Abstract
The RecB and RecD subunits of the RecBCD enzyme of Escherichia coli contain amino acid sequences similar to a consensus mononucleotide binding motif found in a large number of other enzymes. We have constructed by site-directed mutagenesis a lysine-to-glutamine mutation in this sequence in the RecB protein. The mutant enzyme (RecB-K29Q-CD) has essentially no nuclease or ATP hydrolysis activity on double-stranded DNA, showing the importance of RecB for unwinding double-stranded DNA. However, ATP hydrolysis stimulated by single-stranded DNA is reduced by only about 5-8-fold compared to the wild-type, nuclease activity on single-stranded DNA is reduced by less than 2-fold, and the nuclease activity of the RecB-K29Q-CD enzyme requires ATP. The effects of the RecB mutation suggest that the RecD protein hydrolyzes ATP and can stimulate the RecBCD enzyme nuclease activity on single-stranded DNA.
Full text
PDF

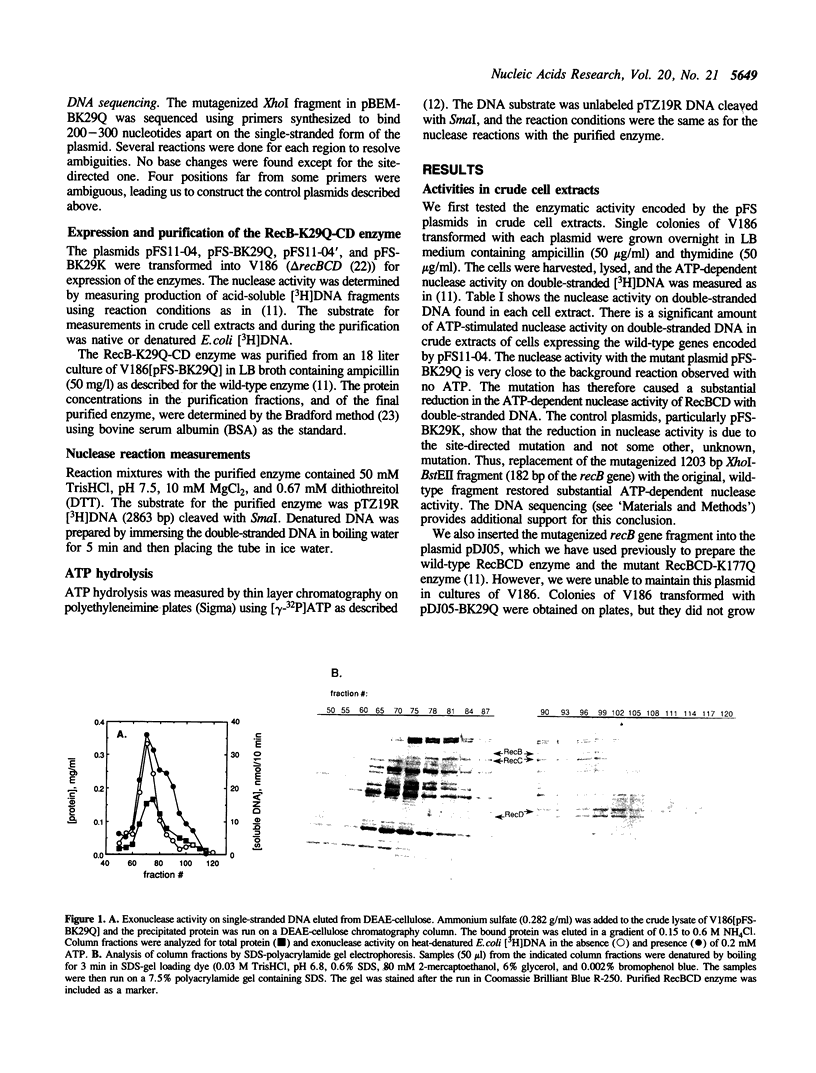
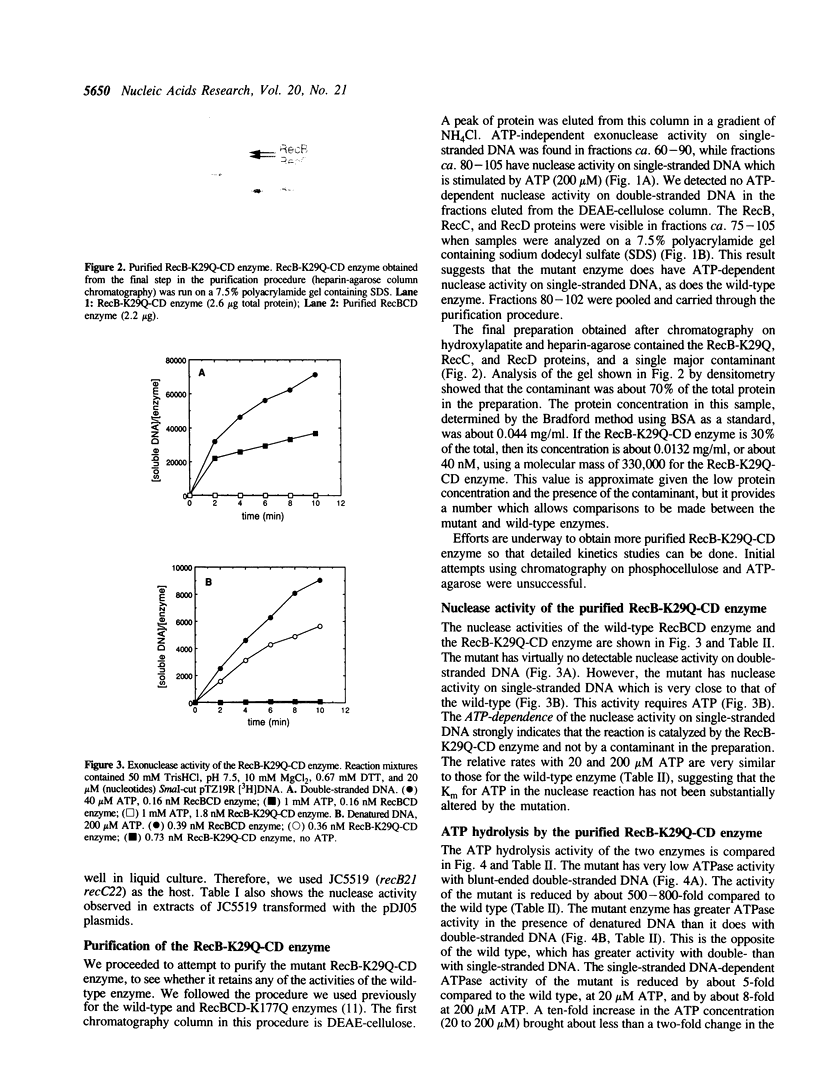
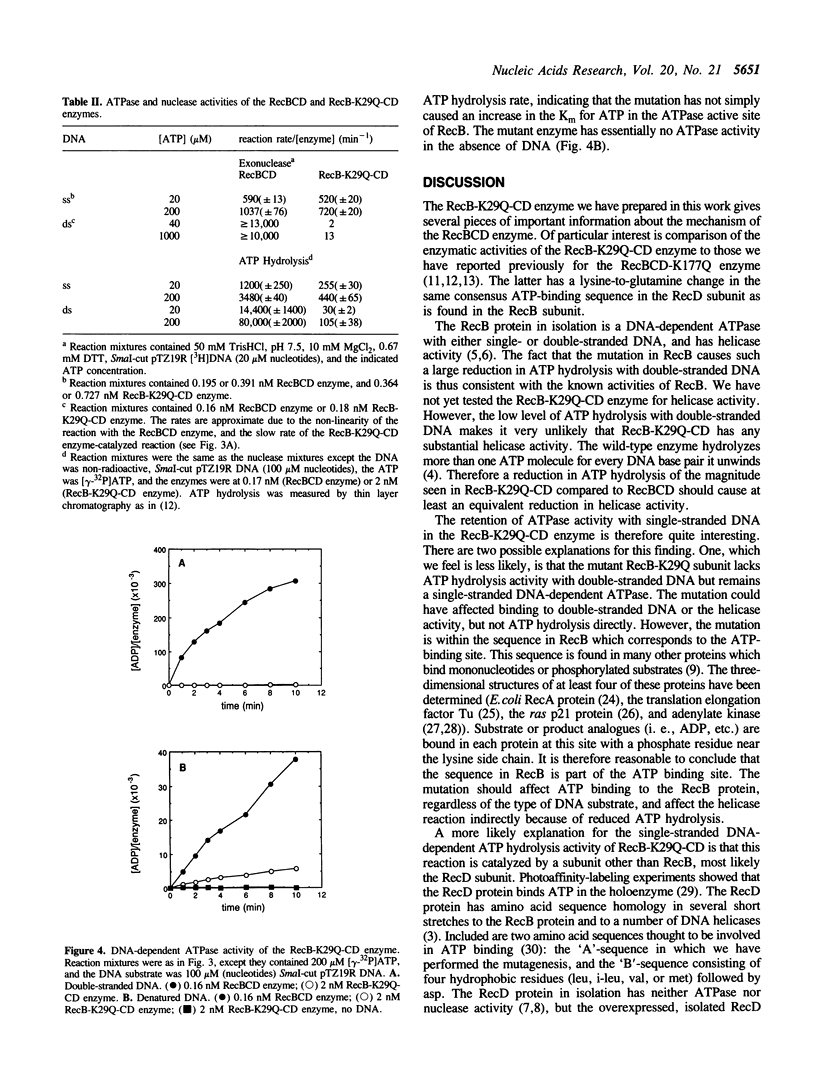


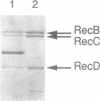
Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Boehmer P. E., Emmerson P. T. The RecB subunit of the Escherichia coli RecBCD enzyme couples ATP hydrolysis to DNA unwinding. J Biol Chem. 1992 Mar 5;267(7):4981–4987. [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Chaudhury A. M., Smith G. R. Escherichia coli recBC deletion mutants. J Bacteriol. 1984 Nov;160(2):788–791. doi: 10.1128/jb.160.2.788-791.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dente L., Cesareni G., Cortese R. pEMBL: a new family of single stranded plasmids. Nucleic Acids Res. 1983 Mar 25;11(6):1645–1655. doi: 10.1093/nar/11.6.1645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Egner U., Tomasselli A. G., Schulz G. E. Structure of the complex of yeast adenylate kinase with the inhibitor P1,P5-di(adenosine-5'-)pentaphosphate at 2.6 A resolution. J Mol Biol. 1987 Jun 5;195(3):649–658. doi: 10.1016/0022-2836(87)90188-4. [DOI] [PubMed] [Google Scholar]
- Finch P. W., Storey A., Chapman K. E., Brown K., Hickson I. D., Emmerson P. T. Complete nucleotide sequence of the Escherichia coli recB gene. Nucleic Acids Res. 1986 Nov 11;14(21):8573–8582. doi: 10.1093/nar/14.21.8573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Finch P. W., Wilson R. E., Brown K., Hickson I. D., Emmerson P. T. Complete nucleotide sequence of the Escherichia coli ptr gene encoding protease III. Nucleic Acids Res. 1986 Oct 10;14(19):7695–7703. doi: 10.1093/nar/14.19.7695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldmark P. J., Linn S. Purification and properties of the recBC DNase of Escherichia coli K-12. J Biol Chem. 1972 Mar 25;247(6):1849–1860. [PubMed] [Google Scholar]
- Gorbalenya A. E., Koonin E. V., Donchenko A. P., Blinov V. M. A novel superfamily of nucleoside triphosphate-binding motif containing proteins which are probably involved in duplex unwinding in DNA and RNA replication and recombination. FEBS Lett. 1988 Aug 1;235(1-2):16–24. doi: 10.1016/0014-5793(88)81226-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hickson I. D., Robson C. N., Atkinson K. E., Hutton L., Emmerson P. T. Reconstitution of RecBC DNase activity from purified Escherichia coli RecB and RecC proteins. J Biol Chem. 1985 Jan 25;260(2):1224–1229. [PubMed] [Google Scholar]
- Julin D. A., Lehman I. R. Photoaffinity labeling of the recBCD enzyme of Escherichia coli with 8-azidoadenosine 5'-triphosphate. J Biol Chem. 1987 Jul 5;262(19):9044–9051. [PubMed] [Google Scholar]
- Jurnak F. Structure of the GDP domain of EF-Tu and location of the amino acids homologous to ras oncogene proteins. Science. 1985 Oct 4;230(4721):32–36. doi: 10.1126/science.3898365. [DOI] [PubMed] [Google Scholar]
- Karu A. E., Linn S. Uncoupling of the recBC ATPase from DNase by DNA crosslinked with psoralen. Proc Natl Acad Sci U S A. 1972 Oct;69(10):2855–2859. doi: 10.1073/pnas.69.10.2855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Korangy F., Julin D. A. A mutation in the consensus ATP-binding sequence of the RecD subunit reduces the processivity of the RecBCD enzyme from Escherichia coli. J Biol Chem. 1992 Feb 15;267(5):3088–3095. [PubMed] [Google Scholar]
- Korangy F., Julin D. A. Alteration by site-directed mutagenesis of the conserved lysine residue in the ATP-binding consensus sequence of the RecD subunit of the Escherichia coli RecBCD enzyme. J Biol Chem. 1992 Jan 25;267(3):1727–1732. [PubMed] [Google Scholar]
- Korangy F., Julin D. A. Enzymatic effects of a lysine-to-glutamine mutation in the ATP-binding consensus sequence in the RecD subunit of the RecBCD enzyme from Escherichia coli. J Biol Chem. 1992 Jan 25;267(3):1733–1740. [PubMed] [Google Scholar]
- Lieberman R. P., Oishi M. The recBC deoxyribonuclease of Escherichia coli: isolation and characterization of the subunit proteins and reconstitution of the enzyme. Proc Natl Acad Sci U S A. 1974 Dec;71(12):4816–4820. doi: 10.1073/pnas.71.12.4816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Livneh Z. Directed mutagenesis method for analysis of mutagen specificity: application to ultraviolet-induced mutagenesis. Proc Natl Acad Sci U S A. 1983 Jan;80(1):237–241. doi: 10.1073/pnas.80.1.237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MacKay V., Linn S. The mechanism of degradation of duplex deoxyribonucleic acid by the recBC enzyme of Escherichia coli K-12. J Biol Chem. 1974 Jul 10;249(13):4286–4294. [PubMed] [Google Scholar]
- Masterson C., Boehmer P. E., McDonald F., Chaudhuri S., Hickson I. D., Emmerson P. T. Reconstitution of the activities of the RecBCD holoenzyme of Escherichia coli from the purified subunits. J Biol Chem. 1992 Jul 5;267(19):13564–13572. [PubMed] [Google Scholar]
- Pai E. F., Kabsch W., Krengel U., Holmes K. C., John J., Wittinghofer A. Structure of the guanine-nucleotide-binding domain of the Ha-ras oncogene product p21 in the triphosphate conformation. Nature. 1989 Sep 21;341(6239):209–214. doi: 10.1038/341209a0. [DOI] [PubMed] [Google Scholar]
- Palas K. M., Kushner S. R. Biochemical and physical characterization of exonuclease V from Escherichia coli. Comparison of the catalytic activities of the RecBC and RecBCD enzymes. J Biol Chem. 1990 Feb 25;265(6):3447–3454. [PubMed] [Google Scholar]
- Roman L. J., Eggleston A. K., Kowalczykowski S. C. Processivity of the DNA helicase activity of Escherichia coli recBCD enzyme. J Biol Chem. 1992 Feb 25;267(6):4207–4214. [PubMed] [Google Scholar]
- Roman L. J., Kowalczykowski S. C. Characterization of the adenosinetriphosphatase activity of the Escherichia coli RecBCD enzyme: relationship of ATP hydrolysis to the unwinding of duplex DNA. Biochemistry. 1989 Apr 4;28(7):2873–2881. doi: 10.1021/bi00433a019. [DOI] [PubMed] [Google Scholar]
- Saraste M., Sibbald P. R., Wittinghofer A. The P-loop--a common motif in ATP- and GTP-binding proteins. Trends Biochem Sci. 1990 Nov;15(11):430–434. doi: 10.1016/0968-0004(90)90281-f. [DOI] [PubMed] [Google Scholar]
- Sasaki M., Fujiyoshi T., Shimada K., Takagi Y. Fine structure of the recB and recC gene region of Escherichia coli. Biochem Biophys Res Commun. 1982 Nov 30;109(2):414–422. doi: 10.1016/0006-291x(82)91737-5. [DOI] [PubMed] [Google Scholar]
- Story R. M., Steitz T. A. Structure of the recA protein-ADP complex. Nature. 1992 Jan 23;355(6358):374–376. doi: 10.1038/355374a0. [DOI] [PubMed] [Google Scholar]
- Tsai M. D., Yan H. G. Mechanism of adenylate kinase: site-directed mutagenesis versus X-ray and NMR. Biochemistry. 1991 Jul 16;30(28):6806–6818. doi: 10.1021/bi00242a002. [DOI] [PubMed] [Google Scholar]
- Walker J. E., Saraste M., Runswick M. J., Gay N. J. Distantly related sequences in the alpha- and beta-subunits of ATP synthase, myosin, kinases and other ATP-requiring enzymes and a common nucleotide binding fold. EMBO J. 1982;1(8):945–951. doi: 10.1002/j.1460-2075.1982.tb01276.x. [DOI] [PMC free article] [PubMed] [Google Scholar]