Abstract
Food allergy is a major public health problem for which there is no effective treatment. We examined the immunological changes that occurred in a group of children with significant cow’s milk allergy undergoing a novel and rapid high dose oral desensitization protocol enabled by treatment with omalizumab (anti-IgE mAb). Within a week of treatment, the CD4+ T cell response to milk was nearly eliminated, suggesting anergy in, or deletion of, milk-specific CD4+ T cells. Over the following three months while the subjects remained on high doses of daily oral milk, the CD4+ T cell response returned, characterized by a shift from IL-4 to IFN-γ production. Desensitization was also associated with reduction in milk-specific IgE and a 15-fold increase in milk-specific IgG4. These studies suggest that high dose oral allergen desensitization may be associated with deletion of allergen-specific T cells, without the apparent development of allergen-specific Foxp3+ regulatory T cells.
Introduction
Although allergen desensitization in patients allergic to environmental agents such as pollens, dust mite, or cat dander has been performed for more than 100 years 1, the precise immunological mechanisms of allergen desensitization remain controversial. The development of blocking IgG antibody was initially thought to play an important role in mediating the allergen desensitization and tolerance processes 2. However, other mechanisms such as immune suppression, mediated by allergen-specific adaptive regulatory T (TReg) cells, have received intense investigation as the cell type that accounts for the success of immunotherapy 3–6. The development of allergen-specific Foxp3+ TReg cells during desensitization with inhalant allergen or bee venom is consistent with the idea that tolerance induced with low antigen doses (microgram amounts), such as those used in subcutaneous desensitization protocols, is associated with the development of allergen-specific TReg cells 7.
Protocols for oral allergen desensitization in patients with immunoglobulin (Ig)E-mediated food allergy are currently being developed, but little is known about the mechanisms of oral desensitization, during which high doses (gram amounts) of oral antigen are generally administered. In mouse models of oral tolerance, administration of high doses of oral antigen in naïve animals is associated with the development of anergy and deletion of antigen-specific T cells, whereas administration of low doses of oral antigen is associated with TReg cell development 8. On the other hand, much less is known about the mechanisms of oral desensitization in the setting of established allergy in humans.
In humans, the examination of the immunological mechanisms of oral desensitization for food allergy has been hampered by several problems. First, unpredictable allergic reactions in patients have resulted in desensitization courses that vary from months to years. In addition, food-specific T cells, which constitute a very small fraction (<1%) of peripheral T cells, are difficult to study, and have been examined primarily via bulk population activation, or by cloning (or establishing cell lines), which typically involves several cycles of antigen stimulation with exogenous cytokines 9–11, although in some studies, peanut specific T cells were examined more directly using a dye dilution method 12, 13. Nevertheless, studies of oral food desensitization consistently demonstrate a decrease in allergen-specific IgE and an increase in allergen-specific IgG4, suggesting that oral desensitization induces allergen-specific immune deviation 14–18.
To better understand the immunological changes that occur during oral food desensitization, we examined a small cohort of children with IgE-mediated milk allergy in a clinical trial of oral cow’s milk desensitization performed in combination with a 16-week course of omalizumab (anti-IgE monoclonal antibodies, Xolair). The oral desensitization protocol, which was the first to use omalizumab to limit allergic reactions, allowed patients to be rapidly (over a 7–11 week period of time) desensitized to high doses of milk with a uniform dosing schedule. The clinical findings of this study, showing that 10 of the 11 patients with significant milk allergy were desensitized, have been briefly described elsewhere 19. Herein, we describe the dynamic milk-specific immunological changes that occurred in these patients, analyzed in several subgroups of patients, over the course of the oral milk desensitization protocol.
Results
All subjects in the clinical trial began treatment with omalizumab at week 0, and started oral desensitization with milk at week 9 19. 12 doses of milk were given on the first day of desensitization, increasing from 0.1 mg to 1,000 mg every 30 minutes (cumulative dose on the first day, 2,000 mg milk protein). The patients then continued on a daily dose of milk (1,000 mg/day), which increased every 7 days until a dose of 2,000 mg milk/day was achieved. At week 16, the omalizumab was discontinued, but patients remained on a daily dose (2,000 mg/day) of milk. At week 24, nine of the 10 patients remaining in the study passed a double blind food challenge and began taking >8,000 mg of milk per day.
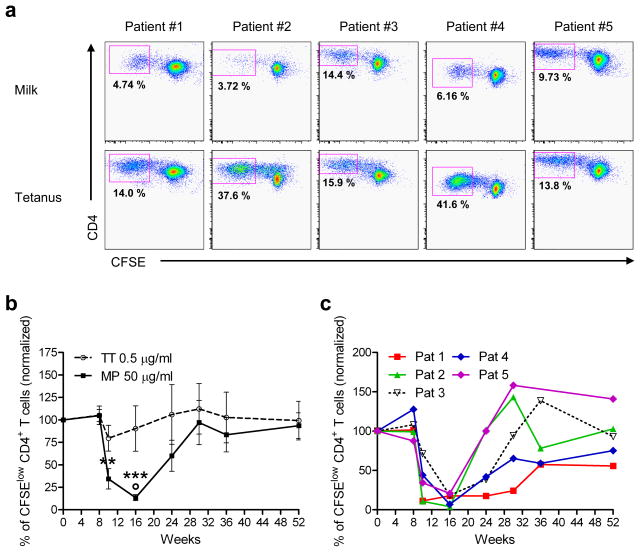
Milk-induced CD4 T cell proliferation is reduced during rush desensitization
Peripheral blood mononuclear cells (PBMC) from a subset of 5 patients were isolated at multiple time points during the desensitization protocol (weeks 0, 9, 10, 12, 14, 16, 24, 30, 36 and 52), and frozen in aliquots in liquid nitrogen for later examination. To characterize the response to milk, PBMC from multiple time points for each patient were thawed, labeled with carboxyfluorescein succinimidyl ester (CFSE) and cultured in the presence of milk proteins (MP) (or tetanus toxoid (TT)) for 7 days. In all patients examined, there was a vigorous proliferative response to milk proteins (and TT) at week 0, as demonstrated by flow cytometry, which showed a significant number of CD4+ T cells with low levels of CFSE, representing cells that underwent multiple cell divisions (Figure 1a) (mean milk-specific CD4+ T cells=7.75%; mean tetanus-specific cells=24.6%; mean, no antigen=0.62%). However, a striking decrease in milk-specific CD4+ T cell proliferation was observed 1 week after initiation of desensitization in all 5 patients examined, which persisted as the daily dose of milk increased (during weeks 8 to 16) (Figures 1b and c). This effect was specific for the milk-induced response, and was not observed with the TT response. Importantly, the response to milk returned over the ensuing 3–4 months, while the subjects remained on daily oral milk. Although omalizumab treatment was stopped at week 16 and coincided with the increase in milk specific proliferation, there was no appreciable change in the tetanus toxoid response.
Figure 1. Milk-induced CD4+ T cell proliferation is greatly reduced during the rush desensitization phase.
(a) Frozen PBMC, isolated from 5 milk allergic patients undergoing rush milk desensitization, were thawed and labeled with CFSE, and cultured in the presence of tetanus toxoid (TT) or milk proteins (MP) for 7 days. Cells were then collected, stained with anti-CD4 mAb, and analyzed by flow cytometry. The number in each panel represents the fraction of total CD4+ cells that are within the CFSElow CD4+ gate (antigen-specific proliferating cells) on day 0. Mean CD4+ milk-specific proliferating cells=7.75%; mean CD4+ tetanus specific cells=24.6%. Mean CD4+ proliferating cells without antigen=0.62%.
(b) Each line represents data for all 5 patients combined (solid line, milk protein (MP); dashed line, tetanus toxoid (TT)). Data represent mean % antigen-specific cells (CFSElow CD4+ cells) of total CD4+ cells, normalized to day 0 for each patient ± SEM, over the course of the study. ** p < 0.01, *** p < 0.001 versus baseline and week 8, determined using paired t-test. ° p < 0.05 versus TT response, determined using two-way ANOVA for matched values with Bonferroni’s post hoc test.
(c) Each line represents data for an individual patient, of % milk-specific CFSElow CD4+ cells of total CD4+ cells, normalized to day 0 for each patient, over the course of the study.
Absence of functional milk specific TReg cells during the desensitization
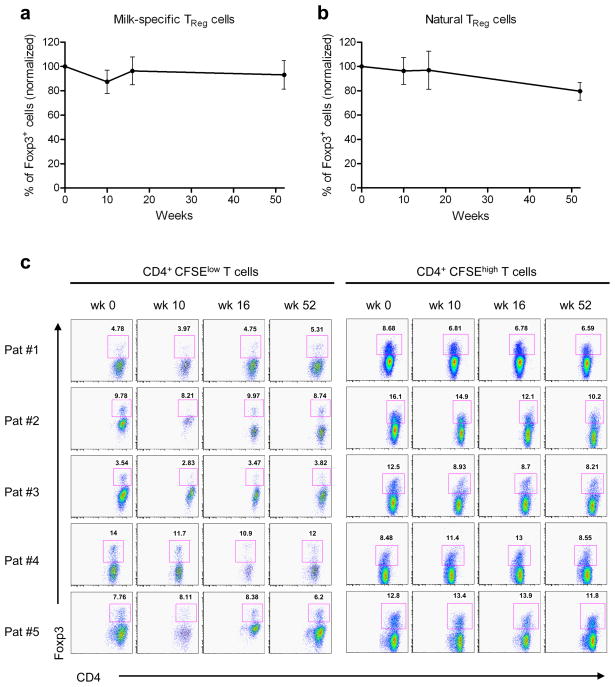
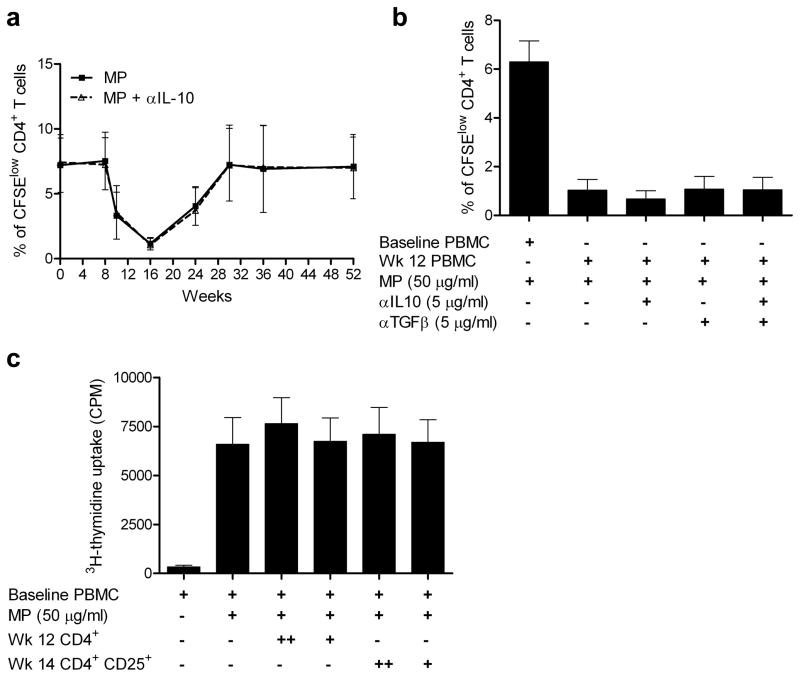
We examined possible mechanisms for the initial transient reduction in the milk-specific proliferative response. Surprisingly, we found no evidence for an increase in the number of milk-specific TReg cells (CFSElow CD4+ Foxp3+) in the cultures stimulated with milk proteins (Figure 2a). Since TReg cells may not proliferate, we also examined CFSEhigh (non-proliferating cells) and found no increase in the number of CD4+ Foxp3+ TReg cells (Figure 2b and c). Furthermore, we showed that IL-10 secreting suppressive/regulatory T cells were not responsible for the reduced proliferation, since no increase in milk-induced CD4+ T cell proliferation was observed in the presence of anti-IL-10 mAb (Figure 3a). Moreover, addition of anti-10 and anti-TGF-β (5 μg/ml) mAb together did not increase the milk-induced proliferative response at any time point, indicating that IL-10/TGF-β producing regulatory T cells were not responsible for the reduction in the response to milk (data for week 12, Figure 3b). Finally, we performed mixing experiments, in which purified CD4+ T cells or CD4+ CD25+ T (TReg) cells from PBMC collected at weeks 12 and 14, when the proliferative response was low, were cultured with PBMC from week 0. In these experiments, milk-induced proliferation was assessed by [3H]-thymidine incorporation during the last 16 hours of a 6-day culture. Neither CD4+ T cells nor CD4+ CD25+ cells were able to reduce the week 0 PBMC response to milk (Figure 3c). Together, these results suggest that Treg cells were not involved in the reduction of the milk-specific CD4+ T cell proliferation observed during desensitization at weeks 10–14.
Figure 2. Milk specific Foxp3+ T cells are not detected during the milk desensitization.
(a–c) CFSE-labeled PBMC isolated from 5 milk allergic patients undergoing milk desensitization were cultured in the presence of milk proteins for 7 days. Cells were then collected, fixed and permeabilized, stained with anti-CD4 and anti–Foxp3, and analyzed by flow cytometry.
(a–b) Figures represent combined percentages of CD4+ Foxp3+ cells of total cells normalized to day 0. Data represent mean ± SEM.
(a) Normalized CD4+ CFSElow cells (proliferating cells, representing milk-specific TReg cells).
(b) Normalized CD4+ CFSEhigh cells (nonproliferating cells, representing natural TReg cells).
(c) Individual dot plots of flow cytometry analysis are shown. CD4+CFSElow T cells (left panels) represent milk-specific TReg cells, which did not increase over time. CD4+CFSEhigh T cells (right panels) represent natural TReg cells, which did not change over time.
Figure 3. Functional regulatory T cells are not detected during the milk desensitization.
(a–b) CFSE-labeled PBMC isolated from 5 patients undergoing milk desensitization were cultured in the presence of milk (MP) for 7 days. Cells were then collected, stained with anti-CD4 mAb, and analyzed by flow cytometry.
(a) Frozen samples from multiple time points were thawed and cultured with or without anti-IL-10 mAb and milk proteins. Percentages of the milk-specific cell populations (CFSElow) within the CD4+ gate are shown. Results with and without anti-IL-10 mAb overlap, indicating an absence of IL-10 mediated suppression.
(b) PBMC from week 12 were cultured with or without anti-IL-10, anti-TGF-β mAb or both and compared to baseline PBMC. Figures represent percentages of the milk-specific cell populations (CFSElow) within the CD4+ gate. No increase in proliferation was observed with the mAbs, indicating an absence of IL-10 or TGF-β mediated suppression.
(c) CD4+ T cells and regulatory T cells were purified from week 12 and week 14 PBMC, respectively. 1× 104 (+) or 5 × 104 (++) CD4+ CD25+ cells, or 1.5 × 104 (+) or 7.5 × 104 (++) CD4+ cells were then cocultured with 2 × 105 baseline PBMC in the presence of milk proteins. The proliferation was measured as [3H]thymidine incorporation during the last 16 hours of a 6-day culture.
Evidence for milk specific T cell anergy
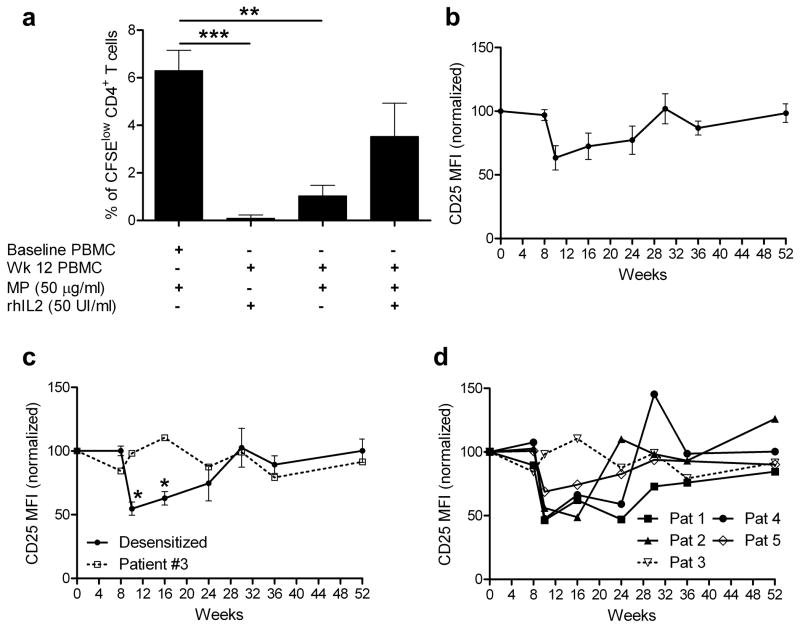
We next examined whether anergy, defined by lack of responsiveness to antigen that can be restored with IL-2 and associated with the absence of suppressive activity 20, could explain the reduced milk-specific T cell proliferative response. First, treatment of milk-activated CD4+ T cells isolated at week 12 with IL-2 partially restored the proliferation of milk-specific CD4+ T cells, although this increase only trended towards statistical significance (p=0.07) (Figure 4a). Furthermore, we found that during the initial desensitization period (weeks 10 and 16), the limited number of CD4+ T cells responding to milk expressed lower levels of CD25 (an activation antigen) compared to that at week 0 (Figure 4b), suggesting that the milk-specific cells at these time points could not be fully activated. In contrast, milk-specific T cells from patient #3, who was only partially desensitized (reacted at the 1,000 mg dose during the double blind placebo controlled food challenge (DBPCFC) at week 24), and who had less of a reduction in milk-induced proliferation at week 10 (Figure 1c), had no reduction in CD25 expression at weeks 10 and 16 compared to week 0 (Figure 4c and d). Moreover, in the one patient in whom PD-1, an antigen expressed by anergic or exhausted T cells 21, 22, was examined (patient #5, who was successfully desensitized), we found that milk-specific CD4+ T cells expressed a significant increase in PD-1 at weeks 10 and 16 (as well as reduced levels of CD25) compared to week 0 (Supplementary Figure S1). These results together suggest that anergy could, at least in part, explain the reduction of milk-specific CD4+ T cell proliferation observed after the rush desensitization phase.
Figure 4. Evidence of milk-specific CD4+ T cell anergy.
(a). PBMC were isolated from week 12 samples of 5 milk allergic patients undergoing milk desensitization protocol, labeled with CFSE, cultured in the presence of milk proteins (MP) for 7 days and treated with or without IL-2. Cells were then collected and stained with anti-CD4 mAb. Proliferation was analyzed by flow cytometry and compared to PBMC proliferation from baseline samples. Figure shows percentages of the milk-specific cells (CFSElow) within the CD4+ gate as mean ± SEM for 5 patients. ** p < 0.01, *** p < 0.001. Significance was determined using paired t-test.
(b–d). Phenotype of the milk specific CD4+ T cells during desensitization. CFSE-labeled PBMC isolated from all 5 of the milk allergic patients undergoing milk desensitization were cultured in the presence of milk proteins for 7 days, then analyzed by flow cytometry.
(b) Data represent CD25 expression (mean CD25 MFI (mean fluorescence intensity)) by milk specific (CD4+ CFSElow) cells ± SEM for the 5 patients evaluated, normalized to results on day 0.
(c) Solid line represents CD25 expression (mean MFI) for milk specific cells from the 4 patients who were desensitized and who passed the DBPCFC, normalized to results on day 0. Dashed line represents CD25 expression (MFI) by milk specific cells from patient #3, who was only partially desensitized, normalized to results on day 0. * p < 0.05 versus baseline and week 8, using paired t-test.
(d) Each line represents CD25 expression (MFI) on milk specific cells from each patient normalized to results on day 0. Patient #3 was only partially desensitized.
Evidence for immune deviation during milk desensitization
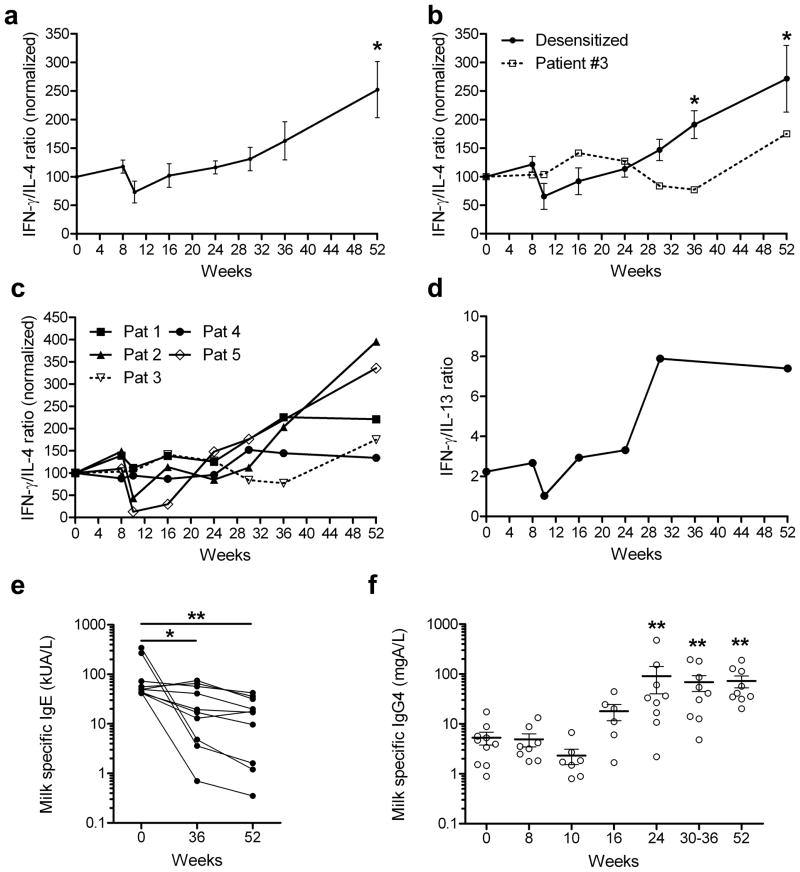
While the cytokine profile of the milk-specific CFSElow CD4+ T cells did not change appreciably during the first 30 weeks, the ratio of IFN-γ/IL-4 production of the milk-specific T cells increased substantially between weeks 36 and 52 (Figure 5a). This increase in the IFN-γ/IL-4 ratio was observed in patients who passed the DBPCFC and were fully desensitized (Figures 5b and c), but was delayed for patient #3 who was only partially desensitized (Figure 5b). IL-13 production in the milk specific CD4+ T cells was examined in one patient (#5), and found to follow IL-4 production, in that the IFN-γ/IL-13 ratio increased after week 30 (Figure 5d). The increase in the IFN-γ/IL-4 ratio in the milk-specific CD4+ T cells suggests that immune deviation developed late during the desensitization, consistent with the significant reduction in milk-specific IgE at weeks 36 and 52 observed in all 10 patients examined (Figure 5e), and by a 15 fold increase in milk-specific IgG4 seen after week 24 in all 10 patients (Figure 5f).
Figure 5. Evidence for immune deviation during milk desensitization.
(a–c) PBMC isolated from 5 milk allergic patients undergoing milk desensitization, labeled with CFSE, and cultured in the presence of antigens for 7 days, were restimulated with PMA and ionomycin in the presence of monensin for 5 h. Cytokine production was then assessed using intracellular IL-4, IFN-γ and IL-13 staining.
(a) Data for all 5 patients. IFN-γ/IL-4 production ratio for the antigen-specific (CFSElow) CD4+ T cells, normalized to day 0 is shown (mean ± SEM).
(b) Solid line represents data from (A), for desensitized patients (patients #1, 2, 4 and 5), and dotted line represents data from (A), for the partially desensitized patient (patient #3). * p < 0.05 versus baseline and week 8, using paired t-test.
(c) Each line represents data for an individual patient, representing IFN-γ/IL-4 production ratio by the antigen-specific CD4+ T cells, normalized to day 0.
(d) IL-13 production was also assessed in CD4+ T cells from patient #5. Data represents IFN-γ/IL-13 production by milk-specific CD4+ T cells.
(e–f) Serum milk-specific IgE (e) and IgG4 (f) levels were determined for the 10 patients who completed the study. Points represent data from different individuals and bars show mean values ± SEM. * p < 0.05, ** p < 0.01 versus baseline, determined using nonparametric Wilcoxon matched-pairs test.
Milk specific basophil activation is reduced by milk desensitization
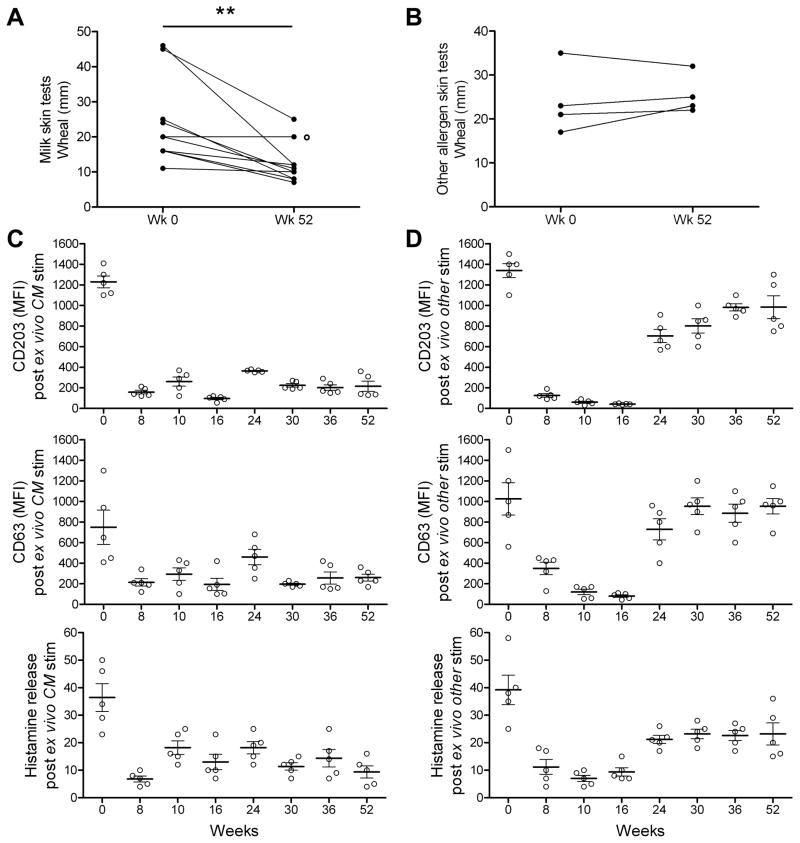
On enrollment, all 10 patients who completed the study had significant immediate skin reactivity to milk (week 0). However, by week 52 of the study, immediate skin prick test responses to cow’s milk were reduced compared week 0 in 9 of the 10 patients (p<0.01) (Figure 6a). In contrast, there was no statistically significant change in immediate skin testing to other allergens (cashew or egg) at week 52 compared to week 0, in the four patients who were skin tested to these other allergens (Figure 6b).
Figure 6. Milk skin test reactivity and in vitro basophil activation are reduced by oral milk desensitization.
(a) Skin prick test wheal with cow’s milk allergen extract (n=10) was reduced at week 52 compared to week 0. ** P < 0.01 determined using paired t-test. ° patient #3, who was slow to become desensitized.
(b) Skin prick test wheal with cashew or ovalbumin did not change, comparing week 0 and week 52 time points in 4 patients known to have those allergies.
(c) Basophils in whole blood, taken on day 0, but not subsequently, when stimulated ex vivo for 20 minutes with cow’s milk (CM), were activated, as assessed by expression of CD203c, CD63 and histamine release (n=5). Treatment with vehicle/glycerine showed no shift in CD203c or CD63 expression (data not shown). MFI = median fluorescence intensity.
(d) Basophil activation with cashew or ovalbumin was blocked by in vivo treatment with omalizumab, as assessed on weeks 8, 9, 10 and 16. Prior to and after omalizumab treatment, basophil activation with cashew or ovalbumin was robust, as assessed by expression of CD203c, CD63 and histamine release.
Similarly, in in vitro studies, we found that cow’s milk antigen, but not vehicle, activated peripheral blood basophils taken on enrollment (week 0), as demonstrated by an increase in basophil expression of CD203c and CD63 (basophil activation markers); in histamine release (p<0.001) (Figure 6c); and in phospho-Syk expression, Supplementary Figure S2). However, treatment of the patients with omalizumab, beginning on week 0 and continuing through week16, greatly reduced milk-induced basophil activation (Figure 6c, Supplementary Figure S2, weeks 8–16). Importantly, the reduction in milk-induced basophil activation persisted after omalizumab was discontinued (weeks 24–52), presumably reflecting a milk-desensitized state, and consistent with the reduction of milk-specific skin test reactivity at week 52 and reduced milk-specific IgE at weeks 36 and 52. The reduction in milk-induced basophil activation at weeks 24–52 was antigen specific, since egg and cashew antigens activated the basophils obtained at weeks 24–52, after discontinuation of omalizumab treatment, as demonstrated by increased antigen-induced expression of CD203c, CD63, increase in histamine release (Figure 6d); and increase in phospho-Syk expression (Supplementary Figure S2). The basophil response to egg and cashew at weeks 24–52 were not quite as robust as at week 0, but were clearly much greater than the very limited basophil responses observed at weeks 8–16, when the patients were being treated with omalizumab. These results together indicated that the mast cell compartment (i.e., mast cells in the skin and basophils in the peripheral blood) was greatly inhibited in an allergen-non-specific fashion during omalizumab treatment, and in an antigen-specific fashion after completion of the oral milk desensitization protocol.
Discussion
Food allergy is a major public health problem that has increased dramatically in prevalence in industrialized countries over the past 10 to 15 years 23, 24. Unfortunately for patients with food allergy, there is no effective treatment except to avoid the offending foods, and to have ready access to self-injectable epinephrine 25. Recently however, several reports indicate that oral desensitization may be effective in patients with allergy to several different foods, including milk, egg, peanut and hazelnut 14–18, 26–30. The protocols for desensitization are diverse, and even with a given protocol, the pace at which the dose of allergen increases varies considerably between patients, due to unpredictable allergic reactions, which confounds the analysis of mechanisms that mediate oral desensitization. As a result, the mechanisms of oral desensitization in patients with food allergy are poorly understood.
In this study, we examined the mechanisms of oral desensitization in a small group of children, who tolerated a novel, relatively rapid and uniform, presumably high dose oral desensitization protocol, enabled with omalizumab, which was used in this setting for the first time 19. After stopping omalizumab, 9 of the children added normal amounts of milk (>8–12 g/day, equivalent to >240–360 ml/day) to their diets 15 to 19 weeks after starting the oral desensitization, and the 10th child was able to consume 1–2 g of milk daily by week 52. Because enrollment in this study was restricted to children with high levels of serum milk-specific IgE (median milk-specific IgE 50 kUA/L, mean 98 kUA/L)), which are associated with milk allergy that is less likely to resolve 31, we believe that the immunological changes observed in these patients are highly significant.
Using CFSE labeling of PBMC in our analysis, we focused on milk-specific CD4+ T cells and found that prior to desensitization, the milk-specific CD4+ T cells were T helper (Th)2-skewed, as expected 9, 11, 12, but that rapid high dose allergen administration greatly but temporarily decreased milk-induced, but not tetanus-induced, CD4+ T cell proliferation. Surprisingly, the acute reduction in milk-specific T cell proliferation in the patients examined was not associated with the development of allergen-specific Foxp3+ TReg cells, nor was there evidence of suppression, as shown by coculture experiments. Moreover, the reduction in milk-specific CD4+ T cell proliferation that occurred during the rapid desensitization period could be partially reversed with the addition of IL-2 to the cultures. This suggested that high dose milk administration resulted initially in the development of milk-specific CD4+ T cell anergy and possibly deletion 18, although additional studies must be performed to fully understand the mechanisms of high dose oral desensitization, particularly because of the limited number of patients analyzed.
Rapid reductions in allergen-specific CD4+ T cell responses have been previously observed in beekeepers exposed to multiple bee stings due to the development of venom specific TReg cells 7. In these subjects, venom-specific IL-10-producing TReg cells developed with repeated bee stings (average cumulative antigen dose over 7 months was <1–4 mg). Similarly, subcutaneous immunotherapy with grass pollen and bee venom, in which the allergen doses are low, ranging from 4–100 μg/dose, as well as with cat allergen peptides, induced allergen-specific tolerance associated with the development of allergen-specific TReg cells producing IL-106, 32–35. There results are consistent with the idea that administration of low doses of antigen during immunotherapy induces allergen-specific TReg cells 8.
In contrast, in our study of oral desensitization, we used much higher doses of antigen (gram amounts daily), and surprisingly, desensitization was not associated with IL-10 producing milk-specific TReg cells. Furthermore, we failed to observe an increase in the number of Foxp3+, natural TReg cells, which are thought to primarily prevent the development of autoimmunity 36. Several studies have shown that the frequency of natural Foxp3+ CD4+ CD25+ TReg cells increased after peanut oral immunotherapy 37, 38, although this was not observed in other studies 39–41. It is possible that in our patients, milk-specific TReg cells and/or natural Foxp3+ CD4+ CD25+ TReg cells were present only in the gastrointestinal tract, or could develop later, e.g., after 12–18 months of daily oral milk maintenance. Alternatively, it is possible that milk-specific TReg cells develop only with low antigen exposure, e.g., induced by exposure to small amounts of milk, and prior to ingestion of full dietary quantities of milk 13.
In our patients, after 4–6 months of daily high doses of oral milk, we found that a vigorous milk-specific CD4+ T cell response returned, but still without evidence of milk-specific Foxp3+ TReg cells. This robust milk-specific T cell response, associated with increased production of IFN-γ, a 15 fold increase in milk-specific IgG4, as well as reduced milk-specific IgE, suggested that high dose oral desensitization evolved into a form of immune deviation. A similar Th1-like allergen-specific response was reported in peanut allergic individuals who had naturally outgrown their sensitivity 12, and in milk-specific Peyer’s patch T cells from non-allergic individuals 42. In those studies, like in our desensitized subjects, the presence of food-specific Foxp3+ TReg cells was also not observed, suggesting that high dose allergen-specific tolerance may not always be associated with allergen-specific Foxp3+ TReg cells. However, is possible that the milk-specific CD4+ T cells producing IFN-γ that we observed may exert inhibitory activity on IgE-mediated milk-induced symptoms through unclear mechanisms 43, 44, particularly since milk-specific IgE was still present after the desensitization in most of our patients. Alternatively, it is possible that milk-specific IgG4 ay play a very dominant role in inhibiting IgE-mediated symptoms and inflammation 3.
Currently, all of our subjects have fully completed the 52 week study, and remain on daily oral milk to maintain their desensitized state. The patients continue to be followed, and over time it is possible that a more permanent tolerant state may develop, such that the capacity to tolerate milk may persist even after oral milk dosing is discontinued 45, perhaps indicating a clinical “cure” of the allergy. However, based on the experience with subcutaneous immunotherapy with inhalant allergens and bee venom, in which long-lived symptom control and allergen “tolerance” is achieved only after completing 3–5 years of “maintenance” therapy 46, 47, complete milk specific immunological “tolerance” may require several years of frequent milk exposure. On the other hand, because the antigen doses with oral desensitization are much greater than that of subcutaneous immunotherapy, it is possible that immunological tolerance might develop sooner with oral immunotherapy, as has been suggested 18, 48.
In summary, we examined milk-specific immune responses in a small number of patients who completed a novel, rapid and uniform high dose oral milk immunotherapy protocol enabled with omalizumab. Within a week of the initial rapid oral desensitization, we observed an acute and significant reduction of milk-specific T cell responses, without evidence of Foxp3+ TReg cell development, suggesting the induction of anergy in, or deletion of, effector T cells. The reduced or anergic response was later replaced by a vigorous CD4+ T cell response, characterized by a shift from IL-4 to greater IFN-γ production, and associated with reduced milk-specific IgE production, reduced milk-specific basophil degranulation, and increased milk-specific IgG4 production. Although our study included a very small number of patients, and requires replication and confirmation in a much larger patient population, the immunological changes observed in the desensitized patients were surprisingly uniform and statistically significant. These results suggest that rapid high dose oral desensitization induces allergen-specific immune deviation not associated with Foxp3+ allergen-specific TReg cells, reminiscent of allergen-specific immunity associated with the natural resolution of food allergy 12. In addition, these results provide an improved understanding of high dose oral desensitization in humans, and could lead to better therapies and diagnostic methods for patients with food allergies.
Methods
Study Population
Eleven patients with a history of significant IgE-mediated milk allergy (defined as having a significant immediate reaction on accidental ingestion of milk, including generalized urticaria, vomiting and/or anaphylaxis) were enrolled in the study at two sites, the Children’s Hospital Boston and at Stanford University School of Medicine. The subjects included 7 boys and 4 girls. 10 of 11 subjects had a past or current history of eczema, asthma, or both, 10 had a history of at least 1 other food allergy, and 6 had 2 or more additional food allergies. The mean age of the patients was 10.2 years (median, 8, range 7–17 years), the median milk-specific IgE was 50 kUA/L (mean 98, range 42 to 342 kUA/L) (normal <0.35) (Phadia ImmunoCAP System), and the median skin prick test (SPT) wheal responses to milk extract was 22 mm (mean 20, range 11–46 mm) 19. The institutional review boards of both institutions approved the clinical protocols, and all participants and/or their parents provided written informed consent. The trial was registered at www.clinicaltrials.gov (NCT00968110).
Clinical protocol
Patients were pre-treated with omalizumab for 9 weeks, allowing IgE to detach from FcεR1 on the surface of mast cells and bind to omalizumab, before starting the oral desensitization to cow’s milk 19. At week 9, the oral desensitization began with 0.1 mg of cow’s milk protein (dried nonfat powdered milk, Carnation Instant Milk, Nestle) mixed in water. There were 12 doses on the first day of desensitization (0.1, 0.5, 1.5, 3, 7, 15, 30, 60, 125, 250, 500, 1,000 mg) given every 30 minutes (cumulative dose 1,992 mg). One subject voluntarily discontinued the study after the 5th oral dose due to vomiting, later diagnosed as abdominal migraines. The day after the rush desensitization, patients returned to start the slower dose escalation, build-up phase, and received the highest dose of milk achieved on the previous day, up to 1,000 mg. Subjects received the same daily dose at home for the next 6 days. The subjects returned weekly for an increase in the oral dose (increase of 12.5%) until a daily oral dose of 2,000 mg was attained (over a period of 7–11 weeks). At week 16, omalizumab treatment was discontinued, while daily oral milk continued. Treatment with omalizumab was extended by 2–4 weeks in 2 of 10 remaining subjects, to allow the milk dose to increase to 2,000 mg. At week 24 (8 weeks after discontinuing the omalizumab), a DBPCFC to milk was conducted. The challenge consisted of 5 doses administered orally every 15 minutes: 500 mg, 750 mg, 1,000 mg, 2,000 mg and 3,000 mg (cumulative dose 7,250 mg). 9 of the 10 remaining subjects passed the DBPCFC, and an open challenge of 4,000–8,000 mg (120 – 240 ml) of milk was given 2–16 h later.
Patient samples
Blood was taken at multiple time points before, during and after the desensitization protocol (week 0, 8, 9, 10, 12, 14, 16, 24, 30, 36 and 52). PBMC were isolated from the blood of 5 patients (at the Children’s Hospital Boston site) by density-gradient separation using Histopaque-1077. The PBMC were frozen in aliquots and stored in liquid nitrogen for later examination. In all experiments, PBMC aliquots from multiple time points for each patient were thawed and studied together so that changes in the phenotype/function of the cells over time could be assessed accurately. Fresh blood samples from 5 patients (at the Stanford site) were used to study basophil function.
CFSE labeling and cell culture
PBMC were labeled with CFSE by incubating PBMC (2 × 107 cells/ml) in RPMI with 2.5 μM CFSE for 8 minutes at 37°C with gentle shaking, then washing excess dye away. CFSE-labeled PBMC were then cultured in the presence of milk proteins (α-casein, β-casein, α-lactalbumin, β-lactoglobulin (Sigma-Aldrich), each at a concentration of 12.5 μg/ml), tetanus toxoid (TT, 0.5 μg/ml) or left unstimulated in RPMI-1640 containing gentamycin and supplemented with 10% AB+ serum and glutamine. Cells were distributed in 48-well flat-bottom plates, at a concentration of 1 × 106/800 μl medium per well. Some of the cultures were also treated with rhIL2 (50 IU/ml). After 7 days of culture, PBMC were collected and stained with PE-conjugated anti-CD4 (RPA-T4; Biolegend), PerCP/Cy5.5-conjugated anti-CD8 (SK1; Biolegend), APC-conjugated anti-CD25 (BC96; Biolegend) and APC-eFluor 780-conjugated anti-CD3 (UCHT1; eBioscience). Some samples were also stained with PerCP/Cy5.5-conjugated anti-CD279 (PD-1) (EH12.2H7; Biolegend), APC-conjugated anti-CD152 (CTLA-4) (L3D10; Biolegend) and APC-Cy7-conjugated anti-HLA-DR (L243; Biolegend).
Detection of cytokine production by flow cytometry
After 7 days of culture with milk proteins, PBMC were restimulated with phorbol myristate acetate (PMA) (25 ng/ml), ionomycin (0.5 μg/ml) and monensin for 5 hours, then fixed and permeabilized with BD Cytofix/Cytoperm Fixation/Permeabilization Kit (BD Biosciences), and stained with PE-conjugated anti-CD4 (RPA-T4, Biolegend), APC-conjugated anti–IL-4 (8D4-8; Biolegend) and Alexa Fluor 700- conjugated anti-IFN-γ (4S.B3; Biolegend). Some cultures were also stained with APC-conjugated anti-IL-10 (JES3-19F1; BD Biosciences) and anti-IL-13 (JES10-5A2; Biolegend). PBMC cultured in the presence of the same antigens, but not restimulated with PMA and ionomycin were used as negative controls.
Detection of regulatory T cells by flow cytometry
After 7 days of culture with milk proteins, PBMC were harvested, fixed and permeabilized with Foxp3 Staining Buffer kit (eBioscience), and stained with PE-conjugated anti-CD4 (RPA-T4, Biolegend) and APC-conjugated anti–FoxP3 (236A/E7; eBioscience).
Study of regulatory T cell function
CFSE-labeled PBMCs were cocultured in the presence of milk proteins and treated or not with anti-IL-10 (JES3-9D7, 5 μg/ml; Biolegend) and/or anti-TGF-β (5 μg/ml) mAbs. In another series of experiments, regulatory T cells and untouched CD4+ T cells were purified from PBMC using the CD4+CD25+ Regulatory T Cell Isolation Kit (Miltenyi Biotec). CD4+ CD25+ cells (1 or 5 × 104 cells) or CD4+ cells (1.5 or 7.5 × 104 cells) were then cocultured with 2 × 105 baseline PBMC in 96-well plates in the presence of milk proteins. The proliferation was measured as [3H]thymidine incorporation during the last 16 hours of a 6-day culture.
Examination of basophil function
Basophil function was studied as previously described 49. Briefly, basophils, identified by gating on at least 500 Live/DeadloCD3−CD16−CD20−CD56−CD66b−HLA-DR−CD123+ cells, were assessed by flow cytometry for changes in expression of CD203 and CD63. Antibodies used were CD3 (UCHT1, Alexa 488), CD41a (96.2C1, Alexa 488), CD66b (G10F5, Alexa 488), CD123 (7G3, Alexa 647) and HLA-DR (L243, Alexa 488), from BD Bio sciences (San Jose, CA), and CD203c (NP4D6, purified) from BioLegend (San Diego, CA). CD203c was custom-conjugated to Pacific Blue (Invitrogen) as per the manufacturer’s protocol. Histamine released into culture supernatants was detected using standardized ELISA methods in quadruplicate.
Prick skin testing
Skin prick tests were conducted via a standardized procedure by a trained, licensed specialist in allergy, using allergens (milk, egg, cashew) from Greer Laboratories.
Determination of serum levels of milk-specific IgE and IgG4
Total milk allergen-specific IgE was quantified in sera collected at time points when patients were not being treated with omalizumab using the ImmunoCAP System (Phadia, Kalamazoo, MI). IgG4 anti-milk was assessed in sera using the UniCAP 100 (Phadia, Kalamazoo, MI) by separately measuring IgG4 antibody levels to the three principal milk allergenic components (casein, α-lactalbumin and β-lactoglobulin). These were summed to provide a measure in μg/ml of IgG4 anti-milk, since IgG anti-milk could not be directly measured in serum using the milk-extract CAP reagent due to the high prevalence of BSA-reactive IgG antibodies in many sera from healthy humans.
Statistics
For most comparisons, we used the paired t-test or 2 way anova for matched values. For immunoglobulin levels (not distributed in a Gaussian manner), we used nonparametric Wilcoxon tests. Differences were considered significant with P <0.05.
Supplementary Material
Samples from Patient #5 were treated and stained as described in Figure 4b (7-day culture in the presence of milk proteins), and further stained with anti-CD152 (PD-1). Data represent PD-1 expression (mean fluorescence intensity, MFI) by milk-specific (CSFElow CD4+) T cells. There was no difference in the expression of MHC class II (HLA-DR) or CTLA-4 at these time points compared to day 0.
Blood basophil phospho-Syk profiling.
(a) Basophils were stimulated with or without (not shown) cow’s milk allergen for 20 minutes (n=5). Basophil phospho-Syk was then quantified by flow cytometry. MFI = median fluorescence intensity.
(b) Basophils were stimulated with control allergens (cashew or ovalbumin) (n=4).
Acknowledgments
Supported by NIH UL1 RR 025758 Clinical and Translational Science Center/Harvard Catalyst; NIH UL1 RR 025744 Stanford University; Children’s Hospital Boston Translational Research Program; the Bunning Food Allergy Project; the Food Allergy Initiative; Stanford Institute of Immunity, Transplantation, and Infectious Diseases; and the Fund for Food Allergy Research at Stanford. DB was supported by a fellowship from the Belgian American Educational Foundation and later by a fellowship from the Thrasher Research Fund. We thank Genentech for generously providing omalizumab for the clinical trial.
Footnotes
Conflict of interest: the authors declare that no conflict of interest exists.
References
- 1.Noon L. Prophylactic inoculation for hay fever. Lancet. 1911;1:1572. [Google Scholar]
- 2.Larche M, Akdis CA, Valenta R. Immunological mechanisms of allergen-specific immunotherapy. Nat Rev Immunol. 2006;6:761–71. doi: 10.1038/nri1934. [DOI] [PubMed] [Google Scholar]
- 3.Jutel M, Akdis CA. Immunological mechanisms of allergen-specific immunotherapy. Allergy. 2011 doi: 10.1111/j.1398-9995.2011.02589.x. in press. [DOI] [PubMed] [Google Scholar]
- 4.Secrist H, Chelen CJ, Wen Y, Marshall JD, Umetsu DT. Allergen immunotherapy decreases interleukin 4 production in CD4+ T cells from allergic individuals. J Exp Med. 1993;178:2123–30. doi: 10.1084/jem.178.6.2123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Varney VA, Hamid QA, Gaga M, Ying S, Jacobson M, Frew AJ, et al. Influence of grass pollen immunotherapy on cellular infiltration and cytokine mRNA expression during allergen-induced late-phase cutaneous responses. J Clin Invest. 1993;92:644–51. doi: 10.1172/JCI116633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Akdis CA, Blesken T, Akdis M, Wuthrich B, Blaser K. Role of interleukin 10 in specific immunotherapy. J Clin Invest. 1998;102:98–106. doi: 10.1172/JCI2250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Meiler F, Zumkehr J, Klunker S, Ruckert B, Akdis CA, Akdis M. In vivo switch to IL-10-secreting T regulatory cells in high dose allergen exposure. J Exp Med. 2008;205:2887–98. doi: 10.1084/jem.20080193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Weiner HL. Oral tolerance for the treatment of autoimmune diseases. Ann Rev Med. 1997;48:341–51. doi: 10.1146/annurev.med.48.1.341. [DOI] [PubMed] [Google Scholar]
- 9.de Jong EC, Spanhaak S, Martens BP, Kapsenberg ML, Penninks AH, Wierenga EA. Food allergen (peanut)-specific TH2 clones generated from the peripheral blood of a patient with peanut allergy. J Allergy Clin Immunol. 1996;98:73–81. doi: 10.1016/s0091-6749(96)70228-2. [DOI] [PubMed] [Google Scholar]
- 10.Higgins JA, Lamb JR, Lake RA, O’Hehir RE. Polyclonal and clonal analysis of human CD4+ T-lymphocyte responses to nut extracts. Immunology. 1995;84:91–7. [PMC free article] [PubMed] [Google Scholar]
- 11.Dorion BJ, Burks AW, Harbeck R, Williams LW, Trumble A, Helm RM, et al. The production of interferon-gamma in response to a major peanut allergy, Ara h II correlates with serum levels of IgE anti-Ara h II. J Allergy Clin Immunol. 1994;93:93–9. doi: 10.1016/0091-6749(94)90237-2. [DOI] [PubMed] [Google Scholar]
- 12.Turcanu V, Maleki SJ, Lack G. Characterization of lymphocyte responses to peanuts in normal children, peanut-allergic children, and allergic children who acquired tolerance to peanuts. J Clin Invest. 2003;111:1065–72. doi: 10.1172/JCI16142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Shreffler WG, Wanich N, Moloney M, Nowak-Wegrzyn A, Sampson HA. Association of allergen-specific regulatory T cells with the onset of clinical tolerance to milk protein. J Allergy Clin Immunol. 2009;123:43–52. e7. doi: 10.1016/j.jaci.2008.09.051. [DOI] [PubMed] [Google Scholar]
- 14.Pajno GB, Caminiti L, Ruggeri P, De Luca R, Vita D, La Rosa M, et al. Oral immunotherapy for cow’s milk allergy with a weekly up-dosing regimen: a randomized single-blind controlled study. Ann Allergy Asthma Immunol. 2010;105:376–81. doi: 10.1016/j.anai.2010.03.015. [DOI] [PubMed] [Google Scholar]
- 15.Buchanan AD, Green TD, Jones SM, Scurlock AM, Christie L, Althage KA, et al. Egg oral immunotherapy in nonanaphylactic children with egg allergy. J Allergy Clin Immunol. 2007;119:199–205. doi: 10.1016/j.jaci.2006.09.016. [DOI] [PubMed] [Google Scholar]
- 16.Nash S, Steele P, Kamilaris J, Pons L, Kulis M, Lee L, et al. Oral peanut immunotherapy for children with peanut allergy. J Allergy Clin Immunol. 2008;121:S147. [Google Scholar]
- 17.Hofmann AM, Scurlock AM, Jones SM, Palmer KP, Lokhnygina Y, Steele PH, et al. Safety of a peanut oral immunotherapy protocol in children with peanut allergy. J Allergy Clin Immunol. 2009;124:286–91. 91, e1–6. doi: 10.1016/j.jaci.2009.03.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Blumchen K, Ulbricht H, Staden U, Dobberstein K, Beschorner J, de Oliveira LC, et al. Oral peanut immunotherapy in children with peanut anaphylaxis. J Allergy Clin Immunol. 2010;126:83–91. e1. doi: 10.1016/j.jaci.2010.04.030. [DOI] [PubMed] [Google Scholar]
- 19.Nadeau K, Schneider L, Hoyte E, Borras I, Umetsu D. Rapid oral desensitization in combination with omalizumab therapy in patients with cow’s milk allergy. J Allergy Clin Immunol. 2011;127:1722–4. doi: 10.1016/j.jaci.2011.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Faria AM, Weiner HL. Oral tolerance. Immunol Rev. 2005;206:232–59. doi: 10.1111/j.0105-2896.2005.00280.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hatachi S, Iwai Y, Kawano S, Morinobu S, Kobayashi M, Koshiba M, et al. CD4+ PD-1+ T cells accumulate as unique anergic cells in rheumatoid arthritis synovial fluid. J Rheumatol. 2003;30:1410–9. [PubMed] [Google Scholar]
- 22.Sharpe AH, Wherry EJ, Ahmed R, Freeman GJ. The function of programmed cell death 1 and its ligands in regulating autoimmunity and infection. Nat Immunol. 2007;8:239–45. doi: 10.1038/ni1443. [DOI] [PubMed] [Google Scholar]
- 23.Sicherer SH, Sampson HA. 9 Food allergy. J Allergy Clin Immunol. 2006;117:S470–5. doi: 10.1016/j.jaci.2005.05.048. [DOI] [PubMed] [Google Scholar]
- 24.Boyce JA, Assa’ad A, Burks AW, Jones SM, Sampson HA, Wood RA, et al. Guidelines for the Diagnosis and Management of Food Allergy in the United States: Summary of the NIAID-Sponsored Expert Panel Report. J Allergy Clin Immunol. 126:1105–18. doi: 10.1016/j.jaci.2010.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sicherer SH, Sampson HA. Food allergy. J Allergy Clin Immunol. 2010;125:S116–25. doi: 10.1016/j.jaci.2009.08.028. [DOI] [PubMed] [Google Scholar]
- 26.Meglio P, Bartone E, Plantamura M, Arabito E, Giampietro PG. A protocol for oral desensitization in children with IgE-mediated cow’s milk allergy. Allergy. 2004;59:980–7. doi: 10.1111/j.1398-9995.2004.00542.x. [DOI] [PubMed] [Google Scholar]
- 27.Longo G, Barbi E, Berti I, Meneghetti R, Pittalis A, Ronfani L, et al. Specific oral tolerance induction in children with very severe cow’s milk-induced reactions. J Allergy Clin Immunol. 2008;121:343–7. doi: 10.1016/j.jaci.2007.10.029. [DOI] [PubMed] [Google Scholar]
- 28.Staden U, Blumchen K, Blankenstein N, Dannenberg N, Ulbricht H, Dobberstein K, et al. Rush oral immunotherapy in children with persistent cow’s milk allergy. J Allergy Clin Immunol. 2008;122:418–9. doi: 10.1016/j.jaci.2008.06.002. [DOI] [PubMed] [Google Scholar]
- 29.Clark AT, Islam S, King Y, Deighton J, Anagnostou K, Ewan PW. Successful oral tolerance induction in severe peanut allergy. Allergy. 2009;64:1218–20. doi: 10.1111/j.1398-9995.2009.01982.x. [DOI] [PubMed] [Google Scholar]
- 30.Enrique E, Pineda F, Malek T, Bartra J, Basagana M, Tella R, et al. Sublingual immunotherapy for hazelnut food allergy: a randomized, double-blind, placebo-controlled study with a standardized hazelnut extract. J Allergy Clin Immunol. 2005;116:1073–9. doi: 10.1016/j.jaci.2005.08.027. [DOI] [PubMed] [Google Scholar]
- 31.Skripak JM, Matsui EC, Mudd K, Wood RA. The natural history of IgE-mediated cow’s milk allergy. J Allergy Clin Immunol. 2007;120:1172–7. doi: 10.1016/j.jaci.2007.08.023. [DOI] [PubMed] [Google Scholar]
- 32.Francis J, Till S, Durham S. Induction of IL-10+CD4+CD25+ T cells by grass pollen immunotherapy. J Allergy Clin Immunol. 2003;111:1255–61. doi: 10.1067/mai.2003.1570. [DOI] [PubMed] [Google Scholar]
- 33.Oldfield W, Larche M, Kay A. Effect of T-cell peptides derived from Fel d 1 on allergic reactions and cytokine production in patients sensitive to cats: a randomised controlled trial. Lancet. 2002;360:47–53. doi: 10.1016/s0140-6736(02)09332-7. [DOI] [PubMed] [Google Scholar]
- 34.Verhoef A, Alexander C, Kay AB, Larche M. T cell epitope immunotherapy induces a CD4+ T cell population with regulatory activity. PLoS Med. 2005;2:e78. doi: 10.1371/journal.pmed.0020078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Aslam A, Chan H, Warrell DA, Misbah S, Ogg GS. Tracking antigen-specific T-cells during clinical tolerance induction in humans. PLoS One. 2010;5:e11028. doi: 10.1371/journal.pone.0011028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Sakaguchi S, Miyara M, Costantino CM, Hafler DA. FOXP3+ regulatory T cells in the human immune system. Nat Rev Immunol. 2010;10:490–500. doi: 10.1038/nri2785. [DOI] [PubMed] [Google Scholar]
- 37.Varshney P, Jones SM, Scurlock AM, Perry TT, Kemper A, Steele P, et al. A randomized controlled study of peanut oral immunotherapy: clinical desensitization and modulation of the allergic response. J Allergy Clin Immunol. 2011;127:654–60. doi: 10.1016/j.jaci.2010.12.1111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Jones SM, Pons L, Roberts JL, Scurlock AM, Perry TT, Kulis M, et al. Clinical efficacy and immune regulation with peanut oral immunotherapy. J Allergy Clin Immunol. 2009;124:292–300. e1–97. doi: 10.1016/j.jaci.2009.05.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Mori F, Bianchi L, Pucci N, Azzari C, De Martino M, Novembre E. CD4+CD25+Foxp3+ T regulatory cells are not involved in oral desensitization. Int J Immunopathol Pharmacol. 23:359–61. doi: 10.1177/039463201002300136. [DOI] [PubMed] [Google Scholar]
- 40.Kim EH, Bird JA, Kulis M, Laubach S, Pons L, Shreffler W, et al. Sublingual immunotherapy for peanut allergy: clinical and immunologic evidence of desensitization. J Allergy Clin Immunol. 2011;127:640–6. e1. doi: 10.1016/j.jaci.2010.12.1083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Tiemessen MM, Van Hoffen E, Knulst AC, Van Der Zee JA, Knol EF, Taams LS. CD4 CD25 regulatory T cells are not functionally impaired in adult patients with IgE-mediated cow’s milk allergy. J Allergy Clin Immunol. 2002;110:934–6. doi: 10.1067/mai.2002.128856. [DOI] [PubMed] [Google Scholar]
- 42.Nagata S, McKenzie C, Pender SL, Bajaj-Elliott M, Fairclough PD, Walker-Smith JA, et al. Human Peyer’s patch T cells are sensitized to dietary antigen and display a Th cell type 1 cytokine profile. J Immunol. 2000;165:5315–21. doi: 10.4049/jimmunol.165.9.5315. [DOI] [PubMed] [Google Scholar]
- 43.Stock P, Kallinich T, Akbari O, Quarcoo D, Gerhold K, Wahn U, et al. CD8(+) T cells regulate immune responses in a murine model of allergen-induced sensitization and airway inflammation. Eur J Immunol. 2004;34:1817–27. doi: 10.1002/eji.200324623. [DOI] [PubMed] [Google Scholar]
- 44.Dominguez-Villar M, Baecher-Allan CM, Hafler DA. Identification of T helper type 1-like, Foxp3(+) regulatory T cells in human autoimmune disease. Nat Med. 2011;17:673–5. doi: 10.1038/nm.2389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Scurlock AM, Vickery BP, Hourihane JO, Burks AW. Pediatric food allergy and mucosal tolerance. Mucosal Immunol. 3:345–54. doi: 10.1038/mi.2010.21. [DOI] [PubMed] [Google Scholar]
- 46.van Halteren HK, van der Linden PW, Burgers JA, Bartelink AK. Discontinuation of yellow jacket venom immunotherapy: follow-up of 75 patients by means of deliberate sting challenge. J Allergy Clin Immunol. 1997;100:767–70. doi: 10.1016/s0091-6749(97)70271-9. [DOI] [PubMed] [Google Scholar]
- 47.Cox L, Cohn JR. Duration of allergen immunotherapy in respiratory allergy: when is enough, enough? Ann Allergy Asthma Immunol. 2007;98:416–26. doi: 10.1016/S1081-1206(10)60755-7. [DOI] [PubMed] [Google Scholar]
- 48.Varshney P, Jones S, Pons L, Kulis M, Steele P, Kemper A, et al. Peanut Oral Immunotherapy (OIT) induces immunologic changes supporting the development of tolerance. J Allergy Clin Immunol. 2010;125:AB59 (Abstract). [Google Scholar]
- 49.Gernez Y, Tirouvanziam R, Yu G, Ghosn EE, Reshamwala N, Nguyen T, et al. Basophil CD203c Levels Are Increased at Baseline and Can Be Used to Monitor Omalizumab Treatment in Subjects with Nut Allergy. Int Arch Allergy Immunol. 2010;154:318–27. doi: 10.1159/000321824. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Samples from Patient #5 were treated and stained as described in Figure 4b (7-day culture in the presence of milk proteins), and further stained with anti-CD152 (PD-1). Data represent PD-1 expression (mean fluorescence intensity, MFI) by milk-specific (CSFElow CD4+) T cells. There was no difference in the expression of MHC class II (HLA-DR) or CTLA-4 at these time points compared to day 0.
Blood basophil phospho-Syk profiling.
(a) Basophils were stimulated with or without (not shown) cow’s milk allergen for 20 minutes (n=5). Basophil phospho-Syk was then quantified by flow cytometry. MFI = median fluorescence intensity.
(b) Basophils were stimulated with control allergens (cashew or ovalbumin) (n=4).