Abstract
Objectives:
Punica granatum L., (Family: Punicaceae) is used in Indian Unani medicine for treatment of diabetes mellitus. Therefore, the present study was done to evaluate the antidiabetic and antihyperlipidemic effects of ethanolic extract of leaves of P. granatum in alloxan-induced diabetic rats.
Materials and Methods:
Healthy Wistar albino rats (100-150 g) were divided into four groups of six animals each. Groups A and B received normal saline [(10 ml/kg/day/per oral (p.o.)]; group C received ethanolic extract of leaves of P. granatum (500 mg/kg/p.o.); and group D received glibenclamide (0.5 mg/kg/day/p.o.). The extracts were given for 1 week in all groups. To induce diabetes, alloxan 150 mg/kg, intraperitoneal (i.p.) single dose was administered to groups B, C, and D. Blood glucose and serum lipids [Total Cholesterol (TC), Triglycerides (TG), Low Density Lipoproteins (LDL), and High Density Lipoproteins (HDL)] and the atherogenic index were estimated after one week. For mechanism of antidiabetic action glycogen estimation on the liver, cardiac and skeletal muscle, and intestinal glucose absorption was done.
Results:
Group B showed a significant (P<0.01) increase in blood glucose as compared to group A. Groups C and D showed significant decrease (P<0.01) in blood glucose level in comparison to group B. The test drug showed a significant (P<0.01) increase in glycogen content in the liver, cardiac, and skeletal muscle; it significantly (P<0.01) reduced intestinal glucose absorption. Groups C and D showed significant (P<0.01) decrease in serum TC, TG, LDL, and AI as compared to Group B, which showed a significant (P<0.01) increase. Groups C and D showed significant (P<0.01) increase in serum HDL as compared to Group B, which showed a significant (P<0.01) decrease in all values.
Conclusion:
P. granatum leaves possess significant antidiabetic and antihyperlipidemic activity.
KEY WORDS: Antioxidant, atherogenic index, blood glucose, flavonoids, glycogen
Introduction
Non-Insulin Dependent Diabetes Mellitus (NIDDM) is a multifactorial disease characterised by hyperglycemia and lipoprotein abnormalities. These traits are hypoyhetized to damage cell membranes, which results in excessive generation of reactive oxygen species. NIDDM has also been associated with an increased risk for developing premature atherosclerosis due to an increase in Triglyceride (TG) and Low Density Lipoproteins (LDL) and decrease in High Density Lipoprotein (HDL) levels.[1]
The pomegranate, Punica granatum (L.), an ancient, mystical, and highly distinctive fruit, belongs to Punicaceae family. The pomegranate tree typically grows 12-16 feet tall. The leaves are glossy and lance-shaped. The pomegranate is a native of the Himalayas in northern India to Iran, but it has been cultivated and naturalized since ancient times over the entire Mediterranean region. It is also found in India and more arid regions of Southeast Asia, the East Indies, and tropical Africa.[2] Pomegranate juice (fruit) prevents age-related vascular complications like atherosclerosis by decreasing lipid peroxidation and increasing antioxidant enzymes. Pomegranate fruits are widely consumed fresh and in beverage forms as juice and wine. Commercial Pomegranate juice contains high amount of polyphenols such as ellagic acid, gallotannins, anthocyanins, and other flavonoids (quercetin, kaempferol, and luteolin glycosides).[3] Pomegranate also serves as a remedy for diabetes in the Unani system of medicine practiced in the Middle East and India.[2] Chemical constituents of the leaf extract of P. granatum are almost similar to those of the fruit or seed, eg, ellagic acid,[4] tannins (punicalin and punicafolin), and flavone glycosides, including luteolin and apigenin.[2] In our previous study, the antidiabetic and antihyperlipidemic activities of the fruit of P. granatum have been observed. But, thus far, no scientific reports are available regarding the antidiabetic and antihyperlipidemic activities of the leaves of this plant. Therefore, the present study was carried out to evaluate the antidiabetic and antihyperlipidemic effects of ethanolic extract of leaves of P. granatum in alloxan-induced diabetic rats.
Materials and Methods
Drugs Used
Leaves of P. granatum (PG) were collected in the month of April-May from Assam Medical College and Hospital (AMCH) campus, Dibrugarh, and authenticated by Dr. M Islam, Professor, Department of Life Science, Dibrugarh University. A voucher specimen (No. DU/LS/211) was deposited at Dibrugarh University. Alloxan Monohydrate was obtained from Sigma Aldrich, Bangalore. Crude powder of glibenclamide and metformin was obtained from Aventis Pharma Limited, Goa. Qualigens-Diagnostics Cholesterol, Triglyceride, and HDL-cholesterol kits were obtained from Sigma Diagnostics (India) Pvt Ltd, Vadodara, Gujarat.
Preparation of Plant Extract
The leaves were washed thoroughly with distilled water, air-dried, powdered with an electrical grinder, and soaked in 90% ethanol for 6 hours in a tightly covered container. The extract was transferred to a percolator with 90% ethanol and percolation was allowed to proceed slowly till the drug was completely exhausted. Ethanol was evaporated to a soft extract at a temperature not exceeding 60°C and transferred to a vacuum desicator.[5] A net yield of 30.6 g was obtained by percolating 250 g of dry leaf powder (12.24%). The extract collected was stored in air-tight glass containers in a refrigerator at 2-8°C for use in the experiments.
Animals
Healthy albino rats of Wistar strain weighing 100-150 g (weight of rats somewhat lower than standard, but the rats were otherwise healthy, 6-8 weeks old, took normal average daily diet, displayed normal daily activity and behavior). Rats of either sex were used (because there may be some sex-related variations in the pharmacokinetics and pharmacodynamics of a drug). Thus, to identify the type of variations, we selected the animals of both sexes for the experiment. They were obtained from the Central Animal House, AMCH, Dibrugarh. The animals were housed in standard polypropylene cages and maintained under controlled room temperature (25 ± 5°C) and humidity (55 ± 5%) with 12:12 hour light and dark cycle. The rats were fed commercially available rat normal pellet diet and water ad libitum. Permission from the Institutional Animal Ethics Committee (Regd. No.634/02/a/CPCSEA) was obtained prior to commencing the study.
Acute Oral Toxicity Studies
Albino rats of either sex were used for acute oral toxicity test according to the Organization for Economic Cooperation and Development (OECD) guidelines 425 (because no previous toxicity study was done for pomegranate leaves, so according to the standard protocol, we did the acute toxicity test to select the dose of ethanolic extract of P. granatum leaves). One-fourth of LD50 (>2000) was taken for the study. A total of five animals were used, which received a single oral dose (2000 mg/kg body weight) of ethanolic extract of leaves of P. granatum (EELPG) after overnight fasting. After administration of EELPG, food was withheld for further 3-4 h. Animals were observed individually at least once during the first 30 min after dosing, periodically during the first 24 h (with special attention during the first 4 h), and daily thereafter for 14 days. At the end of the study, the animals were observed for general toxic signs, morphological behavior, and mortality. One-fourth of LD50 (>2000) was taken for the study.[6]
Experimental Design for Anti-Diabetic Study[7]
A total of 30 animals were equally divided into five groups with six animals in each group:
The above drugs were administered orally once daily for one week.
Induction of diabetes
Leaving aside six rats for normal control group, diabetes was induced in 24 rats by a single intraperitoneal injection of alloxan monohydrate in the dose of 150 mg/kg body weight. The fasting blood glucose was determined after 72 hours.[8] Eighteen rats showing a blood glucose level of >200 mg/100 ml were taken for the study.
Blood samples were collected from the orbital sinus of rats on the eighth day. For seperation of serum, blood was collected in a separate plain vial. The serum thus obtained was used for biochemical estimations.
Method of blood glucose estimation
Blood glucose estimation was done by Glucose Oxidase Method[9] using glucose kit manufactured by Sigma Diagnostic (India) Pvt Ltd, Baroda, Gujarat.
Biochemical estimations
Total serum cholesterol was estimated by using Qualigens-Diagnostics Cholesterol Kit.[10] Triglycerides were measured by enzyme colorimetric method using Qualigens-Diagnostics Triglyceride Reagent GPO.[11] HDL–cholesterol was assayed by using Qualigens-Diagnostics HDL-cholesterol Kit.[12] LDL–cholesterol was measured by using the formula of Friedewald et al.[13] The atherogenic index was calculated using the formula of Schulpis et al.[14]
(1) Glycogen estimation of liver, skeletal muscle and cardiac muscle[15]
The rats were divided into four groups with six animals in each as before.

After two hours of administration of the above-mentioned drugs, the animals were sacrificed by decapitation. The liver, leg muscle, and heart tissues were taken out carefully and their glycogen content was estimated by the method of Carroll et al.[15]
Tissues were then placed in an efficient blender under an appropriate volume of Trichloroacetic Acid (TCA) and homogenized for 3 minutes. The homogenate was poured into a suitable centrifuge tube and centrifuged for 15 minutes. The supernatant fluid was decanted upon acid-washed filter paper placed in a funnel draining into a graduated cylinder. The residue was transferred quantitatively to the blender with an appropriate volume of TCA and homogenized again for 1 minute. The mixture was centrifuged again and the supernatant fluid was poured through the same filter paper. The desired volume was made up with 5% TCA and mixed thoroughly. One millilitre of the TCA filtrate was pipetted into a 15-ml Pyrex centrifuge tube. To obtain the most reliable results, duplicate samples of each unknown were analyzed. Five volumes of 95% ethanol were added to each tube with careful blowing to ensure a thorough mixing. These tube were capped with clean rubber stoppers and allowed to stand overnight at room temperature. After precipitation was complete, the tubes were centrifuged at 3,000 rpm for 15 minutes. The clear liquid was gently decanted from the packed glycogen and the tubes were allowed to drain in an inverted position for 10 minutes. The glycogen was dissolved in 2 ml of distilled water, taking care to wash the side of test tube. A reagent blank was prepared by pipetting 2 ml of distilled water in a clean centrifuge tube. A standard was prepared by pipetting 2 ml of standard glucose solution containing 0.1 mg of glucose into a similar tube.
At this point, 10 ml of anthrone reagent was delivered into each tube with vigorous blowing consistently. This stream of anthrone reagent was directed into the center of the tube to secure good mixing. After the tubes received the anthrone reagent, they were tightly capped with air condensers and placed in cold tap water bath.
After all tubes had reached the temperature of cold water, they were put into a boiling water bath for 15 minutes. The tubes were placed in such a way that the level of boiling water was little above the level of liquid in the tube. The tubes were then removed to a cold water bath and cooled to room temperature. The tubes and stoppers were cleaned dry and the content of each was transferred to a colorimeter tube and read at 620 micrometer after adjusting the colorimeter with the reagent blank. Care was taken to avoid introduction of lint or contaminating carbohydrate into anthrone reagent.
(2) Evaluation of effect of EELPG on intestinal glucose absorption by estimation of glucose uptake[16]
The efect of EELPG was studied by the method described by Das (2001)[16] with some modification. Das had made five to six intestinal loops of roughly equal size lying between the proximal jejunum and distal iluem. In the present study, an intestinal loop of 8 cm from the pyloric end was made.
Eighteen albino rats were divided into three groups, with six animals in each group:

The treatments were given for 7 days, following which the animals were kept fasting for 18 hours. Subsequently, all animals were anaesthetized with thiopentone sodium 40 mg/kg i.p. and the abdomen was opened through midline incision and an intestinal loop of 8 cm from the pyloric end was made. D-glucose 2.5 mg (1 ml of 250 mg% in normal saline at 37°C) was given in the loop by a tuberculin syringe. The animals were sacrificed after 15 minutes and the intact loop was excised and weighed before and after draining the contents of loop to assess the final volume. After constant dilution, resultant fluid was estimated for glucose content by glucose oxidase method.[9] The absorption was expressed in terms of mg/g dry weight/hour. Dry weight of the intestinal segment was measured after dehydrating the loop in ethyl alcohol for 24 hours, and then drying in hot air oven at 110-120°C for 2 hours.
Statistical Analysis
The statistical significance between the groups was analyzed separately using One–way Analysis Of Variance (ANOVA), followed by Dunnett's multiple comparison test. The significance was expressed by P values, as mentioned in the tables. P<0.05 was considered as significant.
Results
Acute Toxicity Test
No mortality was recorded among the rats at the dose of 2000 mg/kg. Hence, one-fourth of the dose tested, ie, 500 mg/kg body weight was selected for the study.
Effect on Fasting Blood Sugar Level in Diabetic Rats
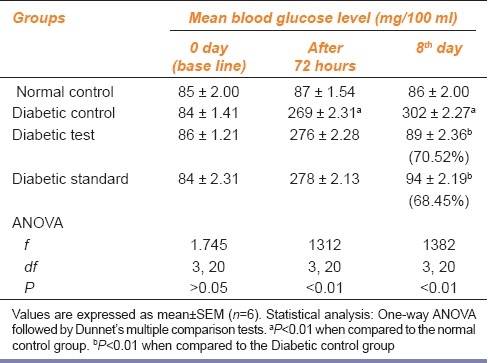
On repeated administration of the extract for one week, a significant (P<0.01) decrease in blood sugar was found in Groups C and D as compared to Group B, which showed a significant (P<0.01) rise in blood sugar as compared to that in Group A [Table 1]. The percentage of reduction in blood glucose in groups C and D on the eighth day was 70.52% and 68.45%, respectively.
Table 1.
Effect of ethanolic extract of leaves of Punica granatum on fasting blood glucose level of alloxan-induced diabetic rats
Effect on Glycogen Estimation
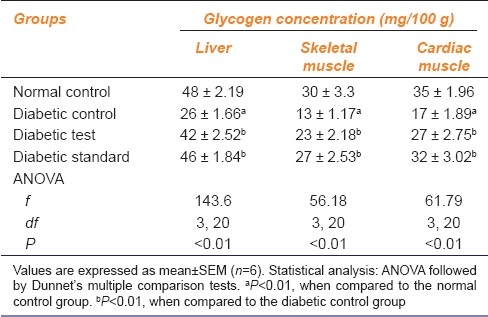
A significant (P<0.01) increase was noted in the glycogen content of liver, skeletal muscle, and cardiac muscle in Groups C and D as compared to Group B, which showed a significant (P<0.01) reduction in glycogen content in the above tissues as compared to Group A [Table 2]. The increase in glycogen content was more in group D than in group C.
Table 2.
Effect of ethanolic extract of leaves of Punica granatum on glycogen concentration in liver, skeletal muscle, and cardiac muscle
Effect on Intestinal Glucose Absorption
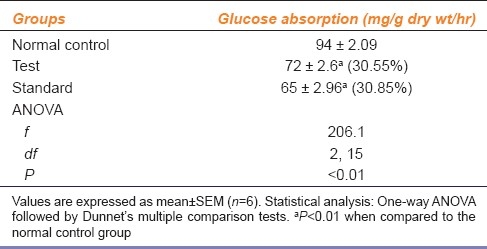
The test and standard group showed significant (P<0.01) reduction in intestinal glucose absorption as compared to normal control group [Table 3]. The reduction in absorption was more in the standard group than in the test group.
Table 3.
Effect of ethanolic extract of leaves of Punica granatum on intestinal glucose absorption
Effect of EELPG on Serum Lipids and Atherogenesis in Alloxan-induced Diabetic Rats
Table 4 shows mean serum levels (in mg/100 ml) of lipid parameters, ie, serum total cholesterol, serum triglyceride, serum HDL cholesterol, serum LDL, and also the atherogenic index, in different groups at the end of 7 days of drug administration.
Table 4.
Effect of ethanolic extract of leaves of Punica granatum on serum lipids on the eighth day of the experiment
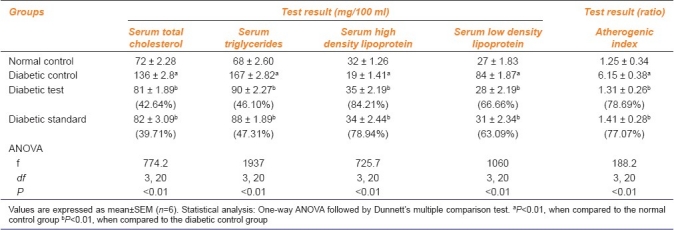
A significant (P<0.01) decrease was observed in serum cholesterol in Groups C and D as compared to Group B, which showed a significant (P<0.01) increase in serum cholesterol as compared to Group A. The percentage of reduction in serum cholesterol in diabetic test and diabetic standard was 42.64% and 39.71%, respectively, as compared to that in the diabetic control group.
There was a significant (P<0.01) decrease in serum triglyceride in Groups C and D as compared to Group B, which showed a significant (P<0.01) increase as compared to Group A. The percentage of reduction of serum triglyceride in diabetic test and diabetic standard was 46.10% and 47.31%, respectively. The percentage of reduction of serum triglyceride in diabetic test and diabetic standard was 65.44% and 72.81%, respectively.
There was a significant (P<0.01) increase in serum HDL cholesterol in Groups C and D as compared to Group B, which showed a significant (P<0.01) decrease as compared to Group A [Table 4]. The percentage of increase of serum HDL cholesterol in the diabetic test and diabetic standard was 84.21% and 78.94%, respectively.
A significant (P<0.01) decrease was noted in serum LDL cholesterol in Groups C and D as compared to Group B, which showed a significant (P<0.01) increase as compared to Group A [Table 4]. The percentage of reduction in the serum LDL cholesterol in diabetic test and diabetic standard was 66.66% and 63.09%, respectively.
Discussion
Alloxan, a cytotoxic agent induces chemical diabetes (alloxan diabetes) in various animal species through destruction of islets of Langerhans of the pancreas. After administration, it is rapidly and selectively taken up by the β-cells of the pancreas, following which there is formation of redox cycle for generation of Reactive Oxygen Species (ROS), superoxide radicals, and hydrogen peroxide.[17]
In type 2 diabete melitus patients, insulin deficiency causes an impairment in glycogen synthase activation in the skeletal muscle due to the inability of insulin to phosphorylate Insulin Receptor Substrate-1 (IRS-1); this causes a decreased activation of the enzyme Phosphatidylinositol 3-Kinase (PI-3K), leading to decreased expression and translocation of GLUT-4 glucose transporters.[18] EELPG, due to its insulin-like action of its phytochemicals probably increased PI-3K activation, thus leading to stimulation of muscle glycogen synthase. The increase in glycogen concentration in the skeletal and cardiac muscle might also be due to increased expression and translocation of GLUT-4 glucose transporters as a result of increased activation of PI-3K, thus leading to increased peripheral glucose uptake. The increase in liver glycogen content due to increase in synthesis of glycogen synthase in alloxan-induced diabetic rats has been reported.[19] Therefore, the increase in liver glycogen content caused by EELPG may also be due to increased synthesis of liver glycogen synthase enzyme.
Tannic acid is a potent inhibitor of glucose uptake, which produced a marked loss in glucose transport capacity in isolated rat intestinal brush border membrane vesicles by dissipation of the Na+ electrochemical gradient that provides the driving force for active glucose accumulation.[20] The reduced absorption of glucose as shown by EELPG might be due to the presence of tannins, which may have acted by a similar mechanism.
Plants that contain the active principles such as glycosides and flavonoids have antioxidant activity and are claimed to possess antidiabetic effects.[21] The antidiabetic activity of EELPG may be due to the presence of phytochemicals (flavonoids, tannins, glycosides) in it, which are strong antioxidants.[2] Moreover, flavonoids are known to regenerate the damaged β-cells in alloxan-induced diabetic rats.[22]
Alloxan significantly increased serum total cholesterol, tryglycerides, LDL levels, and atherogenic index in alloxan-induced diabetic rats. Administration of P. granatum leaf extract to diabetic rats reversed all the above-mentioned parameters and improved the HDL levels. High LDL levels are usually associated with atherosclerosis. High HDL levels reduce this risk.[23] A possible mechanism of EELPG may be due to the presence of flavonoids, which significantly increased LDL receptor mRNA levels, which, in turn, increase hepatic uptake and degradation of LDL causing a decrease in serum LDL levels.[24] A decrease in atherogenic index is believed to be beneficial, since the HDL level is inversely correlated with coronary heart disease and its elevation is considered as an antiatherosclerotic factor.[25] Thus, its antihyperlipidemic effect could play a protective role against the development of atherosclerosis and cardiovascular complications in diabetes mellitus.
Conclusion
It is concluded that the ethanolic extract of leaves of P. granatum at the dose of 500 mg/kg body weight produced significant antidiabetic activity in alloxan-induced NIDDM albino rats. It is also found to be highly effective in managing the complications associated with diabates mellitus, such as hyperlipidaemia, and prevents the defects in lipid metabolism. Therefore, P. granatum leaves show therapeutic promise as a protective agent against the development and progression of atherosclerosis and possible related cardiovascular complications in diabetes mellitus. However, further studies have to be undertaken to work out the exact mechanism by which EELPG could be exerting its effects.
Footnotes
Source of Support: Nil.
Conflict of Interest: None declared.
References
- 1.Rajagopal K, Sasikala K. Antihyperglycaemic and antihyperlipidaemic effects of Nymphaea stellata in alloxan-induced diabetic rats. Singapore Med J. 2008;49:137–41. [PubMed] [Google Scholar]
- 2.Jurenka JS. Therapeutic applications of pomegranate (Punica granatum L.): A review. Altern Med Rev. 2008;13:128–44. [PubMed] [Google Scholar]
- 3.Nishigaki I, Rajendran P, Venugopal R, Ekambaram G, Sakthisekaran D, NishigaKi Y. Effect of extract of Pomegranate (Punica granatum L.) on Glycated Protein-iron Chelate-induced Toxicity: An In Vitro Study on Human Umbilical-vein Endothelial Cells. J Health Sci. 2008;54:441–9. [Google Scholar]
- 4.Lansky EP, Newman RA. Punica granatum (pomegranate) and its potential for prevention and treatment of inflammation and cancer. J Ethnopharmacol. 2007;109:177–206. doi: 10.1016/j.jep.2006.09.006. [DOI] [PubMed] [Google Scholar]
- 5.Nairn JG. Solutions, emulsions, suspensions and extracts. In: Gennaro A, Marderosian AD, Hanson GR, Medwick T, Popovich NG, Schnaare RL, editors. Remington: The science and practice of pharmacy. 20th ed. Philadelphia: Lippincott Williams and Wilkins; 2000. pp. 721–52. [Google Scholar]
- 6.OECD Guidelines for Testing of Chemicals [Internet] France: OECD Publishing; 2006. Jul 11, [Adopted 2006 Mar 23, cited 2009 Mar 22]. Organization for Economic Cooperation and Development (OECD) pp. 1–27. Section 4, Health Effects: Test No. 425: Acute Oral Toxicity: Up-and-Down Procedure; 2006 http://www.oecdbookshop.org/oecd/index.asp/langen . [Google Scholar]
- 7.Akhtar MA, Rashid M, Wahed MI, Islam MR, Shaheen SM, Islam MA, et al. Comparison of long–term antihyperglycemic and hypolipidemic effects between Coccinia cordifolia (Linn.) and Catharanthus roseus (Linn.) and Catharanthus roseus (Linn.) in alloxan–induced diabetic rats. Res J Med Med Sci. 2007;2:29–34. [Google Scholar]
- 8.Kulkarni SK. Handbook of Experimental Pharmacology. 3rd ed. Delhi: Vallabh Prakashan; 2005. Commonly used drugs, their doses and nature of action in laboratory animals; pp. 190–5. [Google Scholar]
- 9.Sood R. Medical laboratory technology methods of interpretations. 5th ed. New Delhi: Japyee Brothers Medical Publishers (P) Ltd; 1999. Clinical chemistry, blood sugar estimation; pp. 433–6. [Google Scholar]
- 10.Allain CC, Poon LS, Chan CG, Richmond W, Fu PC. Enzymatic determination of total serum cholesterol. Clin Chem. 1974;20:470–5. [PubMed] [Google Scholar]
- 11.Fossati P, Lorenzo P. Serum triglycerides determined colorimetrically with an enzyme that produce hydrogen peroxide. Clin Chem. 1982;28:2077–80. [PubMed] [Google Scholar]
- 12.Izzo C, Grillo F, Murador E. Improved method for determination of high–density–lipoprotein cholesterol I. Isolation of high–density lipoproteins by use of polyethylene glycol 6000. Clin Chem. 1981;27:371–4. [PubMed] [Google Scholar]
- 13.Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low–density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem. 1972;18:499–502. [PubMed] [Google Scholar]
- 14.Schulpis K, Karikas GA. Serum cholesterol and triglyceride distribution in 7767 School–aged Greek children. Paediatrics. 1998;101:861–4. doi: 10.1542/peds.101.5.861. [DOI] [PubMed] [Google Scholar]
- 15.Caroll NV, Longley RW, Roe JH. The determination of glycogen in liver and muscle by use of anthrone reagent. J Biol Chem. 1956;200:583–93. [PubMed] [Google Scholar]
- 16.Das S, Jadav RK, Nagchoudhuri J. Effect of fasting on the intestinal absorption of D-glucose and D-xylose in rats in vivo. Indian J Physiol Pharmacol. 2001;45:451–6. [PubMed] [Google Scholar]
- 17.Szkudelski T. The mechanism of alloxan and streptozotocin action in B cells of the rat pancreas. Physiol Res. 2001;50:536–46. [PubMed] [Google Scholar]
- 18.DeFronzo RA. Pathogenesis of type 2 diabetes mellitus. Med Clin North Am. 2004;88:787–835. doi: 10.1016/j.mcna.2004.04.013. [DOI] [PubMed] [Google Scholar]
- 19.Kumar GP, Arulselvan P, Kumar DS. Antidiabetic activity of fruits of Terminalia chebula on streptozotocin induced diabetic rats. J Health Sci. 2006;52:283–91. [Google Scholar]
- 20.Welsch CA, Lachahce PA, Wasserman BP. Dietary phenolic compounds: Inhibition of Na-dependent D-glucose uptake in rat intestinal brush border membrane vesicles. J Nutr. 1989;119:1698–704. doi: 10.1093/jn/119.11.1698. [DOI] [PubMed] [Google Scholar]
- 21.El–Soud NA, Khalil MY, Hussein JS, Oraby FH, Farrag AH. Antidiabetic effects of Fenugreek alkaloid extract in streptozotocin induced hyperglycemic rats. J Appl Sci Res. 2007;3:1073–83. [Google Scholar]
- 22.Hakkim FL, Girija S, Kumar RS, Jalaluddeen MD. Effect of aqueous and ethanol extracts of Cassia auriculata L. flowers on diabetes using alloxan induced diabetic rats. Int J Diabetes Metab. 2007;15:100–6. [Google Scholar]
- 23.Bopanna KN, Kannan J, Gadgil S, Balaraman R, Rathod SP. Antidiabetic and antihyperlipaemic effects of neem seed Kernel powder on alloxan diabetic rabbits. Indian J Pharmacol. 1997;29:162–7. [Google Scholar]
- 24.Wilcox LJ, Borradaile NM, de Dreu LE, Huff MW. Secretion of hepatocyte apo B is inhibited by the flavonoids, naringenin and hesperetin, via reduced activity and expression of ACAT 2 and MTP. J Lipid Res. 2001;42:725–34. [PubMed] [Google Scholar]
- 25.Gebhardt R. Inhibition of Cholesterol Biosynthesis in Primary Cultured Rat Hepatocytes by Artichoke (Cynara scolymus L.) Extracts. J Pharmacol Exp Ther. 1998;286:1122–8. [PubMed] [Google Scholar]


