Abstract
Heart failure (HF) is characterized by the inability of systemic perfusion to meet the body's metabolic demands and is usually caused by cardiac pump dysfunction and may occasionally present with symptoms of a noncardiac disorder such as hepatic dysfunction. The primary pathophysiology involved in hepatic dysfunction from HF is either passive congestion from increased filling pressures or low cardiac output and the consequences of impaired perfusion. Passive hepatic congestion due to increased central venous pressure may cause elevations of liver enzymes and both direct and indirect serum bilirubin. Impaired perfusion from decreased cardiac output may be associated with acute hepatocellular necrosis with marked elevations in serum aminotransferases. Cardiogenic ischemic hepatitis (“shock liver”) may ensue following an episode of profound hypotension in patients with acute HF. We discuss pathophysiology and identification of liver abnormalities that are commonly seen in patients with HF.
Keywords: Heart failure, liver dysfunction, cardiovascular disease, pulsatile liver, hepatic congestion, shock liver, cardiac output
Heart failure (HF) is a clinical syndrome characterized by the inability of systemic perfusion to meet the body's metabolic demands and is usually caused by cardiac pump dysfunction. HF is subdivided into systolic and diastolic HF. Systolic failure presents reduced cardiac contractility whereas diastolic failure exhibits impaired cardiac relaxation with abnormal ventricular filling. HF can result from several structural or functional congenital and acquired cardiac disorders that impairs the ability of the ventricle to fill with or eject blood.1 Clinically, HF may present with a syndrome of decreased exercise tolerance due to dyspnea and/or fatigue related to impaired cardiac output or may present with a syndrome of fluid retention from elevated filling pressure.2 A spectrum of hepatic derangements can also occur in HF particularly in the setting of right heart failure (RHF). Any cause of right ventricular dysfunction can be associated with severe hepatic congestion; patients with hepatic congestion are usually asymptomatic and this entity may be suggested only by abnormal liver function tests (LFTs) during routine laboratory analysis. The primary pathophysiology involved in hepatic dysfunction is either passive congestion from increased filling pressures or low cardiac output and the consequences of impaired perfusion. Passive hepatic congestion due to increased central venous pressure (CVP) may cause elevations of liver enzymes and both direct and indirect serum bilirubin. Impaired perfusion from decreased cardiac output may be associated with acute hepatocellular necrosis with marked elevations in serum aminotransferases. Cardiogenic ischemic hepatitis (“shock liver”) may ensue following an episode of profound hypotension in patients with acute HF. Bridging fibrosis or cardiac cirrhosis can result from prolonged hemodynamic abnormalities, resulting in an impaired hepatic function with impaired coagulation, decreased albumin synthesis, and alteration in the metabolism of several cardiovascular drugs, which can lead to unwanted toxicity. Dosage adjustments are necessary in some of these agents, but precise guidelines for dosing are difficult because unlike in renal impairment, alterations in hepatic drug disposition do not always correlate well with routine laboratory markers of liver dysfunction. In this article we discuss pathophysiology and identification of liver abnormalities that are seen in patients with HF. Further research into the complex relationship between cardiac and hepatic function in HF may improve our understanding of the pathophysiology of the disease process and improve clinical care of HF patients.
PATHOPHYSIOLOGY AND HISTOPATHOLOGY
Hepatic dysfunction due to passive congestion is particularly common in patients with right-sided HF with elevated right ventricular (RV) pressure. Any cause of right-sided HF can result in hepatic congestion, including constrictive pericarditis, severe pulmonary arterial hypertension (PAH), mitral stenosis, tricuspid regurgitation (TR), cor pulmonale, cardiomyopathy, and as a postoperative consequence of the Fontan procedure for pulmonary atresia and the hypoplastic left heart syndrome. TR is particularly prone to result in passive congestion because pressure from the RV is transmitted directly to the hepatic veins and sinusoids.3 This increase in venous pressure caused by RV dysfunction leads to atrophy of the hepatocytes and causes perisinusoidal edema that can impair diffusion of oxygen and nutrients to the hepatocytes.4,5 As a result from this hepatic congestion, mild jaundice, abnormalities in liver enzymes, and derangements in hepatic drug metabolisms ensues. On gross examination the congestive liver is enlarged, with a purple or reddish hue with prominent hepatic veins. The cut surface shows the classic nutmeg appearance, reflecting the alternating pattern of hemorrhage and necrosis of zone 3 with the normal or slightly steatotic areas in zones 1 and 2. Microscopically, the hallmark features of hepatic venous hypertension are prominence of the central veins, central vein hemorrhage, and sinusoidal engorgement.3,6,7 Untreated, long-standing congestion can lead to cardiac fibrosis and, ultimately cardiac cirrhosis.8 In contrast, low cardiac output (forward failure) is associated with some degree of perfusion abnormality that is not necessarily evident. Acute hypoxic hepatitis most commonly arises in the context of profound systemic hypotension from acute cardiopulmonary collapse after myocardial infarction, exacerbation of HF, or pulmonary embolism. In the absence of established hypotension ischemic hepatitis has been shown in instances of severe hypoxemia, such as obstructive sleep apnea, respiratory failure, and in conditions of increased metabolic demand, as seen in toxic/septic shock.9,10,11 The term hepatitis is misnomer because histological evidence of inflammation is absent. Instead, ischemic liver injury is characterized by centrolobular necrosis of zone 3 hepatocytes.12,13,14,15,16,17 Oxygen consumption can be easily increased when hepatic blood flow is decreased. The mechanism by which the liver protects itself from damage in hypoxia is increasing oxygen extraction by the hepatocytes up to 95% as the blood passes through the liver. When inadequate end-organ perfusion and tissue hypoxia is persistent or when acute shock develops this protecting mechanism against hypoxic liver damage is overwhelmed. Hepatocellular injury ensues, accompanied by a sharp elevation of the serum alanine aminotransferase (ALT), aspartate aminotransferase (AST), lactic dehydrogenase (LDH), prolongation of the prothrombin time, and occasionally functional renal impairment. These abnormalities reach their peak 1 to 3 days after the onset of cardiogenic ischemic hepatitis and return to normal within 5 to 10 days from onset of the disorder.
CLINICAL SYMPTOMS AND FINDINGS
Patients with backward HF may present signs and symptoms that can be divided into left- and right-sided. Left-sided HF is associated with symptoms that are predominantly respiratory in nature. Dyspnea on exertion, orthopnea, nocturnal paroxysmal dyspnea, wheezing, and easy fatigability are the most common complaints. Right-sided HF on the other hand is mainly associated with peripheral edema, ascites, and hepatomegaly. Only a few patients experience mild, dull right-upper quadrant pain that is caused by stretching of the liver capsule. Hepatomegaly with a firm, tender liver edge and peripheral edema are typically the most prominent finding in patients with chronic right-sided HF, but it may also occur rapidly in acute HF. Ascites may be present in up to 25% of these patients, but even in the presence of ascites and lower extremity edema, splenomegaly is characteristically absent.18 Jaundice is not commonly reported. In patients with considerable TR, a prominent systolic pulsation of the liver, attributable to an enlarged right atrial V wave, is often noted. A presystolic pulsation of the liver, attributable to an enlarged right atrial A wave, can occur in tricuspid stenosis, constrictive pericarditis, restrictive cardiomyopathy involving the RV, and pulmonary hypertension (primary or secondary). Ischemic hepatitis is usually benign and self-limited. Clinical diagnosis of liver injury is almost always incidental when liver enzymes are found to be massively elevated 1 to 3 days after an episode of systemic hypotension. The effects of systemic hypoperfusion are not isolated to the liver, increased in creatinine level from acute tubular necrosis is nearly universal in the clinical course. Although, there are not unique clinical and physical examination findings for ischemic hepatitis, patients may present with symptoms of nausea, vomiting, anorexia, malaise, right-upper quadrant pain, jaundice, oliguria, and flapping tremor representing cerebral hypoperfusion rather hepatic encephalopathy.
LABORATORY ABNORMALITIES
Congestive hepatomegaly is often associated with impaired hepatic function; biochemical parameters of liver function are moderately elevated 2 to 3 times the upper normal reference level. These parameters include AST, ALT, LDH, gamma-glutamyl transpeptidase (GGT), and alkaline phosphatase (ALP). Hyperbilirubinemia, secondary to an increase in both the directly and indirectly reacting bilirubin, is also common. Total bilirubin level is rarely greater than 3 mg/dL. Impaired perfusion from low cardiac output results in cardiogenic ischemic hepatitis. The hallmark findings of ischemic hepatitis are severe jaundice, with a bilirubin level as high as 15 to 20 mg/dL, elevation of AST to more than 10 times the upper reference range limit, marked increase in serum LDH, elevated ALP level, and prolongation of the prothrombin time. Increases in LDH tend to be massive and ALT/LDH ratio of less than 1.5 help distinguishing ischemic injury from other forms of acute hepatitis.19,20 Increases in international normalized ratio (INR) is not common, yet can be seen in cases of severe ischemic injury. Both the clinical and the laboratory pictures may resemble acute viral hepatitis, but the impairment of hepatic function is rapidly resolved by successful treatment of HF. In patients with long-standing HR, albumin synthesis may be impaired, leading to hypoalbuminemia and intensifying the accumulation of fluid. Table 1 summarizes the key laboratory abnormalities encountered in patients with HF based on the primary mechanism.
Table 1.
Key Laboratory Abnormalities Encountered in Patients with HF Based on the Primary Mechanism
| Laboratory Parameter | Elevated Filling Pressure | Low Cardiac Output |
|---|---|---|
| AST | -/↑ | ↑↑ |
| ALT | -/↑ | ↑/↑↑ |
| Bilirubin | -/↑ | ↑/↑↑ |
| GGT | ↑↑ | -/↑ |
| ALP | ↑/↑↑ | -/↑ |
| LDH | ↑/↑↑ | -/↑↑ |
HF, heart failure, AST, aspartate aminotransferase; ALT, alanine aminotransferase; GGT, gamma-glutamyl transpeptidase; ALP, alkaline phosphatase; LDH, lactate dehydrogenase.
AVAILABLE DATA
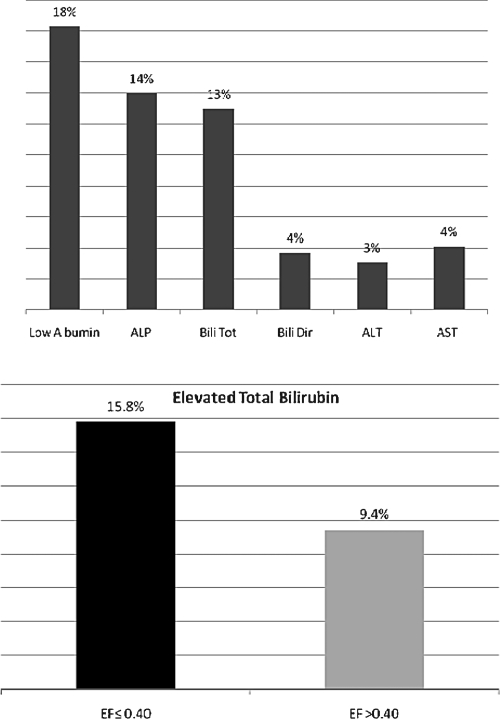
Allen et al characterized liver function tests of 2679 patients with symptomatic chronic HF in the Candesartan in Heart Failure: Assessment of Reduction in Mortality and Morbidity (CHARM) program.21 They used multivariable modeling to assess the relationships between baseline liver function values and long-term outcomes. In their analysis, liver function test abnormalities were common in patients with chronic HF, ranging from ALT elevation in 3.1% of patients to low albumin in 18.3% of patients; total bilirubin was elevated in 13% of patients (Fig. 1). The proportion of patients with impaired left ventricular dysfunction (ejection fraction ≤0.40, n = 1594) who had elevations in total bilirubin (15.8%) was almost double that of patients with preserved left ventricular dysfunction (ejection fraction >0.40, n = 1085; elevated total bilirubin in 9.4%; p < 0.001 for the comparison).
Figure 1.
Frequency of liver function test abnormalities in patients with HF and bilirubin elevation in relation to the ejection fraction.
In multivariable analysis, elevated total bilirubin was the strongest predictor of adverse outcome for both the composite outcome of cardiovascular death or HF hospitalization (HR 1.21 per 1 SD increase, p < 0.0001) and all-cause mortality (HR 1.19 per 1 SD increase, p < 0.0001). Even after adjustment for other variables, elevated total bilirubin remained one of the strongest independent predictors of poor prognosis (by global chi-square). Shinagawa et al similarly suggested that increased bilirubin predicts a worse long-term prognosis of HF, presumably through the potential liability to both congestion and tissue hypoperfusion simultaneously when HF deteriorates.22
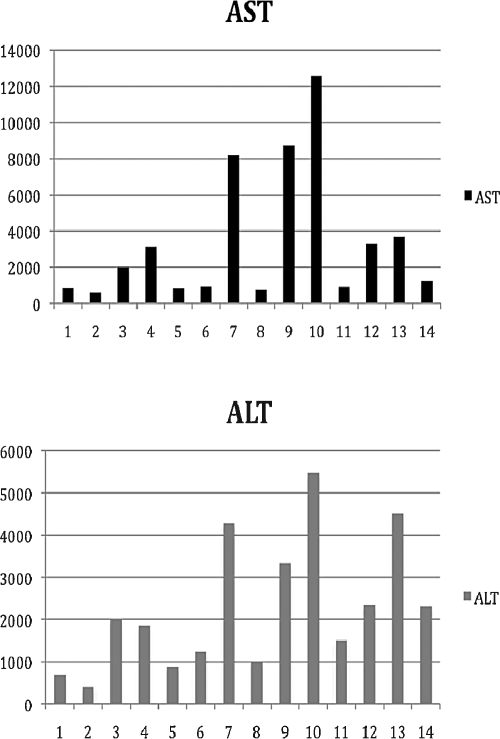
Denis et al described a series of 14 consecutive patients admitted to the intensive care unit of the hepatology department of their hospital for suspected fulminant hepatitis.23 Only six of these patients had history of cardiac disease. In addition to elevation of transaminases, AST and ALT (even more than 100-fold) as shown in Fig. 2, electrocardiographic (ECG) abnormalities were found in all patients and most of them (93%, n = 13) had echocardiographic findings. Elevation of transaminases may not only be due to acute viral or toxic hepatitis, but also due to acute hypoxic hepatitis, and the latter diagnosis should be seriously considered in patients with cardiac disease. Normal ECG virtually eliminates the diagnosis of acute hypoxic hepatitis, whereas echocardiography reveals the diagnosis.
Figure 2.
Liver function abnormalities in patients with acute hypoxic hepatitis.
A retrospective chart review done by van Deursen et al demonstrated that levels of liver function abnormalities increased with decreasing cardiac index (CI; CI = cardiac output divided by the body surface area) and increasing CVP.24 Most liver parameters of liver function were predominantly related to increased CVP, particularly elevated GGT and direct bilirubin, whereas only AST, ALT, and total bilirubin were related to a reduced CI. As these abnormalities often coexist, this finding seems to be a reflection of the pathophysiology of liver function abnormalities in HF being a combination of both congestion and reduced cardiac output.
To investigate the type of liver dysfunction in HF and its relation to the severity of TR, the records of all patients admitted to the cardiology unit of a tertiary referral hospital with a diagnosis of left, right, or congestive HF during a 16-month period were examined by Lau et al.25 There were 72 men and 38 women (aged between 41 and 72 years) studied. The most common abnormalities were elevation of the cholestatic liver enzyme profile, which includes ALP, GGT, bilirubin, and hypoalbuminemia; each of these was significantly more common (all p < 0.001) than the increase of a hepatic profile (increased ALT and AST). The elevation of each of the cholestatic LFTs was significantly associated with the degree of tricuspid incompetence. Patients with moderate or severe TR had significantly greater ALP, GGT, and bilirubin than those with no or mild TR. Differentiating moderate and severe TR from lesser grades can be considered clinically and pathologically useful. Table 2 summarizes the changes on LFTs seen in patients with moderate to severe TR.25
Table 2.
Effect of TR on LFTs
| LFT (Reference Range) | ALP (30–130 U/L) | GGT (11–60 U/L) | ALT (0–60 U/L) | AST (14–50 U/L) | Bilirubin (0–20 mmol/L) | Albumin (33–47 g/L) | |
|---|---|---|---|---|---|---|---|
| No TR (n = 29) | 94 ± 5 | 58 ± 10 | 31 ± 4 | 26 ± 2 | 15 ± 1 | 32 ± 1 | |
| Mild TR (n = 52) | 105 ± 5 | 50 ± 5 | 28 ± 2 | 26 ± 2 | 14 ± 1 | 34 ± 1 | |
| Moderate TR (n = 22) | 136 ± 15 | 110 ± 20 | 26 ± 3 | 24 ± 2 | 20 ± 2 | 35 ± 1 | |
| Severe TR (n = 7) | 154 ± 18 | 158 ± 40 | 21 ± 3 | 25 ± 2 | 21 ± 3 | 25 ± 2 | |
| p = 0.000 | p = 0.001 | p = 0.187 | p = 0.944 | p = 0.012 | p = 0.118 | ||
TR, tricuspid regurgitation; LFTs, liver function tests; ALP, alkaline phosphatase; GGT, gamma-glutamyl transpeptidase; ALT, alanine aminotransferase; AST, aspartate aminotransferase.
Some degree of liver dysfunction is also seen in patients who underwent Fontan procedure for tricuspid or pulmonary atresia, and hypoplastic left heart syndrome. Between December 2003 and September 2004, Camposilvan et al26 established the characteristic pattern of liver dysfunction in patients who had previously undergone a Fontan procedure at their institution and were attending the follow-up program. A total of 34 patients were enrolled in the study. Of these 34, 20 were males and 14 females; the median age at assessment was 14.7 years (range, 4.1 to 26.7). The rationale for the Fontan procedure rests on the premise that systemic venous blood can pass through the lungs without the assistance of a pumping RV. But of the consequences of the Fontan procedure, is a significantly increased in systemic venous pressure that chronically leads to hepatic congestion and its associated cholestatic hepatopathy.26 Reports of laboratory test are reported in Table 3.
Table 3.
Influence of Fontan Procedure on LFTs
| Number of Patients Tested | AST (15–40 U/L) 33 | ALT (5–40 U/L) 33 | GGT (3–40 U/L) 31 | Bilirubin (1.7–20 μmol/L) 29 | Albumin (35–46 g/L) 28 |
|---|---|---|---|---|---|
| Median (range) | 35 (20–69) | 31 (13–62) | 44 (3–184) | 17 (1–32) | 45 (16.6–64) |
| Number abnormal (%) | 6 (18) | 8 (24) | 19 (61) | 10 (32) | 10 (36) |
LFTs, liver function tests; AST, aspartate aminotransferase; ALT, alanine aminotransferase; GGT, gamma-glutamyl transpeptidase; ALP, alkaline phosphatase.
A particularly challenging subset of patients may be those with HF related to constrictive heart disease.27 Cryptogenic cirrhosis is a relatively common presentation of constrictive pericarditis, and primary liver disease is initially mistakenly diagnosed in many such patients. Occasionally, these patients undergo liver biopsy and histological findings are usually nonspecific with diffuse centrilobular congestion, necrosis and fibrosis, or patchy fibrosis without congestion.28 Patients with appreciable sinusoidal dilatation and hemorrhagic necrosis together with hepatic venular thrombus may be misdiagnosed as Budd-Chiari syndrome.29,30 Clinical clues to the correct diagnosis include elevated jugular venous pressure and pericardial knock in constrictive pericarditis. The pericardial knock, characteristic of constriction, occurs earlier in diastole and is of higher frequency than the third heart sound heard in restrictive cardiomyopathy.
DRUG TREATMENT OF HF IN HEPATIC DYSFUNCTION
The liver plays a central role in the absorption, distribution, and elimination of the majority of drugs. Drugs are biotransformed in the liver by enzymatic conversion into active, inactive, or even toxic metabolites. Hepatic impairment can alter the pharmacokinetic profiles of cardiovascular drugs, which can lead to unwanted toxicity. Therefore, dosage adjustments are necessary in some of these agents. Levels and effects for an individual drug are unpredictable and do not correlate well with the type of liver injury, its severity, or liver function test results. Thus, no general rules are available for modifying drug dosage in patients with liver disease.
Angiotensin-Converting Enzyme (ACE) Inhibitors
Angiotensin-converting enzyme (ACE) inhibitors are the first-line therapy in the therapy of HF. They increase cardiac output and decrease LV filling pressure due to their vasodilatory effect. Some of the ACE inhibitors are prodrugs, which require transformation by the liver into active metabolites. These drugs include enalapril, ramipril, fosinopril, trandolapril, quinapril, benazepril, and moexipril. With liver dysfunction, decreases in prodrug transformation and inactivation of active drug may occur. Usual dose with frequent monitoring is recommended for these compounds.31 Lisinopril undergoes no metabolism; therefore, dosage adjustments are unnecessary. Although the liver metabolizes captopril, no particular considerations are required for its use in hepatic impairment.32
Angiotensin II Receptor Blockers (ARB)
As with ACE inhibitors, angiotensin II receptor blockers (ARBs) have shown to reduce morbidity and mortality in patients with systolic HF. The primary reason to use an ARB instead of an ACE inhibitor is to avoid the side effect of cough. Losartan undergoes extensive first-pass metabolism with an oral bioavailability of ~33%. It is metabolized to the active metabolite via hepatic carboxylation. In patients with hepatic impairment, the bioavailability is double and the total plasma clearance is halved. Therefore, lower initial doses are recommended. Valsartan undergoes little metabolic conversion. Caution is recommended in patients with mild to moderate liver dysfunction but dosage adjustments are generally not needed. Similar to valsartan, irbesartan does not require biotransformation, thus dosage modification is not necessary.33,34,35,36,37
Beta-Blockers
Beta-blockers are now part of the standard therapy for HF. The use of β-blocker is associated with 30% reduction in total mortality. Propanolol goes through extensive hepatic first-pass metabolism; hence it should be administered cautiously in patients with hepatic impairment. A significant decrease of clearance and increase in half-life of propanolol has been noticed. No dose adjustments are necessary for atenolol, nadolol, esmolol, sotalol, and acebutalol.38,39,40
Diuretics
Loop diuretics, such as furosemide, bumetanide, and torsemide, are used for volume management in HF because of their superior natriuretic effects compared with other classes of diuretics. Loop diuretics are metabolized mostly hepatically but extrahepatic component by renal glucuronidation has been also identified. For unclear reasons, the pharmacologic response in patients with liver dysfunction and HF is diminished, and there is a net decreased in sodium excretion when compared with healthy individuals taking the same dose. No adjustments are necessary if renal function is normal.41
Digoxin
Digitalis glycosides are indicated for the therapy of mild to moderate HF. Their inotropic action results in the improvement of cardiac output and improvement of signs and symptoms of hemodynamic insufficiency. Digoxin is not hepatically metabolized, but in patients with hepatic impairments the volume of distribution may be reduced; thus lower doses are necessary to avoid toxicity.
Warfarin
Although data are limited, warfarin may be used in the prevention of thromboembolic events in patients with HF due to systolic dysfunction. Warfarin is metabolized by hepatic cytochrome P-450 to inactive hydroxylated metabolites, which are excreted in the bile. In patients with hepatic dysfunction the response to warfarin is potentiated through impaired synthesis of clotting factors and decreased liver metabolism. Initial warfarin dose should be less than or equal to 5 mg and adjusted based on the INR. Careful monitoring is recommended.
Amiodarone
In patients with severe HF, amiodarone has proven to be effective for suppressing ventricular arrhythmias, reducing sudden death and cardiac mortality, and improving exercise tolerance and ejection fraction. It undergoes extensive hepatic metabolism to active metabolite, but no dosage reduction is indicated in hepatic impairment.42,43
Statins
The 3-hydroxy-3-methylglutaryl-coenzyme A (HMG CoA) reductase inhibitor therapy reduces the risks of all-cause mortality and hospitalization among patients with chronic HF. All HMG CoA-reductase inhibitor undergo extensive hepatic metabolism. In patients with active liver disease or persistent unexplained elevations in serum transaminases to above 3 times the upper limit of normal, the use of statins is contraindicated as they may worsen liver function.44
CLINICAL IMPLICATIONS
These studies have several important implications for practicing clinicians. First, elevated liver enzymes with negative viral serology should make the clinician suspect HF as a possibility and proceed with appropriate clinical work-up. ECG and Doppler echocardiography should therefore be undertaken routinely in every patient in whom a diagnosis of fulminant hepatitis is suspected, in particular before liver biopsy. Elevated liver enzymes, particularly bilirubin, appears to portend a poor prognosis and there is some evidence that increased bilirubin coincident with HF decompensation can be a useful marker indicating the need for intravenous inotropic agent administration.45 AST and ALT is typically mildly elevated in patients with elevated filling pressure and passive congestion but more markedly elevated in low output states most likely related to hepatic hypoxia. LDH is less specific than AST and ALT as a marker of hepatocyte injury, but LDH may be disproportionately elevated after an ischemic liver injury. Finally, patients with restrictive-constrictive HF may present with hepatosplenomegaly and ascites suggestive of primary liver disease, and prior to potentially hazardous investigations, such as liver biopsy, a thorough physical examination may point to the correct diagnosis.27 We hope that further research and better characterization of hepatic dysfunction in HF may improve our understanding of the pathophysiology of the disease process and ultimately improve clinical care of HF patients. For now, individuals with marked hepatic dysfunction in the setting of HF should be considered particularly high risk and treated aggressively.
References
- Hunt S A, American College of Cardiology. American Heart Association Task Force on Practice Guidelines (Writing Committee to Update the 2001 Guidelines for the Evaluation and Management of Heart Failure) ACC/AHA 2005 guideline update for the diagnosis and management of chronic heart failure in the adult: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to update the 2001 Guidelines for the Evaluation and Management of Heart Failure) J Am Coll Cardiol. 2005;46(6):e1–e82. doi: 10.1016/j.jacc.2005.08.022. [DOI] [PubMed] [Google Scholar]
- Jessup M, Abraham W T, Casey D E, et al. 2009 focused update: ACCF/AHA Guidelines for the diagnosis and management of heart failure in adults: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines: developed in collaboration with the International Society for Heart and Lung Transplantation. Circulation. 2009;119(14):1977–2016. doi: 10.1161/CIRCULATIONAHA.109.192064. [DOI] [PubMed] [Google Scholar]
- Sherlock S. The liver in heart failure; relation of anatomical, functional, and circulatory changes. Br Heart J. 1951;13(3):273–293. doi: 10.1136/hrt.13.3.273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Safran A P, Schaffner F. Chronic passive congestion of the liver in man. Electron microscopic study of cell atrophy and intralobular fibrosis. Am J Pathol. 1967;50(3):447–463. [PMC free article] [PubMed] [Google Scholar]
- Dunn G D, Hayes P, Breen K J, Schenker S. The liver in congestive heart failure: a review. Am J Med Sci. 1973;265(3):174–189. doi: 10.1097/00000441-197303000-00001. [DOI] [PubMed] [Google Scholar]
- Safran A P, Schaffner F. Chronic passive congestion of the liver in man. Electron microscopic study of cell atrophy and intralobular fibrosis. Am J Pathol. 1967;50(3):447–463. [PMC free article] [PubMed] [Google Scholar]
- Lefkowitch J H, Mendez L. Morphologic features of hepatic injury in cardiac disease and shock. J Hepatol. 1986;2(3):313–327. doi: 10.1016/s0168-8278(86)80043-5. [DOI] [PubMed] [Google Scholar]
- Weisberg I S, Jacobson I M. Cardiovascular diseases and the liver. Clin Liver Dis. 2011;15(1):1–20. doi: 10.1016/j.cld.2010.09.010. [DOI] [PubMed] [Google Scholar]
- Henrion J, Minette P, Colin L, Schapira M, Delannoy A, Heller F R. Hypoxic hepatitis caused by acute exacerbation of chronic respiratory failure: a case-controlled, hemodynamic study of 17 consecutive cases. Hepatology. 1999;29(2):427–433. doi: 10.1002/hep.510290202. [DOI] [PubMed] [Google Scholar]
- Mathurin P, Durand F, Ganne N, et al. Ischemic hepatitis due to obstructive sleep apnea. Gastroenterology. 1995;109(5):1682–1684. doi: 10.1016/0016-5085(95)90659-2. [DOI] [PubMed] [Google Scholar]
- Henrion J, Schapira M, Luwaert R, Colin L, Delannoy A, Heller F R. Hypoxic hepatitis: clinical and hemodynamic study in 142 consecutive cases. Medicine (Baltimore) 2003;82(6):392–406. doi: 10.1097/01.md.0000101573.54295.bd. [DOI] [PubMed] [Google Scholar]
- Ellenberg M, Osserman K E. The role of shock in the production of central liver cell necrosis. Am J Med. 1951;11(2):170–178. doi: 10.1016/0002-9343(51)90102-7. [DOI] [PubMed] [Google Scholar]
- Killip T, III, Payne M A. High serum transaminase activity in heart disease. Circulatory failure and hepatic necrosis. Circulation. 1960;21:646–660. doi: 10.1161/01.cir.21.5.646. [DOI] [PubMed] [Google Scholar]
- Clarke W T. Centrilobular hepatic necrosis following cardiac infarction. Am J Pathol. 1950;26(2):249–255. [PMC free article] [PubMed] [Google Scholar]
- Giallourakis C C, Rosenberg P M, Friedman L S. The liver in heart failure. Clin Liver Dis. 2002;6(4):947–967. viii–ix. doi: 10.1016/s1089-3261(02)00056-9. [DOI] [PubMed] [Google Scholar]
- Seeto R K, Fenn B, Rockey D C. Ischemic hepatitis: clinical presentation and pathogenesis. Am J Med. 2000;109(2):109–113. doi: 10.1016/s0002-9343(00)00461-7. [DOI] [PubMed] [Google Scholar]
- Birgens H S, Henriksen J, Matzen P, Poulsen H. The shock liver. Clinical and biochemical findings in patients with centrilobular liver necrosis following cardiogenic shock. Acta Med Scand. 1978;204(5):417–421. [PubMed] [Google Scholar]
- Myers R P, Cerini R, Sayegh R, et al. Cardiac hepatopathy: clinical, hemodynamic, and histologic characteristics and correlations. Hepatology. 2003;37(2):393–400. doi: 10.1053/jhep.2003.50062. [DOI] [PubMed] [Google Scholar]
- Gitlin N, Serio K M. Ischemic hepatitis: widening horizons. Am J Gastroenterol. 1992;87(7):831–836. [PubMed] [Google Scholar]
- Cassidy W M, Reynolds T B. Serum lactic dehydrogenase in the differential diagnosis of acute hepatocellular injury. J Clin Gastroenterol. 1994;19(2):118–121. doi: 10.1097/00004836-199409000-00008. [DOI] [PubMed] [Google Scholar]
- Allen L A, Felker G M, Pocock S, et al. CHARM Investigators Liver function abnormalities and outcome in patients with chronic heart failure: data from the Candesartan in Heart Failure: Assessment of Reduction in Mortality and Morbidity (CHARM) program. Eur J Heart Fail. 2009;11(2):170–177. doi: 10.1093/eurjhf/hfn031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shinagawa H, Inomata T, Koitabashi T, et al. Prognostic significance of increased serum bilirubin levels coincident with cardiac decompensation in chronic heart failure. Circ J. 2008;72(3):364–369. doi: 10.1253/circj.72.364. [DOI] [PubMed] [Google Scholar]
- Denis C, De Kerguennec C, Bernuau J, Beauvais F, Cohen Solal A. Acute hypoxic hepatitis (‘liver shock’): still a frequently overlooked cardiological diagnosis. Eur J Heart Fail. 2004;6(5):561–565. doi: 10.1016/j.ejheart.2003.12.008. [DOI] [PubMed] [Google Scholar]
- Deursen V M van, Damman K, Hillege H L, Beek A P van, Veldhuisen D J van, Voors A A. Abnormal liver function in relation to hemodynamic profile in heart failure patients. J Card Fail. 2010;16(1):84–90. doi: 10.1016/j.cardfail.2009.08.002. [DOI] [PubMed] [Google Scholar]
- Lau G T, Tan H C, Kritharides L. Type of liver dysfunction in heart failure and its relation to the severity of tricuspid regurgitation. Am J Cardiol. 2002;90(12):1405–1409. doi: 10.1016/s0002-9149(02)02886-2. [DOI] [PubMed] [Google Scholar]
- Camposilvan S, Milanesi O, Stellin G, Pettenazzo A, Zancan L, D'Antiga L. Liver and cardiac function in the long term after Fontan operation. Ann Thorac Surg. 2008;86(1):177–182. doi: 10.1016/j.athoracsur.2008.03.077. [DOI] [PubMed] [Google Scholar]
- Lowe M D, Harcombe A A, Grace A A, Petch M C. Lesson of the week: restrictive-constrictive heart failure masquerading as liver disease. BMJ. 1999;318(7183):585–586. doi: 10.1136/bmj.318.7183.585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kirsch M, Fleshler B. Deceptive liver histology delays diagnosis of cardiac ascites. South Med J. 1992;85(11):1151–1152. doi: 10.1097/00007611-199211000-00023. [DOI] [PubMed] [Google Scholar]
- Solano F X, Jr, Young E, Talamo T S, Dekker A. Constrictive pericarditis mimicking Budd-Chiari syndrome. Am J Med. 1986;80(1):113–115. doi: 10.1016/0002-9343(86)90058-6. [DOI] [PubMed] [Google Scholar]
- Arora A, Tandon N, Sharma M P, Acharya S K. Constrictive pericarditis masquerading as Budd-Chiari syndrome. J Clin Gastroenterol. 1991;13(2):178–181. doi: 10.1097/00004836-199104000-00012. [DOI] [PubMed] [Google Scholar]
- Sokol S I, Cheng A, Frishman W H, Kaza C S. Cardiovascular drug therapy in patients with hepatic diseases and patients with congestive heart failure. J Clin Pharmacol. 2000;40(1):11–30. doi: 10.1177/00912700022008649. [DOI] [PubMed] [Google Scholar]
- Daskalopoulos G, Pinzani M, Murray N, Hirschberg R, Zipser R D. Effects of captopril on renal function in patients with cirrhosis and ascites. J Hepatol. 1987;4(3):330–336. doi: 10.1016/s0168-8278(87)80542-1. [DOI] [PubMed] [Google Scholar]
- New Horizons in Antihypertensive Therapy. Gopten (trandolapril). Ludwigshafen: Knoll AG. 1992 [Google Scholar]
- Brookman L J, Rolan P E, Benjamin I S, et al. Pharmacokinetics of valsartan in patients with liver disease. Clin Pharmacol Ther. 1997;62(3):272–278. doi: 10.1016/S0009-9236(97)90029-1. [DOI] [PubMed] [Google Scholar]
- Gillis J C, Markham A. Irbesartan. A review of its pharmacodynamic and pharmacokinetic properties and therapeutic use in the management of hypertension. Drugs. 1997;54(6):885–902. doi: 10.2165/00003495-199754060-00007. [DOI] [PubMed] [Google Scholar]
- Markham A, Goa K L. Valsartan. A review of its pharmacology and therapeutic use in essential hypertension. Drugs. 1997;54(2):299–311. doi: 10.2165/00003495-199754020-00009. [DOI] [PubMed] [Google Scholar]
- Marino M R, Langenbacher K M, Raymond R H, Ford N F, Lasseter K C. Pharmacokinetics and pharmacodynamics of irbesartan in patients with hepatic cirrhosis. J Clin Pharmacol. 1998;38(4):347–356. doi: 10.1002/j.1552-4604.1998.tb04434.x. [DOI] [PubMed] [Google Scholar]
- Kirch W, Schäfer-Korting M, Mutschler E, Ohnhaus E E, Braun W. Clinical experience with atenolol in patients with chronic liver disease. J Clin Pharmacol. 1983;23(4):171–177. doi: 10.1002/j.1552-4604.1983.tb02721.x. [DOI] [PubMed] [Google Scholar]
- Buchi K N, Rollins D E, Tolman K G, Achari R, Drissel D, Hulse J D. Pharmacokinetics of esmolol in hepatic disease. J Clin Pharmacol. 1987;27(11):880–884. doi: 10.1002/j.1552-4604.1987.tb05583.x. [DOI] [PubMed] [Google Scholar]
- Zaman R, Jack D B, Wilkins M R, Kendall M J. Lack of effect of liver disease on the pharmacokinetics of acebutolol and diacetolol: a single dose study. Biopharm Drug Dispos. 1985;6(2):131–137. doi: 10.1002/bdd.2510060204. [DOI] [PubMed] [Google Scholar]
- Fredrick M J, Pound D C, Hall S D, Brater D C. Furosemide absorption in patients with cirrhosis. Clin Pharmacol Ther. 1991;49(3):241–247. doi: 10.1038/clpt.1991.23. [DOI] [PubMed] [Google Scholar]
- Nul D R, Doval H C, Grancelli H O, et al. Heart rate is a marker of amiodarone mortality reduction in severe heart failure. The GESICA-GEMA Investigators. Grupo de Estudio de la Sobrevida en la Insuficiencia Cardiaca en Argentina-Grupo de Estudios Multicéntricos en Argentina. J Am Coll Cardiol. 1997;29(6):1199–1205. doi: 10.1016/s0735-1097(97)00066-1. [DOI] [PubMed] [Google Scholar]
- Garguichevich J J, Ramos J L, Gambarte A, et al. Effect of amiodarone therapy on mortality in patients with left ventricular dysfunction and asymptomatic complex ventricular arrhythmias: Argentine Pilot Study of Sudden Death and Amiodarone (EPAMSA) Am Heart J. 1995a;130(3 Pt 1):494–500. doi: 10.1016/0002-8703(95)90357-7. [DOI] [PubMed] [Google Scholar]
- Go A S, Lee W Y, Yang J, Lo J C, Gurwitz J H. Statin therapy and risks for death and hospitalization in chronic heart failure. JAMA. 2006;296(17):2105–2111. doi: 10.1001/jama.296.17.2105. [DOI] [PubMed] [Google Scholar]
- Shinagawa H, Inomata T, Koitabashi T, et al. Increased serum bilirubin levels coincident with heart failure decompensation indicate the need for intravenous inotropic agents. Int Heart J. 2007;48(2):195–204. doi: 10.1536/ihj.48.195. [DOI] [PubMed] [Google Scholar]