Abstract
Background/Aim:
We report our experience with the use of octreotide as primary or adjunctive therapy in children with various gastrointestinal disorders.
Patients and Methods:
A pharmacy database identified patients who received octreotide for gastrointestinal diseases. Indications for octreotide use, dosing, effectiveness, and adverse events were evaluated by chart review.
Results:
A total of 21 patients (12 males), aged 1 month to 13 years, were evaluated. Eleven received octreotide for massive gastrointestinal bleeding caused by portal hypertension-induced lesions (n=7), typhlitis (1), Meckel's diverticulum (1), and indefinite source (2). Blood transfusion requirements were reduced from 23±9 mL/kg (mean±SD) to 8±15 mL/kg (P<0.01). Four patients with pancreatic pseudocyst and/or ascites received octreotide over 14.0±5.7 days in 2 patients. In 3 children, pancreatic pseudocyst resolved in 12±2 days and pancreatic ascites resolved in 7 days in 2. Three patients with chylothorax received octreotide for 14±7 days with complete resolution in each. Two infants with chronic diarrhea received octreotide over 11±4.2 months. Stool output decreased from 85±21 mL/kg/day to 28±18 mL/kg/day, 3 months after initiation of octreotide. The child with dumping syndrome responded to octreotide in a week. Adverse events developed in 4 patients: Q-T interval prolongation and ventricular fibrillation, hyperglycemia, growth hormone deficiency, and hypertension.
Conclusion:
Octreotide provides a valuable addition to the therapeutic armamentum of the pediatric gastroenterologist for a wide variety of disorders. Serious adverse events may occur and patients must be closely monitored.
Keywords: Chronic diarrhea, dumping syndrome, gastrointestinal bleeding, octreotide, pancreas
Octreotide is a synthetic peptide analog of somatostatin with the same pharmacologic effects. Its primary advantages over somatostatin are a longer half-life in the circulation, a higher potency, and good bioavailability after subcutaneous administration.[1–3] The half-life of somatostatin is 2–3 minutes compared with 1–2 h for octreotide. Consequently, octreotide has largely supplanted somatostatin as a therapeutic agent for several diseases. The effects of somatostatin and octreotide occur as a result of binding to G protein receptors found throughout the body, including the gastrointestinal tract, pancreas, and the central nervous system.[4] Octreotide decreases the production of gastrointestinal peptides, such as gastrin, secretin, gastric inhibitory peptide, cholecystokinin, neurotensin, motilin, and pancreatic polypeptide.[3,5,6] This reduction in gastrointestinal peptides leads to multiple pharmacologic alterations in intestinal function.
The experience with octreotide for the treatment of gastrointestinal disorders in children is limited, with the vast majority of data in the form of case reports, small case series, or reviews of these reports.[7–9] We report our experience with the use of octreotide as primary or adjunctive therapy for infants and children with multiple gastrointestinal disorders.
PATIENTS AND METHODS
A pharmacy database was used to identify all patients up to 18 years of age, who received octreotide (Novartis Pharmaceutical, Montreal, Canada) at the Alberta Children's Hospital in Calgary, from January 1998 to December 2004. Patients who received octreotide as primary or adjunctive therapy for gastrointestinal disorders were included. Exclusion criterion was the use of octreotide as a primary therapy for nongastrointestinal endocrine disorders. A retrospective review of medical records was performed and demographic data were collected. These included age, weight, gender, and underlying medical condition necessitating octreotide use. Octreotide dosing, duration of use, mode of administration (intermittent or continuous), route of administration (intravenous (IV) or subcutaneous (SC)) and adverse effects attributed to octreotide were also recorded.
The effectiveness of octreotide was evaluated by the assessment of certain measurable variables based on the indication for its use. In patients with gastrointestinal bleeding, packed red blood cell requirement during the 24-h period before and 48-h period following administration of octreotide was obtained. Cessation of bleeding was defined as the absence of bleeding for 48 h after the administration of octreotide with stabilization of vital signs and hemoglobin. Rebleeding was defined as the recurrence of hematemesis, bright red blood in the nasogastric tube, rectum, or melena with hemoglobin drop of 2 gm/dL and deterioration of vital signs after cessation of or attempt to wean octreotide. Failure of therapy was defined as the persistence of bleeding during octreotide therapy. In patients with pancreatic disorders, data were collected based on imaging (ultrasound or computed tomography of abdomen) measurements of pseudocyst size, abdominal drain of pancreatic ascites, or serum lipase levels before and during octreotide administration. For patients with chronic diarrhea, the amount of stool output and total fluid intake were obtained before and during octreotide therapy. In cases of chylothorax, data were collected about chest drain output before and during octreotide administration. For the patient with dumping syndrome, symptomatic response to octreotide therapy was evaluated.
Blood requirements before and during octreotide therapy were compared by paired t-test. Values are expressed as mean±standard deviation. The study was approved by the institutional review board.
RESULTS
Octreotide was administered to 26 patients. Five of 26 patients were excluded: 3 patients with hyperinsulinemic hypoglycemia and 2 with acromegaly. A total of 21 patients aged 1 month to 13 years (5±4.5 years) were included (9 girls, 12 boys). The indications of octreotide use included massive gastrointestinal bleeding (n=11), complications of pancreatitis (n=4), chylothorax postcardiac surgery (n=3), chronic diarrhea (n=2), and dumping syndrome (n=1) [Table 1].
Table 1.
Clinical and therapeutic characteristics of patients with gastrointestinal disorders on octreotide therapy

The causes of gastrointestinal bleeding included esophageal varices (n=4), gastric varix (n=1), portal hypertensive gastropathy (n=2), typhlitis (n=1), Meckel's diverticulum (n=1), and indefinite source (n=2). The mean hemoglobin drop at presentation was 3.31±0.45 g/L. The mean of the initial octreotide bolus administered was 2.2±1.0 μg/kg, and the mean continuous infusion was 2.2±1.0 μg/kg/h. Octreotide infusion rate was tapered by half for 24 h and was stopped when there was no active bleeding. Bleeding ceased in 8 patients (72%) and those with esophageal varices underwent esophageal sclerotherapy or banding once stabilized.
Patients 1, 2, and 9 failed octreotide therapy (28%). Patient 1 underwent a negative upper endoscopy and colonoscopy. Meckel's scan was positive and a Meckel's diverticulum was resected. Nonsteroidal anti-inflammatory drug use for joint pains in patient 2 raises the possibility of proximal small intestinal ulceration or erosion in view of negative upper endoscopy. This child with chronic renal failure did not respond to IV proton pump inhibitor and died 48 h after admission due to cardiac arrhythmia, hyperkalemia, and pulmonary edema. Patient 9 had endstage liver disease secondary to prolonged total parenteral nutrition (TPN) use for short gut, with severe coagulopathy unresponsive to vitamin K injections or fresh frozen plasma and bled persistently despite continuous octreotide infusion for 14 days. The family preferred palliative management rather than the option of combined liver and bowel transplantation.
Bleeding recurred in patients 6, 7, 8, and 10 (50%). Six months after the first gastrointestinal bleed, patient 6 experienced recurrent bleeding esophageal varices with no response to octreotide, and needed emergent portacaval shunt with no recurrence of bleeding. Initially patient 7 with aplastic anemia developed a massive gastrointestinal bleed with oozing from the small intestine that was not controlled with blood products. Octreotide therapy was instituted as outlined in Table 1 and bleeding ceased in 24 h. Therapy was discontinued at 72 h. Nine days after discontinuation of octreotide, massive lower gastrointestinal bleeding recurred that did not respond to reinstitution of octreotide infusion at 2 μg/kg/h for 3 days and then 4 μg/kg/h for 2 days. He developed signs of typhlitis, underwent a right hemicolectomy and had no recurrence of gastrointestinal bleeding. The resected specimen demonstrated extensive cecal necrosis and ulceration. Patient 8 had recurrence of esophageal variceal bleed on two occasions, one month apart. The second bleed occurred two months after the first one and responded to reinstitution of octreotide bolus at 2 μg/kg and 2 μg/kg/h for 3 days. In patient 10, 2 attempts to wean octreotide to 1 μg/kg/hr were associated with recurrence of bleeding. Injection of gastric varix with cyanoacrylate was performed on day 7 of octreotide therapy and octreotide was successfully weaned. Patients 4, 5, 6, and 8 had sclerotherapy of esophageal varices on day 1 of octreotide. Figure 1 demonstrates a significant reduction in packed red blood cell requirement from 23±9 mL/kg before treatment to 8±15 mL/kg after treatment (P<0.01 by paired t test).
Figure 1.
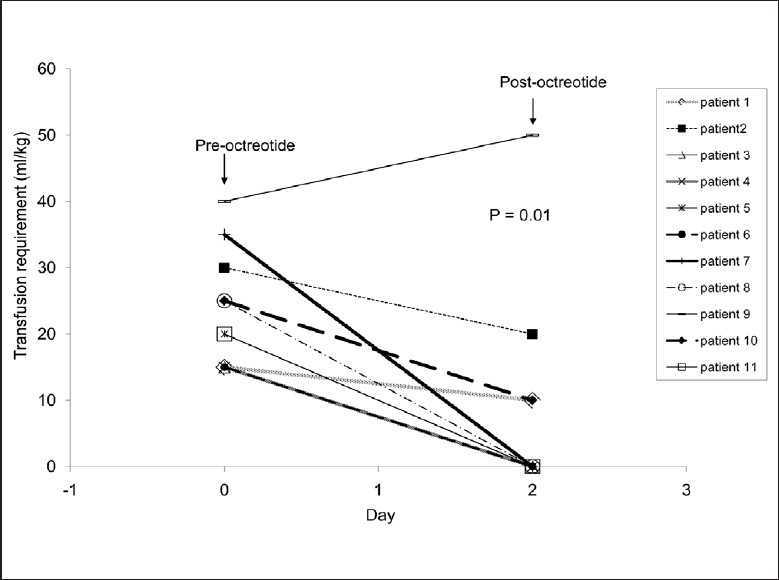
Effect of octreotide on the amount of blood transfusion in gastrointestinal bleeding
Four patients [Table 1, patients 12–15] developed pancreatic pseudocysts and/or ascites within 3 weeks after onset of severe pancreatitis. Pancreatic pseudocysts in patients 12, 14, and 15 resolved completely over a mean duration of 12±2 days and did not recur. Serum lipase decreased from 7682±9064 IU/L (Normal, <60 IU/L) to 347±263 IU/L 2 weeks after initiation of octreotide therapy. In patients 12 and 13, the abdominal drain output of pancreatic fluid at a rate of 12 and 36 mL/kg/day, respectively, resolved completely and drains were removed at day 7 of octreotide therapy. In addition, patient 12 developed watery diarrhea with a stool lipase level >3300 IU/L, thought to be due to an enteropancreatic fistula. The diarrhea resolved 3 days after initiation of octreotide therapy. Patient 15 had endoscopic transgastric drainage of infected pseudocyst on day 1 of octreotide therapy. Culture of cyst fluid grew pseudomonas organisms treated by IV antibiotics. Patient 13, a child with traumatic pancreatitis and ascites, developed severe abdominal pain unresponsive to pain control, bowel rest, and TPN. Pain and ascites resolved within 5 days of octreotide therapy.
Patients 16, 17, and 18 [Table 1] each developed a chylothorax that required placement of a chest tube within 2–3 weeks after cardiac surgery. The resolution of chylothorax was confirmed on chest X-ray. Figure 2 demonstrates the dramatic reduction in chest tube drainage after administration of octreotide in each child.
Figure 2.
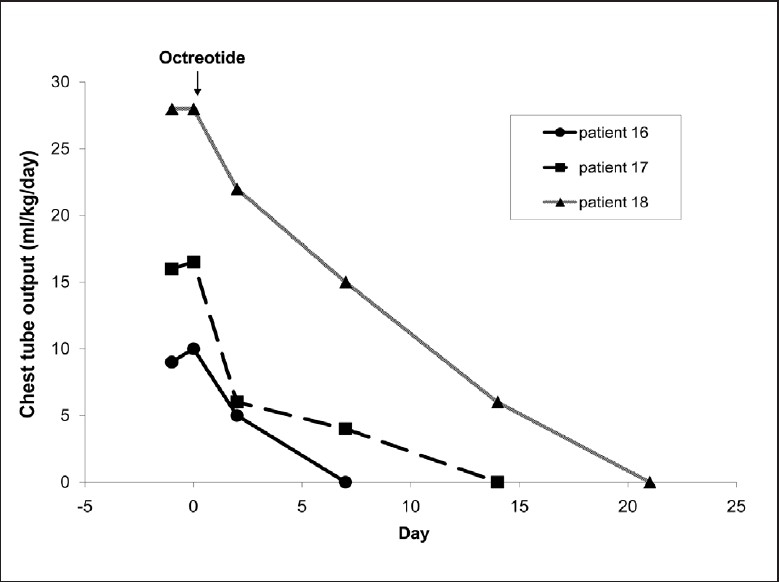
Effect of octreotide on chylothorax postcardiac surgery
Patient 19, an infant with intractable diarrhea due to epithelial dysplasia, displayed a reduction of stool output from a range of 53–73 mL/kg/d prior to octreotide therapy to 25 and 15 mL/kg/d by days 30 and 90 of octreotide therapy, respectively. TPN requirement progressively declined from 85% to 70% and 50% of total daily caloric intake by days 30 and 90 of octreotide therapy, respectively. Fourteen months after initiating octreotide therapy, this patient was receiving 70% of total daily intake by enteral feeds and she grew continuously along 5th centile for both weight and height. Patient 20, a child with short gut after necrotizing enterocolitis, demonstrated a progressive decrease in stool output from 85–105 mL/kg/d to 55 (day 30) and 35 mL/kg/ (day 90) after initiating octreotide therapy. In parallel to the reduction of stool output, TPN requirement decreased from 100% to 80% and 50% of total daily caloric intake at days 30 and 90 of octreotide therapy, respectively. Weight was maintained along the 20th centile.
Patient 21 [Table 1] presented with symptoms suggestive of dumping syndrome that developed 2 months postfundoplication for gastroesophageal reflux disease. Bolus feeding caused a rise of serum glucose from 5.4 to 16 mmol/L at 30 min followed by a dramatic reduction to 2 mmol/L at 90 min postfeeding. The glucose reduction was associated with onset of lethargy, sweating, and diarrhea. Failure to respond to a continuous feeding led to a trial of octreotide, with alleviation of the symptoms within a week.
Side effects of octreotide developed in 4 of 21 patients. Patient 7 suffered bradycardia that progressed into ventricular fibrillation, and was successfully resuscitated. Electrocardiography demonstrated prolonged corrected Q–T interval (Q–Tc) of 0.46–0.48 second (normal, <0.45 second). Echocardiogram and serum electrolytes were normal. At that point, the patient was receiving octreotide at 4 μg/kg/h in an attempt to control lower gastrointestinal bleeding. The dose was reduced to 1 μg /kg/h and the patient was started on oral Propranolol without recurrence of cardiac arrhythmia. Repeated electrocardiography, weeks and months after discontinuation of octreotide therapy, persistently showed borderline Q–Tc intervals of 0.44–0.45 second which raises the probability of pre-existing congenital Q–T prolongation. Apart from octreotide, there was no obvious predisposing factor to explain the occurrence of the arrhythmia. Patient 10 developed hyperglycemia (blood glucose, 18 mmol/L) 1 week after initiating octreotide therapy that was controlled with insulin for 10 days. The insulin was discontinued with no recurrence of hyperglycemia. Patient 20 suffered recurrent hypoglycemia (serum glucose<2.5 mmol/L) 5 months after starting octreotide therapy. Hormonal analysis at the time of hypoglycemia revealed an abnormally low response of serum growth hormone of 1.1 mg/L (Normal, 0.2–7 mg/L), normal serum cortisol, adrenocorticotropin, and thyroid stimulating hormone, and a normally suppressed serum insulin at <21 pmol/L. Provocative growth hormone testing using IV arginine revealed an abnormally low serum growth hormone of 1.2 mg/L (Normal, >3 mg/L). The hypoglycemic episodes responded to growth hormone injections 6 times/week. Hypertension requiring medical therapy developed in patient 21, 3 weeks after initiation of octreotide therapy. Extensive workup for organic causes of hypertension was negative. Hypertension was resolved 2 weeks after discontinuation of octreotide therapy.
DISCUSSION
Octreotide alters multiple aspects of gastrointestinal physiology and is a valuable therapeutic addition in the treatment of a variety of gastrointestinal disorders. In adults, octreotide therapy worked in the management of variceal hemorrhage, secretory diarrhea and profound excessive ileostomy losses, gastroenteropancreatic tumors, dumping syndrome, pancreatic and enterocutaneous fistulas, and pancreatitis.[10,11] Although experience with octreotide is more limited in pediatrics, our data indicate that octreotide is extremely useful to treat several gastrointestinal disorders that remain difficult to manage in children.
Octreotide significantly reduced the amount of blood transfusions in children with severe gastrointestinal bleeding and hemodynamic instability. Because of its potential to reduce portal blood flow, hepatic venous pressure gradient and azygos blood flow,[12,13] the effectiveness of octreotide in controlling upper gastrointestinal bleed in adults has been largely reported in cases of variceal hemorrhage secondary to portal hypertension.[14,15] In this study, 87% of responders had portal hypertension-induced bleeding lesions and octreotide therapy allowed them to be managed with banding or sclerotherapy after hemodynamic stabilization. Randomized controlled trials in adults demonstrate that a combination of endoscopic sclerotherapy/banding and octreotide is more effective than sclerotherapy or banding done alone in controlling acute variceal bleeding and preventing early rebleeding episodes.[15,16] Although octreotide dose for acute variceal hemorrhage is well established in adults with 50–100 μg IV bolus followed by continuous infusion at 25–50 μg/h,[16,17] no pediatric dose has been tested. From results in this study and other pediatric case series,[9,18] it seems that 1–2 μg/kg bolus followed by a continuous infusion of 1–2 μg/kg/h for 2–5 days is effective and safe. No data indicate that a higher dose or longer duration is more effective and free of adverse effects. Higher octreotide dose in patient 7 and longer duration of therapy in patient 10 were associated with major adverse effects.
While the role of octreotide in the treatment of nonvariceal gastrointestinal mucosal lesions is not well established, it has been successfully used to treat severe nonvariceal bleeding in adults and children.[19–21] Octreotide successfully reduced bleeding in a patient with typhlitis and cecal ulceration prior to surgery. Octreotide failed to control massive bleeding in a child with a Meckel's diverticulum, Non-steroidal anti-inflammatory drug induced bleeding in chronic renal failure or in a child with hepatic failure induced coagulopathy. The available data from randomized controlled trials in adults, about the efficacy of octreotide in controlling bleeding peptic ulcers are controversial.[22,23] Octreotide may reduce massive Gastrointestinal bleeding in patients with unknown and potentially treatable causes of bleeding while diagnostic and therapeutic maneuvers are being conducted. It is not a substitute for definitive therapy in these situations.
Historically, pediatric pancreatic pseudocysts have been treated with operative or percutaneous drainage procedures, and occasionally with conservative management.[24] In our study, the use of octreotide has successfully resulted in complete resolution of the pseudocysts over a mean of 15 days, thereby preventing surgery and facilitating wean of TPN. Similar results have been obtained in 4 pediatric case reports.[24–26] Pancreatic fistula and ascites result from pancreatic ductal disruption with subsequent leakage of pancreatic juice. Octreotide inhibited pancreatic secretions and allowed healing of pancreatic duct with the resolution of ascites in 2 of our patients and in a case report.[27] Although the published adult data on the use of octreotide in the treatment of acute pancreatitis are disappointing,[28,29] octreotide dramatically decreased serum lipase levels in all 4 patients and facilitated withdrawal of morphine in patient 13 with good control of pain. Octreotide was used to relieve severe pain associated with chronic pancreatitis perhaps by decreasing intraductal pressure caused by pancreatic secretions.[30] In multicenter controlled therapeutic trials, somatostatin significantly reduced serum lipase levels[28] and reduced the clinical need for analgesics.[31,32] However, it did not reduce mortality. So although there may be benefit in administering octreotide in cases of severe pancreatitis, the substantial number of studies in which no benefit could be demonstrated should also be noted. Diverse therapeutic methods were used and this may be one of the main reasons for the inconsistency of the results.
Somatostatin inhibits pancreatic secretion in a dose-dependent way.[33,34] Pseudocysts in our patients resolved at different time intervals after initiation of octreotide in an inverse relation to the dose of octreotide used. Other pediatric case reports support this observation.[24–26] A lower SC dose of octreotide (2.5 μg/kg/d) that is similar to the octreotide dose used in patient 15, led to resolution of similarly sized pancreatic pseudocysts in 2 children in 4 weeks.[26] At higher doses, 10.5 and 7.5 μg/kg/d, we achieved complete resolution of pseudocysts in patients 12 and 14, respectively, over a shorter time (10–14 days). A continuous IV infusion of octreotide at 1 μg/kg/h was associated with clinical improvement in 3 children with acute pancreatitis.[35] In adults, SC doses of octreotide at 100, 250, and 500 μg three times daily (TID) were used with clinical benefit.[36,37] The minimal effective dosage of octreotide remains to be determined. Low doses may be not sufficient, yet high doses decrease splanchnic blood flow and pancreatic perfusion, which could be associated with aggravation of pancreatitis.[38,39] Octreotide has excellent bioavailability after SC administration[40–42] and is the preferred mode of therapy for the prolonged treatment. However, IV route may be advantageous for acutely ill patients with hypovolemia and circulatory failure when SC absorption of drugs is suboptimal. For the management of complications of pancreatitis, an initial SC dose of 7.5 μg/kg/d divided TID is recommended.
Chylothorax may have deleterious effects on the immunologic and nutritional status of patients. Standard management of chylothorax includes medium chain triglyceride-based feeds, bowel rest with TPN, and surgical intervention, in the form of thoracic duct ligation, pleurodesis or placement of pleuroperitoneal shunt, if medical treatment fails.[40] Such an approach has been associated with significant morbidity, prolonged hospitalization and mortality.[42,43] Data from our study and case reports in children[43–47] indicate that octreotide can treat chylothorax as evidenced by shortening of TPN duration, hospital stay, and avoiding surgery. The mechanism of action of octreotide is not understood. Octreotide may reduce thoracic duct lymph flow,[48] or may act on somatostatin receptors in the splanchnic circulation to reduce lymph fluid absorption.[45] Our mean octreotide dose, 10 μg/kg/d divided b.i.d. SC, was used by others with good results.[48,49] A continuous IV infusion of octreotide at 1–3.5 μg/kg/hr also was effective.[44,47,50]
Octreotide is effective in reducing stool output in children with a variety of disorders, including massive ileostomy loss, intestinal fistula, congenital microvillus atrophy, idiopathic secretory diarrhea, carcinoid tumour, cryptosporidium diarrhea and watery diarrhea hypokalemia achlorohydria syndrome.[51–58] The precise mechanisms by which octreotide exerts its beneficial effect is uncertain. Some studies demonstrated normal gastrointestinal hormonal secretions throughout octreotide therapy, suggesting that the effect of octreotide on stool losses was likely due to the direct inhibition of intestinal secretion and increased intestinal fluid absorption.[51,55] In two children in this case series, stool losses were reduced by 78% and 59% at 3 months post-initiation of octreotide at doses ranging from 8 to 60 μg/kg/d SC divided either b.i.d. or t.i.d. The decrease in stool output was accompanied by tolerance of increasing volumes of enteral feeds and parallel reduction of TPN while maintaining good weight gain. As a consequence, both patients remained free of TPN-associated cholestasis. In the literature, multiple doses of octreotide were used, ranging from 3.8 to 34.6 μg/kg/d SC b.i.d. or t.i.d. for a variable length of time.[51–55]
In adults, octreotide has been proposed as a possible therapeutic agent in patients with dumping syndrome, with doses ranging from 50 to 100 μg b.i.d–q.i.d. SC 15 to 30 min before meals.[59–62] Its therapeutic benefit probably relates both to slowing of gastric emptying,[59,63] increasing small bowel transit time[51] and inhibiting the release of mediators of the vasomotor symptoms.[64] Our patient is the first pediatric case with successful use of octreotide in dumping syndrome. Octreotide before meals appears to be promising in children with dumping syndrome unresponsive to continuous enteral feed and dietary modification.
Four of 21 patients suffered significant adverse effects. Q–T interval prolongation is an extremely rare adverse effect of octreotide.[65] A high octreotide infusion rate (4 μg/kg/h) in patient 7 with probable pre-existing congenital Q–T prolongation might have well triggered the development of bradycardia and cardiac arrhythmia, which are rare side effects of octreotide.[66] Octreotide should be avoided in patients with diagnosed or suspected congenital long Q–Tc syndrome and cautiously used in conjunction with drugs that prolong Q–T interval. Electric cardiography prior to initiation of octreotide therapy is strongly indicated. Prolonged use of octreotide (>14 days) in patients 10, 20, and 21 was a possible determining factor in the development of adverse effects in these patients. In patient 21, a lack of other possible causes of hypertension and normalization of blood pressure after discontinuation of octreotide, make octreotide the most likely cause of hypertension, another recognised cardiovascular effect of octreotide.[62] Therefore, regular check of blood pressure is recommended during octreotide therapy. Because of its inhibitory action on insulin, octreotide has been associated with glucose intolerance and hyperglycemia that may necessitate insulin therapy.[18] This indicates close monitoring of blood glucose during octreotide therapy. In adults, octreotide has been frequently used in the treatment of acromegaly, due to its inhibitory actions on growth hormone and insulin-like growth factor 1 secretion.[67] Growth hormone level was suppressed in 2 children treated with octreotide for 3–4 weeks.[24,51] The concern that octreotide may cause growth retardation on long-term use has been refuted by several studies.[54,68,69] The development of symptomatic growth hormone deficiency in patient 20 indicates that close monitoring of growth hormone level and growth velocity would be required. None of our patients developed cholelithiasis, a complication reported in long-term users of octreotide.[70]
CONCLUSION
In conclusion, although our study is retrospective and uncontrolled, it indicates that octreotide provides a valuable addition to the therapeutic armamentum of the pediatric gastroenterologists for a wide variety of disorders. Large prospective, placebo-controlled and multicenter trials are needed to determine octreotide pharmacokinetics, therapeutic doses and clinical efficacy in children. Serious adverse events may occur at high doses or long-term use of octreotide and patients must be closely monitored.
Footnotes
Source of Support: Nil
Conflict of Interest: None declared.
REFERENCES
- 1.Gyr KE, Meier R. Pharmacodynamic effects of Sandostatin in the gastrointestinal tract. Digestion. 1993;54(Suppl 1):14–9. doi: 10.1159/000201070. [DOI] [PubMed] [Google Scholar]
- 2.Lucey MR, Yamada T. Biochemistry and physiology of gastrointestinal somatostatin. Dig Dis Sci. 1989;34(3 Suppl):5S–13S. doi: 10.1007/BF01536041. [DOI] [PubMed] [Google Scholar]
- 3.Whitehouse I, Beglinger C, Ruttimann G, Gyr K. Inhibition of pentagastrin-stimulated acid secretion after subcutaneous administration of a new somatostatin analogue. Gut. 1986;27:141–6. doi: 10.1136/gut.27.2.141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Polak JM, Broom SR. Somatostatin localisation in tissues. Scand J Gastroenterol Suppl. 1986;119:11–21. doi: 10.3109/00365528609087427. [DOI] [PubMed] [Google Scholar]
- 5.Kraenzlin ME, Wood SM, Neufeld M, Adrian TE, Bloom SR. Effect of long acting somatostatin-analogue, SMS 201-995, on gut hormone secretion in normal subjects. Experientia. 1985;41:738–40. doi: 10.1007/BF02012573. [DOI] [PubMed] [Google Scholar]
- 6.Lembcke B, Creutzfeldt W, Schleser S, Ebert R, Shaw C, Koop I. Effect of the somatostatin analogue sandostatin (SMS 201-995) on gastrointestinal, pancreatic and biliary function and hormone release in normal men. Digestion. 1987;36:108–24. doi: 10.1159/000199408. [DOI] [PubMed] [Google Scholar]
- 7.Tauber MT, Harris AG, Rochiccioli P. Clinical use of the long acting somatostatin analogue octreotide in pediatrics. Eur J Pediatr. 1994;153:304–10. doi: 10.1007/BF01956407. [DOI] [PubMed] [Google Scholar]
- 8.Heikenen JB, Pohl JF, Werlin SJ, Bucuvalas JC. Octreotide in pediatric patients. J Pediatr Gasroenterol Nutr. 2002;35:600–9. doi: 10.1097/00005176-200211000-00003. [DOI] [PubMed] [Google Scholar]
- 9.Eroglu Y, Emerick KM, Whitington PF, Alonso EM. Octreotide therapy for control of acute gastrointestinal bleeding in children. J Pediatr Gastroenterol Nutr. 2004;38:41–7. doi: 10.1097/00005176-200401000-00011. [DOI] [PubMed] [Google Scholar]
- 10.Grosman I, Simon D. Potential gastrointestinal uses of somatostatin and its synthetic analogue octreotide. Am J Gastroenterol. 1990;85:1061–71. [PubMed] [Google Scholar]
- 11.Mosdell KW, Visconti JA. Emerging indications for octreotide therapy, Part 1. Am J Hosp Pharm. 1994;51:1184–92. [PubMed] [Google Scholar]
- 12.Hadengue A. Somatostatin or octreotide in acute variceal bleeding. Digestion. 1999;60(Suppl 2):31–41. doi: 10.1159/000051478. [DOI] [PubMed] [Google Scholar]
- 13.McCormick PA, Dick R, Siringo S. Octreotide reduces azygous blood flow in cirrhotic patients with portal hypertension. Eur J Gastroenterol Hepatol. 1990;2:489–92. [Google Scholar]
- 14.Hwang SJ, Lin HC, Chang CF, Lee FY, Lu CW, Hsia HC, et al. A randomized controlled trial comparing octreotide and vasopressin in the control of acute oesophageal bleeding. J Hepatol. 1992;16:320–5. doi: 10.1016/s0168-8278(05)80663-4. [DOI] [PubMed] [Google Scholar]
- 15.Besson I, Ingrand P, Person B, Boutroux D, Heresbach D, Bernard P, et al. Sclerotherapy with or without octreotide for acute variceal bleeding. N Engl J Med. 1995;333:555–60. doi: 10.1056/NEJM199508313330904. [DOI] [PubMed] [Google Scholar]
- 16.Sung JJ, Chung SC, Yung MY, Lai CW, Lau JY, Lee YT, et al. Prospective randomised study of effect of octreotide in rebleeding from oesophageal varices after endoscopic ligation. Lancet. 1995;346:1666–9. doi: 10.1016/s0140-6736(95)92840-5. [DOI] [PubMed] [Google Scholar]
- 17.Sadowski DC. Use of octreotide in the acute management of bleeding oesophageal varices. Can J Gastroenterol. 1997;11:339–43. doi: 10.1155/1997/156920. [DOI] [PubMed] [Google Scholar]
- 18.Siafakas C, Fox VL, Nurko S. Use of octreotide for the treatment of severe gastrointestinal bleeding in children. J Pediatr Gastroenterol Nutr. 1998;26:356–9. doi: 10.1097/00005176-199803000-00022. [DOI] [PubMed] [Google Scholar]
- 19.Jenkins SA, Taylor BA, Nott DM, Ellenbogen S, Haggie J, Shields R. Management of massive gastrointestinal haemorrhage from multiple sites of peptic ulceration with somatostatin and octreotide- A report of five cases. Gut. 1992;33:404–7. doi: 10.1136/gut.33.3.404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lamberts SW, van der Lely AJ, de Herder WW, Hofland LJ. Octreotide. N Eng J Med. 1996;334:246–54. doi: 10.1056/NEJM199601253340408. [DOI] [PubMed] [Google Scholar]
- 21.Nikolopoulou VN, Thomopoulos KC, Katsakoulis EC, Vasilopoulos AG, Margaritis VG, Vagianos CE. The effect of octreotide as an adjunct treatment in active nonvariceal upper gastrointestinal bleeding. J Clin Gastroenterol. 2004;38:243–7. doi: 10.1097/00004836-200403000-00009. [DOI] [PubMed] [Google Scholar]
- 22.Christiansen J, Ottenjann R, Von Arx F. Placebo-controlled trial with the somatostatin analogue SMS 201-995 in peptic ulcer bleeding. Gastroenterology. 1989;97:568–74. doi: 10.1016/0016-5085(89)90626-4. [DOI] [PubMed] [Google Scholar]
- 23.Lin HJ, Perng CL, Wang K, Lee CH, Lee SD. Octreotide for arrest of peptic ulcer haemorrhage – a prospective, randomized controlled trial. Hepatogastroenterology. 1995;42:856–60. [PubMed] [Google Scholar]
- 24.Mulligan C, Howell C, Hatley R, Martindale R, Clark J. Conservative management of pediatric pancreatic pseudocyst using octreotide acetate. Am Surg. 1995;61:206–9. [PubMed] [Google Scholar]
- 25.Bosman-Vermeeren JM, Veereman-Wauters G, Broos P, Eggermant E. Somatostatin in the treatment of pancreatic pseudocyst in a child. J Pediatr Gastroenterol Nutr. 1996;23:422–5. doi: 10.1097/00005176-199611000-00011. [DOI] [PubMed] [Google Scholar]
- 26.Tissieres P, Bugmann P, Rimensberger PC, Mentha G, Lencoultre C, Belli DC. Somatostatin in the treatment of pancreatic pseudocyst complicating acute pancreatitis in a child with liver transplantation. J Pediatr Gastroenterol Nutr. 2000;31:445–7. doi: 10.1097/00005176-200010000-00022. [DOI] [PubMed] [Google Scholar]
- 27.Rushforth JA, Beck JM, McMahon M, Puntis JW. Resolution of pancreatic ascites with octreotide. Arch Dis Child. 1993;68:135–6. doi: 10.1136/adc.68.1.135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Cavallini G, Frulloni L. Somatostatin and octreotide in acute pancreatitis: the never-ending story. Dig Liver Dis. 2001;33:192–201. doi: 10.1016/s1590-8658(01)80077-3. [DOI] [PubMed] [Google Scholar]
- 29.Greenberg R, Haddad R, Kashtan H, Kaplan O. The effects of somatostatin and octreotide on experimental and human acute pancreatitis. J Lab Clin Med. 2000;135:112–21. doi: 10.1067/mlc.2000.104457. [DOI] [PubMed] [Google Scholar]
- 30.Malfertheiner P, Mayer D, Buchler M, Dominquesz-Munoz JE, Schiefer B, Ditschuneit H. Treatment of pain in chronic pancreatitis by inhibition of pancreatic secretion with octreotide. Gut. 1995;36:450–4. doi: 10.1136/gut.36.3.450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Usadel KH, Uberla KK, Leuschner U. Treatment of acute pancreatitis with somatostatin. Results of the multi-center double-blind trial (APTS-Study) Dig Dis Sci. 1985;30:992. [Google Scholar]
- 32.D’Amico D, Favia G, Biasiato R, Casaccia M, Falcome F, Fersini M, et al. The use of somatostatin in acute pancreatitis: Results of a multi-center trial. Hepatogastroenterology. 1990;37:92–8. [PubMed] [Google Scholar]
- 33.Choi TK, Mok F, Zhan WH, Fan ST, Lai ECS, Wong J. Somatostatin in the treatment of acute pancreatitis: a prospective randomised controlled trial. Gut. 1989;30:223–7. doi: 10.1136/gut.30.2.223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Gullo L, Priori P, Scarpignato C, Baldoni F, Mattioli G, Barbara L. Effect of somatostatin 14 on pure human pancreatic secretion. Dig Dis Sci. 1987;32:1065–70. doi: 10.1007/BF01300189. [DOI] [PubMed] [Google Scholar]
- 35.Lam JC, Aters S, Tobias JD. Initial experience with octreotide in the pediatric population. Am J Ther. 2001;8:409–15. doi: 10.1097/00045391-200111000-00005. [DOI] [PubMed] [Google Scholar]
- 36.Paran H, Neufeld D, Mayo A, Shwartz I, Singer P, Kaplan O, et al. Preliminary report of a prospective study in the treatment of severe acute pancreatitis. J Am Coll Surg. 1995;181:121–4. [PubMed] [Google Scholar]
- 37.Binder M, Uhl W, Friess H, Malfertheiner P, Bachler MW. Octreotide in the treatment of acute pancreatitis: results of a unicentric prospective trial with three different octreotide dosages. Digestion. 1994;55(Suppl 1):20–3. doi: 10.1159/000201184. [DOI] [PubMed] [Google Scholar]
- 38.Conway DR, Djuricin G, Prinz RA. The effect of somatostatin analogue (SMS 201-995) on pancreatic blood flow. Surgery. 1988;104:1024–30. [PubMed] [Google Scholar]
- 39.Klar E, Rattner DW, Compton C, Stanford G, Chernow B, Warshow AL. Adverse effect of therapeutic vasoconstrictors in experimental acute pancreatitis. Ann Surg. 1991;214:168–74. doi: 10.1097/00000658-199108000-00011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Beghetti M, La Scala G, Belli D, Bugmann P, Kalangos A, Le Coultre C. Etiology and management of pediatric chylothorax. J Pediatr. 2000;136:653–58. doi: 10.1067/mpd.2000.104287. [DOI] [PubMed] [Google Scholar]
- 41.DeLedinghen V, Ripault MP, Silvain C, Beauchant M, Raffoux N, Bouquet S. Subcutaneous octreotide for the prevention of early variceal bleeding. Hepatology. 1996;23:938–39. doi: 10.1002/hep.510230443. [DOI] [PubMed] [Google Scholar]
- 42.Cerfolio RJ, Allen MS, Deschamps C, Trastek VF, Pairolero PC. Postoperative chylothorax. J Thorac Cardiovasc Surg. 1996;112:1361–6. doi: 10.1016/S0022-5223(96)70152-6. discussion 1365-6. [DOI] [PubMed] [Google Scholar]
- 43.Browse NL, Wilson NM, Russo F, al-Hassan H, Allen DR. Aetiology and treatment of chylous ascites. Br J Surg. 1992;79:1145–50. doi: 10.1002/bjs.1800791110. [DOI] [PubMed] [Google Scholar]
- 44.Rosti L, Bini RM, Chessa M, Butera G, Drago M, Carminati M. The effectiveness of octreotide in the treatment of post-operative chylothorax. Eur J Pediatr. 2002;161:149–50. doi: 10.1007/s00431-001-0891-7. [DOI] [PubMed] [Google Scholar]
- 45.Cheung Y, Leung MP, Yip M. Octreotide for treatment of post-operative chylothorax. J Pediatr. 2001;139:157–9. doi: 10.1067/mpd.2001.115572. [DOI] [PubMed] [Google Scholar]
- 46.Goyal A, Smith NP, Jesudason EC, Kerr S, Losty PD. Octreotide for treatment of chylothorax after repair of congenital diaphragmatic hernia. J Pediatr Surg. 2003;38:E19–20. doi: 10.1016/s0022-3468(03)00294-x. [DOI] [PubMed] [Google Scholar]
- 47.Au M, Weber TR, Fleming RE. Successful use of somatostatin in a case of neonatal chylothorax. J Pediatr Surg. 2003;38:1106–7. doi: 10.1016/s0022-3468(03)00205-7. [DOI] [PubMed] [Google Scholar]
- 48.Nakabayashi H, Sagara H, Usukura N, Yoshimitsu K, Imamura T, Seta T, et al. Effect of somatostatin on the flow rate and triglyceride levels of thoracic duct lymph in normal and vagotomized dogs. Diabetes. 1981;30:440–45. doi: 10.2337/diab.30.5.440. [DOI] [PubMed] [Google Scholar]
- 49.Bhatia C, Pratap U, Slavik Z. Octreotide therapy: a new horizon in treatment of iatrogenic chyloperitoneum. Arch Dis Child. 2001;85:234–5. doi: 10.1136/adc.85.3.234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Ballinger AB, Farthing MJ. Octreotide in the treatment of intestinal lymphangiectasia. Eur J Gastroenterol Hepatol. 1998;10:699–702. [PubMed] [Google Scholar]
- 51.Ohlbaum P, Galperine RI, Demarquez JL, Vergnes P, Martin C. Use of a long-acting somatostatin analogue (SMS 201-995) in controlling a significant ileal output in a 5-year-old child. J Pediatr Gastroenterol Nutr. 1987;6:466–70. doi: 10.1097/00005176-198705000-00026. [DOI] [PubMed] [Google Scholar]
- 52.Lamireau T, Galpérine RI, Ohlbaum P, Demarquez JL, Vergnes P, Kurzenne Y, et al. Use of a long acting somatostatin analogue in controlling ileostomy diarrhoea in infants. Acta Pediatr Scand. 1990;79:871–2. doi: 10.1111/j.1651-2227.1990.tb11572.x. [DOI] [PubMed] [Google Scholar]
- 53.Inamdar S, Slim MS, Bostwick H, Godine L. Treatment of duodenocutaneous fistula with somatostatin analogue in a child with dermatomyositis. J Pediatr Gastroenterol Nutr. 1990;10:402–4. doi: 10.1097/00005176-199004000-00022. [DOI] [PubMed] [Google Scholar]
- 54.Couper RTL, Berzen A, Berall G, Sherman PM. Clinical response to the long acting somatostatin analogue SMS 201-995 in a child with congenital microvillus atrophy. Gut. 1989;30:1020–4. doi: 10.1136/gut.30.7.1020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Jaros W, Biller J, Greer S, O’dorisio T, Grand R. Successful treatment of idiopathic secretory diarrhea of infancy with the somatostatin analogue SMS 201-995. Gastroenterology. 1988;94:189–93. doi: 10.1016/0016-5085(88)90629-4. [DOI] [PubMed] [Google Scholar]
- 56.Helfer N, Wyss M, Friedli B, Sizonenko PC. Tumeur carcinoide bronchique associee a une secretion exageree de prolactine (PRL) et d’hormone de croissance (STH); traitement par la sandostatine et la bromocriptine. Schweiz Med Wochenschr. 1990;120(Suppl 34):26. [Google Scholar]
- 57.Guarino A, Berni Canani R, Spagnuolo MI, Bisceglia M, Boccia MC, Rubino A. In vivo and in vitro efficacy of octreotide for treatment of enteric cryptosporidiosis. Dig Dis Sci. 1998;43:436–41. doi: 10.1023/a:1018839329759. [DOI] [PubMed] [Google Scholar]
- 58.Smith SS, Shulman DI, O’Dorisio TM, McClenathan DT, Borger JA, Bercu BB, et al. Watery diarrhea, hypokalemia, achlorohydria syndrome in an infant: effect of the long-acting somatostatin analogue SMS 201-995 on the disease and linear growth. J Pediatr Gastroenterol Nutr. 1987;6:710–16. [PubMed] [Google Scholar]
- 59.Geer RJ, Richards WO, O’Dorisio TM, Woltering EO, Williams S, Rice D, et al. Efficacy of octreotide acetate in treatment of severe postgastrectomy dumping syndrome. Ann Surg. 1990;212:678–87. doi: 10.1097/00000658-199012000-00005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Hopman WP, Wolberink RG, Lamers CB, Van Tongeren JH. Treatment of the dumping syndrome with the somatostatin analogue SMS 201-995. Ann Surg. 1988;207:155–9. doi: 10.1097/00000658-198802000-00007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Primrose JN, Johnston D. Somatostatin analogue SMS 201-995 (octreotide) as a possible solution to the dumping syndrome after gastrectomy or vagotomy. Br J Surg. 1989;76:140–4. doi: 10.1002/bjs.1800760212. [DOI] [PubMed] [Google Scholar]
- 62.Tulassay Z, Tulassay T, Gupta R, Cierny G. Long acting somatostatin analogue in dumping syndrome. Br J Surg. 1989;76:1294–95. doi: 10.1002/bjs.1800761223. [DOI] [PubMed] [Google Scholar]
- 63.Petersen JM, Saltzman M, Sherwin RS, Lange R, McCallum RW. Somatostatin inhibits gastric emptying of solids and liquids in man (Abstract) Dig Dis Sci. 1984;29:A64. [Google Scholar]
- 64.Hasler WL, Soudah HC, Owyang C. Mechanisms by which octreotide ameliorates symptoms in the dumping syndrome. J Pharmacol Exp Ther. 1996;277:1359–65. [PubMed] [Google Scholar]
- 65.Fatti LM, Scacchi M, Lavezzi E, Pecori Giraldi F, De Martin M, Toja P, et al. Effects of treatment with somatostatin analogues on QT interval duration in acromegalic patients. Clin Endocrinol (Oxf) 2006;65:626–30. doi: 10.1111/j.1365-2265.2006.02639.x. [DOI] [PubMed] [Google Scholar]
- 66.Herrington AM, George KW, Moulds CC. Octreotide-induced bradycardia. Pharmacotherapy. 1998;18:413–6. [PubMed] [Google Scholar]
- 67.Battershill PE, Clissold SP. Octreotide: A review of its pharmacokinetic properties and therapeutic potential in conditions associated with excessive peptide secretion. Drugs. 1989;38:658–702. doi: 10.2165/00003495-198938050-00002. [DOI] [PubMed] [Google Scholar]
- 68.DeClue TJ, Malone JI, Bercu BB. Linear growth during long-term treatment with somatostatin analog (SMS 201-995) for persistent hyperinsulinemic hypoglycemia of infancy. J Pediatr. 1990;116:747–50. doi: 10.1016/s0022-3476(05)82665-4. [DOI] [PubMed] [Google Scholar]
- 69.Glaser B, Landau H, Smilovici A, Nesher R. Persistent hyperinsulinaemic hypoglycaemia of infancy: long-term treatment with the somatostatin analogue Sandostatin. Clin Endocrinol. 1989;31:71–80. doi: 10.1111/j.1365-2265.1989.tb00455.x. [DOI] [PubMed] [Google Scholar]
- 70.Dowling RH, Hussaini SH, Murphy GM, Besser GM, Wass JA. Gallstones during octreotide therapy. Metabolism. 1992;41(9 Suppl 2):22–33. doi: 10.1016/0026-0495(92)90027-8. [DOI] [PubMed] [Google Scholar]


