The JAK2 V617F mutation is present in the vast majority of polycythemia vera patients, and in about half of patients with primary myelofibrosis and essential thrombocythemia.1 A correlation between MPN disease phenotype and the proportion of JAK2 V617F mutant alleles2, 3 has lead to the need for sensitive and reproducible molecular techniques to assess the so-called JAK2 V617F allele burden in patient samples. A 2009 multicenter study conducted by Lippert et al.4 assessed the concordance of JAK2 V617F allele burden quantitation from various diagnostic laboratories. The study demonstrated the importance of using well-defined and accurate standards to calibrate JAK2 V617F quantitative assays. To that end, the authors suggested the use of plasmid DNA dilutions along with a known genomic DNA sample as an internal control to allow for the most precise quantification of JAK2 V617F allele burden. As an alternative to plasmids, the diploid UKE-1 cell line, having two copies of mutated JAK2 per cell, was suggested as a useful genomic DNA standard.4
There are several JAK2 V617F-positive cell lines derived from patients with history of myeloproliferative or myelodysplasic disorders (HEL, MB-02, MUTZ-8, SET-2 and UKE-1) that differ in number of JAK2 mutant alleles. These cell lines are good research tools for elucidating the pathobiology of MPN,5 and are potential standard material as well. However, HEL cells have more than two copies of JAK2 and should not be used as a standard for quantification of JAK2 V617F allele burden.3, 4 The UKE-1 cell line has recently been shown to be homozygous for the JAK2 V617F mutation4, 5 and may therefore be a useful as a genomic DNA standard. UKE-1 was derived from an essential thrombocythemia patient transformed into acute leukemia in 1997.6 Phenotypic analysis of leukemic cells revealed coexpression of myeloid and endothelial antigens. Cytogenetic analysis of cells showed biclonality with the following karyotype: 48,XX,+8,+14[14]/45, XX,-7,del(11)(p14)[8]. UKE-1 cells were cultured in Iscove modified Dulbecco medium supplemented with 10% fetal calf serum, 10% horse serum and 1 μM hydrocortisone. Morphologically, UKE-1 cells were described as heterogeneous, with coexistence of large, adherent cells with prominent vacuoles and smaller nonadherent cells.6
Here, we show that UKE-1 cells in our hands underwent clonal evolution during in vitro culture, leading to an increase of JAK2 gene copies. Use of these cells as a standard would cause an underestimation of the JAK2 V617F allele burden.
The UKE-1 cells used in this study had been cultured for 5 months, frozen for 16 months, then thawed. We then cultivated the UKE-1 cells and a human myeloid cell line, HL60, for 3 weeks before making serial dilutions of UKE-1 in HL60 (100% UKE-1 through 0% UKE-1). This widely used human myeloid cell line derives from leukocytes of a patient with acute promyelocytic leukemia in 1979. Initial cytogenetic analysis revealed 75% of aneuploid cells (44 chromosomes) with structural anomalies on chromosomes 7, 9, 10 and number anomalies on chromosomes 5, 8, 18, X.7 The cell mixtures were washed twice in 1 × phosphate-buffered saline and pelleted. Genomic DNA was extracted with the FlexiGene DNA kit (Qiagen, Courtaboeuf, France), and JAK2 V617F quantification was performed using the plasmid based JAK2 Mutaquant kit (Ipsogen SA, Marseille, France) according to manufacturer recommendations.
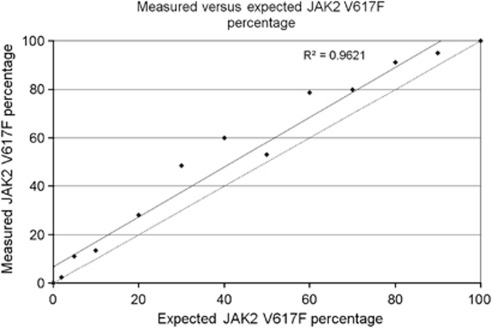
Throughout the range of dilutions, we found that the measured JAK2 V617F allele burden in the UKE-1/HL60 mixtures was consistently higher than expected (Figure 1). Similar findings were found in another lab. It was found that the JAK2 V617F allele burden was consistently overestimated when Mutaquant kit was used on a range of dilution of the line UKE-1: 2.4% for 1%, 67% for 50% in mean (Cassinat, personal data). One possible explanation for this discrepancy is an abnormal JAK2 copy number in one of the cell lines. We therefore performed conventional and molecular cytogenetic analysis of the HL60 and UKE-1 cells. Karyotypes were determined using standard cytogenetic techniques and described according to the International System for Human Cytogenetic Nomenclature (ISCN 2009).8 In order to assess JAK2 copy number, fluorescence in situ hybridization with bacterial artificial chromosomes (FISH-BAC) was performed as previously described.9 A BAC-targeting JAK2 (RP11-982A21 on 9p24.1 band) labeled in SpectrumGreen (Abbott, Rungis, France) and a reference BAC (RP11-115G2 located on 9p21.2) labeled in SpectrumOrange (Abbott) were applied to metaphase preparations and analyzed.
Figure 1.
Measured versus expected JAK2 V617F percentage. Serial dilutions of UKE-1 cells (supposedly homozygous for mutant JAK2 V617F) in HL60 cells (homozygous for wild-type JAK2) were made and the percentage of JAK2 V617F in each dilution was quantified using the JAK2 Mutaquant Ipsogen kit. For each dilution, the measured JAK2 V617F percentage was significantly greater than the theoretical ratio shown by the dotted line (P=0.002).
The karyotype of the HL60 cell line was 45,X,del(3)(p2?),add(5)(q2?3),der(8)t(4;8)(q2?;q24),del(9)(p1?2),del(10)(p1?2), add(11)(q2?3),i(13)(q10),add(17)(p11),del(18)(q1?)[22] (data not shown). FISH-BAC with JAK2 probe found an interstitial deletion of chromosome 9 without JAK2 deletion (data not shown) in HL60 lineage.
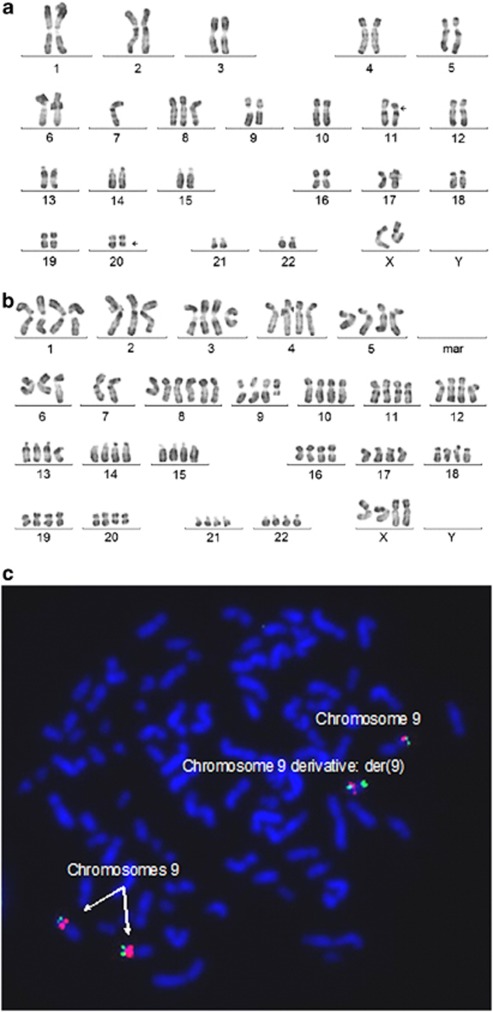
Cytogenetic analysis of UKE-1 revealed two clones: a pseudo-diploid clone (previously observed in 2008 in our lab) (Figure 2a) and a tetraploid clone (observed for the first time) (Figure 2b). The karyotype of UKE-1 was 46,XX,-7,+8,del(11)(p1?1p1?3),del(20q)[12]/92,idemx2,del(9)(q2?1)[8]. The tetraploid clone had three normal chromosomes 9 and a derivative chromosome 9 having a partial deletion of the long arm. FISH-BAC analysis showed the expected two copies of JAK2 in the pseudo-diploid clone (data not shown) and five copies of JAK2 in the tetraploid clone: one copy on each of the three normal chromosomes 9 and two copies of JAK2 on abnormal chromosome 9 (Figure 2c). As a consequence, there is an unbalanced ratio of wild-type JAK2 and JAK2 V617F within the quantification standard, leading to an underestimation of the JAK2 mutational load when using this UKE-1 cell line as the standard in the analysis of patient samples.
Figure 2.
Cytogenetic analysis of the UKE-1 cell line used in this experiment. We observed two clones and determined the karyotype to be 46,XX,-7,+8,del(11)(p1?1p1?3),del(20q)[12]/92,idemx2,del(9) (q2?1)[8]. (a) RHG banding of the pseudo-diploid clone. (b) RHG banding of the tetraploid clone. Note the partial deletion of the long arm of chromosome 9. (c) FISH-BAC analysis on the UKE-1 cell line using a JAK2 probe labeled with spectrumGreen and a reference probe on 9p21.2 labeled with spectrumOrange. The metaphase FISH shows five copies of JAK2 in the tetraploid clone: three normal chromosomes 9, each having one copy of JAK2 (green) and a chromosome 9 derivative having two copies of JAK2.
These results highlight the possibility of clonal evolution of the UKE-1 cell line with an increase in JAK2 gene copy number. The cell line has deviated following culture in our hands because the tetraploid clone was not present when supplying in 2008 and only two copies of JAK2 where detectable at this time. Mechanisms of this genetic instability may be related to an excessive reactive oxygen species production that promotes DNA double-strand breaks and altered repair, as this has been reported in several myeloid diseases and cell lines.10 In MPN, it has been shown that the JAK2 V617F mutation induces an increase in spontaneous homologous recombination leading to a hyperrecombination state.11 JAK2 V617F mutation may also affect p53 response to DNA damage.12
Several clinical studies show that measuring the JAK2 V617F allele burden may be important to assess prognosis and adjust treatment in MPN patients.2, 3, 13, 14 Accurate quantification of allele burden requires a well-defined genetic standard for calibration. Besides, cooperative works are underway to standardize and harmonize the q-PCR techniques. This study shows that using the UKE-1 cell line as a standard for JAK2 V617F quantification must be avoided, as cells in culture can acquire multiple copies of the JAK2 gene.
Acknowledgments
This work was supported by grants from the Ligue contre le Cancer (Comite Departemental du Finistere) and the Canceropole Grand Ouest. We thank Nadine Couchouron and the Biological Resource Centre (BRC) of Brest for technical help.
The authors declare no conflict of interest.
References
- Tefferi A, Vardiman JW. Classification and diagnosis of myeloproliferative neoplasms: the 2008 World Health Organization criteria and point-of-care diagnostic algorithms. Leukemia. 2008;22:14–22. doi: 10.1038/sj.leu.2404955. [DOI] [PubMed] [Google Scholar]
- Vannucchi AM, Antonioli E, Guglielmelli P, Pardanani A, Tefferi A. Clinical correlates of JAK2V617F presence or allele burden in myeloproliferative neoplasms: a critical reappraisal. Leukemia. 2008;22:1299–1307. doi: 10.1038/leu.2008.113. [DOI] [PubMed] [Google Scholar]
- Tefferi A, Strand JJ, Lasho TL, Knudson RA, Finke CM, Gangat N, et al. Bone marrow JAK2V617F allele burden and clinical correlates in polycythemia vera. Leukemia. 2007;21:2074–2075. doi: 10.1038/sj.leu.2404724. [DOI] [PubMed] [Google Scholar]
- Lippert E, Girodon F, Hammond E, Jelinek J, Reading NS, Fehse B, et al. Concordance of assays designed for the quantification of JAK2V617F: a multicenter study. Haematologica. 2009;94:38–45. doi: 10.3324/haematol.13486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quentmeier H, MacLeod RA, Zaborski M, Drexler HG. JAK2 V617F tyrosine kinase mutation in cell lines derived from myeloproliferative disorders. Leukemia. 2006;20:471–476. doi: 10.1038/sj.leu.2404081. [DOI] [PubMed] [Google Scholar]
- Fiedler W, Henke RP, Ergun S, Schumacher U, Gehling UM, Vohwinkel G, et al. Derivation of a new hematopoietic cell line with endothelial features from a patient with transformed myeloproliferative syndrome: a case report. Cancer. 2000;15:344–351. doi: 10.1002/(sici)1097-0142(20000115)88:2<344::aid-cncr14>3.0.co;2-6. [DOI] [PubMed] [Google Scholar]
- Gallagher R, Collins S, Trujillo J, McCredie K, Ahearn M, Tsai S, et al. Characterization of the continuous, differentiating myeloid cell line (HL-60) from a patient with acute promyelocytic leukemia. Blood. 1979;54:713–733. [PubMed] [Google Scholar]
- Basel SK. An International System for Human Cytogenetic Nomenclature. 2009.
- De Braekeleer E, Douet-Guilbert N, Basinko A, Morel F, Le Bris M-J, et al. Using bacterial artificial chromosomes in leukemia research: the experience at the University Cytogenetics Laboratory in Brest, France. J Biomed Biotechnol. 2011;2011:329471. doi: 10.1155/2011/329471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sallmyr A, Fan J, Rassool FV. Genomic instability in myeloid malignancies: increased reactive oxygen species (ROS), DNA double strand breaks (DSBs) and error-prone repair. Cancer Lett. 2008;270:1–9. doi: 10.1016/j.canlet.2008.03.036. [DOI] [PubMed] [Google Scholar]
- Plo I, Nakatake M, Malivert L, de Villartay JP, Giraudier S, Villeval JL, et al. JAK2 stimulates homologous recombination and genetic instability: potential implication in the heterogeneity of myeloproliferative disorders. Blood. 2008;112:1402–1412. doi: 10.1182/blood-2008-01-134114. [DOI] [PubMed] [Google Scholar]
- Nakatake M, Monte-Mor B, Debili N, Casadevall N, Ribrag V, Solary E, et al. JAK2(V617F) negatively regulates p53 stabilization by enhancing MDM2 via La expression in myeloproliferative neoplasms. Oncogene. 2012;31:1323–1333. doi: 10.1038/onc.2011.313. [DOI] [PubMed] [Google Scholar]
- Kiladjian JJ, Cassinat B, Chevret S, Turlure P, Cambier N, Roussel M, et al. Pegylated interferon-alfa-2a induces complete hematologic and molecular responses with low toxicity in polycythemia vera. Blood. 2008;112:3065–3072. doi: 10.1182/blood-2008-03-143537. [DOI] [PubMed] [Google Scholar]
- Kuriakose E, Vandris K, Wang YL, Chow W, Jones AV, Christos PJ, et al. Decrease in JAK2V617F allele burden is not a prerequisite to clinical response in patients with polycythemia vera Haematologica 2011. e-pub ahead of print 18 November 2011 doi: 10.3324/haematol.2011.053348 [DOI] [PMC free article] [PubMed]