Abstract
Rod-shaped bacteria such as Escherichia coli accurately maintain their shape from generation to generation. The cytoskeletal proteins MreB and FtsZ, which respectively guide parallel growth of the sidewall and perpendicular growth of the division septum, are important to maintain a straight sidewall and uniformly rounded cell poles. FtsZ normally assembles into a ring at the cell midpoint, called the Z ring, which is oriented perpendicular to the cell’s axis and is thus in perfect position to guide growth of a perpendicular septum. In this issue of Molecular Microbiology, Potluri et al. show that low molecular weight penicillin binding proteins, particularly PBP5, have a role in maintaining the perpendicular geometry of the Z ring and subsequent septum in E. coli. When these factors are absent or perturbed, division septa are readily deformed, which result in abnormal cell poles that often bifurcate over time to generate branches. The data suggest that cellular branching in E. coli is specifically induced by aberrant septation events caused by mis-oriented Z rings and not by deformation of a growing cell pole or emergence of new tips from the sidewall, which are likely mechanisms of branching in other bacterial families.
Watching normal Escherichia coli cells divide by binary fission, one is struck by how uniform their shape remains from one generation to the next. Every cell looks like a gelatin capsule, with flat sides and uniformly hemispherical cell poles (Nanninga, 1998) (den Blaauwen et al., 2008). The faithful partitioning of chromosomes and proper localization of protein machines likely depends on ensuring the same cell shape over and over again. Although E. coli can be forced to change its shape in response to physical constraints (Mannik et al., 2009), the dimensions of normal cells fall within a fairly narrow range at a given growth rate. How is this precision maintained over many generations? Because the shape of bacteria with cell walls is governed by how the wall is built (Koch, 1991), we can learn about cell shape by understanding how two major cytoskeletal proteins guide peptidoglycan (PG) synthesis (Typas et al., 2011).
One of these cytoskeletal proteins is MreB, a bacterial actin homolog (van den Ent et al., 2001). MreB is clearly important for guiding growth of the sidewall in rod shaped cells that have it, such as E. coli and Bacillus subtilis. Recent evidence suggests that complexes of MreB are distributed throughout the cell and shuttle in roughly circumferential directions in response to PG synthesis (van Teeffelen et al.) (Garner et al., 2011) (Dominguez-Escobar et al., 2011). As MreB can polymerize, it is likely that short, highly dynamic MreB polymers assemble on the inner surface of the cytoplasmic membrane, attached by N-terminal amphipathic membrane targeting domains (Salje et al.). Such MreB polymers likely reinforce proper directionality on the PG synthesis machinery via direct protein-protein interactions across the membrane (White et al., 2011). This is consistent with the roughly circumferential orientation of glycan strands in the sidewall (Gan et al., 2008). When MreB polymerization is perturbed, cell wall synthesis continues but the cell diameter is no longer constrained. Mutants of E. coli that lack MreB propagate as spheres, and resemble cocci such as Staphylococci or Neisseria, which naturally lack MreB. Interestingly, however, some rod-shaped bacterial species also lack MreB. The few species that have been tested so far grow by tip extension only (Brown et al., 2011); their sidewall no longer grows, and thus does not need MreB to regulate its growth (Fig. 1). How these species regulate the diameter of the growing tips is unknown, but they may rely on the existing shape of their cell wall as a template.
Fig. 1. Examples of rod-shaped bacteria that form branches naturally or under abnormal conditions.
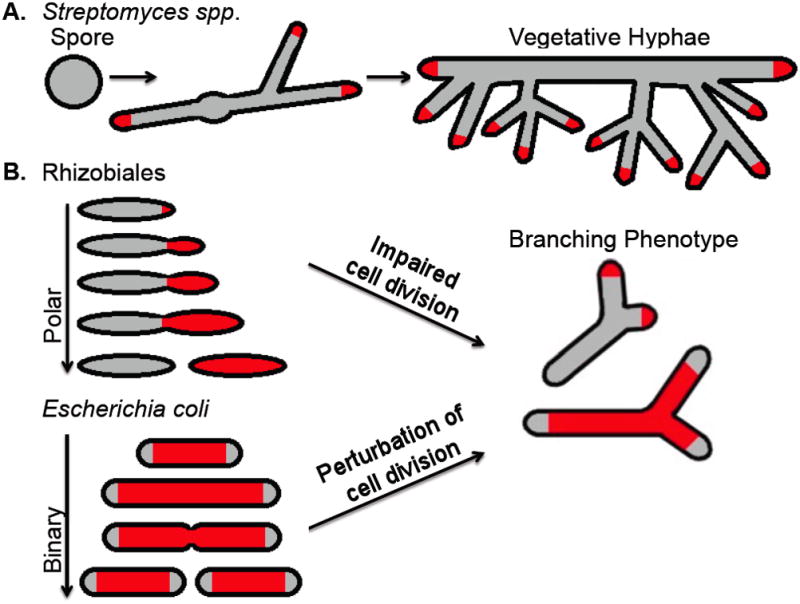
A. Streptomyces spores germinate and grow by tip extension (dependent on the DivIVA protein) and naturally form branched vegetative hyphae similar to fungi. B. Some Rhizobiales species (Agrobacterium tumefaciens, Sinorhizobium meliloti, and Brucella abortus) lack MreB and DivIVA and grow at only one cell pole (Brown et al., 2011); they exhibit a branched cell phenotype when their cell cycle is perturbed. Although they grow completely differently, Escherichia coli can also branch when their cell division cycle is perturbed. Active PG growth is denoted by red shading.
The other cytoskeletal protein with a major role in cell shape is the bacterial tubulin, FtsZ. Although it has a minor MreB-independent role in sidewall synthesis (Varma and Young, 2009), FtsZ’s main function is to orchestrate the 90° switch from synthesis of the sidewall to synthesis of the division septum (Margolin, 2009). It does so by assembling into a polymeric Z ring at mid-cell and recruiting a large protein machine, often called the divisome, that includes the septal transpeptidase PBP3 and cell wall amidases that help daughter cells to separate (Uehara and Bernhardt). Some components of the sidewall synthesis machine are also borrowed for septal synthesis, suggesting that the two machines directly compete for parts (Typas et al., 2011). After cell separation, former division septa become cell poles and no longer are active in PG synthesis; this inactive wall is called inert PG (de Pedro et al., 1997). It is notable the Z ring also directs the deposition of preseptal PG prior to the divisome-dependent septal PG (Rothfield, 2003). Inactivation of FtsZ causes E. coli cells to form long, septum-free filaments that maintain their rod shape over long distances, indicating that MreB-dependent sidewall synthesis is not significantly affected.
Nevertheless, altering FtsZ activity sometimes affects overall rod shape of E. coli. For example, a mutant FtsZ that makes spiral-shaped Z rings also has spiral-shaped division septa and cell poles (Addinall and Lutkenhaus, 1996). Cells overproducing another mutant FtsZ defective in assembly dynamics tend to form branches (Yu and Margolin, 2000), and inhibiting FtsZ assembly in the absence of low molecular weight penicillin-binding proteins (LWM PBPs) induces cells to grow as striking spirillum-like spirals (Varma and Young, 2004). These observations lead to several questions: How might FtsZ be influencing sidewall growth? How do extra cell poles, visible as branches, occur in E. coli cells that normally have only two poles.
Clues for answering these questions come from the LMW PBPs, which function to modify PG by trimming amino acid linkages from the glycan side chains. For example, PBP5, PBP6 and DacD are a D,D carboxypeptidases that remove terminal D-alanines from those side chains, whereas PBP4 and 7 are endopeptidases (Ghosh et al., 2008). Although they are not essential for viability, inactivation of one or more of these proteins induces significant shape changes in otherwise wild-type cells. Removal of PBP5 has the strongest effect, and increases the effects of removing one or more of the others (Nelson and Young, 2001). Cells of multiple PBP mutants are often bizarrely shaped, with branches being very frequent (Young, 2011). While branching is common in filamentous fungi and bacteria such as Streptomyces, it is normally not observed at significant frequencies in E. coli (Fig. 1). Therefore, one reasonable hypothesis is that a major function of LMW PBPs in E. coli is to prevent unwanted ectopic cell poles from emerging, and this function is somehow tied with normal activity and/or levels of FtsZ.
Potluri et al. first tested this hypothesis by asking whether increasing levels of FtsZ in cells defective for one or more of the LMW PBPs could increase branching. Surprisingly, they found that ~3-fold excess of FtsZ in otherwise wild-type cells increased the chances of branch formation. Similar levels of FtsZ in PBP knockout mutants, particularly cells lacking PBP5, significantly increased chances of branching and even formation of multiple branches per cell. Previous studies of cells with excess wild-type FtsZ did not observe significant numbers of branched cells, possibly because these cells were too short to see branches as they divide both at the proper mid-cell location and at the cell poles. In addition, at much higher levels of FtsZ, cell division is completely inhibited, and branches are not observed. This prompted Potluri et al. to test if the act of cell division under altered FtsZ levels might give rise to branching, with the absence of LMW PBPs an additional trigger. Cell division was originally hypothesized to be a prerequisite for branching (Woldringh et al., 1994), but a subsequent study suggested that branching was independent of FtsZ (Gullbrand et al., 1999).
Potluri et al. explored this issue in more depth by careful examination of mutant cells and their progeny during time-lapse growth under the microscope. If aberrant septation did indeed cause branching, the prediction would be that all aberrant septa would eventually give rise to a branch, while normal septa would not. Moreover, inhibiting cell division should block further production of branches. By following cell lineages in which an asymmetric septum was clearly detectable, the authors made a strong case that cell wall abnormalities resulting from these asymmetric septation events eventually caused an ectopic cell pole to emerge.
How do these asymmetric septa arise in the first place? As the shape of the invaginating septum is guided by the Z ring, it would make sense that Z ring geometry might be more frequently abnormal in the mutants that lead to branched cells. Indeed, using fluorescently tagged FtsZ, Potluri et al. found many of the Z rings in the mutant cells were clearly slanted at an angle. In contrast, Z rings in cells with normal LMW PBPs are uniformly perpendicular, even when spatial regulation by the Min and nucleoid occlusion systems are absent (Yu and Margolin, 1999). Therefore, the data suggest that LMW PBPs help to maintain the perpendicular orientation of the Z ring and, ultimately, ensure that the division septum and new cell poles are symmetrical. One potential complication is that many LMW PBP mutant cells that have slanted Z rings are also wider than normal, and this increase in cell diameter might indirectly increase the tendency of Z rings to form at odd angles. Nonetheless, PBP5 has been shown to localize to the lateral cell envelope and the septum when FtsZ is present (Potluri et al., 2011); moreover, PBP6 and DacD can localize to the septum if PBP5 is absent, supporting the idea that LMW PBPs can contact the Z ring directly. It is unlikely that excess FtsZ competes with LMW PBPs, as excess FtsZ exacerbates branching even in the their absence. It is more likely that LMW PBPs exert feedback control on the Z ring as it synthesizes preseptal PG. Such control is plausible, as there is recent evidence that septal PG hydrolases may feed back on the Z ring via the FtsEX ATPase complex (Yang et al., 2011). At this point it is unknown how preseptal PG is made or how the Z ring switches from preseptal to septal PG mode.
Finally, how do asymmetric septa lead to formation of branches? The authors propose an intriguing model in which aberrantly oriented Z rings transiently synthesize preseptal PG at off-center sites. If these slanted Z rings subsequently reorient during septal PG synthesis, the originally deposited preseptal PG may become inert. Upon cell division, these extra sites of inert PG will act like a false cell pole, causing new PG to be synthesized around it, much like a tree growing around a rock or a fencepost. This surrounding PG growth will be anisotropic, extending the ectopic pole until it becomes the tip of a new branch. Interestingly, cells with excess FtsZ and multiple Z rings often appear bumpy along their sidewalls. This may reflect many foci of inert PG resulting from preseptal but not septal PG synthesis. Why every focus of inert PG does not form a branch remains unclear.
In summary, Potluri et al. have proposed the first convincing molecular explanation for cellular branching in E. coli. Further studies are necessary to determine what other mechanisms E. coli and other rod-shaped bacteria use that keeps them from getting bent out of shape.
Acknowledgments
Work in our laboratory is supported by grants from the NIGMS (GM61074) and the Human Frontier Science Program.
References
- Addinall SG, Lutkenhaus J. FtsZ-spirals and -arcs determine the shape of the invaginating septa in some mutants of Escherichia coli. Mol Microbiol. 1996;22:231–237. doi: 10.1046/j.1365-2958.1996.00100.x. [DOI] [PubMed] [Google Scholar]
- Brown PJ, de Pedro MA, Kysela DT, Van der Henst C, Kim J, De Bolle X, Fuqua C, Brun YV. Polar growth in the Alphaproteobacterial order Rhizobiales. Proc Natl Acad Sci USA. 2011;109:1697–1701. doi: 10.1073/pnas.1114476109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Pedro MA, Quintela JC, Holtje JV, Schwarz H. Murein segregation in Escherichia coli. J Bacteriol. 1997;179:2823–2834. doi: 10.1128/jb.179.9.2823-2834.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- den Blaauwen T, de Pedro MA, Nguyen-Disteche M, Ayala JA. Morphogenesis of rod-shaped sacculi. FEMS Microbiol Rev. 2008;32:321–344. doi: 10.1111/j.1574-6976.2007.00090.x. [DOI] [PubMed] [Google Scholar]
- Dominguez-Escobar J, Chastanet A, Crevenna AH, Fromion V, Wedlich-Soldner R, Carballido-Lopez R. Processive movement of MreB-associated cell wall biosynthetic complexes in bacteria. Science. 2011;333:225–228. doi: 10.1126/science.1203466. [DOI] [PubMed] [Google Scholar]
- Gan L, Chen S, Jensen GJ. Molecular organization of Gram-negative peptidoglycan. Proc Natl Acad Sci U S A. 2008;105:18953–18957. doi: 10.1073/pnas.0808035105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garner EC, Bernard R, Wang W, Zhuang X, Rudner DZ, Mitchison T. Coupled, circumferential motions of the cell wall synthesis machinery and MreB filaments in B. subtilis. Science. 2011;333:222–225. doi: 10.1126/science.1203285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghosh AS, Chowdhury C, Nelson DE. Physiological functions of D-alanine carboxypeptidases in Escherichia coli. Trends Microbiol. 2008;16:309–317. doi: 10.1016/j.tim.2008.04.006. [DOI] [PubMed] [Google Scholar]
- Gullbrand B, Åkerlund T, Nordström K. On the origin of branches in Escherichia coli. J Bacteriol. 1999;181:6607–6614. doi: 10.1128/jb.181.21.6607-6614.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koch AL. The wall of bacteria serves the roles that mechano-proteins do in eukaryotes. FEMS Microbiol Rev. 1991;88:15–26. doi: 10.1111/j.1574-6968.1991.tb04954.x. [DOI] [PubMed] [Google Scholar]
- Mannik J, Driessen R, Galajda P, Keymer JE, Dekker C. Bacterial growth and motility in sub-micron constrictions. Proc Natl Acad Sci USA. 2009;106:14861–14866. doi: 10.1073/pnas.0907542106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Margolin W. Sculpting the bacterial cell. Curr Biol. 2009;19:R812–822. doi: 10.1016/j.cub.2009.06.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nanninga N. Morphogenesis of Escherichia coli. Microbiol Mol Biol Rev. 1998;62:110–129. doi: 10.1128/mmbr.62.1.110-129.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nelson DE, Young KD. Contributions of PBP 5 and DD-carboxypeptidase penicillin binding proteins to maintenance of cell shape in Escherichia coli. J Bacteriol. 2001;183:3055–3064. doi: 10.1128/JB.183.10.3055-3064.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Potluri L, Karczmarek A, Verheul J, Piette A, Wilkin JM, Werth N, Banzhaf M, Vollmer W, Young KD, Nguyen-Disteche M, den Blaauwen T. Septal and lateral wall localization of PBP5, the major D,D-carboxypeptidase of Escherichia coli, requires substrate recognition and membrane attachment. Mol Microbiol. 2011;77:300–323. doi: 10.1111/j.1365-2958.2010.07205.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rothfield L. New insights into the developmental history of the bacterial cell division site. J Bacteriol. 2003;185:1125–1127. doi: 10.1128/JB.185.4.1125-1127.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salje J, van den Ent F, de Boer P, Löwe J. Direct membrane binding by bacterial actin MreB. Mol Cell. 43:478–487. doi: 10.1016/j.molcel.2011.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Typas A, Banzhaf M, Gross CA, Vollmer W. From the regulation of peptidoglycan synthesis to bacterial growth and morphology. Nat Rev Microbiol. 2011;10:123–136. doi: 10.1038/nrmicro2677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uehara T, Bernhardt TG. More than just lysins: peptidoglycan hydrolases tailor the cell wall. Curr Opin Microbiol. 2011;14:698–703. doi: 10.1016/j.mib.2011.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van den Ent F, Amos LA, Löwe J. Prokaryotic origin of the actin cytoskeleton. Nature. 2001;413:39–44. doi: 10.1038/35092500. [DOI] [PubMed] [Google Scholar]
- van Teeffelen S, Wang S, Furchtgott L, Huang KC, Wingreen NS, Shaevitz JW, Gitai Z. The bacterial actin MreB rotates, and rotation depends on cell-wall assembly. Proc Natl Acad Sci U S A. 108:15822–15827. doi: 10.1073/pnas.1108999108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Varma A, Young KD. FtsZ Collaborates with Penicillin Binding Proteins To Generate Bacterial Cell Shape in Escherichia coli. J Bacteriol. 2004;186:6768–6774. doi: 10.1128/JB.186.20.6768-6774.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Varma A, Young KD. In Escherichia coli, MreB and FtsZ direct the synthesis of lateral cell wall via independent pathways that require PBP 2. J Bacteriol. 2009;191:3526–3533. doi: 10.1128/JB.01812-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- White CL, Kitich A, Gober JW. Positioning cell wall synthetic complexes by the bacterial morphogenetic proteins MreB and MreD. Mol Microbiol. 2011;76:616–633. doi: 10.1111/j.1365-2958.2010.07108.x. [DOI] [PubMed] [Google Scholar]
- Woldringh CL, Zaritsky A, Grover NB. Nucleoid partitioning and the division plane of Escherichia coli. J Bacteriol. 1994;176:6030–6038. doi: 10.1128/jb.176.19.6030-6038.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang DC, Peters NT, Parzych KR, Uehara T, Markovski M, Bernhardt TG. An ATP-binding cassette transporter-like complex governs cell-wall hydrolysis at the bacterial cytokinetic ring. Proc Natl Acad Sci U S A. 2011;108:E1052–1060. doi: 10.1073/pnas.1107780108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young KD. Bacterial shape: two-dimensional questions and possibilities. Annu Rev Microbiol. 2011;64:223–240. doi: 10.1146/annurev.micro.112408.134102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu XC, Margolin W. FtsZ ring clusters in min and partition mutants: Role of both the Min system and the nucleoid in regulating FtsZ ring localization. Mol Microbiol. 1999;32:315–326. doi: 10.1046/j.1365-2958.1999.01351.x. [DOI] [PubMed] [Google Scholar]
- Yu XC, Margolin W. Deletion of the min operon results in increased thermosensitivity of an ftsZ84 mutant and abnormal FtsZ ring assembly, placement, and disassembly. J Bacteriol. 2000;182:6203–6213. doi: 10.1128/jb.182.21.6203-6213.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]


