinetics opposite abasic lesions in DNA. J Biol Chem. 1987 May 15;262(14):6864–6870. [PubMed] [Google Scholar]
- Sagher D., Strauss B. Insertion of nucleotides opposite apurinic/apyrimidinic sites in deoxyribonucleic acid during in vitro synthesis: uniqueness of adenine nucleotides. Biochemistry. 1983 Sep 13;22(19):4518–4526. doi: 10.1021/bi00288a026. [DOI] [PubMed] [Google Scholar]
- Schaaper R. M., Kunkel T. A., Loeb L. A. Infidelity of DNA synthesis associated with bypass of apurinic sites. Proc Natl Acad Sci U S A. 1983 Jan;80(2):487–491. doi: 10.1073/pnas.80.2.487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schaaper R. M., Loeb L. A. Depurination causes mutations in SOS-induced cells. Proc Natl Acad Sci U S A. 1981 Mar;78(3):1773–1777. doi: 10.1073/pnas.78.3.1773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strauss B., Rabkin S., Sagher D., Moore P. The role of DNA polymerase in base substitution mutagenesis on non-instructional templates. Biochimie. 1982 Aug-Sep;64(8-9):829–838. doi: 10.1016/s0300-9084(82)80138-7. [DOI] [PubMed] [Google Scholar]
- Vinograd J., Lebowitz J., Radloff R., Watson R., Laipis P. The twisted circular form of polyoma viral DNA. Proc Natl Acad Sci U S A. 1965 May;53(5):1104–1111. doi: 10.1073/pnas.53.5.1104. [DOI] [PMC free article] [PubMed] [Google Scholar]