Abstract
The title compound, C30H36N2O6, was prepared via twofold Suzuki coupling of a diboronic acid with bromonitrobenzene. The molecule is located on a crystallographic inversion centre. The lateral benzene ring and the central ring make a dihedral angle of 48.75 (14)° and the nitro group is twisted by 41.47 (13)° out of the plane of the benzene ring. The nitro and hexyloxy groups are in close proximity and the hexyloxy chain adopts an all-anti conformation.
Related literature
For the synthesis of carbazoles and heteroanalogous carbazoles, see: Letessier et al. (2011 ▶); Dassonneville et al. (2011 ▶); Nissen & Detert (2011 ▶); Letessier & Detert (2012 ▶). For the Cadogan reaction, see: Cadogan (1962 ▶). For Suzuki cross-couplings see Miyaura & Suzuki (1995 ▶). For π-systems for optoelectronic applications, see: Nemkovich et al. (2009 ▶). For structures of substituted p-terphenyls, see: Jones et al. (2005 ▶), Moschel et al. (2011 ▶). For torsion in biphenyls, see: Miao et al. (2009 ▶); Fischer et al. (2007 ▶).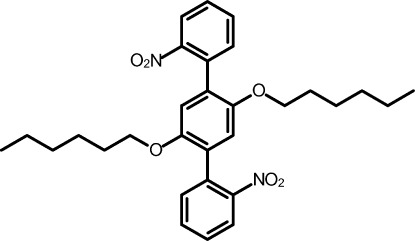
Experimental
Crystal data
C30H36N2O6
M r = 520.61
Monoclinic,

a = 7.9314 (4) Å
b = 19.2029 (17) Å
c = 9.1247 (5) Å
β = 96.368 (5)°
V = 1381.17 (16) Å3
Z = 2
Mo Kα radiation
μ = 0.09 mm−1
T = 193 K
0.44 × 0.30 × 0.20 mm
Data collection
Stoe IPDS 2T diffractometer
8154 measured reflections
3331 independent reflections
2610 reflections with I > 2σ(I)
R int = 0.026
Refinement
R[F 2 > 2σ(F 2)] = 0.044
wR(F 2) = 0.120
S = 1.07
3331 reflections
173 parameters
H-atom parameters constrained
Δρmax = 0.31 e Å−3
Δρmin = −0.27 e Å−3
Data collection: X-AREA (Stoe & Cie, 2011 ▶); cell refinement: X-AREA; data reduction: X-RED (Stoe & Cie, 2011 ▶); program(s) used to solve structure: SIR97 (Altomare et al., 1999 ▶); program(s) used to refine structure: SHELXL97 (Sheldrick, 2008 ▶); molecular graphics: PLATON (Spek, 2009 ▶); software used to prepare material for publication: PLATON.
Supplementary Material
Crystal structure: contains datablock(s) I, global. DOI: 10.1107/S1600536812009944/bt5839sup1.cif
Structure factors: contains datablock(s) I. DOI: 10.1107/S1600536812009944/bt5839Isup2.hkl
Supplementary material file. DOI: 10.1107/S1600536812009944/bt5839Isup3.cml
Additional supplementary materials: crystallographic information; 3D view; checkCIF report
Acknowledgments
The authors are grateful to Heinz Kolshorn for invaluable discussions and the NMR spectra.
supplementary crystallographic information
Comment
As part of a larger project on the synthesis of carbazoles and heteroanalogous carbazoles (Letessier et al. 2011, Dassonneville et al. 2011, Nissen & Detert 2011, Letessier & Detert 2012) the Cadogan reaction (Cadogan 1962) appeared to be a suitable method for the construction of larger planar π-systems for optoelectronic applications (Nemkovich et al. 2009). The title compond was prepared as an intermediate for the synthesis of dihexyloxy-indolocarbazole.
The title compound crystallizes in a centrosymmetrical conformation with a highly twisted dinitroterphenyl core and hexyloxy chains in an all-anti conformation. The dihedral angle of the mean planes of the central and the lateral ring is 131.25 (14)° with the ortho-substituents nitro- and hexyloxy in close proximity. The distance N10 - O13 (nitro-hexyloxy) is only 2.710 (2) Å. The nitro group is twisted out of the plane of the adjacent benzene ring, the dihedral angle is 138.53 (13)° pointing towards the adjacent o-hexyloxy group. A o-methyl substitution on on a biphenyl linkage is sufficient to open the dihedral angle from 9.45 ° (Fischer et al. 2007) to more than 63° (Jones et al. 2005). The twist (131.25°) found in the title compound - though o,o-disubstituted on both biphenyl linkages - is significantly smaller. This can result from an electronic attraction between N10 (nitro) and O13 (hexyloxy). Miao et al. (2009) reported a dihedral angle of 60.5° in the fourfold o-substituted 2,2-dimethoxy-6,6-dinitrobiphenyl.
Experimental
Synthesis: A mixture of 2,5-dihexyloxy-1,4-phenylenediboronic acid (500 mg, 1.37 mmol), 1-bromo-2-nitrobenzene (553 mg, 2.74 mmol), Pd(PPh3)3 (79 mg, 0.067 mmol) in dimethoxyethane (10 ml) was stirred for 45 min at 298 K. An aqueous solution of Na2CO3 (1M, 8.2 ml) was added and the mixture heated to 353 K for 18 h. The cooled mixture was poured into water (40 ml) and the product was isolated by extraction with dichloromethane (3 x 15 ml), washing the pooled solutions with brine (2 x 10 ml), drying (Na2SO4) and crystallization from chloroform/pentane. Yield: 495 mg (70%) of a yellow solid with m. p. 438 - 440 K. Rf = 0.41 (silica gel, petroleum ether/ethyl acetate 9/1).
Refinement
Hydrogen atoms were placed at calculated positions with C—H = 0.95 Å (aromatic) or 0.98–0.99 Å (sp3 C-atom). All H atoms were refined in the riding-model approximation with isotropic displacement parameters set at 1.2–1.5 times of the Ueq of the parent atom.
Figures
Fig. 1.
View of compound I. Displacement ellipsoids are drawn at the 50% probability level. Second part of the molecule labeled with a generated applying symmetry code 1 - x, 1 - y, 1 - z.
Crystal data
| C30H36N2O6 | F(000) = 556 |
| Mr = 520.61 | Dx = 1.252 Mg m−3 |
| Monoclinic, P21/n | Mo Kα radiation, λ = 0.71073 Å |
| Hall symbol: -P 2yn | Cell parameters from 7928 reflections |
| a = 7.9314 (4) Å | θ = 3.2–29.1° |
| b = 19.2029 (17) Å | µ = 0.09 mm−1 |
| c = 9.1247 (5) Å | T = 193 K |
| β = 96.368 (5)° | Block, yellow |
| V = 1381.17 (16) Å3 | 0.44 × 0.30 × 0.20 mm |
| Z = 2 |
Data collection
| Stoe IPDS 2T diffractometer | 2610 reflections with I > 2σ(I) |
| Radiation source: sealed X-ray tube, 12 x 0.4 mm long-fine focus | Rint = 0.026 |
| Plane graphite monochromator | θmax = 28.0°, θmin = 3.2° |
| Detector resolution: 6.67 pixels mm-1 | h = −10→10 |
| ω scan | k = −25→22 |
| 8154 measured reflections | l = −12→10 |
| 3331 independent reflections |
Refinement
| Refinement on F2 | Primary atom site location: structure-invariant direct methods |
| Least-squares matrix: full | Secondary atom site location: difference Fourier map |
| R[F2 > 2σ(F2)] = 0.044 | Hydrogen site location: inferred from neighbouring sites |
| wR(F2) = 0.120 | H-atom parameters constrained |
| S = 1.07 | w = 1/[σ2(Fo2) + (0.0562P)2 + 0.4038P] where P = (Fo2 + 2Fc2)/3 |
| 3331 reflections | (Δ/σ)max < 0.001 |
| 173 parameters | Δρmax = 0.31 e Å−3 |
| 0 restraints | Δρmin = −0.27 e Å−3 |
Special details
| Experimental. 1H-NMR (400 MHz, CDCl3): δ = 7.95 (dd, 3J = 8.5 Hz, 4J= 1.2 Hz, 2 H, 3-H); 7.64 (dt, 3J = 7.5 Hz, 4J = 1.4 Hz, 2 H, 4-H); 7.49 - 7.45 (m, 4 H); 6.83 (s, 2 H, 2-H); 3.81 (bs (t), 4 H, O—CH2); 1.59 - 1.54 (m, 4 H); 1.25 - 1.18 (m, 12 H); 0.80 (t, 3J = 6.9 Hz, 6 H, CH3).13C-NMR (75 MHz, CDCl3): δ = 149.7 (s, 2-C), 149.5 (s), 132.9 (s), 132.6 (d), 132.5 (d), 128.1 (d), 127.8 (s), 123.9 (d), 113.4 (d), 69.1 (t), 31.3 (t), 28.8 (t), 25.4 (t), 22.5 (t), 13.8 (q).IR (ATR): ν = 3734, 3585, 3070, 2944, 2869, 2855, 2363, 2334, 1608, 1573, 1530, 1510, 1469, 1441, 1387, 1358, 1290, 1255, 1209, 1165, 1144, 1025, 997, 870, 860.MS (EI): m/z = 520 (100%, M+.). |
| Geometry. All e.s.d.'s (except the e.s.d. in the dihedral angle between two l.s. planes) are estimated using the full covariance matrix. The cell e.s.d.'s are taken into account individually in the estimation of e.s.d.'s in distances, angles and torsion angles; correlations between e.s.d.'s in cell parameters are only used when they are defined by crystal symmetry. An approximate (isotropic) treatment of cell e.s.d.'s is used for estimating e.s.d.'s involving l.s. planes. |
| Refinement. Refinement of F2 against ALL reflections. The weighted R-factor wR and goodness of fit S are based on F2, conventional R-factors R are based on F, with F set to zero for negative F2. The threshold expression of F2 > σ(F2) is used only for calculating R-factors(gt) etc. and is not relevant to the choice of reflections for refinement. R-factors based on F2 are statistically about twice as large as those based on F, and R- factors based on ALL data will be even larger. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2)
| x | y | z | Uiso*/Ueq | ||
| C1 | 0.46215 (15) | 0.52525 (7) | 0.35512 (14) | 0.0240 (3) | |
| C2 | 0.35820 (15) | 0.54228 (7) | 0.46442 (14) | 0.0252 (3) | |
| C3 | 0.60362 (16) | 0.48321 (8) | 0.39358 (14) | 0.0264 (3) | |
| H3 | 0.6756 | 0.4717 | 0.3206 | 0.032* | |
| C4 | 0.43305 (15) | 0.55452 (7) | 0.20336 (14) | 0.0232 (3) | |
| C5 | 0.57076 (16) | 0.58310 (8) | 0.14150 (15) | 0.0299 (3) | |
| H5 | 0.6801 | 0.5811 | 0.1956 | 0.036* | |
| C6 | 0.55321 (18) | 0.61424 (8) | 0.00394 (16) | 0.0338 (3) | |
| H6 | 0.6502 | 0.6318 | −0.0362 | 0.041* | |
| C7 | 0.39481 (18) | 0.61991 (8) | −0.07522 (16) | 0.0309 (3) | |
| H7 | 0.3820 | 0.6432 | −0.1676 | 0.037* | |
| C8 | 0.25493 (17) | 0.59149 (7) | −0.01925 (15) | 0.0273 (3) | |
| H8 | 0.1456 | 0.5945 | −0.0732 | 0.033* | |
| C9 | 0.27660 (15) | 0.55862 (7) | 0.11640 (14) | 0.0225 (3) | |
| N10 | 0.12608 (13) | 0.52343 (6) | 0.16019 (12) | 0.0271 (3) | |
| O11 | −0.01207 (12) | 0.55180 (6) | 0.13048 (12) | 0.0381 (3) | |
| O12 | 0.14470 (14) | 0.46637 (6) | 0.21901 (12) | 0.0380 (3) | |
| O13 | 0.22425 (12) | 0.58564 (6) | 0.42244 (10) | 0.0314 (2) | |
| C14 | 0.11269 (18) | 0.60345 (9) | 0.52871 (16) | 0.0330 (3) | |
| H14A | 0.1748 | 0.6300 | 0.6106 | 0.040* | |
| H14B | 0.0665 | 0.5607 | 0.5699 | 0.040* | |
| C15 | −0.02962 (17) | 0.64694 (9) | 0.45359 (16) | 0.0330 (3) | |
| H15A | 0.0189 | 0.6874 | 0.4055 | 0.040* | |
| H15B | −0.0946 | 0.6188 | 0.3758 | 0.040* | |
| C16 | −0.1481 (2) | 0.67270 (12) | 0.5597 (2) | 0.0560 (6) | |
| H16A | −0.1793 | 0.6328 | 0.6201 | 0.067* | |
| H16B | −0.0862 | 0.7067 | 0.6274 | 0.067* | |
| C17 | −0.30865 (18) | 0.70652 (8) | 0.49024 (18) | 0.0344 (3) | |
| H17A | −0.2781 | 0.7465 | 0.4299 | 0.041* | |
| H17B | −0.3715 | 0.6726 | 0.4231 | 0.041* | |
| C18 | −0.4239 (3) | 0.73189 (14) | 0.5993 (3) | 0.0670 (7) | |
| H18A | −0.4492 | 0.6924 | 0.6631 | 0.080* | |
| H18B | −0.3626 | 0.7675 | 0.6632 | 0.080* | |
| C19 | −0.5889 (2) | 0.76264 (11) | 0.5319 (3) | 0.0602 (6) | |
| H19A | −0.5662 | 0.8048 | 0.4767 | 0.090* | |
| H19B | −0.6592 | 0.7745 | 0.6101 | 0.090* | |
| H19C | −0.6488 | 0.7287 | 0.4650 | 0.090* |
Atomic displacement parameters (Å2)
| U11 | U22 | U33 | U12 | U13 | U23 | |
| C1 | 0.0190 (5) | 0.0341 (7) | 0.0187 (6) | 0.0020 (5) | 0.0005 (4) | −0.0030 (5) |
| C2 | 0.0172 (5) | 0.0358 (7) | 0.0218 (6) | 0.0056 (5) | −0.0005 (4) | −0.0031 (5) |
| C3 | 0.0204 (6) | 0.0391 (7) | 0.0199 (6) | 0.0049 (5) | 0.0027 (5) | −0.0037 (5) |
| C4 | 0.0202 (5) | 0.0296 (6) | 0.0198 (6) | 0.0027 (5) | 0.0023 (4) | −0.0032 (5) |
| C5 | 0.0189 (6) | 0.0436 (8) | 0.0271 (7) | −0.0031 (5) | 0.0017 (5) | −0.0039 (6) |
| C6 | 0.0302 (7) | 0.0441 (8) | 0.0285 (7) | −0.0111 (6) | 0.0094 (6) | −0.0021 (6) |
| C7 | 0.0375 (7) | 0.0326 (7) | 0.0229 (6) | −0.0045 (6) | 0.0040 (5) | 0.0024 (5) |
| C8 | 0.0260 (6) | 0.0322 (7) | 0.0228 (6) | 0.0003 (5) | −0.0010 (5) | 0.0010 (5) |
| C9 | 0.0186 (5) | 0.0268 (6) | 0.0219 (6) | −0.0002 (5) | 0.0020 (4) | −0.0012 (5) |
| N10 | 0.0210 (5) | 0.0398 (7) | 0.0201 (5) | −0.0036 (5) | 0.0009 (4) | −0.0006 (5) |
| O11 | 0.0178 (4) | 0.0605 (7) | 0.0353 (6) | 0.0027 (4) | 0.0006 (4) | −0.0021 (5) |
| O12 | 0.0358 (5) | 0.0425 (6) | 0.0351 (6) | −0.0098 (5) | 0.0014 (4) | 0.0114 (5) |
| O13 | 0.0239 (4) | 0.0492 (6) | 0.0213 (5) | 0.0155 (4) | 0.0029 (4) | 0.0003 (4) |
| C14 | 0.0277 (6) | 0.0471 (9) | 0.0254 (7) | 0.0141 (6) | 0.0078 (5) | 0.0025 (6) |
| C15 | 0.0248 (6) | 0.0453 (8) | 0.0288 (7) | 0.0118 (6) | 0.0030 (5) | 0.0009 (6) |
| C16 | 0.0502 (10) | 0.0811 (14) | 0.0402 (9) | 0.0427 (10) | 0.0203 (8) | 0.0197 (9) |
| C17 | 0.0254 (6) | 0.0330 (7) | 0.0459 (9) | 0.0059 (6) | 0.0090 (6) | 0.0019 (6) |
| C18 | 0.0543 (11) | 0.0902 (16) | 0.0615 (13) | 0.0435 (11) | 0.0297 (10) | 0.0211 (12) |
| C19 | 0.0324 (8) | 0.0556 (12) | 0.0951 (17) | 0.0150 (8) | 0.0188 (10) | 0.0009 (11) |
Geometric parameters (Å, º)
| C1—C3 | 1.3952 (18) | O13—C14 | 1.4250 (15) |
| C1—C2 | 1.4014 (17) | C14—C15 | 1.5065 (19) |
| C1—C4 | 1.4886 (18) | C14—H14A | 0.9900 |
| C2—O13 | 1.3705 (15) | C14—H14B | 0.9900 |
| C2—C3i | 1.3867 (19) | C15—C16 | 1.506 (2) |
| C3—C2i | 1.3867 (19) | C15—H15A | 0.9900 |
| C3—H3 | 0.9500 | C15—H15B | 0.9900 |
| C4—C5 | 1.3963 (18) | C16—C17 | 1.505 (2) |
| C4—C9 | 1.3990 (17) | C16—H16A | 0.9900 |
| C5—C6 | 1.383 (2) | C16—H16B | 0.9900 |
| C5—H5 | 0.9500 | C17—C18 | 1.505 (2) |
| C6—C7 | 1.382 (2) | C17—H17A | 0.9900 |
| C6—H6 | 0.9500 | C17—H17B | 0.9900 |
| C7—C8 | 1.3838 (19) | C18—C19 | 1.503 (3) |
| C7—H7 | 0.9500 | C18—H18A | 0.9900 |
| C8—C9 | 1.3831 (18) | C18—H18B | 0.9900 |
| C8—H8 | 0.9500 | C19—H19A | 0.9800 |
| C9—N10 | 1.4655 (16) | C19—H19B | 0.9800 |
| N10—O12 | 1.2218 (16) | C19—H19C | 0.9800 |
| N10—O11 | 1.2268 (15) | ||
| C3—C1—C2 | 118.44 (12) | O13—C14—H14B | 110.0 |
| C3—C1—C4 | 119.33 (11) | C15—C14—H14B | 110.0 |
| C2—C1—C4 | 122.08 (11) | H14A—C14—H14B | 108.4 |
| O13—C2—C3i | 123.90 (11) | C16—C15—C14 | 112.27 (12) |
| O13—C2—C1 | 116.22 (11) | C16—C15—H15A | 109.2 |
| C3i—C2—C1 | 119.85 (12) | C14—C15—H15A | 109.2 |
| C2i—C3—C1 | 121.70 (11) | C16—C15—H15B | 109.2 |
| C2i—C3—H3 | 119.2 | C14—C15—H15B | 109.2 |
| C1—C3—H3 | 119.2 | H15A—C15—H15B | 107.9 |
| C5—C4—C9 | 115.65 (12) | C17—C16—C15 | 115.45 (14) |
| C5—C4—C1 | 118.49 (11) | C17—C16—H16A | 108.4 |
| C9—C4—C1 | 125.82 (11) | C15—C16—H16A | 108.4 |
| C6—C5—C4 | 122.16 (12) | C17—C16—H16B | 108.4 |
| C6—C5—H5 | 118.9 | C15—C16—H16B | 108.4 |
| C4—C5—H5 | 118.9 | H16A—C16—H16B | 107.5 |
| C7—C6—C5 | 120.15 (12) | C18—C17—C16 | 114.11 (15) |
| C7—C6—H6 | 119.9 | C18—C17—H17A | 108.7 |
| C5—C6—H6 | 119.9 | C16—C17—H17A | 108.7 |
| C6—C7—C8 | 119.71 (13) | C18—C17—H17B | 108.7 |
| C6—C7—H7 | 120.1 | C16—C17—H17B | 108.7 |
| C8—C7—H7 | 120.1 | H17A—C17—H17B | 107.6 |
| C9—C8—C7 | 119.02 (12) | C19—C18—C17 | 114.93 (19) |
| C9—C8—H8 | 120.5 | C19—C18—H18A | 108.5 |
| C7—C8—H8 | 120.5 | C17—C18—H18A | 108.5 |
| C8—C9—C4 | 123.21 (12) | C19—C18—H18B | 108.5 |
| C8—C9—N10 | 115.49 (11) | C17—C18—H18B | 108.5 |
| C4—C9—N10 | 121.17 (11) | H18A—C18—H18B | 107.5 |
| O12—N10—O11 | 123.84 (12) | C18—C19—H19A | 109.5 |
| O12—N10—C9 | 118.09 (11) | C18—C19—H19B | 109.5 |
| O11—N10—C9 | 118.01 (12) | H19A—C19—H19B | 109.5 |
| C2—O13—C14 | 118.44 (10) | C18—C19—H19C | 109.5 |
| O13—C14—C15 | 108.30 (11) | H19A—C19—H19C | 109.5 |
| O13—C14—H14A | 110.0 | H19B—C19—H19C | 109.5 |
| C15—C14—H14A | 110.0 | ||
| C3—C1—C2—O13 | 177.82 (12) | C7—C8—C9—N10 | −173.60 (12) |
| C4—C1—C2—O13 | 2.17 (19) | C5—C4—C9—C8 | −3.0 (2) |
| C3—C1—C2—C3i | −0.7 (2) | C1—C4—C9—C8 | 174.57 (13) |
| C4—C1—C2—C3i | −176.30 (13) | C5—C4—C9—N10 | 172.61 (12) |
| C2—C1—C3—C2i | 0.7 (2) | C1—C4—C9—N10 | −9.8 (2) |
| C4—C1—C3—C2i | 176.44 (13) | C8—C9—N10—O12 | 138.53 (13) |
| C3—C1—C4—C5 | −44.37 (18) | C4—C9—N10—O12 | −37.39 (18) |
| C2—C1—C4—C5 | 131.25 (14) | C8—C9—N10—O11 | −38.71 (17) |
| C3—C1—C4—C9 | 138.15 (14) | C4—C9—N10—O11 | 145.37 (13) |
| C2—C1—C4—C9 | −46.2 (2) | C3i—C2—O13—C14 | −2.8 (2) |
| C9—C4—C5—C6 | 0.8 (2) | C1—C2—O13—C14 | 178.78 (13) |
| C1—C4—C5—C6 | −176.95 (13) | C2—O13—C14—C15 | −176.02 (12) |
| C4—C5—C6—C7 | 2.1 (2) | O13—C14—C15—C16 | −175.95 (16) |
| C5—C6—C7—C8 | −2.9 (2) | C14—C15—C16—C17 | −170.43 (16) |
| C6—C7—C8—C9 | 0.8 (2) | C15—C16—C17—C18 | −179.81 (19) |
| C7—C8—C9—C4 | 2.2 (2) | C16—C17—C18—C19 | −177.1 (2) |
Symmetry code: (i) −x+1, −y+1, −z+1.
Footnotes
Supplementary data and figures for this paper are available from the IUCr electronic archives (Reference: BT5839).
References
- Altomare, A., Burla, M. C., Camalli, M., Cascarano, G. L., Giacovazzo, C., Guagliardi, A., Moliterni, A. G. G., Polidori, G. & Spagna, R. (1999). J. Appl. Cryst. 32, 115–119.
- Cadogan, J. I. G. (1962). Q. Rev. 16, 208–239.
- Dassonneville, B., Witulski, B. & Detert, H. (2011). Eur. J. Org. Chem. pp. 2836–2844.
- Fischer, A., Yathirajan, H. S., Ashalatha, B. V., Narayana, B. & Sarojini, B. K. (2007). Acta Cryst. E63, o1357–o1358.
- Jones, P. G., Kuś, P. & Pasewicz, A. (2005). Acta Cryst. E61, o1895–o1896.
- Letessier, J. & Detert, H. (2012). Synthesis, 44, 290–296.
- Letessier, J., Schollmeyer, D. & Detert, H. (2011). Acta Cryst. E67, o2494. [DOI] [PMC free article] [PubMed]
- Miao, S.-B., Deng, D.-S., Liu, X.-M. & Ji, B.-M. (2009). Acta Cryst. E65, o2314. [DOI] [PMC free article] [PubMed]
- Miyaura, N. & Suzuki, A. (1995). Chem. Rev. 95, 2457–2483.
- Moschel, S., Schollmeyer, D. & Detert, H. (2011). Acta Cryst. E67, o1425. [DOI] [PMC free article] [PubMed]
- Nemkovich, N. A., Kruchenok, Yu. V., Sobchuk, A. N., Detert, H., Wrobel, N. & Chernyavskii, E. A. (2009). Opt. Spectrosc. 107, 275–281.
- Nissen, F. & Detert, H. (2011). Eur. J. Org. Chem. pp. 2845–2854.
- Sheldrick, G. M. (2008). Acta Cryst. A64, 112–122. [DOI] [PubMed]
- Spek, A. L. (2009). Acta Cryst. D65, 148–155. [DOI] [PMC free article] [PubMed]
- Stoe & Cie (2011). X-AREA and X-RED Stoe & Cie, Darmstadt, Germany.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Crystal structure: contains datablock(s) I, global. DOI: 10.1107/S1600536812009944/bt5839sup1.cif
Structure factors: contains datablock(s) I. DOI: 10.1107/S1600536812009944/bt5839Isup2.hkl
Supplementary material file. DOI: 10.1107/S1600536812009944/bt5839Isup3.cml
Additional supplementary materials: crystallographic information; 3D view; checkCIF report



