Abstract
Objective
To compare a semi-quantitative and a quantitative morphological score for assessment of early osteoarthritis (OA) evolution.
Materials and methods
3.0 T MRI of the knee was performed in 60 women, 30 with early OA (each 15 with Kellgren–Lawrence grade 2 and 3) and 30 age-matched controls at baseline and at 12 and 24 months. Pathological condition was assessed with the whole-organ magnetic resonance imaging score (WORMS). Cartilage abnormalities and bone marrow edema pattern (BMEP) were also quantified using a previously introduced morphological quantitative score. These data were correlated with changes in clinical parameters and joint space width using generalized estimation equations (GEE).
Results
At baseline, OA patients had significantly (p<0.05) more and larger cartilage lesions and BMEP. During follow-up, cartilage lesions increased significantly (p<0.05) in the patients compared with controls: WORMS showed progression only at the lateral patella, whereas the quantitative score revealed progression additionally at the trochlea and at the medial compartment. Both scores showed a significant (p<0.05) increase in BMEP at the lateral femur in OA patients. In addition, quantitative scores of BMEP of the whole knee decreased significantly (p<0.05) after 12 months and increased after 24 months in the patients, but showed an increase in controls at all follow-up examinations. Only weak correlations between structural imaging findings and clinical parameters were observed.
Conclusion
Quantitative assessment of cartilage lesions and BMEP is more sensitive to changes during the course of the disease than semi-quantitative scoring. However, structural imaging findings do not correlate well with the clinical progression of OA.
Keywords: Osteoarthritis, Magnetic resonance imaging, Cartilage lesion, Bone marrow edema pattern, Follow-up, Lesion size
Introduction
As the most common form of arthritis and a leading cause of disability, osteoarthritis (OA) affects at least 12 % (more than 20 million) of the United States population and is associated with substantial socioeconomic costs [1–4]. Most people over the age of 60 will have some form of OA and about half will have symptoms [5, 6]. Given its high prevalence and associated limitation of activity in an aging population, improved diagnostic tools are required for evaluation of the disease in order to detect it in the earlier stages, monitor disease progression more sensitively, and potentially analyze the success of therapeutic interventions early during the course of the disease.
The most widely used method that is currently accepted to monitor progression of knee OA is the sequential assessment of the joint space width with radiographs. However, radiographs are a relatively insensitive method incompletely assessing morphological degeneration of cartilage, menisci, and other joint tissues; in addition, large studies of 18–24 months’ duration may be required to demonstrate changes [7]. There is also an imperfect correlation of radiographic findings with clinical symptoms [7–10].
Currently, magnetic resonance imaging (MRI) is the best imaging technique to qualitatively and quantitatively analyze joint tissues affected by OA at the knee. Significant associations between MR imaging findings and clinical scores [9, 10] have been found, indicating that OA is a whole-organ degenerative process with articular and periarticular abnormalities contributing to the clinical expression of the disease [11]. Several studies have used MR imaging for the diagnosis and longitudinal follow-up of OA [9, 10, 12, 13]. A number of signs and parameters have been identified, such as cartilage loss and bone marrow edema pattern (BMEP), which may act as potential structural risk factors, demonstrate preclinical changes, or predict disease progression.
Since cartilage lesions and BMEP are hallmarks of OA the assessment of the disease can be performed by the sizing of these findings. For this, several approaches exist: the whole-organ magnetic resonance imaging score (WORMS, [11]) is a semi-quantitative method that has been widely used in clinical and epidemiological studies [14–18]. Quantitative MRI (qMRI) of cartilage morphology has shown promise in the sensitive detection of changes in cartilage thickness during the course of OA [19, 20]. However, this technique requires dedicated software and is time-consuming, since segmentation has to be done manually or semi-automated. Recently, a quantitative morphological score [21, 22] has been introduced that approximates the lesion volume from knee MRIs and can be performed within a relatively short time.
Thus, the aim of this study was to assess the feasibility of this quantitative morphological score for its potential applicability for study evaluation and usage in clinical settings. Therefore we first compared the quantitative morphological score and the semi-quantitative WORMS for analyzing disease progression in a subset of the A9001140-study and second correlated their specific results of structural imaging abnormalities with progression of clinical scores and radiographically determined joint space width.
The study was performed with high-field (HF) MRI with a field strength of 3.0 Tesla (T), since it has been shown thatMR imaging at higher field strengths provides improved signal-to-noise ratio, improved contrast-to-noise ratio and potential shortening of acquisition times, which results in improved visualization of joint morphology [23–26].
Materials and methods
Subjects
Thirty female patients with OA (57.5±8.5 years) and 30 female age-matched controls (54.5±6.8 years) were randomly selected from the A9001140 study after completion of the trial.
The study is described in detail in [20, 27–29]. In brief, it was performed at seven clinical sites and initially included 180 women, of which 152 (age 56.7±8.6 years) completed the trial. Inclusion criteria for all subjects were age≥40 years; good health by medical history, physical examination, and clinical laboratories as well as no contraindications for the use of MRI. Additional inclusion criteria for the patients were radiographic signs of OA (grade 2–3 based on the Kellgren–Lawrence [KL] score [30]), clinical symptoms of knee OA, and a body mass index (BMI)≥30. Patients with inflammatory arthritis were excluded and also those with knee OA secondary to other causes (acute or chronic infection, metabolic abnormalities, previous surgery or history of intra-articular fracture). Controls had no clinical and radiological (KL score=0) evidence of knee OA and a body mass index<30. The focus on obese women in this study was driven by the desire to maximize risk of progression in this relatively small cohort, but not to marginalize men and the non-obese, as female sex and obesity have been reported to be risk factors of OA progression [31–33]. Written informed consent was obtained from all patients after the nature of the examinations had been fully explained. All examinations were performed in accordance with the rules and regulations from the local Human Research Committee.
The 60 subjects for our retrospective analysis were chosen from those four clinical centers of the A9001140 study where MRI was performed with Signa Excite magnets to avoid methodological bias.
Clinical and radiographic assessment of OA
All subjects were evaluated at baseline (BL) and after 12 (follow-up 1, FU1) and 24 (follow-up 2, FU2) months between September 2004 and July 2007. At these time points, several clinical scores and outcome questionnaires were obtained.
A standardized questionnaire (Western Ontario and McMaster Universities Pain, Stiffness, and Physical Function scales, WOMAC) for measuring the degree of pain, functional impairment, and stiffness in all subjects through a 5-point scale (none, slight, moderate, severe, and extreme) was used [34]. The subjects also completed the Short Form General Health Survey (SF-36, [35]), which measures the health-related quality of life. The SF-36 is a validated and widely used instrument that allows quantification of limitation of health quality in eight domains: physical activity, role activity due to physical problems, role activity due to emotional problems, vitality, general mental health, social activity, bodily pain, and general health. Two summary measures of physical (physical component summary scale, PCS) and mental (mental component summary scale, MCS) health are constructed from the eight scales, with higher scores indicating a better health status.
The subjects assessed the severity of arthritis pain in their signal knee at the time of the visit by using a 100-mm Visual Analog Scale (VAS) between 0 (no pain) and 100 (most severe pain).
Lyon–Schuss radiographs [36] were acquired at each visit. Independent quantitative measurements of the minimum joint space width were obtained once in the medial femorotibial compartment of all radiographs by an experienced observer, using digitized image analysis software (Holy’s software, UCLB, Lyon, France, [37])
MRI
Magnetic resonance imaging of the knee joint of the signal knee was performed on a 3.0-Tesla system (Signa; GE Medical systems, Waukesha, WI, USA) using a dedicated knee coil, specifically developed for this study (Clinical MR Solutions, Brookfield, WI, USA) at BL, at FU1, and at FU2. The morphology of cartilage, menisci, ligaments and other knee structures was assessed on three pulse sequences:
A sagittal 2D dual-echo fast spin echo (FSE) sequence with TR/TE1/TE2 3600/8.5/34.1 ms, echo train length of 6, matrix of 256 × 256, in-plane resolution of 0.625 × 0.625 mm2, FOV of 16 cm, one signal average, and a slice thickness of 3 mm
A coronal T1-weighted 3D high-spatial-resolution volumetric water-excite spoiled gradient-echo (SPGR) sequence. Imaging parameters were as follows: TR/TE 17/8.5 ms, flip angle of 12° and number of excitations 0.75. A total of 116 sections were acquired with a field of view (FOV) of 15 cm (matrix 512 × 512) with an in-plane spatial resolution of 0.293 × 0.293 mm2 and a slice thickness of 1.0 mm
A sagittal gradient recalled acquisition in steady state sequence (GRASS) with TR/TE 25/14 ms, flip angle of 40°, number of excitations 0.75, matrix of 512 × 512, in-plane resolution of 0.293 × 0.293, FOV of 15 cm, slice thickness of 1.5 mm and 76 sections were acquired
All images were reviewed on PACS workstations (Agfa, Ridgefield Park, NJ, USA). Three radiologists with experience in musculoskeletal imaging of 19, 7, and 5 years assessed pathological findings of the knee joint independently. If scores were not identical consensus readings by these radiologists were performed in subsequent sessions.
Semi-quantitative morphological score
Pathological findings at the knee joint were analyzed using a modified WORMS [11]. Since only a relatively small number of lesions were expected in these subjects with mild, moderate, and no OA, the number of anatomical compartments was reduced from 15 to 7 compartments and included: the medial and lateral tibia, trochlea, medial and lateral femur, and the medial and lateral patella. Cartilage signal and morphology, osteophytes, bone attrition, medial and lateral meniscal integrity, synovitis/effusion, intra-articular loose bodies, and periarticular cysts/bursitis, anterior and posterior cruciate ligament integrity, as well as medial and lateral collateral ligament integrity were scored as suggested by the original WORMS methodology. To characterize the extent of the regional involvement of bone marrow edema and subchondral cysts, the original partitions of the femoral condyles and tibial plateaus in anterior, central, and posterior regions were used. Any other pathologies not included in a WORMS category were also documented.
Quantitative morphological score
To better quantify small cartilage lesions, the volume of cartilage inhomogeneity (WORMS=1) and the volume of cartilage defects (WORMS≥2) were calculated as previously described [22]. Briefly, the original WORMS method assigns a score of 1 to areas with normal cartilage thickness, but increased signal on T2-weighted images. Accordingly, we defined an area of elevated signals on the sagittal FSE images as “cartilage inhomogeneity” (Fig. 1) and quantified its size by multiplying its largest diameter with the number of slices that it covered (slice thickness in millimeters including the section gap). The volume of focal cartilage defects (WORMS≥2) was approximated on the sagittal FSE images by the following equation:
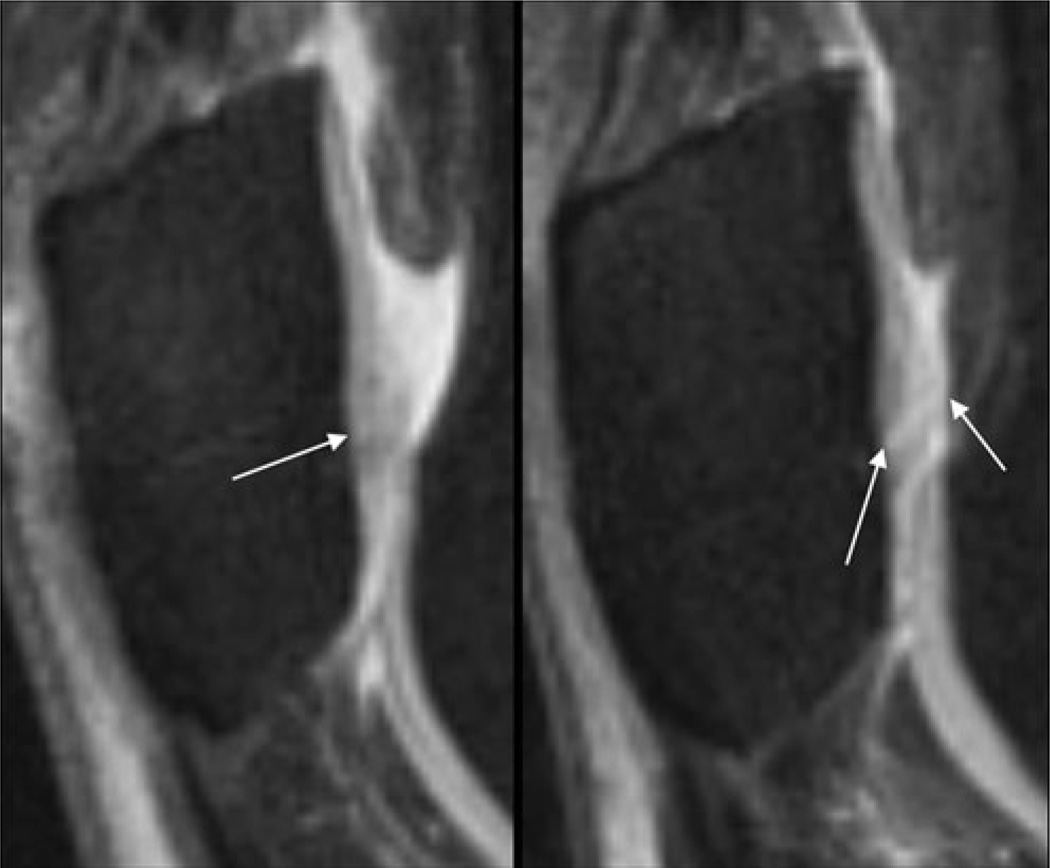
Fig. 1.
Sagittal T2w-FSE images (TR/TE 3,600/8.5 ms) of the lateral left patella in a 63-year-old female osteoarthritis (OA) patient at baseline (BL, left) and after 12 months (FU1, right). At BL, an area of cartilage signal inhomogeneity (arrow) was visible without a clear cartilage defect; therefore, a whole-organ magnetic resonance imaging score (WORMS) of 1 (signal inhomogeneity with normal cartilage thickness) was assigned. At FU1, a fissure of the cartilage is clearly depicted (the maximal extent in the sagittal orientation is outlined by the arrows) yielding a WORMS of 2.5. Semi-quantitative estimated lesion size was 117 mm3 at BL and 170 mm3 at FU1
The depth of the lesion was determined by dividing the surrounding normal cartilage into three layers with equal thickness. If the maximum lesion depth did not exceed the thickness of the superficial layer it was assigned as one third. It became two thirds or three thirds if the maximal lesion depth comprised also the medium or the lowest layer respectively. The shape factor was 1 if more than 50% of the lesion had the largest depth (assessed in the section with the largest diameter), otherwise a shape factor of 0.5 was assigned. Extent of the bone marrow edema pattern was assessed on the FSE images by multiplying the two largest diameters with the number of sections, where it was visualized (slice thickness including gap).
Reproducibility of the quantitative and semi-quantitative measures
The precision of the three radiologists in the quantification of the extent of cartilage lesions and bone marrow edema as well as in the determination of WORMS cartilage and BMEP subscores was expressed by calculating intraclass correlation coefficients (ICCs, [38]) in a sample of ten OA patient image datasets.
Statistical analysis
All the data analysis was performed with PASW Statistics 18, Release Version 18.0.2 (SPSS, Chicago, IL, USA, www.spss.com). Data were initially assessed for normality by visual inspection of their histograms. On the basis of these results mean and standard deviation (SD) by visit and groups were calculated for all numerical variables comprising age, weight, BMI, SF-36 physical and mental component summary scale, and JSW. For the remaining clinical outcome variables (WOMAC, VAS) and the MRI measurements (modified WORMS; quantified sizes of cartilage lesions and BMEP) the median values and the 10% and 90% percentiles are reported. Fisher’s exact test, the t test, and the Mann–Whitney U test were used to compare the significance of individual characteristic variables between the two groups at baseline. In the case of normally distributed data the difference in the mean between the groups and its 95% confidence interval are reported.
The annual change in the measurement and outcome variables over time were assessed by generalized estimation equations (GEE). Fixed effects were given by group, time, interaction of group and time and the adjustment for age and BMI. Random effects were given by subject ID repeated by time. The correlation structure was chosen to be autoregressive first-order. Depending on the distribution of the response, linear or ordinal logistic models were fitted to the data. In a second analysis with GEE models the annual change in the longitudinal data was assessed for all compartments. There was no necessity for adaptation of the correlation structure under the assumption that correlation of observations remained equal among all time points and compartments.
Furthermore, correlation and partial correlation with visit time were analyzed between the measurement variables by compartments and the subject characteristics or outcome variables. This was performed with the Spearman rank correlation coefficient. Findings were not corrected for multiple testing due to the exploratory design of the study. A level of significance of α=0.05 was used throughout the study.
Results
Patient characteristics
Table 1 shows the baseline characteristics of all subjects. Given the subject inclusion criteria, age was not different between the groups whereas OA patients had significantly (p<0.0001) higher weight and BMI.
Table 1.
Subject characteristics at baseline
| Patients | Controls | Diff. mean (95% CI) | p values | |
|---|---|---|---|---|
| n | 30 | 30 | ||
| Age (years) | 57.5±8.5 | 54.5±6.8 | 3.0 (−1.0, 6.9) | 0.143 |
| Weight (kg) | 94.2±14.6 | 64.8±10.2 | 29.4 (22.9, 35.9) | < 0.0001 |
| BMI (kg/m2) | 35.3±4.8 | 24.2±3.2 | 11.1 (9.0, 13.2) | < 0.0001 |
| Kellgren–Lawrence grade (n) | ||||
| Grade 0 | 0 | 30 | ||
| Grade 2 | 15 | 0 | ||
| Grade 3 | 15 | 0 |
Age, weight, and body mass index (BMI) values are reported as means ± standard deviations
Intergroup comparison was performed with t-tests
p values in bold indicate significant group differences
Diff. mean: difference in the mean between the groups; CI: confidence interval
The mean time between BL and FU1 examination was 367.7±8.4 days (95% CI: 362.3; 373.0) in the OA group and 365.8±7.0 days (95% CI: 362.0; 369.5) in the control group. Between FU1 and FU2, 361.0±8.1 days (95% CI: 355.9; 366.2) in the OA group and 367.2±10.0 days (95% CI: 361.8; 372.5) in the control group have passed. These data were not significantly different between the groups (p>0.100).
Clinical and radiographic assessment of OA
Osteoarthritis patients had significantly higher (p<0.0001) values in the WOMAC total score, as well as in the subgroups for pain, stiffness, and function (Table 2). OA patients reported significantly more (p<0.0001) arthritis pain compared with controls when assessed with the visual analog score (VAS). The SF-36 physical component summary scale was significantly (p<0.0001) lower in the patients, whereas the mental component summary scale did not differ between the two groups. Within the OA patients there was no significant difference in the distribution of the clinical OA scores for different KL grades: VAS yielded 19 (0; 68.4) in KL 2 vs 15 (1.5; 68.5) in KL 3 subjects (p=0.880); SF-36 (PCS): 44.0 (25.6; 56.1) vs 42.8 (28.8; 51.3), p=0.949; SF-36 (MCS): 56.3 (32.3; 65.2) vs 59.4 (41.0; 66.9), p=0.290; WOMAC pain: 2 (1.6; 11.4) vs 5 (0; 9.5), p=0.533; WOMAC stiffness: 2 (1; 5.2) vs 2 (0; 5), p=0.999; WOMAC function: 14 (0.6; 48.4) vs 14.5 (1.0; 34.0), p=0.847; and WOMAC total: 23.0 (3.6; 64.4.) vs 22 (2; 47), p=0.539.
Table 2.
Subject characteristics in clinical pain assessment/quality of life scores and joint space width at baseline and after 24 months
| Baselinea | Change from baselineb | ||||||
|---|---|---|---|---|---|---|---|
| Patients | Controls | Diff. mean (95% CI) | p values | Patients | Controls | p values | |
| Pain/quality of life scores | |||||||
| Subject’s assessment of arthritis pain (VAS) | 16 (0; 66) | 1 (0; 12) | <0.0001 | 0.8 | 0.0 | 0.060 | |
| SF-36 physical component summary scale (PCS) | 42.69±10.43 | 53.73±6.38 | −11.0 (−15.5; −6.6) | <0.0001 | −0.77±9.55 | −0.49±5.39 | 0.891 |
| SF-36 mental component summary scale (MCS) | 54.99±10.45 | 54.88±5.50 | 0.1 (−4.2; 4.4) | 0.961 | −2.15±1.66 | −0.73±1.06 | 0.465 |
| WOMAC osteoarthritis index | |||||||
| Pain | 4 (0; 12) | 0 (0; 1) | <0.0001 | 1.5 | 1.0 | 0.293 | |
| Stiffness | 2 (0; 5) | 0 (0; 1) | <0.0001 | 1.0 | 1.0 | 0.397 | |
| Function | 14 (1; 40) | 0 (0; 2) | <0.0001 | 1.1 | 1.0 | 0.490 | |
| Total | 21.5 (0; 58.1) | 0 (0; 4) | <0.0001 | 1.1 | 1.0 | 0.090 | |
| Minimal joint space width (mm) | 3.78±1.01 | 4.05±0.54 | −0.3 (−0.7; 0.1) | 0.189 | −0.37±0.14 | −0.09±0.04 | 0.021 |
Values at baseline are mean ± SD in the case of normally distributed data. Otherwise, median values and 10th and 90th percentiles are reported. Intergroup comparison of the absolute values was conducted with a t-test or a Mann-Whitney-U-test, respectively.
The rate of change during the 24 month-follow-up is reported as mean ± standard error of the mean in the case of normally distributed data. Otherwise, the factor of change of the median in comparison to its value at baseline is shown. Comparison of the slope was performed with generalized estimation equation (GEE) analysis after adjusting for age and BMI
P-values in bold indicate significant group differences
None of the above quoted scores changed significantly at follow-up within the groups.
The minimal joint space width obtained with the modified Lyon–Schuss technique [39, 40] at baseline was slightly, but not significantly, lower in OA patients compared with that of the controls. The amount of change in the JSW after 2 years was significantly (p=0.021) higher in OA patients (−0.37±0.14 mm) than in controls (−0.09±0.04 mm).
Quantitative and semi-quantitative assessment of focal cartilage lesions
Two hundred and ten compartments of the knee were studied in each subject group. At BL a total of 56 (26.7%) compartments in the controls and 119 (56.7%) in the OA patients had cartilage lesions. The distribution of median WORMS of the cartilage lesions and their size is shown in Table 3. Compared with controls, OA patients exhibited at BL significantly higher (p<0.014) cartilage lesion WORMS and sizes in all compartments and in the whole knee (median value [10th percentile, 90th percentile]: WORMS: 14 [6.5, 21] in OA patients vs 2 [0, 10.2] in controls; size: 353 [146, 1, 339] in OA patients vs 54.4 [0, 253.8] in controls). These significant differences between the groups persisted at all follow-up time points.
Table 3.
Distribution of cartilage lesions in terms of their specific semi-quantitative whole-organ magnetic resonance imaging score (WORMS) and quantitative lesion sizes in the subject groups
| Compartment | Month | Semiquantitative score (WORMS) | Quantitative score (size) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Patients | Controls | p values | Patients | Controls | p values | ||||
| Slopea | BLb | Slopea | BLb | ||||||
| Patella | |||||||||
| Lateral | BL | 4 (0; 6) | 1 (0; 4.2) | 0.004 | 66 (0; 408) | 6.5 (0; 156.9) | 0.009 | ||
| 12 | 4 (0; 6) | 1 (0; 4.2) | 0.003 | 80 (0; 420) | 8 (0; 169.2) | 0.010 | |||
| 24 | 4 (0; 6) | 1 (0; 5.1) | 80 (0; 420) | 8.5 (0; 180) | |||||
| Medial | BL | 2.5 (0; 6) | 0 (0; 4.1) | 64 (0; 396) | 0 (0; 121.5) | 0.014 | |||
| 12 | 2.5 (0; 6) | 0 (0; 4.1) | 0.533 | 42 (0; 396) | 0 (0; 117.3) | 0.051 | |||
| 24 | 2 (0; 6) | 0 (0; 4.1) | 51 (0; 396) | 0 (0; 126.9) | |||||
| Femur | |||||||||
| Lateral | BL | 0 (0; 5) | 0 (0; 0) | 0.011 | 0 (0; 189) | 0 (0; 0) | 0.008 | ||
| 12 | 0 (0; 5) | 0 (0; 0) | 0.919 | 0 (0; 189) | 0 (0; 0) | 0.054 | |||
| 24 | 0 (0; 5) | 0 (0; 0) | 0 (0; 189) | 0 (0; 0) | |||||
| Trochlea | BL | 2 (0; 5) | 0 (0; 3.2) | 0.008 | 45 (0; 357) | 0 (0; 60.3) | 0.001 | ||
| 12 | 2.5 (0; 5) | 0 (0; 3.2) | 0.066 | 40 (0; 384) | 0 (0; 63.9) | 0.002 | |||
| 24 | 3 (0; 5) | 0 (0; 3.2) | 88 (0; 408) | 0 (0; 65.1) | |||||
| Medial | BL | 3 (0; 5) | 0 (0; 0) | <0.0001 | 10 (0; 345) | 0 (0; 0) | <0.0001 | ||
| 12 | 3 (0; 5) | 0 (0; 0) | 0.120 | 38 (0; 338) | 0 (0; 0) | 0.008 | |||
| 24 | 3 (0; 5) | 0 (0; 0) | 36 (0; 360) | 0 (0; 0) | |||||
| Tibia | |||||||||
| Lateral | BL | 1 (0; 3) | 0 (0; 1) | 0.002 | 12 (0; 144) | 0 (0; 72.6) | 0.006 | ||
| 12 | 1 (0; 4) | 0 (0; 1) | 0.132 | 14 (0; 120) | 0 (0; 74.4) | 0.053 | |||
| 24 | 1 (0; 4) | 0 (0; 1) | 15 (0; 135) | 0 (0; 58.2) | |||||
| Medial | BL | 0 (0; 5) | 0 (0; 0) | <0.0001 | 0 (0; 96) | 0 (0; 0) | 0.001 | ||
| 12 | 0 (0; 5) | 0 (0; 0) | 0.551 | 0 (0; 90) | 0 (0; 0) | 0.022 | |||
| 24 | 0 (0; 5) | 0 (0; 0) | 0 (0; 90) | 0 (0; 0) | |||||
| Whole knee | BL | 14 (6.5; 21) | 2 (0; 10.2) | <0.0001 | 353 (146; 1,339) | 54.5 (0; 253.8) | <0.0001 | ||
| 12 | 14 (8; 21) | 2 (0; 10.2) | 0.082 | 356 (157; 1,384) | 55.5 (0; 249.6) | 0.026 | |||
| 24 | 14 (9; 21) | 2 (0; 10.2) | 397 (176; 1,402) | 76 (0; 278.7) | |||||
Results are median values and the 10th and 90th percentiles
p values in bold indicate significant group differences
Intergroup comparison of the slope was performed with generalized estimation equations (GEE) after adjustment for age and BMI
Intergroup comparison of the absolute values at baseline (BL) was conducted with Mann–Whitney U tests
In the GEE model the OA patients had a significant (p<0.010) increase in cartilage WORMS and cartilage lesion size at the lateral patella in comparison to the controls (Fig. 1). In addition, the cartilage lesion size increased significantly (p<0.026) over time at the trochlea (Fig. 2), the medial femur, the medial tibia, and in the whole knee in the OA patients.
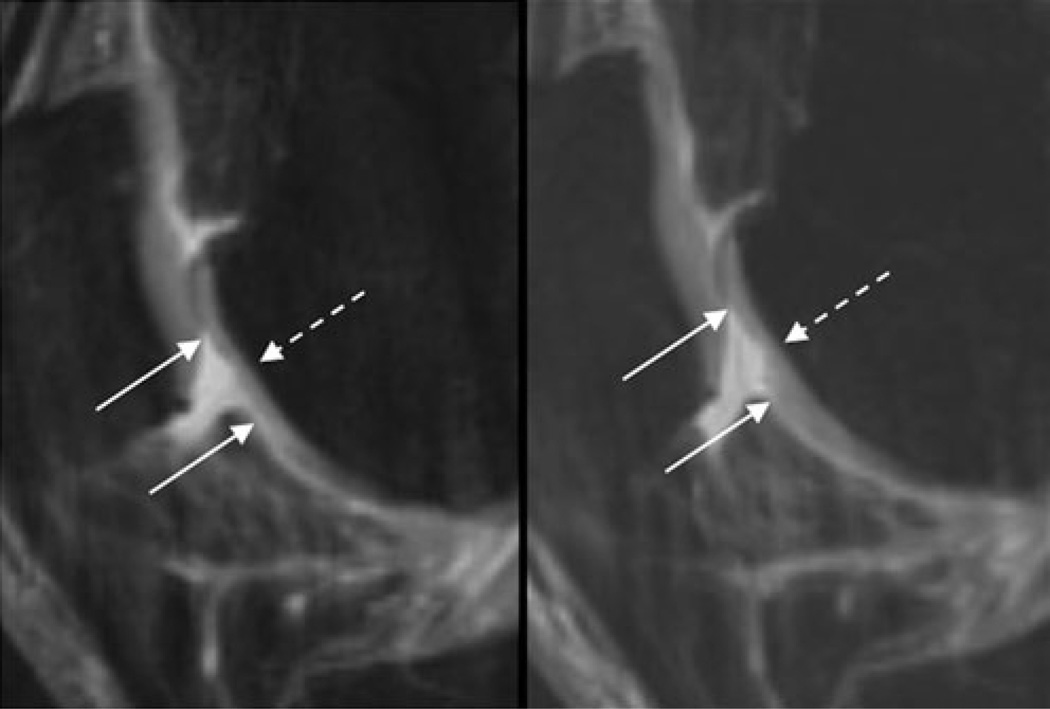
Fig. 2.
Sagittal T2w-FSE images (TR/TE 3,600/8.5 ms) of the right trochlea in a 50-year-old female OA patient at BL (left) and after 12 months (FU1, right). At both time points, a 12-mm-wide partial thickness defect of the cartilage is shown (the maximal extent of this lesion in cranio-caudal extent is outlined by the solid arrows); therefore, a WORMS of 3 has been assigned. At BL, only the superficial layer of the cartilage is affected, whereas at FU1 the middle and parts of the basal layer are also impaired, resulting in a semi-quantitative estimated lesion size of 53 mm3 at BL and 126 mm3 at FU1
Using the WORMS 3.8% of the cartilage lesions in OA patients increased, while 96.2% of the lesions remained unchanged. In controls, 2.5% of the cartilage lesions increased; for the remaining, no change was observed. With the quantitative score an increase in cartilage lesion size was shown in 23.8% of the lesions in OA patients, no change occurred in 61.9%, and a decrease was observed in 14.2%. For controls, these values were 6.6%, 89.8%, and 3.6% respectively.
Quantitative and semi-quantitative assessment of BMEP
Eighteen out of 210 (8.6%) compartments in controls and 49 out of 210 (23.3%) compartments in OA patients had a BMEP at BL. The OA group had significantly (p<0.012) higher BMEP WORMS and BMEP sizes at BL at the lateral tibia and in the whole knee (WORMS: 2 [0, 6] in OA patients vs 0 [0, 2.2] in controls; size: 291 [0, 4, 788] in OA patients vs 0 [0, 537.9] in controls; Table 4). In addition, the BMEP size was significantly (p=0.002) higher at the medial tibia.
Table 4.
Distribution of bone marrow edema pattern (BMEP) in terms of their semi-quantitative WORMS and quantitative lesion sizes in the subject groups
| Compartment | Month | Semiquantitative score (WORMS) | Quantitative score (size) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Patients | Controls | p values | Patients | Controls | p values | ||||
| Slopea | BLb | Slopea | BLb | ||||||
| Patella | |||||||||
| Lateral | BL | 0 (0; 2) | 0 (0; 1.1) | 0.284 | 0 (0; 360) | 0 (0; 333.9) | 0.300 | ||
| 12 | 0 (0; 1) | 0 (0; 1.1) | 0.055 | 0 (0; 495) | 0 (0; 205.2) | 0.013 | |||
| 24 | 0 (0; 1) | 0 (0; 2) | 0 (0; 576) | 0 (0; 381.6) | |||||
| Medial | BL | 0 (0; 2) | 0 (0; 1) | 0.460 | 0 (0; 240) | 0 (0; 206.1) | 0.468 | ||
| 12 | 0 (0; 2) | 0 (0; 1) | 0.158 | 0 (0; 216) | 0 (0; 129.6) | 0.906 | |||
| 24 | 0 (0; 1) | 0 (0; 1) | 0 (0; 192) | 0 (0; 135) | |||||
| Femur | |||||||||
| Lateral | BL | 0 (0; 1) | 0 (0; 0) | 0.172 | 0 (0; 288) | 0 (0; 0) | 0.158 | ||
| 12 | 0 (0; 1) | 0 (0; 0) | 0.042 | 0 (0; 864) | 0 (0; 0) | 0.001 | |||
| 24 | 0 (0; 2) | 0 (0; 0) | 0 (0; 1,248) | 0 (0; 0) | |||||
| Trochlea | BL | 0 (0; 2) | 0 (0; 1.1) | 0.091 | 0 (0; 630) | 0 (0; 126) | 0.076 | ||
| 12 | 0 (0; 2) | 0 (0; 1) | 0.721 | 0 (0; 975) | 0 (0; 180.9) | 0.001 | |||
| 24 | 0 (0; 2) | 0 (0; 1) | 0 (0; 1,260) | 0 (0; 198) | |||||
| Medial | BL | 0 (0; 1) | 0 (0; 0) | 0.177 | 0 (0; 108) | 0 (0; 0) | 0.187 | ||
| 12 | 0 (0; 1) | 0 (0; 0) | 0.215 | 0 (0; 294) | 0 (0; 0) | 0.368 | |||
| 24 | 0 (0; 1) | 0 (0; 0) | 0 (0; 294) | 0 (0; 0) | |||||
| Tibia | |||||||||
| Lateral | BL | 0 (0; 2) | 0 (0; 0) | 0.012 | 0 (0; 3078) | 0 (0; 0) | 0.011 | ||
| 12 | 0 (0; 2) | 0 (0; 0) | 0.552 | 0 (0; 2160) | 0 (0; 0) | <0.0001 | |||
| 24 | 0 (0; 2) | 0 (0; 1) | 0 (0; 2352) | 0 (0; 33.3) | |||||
| Medial | BL | 0 (0; 1) | 0 (0; 0.1) | 0.051 | 0 (0; 486) | 0 (0; 21.6) | 0.002 | ||
| 12 | 0 (0; 2) | 0 (0; 0.1) | 0.947 | 0 (0; 450) | 0 (0; 1.2) | 0.699 | |||
| 24 | 0 (0; 2) | 0 (0; 1) | 0 (0; 702) | 0 (0; 67.5) | |||||
| Whole knee | BL | 2 (0; 6) | 0 (0; 2.2) | <0.0001 | 291 (0; 4788) | 0 (0; 537.9) | 0.002 | ||
| 12 | 2 (0; 8) | 0 (0; 2.2) | 0.174 | 288 (0; 4311) | 0 (0; 559.2) | 0.047 | |||
| 24 | 2 (0; 8) | 0 (0; 3.1) | 432 (0; 6924) | 63 (0; 1,377) | |||||
Results are median values and 10th/90th percentiles
p values in bold indicate significant group differences
Intergroup comparison of the slope was performed with generalized estimation equations (GEE) after adjustment for age and BMI
Intergroup comparison of the absolute values at baseline (BL) was conducted with Mann–Whitney U tests
The change over time demonstrated mixed results in the GEE model: a significant (p<0.042) increase in BMEP WORMS and BMEP size was observed in OA patients at the lateral femur. At the lateral tibia, BMEP size in OA patients revealed a significant (p<0.0001) decrease at FU1 and an increase at FU2, whereas controls had a slight increase at each follow-up examination (Fig. 3). At the trochlea, OA patients showed a significantly (p=0.001) higher increase in BMEP size during the follow-up than controls. At the lateral patella, controls had a significant (p=0.013) decrease in BMEP size at FU1 and an increase at FU2, whereas OA patients exhibited an increase over time. The BMEP size values comprising the whole knee yielded a significant (p=0.047) increase in controls, whereas in OA patients the size decreased at FU1 and increased at FU2.
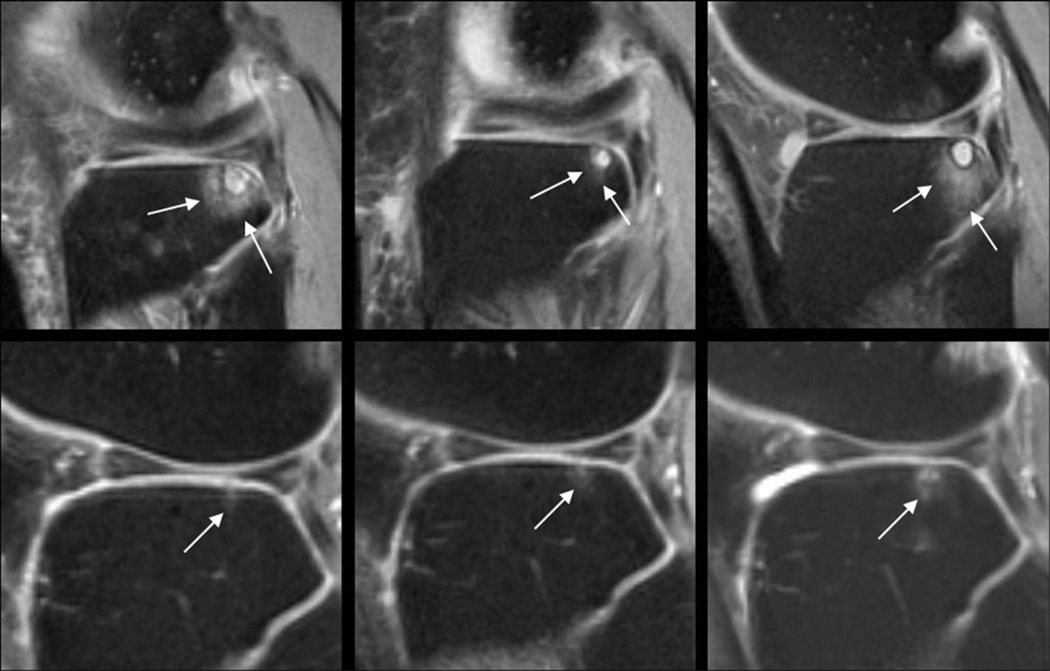
Fig. 3.
Sagittal T2w-FSE images (TR/TE 3,600/8.5 ms) of the lateral tibia at BL (left), FU1 (center), and at FU2 (right) of the left knee in a 60-year-old female OA patient (upper row) and a 59-year-old female OA control (lower row). The patient had a large area of bone marrow edema pattern (BMEP) at BL (WORMS of 2 and 4,095 mm3 in size), which encompasses a subchondral cyst. The BMEP decreased at FU1 (WORMS 1; size 504 mm3) and increased again at FU2 (WORMS 2; size 6,318 mm3). The control subject had a small BMEP at BL, which showed a slight increase at the follow-up examinations. The WORMS was 1 for all time points; the size was 18 mm3, 48 mm3, and 90 mm3 at BL, FU1, and FU2 respectively
Applying the WORMS, BMEP decreased in 3.4% and increased in 4.9% of the lesions in the OA patients. These values were 9.2% and 1.8% for controls respectively. Using the quantitative score in OA patients, an increase in 12.8%, no change in 73.4%, and a decrease in 13.8% of the lesions were observed. For controls, these values were 9.8%, 88.3%, and 2.6% respectively.
Changes in focal cartilage lesions and BMEP were not statistically significant (p>0.165) between OA patients with KL grade 3 and KL grade 2, irrespective of which scoring system was used.
Correlation between quantitative/semi-quantitative scores and clinical/radiographic parameters of OA during follow-up
In the controls, changes in the SF-36 PCS score significantly positively correlated with changes in the quantitative BMEP values (WORMS: 0.400, p=0.035; size: r=0.520, p=0.005) for the whole knee.
In OA patients, several significantly positive correlations between changes in the assessed parameters were observed: WOMAC pain scores with quantitative cartilage lesion size at the lateral tibia (r=0.409; p=0.034) and at the lateral patella (r=0.450; p=0.018) as well as with cartilage WORMS at the lateral tibia (r=0.380, p=0.042); WOMAC stiffness scores with cartilage WORMS at the lateral tibia (r=0.412, p=0.027), and the lateral patella (r=0.411, p=0.033) as well as with quantitative BMEP size at the lateral patella (r=0.382, p=0.041); WOMAC function scores with quantitative BMEP size at the medial tibia (r=0.419, p=0.030), and quantitative cartilage lesion size at the lateral femur (r=0.397, p=0.044); WOMAC total scores with the surface WORMS of the lateral femur (r=0.388, p=0.046); SF-36 PCS with quantitative BMEP size at the medial tibia (r=0.453; p=0.018) and at the whole knee (r=0.435, p=0.023); VAS with quantitative cartilage lesion size at the lateral patella (r=0.397, p=0.040); JSW with surface WORMS of the trochlea (r=0.418, p=0.033); and WORMS cartilage lesion size at the whole knee (r=0.406, p=0.039).
Whole knee pathology
The median global knee WORMS at BL (median value [10th percentile, 90th percentile]) was 35.25 (22.1; 51.3) in OA patients and 7 (2; 15) in controls, which was also significantly (p<0.0001) different between the groups for the follow-up. For OA patients, the values were 35.5 (24; 53.4) at FU1and 35.5 (24; 54) at FU2. Controls had a global median knee WORMS of 8 (2; 15) at each follow-up examination. The slope of these values between the subject groups was not significantly (p=0.179) different in the GEE model.
Reproducibility of the quantitative MRI measurements
Results of the interobserver reliability assessments in the subset of the OA data indicated good agreement: ICCs for cartilage lesion size and BMEP size were 0.84 and 0.86. For the WORMS cartilage and BMEP subscores the ICCs were 0.82 and 0.89 respectively.
Discussion
In our study we sought to assess the feasibility of a quantitative score for monitoring changes in cartilage lesion and BMEP size at the knee joint in subjects with and without early OA using 3.0 T MRI. Findings were compared with the semi-quantitative WORMS, which was developed as a multi-feature, whole organ evaluation method of the knee in OA. Results were correlated with clinical and radiographic findings. A subset of the A9001140 study subjects was retrospectively analyzed. The study comprised an observation period of 2 years with a total of three surveys including 12- and 24-month follow-up examinations.
As expected, OA patients had higher clinical parameters of OA and higher WORMS global scores of the whole knee. They presented with significantly higher semi-quantitative cartilage WORMS scores and quantitative determined cartilage lesion sizes in all compartments of the knee at BL. During follow-up, a significant increase in the cartilage lesion WORMS score and size in OA patients compared with the controls occurred at the lateral patella. These results could be explained by increasing cartilage degeneration during OA progression, including preceding thinning and erosion. It is in agreement with previous findings, with the patellofemoral compartment being the most affected [41], and also in concordance with the radiographic findings, showing a significant reduction in JSW in our patients. However, applying the quantitative score an increase in cartilage lesion size was additionally observed at the trochlea, at the medial compartment, and in the whole knee, indicating a higher sensitivity to subtle cartilage changes of this approach than the WORMS. This is supported by the findings of Le Graverand et al. [20], who analyzed 145 subjects from the A9001140 study and revealed a significant decrease in cartilage thickness in the central medial femorotibial compartment in OA patients with a KL grade of 3.
Using the semi-quantitative WORMS, 3.8% of the cartilage lesions in OA patients and 2.5% in controls increased. However, by using the quantitative score the rate of change was also more sensitively monitored than with the WORMS score: an increase in cartilage lesion size was shown in 23.8% of the lesions in OA patients and in 6.6% of the controls; a decrease was observed in 14.2% of the OA patients and in 3.6% of the controls.
Bone marrow edema pattern had significantly higher WORMS scores and sizes in OA patients when evaluated for the whole knee. The frequency of this pathology at BL was 23.3% in OA patients and 8.6% in controls. This is in agreement with the literature and reflects the high prevalence of this abnormality in OA, but also to a lesser extent in healthy subjects [9, 12, 13, 16, 42–47]. The change at the follow-up examinations yielded mixed results: an increase over all time points was observed in OA patients at the lateral femur with both scores. The quantitative score revealed in addition in OA patients a significant increase at the trochlea and at the lateral patella as well as a temporary reduction at the lateral tibia; controls had a small increase in BMEP size at the lateral tibia and a small decrease at FU1 at the lateral patella.
Applying the WORMS, BMEP decreased in 3.4% and increased in 4.9% of the lesions in our OA patients, whereas a decrease was found in 13.8% and an increase in 12.8% when the lesions were assessed with the quantitative score. For controls, these numbers were 9.2% and 1.8% applying the WORMS in comparison to 9.8% and 2.6% for the quantitative score. Therefore—similar to cartilage lesions—the quantitative score is more sensitive to changes in the BMEP sizes than the WORMS score. However, the fluctuation of BMEP parallels that of other studies, where in OA patients after 2 years a significant change in up to 66–80% of the lesions is reported [13, 47]. It illustrates the dynamic process of this imaging parameter in the course of the disease. Therefore, care should be taken when BMEP is utilized as a surrogate endpoint.
We demonstrated low correlations between changes in the WOMAC total score and its subscores with changes in cartilage and BMEP morphology indices in the OA patients in several subregions of the knee, whereby mostly the lateral compartments were affected. Changes in SF-36 PCS and VAS scores correlated in a considerably lower amount of compartments with changes in cartilage or BMEP morphology. We conclude that the use of a disease-specific health score like WOMAC is more sensitive in the evaluation of changes in the early disease processes in contrast to the utilization of only general outcomes scores like SF-36 or VAS. However, since changes in volume approximation of cartilage and BMEP lesions were almost equally correlated with changes in the clinical parameters, like changes in the respective WORMS scores we think that both methods have a comparable sensitivity to draw conclusions from morphology as to the clinical status of the disease. The overall lack of strong correlations of clinical OA scores with morphological findings may be related to the fact that the knee becomes more accustomed to cartilage damage. Link et al. [10] found most symptoms in patients with lower grade cartilage lesions and concluded that clinical symptoms are more substantial at the onset of OA. Furthermore, controversy still exists about the cause of knee pain in OA patients. Felson et al. [43] found a moderately strong association between the change in BMEP with the development of frequent knee pain, whereas this association was not found by Link et al. [10]. Kornaat et al. [48] found that the presence of large central osteophytes are associated with pain. Hunter et al. [49] reported that alterations in patella volume are associated with pain and other subscores of the WOMAC. Since hyaline cartilage does not contain pain fibers it is hypothesized that other factors like inflammation within the joint or enlargement of effusions might also play an important role [43].
Our study was conducted with HF-MRI at 3.0 Tesla. Visualization of the morphological and pathological features of cartilage has been shown to be improved at 3.0 T compared with the clinically widely-used 1.5-T systems in cadaver studies [23, 24] and in specimens [23, 26]. One recent larger scale clinical study [25] utilizing MRI at 3.0 T and validation of the findings with arthroscopy has shown further promise with higher specificity, higher accuracy, and higher diagnostic confidence for detecting cartilage lesions, but not for higher sensitivity. However, Wong et al. [50] found even increased sensitivity in a similar setting with a smaller sample size. We therefore assume that detection of lesions and estimation of their size in our study was improved in comparison to using lower field strengths.
Several limitations can be identified in our study. First is the small sample size of subjects in the OA patient and control groups. Second, there was no true gold standard in the study; none of the study subjects underwent arthroscopy, which could not be ethically justified in healthy subjects. Also, no histopathological examination has been performed. However, the subjects were thoroughly monitored. Careful simultaneous assessment of accepted clinical and radiographic progress indicators of OA was also performed. Third, obesity is a known risk factor for OA. Although differences in BMI were outlined in the inclusion criteria, BMI was adjusted for the analysis of each group given the presence of confounding. Fourth, reducing WORMS partitioning to seven compartments could have potentially affected the number of grade 4 or grade 6 lesions. Grade 6 lesions, however, were not expected in this cohort with early disease and grade 4 lesions are very rare, as usually full thickness lesions are present before >75% partial thickness lesions in one subcompartment occur. Finally, our study was limited to women, so it is questionable whether the results could be extrapolated to men.
In summary, with our retrospective analysis of longitudinal data at 3.0 T by application of a quantitative scoring system, we were able to detect changes in cartilage and BMEP morphology between OA patients and controls within a follow-up period of 24 months. More compartments of the knee in OA patients are affected by changes in comparison to the semi-quantitative WORMS score. The amount of lesions that experience changes is also higher with the quantitative scoring system. Since a progression of the disease in our OA patients is indicated by significant narrowing of the JSW, we conclude that the quantitative score is more sensitive to changes in cartilage and BMEP morphology in the course of the disease than the semi-quantitative technique. Since it has a good reproducibility and can be applied relatively quickly, we believe that the quantitative score is feasible for study evaluation and has potential usage in clinical settings. However, verification in a larger number of patients is needed to ascertain whether the method could be used as an alternative to time-consuming approaches like qMRI. Only weak associations were found when the rate of change of the lesions correlated with health-related quality of life, as assessed by the WOMAC and SF-36 scores. But these findings were not unexpected and had been demonstrated by previous studies [9, 10], indicating that structural imaging findings do not correlate well with the clinical progression of OA.
Acknowledgement
This work was supported by Pfizer Inc., Eastern Point Road, Groton, CT, 06340, USA. We are grateful to the dedicated group of A9001140 investigators: Cecil Charles, Julia Crim, David Hunter, Gary Hutchins, Chris Jackson, Virginia Byers Kraus, Steve Mazzuca, Prasad Pottumarthi, Thomas Schnitzer, Mihra Taljanovic, and Berchman Vaz. Also to the dedicated study coordinators whose skills were essential in ensuring the successful conduct of this research: Emily Brown, Sandra Chapman, Eugene Dunkle, Kristen Fredley, Donna Gilmore, Joyce Goggins, Mohsen Haddad-Kaveh, Norine Hall, Thelma Munoz, and Kim Tally. We would also like to express our thanks to the dedicated MRI technologists, the Duke Image Analysis Laboratory staff: Cecil Charles, Maureen Ainslie, April Davis, Allison Fowlkes, Mark Ward, Scott White, and the Pfizer A9001140 Team: Lydia Brunstetter, Peggy Coyle, Yevgenia Davidoff, Charles Packard, Ann Remmers, Mark Tengowski, Jeff Evelhoch (now Merck,West Point, PA, USA), and John Kotyk (now Washington University, St. Louis, MI, USA).
Conflicts of interest/funding and grants Dr. Hellio Le Graverand-Gastineau and B. Wyman are employees of Pfizer. Drs. Majumdar and Link were partially funded by Pfizer. All investigators worked under grants received by Pfizer to conduct the A9001140 study.
Contributor Information
Robert Stahl, Email: Robert.Stahl@med.uni-muenchen.de, Department of Radiology, University of California, San Francisco, 185 Berry Street, Suite 350, San Francisco, CA 94107–0946, USA; Department of Clinical Radiology, University Hospitals, Campus Grosshadern, Ludwig Maximilians University of Munich, Marchioninistrasse 15, 81377 Munich, Germany.
Sapna K. Jain, Department of Radiology, University of California, San Francisco, 185 Berry Street, Suite 350, San Francisco, CA 94107–0946, USA
Jürgen Lutz, Department of Neuroradiology, University Hospitals, Campus Grosshadern, Ludwig Maximilians University of Munich, Marchioninistrasse 15, 81377 Munich, Germany.
Bradley T. Wyman, Pfizer Inc., Eastern Point Road, Groton, CT 06340, USA
Marie-Pierre Hellio Le Graverand-Gastineau, Pfizer Inc., Eastern Point Road, Groton, CT 06340, USA.
Eric Vignon, Claude Bernard University Lyon I, 69495 Lyon, France.
Sharmila Majumdar, Department of Radiology, University of California, San Francisco, 185 Berry Street, Suite 350, San Francisco, CA 94107–0946, USA.
Thomas M. Link, Department of Radiology, University of California, San Francisco, 185 Berry Street, Suite 350, San Francisco, CA 94107–0946, USA
References
- 1.Centers for Disease Control and Prevention (CDC) Prevalence of disabilities and associated health conditions among adults—United States, 1999. MMWR Morb Mortal Wkly Rep. 2001;50(7):120–125. [PubMed] [Google Scholar]
- 2.Centers for Disease Control and Prevention (CDC) Update: direct and indirect costs of arthritis and other rheumatic conditions–United States, 1997. MMWR Morb Mortal Wkly Rep. 2004;53(18):388–389. [PubMed] [Google Scholar]
- 3.Centers for Disease Control and Prevention (CDC) Racial/ethnic differences in the prevalence and impact of doctor-diagnosed arthritis—United States, 2002. MMWR Morb Mortal Wkly Rep. 2005;54(5):119–123. [PubMed] [Google Scholar]
- 4.Felson DT, Lawrence RC, Dieppe PA, Hirsch R, Helmick CG, Jordan JM, et al. Osteoarthritis: new insights. I. The disease and its risk factors. Ann Intern Med. 2000;133(8):635–646. doi: 10.7326/0003-4819-133-8-200010170-00016. [DOI] [PubMed] [Google Scholar]
- 5.Centers for Disease Control and Prevention (CDC) Prevalence of self-reported arthritis or chronic joint symptoms among adults—United States, 2001. MMWR Morb Mortal Wkly Rep. 2002;51(42):948–950. [PubMed] [Google Scholar]
- 6.Reginster JY. The prevalence and burden of arthritis. Rheumatology (Oxford) 2002;41(Supp 1):3–6. [PubMed] [Google Scholar]
- 7.Buckland-Wright JC. Quantitative radiography of osteoarthritis. Ann Rheum Dis. 1994;53(4):268–275. doi: 10.1136/ard.53.4.268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hannan MT, Felson DT, Pincus T. Analysis of the discordance between radiographic changes and knee pain in osteoarthritis of the knee. J Rheumatol. 2000;27(6):1513–1517. [PubMed] [Google Scholar]
- 9.Hayes CW, Jamadar DA, Welch GW, Jannausch ML, Lachance LL, Capul DC, et al. Osteoarthritis of the knee: comparison of MR imaging findings with radiographic severity measurements and pain in middle-aged women. Radiology. 2005;237(3):998–1007. doi: 10.1148/radiol.2373041989. [DOI] [PubMed] [Google Scholar]
- 10.Link TM, Steinbach LS, Ghosh S, Ries M, Lu Y, Lane N, et al. Osteoarthritis: MR imaging findings in different stages of disease and correlation with clinical findings. Radiology. 2003;226(2):373–381. doi: 10.1148/radiol.2262012190. [DOI] [PubMed] [Google Scholar]
- 11.Peterfy CG, Guermazi A, Zaim S, Tirman PF, Miaux Y, White D, et al. Whole-Organ Magnetic Resonance Imaging Score (WORMS) of the knee in osteoarthritis. Osteoarthritis Cartilage. 2004;12(3):177–190. doi: 10.1016/j.joca.2003.11.003. [DOI] [PubMed] [Google Scholar]
- 12.Garnero P, Peterfy C, Zaim S, Schoenharting M. Bone marrow abnormalities on magnetic resonance imaging are associated with type II collagen degradation in knee osteoarthritis: a three-month longitudinal study. Arthritis Rheum. 2005;52(9):2822–2829. doi: 10.1002/art.21366. [DOI] [PubMed] [Google Scholar]
- 13.Phan CM, Link TM, Blumenkrantz G, Dunn TC, Ries MD, Steinbach LS, et al. MR imaging findings in the follow-up of patients with different stages of knee osteoarthritis and the correlation with clinical symptoms. Eur Radiol. 2006;16(3):608–618. doi: 10.1007/s00330-005-0004-5. [DOI] [PubMed] [Google Scholar]
- 14.Amin S, LaValley MP, Guermazi A, Grigoryan M, Hunter DJ, Clancy M, et al. The relationship between cartilage loss on magnetic resonance imaging and radiographic progression in men and women with knee osteoarthritis. Arthritis Rheum. 2005;52(10):3152–3159. doi: 10.1002/art.21296. [DOI] [PubMed] [Google Scholar]
- 15.Carbone LD, Nevitt MC, Wildy K, Barrow KD, Harris F, Felson D, et al. The relationship of antiresorptive drug use to structural findings and symptoms of knee osteoarthritis. Arthritis Rheum. 2004;50(11):3516–3525. doi: 10.1002/art.20627. [DOI] [PubMed] [Google Scholar]
- 16.Felson DT, Niu J, Guermazi A, Roemer F, Aliabadi P, Clancy M, et al. Correlation of the development of knee pain with enlarging bone marrow lesions on magnetic resonance imaging. Arthritis Rheum. 2007;56(9):2986–2992. doi: 10.1002/art.22851. [DOI] [PubMed] [Google Scholar]
- 17.Neogi T, Felson D, Niu J, Lynch J, Nevitt M, Guermazi A, et al. Cartilage loss occurs in the same subregions as subchondral bone attrition: a within-knee subregion-matched approach from the Multicenter Osteoarthritis Study. Arthritis Rheum. 2009;61(11):1539–1544. doi: 10.1002/art.24824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Roemer FW, Neogi T, Nevitt MC, Felson DT, Zhu Y, Zhang Y, et al. Subchondral bone marrow lesions are highly associated with, and predict subchondral bone attrition longitudinally: the MOST study. Osteoarthritis Cartilage. 2010;18(1):47–53. doi: 10.1016/j.joca.2009.08.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Eckstein F, Burstein D, Link TM. Quantitative MRI of cartilage and bone: degenerative changes in osteoarthritis. NMR Biomed. 2006;19(7):822–854. doi: 10.1002/nbm.1063. [DOI] [PubMed] [Google Scholar]
- 20.Le Graverand MP, Buck RJ, Wyman BT, Vignon E, Mazzuca SA, Brandt KD, et al. Change in regional cartilage morphology and joint space width in osteoarthritis participants versus healthy controls: a multicentre study using 3.0 Tesla MRI and Lyon-Schuss radiography. Ann Rheum Dis. 2010;69(1):155–162. doi: 10.1136/ard.2008.099762. [DOI] [PubMed] [Google Scholar]
- 21.Stahl R, Krug R, Kelley DA, Zuo J, Ma CB, Majumdar S, et al. Assessment of cartilage-dedicated sequences at ultra-high-field MRI: comparison of imaging performance and diagnostic confidence between 3.0 and 7.0 T with respect to osteoarthritis-induced changes at the knee joint. Skeletal Radiol. 2009;38(8):771–783. doi: 10.1007/s00256-009-0676-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Stahl R, Luke A, Ma CB, Krug R, Steinbach L, Majumdar S, et al. Prevalence of pathologic findings in asymptomatic knees of marathon runners before and after a competition in comparison with physically active subjects—a 3.0 T magnetic resonance imaging study. Skeletal Radiol. 2008;37(7):627–638. doi: 10.1007/s00256-008-0491-y. [DOI] [PubMed] [Google Scholar]
- 23.Bauer JS, Barr C, Henning TD, Malfair D, Ma CB, Steinbach L, et al. Magnetic resonance imaging of the ankle at 3.0 Tesla and 1.5 Tesla in human cadaver specimens with artificially created lesions of cartilage and ligaments. Invest Radiol. 2008;43(9):604–611. doi: 10.1097/RLI.0b013e31817e9ada. [DOI] [PubMed] [Google Scholar]
- 24.Fischbach F, Bruhn H, Unterhauser F, Ricke J, Wieners G, Felix R, et al. Magnetic resonance imaging of hyaline cartilage defects at 1.5 T and 3.0 T: comparison of medium T2-weighted fast spin echo, T1-weighted two-dimensional and three-dimensional gradient echo pulse sequences. Acta Radiol. 2005;46(1):67–73. doi: 10.1080/02841850510012625. [DOI] [PubMed] [Google Scholar]
- 25.Kijowski R, Blankenbaker DG, Davis KW, Shinki K, Kaplan LD, De Smet AA. Comparison of 1.5- and 3.0-T MR imaging for evaluating the articular cartilage of the knee joint. Radiology. 2009;250(3):839–848. doi: 10.1148/radiol.2503080822. [DOI] [PubMed] [Google Scholar]
- 26.Masi JN, Sell CA, Phan C, Han E, Newitt D, Steinbach L, et al. Cartilage MR imaging at 3.0 versus that at 1.5 T: preliminary results in a porcine model. Radiology. 2005;236(1):140–150. doi: 10.1148/radiol.2361040747. [DOI] [PubMed] [Google Scholar]
- 27.Eckstein F, Buck RJ, Burstein D, Charles HC, Crim J, Hudelmaier M, et al. Precision of 3.0 Tesla quantitative magnetic resonance imaging of cartilage morphology in a multicentre clinical trial. Ann Rheum Dis. 2008;67(12):1683–1688. doi: 10.1136/ard.2007.076919. [DOI] [PubMed] [Google Scholar]
- 28.Eckstein F, Buck RJ, Wyman BT, Kotyk JJ, Le Graverand MP, Remmers AE, et al. Quantitative imaging of cartilage morphology at 3.0 Tesla in the presence of gadopentate dimeglumine (Gd-DTPA) Magn Reson Med. 2007;58(2):402–406. doi: 10.1002/mrm.21290. [DOI] [PubMed] [Google Scholar]
- 29.Eckstein F, Wyman BT, Buck RJ, Wirth W, Maschek S, Hudelmaier M, et al. Longitudinal quantitative MR imaging of cartilage morphology in the presence of gadopentetate dimeglumine (Gd-DTPA) Magn Reson Med. 2009;61(4):975–980. doi: 10.1002/mrm.21929. [DOI] [PubMed] [Google Scholar]
- 30.Kellgren JH, Lawrence JS. Radiological assessment of osteoarthrosis. Ann Rheum Dis. 1957;16(4):494–502. doi: 10.1136/ard.16.4.494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Doherty M. Risk factors for progression of knee osteoarthritis. Lancet. 2001;358(9284):775–776. doi: 10.1016/S0140-6736(01)06006-8. [DOI] [PubMed] [Google Scholar]
- 32.Issa SN, Sharma L. Epidemiology of osteoarthritis: an update. Curr Rheumatol Rep. 2006;8(1):7–15. doi: 10.1007/s11926-006-0019-1. [DOI] [PubMed] [Google Scholar]
- 33.Lohmander LS, Felson D. Can we identify a ‘high risk’patient profile to determine who will experience rapid progression of osteoarthritis? Osteoarthritis Cartilage. 2004;12(Suppl A):S49–S52. doi: 10.1016/j.joca.2003.09.004. [DOI] [PubMed] [Google Scholar]
- 34.Bellamy N, Buchanan WW, Goldsmith CH, Campbell J, Stitt LW. Validation study of WOMAC: a health status instrument for measuring clinically important patient relevant outcomes to antirheumatic drug therapy in patients with osteoarthritis of the hip or knee. J Rheumatol. 1988;15(12):1833–1840. [PubMed] [Google Scholar]
- 35.Ware J, Snow K, Kosinksi M. SF-36 health survey: manual and interpretation guide. Boston, MA: The Health Institute, New England Medical Center; 1993. [Google Scholar]
- 36.Le Graverand MP, Vignon EP, Brandt KD, Mazzuca SA, Piperno M, Buck R, et al. Head-to-head comparison of the Lyon Schuss and fixed flexion radiographic techniques. Long-term reproducibility in normal knees and sensitivity to change in osteoarthritic knees. Ann Rheum Dis. 2008;67(11):1562–1566. doi: 10.1136/ard.2007.077834. [DOI] [PubMed] [Google Scholar]
- 37.Conrozier T, Favret H, Mathieu P, Piperno M, Provvedini D, Taccoen A, et al. Influence of the quality of tibial plateau alignment on the reproducibility of computer joint space measurement from Lyon schuss radiographic views of the knee in patients with knee osteoarthritis. Osteoarthritis Cartilage. 2004;12(10):765–770. doi: 10.1016/j.joca.2004.06.003. [DOI] [PubMed] [Google Scholar]
- 38.Shrout PE, Fleiss JL. Intraclass correlations: uses in assessing rater reliability. Psychol Bull. 1979;86(2):420–428. doi: 10.1037//0033-2909.86.2.420. [DOI] [PubMed] [Google Scholar]
- 39.Piperno M, Hellio Le Graverand MP, Conrozier T, Bochu M, Mathieu P, Vignon E. Quantitative evaluation of joint space width in femorotibial osteoarthritis: comparison of three radiographic views. Osteoarthritis Cartilage. 1998;6(4):252–259. doi: 10.1053/joca.1998.0118. [DOI] [PubMed] [Google Scholar]
- 40.Vignon E, Piperno M, Le Graverand MP, Mazzuca SA, Brandt KD, Mathieu P, et al. Measurement of radiographic joint space width in the tibiofemoral compartment of the osteoarthritic knee: comparison of standing anteroposterior and Lyon schuss views. Arthritis Rheum. 2003;48(2):378–384. doi: 10.1002/art.10773. [DOI] [PubMed] [Google Scholar]
- 41.Ding C, Cicuttini F, Scott F, Cooley H, Boon C, Jones G. Natural history of knee cartilage defects and factors affecting change. Arch Intern Med. 2006;166(6):651–658. doi: 10.1001/archinte.166.6.651. [DOI] [PubMed] [Google Scholar]
- 42.Brem MH, Schlechtweg PM, Bhagwat J, Genovese M, Dillingham MF, Yoshioka H, et al. Longitudinal evaluation of the occurrence of MRI-detectable bone marrow edema in osteoarthritis of the knee. Acta Radiol. 2008;49(9):1031–1037. doi: 10.1080/02841850802339413. [DOI] [PubMed] [Google Scholar]
- 43.Felson DT, Chaisson CE, Hill CL, Totterman SM, Gale ME, Skinner KM, et al. The association of bone marrow lesions with pain in knee osteoarthritis. Ann Intern Med. 2001;134(7):541–549. doi: 10.7326/0003-4819-134-7-200104030-00007. [DOI] [PubMed] [Google Scholar]
- 44.Felson DT, McLaughlin S, Goggins J, LaValley MP, Gale ME, Totterman S, et al. Bone marrow edema and its relation to progression of knee osteoarthritis. Ann Intern Med. 2003;139(5 Pt 1):330–336. doi: 10.7326/0003-4819-139-5_part_1-200309020-00008. [DOI] [PubMed] [Google Scholar]
- 45.Kornaat PR, Kloppenburg M, Sharma R, Botha-Scheepers SA, Le Graverand MP, Coene LN, et al. Bone marrow edema-like lesions change in volume in the majority of patients with osteoarthritis; associations with clinical features. Eur Radiol. 2007;17(12):3073–3078. doi: 10.1007/s00330-007-0711-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Raynauld JP, Martel-Pelletier J, Berthiaume MJ, Beaudoin G, Choquette D, Haraoui B, et al. Long term evaluation of disease progression through the quantitative magnetic resonance imaging of symptomatic knee osteoarthritis patients: correlation with clinical symptoms and radiographic changes. Arthritis Res Ther. 2006;8(1):R21. doi: 10.1186/ar1875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Roemer FW, Guermazi A, Javaid MK, Lynch JA, Niu J, Zhang Y, et al. Change in MRI-detected subchondral bone marrow lesions is associated with cartilage loss: the MOST Study. A longitudinal multicentre study of knee osteoarthritis. Ann Rheum Dis. 2009;68(9):1461–1465. doi: 10.1136/ard.2008.096834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Kornaat PR, Bloem JL, Ceulemans RY, Riyazi N, Rosendaal FR, Nelissen RG, et al. Osteoarthritis of the knee: association between clinical features and MR imaging findings. Radiology. 2006;239(3):811–817. doi: 10.1148/radiol.2393050253. [DOI] [PubMed] [Google Scholar]
- 49.Hunter DJ, March L, Sambrook PN. The association of cartilage volume with knee pain. Osteoarthritis Cartilage. 2003;11(10):725–729. doi: 10.1016/s1063-4584(03)00160-2. [DOI] [PubMed] [Google Scholar]
- 50.Wong S, Steinbach L, Zhao J, Stehling C, Ma CB, Link TM. Comparative study of imaging at 3.0 T versus 1.5 T of the knee. Skeletal Radiol. 2009;38(8):761–769. doi: 10.1007/s00256-009-0683-0. [DOI] [PMC free article] [PubMed] [Google Scholar]