Abstract
Mupirocin has been used in nursing homes to prevent the spread of methicillin-resistant Staphylococcus aureus (MRSA), despite the lack of controlled trials. In this double-blind, randomized study, the efficacy of intranasal mupirocin ointment versus that of placebo in reducing colonization and preventing infection was assessed among persistent carriers of S. aureus. Twice-daily treatment was given for 2 weeks, with a follow-up period of 6 months. Staphylococcal colonization rates were similar between residents at the Ann Arbor Veterans Affairs (VA) Extended Care Center, Michigan (33%), and residents at a community-based long-term care facility in Ann Arbor (36%), although those at the VA Center carried MRSA more often (58% vs. 35%; P = .017). After treatment, mupirocin had eradicated colonization in 93% of residents, whereas 85% of residents who received placebo remained colonized (P < .001). At day 90 after study entry, 61% of the residents in the mupirocin group remained decolonized. Four patients did not respond to mupirocin therapy; 3 of the 4 had mupirocin-resistant S. aureus strains. Thirteen (86%) of 14 residents who became recolonized had the same pretherapy strain; no strain recovered during relapse was resistant to mupirocin. A trend toward reduction in infections was seen with mupirocin treatment.
Colonization and infection with Staphylococcus aureus, which are common in older persons, are perhaps associated with chronic disease and debility [1]. Mortality due to staphylococcal bacteremia is greater among older persons and is >60% among residents of long-term care facilities (LTCFs) and hospitalized older adults [2–4]. For staphylococcal carriers, the risk of infection with S. aureus is significantly higher than it is for noncarriers, and infection is usually caused by the colonizing strain [5, 6]. Carriage of methicillin-resistant S. aureus (MRSA) has been shown to be a marker for increased risk of infection and mortality among LTCF residents [7–9].
Treatment of persistent staphylococcal carriage with the topical antibiotic mupirocin has been shown to decrease staphylococcal infections among patients undergoing hemodialysis and those who have undergone elective surgery [10–14]. Several noncontrolled studies have shown that mupirocin alone or in combination with other measures decreases S. aureus colonization among residents of LTCFs [15–18]. However, there are no controlled trials involving such individuals that have definitively shown that mupirocin decreases S. aureus colonization and infection.
This study, which was performed in both community and Veterans Affairs (VA) LTCFs in Ann Arbor, Michigan, assessed whether a 2-week course of mupirocin therapy led to prolonged reduction in colonization and prevented S. aureus infection. Recolonization with S. aureus and acquisition of mupirocin resistance were evaluated.
STUDY PARTICIPANTS AND METHODS
Study population
The Ann Arbor VA Extended Care Center is a 50-bed facility attached to an acute care hospital. Residents are admitted for long-term care (10 beds), rehabilitation (10 beds), and geriatric evaluation (30 beds). Glacier Hills Nursing Center is a 163-bed community LTCF located within a few miles of the VA Medical Center. It has a 127-bed skilled nursing unit and a 36-bed dementia unit.
Eligibility
All residents of the VA and community LTCFs were eligible. Appropriate informed consent was obtained, and guidelines for human experimentation and the conduct of clinical research were followed, as required by the University of Michigan and Veterans Affairs Ann Arbor Healthcare System institutional review boards. After obtaining written informed consent, specimens were obtained from the nares and, when present, wounds of all patients. Residents for whom culture results were positive for S. aureus on 2 consecutive cultures performed ≤2 weeks apart were considered persistent carriers and were enrolled into the treatment trial.
Exclusion criteria
Residents were not enrolled if they were receiving systemic antibiotic therapy or topical antibiotic therapy for wounds, had active S. aureus infection, or were judged unable to cooperate with the study. Known hypersensitivity to mupirocin and the presence of large wounds (i.e., a surface area of >10 × 10 cm and a depth of >3 cm) were also exclusion factors. Residents who were initially found not to be carriers were screened again if they required admission to the hospital and then returned to the LTCF.
Group assignment
Enrolled residents were randomly assigned to study groups by stratification according to LTCF type and presence of wounds. Randomization was performed separately on the basis of residence type and presence of wounds in blocks of 2 to assure that the number of patients assigned to study groups using these strata was equal. Investigators, nursing staff, and study participants were blinded to the treatment group.
Treatment
Mupirocin therapy or placebo was administered twice daily for 14 days by study personnel who wore gloves after appropriate hand disinfection and who were blinded to results of microbiological tests. The study drugs were placed in identical containers labeled “A” or “B” by the study pharmacist. Mupirocin 2% ointment in polyethylene glycol (PEG) base or plain PEG ointment (placebo) was applied to each anterior naris, and the nose was massaged gently. Ointment was applied over wound surfaces in a thin layer.
Samples were obtained from nares and wounds every other day during the treatment period. On day 15 of the study—the day after treatment ended—a sample was obtained. Successive samples were obtained every week during the next 4 weeks, every 2 weeks for 2 months, and monthly for an additional 2.5 months, as long as the patient remained in the facility. Enrolled residents were observed for the development of staphylococcal infection during the same 6-month period, as long as they were residents of the facility.
Permitted local wound care included whirlpool therapy, debridement, and application of hydrogen peroxide, Dakin’s solution (sodium hypochlorite 5.25%), and dressings. Wound therapy was provided before the study drug or placebo was administered. No topical or systemic antibiotic therapy with activity against S. aureus was allowed.
Assessment
Demographic characteristics and risk factors for S. aureus colonization were assessed at study entry. Dimensions of decubitus or vascular ulcers, other wounds, skin conditions, and devices were recorded. Functional status was measured using a modified Katz scale [19].
Enrolled residents were monitored daily for S. aureus infection, on the basis of the Centers for Disease Control and Prevention definitions [20]. All cases were reviewed by a panel of 3 consultants who specialized in infectious diseases and geriatrics, all of whom were blinded to colonization data and treatment regimens.
Outcomes
Outcomes were categorized as cure (i.e., no S. aureus was recovered from any site) or failure (i.e., persistence of S. aureus at the end of treatment or recolonization after negative culture results on day 15). Recolonization was further defined as relapse (i.e., recolonization by the previously colonizing S. aureus strain) or reinfection (i.e., acquisition of a new S. aureus strain).
To test the hypothesis that mupirocin therapy led to eradication of S. aureus colonization, short-term (15 days after study entry) and long-term (90 days after study entry) responses were assessed, with S. aureus colonization being the primary outcome variable. Recolonization, emergence of mupirocin-resistant strains, and reduction in S. aureus infections in residents treated with mupirocin were secondary outcomes.
Microbiological methods
Nares and wounds were swabbed with sterile rayon-tipped applicator sticks that were then placed into Stuart’s transport medium. Samples were obtained before daily local wound care and application of mupirocin. Swabs were streaked onto colistin–nalidixic acid agar with 5% sheep blood (BBL; Becton Dickinson) and incubated at 35°C for 24 h. S. aureus identification and methicillin-susceptibility testing were performed using standard methods [15, 16]. If a patient was found to have both MRSA and methicillin-susceptible S. aureus (MSSA), methicillin resistance was verified by a latex agglutination test for penicillin-binding protein 2a (Oxoid). Isolates were stored at −70°C for later analysis.
Each isolate was screened for mupirocin resistance by inoculation onto Mueller-Hinton agar plates containing 2 μg/mL mupirocin. The MICs of mupirocin for isolates that grew on the screening plates were determined by Etest (AB Biodisk) [21]. Mupirocin resistance was defined as an MIC of ≥4 μg/mL, and high-level resistance was defined as an MIC of ≥500 μg/mL [22].
S. aureus strains recovered from residents who were recolonized after therapy and from those who did not respond to therapy were typed by PGFE to determine whether recolonization or therapy failure was due to the previous colonizing strain or a new strain. Genomic DNA fragments obtained after digestion with SmaI (New England BioLabs) were separated by PFGE using a CHEF III system (BioRad) [23, 24]. Gels were stained and photographed, and the banding patterns of different isolates were compared visually. Strain relatedness was determined as described elsewhere [25].
Statistical methods
Data were entered into EpiInfo 6.0 (Centers for Disease Control and Prevention) and analyzed using SAS software. Demographic and clinical characteristics at enrollment that predisposed residents to S. aureus colonization were compared between VA and community LTCFs and between treatment groups. Depending on the scale by which variables were measured, the following standard 2-group comparison methods were used: 2-sample Student’s t test, for normally distributed variables; Mann-Whitney U test, for continuous nonnormally distributed variables; or χ2 test, for categorical variables. A P value of <.05 was considered to be statistically significant.
The proportion of residents in each treatment arm who were colonized with S. aureus was compared after treatment was stopped (i.e., 15 days after study entry) and on day 90 using the standard Cochran-Mantel-Haenszel (CMH) test on day 15 and a modified CMH test on day 90 (because of residents lost to follow-up) [26, 27]. The Andersen-Gill model was used to analyze data on time to decolonization. The model, an extension of the classical Cox regression model, accounts for potential spontaneous clearance and recolonization.
RESULTS
Demographic and clinical characteristics of the study population at enrollment
Of the 427 residents screened for colonization with S. aureus, 254 (59%) were from the VA LTCF and 173 (41%) were from the community LTCF (table 1). Overall, 83 (33%) of VA and 62 (36%) of community LTCF residents were persistently colonized with S. aureus and eligible for enrollment. Of the patients eligible to participate, 127 (85%) were enrolled into the treatment trial; 73 (57%) were from the VA LTCF, and 54 (43%) were from the community LTCF.
Table 1.
Demographic and clinical characteristics of residents of 2 long-term care facilities (LTCFs) in Ann Arbor, Michigan, who were enrolled into the study.
| Variable | LTCF type
|
P | |
|---|---|---|---|
| Veterans Affairs hospital | Community-based nursing center | ||
| No. of residents screened | 254 | 173 | … |
| Persistent staphylococcal carriers | 83 (33) | 62 (36) | .5 |
| No. of residents enrolled | 73 | 54 | .6 |
| Age, mean years ± SD | 69 ± 9.9 | 86 ± 7.1 | <.001 |
| Male sex | 70 (96) | 13 (24) | <.001 |
| Diabetes mellitus | 26 (37) | 14 (26) | .25 |
| Acute carea | 63 (86) | 22 (41) | <.001 |
| Antibiotic usea | 43 (59) | 13 (24) | <.001 |
| ADL | |||
| Class 1 | 39 (53) | 7 (13) | <.001 |
| Class 2 | 30 (41) | 34 (63) | … |
| Class 3 | 4 (6) | 13 (24) | … |
| Device use | 35 (48) | 12 (22) | .003 |
| Renal insufficiency | 11 (15) | 5 (9) | .33 |
| Peripheral vascular disease | 23 (32) | 4 (6) | .001 |
| Injectable medication use | 18 (25) | 6 (11) | .05 |
| Skin disease | 9 (12) | 1 (2) | .03 |
| Wounds | 7 (10) | 1 (2) | .08 |
NOTE. Data are no. or no. (%) of individuals, unless otherwise indicated. ADL, activities of daily living.
During the 30-day period before enrollment.
A total of 83 enrolled residents (65%) were men, and 44 (35%) were women. The majority of men (84%) were VA LTCF residents, and almost all women (93%) were community LTCF residents. The mean age (±SD) of study participants was 76.2 ± 12.3 years (range, 45–102 years). The VA LTCF study population was significantly younger than community LTCF study population (69 ± 9.9 vs. 86 ± 7.1 years; P <.001).
Among the 73 VA LTCF residents, colonization with MRSA alone occurred in 42 (58%); an additional 2 (3%) were colonized with both MRSA and MSSA at enrollment. Among the 54 community LTCF residents, 19 (35%) were colonized with MRSA only, and 1 (2%) was colonized with both MRSA and MSSA at enrollment; for 2 residents, methicillin susceptibility was not determined because isolates recovered at enrollment were lost. The difference in the prevalence of MRSA carriage was significantly different between the 2 facilities (P = .017).
Risk factors for colonization with S. aureus
The most common risk factors for persistent S. aureus carriage included recent hospitalization (67% of study participants), antibiotic use in the 30 days before study entry (44%), presence of devices (37%), and diabetes mellitus (32%). Only 8 (6%) of enrolled residents had wounds at enrollment. VA LTCF study participants were significantly more likely to have been hospitalized or to have received antibiotics, to have a device, peripheral vascular disease, or skin disease, and to be functionally more independent than were community LTCF study participants (table 1).
Response to treatment with study drug
Sixty-four and 63 LTCF residents were randomized to the mupirocin and placebo groups, respectively (table 2). No significant differences were noted between the groups, with the exception of functional status, which was slightly better in the mupirocin group (P = .05) (table 2). Among study participants who received mupirocin, 36 (56%) were at the VA LTCF, and 28 (44%) were at the community-based LTCF. Colonization with MRSA was noted in 27 (44%) of those in the mupirocin group and in 36 (57%) of those in the placebo group (P = .07).
Table 2.
Demographic and clinical characteristics of long-term care facility residents randomized to received mupirocin therapy or placebo.
| Variable | Mupirocin group (n = 64) | Placebo group (n = 63) | P |
|---|---|---|---|
| Age, mean years ± SD | 76.3 ± 12.8 | 76.0 ± 12.0 | .9 |
| Male sex | 43 (67) | 40 (63) | .7 |
| Diabetes mellitus | 17 (27) | 23 (37) | .2 |
| Acute carea | 42 (66) | 43 (68) | .8 |
| Antibiotic usea | 28 (44) | 28 (44) | .9 |
| ADL | |||
| Class 1 | 27 (40) | 19 (30) | .05 |
| Class 2 | 33 (52) | 31 (49) | … |
| Class 3 | 4 (8) | 13 (21) | … |
| Device use | 22 (35) | 25 (40) | .5 |
| Renal insufficiency | 7 (11) | 9 (14) | .6 |
| Peripheral vascular disease | 13 (20) | 14 (22) | .8 |
| Injectable medication use | 10 (16) | 14 (22) | .3 |
| Skin disease | 7 (11) | 3 (5) | .2 |
| Wounds | 5 (8) | 3 (5) | .5 |
NOTE. Data are no. (%) of study participants, unless otherwise indicated. ADL, activities of daily living.
During the 30-day period before enrollment.
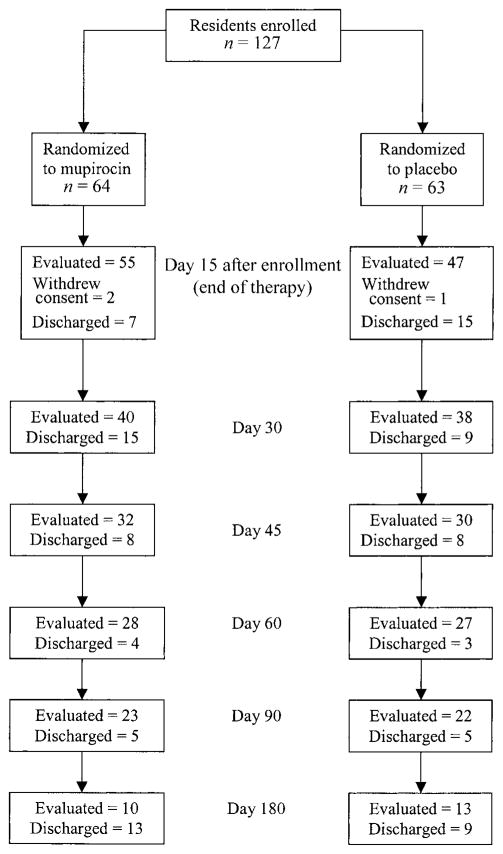
Of the group of 127 residents who were treated, 102 (80%) completed 14 days of therapy and were evaluated for treatment efficacy (figure 1). Twenty-two study participants were discharged before completing therapy, and 3 refused to complete therapy. Figure 1 shows data on the number of participants who were evaluated at each of the subsequent time points after study entry.
Figure 1.
Number of study participants evaluated for Staphylococcus aureus decolonization, by time point.
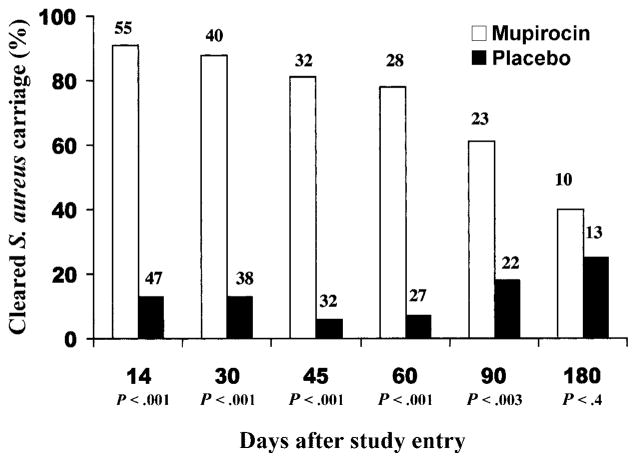
On day 15 of the study (the day after treatment ended), 51 (93%) of 55 study participants who were randomized to receive mupirocin were no longer colonized with S. aureus, compared with 7 (15%) of 47 in the placebo group (P < .001) (figure 2). Among 8 study participants with wounds, 3 did not complete 14 days of therapy and could not be evaluated; all 4 who had received mupirocin were no longer colonized with S. aureus, and 1 who had received placebo remained colonized.
Figure 2.
Proportion of long-term care residents free of colonization with Staphylococcus aureus after receiving mupirocin therapy (unshaded regions) or placebo (shaded regions) for 14 days. The number of patients remaining in the study at each time point is indicated above each bar.
At 30 days after study entry (2 weeks after treatment was stopped), 35 (88%) of the 40 residents who received mupirocin were free of S. aureus, compared with 5 (13%) of 38 of those who had received placebo (P <.001). Three residents in the mupirocin group and 1 in the placebo group had become recolonized with S. aureus. At day 60, 22 (79%) of 28 of the study participants remained decolonized, and, at day 90, 14 (61%) of 23 who had received mupirocin remained free of S. aureus. Among residents in the placebo group, decolonization was uncommon, occurring in 7%–18% at the time points at which samples were obtained. S. aureus colonization did not differ between the 2 groups at 180 days, but too few residents remained in the study to draw conclusions about this time period. Over the entire duration of the study, the hazard rate of colonization was 4.12 times higher for the placebo group (95% CI, 2.68–6.31; P < .0001).
Four residents in the mupirocin group—all of whom were colonized with MRSA—did not respond to decolonization therapy (table 3). However, decolonization rates did not differ statistically between those with MRSA and those with MSSA (P = .08). All 4 remained colonized with the strain recovered at enrollment. One resident each had a low-level and a high-level mupirocin-resistant strain at enrollment that persisted, despite receipt of therapy. Another was colonized with a strain that developed high-level mupirocin resistance during the course of treatment, and the fourth remained colonized with a mupirocin-susceptible strain throughout treatment.
Table 3.
MICs of mupirocin for Staphylococcus aureus isolates recovered from long-term care residents who did not respond to treatment with mupirocin.
| Patient no. | Mupirocin MIC, μg/mL (no. of isolates)
|
||
|---|---|---|---|
| Before therapy | During therapya | After therapyb | |
| 1 | <0.98 (2) | <0.98 (2) | >500 (2) |
| 2 | 16–32 (4) | 16–64 (8) | 16–32 (9) |
| 3 | <0.98 (1) >500 (1) |
>500 (2) | >500 (2) |
| 4 | <0.98 (1) | <0.98 (4) | <0.98 (2) |
Isolates recovered on days 1–13.
Isolates recovered on day 15 and thereafter.
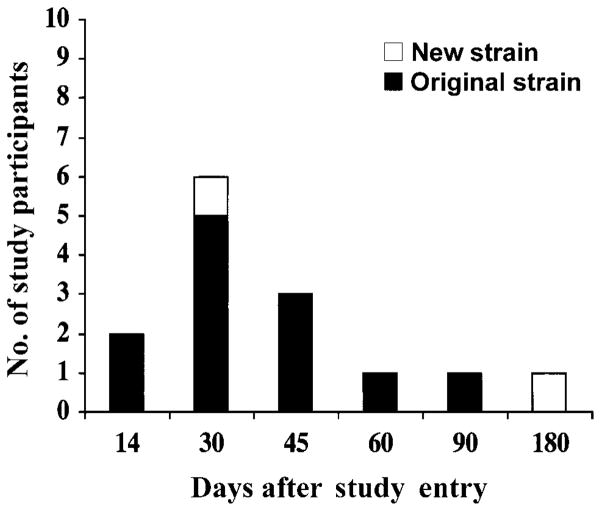
Of the 14 study participants who became recolonized with S. aureus after initial decolonization, 12 (86%) relapsed with the same strain and 2 acquired a new strain (figure 3). All recolonizing strains remained mupirocin susceptible. The PEG formulation was well tolerated; failure to complete therapy because of side effects occurred in only 1 placebo recipient, who complained of nasal stuffiness.
Figure 3.
Study participants (n = 14) who were recolonized with Staphylococcus aureus at various time points after initial decolonization. Patients either relapsed with the same pretherapy strain (shaded regions) or acquired a new strain (unshaded regions), as defined by PFGE.
Infections
Ten (10%) of 102 residents who finished the 14-day treatment course developed laboratory-confirmed or probable infection with S. aureus. Three (5%) of 55 residents treated with mupirocin and 7 (15%) of 47 who received placebo developed infection (P = .10). In the placebo group, 3 and 4 residents, respectively, developed infection during treatment and after the regimen was stopped. Cellulitis occurred in 3 study participants, conjunctivitis in 2, a perirectal abscess in 1, and a urinary tract infection in 1. Of the 3 residents in the mupirocin group who developed infection, all had cellulitis that developed 5–38 days after treatment had stopped; all 3 had cleared staphylococcal nasal carriage and remained free of S. aureus in the nares at the time infection occurred. Culture results for 2 patients were positive for MRSA, and culture was not performed for the third, although the infection in this patient responded to cephalexin.
DISCUSSION
In this study of LTCF residents, mupirocin was highly effective in decolonizing persistent S. aureus colonization in the nares, compared with placebo. At the end of therapy, 93% of residents in the mupirocin group had cleared S. aureus from the nares. Previous studies of mupirocin in LTCFs—although they were not placebo controlled—showed similar clearance rates [15, 16]. Clearance rates of 90% are similar to those noted for other patient populations [12, 13, 28, 29] and health care workers [30].
Studies to assess decolonization strategies in older persons have been infrequent [15, 16, 18, 31], and some regimens have been less effective in such patients [31–33]. In an older population of hospitalized patients, mupirocin was no more effective than was placebo for clearing MRSA carriage [33]. In an uncontrolled study in which oral antibiotics were administered with or without rifampin, persistent or recurrent colonization within 30 days after treatment completion was demonstrated in 56% of LTCF residents [31], and another study in which oral antibiotics and rifampin were administered noted age as a risk factor for failure to clear MRSA [32].
We found that decolonization due to mupirocin persisted for 45 days after treatment had ended (day 60 after study entry) and that, although the duration of decolonization was still significantly different from that associated with placebo, decolonization efficacy had begun to decrease by day 90 after study entry. Few studies have assessed the effect of decolonization regimens on the persistence of clearance for >30 days after treatment was stopped. An earlier uncontrolled study in our facility noted that 56% of residents remained decolonized for 60 days after mupirocin therapy was stopped [15], and a placebo-controlled study of intranasal bacitracin and oral rifampin demonstrated that 44% of patients who were undergoing hemodialysis remained decolonized for 90 days [34].
In this study, recolonization in 12 (86%) of 14 residents involved the original strain. In contrast, health care workers who were treated with mupirocin were as likely to relapse with a new strain as with their pretherapy strain [35]. Relapse of S. aureus carriage in residents of LTCFs is likely due to persistent risk factors that contribute to initial colonization [9, 36, 37]. Relapse of S. aureus carriage in our study was not associated with the development of mupirocin resistance.
One approach to decrease recolonization is intermittent mupirocin treatment. Thrice-weekly, weekly, and twice-monthly regimens have been used to maintain nasal decolonization in patients undergoing dialysis [38, 39], without development of mupirocin resistance. However, use of a weekly maintenance regimen of mupirocin in MRSA-colonized residents of LTCFs was less efficacious and was associated with mupirocin resistance [16]. On the basis of the results of this study, an alternative approach might be pulse therapy with 14 days of mupirocin every 2–3 months to prevent relapse and decrease the likelihood of resistance. A similar approach has been successful in patients with furunculosis (mupirocin was administered for 5 days each month) [40] and has been suggested for use in patients undergoing hemodialysis [41].
Three of 4 residents who did not respond to therapy were found to carry mupirocin-resistant S. aureus at the end of the treatment period, only 1 of whom had a strain that developed resistance. All 4 mupirocin failures in this study occurred in MRSA-colonized residents, but the rate of failure was not statistically different from that for MSSA-colonized residents. MRSA strains are no more likely to harbor or acquire mupirocin resistance elements than are MSSA strains [22]. It is likely that patient factors that select for MRSA—and not only for mupirocin resistance—play a role in failure of decolonization.
Mupirocin PEG ointment has been used extensively for de-colonization of staphylococcal carriage [15–17, 42–45]. Different formulations were developed because of concern for nasal side effects and absorption of PEG in neonates and in large burns wounds [14]. In our study, the PEG formulation was well tolerated.
It has been questioned whether data on S. aureus colonization obtained from VA LTCFs are applicable to community LTCFs. As a group, the VA LTCF population is younger, predominantly male, and more functionally independent [8, 36]. We found no differences in colonization rates for S. aureus between the 2 facilities, but the proportion of S. aureus isolates that were methicillin resistant was higher in VA LTCF study participants, confirming previous reports [8]. The colonization rates in the community facility were higher than we anticipated. This could reflect increasing MRSA rates in the community [46, 47] or increasing trends toward transferring sicker patients from the hospital to the LTCF [48].
Approximately 20% of the study participants did not complete the 14-day therapy course; this hampered our ability to detect differences in infection rates. The number of residents in each treatment arm was too small to show a significant difference in infection rates, although a trend toward decreased infection rates was seen in the mupirocin group. Some S. aureus carriers may be at greater risk of infection than others [1, 9, 36]. Mupirocin decolonization might prove more efficacious if residents who were most dependent or who had devices in place, diabetes mellitus, or peripheral vascular occlusive disease were treated.
In conclusion, mupirocin was effective in decolonizing S. aureus in persistent carriers enrolled from 2 LTCF populations. Although recolonization occurred, development of resistance was uncommon. There was a trend towards reduction in infections with mupirocin use. Multicenter trials that focus on persistent S. aureus carriers—who are at greatest risk—are needed to assess the effectiveness of mupirocin in reducing infections in LTCFs.
Acknowledgments
Financial support: National Institutes on Aging and Claude D. Pepper Older Americans Independence Centers (AG08808).
We thank SmithKline Beecham for providing Etest strips and mupirocin powder.
References
- 1.Bradley SF. Staphylococcus aureus infections and antibiotic resistance in older adults. Clin Infect Dis. 2002;34:211–6. doi: 10.1086/338150. [DOI] [PubMed] [Google Scholar]
- 2.McClelland RS, Fowler VG, Sanders LL, et al. Staphylococcus aureus bacteremia among elderly vs. younger adult patients: comparison of clinical features and mortality. Arch Intern Med. 1999;159:1244–7. doi: 10.1001/archinte.159.11.1244. [DOI] [PubMed] [Google Scholar]
- 3.Muder RR, Brennen C, Wagener MM, et al. Bacteremia in a long-term care facility: a five-year prospective study of 163 consecutive episodes. Clin Infect Dis. 1992;14:647–54. doi: 10.1093/clinids/14.3.647. [DOI] [PubMed] [Google Scholar]
- 4.Whitelaw DA, Rayner BL, Willcox PA. Community-acquired bacteremia in the elderly: a prospective study of 121 cases. J Am Geriatr Soc. 1992;40:996–1000. doi: 10.1111/j.1532-5415.1992.tb04475.x. [DOI] [PubMed] [Google Scholar]
- 5.Kluytmans J, van Belkam A, Verbrugh H. Nasal carriage of Staphylococcus aureus: epidemiology, underlying mechanisms, and associated risks. Clin Microbiol Rev. 1997;10:505–20. doi: 10.1128/cmr.10.3.505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Von Eiff C, Becker K, Machka K, et al. Nasal carriage as a source of Staphylococcus aureus bacteremia. N Engl J Med. 2001;344:11–6. doi: 10.1056/NEJM200101043440102. [DOI] [PubMed] [Google Scholar]
- 7.Niclaes L, Buntinx F, Banuro F, Lesaffre E, Heyrman J. Consequences of MRSA carriage in nursing home residents. Epidemiol Infect. 1999;122:235–9. doi: 10.1017/s0950268898001770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mulhausen PL, Harrell LJ, Weinberger M, Kochersberger GG, Feussner JR. Contrasting methicillin-resistant Staphylococcus aureus colonization in Veterans Affairs and community nursing homes. Am J Med. 1996;100:24–31. doi: 10.1016/s0002-9343(96)90007-8. [DOI] [PubMed] [Google Scholar]
- 9.Muder RR, Brennen C, Wagener MM, et al. Methicillin-resistant staphylococcal colonization and infection in a long-term care facility. Ann Intern Med. 1991;114:107–12. doi: 10.7326/0003-4819-114-2-1-107. [DOI] [PubMed] [Google Scholar]
- 10.Perl TM, Cullen JJ, Wenzel RP, et al. Intranasal mupirocin to prevent postoperative Staphylococcus aureus infections. N Engl J Med. 2002;346:1871–7. doi: 10.1056/NEJMoa003069. [DOI] [PubMed] [Google Scholar]
- 11.Peacock SJ, Mandal S, Bowler IC. Preventing Staphylococcus aureus infection in the renal unit. QJM. 2002;95:405–10. doi: 10.1093/qjmed/95.6.405. [DOI] [PubMed] [Google Scholar]
- 12.Kluytmans JA, Manders MJ, van Bommel E, Verbrugh H. Elimination of nasal carriage of Staphylococcus aureus in hemodialysis patients. Infect Control Hosp Epidemiol. 1996;17:793–7. doi: 10.1086/647238. [DOI] [PubMed] [Google Scholar]
- 13.Kluytmans JA, Mouton JW, VandenBergh MF, et al. Reduction of surgical-site infections in cardiothoracic surgery by elimination of nasal carriage of Staphylococcus aureus. Infect Control Hosp Epidemiol. 1996;17:780–5. doi: 10.1086/647236. [DOI] [PubMed] [Google Scholar]
- 14.Bradley SF. Effectiveness of mupirocin in the control of methicillin-resistant Staphylococcus aureus. Infections in Medicine. 1993;10:23–31. [Google Scholar]
- 15.Cederna JE, Terpenning MS, Ensberg M, Bradley SF, Kauffman CA. Staphylococcus aureus nasal colonization in a nursing home: eradication with mupirocin. Infect Control Hosp Epidemiol. 1990;11:13–6. doi: 10.1086/646072. [DOI] [PubMed] [Google Scholar]
- 16.Kauffman CA, Terpenning MS, He X, et al. Attempts to eradicate methicillin-resistant Staphylococcus aureus from a long-term care facility with the use of mupirocin ointment. Am J Med. 1993;94:371–8. doi: 10.1016/0002-9343(93)90147-h. [DOI] [PubMed] [Google Scholar]
- 17.Simor AE, Augustin A, Ng J, Betschel S, McArthur M. Control of MRSA in a long-term care facility. Infect Control Hosp Epidemiol. 1994;15:69–70. doi: 10.1086/646861. [DOI] [PubMed] [Google Scholar]
- 18.Kotilainen P, Routamaa M, Peltonen R, et al. Eradication of methicillin-resistant Staphylococcus aureus from a health center ward and associated nursing home. Arch Intern Med. 2001;161:859–63. doi: 10.1001/archinte.161.6.859. [DOI] [PubMed] [Google Scholar]
- 19.Katz S, Ford AB, Moskowitz RW, et al. Studies of illness in the aged. The index of ADL: a standardized measure of biological and psychological function. JAMA. 1963;185:914–9. doi: 10.1001/jama.1963.03060120024016. [DOI] [PubMed] [Google Scholar]
- 20.Garner JS, Jarvis WR, Emori TG, Horan TC, Hughes JM. CDC definitions for nosocomial infections. Am J Infect Control. 1988;16:128–40. doi: 10.1016/0196-6553(88)90053-3. [DOI] [PubMed] [Google Scholar]
- 21.Finlay JE, Miller LA, Poupard JA. Interpretive criteria for testing susceptibility of staphylococci to mupirocin. Antimicrob Agents Chemother. 1997;41:1137–9. doi: 10.1128/aac.41.5.1137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bradley SF, Ramsey MA, Morton TM, Kauffman CA. Mupirocin resistance: clinical and molecular epidemiology. Infect Control Hosp Epidemiol. 1995;16:354–8. doi: 10.1086/647125. [DOI] [PubMed] [Google Scholar]
- 23.Ramsey MA, Bradley SF, Kauffman CA, Morton TM. Identification of a chromosomal location of the mupA gene encoding low level mupirocin resistance in staphylococcal isolates. Antimicrob Agents Chemother. 1996;40:2820–3. doi: 10.1128/aac.40.12.2820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Struelens MJ, Deplanno A, Godard C, Maes N, Surruys E. Epidemiologic typing and delineation of genetic relatedness of methicillin-resistant Staphylococcus aureus by macrorestriction analysis of genomic DNA by using pulsed-field electrophoresis. J Clin Microbiol. 1992;30:2599–605. doi: 10.1128/jcm.30.10.2599-2605.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Tenover FC, Arbeit RD, Goering RV, et al. Interpreting chromosomal DNA restriction patterns produced by pulsed-field gel electrophoresis: criteria for bacterial strain typing. J Clin Microbiol. 1995;33:2233–9. doi: 10.1128/jcm.33.9.2233-2239.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Andersen PK, Gill RD. Cox’s regression model for counting processes: a large sample study. Ann Statistics. 1982;10:1100–20. [Google Scholar]
- 27.Therneau TM, Grambsch PM. Modeling survival data: extending the Cox model. New York: Springer; 2000. [Google Scholar]
- 28.Parras F, Guerrero DC, Bouza E, et al. Comparative study of mupirocin and oral co-trimoxazole plus topical fusidic acid in eradication of nasal carriage of methicillin-resistant Staphylococcus aureus. Antimicrob Agents Chemother. 1995;39:175–9. doi: 10.1128/aac.39.1.175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Martin JN, Perdreau-Remington F, Kartalija M, et al. A randomized clinical trial of mupirocin in the eradication of Staphylococcus aureus nasal carriage in human immunodeficiency virus disease. J Infect Dis. 1999;180:896–9. doi: 10.1086/314949. [DOI] [PubMed] [Google Scholar]
- 30.Reagan DR, Doebbling BN, Pfaller MA, et al. Elimination of coincident Staphylococcus aureus nasal and hand carriage with intranasal application of mupirocin calcium ointment. Ann Intern Med. 1991;114:101–6. doi: 10.7326/0003-4819-114-2-101. [DOI] [PubMed] [Google Scholar]
- 31.Strausbaugh LJ, Jacobson C, Sewell DL, Potter S, Ward TT. Antimicrobial therapy for methicillin-resistant Staphylococcus aureus colonization in residents and staff of a Veterans Affairs nursing home care unit. Infect Control Hosp Epidemiol. 1992;13:151–9. doi: 10.1086/646499. [DOI] [PubMed] [Google Scholar]
- 32.Walsh TJ, Standiford HC, Reboli AC, et al. Randomized double-blinded trial of rifampin with either novobiocin or trimethoprim-sulfamethoxazole against methicillin-resistant Staphylococcus aureus colonization: prevention of antimicrobial resistance and effect of host factors on outcome. Antimicrob Agents Chemother. 1993;37:1334–42. doi: 10.1128/aac.37.6.1334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Harbarth S, Dharan S, Liassine N, Herrault P, Auckenthaler R, Pittet D. Randomized, placebo-controlled, double-blind trial to evaluate the efficacy of mupirocin for eradicating carriage of methicillin-resistant Staphylococcus aureus. Antimicrob Agents Chemother. 1999;43:1412–6. doi: 10.1128/aac.43.6.1412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Yu VL, Goetz A, Wagener M, et al. Staphylococcus aureus nasal carriage and infection in patients on hemodialysis: efficacy of antibiotic prophylaxis. N Engl J Med. 1986;315:91–6. doi: 10.1056/NEJM198607103150204. [DOI] [PubMed] [Google Scholar]
- 35.Doebbling BN, Reagen DR, Pfaller MA, Houston AK, Hollis RJ, Wenzel RP. Long-term efficacy of intranasal mupirocin ointment: a prospective cohort study of Staphylococcus aureus carriage. Arch Intern Med. 1994;154:1505–8. [PubMed] [Google Scholar]
- 36.Terpenning MS, Bradley SF, Wan JY, Chenoweth CE, Jorgensen KA, Kauffman CA. Colonization and infection with antibiotic-resistantbacteria in a long-term care facility. J Am Geriatr Soc. 1994;42:1062–9. doi: 10.1111/j.1532-5415.1994.tb06210.x. [DOI] [PubMed] [Google Scholar]
- 37.Bradley SF, Terpenning MS, Ramsey M, et al. Methicillin-resistant Staphylococcus aureus: colonization and infection in a long-term care facility. Ann Intern Med. 1991;115:417–22. doi: 10.7326/0003-4819-115-6-417. [DOI] [PubMed] [Google Scholar]
- 38.Boelaert JR, Van Landuyt HW, Godard CA, et al. Nasal mupirocin ointment decreases the incidence of Staphylococcus aureus bacteraemias in haemodialysis patients. Nephrol Dial Transplant. 1993;8:235–9. [PubMed] [Google Scholar]
- 39.Mylotte JM, Kahler L, Jackson E. Pulse nasal mupirocin maintenance regimen in patients undergoing continuous ambulatory peritoneal dialysis. Infect Control Hosp Epidemiol. 1999;20:741–5. doi: 10.1086/501575. [DOI] [PubMed] [Google Scholar]
- 40.Raz R, Miron D, Colodner R, et al. A 1-year trial of nasal mupirocin in the prevention of recurrent staphylococcal nasal colonization and skin infection. Arch Intern Med. 1996;156:1109–12. [PubMed] [Google Scholar]
- 41.Chow JW, Yu VL. Staphylococcus aureus nasal carriage in hemodialysis: its role in infection and approaches to prophylaxis. Arch Intern Med. 1989;149:1258–62. [PubMed] [Google Scholar]
- 42.Watanakunakorn C, Brandt J, Durkin P, Santore S, Bota B, Stahl CJ. The efficacy of mupirocin ointment and chlorhexidine body scrubs in the eradication of nasal carriage of Staphylococcus aureus among patients undergoing long-term hemodialysis. Am J Infect Control. 1992;20:138–41. doi: 10.1016/s0196-6553(05)80180-4. [DOI] [PubMed] [Google Scholar]
- 43.Dacre J, Emmerson AM, Jenner EA. Gentamicin-methicillin–resistant Staphylococcus aureus: epidemiology and containment of an outbreak. J Hosp Infect. 1986;7:130–6. doi: 10.1016/0195-6701(86)90055-1. [DOI] [PubMed] [Google Scholar]
- 44.Perez-Fontan M, Garcia-Falcon T, Rosales M, et al. Treatment of Staphylococcus aureus nasal carriers in continuous ambulatory peritoneal dialysis with mupirocin: long-term results. Am J Kidney Dis. 1993;22:708–12. doi: 10.1016/s0272-6386(12)80434-3. [DOI] [PubMed] [Google Scholar]
- 45.Tuffnell DJ, Croton RS, Hemingway DM, Hartley MN, Wake PN, Garvey RJ. Methicillin resistant Staphylococcus aureus; the role of antisepsis in the control of an outbreak. J Hosp Infect. 1987;10:255–9. doi: 10.1016/0195-6701(87)90006-5. [DOI] [PubMed] [Google Scholar]
- 46.Naimi TS, LeDell KH, Boxrud DJ, et al. Epidemiology and clonality of community-acquired methicillin-resistant Staphylococcus aureus in Minnesota, 1996–1998. Clin Infect Dis. 2001;33:990–6. doi: 10.1086/322693. [DOI] [PubMed] [Google Scholar]
- 47.Cookson BD. Methicillin-resistant Staphylococcus aureus in the community: new battlefronts, or are the battles lost? Infect Control Hosp Epidemiol. 2000;21:398–403. doi: 10.1086/501781. [DOI] [PubMed] [Google Scholar]
- 48.Report of the Council on Scientific Affairs. American Medical Association white paper on elderly health. Arch Intern Med. 1990;150:2459–72. [PubMed] [Google Scholar]