Abstract
Acute myeloid leukemia (AML) is an aggressive malignancy that leads to marrow failure and death. There is a desperate need for new therapies. The novel fluoropyrimidine, FdUMP[10], was highly active against both human AML cell lines, (IC50 values, 3.4nM-21.5nM) and murine lines (IC50 values, 123.8pM-131.4pM). In all cases, the IC50 of FdUMP[10] was lower than for cytarabine and ∼ 1000 times lower than 5-fluorouracil (5-FU). FdUMP[10] remained effective against cells expressing the Flt3 internal tandem duplication, BCR-ABL, MN1, and an shRNA against p53. It had activity against patient samples at concentrations that did not affect normal hematopoietic cells. FdUMP[10] inhibited thymidylate synthase (TS) and trapped topoisomerase I cleavage complexes (Top1CCs), leading to DNA damage and apoptosis. All cell lines and nearly all primary AML samples examined expressed both TS and Top1. In vivo, FdUMP[10] was active against a syngeneic AML model with a survival advantage equivalent to doxorubicin plus cytarabine. 5-FU treatment was toxic and did not improve survival. FdUMP[10] was better tolerated than 5-FU or cytarabine plus doxorubicin and did not affect normal HSCs, while 5-FU dramatically impaired their ability to engraft. In summary, FdUMP[10] was highly efficacious and better tolerated than standard therapies.
Introduction
Acute myelogenous leukemia (AML) is an aggressive malignancy that leads to marrow failure and death.1 AML affects ∼ 12 000 people per year in the United States, causing 9000 deaths.1 Despite decades of research, standard therapy has not changed, and the overall 5-year survival rate is 30%-40%.2 The current standard of care for patients with AML is induction chemotherapy with cytarabine (Ara-C) and an anthracycline.2 Most patients treated this way will achieve a complete, but transient, remission. Once relapsed, the disease is increasingly resistant to further therapy. Age is an important prognostic factor in AML.3 For patients 60 years of age or older the prognosis is grave. There are biologic and clinical differences in older patients, resulting in a 5-year survival rate of < 10%.4 These differences include increased comorbidities that result in higher early death rates, more patients with high-risk cytogenetic profiles, and multidrug resistance phenotypes.2 This is compounded by the fact that AML is a disease of the elderly with the median age of onset of 72 years.5 The high rate of early mortality and resistance has led some to question whether elderly patients with AML benefit from therapy at all.6 There is a clear need for additional therapies with acceptable toxicity profiles.
The novel fluoropyrimidine FdUMP[10] is a polymer of the thymidylate synthase (TS)–inhibitory fluoropyrimidine (FP) metabolite 5-fluoro-2′-deoxyuridine-5′-O-monophosphate (FdUMP). Since its discovery, 5-fluorouracil (5-FU) has been widely used to treat multiple solid tumors, including breast, colon, and head/neck cancers7; however, it is seldom used in hematologic malignancies, including the acute leukemias. The mechanism of action for 5-FU is thought to involve both the inhibition of TS and disruption of RNA processing. Toxicities associated with 5-FU treatment include myelosuppression, diarrhea, and mucositis. These toxicities overlap with the drugs currently used in AML; thus, 5-FU has not been considered an attractive candidate for AML treatment. In contrast, FdUMP[10] was 338-fold more potent in the National Cancer Institute (NCI) 60 cell line screen relative to 5-FU.8 FdUMP[10] uniquely targets both TS and topoisomerase 1 (Top1). FdUMP[10], but not 5-FU, is highly active toward the human leukemia cell lines included in the NCI 60 and has shown enhanced potency and lower toxicity relative to 5-FU in mouse xenograft models.8,9 The favorable safety profile for FdUMP[10] combined with its enhanced activity led us to investigate whether this compound has activity against AML.
Methods
Cell culture and viability assays
OCI-AML3 cells were a kind gift of Dr Mark Minden at the University of Toronto. All human cell lines were maintained in RPMI media (Gibco) supplemented with 10% FBS, penicillin, and streptomycin. Cells were grown at 37°C with 5% CO2. Viability assays were done with the use of the Cell Titer-Glo assay (Promega) according to the manufacturer's protocol or by Trypan blue exclusion assay with the use of the Countess cell counting system (Invitrogen). All murine cells were derived from fetal liver cells infected with MLL-ENL alone or with NRasG12D or Flt3-ITD (internal tandem duplication) expressing vectors.10 Murine cells were maintained in stem cell media (40% DMEM, 40% IMDM, 20% FBS, supplemented with murine SCF to 10 ng/mL, murine IL-6 to 2 ng/mL, and murine IL-3 to 0.4 ng/mL).
Primary AML samples and normal human HSCs
All samples were collected under a protocol approved by the Wake Forest University Institutional Review Board. Primary AML samples were from 15 patients with confirmed AML (supplemental Table 1, available on the Blood Web site; see the Supplemental Materials link at the top of the online article). All patients gave written informed consent. All samples were obtained during clinically indicated procedures. Cells were collected by centrifugation, resuspended in ACK lysis buffer (150mM NH4Cl, 10mM KHCO3, 0.1mM EDTA) at room temperature for 5 minutes, centrifuged again, washed with PBS, and stored at −80°C until use. Alternatively, cells were isolated by Ficoll-gradient centrifugation and stored as above. Normal HSCs were obtained from healthy allogeneic stem cell transplant donors. Cells were obtained from GM-CSF–primed apheresis of peripheral blood, Ficoll separated, and stored as above.
Colony formation assays
Human cell lines, primary patient samples, or normal human HSCs were thawed and incubated in Stem SpanSFEM media supplemented with Stem Span CC100 cytokine cocktail (StemCell Technologies) for 24 hours with the indicated drug. Cells were then placed in ColonyGEL High Cytokine Formulation media (ReachBio). Experiments were performed in triplicate. Colonies of ≥ 8 cells were counted on or after day 7 as in Shankar et al.11 This criterion was based on the observation that primary AML samples typically yield a mixture of larger and smaller cell clusters with many clusters as small as 8 cells. More traditional criteria of 20- or 50-cell colonies would have missed a significant number of blast colonies. We counted all clusters, CFU-GMs, and BFU-E colonies. To avoid bias we used identical criteria to count colonies from healthy patient samples.
Western blot analyses and immunofluorescence
Samples were lysed in Laemmli buffer, separated by SDS-PAGE, and transferred to an Immobilon polyvinylidene difluoride membrane (Millipore). Abs against p53 (IMX25, 1:1000; Leica Microsystems), TS (#35-5800, 1:1000; Invitrogen), topoisomerase I (556597, 1:2000; BD PharMingen), caspase 3 (9662, 1:2000; Cell Signaling), and actin (AC-15, 1:5000; Abcam) were used. For ECL Western analyses secondary Abs were anti–mouse (7076, 1:5000; Cell Signaling) or anti–rabbit (7074, 1:5000; Cell Signaling). For immunofluorescence studies of phosphorylated γH2AX, cells were fixed with 4% neutral-buffered formalin, permeabilized with PBS containing 0.2% Triton X-100, and probed with anti-phosphoH2AX (#2577, 1:100; Cell Signaling Technologies) followed by donkey anti–rabbit Alexa Fluor 594–conjugated Ab (1:500, A-21207; Invitrogen) and visualized with fluorescence microscopy.
In vivo treatment studies
The Wake Forest University Institutional Animal Care and Use Committee approved all mouse experiments. Luciferase-tagged leukemia cells were transplanted into 6- to 8-week-old sublethally irradiated recipient mice (4.5 Gy) by tail vein injection of 1 × 106 viable cells. Mice were monitored by bioluminescent imaging on day 7. Imaging was performed with an IVIS100 imaging system (Caliper LifeSciences). Mice were injected with 150 mg/kg D-Luciferin (Gold Biotechnology), anesthetized with isoflurane, and imaged for 2 minutes. Chemotherapy was initiated on detection of clear signals. Mice were treated with 300 mg/kg FdUMP[10] or 5-FU at 121 mg/kg (APP Pharmaceuticals) or the combination of 125 mg/kg Ara-C and 3.75 mg/kg doxorubicin (Dox; both from Bedford Laboratories) by jugular or tail vein injection. Control animals were injected with PBS. Repeat luciferase imaging was performed on day 6 of treatment.
Toxicology studies and murine BM transplantation
Normal C57/Bl6 mice were treated with identical dose, schedule, and route of each drug as in the efficacy studies (ie, days 1, 3, 5, and 7). Seventy-two hours after the last dose, animals were killed, bilateral femoral cells harvested, and organs were fixed in 10% neutral-buffered formalin followed by routine tissue processing and sectioning for H&E staining. Slides were then reviewed by a veterinary pathologist with the use of a Nikon Eclipse 50i light microscope. Photographs of tissues were taken with the NIS Elements D3.10 camera and software system. For transplantation studies Ly5.1+ C57/Bl6 recipient animals were irradiated to 8 Gy and injected with 1 × 106 femur cells or, when 1 × 106 cells were not isolated, then 90% of all cells isolated by tail vein injection. Three weeks later recipient femoral BM was harvested and stained with allophycocyanin-conjugated anti-Ly5.2 Ab (BD PharMingen) and analyzed by flow cytometry.
Apoptosis assays
Cells were seeded in 6-well plates at 25 000 cells/mL in 3 mL, grown for 2 days, and treated with the indicated drug for 16, 24, 36, or 48 hours. After centrifugation and washing in cold PBS cells were stained with PI (Sigma-Aldrich) and allophycocyanin-conjugated annexin V in a binding buffer (0.1M HEPES [pH 7.4], 1.4M NaCl, and 25mM CaCl2 solution; BD PharMingen) according to the manufacturer's protocol. Flow cytometric analysis was conducted on a BD FACSCantoII cytometer with the BD FACSDiva Version 5.0.3 software (BD Biosciences).
TS catalytic activity
Cells were plated at 1.5 × 106 cells in 100-mm2 plates and grown overnight in RPMI 1640 medium with 20% FBS. Cells were exposed to the indicated drug for 8, 16, 24, or 48 hours. Cells were then harvested in 25mM Tris-HCl, pH 7.4, with Complete Protease Inhibitor Cocktail (Roche), put through 2 freeze/thaw cycles, and vortexed. The lysates were centrifuged at 10 000g for 10 minutes at 4°C. TS assays were performed in a final volume of 200 μL containing 75μM 5,10 methylene tetrahydrofolate in 0.5M NaOH (Schircks Laboratories), 10μM dUMP, 200 000dpm of 3H-dUMP (Moravek Biochemicals), 100μM 2-mercaptoethanol, and 25mM KH2PO4, pH 7.4. Cell lysate (400 μg of protein) was added to the reaction buffer. Reactions were incubated at 37°C for 30 minutes and stopped by the addition of 100 μL of 20% TCA, incubated for 5 minutes on ice. Two hundred microliters of activated charcoal solution (10 g of activated charcoal, 0.25 g of BSA, 0.25 g of dextran sulfate, in 100 mL of water) was added, vortexed, and maintained at room temperature for 10 minutes. Reactions were centrifuged at 10 000g for 30 minutes. Aliquots (200 μL) of the supernatant fluid were read by scintillation counting. All reactions were repeated a minimum of 3 times.
ICE bioassay/Top1 cleavage complex detection
Cells were plated at 1.5 × 106 cells in 100-mm2 plates and grown in RPMI 1640 medium with 20% FBS. Either 100nM 5-FU or 1nM FdUMP[10] was added and incubated for 24 or 48 hours. Cells were lysed in 2 mL of 1% sarkosyl in 1× TE, pH 7.5, and frozen at −20°C. Samples were homogenized ∼ 25 times. The addition of 1 mL of sarkosyl was used to wash the homogenizer. CsCl gradients were made as described in Liao et al.9 The samples were overlaid and ultracentrifuged in a SW41T rotor. Centrifugation was at 36K for 20-22 hours at 20°C; 500-mL fractions were collected. Fractions 6 through 11 were used for analysis. Two hundred microliters of each fraction and an equal volume of 25mM NaPO4 buffer (pH 6.5) were loaded onto a nitrocellulose membrane with the use of a Schleicher and Schuell Minifold II slot blotter. The membrane was blocked with 5% milk in TBST. Primary Ab was added at 1:500 dilution, overnight at 4°C. Secondary Ab was used in the same manner. ECL Lightning Plus (Perkin-Elmer) was used for detection of the Top1 cleavage complex.
Competition assays
Cells were infected with ecotropic retrovirus engineered to express thymidylate synthase in a bicistronic message with green fluorescent protein (GFP). Partially infected populations were exposed to therapy for 72 hours, the viable cells were gated on forward scatter versus side scatter plots, and the percentage of GFP+ cells was determined as in Pardee et al.10
Statistical analysis
All means were compared by 2-tailed Student t test. Survival curves were estimated by the Kaplan-Meier method, and P values were determined by the log-rank test. P values < .05 were considered significant. Analysis was performed with Graph Pad Prism Version 5.02 (GraphPad Software).
Results
FdUMP[10] is highly active in vitro against multiple human leukemia cell lines
To assess the activity of FdUMP[10] against human leukemia cell lines, we performed in vitro cytotoxicity assays. We exposed HL60, Molm-13, Jurkat, THP-1, K562, and KG1a cells to various concentrations of FdUMP[10] for 72 hours and then determined viability. All human leukemia lines had IC50 values in the low nanomolar range (Table 1). To compare activity of FdUMP[10] with other standard chemotherapies with the use of this assay, we also treated cells with 5-FU, Ara-C, or Dox. We chose to use Dox as the anthracycline because it is the best-tolerated anthracycline in C57/Bl6 mice12 and would allow for comparisons between in vivo and in vitro studies. The IC50 values for 5-FU were all ∼ 1000 times higher than for FdUMP[10] despite that on a molar basis there is only a 10-fold increase FP content (Table 1). Although we observed a wide range of sensitivities to Ara-C and Dox, all cell lines had similar responses to FdUMP[10], implying a lack of cross-resistance. The assays in Table 1 cells were cultured in FBS. To determine whether would observe similar results when cells were grown in human serum, we compared FdUMP[10] activity against HL60 and KG1a cells grown in 10% human serum or 10% FBS. We saw no difference in response to FdUMP[10] when human serum was used, suggesting FdUMP[10] activity is not altered by human serum (supplemental Figure 1). These data show that FdUMP[10] is more active against a variety of human leukemia cell lines than 5-FU, Ara-C, or Dox.
Table 1.
IC50 values for FdUMP[10], 5-FU, Dox, and Ara-C
| Chemotherapy | HL60 | Jurkat | THP-1 | K562 | KG1a | Molm-13 |
|---|---|---|---|---|---|---|
| FdUMP[10], nM | 3.4 (2.984-3.825) | 5.4 (4.609-6.417) | 4.1 (3.413-4.907) | 21.5 (14.85-30.99) | 4.9 (3.995-6.043) | 10.01 (8.726-11.49) |
| 5-FU, nM | 5027 (3804-6643) | 7,492 (6,169-9,100) | 1157 (943-1421) | 7011 (3270-15 030) | 5277 (3902-7139) | 943.4 (895-994) |
| Dox, nM | 21.7 (18.90-24.97) | 75.2 (48.16-117.47) | 160.5 (75.7-340.5) | 252.9 (200.69-319.14) | 1337 (1190-1510) | 8.18 (6.41-10.43) |
| Ara-C, nM | 144.9 (85.00-247.0) | 15.5 (14.44-16.67) | 6,700 (5890-7534) | 83.7 (72.24-96.94) | 195.8 (163.5-234.4) | 41.24 (12.95-131.3) |
Values are mean (95% CI).
FdUMP[10] is highly active against murine AML models expressing different poor prognostic markers
The human cell lines tested have different adverse prognostic factors, yet they all had similar IC50 values for FdUMP[10] (Table 1), suggesting that these factors may not modulate response to FdUMP[10]. To determine the effects of these markers we used a genetically defined mouse model system. This model is based on expression of MLL-ENL and has been shown to alter its chemotherapy sensitivity in response to expression of different prognostic markers.10 FdUMP[10] was highly active against multiple MLL-ENL–expressing cell lines with IC50 vales in the picomolar range (Table 2). This included a cell line expressing the human FLT3 receptor containing an internal tandem mutation (Flt3 ITD) shown to confer a worse prognosis in patients with AML13 and to alter chemotherapy response.10
Table 2.
IC50 values of FdUMP[10], 5-FU, Dox, and Ara-C
| Chemotherapy | MFL2 | MR2 | M1 |
|---|---|---|---|
| FdUMP[10], pM | 131.4 (108.3-159.3) | 124.2 (97.9-157.7) | 123.8 (111.5-137.6) |
| 5-FU, nM | 292.3 (251.8-339.4) | 214.0 (178.9-255.9) | ND |
| Dox, nM | 24.6 (15.9-38.1) | 11.4 (7.2-18.2) | 3.7 (3.4-4.2) |
| Ara-C, nM | 54.4 (58.4-182.7) | 71.8 (75.9-166.5) | 46.8 (43.5-50.4) |
Values are mean (95% CI).
MFL2 indicates MLL-ENL + Flt3 ITD; MR2, MLL-ENL + NRasG12D; M1, MLL-ENL alone; and ND, not done.
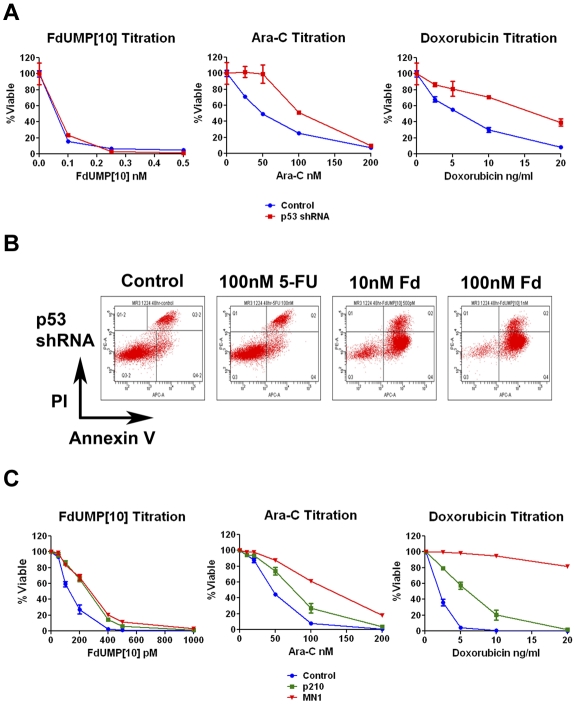
Most of the human leukemia lines tested have deleted or mutated p53. p53 activation is important for response to Ara-C14 and Dox,15 and its loss is associated with a worse prognosis in AML.16,17 To determine whether p53 is important in response to FdUMP[10], we infected an MLL-ENL– and FLT3 ITD–expressing cell line with either a control or p53-targeting shRNA vector. Cells expressing the shRNA showed loss of p53 induction, indicating efficient knockdown (supplemental Figure 2). Loss of p53 response led to resistance to both Dox and Ara-C (Figure 1A). However, no change was observed for FdUMP[10], suggesting that p53 is not required for response. To determine whether p53-suppressing cells are undergoing apoptosis in response to FdUMP[10], we performed an annexin V assay. Cells treated with FdUMP[10] were positive for annexin V binding, suggesting a p53-independent apoptotic pathway (Figure 1B).
Figure 1.
FdUMP[10] is active against cells expressing adverse prognostic factors. (A) Cytotoxicity assays of murine AML cells that knockdown p53. Cells were exposed to the indicated drugs for 72 hours and then assessed for viability. Viability is shown as percentage of control; error bars represent the SE. (B) Flow cytometry of annexin V assay. Cells expressing the p53 shRNA were exposed to the indicated drug for 48 hours and then labeled with annexin V and propidium iodide (PI). (C) Cytotoxicity assays of murine AML cells expressing either MN1 or BCR-ABL (p210). Cells were exposed to the indicated drugs for 72 hours and then assessed for viability. Viability is shown as percentage of control; error bars represent the SE.
In addition to p53 loss, several other adverse prognostic factors are expressed in the cell lines tested. K562 cells express the fusion gene BCR-ABL that has been implicated in chemotherapy resistance18 and prognosis in AML.19 THP-1 cells express high levels of MN1,20 shown to confer an adverse prognosis.21,22 To test the effects of these genes on FdUMP[10] response we infected a murine AML cell line with vectors expressing human MN1 or the p210 BCR-ABL (p210) fusion protein. Cells expressing either p210 or MN1 displayed resistance to Dox with IC50 values that were 2.95- and > 10-fold higher than the parental leukemia cells and to Ara-C with MN1-expressing cells having a 3.2-fold increase in IC50 value (Figure 1C). In contrast FdUMP[10] remained highly effective for cells expressing either p210 or MN1 with IC50 values that changed by only 1.98- or 2.07-fold, respectively (Figure 1C). These results indicate that factors conferring resistance to Ara-C and Dox minimally affect response to FdUMP[10] and suggest FdUMP[10] probably has activity in patients with these poor prognostic markers.
FdUMP[10] is highly active against primary patient AML samples while sparing normal hematopoietic cells
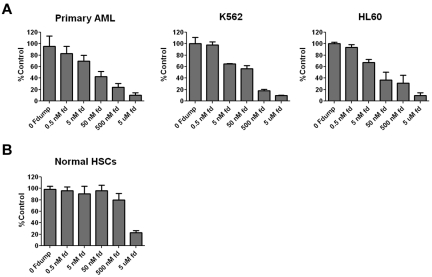
To determine whether FdUMP[10] had activity against primary patient samples, we performed colony formation assays with the use of 3 primary patient samples (samples A10-A12; supplemental Table 1) as well as K562 and HL60 cells. For all primary patient samples leukemic blasts made up the majority of the cells in the sample (> 55%, 80%-90%, and > 90%). FdUMP[10] was highly effective in suppressing colony formation from the cell lines and primary patient samples (Figure 2A), showing that FdUMP[10] has activity against human leukemia colony-forming cells. To establish the effect of FdUMP[10] on normal hematopoietic cells we performed colony assays on HSCs from 4 separate healthy allogeneic transplant donors. In contrast to its effect on AML samples, FdUMP[10] only suppressed HSC colony formation at the highest dose tested (Figure 2B), suggesting a large therapeutic window. The existence of LSCs has been widely established, and their persistence in treated patients is thought to be responsible for relapse (reviewed in Chan and Huntly23). To determine whether FdUMP[10] had activity against LSCs we incubated luciferase tagged, MLL-ENL and Flt3 ITD (MFL2) or MLL-ENL and NrasG12D (MR2) expressing murine AML cells with 0.5nM or 1nM FdUMP[10] for 24 hours. After incubation 1 × 106 viable cells were injected into syngeneic recipients. FdUMP[10]-treated cells displayed a dose-dependent reduction in the ability to engraft consistent with a reduction in the LSC population (supplemental Figure 3A), and mice injected with treated cells had a significant increase in survival (supplemental Figure 3B). These data suggest FdUMP[10] is active against primary patient samples and murine LSCs and spares normal hematopoietic cells.
Figure 2.
FdUMP[10] is active against leukemia stem cells (LSCs) from cell lines and primary patient samples. (A) Colony formation assays. Primary patient samples or cell lines were incubated with the indicated drug for 24 hours and placed in methylcellulose media. Plates were read on or after day 7. All experiments were done in triplicate. Primary AML is the combined results of 3 independent primary patient samples. Colony numbers are normalized to controls, and error bars represent the SE. (B) Colony formation assays. As in panel A, the result shown is the combination of 4 separate normal HSC donors.
FdUMP[10] causes inhibition of TS and trapped Top1 cleavage complexes in human leukemia cells
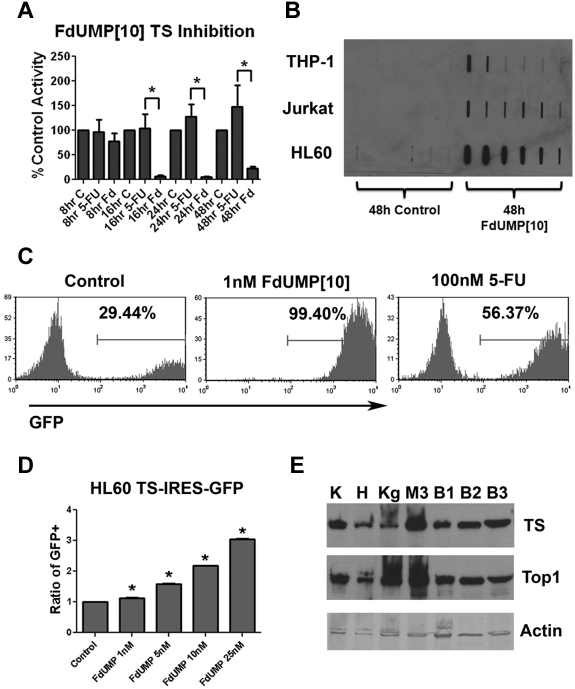
Previous work in human and murine lymphoblastic leukemia cell lines has shown that FdUMP[10] induces TS inhibition and trapped Top1 cleavage complexes (Top1CC).9 To determine whether FdUMP[10] inhibited TS in human AML cell lines, we exposed HL60 cells to 10nM FdUMP[10] or 100nM 5-FU and performed assays for activity. FdUMP[10] caused profound and prolonged TS inhibition compared with a 10-fold greater concentration of 5-FU (Figure 3A). To determine whether FdUMP[10] trapped Top1CC, THP-1, Jurkat, and HL60 cells were exposed to 100nM FdUMP[10] for 48 hours and assessed for the presence of Top1CC. After 48 hours, Top1 was detected co-migrating with genomic DNA, consistent with trapped complexes (Figure 3B). To further establish the role of TS in FdUMP[10] response we performed competition assays with the use of a retroviral vector engineered to express TS in a bicistronic message with GFP. Murine AML cells expressing TS were highly enriched in the presence of 1nM FdUMP[10] compared with controls. In contrast treatment with 100nM 5-FU resulted in only partial enrichment (Figure 3C). A significant and dose-dependent increase in TS-expressing cells was also seen when the human AML cell line HL60 was used (Figure 3D). Importantly, both viability and trapped Top1CCs could be rescued by the addition of thymidine to the culture media, consistent with TS inhibition and Top1CC trapping being central to FdUMP[10] cytotoxicity (data not shown). These data have shown that FdUMP[10] causes profound TS inhibition and traps Top1CC. This is in contrast to 5-FU, which did not significantly inhibit TS at 10 times the concentration of FdUMP[10] despite that the amount of fluoropyrimidine delivered is identical. This would suggest that FdUMP[10] is not simply a more-concentrated version of 5-FU but rather has distinct biochemical effects.
Figure 3.
FdUMP[10] is a potent inhibitor of TS and traps Top1CCs. (A) TS inhibition assay. HL60 cells were exposed to 10nM FdUMP[10] or 100nM 5-FU for the indicated time, lysed, and assayed for TS activity. Activity is plotted as percentage of control. (B) ICE bioassay for Top1CCs. THP-1, Jurkat, and HL60 cells were incubated with 100nM FdUMP[10] for 48 hours. Cells were lysed and subjected to ICE bioassay (see “ICE bioassay/Top1 cleavage complex detection”), and DNA-containing fractions were blotted for Top1. (C) Murine AML TS competition assays. Murine AML cells were partially infected with TS and GFP-expressing retrovirus and treated with the indicated drug for 72 hours. All experiments were performed in triplicate. The GFP+ percentage in the viable cell population is indicated; representative histograms are shown. (D) HL60 TS competition assays. HL60 cells were partially infected with TS and GFP-expressing retrovirus and exposed to the indicated drug for 72 hours. Percentage of GFP+ cells was determined as in panel C and normalized to untreated controls. All experiments were done in triplicate. (E) Results of TS and Top1 Western blot analyses. K562 (K), HL60 (H), Kg1a (Kg), MLL-ENL murine AML (M3), or 3 primary AML patient samples (B1-3) were blotted with the indicated Ab. Error bars represent SE. *P < .05.
To determine whether TS and Top1 are commonly expressed in AML cells we performed a Western blot analysis for TS and Top1 in HL60, K562, KG1a, 3 murine AML lines, and 12 patient samples. Patient characteristics are shown in supplemental Table 1. We found wide variability in the expression levels of TS and Top1 (Figure 3E; supplemental Figure 4). However, no clear correlation was observed between TS or Top1 level and FdUMP[10] sensitivity because K562 cells had the highest levels of TS and Top1 expression, despite having an IC50 ∼ 7 times higher than HL60 cells (Table 1). Importantly, both proteins were detectable in all but one primary patient sample. These data show that TS and Top1 are commonly expressed in human AML cells and suggest that FdUMP[10] will have broad activity in AML.
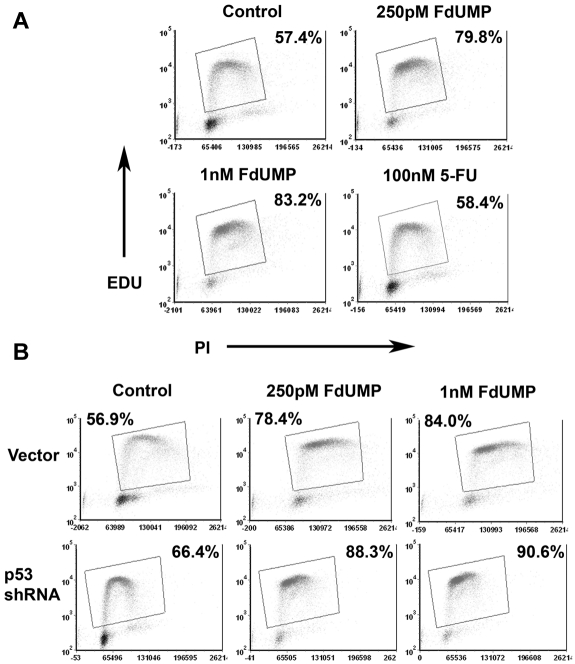
FdUMP[10] arrests cells in S phase
Our results suggest that Top1 complexes at replication forks are trapped by incorporated FdUMP or dUMP, leading to stalled replication forks and ultimately DNA strand breaks. This, coupled with the lack of thymidine, would lead to futile cycles of attempted repair and recurrent trapping of Top1.9 If so, cells exposed to FdUMP[10] should accumulate in S phase. To assess this possibility, we determined the cell cycle distribution of murine AML cells after exposure to FdUMP[10] for 8 hours. As predicted, cells accumulated in the S phase after exposure to FdUMP[10] (Figure 4A). 5-FU was much less efficient at arresting cells in S phase, with essentially no difference in the number of cells in S phase from control even at 100 times the FdUMP[10] concentration (Figure 4A). An intact p53 response was not necessary for the observed S-phase arrest, because cells expressing a p53-targeting shRNA were also efficiently arrested in S phase (Figure 4B). These data confirm that the TS inhibition and trapped Top1CC induced by FdUMP[10] lead to the accumulation of cells in S phase. The lack of significant accumulation in S phase of cells exposed to even 100 times higher concentration of 5-FU, again, show that FdUMP[10] has distinct biochemical activities and is not simply converted to 5-FU.
Figure 4.
FdUMP[10] causes S-phase arrest regardless of p53 status. (A) EdU (5-ethynyl-2′-deoxyuridine) incorporation assays. MLL-ENL–driven murine AML cells were incubated with the indicated drug for 8 hours and subjected to an EdU incorporation assay. S-phase cells were gated as shown. Percentages shown are for cells in S phase. (B) EdU incorporation assay as in panel A. Cells were infected with either a p53-targeting shRNA or a control vector and exposed to the indicated drug for 8 hours.
FdUMP[10] induces DNA damage and apoptosis in AML cells
We have shown that FdUMP[10] induces profound TS inhibition, trapped Top1CC, and accumulation of cells in S phase. This combination of stalled replication, lack of thymidine, and trapped Top1CC should lead to the accumulation of DNA double-strand breaks (DSBs). Consistent with this, previous work has shown that FdUMP[10] induces DNA damage.9 To determine whether FdUMP[10] induces DSBs in human AML cells, we performed immunofluorescence assays against γH2AX. K562 cells were exposed to 10nM or 100nM FdUMP[10] or 1μM 5-FU for 24 hours and then assessed for γH2AX foci. Exposure to FdUMP[10] at either concentration resulted in the formation of multiple γH2AX foci (Figure 5A), indicating the induction of DNA DSBs. In contrast, exposure to 1μM 5-FU under identical conditions did not result in the formation of γH2AX foci, suggesting that FdUMP[10] is much more efficient than 5-FU at inducing DNA damage. A similar result was obtained with the use of Jurkat cells (supplemental Figure 5A). To determine that the observed DNA damage was a direct effect of FdUMP[10] and not a consequence of apoptosis, we repeated the experiment in the presence of the pan caspase inhibitor Z-VAD-FMK. Caspase inhibition had no effect on the number of γ-H2AX foci (supplemental Figure 5B).
Figure 5.
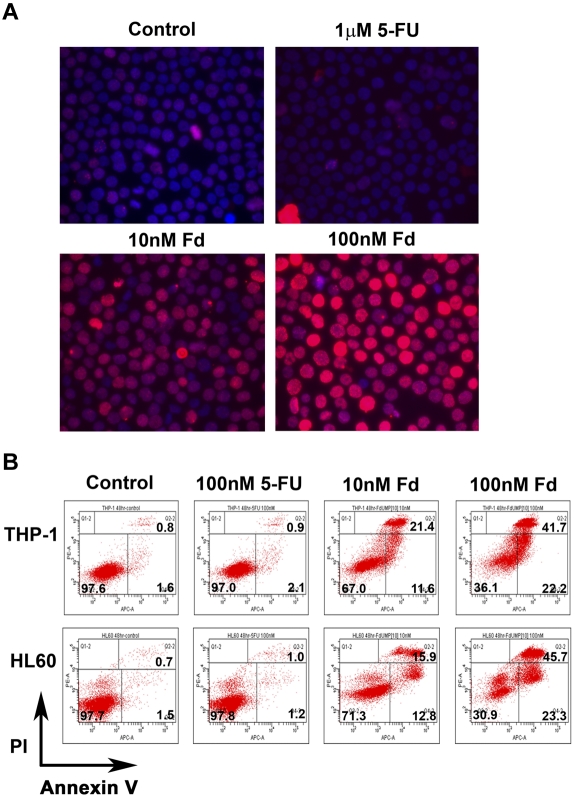
FdUMP[10] causes DNA damage and induces apoptosis. (A) Immunofluorescence for γH2AX foci. K562 cells were treated with the indicated drug for 24 hours and then assayed for the presence of γH2AX foci. Secondary Ab was conjugated with Alexa Fluor 594, and images were captured with an Olympus IX70 inverted fluorescent microscope equipped with a Retiga 2000R digital color camera and using an LPlanFl 40×/0.40 objective. Images were analyzed with Image Pro Plus 5.1 software. (B) Flow cytometry of annexin V assay. THP-1 or HL60 cells were treated with the indicated drug for 48 hours and then labeled with annexin V and propidium iodide (PI) and analyzed by flow cytometry. Numbers in bold denote percentage of cells in each quadrant.
The futile repair cycles and DNA damage induced by FdUMP[10] would be predicted to lead to cell death, possibly by apoptosis. Indeed, previous work has shown that FdUMP[10] induces apoptosis in human colorectal cancer cells.24 To determine whether exposure to FdUMP[10] culminates in apoptosis, we treated THP-1 and HL60 cells with either 10nM or 100nM FdUMP[10] and assessed apoptosis induction by annexin V and propidium iodide staining. At 48 hours, most cells were stained positive, indicating apoptosis is the predominant cell death mechanism (Figure 5B). In addition, intact caspase 3 was lost in a dose-dependent manner in HL60 cells treated with FdUMP[10], indicating induction of apoptosis (supplemental Figure 6A). Consistent with the IC50 data, 5-FU was far less efficient at inducing apoptosis with only minimal annexin V–positive cells, even at 100nM (Figure 5B). FdUMP[10] also efficiently induced apoptosis in 2 separately derived murine AML cell lines (supplemental Figure 6B). As with the human lines, FdUMP[10] was far more efficient at inducing apoptosis than 5-FU or Ara-C (supplemental Figure 6C). When treated AML cells were followed by viable cell number, a cytostatic phenomenon was observed at lower doses of FdUMP[10] with delayed cell growth presumably from S-phase arrest, and FdUMP[10] was metabolized before resumption of cell growth could occur (supplemental Figure 7A). At higher concentrations of FdUMP[10] there was predominantly an apoptotic response with no resumption of cell growth when HL60 cells were treated with 50nM or when the more sensitive murine cells were treated at 0.5nM or 1nM FdUMP[10] (supplemental Figure 7A-B). This is consistent with our data that showed 28.7% of HL60 cells treated with 10nM FdUMP[10] became positive for annexin V versus 69.1% of cells treated at 100nM (Figure 5B). Likewise HL60 cells treated with 10nM FdUMP[10] retained ∼ 74% of control levels of intact caspase 3 compared with only ∼ 25% of 50nM of treated cells (supplemental Figure 6A). Taken together, these data show that FdUMP[10], but not 5-FU, efficiently induces DNA damage and apoptosis in human and murine leukemia cells, indicating a distinct biochemical mechanism for FdUMP[10] relative to 5-FU.
FdUMP[10] is highly active against murine AML in vivo
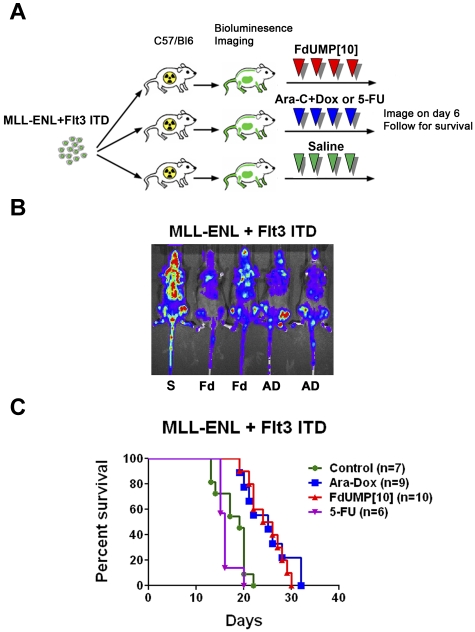
Recent studies have shown that the BM microenvironment can have a profound effect on chemotherapy response in AML.25 In addition, the immune system can affect the degree of response to a given therapy in vivo.26 To assess the activity of FdUMP[10] in a setting that would incorporate these important interactions, we injected sublethally irradiated C57/Bl6 mice with the MR2 cell line. Once leukemic engraftment was confirmed by bioluminescence imaging, mice were treated. When mice were treated with FdUMP[10] at 300 mg/kg on days 1 and 4, there was reduction in disease burden (supplemental Figure 8A) and a statistically significant survival benefit compared with untreated controls (supplemental Figure 8B). FdUMP[10]-treated mice showed no signs of toxicity. To determine whether we could improve survival, we treated MR2-injected mice with FdUMP[10] at 300 mg/kg every other day for 4 doses. This increased dosing regimen was well tolerated and resulted in a significant survival benefit (supplemental Figure 8C). To establish the efficacy of FdUMP[10] in a second MLL-ENL leukemia and to compare it with 5-FU or Ara/Dox treatment, mice were injected with the MFL2 cell line. Treatment consisted of either FdUMP[10] (300 mg/kg), 5-FU (121 mg/kg), or Ara-C (125 mg/kg) plus Dox (3.75 mg/kg) on days 1, 3, 5, and 7 (Figure 6A). The 5-FU dose was calculated to deliver the same FP content as FdUMP[10] dosed at 300 mg/kg. The Ara/Dox doses were designed to deliver the same amount of both drugs as in the previously published regimen shown to be tolerable and efficacious.27 FdUMP[10] treatment resulted in reduced disease burden as determined by bioluminescent imaging (Figure 6B) and improved survival compared with controls (P = .0002; Figure 6C). No difference in survival was observed between FdUMP[10]-treated and Ara/Dox-treated animals (P = .604; Figure 6C), suggesting comparable efficacy. In contrast treatment with 5-FU resulted in early deaths and no survival benefit over controls (P = .24; Figure 6C). These data show that FdUMP[10] as a single agent confers a survival benefit comparable with that seen with a previously established combination regimen of Ara/Dox. The increased toxicity and early deaths seen with 5-FU but not FdUMP[10] treatment show that in vivo FdUMP[10] is not simply catabolized to the 5-FU moiety but rather behaves as a distinct chemical entity.
Figure 6.
FdUMP[10] confers a survival benefit equivalent to the combination of Ara-C and Dox. (A) Schema of treatment trial. C57/Bl6 mice were sublethally irradiated to 4.5 Gy and injected with an MLL-ENL and Flt3 ITD syngeneic leukemia. Once engraftment was established by bioluminescence imaging, mice were treated with either saline (S), FdUMP[10] at 300 mg/kg (Fd), 5-FU at 121 mg/kg or Ara-C at 125 mg/kg plus Dox at 3.75 mg/kg (AD) on days 1, 3, 5, and 7. (B) Bioluminescent image of mice on day 6 after treatment. (C) Kaplan-Meier curves for animals treated with Fd, 5-FU, or AD as in panel B.
FdUMP[10] causes less toxicity than either 5-FU or Ara-C plus Dox and spares normal HSCs
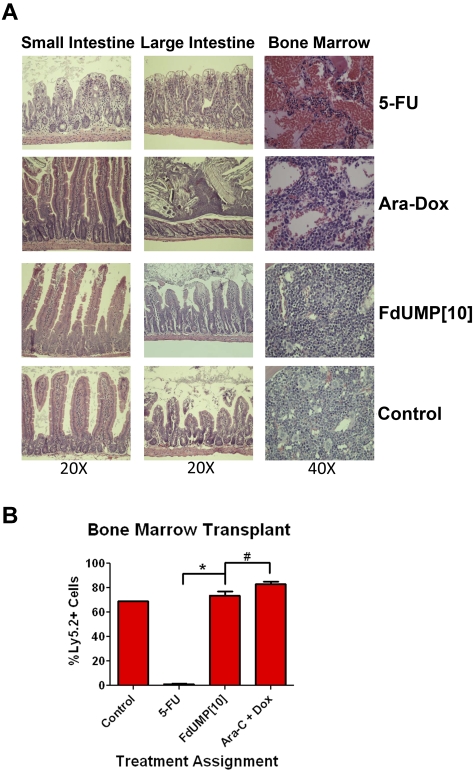
Previous studies have shown that the gastrointestinal (GI) tract toxicity caused by 5-FU is at least in part mediated by RNA-directed effects.28 Because FdUMP[10] is primarily metabolized to the DNA-directed metabolite, FdUMP, it should exhibit reduced GI tract toxicity compared with 5-FU. To compare the toxicity of FdUMP[10] with 5-FU and Ara/Dox, we treated C57/Bl6 mice with these agents as in the efficacy studies with animals dosed on days 1, 3, 5, and 7. Three days after the last treatment; animals were killed; bilateral femur marrow cells were collected; and organs were harvested, sectioned, and stained. Tissues most affected were the small intestine, colon, and the BM. In the GI tract, the 5-FU–treated animals had severe villous blunting and fusion with crypt necrosis in both large and small intestine (Figure 7A). In contrast, FdUMP[10]-treated animals had only mild crypt epithelial apoptosis with mitoses. The 5-FU and Ara/Dox groups had severe pancytopenia in the marrow compared with FdUMP[10]-treated animals that showed only minimal-to-mild apoptosis (Figure 7A). To assess the effect of FdUMP[10] on normal murine HSCs, harvested femur cells were counted, and 1 × 106 cells were injected into Ly5.1+ C57/Bl6 mice irradiated to 8 Gy. In the 5-FU–treated animals 1 × 106 cells could not be harvested, so 90% of all cells collected were injected. Three weeks later the recipients were killed, and BM was harvested and stained for Ly5.2 to determine the extent of engraftment. Cells from mice treated with FdUMP[10] engrafted to the same extent as the control animals, and the level of engraftment was not significantly different from animals treated with Ara/Dox (P = .083; Figure 7B). In contrast, the cells from the 5-FU–treated animals showed only minimal engraftment. These data suggest that, despite the enhanced activity of FdUMP[10] against AML, there is in fact less toxicity compared with 5-FU or Ara/Dox. The fact that BM cells from FdUMP[10]-treated animals efficiently engrafted in recipients suggests a large therapeutic window and provides further evidence that its mechanism of action is distinct from 5-FU.
Figure 7.
FdUMP[10] induces less toxicity than either 5-FU or Ara-C plus Dox. (A) H&E staining of organs from mice treated with either FdUMP[10], 5-FU, or Ara/Dox. Mice were treated in the same manner as in Figure 6 and killed 72 hours after the last dose, and organs were harvested, fixed, paraffin-embedded, sectioned, and stained. Slides were imaged with a Nikon Eclipse 50i light microscope, magnification as indicated. Photographs of tissues were taken with the NIS Elements D3.10 camera and software system. (B) BM transplantation. Donor mice were treated with 5-FU, FdUMP[10], or Ara-C/Dox as in panel A, and 72 hours after last doses animals were killed and b/l femur cells were harvested. Ly5.1+ C57/Bl6 recipient mice were irradiated to 8 Gy and injected with donor cells. After 3 weeks recipients were harvested, and degree of femur engraftment by donor cells was determined by staining with Ly5.2 Ab and analyzed by flow cytometry. *P < .0001 and #P = .0827.
FdUMP[10] is synergistic with Ara-C and Dox in vitro
Given that standard therapy for AML is combination chemotherapy and the impressive efficacy and tolerability data of FdUMP[10], we sought to determine whether FdUMP[10] would synergize with either Ara-C or Dox. HL60 cells were treated with Ara-C or Dox with or without the addition of FdUMP[10], and viability was determined. The degree of synergy was assessed with the combinatorial index method of Chou and Talalay.29 We found FdUMP[10] was synergistic with both Dox and Ara-C with combinatorial index values as low as 0.758 (± 0.015) for Dox and 0.3765 (± 0.014) for Ara-C (supplemental Figure 9). These data suggest that FdUMP[10] is synergistic with anthracyclines and Ara-C. Further studies to evaluate the efficacy of FdUMP[10] in combination with traditional therapies in vitro and in vivo are ongoing.
Discussion
AML is an aggressive malignancy with poor outcomes. Despite decades of research, therapy has remained largely unchanged. AML is primarily a disease of the elderly, with a median age of onset of 72 years5; remissions come at the cost of considerable toxicity. In a recent report of older patients treated with standard therapy, 32% of patients either died or could not receive additional therapy because of severe morbidity.4 In elderly patients who do not die of treatment-related toxicity or resistant disease, remissions are short lived, with median survival times of only ∼ 12 months.5 Novel agents are critically needed. To this end we tested the activity of the novel polymeric fluoropyrimidine, FdUMP[10]. This compound is a deoxyoligonucleotide made up entirely of the TS-inhibitory metabolite of 5-FU, FdUMP. As a deoxyoligonucleotide it would be predicted to have distinct uptake and metabolic breakdown products with an increase in the production of the DNA-directed metabolite, FdUMP, compared with 5-FU. Consistent with this, FdUMP[10] showed a 338-fold increase in potency against the NCI 60 cell line screen and decreased toxicity compared with 5-FU.8,30 In addition, FdUMP[10] has shown efficacy against malignancies not typically treated with 5-FU.9,31 When we tested FdUMP[10] against several preclinical models of AML we made several important observations.
First, FdUMP[10] was highly cytotoxic against multiple human leukemia cell lines with IC50 values lower than those for Dox or Ara-C. Importantly, it showed an ∼ 1000-fold increased potency compared with 5-FU despite having only 10 times the FP content, suggesting a distinct mechanism of action. Second, the cytotoxicity of FdUMP[10] was not dramatically affected by several factors shown to contribute to resistance to standard therapies or confer an adverse prognosis. FdUMP[10] was extremely potent in MLL-ENL–expressing murine AML cells, including those expressing the Flt3 ITD. The presence of an shRNA that targets p53 did not affect response to FdUMP[10] although it conferred resistance to both Dox and Ara-C. Likewise, when murine AML cells expressed high levels of MN1 or BCR-ABL, conferring resistance to both Ara-C and Dox, only minimal changes were noted in FdUMP[10] response. Third, FdUMP[10] inhibited leukemia stem cell engraftment in a murine AML model as well as colony formation in both human cell lines and primary patient samples while sparing normal human hematopoietic cells. Fourth, FdUMP[10] showed profound TS inhibition, trapping of Top1CC, DNA damage, and induction of apoptosis. Importantly 5-FU did not display these activities despite being used at doses that would deliver the same or more FP content, showing that FdUMP[10] exhibits a distinct mechanism of action with dual TS- and Top1-directed activity. All human leukemia cell lines and virtually all patient samples expressed both targets, suggesting FdUMP[10] probably has broad activity in AML. Finally, FdUMP[10] was found to have in vivo activity against 2 MLL-ENL–expressing, syngeneic mouse leukemias which was comparable with a combination of Ara-C and Dox. This activity did not come at the cost of increased toxicity because animals treated with FdUMP[10] showed the least GI tract and BM toxicity compared with Ara-C and Dox or 5-FU–treated animals. Indeed marrow cells taken from FdUMP[10]-treated animals engrafted well into irradiated recipients, in contrast to 5-FU–treated animals whereby little to no engraftment was detected.
These data show that FdUMP[10] is not simply a delivery vehicle for 5-FU but has a distinct mechanism of action. This is in agreement with previous studies,8,30,32 including a recent genome-wide profiling study for determinants of sensitivity to FdUMP[10] that found expression of genes involved in endocytosis as important for activity, arguing the molecule is taken up whole.31 Consistent with FdUMP[10] having a unique mechanism of action we showed that it efficiently traps Top1CC, an activity not ascribed to 5-FU. The notion that FdUMP[10] acts at least in part as a Top1 poison is supported by the fact that a COMPARE analysis done with the NCI 60 cell line panel found that the top 4 similar drugs were all camptothecin derivatives.30 This is consistent with the fact that camptothecins have activity in AML, as shown in several clinical trials.33,34
The data support the hypothesis that FdUMP[10] is taken up by AML cells, converted to the TS inhibitory metabolite FdUMP, and causes simultaneous DNA damage and nucleotide imbalances that make repair impossible. This dual activity suggests that cells with impaired DNA damage responses that can confer resistance to DNA-damaging agents will not be resistant to FdUMP[10] because the damage must be repaired to resolve the trapped Top1CCs before cells can complete S phase. The fact that p53 loss did not affect response to FdUMP[10] is consistent with this hypothesis.
The improved toxicity profile of FdUMP[10] may be explained by the fact that Top1-trapping activity requires incorporation of FdUMP or dUMP into DNA by actively replicating cells. This is in contrast to 5-FU, which causes ribosomal stress and inhibits RNA processing, and camptothecins, which cause DNA DSBs as a result of trapping Top1 cleavage complexes in transcriptionally active, nonreplicating cells. Similarly, the anthracyclines can induce DNA damage in cells regardless of cell-cycle phase, leading to increased toxicity. The reduced systemic toxicity for FdUMP[10] relative to 5-FU observed in our studies probably results from reduced RNA-directed effects.
In the past 3 decades the standard therapy for remission induction in AML has not changed.2 The unique mechanism of action for FdUMP[10] with dual targeting of TS and Top1 results in strong efficacy even when cells expressed several markers of poor prognosis. This, taken together with the reduced systemic toxicity, makes FdUMP[10] an ideal candidate for translation to the clinic. Thus, FdUMP[10] is probable to have broad clinical activity in AML and to provide a less-toxic alternative to the current standard of care applicable to most, if not all, patients with AML.
Supplementary Material
Acknowledgments
The authors thank Dr Mark Mendin for his generous gift of the OCI-AML3 cell line; Melisa Tanverdi, Erica Peronto, and Teresa Mascenik for technical assistance; and Karen Klein for help editing the manuscript.
This work was supported by the Doug Coley Foundation for Leukemia Research, the Frances P. Tutwiler Fund, the Translational Science Institute of Wake Forest University Comprehensive Cancer Center, the National Cancer Institute (award P30CA012197; T.S.P.), and the National Institutes of Health (grant CA102532; W.H.G.).
The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Cancer Institute or the National Institutes of Health.
Footnotes
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Authorship
Contribution: T.S.P. designed and performed research, analyzed data, and wrote the manuscript; E.G. performed research; J.J.-G. performed research; D.C. analyzed pathology specimens; and W.H.G. helped write the manuscript, analyzed data, and designed research.
Conflict-of-interest disclosure: W.H.G. is the president of Salzburg Therapeutics. The remaining authors declare no competing financial interests.
Correspondence: Timothy S. Pardee, Section on Hematology and Oncology, Department of Internal Medicine, Wake Forest University Health Sciences, 1 Medical Center Blvd, Winston-Salem, NC 27157; e-mail: tspardee@wakehealth.edu.
References
- 1.Licht JD, Sternberg DW. The molecular pathology of acute myeloid leukemia. Hematology Am Soc Hematol Educ Program. 2005:137–142. doi: 10.1182/asheducation-2005.1.137. [DOI] [PubMed] [Google Scholar]
- 2.Dohner H, Estey EH, Amadori S, et al. Diagnosis and management of acute myeloid leukemia in adults: recommendations from an international expert panel, on behalf of the European LeukemiaNet. Blood. 2010;115(3):453–474. doi: 10.1182/blood-2009-07-235358. [DOI] [PubMed] [Google Scholar]
- 3.Appelbaum FR, Gundacker H, Head DR, et al. Age and acute myeloid leukemia. Blood. 2006;107(9):3481–3485. doi: 10.1182/blood-2005-09-3724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Rollig C, Thiede C, Gramatzki M, et al. A novel prognostic model in elderly patients with acute myeloid leukemia: results of 909 patients entered into the prospective AML96 trial. Blood. 2010;116(6):971–978. doi: 10.1182/blood-2010-01-267302. [DOI] [PubMed] [Google Scholar]
- 5.Juliusson G, Antunovic P, Derolf A, et al. Age and acute myeloid leukemia: real world data on decision to treat and outcomes from the Swedish Acute Leukemia Registry. Blood. 2009;113(18):4179–4187. doi: 10.1182/blood-2008-07-172007. [DOI] [PubMed] [Google Scholar]
- 6.Kantarjian H, Ravandi F, O'Brien S, et al. Intensive chemotherapy does not benefit most older patients (age 70 years or older) with acute myeloid leukemia. Blood. 2010;116(22):4422–4429. doi: 10.1182/blood-2010-03-276485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Longley DB, Harkin DP, Johnston PG. 5-fluorouracil: mechanisms of action and clinical strategies. Nat Rev Cancer. 2003;3(5):330–338. doi: 10.1038/nrc1074. [DOI] [PubMed] [Google Scholar]
- 8.Gmeiner WH, Skradis A, Pon RT, Liu J. Cytotoxicity and in-vivo tolerance of FdUMP[10]: a novel pro-drug of the TS inhibitory nucleotide FdUMP. Nucleosides Nucleotides. 1999;18(6-7):1729–1730. doi: 10.1080/07328319908044836. [DOI] [PubMed] [Google Scholar]
- 9.Liao ZY, Sordet O, Zhang HL, et al. A novel polypyrimidine antitumor agent FdUMP[10] induces thymineless death with topoisomerase I-DNA complexes. Cancer Res. 2005;65(11):4844–4851. doi: 10.1158/0008-5472.CAN-04-1302. [DOI] [PubMed] [Google Scholar]
- 10.Pardee TS, Zuber J, Lowe SW. The Flt3 internal tandem duplication alters chemotherapy response in vitro and in vivo in a p53-dependent manner. Exp Hematol. 2011;39(4):473–485.e4. doi: 10.1016/j.exphem.2011.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Shankar DB, Li J, Tapang P, et al. ABT-869, a multitargeted receptor tyrosine kinase inhibitor: inhibition of FLT3 phosphorylation and signaling in acute myeloid leukemia. Blood. 2007;109(8):3400–3408. doi: 10.1182/blood-2006-06-029579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lee BD, Sevcikova S, Kogan SC. Dual treatment with FLT3 inhibitor SU11657 and doxorubicin increases survival of leukemic mice. Leuk Res. 2007;31(8):1131–1134. doi: 10.1016/j.leukres.2006.11.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Schlenk RF, Dohner K, Krauter J, et al. Mutations and treatment outcome in cytogenetically normal acute myeloid leukemia. N Engl J Med. 2008;358(18):1909–1918. doi: 10.1056/NEJMoa074306. [DOI] [PubMed] [Google Scholar]
- 14.Yin B, Kogan SC, Dickins RA, Lowe SW, Largaespada DA. Trp53 loss during in vitro selection contributes to acquired Ara-C resistance in acute myeloid leukemia. Exp Hematol. 2006;34(5):631–641. doi: 10.1016/j.exphem.2006.01.015. [DOI] [PubMed] [Google Scholar]
- 15.Ravizza R, Gariboldi MB, Passarelli L, Monti E. Role of the p53/p21 system in the response of human colon carcinoma cells to Doxorubicin. BMC Cancer. 2004;4:92. doi: 10.1186/1471-2407-4-92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Seifert H, Mohr B, Thiede C, et al. The prognostic impact of 17p (p53) deletion in 2272 adults with acute myeloid leukemia. Leukemia. 2009;23(4):656–663. doi: 10.1038/leu.2008.375. [DOI] [PubMed] [Google Scholar]
- 17.Nahi H, Lehmann S, Bengtzen S, et al. Chromosomal aberrations in 17p predict in vitro drug resistance and short overall survival in acute myeloid leukemia. Leuk Lymphoma. 2008;49(3):508–516. doi: 10.1080/10428190701861645. [DOI] [PubMed] [Google Scholar]
- 18.Cotter TG. BCR-ABL: an anti-apoptosis gene in chronic myelogenous leukemia. Leuk Lymphoma. 1995;18(3-4):231–236. doi: 10.3109/10428199509059612. [DOI] [PubMed] [Google Scholar]
- 19.Soupir CP, Vergilio JA, Dal Cin P, et al. Philadelphia chromosome-positive acute myeloid leukemia: a rare aggressive leukemia with clinicopathologic features distinct from chronic myeloid leukemia in myeloid blast crisis. Am J Clin Pathol. 2007;127(4):642–650. doi: 10.1309/B4NVER1AJJ84CTUU. [DOI] [PubMed] [Google Scholar]
- 20.Liu T, Jankovic D, Brault L, et al. Functional characterization of high levels of meningioma 1 as collaborating oncogene in acute leukemia. Leukemia. 2010;24(3):601–612. doi: 10.1038/leu.2009.272. [DOI] [PubMed] [Google Scholar]
- 21.Heuser M, Beutel G, Krauter J, et al. High meningioma 1 (MN1) expression as a predictor for poor outcome in acute myeloid leukemia with normal cytogenetics. Blood. 2006;108(12):3898–3905. doi: 10.1182/blood-2006-04-014845. [DOI] [PubMed] [Google Scholar]
- 22.Langer C, Marcucci G, Holland KB, et al. Prognostic importance of MN1 transcript levels, and biologic insights from MN1-associated gene and microRNA expression signatures in cytogenetically normal acute myeloid leukemia: a cancer and leukemia group B study. J Clin Oncol. 2009;27(19):3198–3204. doi: 10.1200/JCO.2008.20.6110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Chan WI, Huntly BJ. Leukemia stem cells in acute myeloid leukemia. Semin Oncol. 2008;35(4):326–335. doi: 10.1053/j.seminoncol.2008.04.003. [DOI] [PubMed] [Google Scholar]
- 24.Gmeiner WH, Trump E, Wei C. Enhanced DNA-directed effects of FdUMP[10] compared to 5FU. Nucleosides Nucleotides Nucleic Acids. 2004;23(1-2):401–410. doi: 10.1081/ncn-120028336. [DOI] [PubMed] [Google Scholar]
- 25.Nervi B, Ramirez P, Rettig MP, et al. Chemosensitization of acute myeloid leukemia (AML) following mobilization by the CXCR4 antagonist AMD3100. Blood. 2009;113(24):6206–6214. doi: 10.1182/blood-2008-06-162123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Majeti R, Chao MP, Alizadeh AA, et al. CD47 is an adverse prognostic factor and therapeutic antibody target on human acute myeloid leukemia stem cells. Cell. 2009;138(2):286–299. doi: 10.1016/j.cell.2009.05.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Pritchard DM, Watson AJ, Potten CS, Jackman AL, Hickman JA. Inhibition by uridine but not thymidine of p53-dependent intestinal apoptosis initiated by 5-fluorouracil: evidence for the involvement of RNA perturbation. Proc Natl Acad Sci U S A. 1997;94(5):1795–1799. doi: 10.1073/pnas.94.5.1795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Liu J, Skradis A, Kolar C, et al. Increased cytotoxicity and decreased in vivo toxicity of FdUMP[10] relative to 5-FU. Nucleosides Nucleotides. 1999;18(8):1789–1802. doi: 10.1080/07328319908044843. [DOI] [PubMed] [Google Scholar]
- 29.Chou TC, Talalay P. Quantitative analysis of dose-effect relationships: the combined effects of multiple drugs or enzyme inhibitors. Adv Enzyme Regul. 1984;22:27–55. doi: 10.1016/0065-2571(84)90007-4. [DOI] [PubMed] [Google Scholar]
- 30.Gmeiner WH, Reinhold WC, Pommier Y. Genome-wide mRNA and microRNA profiling of the NCI 60 cell-line screen and comparison of FdUMP[10] with fluorouracil, floxuridine, and topoisomerase 1 poisons. Mol Cancer Ther. 2010;9(12):3105–3114. doi: 10.1158/1535-7163.MCT-10-0674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Liu C, Willingham M, Liu J, Gmeiner WH. Efficacy and safety of FdUMP[10] in treatment of HT-29 human colon cancer xenografts. Int J Oncol. 2002;21(2):303–308. [PubMed] [Google Scholar]
- 32.Minderman H, O'Loughlin KL, Smith PF, et al. Sequential administration of irinotecan and cytarabine in the treatment of relapsed and refractory acute myeloid leukemia. Cancer Chemother Pharmacol. 2006;57(1):73–83. doi: 10.1007/s00280-005-0017-4. [DOI] [PubMed] [Google Scholar]
- 33.Weihrauch MR, Staib P, Seiberlich B, Hoffmann M, Diehl V, Tesch H. Phase I/II clinical study of topotecan and cytarabine in patients with myelodysplastic syndrome, chronic myelomonocytic leukemia and acute myeloid leukemia. Leuk Lymphoma. 2004;45(4):699–704. doi: 10.1080/10428190310001593175. [DOI] [PubMed] [Google Scholar]
- 34.Bolanos-Meade J, Guo C, Gojo I, Karp JE. A phase II study of timed sequential therapy of acute myelogenous leukemia (AML) for patients over the age of 60: two cycle timed sequential therapy with topotecan, ara-C and mitoxantrone in adults with poor-risk AML. Leuk Res. 2004;28(6):571–577. doi: 10.1016/j.leukres.2003.10.026. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.