Abstract
One consequence of central nervous system injury or disease is the impairment of neural control of movement, resulting in spasticity and paralysis. To enhance recovery, restorative neurology procedures modify altered, yet preserved nervous system function. This review focuses on functional electrical stimulation (FES) and spinal cord stimulation (SCS) that utilize remaining capabilities of the distal apparatus of spinal cord, peripheral nerves and muscles in upper motor neuron dysfunctions. FES for the immediate generation of lower limb movement along with current rehabilitative techniques is reviewed. The potential of SCS for controlling spinal spasticity and enhancing lower limb function in multiple sclerosis and spinal cord injury is discussed. The necessity for precise electrode placement and appropriate stimulation parameter settings to achieve therapeutic specificity is elaborated. This will lead to our human work of epidural and transcutaneous stimulation targeting the lumbar spinal cord for enhancing motor functions in spinal cord injured people, supplemented by pertinent human research of other investigators. We conclude that the concept of restorative neurology recently received new appreciation by accumulated evidence for locomotor circuits residing in the human spinal cord. Technological and clinical advancements need to follow for a major impact on the functional recovery in individuals with severe damage to their motor system.
Keywords: Functional electrical stimulation, Locomotion, Motor control, Multiple sclerosis, Neuromodulation, Restorative neurology, Spinal cord injury, Spinal cord stimulation
1. Introduction
The effects of central nervous system (CNS) injury or disease include altered sensation and pain and impaired control of movement, paralysis and spasticity. Contemporary emergency and rehabilitative medical care has developed to successfully prevent and treat secondary medical complications of neurological disorders providing a near-normal life expectancy for people with neurological damage. Medicine at present, however, cannot anatomically repair the affected parts of the CNS to return normal neurological function. This results in a large population of patients who must endure chronic sensory and motor disabilities and need solutions that improve their quality of life.
To enhance recovery from CNS injury or disease, restorative neurology is a discipline that works through the modification of residual, altered, yet remaining nervous system function. Neuromodulation is a major category of interventions used in restorative neurology. It evolved from physiological and biomedical engineering advances in the 1960s. The therapeutic paradigm was the modulation of CNS activity rather than the irreversible (destructive) modification of the CNS structure. An example of this development was Vladimir Liberson's functional electrotherapy, later renamed functional electrical stimulation (FES), for patients with foot drop after stroke [1]. The availability of portable stimulators further led to multi-site FES to generate standing and some stepping-movements in paraplegic patients and to augment gait rehabilitation in patients with incomplete spinal cord injury (SCI). In 1967, Norman Shealy implanted the first neuroaugmentive device for the relief of intractable pain which he termed dorsal column stimulator [2]. The potential of this technique of spinal cord stimulation (SCS) in movement disorders was recognized soon thereafter [3]. The commercial availability of implantable stimulators paved the way for the current neuromodulation therapies for altered sensation and pain, peripheral and cardiac ischemia, severe depression and psychiatric conditions [4,5].
In movement disorders, contemporary neuromodulation therapies target brain structures, cranial nerves, spinal cord, and peripheral nerves and influence the nervous system activity by the interaction of an electrical or neurochemical input with specific neural circuits [5–7]. Deep brain stimulation with implanted leads is applied in Parkinson's disease [8], in dystonia [9] and as a treatment for essential tremor [10]. Vagus nerve stimulation via an implanted electrical stimulator is applied in the treatment of intractable epilepsy [11]. Electrical epidural SCS can alleviate various disorders of the motor system in patients with SCI, multiple sclerosis (MS), cerebral palsy, and people with post-traumatic and post-stroke brain injury [12]. Sacral nerve stimulation with an implanted lead, also termed sacral neuromodulation, is an accepted treatment method for urinary pelvic disorders and incontinence [13]. The development of implantable drug pumps allowed for the chronic delivery of medication to the physiological target site of the spinal cord to modulate motor function. Intrathecal delivery of baclofen is well-established in the treatment of spinal spasticity caused by MS or SCI [14] and spasticity of cerebral origin [15].
Methods for the restoration of the neuromuscular lower limb functions will be the focus of the present review. These approaches utilize the intact peripheral nerves and the capacity of the spinal cord of neural signal processing that remain after impairment of the CNS [16]. FES for the generation of lower limb movement will be reviewed first. Then, SCS for the improvement of lower limb function in motor disorders (primarily) with spinal cord involvement will be discussed, i.e. the improvement of spinal spasticity and altered motor control due to MS and SCI. These topics will lead to the report on our human work of epidural and transcutaneous stimulation specifically targeting the lumbar spinal cord for enhancing motor functions in people with SCI, supplemented by pertinent human research of other investigators. The reviewed literature was partially selected following the development process of the recently published monograph on restorative neurology of motor control after SCI [17], and from a comprehensive reference work encompassing the various aspects of neuromodulation [18].
2. Functional electrical stimulation for the restoration of lower limb functions
The first modern applications of FES to generate movements of paralyzed lower limbs in SCI and hemiplegic subjects were reported in the early 1960s [1,19]. FES is applied to produce patterned muscle contractions and movements for specific function in people with motor control impaired by upper motor neuron dysfunction. FES methodologies use timed sequences of short bursts of electrical pulses, rather than sustained stimulation commonly applied in other neuromodulation therapies [6]. Muscles are either activated by electrical stimulation of the respective lower motoneurons, usually near the corresponding motor endplate region [6], or by peripheral afferent stimulation and the resulting spinal reflex [20].
FES systems for the restoration of lower limb movement can be divided into applications for the facilitation of ankle dorsiflexion in hemiplegia [21] and the generation of standing and stepping-movements in SCI individuals [22]. Both, surface stimulators and implantable devices have been developed. In the therapeutic treatment of foot drop after stroke, footswitch- or tilt sensor-triggered peroneal nerve stimulation elicits a flexion reflex that generates hip, knee, and ankle movements and enhances the swing phase during gait [1,23,24]. A review that evaluated seven case series and one randomized clinical trial of FES in stroke patients with foot drop suggested a positive effect of FES on walking speed, with a pooled improvement by 38% [25]. Commercially available and FDA approved surface stimulation systems primarily designed for foot drop correction are the WalkAide System (Innovative Neurotronics Inc., Austin, TX), the Odstock Dropped Foot Stimulator (Odstock Medical Limited, Salisbury District Hospital, Salisbury, UK), and the Ness L300 Foot Drop System (Bioness Inc., Valencia, CA). Implantable FES devices were designed for more selective control of the evoked muscle activities by stimulating separate groupings of fibers within a nerve trunk. The ActiGate system (Neurodan A/S, Aalborg, Denmark) is based on an implanted cuff electrode with four channels positioned to activate different nerve fibers within the common peroneal nerve and can elicit dorsal and plantar flexion as well as inversion and eversion. Safety and the effect on walking of the ActiGate system in hemiplegia were evaluated in a phase II trial [26]. The authors reported long-term improvements in walking speed by 19% and in the distance walked in 4 min. The STIMuSTEP (Finetech Medical Ltd, Welwyn Garden City, UK) is a two-channel implantable peroneal nerve stimulator. Randomized controlled trials of the effects of the STIMuSTEP in hemiplegia demonstrated an increased walking speed by 23% [27] and improvements of spatiotemporal gait parameters and kinematics [28]. Apart from the immediate effect of stimulation, training with the FES system can (temporarily) enable patients to dorsiflex their foot volitionally to ambulate with less fatigue [1,29].
In paraplegic individuals with thoracic SCI, multi-site FES can generate standing and short-distance ambulation by stimulating the quadriceps, sometimes together with the gluteal muscles, to produce lower limb extension during stance phases, and the peroneal nerve to initiate a swing-like motion [30–32]. The stimulation sequences are controlled by a microcomputer and are manually initiated by the user, and a walker or crutches are used for weight and balance support. Forward movement is predominantly achieved by sliding the walker in the double support phase or with the help of the crutches. The walking FES systems can be used in combination with mechanical braces to reduce the number of muscles to be controlled and to provide additional joint stability [33]. The Parastep (Sigmedics, Inc., Faiborn, OH) is an FDA approved surface FES system composed of a six-channel, microprocessor-controlled stimulator, skin electrodes, and a modified walking frame [30]. This system has been evaluated for ambulation and other beneficial medical effects in several studies [34–37] and is widely available. Research-based implantable multi-channel FES devices for the restoration of lower limb movement in motor-complete SCI subjects have been developed by several research groups [22]. The feasibility of the application of implants utilizing either epimysial electrodes or cuff electrodes for the generation of standing and some stepping movements was tested in a few SCI subjects [38–40]. Generally, the walking FES systems for motor-complete SCI subjects are not a substitute for a wheelchair, but used as an active means of exercise they provide many benefits including improved blood flow to the lower limbs, increased muscle mass and strength, reduced spasticity, improvement in skin trophic, and cardiovasular benefits.
In people with motor-incomplete SCI, timed peroneal nerve stimulation can be used to facilitate the swing phase during locomotor training on a treadmill to augment gait rehabilitation in the early phase after injury [41]. Candidates for chronic use of peroneal electrical stimulators are SCI individuals, who have sufficient trunk and knee extensor function but weak flexor activity of the lower limbs [42]. In the chronic phase of incomplete SCI, the diversity of injury profiles and residual motor function results in a broad variety of impaired gait patterns. By supplementing the individual residual motor control, tailored multi-site FES can improve these altered patterns of movements [43]. Such applications can exploit all of the characteristic advantages of FES to either achieve local or more widespread synergistic responses by stimulation of the appropriate peripheral nerves, to produce immediate muscle force by efferent stimulation or to utilize central mechanisms following the sensory input produced either directly or secondary to the evoked movements [44,45].
3. Epidural spinal cord stimulation in multiple sclerosis
Epidural SCS is generally associated with the treatment of chronic neurogenic pain conditions and in fact, SCS for pain control is the most common modality of all neuromodulation therapies [5]. Yet, SCS can be an effective method in the alleviation of various disorders of the motor system. The application of SCS in motor disorders resulted from observations made by Cook and Weinstein when treating an MS patient for pain [3]. SCS controlled the pain and, in addition, improved voluntary motor function. Cook's work was repeated and the finding of improved motor control and spinal spasticity in patients with MS was confirmed [46–48]. Illis and colleagues observed immediate and profound improvements of motor, sensory and bladder function in 2 patients with MS treated by temporary epidural SCS [47]. The first patient with signs of an upper motor neuron lesion with spasticity regained unaided walking capability with SCS. Siegfried et al. applied SCS in the attempt to control spinal spasticity in 10 patients [49]. Six of these patients had MS. Electrodes were positioned between the C3/C4 and T1/T2 vertebral levels and intermittent stimulation was applied at a frequency between 50 Hz and 100 Hz. Such stimulation elicited paraesthesiae in the cervical or thoracic dermatomes corresponding to the segmental level of the epidural electrodes and, with increasing stimulus intensities, in both lower limbs or in all four extremities. In all cases, a striking improvement of the spasticity of the lower (and the upper) limbs was confirmed by neurophysiological evaluation, with the best results observed in the MS patients.
In subsequent studies of separate groups with increasing numbers of individuals treated it became clear that SCS augmented motor function only in a portion of the tested patient populations with inter-individually varying improvements [50]. Davis et al. reported improvements of ambulation with increased endurance and muscle strength in 44 of 69 patients with MS [51]. Illis et al. found significant improvement in 5 of 18 MS patients with mobility problems treated with SCS [52]. The beneficial responses included a feeling of lightness of the legs, the ability to stand and walk more easily, increased endurance, and regained unaided walking capability. The stimulation was carried out at 33 Hz and the electrodes were placed between C6/C7 and T9/T10 vertebral levels. Stimulation which produced improvements had to be adjusted to generate bilateral sensation into the legs (by adjusting the medio-lateral electrode position and the stimulus intensity). The variability of the effects of SCS is also reflected by a later work of the same group suggesting that SCS did not have a major quantifiable impact upon motor disability in MS [53]. The effect of SCS in a large number of patients with various motor disorders, including 130 MS patients treated with SCS, was discussed by Waltz [54]. Among the MS patients, 82% had walking difficulties, partially attributable to spasticity and weakness. Spasticity was abolished or significantly decreased in 58%, with increased ease in carrying out voluntary motor function. Weakness improved in 33%, most markedly in the hip flexors, with a positive impact on gait. Electrodes were placed between the vertebral levels C2 and C4 and frequencies of 100–1500 Hz were applied, with most patients responding best to 100–200 Hz.
The physiological mechanisms of epidural SCS in MS are not yet elucidated. Electrophysiological studies of the effects of SCS during the treatment of MS, including recordings of cervical somatosensory and brainstem evoked potentials, indicated that SCS acted at spinal cord and brainstem levels [55]. The detected changes in the evoked potentials in association with SCS were interpreted as variations of the efficacy of afferent volleys in engaging neural circuits, as in the dorsal horn of the spinal cord and the nucleus cuneatus. Gybels and van Roost suggested in their review of numerous studies of SCS in motor disorders that the working mechanisms of SCS are beyond local effects and rather due to an increased descending inhibition of excessive spinal reflex activity [50]. Waltz proposed the augmentation or modulation of ascending and descending tracts [54]. Illis hypothesized that SCS might work through modulation of the central excitatory state and neurotransmitter release and of the afferent inflow [52].
The variety of rostro-caudal sites of SCS in the studies reviewed above, ranging from C2 to T10 vertebral levels, indeed suggests the activation of a rather diffuse system involving ascending and descending tracts (the latter by synaptic activation) and segmental circuits. This fact together with the pathophysiological complexity of MS might have contributed to the variety of results reported.
Our understanding is that the therapeutic specificity of SCS hinges on the direct involvement of specific segmental spinal circuits through the precise placement of the implanted electrodes. With regard to lower limb functions, considerable progress has been made in recent years in recognizing the remarkable capabilities of the lumbar spinal cord circuitry (located at T11–T12 vertebral levels) to control motor function and how they can be utilized by lumbar SCS [56–60]. Studies on the effect of SCS targeting these circuits involved in the control of muscle tone and lower limb movement will be presented in the following sections.
4. Epidural spinal cord stimulation in spasticity after spinal cord injury
The positive effects of SCS in MS [3,46] motivated its application in SCI. Richardson, McLone and colleagues described the alleviation of severe spasticity and flexor spasms in 6 individuals with post-traumatic, complete thoracic SCI by SCS in a series of case reports [61,62]. Epidural electrodes were placed below the injury between the L1 and L4 vertebral levels, and the effective stimulation frequencies were 33–75 Hz. Subsequent studies had variable results. Siegfried et al. did not confirm a satisfactory effect of SCS on spasticity in 15 chronic SCI patients [63], with epidural electrodes placed rostral to the lesion level (see personal communication of Dimitrijevic in [64]). Barolat and colleagues, on the other hand, concluded in a larger series of cases that epidural SCS is effective and safe in the management of spasticity and spasms in SCI individuals [65,66]. Waltz found improved motor function in 65% of 303 SCI patients treated by cervical SCS [54]. No specific SCS frequency preference within the range of 100–1500 Hz was reported. Benefits included decreased spasticity and spasms and augmented function in muscle groups retaining some voluntary movement. Dimitrijevic and co-workers investigated the efficacy of SCS for spasticity control in 59 SCI patients and obtained marked or moderate effects in 63% of the patients [64,67]. The epidural electrodes were placed at C2 to T12 vertebral levels and continuous stimulation was applied at 30–50 Hz. The variability of the efficacy of stimulation did not depend on the severity of spasticity, but on the rostro-caudal electrode site. Considering their own results and the reports of Richardson et al. [61,62] and Siegfried et al. [63], they concluded that the optimal position for epidural leads in patients with thoracic SCI was below the level of the lesion [64]. It was suggested that such thoraco-lumbar SCS would lead to antidromic activation of residual, longitudinal structures of the spinal cord below the injury with resulting modulation of segmental spinal circuits’ activity involved in the generation of spasticity. In incomplete SCI patients with partial functional preservation of ascending and descending neural tracts, long loop dorsal column–brainstem–spinal mechanisms modulating the excitability of segmental circuits were suggested as additional mechanism [64].
In view of the motor control capabilities of the upper lumbar spinal cord circuitry [56] (see also below), Pinter and colleagues revisited the use of SCS in chronic SCI individuals with severe lower limb spasticity [57]. Rather than just placing the epidural lead at some level below the SCI, they precisely positioned the electrodes at the segmental levels of the upper lumbar spinal cord. The positioning of the lead was controlled under fluoroscopy and guided by intraoperative elicitation of muscle twitch responses (see [58,68] and cf. posterior root-muscle reflexes, below). Based on the segmental muscle innervations, the target position required the lowest thresholds for responses evoked in the adductors and quadriceps. Eight subjects with chronic, post-traumatic SCI and severe lower limb spasticity were studied. Lesions were at C5–C6 (n = 3) and T3–T6 (n = 5) levels and were classified as grade A (n = 5), B (n = 2) and C (n = 1) according to the American Spinal Injury Association impairment scale (AIS), respectively [69]. The neurophysiological assessment [70] showed a significant suppression of lower limb spasticity in all subjects, when the lead was over the target site and the stimulation frequency in a range of 50–100 Hz. Furthermore, the same study demonstrated that there was no modification of lower limb spasticity when the epidural contacts were located over the lowest thoracic spinal cord segments [57]. At this more rostral stimulation site, hypertonia remained unchanged even when various stimulation intensities and frequencies were tested. The specificity of the segmental position of the stimulating epidural contact strongly suggested that the inhibitory mechanism were due to the stimulation of segmental afferents within the closest posterior roots that in turn trans-synaptically activated local neural processors in the lumbar spinal cord [57]. Note, that the posterior roots contain the whole range of sensory fibers, including Group I afferent fibers that arise in muscles, whereas there are differences in the intramedullary projections of Group I fibers and Group II cutaneous afferent fibers within the posterior columns [71].
5. Epidural lumbar spinal cord stimulation for the generation of lower limb motor activity
The control of lower limb spasticity by epidural stimulation of the upper lumbar spinal cord requires individual identification of effective stimulation parameters, involving stimulation through different contacts along the epidural lead (Fig. 1A) and the use of different stimulus intensities and frequencies [57,64]. During such testing in the clinical practice of SCS it became obvious that the stimulation did not only attenuate spinal cord activity, but also produced a variety of non-patterned and patterned motor outputs to the lower limb muscles, depending on the stimulation parameters.
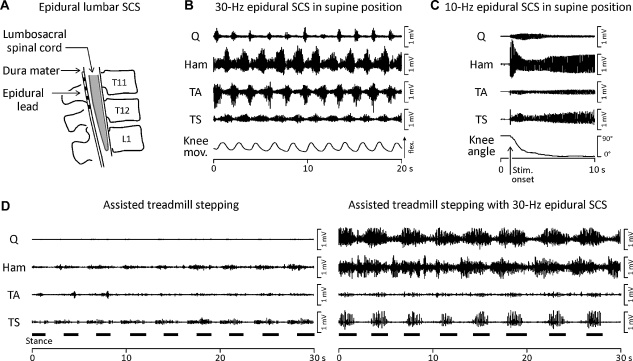
Fig. 1.
Lower limb motor activity generated by epidural lumbar SCS. (A) Drawing of a cylindrical epidural lead with four contacts (black rectangles) placed over the posterior aspect of the lumbar spinal cord. (B) Locomotor-like EMG activities of paralyzed lower limb muscles elicited by sustained epidural lumbar SCS at 25 Hz and 10 V in a supine position. Q: quadriceps, Ham: hamstrings, TA: tibialis anterior, TS: triceps surae. Induced relative knee movements (Knee mov.) are documented by a position sensor trace; deflection up indicates knee flexion. Data derived from a subject with chronic, motor-complete (AIS B), mid-thoracic (motor level: T8) traumatic SCI [56]. (C) Extension movement and associated EMG activity induced by epidural lumbar SCS at 10 Hz and 10 V. The subject's lower limb had been placed in a flexed position prior to the onset of stimulation (see vertical arrow along the time-axis). The goniometer trace (Knee angle) illustrates the generated extension movement. Data derived from a subject with chronic, motor-complete (AIS A), mid-thoracic (motor level: T6) traumatic SCI in supine position [75]. (D) Lower limb EMG activity induced by manually assisted, body-weight supported treadmill walking without (left side) and with additional epidural lumbar SCS. The treadmill belt speed was 0.36 m/s. The subject wore a parachute harness connected to counterweights which supported him vertically over the treadmill and provided 50% body weight support. Two physiotherapists manually imposed the stepping motions on the moving treadmill belt. No independent functional movements were produced. Black horizontal bars indicate stance phases. Subject with chronic, motor-complete (AIS A), low-cervical (motor level: C6) traumatic SCI [78].
Dimitrijevic and colleagues reported in chronic, motor complete SCI subjects that lumbar SCS could produce rhythmic activities in the paralyzed lower limbs [56]. An example of locomotor-like, alternating electromyographic (EMG) activity in groups of antagonistic muscles generated by lumbar SCS is shown in Fig. 1B. The functional separation of the lumbar spinal cord from supraspinal structures was demonstrated in these subjects by clinical and neurophysiological assessment [56,70]. The influence of proprioceptive feedback in the rhythm-generation of the muscle activity could be assumed to be insignificant, since all subjects were studied in a supine position that limited lower limb loading and hip extension. It was thus demonstrated that the human lumbar spinal cord contains neural networks that can generate coordinated oscillating activity in response to tonic neural input signals, even after chronic disconnection from supraspinal structures. For the generation of rhythmic motor outputs to the lower limbs, SCS frequency had to be within the range of 25–60 Hz. Subsequent studies repeated and verified these results [72–74].
In addition, it was found that non-patterned electrical stimulation of the lumbar spinal cord at 5–15 Hz could generate and retain bilateral lower-limb extension in motor complete SCI subjects in supine position [58,75]. When the subjects’ lower extremities were manually moved to a position of hip and knee flexion, and SCS was subsequently applied at 5–15 Hz, a brisk and strong extension movement of the lower limbs was produced. The associated EMG activities from the lower limb muscles revealed characteristic and reproducible temporal amplitude modulations (Fig. 1C). When the position of full extension was reached, and stimulation was continued, the limbs further remained in this position, with the muscles clearly contracting. In the same subjects, the extension-like muscle activities were replaced by rhythmic ones when the SCS frequency was increased to 30 Hz, without changing the site or intensity of stimulation [75].
Electrophysiological [68,74] and computational [76,77] studies revealed that the structures directly, electrically stimulated by epidural lumbar SCS are predominantly afferent fibers of the posterior roots. The tonic neural drive to the lumbar spinal cord via the posterior root afferents was suggested to produce parallel effects. The volleys elicit segmental muscle responses in the lower limbs, so-called posterior root-muscle reflexes, and co-activate lumbar interneuronal circuits via synaptic projections [58,74,75]. Their integration into the generation of motor outputs most probably relies on temporal and spatial summation processes of the postsynaptic potentials produced.
Despite the above discussed findings and the clear potential for clinical application, few human studies have capitalized on the intrinsic motor control mechanisms of the lumbar spinal cord that remain below a spinal cord lesion. In 2 cases of chronic, motor complete SCI individuals, the rhythmic EMG activity produced by partial body weight-supported, manually assisted treadmill-stepping was immediately augmented when epidural lumbar SCS at 20–50 Hz was supplied (Fig. 1D) [58,78]. It was thus shown that the rhythmic sequences of proprioceptive feedback associated with the mechanical events during passive stepping and the tonic neural drive produced by SCS could be integrated by the lumbar spinal cord in the generation of motor output. Furthermore, rhythmic activity could be produced in muscles that did not respond to manually assisted treadmill-stepping alone, and the timing of the activity relative to the gait cycle could be shifted by the stimulation. Independent functional stepping, however, was not achieved. A similar augmentation of rhythmic muscle activities on a moving treadmill belt by SCS was recently demonstrated in a case study of a subject with a clinically motor complete and sensory incomplete SCI, but this subject had undergone an extensive locomotor training [60]. Rhythmic EMG activity was generated when epidural lumbar SCS at 30–40 Hz and step-related proprioceptive feedback were provided. Independent gait-like movements were not achieved in this study either. Yet, SCS could generate full weight-bearing standing without manual facilitation when the stimulation frequency was set at 15 Hz. Additionally, 7 months after implantation, epidural SCS revealed residual voluntary control of some lower limb movements that was only present when stimulation was supplied (see also [79]).
In 2 wheelchair-dependent individuals with motor and sensory incomplete SCI classified as AIS C, Herman and his team investigated a combined therapy of locomotor training and SCS [80–83]. They applied locomotor training until the participant reached a plateau in treadmill gait performance and subsequently employed epidural SCS in conjunction with locomotor training. The initial locomotor training without SCS improved the subjects’ ability to step on the treadmill, however, the improvements did not translate successfully into over-ground ambulation. Acute effects of SCS when applied during treadmill stepping included improved muscle activation patterns and an enhanced level of EMG activity. After the period of combined training, both subjects were able to transfer the coordinated movements acquired through locomotor training with SCS into functional over-ground ambulation. In both subjects, 2 epidural leads were placed over the thoraco-lumbar spinal cord in parallel, each located few millimeters off the physiological midline on either side, and stimulation frequency was between 20 and 60 Hz. The thoraco-lumbar stimulation might have accessed the lumbar locomotor circuits by antidromic activity in afferent branches within the posterior columns of the spinal cord white matter or by stimulation of the respective segmental posterior roots due to current spread [83]. Brain stem-spinal pathways could have been also involved.
In summary, there is clear evidence for the presence of a remarkable motor control capacity within the human lumbar spinal cord that remains operational even after chronic separation from supraspinal structures. This remaining functional capacity can be set into operation by non-patterned stimulation delivered via intact segmental input pathways. In spite of being provided through afferent projections, such tonic activity is characteristic for descending neural drive rather than for proprioceptive feedback. The activated neural circuits can generate a variety of motor outputs [56,84]. Their operation strongly depends on the site, intensity and frequency of SCS. Frequencies of 5–15 Hz promote extension [58,60,75], stimulation at 25–60 Hz can generate or facilitate rhythmic activity of the lower limbs [56,60,73,74,78,82], and 50–100 Hz can attenuate exaggerated spinal activity affecting the lower extremities [57,62].
6. Transcutaneous lumbar spinal cord stimulation for modification of altered neural control following spinal cord injury
Epidural lumbar SCS does not activate spinal cord circuitries directly, but trans-synaptically through the electrical stimulation of posterior root and spinal cord afferents [58,76,85]. Posterior root afferents can be stimulated by non-invasive methods as well. Posterior root fibers of the L2 to S2 spinal cord segments can be consistently stimulated by transcutaneous SCS with skin electrodes [86–88]. The technique uses stimulating electrodes over the paravertebral skin between the T11 and T12 spinous processes, and large reference electrodes over the lower abdomen (Fig. 2A). The biophysics leading to the specific, localized depolarization of posterior root fibers in spite of the distant stimulation is well-understood [77,89,90]. The ligaments between the spinous processes and laminae and the intervertebral discs between the vertebral bodies reduce the transversal electrical resistance of the thoraco-lumbar spine and allow some current produced by transcutaneous SCS to flow through the vertebral canal. Within the vertebral canal, the current density is relatively high in the well-conducting cerebrospinal fluid. There, the excitation threshold of the posterior root fibers is considerably reduced by non-uniformities of the anatomy along the fiber paths and changes of the fiber path direction with respect to the generated field.
Fig. 2.
Lower limb motor activity generated by transcutaneous lumbar SCS. (A) Stimulation method of transcutaneous SCS. Drawings of the stimulating paravertebral and abdominal reference electrodes with respect to the spine and spinal cord. (B) EMG activities generated in paralyzed lower limb muscles by partial (50%) body-weight supported and manually assisted treadmill stepping without (left) and with sub-motor transcutaneous SCS. EMG recordings were derived from the right quadriceps (Q), hamstrings (Ham), tibialis anterior (TA), and triceps surae (TS) along with goniometric data from the knee (Knee angle). Black bars mark stance phases; treadmill speed was 0.33 m/s. Continuous transcutaneous SCS at 30 Hz and 25 V produced rhythmic gait-synchronized EMG activities. Lower limb motor threshold was 28 V in a supported standing position. Subject with chronic, motor-complete (AIS A), mid-thoracic (motor level: T6) traumatic SCI. (C) EMG activities generated in paralyzed lower limbs by partial (50%) body-weight supported and manually assisted treadmill stepping without (left) and with above-motor threshold transcutaneous SCS; treadmill belt speed was 0.39 m/s. Transcutaneous SCS at 30 Hz and 35 V produced burst-like activities in all right lower limb muscles, with in-phase oscillation that were not synchronized to the manually controlled step-cycle. Lower limb motor threshold was 25 V in a passive supported standing position. Subject with chronic, motor-complete (AIS B), low-cervical (motor level: C6) traumatic SCI.
Potential therapeutic applications of tonic transcutaneous SCS for the alleviation of spinal spasticity and the enhancement of neural control of locomotion after SCI are currently being investigated. In a pilot study, it was explored whether lumbar transcutaneous SCS could temporarily modify spinal spasticity in 3 incomplete SCI subjects classified as AIS D [88,91]. Stimulation was applied in a supine position for 30 min at 50 Hz and an intensity producing paraesthesiae in lower limbs. Before and immediately after stimulation, the subjects’ spasticity and residual voluntary motor control were assessed clinically and neurophysiologically. In the post-stimulation evaluation, EMG associated with Achilles clonus, passive knee movement and mechanical plantar stimulation were reduced, and volitional rhythmic ankle movement was improved. The pendulum test demonstrated a statistically significant decline in spastic hypertonia. Two subjects (one using a walker, the other one 2 crutches) considerably increased their walking speed after SCS.
Early exploratory studies have shown that tonic transcutaneous lumbar SCS applied during treadmill stepping generated or modulated rhythmic lower limb EMG activities in motor complete and incomplete SCI [88,92]. In the subjects with motor complete SCI, manually assisted and body-weight supported treadmill stepping (without stimulation) produced only low-amplitude EMG activity in the paralyzed lower limbs [93]. When tonic transcutaneous SCS at 30 Hz was additionally supplied, rhythmic patterns of extensive EMG amplitudes could be produced (Fig. 2B and C). That the induced rhythmic activities were largely caused by the tonic transcutaneous SCS was reflected by the fact that the EMG consisted of series of stimulus-triggered posterior root-muscle reflexes. While the first results of the combined application of transcutaneous SCS and assisted treadmill stepping were very encouraging in motor complete SCI subjects, independent functional gait-like movements were not yet produced. Remarkable effects were achieved in 4 individuals classified as AIS D. Continuous transcutaneous SCS at 30 Hz and a sub-motor threshold level producing paraesthesiae in the lower limb dermatomes immediately modified the voluntarily generated lower limb activities in a gait phase appropriate way. The consciously controlled, yet augmented muscle activity led to changes of the stepping kinematics as well, including increased ranges of hip and knee movement and stride lengths [88,92].
Transcutaneous SCS combines the characteristics of FES and neuromodulation techniques and has all advantages of a non-invasive method. Using skin electrodes, it can be applied for repetitive interventions rather than for chronic stimulation therapies. Studies in progress will clarify the role of transcutaneous SCS in restorative neurology, particularly its efficacy in enhancing remaining motor capacity in people with various upper motor neuron dysfunctions.
7. Principles of neuromodulation in restorative neurology and future directions
Numerous research efforts are being conducted for the biological repair of the damaged CNS that focus on the lesion site and the interrupted neural pathways [94]. Even after severe CNS damage, however, there remains a distal, potentially functional apparatus of spinal cord, peripheral nerves and muscles [16]. In the present review we discussed FES and SCS as restorative neurology interventions in motor disorders that utilize this residual system. FES can provide for the immediate control of motor functions and generate sufficient force to produce movement. SCS modifies the steady-state of neural circuits’ operation and their response to physiological input signals—a common principle of neuromodulation therapies [7].
The concept of restorative neurology to augment surviving CNS capabilities is not new [95–97]. Yet, it received new appreciation by the accumulated evidence for the existence of neural circuits within the human lumbar spinal cord that can execute complex stereotyped motor tasks in response to rather unspecific stimuli [16,60,98]. Being deprived of (sufficient) supraspinal drive, neural processing and pattern generating networks caudal to a spinal cord lesion lose an adequate, sustainable state of excitability to be fully operational. Furthermore, the altered balance of excitation and inhibition [99] contribute to the clinical pictures of spasticity [100]. SCS provides a multi-segmental tonic neural drive to these circuitries and ‘tunes’ their physiological state to a more functional level.
These concepts need to be followed by technological and clinical advancements for the benefit of those who suffer from CNS damage. Epidural electrode arrays covering the dorsal columns as well as the dorsal root entry zones of the lumbosacral spinal cord in combination with multi-channel pulse generators are needed to allow for the spatially selective stimulation (of multiple targets) using field-steering methods. To ‘mimic’ altered or absent supraspinal drive, stimulation modes with more flexible control of time parameters (e.g. modulated stimulation frequencies) need to be tested for the generation of locomotor activity. Specific adaptations could build on such basic walking output by the combined utilization of SCS with methods of FES or intraspinal microstimulation [101].
SCS in combination with repetitive, task-specific training can have a major impact on the functional recovery in patients with severe damage to their motor system [60,78,92,98]. The confirmation of this potential by clinical trials with statistically appropriate subject populations or by alternative objective approaches [102] will be one of the future challenges. Such clinical programs will require the combined efforts of neurologists, human neurophysiologists, therapists and biomedical engineers at centers where comprehensive evaluation and long-term follow-up can be carried out.
Acknowledgements
The authors acknowledge the support of the Austrian Science Fund (FWF), Project No. L512-N13, the Vienna Science and Technology Fund (WWTF), Project No. LS11-057, and the Wings for Life Spinal Cord Research Foundation (WfL), Project No. WFL-AT-007/11.
The authors are grateful to Milan R. Dimitrijevic, W. Barry McKay and Manfred Bijak for their insightful discussions of the manuscript.
References
- 1.Liberson W.T., Holmquest H.J., Scot D., Dow M. Functional electrotherapy: stimulation of the peroneal nerve synchronized with the swing phase of the gait of hemiplegic patients. Arch Phys Med Rehabil. 1961;42:101–105. [PubMed] [Google Scholar]
- 2.Shealy C.N., Mortimer J.T., Reswick J.B. Electrical inhibition of pain by stimulation of the dorsal columns: preliminary clinical report. Anesth Analg. 1967;46:489–491. [PubMed] [Google Scholar]
- 3.Cook A.W., Weinstein S.P. Chronic dorsal column stimulation in multiple sclerosis. Preliminary report. NY State J Med. 1973;73:2868–2872. [PubMed] [Google Scholar]
- 4.Gildenberg P.L. Neuromodulation: a historical perspective. In: Krames E.S., Peckham P.H., Rezai A.R., editors. Neuromodulation. Elsevier-Academic Press; London: 2009. pp. 9–20. [Google Scholar]
- 5.Krames E.S., Rezai A.R., Peckham P.H., Aboelsaad F. What is neuromodulation? In: Krames E.S., Peckham P.H., Rezai A.R., editors. Neuromodulation. Elsevier-Academic Press; London: 2009. pp. 3–8. [Google Scholar]
- 6.Holsheimer J. Concepts and methods in neuromodulation and functional electrical stimulation: an introduction. Neuromodulation. 1998;1:57–61. doi: 10.1111/j.1525-1403.1998.tb00018.x. [DOI] [PubMed] [Google Scholar]
- 7.Holsheimer J. Letters to the Editor: what is neuromodulation? Neuromodulation. 2003;6:270–272. doi: 10.1046/j.1525-1403.2003.03036.x. [DOI] [PubMed] [Google Scholar]
- 8.Sillay K.A., Starr P.A. Deep brain stimulation in Parkinson's disease. In: Krames E.S., Peckham P.H., Rezai A.R., editors. Neuromodulation. Elsevier-Academic Press; London: 2009. pp. 539–548. [Google Scholar]
- 9.Alterman R.L., Tagliati M. Deep brain stimulation for torsion dystonia. In: Krames E.S., Peckham P.H., Rezai A.R., editors. Neuromodulation. Elsevier-Academic Press; London: 2009. pp. 571–578. [Google Scholar]
- 10.Burdick A.P., Okun M.S., Foote K.D. Deep brain stimulation for tremor. In: Krames E.S., Peckham P.H., Rezai A.R., editors. Neuromodulation. Elsevier-Academic Press; London: 2009. pp. 549–559. [Google Scholar]
- 11.Amar A.P., Levy M.L., Liu C.Y., Apuzzo M.L.J. Vagus nerve stimulation. In: Krames E.S., Peckham P.H., Rezai A.R., editors. Neuromodulation. Elsevier-Academic Press; London: 2009. pp. 625–637. [Google Scholar]
- 12.Waltz J.M., Reynolds L.O., Riklan M. Multi-lead spinal cord stimulation for control of motor disorders. Appl Neurophysiol. 1981;44:244–257. doi: 10.1159/000102207. [DOI] [PubMed] [Google Scholar]
- 13.McAchran S., Rackley R., Vasavada S. Neuromodulation for voiding dysfunction. In: Krames E.S., Peckham P.H., Rezai A.R., editors. Neuromodulation. Elsevier-Academic Press; London: 2009. pp. 945–956. [Google Scholar]
- 14.Penn R.D., Savoy S.M., Corcos D., Latash M., Gottlieb G., Parke B. Intrathecal baclofen for severe spinal spasticity. N Engl J Med. 1989;320:1517–1521. doi: 10.1056/NEJM198906083202303. [DOI] [PubMed] [Google Scholar]
- 15.Meythaler J.M., Guin-Renfroe S., Brunner R.C., Hadley M.N. Intrathecal baclofen for spastic hypertonia from stroke. Stroke. 2001;32:2099–2109. doi: 10.1161/hs0901.095682. [DOI] [PubMed] [Google Scholar]
- 16.Illis L.S. Central nervous system regeneration does not occur. Spinal Cord. 2011 doi: 10.1038/sc.2011.132. [DOI] [PubMed] [Google Scholar]
- 17.Dimitrijevic M.R., Kakulas B.A., McKay W.B., Vrbova G. Oxford University Press; New York: 2011. Restorative neurology of spinal cord injury. 336 pp. [Google Scholar]
- 18.Krames E.S., Peckham P.H., Rezai A.R. Elsevier-Academic Press; London: 2009. Neuromodulation. 1063 pp. [Google Scholar]
- 19.Kantrowitz A. Maimonides Hospital; New York, Brooklyn: 1960. Electronic physiologic aids. p. 4–5. [Google Scholar]
- 20.Kralj A., Bajd T., Turk R., Krajnik J., Benko H. Gait restoration in paraplegic patients: a feasibility demonstration using multichannel surface electrode FES. J Rehabil R D. 1983;20:3–20. [PubMed] [Google Scholar]
- 21.Chae J., Knutson J., Sheffler L.R. Stimulation for return of function after stroke. In: Krames E.S., Peckham P.H., Rezai A.R., editors. Neuromodulation. Elsevier-Academic Press; London: 2009. pp. 743–751. [Google Scholar]
- 22.Kilgore K.L., Keith M.W., Peckham P.H. Stimulation for return of upper and lower extremity function. In: Krames E.S., Peckham P.H., Rezai A.R., editors. Neuromodulation. Elsevier-Academic Press; London: 2009. pp. 767–776. [Google Scholar]
- 23.Vodovnik L., Kralj A., Stanic U., Acimovic R., Gros N. Recent applications of functional electrical stimulation to stroke patients in Ljubljana. Clin Orthop Relat Res. 1978;131:64–70. [PubMed] [Google Scholar]
- 24.Weber D.J., Stein R.B., Chan K.M., Loeb G., Richmond F., Rolf R. BIONic WalkAide for correcting foot drop. IEEE Trans Neural Syst Rehabil Eng. 2005;13:242–246. doi: 10.1109/TNSRE.2005.847385. [DOI] [PubMed] [Google Scholar]
- 25.Kottink A.I., Oostendorp L.J., Buurke J.H., Nene A.V., Hermens H.J., IJzerman M.J. The orthotic effect of functional electrical stimulation on the improvement of walking in stroke patients with a dropped foot: a systematic review. Artif Organs. 2004;28:577–586. doi: 10.1111/j.1525-1594.2004.07310.x. [DOI] [PubMed] [Google Scholar]
- 26.Burridge J.H., Haugland M., Larsen B., Pickering R.M., Svaneborg N., Iversen H.K. Phase II, trial to evaluate the ActiGait implanted drop-foot stimulator in established hemiplegia. J Rehabil Med. 2007;39:212–218. doi: 10.2340/16501977-0039. [DOI] [PubMed] [Google Scholar]
- 27.Kottink A.I., Hermens H.J., Nene A.V., Tenniglo M.J., van der Aa H.E., Buschman H.P. A randomized controlled trial of an implantable 2-channel peroneal nerve stimulator on walking speed and activity in poststroke hemiplegia. Arch Phys Med Rehabil. 2007;88:971–978. doi: 10.1016/j.apmr.2007.05.002. [DOI] [PubMed] [Google Scholar]
- 28.Kottink A.I., Tenniglo M.J., de Vries W.H., Hermens H.J., Buurke J.H. Effects of an implantable two-channel peroneal nerve stimulator versus conventional walking device on spatiotemporal parameters and kinematics of hemiparetic gait. J Rehabil Med. 2012;44:51–57. doi: 10.2340/16501977-0909. [DOI] [PubMed] [Google Scholar]
- 29.Stein R.B., Chong S., Everaert D.G., Rolf R., Thompson A.K., Whittaker M. A multicenter trial of a footdrop stimulator controlled by a tilt sensor. Neurorehabil Neural Repair. 2006;20:371–379. doi: 10.1177/1545968306289292. [DOI] [PubMed] [Google Scholar]
- 30.Graupe D., Kohn K.H. Functional neuromuscular stimulator for short-distance ambulation by certain thoracic-level spinal-cord-injured paraplegics. Surg Neurol. 1998;50:202–207. doi: 10.1016/s0090-3019(98)00074-3. [DOI] [PubMed] [Google Scholar]
- 31.Bijak M., Mayr W., Rakos M., Hofer C., Lanmüller H., Rafolt D. The Vienna functional electrical stimulation system for restoration of walking functions in spastic paraplegia. Artif Organs. 2002;26:224–227. doi: 10.1046/j.1525-1594.2002.06935.x. [DOI] [PubMed] [Google Scholar]
- 32.Bijak M., Rakos M., Hofer C., Mayr W., Strohhofer M., Raschka D. Stimulation parameter optimization for FES supported standing up and walking in SCI patients. Artif Organs. 2005;29:220–223. doi: 10.1111/j.1525-1594.2005.29039.x. [DOI] [PubMed] [Google Scholar]
- 33.Popovic M.R., Keller T., Pappas I.P., Dietz V., Morari M. Surface-stimulation technology for grasping and walking neuroprosthesis. IEEE Eng Med Biol Mag. 2001;20:82–93. doi: 10.1109/51.897831. [DOI] [PubMed] [Google Scholar]
- 34.Gallien P., Brissot R., Eyssette M., Tell L., Barat M., Wiart L. Restoration of gait by functional electrical stimulation for spinal cord injured patients. Paraplegia. 1995;33:660–664. doi: 10.1038/sc.1995.138. [DOI] [PubMed] [Google Scholar]
- 35.Chaplin E. Functional neuromuscular stimulation for mobility in people with spinal cord injuries. The Parastep I System. J Spinal Cord Med. 1996;19:99–105. [PubMed] [Google Scholar]
- 36.Klose K.J., Jacobs P.L., Broton J.G., Guest R.S., Needham-Shropshire B.M., Lebwohl N. Evaluation of a training program for persons with SCI paraplegia using the Parastep 1 ambulation system: part 1. Ambulation performance and anthropometric measures. Arch Phys Med Rehabil. 1997;78:789–793. doi: 10.1016/s0003-9993(97)90188-x. [DOI] [PubMed] [Google Scholar]
- 37.Graupe D., Cerrel-Bazo H., Kern H., Carraro U. Walking performance, medical outcomes and patient training in FES of innervated muscles for ambulation by thoracic-level complete paraplegics. Neurol Res. 2008;30:123–130. doi: 10.1179/174313208X281136. [DOI] [PubMed] [Google Scholar]
- 38.Davis J.A., Jr., Triolo R.J., Uhlir J., Bieri C., Rohde L., Lissy D. Preliminary performance of a surgically implanted neuroprosthesis for standing and transfers—where do we stand? J Rehabil Res Dev. 2001;38:609–617. [PubMed] [Google Scholar]
- 39.Davis R., Patrick J., Barriskill A. Development of functional electrical stimulators utilizing cochlear implant technology. Med Eng Phys. 2001;23:61–68. doi: 10.1016/s1350-4533(01)00023-6. [DOI] [PubMed] [Google Scholar]
- 40.von Wild K., Rabischong P., Brunelli G., Benichou M., Krishnan K. Computer added locomotion by implanted electrical stimulation in paraplegic patients (SUAW) Acta Neurochir Suppl. 2002;79:99–104. doi: 10.1007/978-3-7091-6105-0_22. [DOI] [PubMed] [Google Scholar]
- 41.Cikajlo I., Matjacić Z., Bajd T., Futami R. Sensory supported FES control in gait training of incomplete spinal cord injury persons. Artif Organs. 2005;29:459–461. doi: 10.1111/j.1525-1594.2005.29077.x. [DOI] [PubMed] [Google Scholar]
- 42.Bajd T., Kralj A., Stefancic M., Lavrac N. Use of functional electrical stimulation in the lower extremities of incomplete spinal cord injured patients. Artif Organs. 1999;23:403–409. doi: 10.1046/j.1525-1594.1999.06360.x. [DOI] [PubMed] [Google Scholar]
- 43.Dimitrijevic M.M., Dimitrijevic M.R. Clinical elements for the neuromuscular stimulation and functional electrical stimulation protocols in the practice of neurorehabilitation. Artif Organs. 2002;26:256–259. doi: 10.1046/j.1525-1594.2002.06946.x. [DOI] [PubMed] [Google Scholar]
- 44.Vodovnik L., Bowman B.R., Hufford P. Effects of electrical stimulation on spinal spasticity. Scand J Rehabil Med. 1984;16:29–34. [PubMed] [Google Scholar]
- 45.Stein R.B. Functional electrical stimulation after spinal cord injury. J Neurotrauma. 1999;16:713–717. doi: 10.1089/neu.1999.16.713. [DOI] [PubMed] [Google Scholar]
- 46.Cook A.W. Electrical stimulation in multiple sclerosis. Hosp Pract. 1976;11:51–58. doi: 10.1080/21548331.1976.11706516. [DOI] [PubMed] [Google Scholar]
- 47.Illis L.S., Oygar A.E., Sedgwick E.M., Awadalla M.A. Dorsal-column stimulation in the rehabilitation of patients with multiple sclerosis. Lancet. 1976;1:1383–1386. doi: 10.1016/s0140-6736(76)93030-0. [DOI] [PubMed] [Google Scholar]
- 48.Dooley D.M., Sharkey J. Electrostimulation of the nervous system for patients with demyelinating and degenerative diseases of the nervous system and vascular diseases of the extremities. Appl Neurophysiol. 1977;40:208–217. doi: 10.1159/000102444. [DOI] [PubMed] [Google Scholar]
- 49.Siegfried J., Krainick J.U., Haas H., Adorjani C., Meyer M., Thoden U. Electrical spinal cord stimulation for spastic movement disorders. Appl Neurophysiol. 1978;41:134–141. doi: 10.1159/000102409. [DOI] [PubMed] [Google Scholar]
- 50.Gybels J., van Roost D. Spinal cord stimulation for the modification of dystonic and hyperkinetic conditions: a critical review. In: Eccles J., Dimitrijevic M.R., editors. vol. 1. S Karger AG; Basel: 1985. pp. 58–70. (Upper motor neuron functions and dysfunctions. Recent achievements in restorative neurology). [Google Scholar]
- 51.Davis R., Gray E., Kudzma J. Beneficial augmentation following dorsal column stimulation in some neurological diseases. Appl Neurophysiol. 1981;44:37–49. doi: 10.1159/000102182. [DOI] [PubMed] [Google Scholar]
- 52.Illis L.S., Sedgwick E.M., Tallis R.C. Spinal cord stimulation in multiple sclerosis: clinical results. J Neurol Neurosurg Psychiatry. 1980;43:1–14. doi: 10.1136/jnnp.43.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Tallis R.C., Illis L.S., Sedgwick E.M. The quantitative assessment of the influence of spinal cord stimulation on motor function in patients with multiple sclerosis. Int Rehabil Med. 1983;5:10–16. doi: 10.3109/09638288309166928. [DOI] [PubMed] [Google Scholar]
- 54.Waltz J.M. Chronic stimulation for motor disorders. In: Gindelberg P.L., Tasker R.R., editors. Textbook for stereotactic and functional neurosurgery. McGraw-Hill; New York: 1998. pp. 1087–1099. [Google Scholar]
- 55.Sedgwick E.M., Illis L.S., Tallis R.C., Thornton A.R., Abraham P., El-Negamy E. Evoked potentials and contingent negative variation during treatment of multiple sclerosis with spinal cord stimulation. J Neurol Neurosurg Psychiatry. 1980;43:15–24. doi: 10.1136/jnnp.43.1.15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Dimitrijevic M.R., Gerasimenko Y., Pinter M.M. Evidence for a spinal central pattern generator in humans. Ann NY Acad Sci. 1998;860:360–376. doi: 10.1111/j.1749-6632.1998.tb09062.x. [DOI] [PubMed] [Google Scholar]
- 57.Pinter M.M., Gerstenbrand F., Dimitrijevic M.R. Epidural electrical stimulation of posterior structures of the human lumbosacral cord: 3. Control of spasticity. Spinal Cord. 2000;38:524–531. doi: 10.1038/sj.sc.3101040. [DOI] [PubMed] [Google Scholar]
- 58.Minassian K., Persy I., Rattay F., Pinter M.M., Kern H., Dimitrijevic M.R. Human lumbar cord circuitries can be activated by extrinsic tonic input to generate locomotor-like activity. Hum Mov Sci. 2007;26:275–295. doi: 10.1016/j.humov.2007.01.005. [DOI] [PubMed] [Google Scholar]
- 59.Gerasimenko Y., Roy R.R., Edgerton V.R. Epidural stimulation: comparison of the spinal circuits that generate and control locomotion in rats, cats and humans. Exp Neurol. 2008;209:417–425. doi: 10.1016/j.expneurol.2007.07.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Harkema S., Gerasimenko Y., Hodes J., Burdick J., Angeli C., Chen Y. Effect of epidural stimulation of the lumbosacral spinal cord on voluntary movement, standing, and assisted stepping after motor complete paraplegia: a case study. Lancet. 2011;377:1938–1947. doi: 10.1016/S0140-6736(11)60547-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Richardson R.R., McLone D.G. Percutaneous epidural neurostimulation for paraplegic spasticity. Surg Neurol. 1978;9:153–155. [PubMed] [Google Scholar]
- 62.Richardson R.R., Cerullo L.J., McLone D.G., Gutierrez F.A., Lewis V. Percutaneous epidural neurostimulation in modulation of paraplegic spasticity. Six case reports. Acta Neurochir (Wien) 1979;49:235–243. doi: 10.1007/BF01808963. [DOI] [PubMed] [Google Scholar]
- 63.Siegfried J., Lazorthes Y., Broggi G. Electrical spinal cord stimulation for spastic movement disorders. Appl Neurophysiol. 1981;44:77–92. doi: 10.1159/000102187. [DOI] [PubMed] [Google Scholar]
- 64.Dimitrijevic M.M., Dimitrijevic M.R., Illis L.S., Nakajima K., Sharkey P.C., Sherwood A.M. Spinal cord stimulation for the control of spasticity in patients with chronic spinal cord injury: I. Clinical observations. Cent Nerv Syst Trauma. 1986;3:129–144. doi: 10.1089/cns.1986.3.129. [DOI] [PubMed] [Google Scholar]
- 65.Barolat G., Myklebust J.B., Wenninger W. Effects of spinal cord stimulation on spasticity and spasms secondary to myelopathy. Appl Neurophysiol. 1988;51:29–44. doi: 10.1159/000099381. [DOI] [PubMed] [Google Scholar]
- 66.Barolat G., Singh-Sahni K., Staas W.E., Jr., Shatin D., Ketcik B., Allen K. Epidural spinal cord stimulation in the management of spasms in spinal cord injury: a prospective study. Stereotact Funct Neurosurg. 1995;64:153–164. doi: 10.1159/000098744. [DOI] [PubMed] [Google Scholar]
- 67.Dimitrijevic M.R., Illis L.S., Nakajima K., Sharkey P.C., Sherwood A.M. Spinal cord stimulation for the control of spasticity in patients with chronic spinal cord injury: II. Neurophysiologic observations. Cent Nerv Syst Trauma. 1986;3:145–152. doi: 10.1089/cns.1986.3.145. [DOI] [PubMed] [Google Scholar]
- 68.Murg M., Binder H., Dimitrijevic M.R. Epidural electric stimulation of posterior structures of the human lumbar spinal cord: 1. Muscle twitches – a functional method to define the site of stimulation. Spinal Cord. 2000;38:394–402. doi: 10.1038/sj.sc.3101038. [DOI] [PubMed] [Google Scholar]
- 69.Marino R.J., Barros T., Biering-Sorensen F., Burns S.P., Donovan W.H., Graves D.E. International standards for neurological classification of spinal cord injury. J Spinal Cord Med. 2003;26:S50–S56. doi: 10.1080/10790268.2003.11754575. ASIA Neurological Standards Committee 2002. [DOI] [PubMed] [Google Scholar]
- 70.Sherwood A.M., McKay W.B., Dimitrijevic M.R. Motor control after spinal cord injury: assessment using surface EMG. Muscle Nerve. 1996;19:966–979. doi: 10.1002/(SICI)1097-4598(199608)19:8<966::AID-MUS5>3.0.CO;2-6. [DOI] [PubMed] [Google Scholar]
- 71.Davidoff R.A. The dorsal columns. Neurology. 1989;39:1377–1385. doi: 10.1212/wnl.39.10.1377. [DOI] [PubMed] [Google Scholar]
- 72.Gerasimenko Y., Daniel O., Regnaux J., Combeaud M., Bussel B. Mechanisms of locomotor activity generation under epidural spinal cord stimulation. In: Dengler R., Kossev A.R., editors. Sensorimotor control. IOS Press; Washington, DC: 2001. pp. 164–171. [Google Scholar]
- 73.Gerasimenko Y.P., Makarovskii A.N., Nikitin O.A. Control of locomotor activity in humans and animals in the absence of supraspinal influences. Neurosci Behav Physiol. 2002;32:417–423. doi: 10.1023/a:1015836428932. [DOI] [PubMed] [Google Scholar]
- 74.Minassian K., Jilge B., Rattay F., Pinter M.M., Binder H., Gerstenbrand F. Stepping-like movements in humans with complete spinal cord injury induced by epidural stimulation of the lumbar cord: electromyographic study of compound muscle action potentials. Spinal Cord. 2004;42:401–416. doi: 10.1038/sj.sc.3101615. [DOI] [PubMed] [Google Scholar]
- 75.Jilge B., Minassian K., Rattay F., Pinter M.M., Gerstenbrand F., Binder H. Initiating extension of the lower limbs in subjects with complete spinal cord injury by epidural lumbar cord stimulation. Exp Brain Res. 2004;154:308–326. doi: 10.1007/s00221-003-1666-3. [DOI] [PubMed] [Google Scholar]
- 76.Rattay F., Minassian K., Dimitrijevic M.R. Epidural electrical stimulation of posterior structures of the human lumbosacral cord: 2. Quantitative analysis by computer modeling. Spinal Cord. 2000;38:473–489. doi: 10.1038/sj.sc.3101039. [DOI] [PubMed] [Google Scholar]
- 77.Ladenbauer J., Minassian K., Hofstoetter U.S., Dimitrijevic M.R., Rattay F. Stimulation of the human lumbar spinal cord with implanted and surface electrodes: a computer simulation study. IEEE Trans Neural Syst Rehabil Eng. 2010;18:637–645. doi: 10.1109/TNSRE.2010.2054112. [DOI] [PubMed] [Google Scholar]
- 78.Minassian K., Persy I., Rattay F., Dimitrijevic M.R. Effect of peripheral afferent and central afferent input to the human lumbar spinal cord isolated from brain control. Biocyber Biomed Eng. 2005;25:11–29. [Google Scholar]
- 79.Barolat G., Myklebust J.B., Wenninger W. Enhancement of voluntary motor function following spinal cord stimulation—case study. Appl Neurophysiol. 1986;49:307–314. doi: 10.1159/000100160. [DOI] [PubMed] [Google Scholar]
- 80.Herman R., He J., D’Luzansky S., Willis W., Dilli S. Spinal cord stimulation facilitates functional walking in a chronic, incomplete spinal cord injured. Spinal Cord. 2002;40:65–68. doi: 10.1038/sj.sc.3101263. [DOI] [PubMed] [Google Scholar]
- 81.Carhart M.R., He J., Herman R., D’Luzansky S., Willis W.T. Epidural spinal-cord stimulation facilitates recovery of functional walking following incomplete spinal-cord injury. IEEE Trans Neural Syst Rehabil Eng. 2004;12:32–42. doi: 10.1109/TNSRE.2003.822763. [DOI] [PubMed] [Google Scholar]
- 82.Ganley G.J., Willis W.T., Carhart M.R., He J., Herman R.M. Epidural spinal cord stimulation improves locomotor performance in low ASIA C, wheelchair-dependent, spinal cord-injured individuals: Insights from metabolic response. Top Spinal Cord Inj Rehabil. 2005;11:50–63. [Google Scholar]
- 83.Huang H., He J., Herman R., Carhart M.R. Modulation effects of epidural spinal cord stimulation on muscle activities during walking. IEEE Trans Neural Syst Rehabil Eng. 2006;14:14–23. doi: 10.1109/TNSRE.2005.862694. [DOI] [PubMed] [Google Scholar]
- 84.Danner S., Rattay F., Bijak M., Mayr W., Minassian K., Hofstoetter U.S. Society for Neuroscience; Washington, DC: 2011. Human lumbar cord can process spinal cord stimulation of different frequencies. Program no. 182. 06. Neuroscience meeting planner. [Online] [Google Scholar]
- 85.Holsheimer J. Which neuronal elements are activated directly by spinal cord stimulation. Neuromodulation. 2002;5:25–31. doi: 10.1046/j.1525-1403.2002._2005.x. [DOI] [PubMed] [Google Scholar]
- 86.Minassian K., Persy I., Rattay F., Dimitrijevic M.R., Hofer C., Kern H. Posterior root-muscle reflexes elicited by transcutaneous stimulation of the human lumbosacral cord. Muscle Nerve. 2007;35:327–336. doi: 10.1002/mus.20700. [DOI] [PubMed] [Google Scholar]
- 87.Hofstoetter U.S., Minassian K., Hofer C., Mayr W., Rattay F., Dimitrijevic M.R. Modification of reflex responses to lumbar posterior root stimulation by motor tasks in healthy subjects. Artif Organs. 2008;32:644–648. doi: 10.1111/j.1525-1594.2008.00616.x. [DOI] [PubMed] [Google Scholar]
- 88.Minassian K., Hofstoetter U.S., Rattay F. Transcutaneous lumbar posterior root stimulation for motor control studies and modification of motor activity after spinal cord injury. In: Dimitrijevic M.R., Kakulas B.A., McKay W.B., Vrbova G., editors. Restorative neurology of spinal cord injury. Oxford University Press; New York: 2011. pp. 226–255. [Google Scholar]
- 89.Danner S.M., Hofstoetter U.S., Ladenbauer J., Rattay F., Minassian K. Can the human lumbar posterior columns be stimulated by transcutaneous spinal cord stimulation? A modeling study. Artif Organs. 2011;35:257–262. doi: 10.1111/j.1525-1594.2011.01213.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Szava Z., Danner S.M., Minassian K. VDM Verlag Dr. Müller; Saarbrücken: 2011. Transcutaneous electrical spinal cord stimulation: biophysics of a new rehabilitation method after spinal cord injury. 104 pp. [Google Scholar]
- 91.Hofstoetter U., Mayr W., Rattay F., Dimitrijevic M.R., Minassian K. Society for Neuroscience; Washington, DC: 2011. Effects of transcutaneous spinal cord stimulation on spasticity electrophysiologically evaluated in spinal cord injured individuals. Program No. 808. 02. Abstract Viewer/Itinerary Planner. [Google Scholar]
- 92.Minassian K., Hofstoetter U., Tansey K., Rattay F., Mayr W., Dimitrijevic M.R. Society for Neuroscience; San Diego, CA: 2010. Transcutaneous stimulation of the human lumbar spinal cord: Facilitating locomotor output in spinal cord injury. Program No. 286. 19. Neuroscience Meeting Planner. [Online] [Google Scholar]
- 93.Dietz V., Colombo G., Jensen L., Baumgartner L. Locomotor capacity of spinal cord in paraplegic patients. Ann Neurol. 1995;37:574–582. doi: 10.1002/ana.410370506. [DOI] [PubMed] [Google Scholar]
- 94.Vrbova G., Slawinska U. Summary of strategies used to repair the injured spinal cord. In: Dimitrijevic M.R., Kakulas B.A., McKay W.B., Vrbova G., editors. Restorative neurology of spinal cord injury. Oxford University Press; New York: 2011. pp. 93–133. [Google Scholar]
- 95.Dimitrijevic M.R., Larsson L.E. Neural control of gait: clinical neurophysiological aspects. Appl Neurophysiol. 1981;44:152–159. doi: 10.1159/000102195. [DOI] [PubMed] [Google Scholar]
- 96.Eccles J., Dimitrijevic M.R. vol. 1. S Karger AG; Basel: 1985. (Recent achievements in restorative neurology, upper motor neuron functions and dysfunctions). 346 pp. [Google Scholar]
- 97.Illis L.S. Is there a central pattern generator in man? Paraplegia. 1995;33:239–240. doi: 10.1038/sc.1995.54. [DOI] [PubMed] [Google Scholar]
- 98.Courtine G., van den Brand R., Musienko P. Spinal cord injury: time to move. Lancet. 2011;377:1896–1898. doi: 10.1016/S0140-6736(11)60711-3. [DOI] [PubMed] [Google Scholar]
- 99.Edgerton V.R., Leon R.D., Harkema S.J., Hodgson J.A., London N., Reinkensmeyer D.J. Retraining the injured spinal cord. J Physiol. 2001;533:15–22. doi: 10.1111/j.1469-7793.2001.0015b.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Dimitrijevic M.R. Chronic spinal cord stimulation in spasticity. In: Gindelberg P.L., Tasker R.R., editors. Textbook for stereotactic and functional neurosurgery. McGraw-Hill; New York: 1998. pp. 1267–1273. [Google Scholar]
- 101.Bamford J.A., Mushahwar V.K. Intraspinal microstimulation for the recovery of function following spinal cord injury. Prog Brain Res. 2011;194:227–239. doi: 10.1016/B978-0-444-53815-4.00004-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Edgerton V.R., Harkema S. Epidural stimulation of the spinal cord in spinal cord injury: current status and future challenges. Expert Rev Neurother. 2011;11:1351–1353. doi: 10.1586/ern.11.129. [DOI] [PMC free article] [PubMed] [Google Scholar]