Abstract
Oligonucleotide (2-aminoethyl)phosphonates in which the backbone consisted of isomerically pure, alternating (2-aminoethyl)-phosphonate and phosphodiester linkages have been prepared and characterized. One of these single isomer oligonucleotides (Rp) formed a more stable duplex with DNA or RNA than its corresponding natural counterpart. Hybrid stability was more pH-dependent, but less salt-dependent than a natural duplex. The specificity of hybridization was examined by hybridization of an oligonucleotide containing one (2-aminoethyl)phosphonate to oligonucleotides possessing mismatches in the region opposite to the aminoethyl group. In contrast to oligonucleotides containing (aminomethyl)-phosphonate linkages, oligonucleotide (2-aminoethyl)phosphonates were completely stable to hydrolysis in aqueous solution. These oligonucleotides were resistant to nuclease activity but did not induce RNase H mediated cleavage of a complementary RNA strand. Incubation in a serum-containing medium resulted in minimal degradation over 24 hours. Studies of cell uptake by flow cytometry and confocal microscopy demonstrated temperature dependent uptake and intracellular localization. (2-Aminoethyl)phosphonates represent a novel approach to the introduction of positive charges into the backbone of oligonucleotides.
Full text
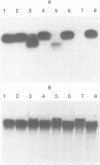
PDF







Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Arnold L. J., Jr Polylysine-drug conjugates. Methods Enzymol. 1985;112:270–285. doi: 10.1016/s0076-6879(85)12023-9. [DOI] [PubMed] [Google Scholar]
- Fathi R., Huang Q., Syi J. L., Delaney W., Cook A. F. (Aminomethyl)phosphonate derivatives of oligonucleotides. Bioconjug Chem. 1994 Jan-Feb;5(1):47–57. doi: 10.1021/bc00025a007. [DOI] [PubMed] [Google Scholar]
- Froehler B., Ng P., Matteucci M. Phosphoramidate analogues of DNA: synthesis and thermal stability of heteroduplexes. Nucleic Acids Res. 1988 Jun 10;16(11):4831–4839. doi: 10.1093/nar/16.11.4831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumagai A. K., Eisenberg J. B., Pardridge W. M. Absorptive-mediated endocytosis of cationized albumin and a beta-endorphin-cationized albumin chimeric peptide by isolated brain capillaries. Model system of blood-brain barrier transport. J Biol Chem. 1987 Nov 5;262(31):15214–15219. [PubMed] [Google Scholar]
- Lemaitre M., Bayard B., Lebleu B. Specific antiviral activity of a poly(L-lysine)-conjugated oligodeoxyribonucleotide sequence complementary to vesicular stomatitis virus N protein mRNA initiation site. Proc Natl Acad Sci U S A. 1987 Feb;84(3):648–652. doi: 10.1073/pnas.84.3.648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lesnikowski Z. J., Jaworska M., Stec W. J. Octa(thymidine methanephosphonates) of partially defined stereochemistry: synthesis and effect of chirality at phosphorus on binding to pentadecadeoxyriboadenylic acid. Nucleic Acids Res. 1990 Apr 25;18(8):2109–2115. doi: 10.1093/nar/18.8.2109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Löschner T., Engels J. W. Diastereomeric dinucleoside-methylphosphonates: determination of configuration with the 2-D NMR ROESY technique. Nucleic Acids Res. 1990 Sep 11;18(17):5083–5088. doi: 10.1093/nar/18.17.5083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller P. S., Dreon N., Pulford S. M., McParland K. B. Oligothymidylate analogues having stereoregular, alternating methylphosphonate/phosphodiester backbones. Synthesis and physical studies. J Biol Chem. 1980 Oct 25;255(20):9659–9665. [PubMed] [Google Scholar]
- Pardridge W. M., Boado R. J. Enhanced cellular uptake of biotinylated antisense oligonucleotide or peptide mediated by avidin, a cationic protein. FEBS Lett. 1991 Aug 19;288(1-2):30–32. doi: 10.1016/0014-5793(91)80996-g. [DOI] [PubMed] [Google Scholar]
- Pardridge W. M., Kumagai A. K., Eisenberg J. B. Chimeric peptides as a vehicle for peptide pharmaceutical delivery through the blood-brain barrier. Biochem Biophys Res Commun. 1987 Jul 15;146(1):307–313. doi: 10.1016/0006-291x(87)90726-1. [DOI] [PubMed] [Google Scholar]
- Pieken W. A., Olsen D. B., Benseler F., Aurup H., Eckstein F. Kinetic characterization of ribonuclease-resistant 2'-modified hammerhead ribozymes. Science. 1991 Jul 19;253(5017):314–317. doi: 10.1126/science.1857967. [DOI] [PubMed] [Google Scholar]
- Quartin R. S., Brakel C. L., Wetmur J. G. Number and distribution of methylphosphonate linkages in oligodeoxynucleotides affect exo- and endonuclease sensitivity and ability to form RNase H substrates. Nucleic Acids Res. 1989 Sep 25;17(18):7253–7262. doi: 10.1093/nar/17.18.7253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rychlik W., Rhoads R. E. A computer program for choosing optimal oligonucleotides for filter hybridization, sequencing and in vitro amplification of DNA. Nucleic Acids Res. 1989 Nov 11;17(21):8543–8551. doi: 10.1093/nar/17.21.8543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shoji Y., Akhtar S., Periasamy A., Herman B., Juliano R. L. Mechanism of cellular uptake of modified oligodeoxynucleotides containing methylphosphonate linkages. Nucleic Acids Res. 1991 Oct 25;19(20):5543–5550. doi: 10.1093/nar/19.20.5543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stevenson M., Iversen P. L. Inhibition of human immunodeficiency virus type 1-mediated cytopathic effects by poly(L-lysine)-conjugated synthetic antisense oligodeoxyribonucleotides. J Gen Virol. 1989 Oct;70(Pt 10):2673–2682. doi: 10.1099/0022-1317-70-10-2673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wagner E., Zenke M., Cotten M., Beug H., Birnstiel M. L. Transferrin-polycation conjugates as carriers for DNA uptake into cells. Proc Natl Acad Sci U S A. 1990 May;87(9):3410–3414. doi: 10.1073/pnas.87.9.3410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao Q., Matson S., Herrera C. J., Fisher E., Yu H., Krieg A. M. Comparison of cellular binding and uptake of antisense phosphodiester, phosphorothioate, and mixed phosphorothioate and methylphosphonate oligonucleotides. Antisense Res Dev. 1993 Spring;3(1):53–66. doi: 10.1089/ard.1993.3.53. [DOI] [PubMed] [Google Scholar]