EMBO reports advance online publication April 10 2012; doi:; DOI: 10.1038/embor.2012.50
We all exist because of our parent's fertility; yet in the grand scheme of evolution, fertility is a crucial selection factor that has determined the future of our own and many other species. By animal standards, humans have remarkably poor fertility, although we have nonetheless managed to ‘overpopulate’ the planet. However, we now face the prospect that the population of the world, and of individual countries, will begin to contract as more and more countries move below the ‘population replacement’ level for birth rates. This is the situation for the UK, the EU, and most developed and developing nations [1]. As a country develops in terms of public health, economic progress and education rate, so its birth-rate drops, and this drop has been happening ever-faster in developing countries in recent decades with the advent of improved communication and education.
One aspect that compounds the negative impact of a low sperm count is that it is also frequently associated with reduced sperm quality
At face value, fewer humans on the planet sounds like an attractive prospect: for example, there will be less pressure on resources and the environment. But it will also bring unprecedented social and financial challenges as the age structure of the population bulges at the aged end rather than at the young end. The dependence of the increasingly longer-living aged on the fewer young for financial, social and health support will create problems for modern societies the consequences of which are unknown. Equally important, there will be fewer taxpayers to provide governments with the necessary finances for running their countries.
This ‘brave new world’ scenario is an appropriate way to introduce the subject of couple fertility—male fertility in particular—because it illustrates that although fertility or infertility is an issue of huge personal importance for couples and individuals, it is even more important for nations and, indeed, the human race. What this article hopes to demonstrate is that in addition to the social trends above, a biological factor is now playing a role—at least in Europe—and that is declining male sperm counts, which might exacerbate the ongoing socially determined changes. Falling sperm counts have the potential to distort and worsen the fertility and ultimate birth rate of EU nations.
Yet the effect of declining sperm counts in men on couple fertility has been obscured by prominent social changes, such as the career aspirations of women, which have had a major negative impact on birth rates and family size. Only one study has so far sought and found evidence that declining sperm counts are impairing conception rates [2], but as this article demonstrates, this hidden decline is predictable, given all that we know about the determinants of couple fertility.
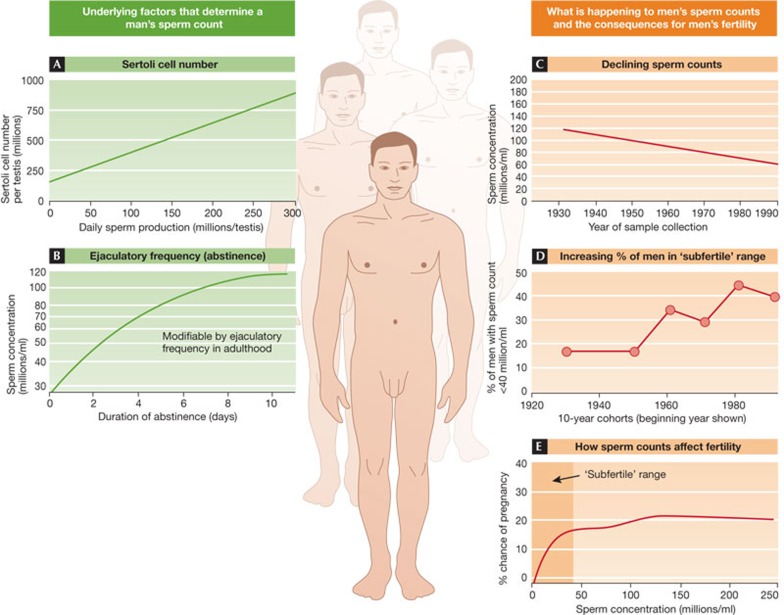
There are two main factors that determine a man's sperm count at any given time. These are the number of Sertoli cells in his testes (Fig 1A) and the time since last ejaculation (abstinence) (Fig 1B). Both clearly have major effects on sperm count, but the big difference is that abstinence is variable, whereas Sertoli cell number is fixed early in development [3].
Figure 1. Sperm counts—the key factors and issues.
On the left are shown the two main factors (A,B) determining sperm count in an individual, while the right panels show the temporal change in sperm counts (C,D) and the relationship between sperm count and couple fertility (E). Full details are given in the text. The illustrated data have been adapted from the following references: A [28], B [29], C [6], D [5,9], E [4].
Sperm are produced continuously in the testes after puberty, with each sperm taking approximately 10 weeks to manufacture—this can be termed the ‘supply side’. The frequency of ejaculation determines the rate at which these sperm are used up, and it is the balance between supply and demand (ejaculation) that determines sperm count at any point in time in a man. In most animals, the situation is different because there is a third factor at work: sperm storage. In many species, sperm are stored to maintain a uniformly high sperm count even with a high ejaculatory frequency. In man, there is no storage, so sperm count is essentially a reflection of production rate, albeit modified by abstinence period [3].
This difference between humans and other animals might sound incidental, but it is fundamentally important with regard to fertility. As shown in Fig 1E, as sperm counts in men rise from zero to40 million/ml of ejaculate, there is a progressive increase in the chances of their partner becoming pregnant; at sperm counts greater than 40 million/ml there is no further benefit regarding fertility [4]. So, for example, the study on which Fig 1E is based, found that during the six-month period of the study of 430 couples, 65% of men with sperm counts >40 million/ml impregnated their partners, while for men with counts <40 million/ml, the rate was 51.2%; corresponding figures for sperm counts >20 or <20 million/ ml were 65.0% and 36.4% [4]. Therefore, having a low sperm count makes you less fertile, although it does not exclude the possibility that you will impregnate your partner over a span of time, unless your sperm count is zero.
One aspect that compounds the negative impact of a low sperm count is that it is also frequently associated with reduced sperm quality, including less motility or abnormal shape. It should also be mentioned that even in a normal fertile man, only a minority of sperm can be classed as morphologically normal (5–15% depending on the criteria used), in comparison with values usually in excess of 90% in most animals. With this statistic in mind, it is not difficult to see why humans have poor fertility.
As shown In Fig 1C, average sperm counts reported in large numbers of men in 101 studies across the world have shown a progressive decline since the 1930s–1940s [5,6]. These data derive only from studies of men without known fertility problems, and might therefore overestimate average sperm counts in the population. Perhaps a more relevant statistic is the change in proportion of men in these studies with a sperm count <40 million/ml, which has increased from around 15% in the 1930s to around 40% in the 1990s–2000 (Fig 1D). The obvious conclusion to draw is that an increased percentage of men will probably experience difficulty in impregnating their partners compared with more than 50 years ago, even if it only means a longer time spent trying for a pregnancy. So does it matter?
Today, a single sperm is sufficient for fertilization if it is literally injected into the egg using ICSI (intracytoplasmic sperm injection), so the question takes on new meaning. Infertility, especially when associated or caused by low sperm count, is widely viewed as a solvable problem—never mind that ICSI fails more often than it succeeds, is hugely expensive and creates considerable psychological and other stresses for the couple [7]. Thus, although widely used, ICSI does not resolve infertility for many couples. This ‘problem solved’ mindset might be one reason why, 20 years on from initial reports of ‘falling sperm counts’, the ‘problem’ has been repeatedly challenged, whether on grounds of validity or importance [8].
Even so, no viable, evidence-based alternative explanation has emerged for the declining sperm counts shown in Fig 1C. Moreover, a large series of standardized, prospective studies in Europe involving men between 18 and 25 years old from seven countries has confirmed that although average sperm counts are in the range of 45–65 million/ml, depending on the country, the proportion of young men with a sperm count of less than 20 million/ml is still close to 20% [9]. Therefore, irrespective of whether sperm counts have decreased, a substantial number of young men in the next generation have sperm counts within the ‘subfertile range’. Yet even the relevance of this has been challenged [8], despite the fact that one in seven couples experience ‘infertility’ problems—that is, no pregnancy after 12–18 months of trying—and that in at least half of these cases, the problem is identified as a ‘male factor’: most commonly a low sperm count.
This raises the frequently overlooked point that infertility involves two people. Male factor issues such as low sperm count have to be seen in the context of female fertility, which is unarguably also on the decline for social and career reasons; this is reflected in the progressive increase in age at first pregnancy across the developed world.
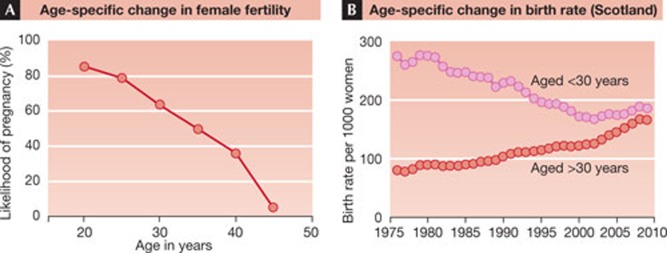
Consider the following present-day scenario of a modern infertile couple. They are both 37, and he has a low sperm count of 20 million/ml. At 37, her likelihood of becoming pregnant is about 50%, compared with roughly 80% in her 20s (Fig 2A). With time, her partner might impregnate her, but time is not on her side as her fertility is already declining. As a couple they are presently infertile, but if she had a partner with a normal or high sperm count, she might conceive more easily. Similarly, if he was partnered with a younger woman with high fertility, he might also be fertile as part of that couple. So, inadvertently, the recent societal trend towards later age of first and later pregnancies in women exacerbates the impact of the high prevalence of low sperm counts in men. As seen in Fig 2B—which shows time trends in age-specific birth rates for Scotland—in 1976, births in women below 30 outnumbered births in women over 30 by more than a factor of 3. Yet, just 30 years later, the number of births in these two age groups is nearly equivalent. Viewed another way, an increasing number of women are waiting to start a family until an age when their fertility is declining.
Figure 2. Age-specific change in female fertility.
(A) in relation to temporal changes in age-specific birth rates in women (B). Data in A are based on several studies in the literature whereas data in B are extracted from the Information Services Division website of the general registry office for Scotland.
So the answer to the question ‘does sperm count matter?’ is yes, it does matter, and probably more than a few decades ago when most women embarked on families at an earlier age. But sperm count also matters for men for reasons even more fundamental than fertility. It is a barometer of overall health; the lower your sperm count the greater your risk of dying [10]. The other barometer of healthy testis function—testosterone levels in blood—shows a similar relationship [11]. Therefore, irrespective of fertility issues, healthy testis function and a high sperm count is a measure of population health for men. The fact that both sperm counts (Fig 1C) and testosterone levels [12] have been declining in men in recent decades suggests that male health might have also declined, which does not bode well for today's young men. Thus, there is a great incentive to understand what has caused lower sperm counts in men, and to establish whether this trend can be reversed or prevented.
Falling sperm counts have been presented by the media as a scare story about environmental chemical pollution, although, in reality, the causes remain unknown. The fact that sperm counts have fallen across a short timescale of 50–70 years (Fig 1C) suggests that the causes must be lifestyle and environmental, rather than genetic. This also means that the decline is probably preventable, and possibly reversible. For this to happen, the problem has to be recognized, its causes elucidated and appropriate intervention or prevention implemented. To identify the causes requires that we know where and when to look. As men do not begin to produce sperm until mid-puberty, an obvious place to start looking would be at changes in the lifestyles of young men. However, there are few such changes that are proven to have any impact on sperm production (reviewed in [13]), although one recent study has shown that eating a diet high in saturated fat (a ‘Western’ diet) might be such a factor, at least in men with fertility problems [14]. Instead, the spotlight has fallen on the possibility of effects much earlier in life, in the period six months before and after birth—decades before sperm are even made.
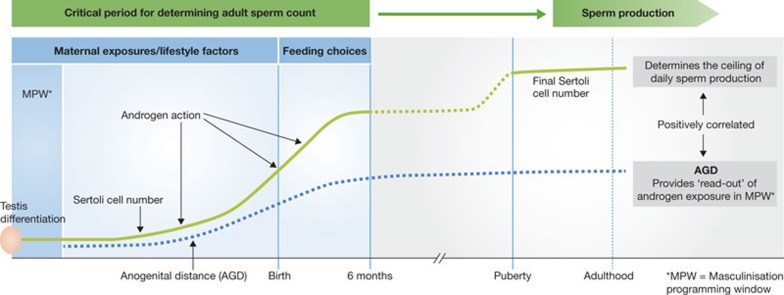
During their development into sperm, germ cells depend on Sertoli cells for physical and metabolic support [3,13]. Each Sertoli cell can only support a fixed number of germ cells, with the result that the number of Sertoli cells per testis determines the overall level of sperm production (Fig 1A). Adverse effects in adulthood, such as elevated scrotal temperature, disease or toxic chemical exposures, as well as increasing age, might reduce the final number of sperm produced per Sertoli cell, but nothing can increase it [13]. Moreover, the number of Sertoli cells itself cannot increase after puberty, and the best evidence suggests that the crucial period for determining Sertoli cell number is probably well before this [3,13], during the six months either side of birth (Fig 3). On the basis of experimental studies in animals, it seems that the ultimate size of the adult testis, which equates to the level of sperm production, is predetermined by the actions of androgens during what is termed the ‘masculinization programming window’ (MPW; Fig 3; [15]).
…sperm count also matters for men for reasons even more fundamental than fertility. It is a barometer of overall health; the lower your sperm count the greater your risk of dying
Figure 3. Perinatal change in Sertoli cell number.
The number of Sertoli cells is in relation to testis differentiation, birth and androgen action in the masculinization programming window (MPW), as reflected by anogenital distance (AGD) in the human. In adulthood AGD is positively related to testis size, sperm count and fertility. Further details are given in the text.
In humans, the MPW is thought to occur between the 8th and 14th weeks of gestation [15], immediately after testis differentiation and when the fetus is tiny. This is of significance because androgen action within the MPW also determines the normal development of all the male reproductive organs including the penis, prostate and seminal vesicles. Common reproductive development disorders in boys—namely incomplete testis descent (cryptorchidism) and abnormal opening of the penile urethra (hypospadias)—can both be caused by deficient androgen action within the MPW [15,16]. This understanding has come about largely because of the demonstration in rats that the level of fetal androgen exposure during the MPW can be determined, retrospectively, at any age after birth by measuring anogenital distance (AGD), as this is also programmed by androgen action within the MPW [15]. Subsequently, it has been shown in both rats and humans that AGD is positively correlated with penis length and testis size and inversely related to risk of cryptorchidism and hypospadias [15,16,17]; in men, AGD is positively related to sperm count and fertility and is the strongest predictor of sperm count [18].
Exactly why AGD is related to testis size and sperm count is unclear, but it is presumed to reflect differences in Sertoli cell number, because proliferation of Sertoli cells perinatally is at least partly driven by testosterone (androgens) produced by Leydig cells within the fetal testis (Fig 3). The attractive resulting hypothesis is that maternal lifestyle and/or environmental chemical exposure during the MPW—perhaps in later gestation or even during the six months after birth when feeding choice might be important—affects androgen production and action, leading to reduced Sertoli cell number and hence to an irreversible reduction in sperm-producing capacity and sperm count in adulthood (Fig 3). This would also fit with the ‘testicular dysgenesis syndrome’ hypothesis, which proposes a common fetal origin for all of the male reproductive disorders mentioned above [19].
There is strong evidence to show that maternal lifestyle during pregnancy can adversely affect sperm counts in adulthood. Prime among these is smoking, as several large studies have shown that moderate to heavy smoking during pregnancy reduces the testis size and sperm count of resulting offspring in adulthood by 20–40% [13,20]. Given that around 25% of all women in the UK today smoke throughout pregnancy, the potential scale of such effects should not be underestimated. Indirect evidence points towards reduced Sertoli cell number as being the explanation for reduced sperm counts as a result of maternal smoking, but how this affects Sertoli cell proliferation and number is unknown. There is experimental evidence in animals that nicotine might inhibit testosterone production, which could be important (Fig 3), but other possibilities could be more probable.
Detailed studies, for example, have shown that exposure to diesel exhaust in pregnancy reduces testis size and sperm production, probably by reducing Sertoli cell number; an effect that works at least in part through the aryl hydrocarbon receptor (AhR) [21]. The AhR is also probably affected by cigarette smoking and by exposure to other smoke sources, including atmospheric pollution, so there could be a common mechanism. This hypothesis is reinforced by a study of sons born to mothers exposed to high levels of dioxin as a result of the Seveso accident in Italy in 1976. The study showed that high exposure to 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) before and after birth, resulted in around a 40% reduction in sperm count in adulthood compared with unexposed breastfed controls [22]. Again, the indirect evidence points towards reduced Sertoli cell number as the underlying explanation. However, the mechanistic link from AhR activation to reduced Sertoli cell number is unclear, and it remains a glaring information gap.
There is also limited evidence that both alcohol consumption and obesity during pregnancy result in lower sperm counts in offspring in adulthood, but there are presently insufficient data to gauge how important these factors might be, or the scale of their effects. An arguably more worrying observation is that bottle-feeding with either standard formula or soy formula milk after birth results in approximately 20% smaller testes at four months of age in boys compared with breastfeeding [23]. Four months is an age at which Sertoli cell number/proliferation might have completed its most important phases (Fig 3). However, as this was a small study, it is premature to draw any strong conclusions. Even so, given how important we know perinatal life can be in determining adult sperm counts, it is remarkable that we do not know whether breast- or bottle-feeding can affect this, as it has population-level implications.
In the public eye there is probably no doubt that perinatal exposure to environmental chemicals, in particular to ‘endocrine disruptors’, accounts for falling sperm counts. Conceptually attractive though this might be, the scientific reality is that there is only limited supporting evidence. This might in part be a consequence of the extreme difficulties in retrospectively linking low sperm counts in adulthood with chemical exposure in the mother two to three decades earlier [13]. It was possible to make this link with maternal smoking because there is extremely good recall of smoking during pregnancy. To determine exposure to environmental chemicals, however, would require samples—maternal blood, milk, amniotic fluid and placenta—from a prospective cohort study to be analysed for contaminants, and adult offspring willing to undergo sperm count analysis. There are such cohorts, but it will probably take a few more years before definitive data emerge that are able to support or refute the possibility of chemical-induced reduction in sperm counts.
However, one intriguing piece of evidence is already available and it concerns the differences in incidence of male reproductive disorders in Finns (low incidence) and Danes (high incidence) [19]. Moreover, it is established that Finns have higher sperm counts than Danes [9], and Finnish newborn boys grow their testes faster in the first few months after birth than Danish newborns [24], pointing to perinatal origins of the sperm count difference. Analysis of breast milk samples from mothers has shown that the environmental chemical ‘signature’ is clearly different between the two countries [25]. Whether this difference is incidental or significant in the context of reproductive development of males is unknown, in particular whether it has any relationship to eventual testis size and sperm count. There is also no ready explanation for the difference in chemical exposure in the two countries.
The fact that sperm counts have fallen across a short timescale (50–70 years […]) suggests that the causes must be lifestyle and environmental, rather than genetic
While waiting for definitive human data, we can forecast what will be found by referring to numerous animal studies. These have shown that fetal exposure to a range of common environmental chemicals can induce reductions in adult testis size and sperm production, as well as inducing other disorders such as cryptorchidism and hypospadias in the male offspring. However, virtually all such studies have used levels of exposure that are far higher than would occur in humans, thus limiting their relevance. Nevertheless, more sophisticated studies have shown that at doses at which individual compounds have no effects, mixtures of 4–10 compounds have huge adverse effects in combination [13]. This does not prove that such mixture effects will be sufficient to induce effects in humans, but humans are certainly exposed to a complex cocktail of hundreds of such chemicals.
In this regard, one recent study in sheep has tried to assess the effects of a complex chemical cocktail [26]. Pregnant sheep were reared on pastures fertilized with either standard fertilizer or sewage sludge—which is the recommended means of disposing of this material within the EU. Sewage sludge contains a large number of compounds and broadly reflects overall human chemical exposure as it is largely derived from our waste. In this regard it does not just contain environmental chemicals, but also pharmaceuticals and personal care products; the latter are now a far more important source of female chemical exposure than in previous decades, and there is a suspicion that they might be more important than environmental contaminants.
Sewage sludge exposure of the mother resulted in 40% of the male offspring having substantially reduced sperm production in adulthood, while the remainder of the animals were unaffected [26]; earlier studies had shown effects in fetal life on testosterone levels and Sertoli cell numbers. The fact that not all adult animals were affected by perinatal sewage sludge exposure is unsurprising as sheep, similarly to humans, are outbred, in contrast with laboratory animals. The findings raise several important points that need to be factored into thinking about falling sperm counts in humans and how to investigate them. First, any study searching for evidence of an association between perinatal chemical exposure and adult sperm count in humans might expect that, if there is an adverse effect of chemical exposure, it might not affect all exposed individuals equally or at all; this would be a major confounding factor. For example, in reflecting on the Danish–Finnish difference in testis development in boys [24], it could mean that even if Finnish boys were exposed to similar or higher levels of chemicals as the Danish boys, they might be less affected. Second, the sheep studies remind us of a basic principle that we already accept, namely that disease in an individual is a product of their genotype and their environment. This needs to be factored into not only our thinking and interpretation, but also the design and planning of studies. In this regard, making detailed use of the differences between Finns and Danes would provide an obvious foundation for studies aimed at teasing apart genotype–environment/lifestyle interactions.
There is now widespread acceptance of the principles of ‘fetal programming’ of adult disease; indeed of the most common complex diseases in Western countries. In this context, the notion of perinatal origins of disorders of sperm production in humans, and the potential causal involvement of maternal lifestyle and environmental exposures described above is anything but ‘new ground’. A more troubling spin-off from the fetal programming studies has been the discovery that some aspects are transmissible to future generations, most probably through epigenetic reprogramming [27].
Of course, the reduced fertility of future generations is not the only impact the present decline in sperm counts will have on unborn children. There will also be the social impact of an ageing population, and an existential impact of whether future generations will come into being at all. As mentioned in the introduction, the social consequences of fewer young people and many older people will be unprecedented and difficult to predict.
Declining sperm counts and social trends will also have severe consequences for couples and individuals that are affected by infertility. The current trend for couples to have children later in life seems likely to continue in the foreseeable future, which makes it easy to predict that couple fertility problems that are already common are likely to become more so. Declining sperm counts in men will increasingly interact with declining fertility in many women, who will wait longer to try to become mothers.
These are real concerns, but perhaps the most disturbing conclusion is that the evidence for low and falling sperm counts points to a wider issue of the subtle dysfunction of the process that makes men male. If this process is affected by maternal lifestyle and environmental exposures, which a growing body of evidence suggests is the case—and for which falling sperm counts is but one symptom—then what other consequences for the programming of behaviour and disease risks will it also bring?
Science & Society Series on Sex and Science.
Sex is the greatest invention of all time: not only has sexual reproduction facilitated the evolution of higher life forms, it has had a profound influence on human history, culture and society. This series explores our attempts to understand the influence of sex in the natural world, and the biological, medical and cultural aspects of sexual reproduction, gender and sexual pleasure

Richard M Sharpe
Acknowledgments
I am grateful to Natasha Fletcher and Jim Chalmers for access to Scottish pregnancy data. The author was supported by Programme Grant G1100358/1 from the UK Medical Research Council and by Grant FP7-ENV-2007-1-212844 from the European Union.
Footnotes
The author declares that he has no conflict of interest.
References
- Skakkebaek NE et al. (2006) Is human fecundity declining? Int J Androl 29: 2–11 [DOI] [PubMed] [Google Scholar]
- Jensen TK, Sbotka T, Hansen MA, Pedersen AT, Lutz W, Skakkebaek NE (2008) Declining trends in conception rates in recent birth cohorts of native Danish women: a possible role of deteriorating male reproductive health. Int J Androl 31: 81–92 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharpe RM, McKinnell C, Kivlin C, Fisher JS (2003) Proliferation and functional maturation of Sertoli cells, and their relevance to disorders of testis function in adulthood. Reproduction 125: 769–784 [DOI] [PubMed] [Google Scholar]
- Bonde JP, Ernst E, Jensen TK, Hjollund NH, Kolstad H, Henriksen TB, Scheike T, Giwercman A, Olsen J, Skakkebaek NE (1998) Relation between semen quality and fertility: a population-based study of 430 first-pregnancy planners. Lancet 354: 1172–1177 [DOI] [PubMed] [Google Scholar]
- Carlsen E, Giwercman A, Keiding N, Skakkebaek NE (1992) Evidence for decreasing quality of semen during past 50 years. BMJ 305: 609–613 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swan SH, Elkin EP, Fenster L (2000) The question of declining sperm density revisited: an analysis of 101 studies published 1934–1996. Environ Health Perspect 108: 961–966 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oehninger S (2011) Clinical management of male infertility in assisted reproduction: ICSI and beyond. Int J Androl 34: e319–e329 [DOI] [PubMed] [Google Scholar]
- Bonde JP, Ramlau-Hansen CH, Olsen J (2011) Trends in sperm counts: the saga continues. Epidemiology 22: 878. [DOI] [PubMed] [Google Scholar]
- Jørgensen N, Asklund C, Carlsen E, Skakkebaek NE (2006) Coordinated European investigations of semen quality: results from studies of Scandinavian young men is a matter of concern. Int J Androl 29: 54–61 [DOI] [PubMed] [Google Scholar]
- Jensen TK, Jacobsen R, Christensen K, Nielsen NC, Bostofte E (2009) Good semen quality and life expectancy: a cohort study of 43,277 men. Am J Epidemiol 170: 559–565 [DOI] [PubMed] [Google Scholar]
- Laughlin GA, Barrett-Connor E, Bergstrom J (2008) Low serum testosterone and mortality in older men. J Clin Endocrinol Metab 93: 68–75 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Travison TG, Araujo AB, O'Donnell AB, Kupelian V, McKinlay JB (2007) A population-level decline in serum testosterone levels in American men. J Clin Endocrinol Metab 92: 196–202 [DOI] [PubMed] [Google Scholar]
- Sharpe RM (2010) Environmental/lifestyle effects on spermatogenesis. Phil Trans R Soc Lond Ser B 365: 1697–1712 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Welsh M, Saunders PTK, Fisken M, Scott HM, Hutchison GR, Smith LB, Sharpe RM (2008) Identification in rats of a programming window for reproductive tract masculinization, disruption of which leads to hypospadias and cryptorchidism. J Clin Invest 118: 1479–1490 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Attaman JA, Toth TL, Furtado J, Campos H, Hauser R, Chavarro JE (2012) Dietary fat and semen quality among men attending a fertility clinic. Hum Reprod [Epub ahead of print] doi:; DOI: 10.1093/humrep/des065 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Drake AJ, Van den Driesche S, Scott HM, Hutchison G, Seckl JR, Sharpe RM (2009) Glucocorticoids amplify dibutyl phthalate-induced disruption of fetal testosterone production and male reproductive development. Endocrinology 150: 5055–5064 [DOI] [PubMed] [Google Scholar]
- Swan SH et al. and the Study for Future Families Research Team (2005) Decrease in anogenital distance among male infants with prenatal phthalate exposure. Environ Health Perspect 113: 1056–1061 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eisenberg ML, Hsieh MH, Walters RC, Krasnow R, Lipshultz LI (2011) The relationship between anogenital distance, fatherhood and fertility in adult men. PLoS ONE 6: e18973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Skakkebaek NE, Rajpert-De Meyts E, Main KM (2001) Testicular dysgenesis syndrome: an increasingly common developmental disorder with environmental aspects. Hum Reprod 16: 972–978 [DOI] [PubMed] [Google Scholar]
- Jensen TK et al. (2004) Association of in utero exposure smoking with reduced semen quality and testis size in adulthood: a cross-sectional study of 1,770 young men from the general population in five European countries. Am J Epidemiol 159: 49–58 [DOI] [PubMed] [Google Scholar]
- Izawa H, Kohara M, Watanbe G, Taya K, Sagai M (2007) Effects of diesel exhaust particles on the male reproductive system in strains of mice with different aryl hydrocarbon receptor responsiveness. J Reprod Dev 53: 1191–1197 [DOI] [PubMed] [Google Scholar]
- Mocarelli P et al. (2011) Perinatal exposure to low doses of dioxin can permanently impair human semen quality. Environ Health Perspect 119: 713–718 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gilchrist JM, Moore MB, Andres A, Estroff JA, Badger TM (2010) Ultrasonographic patterns of reproductive organs in infants fed soy formula: comparisons to infants fed breast milk and milk formula. J Pediatr 156: 215–220 [DOI] [PubMed] [Google Scholar]
- Main KM et al. (2006) Larger testes and higher inhibin B levels in Finnish than in Danish newborn boys. J Clin Endocrinol Metab 91: 2732–2737 [DOI] [PubMed] [Google Scholar]
- Krysiak-Baltyn K et al. (2010) Country-specific chemicals signatures of persistent environmental compounds in breast milk. Int J Androl 33: 270–278 [DOI] [PubMed] [Google Scholar]
- Bellingham M, McKinnell C, Fowler PA, Amezaga MR, Zhang Z, Rhind SM, Cotinot C, Mandon-Pepin B, Evans NP, Sharpe RM (2012) Fetal and postnatal exposure to sewage sludge chemicals disrupts sperm production in adulthood in a subset of animals. Int J Androl [Epub ahead of print] doi: ; DOI: 10.1111/j.1365–26052011.01234.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Drake AJ, Seckl JR (2011) Transmission of programming effects across generations. Pediatr Endocrinol Rev 9: 566–578 [PubMed] [Google Scholar]
- Johnson L, Zane RS, Petty CS, Neaves WB (1984) Quantification of the human Sertoli cell population: its distribution, relation to germ cell numbers and age-related decline. Biol Reprod 31: 785–795 [DOI] [PubMed] [Google Scholar]
- Carlsen E, Petersen JH, Andersson AM, Skakkebaek NE (2004) Effects of ejaculatory frequency and season on variations in semen quality. Fertil Steril 82: 358–366 [DOI] [PubMed] [Google Scholar]