Abstract
We present the case of a 58-year-old woman with a renin secreting typical bronchopulmonary carcinoid. This patient showed hypotension, constipation and fatigue due to extensive hypokaliemia (K =1.9 meq/L). Aldosterone (102.7 ng/100 mL) and renin (46 ng/mL) were excessively elevated at that time, but cortisol level was normal. Routine chest roentgenography and computed tomography revealed a nodular lesion in the upper left lung lobe, which was suspicious for a neurosecretory pulmonary tumor. The final diagnosis was made by using bronchoscopic procedures and the histologically diagnosis was compatible as a typical pulmonary carcinoid. The tumor was resected curatively, and the renin and aldosterone level became normal. A year after the patient looks healthy.
Keywords: pulmonary carcinoid, renin, hypokaliemia, neurosecretory pulmonary tumor
Case Report
A 58 year-old woman entered our hospital, with symptoms of constipation, excessive urine secretion (over 2 liter/day—polyouria) and severe muscle fatigue. She had a negative medical history and she hadn’t used alcohol or drugs of any type. She complained of persistent dry cough and anorexia— her body weight had decreased 10 kgs during the last two months period. On evaluation, the patient looked sick; she was pale, her temperature was 36.1 °C, her blood pressure was 100/60 mmHg, and her pulse rate was 70 bpm. The clinical examination revealed total muscle fatigue of arms and legs, without specific neurological signs. The patient couldn’t walk or stand without help. She said that she had remained in bed during the main part of the day for the last week.
The initial laboratory data were: Hct =31%, Hb =10.9 g/dL, Wbc =20.250 k/μL; (91% were granulocytes and 9% were lymphocytes), Platelets =438.000/μL, Glu =140 mg/dL, BUN =44 mg/dL, Creatine =0.9 mg/dL, K =1.9 meq/L, Na =140 meq/L, Ca =9.6 mg/dL, SGOT =11 IU/L, SGPT =15 IU/L, ALP =85 IU/L, LDH =147 IU/L, CPK =53 IU/L, Bill =0,4 mg/dL. Her arterial blood gasses were pH =7.48, pO2 =73 mmHg, pCO2 =44 mmHg, HCO3 =36.5, SpO2 =94%. All these demonstrated metabolic alkalosis.
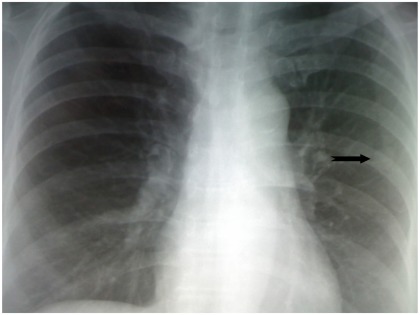
The chest X-ray that was performed, demonstrated opacity without cavitation to the left upper lung lobe (Fig. 1). During next day, the patient was extremely tired, febrile and her breath frequency exceeded 22 per minute (tachypnea). Finally, she got intubated because of severe respiratory (hypercapnic- elevated PCO2) acidosis. The measurement of 24 hour-urine secretion of sodium (108,2 mEq/L) and potassium (34,8 mEq/L), showed extensive hyperkaliurea. On the contrary, urine 5-HIAA was normal (5 mg; normal range: 5–100 mg). After 3 days, the patient extubated, without respiratory muscle weakness, while she had received intravenously high amount of solutions with potassium. The patient remained with low serum potassium (hypokaliemia), despite the extensive intravenous replacement. We measured the serum levels of aldosterone, aldolase, cortisone, and renin in order to find out a responsible specific disease for this condition. The laboratory data are presented in Table 1. We considered as possible the existence of paraneoplasmatic syndrome.
Figure 1.
Chest X-ray revealed a nodule without cavitations to the upper lung lobe of the left lung.
Table 1.
Levels of commonest hormones which are associated with potassium homeostasis.
| 1st measurement | 2nd measurement (after therapy) | Normal values | |
|---|---|---|---|
| Aldosterone | 102.7 ng/100 mL | 5.6 ng/100 mL | 0.7–15 |
| ACTH | 14 ng/100 mL | 23 ng/100 mL | 5–25 |
| Cortisol | 15 μg/100 mL | 22.7 μg/100 mL | 5–25 |
| Renin | 46 ng/mL | 2.7 ng/mL | 0.2–2.7 |
The CT scanning of the thorax revealed a lung nodule, at the upper anterior left lung lobe, without collateral lymphadenopathy (Fig. 2). The CT scanning of the brain, and upper and lower abdomen departments, were negative for any kinds of lesions (secondary metastatic sites) and excluded the coexistence of juxtaglomerular tumors.
Figure 2.
Thorax CT scanning revealed a left lung nodule (diameter 21 ×14 mm), at the upper anterior lung lobe without collateral lymphadenopathy.
The values of tumor markers were within normal range. Urine examination was also normal. Smear examination and sputum culture were negative for Mucobacterium tuberculosis and the tuberculin skin test was negative too.
The patient underwent a fiberoptic bronchoscopy. We found a highly vascular endobronchial mass with a reddish smooth-looking surface, into the right anterior sub segmental bronchus of the left upper lung lobe. The bronchial biopsy revealed numerous membrane-bound neurosecretory granules characteristic of neuroendocrine tumor (pulmonary carcinoid tumor).
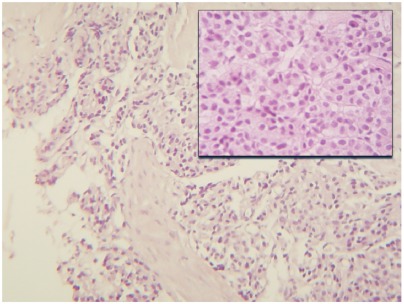
Three days later, we planned the surgical wedge resection of the carcinoid. The extended histopathology study revealed the pattern of the typical carcinoid tumor of the lung, because of the absence of necrosis. (Fig. 3).
Figure 3.
Photomicrograph shows small nests of uniform high-grade epithelial endocrine cells, without necrosis or mitosis (<2 mm2), classified as a typical carcinoid tumor of the lung (original magnification, ×100 and ×300; hematoxylin-eosin stain).
Two weeks after the surgical procedure, the patient’s blood pressure was measured and was found normal and the renin activity-levels serum were found normal, too (Table 1).
Discussion
We present, for the first time, a female patient with hypokalemia due to renin secretion from a bronchial carcinoid tumor. After surgical resection of the tumour, the patient became asymptomatic, and the serum renin level was decreased to normal values; these findings strongly suggested that the clinical symptoms and signs were due to tumor over-secretion of renin.
The estimated annual incidence of carcinoid tumors is 1–2 cases per 100,000 people.1 In fact, the overall incidence of carcinoid tumors seems to have sharply increased over the past few decades, perhaps, due to a more accurate diagnosis, supported by new diagnostic techniques.2,3 The sex distribution of overall incidence of carcinoid tumors is almost equal. However, in those aged <50 years, the carcinoid incidence in females is almost twice than in males.3
Bronchial carcinoid tumors are considered a low-grade malignant neoplasm comprised of neuroendocrine cells. These tumors comprise 25% of all carcinoid tumors but only 5% of all primary lung cancers.4,5 About 90% of the carcinoid tumors are well differentiated with rare mitoses, pleomorphism, and necrosis.6 These are referred as “typical” carcinoid tumors7 and the other 10% is referred as “atypical”.
Carcinoid tumours have the ability to secrete vasoactive peptides, which are initially responsible for clinical presentation and complications. Carcinoids have been found to secrete serotonin (5-hydroxytryptamine; 5-HT), bradykinins, tachykinins, histamine, substance P, adrenocorticotropic hormone, prostaglandins, kallikrein, dopamine and several other peptides, although 5-HT production is the most prominent.1,8 There was no data related to renin secretion from these tumors,9 as was in the case with our patient.
The carcinoid syndrome—a paraneoplasmatic syndrome—is very rare in patients with pulmonary carcinoids, occurring in approximately 2% of cases. Its characteristic symptoms are diarrhoea, flushing, palpitations and, less frequently, wheezing. The severity of these symptoms is related both to the location of the tumour (related to drainage to systemic circulation, especially in liver metastases) and to the tumour mass.2,3 The 5-HT is the most frequently detected peptide responsible for carcinoid syndrome. In rare cases, carcinoid crisis is induced by the release of a large concentration of vasoactive substances into the systemic circulation, after tumour manipulation, induction of anaesthesia or intubation.1–3 Contradicting these clinical signs, our patient had marked hypotension, without any hypertension crisis, and her serum levels of the 5-HT were in a normal range. Carcinoid syndrome with hypertension crisis due to tumor release of enormous amount of renin has never been described before.10,11
Another paraneoplastic syndrome is Cushing’s Syndrome (CS), which is present in approximately 2% of patients with pulmonary carcinoid. This is characterized by ectopic adrenocorticotropic hormone (ACTH) production, resulting in hypercortisolism. Ectopic ACTH production resulting in CS is seen in a small number of malignancies. Bronchial carcinoid is the most frequent cause (25%), whereas small cell lung cancer (11%) is the second one.12 Frequent signs of hypercortisolism are weakness, hypertension, glucose intolerance, hypokalemia, alkalosis, weight loss, anemia, and hyperpigmentation. The typical features of CS are more frequent in slowly progressing tumors. Our patients had no signs of CS and her ACTH levels were in the normal range.
The renin-angiotensin-aldosterone axis regulates the molecular pathway of sodium reabsorption in the cortical collecting duct (CD). The CD, which is sited at the end of the nephron, played the critical role in the reabsorption of sodium via the aldosterone-activated mineralocorticoid receptor (MR). Aldosterone, in response to renin and angiotensin governs the overall expression of critical proteins for this purpose. In addition, both the CD and the Thick Ascending Limb of the Loop of Henle (TALLH) has the main role of potassium and protons secretion.13 Pathophysiologically, hypokalemia and metabolic alkalosis can be noted due to the compensatory hyperreninemic hyperaldosteronism, as was the case with our patient (See Fig. 4).
Figure 4.
The renin-angiotensin-aldosterone axis and molecular pathways of sodium reabsorption in the nephron.
Notes: A cartoon of a nephron is shown, with sodium-reabsorbing cells of the thick ascending limb of the loop of Henle (TALLH), distal convoluted tubule (DCT), and collecting duct (CD). The latter is responsible for fine regulation via the aldosterone-activated mineralocorticoid receptor (MR). In all cells, sodium exits the basolateral compartment via the Na/K-ATPase (not shown).19 Renin levels stimulate the the axis initiating the transformation of angiotensinogen to angiotensin I. Aldosterone synthesis is controlled by angiotensin II. The figure taken and adjusted from reference 19.
Abbreviations: Na, sodium; K, potassium; Cl, chloride.
The appropriate therapeutic decision for patients with bronchial carcinoid tumors is made on the basis of three components: histologic findings, clinical staging with bronchoscopy and computed tomography, and, in some instances, is based on intraoperative lymph node staging. Most tumors are localized and the likelihood of local recurrence after resection is small. This resulted in a 5-year survival nearly 90% of patients with localized disease and 70% in patients with regionally advanced or metastatic disease.14–16 Limited resections such as wedge resection or segmentectomy for peripheral tumors and isolated bronchial sleeve resection or sleeve lobectomy for proximal tumors should be considered, when feasible for early stage typical bronchial carcinoid tumors, because local recurrence is unlikely and survival is excellent.17,18 Our patient after the wedge resection procedure was healthy and needed no additional treatment. After one year follow up, the patient’s x-ray chest film was without abnormal findings, and her renin serum level was normal.
Footnotes
Author Contributions
VP, DD, LS and SM were responsible for data collection/entry/analysis and documentation. KA and KI were responsible for the study design and preparation of the manuscript. GA and ST were responsible for the coordination of all authors and for the final preparation of the manuscript. All authors read and approved the final manuscript.
Disclosures and Ethics
As a requirement of publication author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest.
References
- 1.Kulke MH, Mayer RJ. Carcinoid tumours. N Engl J Med. 1999;340:858–68. doi: 10.1056/NEJM199903183401107. [DOI] [PubMed] [Google Scholar]
- 2.Zuetenhorst JM, Taal BG. Metastatic carcinoid tumours: a clinical review. Oncologist. 2005;10:123–31. doi: 10.1634/theoncologist.10-2-123. [DOI] [PubMed] [Google Scholar]
- 3.Hage R, de la Rivière AB, Seldenrijk CA, van den Bosch JMM. Update in pulmonary carcinoid tumours: a review article. Ann Surg Oncol. 2003;10:697–704. doi: 10.1245/aso.2003.09.019. [DOI] [PubMed] [Google Scholar]
- 4.Davila DG, Dunn WF, Tazelaar HD, Pairolero PC. Bronchial carcinoid tumors. Mayo Clin Proc. 1993;68:795–803. doi: 10.1016/s0025-6196(12)60641-7. [DOI] [PubMed] [Google Scholar]
- 5.Modlin IM, Sandor A. An analysis of 8305 cases of carcinoid tumors. Cancer. 1997;79:813–29. doi: 10.1002/(sici)1097-0142(19970215)79:4<813::aid-cncr19>3.0.co;2-2. [DOI] [PubMed] [Google Scholar]
- 6.Davila DG, Dumm WF, Tazelaar HD, et al. Bronchial carcinoid tumors. Mayo Clin Proc. 1993;68:795–803. doi: 10.1016/s0025-6196(12)60641-7. [DOI] [PubMed] [Google Scholar]
- 7.Hamperl H. Über gutartige bronchialtumoren (cylindrome und carcinoid) Virchows Arch Pathol Anat. 1937;300:46–88. [Google Scholar]
- 8.Lips CJM, Lentjes EGWM, Höppener JWM. The spectrum of carcinoid tumours and carcinoid syndromes. Ann Clin Biochem. 2003;40:612–27. doi: 10.1258/000456303770367207. [DOI] [PubMed] [Google Scholar]
- 9.Fink G, Krelbaum T, Yellin A, et al. pulmonary carcinoid. Presentation, diagnosis, and outcome in 142 cases in Israel and review of 640 cases from the literature. Chest. 2001;119:1647–51. doi: 10.1378/chest.119.6.1647. [DOI] [PubMed] [Google Scholar]
- 10.Rea F, Binda F, Spreafico G, et al. Bronchial carcinoids: a review of 60 patients. Annals of Thoracic Surgery. 1989;47:412–4. doi: 10.1016/0003-4975(89)90383-4. [DOI] [PubMed] [Google Scholar]
- 11.Vadasz P, Palffy G, Egervary M, Schaff Z. Diagnosis and treatment of bronchial carcinoid tumors: clinical and pathological review of 120 operated patients. European Journal of Cardio-Thoracic Surgery. 1993;7:8–11. doi: 10.1016/1010-7940(93)90140-7. [DOI] [PubMed] [Google Scholar]
- 12.Aniszewski JP, Young WF, Jr, Thompson GB, Grant CS, Van Heerden JA. Cushing syndrome due to ectopic adrenocorticotropic hormone secretion. World J Surg. 2001;25:934–40. doi: 10.1007/s00268-001-0032-5. [DOI] [PubMed] [Google Scholar]
- 13.Kleta R, Bockenhauer D. Bartter Syndromes and Other Salt-Losing Tubulopathies. Nephron Physiol. 2006;104:73–80. doi: 10.1159/000094001. [DOI] [PubMed] [Google Scholar]
- 14.Ferguson MK, Landreneau RJ, Hazelrigg SR, et al. Long-term outcome after resection for bronchial carcinoid tumors. Eur J Cardiothorac Surg. 2000 Aug;18(2):156–61. doi: 10.1016/s1010-7940(00)00493-0. [DOI] [PubMed] [Google Scholar]
- 15.Hurt R, Bates M. Carcinoid tumours of the bronchus: a 33 year experience. Thorax. 1984;39:617–23. doi: 10.1136/thx.39.8.617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Perkins P, Kemp BL, Putnam JB, Jr, Cox JD. Pretreatment characteristics of carcinoid tumors of the lung which predict aggressive behavior. Am J Clin Oncol. 1997;20:285–8. doi: 10.1097/00000421-199706000-00016. [DOI] [PubMed] [Google Scholar]
- 17.Travis WD, Rush W, Flieder DB, et al. Survival analysis of 200 pulmonary neuroendocrine tumors with clarification of criteria for atypical carcinoid and its separation from typical carcinoid. Am J Surg Pathol. 1998;22:934–44. doi: 10.1097/00000478-199808000-00003. [DOI] [PubMed] [Google Scholar]
- 18.Chughtai TS, Morin JE, Sheiner NM, Wilson JA, Mulder DS. Bronchial carcinoid—twenty years’ experience defines a selective surgical approach. Surgery. 1997;122:801–8. doi: 10.1016/s0039-6060(97)90090-8. [DOI] [PubMed] [Google Scholar]
- 19.O’Shaughnessy Kevin M, Karet Fiona E. Salt handling and hypertension. J Clin Invest. 2004;113(8):1075–81. doi: 10.1172/JCI21560. [DOI] [PMC free article] [PubMed] [Google Scholar]