ABSTRACT
Purpose: Best rehabilitation practices after hip fracture for people with dementia have not been established. A systematic review was conducted to determine current evidence for rehabilitation in this population, including residents in continuing care. Methods: Standardized review methodology was used to search eight databases for literature on hip-fracture rehabilitation for people with dementia. Eligible studies included participants with dementia who had a hip fracture; performed a rehabilitation intervention; and evaluated one or more of function, ambulation, discharge location, or falls. The Newcastle–Ottawa Scale was used to assess validity. Results: A total of 13 studies were included: five randomized controlled trials (RCTs), seven prospective cohort series, and one retrospective cohort study. Average quality ratings for RCTs and cohort studies were good and fair respectively. Participants with mild to moderate dementia receiving rehabilitation showed similar relative gains in function to those without dementia. Only one study examined the effect of rehabilitation among residents in continuing care. Conclusions: People with mild or moderate dementia may show improved function and ambulation and decreased fall risk after rehabilitation post hip fracture, similar to gains achieved by those without dementia. More research is required to ascertain the effect of rehabilitation in people with moderate to severe dementia, including those residing in continuing-care settings.
Key Words: dementia, geriatrics, hip fractures, rehabilitation
RÉSUMÉ
Objectif : Il n'y a pas de pratiques exemplaires d'établies pour la réadaptation après une fracture de la hanche chez les personnes aux prises avec la démence. Nous avons procédé à une revue systématique en vue de recueillir les faits cliniques relatifs à la réadaptation chez ce segment de la population, y compris les personnes en soins prolongés. Méthode : Une méthodologie d'examen de documents normalisée a été utilisée pour effectuer une recherche dans huit bases de données afin de recueillir de la documentation sur la réadaptation après une fracture de la hanche chez les personnes souffrant de démence. Les études admissibles traitaient de patients avec démence qui avaient subi une fracture de la hanche; auprès de qui on avait procédé à une intervention en réadaptation; et où au moins une fonction, la marche, le site du congé ou les risques de chutes avaient été évalués. L'échelle de Newcastle–Ottawa a été utilisée aux fins d'évaluation de la validité de ces études. Résultats : Au total, 13 études ont été répertoriées; cinq essais contrôlés randomisés (ECR), sept études de cohorte prospective et une étude de cohorte rétrospective. La qualité moyenne des ECR et des études de cohortes étaient respectivement bonne à moyenne. Les participants avec démence légère à modérée qui recevaient des traitements de réadaptation ont démontré des gains relatifs de fonction similaires à ceux qui ne souffraient pas de démence. Une seule de ces études s'est penchée sur les effets de la réadaptation chez les résidents d'établissements de soins prolongés. Conclusions : Les personnes souffrant de démence légère ou modérée ont démontré une fonction et une ambulation améliorées, de même qu'une réduction des risques de chutes après des soins en réadaptation à la suite d'une fracture de la hanche; ces gains étaient similaires chez les personnes non affectées par la démence. D'autres recherches seront nécessaires pour étudier les effets de la réadaptation chez les personnes souffrant de démence modérée à grave, y compris celles qui résident dans des établissements de soins prolongés.
Mots clés : démence, fracture de la hanche, maladie d'Alzheimer, patients gériatriques, réadaptation
Canada, like the rest of the Western world, is nearing what has been termed “an impending health care crisis”1 involving the ageing population. Projections indicate that by 2056, 29.5% of the Canadian population, or approximately 10.5 million people, will be 65 and over, more than double the population in the same age group in 2006.2
Hip fractures are common in the elderly population, and the absolute number of hip fractures is expected to rise as the number of older adults continues to increase.3 Between 2003 and 2004, 23,631 Canadians over 59 years of age were discharged from acute-care hospitals after being treated for a hip fracture.4 Indeed, hip fractures are one of the main causes of hospitalization in people over 655 and, relative to other types of osteoporotic fractures, result in the longest hospital stays and the highest costs.6 Furthermore, when patients with hip fractures leave the hospital, especially patients over 85, they are more likely to be dependent on others for activities of daily living (ADL) and less able to get around on their own.7
The current exponential increase in the proportion of individuals with dementia in Canada is also of particular concern in relation to the issue of hip rehabilitation, as cognitive impairment increases fall risk among community-dwelling older adults.8 In 2006, 2.0% of Canadians were diagnosed with dementia,2 and this proportion is expected to increase. People with moderate to severe dementia often become unable to live independently and require ongoing care in continuing-care facilities (e.g., nursing homes, auxiliary hospitals). In 2008–2009, between 35% and 60% of residents in continuing-care facilities in Canada had been diagnosed with dementia.9 Residents with dementia in continuing-care facilities have been reported to fall more often than those without dementia.10 Since up to 25% of hip fractures are reported to occur in continuing care facilities, this group represents a substantial proportion of the hip-fracture population.11,12
Further, people with moderate to severe dementia have been reported to have poor mobility and discharge outcomes following hip fracture.13 They have also been reported to have shorter hospital stays and to receive less rehabilitation, and specifically less physical therapy (PT).11,14 A study by Bellelli and colleagues15 showed that among patients with hip fracture admitted to a rehabilitation unit, those who were older and had a higher level of cognitive impairment received fewer rehabilitation interventions. Despite these reports, however, there exists a consensus that effective rehabilitation with respect to mobility and function for people who have sustained hip fractures, whether or not they also have dementia, is crucial to their return to their previous dwelling places in the community.16–22 Several strategies have been suggested for health care professionals to reduce the negative impact of cognitive and behavioural symptoms, thereby improving prognoses for these patients.23,24 However, not all health care professionals are aware of strategies for working with patients with dementia or put these strategies into practice.25
Given the growing population of older adults in Canada, and the resultant increase in the population with dementia, a greater understanding of effective rehabilitation following a hip fracture for this population will benefit both individual patients and Canadian society as a whole. Our research therefore sought to determine the current state of the evidence for rehabilitation in patients with dementia who have had a hip fracture by performing a systematic literature review. Our primary objective was to examine the best available evidence on rehabilitation practices for this population. In addition, we aimed to determine what evidence exists specific to rehabilitation management of patients with dementia who reside in continuing-care settings, since more than half of individuals with dementia have been reported to live in continuing-care facilities.1,26
METHODS
Literature Search Strategy
We conducted a systematic review to answer the following research question: In elderly hip fracture patients with dementia, what is the evidence for rehabilitation interventions, in addition to only medical management, with respect to (a) regaining function, (b) ambulation, (c) discharge location, and (d) reducing fall risk?
There was no restriction on study design, as we expected to find few published papers in this area. The Medline, CINAHL, Cochrane, Embase, PEDro, PsychINFO, Web of Science, and Scopus databases were searched for potential articles published up to November 2010. A health sciences librarian assisted in developing the search strategy (outlined in Appendix 1). An expert in the field was contacted to identify additional relevant articles not retrieved through the database search. Studies were included if they (1) involved hip-fracture patients with dementia; (2) investigated rehabilitation interventions in the fields of physical (PT), occupational (OT), or recreational therapy; and (3) evaluated at least one of the following outcomes: function, ambulation, discharge location, or falls. Articles were excluded if they (1) replied to or commented on an intervention article or (2) were solely descriptive in nature and did not describe a rehabilitation intervention.
Article Evaluation
Articles identified in the literature search were blinded to allow viewing of the title only, and three independent reviewers evaluated all titles to determine whether they met the inclusion criteria. When titles did not give sufficient information to make a decision, or when reviewers disagreed, the articles were carried forward to abstract evaluation. Articles that passed the initial title evaluation were again blinded to allow viewing of the abstract only. These articles were divided into two groups, and each was evaluated by two independent reviewers, using a standardized data sheet, to determine whether they met the inclusion criteria. When abstracts were not available for review or did not give sufficient information to make a decision, or when there was disagreement between reviewers, the articles were included in the full-text evaluation. At this stage the remaining articles were again divided into two groups, and two independent reviewers evaluated each article to determine inclusion. When the reviewers disagreed, a third party was introduced to determine the article's eligibility. Kappa values27 were calculated for agreement between reviewers at the abstract and full-text evaluation levels.
Data Extraction and Validity Assessment
For articles included after the full-text review, data on study methodology and results were extracted using a standardized data-extraction form. Validity assessment was performed using the Newcastle–Ottawa Quality Assessment Scale for Cohort Studies (NOS),28 as cohort studies were the primary study type found. We chose the NOS because it can be used for both randomized and non-randomized studies, which allowed us to use a single rating scale across all studies for more direct quality comparison. We followed the QUOROM guidelines 29 and did not use a formal risk-of-bias tool.
RESULTS
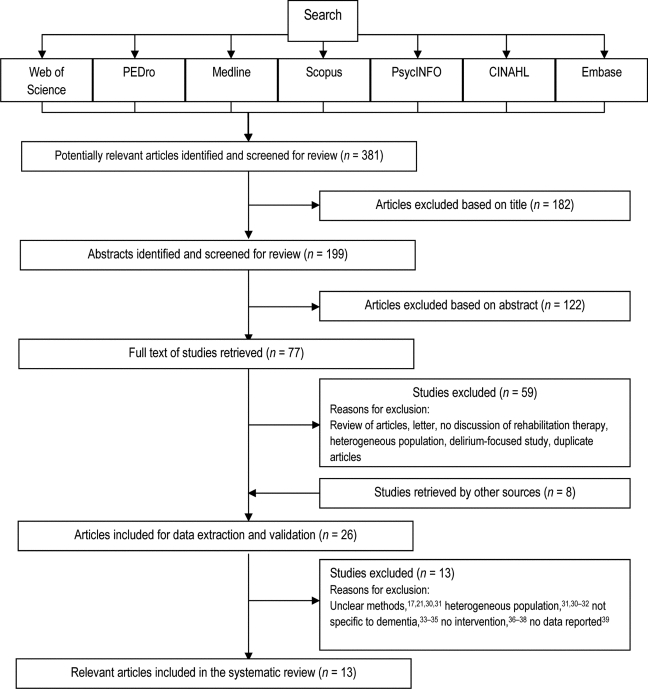
A total of 381 articles were identified after the initial search (see Figure 1). Of these, 199 abstracts were identified as potentially relevant studies based on key search terms and an initial title screening. Following this initial screening, 77 articles were carried forward to the full-text review, and 18 were retrieved for detailed analysis. Because of the large number of geriatric, internal medicine, and rehabilitation journals that publish these studies, hand searching was not undertaken. Through contact with an expert in the field, 8 additional studies were included for data extraction and validation, for a total of 26. Of these 26 articles, 13 met the inclusion criteria: five randomized clinical trials (RCTs), seven prospective cohort series, and one retrospective cohort study. Articles carried forward to full-text review were excluded for having unclear methods,17,21,30,31 using heterogeneous populations,21,30–32 not reporting data specifically for dementia patients,33–35 not describing a rehabilitation intervention,36–38 or not reporting sufficient outcome data related to our patient population of interest.39 Agreement between reviewers at the abstract level was moderate for pair 1 (κ=0.53) and fair for pair 2 (κ=0.31). Agreement between reviewers at full-text evaluation was almost perfect for pair 1 (κ=0.94) and substantial for pair 2 (κ=0.75).
Figure 1.
Flow Diagram of Study Selection.
Methodological details of the included studies are presented in Table 1, and NOS scores from the methodological evaluation in Table 2. Scores were out of a possible 8 points; studies scoring ≥7 were considered good-quality studies, those scoring 5–6 of fair quality, and those scoring ≤4 of poor quality. The average scores of the RCTs and cohort studies were 7.5/8 and 6.1/8 respectively.
Table 1.
Methodological Details of Studies Included in Systematic Review
| Study | Design | Sample size | Mean age, y | Sex; no. of female participants and (%) |
Dementia assessment |
Follow-up |
|---|---|---|---|---|---|---|
| Giusti et al.45 | Prospective longitudinal cohort |
n=96 (home-based rehabilitation, n=41; institution-based rehabilitation, n=55); 96 patients with dementia | Home-based group: 84.1 (SD 5.4); institution-based group: 84.4 (SD 6.9) |
Home-based group: 36 (88); institution-based group: 49 (89) | SPMSQ | 3, 6, and 12 mo |
| Goldstein et al.41 | Prospective longitudinal cohort |
n=58 (35 with impaired cognition [18 mild, 9 moderate, 8 severe]) |
84.0 (SD 6.7); 71–99 | 48 (83) | MDRS | ∼1 mo |
| Heruti et al.47 | Prospective longitudinal cohort |
n=204 (173 with reported outcomes); 54 with impaired cognition; 199 normal cognition | 80 (SD 7.1); 65–97 | 156 (76) | MMSE | ∼1 mo |
| Horgan and Cunningham13 |
Prospective | n=59 | No mean provided; median=80; IQR 73–87 |
54 (92) | AMTS | None |
| Huusko et al.42 | Randomized controlled trial |
n=243 (132 with cognitive impairment [68 mild, 36 moderate, 28 severe]; 97 normal cognition) | Intervention group: 80 (67–92); control group: 80 (66–97) | Intervention group: 84 (70), control group: 90 (73) | MMSE | 3 mo and 1 y |
| Lenze et al.19 | Prospective cohort |
n=97 (20 [51%] SNF impaired cognition; 18 [31%] IRF impaired cognition) | 81.7 (SD 8.8) | 79 (81) | MMSE | 2 and 12 wk |
| McGilton et al.20 | Longitudinal retrospective |
n=31 (17 impaired cognition; 14 normal cognition) | 87 | 18 (58) | MMSE | None |
| Naglie et al.43 | Randomized controlled trial |
n=280 (74 impaired cognition; 205 normal cognition) | Interdisciplinary care group: 83.8 (SD 6.9); usual care group: 84.6 (SD 7.3) | Interdisciplinary care group: 109 (77); usual care group: 114 (83) | SPMSQ | 3 and 6 mo |
| Penrod et al.48 | Prospective cohort |
n=443 (93 impaired cognition; 350 normal cognition) | 81.4 (SD 8.7); 53–101 | 363 (82) | Self-reported as present or not present | 2 and 6 mo |
| Rolland et al.22 | Prospective cohort |
n=61 (8 impaired cognition [MMSE <20]; 23 possible impaired cognition [MMSE 20–27]; 10 without cognitive impairment [MMSE >27]) | MMSE <20: 87.6 (SD 7.2); MMSE 20–27: 83.9 (SD 6.8); MMSE >27: 77.6 (SD 7.4) |
MMSE <20: 25 (41); MMSE 20–27: 17 (28); MMSE >27: 8 (13) | MMSE | ∼2 mo |
| Stenvall et al.46 | Randomized controlled trial |
n=199 (64 impaired cognition; 135 normal cognition) | Intervention group: 82.3 (SD 6.6); control group: 82.0 (SD 5.9) | Intervention group: 74 (73); control group: 74 (76) | MMSE | Hospitalization period |
| Uy et al.40* | Randomized controlled trial |
n=11* (11 impaired cognition) | Intervention group 83; control group 80 | 11 (100) | SPMSQ | 1 and 4 mo |
| Vidan et al.44 | Randomized controlled trial |
n=319 (78 impaired cognition; 241 normal cognition; intervention group 39 [25%]; usual-care group 39 [24%]) | Intervention group: 81.7 (SD 7.8); usual care group: 82.6 (SD 7.4) | Intervention group: 131 (85); usual care group: 129 (79) | Goldman Scale | 3, 6, and 12 mo |
Restricted to nursing-home residents with cognitive impairment.
SPMSQ=Short Portable Mental Status Questionnaire; MDRS=Mattis Dementia Rating Scale; MMSE=Mini Mental State Exam; AMTS=Abbreviated Mental Test Score; IRF=in-patient rehabilitation facility; SNF=skilled nursing facility.
Table 2.
Newcastle–Ottawa Scale Scores from the Methodological Evaluation of Studies Included in Systematic Review
| Study | Selection (/4) | Comparability (/1) | Outcome (/3) | Total (/8) |
|---|---|---|---|---|
| Giusti et al.45 | 4 | 1 | 2 | 7 |
| Goldstein et al.41 | 3 | 1 | 1 | 5 |
| Heruti et al.47 | 4 | 1 | 0 | 5 |
| Horgan and Cunningham13 | 4 | 1 | 1 | 6 |
| Huusko et al.42 | 4 | 1 | 3 | 8 |
| Lenze et al.19 | 4 | 1 | 2 | 7 |
| McGilton et al.20 | 4 | 0 | 2 | 6 |
| Naglie et al.43 | 4 | 1 | 3 | 8 |
| Penrod et al.48 | 4 | 0 | 2 | 6 |
| Rolland et al.22 | 4 | 1 | 2 | 7 |
| Stenvall et al.46 | 4 | 1 | 2 | 7 |
| Uy et al.40 | 4 | 1 | 2 | 7 |
| Vidan et al.44 | 4 | 0.5 | 3 | 7.5 |
Assessment of Cognitive Impairment
Cognitive impairment was assessed using multiple tools (see Table 1). The Mini Mental State Examination (MMSE), the most common scale, was used in six studies. Cut-off scores to indicate impairment on the MMSE were not consistent across studies; further, some studies combined all levels of cognitive impairment into a single group, whereas others separated participants into multiple groups based on severity of impairment. In only one study40 were all participants cognitively impaired. Other assessment tools used in the literature reviewed were the Short Portable Mental Status Questionnaire (SPMSQ), Abbreviated Mental Test Score (AMTS), Goldman Scale, and Mattis Dementia Rating Scale (MDRS).
Interventions
Interventions most often took the form of interdisciplinary or multidisciplinary care; however, the details of the interventions were often not well described. Intervention data are listed in Table 1. Specialized interventions involved combinations of geriatrician internists, general practitioners, nurses, social workers, neuropsychologists, and occupational, physical, and speech therapists.41–44 Patients involved in specialized interventions participated in PT and OT at varying levels of intensity, frequencies, and durations. Three interventions included PT sessions carried out twice daily 5 days a week.22,42,43 Interventions specific to PT included strengthening and range-of-motion exercises and instruction to patients in gait and transfer training.45 Early mobilization within the first 24 hours following surgery was included in two studies.43,46 Fall risk factors were a focus of rehabilitation in one study.46 Other features of the interventions described included early participation in self-care43 and practising activities throughout the day with nurses.42
Function
Eight studies examined function following hip fracture in dementia patients (see Table 3). Giusti and colleagues45 compared institution-based rehabilitation with home-based rehabilitation for community-dwelling persons with dementia who sustained a hip fracture; they found similar recovery levels with respect to activities of daily living (ADL), as measured by the Barthel and Lawton indices, for both groups. Moreover, those receiving home-based rehabilitation showed a slightly lower degree of functional decline than those receiving institution-based rehabilitation. Lenze and colleagues19 studied the difference in functional outcomes, based on Functional Independence Measure (FIM) motor scores, between patients from in-patient rehabilitation facilities and those from skilled nursing facilities. They found no overall effect of cognitive impairment on functional outcomes for either type of rehabilitative facility. Previously, in 1997, Goldstein and colleagues41 examined FIM motor scores in dementia patients following hip fracture (at admission and discharge) and found that cognitively impaired patients displayed gains in specific FIM areas of self-care, sphincter control, and locomotion similar to those of cognitively intact patients. Patients with cognitive impairment showed improvement in mobility, but cognitively intact patients exhibited significantly greater absolute gains in mobility. Similarly, Heruti and colleagues47 found that higher cognitive status at admission was related to higher motor FIM scores at discharge, although even the patients with the greatest cognitive impairments showed gains in motor FIM scores. Motor FIM scores were lower in cognitively impaired patients, and the relative functional gain in patients with the lowest MMSE scores was significantly lower. McGilton and colleagues,20 also using the FIM as a measure of mobility in relation to cognitive status, found similar results in their longitudinal retrospective study of people undergoing in-patient rehabilitation. Rolland and colleagues22 found analogous results using the FIM and the Montebello Rehabilitation Factor Score (MRFS). Although cognitively intact patients scored higher on the MRFS than cognitively impaired patients at both admission and discharge, after adjustments, relative functional gain was not significantly different between the two groups. Vidan and colleagues44 found parallel results. In their study, the effect of geriatric intervention on short-term functional recovery was greater for cognitively intact patients than for those with dementia: although those without dementia demonstrated a greater improvement in function than those with dementia, both groups improved.
Table 3.
Interventions as Outcome Measures of Rehabilitation Following Hip Fracture in Dementia Patients in Studies Included in Systematic Review
| Study | Intervention | Outcomes assessed | Follow-up | Results |
|---|---|---|---|---|
| Function | ||||
| Giusti et al.45 | Home rehabilitation programme vs. institution-based rehabilitation | Functional recovery (BI) | 3, 6, and 12 mo | Comparable recovery of ADL in patients with dementia receiving either home-based rehabilitation or institutional rehabilitation |
| Goldstein et al.41 | In-patient geriatric rehabilitation unit | Functional recovery (FIM) | ∼1 mo | Similar gains in FIM scores between patients with impaired and normal cognition |
| Heruti et al.47 | Comprehensive in-patient rehabilitation | Functional recovery (FIM motor sub-scale) | ∼1 mo | Motor FIM scores were lower in cognitively impaired patients. Absolute FIM motor gains were similar in patients with impaired and normal cognition, but relative functional gain of patients in the lowest MMSE quartile was significantly lower. |
| Lenze et al.19 | Rehabilitation at skilled nursing or in-patient rehabilitation facility | Functional recovery (FIM) | 2 and 12 wk | No overall effect of cognitive impairment on functional outcomes, nor any effect of cognition on superior functional outcomes, in in-patient rehabilitation facilities compared with skilled nursing facilities |
| McGilton et al.20 | In-patient rehabilitation | Functional recovery (FIM motor sub-scale) |
Discharge | All patients improved in motor function post rehabilitation. |
| Rolland et al.22 | Rehabilitation in geriatric in-patient unit | Functional recovery (FIM, MRFS) |
∼2 mo | No difference in change in FIM scores between subjects with impaired cognition and those with normal cognition; absolute scores lower in cognitively impaired patients |
| Uy et al.40 | In-patient multidisciplinary rehabilitation programme | Functional recovery (BI) | 1 and 4 mo | Non-significant improvement in BI in the intervention group |
| Vidan et al.44 |
Intensive multidisciplinary geriatric intervention |
Functional independence (Katz ADL; FAC) |
3, 6, and 12 mo |
Those without dementia showed greater improvement in function than those with dementia, but both groups improved. |
| Ambulation | ||||
| Horgan and Cunningham13 | In-patient PT | Ambulation (EMS) | Discharge | Independent of the amount of PT treatment received, patients with mild to moderate dementia made similar gains to those with normal cognition. Patients with moderate to severe dementia did not make similar gains. |
| Naglie et al.43 | In-patient interdisciplinary care vs. usual postoperative care | Recovery to pre-fracture level of ambulation and transfers (modified BI) | 3 and 6 mo | More patients with dementia in the intervention group showed no decline in ambulation or transfers. |
| Penrod et al.48 | Various amounts of PT | Ambulation (FIM locomotion sub-scale) | 2 and 6 mo | More PT in the first days after hip fracture surgery was associated with significantly better locomotion at 2 mo post admission. |
| Uy et al.40 | In-patient multidisciplinary rehabilitation programme | Gait velocity (Timed 2.44 m walk) | 1 and 4 mo | Non-significant improvement in gait velocity in the intervention group |
| Vidan et al.44 |
Intensive multidisciplinary geriatric intervention |
Functional independence (Katz ADL; FAC) |
3, 6, and 12 mo |
Those without dementia had a greater improvement in ambulation than those with dementia, but both groups improved. |
| Discharge location | ||||
| Goldstein et al.41 | In-patient geriatric rehabilitation unit | Discharge location | ∼1 mo | The proportion of patients returning or not returning to the community was similar between cognitively intact and cognitively impaired patients. |
| Horgan and Cunningham13 | In-patient PT | Discharge location | Discharge | Cognitively impaired patients were less likely to be discharged home and more likely to be discharged to a nursing home at the end of acute treatment. |
| Huusko et al.42 | In-patient geriatric rehabilitation vs. usual postoperative care | Return to independent living (place of residence) | 3 mo and 1 y |
More patients with mild to moderate dementia in the intervention group returned to independent living. |
| McGilton et al.20 | In-patient rehabilitation | Discharge location | Discharge | Both groups were predominantly discharged to the community; 80% returned home; 11.8% of cognitively impaired patients were discharged to continuing care facilities. |
| Naglie et al.43 |
In-patient interdisciplinary care vs. usual postoperative care |
Return to pre-fracture residence |
3 and 6 mo |
More patients with dementia in the intervention group showed no change in place of residence at 6 mo. |
| Falls | ||||
| Stenvall et al.46 | Multidisciplinary intervention programme | Falls in overall cohort as well as in predefined sub-groups; injuries associated with falls |
Hospitalization period | Fall incidence and risk was significantly lower in the intervention group; fall incidence was significantly lower among patients with dementia. |
Restricted to nursing-home residents with cognitive impairment.
BI=Barthel Index; FIM=Functional Independence Measure; MRFS=Montebello Rehabilitation Factor Score; Katz ADL=Katz Index of Independence in Activities of Daily Living; FAC=Functional Ambulation Classification; EMS=Elderly Mobility Scale; PT=physiotherapy.
Ambulation
Five articles included ambulation as an outcome measure of rehabilitation gains (see Table 3). One study used the FIM as a measure of mobility in relation to cognitive status. Penrod and colleagues48 showed that dementia was not a significant predictor of ambulatory status, as indicated by FIM locomotion sub-scale scores (obtained via phone interviews at 2 and 6 months after hip fracture), for patients receiving rehabilitation after hip fracture. Other outcome measures used were the Elderly Mobility Scale (EMS), the Functional Ambulation Classification (FAC), the Barthel Index (BI), and gait velocity. In 2003, Horgan and Cunningham13 used the EMS to examine the effect of cognitive status on ambulation following hip fracture. After adjusting for age, gender, baseline EMS score, time to operation, time to start PT intervention after surgery, and amount of PT intervention using a multiple linear regression, they found better cognitive status at both admission and discharge to be significantly related to improvement in mobility. Independent of the amount of PT treatment received, patients with mild to moderate dementia made similar gains on the EMS relative to their cognitively intact counterparts, while patients with moderate to severe dementia did not. In 2005, Vidan and colleagues44 used the FAC to measure ambulation recovery in cognitively impaired patients at 3 and 6 months following discharge. They found that significantly more patients in the intervention group than in the control group reported FAC recovery. Naglie and colleagues43 examined ambulation using mean BI ambulation domain scores and mean instrumental ADL scores. More patients in the intervention group than in the usual care group showed no declines in ambulation status at 3 and 6 months post surgery. Finally, Uy and colleagues40 used the timed 2.44 m walk to measure gains in ambulatory capacity. This study ended prematurely, but the data collected showed a trend toward improved ambulation in cognitively impaired persons residing in nursing homes after they participated in a rehabilitation programme.40
Discharge Location
Place of residence at discharge was examined in five studies (see Table 3). These studies investigated participants' ability to return to their previous place of residence or, in most cases, to return to community living. In a prospective longitudinal study of people receiving in-patient rehabilitation after hip fracture, Goldstein and colleagues41 found that cognitively impaired patients living in the community (either alone or with a spouse or other family members) before admission were as likely as cognitively intact patients to return to their previous living arrangements in the community. Horgan and Cunningham13 also followed patients with and without dementia receiving acute in-patient PT to determine the relationship between cognitive impairment and discharge location. Their main finding was that discharge location differed significantly based on patients' AMTS: lower scores were significantly related to increased likelihood of discharge to a nursing home following treatment.
Three studies examined place of residence after discharge from hospital. Naglie and colleagues43 found that patients with mild to moderate cognitive impairment tended to benefit the most from interdisciplinary care as opposed to usual care and that, 6 months after discharge, these patients showed no difference in residential status relative to their cognitively intact counterparts. Huusko and colleagues42 found similar results when comparing discharge locations of patients with mild to severe dementia receiving a rehabilitation intervention in a geriatric ward to those of patients with mild to severe dementia discharged to a local hospital following surgery (control). Significantly more patients with mild to moderate dementia who received the intervention in the geriatric ward returned to living independently 3 months after discharge. Further, among patients with dementia who received the intervention, those with mild dementia were as successful at returning to their previous living arrangements as those with no dementia. The third study, by McGilton and colleagues,20 found that for both cognitively intact and cognitively impaired patients, the primary discharge location was back to the community, and 80% returned home; only 11.8% of cognitively impaired patients (vs. none of the cognitively intact patients) were discharged to continuing-care facilities.
Falls
One study examined the effect of a postoperative multidisciplinary multi-factorial intervention programme on in-patient falls and fall-related injuries in patients with dementia who sustained a hip fracture (see Table 3).46 The authors found that the incidence of falls was significantly lower in the intervention group, including those patients with dementia, than in the control group.
DISCUSSION
Although hip fractures are relatively common in the frail elderly population, there is limited evidence on how best to optimize recovery through rehabilitation. Further, despite the fact that dementia is common in patients with hip fracture, the evidence for best rehabilitation practices in this large population sub-group is sparse at best. Our systematic review found 13 studies of fair to good methodological quality that examined functional recovery, ambulation, and discharge location in cohorts that included subjects with some degree of cognitive impairment. Because the studies were heterogeneous not only in the rehabilitation interventions they evaluated but also in terms of dementia assessment and severity, study settings, outcome measures, and evaluation periods, no meta-analysis could be performed. Despite this heterogeneity, however, the studies consistently reported that mild to moderate dementia was not an impediment to rehabilitation following hip fracture: subjects with mild to moderate dementia exhibited similar relative gains in functional recovery to their cognitively intact counterparts.
Absolute functional recovery scores were lower for people with cognitive impairment than for the cognitively intact;22 however, similar gains in functional recovery and return to community living were seen between cognitively impaired and cognitively intact patients receiving in-patient rehabilitation services.13,22,41,47 Moreover, persons with cognitive impairment receiving specialized in-patient interdisciplinary or multidisciplinary rehabilitation demonstrated a higher return to independent living and less decline in ambulation and transfers than those with impaired cognition who received usual postoperative care.42,43 Multidisciplinary intervention also reduced both fall risk and the incidence of falls in people with dementia.46 Our findings are similar to those of a recent review by Muir and Yohannes,49 who also reported that adults with cognitive impairment receiving intensive in-patient rehabilitation after surgical repair of a hip fracture appear to show comparable gains in physical function to adults with intact cognition.
Despite this evidence, however, older adults with dementia living in continuing-care settings are often excluded from research studies; as a result, few studies in our review included either individuals living in continuing-care settings or those with severe dementia. People with severe cognitive impairment are more dependent with respect to basic ADL50 and are considered less likely to be able to respond to rehabilitation, as physical disability increases with increasing dementia.51 To date, however, little research has attempted to determine at what level the severity of cognitive impairment precludes any benefit from rehabilitation.
People with mild to moderate dementia are more likely to reside in the community, and this fact is reflected in the current research; most patients included in the studies we reviewed had mild to moderate dementia and were living in the community before their hip fracture. Many individuals with cognitive impairment reside in continuing-care facilities, and rehabilitation efforts do occur in these settings.1,26 When studies are limited to community-dwelling and/or independently living individuals and to those with mild to moderate dementia, a large proportion of people with dementia who may still benefit from rehabilitation following hip fracture are overlooked.
Only one study, by Uy and colleagues,40 focused on cognitively impaired individuals living in nursing homes. Although this study was terminated prematurely when changes in government regulations resulted in a different population of individuals living in nursing homes, the authors found a promising trend for improvement in function and ambulation with a rehabilitation programme; further research with this patient group is therefore warranted.
The amount of rehabilitation provided and the types of activities included in each intervention constitute another area of inconsistency within the literature reviewed. Interventions most often took the form of interdisciplinary or multidisciplinary care in which patients underwent therapy of intense duration (e.g., PT twice per day for 1 hour, 5 times per week). No specific treatments were defined. Similarly, a recent review by Chudyk and colleagues52 found a wide variation in hip-fracture rehabilitation practices: initiation, intensity, and duration of rehabilitation varied, and specific treatments were not well defined. However, the programmes had common goals based on outcomes measured and focused on improving function and, in particular, improving ambulatory status.
Finally, the studies reviewed used a variety of outcome measures; most commonly, the FIM and BI were used to quantify functional gain. Given the heterogeneity of outcome measures, it was difficult to synthesize the evidence. Developing a research toolkit specific to this population would be an effective way to streamline outcome measures.
Our systematic review highlights the paucity of information and the dearth of methodologically sound studies in this area, indicating a need for further research. We found little information on continuing-care rehabilitation practices, as much of the research focused on in-hospital rehabilitation. Understanding how rehabilitation currently occurs in continuing care settings—for instance, determining specific rehabilitation interventions and protocols—would help to guide further research. Moreover, this research would encourage the use of more relevant outcome measures in continuing-care settings.
Our systematic review has some limitations. First, although RCTs provide the strongest evidence, only five relevant RCTs met our inclusion criteria, and these five RCTs and eight cohort studies provide insufficient Level 1 evidence to formulate strong conclusions about the impact of rehabilitation for people with impaired cognition. Second, although we did not complete any formal assessment of risk of bias, it is likely, based on the paucity of research in this area and the lack of randomized evidence, that there is some degree of bias in the current published evidence. Third, the methodological quality of the included studies ranged from poor to good, as scored on the NOS, and although the NOS has been shown to be appropriate for use in systematic reviews, its validity and reliability have not been documented.53 Moreover, while the NOS was constructed primarily for use in cohort studies, in this review it was used to compare both RCTs and cohort studies.
CONCLUSION
Our systematic review found evidence to support the effectiveness of rehabilitation following a hip fracture in patients with mild to moderate dementia. There is little research evidence supporting current rehabilitation practice in continuing-care facilities, although the rehabilitation goals appear similar to those reported in other settings.40 Despite the limited research evidence, however, we saw consistent findings across the studies reviewed: cognitive impairment may require the implementation of strategies to manage cognitive and behavioural issues, but it does not prevent rehabilitation from having a positive effect on recovery of function and ambulation.
More research is needed on standardization of outcome measures and potential simplification of outcome evaluation for patients with dementia, with a focus on function and a return to ambulation. Furthermore, research is needed on the effectiveness of rehabilitation in patients with severe dementia. More research in this area will result in a better understanding of rehabilitation practices in cognitively impaired hip-fracture patients. With additional evidence, this frail elderly population will be less likely to be excluded, on the basis of impaired cognition, from rehabilitation programmes and future research.
KEY MESSAGES
What Is Already Known on This Topic
Hip fractures are common in older adults. Those with dementia are at a higher risk of falling, and thus of sustaining a hip fracture, than those without dementia, but people with dementia are often excluded from studies of hip-fracture recovery.
What This Study Adds
Following a hip fracture, patients with mild to moderate dementia who received rehabilitation show similar relative gains in function to patients without dementia. More research is needed to determine the effect of rehabilitation following hip fracture in patients who reside in continuing-care settings and those with severe dementia.
Appendix 1: Search Strategy
- Medline
- exp Hip Fractures/
- ((hip or hips or trochanter or intertrochanter or proximal femur or proximal femoral) adj5 fractur*).mp. [mp=title, original title, abstract, name of substance word, subject heading word]
- 1 or 2
- exp Dementia/
- exp Delirium, Dementia, Amnestic, Cognitive Disorders/
- 4 or 5
- 6 and 3
- rh.fs. or rehabilitat*.mp. [mp=title, original title, abstract, name of substance word, subject heading word]
- 8 and 7
- limit 9 to (“all aged (65 and over)” or “aged (80 and over)”)
- limit 10 to English language
- CINAHL
- (MH Hip fractures)
- (MH “Hip Fractures+”) or ((hip OR hips OR trochanteric OR intertrochanteric OR proximal femur OR proximal femoral) AND fracture*)
- (MH “Dementia+”) OR (MH “Delirium, Dementia, Amnestic, Cognitive Disorders+”)
- (MH “Rehabilitation+”) or rehab
- (MH “Rehabilitation+”) or rehab* Narrow by Subject Age: −Aged: 65+ years
- S1 and S2 and S3 and S5
- (MH “Rehabilitation+”) or rehab* Narrow by Subject Age: Aged, 80 and over
- S1 and S2 and S3 and S7
- S6 OR S8
- Embase
- exp Hip Fractures/
- ((hip or hips or trochanter or intertrochanter or proximal femur or proximal femoral) adj5 fractur*).mp. [mp=title, abstract, subject headings, heading word, drug trade name, original title, device manufacturer, drug manufacturer name]
- 1 or 2
- rh.fs. or rehabilitat*.mp. [mp=title, abstract, subject headings, heading word, drug trade name, original title, device manufacturer, drug manufacturer name]
- exp confusion/ or exp delirium/ or dementia/ or exp senile dementia/ or exp memory disorder/
- 4 and 3 and 5
- limit 6 to aged <65+ years>
- limit 7 to English language
-
PEDro
Abstract & Title: fractur* dement*, Body Part: Thigh or Hip
- PsychINFO
- exp Injuries/ and exp Hips/ and exp Rehabilitation/
- hip fracture*.mp.
- 1 or 2
- exp Geriatric Patients/
- limit 3 to (“380 aged <age 65 yrs and older>” or “390 very old <age 85 yrs and older>”)
- 4 and 3
- 6 or 5
- exp dementia/
- exp Memory/ or exp Cognitive Impairment/ or exp Alzheimers Disease/
- 8 or 9
- 7 and 10
-
Web of Science
This database will be used for citation tracking after the full text evaluation is complete.
-
Scopus
(TITLE-ABS-KEY((delirium OR dementia* OR amnesi* OR “cognitive disorder” OR “cognitive disorders”) AND rehab*) AND TITLE-ABS-KEY((hip OR hips OR trochanter OR intertrochanter OR “proximal femur” OR “proximal femoral”) AND fracture*))
This was limited to English language.
Physiotherapy Canada 2012; 64(2);190–201; doi:10.3138/ptc.2011-06BH
References
- 1.Macdonald A, Cooper B. Long-term care and dementia services: an impending crisis. Age Ageing. 2007;36(1):16–22. doi: 10.1093/ageing/afl126. doi: 10.1093/ageing/afl126. Medline:17175565. [DOI] [PubMed] [Google Scholar]
- 2.Turcotte M, Schellenberg G. A portrait of seniors in Canada [Internet] Ottawa: Statistics Canada; 2006. [cited 2011 Aug 9]. Available from: http://www.statcan.gc.ca/pub/89-519-x/89-519-x2006001-eng.pdf. [Google Scholar]
- 3.Papadimitropoulos EA, Coyte PC, Josse RG, et al. Current and projected rates of hip fracture in Canada. CMAJ. 1997;157(10):1357–63. Medline:9371065. [PMC free article] [PubMed] [Google Scholar]
- 4.Canadian Community Health Survey. Detailed information for 2003 (Cycle 2.1) [Internet] Ottawa: Statistics Canada; 2004. Jun 15, [cited 2011 Aug 9]. Available from: http://www.statcan.gc.ca/cgi-bin/imdb/p2SV.pl?Function=getSurvey&SurvId=3226&SurvVer=0&SDDS=3226&InstaId=15282&InstaVer=2&lang=en&db=imdb&adm=8&dis=2. [Google Scholar]
- 5.Peel NM, Kassulke DJ, McClure RJ. Population based study of hospitalised fall related injuries in older people. Inj Prev. 2002;8(4):280–3. doi: 10.1136/ip.8.4.280. doi: 10.1136/ip.8.4.280. Medline:12460962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Maravic M, Le Bihan C, Landais P, et al. Incidence and cost of osteoporotic fractures in France during 2001. A methodological approach by the national hospital database. Osteoporos Int. 2005;16(12):1475–80. doi: 10.1007/s00198-005-2031-0. doi: 10.1007/s00198-005-2031-0. Medline:16217587. [DOI] [PubMed] [Google Scholar]
- 7.Stenvall M, Elinge E, von Heideken Wågert P, et al. Having had a hip fracture—association with dependency among the oldest old. Age Ageing. 2005;34(3):294–7. doi: 10.1093/ageing/afi042. doi: 10.1093/ageing/afi042. Medline:15863415. [DOI] [PubMed] [Google Scholar]
- 8.Tinetti ME, Doucette JT, Claus EB. The contribution of predisposing and situational risk factors to serious fall injuries. J Am Geriatr Soc. 1995;43(11):1207–13. doi: 10.1111/j.1532-5415.1995.tb07395.x. Medline:7594153. [DOI] [PubMed] [Google Scholar]
- 9.Continuing Care Reporting System. 2008/2009. Available from: https://secure.cihi.ca/estore/productFamily.htm?pf=PFC1631&locale=en&lang=EN&mediatype=0.
- 10.van Doorn C, Gruber-Baldini AL, Zimmerman S, et al. Epidemiology of Dementia in Nursing Homes Research Group. Dementia as a risk factor for falls and fall injuries among nursing home residents. J Am Geriatr Soc. 2003;51(9):1213–8. doi: 10.1046/j.1532-5415.2003.51404.x. doi: 10.1046/j.1532-5415.2003.51404.x. Medline:12919232. [DOI] [PubMed] [Google Scholar]
- 11.Beaupre LA, Cinats JG, Jones CA, et al. Does functional recovery in elderly hip fracture patients differ between patients admitted from long-term care and the community? J Gerontol Ser A Biol Sci Med Sci. 2007;62(10):1127–33. doi: 10.1093/gerona/62.10.1127. [DOI] [PubMed] [Google Scholar]
- 12.Ronald LA, McGregor MJ, McGrail KM, et al. Hospitalization rates of nursing home residents and community-dwelling seniors in british columbia. Can J Aging. 2008;27(1):109–15. doi: 10.3138/cja.27.1.109. doi: 10.3138/cja.27.1.109. Medline:18492642. [DOI] [PubMed] [Google Scholar]
- 13.Horgan NF, Cunningham CJ. Impact of cognitive impairment on hip fracture outcome in older people … including commentary by Sherrington C. Int J Ther Rehabil. 2003;10(5):228–32. [Google Scholar]
- 14.Crotty M, Miller M, Whitehead C, et al. Hip fracture treatments–what happens to patients from residential care? J Qual Clin Pract. 2000;20(4):167–70. doi: 10.1046/j.1440-1762.2000.00385.x. doi: 10.1046/j.1440-1762.2000.00385.x. Medline:11207957. [DOI] [PubMed] [Google Scholar]
- 15.Bellelli G, Frisoni GB, Pagani M, et al. Does cognitive performance affect physical therapy regimen after hip fracture surgery? Aging Clin Exp Res. 2007;19(2):119–24. doi: 10.1007/BF03324677. Medline:17446722. [DOI] [PubMed] [Google Scholar]
- 16.Barnes C, Conner D, Legault L, et al. Rehabilitation outcomes in cognitively impaired patients admitted to skilled nursing facilities from the community. Arch Phys Med Rehabil. 2004;85(10):1602–7. doi: 10.1016/j.apmr.2004.02.025. doi: 10.1016/j.apmr.2004.02.025. Medline:15468018. [DOI] [PubMed] [Google Scholar]
- 17.Beloosesky Y, Grinblat J, Epelboym B, et al. Functional gain of hip fracture patients in different cognitive and functional groups. Clin Rehabil. 2002;16(3):321–8. doi: 10.1191/0269215502cr497oa. doi: 10.1191/0269215502cr497oa. Medline:12017519. [DOI] [PubMed] [Google Scholar]
- 18.Halbert J, Crotty M, Whitehead C, et al. Hip Fracture Rehabilitation Trial Collaborative Group. Multi-disciplinary rehabilitation after hip fracture is associated with improved outcome: a systematic review. J Rehabil Med. 2007;39(7):507–12. doi: 10.2340/16501977-0102. doi: 10.2340/16501977-0102. Medline:17724548. [DOI] [PubMed] [Google Scholar]
- 19.Lenze EJ, Skidmore ER, Dew MA, et al. Does depression, apathy or cognitive impairment reduce the benefit of inpatient rehabilitation facilities for elderly hip fracture patients? Gen Hosp Psychiatry. 2007;29(2):141–6. doi: 10.1016/j.genhosppsych.2007.01.001. doi: 10.1016/j.genhosppsych.2007.01.001. Medline:17336663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.McGilton KS, Mahomed N, Davis AM, et al. Outcomes for older adults in an inpatient rehabilitation facility following hip fracture (HF) surgery. Arch Gerontol Geriatr. 2009;49(1):e23–31. doi: 10.1016/j.archger.2008.07.012. doi: 10.1016/j.archger.2008.07.012. Medline:18842307. [DOI] [PubMed] [Google Scholar]
- 21.Moncada LV, Andersen RE, Franckowiak SC, et al. The impact of cognitive impairment on short-term outcomes of hip fracture patients. Arch Gerontol Geriatr. 2006;43(1):45–52. doi: 10.1016/j.archger.2005.09.003. doi: 10.1016/j.archger.2005.09.003. Medline:16256217. [DOI] [PubMed] [Google Scholar]
- 22.Rolland Y, Pillard F, Lauwers-Cances V, et al. Rehabilitation outcome of elderly patients with hip fracture and cognitive impairment. Disabil Rehabil. 2004;26(7):425–31. doi: 10.1080/09638280410001663148. doi: 10.1080/09638280410001663148. Medline:15204479. [DOI] [PubMed] [Google Scholar]
- 23.McGilton K, Wells J, Davis A, et al. Rehabilitating patients with dementia who have had a hip fracture: part II: cognitive symptoms that influence care. Top Geriatr Rehabil. 2007;23(2):174–82. [Google Scholar]
- 24.McGilton K, Wells J, Teare G, et al. Rehabilitating patients with dementia who have had a hip frature—part I: behavioral symptoms that influence care. Top Geriatr Rehabil. 2007;23(2):161–73. [Google Scholar]
- 25.World Health Organization. WHO global report on falls prevention in older age [Internet] Geneva: The Organization; 2007. [cited 2011 Aug 9]. Available from: http://www.who.int/ageing/publications/Falls_prevention7March.pdf. [Google Scholar]
- 26.Canadian Institute for Health Information. Facility-based continuing care in Canada, 2004–2005: an emerging portrait of the continuum [Internet] Ottawa: The Institute; 2006. [cited 2010 Sep]. Available from: https://secure.cihi.ca/estore/productFamily.htm?locale=en&pf=PFC552&lang=fr&media=0. [Google Scholar]
- 27.Landis JR, Koch GG. The measurement of observer agreement for categorical data. Biometrics. 1977;33(1):159–74. doi: 10.2307/2529310. Medline:843571. [PubMed] [Google Scholar]
- 28.Wells GA, Shea B, O'Connell D, et al. The Newcastle–Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses [Internet] Ottawa: Ottawa Hospital Research Institute; n.d.. [cited 2011 Aug 9]. Available from: http://www.ohri.ca/programs/clinical_epidemiology/oxford.htm. [Google Scholar]
- 29.Moher D, Cook DJ, Eastwood S, et al. Improving the quality of reports of meta-analyses of randomised controlled trials: the QUOROM statement. Quality of Reporting of Meta-analyses. Lancet. 1999;354(9193):1896–900. doi: 10.1016/s0140-6736(99)04149-5. doi: 10.1016/S0140-6736(99)04149-5. Medline:10584742. [DOI] [PubMed] [Google Scholar]
- 30.Rösler A, Krause T, Niehuus C, et al. Dementia as a cofactor for geriatric rehabilitation-outcome in patients with osteosynthesis of the proximal femur: a retrospective, matched-pair analysis of 250 patients. Arch Gerontol Geriatr. 2009;49(1):1114–20. doi: 10.1016/j.archger.2008.08.003. doi: 10.1016/j.archger.2008.08.003. [DOI] [PubMed] [Google Scholar]
- 31.Resnick B, Daly MP. The effect of cognitive status on outcomes following rehabilitation. Fam Med. 1997;29(6):400–5. Medline:9193910. [PubMed] [Google Scholar]
- 32.Feng L, Scherer SC, Tan BY, et al. Comorbid cognitive impairment and depression is a significant predictor of poor outcomes in hip fracture rehabilitation. Int Psychogeriatr. 2010;22(2):246–53. doi: 10.1017/S1041610209991487. doi: 10.1017/S1041610209991487. Medline:19951458. [DOI] [PubMed] [Google Scholar]
- 33.Adunsky A, Lusky A, Arad M, et al. A comparative study of rehabilitation outcomes of elderly hip fracture patients: the advantage of a comprehensive orthogeriatric approach. J Gerontol Ser A Biol Sci Med Sci. 2003;58A(6):542–7. doi: 10.1093/gerona/58.6.m542. [DOI] [PubMed] [Google Scholar]
- 34.Arinzon Z, Shabat S, Peisakh A, et al. Gender differences influence the outcome of geriatric rehabilitation following hip fracture. Arch Gerontol Geriatr. 2010;50(1):86–91. doi: 10.1016/j.archger.2009.02.004. doi: 10.1016/j.archger.2009.02.004. Medline:19303648. [DOI] [PubMed] [Google Scholar]
- 35.Lenze EJ, Munin MC, Dew MA, et al. Adverse effects of depression and cognitive impairment on rehabilitation participation and recovery from hip fracture. Int J Geriatr Psychiatry. 2004;19(5):472–8. doi: 10.1002/gps.1116. doi: 10.1002/gps.1116. Medline:15156549. [DOI] [PubMed] [Google Scholar]
- 36.Al-Ani AN, Flodin L, Söderqvist A, et al. Does rehabilitation matter in patients with femoral neck fracture and cognitive impairment? a prospective study of 246 patients. Arch Phys Med Rehabil. 2010;91(1):51–7. doi: 10.1016/j.apmr.2009.09.005. doi: 10.1016/j.apmr.2009.09.005. Medline:20103396. [DOI] [PubMed] [Google Scholar]
- 37.Samuelsson BHM, Hedström MI, Ponzer S, et al. Gender differences and cognitive aspects on functional outcome after hip fracture—a 2 years' follow-up of 2,134 patients. Age Ageing. 2009;38(6):686–92. doi: 10.1093/ageing/afp169. doi: 10.1093/ageing/afp169. Medline:19767316. [DOI] [PubMed] [Google Scholar]
- 38.van Dortmont LM, Douw CM, van Breukelen AM, et al. Outcome after hemi-arthroplasty for displaced intracapsular femoral neck fracture related to mental state. Injury. 2000;31(5):327–31. doi: 10.1016/s0020-1383(99)00304-6. doi: 10.1016/S0020-1383(99)00304-6. Medline:10775686. [DOI] [PubMed] [Google Scholar]
- 39.Hamman RJ. Rehabilitation following hip fracture in patients with Alzheimer's disease and related disorders. Am J Alzheimer Dis. 1997;12(5):209–11. doi: 10.1177/153331759701200504. [Google Scholar]
- 40.Uy C, Kurrle SE, Cameron ID. Inpatient multidisciplinary rehabilitation after hip fracture for residents of nursing homes: a randomised trial. Australas J Ageing. 2008;27(1):43–4. doi: 10.1111/j.1741-6612.2007.00277.x. doi: 10.1111/j.1741-6612.2007.00277.x. Medline:18713215. [DOI] [PubMed] [Google Scholar]
- 41.Goldstein FC, Strasser DC. Hip fractures in older adults: cognitive factors related to a positive rehabilitation outcome [dissertation] Atlanta (GA): Emory University School of Medicine; 1997. [Google Scholar]
- 42.Huusko TM, Karppi P, Avikainen V, et al. Randomised, clinically controlled trial of intensive geriatric rehabilitation in patients with hip fracture: subgroup analysis of patients with dementia. BMJ. 2000;321(34):1107–11. doi: 10.1136/bmj.321.7269.1107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Naglie G, Tansey C, Kirkland JL, et al. Interdisciplinary inpatient care for elderly people with hip fracture: a randomized controlled trial. CMAJ. 2002;167(1):25–32. Medline:12137074. [PMC free article] [PubMed] [Google Scholar]
- 44.Vidán M, Serra JA, Moreno C, et al. Efficacy of a comprehensive geriatric intervention in older patients hospitalized for hip fracture: a randomized, controlled trial. J Am Geriatr Soc. 2005;53(9):1476–82. doi: 10.1111/j.1532-5415.2005.53466.x. doi: 10.1111/j.1532-5415.2005.53466.x. Medline:16137275. [DOI] [PubMed] [Google Scholar]
- 45.Giusti A, Barone A, Pioli G. Rehabilitation after hip fracture in patients with dementia. J Am Geriatr Soc. 2007;55(8):1309–10. doi: 10.1111/j.1532-5415.2007.01258.x. doi: 10.1111/j.1532-5415.2007.01258.x. Medline:17661980. [DOI] [PubMed] [Google Scholar]
- 46.Stenvall M, Olofsson B, Lundström M, et al. A multidisciplinary, multifactorial intervention program reduces postoperative falls and injuries after femoral neck fracture. Osteoporos Int. 2007;18(2):167–75. doi: 10.1007/s00198-006-0226-7. doi: 10.1007/s00198-006-0226-7. Medline:17061151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Heruti RJ, Lusky A, Barell V, et al. Cognitive status at admission: does it affect the rehabilitation outcome of elderly patients with hip fracture? Arch Phys Med Rehabil. 1999;80(4):432–6. doi: 10.1016/s0003-9993(99)90281-2. doi: 10.1016/S0003-9993(99)90281-2. Medline:10206606. [DOI] [PubMed] [Google Scholar]
- 48.Penrod JD, Boockvar KS, Litke A, et al. Physical therapy and mobility 2 and 6 months after hip fracture. J Am Geriatr Soc. 2004;52(7):1114–20. doi: 10.1111/j.1532-5415.2004.52309.x. doi: 10.1111/j.1532-5415.2004.52309.x. Medline:15209649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Muir SW, Yohannes AM. The impact of cognitive impairment on rehabilitation outcomes in elderly patients admitted with a femoral neck fracture: a systematic review. J Geriatr Phys Ther. 2009;32(1):24–32. doi: 10.1519/00139143-200932010-00006. doi: 10.1519/00139143-200932010-00006. Medline:19856633. [DOI] [PubMed] [Google Scholar]
- 50.McConnell ES, Pieper CF, Sloane RJ, et al. Effects of cognitive performance on change in physical function in long-stay nursing home residents. J Gerontol Ser A Biol Sci Med Sci. 2002;57(12):M778–84. doi: 10.1093/gerona/57.12.m778. doi: 10.1093/gerona/57.12.M778. [DOI] [PubMed] [Google Scholar]
- 51.Reisberg B, Kenowsky S, Franssen EH, et al. Towards a science of Alzheimer's disease management: a model based upon current knowledge of retrogenesis. Int Psychogeriatr. 1999;11(1):7–23. doi: 10.1017/s1041610299005554. doi: 10.1017/S1041610299005554. Medline:10189596. [DOI] [PubMed] [Google Scholar]
- 52.Chudyk AM, Jutai JW, Petrella RJ, et al. Systematic review of hip fracture rehabilitation practices in the elderly. Arch Phys Med Rehabil. 2009;90(2):246–62. doi: 10.1016/j.apmr.2008.06.036. doi: 10.1016/j.apmr.2008.06.036. Medline:19236978. [DOI] [PubMed] [Google Scholar]
- 53.Deeks JJ, Dinnes J, D'Amico R, et al. International Stroke Trial Collaborative Group; European Carotid Surgery Trial Collaborative Group. Evaluating non-randomised intervention studies. Health Technol Assess. 2003;7(27):iii–x. 1–173. doi: 10.3310/hta7270. Medline:14499048. [DOI] [PubMed] [Google Scholar]