Abstract
Elimination of cisplatin resistant (CR) lung cancer cells remains a major obstacle. We have shown that CR tumors have higher reactive oxygen species (ROS) levels and can be exploited for targeted therapy. Here we show that increased secretion of the antioxidant thioredoxin-1 (TRX1) resulted in lowered intracellular TRX1, and contributed to higher ROS in CR tumors in vivo and in vitro. By reconstitutingTRX1 protein in CR cells, we increased sensitivity to cisplatin but decreased sensitivity to elesclomol (ROS inducer). Conversely, decreased TRX1 protein in parental cells reduced the sensitivity to cisplatin but increased sensitivity to elesclomol. CR cells had increased endogenous oxygen consumption and mitochondrial activity but decreased lactic acid production. They also exhibited higher levels of argininosuccinate synthetase (ASS) and fumarase (FH) mRNA which contributed to oxidative metabolism (OXMET) when compared to parental cells. Restoring intracellular TRX1 protein in CR cells resulted in lowering ASS and FH mRNAs which in turn sensitized them to arginine deprivation. Interestingly, CR cells also possessed significantly higher basal levels of acetyl-CoA-carboxylase (ACC) and fatty acid synthase (FAS). Over-expressing TRX1 lowered ACC and FAS proteins expressions in CR cells. Chemical inhibition and siRNA of ACC resulted in significant cell death in CR compared to parental cells. Conversely, TRX1 over-expressed CR cells resisted to TOFA-induced death. Collectively, lowering TRX1 expression through increased secretion leads CR cells to higher ROS production and increase in dependency on OXMET. These changes raise an intriguing therapeutic potential for future therapy in cisplatin resistant lung cancer.
Keywords: Lung cancer, Cisplatin resistance, ROS, Oxidative metabolism, Thioredoxin-1
Introduction
Cisplatin is a widely used therapeutic agent in the treatment of several types of solid tumors including lung cancer (both small cell and non small cell), head and neck and ovarian cancer. The majority of cancer patients initially respond to cisplatin treatment, however development of drug resistance is expected resulting in disease progression. Mechanisms of resistance are complex and involve multiple molecular pathways (1), making cisplatin resistance difficult to overcome. Thus far, no drugs can reverse cisplatin resistance or selectively kill cisplatin resistant cells.
We have previously discovered that elevated reactive oxygen species (ROS) are found in cisplatin resistant (CR) cell lines (2); however, the question remains as to why this occurs. A number of investigators have shown that cisplatin can inhibit thioredoxin reductase1 (TrxR1), a thioredoxin reducing enzyme, which leads to increased ROS, resulting in further damage of DNA and subsequent cell death (3, 4). In order to avoid cell death caused by cisplatin, cells adapt to survive at high ROS levels and use less TrxR1/thioredoxin-1 but will up-regulate other antioxidant systems (5). We have reported lower thioredoxin (TRX1) in a panel of CR lung cancer cells tested (2). However, it is not known whether decreased TRX1 protein was due to transcriptional down-regulation or increased in secretion. Increased TRX1 secretion has been reported by others as a result of cellular stress and after cisplatin treatment (6–8). We investigated whether decreased TRX1 found in CR cells is regulated at the mRNA or the protein level and report our results herein.
TRX1, a disulfide-reducing dithiol enzyme, is an important antioxidant protein which facilitates the reduction of other enzymes (9). TRX1 also interacts with certain transcription factors which are known to be redox-regulated via the dithiol-disulfide exchange reaction, thereby altering their DNA binding capacity (10, 11). Interestingly, it has been reported recently that TRX1 is involved in adipogenesis in fibroblast cells (12). This study showed that down-regulation of TRX1 can lead to increase in lipogenesis. Thus, it is possible that lower TRX1 found in CR cells may have an impact on the fatty acid (FA) synthesis pathway and other pathways that are involved in oxidative metabolism (OXMET).
In this report, we present evidence that decreased TRX1 expression in cisplatin resistant lung cancer cells is an important mediator of ROS and reprograms lung cancer cells to become more reliant on OXMET. To our knowledge, this is the first report which demonstrates that decreased TRX1 in cisplatin resistant lung cancer can result in increased ROS and alteration in tumor metabolism. These biochemical changes can be used as a target for future treatment of cisplatin resistant cells.
Materials and Methods
Cell Lines and reagents
SCLC1 was derived from the bone marrow of SCLC (small cell lung cancer) patient, SR2 is the cisplatin resistant variant derived from SCLC1 which was generated by intermittent exposure to cisplatin, S (non small cell lung cancer) was established from metastatic adenocarcinoma to the brain, SC is a cisplatin resistant variant derived from S by intermittent exposure to cisplatin. These cell lines have been previously characterized (13–16). Note: SR2 exhibits 20fold resistances to cisplatin and S exhibits 7fold resistance to both cisplatin and carboplatin. Elesclomol was kindly provided by Synta Pharmaceuticals. TOFA (5-(Tetradecyloxy)-2-Furoic Acid) was purchased from Sigma.
Growth inhibition and cytotoxicity assay
Cells were seeded in 24-well dishes and treated with various concentrations of elesclomol or cisplatin for 72 hrs. as described previously (13, 17). At 72 hrs., the culture mediums as well as the trypsinized cells were collected and this admixture was centrifuged at 400 × g for 5 min. The cell pellet was resuspended in 1 mL of Hank's buffer and assayed for live cells and death cells using trypan blue exclusion method.
Western Blot analysis
Cells were seeded at 1 × 105/ml onto 100 mm dishes, treated, collected and lysed. Total protein was separated on an SDS-PAGE, transferred onto a nitrocellulose membrane (Amersham Biosciences) and immunoblotted with ACL, ACC, FAS (Cell Signaling Inc.), TRX1 (BD Bioscience), TRX2 (Santa Cruz), Citrate synthase (Sigma), or Actin (Sigma) (13, 17). The signal intensity was measured using a molecular imager Chemidoc system with Quality One software (Bio-Rad).
Assay of Intracellular ROS/H2O2
As previously described (2), cells were collected and intracellular H2O2 was measured by incubating with 10 µM of acetyl-penta-fluorobenzenesulfonyl fluorescein (APFB) (EMD) at 37 °C for 30 min in the dark. Then the cells were washed once with PBS and centrifuged to remove impermeable reagents. Cells were resuspended in 500 µL of PBS and analyzed in a fluorometer, FLUOstar OPTIMA, BMG Labtech (excitation at 485 nm and emission at 520 nm).
Assay of mitochondrial membrane potential (MMP)
Cells were collected and incubated with 50 nM of tetramethylrhodamine ethyl ester (TMRE) (Invitrogen) at 37 °C for 30 min in the dark. Then the cells were washed once with PBS and centrifuged to remove impermeable reagents. Cells were resuspended in 500 µL of PBS and analyzed in Accuri Flow Cytometer (excitation at 544 nm and emission at 590 nm) for the MMP.
Determination of the concentration of TRX1 in the culture medium or in the plasma
The concentration of TRX1 was determined by sandwich enzyme-linked immunosorbent assay (ELISA) with two TRX1 antibodies. Tetra Methyl Benzidine (TMB) was used as coloring agent (Chromogen). The strength of coloring was proportional to the quantities of human TRX1. Briefly: 100 uL of samples (medium or plasma), standards, or reagent blank were incubated with 96 wells precoated plate for 60 mins at 37°C (IBL, Co., Ltd). The precoated plate was washed vigorously with wash buffer 7 times. 100uL of labeled antibody solution was added to each well and incubated for 30 min at 37°C. The precoated plate was washed with buffer. 100uL of TMB was added to each well incubated for 30 mins at room temperature in the dark. After 30min, stop solution was added into each well, then subjected to ELISA plate reader at 450nm against reagent blank. TRX1 concentration was quantified against the standard curve and normalized with the cell number.
Assay of thioredoxin reductase (TrxR) activity
A TrxR kit (Cayman Chemical) was used to measure total cellular thioredoxin reductase as previously described (2). Briefly, cells were seeded at 4 × 105 and cell lysate was prepared by sonication using the conditions recommended by the manufacturer. Total TrxR activity was detected by measuring the reduction of DTNB with NADPH to TNB by UV spectrophotometer at 405 nm.
RNA interference experiments
8×105 cells were seeded in a 60mm petri dish and incubated for 24hrs. INTERFERin™ transfection reagent (Polyplus) was then used to transfect following siRNAs (Dharmacon): siTRXC1 (GAAAAGUAUUCCAACGUGA), siTRXC2 (GGACGCUGCAGGUGAUAAA), siACC (SMARTpool®siRNA,NM_198836), and siCONTROL® (UAGCGACUAAACACAUCAA) (17).
TRX1 over-expression experiment
TRX1-cDNA with restriction site Sgf I and Mlu l was cloned in to the pCMV6 expression vector (Origene). The plasmid DNA was transfected into cultured cells (8×105) using Fugene®6 (Roche). After 24hrs, 0.5mg/ml of neomycin(G418) was added to the culture media for transfection selection. Two Clonal populations (SR2TRX+C1 and SR2TRX+C2) were selected and maintained in media containing 0.2mg/ml of neomycin.
Assay for lactate production
The lactate assay was carried out with a kit (Biovision). Following the manufacture’s instruction, 200 µl/well of media was filter through 10 kd molecular weight spin filter. 50 µl of supernatant were then added to reaction mix which contained lactate assay buffer, probe, enzyme mix, and incubated for 30 mins. Lactate levels were measured by ELISA plate reader at 570 nm against reagent blank (media alone).
Qualitative real-time PCR
qRTPCR was carried out as previously described (18). Briefly, 1ug of RNA was used for cDNA synthesis. The primers for qRT-PCR are designed with Beacon Designer for SYBR Green fluorophore. 40 cycle amplification was used. The data were analyzed with iQ5 software from Bio-Rad. To calculate the relative mRNA level, we used the ΔΔCt method. The level of mRNA was corrected with that of GAPDH.
Assay of oxygen consumption
Live cells were trypsinized and counted. Cells were then resuspended in 1 ml of RPMI supplemented with 5% FBS and 2 mg/ml of glucose. Each cell line was tested three or more independent times for a minimum of six readings per cell. Oxygen consumption readings were performed as suggested by the manufacturer (Yellow Springs Instruments model 5300).
Immunohistochemistry staining
Immunohistochemical staining was performed according to routine methodology with some modification. We used the target retrieval solution and the biotin block system (both from Dako) to enhance the staining. Samples were incubated overnight with primary antibody (1:200 in antibody dilution solution (Dako S3022)) and washed. Secondary antibody solution (Dako Link, biotinylated antibody. solution) was added for 25 min, washed, and streptavidin conjugated to peroxidase (HRP) solution was added for 25 min. DAB chromogen (Dako) and then hemotoxylin were used for staining. Section with only antibody solution was used as control. A modified protocol without the rehydration steps was used for cells.
Statistical analysis
All statistical analyses were performed from three separate measurements using the two-tailed t-test and the results were expressed as mean ± standard deviation. A p-value of less than 0.05 was considered as statistically significant.
Results
Decreased thioredoxin-1 (TRX-1) in CR cells is due to increased secretion
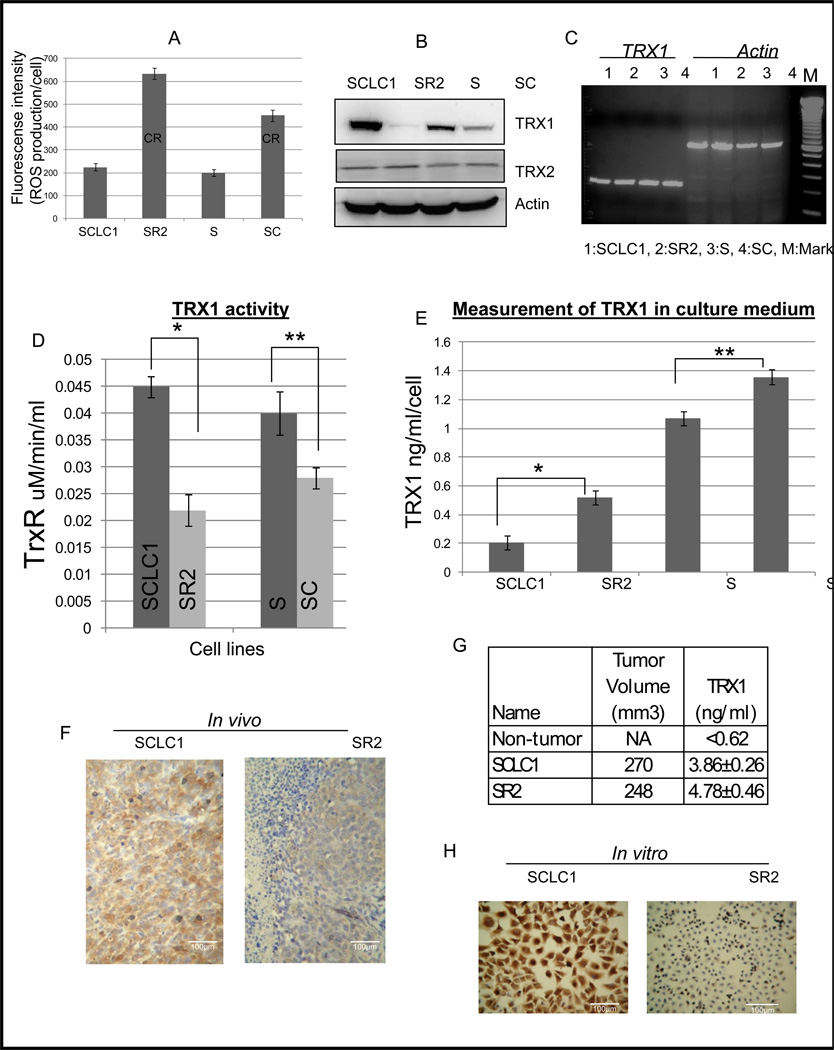
To confirm that CR cells possess higher basal levels of ROS, we assayed for H2O2 production per cell in CR vs. parental cells (Fig. 1A). Our previous results also showed that TRX1 was lower in CR cells and most likely contributes to higher ROS levels. TRX has two functional isoforms; TRX1 localized in the cytoplasm and TRX2 in the mitochondria. In this report, we have clarified that the TRX1 isoforms were decreased while TRX2 showed no changes in all lung cancer cell lines tested (Fig. 1B). We then investigated whether decreased TRX1 protein was due to a decrease in transcription by evaluating the TRX1 mRNA using RT-PCR. TRX1mRNA levels were similar between the parental and CR cells (Fig. 1C), whereas their activities were significantly lower (Fig. 1D). Thus, our data suggest that the decreased TRX1 protein seen in CR cells occurs at the post transcriptional levels. Since it has been reported that TRX1 can be secreted from cells through the leaderless secretory pathway (19), it is possible that lower TRX1 found in CR cells may be due to the excessive secretion of this protein. We therefore compared the concentration of extracellular TRX1 in culture media from the parental vs. CR cells. Results showed that TRX1 concentrations in culture media from CR cells were significantly higher (Fig. 1E). To further investigate whether decreased TRX1 protein expression and increased TRX secretion also occur in vivo, we chose SCLC1 (parental) and SR2 (CR) as our xenograft model in SCID mice and assayed for TRX1 in tumors via immunohistochemical staining (Fig. 1F). Simultaneously, we assayed mouse serum for TRX1 (Fig. 1G). SR2 tumors clearly expressed less TRX1 intracellular protein consistent with the low intracellular expression we detected in vitro (Fig. 1H). Mice with SR2 xenografts had higher TRX1 in serum when compared to mice with SCLC1 xenografts. These findings were consistent with the increased TRX1 secretion we observed in culture (Fig. 1E). Taken together from the in vitro and in vivo models, our findings suggest that CR cells possess lower intracellular TRX1 protein that is not due to a transcriptional consequence, but rather due to an increase in secretion ultimately resulting in higher ROS accumulation.
Figure 1. CR lung cancer cells express lower levels of thioredoxin 1 protein.
(A) Fluorometer analysis of H2O2 in various lung cancer cell lines detected by APFB probe indicates that CR lung cancer cell lines expressed higher basal levels of ROS (Mean SD of three experiments). (B) Immunoblot of TRX1 and TRX2 in lung cancer cell lines showed that resistant variants expressed lower levels of TRX1 while no significant changes in TRX2 occurred. Actin was used as a loading control. (C) RT-PCR of TRX1 in lung cancer cell lines indicated that all 4 cell lines possess similar levels of TRX1 mRNA expression. (D) Using thioredoxin reductase 1 activity assay, CR cells possess lower TRX1 activity when compared to their parental cells (*p<0.001, **p=0.003). (E) The concentration of extracellular TRX1 in culture medium. CR cells secreted greater levels of TRX1 when compared to their parental counterparts (*P=0.001, **P=0.002). (F) Immunohistochemistry of TRX1 in mouse xenograft tissue showed that SR2 possess lower levels of TRX1 protein. (G) The concentration of extracellular TRX1 in mouse serum. Mice bearing SR2 xenografts were found to have higher levels of TRX1 in the serum. The value (ng/ml) represents the average of 3 mice per group. (p=0.03; SCLC1 vs. SR2). (H) Immunocytochemistry of TRX1 in lung cancer cell cultures also showed decreased levels of TRX1 in CR cells (200X).
Down-regulation of TRX1 correlates with increased ROS production, resistance to cisplatin, but sensitivity to elesclomol in a cisplatin sensitive cell line
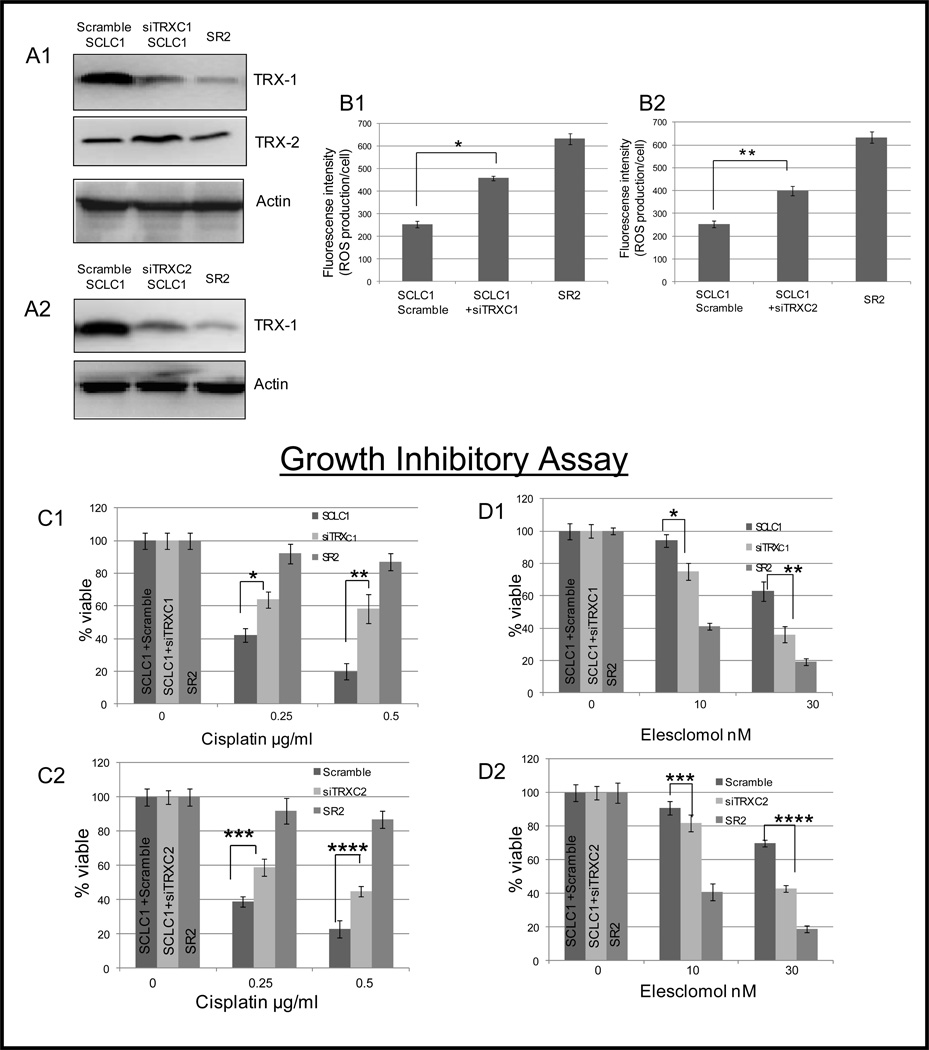
To determine whether TRX1 correlates with ROS levels and sensitivity to cisplatin as well as ROS producing agent (elesclomol), we inhibited TRX1 expression in SCLC1 using two different siRNA (SCLCsiTRXC1 and SCLCsiTRXC2). TRX1 protein was successfully knocked down (about 80% in the transfected cells, SCLCsiTRXC1 and SCLCsiTRXC2 (Fig. 2A1, 2A2) and higher levels of ROS were detected relative to SCLC1 (Fig. 2B1, 2B2). Nevertheless, the levels of ROS in TRX1 knocked down cells were still lower than in SR2 cells. It is conceivable that mitochondrial ROS may also contribute to high ROS in SR2. Since silencing TRX1 may affect TRX2, we studied TRX2 in SCLC1siTRXC1 and found only slightly increased in TRX2 levels (Fig. 2A). Whether TRX2 participates in ROS accumulation is currently unclear.
Figure 2. Down-regulation of TRX-1 results in increased ROS production, sensitivity to elesclomol, and resistance to cisplatin in a SCLC1 cell line.
We selected SCLC1 which possess the highest amount of TRX1 while its cisplatin resistant variant (SR2) had the lowest amount of TRX1 to do the study. (A1&A2) Immunoblot of TRX1 and TRX2 in SCLC1scramble (control), SR2, SCLCsiTRXC1, and SCLCsiTRXC2 cells Two different siRNAs were able to down-regulate TRX1 by 80% at 48 hr. post transfection while no effect occurred in TRX2. Actin was used as a loading control. (B1&B2) Down-regulation of TRX1 resulted in significant ROS production. (*p=0.02, **p=0.035). (C1&C2) Growth inhibitory effect of cisplatin for 72hrs showed that down-regulation of TRX1 in SCLC1 cells resulted in resistance to cisplatin treatment (*p=0.02, **P=0.001, ***p<0.05, ****p=0.04). (D1&D2) Growth inhibitory effect of elesclomol for 72hrs showed that down-regulation of TRX1 in SCLC1 results in increased sensitivity to elesclomol treatment (*p=0.01, **P=0.003, ***p=0.12, ****p=0.04). (Mean SD of three experiments)
Next, we evaluated the viability of these cells under cisplatin treatment. Both siTRX clones were significantly more resistant to cisplatin (Fig. 2C1, 2C2). At 0.5 µg/ml of cisplatin, 60% of the transfected cells were still viable while only 20% of the SCLC1 cells survived. We could not reach the similar cisplatin resistant levels as in SR2 cells which may due to the fact that we cannot knock down TRX1 down to the same levels as in SR2 and other mechanisms of resistance in SR2 were present (see discussion).
We have previously shown that CR cells which possess high ROS levels are sensitive to elesclomol (2). We anticipate that TRX1 knock down cells which have high ROS should be more sensitive to elesclomol. Indeed, we have found that both TRX1 knocked down clones (SCLC1siTRXC1 and SCLC1siTRXC2) were more sensitive to elesclomol (Fig. 2D1, 2D2). At 30 nM of elesclomol, 70% of SCLC1 cells were still viable while only 35% of the knocked down cells survived. These data suggest that TRX1 plays a role in determining elesclomol sensitivity via alteration in ROS levels.
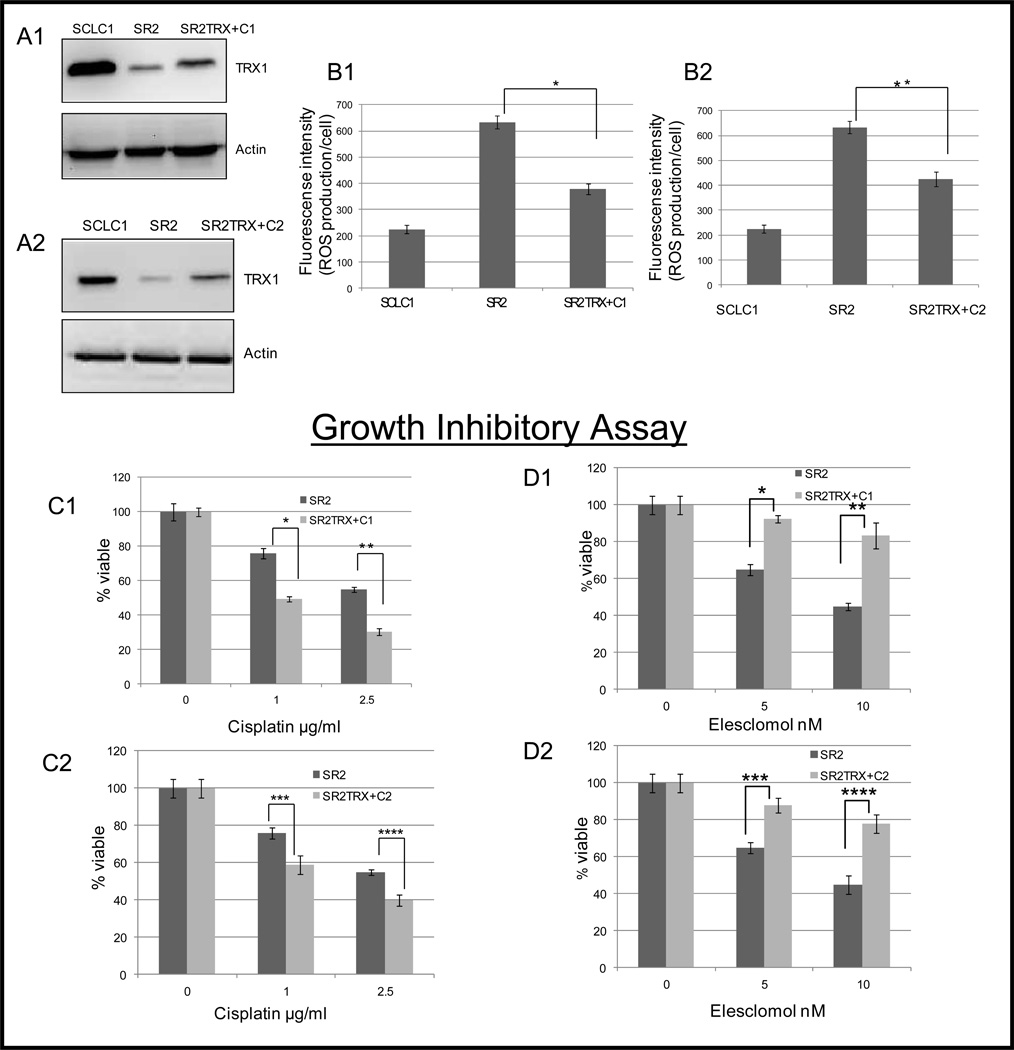
Over-expression of TRX-1 results in decreased ROS production, increased sensitivity to cisplatin treatment, but resistance to elesclomol in CR cells
To further verify the role of TRX1 in cisplatin resistance in lung cancer cells, we over-expressed TRX1 protein in SR2, using the pCMV6 vector containing full length TRX1 cDNA. We were able to generate two stable transfected cells (SR2TRX+C1 and SR2TRX+C2 which possess about 3–4 fold increase in TRX1 protein expression when compared to SR2 (Fig. 3A1, 3A2). These transfectants containing increased levels of TRX1 were used to study ROS production and sensitivity to cisplatin. Results showed that ROS production was significantly reduced in both TRX1over-expressed clones (Fig. 3B1, 3B2) while cisplatin sensitivity was increased (Fig. 3C1, 3C2). At 2.5 µg/ml of cisplatin, 55% of SR2 cells were still viable while about 30% and 40% of SR2TRX+C1 and SR2TRX+C2 survived, respectively. Next we analyzed the sensitivity of both clones to the ROS producing agent, elesclomol. As predicted, elesclomol had a lesser antitumor effect in TRX1 over-expressed cells than in SR2 cells. At 10 nM of elesclomol, only 45% of SR2 survived while in SR2TRX+C1 and C2 about 80% of the cells were still viable (Fig. 3D1, 3D2). Overall, our data clearly indicate that TRX1 levels have a major impact on high ROS levels seen in CR cells and that manipulation of ROS levels through TRX1 expression can alter cisplatin and elesclomol sensitivity. Although further work in this area is needed, it is possible that TRX1 can be used as a marker in the future selection of patients for cisplatin and/or elesclomol treatment.
Figure 3. Over-expression of TRX-1 results in decreased ROS production, resistance to elesclomol, and sensitivity to cisplatin in the SR2 cell line.
(A1&A2) Immunoblot of TRX1 in SCLC1, SR2, SR2TRX+C1, and SR2TRX+C2 cells. Both TRX1 over-expressing clones expressed about 3–4 fold increase in TRX protein when compared to SR2. Actin was used as a loading control. (B1&B2) Over-expressing of TRX1 resulted in decreased ROS production in SR2 cell lines (*p=0.002, **p=0.01). (C1&C2) Growth inhibitory effect of cisplatin for 72hrs showed that over-expressing of TRX1 in SR2 resulted in increased sensitivity to cisplatin treatment (*p=0.001, **p=0.03, ***p<0.05, ****p<0.05). (D1&D2) Growth inhibitory effect of elesclomol for 72hrs indicated that over-expressing of TRX1 in SR2 resulted in resistance to elesclomol treatment (*p=0.007, **P=0.006, ***p=0.008, ****p=0.004). (Mean SD of three experiments)
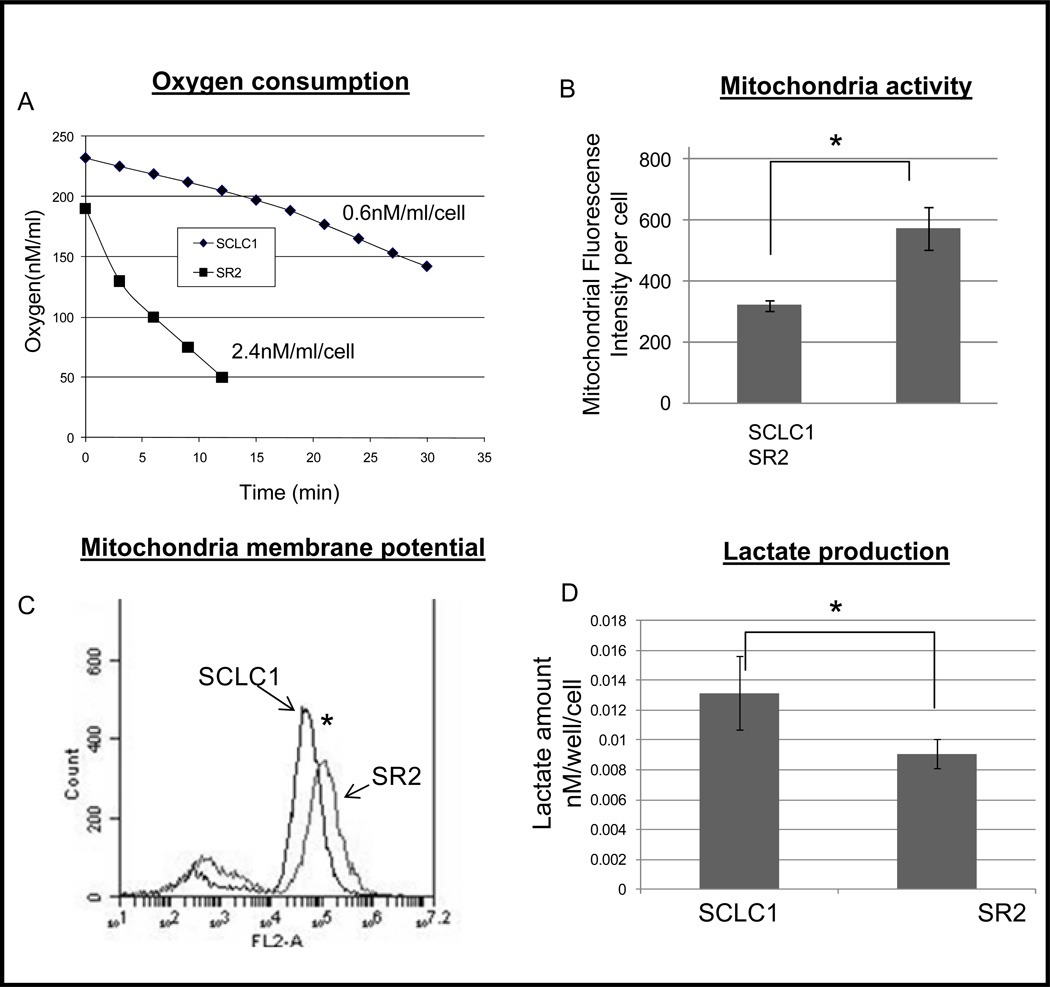
CR cells consume more oxygen and have more mitochondrial activity
Although we have shown that low intracellular TRX1 levels in CR cells contribute to higher ROS levels, it is also possible that these CR cells possess highly active mitochondria which are known to contribute to elevated ROS. To verify this, we first analyzed the amount of oxygen consumed in SCLC1 vs. SR2 cells. SCLC1 consumed 0.6 nM of 02/cell/ml of media while SR2 consumed 2.4 nM of 02/cell/ml of media in the same time period (Fig. 4A). These data indicate that CR cells utilize more oxygen (aerobic) than parental cells. Additionally, under hypoxic conditions (0.5% O2) CR cell growth was inhibited more than parental cell (data not shown). This observation led us to assess mitochondrial activity through mitochondrial staining (Mitotracker). SR2 had more intense fluorescence when compared to SCLC1 which is indicative of higher mitochondrial activity (Fig. 4B and Fig. S1). To further demonstrate that oxidative phosphorylation (OXPHOS) was utilized more in SR2, we measured mitochondrial membrane potential (MMP) using TMRE staining. Higher MMP correlates to more active OXPHOS (20). MMP was significantly higher in SR2 cells (Fig. 4C) than SCLC1, suggesting that CR cells have more highly active mitochondria which can also contribute to increased ROS production. Higher mitochondrial activity implies that CR cells may use more OXMET and less glycolytic flux. To consider this possibility, we assayed for the lactate production, which is a known indicator of glycolytic metabolism. We found lower amounts of lactic acid (nM/well/cell) in media from CR cells (Fig. 4D). Thus, our initial findings suggest that CR cells have higher mitochondrial activity and rely more on OXMET instead of glycolysis.
Figure 4. CR cells consume more oxygen and have higher mitochondrial activity than parental cells.
(A) SCLC1 and SR2 were assayed for baseline oxygen consumption. Live cells were counted and placed in an oxygen chamber. The amount of oxygen consumed per cell in 10min was measured. The rate of oxygen consumption (O2 nM/mL/cell/min) was four times higher in SR2 cells than SCLC1 cells. (B) SCLC1 and SR2 were incubated with 100nM of MitoTracker. Bar graph represents the relative fluorescent units/cell via fluorometer plate reader. SR2 cells possess higher number of active mitochondria when compared to SCLC1 (p<0.001). (C) Flow analysis of MMP in SCLC1 vs. SR2, using 50nM of TMRE. SR2 possess significant higher levels of MMP (p<0.001). (D) Media from the SCLC1 vs. SR2 cells was collected and used in a chromatogenic assay to measure amounts of lactic acid (nM/well/cell). SR2 cells produced less lactic acid than CR cells (Mean SD of three experiments; p<0.05).
CR cells preferentially used OXMET which was partially reversed upon over-expression of TRX1
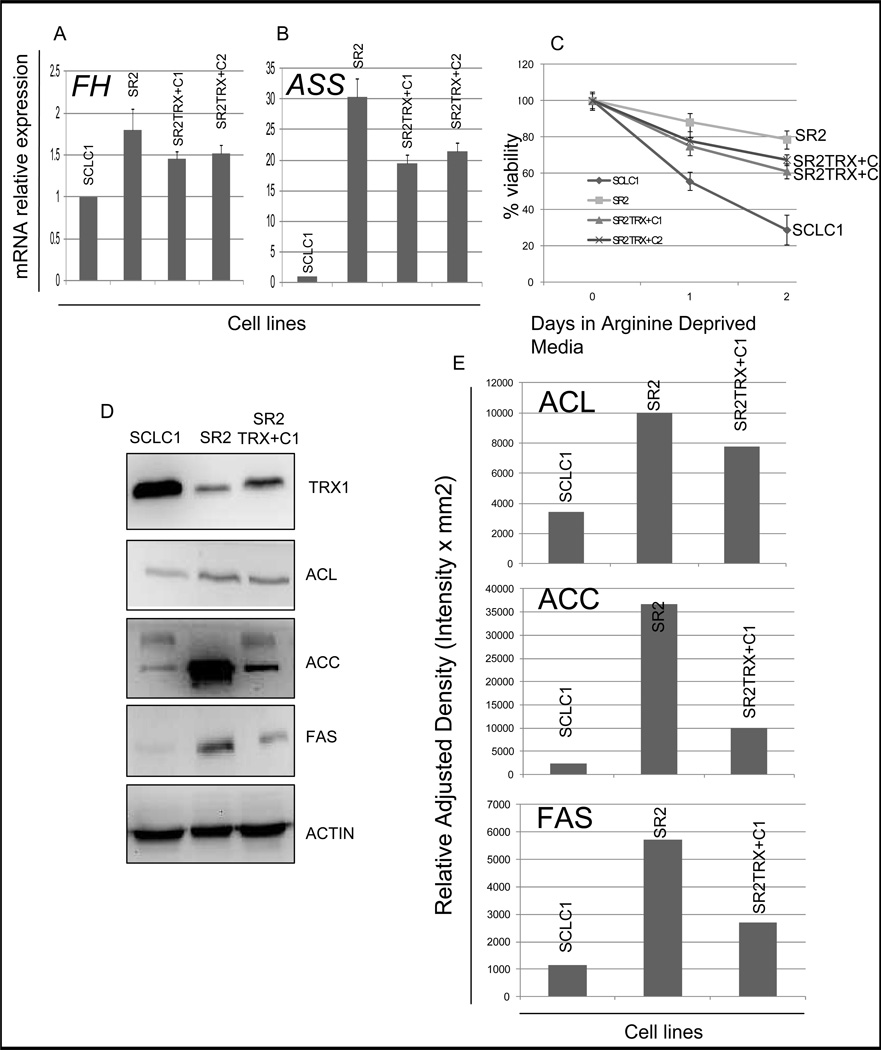
We further validated the involvement of specific OXMET components as well as the impact of TRX1 on cisplatin resistant lung cancer metabolism. To investigate the dependence of CR cells on OXMET and TRX1 as an important factor in this metabolic switching, we studied key proteins in the TCA cycle, the urea cycle, and FA synthesis using our cell lines model. Citrate synthase (CS) is the first rate determining enzyme in the TCA cycle (21, 22), while fumarase (FH) participates in maintaining the equilibrium between succinate and fumarate. This equilibrium impacts the functionality of complex II in the electron transport chain (ETC) (21). We found no significant changes in CS protein in all three cell lines (data not shown); however, FH mRNA was increased in SR2 (1.8fold) and attenuated in TRX1 transfected cells, SR2TRX+C1 and SR2TRX+C2 (Fig. 5A). FH hydrates fumarate to malate, an important intermediate in the TCA cycle; however, the major source of fumarate is from the urea cycle (23). Hence, we investigated two important enzymes in the urea cycle: argininosuccinate synthetase (ASS) and argininosuccinate lyase (ASL) which generate arginine and yield fumarate as by-product. Interestingly, SCLC1 possessed a negligible amount of endogenous ASS mRNA (Fig. 5B) while SR2 had a relatively robust expression (30 fold increase). Both TRX1 over-expressed transfectants showed a decrease in ASS mRNA (Fig. 5B). ASL expression was not significantly different in these three cell culture models, so it was not evaluated further (data not shown). These results imply that SCLC1 will require an exogenous arginine supply for their growth while SR2 should survive better in arginine free media. To confirm the functional role of ASS, we compared the growth sensitivity of these three cell lines to arginine free media supplemented with citrulline, as an ASS substrate. SCLC1 could not withstand arginine deprivation due to lack of ASS expression and hence only 30% cells were viable after 48 hrs in arginine free media. In contrast, 80% of SR2 were still viable. Importantly, over-expression of TRX1 suppressed ASS in SR2 which in turn sensitized them to arginine deprivation (Fig. 5C).
Figure 5. CR cells preferentially used OXMET which partly reversed upon over-expression of TRX1.
(A&B) Relative mRNA levels of FH and ASS. Total RNAs extracted from these cells were reverse-transcribed and subsequently used as template for real-time quantitative PCR. GAPDH was used as internal control. The results shown in the graph were calculated with the ΔΔCt method by setting the FH or ASS mRNA level of SCLC1 as 1. Relative abundance of FH and ASS mRNA were higher in CR cells, but decreased with TRX1 over-expression. (C) Comparison of growth inhibition of SCLC1, SR2, SR2TRX+C1, and SR2TRX+C2 in arginine free media supplement with citrulline. At 72 hrs, only 30% of SCLC1 survive compared to 60–65% of TRX1 over-expressing clones, and 80% of SR2 were viable. (D) Immunoblot of key enzymes (ACL,ACC, FAS) in the FA synthesis pathway. CR cells had relative higher levels of expression in all of these proteins than parental cells. Over-expression TRX-1 protein can partly suppress these FA synthesis enzymes. Actin was used as a loading control. (E) Bar graph indicates the relative adjusted density of indicated protein expressions in each cell line. SR2TRX+C2 yielded similar results (data not shown)
FA synthesis also contributes to OXMET by providing carbon skeleton to β-oxidation in the mitochondria. Recently, it has been reported that over-expression of the Txnip protein, an inhibitor of TRX1, can lead to adipogenesis (12). Thus, it is likely that the relatively lower TRX1 expression found in SR2 may promote FA synthesis. To investigate this concept we assayed three important enzymes in FA synthesis: ATP-citrate lyase (ACL), acetyl-CoA carboxylase (ACC), and fatty acid synthase (FAS). All three protein levels were higher in SR2 cells than in SCLC1 (Fig. 5D); ACL (3 fold), ACC (15 fold), and FAS (5 fold) increased (Fig. 5E). TRX1 over-expression (SR2TRX+C1) significantly decreased the expression of FA synthesis proteins. These data support the notion that CR cells rely on OXMET more than their parental cells. Importantly, the over-expression of TRX1 protein opposes OXMET by down-regulating enzymes involved in the urea cycle and FA synthesis. Similarly, SR2 TRX+C2 also exhibited a decrease in ACC and FAS, when compared to SR2 (data not shown).
FA synthesis is important for survival of CR cells
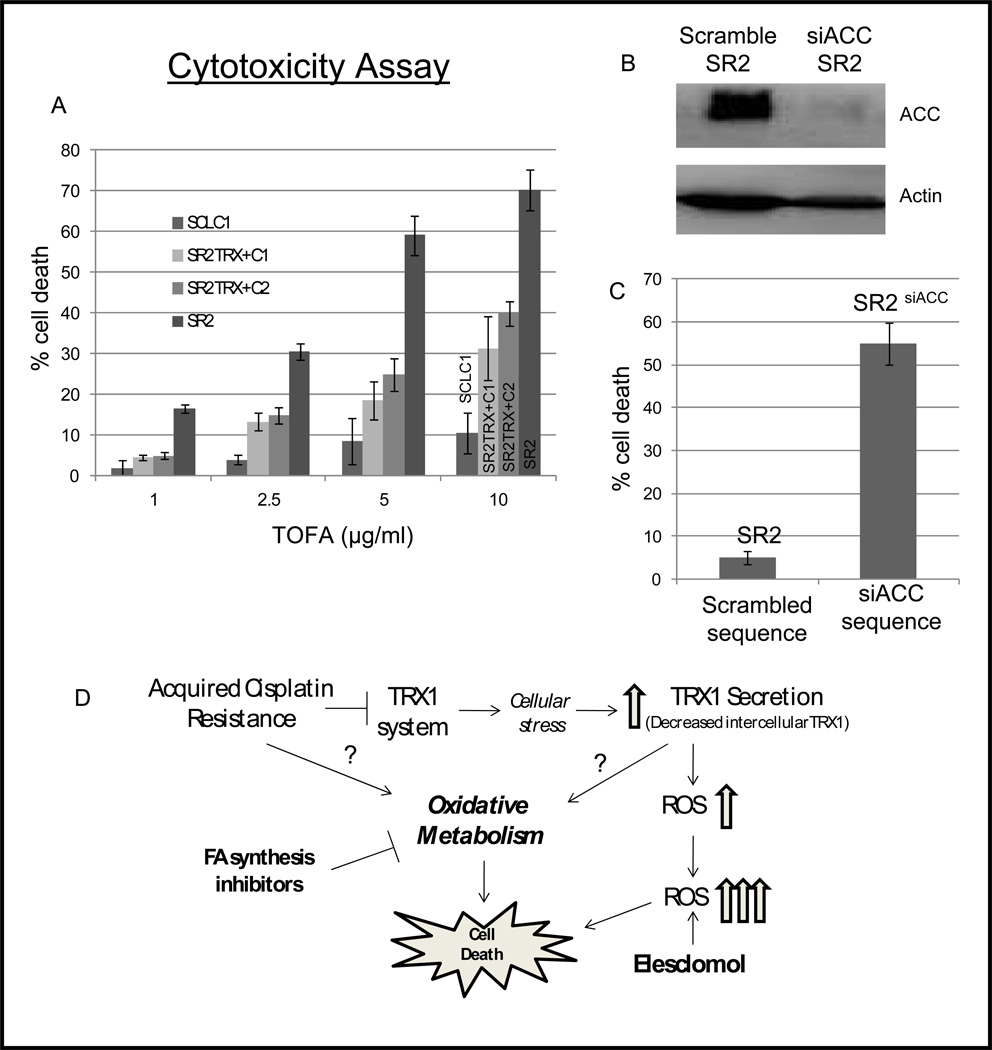
The above data strongly suggest that SR2 depends on FA synthesis pathway for survival since SR2 expressed 15 fold more ACC and 5 fold more FAS when compared to SCLC1. To further verify that FA synthesis plays a vital role on SR2 survival, first we treated SR2, SR2TRX+C1, SR2TRX+C2 and SCLC1 with TOFA (5-(Tetradecyloxy)-2-Furoic Acid), an allosteric inhibitor of ACC that blocks the synthesis of malonyl CoA (24), and then measured for cell death (Fig. 6A). TOFA induced cell death in a dose responsive manner in all cells with SR2 showing the highest sensitivity to TOFA, with 30% cell death at 2.5 ug/ml and 70% at 10ug/ml which was 6–7 fold higher compared to SCLC1. TRX1 over-expression rescued SR2TRX+C1 and SR2TRX+C2 cells from TOFA induced cell death at all doses with about only 30–40% cell death at the highest dose (10µg/ml), which were 2 fold less than SR2. Importantly, knock down of ACC by about 90% using siRNA (SR2siACC) (Fig. 6B) resulted in significant cell death (55%), while the control SR2 cells with scramble sequence can survive with only 6% cell death (Fig. 6C). Thus, our data strongly suggest that SR2 relies on FA synthesis for survival and decreased TRX1 impacts fatty acid metabolism dependency.
Figure 6. Downregulation of ACC enhances cell death in cisplatin resistant cells.
(A) Cells were treated by TOFA to inhibit ACC and cytotoxicity was assessed. Data are shown as % cell death as compared to untreated samples for each cell line. Over-expressing TRX1 protected CR cells against TOFA induced cell death. (B) Down regulation of ACC by siRNA (only 1nM is used to minimize the off-target effect) Immunoblot of ACC in SR2scramble, and SRsiACC cells showed more than 90% decrease in ACC 48hrs post-transfection. Actin was used as a loading control. (C) Comparison of cell death in SR2scramble and SRsiACC, ACC knocked down resulted in significant increase in cell death. (D) A proposed model: Acquired resistance to cisplatin disrupted the redox system through inhibition of TRX1 system (TrxR1/TRX1) causing TRX1 secretion. Decreased TRX1 resulted in accumulation of cellular ROS. Further increased ROS using elesclomol in these cisplatin resistant (CR) cells can push them beyond their tolerance limit which ultimately leads to cell death. Decreased TRX1 levels may involve metabolic reprogramming by switching CR cells from glycolysis toward OXMET. Inhibiting key metabolic enzymes in FA synthesis pathway led to significant cell death in CR cells.
Discussion
We previously reported that cisplatin resistant (CR) cells possess more basal ROS levels when compared to parental cell counterparts. Further increasing ROS using elesclomol in these CR cells can push them beyond their tolerance limit, which ultimately leads to cell death (2). Elesclomol is an investigational drug which chelates copper (Cu) outside the cells and enters as elesclomol-Cu(II), generating ROS via redox cycling of Cu(II) to Cu(I) inside mitochondria (25, 26). This drug has entered into both phase I and II clinical study, and thus far has been very well tolerated (27, 28).
Here we showed that lower TRX1 levels were found in all CR cells tested and this is primarily due to the increase in secretion. High TRX1 levels were found in the media as well as in the serum of the SR2 mouse xenograft model. TRX1, a 12kd protein, is a member of the leaderless protein family and can be effluxed from the cells through the leaderless pathway (6, 19). It has been shown that increased TRX1 secretion is found when cells are under stress (8, 29, 30). Increased plasma TRX1 levels are found in patients who received cisplatin treatment (31, 32). It is very likely that these CR cells excrete higher amounts of TRX1 due to continuous cellular stress, which in turn results in lower intracellular accumulation of TRX1 and consequently increased ROS.
In order to mimic low TRX1 levels observed in CR cells, we used siRNA to decrease TRX1 levels. SCLCsiTRX cells generated more ROS and became resistant to cisplatin, but hypersensitive to elesclomol. Likewise, over-expressing TRX1 in CR cells resulted in decreased ROS and increased sensitivity to cisplatin, but more resistant to elesclomol. Although the differential sensitivity to elesclomol in these transfectants could be explained on the basis of ROS which is influenced by TRX1 levels, it is unclear why TRX1 levels also influence cisplatin sensitivity. It is noteworthy that SR2 has other mechanisms of resistance, such as decreased cisplatin uptake due to decreased copper transport (hCTR1), a known transporter of cisplatin inside the cells and is influenced by the redox system (33–35). Whether TRX1 has any impact on hCTR1 is not known and is currently under investigation.
TRX1 has been reported to be involved in tumor angiogenesis. Welsh et al. have shown that over-expression of TRX1 increased HIF1α while transfection of non-functional TRX1 decreased HIF1α (36). Since HIF1α also regulates enzymes involved in the glycolytic pathway, it is possible that decreased TRX1 found in CR cells also has an impact on glucose metabolism. In fact, we found that basal HIF1α protein expression was lowered in all CR cells tested (Fig. S2). These CR cells possessed lower hexokinase-II level (37) and lower lactic acid production. Thus, it appears that decreased TRX1 levels have a negative impact on the glycolytic pathway. We have further shown that lower TRX1 found in CR cells had other consequences on the key enzyme (ASS) involved in the urea cycle. These CR cells possess significantly higher levels of ASS mRNA, while the parental cells (high TRX1 and HIF1α) expressed negligible amount of ASS mRNA. Interestingly, we have previously reported that HIF1α is a key transcription factor which negatively regulates ASS (38). Here, we showed that CR cells which possess lower TRX1 and decreased HIF1α levels, express increased levels of ASS. Consequently, these CR cells are able to synthesize arginine from citrulline and therefore are able to survive in arginine free media, while its parental cells could not. In addition, over-expression of TRX1 in CR cells resulted in lower ASS mRNA and less tolerance to arginine deprivation, which further confirms the role of TRX1 in ASS mRNA expression. This is an important finding which warrants further study since down regulation of ASS has been reported in multiple tumor types such as melanoma, hepatocellular carcinoma, and small cell lung cancer (39–42). These tumors are all highly sensitive to arginine deprivation. In fact, arginine deprivation therapy using pegylated arginine deiminase has been shown to have antitumor activity in tumors which do not express ASS (18, 41, 43, 44). Thus, alteration of ASS expression by TRX1 could have future clinical implication for treatment.
During the generation of arginine, fumarate is produced as a by-product. We have found that FH (fumarase) was higher in SR2 cells and attenuated with TRX1 over-expression. Interestingly, it has been reported that FH deficiency results in glycolytic addiction, HIF stabilization, and alteration in ROS (45) in renal cell carcinoma which is similar to our parental cells. It is also noteworthy that the majority of renal cells also do not express ASS (39). Increased ASS and FH expression in CR cells implies that more OXPHOS is taking place. This is also confirmed by increased oxygen consumption as well as higher mitochondrial activity found in CR cells. To further support that levels of TRX1 influenced CR cells to utilize more OXMET, we showed that CR cells up-regulate the key enzymes (ACL, ACC, and FAS) in the FA synthesis pathway, while over-expression of TRX1 resulted in attenuated levels of these enzymes. These data strongly suggest that TRX1 may play an important role in lipid metabolism. Interestingly, Chutkow et al. have recently reported that inhibition of TRX1 via Txnip can lead to increase in lipogenesis in fibroblast cells (12). Taken together, these findings further support the role of TRX1 in tumor metabolism. Knock down of ACC in SR2 resulted in significant cell death which indicates that FA synthesis is essential for CR cell survival. This is further confirmed by the finding of significant differential sensitivity to ACC inhibitor (TOFA) in SR2 cells and in SR2TRX+. Although the data presented here are limited to one pair of cisplatin resistant cell line with different clones of transfectants, our previous data has shown that 4 pairs and 2 additional primary cultures derive from patients who failed cisplatin have higher ROS and low TRX1 levels (2). These cell lines also showed alteration in their tumor metabolism either via TCA cycle or FA or by both. The underlying mechanisms are not yet clear and will be forthcoming. We have also established other CR cells from H69 (SCLC) obtained from ATCC and our initial finding suggested that the extent of decreased TRX1 as well as higher ROS accumulation correlates with the resistant levels. Overall, these findings could have future implication for drug development to selectively kill CR cells which possess high ROS and low TRX1 level.
Supplementary Material
Acknowledgements
We particularly thank Dr. Masazumi Nagai, Dr. Yumiko Wada, and Dr. Jim Sang from Synta Pharmaceuticals for Elesclomol, reagents, and SCID mouse xenograft model. The authors thank Dr. Theodore Lampidis for helpful discussion.
Grant support:
This work was support in part by J&E King Biomedical Research, FL Dept. of Health (1KD08) to M.W. VA Merit Review and NIH/NCI (1R01 CA109578-01) to G.T., C.W., M.Y, L.G.F, and N.S. NIH/NCI (1R01CA149260) to M.T.K.
References
- 1.Siddik ZH. Cisplatin: mode of cytotoxic action and molecular basis of resistance. Oncogene. 2003;22:7265–7279. doi: 10.1038/sj.onc.1206933. [DOI] [PubMed] [Google Scholar]
- 2.Wangpaichitr M, Wu C, You M, Maher JC, Dinh V, Feun LG, et al. N1,N3-Dimethyl-N1,N3-bis(phenylcarbonothioyl) Propanedihydrazide (Elesclomol) Selectively Kills Cisplatin Resistant Lung Cancer Cells through Reactive Oxygen Species (ROS) Cancers. 2009;1:23–28. doi: 10.3390/cancers1010023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Saitoh M, Nishitoh H, Fujii M, Takeda K, Tobiume K, Sawada Y, et al. Mammalian thioredoxin is a direct inhibitor of apoptosis signal-regulating kinase (ASK) 1. Embo J. 1998;17:2596–2606. doi: 10.1093/emboj/17.9.2596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Sun Y, Rigas B. The thioredoxin system mediates redox-induced cell death in human colon cancer cells: implications for the mechanism of action of anticancer agents. Cancer Res. 2008;68:8269–8277. doi: 10.1158/0008-5472.CAN-08-2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Witte AB, Anestal K, Jerremalm E, Ehrsson H, Arner ES. Inhibition of thioredoxin reductase but not of glutathione reductase by the major classes of alkylating and platinum-containing anticancer compounds. Free Radic Biol Med. 2005;39:696–703. doi: 10.1016/j.freeradbiomed.2005.04.025. [DOI] [PubMed] [Google Scholar]
- 6.Rubartelli A, Bajetto A, Allavena G, Wollman E, Sitia R. Secretion of thioredoxin by normal and neoplastic cells through a leaderless secretory pathway. J Biol Chem. 1992;267:24161–24164. [PubMed] [Google Scholar]
- 7.Rubartelli A, Bonifaci N, Sitia R. High rates of thioredoxin secretion correlate with growth arrest in hepatoma cells. Cancer Res. 1995;55:675–680. [PubMed] [Google Scholar]
- 8.Miyamoto S, Kawano H, Sakamoto T, Soejima H, Kajiwara I, Hokamaki J, et al. Increased plasma levels of thioredoxin in patients with coronary spastic angina. Antioxid Redox Signal. 2004;6:75–80. doi: 10.1089/152308604771978363. [DOI] [PubMed] [Google Scholar]
- 9.Powis G, Kirkpatrick DL. Thioredoxin signaling as a target for cancer therapy. Current opinion in pharmacology. 2007;7:392–397. doi: 10.1016/j.coph.2007.04.003. [DOI] [PubMed] [Google Scholar]
- 10.Wei SJ, Botero A, Hirota K, Bradbury CM, Markovina S, Laszlo A, et al. Thioredoxin nuclear translocation and interaction with redox factor-1 activates the activator protein-1 transcription factor in response to ionizing radiation. Cancer Res. 2000;60:6688–6695. [PubMed] [Google Scholar]
- 11.Ando K, Hirao S, Kabe Y, Ogura Y, Sato I, Yamaguchi Y, et al. A new APE1/Ref-1-dependent pathway leading to reduction of NF-kappaB and AP-1, and activation of their DNA-binding activity. Nucleic acids research. 2008;36:4327–4336. doi: 10.1093/nar/gkn416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Chutkow WA, Lee RT. Thioredoxin regulates adipogenesis through thioredoxin interacting protein (Txnip) protein stability. J Biol Chem. 2011 doi: 10.1074/jbc.M111.267666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wangpaichitr M, Wu C, You M, Kuo MT, Feun L, Lampidis T, et al. Inhibition of mTOR restores cisplatin sensitivity through down-regulation of growth and anti-apoptotic proteins. Eur J Pharmacol. 2008;591:124–127. doi: 10.1016/j.ejphar.2008.06.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wu C, Wangpaichitr M, Feun L, Kuo MT, Robles C, Lampidis T, et al. Overcoming cisplatin resistance by mTOR inhibitor in lung cancer. Mol Cancer. 2005;4:25. doi: 10.1186/1476-4598-4-25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Savaraj N, Wu C, Wangpaichitr M, Kuo MT, Lampidis T, Robles C, et al. Overexpression of mutated MRP4 in cisplatin resistant small cell lung cancer cell line: collateral sensitivity to azidothymidine. Int J Oncol. 2003;23:173–179. [PubMed] [Google Scholar]
- 16.Xu J, Tyan T, Cedrone E, Savaraj N, Wang N. Detection of 11q13 amplification as the origin of a homogeneously staining region in small cell lung cancer by chromosome microdissection. Genes Chromosomes Cancer. 1996;17:172–178. doi: 10.1002/(SICI)1098-2264(199611)17:3<172::AID-GCC5>3.0.CO;2-1. [DOI] [PubMed] [Google Scholar]
- 17.Wangpaichitr M, Savaraj N, Maher J, Kurtoglu M, Lampidis TJ. Intrinsically lower AKT, mammalian target of rapamycin, and hypoxia-inducible factor activity correlates with increased sensitivity to 2-deoxy-D-glucose under hypoxia in lung cancer cell lines. Mol Cancer Ther. 2008;7:1506–1513. doi: 10.1158/1535-7163.MCT-07-2334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Feun L, You M, Wu CJ, Kuo MT, Wangpaichitr M, Spector S, et al. Arginine deprivation as a targeted therapy for cancer. Curr Pharm Des. 2008;14:1049–1057. doi: 10.2174/138161208784246199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Rubartelli A, Bajetto A, Bonifaci N, Di Blas E, Solito E, Sitia R. A novel way to get out of the cell. Cytotechnology. 1993;11 Suppl 1:S37–S40. doi: 10.1007/BF00746049. [DOI] [PubMed] [Google Scholar]
- 20.Huttemann M, Lee I, Pecinova A, Pecina P, Przyklenk K, Doan JW. Regulation of oxidative phosphorylation, the mitochondrial membrane potential, and their role in human disease. Journal of bioenergetics and biomembranes. 2008;40:445–456. doi: 10.1007/s10863-008-9169-3. [DOI] [PubMed] [Google Scholar]
- 21.McGilvery RW. Biochemistry a functional approach. WB Saunders Company. 1970;Chapter 11 [Google Scholar]
- 22.Srere PA. An eclectic view of metabolic regulation: control of citrate synthase activity. Adv Enzyme Regul. 1970;9:221–233. doi: 10.1016/s0065-2571(71)80046-8. [DOI] [PubMed] [Google Scholar]
- 23.Ratner S, Anslow WP, Jr, Petrack B. Biosynthesis of urea. VI. Enzymatic cleavage of argininosuccinic acid to arginine and fumaric acid. J Biol Chem. 1953;204:115–125. [PubMed] [Google Scholar]
- 24.Halvorson DL, McCune SA. Inhibition of fatty acid synthesis in isolated adipocytes by 5-(tetradecyloxy)-2-furoic acid. Lipids. 1984;19:851–856. doi: 10.1007/BF02534514. [DOI] [PubMed] [Google Scholar]
- 25.Nagai M, Vho N, Cu J, Wada Y. Elesclomol-Cu chelate selectively targets mitochondria to induce oxidative stress; Proceeding AACR 102nd Annual Meeting; 2011. Abstract No. 2093. [Google Scholar]
- 26.Toogood PL. Mitochondrial drugs. Current opinion in chemical biology. 2008;12:457–463. doi: 10.1016/j.cbpa.2008.06.002. [DOI] [PubMed] [Google Scholar]
- 27.Berkenblit A, Eder JP, Jr, Ryan DP, Seiden MV, Tatsuta N, Sherman ML, et al. Phase I clinical trial of STA-4783 in combination with paclitaxel in patients with refractory solid tumors. Clin Cancer Res. 2007;13:584–590. doi: 10.1158/1078-0432.CCR-06-0964. [DOI] [PubMed] [Google Scholar]
- 28.O'Day S, Gonzalez R, Lawson D, Weber R, Hutchins L, Anderson C, et al. Phase II, randomized, controlled, double-blinded trial of weekly elesclomol plus paclitaxel versus paclitaxel alone for stage IV metastatic melanoma. J Clin Oncol. 2009;27:5452–5458. doi: 10.1200/JCO.2008.17.1579. [DOI] [PubMed] [Google Scholar]
- 29.Nakamura H, De Rosa S, Roederer M, Anderson MT, Dubs JG, Yodoi J, et al. Elevation of plasma thioredoxin levels in HIV-infected individuals. International immunology. 1996;8:603–611. doi: 10.1093/intimm/8.4.603. [DOI] [PubMed] [Google Scholar]
- 30.Nakamura H, Vaage J, Valen G, Padilla CA, Bjornstedt M, Holmgren A. Measurements of plasma glutaredoxin and thioredoxin in healthy volunteers and during open-heart surgery. Free radical biology & medicine. 1998;24:1176–1186. doi: 10.1016/s0891-5849(97)00429-2. [DOI] [PubMed] [Google Scholar]
- 31.Sasada T, Nakamura H, Ueda S, Iwata S, Ueno M, Takabayashi A, et al. Secretion of thioredoxin enhances cellular resistance to cis-diamminedichloroplatinum (II) Antioxid Redox Signal. 2000;2:695–705. doi: 10.1089/ars.2000.2.4-695. [DOI] [PubMed] [Google Scholar]
- 32.Sasada T, Nakamura H, Ueda S, Sato N, Kitaoka Y, Gon Y, et al. Possible involvement of thioredoxin reductase as well as thioredoxin in cellular sensitivity to cis-diamminedichloroplatinum (II) Free Radic Biol Med. 1999;27:504–514. doi: 10.1016/s0891-5849(99)00101-x. [DOI] [PubMed] [Google Scholar]
- 33.Chen HH, Song IS, Hossain A, Choi MK, Yamane Y, Liang ZD, et al. Elevated glutathione levels confer cellular sensitization to cisplatin toxicity by up-regulation of copper transporter hCtr1. Mol Pharmacol. 2008;74:697–704. doi: 10.1124/mol.108.047969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kuo MT, Chen HH, Song IS, Savaraj N, Ishikawa T. The roles of copper transporters in cisplatin resistance. Cancer Metastasis Rev. 2007;26:71–83. doi: 10.1007/s10555-007-9045-3. [DOI] [PubMed] [Google Scholar]
- 35.Song IS, Savaraj N, Siddik ZH, Liu P, Wei Y, Wu CJ, et al. Role of human copper transporter Ctr1 in the transport of platinum-based antitumor agents in cisplatin-sensitive and cisplatin-resistant cells. Mol Cancer Ther. 2004;3:1543–1549. [PubMed] [Google Scholar]
- 36.Welsh SJ, Bellamy WT, Briehl MM, Powis G. The redox protein thioredoxin-1 (Trx-1) increases hypoxia-inducible factor 1alpha protein expression: Trx-1 overexpression results in increased vascular endothelial growth factor production and enhanced tumor angiogenesis. Cancer Res. 2002;62:5089–5095. [PubMed] [Google Scholar]
- 37.Sullivan EJ, Kurtoglu M, Wangpaichitr M, Savaraj N, Lampidis T. Metabolic changes associated with acquired cisplatin resistance; Proceeding AACR 102nd Annual Meeting; 2011. Abstract no 4087. [Google Scholar]
- 38.Tsai WB, Aiba I, Lee SY, Feun L, Savaraj N, Kuo MT. Resistance to arginine deiminase treatment in melanoma cells is associated with induced argininosuccinate synthetase expression involving c-Myc/HIF-1alpha/Sp4. Mol Cancer Ther. 2009;8:3223–3233. doi: 10.1158/1535-7163.MCT-09-0794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Yoon CY, Shim YJ, Kim EH, Lee JH, Won NH, Kim JH, et al. Renal cell carcinoma does not express argininosuccinate synthetase and is highly sensitive to arginine deprivation via arginine deiminase. International journal of cancer. 2007;120:897–905. doi: 10.1002/ijc.22322. [DOI] [PubMed] [Google Scholar]
- 40.Szlosarek PW, Klabatsa A, Pallaska A, Sheaff M, Smith P, Crook T, et al. In vivo loss of expression of argininosuccinate synthetase in malignant pleural mesothelioma is a biomarker for susceptibility to arginine depletion. Clin Cancer Res. 2006;12:7126–7131. doi: 10.1158/1078-0432.CCR-06-1101. [DOI] [PubMed] [Google Scholar]
- 41.Ensor CM, Holtsberg FW, Bomalaski JS, Clark MA. Pegylated arginine deiminase (ADI-SS PEG20,000 mw) inhibits human melanomas and hepatocellular carcinomas in vitro and in vivo. Cancer Res. 2002;62:5443–5450. [PubMed] [Google Scholar]
- 42.Prieto V, Curley S, Ensor C, Holzberg F, Bomalaski J, Clak M. Incidence and distribution of argininosuccinate synthetase deficiency in human cancers: A method for identifying cancers sensitive to arginine deprivation by arginine deiminase. Proc Am Soc Clin Oncol. 2003;22 Abstract No. 968. [Google Scholar]
- 43.Savaraj N, You M, Wu C, Wangpaichitr M, Kuo MT, Feun LG. Arginine deprivation, autophagy, apoptosis (AAA) for the treatment of melanoma. Current molecular medicine. 2010;10:405–412. doi: 10.2174/156652410791316995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Ascierto PA, Scala S, Castello G, Daponte A, Simeone E, Ottaiano A, et al. Pegylated arginine deiminase treatment of patients with metastatic melanoma: results from phase I and II studies. J Clin Oncol. 2005;23:7660–7668. doi: 10.1200/JCO.2005.02.0933. [DOI] [PubMed] [Google Scholar]
- 45.Sudarshan S, Sourbier C, Kong HS, Block K, Valera Romero VA, Yang Y, et al. Fumarate hydratase deficiency in renal cancer induces glycolytic addiction and hypoxia-inducible transcription factor 1alpha stabilization by glucose-dependent generation of reactive oxygen species. Mol Cell Biol. 2009;29:4080–4090. doi: 10.1128/MCB.00483-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.